Submitted:
11 May 2023
Posted:
11 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Calcium Phosphates
3. Deposition Methods
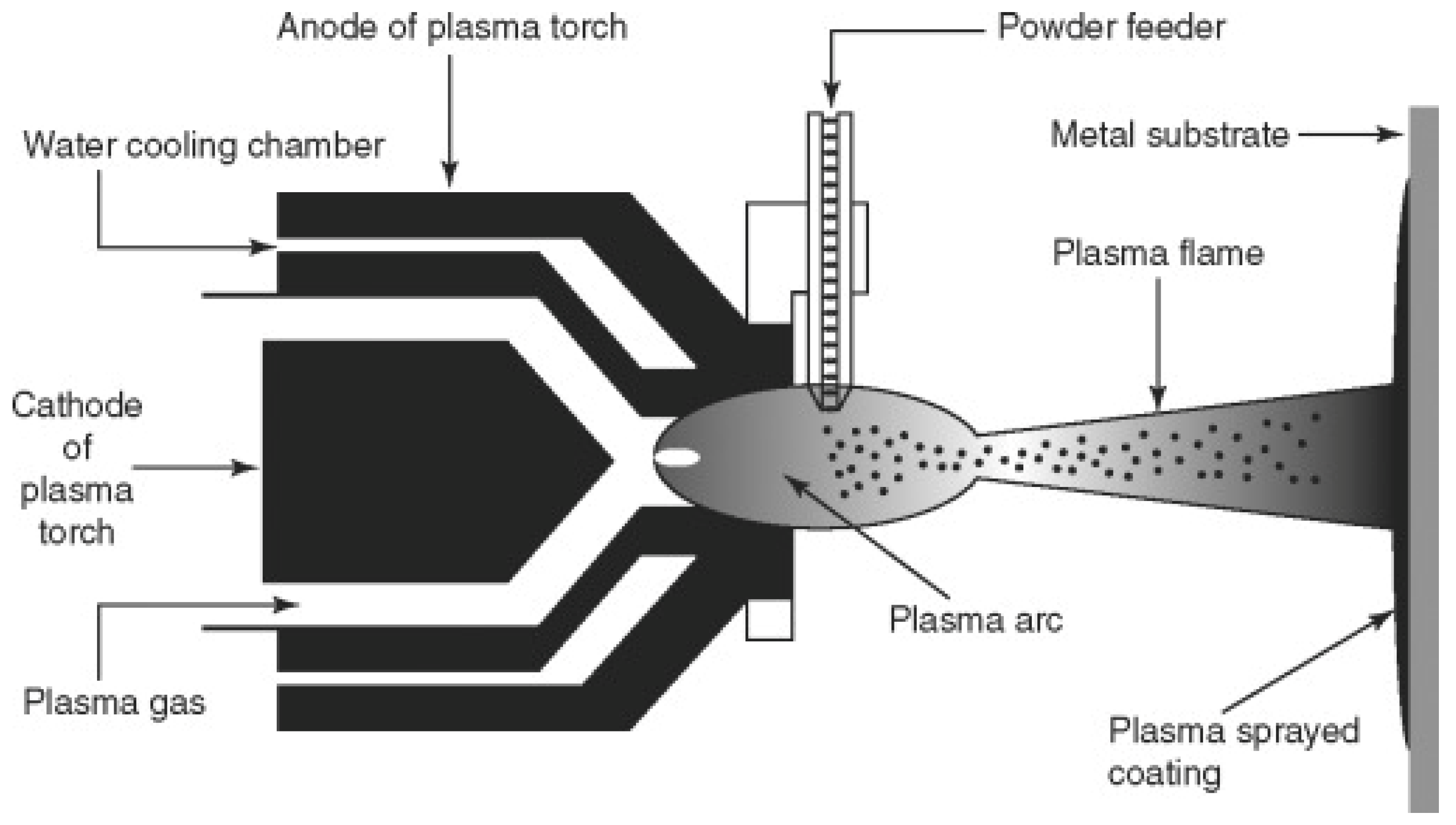
3.1. Plasma Spraying (PS)
| → | + | (1) | |||
| → | + | (2) | |||
| → | + | (3) | |||
| → | + | (4) | |||
| → | + | (5) |
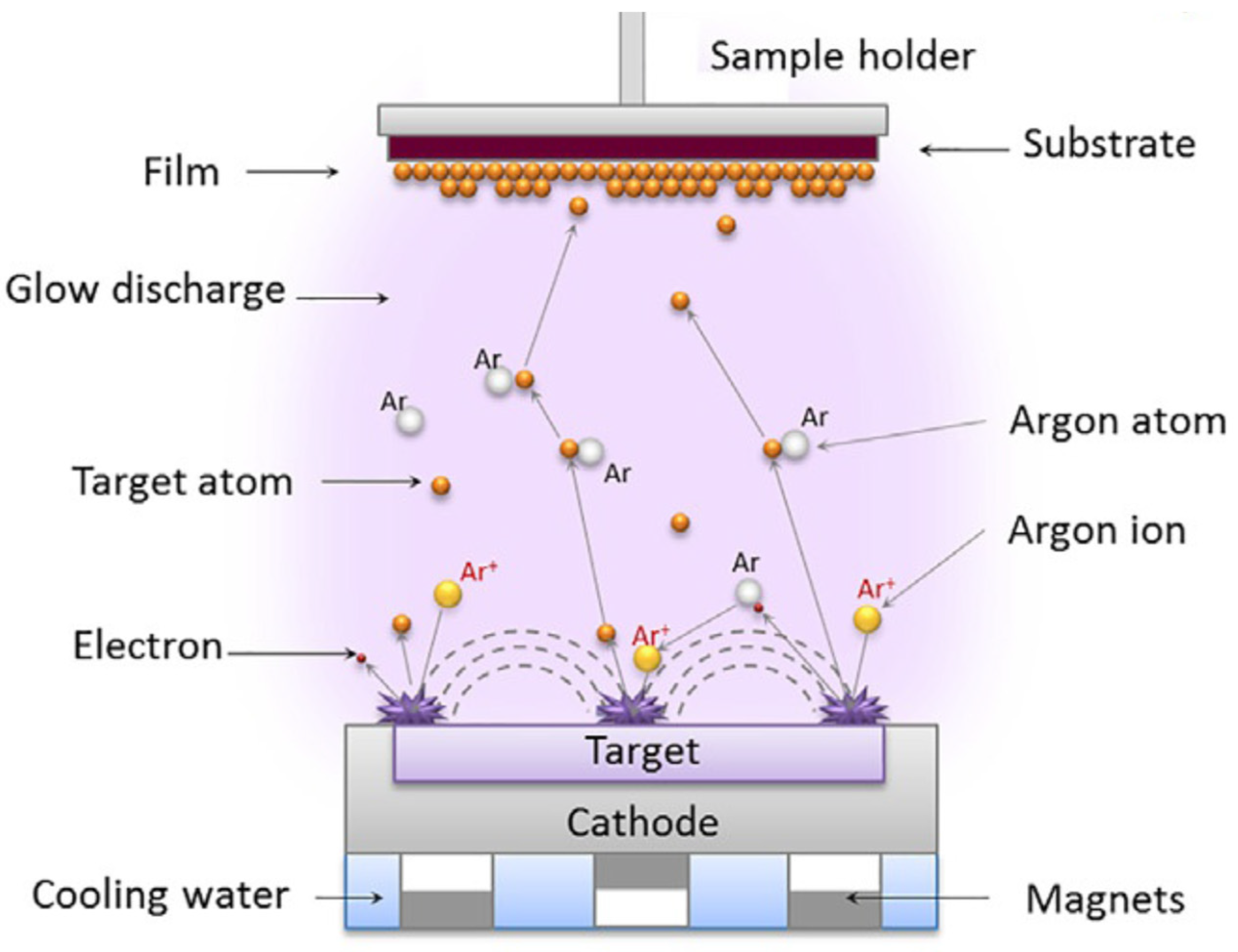
3.2. Magnetron Sputerring (MS)
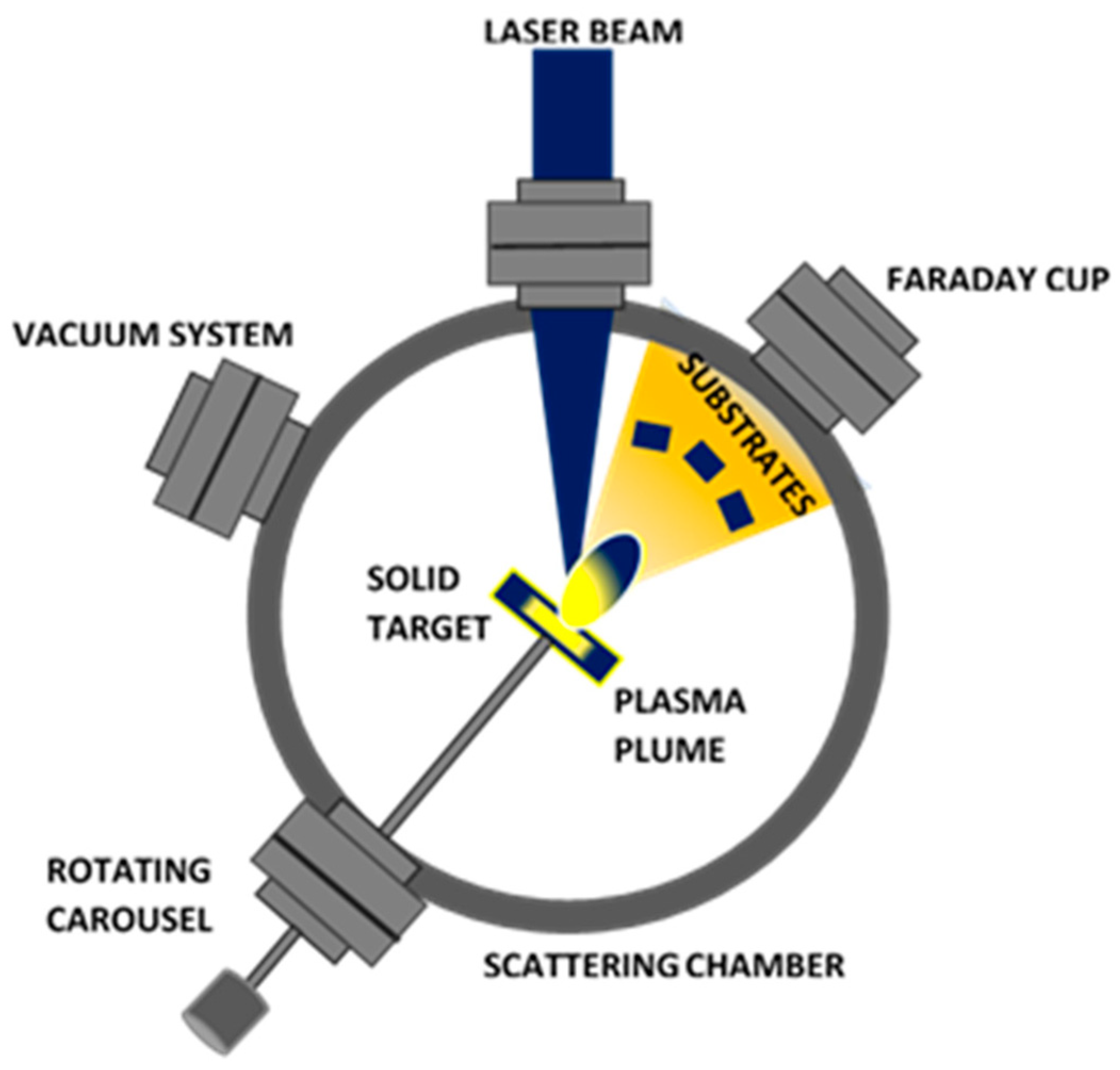
3.3. Pulsed Laser Deposition (PLD)
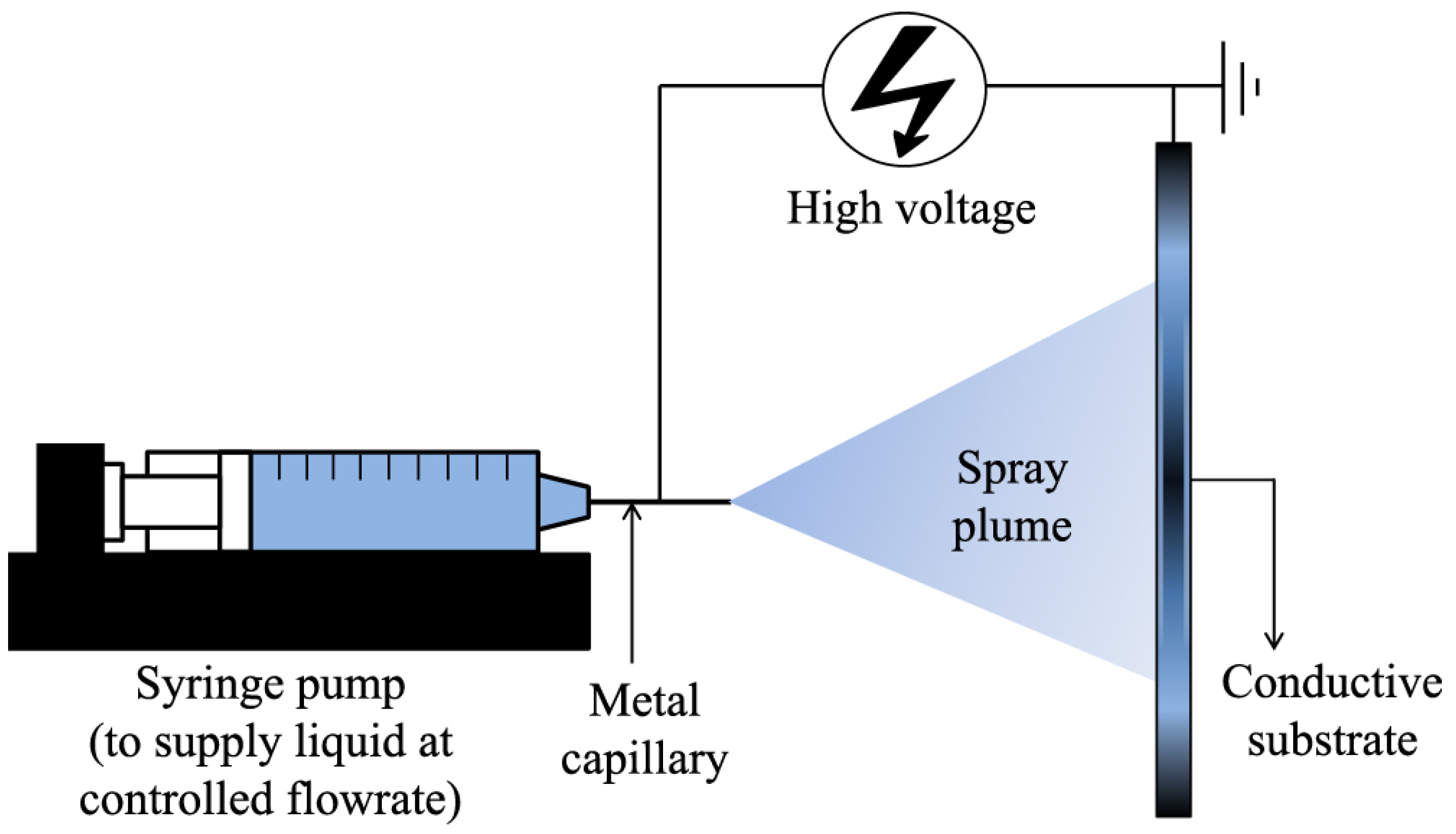
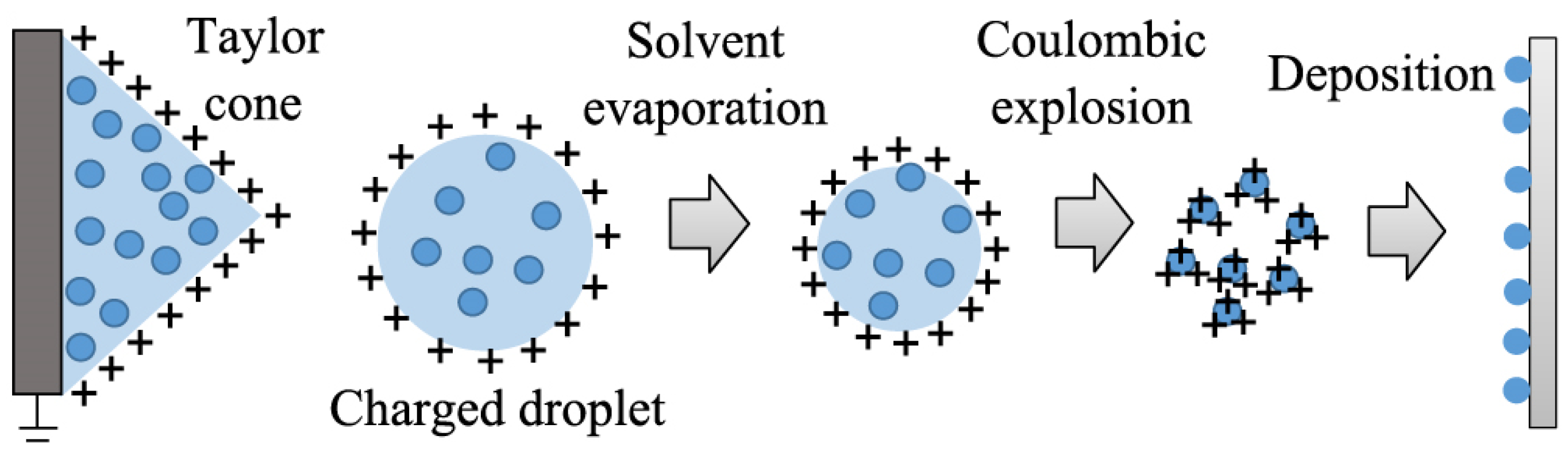
3.4. Electrospray Deposition (ESD)
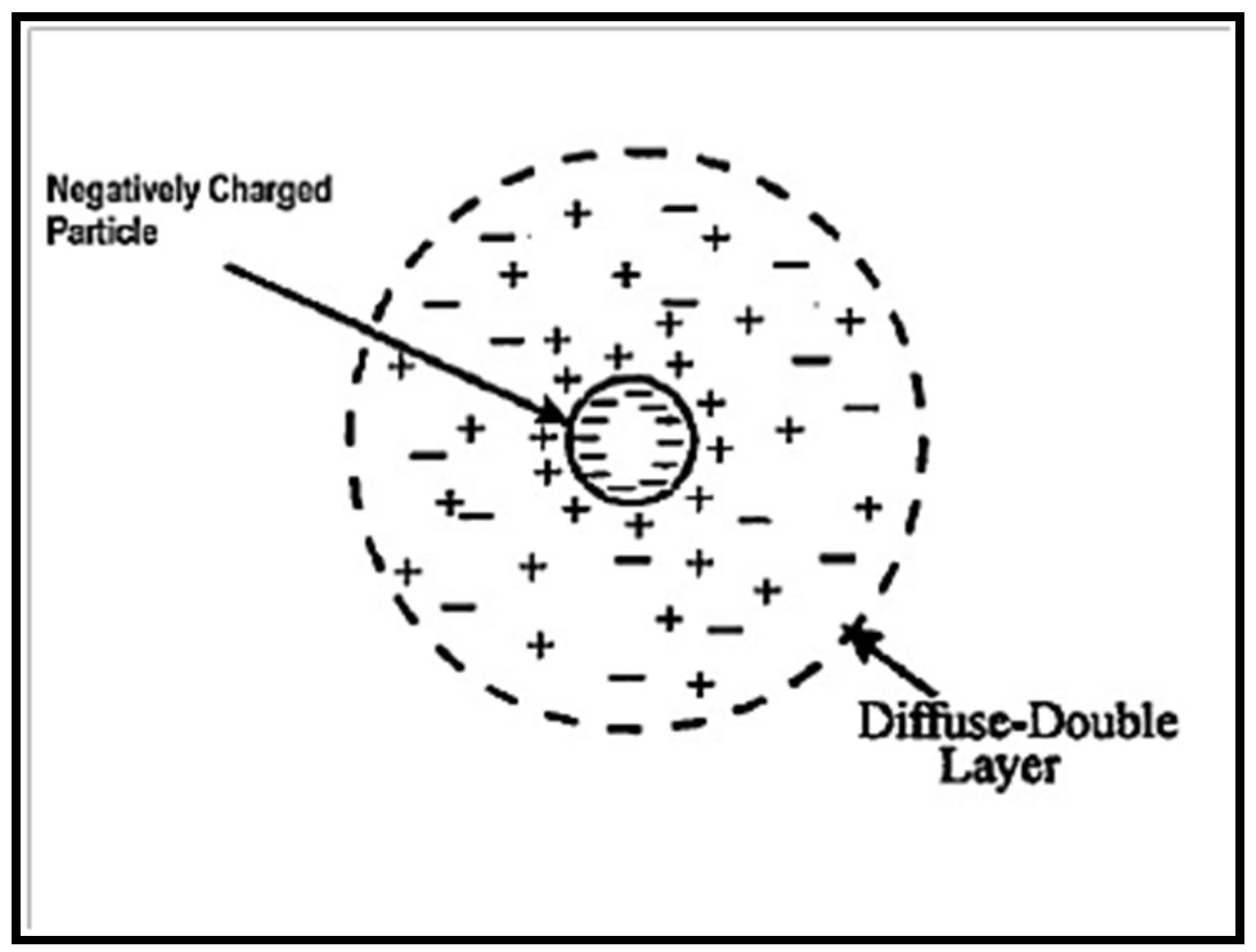
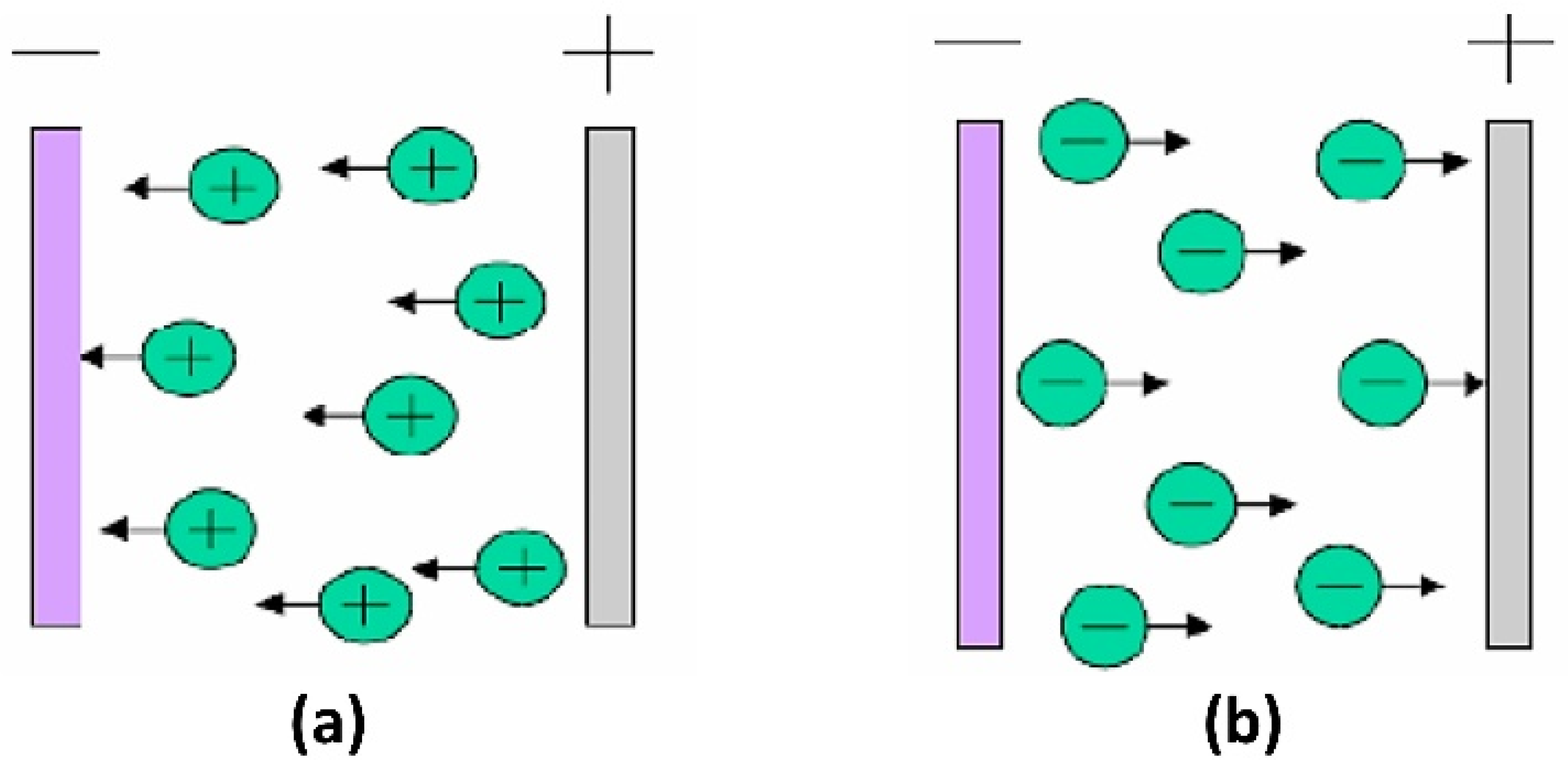
3.5. Electrophoretic Deposition (EPD)
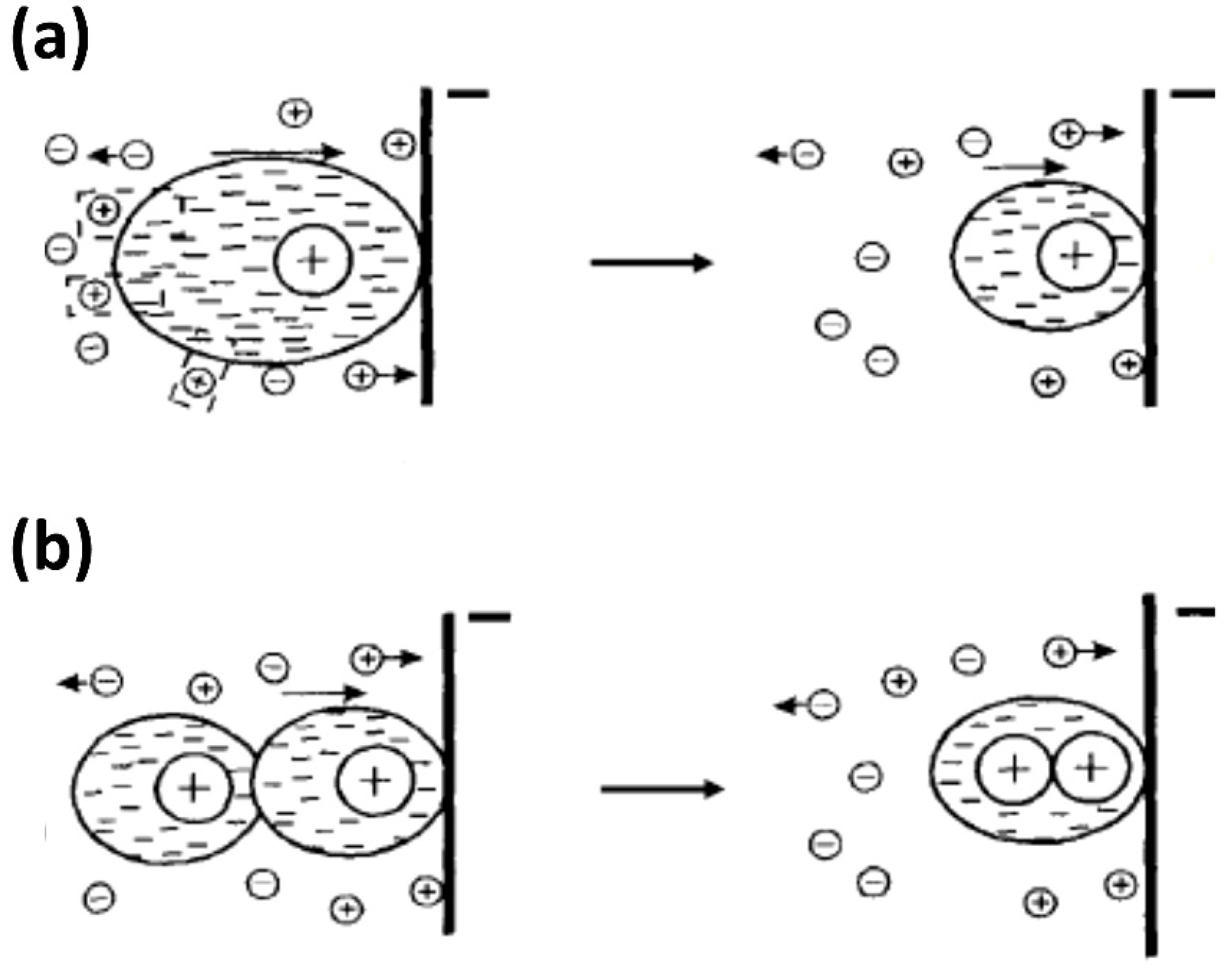
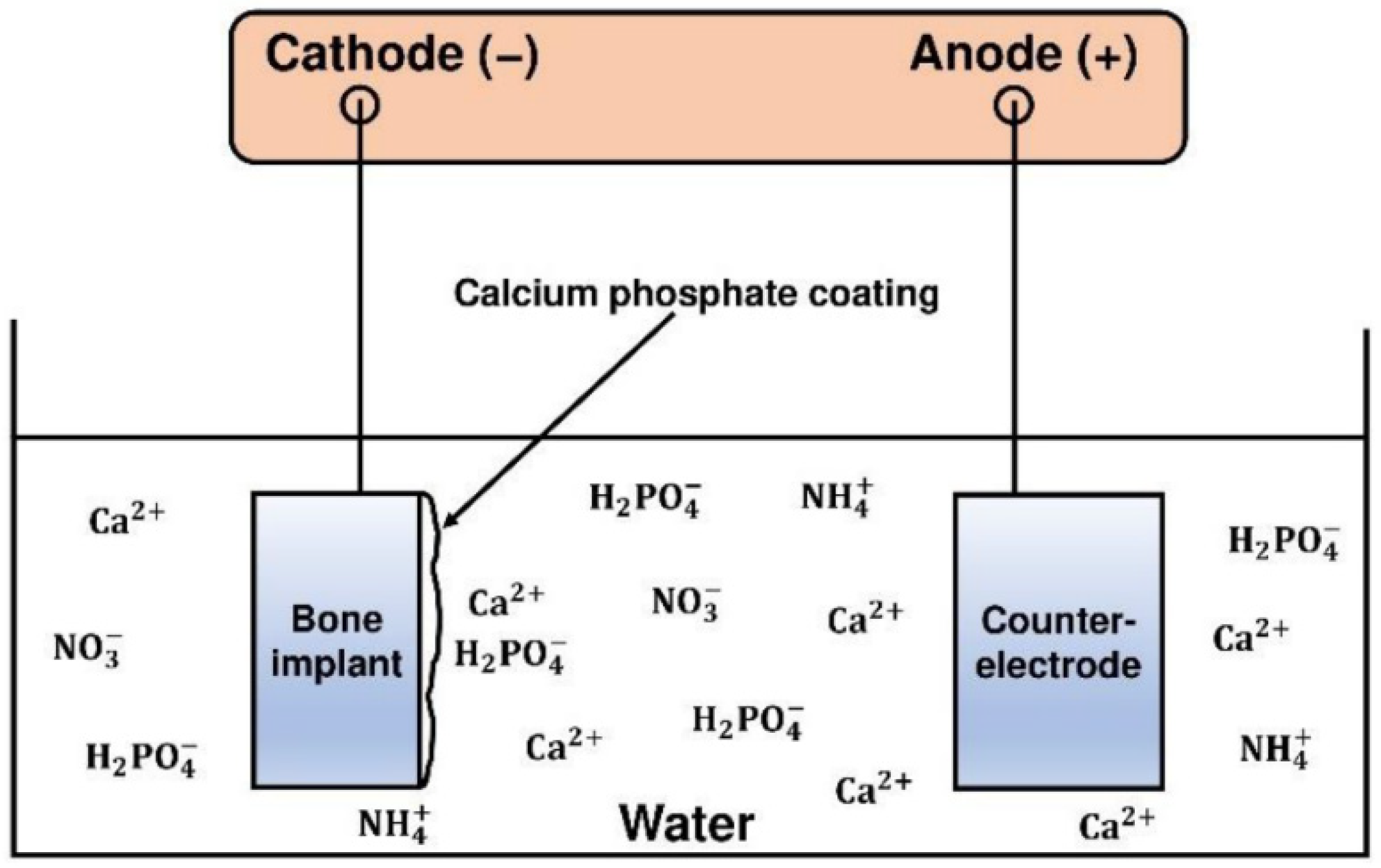
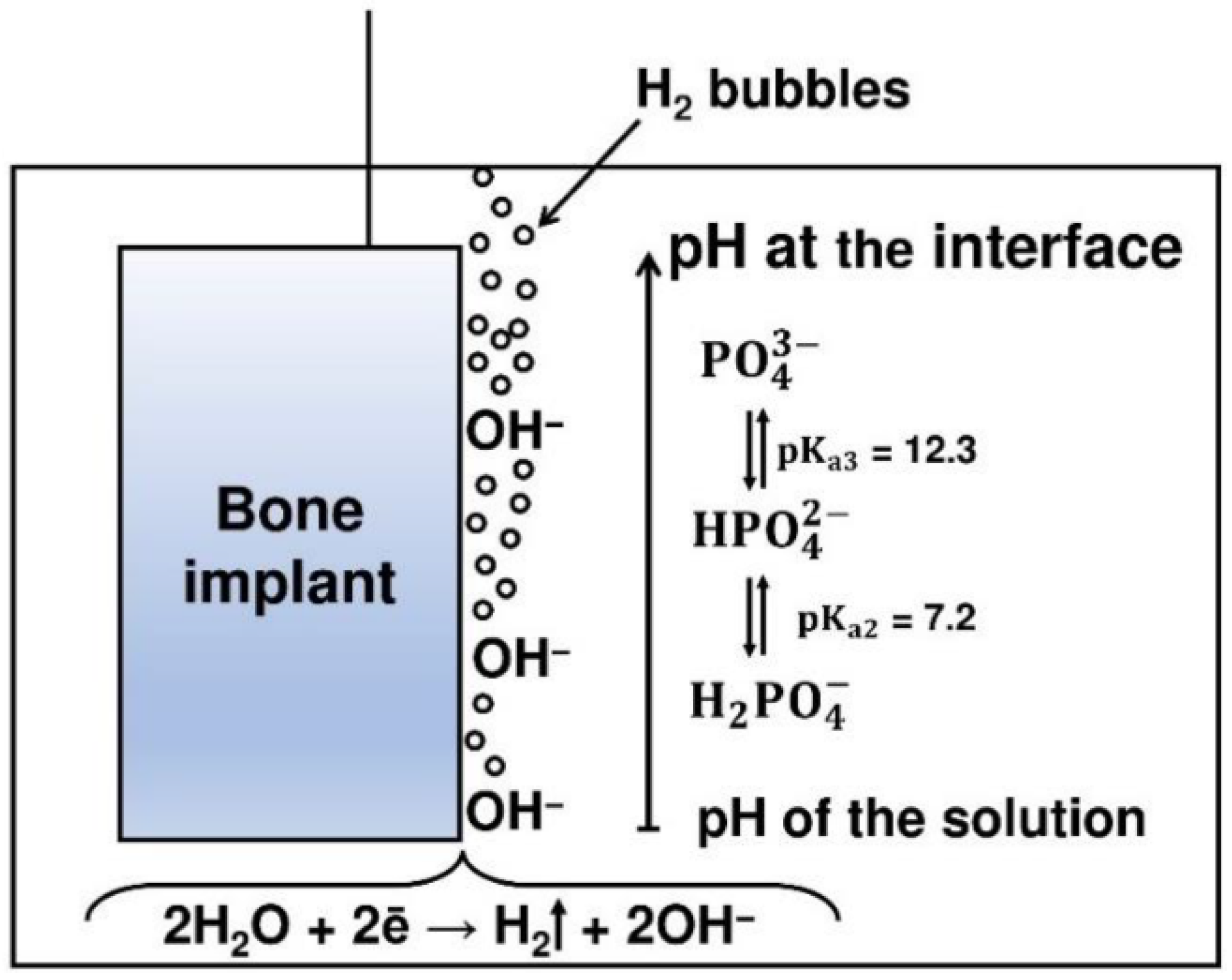
3.6. Electrodeposition (ELD)
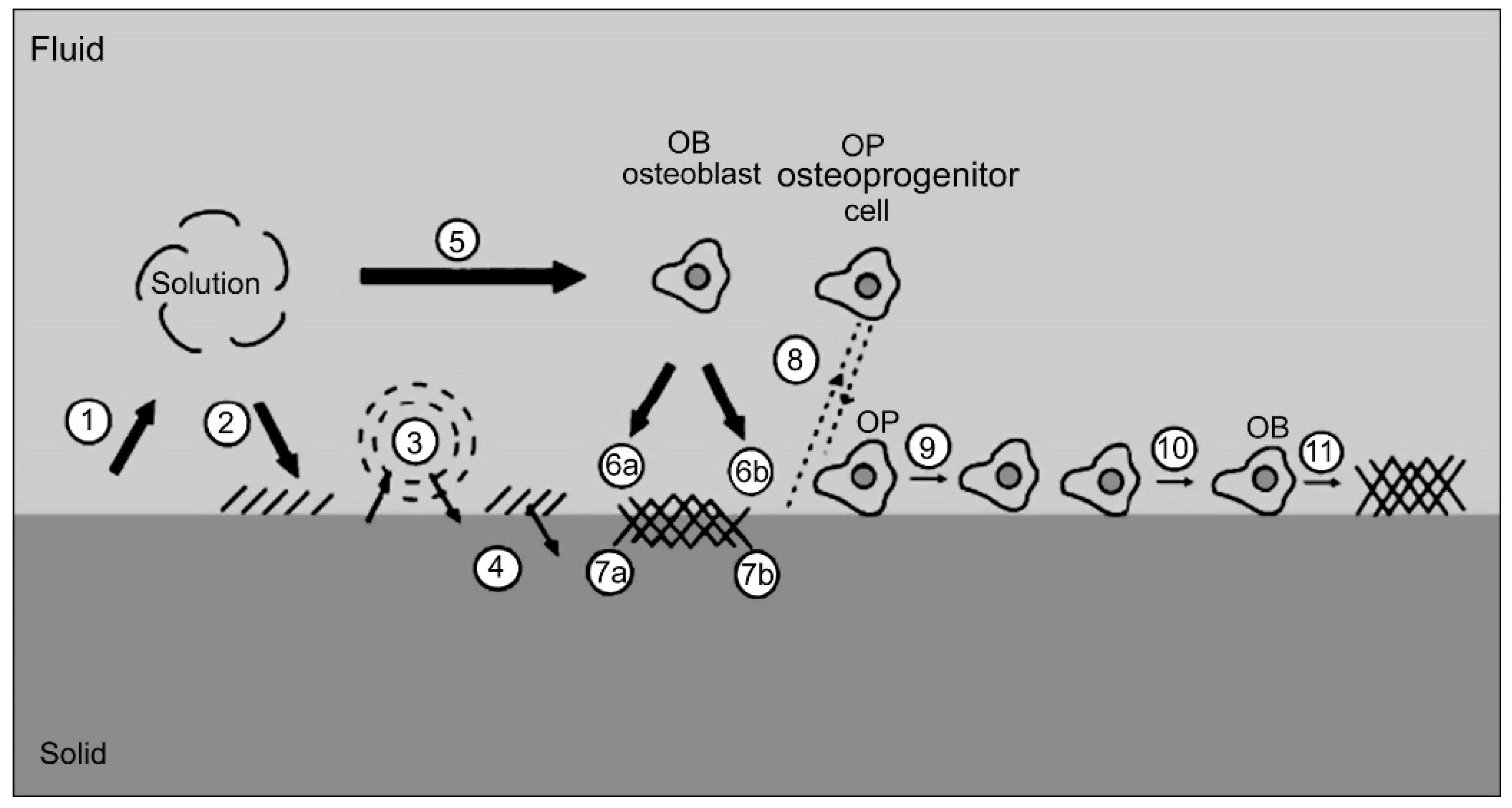
4. Main Properties Impacting the Bioactivity of Calcium Phosphate Coatings
4.1. Crystallinity
4.2. Morphology
4.3. Roughness
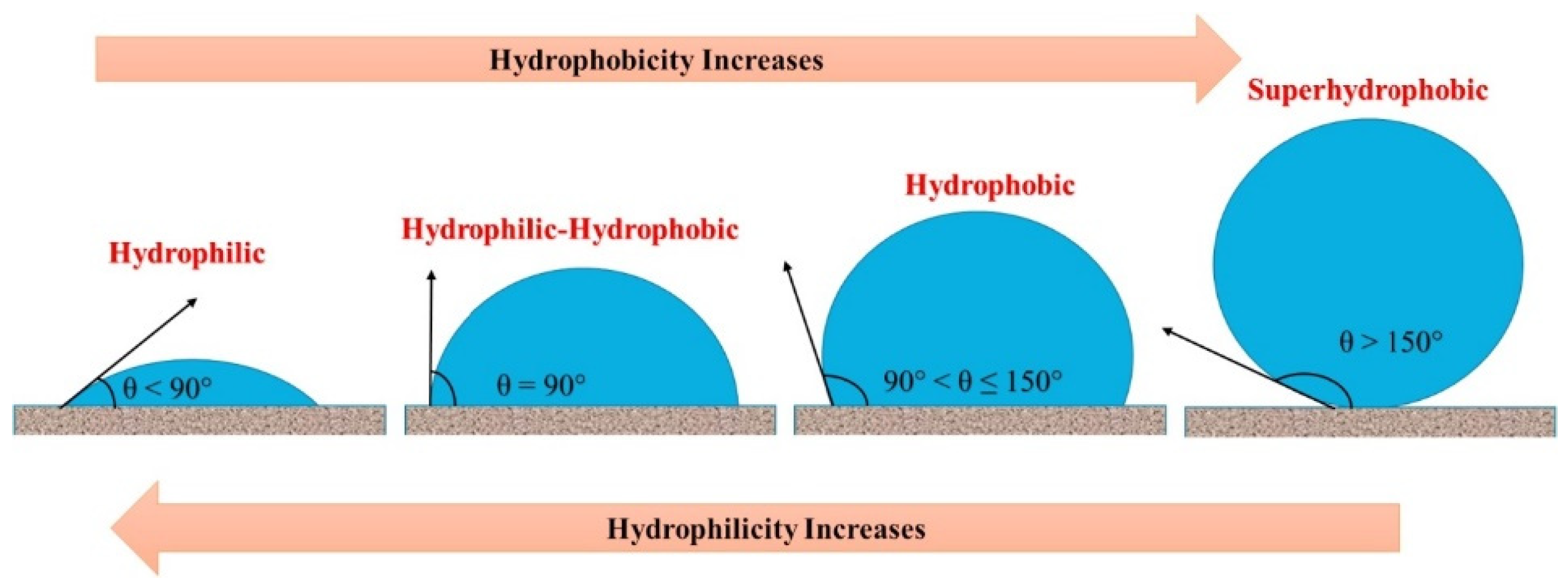
4.4. Wettability
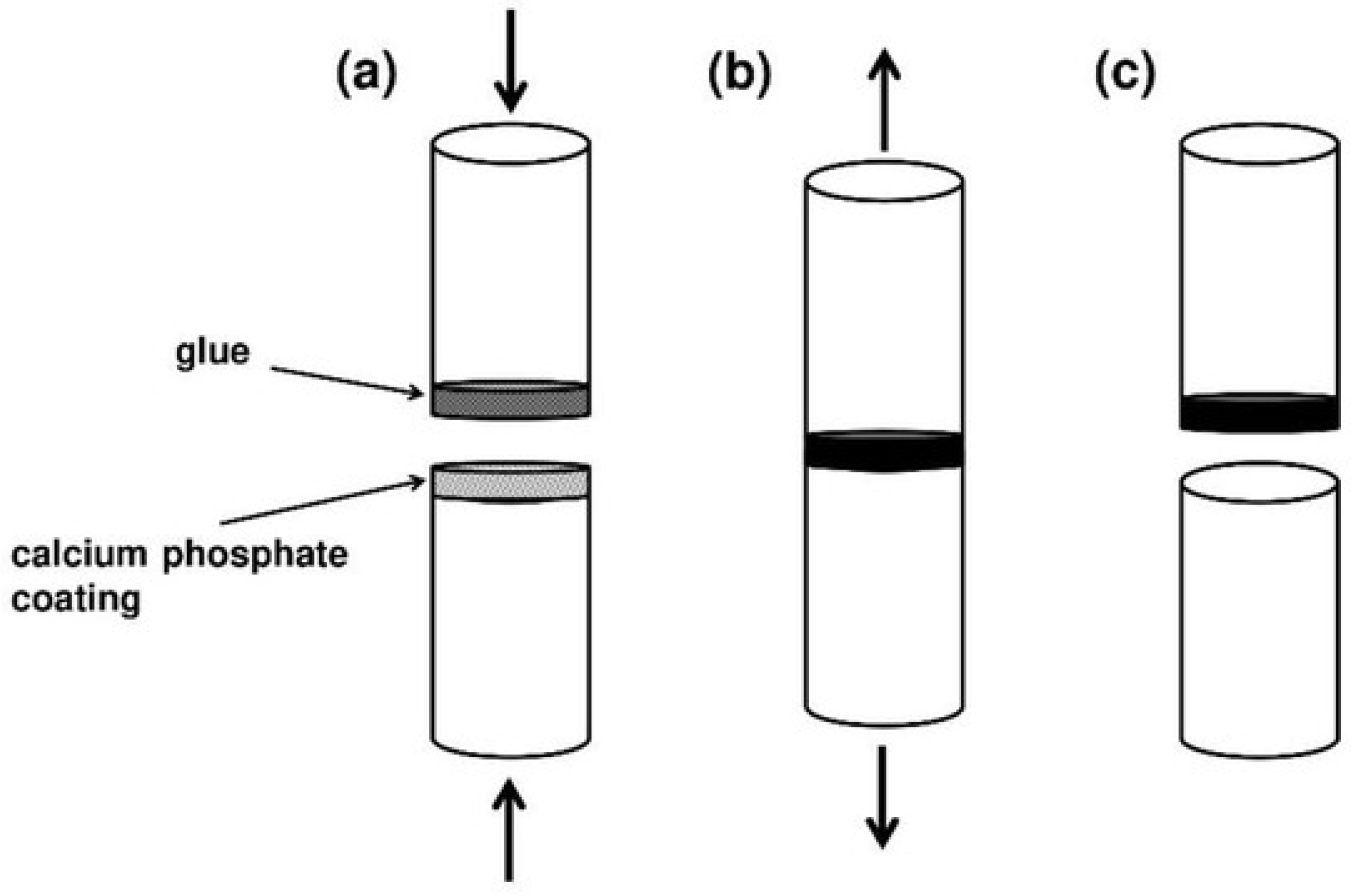
4.5. Adhesion
4.6. Ionic Substitution for Biological Enhancement
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Available online: https://www.who.int/news-room/fact-sheets/detail/ageing-and-health (accessed on 10 May 2023).
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskel. Dis. 2012, 4 (2), 61-76. [CrossRef]
- Gheno, R.; Cepparo, J.M.; Rosca, C.E.; Cotton, A. Musculoskeletal Disorders in the Elderly. J. Clin. Imaging Sci. 2012, 2 (3), 39. [CrossRef]
- U.S. Department of Health and Human Services. Bone Health and Osteoporosis: A Report of the Surgeon General. Rockville, U.S.A., 2004. PMID: 20945569.
- Li, G.; Thabane, L.; Papaioannou, A.; Ioannidis, G.; Levine, M.A.H.; Adachi, J.D. An overview of osteoporosis and frailty in the elderly. BMC Musculoskelet. Disord. 2017, 18, 46. [CrossRef]
- Bal, Z.; Kaito, T.; Korkusuz, F.; Yoshikawa, H. Bone regeneration with hydroxyapatite-based biomaterials. Emergent Mater. 2020, 3, 521-544. [CrossRef]
- Farrakhov, R.; Melnichuk, O.; Parfenov, E.; Mukaeva, V.; Raab, A.; Sheremetyev, V.; Zhukova, Y.; Prokoshkin, S. Comparison of Biocompatible Coatings Produced by Plasma Electrolytic Oxidation on cp-Ti and Ti-Zr-Nb Superelastic Alloy. Coatings 2021, 11, 401. [CrossRef]
- Ijaz, M.F.; Laillé, D.; Héraud, L.; Gordin, D.M.; Castany, P.; Gloriant, T. Design of a novel superelastic Ti-23Hf-3Mo-4Sn biomedical alloy combining low modulus, high strength and large recovery strain. Mater. Lett. 2016, 177, 39-41. [CrossRef]
- Sheremetyev, V.; Lukashevich, L.; Kreitcberg, A.; Kudryashova, A.; Tsaturyants, M.; Galkin, G.; Andreev, V.; Prokoshkin, S.; Brailovski, V. Optimization of a thermomechanical treatment of superelastic Ti-Zr-Nb alloys for the production of bar stock for orthopedic implants. J. Alloys Compd. 2022, 928, 167143. [CrossRef]
- Lukashevich, K.; Sheremetyev, V.; Komissarov, A.; Cheverikin, V.; Andreev, V.; Prokoshkin, S.; Brailovski, V. Effect of Cooling and Annealing Conditions on the Microstructure, Mechanical and Superelastic Behavior of a Rotary Forged Ti-18Zr-15Nb (at. %) Bar Stock for Spinal Implants. J. Funct. Biomater. 2022, 13 (4), 259. [CrossRef]
- He, G.; Hagiwara, M. Ti alloy design strategy for biomedical applications. Mater. Sci. Eng. C 2006, 26 (1), 14-19. [CrossRef]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants - A review. Prog. Mater. Sci. 2009, 54 (3), 397-425. [CrossRef]
- Chen, Q.; Thouas, G.A. Metallic implant biomaterials. Mater. Sci. Eng. R 2015, 87, 1-57. [CrossRef]
- Sherif, E.S.M.; Bahri, Y.A.; Alharbi, H.F.; Ijaz, M.F.; Alnaser, I.A. Influence of Tantalum Addition on the Corrosion Passivation of Titanium-Zirconium Alloy in Simulated Body Fluid. Materials 2022, 15 (24), 8812. [CrossRef]
- Andreucci, C.A.; Alshaya, A.; Fonseca, E.M.M.; Jorge, R.N. Proposal for a New Bioactive Kinetic Screw in an Implant, Using a Numerical Model. Appl. Sci. 2022, 12, 779. [CrossRef]
- Chashmi, M.J.; Fathi, A.; Shirzad, M.; Jafari-Talookolaei, R.A.; Bodaghi, M.; Rabiee, S.M. Design and Analysis of Porous Functionally Graded Femoral Prostheses with Improved Stress Shielding. Designs 2020, 4, 12. [CrossRef]
- Drevet, R.; Zhukova, Y.; Malikova, P.; Dubinskiy, S.; Korotitskiy, A.; Pustov, Y.; Prokoshkin, S. Martensitic Transformations and Mechanical and Corrosion Properties of Fe-Mn-Si Alloys for Biodegradable Medical Implants. Metall. Mater. Trans. A 2018, 49 (3), 1006-1013. [CrossRef]
- Drevet, R.; Zhukova, Y.; Kadirov, P.; Dubinskiy, S.; Kazakbiev, A.; Pustov, Y.; Prokoshkin, S. Tunable corrosion behavior of calcium phosphate coated Fe-Mn-Si alloys for bone implant applications. Metall. Mater. Trans. A 2018, 49 (12), 6553-6560. [CrossRef]
- Prokoshkin, S.; Pustov, Y.; Zhukova, Y.; Kadirov, P.; Dubinskiy, S.; Sheremetyev, V.; Karavaeva, M. Effect of Thermomechanical Treatment on Functional Properties of Biodegradable Fe-30Mn-5Si Shape Memory Alloy. Metall. Mater. Trans. A 2021, 52, 2024–2032. [CrossRef]
- Koumya, Y.; Ait Salam, Y.; Khadiri, M.E.; Benzakour, J.; Romane, A.; Abouelfida, A.; Benyaich, A. Pitting corrosion behavior of SS-316L in simulated body fluid and electrochemically assisted deposition of hydroxyapatite coating. Chem. Pap. 2021, 75(6), 2667-2682. [CrossRef]
- Trincă, L.C.; Burtan, L.; Mareci, D.; Fernández-Pérez, B.M.; Stoleriu, I.; Stanciu, T.; Stanciu, S.; Solcan, C.; Izquierdo, J.; Souto, R.M. Evaluation of in vitro corrosion resistance and in vivo osseointegration properties of a FeMnSiCa alloy as potential degradable implant biomaterial. Mater. Sci. Eng. C 2021, 118, 111436. [CrossRef]
- Tchana Nkonta, D.V.; Simescu-Lazar, F.; Drevet, R.; Aaboubi, O.; Fauré, J.; Retraint, D.; Benhayoune, H. Influence of the surface mechanical attrition treatment (SMAT) on the corrosion behavior of Co28Cr6Mo alloy in Ringer’s solution. J. Solid State Electrochem. 2018, 22 (4), 1091-1098. [CrossRef]
- Chen, Y.; Li, Y.; Kurosu, S.; Yamanaka, K.; Tang, N.; Koizumi, Y.; Chiba, A. Effects of sigma phase and carbide on the wear behavior of CoCrMo alloys in Hanks’ solution. Wear 2014, 310 (1-2), 51-62. [CrossRef]
- Tchana Nkonta, D.V.; Drevet, R.; Fauré, J.; Benhayoune, H. Effect of surface mechanical attrition treatment on the microstructure of cobalt–chromium–molybdenum biomedical alloy. Microsc. Res. Tech. 2021, 84 (2), 238-245. [CrossRef]
- AlMangour, B.; Luqman, M.; Grzesiak, D.; Al-Harbi, H.; Ijaz, F. Effect of processing parameters on the microstructure and mechanical properties of Co–Cr–Mo alloy fabricated by selective laser melting. Mater. Sci. Eng. A 2020, 792, 139456. [CrossRef]
- Yamanaka, K.; Mori, M.; Kurosu, S.; Matsumoto, H.; Chiba, A. Ultrafine grain refinement of biomedical Co-29Cr-6Mo alloy during conventional hot-compression deformation. Metall. Mater. Trans. A 2009, 40 (8), 1980-1994. [CrossRef]
- İbrahim Coşkun, M.; Karahan, İ.H.; Yücel, Y.; Golden, T.D. Optimization of electrochemical step deposition for bioceramic hydroxyapatite coatings on CoCrMo implants. Surf. Coat. Technol. 2016, 301, 42-53. [CrossRef]
- Coşkun, M.I.; Karahan, I.H.; Yücel, Y. Optimized electrodeposition concentrations for hydroxyapatite coatings on CoCrMo biomedical alloys by computational techniques. Electrochim. Acta 2014, 150, 46-54. [CrossRef]
- Ghasemi-Mobarakeh, L.; Kolahreez, D.; Ramakrishna, S.; Williams, D. Key terminology in biomaterials and biocompatibility. Curr. Opin. Biomed. Eng. 2019, 10, 45-50. [CrossRef]
- Williams, D. Revisiting the definition of biocompatibility. Med. Device Technol. 2003, 14 (8), 10-13. PMID: 14603712.
- Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941-2953. [CrossRef]
- Barrère, F.; Mahmood, T.A.; de Groot, K.; van Blitterswijk, C.A. Advanced biomaterials for skeletal tissue regeneration: Instructive and smart functions. Mater. Sci. Eng. R 2008, 59, 38-71. [CrossRef]
- Moniruzzaman, M.d.; O’Neal, C.; Bhuiyan, A.; Egan, P.F. Design and Mechanical Testing of 3D Printed Hierarchical Lattices Using Biocompatible Stereolithography. Designs 2020, 4, 22. [CrossRef]
- Nuswantoro, N.F.; Rahandi Lubis, M.A.; Juliadmi, D.; Mardawati, E.; Antov, P.; Kristak, L.; Seng Hua, L. Bio-Based Adhesives for Orthopedic Applications: Sources, Preparation, Characterization, Challenges, and Future Perspectives. Designs 2022, 6, 96. [CrossRef]
- Williams, D.F. Biocompatibility pathways and mechanisms for bioactive materials: The bioactivity zone. Bioact. Mater. 2022, 10, 306-322. [CrossRef]
- Williams, D.F. On the nature of biomaterials. Biomaterials 2009, 30 (30), 5897-5909. [CrossRef]
- Cao, W.; Hench, L.L. Bioactive materials. Ceram. Int. 1996, 22 (6), 493-507. [CrossRef]
- Albrektsson, T.; Johansson, C. Osteoinduction, osteoconduction and osseointegration. Eur. Spine J. 2001, 10, S96-S101. [CrossRef]
- Andreucci, C.A.; Fonseca, E.M.M.; Jorge, R.N. Bio-Lubricant Properties Analysis of Drilling an Innovative Design of Bioactive Kinetic Screw into Bone. Designs 2023, 7, 21. [CrossRef]
- Shaikh, M.S.; Fareed, M.A.; Zafar, M.S. Bioactive Glass Applications in Different Periodontal Lesions: A Narrative Review. Coatings 2023, 13 (4), 716. [CrossRef]
- Paital, S.R.; Dahotre, N.B. Calcium phosphate coatings for bio-implant applications: Materials, performance factors, and methodologies. Mater. Sci. Eng. R 2009, 66 (1-3), 1-70. [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphate deposits: Preparation, properties and biomedical applications. Mater. Sci. Eng. C 2015, 55, 272-326. [CrossRef]
- Dorozhkin, S.V. Bioceramics of calcium orthophosphates. Biomaterials 2010, 31 (7), 1465-1485. [CrossRef]
- LeGeros, R.Z. Calcium phosphate-based osteoinductive materials. Chem. Rev. 2008, 108 (11), 4742-4753. [CrossRef]
- Hench, L.L. Bioceramics. J. Am. Ceram. Soc. 1998, 81 (7), 1705-1728. [CrossRef]
- Navarrete-Segado, P.; Tourbin, M.; Grossin, D.; Frances, C. Tailoring hydroxyapatite suspensions by stirred bead milling. Ceram. Int. 2022, 48, 24953-24964. [CrossRef]
- Dorozhkin S.V. Calcium Orthophosphate (CaPO4)-Based Bioceramics: Preparation, Properties, and Applications. Coatings 2022, 12, 1380. [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Ivanova, A.A. Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis - A review. Acta Biomater. 2014, 10 (2), 557-579. [CrossRef]
- Vallet-Regí, M.; González-Calbet, J.M. Calcium phosphates as substitution of bone tissues. Prog. Solid State Chem. 2004, 32 (1-2), 1-31. [CrossRef]
- Fiume, E.; Magnaterra, G.; Rahdar, A.; Verné, E.; Baino, F. Hydroxyapatite for Biomedical Applications: A Short Overview. Ceramics 2021, 4, 542–563. [CrossRef]
- Jeong, J.; Kim, J.H.; Shim, J.H.; Hwang, N.S.; Heo, C.Y. Bioactive calcium phosphate materials and applications in bone regeneration. Biomater. Res. 2019, 23, 4. [CrossRef]
- Lu, J.; Yu, H.; Chen, C. Biological properties of calcium phosphate biomaterials for bone repair: A review. RSC Advances 2018, 8, 2015-2033. [CrossRef]
- Drevet, R.; Benhayoune, H. Advanced Biomaterials and Coatings. Coatings 2022, 12, 965. [CrossRef]
- McCabe, A.; Pickford, M.; Shawcross J. The History, Technical Specifications and Efficacy of Plasma Spray Coatings Applied to Joint Replacement Prostheses. Reconstr. Rev. 2016, 6 (4), 19-26. [CrossRef]
- Moseke, C.; Gbureck, U. Tetracalcium phosphate: Synthesis, properties and biomedical applications. Acta Biomater. 2010, 6 (10), 3815-3823. [CrossRef]
- Qin, T.; Xu, Y. Fe-reinforced TTCP biocermet prepared via laser melting: Microstructure, mechanical properties and bioactivity. Ceram. Int. 2021, 47 (12), 17652-17661. [CrossRef]
- Mandal, S.; Meininger, S.; Gbureck, U.; Basu, B. 3D powder printed tetracalcium phosphate scaffold with phytic acid binder: Fabrication, microstructure and in situ X-Ray tomography analysis of compressive failure. J. Mater. Sci. Mater. Med. 2018, 29, 29. [CrossRef]
- LeGeros, R.Z. Properties of Osteoconductive Biomaterials: Calcium Phosphates. Clin. Orthop. Relat. Res. 2002, 395, 81-98. [CrossRef]
- Hench, L.L. Bioceramics: From Concept to Clinic. J. Am. Ceram. Soc. 1991, 74 (7), 1487-1510. [CrossRef]
- Szcześ, A.; Hołysz, L.; Chibowski, E. Synthesis of hydroxyapatite for biomedical applications. Adv. Colloid Interface Sci. 2017, 249, 321-330. [CrossRef]
- Carrodeguas, R.G.; De Aza, S. a-Tricalcium phosphate: Synthesis, properties and biomedical applications. Acta Biomaterialia 2011, 7, 3536-3546. [CrossRef]
- de Aza, P.N.; Luklinska, Z.B.; Mate-Sanchez de Val, J.E.; Calvo-Guirado, J.L. Biodegradation Process of a-Tricalcium Phosphate and a-Tricalcium Phosphate Solid Solution Bioceramics In Vivo: A Comparative Study. Microsc. Microanal. 2013, 19, 1350-1357. [CrossRef]
- Kolmas, J.; Kaflak, A.; Zima, A.; Ślósarczyk, A. Alpha-tricalcium phosphate synthesized by two different routes: Structural and spectroscopic characterization. Ceramics International 2015, 41, 5727-5733. [CrossRef]
- Bohner, M.; Le Gars Santoni, B.; Döbelin, N. β-tricalcium phosphate for bone substitution: Synthesis and properties. Acta Biomater. 2020, 113, 23-41. [CrossRef]
- Chaair, H.; Labjar, H.; Britel, O. Synthesis of b-tricalcium phosphate. Morphologie 2017, 101, 120-124. [CrossRef]
- Drevet, R.; Fauré, J.; Sayen, S.; Marle-Spiess, M., El Btaouri, H.; Benhayoune, H. Electrodeposition of biphasic calcium phosphate coatings with improved dissolution properties. Mater. Chem. Phys. 2019, 236, 121797. [CrossRef]
- Drouet, C. Apatite formation: Why it may not work as planned, and how to conclusively identify apatite compounds. BioMed Res. Int. 2013, 490946. [CrossRef]
- Vallet-Regí, M.; Rodriguez-Lorenzo, L.M.; Salinas, A.J. Synthesis and characterisation of calcium deficient apatite. Solid State Ionics 1997, 101-103, 1279-1285. [CrossRef]
- Hutchens, S.A.; Benson, R.S.; Evans, B.R.; O’Neill, H.M.; Rawn, C.J. Biomimetic synthesis of calcium-deficient hydroxyapatite in a natural hydrogel. Biomaterials 2006, 27, 4661-4670. [CrossRef]
- Teterina, A.Y.; Smirnov, I.V.; Fadeeva, I.S.; Fadeev, R.S.; Smirnova, P.V.; Minaychev, V.V.; Kobyakova, M.I.; Fedotov, A.Y.; Barinov, S.M.; Komlev, V.S. Octacalcium Phosphate for Bone Tissue Engineering: Synthesis, Modification, and In Vitro Biocompatibility Assessment. Int. J. Mol. Sci. 2021, 22, 12747. [CrossRef]
- Suzuki, O.; Hamai, R.; Sakai, S. The material design of octacalcium phosphate bone substitute: Increased dissolution and osteogenecity. Acta Biomaterialia 2023, 158, 1-11. [CrossRef]
- Kovrlija, I.; Locs, J.; Loca, D. Octacalcium phosphate: Innovative vehicle for the local biologically active substance delivery in bone regeneration. Acta Biomaterialia 2021, 135, 27-47. [CrossRef]
- Vasant, S.R.; Joshi, M.J. A review on calcium pyrophosphate and other related phosphate nano bio-materials and their applications. Rev. Adv. Mater. Sci. 2017, 49, 44-57.
- Yan, Y.; Wolke, J.G.C.; De Ruijter, A. ; Yubao, L.; Jansen, J.A. Growth behavior of rat bone marrow cells on RF magnetron sputtered hydroxyapatite and dicalcium pyrophosphate coatings. J. Biomed. Mater. Res. A 2006, 78A (1), 42-49. [CrossRef]
- Golubchikov, D.; Safronova, T.V.; Nemygina, E.; Shatalova, T.B.; Tikhomirova, I.N.; Roslyakov, I.V.; Khayrutdinova, D.; Platonov, V.; Boytsova, O.; Kaimonov, M.; Firsov, D.A.; Lyssenko, K.A. Powder Synthesized from Aqueous Solution of Calcium Nitrate and Mixed-Anionic Solution of Orthophosphate and Silicate Anions for Bioceramics Production. Coatings 2023, 13, 374. [CrossRef]
- Zhou, H.; Yang, L.; Gbureck, U.; Bhaduri, S.B.; Sikder, P. Monetite, an important calcium phosphate compound-Its synthesis, properties and applications in orthopedics. Acta Biomaterialia 2021, 127, 41-55. [CrossRef]
- Prado Da Silva, M.H.; Lima, J.H.C.; Soares, G.A.; Elias, C.N.; de Andrade, M.C.; Best, S.M.; Gibson, I.R. Transformation of monetite to hydroxyapatite in bioactive coatings on titanium. Surf. Coat. Technol. 2001, 137, 270-276. [CrossRef]
- Ling, L.; Xin-bo, X.; Jun, M.; Xin-ye, N.; Xie-rong, Z.; Sial, M.A.Z.G.; Dazhu, C. Post-hydrothermal treatment of hydrothermal electrodeposited CaHPO4 on C/C composites in sodium silicate-containing solution at various temperatures. Ceram. Int. 2019, 45, 5894-5903. [CrossRef]
- Tamimi, F.; Sheikh, Z.; Barralet, J. Dicalcium phosphate cements: Brushite and monetite. Acta Biomaterialia 2012, 8 (2), 474-487. [CrossRef]
- Türk, T.; Altınsoy, İ.; ÇelebiEfe, G.; Ipek, M.; Özacar, M.; Bindal, C. Biomimetric coating of monophasic brushite on Ti6Al4V in new m-5xSBF. Surf. Coat. Technol. 2018, 351, 1-10. [CrossRef]
- Lee, D.W.; Shin, M.C.; Kim, Y.N.; Oh, J.M. Brushite ceramic coatings for dental brace brackets fabricated via aerosol deposition. Ceram. Int. 2017, 43 (1), 1044-1051. [CrossRef]
- Su, Y.; Cockerill, I.; Zheng, Y.; Tang, L.; Qin, Y.X.; Zhu, D. Biofunctionalization of metallic implants by calcium phosphate coatings. Bioact. Mater. 2019, 4, 196-206. [CrossRef]
- Eliaz, N.; Metoki, N. Calcium phosphate bioceramics: A review of their history, structure, properties, coating technologies and biomedical applications. Materials 2017, 10 (4), 334. [CrossRef]
- Dorozhkin, S.V.; Calcium orthophosphates (CaPO4): Occurrence and properties. Prog. Biomater. 2016, 5 (1), 9-70. [CrossRef]
- Huan, Z.; Chang, J. Novel bioactive composite bone cements based on the b-tricalcium phosphate–monocalcium phosphate monohydrate composite cement system. Acta Biomaterialia 2019, 5, 1253-1264. [CrossRef]
- Dorozhkin, S.V.; A detailed history of calcium orthophosphates from 1770s till 1950. Mater. Sci. Eng. C 2013, 33, 3085-3110. [CrossRef]
- Bermúdez, O.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Optimization of a calcium orthophosphate cement formulation occurring in the combination of monocalcium phosphate monohydrate with calcium oxide. J. Mater. Sci. Mater. Med. 1994, 5, 67-71. [CrossRef]
- Ducheyne, P.; Qiu. Q. Bioactive ceramics: The effect of surface reactivity on bone formation and bone cell function. Biomaterials 1999, 20, 2287-2303. [CrossRef]
- Kokubo, T.; Takadama, H.; How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907-2915. [CrossRef]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32 (11), 2757-2774. [CrossRef]
- Bohner, M.; Lemaitre, J.; Can bioactivity be tested in vitro with SBF solution? Biomaterials 2009, 30 (12), 2175-2179. [CrossRef]
- Ho-Shui-Ling, A.; Bolander, J.; Rustom, L.E.; Wagoner Johnson, A.; Luyten, F.P.; Picart, C. Bone regeneration strategies: Engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials 2018, 180, 143-162. [CrossRef]
- Sun, L.; Berndt, C.C.; Gross, K.A.; Kucuk, A. Material Fundamentals and Clinical Performance of Plasma-Sprayed Hydroxyapatite Coatings: A Review. J. Biomed. Mater. Res. 2001, 58 (5), 570-592. [CrossRef]
- Gross, K.A.; Walsh, W.; Swarts, E. Analysis of retrieved hydroxyapatite-coated hip prostheses. J. Therm. Spray Technol. 2004, 13 (2), 190-199. [CrossRef]
- Wang, M. Composite coatings for implants and tissue engineering scaffolds. In Biomedical Composites, 1st ed.; Ambrosio, L., Eds.; Woodhead Publishing Series in Biomaterials: Cambridge, England, 2010; Part 2, Chapter 6, pp. 127-177. [CrossRef]
- Heimann, R.B. Plasma-Sprayed Hydroxylapatite-Based Coatings: Chemical, Mechanical, Microstructural, and Biomedical Properties. J. Therm. Spray Technol. 2016, 25 (5), 827-850. [CrossRef]
- Heimann, R.B. On the self-affine fractal geometry of plasma-sprayed surfaces. J. Therm. Spray Technol. 2011, 20 (4), 898-908. [CrossRef]
- Chambard, M.; Marsan, O.; Charvillat, C.; Grossin, D.; Fort, P.; Rey, C.; Gitzhofer, F.; Bertrand, G. Effect of the deposition route on the microstructure of plasma-sprayed hydroxyapatite coatings. Surf. Coat. Technol. 2019, 371, 68-77. [CrossRef]
- Heimann, R.B. Thermal spraying of biomaterials. Surf. Coat. Technol. 2006, 201 (5), 2012-2019. [CrossRef]
- Heimann, R.B. Structural Changes of Hydroxylapatite during Plasma Spraying: Raman and NMR Spectroscopy Results. Coatings 2021, 11, 987. [CrossRef]
- Heimann, R.B. Functional plasma-sprayed hydroxylapatite coatings for medical application: Clinical performance requirements and key property enhancement. J. Vac. Sci. Technol. A 2021, 39 (5), 050801. [CrossRef]
- Mohseni, E.; Zalnezhad, E.; Bushroa, A.R.; Comparative investigation on the adhesion of hydroxyapatite coating on Ti–6Al–4V implant: A review paper. Int. J. Adhes. Adhes. 2014, 48, 238-257. [CrossRef]
- Meek, J.M. A theory of spark discharge. Phys. Rev. 1940, 57 (8), 722-728. [CrossRef]
- Boyle, W.S.; Kisliuk, P. Departure from Paschen’s law of breakdown in gases. Phys. Rev. 1955, 97 (2), 255-259. [CrossRef]
- Bonafos, C.; Khomenkhova, L.; Gourbilleau, F.; Talbot, E.; Slaoui, A.; Carrada, M.; Schamm-Chardon, S.; Dimitrakis, P.; Normand, P. Nano-composite MOx materials for NVMs. In Metal Oxides for Non-volatile Memory, 1st ed.; Dimitrakis, P., Valov, I., Tappertzhofen, S., Eds.; Elsevier Science: Amsterdam, Netherlands, 2022; Chapter 7, pp. 201-244. [CrossRef]
- Surmenev, R.A.; Ivanova, A.A.; Epple, M.; Pichugin, V.F.; Surmeneva, M.A. Physical principles of radio-frequency magnetron sputter deposition of calcium-phosphate-based coating with tailored properties. Surf. Coat. Technol. 2021, 413, 127098. [CrossRef]
- Ivanova, A.A.; Surmeneva, M.A.; Tyurin, A.I.; Surmenev, R.A. Correlation between structural and mechanical properties of RF magnetron sputter deposited hydroxyapatite coating. Mater. Charact. 2018, 142, 261-269. [CrossRef]
- Nelea, V.; Morosanu, C.; Iliescu, M. Mihailescu, I.N. Microstructure and mechanical properties of hydroxyapatite thin films grown by RF magnetron sputtering. Surf. Coat. Technol. 2003, 173 (2-3), 315-322. [CrossRef]
- Safavi, M.S.; Surmeneva, M.A.; Surmenev, R.A.; Khalil-Allafi, J. RF-magnetron sputter deposited hydroxyapatite-based composite & multilayer coatings: A systematic review from mechanical, corrosion, and biological points of view. Ceram. Int. 2021, 47 (3), 3031-3053. [CrossRef]
- Chernozem, R.V.; Surmeneva, M.A.; Krause, B.; Baumbach, T.; Ignatov, V.P.; Tyurin, A.I.; Loza, K.; Epple, M.; Surmenev, R.A. Hybrid biocomposites based on titania nanotubes and a hydroxyapatite coating deposited by RF-magnetron sputtering: Surface topography, structure, and mechanical properties. Appl. Surf. Sci. 2017, 426, 229-237. [CrossRef]
- Surmeneva, M.A.; Ivanova, A.A.; Tian, Q.; Pittman, R.; Jiang, W.; Lin, J.; Liu, H.H.; Surmenev, R.A. Bone marrow derived mesenchymal stem cell response to the RF magnetron sputter deposited hydroxyapatite coating on AZ91 magnesium alloy. Mater. Chem. Phys. 2019, 221, 89-98. [CrossRef]
- Garcia-Sanz, F.J.; Mayor, M.B.; Arias, J.L.; Pou, J.; Leon, B.; Perez-Amor, M. Hydroxyapatite coatings: A comparative study between plasma-spray and pulsed laser deposition techniques. J. Mater. Sci. Mater. Med. 1997, 8, 861-865. [CrossRef]
- Koch, C.F.; Johnson, S.; Kumar, D.; Jelinek, M.; Chrisey, D.B.; Doraiswamy, A.; Jin, C.; Narayan, R.J.; Mihailescu, I.N. Pulsed laser deposition of hydroxyapatite thin films. Mater. Sci. Eng. C 2007, 27 (2-3), 484-494. [CrossRef]
- Popescu-Pelin, G.; Sima, F.; Sima, L.E.; Mihailescu, C.N.; Luculescu, C.; Iordache, I.; Socol, M.; Socol, G.; Mihailescu, I.N. Hydroxyapatite thin films grown by pulsed laser deposition and matrix assisted pulsed laser evaporation: Comparative study. Appl. Surf. Sci. 2017, 418 (B), 580-588. [CrossRef]
- Cutroneo, M.; Havranek, V.; Flaks, J.; Malinsky, P.; Torrisi, L. Pulsed Laser Deposition and Laser-Induced Backward Transfer to Modify Polydimethylsiloxane. Coatings 2021, 11, 1521. [CrossRef]
- Nishikawa, H.; Hasegawa, T.; Miyake, A.; Tashiro, Y.; Hashimoto, Y.; Blank, D.H.A.; Rijnders, G. Relationship between the Ca/P ratio of hydroxyapatite thin films and the spatial energy distribution of the ablation laser in pulsed laser deposition. Mater. Lett. 2016, 165, 95-98. [CrossRef]
- González-Estrada, O.A.; Pertuz Comas, A.D.; Ospina, R. Characterization of hydroxyapatite coatings produced by pulsed-laser deposition on additive manufacturing Ti6Al4V ELI. Thin solid films 2022, 763, 139592. [CrossRef]
- Duta, L.; Popescu, A.C. Current status on pulsed laser deposition of coatings from animal-origin calcium phosphate sources. Coatings 2019, 9 (5), 335. [CrossRef]
- Saallah, S.; Lenggoro, I.W. Nanoparticles Carrying Biological Molecules: Recent Advances and Applications. KONA Powder Part. J. 2018, 35, 89-111. [CrossRef]
- Leeuwenburgh, S.; Wolke, J.; Schoonman, J.; Jansen, J. Electrostatic spray deposition (ESD) of calcium phosphate coatings. J. Biomed. Mater. Res. A 2003, 66, 330-334. [CrossRef]
- Leeuwenburgh, S.C.G.; Wolke, J.G.C.; Siebers, M.C.; Schoonman, J.; Jansen, J.A. In vitro and in vivo reactivity of porous, electrosprayed calcium phosphate coatings. Biomaterials 2006, 27 (18), 3368-3378. [CrossRef]
- Müller, V.; Pagnier, T.; Tadier, S.; Gremillard, L.; Jobbagy, M.; Djurado, E. Design of advanced one-step hydroxyapatite coatings for biomedical applications using the electrostatic spray deposition. Appl. Surf. Sci. 2021, 541, 148462. [CrossRef]
- Huang, J.; Jayasinghe, S.N.; Best, S.M.; Edirisinghe, M.J.; Brooks, R.A.; Bonfield, W. Electrospraying of a nano-hydroxyapatite suspension. J. Mater. Sci. 2004, 39, 1029-1032. [CrossRef]
- Matsuura, T.; Maruyama, T. Calcium phosphate-polymer hybrid microparticles having functionalized surfaces prepared by a coaxially electrospray technique. Colloids Surf. A Physicochem. Eng. Asp. 2017, 526, 64-69. [CrossRef]
- Boccaccini, A.R.; Keim, S.; Ma, R.; Li, Y.; Zhitomirsky, I. Electrophoretic deposition of biomaterials. J. Royal Soc. Interface 2010, 7 (5), S581-S613. [CrossRef]
- Corni, I.; Ryan, M.P.; Boccaccini, A.R. Electrophoretic deposition: From traditional ceramics to nanotechnology. J. Eur. Ceram. Soc. 2008, 28 (7), 1353-1367. [CrossRef]
- Boccaccini, A.R.; Zhitomirsky, I. Application of electrophoretic and electrolytic deposition techniques in ceramics processing. Curr. Opin. Solid State Mater. Sci. 2002, 6 (3), 251-260. [CrossRef]
- Besra, L.; Liu, M. A review on fundamentals and applications of electrophoretic deposition (EPD). Prog. Mater. Sci. 2007, 52(1), 1-61. [CrossRef]
- Drevet, R.; Ben Jaber, N.; Fauré, J.; Tara, A.; Ben Cheikh Larbi, A.; Benhayoune, H. Electrophoretic deposition (EPD) of Nano-hydroxyapatite coatings with improved mechanical properties on prosthetic Ti6Al4V substrates. Surf. Coat. Technol. 2016, 301, 94-99. [CrossRef]
- Azzouz, I.; Faure, J.; Khlifi, K.; Cheikh Larbi, A.; Benhayoune, H. Electrophoretic Deposition of 45S5 Bioglass® Coatings on the Ti6Al4V Prosthetic Alloy with Improved Mechanical Properties. Coatings 2020, 10, 1192. [CrossRef]
- Akhtar, M.A.; Hadzhieva, Z.; Dlouhý, I.; Boccaccini, A.R. Electrophoretic Deposition and Characterization of Functional Coatings Based on an Antibacterial Gallium (III)-Chitosan Complex. Coatings 2020, 10, 483. [CrossRef]
- Virk, R.S.; Rehman, M.A.U.; Munawar, M.A.; Schubert, D.W.; Goldmann, W.H.; Dusza, J.; Boccaccini, A.R. Curcumin-Containing Orthopedic Implant Coatings Deposited on Poly-Ether-Ether-Ketone/Bioactive Glass/Hexagonal Boron Nitride Layers by Electrophoretic Deposition. Coatings 2019, 9, 572. [CrossRef]
- Bartmański, M.; Pawłowski, Ł.; Strugała, G.; Mielewczyk-Gryń, A.; Zieliński, A. Properties of nanohydroxyapatite coatings doped with nanocopper, obtained by electrophoretic deposition on Ti13Zr13Nb alloy. Materials 2019, 12 (22), 3741. [CrossRef]
- Sakar, P.; Nicholson, P.S. Electrophoretic deposition (EPD): Mechanisms, kinetics and application to ceramics. J. Am. Ceram. Soc. 1996, 79(8), 1987-2002. [CrossRef]
- Ozhukil Kollath, V.; Chen, Q.; Closset, R.; Luyten, L.; Traina, K.; Mullens, S.; Boccaccini, A.R.; Cloots, R. AC vs. DC electrophoretic deposition of hydroxyapatite on titanium. J. Eur. Ceram. Soc. 2013, 33(13-14), 2715-2721. [CrossRef]
- Azzouz, I.; Khlifi, K.; Faure, J.; Dhiflaoui, H.; Larbi, A.B.C.; Benhayoune, H. Mechanical behavior and corrosion resistance of sol-gel derived 45S5 bioactive glass coating on Ti6Al4V synthesized by electrophoretic deposition. J. Mech. Behav. Biomed. Mater. 2022, 134,105352. [CrossRef]
- Shirkhanzadeh, M. Bioactive calcium phosphate coatings prepared by electrodeposition, J. Mater. Sci. Lett. 1991, 10 (23), 1415-1417. [CrossRef]
- Shirkhanzadeh, M. Calcium phosphate coatings prepared by electrocrystallization from aqueous electrolytes. J. Mater. Sci. – Mater. Med. 1995, 6 (2), 90-93. [CrossRef]
- Drevet, R.; Benhayoune, H. Electrochemical Deposition of Calcium Phosphate Coatings on a Prosthetic Titanium Alloy Substrate. In Calcium Phosphate: Structure, synthesis, properties and applications; Heimann, R.B.; Nova Science Publishers, Inc., Hauppauge, New York, USA, 2012, pp. 231-252. ISBN: 978-162257299-1.
- Drevet, R.; Benhayoune, H. Electrodeposition of Calcium Phosphate Coatings on Metallic Substrates for Bone Implant Applications: A Review. Coatings 2022, 12 (4), 539. [CrossRef]
- Redepenning, J.; McIsaac, J.P. Electrocrystallization of Brushite Coatings on Prosthetic Alloys. Chem. Mater. 1990, 2 (6), 625-627. [CrossRef]
- Zhitomirsky, I. Cathodic electrodeposition of ceramic and organoceramic materials. Fundamental aspects. Adv. Colloid Interface Sci. 2002, 97 (1-3), 279-317. [CrossRef]
- Eliaz, N.; Eliyahu, M. Electrochemical processes of nucleation and growth of hydroxyapatite on titanium supported by real-time electrochemical atomic force microscopy. J. Biomed. Mater. Res. A 2007, 80 (3), 621-634. [CrossRef]
- Eliaz, N.; Sridhar, T.M. Electrocrystallization of hydroxyapatite and its dependence on solution conditions. Cryst. Growth Des. 2008, 8 (11), 3965-3977. [CrossRef]
- Kuo, M.C.; Yen, S.K. The process of electrochemical deposited hydroxyapatite coatings on biomedical titanium at room temperature. Mater. Sci. Eng. C 2002, 20 (1-2), 153-160. [CrossRef]
- Zielinski, A.; Bartmanski, M. Electrodeposited biocoatings, their properties and fabrication technologies: A review. Coatings 2020, 10 (8), 782. [CrossRef]
- Lin, S.; LeGeros, R.Z.; LeGeros, J.P. Adherent octacalcium phosphate coating on titanium alloy using modulated electrochemical deposition method. J. Biomed. Mater. Res. A 2003, 66 (4), 819-828. [CrossRef]
- Furko, M.; Balázsi, C. Calcium phosphate based bioactive ceramic layers on implant materials preparation, properties, and biological performance. Coatings 2020, 10 (9), 823. [CrossRef]
- Drevet, R.; Lemelle, A.; Untereiner, V.; Manfait, M.; Sockalingum, G.D.; Benhayoune, H. Morphological modifications of electrodeposited calcium phosphate coatings under amino acids effect. Appl. Surf. Sci. 2013, 268, 343-348. [CrossRef]
- Drevet, R.; Viteaux, A.; Maurin, J.C.; Benhayoune, H. Human osteoblast-like cells response to pulsed electrodeposited calcium phosphate coatings. RSC Advances 2013, 3, 11148-11154. [CrossRef]
- Vidal, E.; Buxadera-Palomero, J.; Pierre, C.; Manero, J.M.; Ginebra, M.P.; Cazalbou, S.; Combes, C.; Rupérez, E.; Rodríguez, D. Single-step pulsed electrodeposition of calcium phosphate coatings on titanium for drug delivery. Surf. Coat. Technol. 2019, 358, 266-275. [CrossRef]
- Jimenez-Garcia, F.N.; Giraldo-Torres, L.R.; Restrepo-Parra, E. Electrochemically deposited calcium phosphate coatings using a potentiostat of in-house design and implementation. Mater. Res. 2021, 25 (5), e20210098. [CrossRef]
- Vidal, E.; Guillem-Marti, J.; Ginebra, M.-P.; Combes, C.; Rupérez, E.; Rodriguez, D. Multifunctional homogeneous calcium phosphate coatings: Toward antibacterial and cell adhesive titanium scaffolds. Surf. Coat. Technol. 2021, 405, 126557. [CrossRef]
- Safavi, M.S.; Walsh, F.C.; Surmeneva, M.A.; Surmenev, R.A.; Khalil-Allafi, J. Electrodeposited hydroxyapatite-based biocoatings: Recent progress and future challenges. Coatings 2021, 11 (1), 110. [CrossRef]
- Gao, A.; Hang, R.; Bai, L.; Tang, B.; Chu, P.K. Electrochemical surface engineering of titanium-based alloys for biomedical application. Electrochim. Acta 2018, 271, 699-718. [CrossRef]
- Ben Jaber, N.; Drevet, R.; Fauré, J.; Demangel, C.; Potiron, S.; Tara, A.; Ben Cheikh Larbi, A.; Benhayoune, H. A new process for the thermal treatment of calcium phosphate coatings electrodeposited on Ti6Al4V substrate. Adv. Eng. Mater. 2015, 17 (11), 1608-1615. [CrossRef]
- Degli Esposti, L.; Markovic, S.; Ignjatovic, N.; Panseri, S.; Montesi, M.; Adamiano, A.; Fosca, M.; Rau, J.V.; Uskoković, V.; Iafisco, M. Thermal crystallization of amorphous calcium phosphate combined with citrate and fluoride doping: A novel route to produce hydroxyapatite bioceramics. J. Mater. Chem. B 2021, 9, 4832-4845. [CrossRef]
- Gerk, S.A.; Golovanova, O.A.; Odazhiu, V.N. Structural, Morphological, and Resorption Properties of Carbonate Hydroxyapatite Prepared in the Presence of Glycine. Inorg. Mater. 2018, 54 (3), 305-314. [CrossRef]
- Hu, Q.; Tan, Z.; Liu, Y.; Tao, J.; Cai, Y. ; Zhang, M. ; Pan, H. ; Xu, X.; Tang, R. Effect of crystallinity of calcium phosphate nanoparticles on adhesion, proliferation, and differentiation of bone marrow mesenchymal stem cells. J. Mater. Chem. 2007, 17, 4690-4698. [CrossRef]
- Implants for surgery - Hydroxyapatite - Part 2: Thermally sprayed coatings of hydroxyapatite 2018, ISO 13779-2.
- Raynaud, S.; Champion, E.; Bernache-Assollant, D. Calcium phosphate apatites with variable Ca/P atomic ratio II. Calcination and sintering. Biomaterials 2002, 23 (4), 1073-1080. [CrossRef]
- Destainville, A.; Champion, E.; Bernache-Assollant, D.; Laborde, E. Synthesis, characterization and thermal behavior of apatitic tricalcium phosphate. Mater. Chem. Phys. 2003, 80 (1), 269-277. [CrossRef]
- Implants for surgery - Hydroxyapatite - Part 3: Analyse chimique et caractérisation du rapport de cristallinité et de la pureté de phase 2018, ISO 13779-3.
- Ducheyne, P.; Qiu, Q. Bioactive ceramics: The effect of surface reactivity on bone formation and bone cell function. Biomaterials 1999, 20 (23-24), 2287-2303. [CrossRef]
- Iwamoto, T.; Hieda, Y.; Kogai, Y. Effect of hydroxyapatite surface morphology on cell adhesion. Mater. Sci. Eng. C 2016, 69, 1263-1267. [CrossRef]
- Drevet R.; Fauré, J.; Benhayoune, H. Structural and morphological study of electrodeposited calcium phosphate materials submitted to thermal treatment. Mater. Lett. 2017, 209, 27-31. [CrossRef]
- Liu, S.; Li, H.; Zhang, L.; Yin, X.; Guo, Y. In simulated body fluid performance of polymorphic apatite coatings synthesized by pulsed electrodeposition. Mater. Sci. Eng. C 2017, 79, 100-107. [CrossRef]
- Lee, W.K.; Lee, S.M.; Kim, H.M. Effect of surface morphology of calcium phosphate on osteoblast-like HOS cell responses. J. Ind. Eng. Chem. 2009, 15 (5), 677-682. [CrossRef]
- Cairns, M.L.; Meenan, B.J.; Burke, G.A.; Boyd, A.R. Influence of surface topography on osteoblast response to fibronectin coated calcium phosphate thin films. Colloids Surf. B 2010, 78 (2), 283-290. [CrossRef]
- Pujari-Palmer, S.; Chen, S.; Rubino, S.; Weng, H.; Xia, W.; Engqvist, H.; Tang, L.; Ott, M.K. In vivo and in vitro evaluation of hydroxyapatite nanoparticle morphology on the acute inflammatory response. Biomaterials 2016, 90, 1-11. [CrossRef]
- Chen, S.; Guo, Y.; Liu, R.; Wu, S.; Fang, J.; Huang, B.; Li, Z.; Chen, Z.; Chen, Z. Tuning surface properties of bone biomaterials to manipulate osteoblastic cell adhesion and the signaling pathways for the enhancement of early osseointegration. Colloids Surf. B 2018, 164, 58-69. [CrossRef]
- Khlusov, I.A.; Dekhtyar, Y.; Sharkeev, Y.P.; Pichugin, V.F.; Khlusova, M.Y.; Polyaka, N.; Tyulkin, F.; Vendinya, V.; Legostaeva, E.V.; Litvinova, L.S.; Shupletsova, V.V.; Khaziakhmatova, O.G.; Yurova, K.A.; Prosolov, K.A. Nanoscale electrical potential and roughness of a calcium phosphate surface promotes the osteogenic phenotype of stromal cells. Materials 2018, 11 (6), 978. [CrossRef]
- Deligianni, D.D.; Katsala, N.D.; Koutsoukos, P.G.; Missirlis, Y.F. Effect of surface roughness of hydroxyapatite on human bone marrow cell adhesion, proliferation, differentiation and detachment strength. Biomaterials 2000, 22 (1), 87-96. [CrossRef]
- Anselme, K.; Bigerelle, M. On the Relation Between Surface Roughness of Metallic Substrates and Adhesion of Human Primary Bone Cells. Scanning 2014, 36, 11-20. [CrossRef]
- Giljean, S.; Bigerelle, M.; Anselme, K. Roughness Statistical Influence on Cell Adhesion Using Profilometry and Multiscale Analysis. Scanning 2014, 36, 2-10. [CrossRef]
- Adeleke, S.A.; Ramesh, S.; Bushroa, A.R.; Ching, Y.C.; Sopyan, I.; Maleque, M.A.; Krishnasamy, S.; Chandran, H.; Misran, H., Sutharsini, U. The properties of hydroxyapatite ceramic coatings produced by plasma electrolytic oxidation. Ceram. Int. 2018, 44 (2), 1802-1811. [CrossRef]
- Maidaniuc, A.; Miculescu, F.; Voicu, S.I.; Andronescu, C.; Miculescu, M.; Matei, M.; Mocanu, A.C.; Pencea, I.; Csaki, I.; Machedon-Pisu, T.; Ciocan, L.T. Induced wettability and surface-volume correlation of composition for bovine bone derived hydroxyapatite particles. Appl. Surf. Sci. 2018, 438, 158-166. [CrossRef]
- Paital, S.R.; Dahotre, N.B. Wettability and kinetics of hydroxyapatite precipitation on a laser-textured Ca-P bioceramic coating. Acta Biomaterialia 2009, 5 (7), 2763-2772. [CrossRef]
- Bodhak, S.; Bose, S.; Bandyopadhyay, A. Role of surface charge and wettability on early stage mineralization and bone cell–materials interactions of polarized hydroxyapatite. Acta Biomaterialia 2009, 5 (6), 2178-2188. [CrossRef]
- Doshi, B.; Sillanpää, M.; Kalliola, S. A review of bio-based materials for oil spill treatment. Water Res. 2018, 135, 262-277. [CrossRef]
- Thian, E.S.; Ahmad, Z.; Huang, J.; Edirisinghe, M.J.; Jayasinghe S.N.; Ireland, D.C.; Brooks, R.A.; Rushton, N.; Bonfield, W.; Best, S.M. The role of surface wettability and surface charge of electrosprayed nanoapatites on the behaviour of osteoblasts. Acta Biomaterialia 2010, 6 (3), 750-755. [CrossRef]
- Aronov, D.; Rosen, R.; Ron, E.Z.; Rosenman, G. Tunable hydroxyapatite wettability: Effect on adhesion of biological molecules. Process Biochem. 2006, 41 (12), 2367-2372. [CrossRef]
- Fornell, J.; Feng, Y.P.; Pellicer, E.; Suriñach, S.; Baró, M.D.; Sort, J. Mechanical behaviour of brushite and hydroxyapatite coatings electrodeposited on newly developed FeMnSiPd alloys. J. Alloys Compd. 2017, 729, 231-239. [CrossRef]
- Fathyunes, L.; Khalil-Allafi, J.; Moosavifar, M. Development of graphene oxide/calcium phosphate coating by pulse electrodeposition on anodized titanium: Biocorrosion and mechanical behavior. J. Mech. Behav. Biomed. Mater. 2019, 90, 575-586. [CrossRef]
- Singh, S.; Prakash, C.; Singh, H. Deposition of HA-TiO2 by plasma spray on β-phase Ti-35Nb-7Ta-5Zr alloy for hip stem: Characterization, mechanical properties, corrosion, and in-vitro bioactivity. Surf. Coat. Technol. 2020, 398, 126072. [CrossRef]
- Drevet, R.; Fauré, J.; Benhayoune, H. Thermal Treatment Optimization of Electrodeposited Hydroxyapatite Coatings on Ti6Al4V Substrate. Adv. Eng. Mater. 2012, 14 (6), 377-382. [CrossRef]
- Harun, W.S.W.; Asri, R.I.M.; Alias, J.; Zulkifli, F.H.; Ghani, S.A.C.; Shariffuddin, J.H.M. A comprehensive review of hydroxyapatite-based coatings adhesion on metallic biomaterials. Ceram. Int. 2018, 44, 1250-1268. [CrossRef]
- Implants for surgery - Hydroxyapatite - Part 4: Determination of coating adhesion strength 2018, ISO 13779-4.
- Uskoković, V. Ion-doped hydroxyapatite: An impasse or the road to follow? Ceram. Int. 2020, 46, 11443-11465. [CrossRef]
- Furko, M.; Balázsi, C. Morphological, chemical, and biological investigation of ionic substituted, pulse current deposited calcium phosphate coatings. Materials 2020, 13 (20), 4690. [CrossRef]
- Ungureanu, E.; Vranceanu, D.M.; Vladescu, A.; Parau, A.C.; Tarcolea, M.; Cotrut, C.M. Effect of doping element and electrolyte’s ph on the properties of hydroxyapatite coatings obtained by pulsed galvanostatic technique. Coatings 2021, 11 (12), 1522. [CrossRef]
- Panda, S.; Biswas, C.K.; Paul, S. A comprehensive review on the preparation and application of calcium hydroxyapatite: A special focus on atomic doping methods for bone tissue engineering. Ceram. Int. 2021, 47 (20), 28122-28144. [CrossRef]
- Schatkoski, V.M.; Larissa do Amaral Montanheiro, T.; Canuto de Menezes, B.R.; Pereira, R.M.; Rodrigues, K.F.; Ribas, R.G.; Morais da Silva, D.; Thim, G.P. Current advances concerning the most cited metal ions doped bioceramics and silicate-based bioactive glasses for bone tissue engineering. Ceram. Int. 2021, 47 (3), 2999-3012. [CrossRef]
- Boanini, E.; Gazzano, M.; Bigi, A. Ionic substitutions in calcium phosphates synthesized at low temperature. Acta Biomater. 2010, 6 (6), 1882-1894. [CrossRef]
- Bigi, A.; Boanini, E.; Gazzano, M. Ion Substitution in Biological and Synthetic Apatites. Biomineralization and Biomaterials: Fundamentals and Applications 2016, pp. 235-266. [CrossRef]
- Wang, W.; Yeung, K.W.K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact. Mater. 2017, 2, 224-247. [CrossRef]
- Panda, S.; Kumar Biswas, C.; Paul, S. A comprehensive review on the preparation and application of calcium hydroxyapatite: A special focus on atomic doping methods for bone tissue engineering. Ceram. Int. 2021, 47, 28122-28144. [CrossRef]
- Arcos, D.; Vallet-Regi, M. Substituted hydroxyapatite coatings of bone implants. J. Mater. Chem. B 2020, 8, 1781-1800. [CrossRef]
- Ratnayake, J.T.B.; Mucalo, M.; Dias, G.J. Substituted hydroxyapatites for bone regeneration: A review of current trends. J. Biomed. Mater. Res. B 2017, 105B, 1285-1299. [CrossRef]
- Dubnika, A.; Loca, D.; Rudovica, V.; Parekh, M.B.; Berzina-Cimdina, L. Functionalized Silver Doped Hydroxyapatite Scaffolds for Controlled Simultaneous Ion and Drug Delivery. Ceram. Int. 2017, 43, 3698-3705. [CrossRef]
- Chen, K.; Ustriyana, P.; Moore, F.; Sahai, N. Biological Response of and Blood Plasma Protein Adsorption on Silver-Doped Hydroxyapatite. ACS Biomater. Sci. Eng. 2019, 5, 561−571. [CrossRef]
- Mokabber, T.; Cao, H.T.; Norouzi, N.; van Rijn, P.; Pei, Y.T. Antimicrobial Electrodeposited Silver-Containing Calcium Phosphate Coatings. ACS Appl. Mater. Interfaces 2020, 12, 5531-5541. [CrossRef]
- Wiesmann, H.P.; Plate, U.; Zierold, K.; Höhling, H.J. Potassium is Involved in Apatite Biomineralization. J. Dent. Res. 1998, 77 (8), 1654-1657. [CrossRef]
- Kannan, S.; Ventura, J.M.G.; Ferreira, J.M.F. Synthesis and thermal stability of potassium substituted hydroxyapatites and hydroxyapatite/b-tricalcium phosphate mixtures. Ceram. Int. 2007, 33, 1489-1494. [CrossRef]
- Kumar, M.; Xie, J.; Chittur, K.; Riley, C. Transformation of modified brushite to hydroxyapatite in aqueous solution: Effects of potassium substitution. Biomaterials 1999, 20, 1389-1399. [CrossRef]
- Kaygili, O.; Keser, S.; Ates, T.; Yakuphanoglu, F. Synthesis and characterization of lithium calcium phosphate ceramics. Ceram. Int. 2013, 39, 7779-7785. [CrossRef]
- Pan, C.; Chen, L.; Wu, R.; Shan, H.; Zhou, Z.; Lin, Y.; Yu, X.; Yan, L.; Wu, C. Lithium-containing biomaterials inhibit osteoclastogenesis of macrophages in vitro and osteolysis in vivo. J. Mater. Chem. B 2018, 6, 8115-8126. [CrossRef]
- Wang, Y.; Yang, X.; Gu, Z.; Qin, H.; Li, L.; Liu, J.; Yu, X. In vitro study on the degradation of lithium-doped hydroxyapatite for bone tissue engineering scaffold. Mater. Sci. Eng. C 2016, 66, 185-192. [CrossRef]
- Li, H.; Zhao, X.; Cao, S.; Li, K.; Chen, M.; Xu, Z.; Lu, J.; Zhang, L. Na-doped hydroxyapatite coating on carbon/carbon composites: Preparation, in vitro bioactivity and biocompatibility. Appl. Surf. Sci. 2012, 263, 163-173. [CrossRef]
- Kannan, S.; Ventura, J.M.G.; Lemos, A.F.; Barba, A.; Ferreira, J.M.F. Effect of sodium addition on the preparation of hydroxyapatites and biphasic ceramics. Ceram. Int. 2008, 34 (1), 7-13. [CrossRef]
- Sang Cho, J.; Um, S.H.; Su Yoo, D.; Chung, Y.C.; Chung, S.H.; Lee, J.C.; Rhee, S.H. Enhanced osteoconductivity of sodium-substituted hydroxyapatite by system instability. J. Biomed. Mater. Res. B 2014, 102 (5), 1046-1062. [CrossRef]
- Tang, C.-M.; Fan, F.-Y.; Ke, Y.-C.; Lin, W.-C. Effects of electrode plate annealing treatment and the addition of hydrogen peroxide on improving the degradation of cobalt hydroxyapatite for bone repair. Mater. Chem. Phys. 2021, 259, 123962. [CrossRef]
- Lin, W.C.; Chuang, C.C.; Wang, P.T.; Tang, C.M. A comparative study on the direct and pulsed current electrodeposition of cobalt-substituted hydroxyapatite for magnetic resonance imaging application. Mater. 2018, 12 (1), 116. [CrossRef]
- Drevet, R.; Zhukova, Y.; Dubinskiy, S.; Kazakbiev, A.; Naumenko, V.; Abakumov, M.; Fauré, J.; Benhayoune, H.; Prokoshkin, S. Electrodeposition of cobalt-substituted calcium phosphate coatings on Ti22Nb6Zr alloy for bone implant applications. J. Alloys Compd. 2019, 793, 576-582. [CrossRef]
- Grass, G.; Rensing, C.; Solioz, M. Metallic copper as an antimicrobial surface. Appl. Environ. Microbiol. 2011, 77 (5), 1541-1547. [CrossRef]
- Wolf-Brandstetter, C.; Oswald, S.; Bierbaum, S.; Wiesmann, H.-P.; Scharnweber, D. Influence of pulse ratio on codeposition of copper species with calcium phosphate coatings on titanium by means of electrochemically assisted deposition. J. Biomed. Mater. Res. B 2014, 102 (1), 160-172. [CrossRef]
- Prosolov, K.A.; Lastovka, V.V.; Khimich, M.A.; Chebodaeva, V.V.; Khlusov, I.A.; Sharkeev, Y.P. RF Magnetron Sputtering of Substituted Hydroxyapatite for Deposition of Biocoatings. Materials 2022, 15, 6828. [CrossRef]
- Farzadi, A.; Bakhshi, F.; Solati-Hashjin, M.; Asadi-Eydivand, M.; Osman, N.A.A. Magnesium incorporated hydroxyapatite: Synthesis and structural properties characterization. Ceram. Int. 2014, 40 (4), 6021-6029. [CrossRef]
- Cacciotti, I.; Bianco, A.; Lombardi, M.; Montanaro, L. Mg-substituted hydroxyapatite nanopowders: Synthesis, thermal stability and sintering behaviour. J. Eur. Ceram. Soc. 2009, 29 (14), 2969-2978. [CrossRef]
- Vranceanu, D.M.; Ionescu, I.C.; Ungureanu, E.; Cojocaru, M.O.; Vladescu, A.; Cotrut, C.M. Magnesium doped hydroxyapatite-based coatings obtained by pulsed galvanostatic electrochemical deposition with adjustable electrochemical behavior. Coatings 2020, 10, 727. [CrossRef]
- Huang, Y.; Qiao, H.; Nian, X.; Zhang, X.; Zhang, X.; Song, G.; Xu, Z.; Zhang, H.; Han, S. Improving the bioactivity and corrosion resistance properties of electrodeposited hydroxyapatite coating by dual doping of bivalent strontium and manganese ion. Surf. Coat. Technol. 2016, 291, 205-215. [CrossRef]
- Huang, Y.; Ding, Q.; Han, S.; Yan, Y.; Pang, X. Characterisation, corrosion resistance and in vitro bioactivity of manganese-doped hydroxyapatite films electrodeposited on titanium. J. Mater. Sci. – Mater. Med. 2013, 24, 1853-1864. [CrossRef]
- Fadeeva, I.V.; Kalita, V.I.; Komlev, D.I.; Radiuk, A.A.; Fomin, A.S.; Davidova, G.A.; Nadezhda K. Fursova, N.K.; Murzakhanov, F.F.; Gafurov, M.R.; Fosca, M.; Antoniac, I.V.; Barinov, S.M., Rau, J.V. In Vitro Properties of Manganese-Substituted Tricalcium Phosphate Coatings for Titanium Biomedical Implants Deposited by Arc Plasma. Mater. 2020, 13 (19), 4411. [CrossRef]
- Pilmane, M.; Salma-Ancane, K.; Loca, D.; Locs, J.; Berzina-Cimdina, L. Strontium and strontium ranelate: Historical review of some of their functions. Mater. Sci. Eng. C 2017, 78, 1222-1230. [CrossRef]
- Boanini, E.; Torricelli, P.; Fini, M.; Bigi, A. Osteopenic bone cell response to strontium-substituted hydroxyapatite. J. Mater. Sci. – Mater. Med. 2011, 22 (9), 2079-2088. [CrossRef]
- Drevet, R.; Benhayoune, H. Pulsed electrodeposition for the synthesis of strontium-substituted calcium phosphate coatings with improved dissolution properties. Mater. Sci. Eng. C 2013, 33, 4260-4265. [CrossRef]
- Capuccini, C.; Torricelli, P.; Sima, F.; Boanini, E.; Ristoscu, C.; Bracci, B.; Socol, G.; Fini, M.; Mihailescu, I.N.; Bigi, A. Strontium-substituted hydroxyapatite coatings synthesized by pulsed-laser deposition: In vitro osteoblast and osteoclast response. Acta Biomaterialia 2008, 4 (6), 1885-1893. [CrossRef]
- Tang, Y.; Chappell, H.F.; Dove, M.T.; Reeder, R.J.; Lee, Y.J. Zinc incorporation into hydroxylapatite. Biomaterials 2009, 30 (15), 2864-2872. [CrossRef]
- Huang, Y.; Zhang, X.; Mao, H.; Li, T.; Zhao, R.; Yan, Y.; Pang, X. Osteoblastic cell responses and antibacterial efficacy of Cu/Zn co-substituted hydroxyapatite coatings on pure titanium using electrodeposition method. RSC Advances 2015, 5 (22), 17076-17086. [CrossRef]
- Furko, M.; Jiang, Y.; Wilkins, T.; Balázsi, C. Development and characterization of silver and zinc doped bioceramic layer on metallic implant materials for orthopedic application. Ceram. Int. 2016, 42, 4924-4931. [CrossRef]
- El Khouri, A.; Zegzouti, A.; Elaatmani, M.; Capitelli, F. Bismuth-substituted hydroxyapatite ceramics synthesis: Morphological, structural, vibrational and dielectric properties. Inorg. Chem. Commun. 2019, 110, 107568. [CrossRef]
- Ciobanu, G.; Bargan, A.M.; Luca, C. New Bismuth-Substituted Hydroxyapatite Nanoparticles for Bone Tissue Engineering. JOM 2015, 67 (11), 2534-2542. [CrossRef]
- Ahmed, M.K.; Mansour, S.F.; Mostafa, M.S.; Darwesh, R.; El-dek, S.I. Structural, mechanical and thermal features of Bi and Sr co-substituted hydroxyapatite. J. Mater. Sci. 2019, 54 (3), 1977-1991. [CrossRef]
- Lin, Y.; Yang, Z.; Cheng, J. Preparation, Characterization and Antibacterial Property of Cerium Substituted Hydroxyapatite Nanoparticles. J. Rare Earths 2007, 25 (4), 452-456. [CrossRef]
- Feng, Z.; Liao, Y.; Ye, M. Synthesis and structure of cerium-substituted hydroxyapatite. J. Mater. Sci. – Mater. Med. 2005, 16 (5), 417-421. [CrossRef]
- Ciobanu, G.; Harja, M. Cerium-doped hydroxyapatite/collagen coatings on titanium for bone implants. Ceram. Int. 2019, 45 (2), 2852-2857. [CrossRef]
- Nisar, A.; Iqbal, S.; Atiq Ur Rehman, M.; Mahmood, A.; Younas, M.; Hussain, S.Z.; Tayyaba, Q.; shah, A. Study of physico-mechanical and electrical properties of cerium doped hydroxyapatite for biomedical applications. Mater. Chem. Phys. 2023, 299, 127511. [CrossRef]
- Alshemary, A.Z.; Akram, M.; Goh, Y.F.; Abdul Kadir, M.R.; Abdolahi, A. Hussain, R. Structural characterization, optical properties and in vitro bioactivity of mesoporous erbium-doped hydroxyapatite. J. Alloys Compds. 2015, 645, 478-486. [CrossRef]
- Neacsu, I.A.; Stoica, A.E.; Vasile, B.S.; Andronescu, E. Luminescent hydroxyapatite doped with rare earth elements for biomedical applications. Nanomaterials 2019, 9 (2), 239. [CrossRef]
- Pham, V.H.; Van, H.N.; Tam, P.D.; Ha, H.N.T. A novel 1540 nm light emission from erbium doped hydroxyapatite/β-tricalcium phosphate through co-precipitation method. Mater. Lett. 2016, 167, 145-147. [CrossRef]
- Yang, P.; Quan, Z.; Li, C.; Kang, X.; Lian, H. Lin, J. Bioactive, luminescent and mesoporous europium-doped hydroxyapatite as a drug carrier. Biomaterials 2008, 29 (32), 4341-4347. [CrossRef]
- Al-Kattan, A.; Santran, V.; Dufour, P.; Dexpert-Ghys, J.; Drouet, C. Novel contributions on luminescent apatite-based colloids intended for medical imaging. J. Biomater. Appl. 2014, 28 (5), 697-707. [CrossRef]
- Graeve, O.A.; Kanakala, R.; Madadi, A.; Williams, B.C.; Glass, K.C. Luminescence variations in hydroxyapatites doped with Eu2+ and Eu3+ ions. Biomaterials 2010, 31 (15), 4259-4267. [CrossRef]
- Singh, R.K.; Srivastava, M.; Prasad, N.K.; Awasthi, S.; Dhayalan, A.; Kannan, S. Iron doped β-Tricalcium phosphate: Synthesis, characterization, hyperthermia effect, biocompatibility and mechanical evaluation. Mater. Sci. Eng. C 2017, 78, 715-726. [CrossRef]
- Singh, R.K.; Srivastava, M.; Prasad, N.K.; Shetty, P.H.; Kannan, S. Hyperthermia effect and antibacterial efficacy of Fe3+ /Co2+ co-substitutions in β-Ca3(PO4)2 for bone cancer and defect therapy. J. Biomed. Mater. Res. B 2018, 106 (3), 1317-1328. [CrossRef]
- Predoi, D.; Iconaru, S.L.; Ciobanu, S.C.; Predoi, S.A.; Nuton, N.; Megier, C.; Beuran, M. Development of Iron-Doped Hydroxyapatite Coatings. Coatings 2021, 11 (2), 186. [CrossRef]
- Melnikov, P.; Teixeira, A.R.; Malzac, A.; Coelho, M.d.B. Gallium-containing hydroxyapatite for potential use in orthopedics. Mater. Chem. Phys. 2009, 117 (1), 86-90. [CrossRef]
- Korbas, M.; Rokita, E.; Meyer-Klaucke, W.; Ryczek, J. Bone tissue incorporates in vitro gallium with a local structure similar to gallium-doped brushite. J. Biol. Inorg. Chem. 2004, 9 (1), 67-76. [CrossRef]
- Mosina, M.; Siverino, C.; Stipniece, L.; Sceglovs, A.; Vasiljevs, R.; Moriarty, T.F.; Locs, J. Gallium-Doped Hydroxyapatite Shows Antibacterial Activity against Pseudomonas aeruginosa without Affecting Cell Metabolic Activity. J. Funct. Biomater. 2023, 14, 51. [CrossRef]
- Paduraru, A.V.; Oprea, O.; Musuc, A.M.; Vasile, B.S.; Iordache, F.; Andronescu, E. Influence of Terbium Ions and Their Concentration on the Photoluminescence Properties of Hydroxyapatite for Biomedical Applications. Nanomaterials 2021, 11, 2442. [CrossRef]
- Jiménez-Flores, Y.; Suárez-Quezada, M.; Rojas-Trigos, J.B.; Lartundo-Rojas, J.B.; Suárez, V., Mantilla, A. Characterization of Tb-doped hydroxyapatite for biomedical applications: Optical properties and energy band gap determination. J. Mater. Sci. 2017, 52, 9990-10000. [CrossRef]
- Demnati, I.; Grossin, D.; Combes, C.; Parco, M.; Braceras, I; Rey, C. A comparative physico-chemical study of chlorapatite and hydroxyapatite: From powders to plasma sprayed thin coatings. Biomed. Mater. 2012, 7 (5), 054101. [CrossRef]
- Navarrete-Segado, P.; Frances, C; Tourbin, M.; Tenailleau, C.; Duployer, B.; Grossin, D. Powder bed selective laser process (sintering/melting) applied to tailored calcium phosphate-based powders. Addit. Manuf. 2022, 50, 102542. [CrossRef]
- Ito, A.; Otsuka, Y.; Takeuchi, M.; Tanaka, H. Mechanochemical synthesis of chloroapatite and its characterization by powder X-ray diffractometory and attenuated total reflection-infrared spectroscopy. Colloid. Polym. Sci. 2017, 295, 2011-2018. [CrossRef]
- Merry, J.C.; Gibson, I.R.; Best, S.M.; Bonfield, W. Synthesis and characterization of carbonate hydroxyapatite. J. Mater. Sci. Mater. Med. 1998, 9, 779-783. [CrossRef]
- Zhang, L.; Li, H.; Li, K.; Song, Q.; Fu, Q.; Zhang, Y.; Liu, S. Electrodeposition of carbonate-containing hydroxyapatite on carbon nanotubes/carbon fibers hybrid materials for tissue engineering application. Ceram. Int. 2015, 41 (3), 4930-4935. [CrossRef]
- Landi, E.; Celotti, G.; Logroscino, G.; Tampieri, A. Carbonated hydroxyapatite as bone substitute. J. Europ. Ceram. Soc. 2003, 23 (15), 2931-2937. [CrossRef]
- Ge, X.; Zhao, J.; Lu, X.; Li, Z.; Wang, K.; Ren, F.; Wang, M.; Wang, Q.; Qian, B. Controllable phase transformation of fluoridated calcium phosphate ultrathin coatings for biomedical applications. J. Alloys Compd. 2020, 847, 155920. [CrossRef]
- Wang, J.; Chao, Y.; Wan, Q.; Zhu, Z.; Yu, H. Fluoridated hydroxyapatite coatings on titanium obtained by electrochemical deposition. Acta Biomater. 2009, 5 (5), 1798-1807. [CrossRef]
- Sun, J.; Wu, T.; Fan, Q.; Hu, Q.; Shi, B. Comparative study of hydroxyapatite, fluor-hydroxyapatite and Si-substituted hydroxyapatite nanoparticles on osteogenic, osteoclastic and antibacterial ability. RSC Adv. 2019, 9, 16106-16118. [CrossRef]
- Wang, Y.; Wang, J.; Hao, H.; Cai, M.; Wang, S.; Ma, J.; Li, Y. Mao, C.; Zhang, S. In Vitro and in Vivo Mechanism of Bone Tumor Inhibition by Selenium-Doped Bone Mineral Nanoparticles. ACS Nano 2016, 10 (11), 9927-9937. [CrossRef]
- Rodríguez-Valencia, C.; López-Álvarez, M.; Cochón-Cores, B.; Pereiro, I.; Serra, J.; González, P. Novel selenium-doped hydroxyapatite coatings for biomedical applications. J. Biomed. Mater. Res. A 2013, 101 (3), 853-861. [CrossRef]
- Tan, H.W.; Mo, H.Y.; Lau, A.T.Y.; Xu, Y.M. Selenium species: Current status and potentials in cancer prevention and therapy. Int. J. Mol. Sci. 2019, 20 (1), 75. [CrossRef]
- Casarrubios, L.; Gómez-Cerezo, N.; Sánchez-Salcedo, S.; Feito, M.J.; Serrano, M.C.; Saiz-Pardo, M.; Ortega, L.; de Pablo, D.; Díaz-Güemes, I.; Fernández-Tomé, B.; Enciso, S.; Sánchez-Margallo, F.M.; Portolés, M.T.; Arcos, D.; Vallet-Regí, M. Silicon substituted hydroxyapatite/VEGF scaffolds stimulate bone regeneration in osteoporotic sheep. Acta Biomaterialia 2020, 101, 544-553. [CrossRef]
- Aboudzadeh, N.; Dehghanian, C.; Shokrgozar, M.A. Effect of electrodeposition parameters and substrate on morphology of Si-HA coating. Surf. Coat. Technol. 2019, 375, 341-351. [CrossRef]
- Dehghanian, C.; Aboudzadeh, N.; Shokrgozar, M.A. Characterization of silicon-substituted nano hydroxyapatite coating on magnesium alloy for biomaterial application. Mater. Chem. Phys. 2018, 203, 27-33. [CrossRef]
- Graziani, G.; Boi, M.; Bianchi, M. A Review on Ionic Substitutions in Hydroxyapatite Thin Films: Towards Complete Biomimetism. Coatings 2018, 8, 269. [CrossRef]
- Mumith, A.; Cheong, V.S.; Fromme, P.; Coathup, M.J.; Blunn, G.W. The effect of strontium and silicon substituted hydroxyapatite electrochemical coatings on bone ingrowth and osseointegration of selective laser sintered porous metal implants. PLoS ONE 2020, 15 (1), e0227232. [CrossRef]
- Robinson, L.; Salma-Ancane, K.; Stipniece, L.; Meenan, B.J.; Boyd, A.R. The deposition of strontium and zinc Co-substituted hydroxyapatite coatings. J. Mater. Sci. – Mater. Med. 2017, 28 (3), 51. [CrossRef]
- Wolf-Brandstetter, C.; Beutner, R; Hess, R.; Bierbaum, S.; Wagner, K.; Scharnweber, D.; Gbureck, U.; Moseke, C. Multifunctional calcium phosphate based coatings on titanium implants with integrated trace elements. Biomed. Mater. 2020, 15, 025006. [CrossRef]
- Liu, S.j.; Li, H.j.; Zhang, L.l.; Feng, L.; Yao, P. Strontium and magnesium substituted dicalcium phosphate dehydrate coating for carbon/carbon composites prepared by pulsed electrodeposition. Appl. Surf. Sci. 2015, 359, 288-292. [CrossRef]
- Kolmas, J.; Groszyk, E.; Kwiatkowska-Rózycka, D. Substituted hydroxyapatites with antibacterial properties. BioMed Res. Int. 2014, 178123. [CrossRef]
- Garbo, C.; Locs, J.; D’este, M.; Demazeau, G.; Mocanu, A.; Roman, C.; Horovitz, O.; Tomoaia-Cotisel, M. Advanced Mg, Zn, Sr, Si multi-substituted hydroxyapatites for bone regeneration. Int. J. Nanomed. 2020, 15, 1037-1058. [CrossRef]
- Bracci, B.; Torricelli, P.; Panzavolta, S.; Boanini, E.; Giardino, R.; Bigi, A. Effect of Mg2+, Sr2+, and Mn2+ on the chemico-physical and in vitro biological properties of calcium phosphate biomimetic coatings. J. Inorg. Biochem. 2009, 103 (12), 1666-1674. [CrossRef]
- Furko, M.; Jiang, Y.; Wilkins, T.A.; Balázsi, C. Electrochemical and morphological investigation of silver and zinc modified calcium phosphate bioceramic coatings on metallic implant materials. Mater. Sci. Eng. C 2016, 62, 249-259. [CrossRef]
- Furko, M.; May, Z.; Havasi, V.; Kónya, Z.; Grünewald, A.; Detsch, R.; Boccaccini, A.R.; Balázsi, C. Pulse electrodeposition and characterization of non-continuous, multi-element-doped hydroxyapatite bioceramic coatings. J. Solid State Electrochem. 2018, 22, 555-566. [CrossRef]
- Furko, M.; Della Bella, E.; Fini, M.; Balázsi, C. Corrosion and biocompatibility examination of multi-element modified calcium phosphate bioceramic layers. Mater. Sci. Eng. C 2019, 95, 381-388. [CrossRef]
- Huang, Y.; Ding, Q.; Pang, X.; Han, S.; Yan, Y. Corrosion behavior and biocompatibility of strontium and fluorine co-doped electrodeposited hydroxyapatite coatings. Appl. Surf. Sci. 2013, 282, 456-462. [CrossRef]
- Bir, F.; Khireddine, H.; Mekhalif, Z.; Bonnamy, S. Pulsed electrodeposition of Ag+ doped prosthetic Fluorohydroxyapatite coatings on stainless steel substrates. Mater. Sci. Eng. C 2021, 118, 111325. [CrossRef]
- Vo, T.H.; Le, T.D.; Pham, T.N.; Nguyen, T.T.; Nguyen, T.P.; Dinh, T.M.T. Electrodeposition and characterization of hydroxyapatite coatings doped by Sr2+, Mg2+, Na+ and F− on 316L stainless steel. Adv. Nat. Sci. Nanosci. Nanotechnol. 2018, 9, 045001. [CrossRef]
- Chambard, M.; Remache, D.; Balcaen, Y.; Dalverny, O.; Alexis, J.; Siadous, R.; Bareille, R.; Catros, S.; Fort, P.; Grossin, D.; Gitzhofer, F.; Bertrand, G. Effect of silver and strontium incorporation route on hydroxyapatite coatings elaborated by rf-SPS. Materialia 2020, 12, 100809. [CrossRef]
| calcium phosphate | abbreviation | chemical formulae | (Ca/P)at. | solubility [-log Ks] | references |
|---|---|---|---|---|---|
| tetracalcium phosphate | TTCP | 2.00 | 38.0-44.0 | [55,56,57] | |
| hydroxyapatite | HAP | 1.67 | 116.8 | [58,59,60] | |
| α-tricalcium phosphate | α-TCP | 1.50 | 25.5 | [61,62,63] | |
| β-tricalcium phosphate | β-TCP | 1.50 | 28.9 | [64,65,66] | |
| calcium-deficient apatite | Ca-def apatite |
with 0 < x < 2 |
1.34-1.66 | 85.1 | [67,68,69] |
| octacalcium phosphate | OCP | 1.33 | 96.6 | [70,71,72] | |
| calcium pyrophosphate | CPP | 1.00 | 18.5 | [73,74,75] | |
| dicalcium phosphate anhydrous, aka monetite | DCPA | 1.00 | 6.9 | [76,77,78] | |
| dicalcium phosphate dihydrate, aka brushite | DCPD | 1.00 | 6.6 | [79,80,81] | |
| monocalcium phosphate anhydrous | MCPA | 0.50 | 1.1 | [82,83,84] | |
| monocalcium phosphate monohydrate | MCPM | 0.50 | 1.1 | [85,86,87] |
| Ions | Biological / Chemical effect | References |
| monovalent cations | ||
| antibacterial activity | [200,201,202] | |
| osteogenesis | [203,204,205] | |
| osteogenesis | [206,207,208] | |
| osteogenesis | [209,210,211] | |
| divalent cations | ||
| angiogenesis | [212,213,214] | |
| antibacterial activity | [215,216,217] | |
| osteogenesis | [218,219,220] | |
| osteogenesis | [221,222,223] | |
| osteogenesis | [224,225,226,227] | |
| osteogenesis/antibacterial/anti-inflammatory | [228,229,230] | |
| trivalent cations | ||
| anticancer/antibacterial | [231,232,233] | |
| antibacterial | [234,235,236,237] | |
| photoluminescence | [238,239,240] | |
| photoluminescence | [241,242,243] | |
| osteogenesis/anticancer/antibacterial | [244,245,246] | |
| anticancer/antibacterial | [247,248,249] | |
| photoluminescence | [250,251] | |
| anions | ||
| osteogenesis | [252,253,254] | |
| osteogenesis | [255,256,257] | |
| antibacterial | [258,259,260] | |
| / | anticancer/antibacterial | [261,262,263] |
| osteogenesis | [264,265,266] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





