Submitted:
12 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
1. The mucosal immune problem: why Th17/Treg balance matters in IBD
Hypothesis Development
2. Bile acid signaling as the proposed mechanistic link
3. Primary sclerosing cholangitis as a disease model for bile acid–immune axis disruption
Mechanistic Framework
4. The OCA dissociation and bile acid sequestrant evidence
5. Two committed dietary configurations and their bile acid environments
5.1. Mediterranean configuration
5.2. Committed ketogenic configuration
Critical caveats on ketogenic lipid and atherosclerosis data
6. The intermediate zone: a question, not a claim
7. Oxysterols, LXR, and the cholesterol–bile acid–immune interface
8. Systems-level coordination: the hepatic-vagal-colonic arc
Experimental Program
9. A staged experimental program
10. Testable predictions
11. Discussion and limitations
Author Contributions
Funding
AI Assistance Disclosure
Data Availability Statement
Ethics Statement
References
- Feagan, BG; Sandborn, WJ; Gasink, C; et al. Ustekinumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. 2016, 375(20), 1946–1960. [Google Scholar] [CrossRef]
- Sands, BE; Peyrin-Biroulet, L; Kierkus, J; et al. Efficacy and safety of mirikizumab in a randomized phase 2 study of patients with Crohn's disease. Gastroenterology 2022, 162(2), 495–508. [Google Scholar] [CrossRef]
- D'Haens, G; Panaccione, R; Baert, F; et al. Risankizumab as induction therapy for Crohn's disease: results from the phase 3 ADVANCE and MOTIVATE induction trials. Lancet 2022, 399(10340), 2015–2030. [Google Scholar] [CrossRef]
- Paik, D; Yao, L; Zhang, Y; et al. Human gut bacteria produce TH17-modulating bile acid metabolites. Nature 2022, 603(7903), 907–912. [Google Scholar] [CrossRef]
- Kummen, M; Thingholm, LB; Rühlemann, MC; et al. Altered gut microbial metabolism of essential nutrients in primary sclerosing cholangitis. Gastroenterology 2021, 160(5), 1784–1798.e0. [Google Scholar] [CrossRef]
- Chan, C; Lemos, M; Finnegan, P; et al. Fecal deoxycholic acid associates with diet, intestinal microbes, and total bilirubin in primary sclerosing cholangitis. JHEP Rep. 2024, 6(12), 101188. [Google Scholar] [CrossRef]
- Mousa, OY; Juran, BD; McCauley, BM; et al. Bile acid profiles in primary sclerosing cholangitis and their ability to predict hepatic decompensation. Hepatology 2021, 74(1), 281–295. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R; Ros, E; Salas-Salvadó, J; et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018, 378(25), e34. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Lista, J; Alcala-Diaz, JF; Torres-Peña, JD; et al. Long-term secondary prevention of cardiovascular disease with a Mediterranean diet and a low-fat diet (CORDIOPREV). Lancet 2022, 399(10338), 1876–1885. [Google Scholar] [CrossRef]
- Bhanpuri, NH; Hallberg, SJ; Williams, PT; et al. Cardiovascular disease risk factor responses to a type 2 diabetes care model including nutritional ketosis induced by sustained carbohydrate restriction at 1 year: an open label, non-randomized, controlled study. Cardiovasc Diabetol. 2018, 17(1), 56. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, AL; Athinarayanan, SJ; Van Tieghem, MR; et al. 5-year effects of a novel continuous remote care model with carbohydrate-restricted nutrition therapy including nutritional ketosis in type 2 diabetes: an extension study. Diabetes Res Clin Pract. 2024, 217, 111898. [Google Scholar] [CrossRef]
- Ang, QY; Alexander, M; Newman, JC; et al. Ketogenic diets alter the gut microbiome resulting in decreased intestinal Th17 cells. Cell. 2020, 181(6), 1263–1275.e16. [Google Scholar] [CrossRef] [PubMed]
- Bravata, DM; Sanders, L; Huang, J; et al. Efficacy and safety of low-carbohydrate diets: a systematic review. JAMA 2003, 289(14), 1837–1850. [Google Scholar] [CrossRef]
- Fante C, Spritzler F, Calabrese L, Laurent N, Roberts C, Deloudi S. The role of β-hydroxybutyrate testing in ketogenic metabolic therapies. Front Nutr. 2025;12:1629921. [COI: all authors affiliated with commercial ketogenic therapy entities including Keto-Mojo/Keto-Check and Virta Health; compliance figures cited as mechanistic context only.]. [CrossRef]
- Budoff, MJ; Manubolu, VS; Kinninger, A; et al. Carbohydrate restriction-induced elevations in LDL-cholesterol and atherosclerosis: the KETO trial. JACC Adv. 2024, 3(8), 101109. [Google Scholar] [CrossRef] [PubMed]
- Soto-Mota A, Norwitz NG, Manubolu VS, et al. Longitudinal data from the KETO-CTA study: Plaque predicts plaque, ApoB does not. JACC Adv. 2025;4(7):101686. [RETRACTED: Full retraction issued by JACC Advances due to methodological concerns.]. [CrossRef]
- Linsalata, M; Russo, F; Riezzo, G; et al. The effects of a very-low-calorie ketogenic diet on the intestinal barrier integrity and function in patients with obesity: a pilot study. Nutrients 2023, 15(11), 2561. [Google Scholar] [CrossRef]
- Hang, S; Paik, D; Yao, L; et al. Bile acid metabolites control TH17 and Treg cell differentiation. Nature 2019, 576(7785), 143–148. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C; McKenney, PT; Konstantinovsky, D; et al. Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature 2020, 581(7809), 475–479. [Google Scholar] [CrossRef] [PubMed]
- Song, X; Sun, X; Oh, SF; et al. Microbial bile acid metabolites modulate gut RORγ+ regulatory T cell homeostasis. Nature 2020, 577(7790), 410–415. [Google Scholar] [CrossRef]
- Won, TH; Arifuzzaman, M; Parkhurst, CN; et al. Host metabolism balances microbial regulation of bile acid signalling. Nature 2025, 638(8049), 216–224. [Google Scholar] [CrossRef]
- Gonzalez, FJ; Jiang, C; Patterson, AD. An intestinal microbiota–farnesoid X receptor axis modulates metabolic disease. Gastroenterology 2016, 151(5), 845–859. [Google Scholar] [CrossRef]
- Schmitt, J; Kong, B; Stieger, B; et al. Protective effects of farnesoid X receptor (FXR) on hepatic lipid accumulation are mediated by hepatic FXR and independent of intestinal FGF15 signal. Liver Int. 2015, 35(4), 1133–1144. [Google Scholar] [CrossRef]
- Kim, DH; Xiao, Z; Kwon, S; et al. A dysregulated acetyl/SUMO switch of FXR promotes hepatic inflammation in obesity. EMBO J 2015, 34(2), 184–199. [Google Scholar] [CrossRef]
- Guo, C; Xie, S; Chi, Z; et al. Bile acids control inflammation and metabolic disorder through inhibition of NLRP3 inflammasome. Immunity 2016, 45(4), 802–816. [Google Scholar] [CrossRef]
- Pols, TWH; Nomura, M; Harach, T; et al. TGR5 activation inhibits atherosclerosis by reducing macrophage inflammation and lipid loading. Cell Metab. 2011, 14(6), 747–757. [Google Scholar] [CrossRef]
- Zhang, Z; Meszaros, G; He, WT; et al. Protein kinase D at the Golgi controls NLRP3 inflammasome activation. J Exp Med. 2017, 214(9), 2671–2693. [Google Scholar] [CrossRef] [PubMed]
- Nagahashi, M; Takabe, K; Liu, R; et al. Conjugated bile acid-activated S1P receptor 2 is a key regulator of sphingosine kinase 2 and hepatic gene expression. Hepatology 2015, 61(4), 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Hou, L; Yang, L; Chang, N; et al. Macrophage sphingosine 1-phosphate receptor 2 blockade attenuates liver inflammation and fibrogenesis triggered by NLRP3 inflammasome. Front Immunol. 2020, 11, 1149. [Google Scholar] [CrossRef]
- Li, W; Hang, S; Fang, Y; et al. A bacterial bile acid metabolite modulates Treg activity through the nuclear hormone receptor NR4A1. Cell Host Microbe 2021, 29(9), 1366–1377.e9. [Google Scholar] [CrossRef] [PubMed]
- Dong, X; Sun, F; Secaira-Morocho, H; et al. The dichotomous roles of microbial-modified bile acids 7-oxo-DCA and isoDCA in intestinal tumorigenesis. Proc Natl Acad Sci USA 2024, 121(47), e2317596121. [Google Scholar] [CrossRef] [PubMed]
- Akagbosu, B; Tayyebi, Z; Shibu, G; et al. Novel antigen-presenting cell imparts Treg-dependent tolerance to gut microbiota. Nature 2022, 610(7933), 752–760. [Google Scholar] [CrossRef]
- Varanasi, SK; Chen, D; Liu, Y; et al. Bile acid synthesis impedes tumor-specific T cell responses during liver cancer. Science 2025, 387(6730), 192–201. [Google Scholar] [CrossRef]
- Poch, T; Krause, J; Casar, C; et al. Single-cell atlas of hepatic T cells reveals expansion of liver-resident naive-like CD4+ T cells in primary sclerosing cholangitis. J Hepatol. 2021, 75(2), 414–423. [Google Scholar] [CrossRef]
- Shaw, DG; Aguirre-Gamboa, R; Vieira, MC; et al. Antigen-driven colonic inflammation is associated with development of dysplasia in primary sclerosing cholangitis. Nat Med. 2023, 29(6), 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- AlRasheed, HA; El-Haggar, SM; Hegazy, SK; et al. Repurposing atorvastatin, HMGCO-A reductase inhibitor, in patients with ulcerative colitis: a randomized controlled study. J Clin Med. 2025, 14(9), 3077. [Google Scholar] [CrossRef]
- Alarfaj, SJ; El-Haggar, SM; Hegazy, SK; et al. Effect of a high dose atorvastatin as adjuvant therapy to mesalamine in attenuating inflammation and symptoms in patients with ulcerative colitis: a randomized controlled pilot study. Front Med. 2025, 11, 1490178. [Google Scholar] [CrossRef] [PubMed]
- Khrieba, MO; Abdulelah, FM; Alsaleh, NA; et al. Atorvastatin as an immunomodulatory adjunct in ulcerative colitis, beyond lipid lowering to inflammation control: a randomized controlled pilot study. Front Pharmacol. 2025, 16, 1690513. [Google Scholar] [CrossRef] [PubMed]
- Younossi, ZM; Ratziu, V; Loomba, R; et al. Obeticholic acid for the treatment of non-alcoholic steatohepatitis: interim analysis from a multicenter, randomized, placebo-controlled phase 3 trial. Lancet 2019, 394(10215), 2184–2196. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Recommendation to revoke conditional marketing authorization for Ocaliva. EMA website. 2024. Accessed 21 March 2026. https://www.ema.europa.eu/en/documents/referral/ocaliva-article-20-procedure-recommendation-revoke-conditional-marketing-authorization-ocaliva_en.pdf.
- Harrison, SA; Bedossa, P; Guy, CD; et al. A phase 3, randomized, controlled trial of resmetirom in NASH with liver fibrosis. N Engl J Med. 2024, 390(6), 497–509. [Google Scholar] [CrossRef]
- The Lipid Research Clinics Coronary Primary Prevention Trial results. I. Reduction in incidence of coronary heart disease. JAMA 1984, 251(3), 351–364. [CrossRef] [PubMed]
- Singh, N; Gurav, A; Sivaprakasam, S; et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 2014, 40(1), 128–139. [Google Scholar] [CrossRef]
- Gao, P; Rinott, E; Dong, D; et al. Gut microbial metabolism of bile acids modifies the effect of Mediterranean diet interventions on cardiometabolic risk in a randomized controlled trial. Gut Microbes 2024, 16(1), 2426610. [Google Scholar] [CrossRef] [PubMed]
- Seethaler, B; Neyrinck, AM; Basrai, M; et al. Elucidating the effect of the Mediterranean diet on fecal bile acids and their mediating role on biomarkers of intestinal barrier function: an exploratory analysis of a randomized controlled trial. Life Sci. 2025, 379, 123855. [Google Scholar] [CrossRef]
- Haskey, N; Estaki, M; Ye, J; et al. A Mediterranean diet pattern improves intestinal inflammation concomitant with reshaping of the bacteriome in ulcerative colitis: a randomized controlled trial. J Crohns Colitis 2023, 17(10), 1569–1578. [Google Scholar] [CrossRef]
- Strauss, JC; Haskey, N; Ramay, HR; et al. Weighted gene co-expression network analysis identifies a functional guild and metabolite cluster mediating the relationship between mucosal inflammation and adherence to the Mediterranean diet in ulcerative colitis. Int J Mol Sci. 2023, 24(8), 7323. [Google Scholar] [CrossRef] [PubMed]
- Godny, L; Elial-Fatal, S; Arrouasse, J; Sharar Fischler, T; Reshef, L; Kutukov, Y; Cohen, S; Pfeffer-Gik, T; et al. Mechanistic implications of the Mediterranean diet in patients with newly diagnosed Crohn's disease: multiomic results from a prospective cohort. Gastroenterology 2025, 168(5), 952–964.e2. [Google Scholar] [CrossRef]
- Bretin, A; Zou, J; Yeoh, BS; et al. Psyllium fiber protects against colitis via activation of bile acid sensor farnesoid X receptor. Cell Mol Gastroenterol Hepatol 2023, 15(6), 1421–1442. [Google Scholar] [CrossRef]
- Li, X; Yang, J; Zhou, X. Ketogenic diet-induced bile acids protect against obesity through reduced calorie absorption . Nat Metab. 2024, 6(7), 1397–1414, [RED under C-11 data quality filter: no BHB verification, non-IBD obese population, carbohydrate intake escalated to 36% energy by week 12. Admissible as mechanistic context for BSH-TGR5 pathway only.]. [Google Scholar] [CrossRef] [PubMed]
- Youm, YH; Nguyen, KY; Grant, RW; et al. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med. 2015, 21(3), 263–269. [Google Scholar] [CrossRef] [PubMed]
- Huang, C; Wang, J; Liu, H; et al. Ketone body β-hydroxybutyrate ameliorates colitis by promoting M2 macrophage polarization through the STAT6-dependent signaling pathway. BMC Med. 2022, 20(1), 148. [Google Scholar] [CrossRef] [PubMed]
- Kong, C; Yan, X; Liu, Y; et al. Ketogenic diet alleviates colitis by reduction of colonic group 3 innate lymphoid cells through altering gut microbiome. Signal Transduct Target Ther. 2021, 6(1), 154. [Google Scholar] [CrossRef] [PubMed]
- Norwitz, NG; Soto-Mota, A. Case report: carnivore–ketogenic diet for the treatment of inflammatory bowel disease: a case series of 10 patients. Front Nutr. 2024, 11, 1467475. [Google Scholar] [CrossRef]
- Shimazu, T; Hirschey, MD; Newman, J; et al. Suppression of oxidative stress by β-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 2013, 339(6116), 211–214. [Google Scholar] [CrossRef] [PubMed]
- Chriett, S; Dąbek, A; Wojtala, M; et al. Prominent action of butyrate over β-hydroxybutyrate as histone deacetylase inhibitor, transcriptional modulator and anti-inflammatory molecule. Sci Rep. 2019, 9(1), 742. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z; Zhang, D; Chung, D; et al. Metabolic regulation of gene expression by histone lysine β-hydroxybutyrylation. Mol Cell. 2016, 62(2), 194–206. [Google Scholar] [CrossRef]
- Qin, J; Huang, X; Gou, S; et al. Ketogenic diet reshapes cancer metabolism through lysine β-hydroxybutyrylation. Nat Metab. 2024, 6(8), 1505–1528. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, BB; Holmbäck, UC; Volpi, E; et al. Malonyl coenzyme A and the regulation of functional carnitine palmitoyltransferase-1 activity and fat oxidation in human skeletal muscle. J Clin Invest. 2002, 110(11), 1687–1693. [Google Scholar] [CrossRef] [PubMed]
- Seidelmann, SB; Claggett, B; Cheng, S; et al. Dietary carbohydrate intake and mortality: a prospective cohort study and meta-analysis. Lancet Public Health 2018, 3(9), e419–e428. [Google Scholar] [CrossRef] [PubMed]
- Angelotti, A; Kowalski, C; Johnson, LK; et al. Restricted carbohydrate diets below 45% energy are not associated with risk of mortality in the National Health and Nutrition Examination Survey, 1999–2018. Front Nutr. 2024, 11, 1225674. [Google Scholar] [CrossRef]
- Parigi, SM; Das, S; Frede, A; et al. Liver X receptor regulates Th17 and RORγt+ Treg cells by distinct mechanisms. Mucosal Immunol. 2021, 14(2), 411–419. [Google Scholar] [CrossRef] [PubMed]
- Jacobse, J; Brown, RE; Li, J; et al. Interleukin-23 receptor signaling impairs the stability and function of colonic regulatory T cells. Cell Rep. 2023, 42(2), 112128. [Google Scholar] [CrossRef] [PubMed]
- Wang, T; Cui, S; Hao, L; et al. Regulation of Th17/Treg balance by 27-hydroxycholesterol and 24S-hydroxycholesterol correlates with learning and memory ability in mice. Int J Mol Sci. 2022, 23(8), 4370. [Google Scholar] [CrossRef]
- Wang, T; Feng, W; Ju, M; et al. 27-hydroxycholesterol causes cognitive deficits by disturbing Th17/Treg balance and the related immune responses in mild cognitive impairment patients and C57BL/6J mice. J Neuroinflammation 2023, 20(1), 305. [Google Scholar] [CrossRef]
- Xiao, J; Wang, S; Chen, L; et al. 25-hydroxycholesterol regulates lysosome AMP kinase activation and metabolic reprogramming to educate immunosuppressive macrophages. Immunity 2024, 57(5), 1087–1104.e7. [Google Scholar] [CrossRef] [PubMed]
- Emgård, J; Kammoun, H; García-Cassani, B; et al. Oxysterol sensing through the receptor GPR183 promotes the lymphoid-tissue-inducing function of innate lymphoid cells and colonic inflammation. Immunity 2018, 48(1), 120–132.e8. [Google Scholar] [CrossRef]
- Ameraoui, H; Bestard-Escalas, J; Roumain, M; et al. Oxysterol-mediated modulation of intestinal inflammation: insights into sex differences and GPR183 signaling. Cell Mol Life Sci. 2025, 83(1), 69. [Google Scholar] [CrossRef]
- Tao, H; Yu, Z; Dong, Y; Liu, L; Peng, L; Chen, X. Lipids, lipid-lowering agents, and inflammatory bowel disease: a Mendelian randomization study. Front Immunol. 2023, 14, 1160312. [Google Scholar] [CrossRef]
- Teratani, T; Mikami, Y; Nakamoto, N; et al. The liver–brain–gut neural arc maintains the Treg cell niche in the gut. Nature 2020, 585(7826), 591–596. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y; Meerschaert, KA; Galvan-Pena, S; et al. A chemogenetic screen reveals that Trpv1-expressing neurons control regulatory T cells in the gut. Science 2024, 385(6708), eadk1679. [Google Scholar] [CrossRef] [PubMed]
- Hogan, AE; Gaoatswe, G; Lynch, L; et al. Glucagon-like peptide 1 analogue therapy directly modulates innate immune-mediated inflammation in individuals with type 2 diabetes mellitus. Diabetologia 2014, 57(4), 781–784. [Google Scholar] [CrossRef]
- Hengist, A; Davies, RG; Walhin, JP; et al. Ketogenic diet but not free-sugar restriction alters glucose tolerance, lipid metabolism, peripheral tissue phenotype, and gut microbiome: RCT. Cell Rep Med. 2024, 5(8), 101667. [Google Scholar] [CrossRef] [PubMed]
- Li, J; Dawson, PA. Animal models to study bile acid metabolism. Biochim Biophys Acta Mol Basis Dis. 2019, 1865(5), 895–911. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, SJ; McKenzie, AL; Williams, PT; et al. Effectiveness and safety of a novel care model for the management of type 2 diabetes at 1 year: an open-label, non-randomized, controlled study. Diabetes Ther. 2018, 9(2), 583–612. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, N; Sasaki, N; Aoki, R; et al. Gut pathobionts underlie intestinal barrier dysfunction and liver T helper 17 cell immune response in primary sclerosing cholangitis. Nat Microbiol. 2019, 4(3), 492–503. [Google Scholar] [CrossRef]
- O'Neill, DF; Westman, EC; Bernstein, RK. The effects of a low-carbohydrate regimen on glycemic control and serum lipids in diabetes mellitus. Metabolic Syndrome and Related Disorders 2003, 1(4), 291–298. [Google Scholar] [CrossRef] [PubMed]
- Soto-Mota, A; Flores-Jurado, Y; Norwitz, NG; Feldman, D; Pereira, MA; Danaei, G; Ludwig, DS. Increased low-density lipoprotein cholesterol on a low-carbohydrate diet in adults with normal but not high body weight: a meta-analysis. Am J Clin Nutr. 2024, 119(3), 740–747. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on sclerosing cholangitis. J Hepatol. 2022, 77(3), 761–806. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y; Yan, W; Zhao, H; et al. Isoallolithocholic acid ameliorates intestinal inflammation via metabolically reprogrammed macrophages. Commun Biol. 2025, 8, 1718. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y; Guzior, DV; Okros, M; et al. Balance between bile acid conjugation and hydrolysis activity can alter outcomes of gut inflammation. Nat Commun. 2025, 16(1), 3434. [Google Scholar] [CrossRef]
- Kabil, A; Nayyar, N; Xu, C; et al. Functional targeting of ILC2s and ILC3s reveals selective roles in intestinal fibrosis and homeostasis. J Exp Med. 2025, 222(7), e20241671. [Google Scholar] [CrossRef] [PubMed]
- García-Mateo, S; Martínez-Domínguez, SJ; Gargallo-Puyuelo, CJ; et al. Healthy lifestyle is a protective factor from moderate and severe relapses and steroid use in inflammatory bowel disease: a prospective cohort study. Inflamm Bowel Dis. 2025, 31(1), 95–104. [Google Scholar] [CrossRef] [PubMed]
- Manns, MP; Bergquist, A; Karlsen, TH; et al. Primary sclerosing cholangitis. Nat Rev Dis Primers 2025, 11(1), 17. [Google Scholar] [CrossRef] [PubMed]
- Toivio, L; Toivio, J; Lindén, J; et al. Free acid β-hydroxybutyrate supplementation does not ameliorate dextran sodium sulfate-induced colitis similar to ketogenic diet in male mice. PharmaNutrition 2025, 31, 100437. [Google Scholar] [CrossRef]
- Westman, EC; Yancy, WS, Jr.; Olsen, MK; Dudley, T; Guyton, JR. Effect of a low-carbohydrate, ketogenic diet program compared to a low-fat diet on fasting lipoprotein subclasses. Int J Cardiol. 2006, 110(2), 212–216. [Google Scholar] [CrossRef]
- Hirschberger, S; Strauß, G; Effinger, D; et al. Very-low-carbohydrate diet enhances human T-cell immunity through immunometabolic reprogramming. EMBO Mol Med. 2021, 13(8), e14323. [Google Scholar] [CrossRef]
- Burén, J; Ericsson, M; Damasceno, NRT; Sjödin, A. A ketogenic low-carbohydrate high-fat diet increases LDL cholesterol in healthy, young, normal-weight women: a randomized controlled feeding trial. Nutrients 2021, 13(3), 814. [Google Scholar] [CrossRef]
- Volek, JS; Noakes, T; Phinney, SD. Rethinking fat as a fuel for endurance exercise. Eur J Sport Sci. 2015, 15(1), 13–20. [Google Scholar] [CrossRef]
- Sayin, SI; et al. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab. 2013, 17(2), 225–235. [Google Scholar] [CrossRef] [PubMed]
- Sun, F; et al. The microbial bile acid metabolite 3-oxo-LCA inhibits colorectal cancer progression. Cancer Res. 2025, 85(24), 4937–4957. [Google Scholar] [CrossRef] [PubMed]
- Kinzig, KP; Honors, MA; Hargrave, SL. Insulin sensitivity and glucose tolerance are altered by maintenance on a ketogenic diet. Endocrinology 2010, 151(7), 3105–3114. [Google Scholar] [CrossRef] [PubMed]
- Webster, CC; et al. Reduced glucose tolerance and skeletal muscle GLUT4 and IRS1 content in cyclists habituated to a long-term low-carbohydrate, high-fat diet. Int J Sport Nutr Exerc Metab. 2020, 30(3), 210–217. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S; et al. Intermittent ketogenic diet promotes Treg differentiation and alleviates Crohn’s disease via the β-hydroxybutyrate driven AHCY–DNMT1–Foxp3 axis. J Crohns Colitis 2026, jjag003. [Google Scholar] [CrossRef]
- Rodrigues, PF; et al. Rorγt-positive dendritic cells are required for the induction of peripheral regulatory T cells in response to oral antigens. Cell. 2025, 188(10), 2720–2737.e22. [Google Scholar] [CrossRef]
- Cabric, V; et al. A wave of Thetis cells imparts tolerance to food antigens early in life. Science 2025, 389(6757), 268–274. [Google Scholar] [CrossRef]
- Abramson, J; Dobeš, J; Lyu, M; Sonnenberg, GF. The emerging family of RORγt+ antigen-presenting cells. Nat Rev Immunol. 2024, 24(1), 64–77. [Google Scholar] [CrossRef] [PubMed]
- Jalil, A; et al. Bile acid 7α-dehydroxylating bacteria accelerate injury-induced mucosal healing in the colon. EMBO Mol Med. 2025, 17(5), 889–908. [Google Scholar] [CrossRef]
- Xiao, J; et al. Clostridium scindens protects against vancomycin-induced cholestasis and liver fibrosis by activating intestinal FXR-FGF15/19 signaling. Adv Sci (Weinh) 2025, 12(5), e2406445. [Google Scholar] [CrossRef]
- Lv, L; et al. Revealing VNN1: An emerging and promising target for inflammation and redox balance. Immun Inflamm Dis. 2025, 13(10), e70274. [Google Scholar] [CrossRef] [PubMed]
- Millet, V; et al. Harnessing the Vnn1 pantetheinase pathway boosts short chain fatty acids production and mucosal protection in colitis. Gut 2023, 72(6), 1115–1128. [Google Scholar] [CrossRef]
- Haskey, N; Ye, J; Josephson, J; Raman, M; Ghosh, S; Gibson, DL. Metabolomic signatures highlight fiber-degrading Bacteroides species in Mediterranean diet response among ulcerative colitis. Gastro Hep Adv. 2025, 4(4), 100606. [Google Scholar] [CrossRef] [PubMed]
- Straniero, S; Laskar, A; Savva, C; Härdfeldt, J; Angelin, B; Rudling, M. Of mice and men: murine bile acids explain species differences in the regulation of bile acid and cholesterol metabolism. J Lipid Res. 2020, 61(4), 480–491. [Google Scholar] [CrossRef] [PubMed]
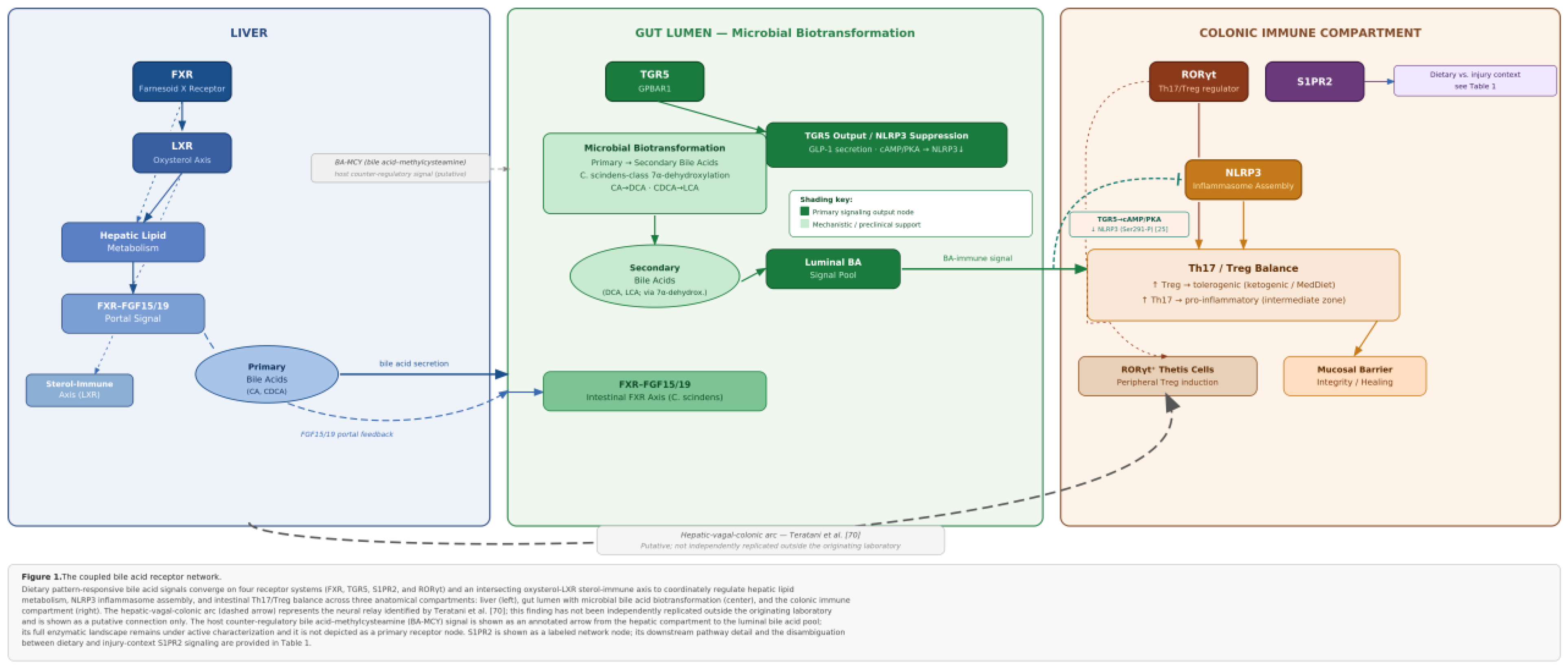
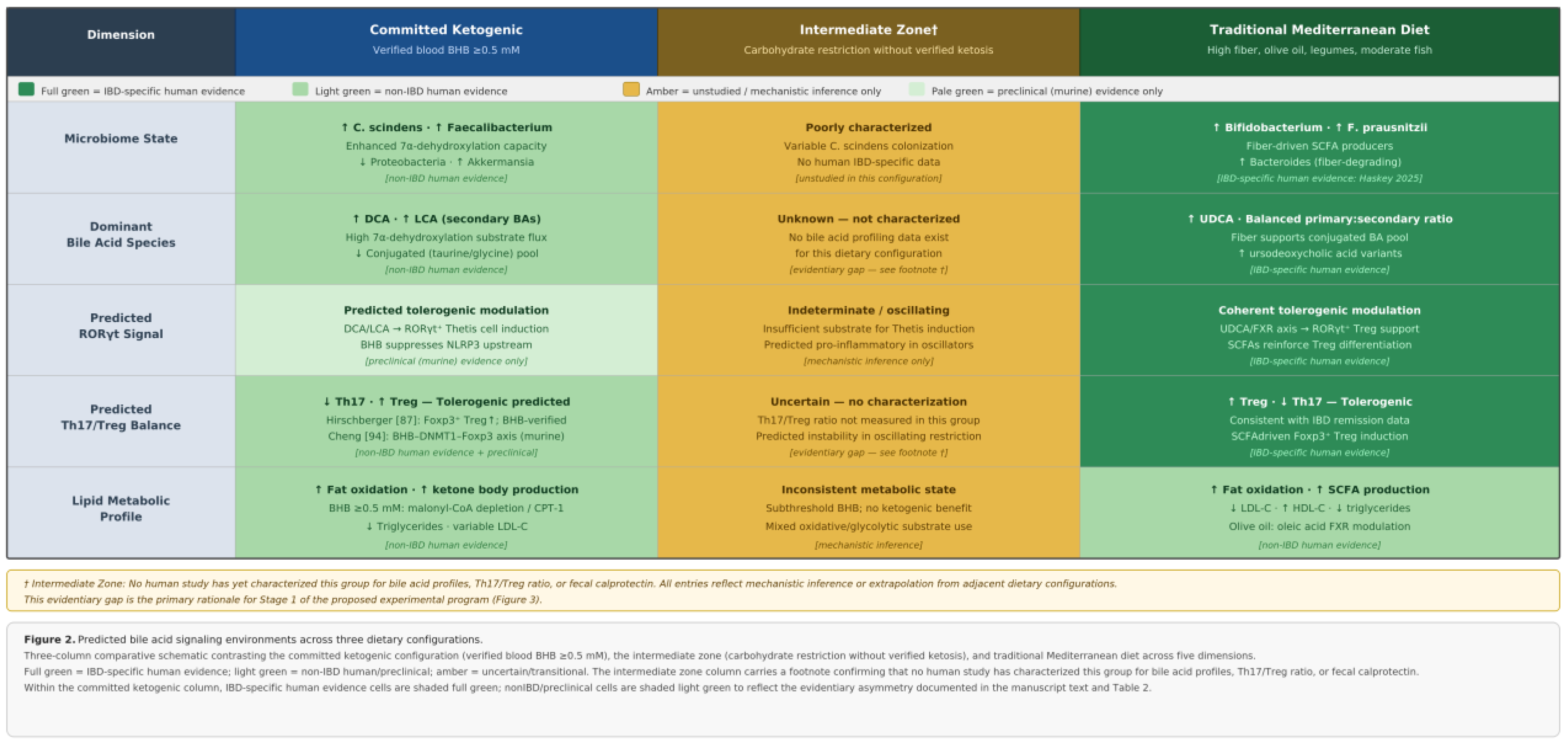
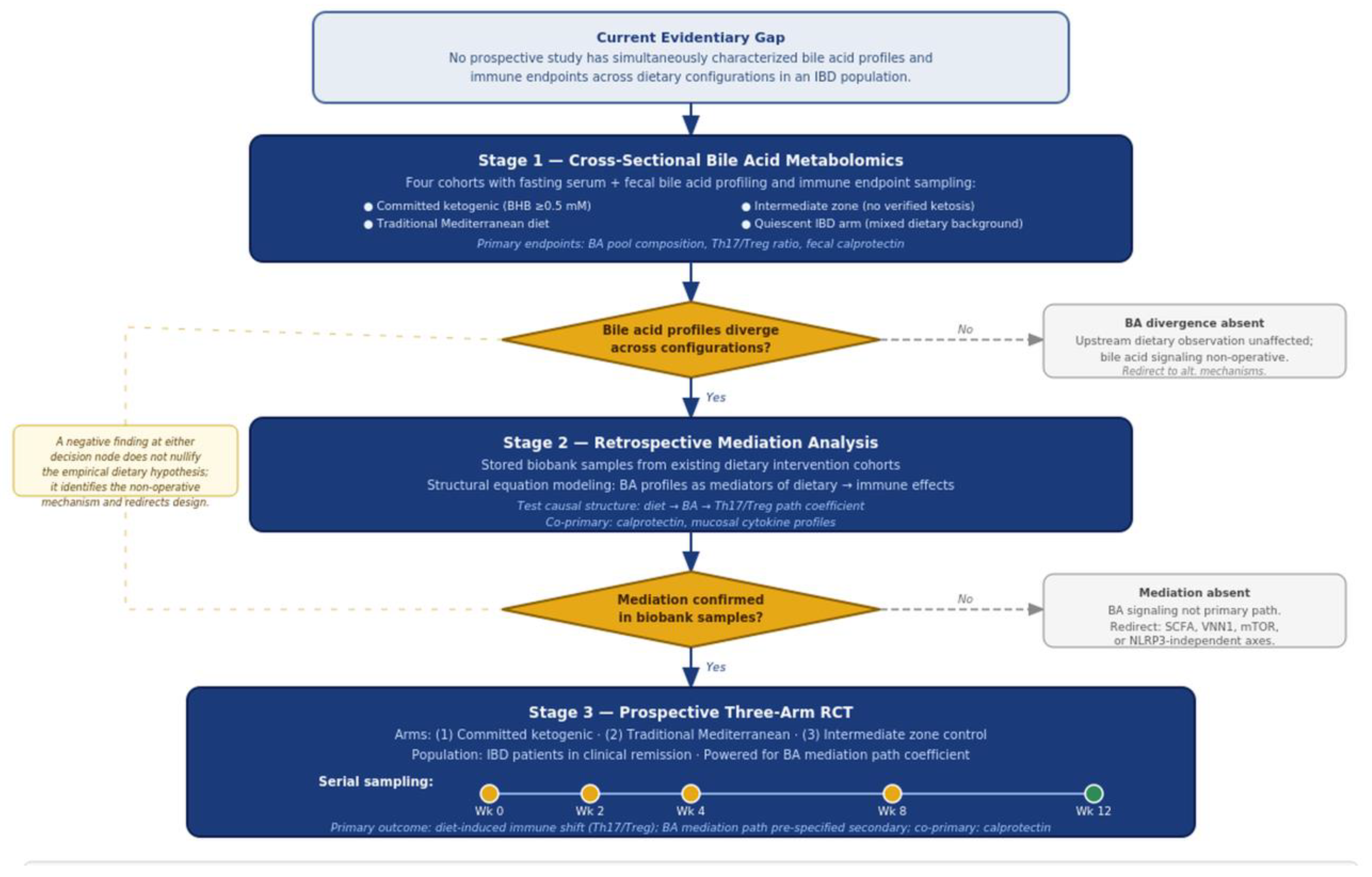
| Receptor | Primary Dietary Ligands | Tissue / Cell Type | Key Downstream Pathway | Supporting Evidence | Predicted Dietary Effect | Key Ref(s) |
|---|---|---|---|---|---|---|
| FXR (NR1H4) — FARNESOID X RECEPTOR | ||||||
|
FXR (hepatic) |
CDCA, DCA, LCA (secondary bile acids) | Hepatocytes | CYP7A1/CYP8B1 repression via SHP → bile acid synthesis suppression; SREBP-1c suppression → reduced lipogenesis; SUMO2-K277 → NF-κB transrepression | OCA (REGENERATE) improved hepatic fibrosis despite atherogenic lipid shift — confirms hepatic FXR engagement at pharmacological concentrations [39]; DIRECT-PLUS [44]: baseline fecal BAs modified MedDiet cardiometabolic response |
KD: elevated secondary BAs activate hepatic FXR; suppresses CYP7A1, reduces VLDL production MedDiet: diverse secondary BA pool maintains hepatic FXR tone Intermediate zone: oscillating BA environment — predicted incomplete FXR activation |
[22,23,24] |
|
FXR (intestinal) |
Secondary bile acids (same ligands; opposing functional context to hepatic FXR) | Intestinal epithelial cells | Intestinal FXR inhibition improves metabolic outcomes in obesity models; FGF15/19 signaling to liver; psyllium → intestinal FXR activation → colitis protection abolished in FXR-KO mice | Bretin et al. 2023 [49]: psyllium protects against DSS and T-cell-transfer colitis through FXR activation; first direct IBD-relevant demonstration of dietary fiber acting via intestinal FXR |
KD: substrate-depleted microbiome alters secondary BA profile at intestinal epithelium MedDiet: psyllium and soluble fiber components activate intestinal FXR; Bretin 2023 confirms FXR-dependent colitis protection |
[22,23,49] |
| TGR5 (GPBAR1) — G PROTEIN-COUPLED BILE ACID RECEPTOR | ||||||
|
TGR5 (L-cell) |
LCA, DCA, TDCA, TUDCA (secondary and taurine-conjugated species) | Enteroendocrine L-cells (ileum and colon) | cAMP → GLP-1 secretion → hepatic lipogenesis suppression, insulin sensitization, innate immune modulation; GLP-1 analogue therapy reduces inflammatory cytokines in immune cells [72] | TDCA and TUDCA elevated in KD-fed mice; correlational support n=416 observational and n=25 interventional human cohorts (Li et al. [50]: RED — no BHB verification; admissible as mechanistic plausibility for BSH-TGR5 pathway) |
KD: elevated TDCA/TUDCA provide TGR5 agonism; predicted enhanced GLP-1 output MedDiet: diverse secondary BA pool maintains TGR5 stimulation Intermediate zone: oscillating environment — predicted incomplete TGR5 activation |
[25,26,50,72] |
|
TGR5 (macrophage) |
LCA, DCA, TLCA (potent agonists) | Macrophages (intestinal and systemic) | cAMP/PKA → NLRP3 phosphorylation (Ser291 mouse; Ser295 human ortholog) → inflammasome assembly blocked [25]; PKD phosphorylates same residue at Golgi in activating context [27] — anti-inflammatory output is pathway- and compartment-dependent | TGR5 activation reduces oxidized LDL uptake and macrophage lipid loading in atherosclerosis models [26] — clearest example of cell-autonomous metabolic-immune coupling in this receptor class |
Both committed patterns: secondary BAs maintain TGR5-mediated NLRP3 suppression Intermediate zone: oscillating BA environment — predicted incomplete inflammasome suppression |
[25,26,27] |
| S1PR2 — SPHINGOSINE-1-PHOSPHATE RECEPTOR 2 | ||||||
|
S1PR2 (hepatocyte, dietary context) |
Conjugated bile acids (taurine- and glycine-conjugated primary and secondary species) | Hepatocytes | SphK2 → nuclear S1P → HDAC1/2 inhibition → SREBP-1c, FAS, LDLR upregulation → lipid metabolism gene activation [28] | Metabolic-protective role under dietary bile acid signaling [28]; distinct from injury-context S1PR2 behavior (see row below; see footnote¹) |
KD: elevated TDCA/TUDCA modulate hepatocyte S1PR2 tone MedDiet: diverse conjugated pool; S1PR2 hepatic signaling maintained |
[28] |
|
S1PR2 (macrophage, injury context — see footnote¹) |
Sphingosine-1-phosphate (S1P) — not a bile acid ligand in this context | Infiltrating macrophages (pathological liver injury) | SphK1 → NLRP3 inflammasome priming → pro-inflammatory cytokine expression [29] | Atorvastatin RCTs in UC [36,37,38]: serum S1P significantly reduced alongside IL-6 and TNF-α, linking HMG-CoA reductase inhibition to S1PR2/SphK1 pathway in colonic macrophages; included for disambiguation only Injury-context findings do not extrapolate to dietary signaling physiology |
Injury-context row; no dietary prediction applicable. Included to make the S1PR2 disambiguation visible at the table level. | [29,36,37,38] |
| RORΓT — RAR-RELATED ORPHAN RECEPTOR GAMMA T | ||||||
|
RORγt (colonic Th17 cell) |
3-oxolithocholic acid (3-oxoLCA), isolithocholic acid (isoLCA) — direct binding; competitive RORγt antagonists; 27-OHC (oxysterol) is a RORγt agonist in functional antagonism | Colonic Th17 cells | 3-oxoLCA and isoLCA bind RORγt → suppress Th17 differentiation [18]; 27-OHC activates RORγt → promotes Th17, suppresses Tregs [64,65] — functional antagonism at same receptor | Paik et al. 2022 [4]: 3-oxoLCA and isoLCA depleted in CD patients across two independent cohorts; depletion inversely correlated with IL-17-related gene expression Wang et al. 2025 [80]: isoalloLCA depleted in pediatric UC in proportion to disease severity |
KD: predicted increased 3-oxoLCA/isoLCA availability under committed microbiome configuration; 27-OHC production may decrease if hepatic cholesterol synthesis reduced (speculative) MedDiet: fiber substrate maintains Coriobacteriaceae; sustained 3-oxoLCA/isoLCA production Intermediate zone: incomplete microbiome restructuring — insufficient or oscillating 3-oxoLCA/isoLCA |
[4,18,64,65,80] |
|
RORγt (Foxp3⁺ Treg / isoalloLCA axis — see footnote²) |
Isoallolithocholic acid (isoalloLCA) — distinct stereoisomer from isoLCA; see footnote² | Foxp3⁺ regulatory T cells (colonic) | isoalloLCA → mitochondrial ROS → Foxp3 CNS3 enhancer → Foxp3⁺ Treg differentiation [18]; NR4A1 identified as downstream effector via distinct bacterial metabolite, independently of FXR and VDR [30] | Hirschberger et al. 2021 [87]: BHB-verified (self-managed ad libitum KD after counseling; BHB ≥0.5 mM confirmed at days 7, 14, 21), 44 healthy volunteers — Foxp3⁺ Treg expansion by flow cytometry, IL-10 upregulation, RNAseq-confirmed immunometabolic reprogramming (3 weeks; below 8-week threshold) Kabil et al. 2025 [82]: ILC3 suppression via RORγt-dependent mechanism attenuates intestinal fibrosis |
KD: committed microbiome may reduce isoalloLCA-producing Coriobacteriaceae; Treg-expanding signal may be partially replaced by BHB-direct immunometabolic reprogramming (Hirschberger [87]) MedDiet: high-fiber substrate maintains Coriobacteriaceae; sustained isoalloLCA production and Foxp3⁺ Treg expansion |
[18,30,82,87] |
| LXR (NR1H3/NR1H2) — LIVER X RECEPTOR (OXYSTEROL–STEROL IMMUNE AXIS) | ||||||
|
LXR-α (CD11c⁺ myeloid, mesenteric) |
Oxysterols: 27-hydroxycholesterol, 25-hydroxycholesterol (derived from cholesterol via CYP27A1/CYP7B1) | CD11c⁺ myeloid cells (mesenteric lymph node) | LXRα deficiency increases mesenteric Th17 cells via isoform-specific mechanism [62]; Jacobse et al. [63]: IL-23R signaling downregulates LXR target genes in colonic Tregs → impairs Treg stability | Parigi et al. 2021 [62]: LXRα deficiency increases mesenteric Th17 cells specifically; LXRβ deficiency increases RORγt⁺ Tregs specifically through CD11c⁺ myeloid signaling (non-overlapping isoform-specific functions); human scRNA-seq confirms IL-23R on colonic Tregs [63] |
KD: sustained malonyl-CoA depletion and β-oxidation predicted to reduce de novo cholesterol synthesis; potential 27-OHC reduction in macrophages (speculative; no direct human data) MedDiet: oxysterol remodeling not directly characterized under MedDiet No study has measured oxysterol profiles under committed dietary patterns in human IBD patients. |
[62,63,64,65,66,67,68] |
|
LXR-β (CD11c⁺ myeloid, mesenteric) |
Same oxysterol ligands as LXRα; broader tissue distribution | CD11c⁺ myeloid cells; also intestinal epithelium | LXRα deficiency increases mesenteric Th17 cells specifically; LXRβ deficiency increases RORγt⁺ Tregs specifically through CD11c⁺ myeloid signaling [62]; FXR-LXR crosstalk at shared gene regulatory networks predicted when bile acid and oxysterol precursor flux both altered | Parigi et al. 2021 [62]: LXRα-null mice show increased Th17 cells; LXRβ-null mice show increased RORγt⁺ Tregs — non-overlapping isoform-specific functions with distinct downstream cell types |
Both LXR isoforms: framework proposes committed dietary patterns shift hepatic cholesterol flux, altering both the bile acid pool and oxysterol landscape simultaneously, engaging FXR, TGR5, LXRα, and LXRβ as co-equal nodes in the coupled network All LXR dietary predictions are speculative; no direct measurement in human IBD under committed dietary conditions |
[62,63] |
| Study / Year | Design | Population (n) | Intervention or Exposure | Bile Acid or Microbiome Outcome | Immune or Clinical Outcome |
|---|---|---|---|---|---|
| IBD AND PSC HUMAN COHORT DATA — BILE ACID AND MICROBIOME EVIDENCE | |||||
|
Paik et al. Nature 2022 |
Cross-sectional; two independent IBD cohorts | CD patients and healthy controls | Gordonibacter pamelaeae and Coriobacteriaceae abundance; 3α/3β-HSDH enzyme gene expression | 3-oxoLCA and isoLCA biosynthetic genes and fecal metabolite levels significantly depleted in CD across both cohorts vs. controls | Depletion inversely correlated with IL-17-related host gene expression; most direct human translational evidence for the bile acid–Th17 regulatory axis in IBD [4] |
|
Kummen et al. Gastroenterology 2021 |
Shotgun metagenomics; multicenter | PSC patients and controls; n≈300 combined | PSC microbiome composition; stratified by concurrent IBD status | Eubacterium spp. and Ruminococcus obeum depleted in PSC independent of IBD status; genera encode 3α/3β-HSDH and 7α-dehydroxylation enzymes for 3-oxoLCA/isoLCA biosynthesis | IBD-independent depletion: bile acid-transforming capacity impaired upstream of diet-microbiome interaction; establishes PSC as human model of the proposed bile acid–immune tolerance breakdown [5] |
|
Mousa et al. Hepatology 2021 |
Population-scale cross-sectional; Mayo Clinic | 400 PSC vs. 302 controls | Fecal and serum bile acid profiles; primary-to-secondary bile acid ratios | Markedly elevated primary-to-secondary bile acid ratios in PSC; quantitative confirmation of deficient secondary bile acid conversion at population scale | Quantitative confirmation of the bile acid environment predicted by microbiome depletion findings; strengthens PSC-as-model-disease argument [7] |
|
Chan et al. JHEP Rep 2024 |
Cross-sectional with 16S microbiome profiling | 26 early-stage PSC patients and controls | Fecal bile acids; gut microbiota; dietary intake; BA synthesis and FXR activity markers | Fecal DCA significantly lower in PSC vs. controls (pₐₑⱼ=0.04); Blautia and Lachnoclostridium abundance positively correlated with fecal DCA; DCA negatively correlated with total bilirubin (p=0.006) | DCA reduction not mediated by BA synthesis or FXR activation; microbiome-driven mechanism; Blautia and Lachnoclostridium as candidate restorative genera under dietary intervention [6] |
| ⚠MEASUREMENT GAP — No published study has directly measured 3-oxoLCA, isoalloLCA, isoLCA, or isoDCA in PSC stool or tissue. The inference chain (depleted secondary BA-producing bacteria → reduced immunomodulatory trace bile acids → Th17/Treg imbalance) is the central untested prediction of this framework. Direct trace bile acid measurement in PSC stool is a Stage 1 priority target. | |||||
| MEDITERRANEAN DIET — RCT AND COHORT EVIDENCE IN IBD-RELEVANT POPULATIONS | |||||
|
Seethaler et al. Life Sci 2025 |
Exploratory analysis of RCT (LIBRE trial; NCT02087592) | 68 women with impaired intestinal barrier (n=33 MedDiet, n=35 control) | Mediterranean diet vs. standard diet; 12 weeks | MedDiet decreased fecal DCA and LCA; increased UDCA; formal mediation analysis confirmed bile acid changes mediated beneficial effects on intestinal barrier integrity (zonulin, LBP) | First RCT demonstrating that MedDiet-induced bile acid compositional shifts mechanistically mediate a gut-specific endpoint; distinct from DIRECT-PLUS (baseline BAs modified response magnitude) [45] |
|
Strauss, Haskey et al. Int J Mol Sci 2023 |
WGCNA metabolomics; randomized pilot (NCT04474561; small n¹) | n=29 quiescent UC; MedDiet n=13 responders, n=16 non-responders | Mediterranean diet vs. Canadian habitual diet; 12 weeks | Bile acid profiles within a WGCNA-identified metabolite cluster mediated the relationship between Mediterranean diet score and fecal calprotectin | Pilot-scale signal for bile acid-mediated immunological response; Faecalibacterium prausnitzii, Dorea longicatena, Roseburia inulinivorans identified as functional mediators [47] |
|
Haskey et al. J Crohns Colitis 2023 |
Randomized controlled trial (NCT03053713; pilot study¹) | n=28 quiescent UC; 12 weeks | Mediterranean diet vs. control diet; 12 weeks | Microbiome reshaped toward Mediterranean-associated taxa; dysbiosis markers improved in MedDiet arm; no direct bile acid metabolomics | 20% of MedDiet participants had fecal calprotectin >100 μg/g vs. 75% of controls; IBD-specific RCT-level evidence for MedDiet effect on mucosal inflammatory activity [46] |
|
Godny et al. Gastroenterology 2025 |
Prospective cohort; dietary recall-based adherence scoring (no dietary assignment) | 271 newly diagnosed CD patients; median 27-month follow-up | Mediterranean diet adherence score; bile acid profiles, kynurenines, Faecalibacterium, SCFAs | Higher MedDiet adherence inversely correlated with primary bile acids and pro-inflammatory kynurenines; positively correlated with Faecalibacterium and SCFAs | MedDiet adherence inversely correlated with CDAI, fecal calprotectin, and CRP; largest IBD-specific cohort with simultaneous favorable shifts in lipid-adjacent and immune markers [48] |
|
DIRECT-PLUS Trial (Gao P et al.) Gut Microbes 2024 |
Multi-omics RCT analysis; n=284 | 284 adults; healthy dietary guidelines or two MedDiet variants; 18-month follow-up | Longitudinal fecal bile acid metabolomics (44 species); gut microbiome shotgun sequencing | Baseline fecal BA levels significantly modified cardiometabolic response to MedDiet; 14 fecal BAs prospectively associated with BMI and lipid profiles | First RCT evidence of bile acid profile-mediated modification of a dietary intervention's cardiometabolic effect; mechanistically distinct from Seethaler 2025 [44] |
| KETOGENIC DIET — PRECLINICAL AND IBD-RELEVANT EVIDENCE | |||||
|
Hirschberger et al. EMBO Mol Med 2021 |
Prospective intervention; BHB-verified (TIER 1) | 44 healthy volunteers; ≤30g/day CHO; BHB verified ≥0.5 mM throughout | Strictly enforced very-low-carbohydrate diet; blood BHB verified ≥0.5 mM throughout; 3 weeks | No bile acid metabolomics conducted | Foxp3⁺ Treg expansion by flow cytometry; IL-10 upregulation; RNAseq-confirmed immunometabolic reprogramming toward oxidative phosphorylation. Only human study with BHB-verified committed ketosis demonstrating Treg expansion; 3 weeks (below 8-week threshold); healthy volunteers, not IBD patients [87] |
|
Westman et al. Int J Cardiol 2006 |
24-week RCT vs. low-fat diet; urinary ketone monitoring (TIER 2 — blood BHB not measured²) | 119 overweight hyperlipidemic adults (n=60 KD, n=59 low-fat); NMR lipoprotein subfraction analysis | Low-carbohydrate ketogenic program (≤20g/day CHO) vs. low-fat diet | Urinary ketones positive throughout; blood BHB not measured — committed-ketosis threshold unconfirmed (TIER 2) | KD arm: large VLDL −78%, small LDL −78%, medium LDL −42%, large HDL +21% by NMR; most detailed lipoprotein subfraction data under strict carbohydrate restriction; no immune outcomes measured [86] |
|
Norwitz and Soto-Mota Front Nutr 2024 |
Case series (AMBER — signal only; no BHB documentation in C-11 format³) | 10 IBD patients (6 UC, 4 CD) | Carnivore-ketogenic diet; clinical outcomes reported; no BHB verification | No bile acid or microbiome measurements; metabolic state not verified by blood BHB meeting C-11 criteria | Universal clinical improvement reported; most discontinued medications; only IBD-specific human clinical signal for the ketogenic pole. Insufficient for clinical inference; provides signal for prospective investigation [54] |
|
Kong et al. Signal Transduct Target Ther 2021 |
Mouse DSS colitis model (RED — murine; 6-hydroxylated BA pool absent in humans) | C57BL/6 mice; DSS colitis model | Ketogenic diet vs. control; fecal microbiota transfer to germ-free mice | KD restructured gut microbiota; fecal microbiota transfer confirmed microbiome-dependent mechanism | KD reduced colonic RORγt⁺CD3⁾ ILC3s and inflammatory cytokines; ILC3 reduction warrants prospective human investigation given ILC3 role in sustaining colonic Th17 programs in IBD [53] |
|
Huang et al. BMC Med 2022 |
Human tissue + murine DSS model (GREEN for IBD relevance of BHB deficit; murine intervention component RED) | IBD patients and healthy controls (tissue); C57BL/6 mice (DSS model) | Colonic mucosal BHB measurement in IBD patients; rectal BHB enema in DSS colitis mice | BHB significantly reduced in colonic mucosa of UC and CD patients; inversely correlated with disease activity | Colonic mucosal BHB deficit in IBD patients is the primary IBD-specific translational finding (GREEN); STAT6/M2 mechanism in mice requires human validation; colonic BHB deficit is distinct from measuring dietary ketosis [52] |
| ⚠MEASUREMENT GAP — No study has simultaneously verified blood BHB ≥0.5 mM, characterized the bile acid and SCFA metabolome, and measured Th17/Treg balance in the same subjects under committed ketosis sustained ≥12 weeks. Hirschberger [87]: BHB-verified and Treg measured, but healthy volunteers, 3 weeks, no bile acid metabolomics. Westman [86]: lipoprotein subfractions documented (urinary ketone compliance only; blood BHB not measured; omega-3 supplementation confound) but no immune outcomes. This three-failure design gap is the primary methodological motivation for the Stage 1 experimental program. | |||||
| CHOLESTEROL–IMMUNE NODE — STATIN RCT EVIDENCE IN UC (INDIRECT SUPPORT FOR OXYSTEROL-LXR AXIS) | |||||
|
AlRasheed, Alarfaj, Khrieba et al. J Clin Med / Front Med / Front Pharmacol 2025 |
Three independent RCTs (same clinical program, Tanta and Horus University) | Active UC; combined n≈300; atorvastatin 80mg added to standard mesalamine | Atorvastatin adjunctive therapy vs. mesalamine alone; multiple biomarker panels | Alarfaj et al. [37]: significant reduction in serum S1P in atorvastatin arm — links HMG-CoA reductase inhibition to S1PR2/SphK1 pathway in colonic macrophages; Mendelian randomization [69]: HMGCR-mediated LDL-C lowering does not increase IBD risk; PCSK9-mediated LDL-C lowering paradoxically does | All three trials: significant reductions in disease activity, IL-6, TNF-α, and fecal calprotectin vs. mesalamine alone; converging evidence that cholesterol pathway interventions alter IBD-relevant inflammatory signals through pleiotropic mechanisms [36,37,38,69] |
| Outcome Domain | H1: Bile Acid Signaling | H2: BHB-Direct NLRP3 Inhibition | H3: Kbhb-mTOR / Epigenetic | H4: Oxysterol-LXR (Exploratory) | Decision Rule |
|---|---|---|---|---|---|
| Immunomodulatory bile acid concentrations (3-oxoLCA, isoalloLCA, isoLCA, isoDCA) | Predicted higher in committed vs. intermediate groups; HMCRI predicted lower. Mechanistic rationale: these species suppress Th17 via RORγt binding and expand Tregs via mitochondrial ROS [18,19]; their depletion is documented in IBD cohorts [4], not in dietary adherence groups. BA-MCY FXR antagonist biology established in mouse and limited human serum data [21]; dietary-group predictions are untested. | No specific prediction; BHB-direct NLRP3 inhibition operates through K⁺ efflux blockade [51]; relationship to bile acid pool composition not tested. | No specific prediction; Kbhb operates through histone modification [57] and ALDOB K108bhb-mediated mTOR inhibition [58]; relationship to bile acid concentrations untested. | Predicted to correlate inversely with 27-OHC via shared RORγt competition; mechanistic rationale from mouse models [62,64]; gut-specific demonstration not established. Exploratory. | Bayesian SEM + LOO-CV |
| Th17/Treg balance (flow cytometry) | Predicted improved in committed vs. intermediate; immunomodulatory bile acid concentrations predicted to mediate this improvement in SEM [18,19]. HMCRI predicted to track direction of effect [21]. Discriminating feature: H1 predicts bile acid mediation of both lipid and immune outcomes simultaneously; neither H2 nor H4 predicts this co-mediation. All dietary-group predictions are untested. | BHB inhibits NLRP3 assembly via K⁺ efflux blockade in macrophages and human monocytes [51]; predicts Th17/Treg benefit in committed KD arm specifically, not Mediterranean arm (BHB concentration-dependent). Bile acid independence inferred, not directly tested. | Not directly testable in Stage 1; BHB concentration serves as indirect proxy for Kbhb substrate availability only [57,58]. Formal test planned for Stage 3 (sorted Treg Kbhb ChIP-seq). | LXR deficiency increases mesenteric Th17 in mouse models [62]; IL-23R suppresses LXR target genes in colonic Tregs [63]. 27-OHC promotes Th17 via RORγt in neurological models [64,65]; gut demonstration not established. Exploratory. | Bayesian SEM + LOO-CV |
| Lipoprotein subfractions (NMR) | Favorable subfraction profile predicted in committed groups. FXR activation suppresses VLDL-TG secretion via SHP-SREBP-1c repression [22,23] and is a candidate partial mediator; malonyl-CoA/CPT-1 disinhibition is the more direct explanation for KD remodeling and is not a bile acid mechanism. H1 predicts both outcomes co-mediated; this is the discriminative claim. | No direct lipid prediction; NLRP3 mechanism is primarily immune. Lipoprotein subfraction differences between groups attributable to malonyl-CoA/CPT-1, not to NLRP3 [51]. | ALDOB K108bhb-mediated mTOR inhibition demonstrated in cancer cell lines [58]; specific lipoprotein subfraction prediction in human dietary contexts is untested. | 27-OHC predicted lower in committed KD via reduced hepatic cholesterol availability; co-directional lipid improvement is a speculative downstream inference. No human dietary oxysterol-lipoprotein data in IBD. Exploratory. | Bayesian SEM + LOO-CV; exploratory for H4 |
| HMCRI (BA-MCY / immunomodulatory secondary BA sum) | Predicted lower in committed vs. intermediate groups; predicted higher in IBD/PSC regardless of dietary pattern (Coriobacteriaceae depletion as driver). BA-MCY FXR antagonist biology established [21]; HMCRI as a named construct is manuscript-derived and requires Stage 1 empirical validation. Reference ranges do not yet exist. | No prediction; BHB-direct NLRP3 mechanism does not engage BA-MCY counter-regulation. | No prediction; Kbhb-mTOR mechanism does not engage BA-MCY counter-regulation. | No prediction; oxysterol-LXR mechanism does not engage BA-MCY counter-regulation. | Bayesian SEM + LOO-CV |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




