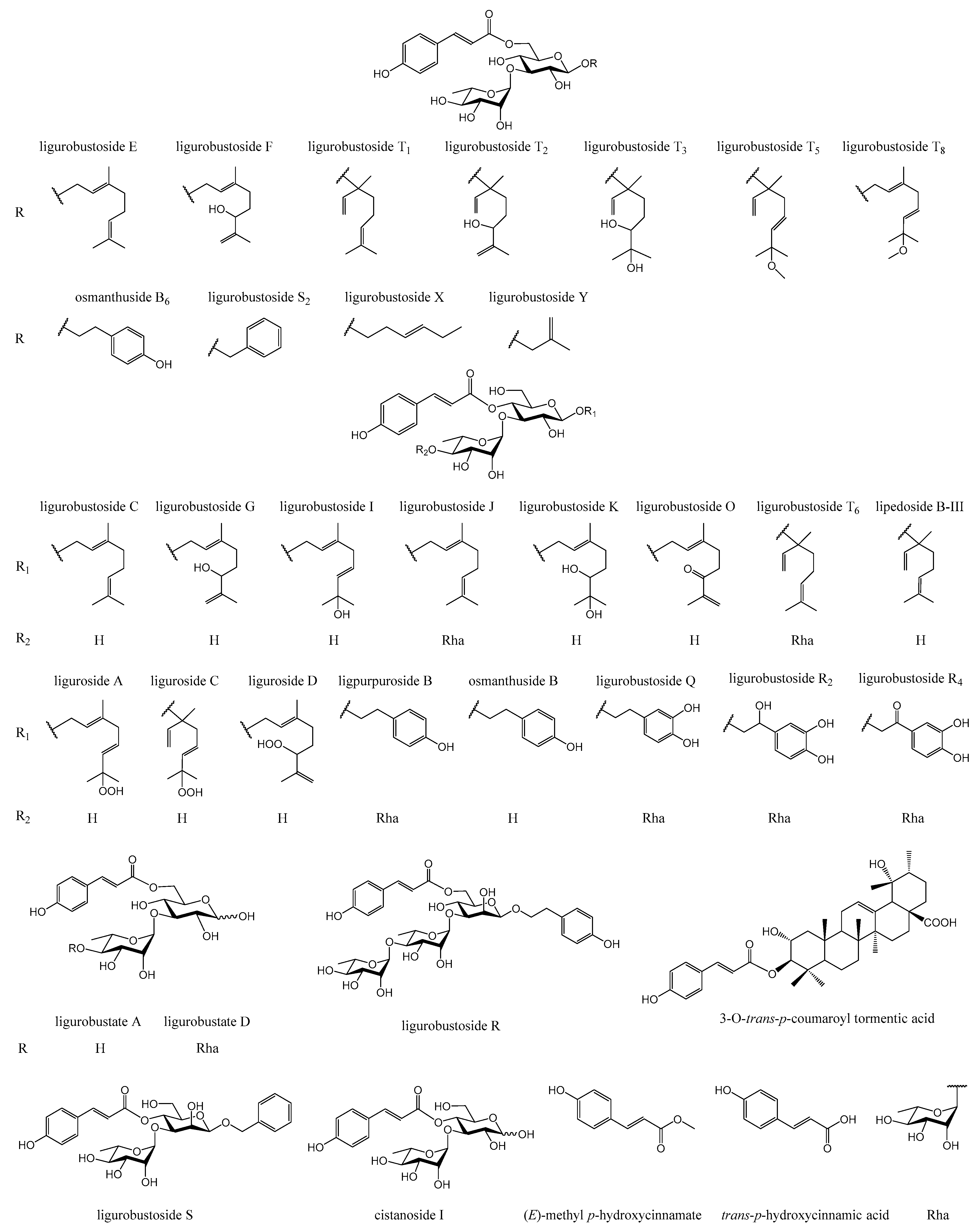
1. Introduction
Ligustrum robustum (Roxb.) Blume, a plant of Oleaceae, is distributed widely in Southwest China, Burma, Vietnam, India and Cambodia [
1]. The leaves of
L. robustum have been used as Ku-Ding-Cha, a functional tea to clear heat and remove toxins, in Southwest China for near 2000 years [
2,
3]. Additionally,
L. robustum was served as a folk medicine to deal with diabetes, obesity, hypertension, and so on [
3,
4].
In the previous phytochemical studies [
2,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17], about 90 chemical constituents, including
trans-
p-hydroxycinnamic acid, 34
trans-
p-hydroxycinnamic acid esters (
Figure 1), flavonoid glycosides, lignan glycosides, and other compositions, were isolated and identified from the leaves of
L. robustum. In our previous biological investigations [
14,
15,
16,
17], the chemical compositions isolated from the leaves of
L. robustum were tested and the results showed that: (1)
trans-
p-hydroxycinnamic acid displayed stronger
α-glucosidase inhibitory activity than the positive control acarbose; (2)
trans-
p-hydroxycinnamic acid and several
trans-
p-hydroxycinnamic acid esters displayed no weaker fatty acid synthase (FAS) inhibitory activities than the positive control orlistat; (3)
trans-
p-hydroxycinnamic acid and a lot of
trans-
p-hydroxycinnamic acid esters revealed stronger 2,2'-azino-bis(3-ethylbenzthiazoline-6-sulphonic acid) ammonium salt (ABTS) radical scavenging effects than the positive control L-(+)-ascorbic acid, and revealed moderate α-amylase inhibitory activities. What is more,
trans-
p-hydroxycinnamic acid esters might be hydrolyzed with catalysis of carboxylesterase or gastric acid in vivo [
18,
19], and release
trans-
p-hydroxycinnamic acid, meaning that
trans-
p-hydroxycinnamic acid esters are the prodrugs of
trans-
p-hydroxycinnamic acid. In addition, it was reported that natural products with inhibitory activities on α-glucosidase, α-amylase, and FAS as well as an antioxidant effect might be a new resource to prevent diabetes and its complications [
17]. Therefore,
trans-
p-hydroxycinnamic acid and its esters isolated from
L. robustum might be a novel resource for preventing diabetes and its complications.
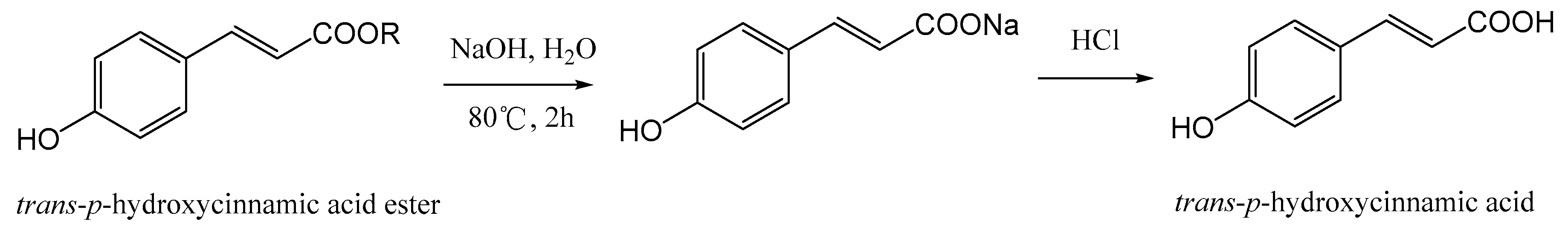
However, the quantitative analytical method for
trans-
p-hydroxycinnamic acid and its esters in the leaves of
L. robustum hasn’t been reported so far. HPLC is a common and accurate method to analyze simultaneously several compositions [
20], but it was difficult and troublesome to determine 34
trans-
p-hydroxycinnamic acid esters by usual HPLC. In addition, there might be other unknown
trans-
p-hydroxycinnamic acid esters in the leaves of
L. robustum. In the present study, thus, a new HPLC-UV method using hydrolyzation with sodium hydroxide for quantitation of
trans-
p-hydroxycinnamic acid and its esters in the leaves of
L.
robustum was developed and validated. Moreover, the proposed method was applied successfully to the determination and comparison of the contents of
trans-
p-hydroxycinnamic acid and its esters in various extracts of the leaves of
L.
robustum.
2. Results and Discussion
2.1. Method Development
The raw powder of the dried leaves of
L.
robustum was extracted with 70% (
v/
v) ethanol (25 mL/g) under reflux for 1 h. The conditions of hydrolyzation of
trans-
p-hydroxycinnamic acid esters in the extracting solution of
L.
robustum were optimized by changing various parameters, i.e., catalyst of hydrolyzation (hydrochloride or sodium hydroxide), incubation temperature (30-90 °C), and period of incubation (1-6 h). As a result (
Supplementary Table S1), the optimal hydrolyzation conditions were as follows: 1 mL sodium hydroxide aqueous solution (1 M) was added into 1 mL the extracting solution of
L.
robustum and incubated at 80 °C for 2 h , and then 1 mL hydrochloride (1 M) solution was added (
Figure 2).
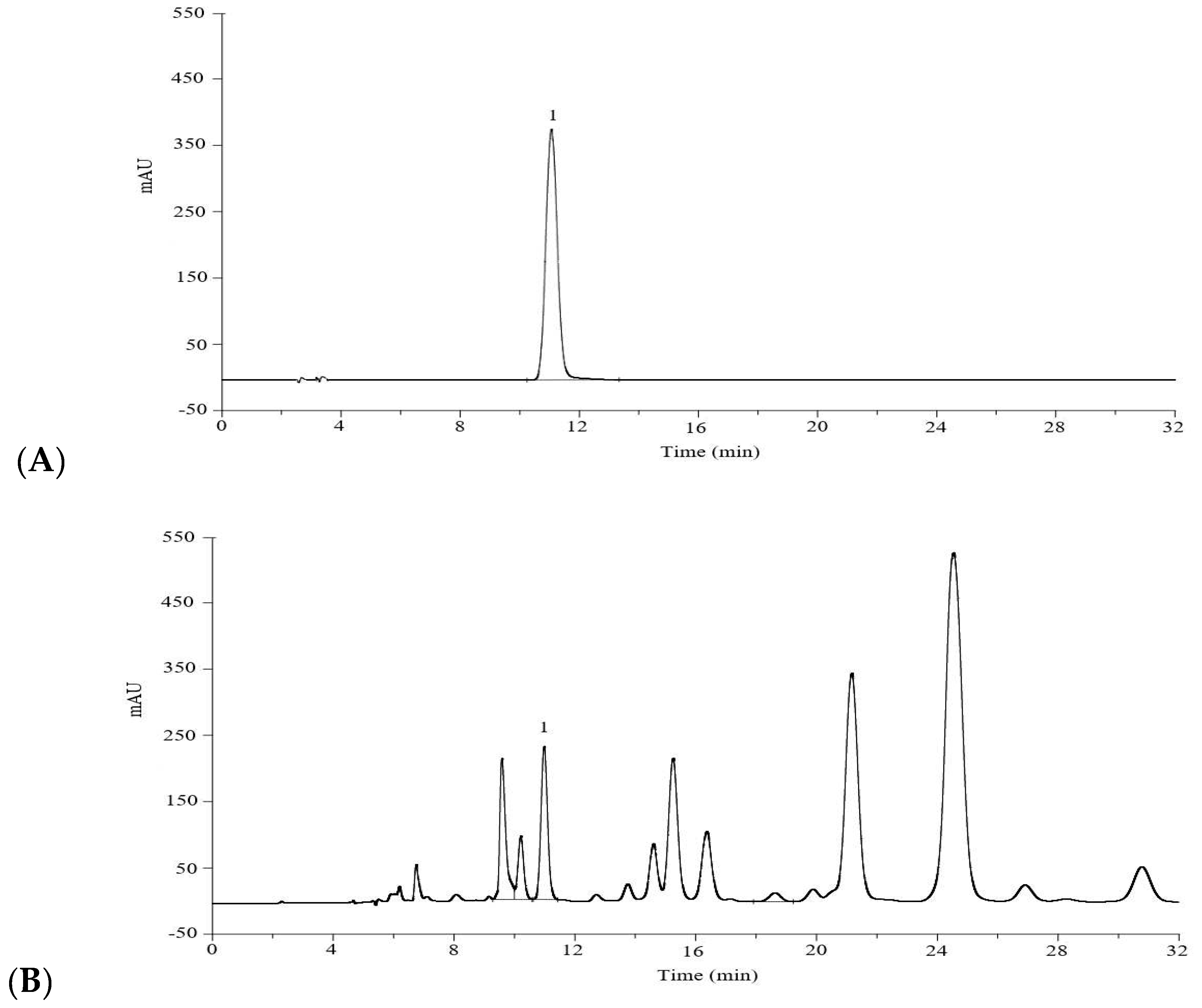
In order to quantify the free
trans-
p-hydroxycinnamic acid in the original extracting solution and the potential
trans-
p-hydroxycinnamic acid in the hydrolyzed extracting solution, several chromatographic items were considered. The first factor was the stationary phase, where C-18 column gave ideal effect if compared to C-8 column. The second factor was the mobile phase. Methanol, acetonitrile and ultrapure water in different volumes were tried, and methanol-water (40:60,
v/
v), without glacial acetic acid, showed acceptable resolution value but with tailed peak. Consequently, glacial acetic acid was added to improve the resolution and peak shape. Furthermore, the UV detector was successfully applied for the detection of the compositions, and the wavelength of 310 nm gave the maximum sensitivity at 30 °C (
Supplementary Figure S1). Generally, the optimal HPLC performance (
Figure 3) was observed when using C-18 column (4.6 mm × 250 mm, 5 μm), eluting with methanol-0.1% acetic acid aqueous solution (40:60,
v/
v) at a flow rate of 1.0 mL/min, and detecting at 310 nm.
The total concentration of
trans-
p-hydroxycinnamic esters (C
t, µmol/mL) in the extracting solution of
L. robustum was calculated by follow equation:
Cp (µmol/mL): The concentration of potential trans-p-hydroxycinnamic acid (molecular weight: 164.16) in the hydrolyzed extracting solution of L. robustum. It was calculated as Cp (µg/mL) / 164.16.
Cf (µmol/mL): The concentration of free trans-p-hydroxycinnamic acid in the original extracting solution of L. robustum. It was calculated as Cf (µg/mL) / 164.16.
Additionally, osmanthuside B (molecular weight: 592.59) was considered as a representation of trans-p-hydroxycinnamic acid esters because of its moderate molecular weight and high content in the leaves of L. robustum. Consequently, Ct (µg/mL) might be calculated approximatively as Ct (µmol/mL) × 592.59.
2.2. Method Validation
2.2.1. Linearity and Calibration Curve
The calibration curve for
trans-
p-hydroxycinnamic acid was obtained by external standard method, using six concentrations of the standard, with three injections per concentration (
Supplementary Table S2). The chromatogram peak areas were plotted against the corresponding concentrations of the standard solutions to establish the calibration curve (
Supplementary Figure S2), and linear regression equation was calculated by the least squares method. This HPLC method showed linear regression at concentrations from 11.0 to 352.0 μg/mL, and the correlation coefficient (
r2) was 1.000 (
Table 1), indicating excellent linearity.
2.2.2. Limit of Detection and Limit of Quantification
The standard deviation of the Y-intercept in the regression equation was used as the residual standard deviation [
21]. Consequently, the limit of detection (LOD) and limit of quantification (LOQ) were 1.55 and 4.69 μg/mL, respectively (
Table 1).
2.2.3. Precision
In order to validate the precision of this HPLC-UV method, the
trans-
p-hydroxycinnamic acid standard was determined at three different concentrations, and the relative standard deviations (RSD) were calculated from the results of repeated measurements for each concentration. The measurement was performed three times in the same day to obtain the intra-day variability, and carried out three times in three different days to give the inter-day variability. The RSDs of intra-day and inter-day variabilities for
trans-
p-hydroxycinnamic acid were less than 2% (
Table 2), indicating the precision of this method was accord with the criterion recommended by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines [
21].
2.2.4. Accuracy
To evaluate the accuracy of this analytical method, a recovery experiment was performed. The raw powder of the dried leaves of
L.
robustum, which was spiked with 100% or 50% of its native content of free
trans-
p-hydroxycinnamic acid, was extracted with 70% (
v/
v) ethanol under reflux. The concentration of free
trans-
p-hydroxycinnamic acid in the original extract was analyzed and calculated by the regression equation showed in
Table 1, and the percentage recovery was calculated by repeated measurements of the analyte. As showed in
Table 3, the percentage recovery of
trans-
p-hydroxycinnamic acid was 101.2% ± 0.8%, demonstrating an acceptable accuracy of this method since the acceptance criteria of the percentage recovery were 90%-107% [
22].
2.2.5. System Suitability Parameters
The resolution of trans-p-hydroxycinnamic acid from other compositions was more than 1.5. The column efficiency for trans-p-hydroxycinnamic acid was more than 15000 N/m.
2.3. Quantification of Trans-p-hydroxycinnamic Acid and Its Esters in Various Extracts of L. robustum.
The dried leaves (10.00 g) of
L. robustum were extracted with 90 mL ethanol aqueous solution (30%, 40%, 50%, 60% ,70%, 80%,
v/
v) under reflux for 60 min, and the contents of
trans-
p-hydroxycinnamic acid and its esters in the extracts were determined by the above developed and validated method. The results are presented in
Table 4. The 60%-70% ethanol extracts of
L. robustum showed the highest contents of free
trans-
p-hydroxycinnamic acid (3.96-3.99 mg/g), while the 30% ethanol extract of
L. robustum showed the lowest content of free
trans-
p-hydroxycinnamic acid (2.26 mg/g) . In addition, the 50%-80% ethanol extracts of
L. robustum displayed the highest contents of
trans-
p-hydroxycinnamic acid esters (202.6-210.6 mg/g), while the 30% ethanol extract of
L. robustum displayed the lowest content of
trans-
p-hydroxycinnamic acid esters (125.8 mg/g). Taken together, 60%-70% ethanol was the optimal extraction solvent for
trans-
p-hydroxycinnamic acid and its esters.
3. Materials and Methods
3.1. Chemicals and Reagents
Methanol (HPLC) was afforded by Saimo Fisher Scientific Co., Ltd. (Shanghai, China). Ethanol (AR) was purchased from Chengdu Kelong Chemical Co., Ltd. (Chengdu, Sichuan, China). Glacial acetic acid, sodium hydroxide, and hydrochloride (AR) were acquired from Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). The standard of
trans-
p-hydroxycinnamic acid (>98%purity) was isolated and identified in our laboratory from the leaves of
L. robustum, as previously described [
14]. Ultrapure water was obtained from an ultra-pure water purifier system (Chengdu Yuechun Scientific Co., Ltd., Chengdu, Sichuan, China).
3.2. Plant Material
The dried leaves of L. robustum (Ku-Ding-Cha) were purchased from Junlian Qing-Shan-Lu-Shui Tea Co., Ltd. (Yibin, Sichuan, China). The material was crushed by hand before extraction.
3.3. Hydrolyzation of Extracting Solution and Preparation of Solutions
Standard solution of trans-p-hydroxycinnamic acid: the stock solution of trans-p-hydroxycinnamic acid was diluted with 40% (v/v) methanol to obtain 6 standard solutions at 11.0, 22.0, 44.0, 88.0, 176.0, 352.0 μg/mL, and filtered through a 0.45 μm PTFE syringe filter (Millipore, Billerica, MA, USA) before HPLC analysis.
Test solution of original extract: the original extracting solution of L. robustum was diluted with 40% (v/v) methanol and percolated using a 0.45 μm PTFE syringe filter prior to HPLC measurement.
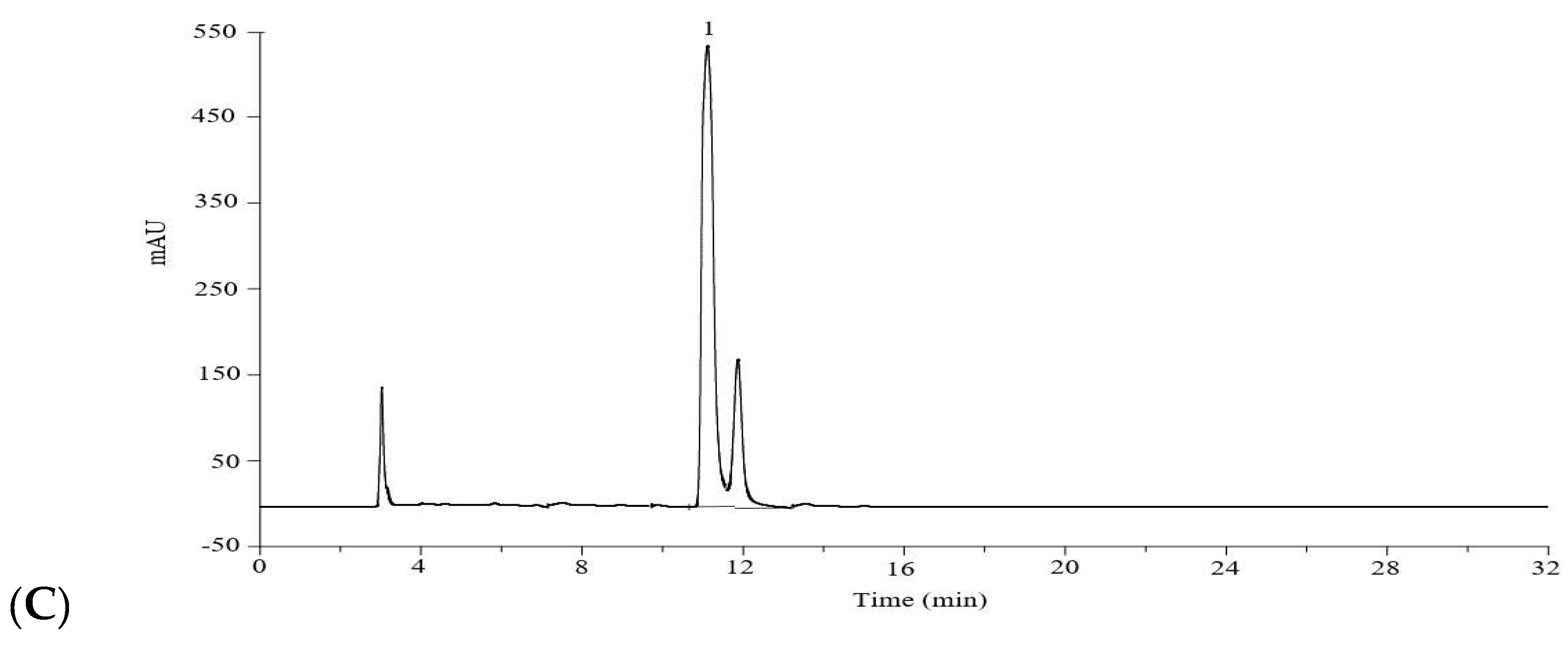
Test solution of hydrolyzed extract: 1 mL sodium hydroxide aqueous solution (1 M) and 1 mL the extracting solution of L. robustum were mixed and incubated at 80 °C for 2 h, then 1 mL hydrochloride (1 M) solution was added. After cooled down to room temperature, the above mixture solution was transferred to 10 mL volumetric flask, and the volume was filled with 40% (v/v) methanol. The diluted solution was filtered with a 0.45 μm PTFE syringe filter prior to HPLC determination.
3.4. HPLC Determination of Trans-p-hydroxycinnamic Acid and Its Esters
HPLC analysis was performed on a LC-20AT HPLC system (Shimadzu Corporation, Kyoto, Japan) with a SPD-20A UV-VIS detector and a binary pump. The standard solution or test solution (20 μL) was injected onto a Phenomenex Luna C18(2) 100A column (4.6 mm × 250 mm, 5 μm) thermostated at 30 °C. The components were eluted with methanol-0.1% acetic acid aqueous solution (40:60, v/v) at a flow rate of 1.0 mL/min in isocratic mode. The signals at 310 nm were monitored. The results were processed in LabSolutions Analysis Station (Shimadzu Corporation, Kyoto, Japan).
3.5. HPLC Validation
The analytical method was validated for linearity, the limit of detection (LOD), the limit of quantification (LOQ), precision and accuracy according to the relevant ICH guidelines [
21].
3.5.1. Linearity
The standard solutions of trans-p-hydroxycinnamic acid at different concentrations (11.0-352.0 μg/mL) were introduced to the HPLC system in triplicate. The calibration curve of trans-p-hydroxycinnamic acid was drawn by plotting the peak areas against the corresponding concentrations. The correlation coefficient (r2) of the regression equation was obtained to validate the linearity.
3.5.2. Limit of Detection and Limit of Quantification
The LOD value was calculated as 3.3σ/S, while the LOQ value was calculated as 10σ/S, in which σ was the residual standard deviation of the regression equation, and S was the calibration curve slope.
3.5.3. Precision
In order to validate the precision of the analytical method, the standard solutions of trans-p-hydroxycinnamic acid at three different concentrations (44.0, 88.0, 176.0 μg/mL) were used to evaluate the intra-day and inter-day variabilities. The standard solutions were analyzed in triplicate in the same day to obtain the intra-day variability, while measured three times in three different days to give the inter-day variability.
3.5.4. Accuracy
In order to validate the accuracy of the analytical method, the recovery of trans-p-hydroxycinnamic acid was determined by the standard addition method. Trans-p-hydroxycinnamic acid standard (4.0 or 2.0 mg) was added into the dried leaves of L. robustum (1.000 g) which contained natively 4.1 mg/g free trans-p-hydroxycinnamic acid, and then extracted with 70% (v/v) ethanol (25 mL/g) under reflux for 1 h. The free trans-p-hydroxycinnamic acid in the original extract was analyzed by the HPLC method. And the percentage recovery of trans-p-hydroxycinnamic acid was obtained from the results.
3.6. Statistical analyses
Statistical analyses were carried out on GraphPad Prism 5.01. Every sample was determined in triplicate. The results are recorded as mean ± standard deviation (SD). Difference of means between groups was analysed by one-way analysis of variance (ANOVA) on statistical package SPSS 25.0. The difference between groups was believed to be significant when P < 0.05.
4. Conclusions
In the present study, a new optimized HPLC-UV method for the quantification of trans-p-hydroxycinnamic acid and its esters in the leaves of L. robustum was developed and validated in accordance with ICH guidelines. Because it was difficult and troublesome to analyze no less than 34 trans-p-hydroxycinnamic acid esters by usual HPLC, these esters were hydrolyzed with sodium hydroxide, and then the potential trans-p-hydroxycinnamic acid was determined by HPLC-UV. The above analytical method was simple and rapid, which simplified greatly the analytical process. Additionally, the methodology validation, including linearity, LOD, LOQ, precision, and accuracy, showed that the new HPLC-UV method was acceptable. To the best of our knowledge, it is the first HPLC method using hydrolyzation for quantification of many carboxylic esters. Finally, the novel method was used successfully to measure the contents of trans-p-hydroxycinnamic acid and its esters in various extracts of the leaves of L. robustum. The method can be applied also to the quality control of the products of L. robustum. Nevertheless, the shortcoming of the method is that the total content of the trans-p-hydroxycinnamic acid esters is clear but the specific content of every ester is not clear.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on
Preprints.org, Table S1: The results of hydrolyzation of
trans-
p-hydroxycinnamic acid esters in the extract of
L. robustum; Table S2: The concentrations and peak areas of
trans-
p-hydroxycinnamic acid standard; Figure S1: UV spectrum of
trans-
p-hydroxycinnamic acid; Figure S2: Calibration curve for
trans-
p-hydroxycinnamic acid.
Author Contributions
Conceptualization, S.-H.L.; methodology, S.-H.L. and X.-N.L.; formal analysis, S.-H.L. and X.-N.L.; investigation, X.-N.L., X.-J.N., R.C., X.-X.L., and S.-H.L.; data curation, S.-H.L.; writing—original draft preparation, S.-H.L. and X.-N.L.; writing review and editing, X.-X.L.; supervision, S.-H.L.; funding acquisition, S.-H.L. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded by the Guangxi Natural Science Foundation Project (grant number: 2020GXNSFAA297129), Guangxi Science and Technology Base and Talents Special Project (grant number: Guike AD21075006), and Youjiang Medical University for Nationalities Science Research Project (grant number: yy2021sk004).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Acknowledgments
The authors are obliged to Ying Li, Jin-Ling Nong, and Yan-Fang Su, College of Pharmacy, Youjiang Medical University for Nationalities, for some help in the work.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Lau, K.M.; He, Z.D.; Dong, H.; Fung, K.P.; But, P.P.-H. Anti-oxidative, anti-inflammatory and hepato-protective effects of Ligustrum robustum. J. Ethnopharmacol. 2002, 83, 63–71. [Google Scholar] [CrossRef]
- He, Z.D.; Lau, K.M.; But, P.P.-H.; Jiang, R.W.; Dong, H.; Ma, S.C.; Fung, K.P.; Ye, W.C.; Sun, H.D. Antioxidative glycosides from the leaves of Ligustrum robustum. J. Nat. Prod. 2003, 66, 851–854. [Google Scholar] [CrossRef]
- Zhu, F.; Cai, Y.Z.; Sun, M.; Ke, J.X.; Lu, D.Y.; Corke, H. Comparison of major phenolic constituents and in vitro antioxidant activity of diverse kudingcha genotypes from Ilex kudingcha, Ilex cornuta, and Ligustrum robustum. J. Agric. Food Chem. 2009, 57, 6082–6089. [Google Scholar] [CrossRef]
- Yang, R.M.; Liu, F.; He, Z.D.; Ji, M.; Chu, X.X.; Kang, Z.Y.; Cai, D.Y.; Gao, N.N. Anti-obesity effect of total phenylpropanoid glycosides from Ligustrum robustum Blume in fatty diet-fed mice via up-regulating leptin. J. Ethnopharmacol. 2015, 169, 459–465. [Google Scholar] [CrossRef]
- Li, L.; Peng, Y.; Xu, L.J.; Wu-Lan, T.N.; Shi, R.B.; Xiao, P.G. Chemical constituents from Ligustrum robustum Bl. Biochem.l Syst. Ecol. 2010, 38, 398–401. [Google Scholar] [CrossRef]
- Li, L.; Peng, Y.; Liu, Y.; Xu, L.J.; Guo, N.; Shi, R.B.; Xiao, P.G. Two new phenethanol glycosides from Ligustrum robustum. Chinese Chem. Lett. 2011, 22, 326–329. [Google Scholar] [CrossRef]
- Tian, J.; Zhang, H.J.; Sun, H.D.; Pan, L.T.; Yao, P.; Chen, D.Y. Monoterpenoid glycosides from Ligustrum robustum. Phytochemistry 1998, 48, 1013–1018. [Google Scholar] [CrossRef]
- Tian, J.; Sun, H.D. New monoterpenoid glycosides from Ligustrum robustum. Chin. J. Appl. Envir. Biol. 1999, 5, 501–506. [Google Scholar]
- Yu, Z.L.; Zeng, W.C. Antioxidant, antibrowning, and cytoprotective activities of Ligustrum robustum (Roxb.) Blume extract. J. Food Sci. 2013, 78, 1354–1362. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.L.; Gao, H.X.; Zhang, Z.; He, Z.; He, Q.; Jia, L.R.; Zeng, W.C. Inhibitory effects of Ligustrum robustum (Roxb.) Blume extract on α-amylase and α-glucosidase. J. Funct. Foods 2015, 19, 204–213. [Google Scholar] [CrossRef]
- Ito, H.; Otsuki, A.; Mori, H.; Li, P.; Kinoshita, M.; Kawakami, Y.; Tsuji, H.; Fang, D.Z.; Takahashi, Y. Two new monoterpene glycosides from Qing Shan Lu Shui tea with inhibitory effects on leukocyte-type 12-lipoxygenase activity. Molecules 2013, 18, 4257–4266. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, Y.; Otsuki, A.; Mori, Y.; Kanzaki, K.; Suzuki-Yamamoto, T.; Fang, D.Z.; Ito, H.; Takahashi, Y. Involvement of the hydroperoxy group in the irreversible inhibition of leukocyte-type 12-lipoxygenase by monoterpene glycosides contained in the Qing Shan Lu Shui tea. Molecules 2019, 24, 304. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Zhang, Z.-W.; Li, C.-C.; Yang, R.-M.; Pan, R.-L. Chemical constituents of alcoholic extract from Ligustrum robustum (Roxb.) Blume. Mod. Chin. Med. 2018, 20, 540–544. [Google Scholar]
- Lu, S.-H.; Zuo, H.-J.; Shi, J.-X.; Li, C.-R.; Li, Y.-H.; Wang, X.; Li, L.-R.; Huang, J. Two new glycosides from the leaves of Ligustrum robustum and their antioxidant activities and inhibitory effects on α-glucosidase and α-amylase. S. Afr. J. Bot. 2019, 125, 521–526. [Google Scholar] [CrossRef]
- Lu, S.-H.; Huang, J.; Zuo, H.-J.; Zhou, Z.-B.; Yang, C.-Y.; Huang, Z.-L. Monoterpenoid glycosides from the leaves of Ligustrum robustum and their bioactivities. Molecules 2022, 27, 3709. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.-H.; Zuo, H.-J.; Huang, J.; Chen, R.; Pan, J.-P.; Li, X.-X. Phenylethanoid and phenylmethanoid glycosides from the leaves of Ligustrum robustum and their bioactivities. Molecules 2022, 27, 7390. [Google Scholar] [CrossRef]
- Lu, S.-H.; Zuo, H.-J.; Huang, J.; Li, W.-N.; Huang, J.-L.; Li, X.-X. Chemical constituents from the leaves of Ligustrum robustum and their bioactivities. Molecules 2023, 28, 362. [Google Scholar] [CrossRef]
- You, Q.-D. Medicinal chemistry, 8th ed.; People’s Medical Publishing House: Beijing, China, 2016; Drug metabolism; pp. 46–62. ISBN 978-7-117-22151-1. [Google Scholar]
- Zhang, Y.-T.; Sun, Y.-M.; Zhang, J.-H.; Huo, Y.; Li, X.; Li, W.-B.; Zhao, A.-P.; Wang, R. Interaction between gut microbiota and drugs. Chin. Pharm. Bull. 2020, 36, 1650–1655. [Google Scholar]
- Orčić, D.; Berežni, S.; Mimica-Dukić, N. Quantitative HPLC-UV study of lignans in Anthriscus sylvestris. Molecules 2022, 27, 6072. [Google Scholar] [CrossRef]
- Kim, T.H.; Shin, H.Y.; Park, S.Y.; Kim, H.; Chung, D.K. Development and validation of a method for determining the quercetin-3-O-glucuronide and ellagic acid content of common evening primrose (Oenothera biennis) by HPLC-UVD. Molecules 2021, 26, 267. [Google Scholar] [CrossRef]
- Pencheva, D.; Teneva, D.; Denev, P. Validation of HPLC method for analysis of gamma-aminobutyric and glutamic acids in plant foods and medicinal plants. Molecules 2023, 28, 84. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).