Submitted:
24 January 2023
Posted:
25 January 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
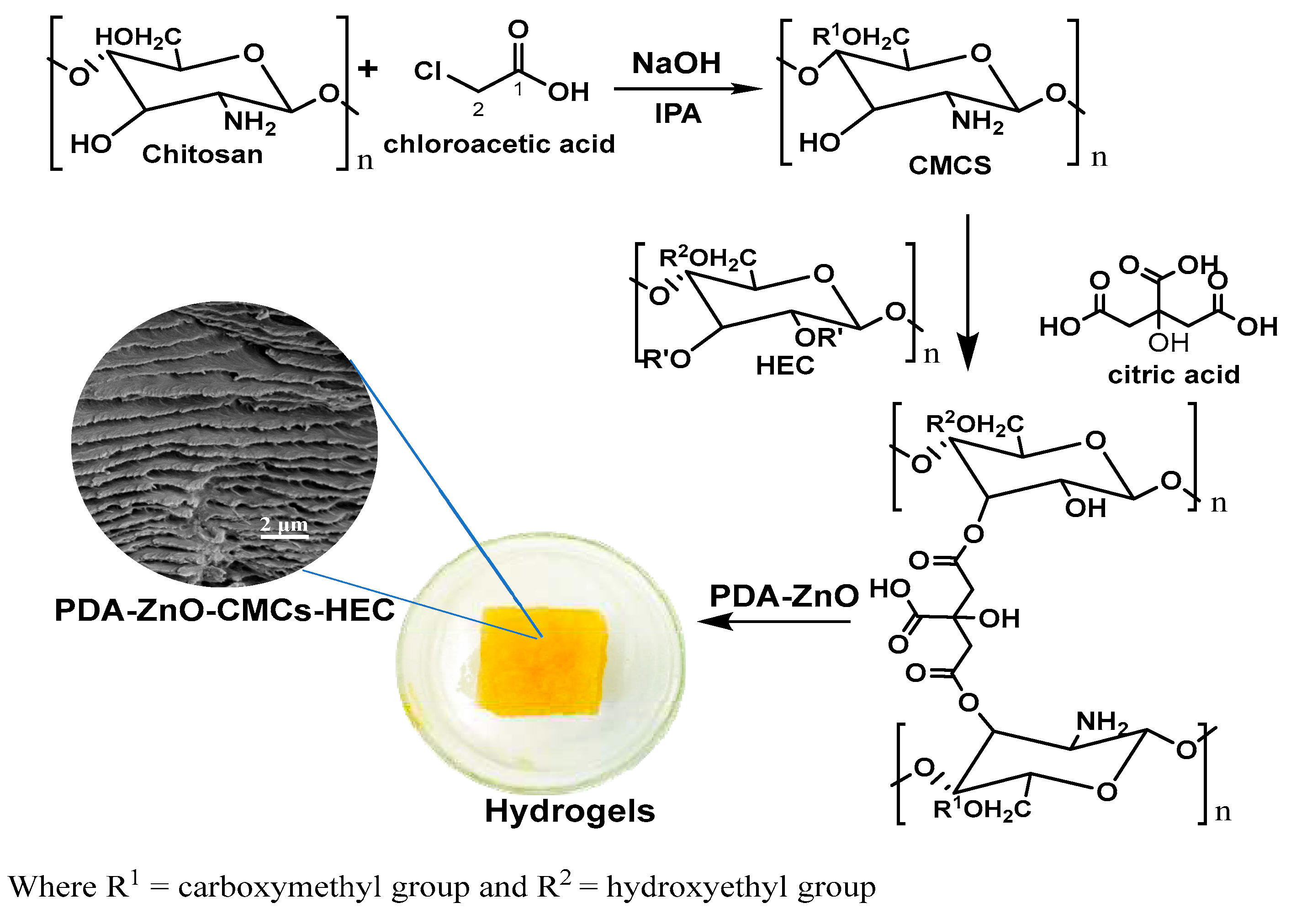
Synthesis of CMCs-HEC Hydrogels
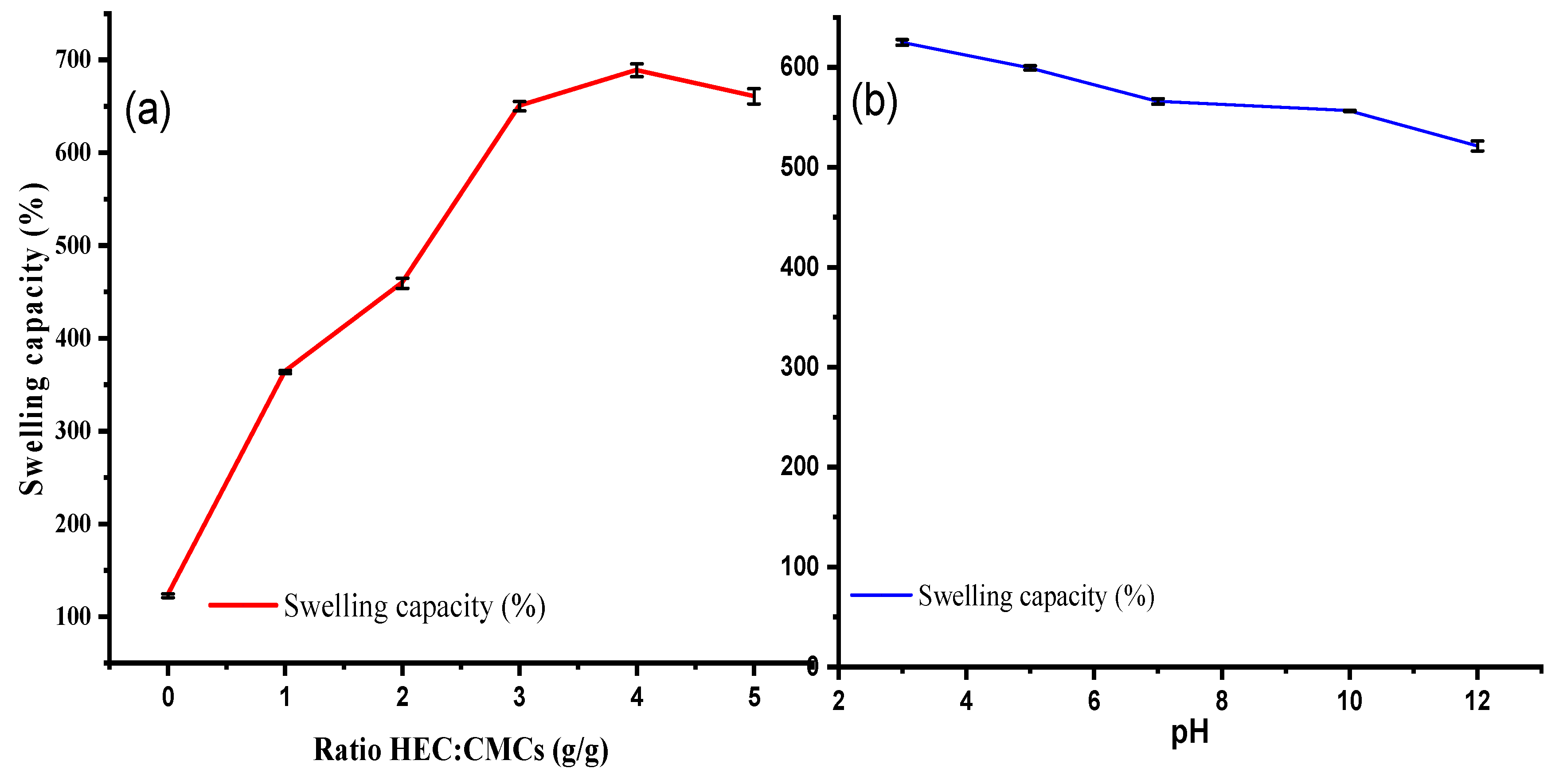
Swelling Measurements
Colorimetric Response to pH Change
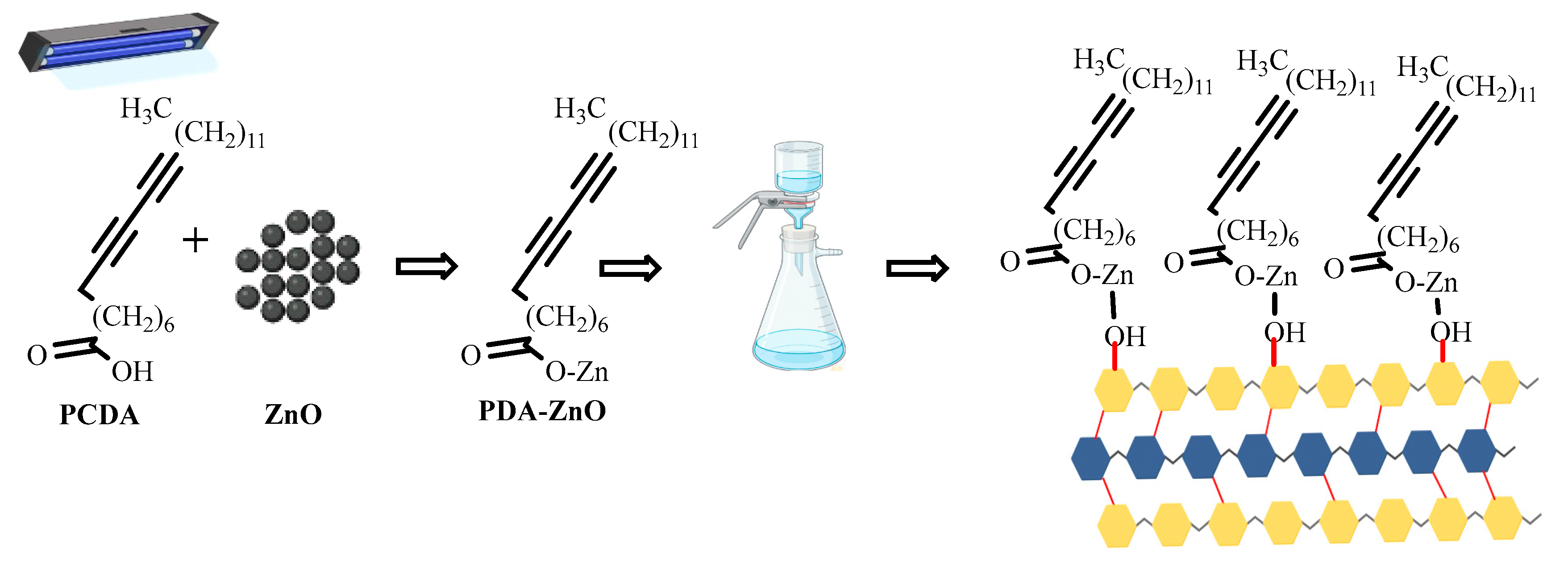
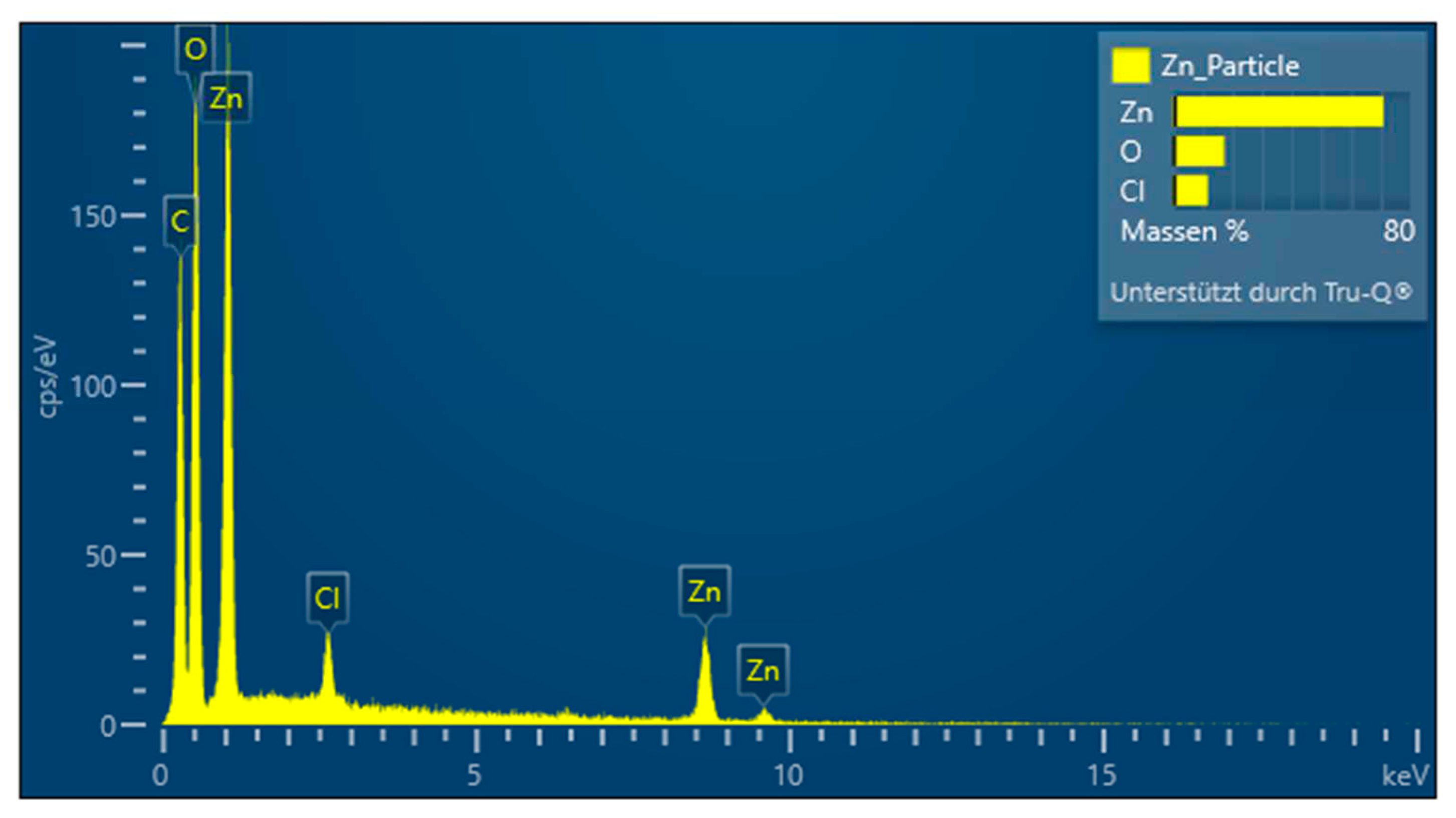
Characterization of PDA-ZnO-CMCs-HEC
Zinc Ion Release Experiments
Killing Curve Assay
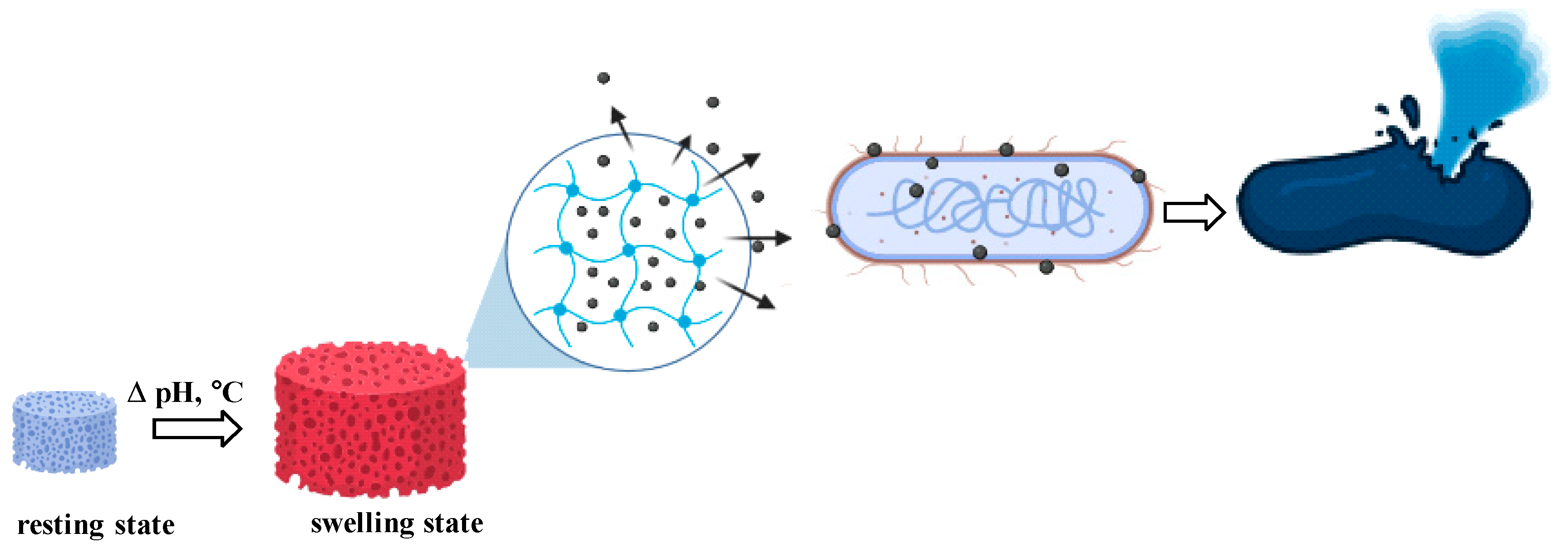
Results and Discussions
Swelling Behaviour of CMC-HEC hydrogels
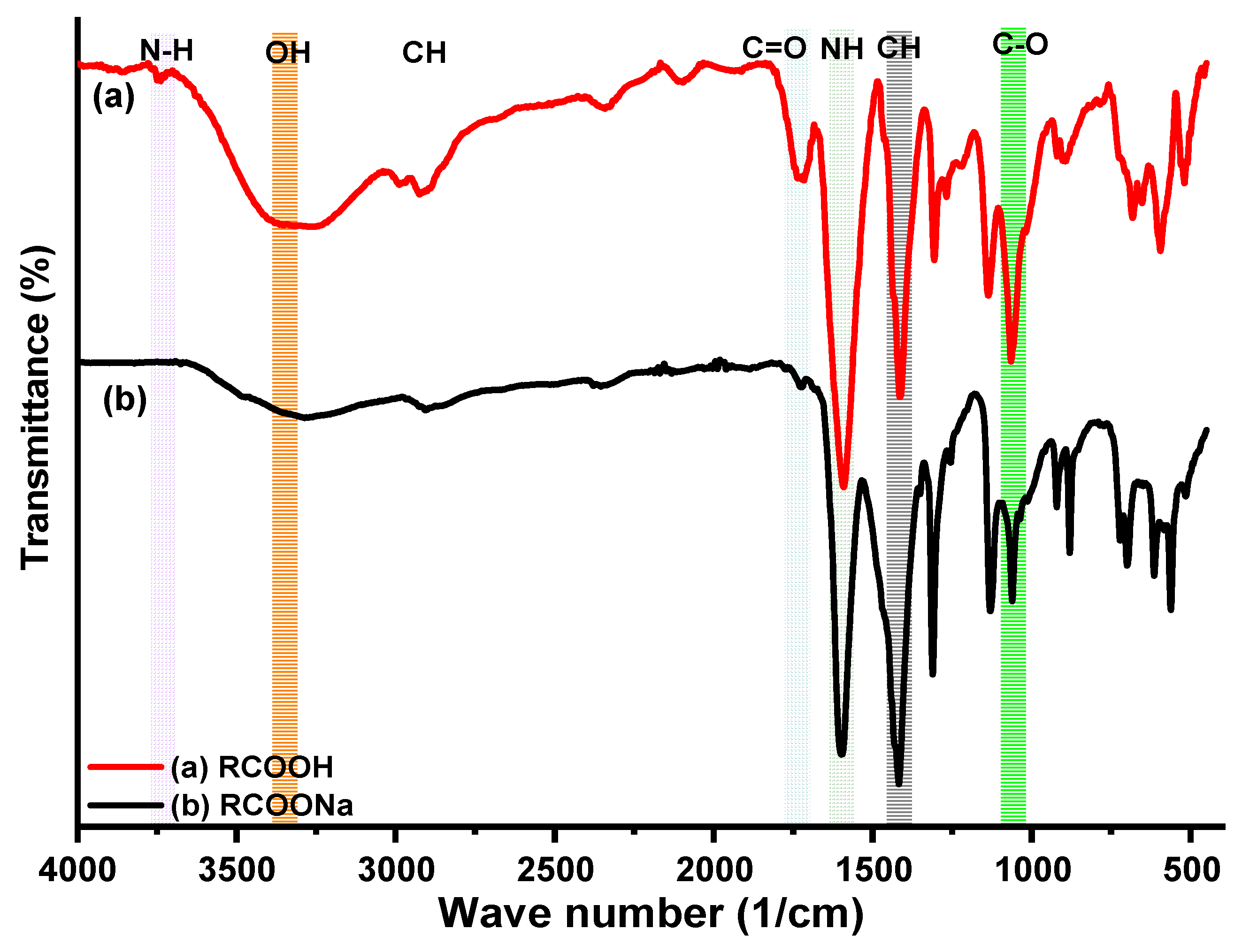
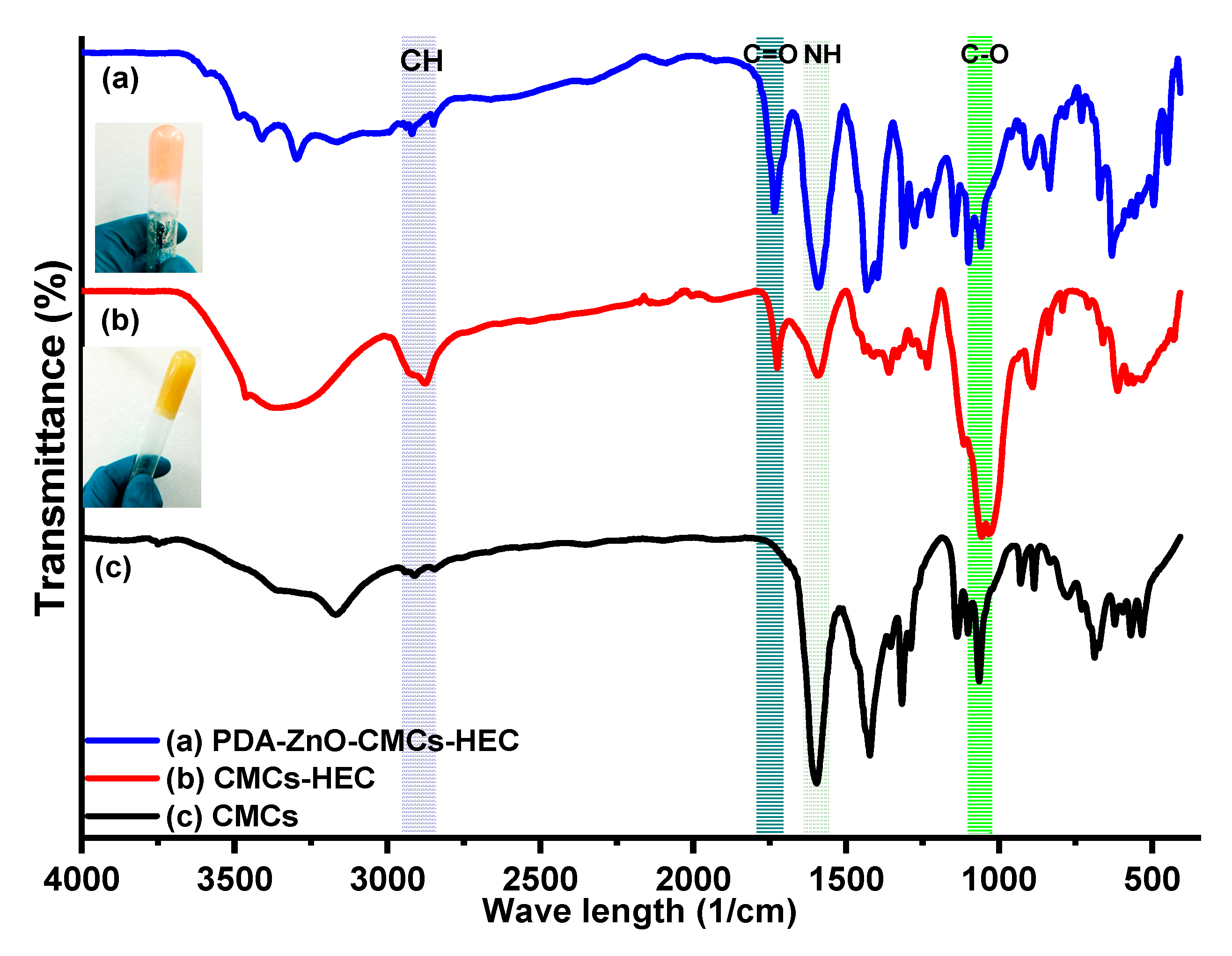
IR results of CMCs, CMCs-HEC Hydrogels
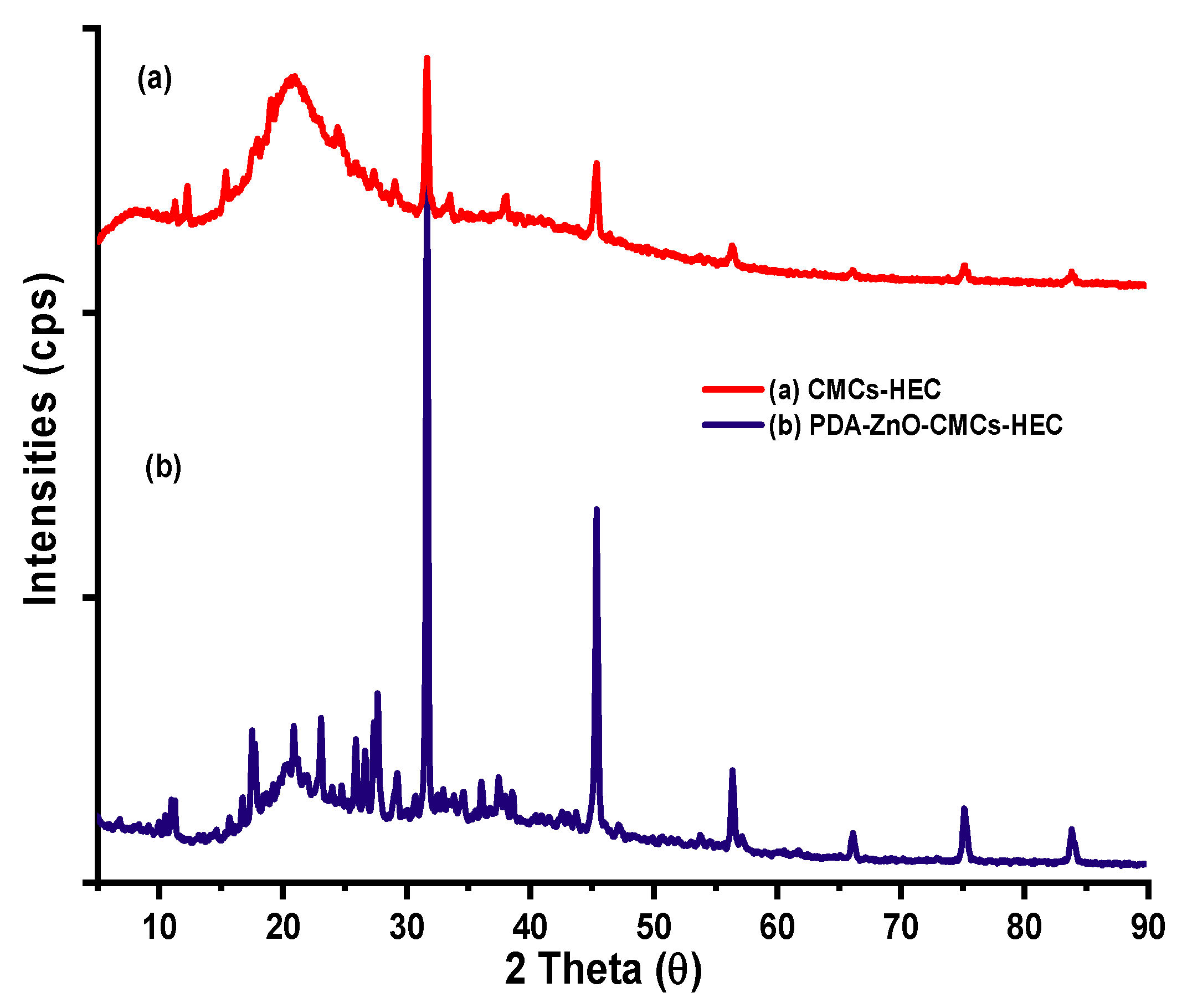
XRD results of CMCS-HEC composite
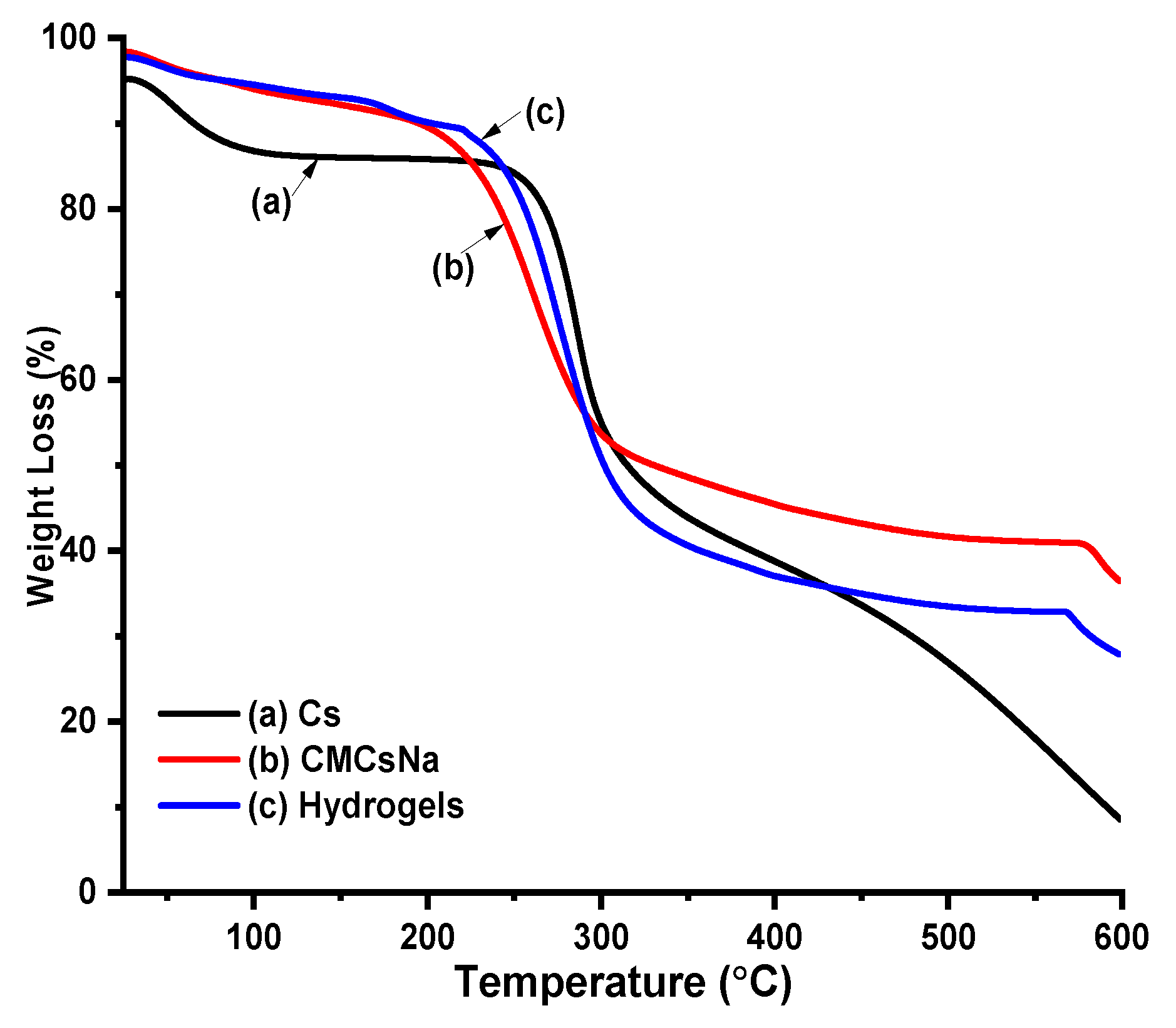
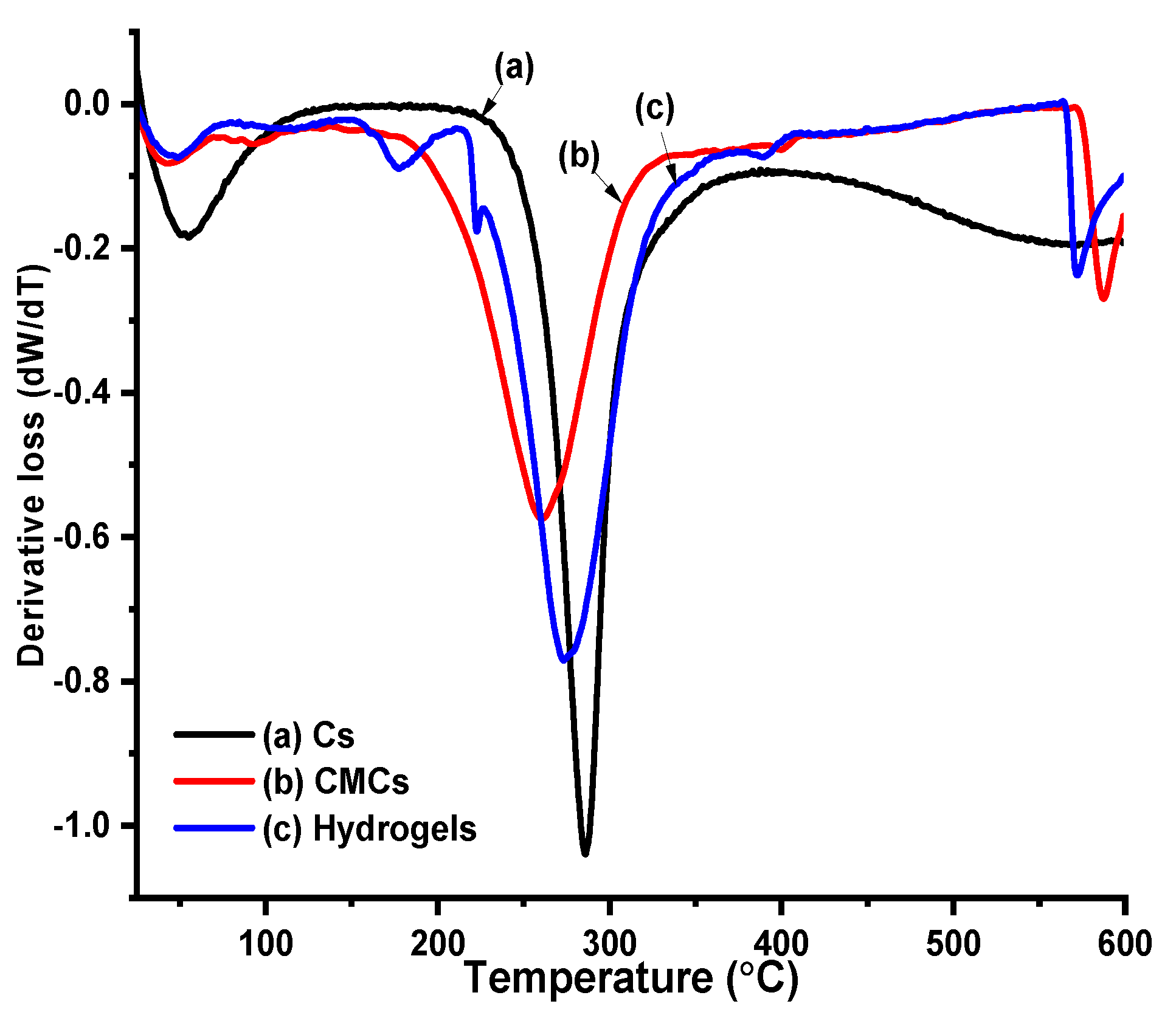
Thermal Profile of Cs, CMCS and Hydrogels
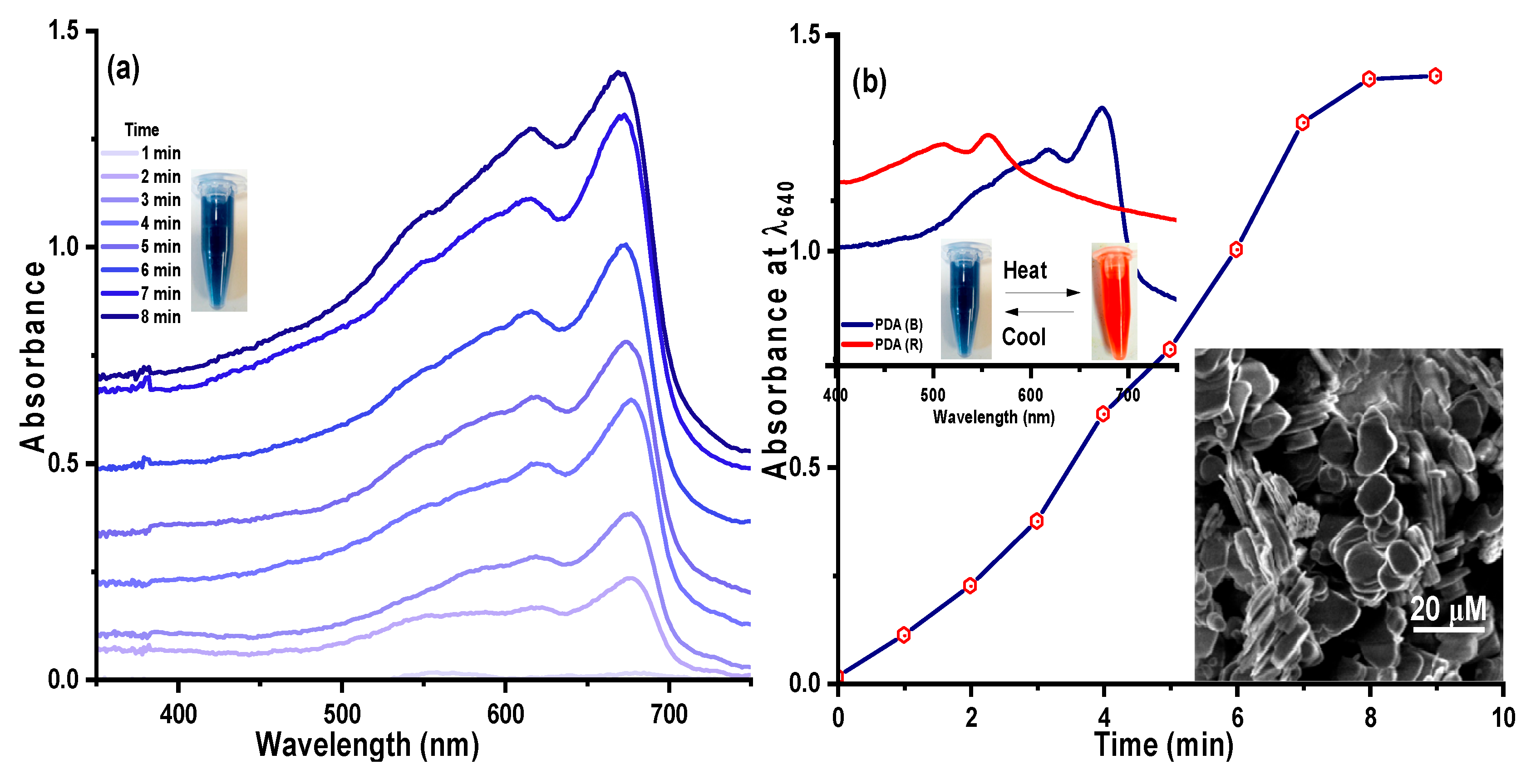
UV-Vis Spectra of polydiacetylene
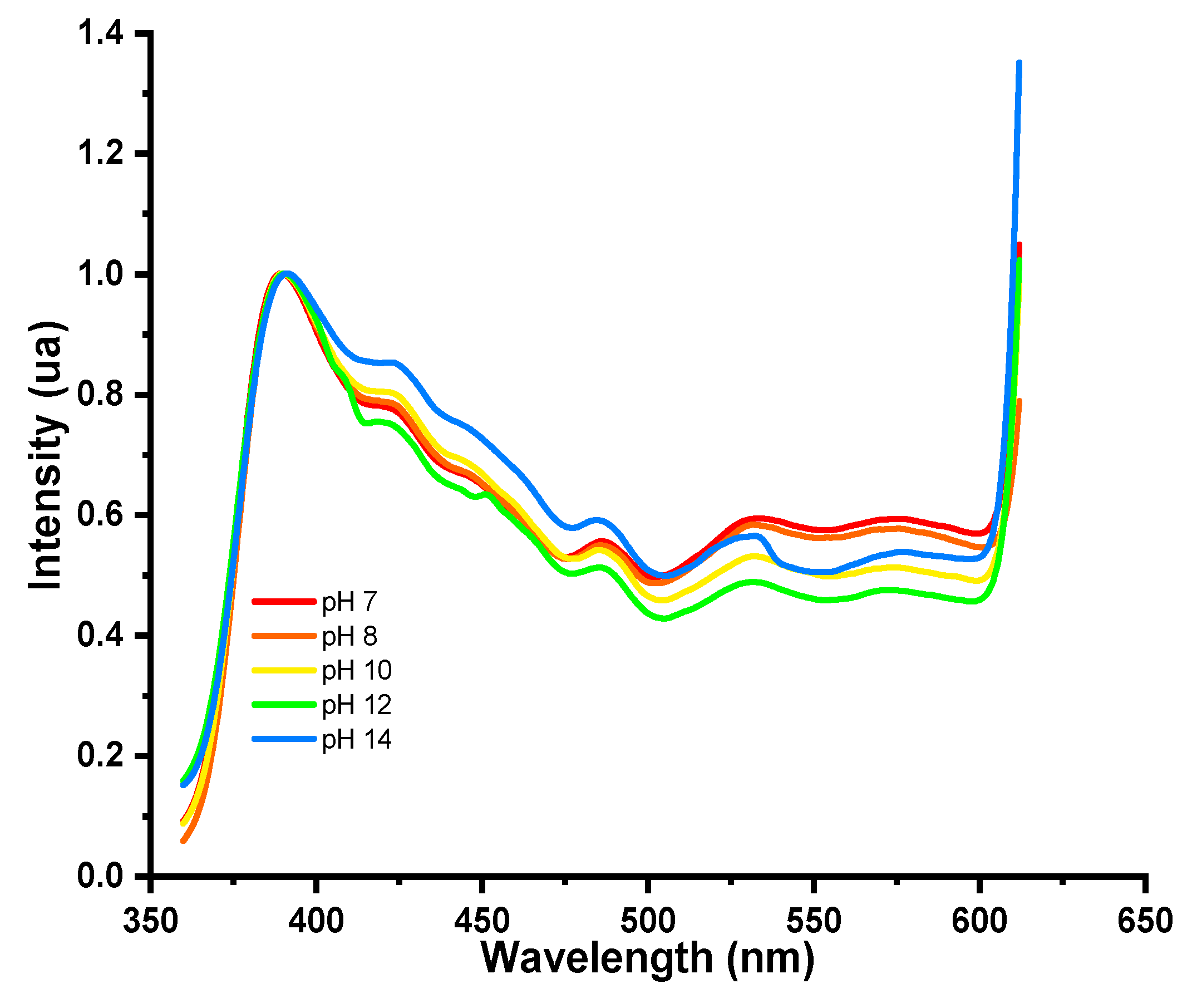
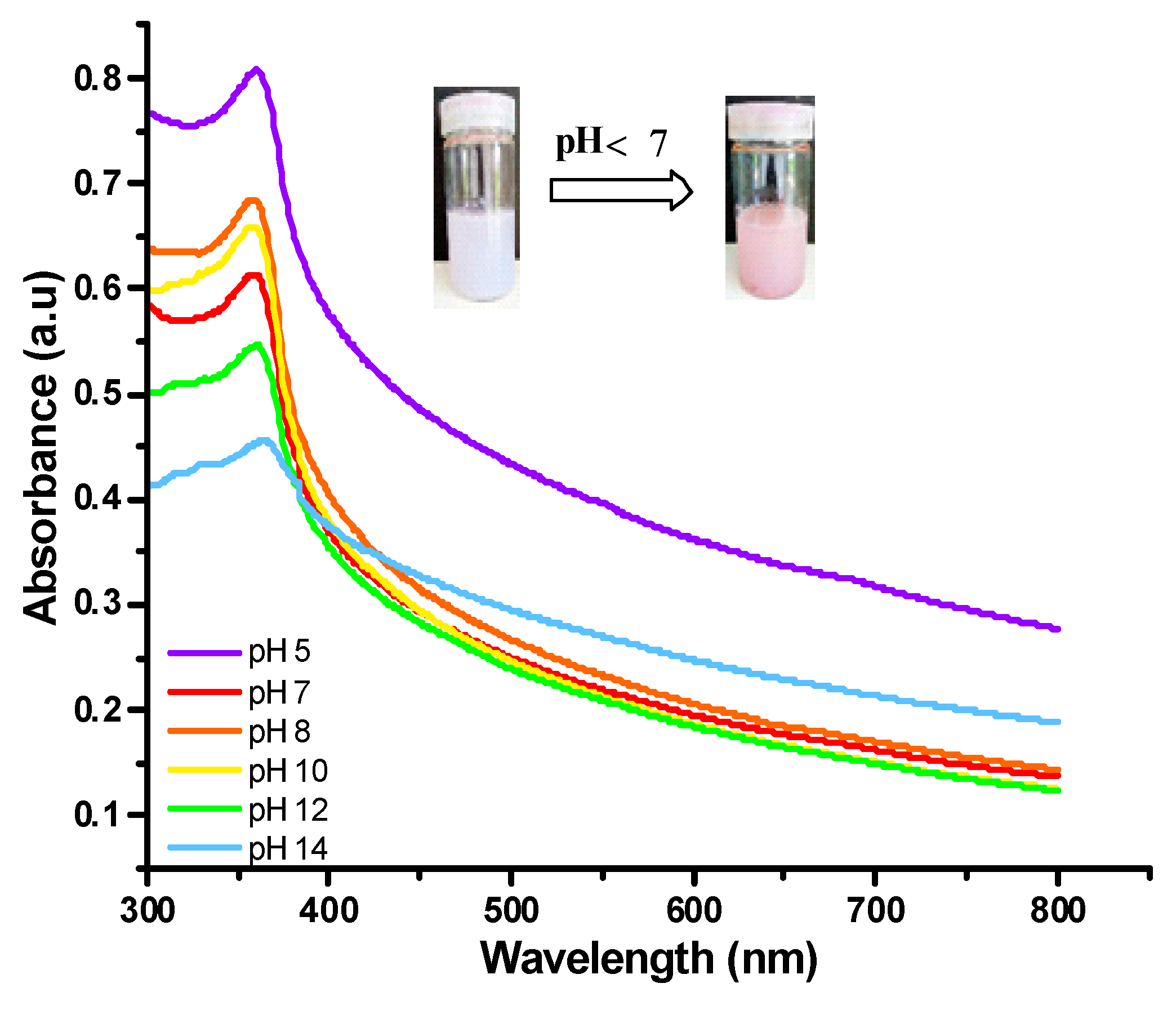
Colorimetric Response of the Hydrogels to pH
SEM Micrographs
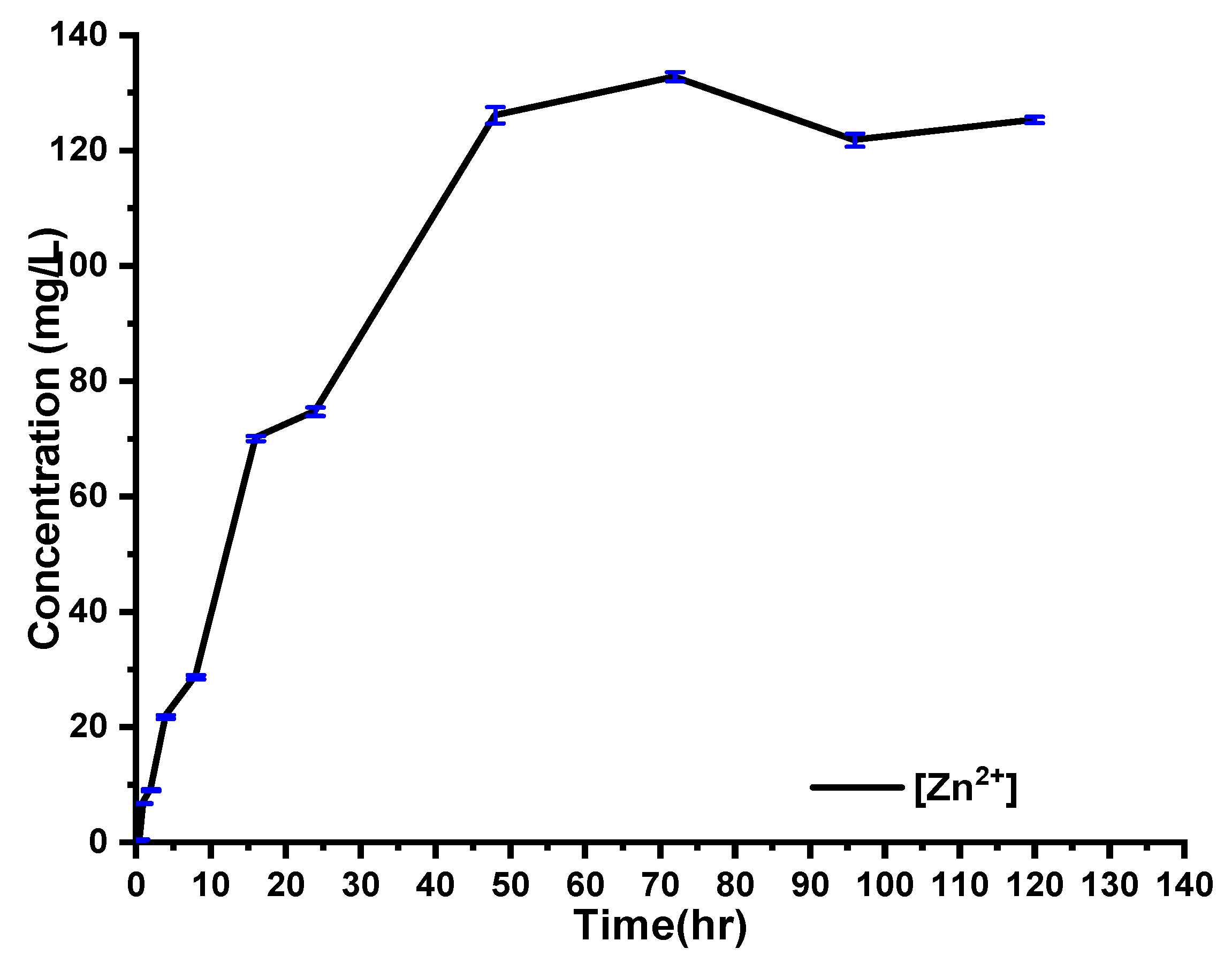
Kinetics of Zn2+ Release from the Nanocomposites
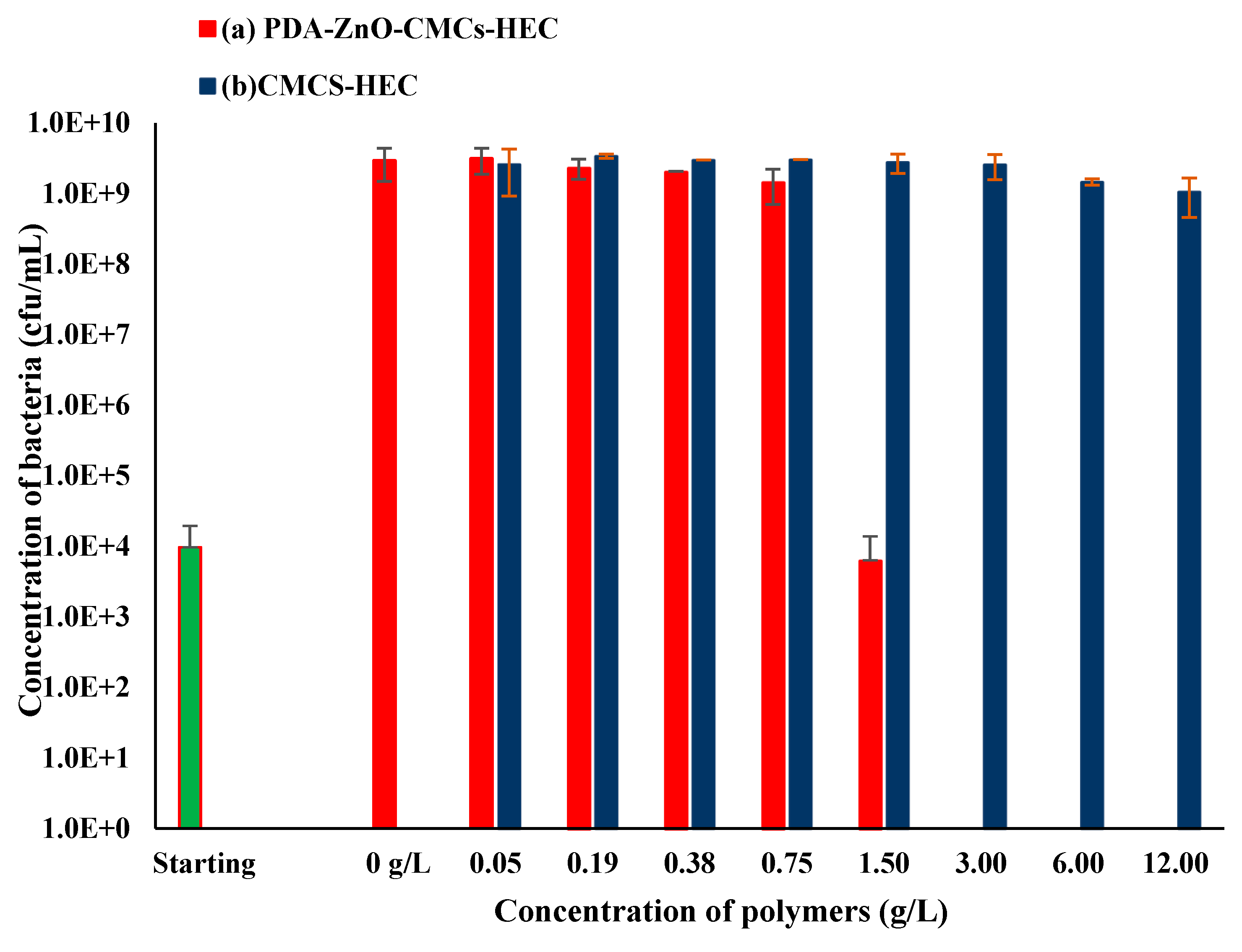
Antimicrobial Assays
Conclusion
Supplementary Materials
Acknowledgements
References
- Lee, H., Lee, J., Lee, S. G. & Doyle, P. S. Hydrogel-Based Colorimetric Assay for Multiplexed MicroRNA Detection in a Microfluidic Device. Anal. Chem. 92, 5750–5755 (2020). [CrossRef]
- Lebègue, E. et al. Responsive polydiacetylene vesicles for biosensing microorganisms. Sensors (Switzerland) vol. 18 599 (2018). [CrossRef]
- Al Sulaiman, D., Shapiro, S. J., Gomez-Marquez, J. & Doyle, P. S. High-Resolution Patterning of Hydrogel Sensing Motifs within Fibrous Substrates for Sensitive and Multiplexed Detection of Biomarkers. ACS Sensors 6, 203–211 (2021). [CrossRef]
- Shapiro, S. J., Dendukuri, D. & Doyle, P. S. Design of Hydrogel Particle Morphology for Rapid Bioassays. Anal. Chem. 90, 13572–13579 (2018). [CrossRef]
- Ning, Y. et al. Glow-type chemiluminescent hydrogels for point-of-care testing (POCT) of cholesterol. Analyst 146, 4775–4780 (2021). [CrossRef]
- Xu, C. et al. Multifunctional manganese-based carboxymethyl chitosan hydrogels for pH-triggered pesticide release and enhanced fungicidal activity. Carbohydr. Polym. 262, (2021). [CrossRef]
- Yapor, J. P. et al. Polydiacetylene Nanofiber Composites as a Colorimetric Sensor Responding to Escherichia coli and pH. ACS Omega 2, 7334–7342 (2017). [CrossRef]
- Suhail, M., Wu, P. C. & Minhas, M. U. Development and characterization of pH-sensitive chondroitin sulfate-co-poly(acrylic acid) hydrogels for controlled release of diclofenac sodium. J. Saudi Chem. Soc. 25, (2021). [CrossRef]
- Ciolacu, D. E., Nicu, R. & Ciolacu, F. Cellulose-based hydrogels as sustained drug-delivery systems. Materials (Basel). 13, 1–37 (2020). [CrossRef]
- Cao, J. et al. Ultrafast Fabrication of Self-Healing and Injectable Carboxymethyl Chitosan Hydrogel Dressing for Wound Healing. ACS Appl. Mater. Interfaces 13, 24095–24105 (2021). [CrossRef]
- Chandran, R., Mohd Tohit, E. R., Stanslas, J., Tuan Mahmood, T. M. & Salim, N. Factors influencing the swelling behaviour of polymethyl vinyl ether-co-maleic acid hydrogels crosslinked by polyethylene glycol. J. Drug Deliv. Sci. Technol. 68, (2022). [CrossRef]
- Alvarez-Lorenzo, C., Blanco-Fernandez, B., Puga, A. M. & Concheiro, A. Crosslinked ionic polysaccharides for stimuli-sensitive drug delivery. Advanced Drug Delivery Reviews vol. 65 1148–1171 (2013). [CrossRef]
- Wang, T., Chen, L., Shen, T. & Wu, D. Preparation and properties of a novel thermo-sensitive hydrogel based on chitosan/hydroxypropyl methylcellulose/glycerol. Int. J. Biol. Macromol. 93, 775–782 (2016). [CrossRef]
- Lin, P. et al. Preparation and properties of carboxymethyl chitosan/oxidized hydroxyethyl cellulose hydrogel. Int. J. Biol. Macromol. 162, 1692–1698 (2020). [CrossRef]
- Akar, E., Altinişik, A. & Seki, Y. Preparation of pH- and ionic-strength responsive biodegradable fumaric acid crosslinked carboxymethyl cellulose. Carbohydr. Polym. 90, 1634–1641 (2012). [CrossRef]
- Lin, G. et al. Elaboration of a feather keratin/carboxymethyl cellulose complex exhibiting pH sensitivity for sustained pesticide release. J. Appl. Polym. Sci. 136, 47160 (2019). [CrossRef]
- Singh, P., Baisthakur, P. & Yemul, O. S. Synthesis, characterization and application of crosslinked alginate as green packaging material. Heliyon 6, e03026 (2020). [CrossRef]
- Zhao, D. et al. 3D Printing Method for Tough Multifunctional Particle-Based Double-Network Hydrogels. ACS Appl. Mater. Interfaces 13, 13714–13723 (2021). [CrossRef]
- Fatimi, A., Okoro, O. V., Podstawczyk, D., Siminska-Stanny, J. & Shavandi, A. Natural Hydrogel-Based Bio-Inks for 3D Bioprinting in Tissue Engineering: A Review. Gels 8, 179 (2022). [CrossRef]
- Tsirigotis-Maniecka, M. Alginate-, Carboxymethyl Cellulose-, and κCarrageenan-Based Microparticles as Storage Vehicles for Cranberry Extract. Molecules 25, (2020). [CrossRef]
- Tavakoli, S., Kharaziha, M., Nemati, S. & Kalateh, A. Nanocomposite hydrogel based on carrageenan-coated starch/cellulose nanofibers as a hemorrhage control material. Carbohydr. Polym. 251, (2021). [CrossRef]
- Barrett-Catton, E., Ross, M. L. & Asuri, P. Multifunctional hydrogel nanocomposites for biomedical applications. Polymers vol. 13 (2021). [CrossRef]
- Mokhtari, H. et al. Recent advances in chemically-modified and hybrid carrageenan-based platforms for drug delivery, wound healing, and tissue engineering. Polymers vol. 13 1744 (2021). [CrossRef]
- Mo, F. et al. Energy-dissipative dual-crosslinked hydrogels for dynamically super-tough sensors. Sci. China Mater. 64, 2764–2776 (2021). [CrossRef]
- Chu, W., Nie, M., Ke, X., Luo, J. & Li, J. Recent Advances in Injectable Dual Crosslinking Hydrogels for Biomedical Applications. Macromol. Biosci. 21, (2021). [CrossRef]
- Pérez-Luna, V. H. & González-Reynoso, O. Encapsulation of biological agents in hydrogels for therapeutic applications. Gels 4, 1–30 (2018). [CrossRef]
- Budianto, E. & Amalia, A. Swelling behavior and mechanical properties of Chitosan-Poly(N-vinyl-pyrrolidone) hydrogels. J. Polym. Eng. 40, 551–560 (2020). [CrossRef]
- Huang, Q., Wu, W., Ai, K. & Liu, J. Highly Sensitive Polydiacetylene Ensembles for Biosensing and Bioimaging. Front. Chem. 8, 1–21 (2020). [CrossRef]
- Saenjaiban, A. et al. Novel color change film as a time–temperature indicator using polydiacetylene/silver nanoparticles embedded in carboxymethyl cellulose. Polymers (Basel). 12, 1–14 (2020). [CrossRef]
- Tang, J. et al. Fabrication of polydiacetylene particles using a solvent injection method. Mater. Adv. 1, 1745–1752 (2020). [CrossRef]
- Sawayama, J., Okitsu, T., Nakamata, A., Kawahara, Y. & Takeuchi, S. Hydrogel Glucose Sensor with In Vivo Stable Fluorescence Intensity Relying on Antioxidant Enzymes for Continuous Glucose Monitoring. iScience 23, 101243 (2020). [CrossRef]
- Kim, C. & Lee, K. Polydiacetylene (PDA) Liposome-Based Immunosensor for the Detection of Exosomes. Biomacromolecules 20, 3392–3398 (2019). [CrossRef]
- Weston, M., Tjandra, A. D. & Chandrawati, R. Tuning chromatic response, sensitivity, and specificity of polydiacetylene-based sensors. Polym. Chem. 11, 166–183 (2020). [CrossRef]
- Kimani, P. K., Kareru, P. G., Madivoli, S. E., Kairigo, P. K. & Maina, E. G. Comparative Study of Carboxymethyl Cellulose Synthesis from Selected Kenyan Biomass. Chem. Sci. Int. J. 17, 1–8 (2016).
- Seki, Y., Altinisik, A., Demircioğlu, B. & Tetik, C. Carboxymethylcellulose (CMC)-hydroxyethylcellulose (HEC) based hydrogels: Synthesis and characterization. Cellulose 21, 1689–1698 (2014). [CrossRef]
- Durpekova, S. et al. A Novel Hydrogel Based on Renewable Materials for Agricultural Application. Int. J. Polym. Sci. 2020, (2020). [CrossRef]
- Muslim, T., Rahman, M. H., Begum, Ho. A. & Rahman, M. A. Chitosan and Carboxymethyl Chitosan from Fish Scales of Labeo rohita. Dhaka Univ. J. Sci. 61, 145–148 (2013). [CrossRef]
- Wen, X. et al. Preparation of CMC/HEC Crosslinked Hydrogels for Drug Delivery. BioResources 10, (2015). [CrossRef]
- Li, Q. et al. A Colorimetric Strip for Rapid Detection and Real-Time Monitoring of Histamine in Fish Based on Self-Assembled Polydiacetylene Vesicles. Anal. Chem. 92, 1611–1617 (2020). [CrossRef]
- Huang, Q., Wu, W., Ai, K. & Liu, J. Highly Sensitive Polydiacetylene Ensembles for Biosensing and Bioimaging. Frontiers in Chemistry vol. 8 1079 (2020). [CrossRef]
- Madivoli, E. S., Kareru, P. G., Gachanja, A. N., Mugo, S. M. & Makhanu, D. S. Synthesis and characterization of dialdehyde cellulose nanofibers from O. sativa husks. SN Appl. Sci. 1, 1–7 (2019). [CrossRef]
- Kian, L. K., Jawaid, M., Ariffin, H. & Alothman, O. Y. Isolation and characterization of microcrystalline cellulose from roselle fibers. Int. J. Biol. Macromol. 103, 931–940 (2017). [CrossRef]
- Otenda, B. V et al. Starch-Hibiscus-Cellulose Nanofibrils Composite Films as a Model Antimicrobial Food Packaging Material. J. Nat. Fibers 1–14 (2022). [CrossRef]
- Priebe, M. et al. Antimicrobial silver-filled silica nanorattles with low immunotoxicity in dendritic cells. Nanomedicine Nanotechnology, Biol. Med. 13, 11–22 (2017). [CrossRef]
- Hérault, N. et al. Silver-containing titanium dioxide nanocapsules for combating multidrug-resistant bacteria. Int. J. Nanomedicine 15, 1267–1281 (2020). [CrossRef]
- Mali, K. K., Dhawale, S. C., Dias, R. J., Dhane, N. S. & Ghorpade, V. S. Citric acid crosslinked carboxymethyl cellulose-based composite hydrogel films for drug delivery. Indian J. Pharm. Sci. 80, 657–667 (2018). [CrossRef]
- Chen, Y.-C., Ho, H.-O., Liu, D.-Z., Siow, W.-S. & Sheu, M.-T. Swelling/Floating Capability and Drug Release Characterizations of Gastroretentive Drug Delivery System Based on a Combination of Hydroxyethyl Cellulose and Sodium Carboxymethyl Cellulose. PLoS One 10, e0116914 (2015). [CrossRef]
- Barbucci, R., Magnani, A. & Consumi, M. Swelling Behavior of Carboxymethylcellulose Hydrogels in Relation to Cross-Linking, pH, and Charge Density. Macromolecules 33, 7475–7480 (2000). [CrossRef]
- Bashir, S. et al. Fundamental concepts of hydrogels: Synthesis, properties, and their applications. Polymers (Basel). 12, 1–60 (2020). [CrossRef]
- Astrini, N., Anah, L. & Haryono, A. Crosslinking Parameter on the Preparation of Cellulose Based Hydrogel with Divynilsulfone. Procedia Chem. 4, 275–281 (2012). [CrossRef]
- Raucci, M. G. et al. Effect of citric acid crosslinking cellulose-based hydrogels on osteogenic differentiation. J. Biomed. Mater. Res. - Part A 103, 2045–2056 (2015). [CrossRef]
- Jing, X. et al. Highly Stretchable, Self-Healable, Freezing-Tolerant, and Transparent Polyacrylic Acid/Nanochitin Composite Hydrogel for Self-Powered Multifunctional Sensors. ACS Sustain. Chem. Eng. 9, 9209–9220 (2021). [CrossRef]
- Cao, J., You, J., Zhang, L. & Zhou, J. Homogeneous synthesis and characterization of chitosan ethers prepared in aqueous alkali/urea solutions. Carbohydr. Polym. 185, 138–144 (2018). [CrossRef]
- Hao, Y., Zhao, W., Zhang, H., Zheng, W. & Zhou, Q. Carboxymethyl chitosan-based hydrogels containing fibroblast growth factors for triggering diabetic wound healing. Carbohydr. Polym. 287, 119336 (2022). [CrossRef]
- Socrates, G. Infrared and Raman characteristic group frequencies, third edition. John Wiley & Sons, LTD (2001). [CrossRef]
- Madivoli, E. S., Kareru, P. G., Gichuki, J. & Elbagoury, M. M. Cellulose nanofibrils and silver nanoparticles enhances the mechanical and antimicrobial properties of polyvinyl alcohol nanocomposite film. Sci. Rep. 12, 1–13 (2022). [CrossRef]
- Kim, J. et al. Solid-state colorimetric polydiacetylene liposome biosensor sensitized by gold nanoparticles. Analyst 146, 1682–1688 (2021). [CrossRef]
- Abreu, F. D. & Campana-Filho, S. P. Preparation and characterization of carboxymethylchitosan. Polimeros 15, 79–83.
- Tian, X., Wu, S., Zhang, Q. & Zou, G. Colorimetric sensor for fine differentiation of organic solvents based on only one kind of polydiacetylene coated on polymer optical fiber. IEEE Sens. J. 12, 1946–1949 (2012). [CrossRef]
- Jang, S. et al. Polydiacetylene-Based Hydrogel Beads as Colorimetric Sensors for the Detection of Biogenic Amines in Spoiled Food. SSRN Electron. J. (2022). [CrossRef]
- Liu, G., Ding, Z., Yuan, Q., Xie, H. & Gu, Z. Multi-Layered Hydrogels for Biomedical Applications. Front. Chem. 6, 1–10 (2018). [CrossRef]
- Jin, L. et al. Research progress in the multilayer hydrogels. Gels 7, (2021). [CrossRef]
- Nie, J., Wang, Z. & Hu, Q. Chitosan Hydrogel Structure Modulated by Metal Ions. Sci. Rep. 6, 1–8 (2016). [CrossRef]
- Masood, N. et al. Silver nanoparticle impregnated chitosan-PEG hydrogel enhances wound healing in diabetes induced rabbits. Int. J. Pharm. 559, 23–36 (2019). [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




