Submitted:
08 March 2023
Posted:
09 March 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Naterstad, I.F.; Joensen, J.; Bjordal, J.M.; Couppé, C.; Lopes-Martins, R.A.B.; Stausholm, M.B. Efficacy of low-level laser therapy in patients with lower extremity tendinopathy or plantar fasciitis: Systematic review and meta-analysis of randomised controlled trials. BMJ Open 2022, 12, e059479. [Google Scholar] [CrossRef] [PubMed]
- Mussttaf, R.A.; Jenkins, D.F.L.; Jha, A.N. Assessing the impact of low level laser therapy (LLLT) on biological systems: A review. Int. J. Radiat. Biol. 2019, 95, 120–143. [Google Scholar] [CrossRef] [PubMed]
- Bjordal, J.M.; Lopes-Martins, R.A.B.; Joensen, J.; Iversen, V.V. The anti-inflammatory mechanism of low level laser therapy and its relevance for clinical use in physiotherapy. Phys. Ther. Rev. 2010, 15, 286–293. [Google Scholar] [CrossRef]
- Dompe, C.; Moncrieff, L.; Matys, J.; Grzech-Leśniak, K.; Kocherova, I.; Bryja, A.; Bruska, M.; Dominiak, M.; Mozdziak, P.; Skiba, T.H.I.; et al. Photobiomodulation - underlying mechanism and clinical applications. J. Clin. Med. 2020, 9, 1724. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, C. Improving extracorporeal shock wave therapy with 904 or 905 nm pulsed, high power laser pretreatment. Preprints 2021, 2021010138. [Google Scholar] [CrossRef]
- Chung, H.; Dai, T.; Sharma, S.K.; Huang, Y.-Y.; Carroll, J.D.; Hamblin, M.R. The nuts and bolts of low-level laser (light) therapy. Ann. Biomed. Eng. 2012, 40, 516–533. [Google Scholar] [CrossRef]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Biol. 2013, 58, R37–R61. [Google Scholar] [CrossRef]
- Kaub, L.; Schmitz, C. More than ninety percent of the light energy emitted by near-infrared laser therapy devices used to treat musculoskeletal disorders is absorbed within the first ten millimeters of biological tissue. Biomedicines 2022, 10. [Google Scholar] [CrossRef]
- Hudson, D.E.; Hudson, D.O.; Wininger, J.M.; Richardson, B.D. Penetration of laser light at 808 and 980 nm in bovine tissue samples. Photomed. Laser Surg. 2013, 31, 163–168. [Google Scholar] [CrossRef]
- Han, D.; Xu, J.; Wang, Z.; Yang, N.; Li, X.; Qian, Y.; Li, G.; Dai, R.; Xu, S. Penetrating effect of high-intensity infrared laser pulses through body tissue. RSC Adv. 2018, 8, 32344–32357. [Google Scholar] [CrossRef]
- Mineroff, J.; Austin, E.; Jagdeo, J. Cutaneous effects of photobiomodulation with 1072 nm light. Arch. Dermatol. Res. 2022. [Google Scholar] [CrossRef]
- Ash, C.; Dubec, M.; Donne, K.; Bashford, T. Effect of wavelength and beam width on penetration in light-tissue interaction using computational methods. Lasers Med. Sci. 2017, 32, 1909–1918. [Google Scholar] [CrossRef]
- Bashkatov, A.N.; Genina, E.A.; Kochubey, V.I.; Tuchin, V.V. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000nm. J. Phys. D App. Phys. 2005, 38, 2543. [Google Scholar] [CrossRef]
- Van Veen, R.; Sterenborg, H.; Pifferi, A.; Torricelli, A.; Cubeddu, R. Determination of VIS- NIR absorption coefficients of mammalian fat, with time- and spatially resolved diffuse reflectance and transmission spectroscopy. In Biomedical Topical Meetings, Miami Beach, FL, USA, 14-17 April 2004; Optica Publishing Group: Washington, DC, USA; p. SF4. 2004; ISBN 1-55752-769-5. [Google Scholar]
- Deng, R.; He, Y.; Qin, Y.; Chen, Q.; Chen, L. Measuring pure water absorption coefficient in the near-infrared spectrum (900—2500 nm). J. Remote Sens. 2012, 16, 192–206. [Google Scholar] [CrossRef]
- Liebert, A.; Waddington, G.; Bicknell, B.; Chow, R.; Adams, R. Quantification of the absorption of low-level 904 nm superpulsed laser, light as a function of skin colour. In Proceedings of the 9th World Association for Laser Therapy Congress, Surfers Paradise, Australia, 28–30 September 2012; Laakso, E.L., Young, C., Eds.; Medimond: Gold Coast, Australia, 2012; pp. 11–15. [Google Scholar]
- Hashmi, J.T.; Huang, Y.-Y.; Sharma, S.K.; Kurup, D.B.; de Taboada, L.; Carroll, J.D.; Hamblin, M.R. Effect of pulsing in low-level light therapy. Lasers Surg. Med. 2010, 42, 450–466. [Google Scholar] [CrossRef] [PubMed]
- Kaub, L.; Schmitz, C. Spread of the optical power emission of three units each of two different laser therapy devices used in sports medicine, which cannot be assessed by the users, shown by means of high-fidelity laser measurement technology. Biomedicines 2023, 11, 585. [Google Scholar] [CrossRef]
- Stolik, S.; Delgado, J.A.; Perez, A.; Anasagasti, L. Measurement of the penetration depths of red and near infrared light in human “ex vivo” tissues. J. Photochem. Photobiol. B 2000, 57, 90–93. [Google Scholar] [CrossRef]
- Arslan, H.; Doluğan, Y.B. Optical penetration depths and fluence distributions in chicken breast and liver tissues. Opt. Spectrosc. 2019, 127, 763–768. [Google Scholar] [CrossRef]
- Barbora, A.; Bohar, O.; Sivan, A.A.; Magory, E.; Nause, A.; Minnes, R. Higher pulse frequency of near-infrared laser irradiation increases penetration depth for novel biomedical applications. PLoS ONE 2021, 16, e0245350. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, R.I.; Guirro, E.C.d.O.; Bachmann, L.; Brandino, H.E.; Guirro, R.R.d.J. Analysis of low-level laser transmission at wavelengths 660, 830 and 904 nm in biological tissue samples. J. Photochem. Photobiol. B 2020, 209, 111914. [Google Scholar] [CrossRef]
- Nussbaum, E.L.; van Zuylen, J.; Jing, F. Transmission of light through human skin folds during phototherapy: Effects of physical characteristics, irradiation wavelength, and skin-diode coupling. Physiother. Can. 2007, 59, 194–207. [Google Scholar] [CrossRef]
- Li, T.; Xue, C.; Wang, P.; Li, Y.; Wu, L. Photon penetration depth in human brain for light stimulation and treatment: A realistic Monte Carlo simulation study. J. Innov. Opt. Health Sci. 2017, 10, 1743002. [Google Scholar] [CrossRef]
- Oshina, I.; Spigulis, J. Beer-Lambert law for optical tissue diagnostics: Current state of the art and the main limitations. J. Biomed. Opt. 2021, 26, 100901. [Google Scholar] [CrossRef]
- Zhao, Z.; Fairchild, P.W. Dependence of light transmission through human skin on incident beam diameter at different wavelengths. In Laser-Tissue Interaction IX, Proceedings of the SPIE 3254, BiOS ’98 International Biomedical Optics Symposium, San Jose, CA, USA, 13 May 1998; SPIE: Manchester, UK, 1998. [Google Scholar] [CrossRef]
- Moriyama, Y.; Nguyen, J.; Akens, M.; Moriyama, E.H.; Lilge, L. In vivo effects of low level laser therapy on inducible nitric oxide synthase. Lasers Surg. Med. 2009, 41, 227–231. [Google Scholar] [CrossRef]
- De Freitas, L.F.; Hamblin, M.R. Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE J. Sel. Top. Quantum Electron. 2016, 22. [Google Scholar] [CrossRef] [PubMed]
- Mezawa, S.; Iwata, K.; Naito, K.; Kamogawa, H. The possible analgesic effect of soft-laser irradiation on heat nociceptors in the cat tongue. Arch. Oral Biol. 1988, 33, 693–694. [Google Scholar] [CrossRef] [PubMed]
- De Freitas Rodrigues, A.; De Oliveira Martins, D.; Chacur, M.; Luz, J.G.C. The effectiveness of photobiomodulation in the management of temporomandibular pain sensitivity in rats: Behavioral and neurochemical effects. Lasers Med. Sci. 2020, 35, 447–453. [Google Scholar] [CrossRef]
- De Oliveira, M.E.; Da Silva, J.T.; Brioschi, M.L.; Chacur, M. Effects of photobiomodulation therapy on neuropathic pain in rats: Evaluation of nociceptive mediators and infrared thermography. Lasers Med. Sci. 2021, 36, 1461–1467. [Google Scholar] [CrossRef]
- Snijdelaar, D.G.; Dirksen, R.; Slappendel, R.; Crul, B.J. Substance P. Eur. J. Pain 2000, 4, 121–135. [Google Scholar] [CrossRef]
- Sann, H.; Pierau, F.K. Efferent functions of C-fiber nociceptors. Z. Rheumatol. 1998, 57 (Suppl. S2), 8–13. [Google Scholar] [CrossRef]
- Foreman, J.C. Peptides and neurogenic inflammation. Br. Med. Bull. 1987, 43, 386–400. [Google Scholar] [CrossRef]
- Julius, D. TRP channels and pain. In Neurobiology of TRP Channels, 1st ed.; Rosenbaum Emir, T.L., Ed.; CRC Press: Milton, ON, USA, 2017; pp. 125–148. ISBN 978-1-4987-5524-5. [Google Scholar]
- Han, D.-S.; Lee, C.-H.; Shieh, Y.-D.; Chen, C.-C. Involvement of substance P in the analgesic effect of low-level laser therapy in a mouse model of chronic widespread muscle pain. Pain Med. 2019, 20, 1963–1970. [Google Scholar] [CrossRef]
- Bjordal, J.M.; Lopes-Martins, R.Á.B.; Iversen, V.V. A randomised, placebo controlled trial of low level laser therapy for activated Achilles tendinitis with microdialysis measurement of peritendinous prostaglandin E2 concentrations. Br. J. Sports Med. 2006, 40, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Bergqvist, F.; Carr, A.J.; Wheway, K.; Watkins, B.; Oppermann, U.; Jakobsson, P.-J.; Dakin, S.G. Divergent roles of prostacyclin and PGE2 in human tendinopathy. Arthritis Res. Ther. 2019, 21, 74. [Google Scholar] [CrossRef]
- Momin, A.; McNaughton, P.A. Regulation of firing frequency in nociceptive neurons by pro-inflammatory mediators. Exp. Brain Res. 2009, 196, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.; Kim, M.; Hwang, S.W. Molecular mechanisms underlying the actions of arachidonic acid-derived prostaglandins on peripheral nociception. J. Neuroinflammation 2020, 17, 30. [Google Scholar] [CrossRef]
- Rizzi, C.F.; Mauriz, J.L.; Freitas Corrêa, D.S.; Moreira, A.J.; Zettler, C.G.; Filippin, L.I.; Marroni, N.P.; González-Gallego, J. Effects of low-level laser therapy (LLLT) on the nuclear factor (NF)-kappaB signaling pathway in traumatized muscle. Lasers Surg. Med. 2006, 38, 704–713. [Google Scholar] [CrossRef]
- Monaco, C.; Andreakos, E.; Kiriakidis, S.; Mauri, C.; Bicknell, C.; Foxwell, B.; Cheshire, N.; Paleolog, E.; Feldmann, M. Canonical pathway of nuclear factor kappa B activation selectively regulates proinflammatory and prothrombotic responses in human atherosclerosis. Proc. Natl. Acad. Sci. USA 2004, 101, 5634–5639. [Google Scholar] [CrossRef]
- Barnes, P.J.; Karin, M. Nuclear factor-kappa B: A pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med. 1997, 336, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Kim, J.; Kim, S.; Karna, S.; Won, J.; Jeon, S.M.; Kim, S.Y.; Choi, Y.; Choi, H.; Kim, O. Modulation of lipopolysaccharide-induced NF-κB signaling pathway by 635 nm irradiation via heat shock protein 27 in human gingival fibroblast cells. Photochem. Photobiol. 2013, 89, 199–207. [Google Scholar] [CrossRef]
- Girasol, C.E.; Braz, G.d.A.; Bachmann, L.; Celli, J.; Guirro, R.R.d.J. Laser light sources for photobiomodulation: The role of power and beam characterization in treatment accuracy and reliability. PLoS ONE 2022, 17, e0266193. [Google Scholar] [CrossRef] [PubMed]
- Parker, S.; Cronshaw, M.; Grootveld, M.; George, R.; Anagnostaki, E.; Mylona, V.; Chala, M.; Walsh, L. The influence of delivery power losses and full operating parametry on the effectiveness of diode visible-near infra-red (445-1064 nm) laser therapy in dentistry-a multi-centre investigation. Lasers Med. Sci. 2022, 37, 2249–2257. [Google Scholar] [CrossRef] [PubMed]

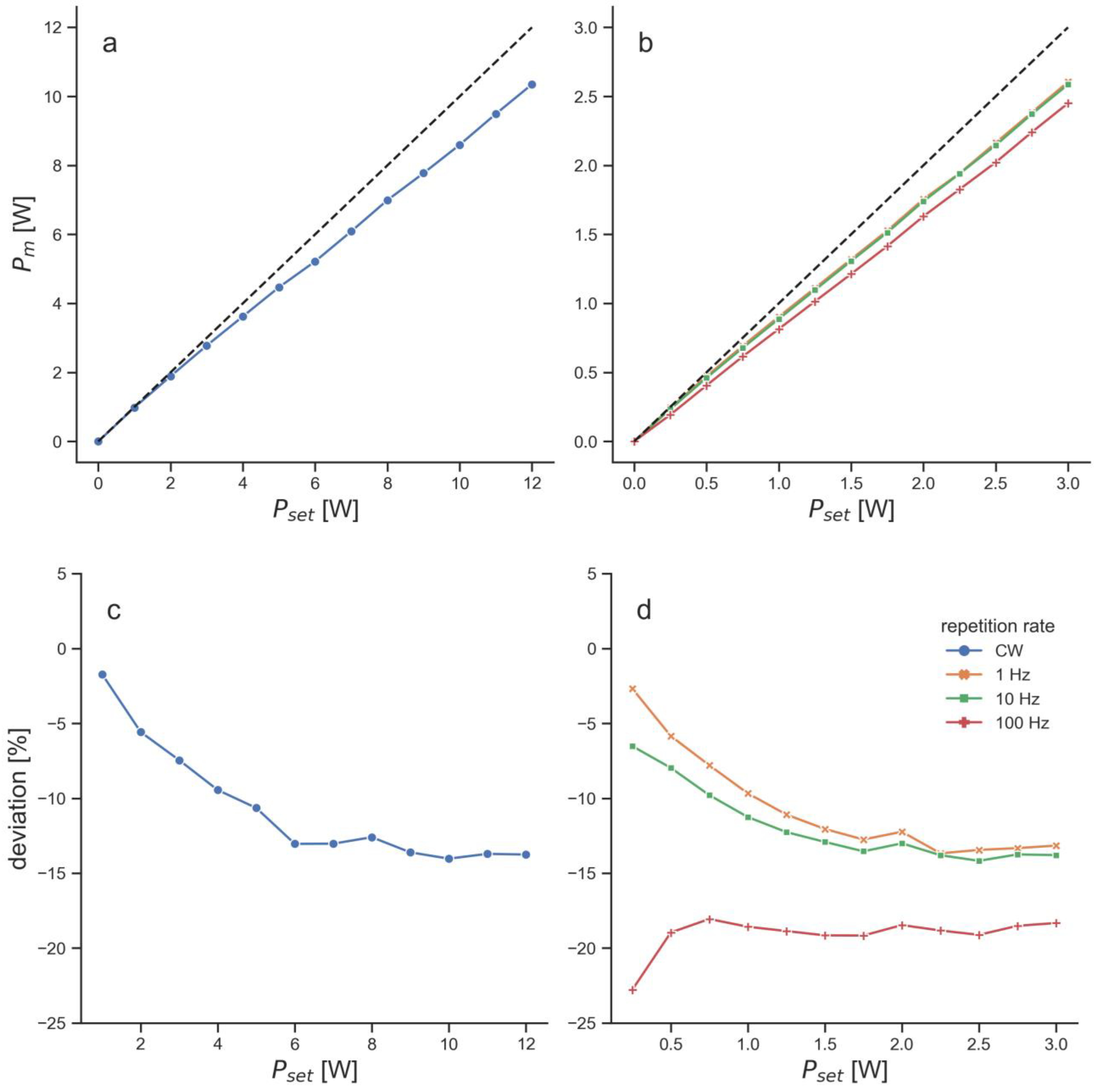
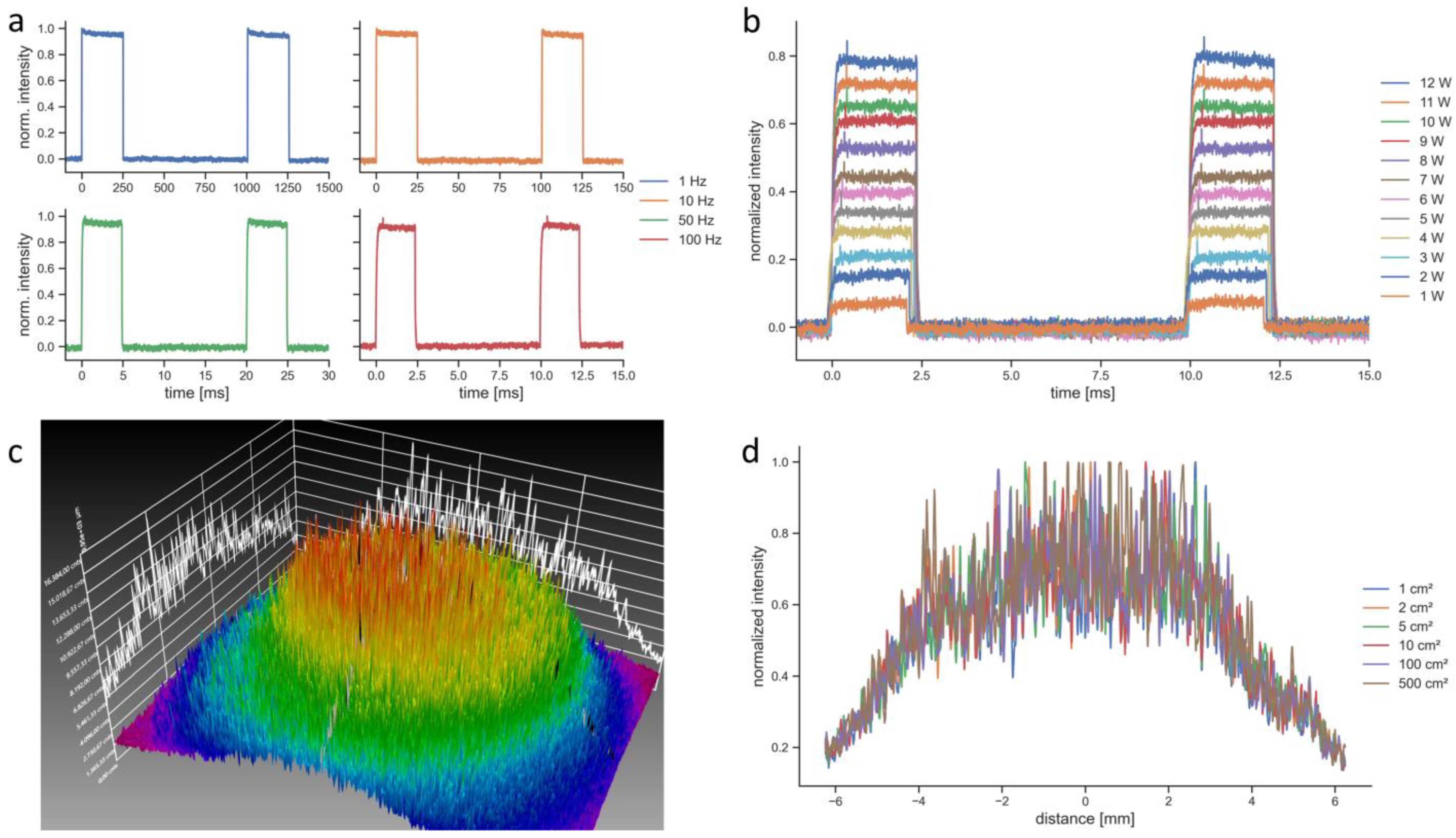
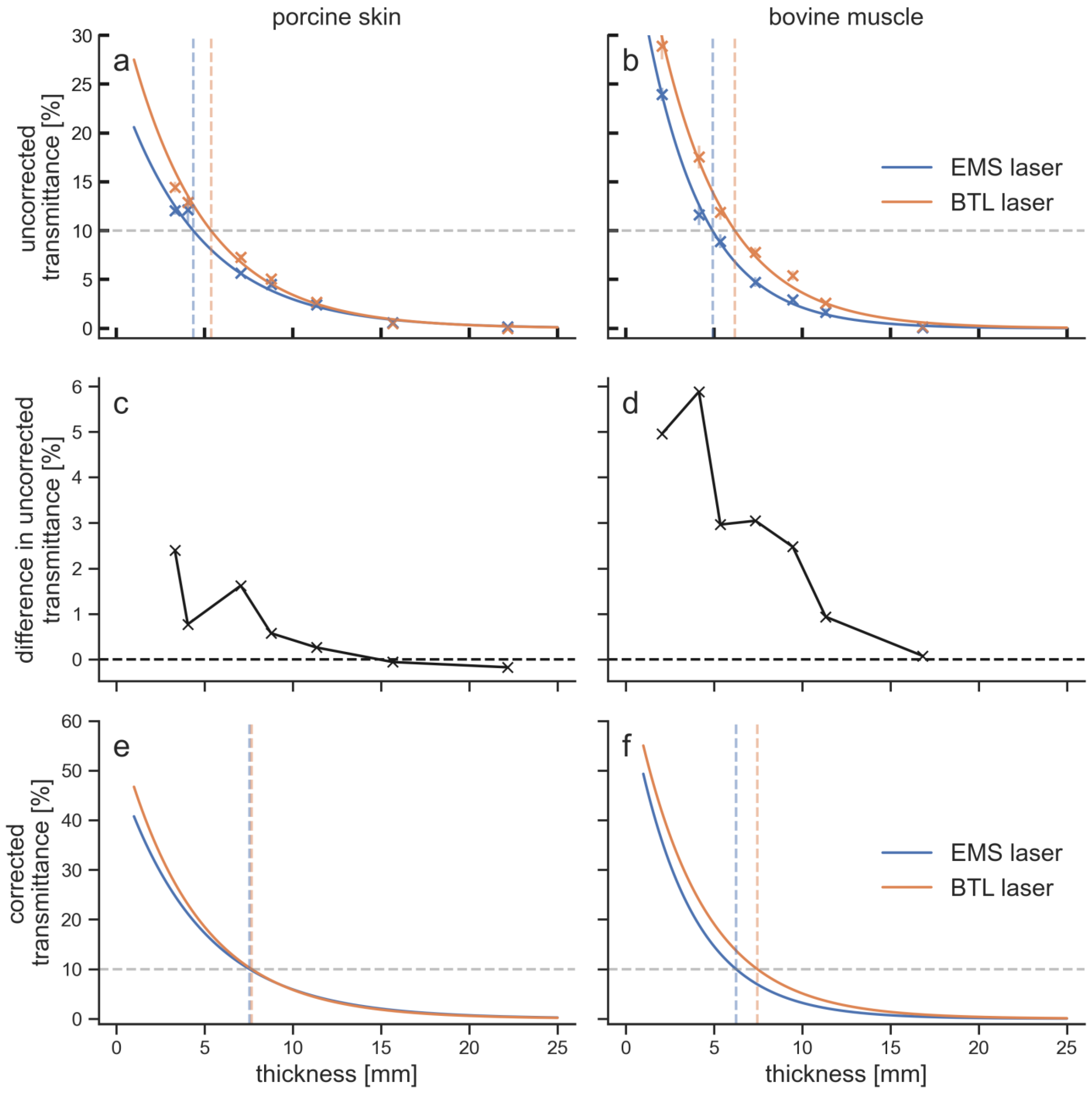
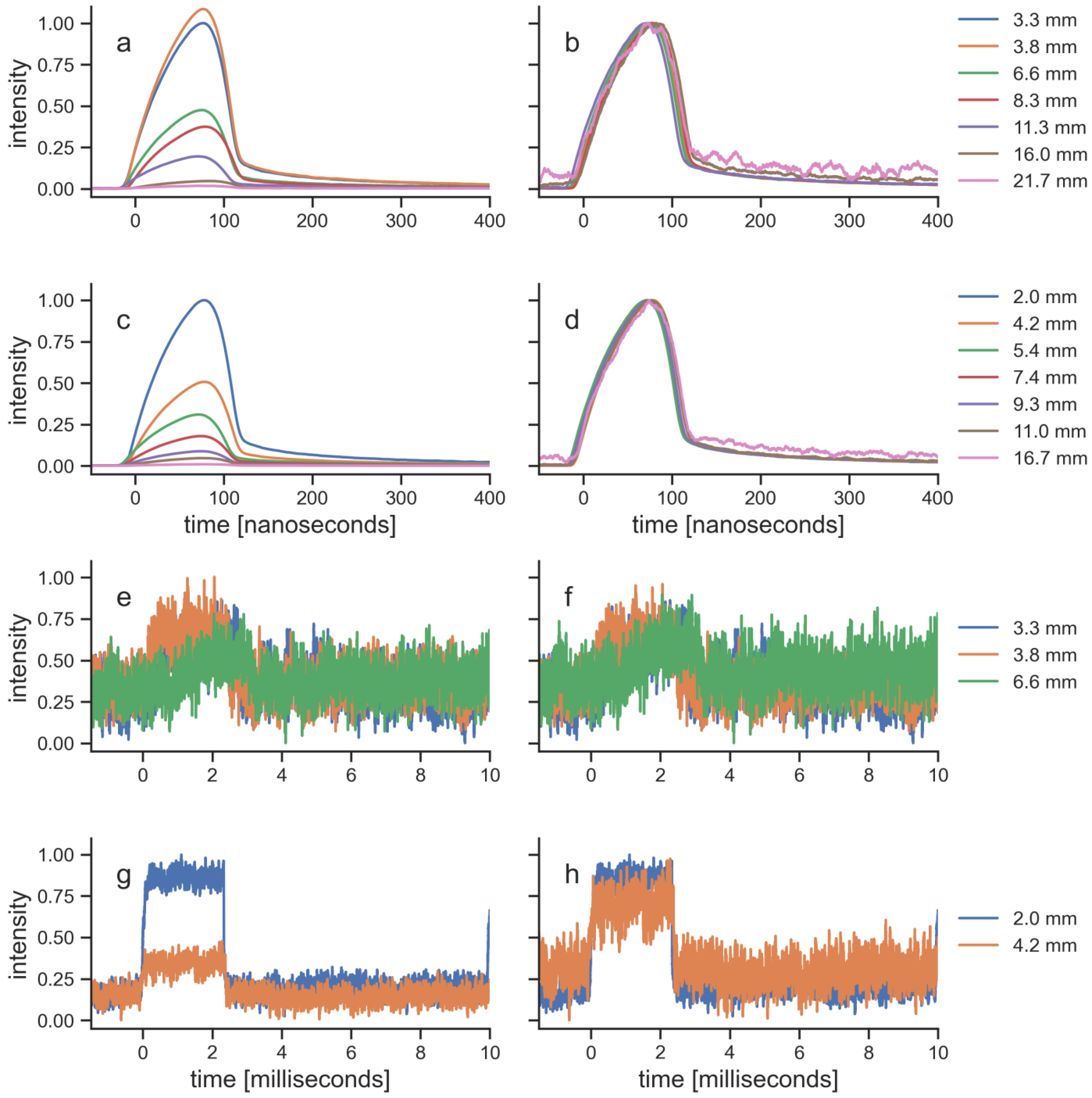
| Laser therapy device | EMS laser | BTL laser |
|---|---|---|
| Wavelength [nm] | 905 | 1064 |
| Modes | PW | CW/PW/SP |
| [W] | 1.2 | 12 |
| [W] | 300 | 12 |
| Pulse length | 100 ns | 2.5 ms – 250 ms |
| Repetition rates [Hz] | 5,000 – 40,000 | 1 – 100 |
| Penetration Depth [mm] for | Transmittance [%] at | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Laser | Tissue | 15% | 10% | 5% | 1% | 5 mm | 10 mm | 15 mm | 20 mm |
| uncorrected | |||||||||
| BTL laser | porcine skin | 3.6 | 5.4 | 8.4 | 15.3 | 10.9 | 3.4 | 1.1 | 0.3 |
| bovine muscle | 4.7 | 6.2 | 8.8 | 14.9 | 13.7 | 3.7 | 1.0 | 0.3 | |
| EMS laser | porcine skin | 2.5 | 4.4 | 7.6 | 15.1 | 8.7 | 3.0 | 1.0 | 0.3 |
| bovine muscle | 3.6 | 4.9 | 7.2 | 12.5 | 9.8 | 2.1 | 0.5 | 0.1 | |
| corrected | |||||||||
| BTL laser | porcine skin | 5.9 | 7.7 | 10.7 | 17.6 | 18.5 | 5.8 | 1.8 | 0.6 |
| bovine muscle | 5.9 | 7.5 | 10.1 | 16.2 | 19.1 | 5.1 | 1.4 | 0.4 | |
| EMS laser | porcine skin | 5.7 | 7.5 | 10.8 | 18.3 | 17.3 | 5.9 | 2.0 | 0.7 |
| bovine muscle | 4.9 | 6.3 | 8.5 | 13.8 | 14.6 | 3.2 | 0.7 | 0.2 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




