1. Introduction
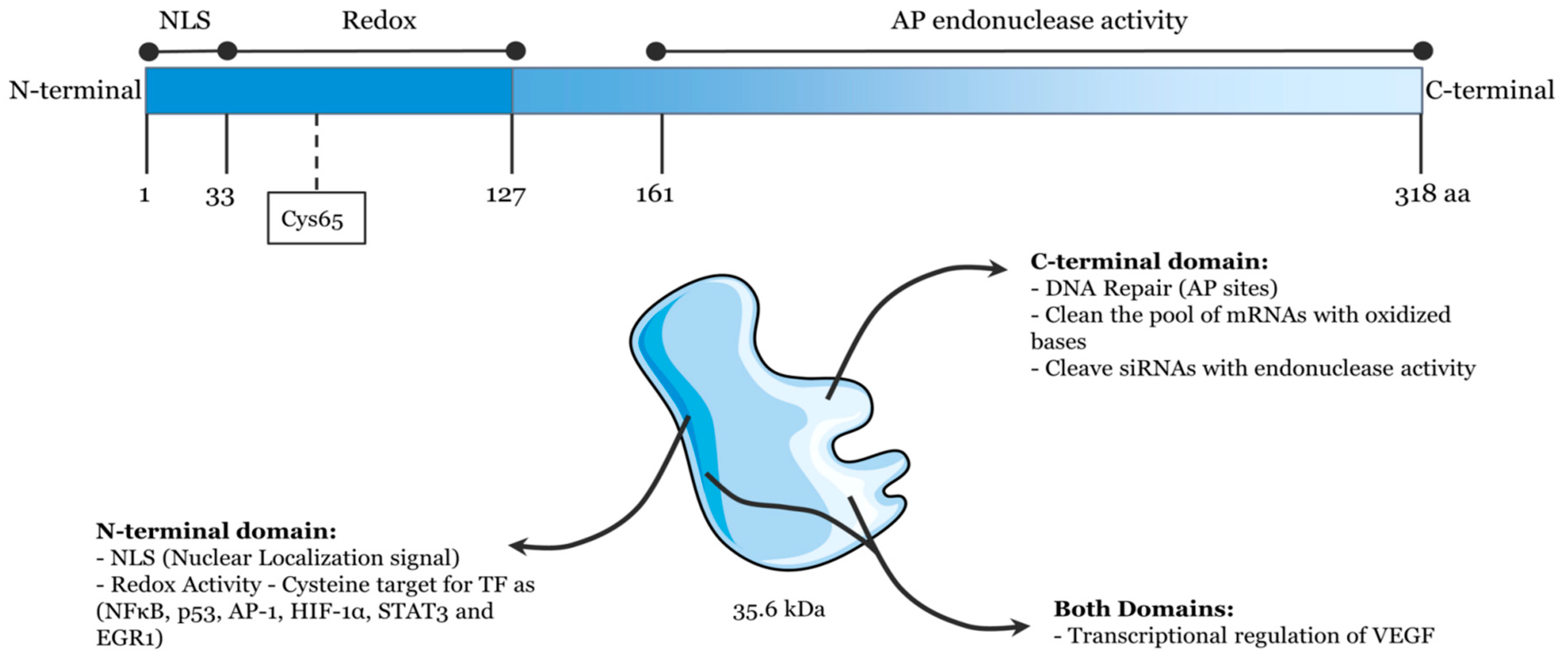
Human APE1, which displays homology to Escherichia coli exonuclease III, consists of a globular domain with 318 amino acid residues [
1,
2,
3]. It is discovered to be a 35.6 kDa cellular multifunctional protein which contains a redox function and an AP endonuclease activity, and both functions are independent in their actions (
Figure 1) [
4,
5,
6,
7]. The redox function of APE1, which is in its N-terminal domain, plays an important role in regulating the expression of genes to activate some transcription factors, such as p53, Egr-1, NF-kB [
8,
9]. According to researches, APE1 redox function is linked to the occurrence of various cancers, such as cutaneous squamous cell, pancreatic cancer, and esophageal adenocarcinoma [
10,
11,
12,
13]. Inhibiting APE1 redox function can decrease cell proliferation, prevent angiogenesis, and inhibit endothelial precursor cell differentiation [
14,
15,
16]. On the other hand, as a central enzyme in BER pathway, APE1 contains AP endonuclease activity in its C-terminal domain [
17,
18,
19,
20]. After APE1 recognizes and cleaves the AP site, it results in a single strand break that may be processed further by the coordinated actions of DNA polymerase, deoxyribose phosphatase, and DNA ligase [
21,
22,
23,
24]. APE1 is mainly present in the nucleus and mitochondria. In addition, it can also be found in the cytoplasm or secreted from cells. The state of disease dynamically regulates APE1 localization [
7,
25,
26].
So far, APE1 has been found to be vital in modern life science, and it is engaged in a variety of biological functions such as apoptosis and cell cycle regulation [
20,
27]. Misfunctioning or disturbed expression of APE1 can lead to a series of diseases [
28], such as immunodeficiency [
29], cancer [
30,
31], cellular senescence [
32]. Therefore, monitoring APE1 activity is extremely desired in order to acquire a complete understanding of the repair process of damaged DNA and make clinical diagnosis.
DNA nanotechnology has become a potent tool for combining therapeutic and imaging in DNA nanostructures [
33]. Our group designed a series of DNA nanostructures for biomarkers detection and cancer therapy, such as: (1) DNA tetrahedron-based nanocarrier with ATP aptamer for ATP detection [
34]; (2) DNA nanotriangle linked with split aptamer probes and loaded doxorubicin (dox) as a platform for diagnosis and treatment [
35]; (3) Y-shaped DNA nanostructure with split i-motif fragments and loaded dox for enhancing cancer imaging and therapy [
36]; (4) three-arm aptamer nanoclaw for enhancing imaging and tumor growth inhibition [
37]; and (5) a hand-in-hand DNA tile assembly loaded with TK1 mRNA and dox for imaging of TK1 mRNA and chemotherapy. In addition, Willner’s group proposed that the formation of self-assembled nanostructures provides a basis for the formation of signal amplifying sensor systems as well as for the bottom-up construction of functional scaffolds [
38]. In fact, DNA nanotechnology also has a good application in the imaging and regulation of APE1.
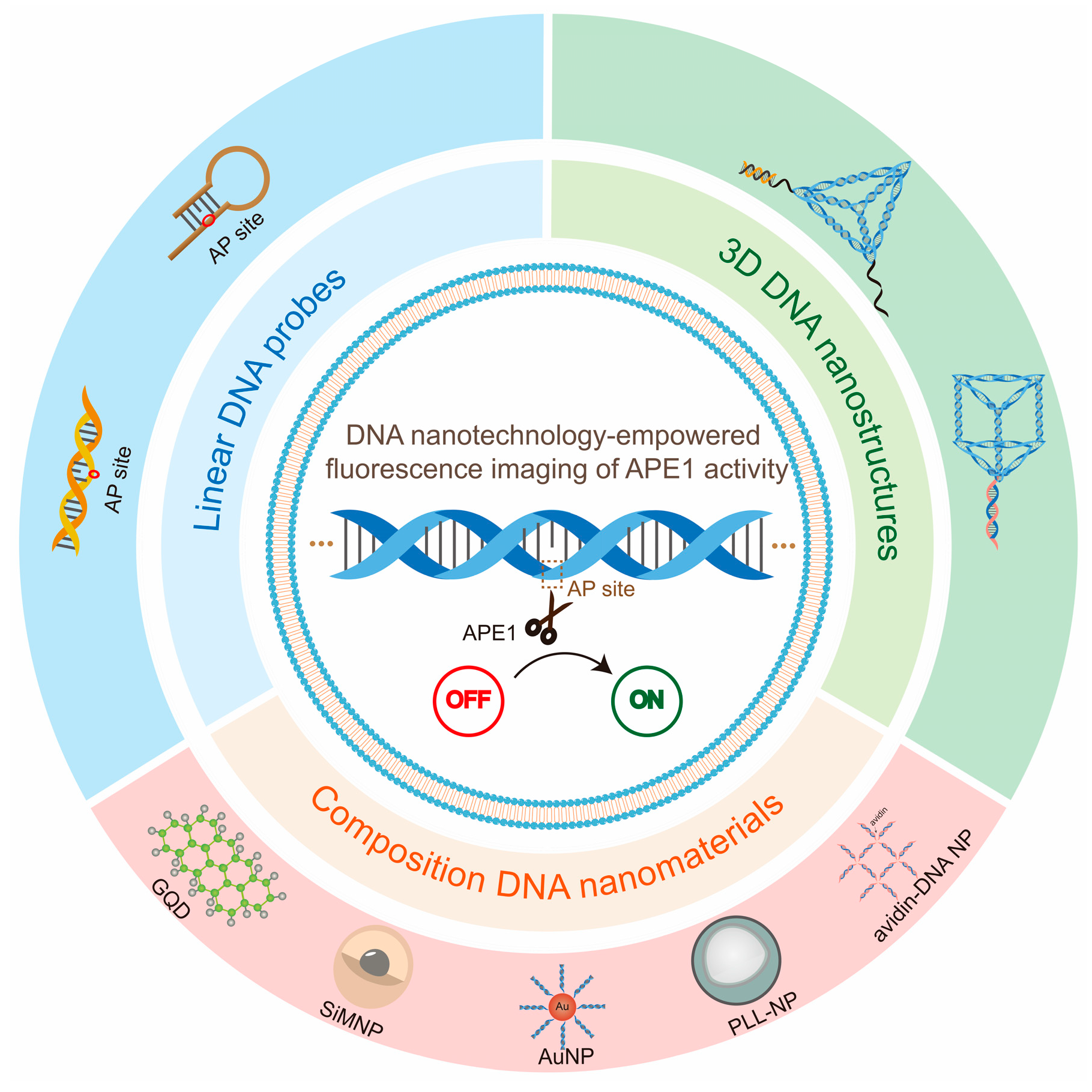
In this review, we will specifically summarize DNA nanotechnology-based fluorescence imaging of APE1 activity (
Figure 2). First, we briefly describe the origin of AP sites and mechanism of APE1 cleavage of the AP site. Then, the most recent achievements of DNA nanotechnology as an excellent tool for APE1 activity fluorescence imaging are described by typical examples. Finally, we summarize the driving effect of DNA nanotechnology-empowered fluorescence imaging of APE1 activity and the future development directions and opportunities. We hope this review will provide new insights into the design and development of new strategies for APE1 activity imaging, acquiring a complete understanding of the repair process of damaged DNA and making clinical diagnosis.
2. Origin of AP Sites and Mechanism of APE1 Cleavage of the AP Site
As one of common lesions in genomic DNA, AP sites can block DNA replication and transcription [
39]. The term "AP sites" refers to DNA damage products that are created when a nucleobase is lost but the deoxyribose is retained, along with the conserved 3'- and 5'-phosphodiester linkages [
40]. As shown in
Figure 3a, there are two main sources of AP sites: (1) spontaneous loss of base; and (2) removal by DNA N-glycosylase [
41,
42]. As the major repair enzyme of the BER pathway, APE1 recognizes the AP site and cleaves it on the 5' side, producing a single strand break with a 3'-hydroxyl end and a 5'-deoxyribose phosphate end (shown in
Figure 3b) [
43,
44]. Studies have shown that increased APE1 levels lead to migration, drug resistance of tumor cells and reduced patient survival. Differences in APE1 expression patterns between normal and cancer cells or inflammatory cells could serve as endogenous triggers. In DNA nanotechnology, the majority of APE1-responsive DNA systems rely on the design of AP site that can be specifically recognized and bound by APE1. With this concept, a variety of DNA nanostructures containing AP sites have been rationally designed to be substrates for imaging of APE1 activity.
3. DNA Nanotechnology-Empowered Fluorescence Imaging of APE1 Activity
3.1. Linear DNA-Empowered Fluorescence Imaging of APE1 Activity
The basic DNA-based probes for intracellular biomarkers detection are linear DNA structures [
45]. As it is simple to design and produce, linear DNA is frequently used as the fundamental unit for arranging different nanomaterials. According to their structural characteristics, linear DNA-based probes used for APE1 imaging can be classified as hairpin type and double-stranded linear type [
46].
3.1.1. Hairpin DNA-Based Nucleic Acid Probes
Hairpin DNA is formed by single-stranded DNA (ssDNA) molecules which bend back to bring complementary base pairs close to each other [
47,
48]. It is widely used in biosensing assays, such as the detection of proteins [
49,
50], metal ions [
51], small molecules [
52,
53,
54] and RNA [
55,
56]. Hairpin DNA probes used for APE1 imaging are typically designed with an AP site at the stem and tagged with fluorophores and quenched groups on both sides of the AP site. APE1 has the ability to cut the 5' end of the AP site, causing the short strand to separate from the rest of the probe and making fluorescence recovery. Furthermore, APE1 can cleave the AP site to form a trigger sequence and combine with DNA signal amplification technology, such as Hybridization Chain Reaction (HCR), Catalytic Hairpin Assembly (CHA), Rolling Circle Amplification (RCA) to achieve signal amplification detection of APE1 activity.
Signal reporters using hairpin DNA, based on Föster resonance energy transfer (FRET), have shown robustness for imaging of APE1 activity [
57,
58,
59]. Lu et al. designed a hairpin DNA with an AP site on the toehold section and a blunt end [
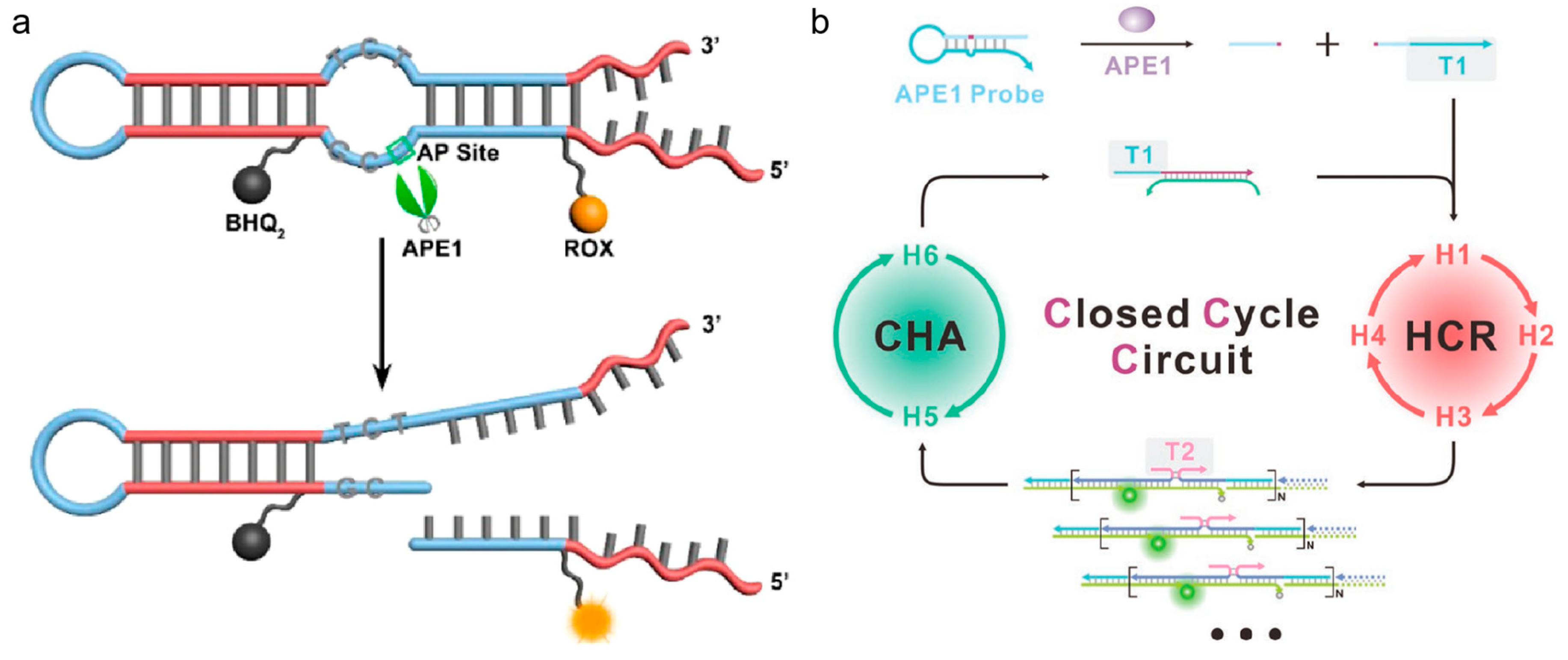
60]. The presence of APE1 could expose the toehold part of the hairpin by cleaving the AP site, then miRNA-21 triggered the CHA, causing fluorescence recovery. With ease of use and low cost, this fluorescent biosensor would support for rapid diagnosis of APE1. To avoid severe interference by other nonspecific endonucleases and distinguish APE1 from similar nucleases, Fang et al. designed a fluorescent probe with a double-loop (shown in
Figure 4a) [
61]. As this structure contained an AP site with a 3’ internal-loop, which enabled effective differentiation between APE1 and nonspecific endonuclease, it was a practical tool for measuring APE1 activity in several biological samples with high throughput. Besides, DNA signal amplification techniques are one of the most effective approaches to improve detection effect, and they were in combination with hairpin DNA for APE1 imaging. The cascade integration of different amplification modes can improve amplification depth and strengthen anti-interference capacity. As shown in
Figure 4b, Chai et al. employed a probe contained AP site and trigger T1, which was complementary to the 5’-toehold of H1 [
62]. APE1 could cleave the AP site and release T1. Then, the T1 activated the cascade cycle of HCR and CHA to provide the amplified signal for quantitative analysis.
However, problems such as degradation of nucleases in cells and non-specific separation of fluorescent groups from quenched groups often result in false positive signals. In comparison to the FRET strategy, the label-free strategy preserves many benefits: (1) great specificity; (2) low time consumption, and (3) stability of the sample activity for a significantly longer period of time. Huang and co-workers used a hairpin-structured probe to detect the activity of APE1 sensitively [
63]. After incised by APE1, with the help of Klenow fragment (KF) polymerase, the products could initiate polymerization. Then, it might make the hairpin structure open, resulting in double-stranded DNA (dsDNA) with a Nt.BbvCI endonuclease recognition site. By means of polymerase-catalyzed strand displacement and binding with N-methylmesoporphyrin IX (NMM), a G-rich segment was released to produce a powerful fluorescence emission. In a subsequent work, Liu et al. reported a hairpin-structured probe (HP) that contained an AP site and a circular DNA probe (CDT) as the template of RCA [
64]. The primer sequence was released by APE1. Then, with the assistance of DNA polymerase and dNTPs, it quickly initiated RCA to produce a long chain DNA. As the production contained a number of repetitive G-quadruplex sequence that could combine with Thioflavin T (ThT), it generated strong fluorescent signals. This experiment successfully developed a simple, label-free and highly sensitive fluorescence method for APE1 detection.
3.1.2. Double-Stranded Linear DNA-Based Nucleic Acid Probes
In addition to hairpin DNA probes, double-stranded linear DNA probes are one of the most widely used tools in the linear DNA probes for APE1 analysis. Simeonov et al. used duplex probe to identify APE1 inhibitions [
65,
66]. Subsequently, Liu and co-workers designed a natural substrate of dsDNA probes for APE1 assay [
67]. As this probe contained one uracil residue, the probes were pretreated with Uracil DNA glycosylase (UDG) and then reduced with NaBH
4 to create an AP site. When APE1 cleaved DNA that labeled with fluorophore (TAMRA), it quickly dissociated, restoring fluorescence. This strategy featured rapidity and high simplicity, which could be easily adaptable to alternative DNA repair enzymes. Conventional dsDNA-based probes are susceptible to nonspecific cleavage by other nucleases. Lu et al. developed a DNA/RNA hybrid-based fluorescent probe. This hybrid-probes contained a 3′-base mismatch that could effectively enhance the activity of APE1 endonuclease. It showed good stability against other nucleases, and it made the APE1 detection in complex matrices possible [
68]. The hybrid probe, which allowed for one-step detection of APE1 subcellular activity, provided a practical and potential tool for APE1 high-throughput measurement subcellular activity. Label-free method can also be used in the dsDNA for the assay of the APE1 activity. Li et al. used the AP site-binding fluorophore 2-Amino-5,6,7-trimethyl-1,8-naphthyridine (ATMND) for APE1 activity imaging [
69]. As a C base was in the opposite location, the fluorescence of ATMND was quenched. After the APE site cleaved by APE1, the ATMND was released and the fluorescence recovered. This label-free technique provided a good sensitivity and selectivity for APE1 activity detection.
Since APE1 can specifically recognize and cleave the AP site at the duplex DNA, Li and co-workers developed a DNA system that contained a motor chain, a track strand and four stator strands to achieve autonomous movement with high controllability and processivity [
46]. As the motor-stator hybrid-probe contained AP site, it could be incised by APE1, while the ssDNA could not cleave. The cleaved short fragment products can activate the toehold domain for motor strand movement. As a proof of concept for developing gene regulators for creating cellular diagnostic tools, this DNA machine showed great potential. Although APE1 is applicable to a variety of DNA substrates containing AP sites, the relative activity of APE1 varied with the sequence. Wu’s group constructed a probe with 2-Aminopurine for APE1 activity imaging [
70]. 2-Aminopurine can produce different fluorescence in different DNA microenvironments (mononucleotide > ssDNA > dsDNA). The 2-Aminopurine fluorescence at the dsDNA is completely extinguished. However, it could be cut by APE1 and released DNA fragment that contained 2-Aminopurine. Then, the fragment was digested by Exo I and released the 2-Aminopurine mononucleotide to make fluorescence recovery. This strategy eliminated the influence from the little exonuclease activity of APE1 in comparison to the standard dual-terminal labeling. It was shown that the relative endonuclease activity of APE1 varied with the close or opposition base sequence. This study provided a reference for sequence design for imaging APE1 activity or APE1-mediated biosensors.
Linear DNA probes are widely used in APE1 imaging. Besides, they are common in many DNA substrates for signal amplification. However, as a hydrophilic molecule with a negative charge, linear DNA is difficult to enter living cells in the absence of transfection agent and vector, because the cell membrane is negatively charged. In the complex cellular environment, linear DNA probes are easily cleaved by other intracellular enzymes, causing false positives [
71,
72]. With the development of nanotechnology, some intelligent and multifunctional nanomaterials have become good gene carriers. Selected functional groups can be modified to efficiently bring large numbers of nucleic acid probes into living cells.
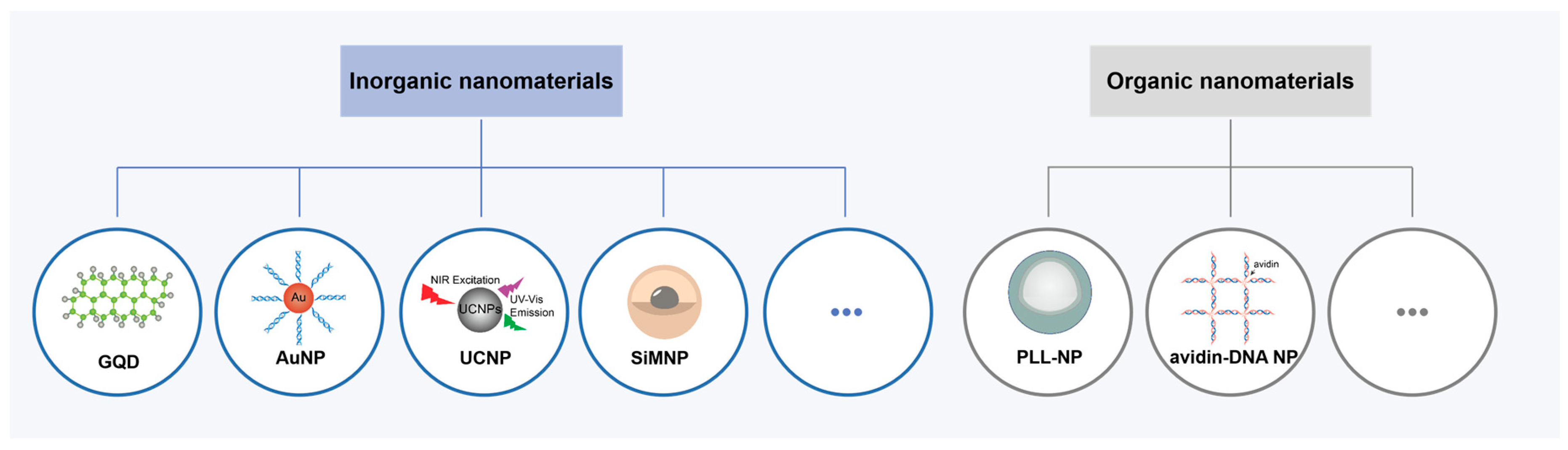
3.2. Composite DNA Nanomaterials-Empowered Fluorescence Imaging of APE1 Activity
Nanoparticles have attracted much attention as a carrier for APE1-probe delivery. According to Willner's group, combining biomolecules with nanoparticles can produce new hybrid nanostructures with distinctive traits that mix the characteristics of the biomolecules and the nanoparticles [
73]. DNA containing AP site is usually linked to nanomaterials by adsorption, covalent coupling and encapsulation to improve the stability of the probe in the plasma and further facilitate their cellular uptake [
71,
72,
74]. According to the properties, nanomaterials are categorized including inorganic nanomaterials and organic nanomaterials (
Figure 5). Inorganic nanomaterials like graphene quantum dots (GQDs), gold nanoparticles (AuNPs), lanthanide-doped upconversion nanoparticles (UCNPs) or other metals are desired as participants in these hybrid materials for APE1 activity imaging [
75,
76]. In addition, organic nanomaterials such as cationic polymer coated nanoparticles (PLL-NPs) and proteins can also be used for the same role, as they can provide predefined and biocompatible cargo carrying and targeting capabilities [
77,
78].
3.2.1. DNA/Inorganic Nanomaterial Composites
GQDs are zero-dimensional graphene nanomaterials with excellent properties and used in various applications [
67,
79]. Nanocomposites constructed of DNA and fine-sized GQDs can be effectively used to detect intracellular APE1 activity. Zhang et al. constructed a biocompatible functional nanocomposite, which was designed combination of single-molecule DNA and fine-sized GQDs for cellular APE1 imaging [
80]. The GQDs moiety of this nanocomposite were designed to be a quench agent that turned off the fluoresce of Cy3 in the DNA before the cleavage by APE1. In addition, they also facilitated the entry of functional nanocomposites into the cytoplasm by endocytosis. This study showed that sensitive, selective, and rapid detection of APE1 activity in living cells was possible by utilizing GQD-based nanocomposites.
AuNPs are also widely used in intracellular imaging of APE1 activity due to their unique properties and ease of molecular functionalization [
81,
82]. Cai and co-workers re-ported a DNA nanomachine to detect miRNA-21 and APE1 concurrently [
83]. The detecting device had two operating modes: OR and AND. The existence of any target could result in fluorescence in the OR mode, which was suited for sensitive detection. In the AND mode, fluorescence could be produced by the presence of all targets which was more suited for precise detection. It had potential in medical application and provided reference for other biomarker detection. In another study, as shown in
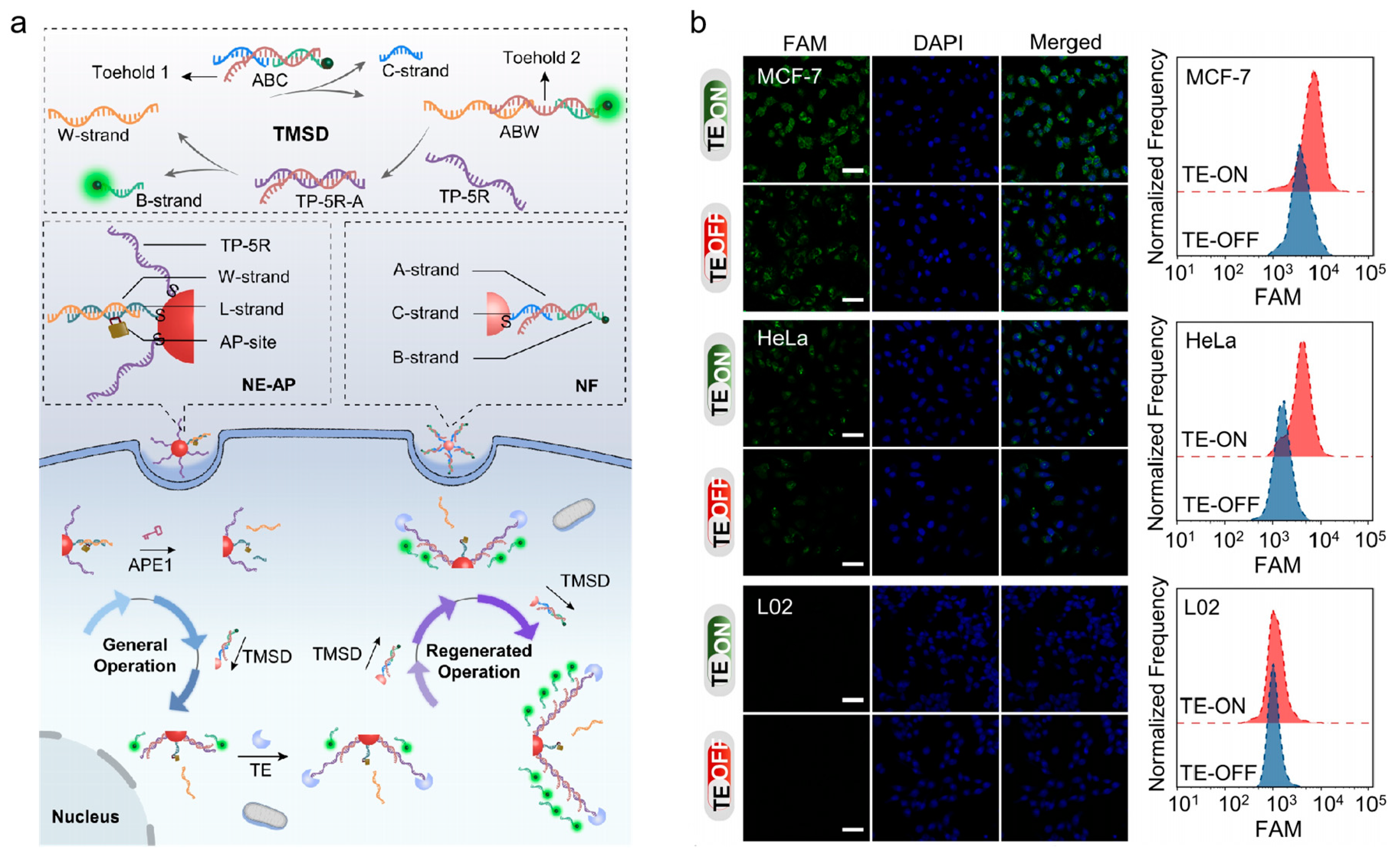
Figure 6a, Zhang et al. presented a DNA nanomachine for signal amplification imaging of APE1 in live cells [
84]. APE1 served as a switch in this DNA nanomachine to get it moving, and telomerase served as an engineer to continually expand new tracks. The DNA nanomachine was powered by the toehold-mediated strand displacement reaction, which provided enough fuel for the walker to travel continuously along each track. The fluorophore-labeled DNA strand was continuously released while the walker walked, enhancing fluorescence signals for APE1 imaging in different cell types (shown in
Figure 6b). Compared to traditional nanomachines, this nanomachine performs regenerative motion and shows higher specificity and sensitivity.
In recent years, UCNPs have been received a lot of attention to offer spatial and temporal control of APE1 activity detection. They can be activated in the biological window of the near-infrared (650-1700 nm) for upconversion luminescence (UCL), which results to reduced bioabsorption and deeper penetration [
85,
86]. Li’s group introduced a sensor technology that allowed in situ localization and photoactivation imaging of APE1 in the nucleus as well as mitochondria (shown in
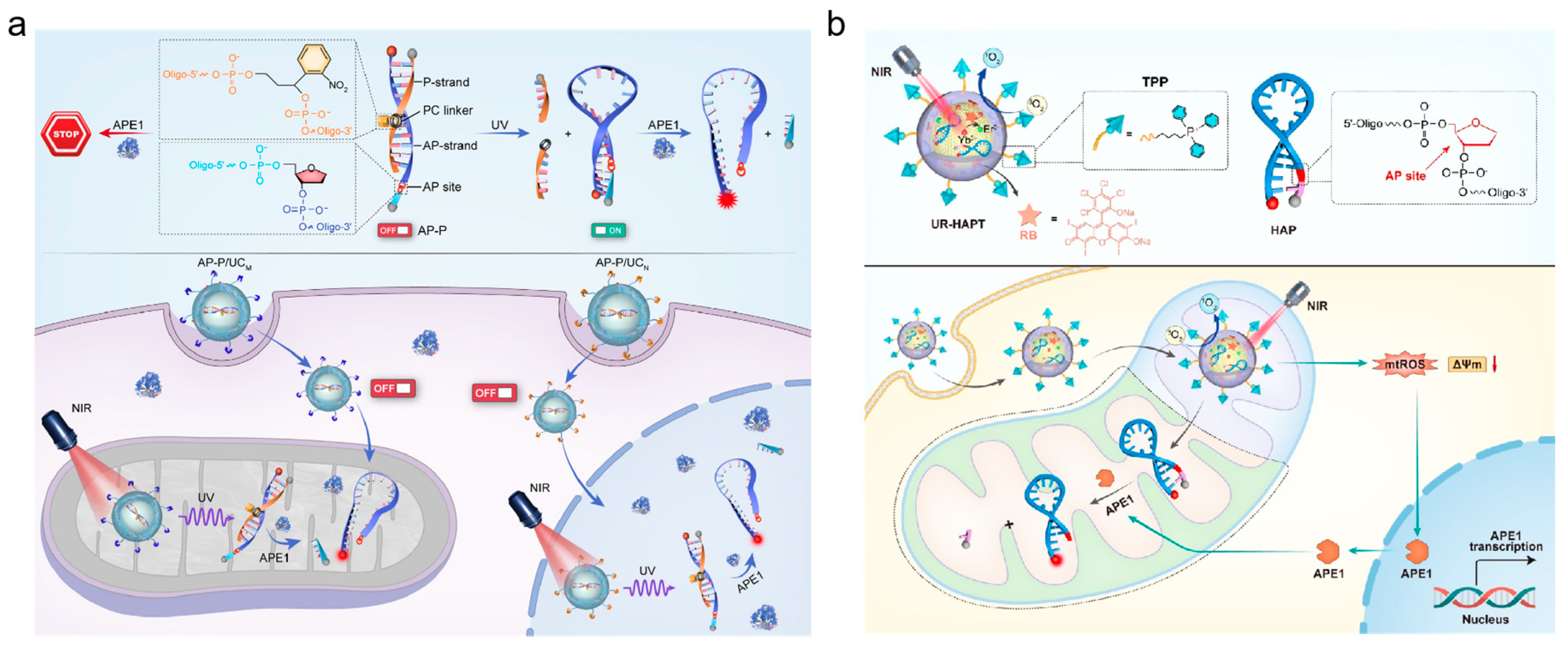
Figure 7a) [
87]. The P-strand contained photocleavable (PC) linker, and it was used to hybridize the AP-strand to block the fluorescent sequence of the target APE1. The UCNPs were used as carriers of DNA probe (AP-P) to improve the capability to penetrate deep tissue. This detection probe AP-P could effectively eliminate potential false-positive signals. Then, Li’s group presented a nanoplatform (UR-HAPT) for dynamic imaging of mitochondrial APE1 (shown in
Figure 7b) [
88]. It was constructed by the combination of an UCNP, rose bengal (RB)-modified UCNP, tri-phenyllphosphonium (TPP) ligands and DNA probe (HAP). Upon the TPP targeting, the nanostructure entered the mitochondria. During photodynamic therapy (PDT), the NIR light irradiation induced subcellular ROS production by the energy transfer from UCNPs to the photosensitizers RB, which increased the mitochondrial APE1 expression. Then, APE1 cleaved the AP site in HAP, and led to a recovery of fluorescence signal. Since PDT could increase the content of APE1 in mitochondria, this system could be used as an index to evaluate the efficacy of PDT in real-time.
For intracellular diagnosis or genetic treatment, silica coated magnetic core-shell NPs (SiMNPs) also employed to carry APE1-DNA probes into different types of cells [
89,
90]. As SiMNPs completely inhibits the interaction between DNA and nucleases, including APE1 [
91], it is not feasible to use SiMNPs only for APE1 activity imaging. Excitingly, interactions with avidin allow it to attract APE1 to DNA on nanoparticles. This allowed avidin-modified SiMNPs to be used for APE1 imaging. Zhai et al. demonstrated that APE1 might precisely and effectively cleave the AP site in DNA, which linked to the avidin-modified surface of SiMNPs. It could resist the nonspecific cleavage by other enzymes [
92]. This study demonstrated the potential application of avidin in the detection of nuclease activity.
3.2.2. DNA/Organic Nanomaterial Composites
It is difficult for naked DNA probes to penetrate the cell membrane, because both DNA and biological cell membranes are negatively charged. However, cationic polymer coated NPs can be employed for delivery of DNA probes as well as their stability improvement. Li and co-workers designed an activatable DNA nanoparticles for imaging of cytochrome c and APE1 [
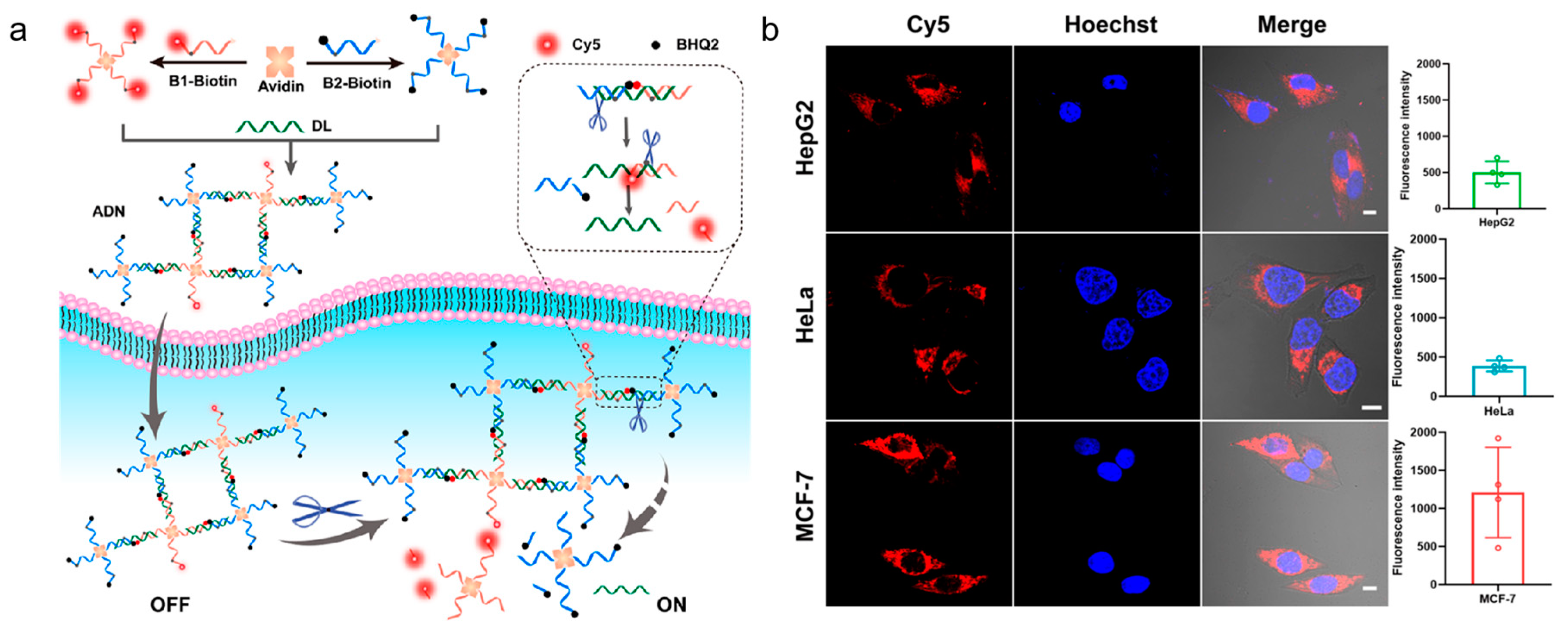
93]. The nanoparticles coated with cationic polymer was used for DNA probe (E-Apt) delivery. After the AP site was cleaved by APE1, it led to restoration of the aptamer recognition ability of cytochrome c. This method allowed for imaging of cytochrome c and APE1 with a DNA probe, which provided a basis for the detection of biological functions of related proteins. Inspired by the strong interaction between avidin and APE1, Li et al. proposed a DNA nanostructure, which consisted of a protein(avidin) as a connecting structure for imaging the activity of APE1 in different live cells (shown in
Figure 8) [
94]. The avidin-scaffolded DNA nanostructure, which was prepared based on the interaction between avidin and biotin, had a relatively loose structure with little steric hindrance. It favored APE1 entry into the AP sites. APE1 cleaved the nanostructure effectively and precisely, resulting in activated fluorescence for APE1 detection. By assembling an avidin-scaffold nanostructure, it could effectively improve the specificity of APE1 detection and reduce nonspecific cleavage by other nucleases.
Organic and inorganic nanomaterials can be used to effectively improve the stability of APE1-probes in the plasma and further facilitate their cellular uptake. On the other hand, composite DNA nanomaterials combine the APE1-responsive DNA systems with extra functions of nanomaterials to improve the performance in APE1 imaging [
72,
95]. Despite the rapid development in composite DNA nanomaterials, the fabrication process of nanomaterials is complex and time-consuming. Different from these nanomaterials, DNA possess many unique properties, including biocompatibility, programmability, ease of synthesis, and controllability on the nanoscale [
96,
97,
98]. Because of the self-assembly ability of DNA, three-dimensional (3D) nanostructures, such as DNA tetrahedron [
99], DNA polyhedron [
100], 3D DNA origami [
101], and DNA nanoflowers [
102,
103] can be created for APE1 activity imaging [
104].
3.3. 3D DNA Nanostructure-empowered fluorescence imaging of APE1 Activity
A strong hydrogen bond holds the 3D DNA nanostructure together. It has great qualities such as: (1) biocompatibility and nontoxicity; (2) precise size and shape management; and (3) facile targeting decorating in numerous places [
105]. Currently, 3D DNA nanostructures for APE1 activity imaging includes DNA prisms and DNA tetrahedrons [
106]. Herein, we concentrate on the usage of DNA tetrahedron for APE1 activity imaging since tetrahedron is extensively employed because to its high productivity and rigid structure [
107].
DNA tetrahedron is typically formed by four ssDNA strands. The four strands can be arranged to create a tetrahedral frame structure with six double-helix edges. Due to their superior mechanical rigidity and structural stability, DNA tetrahedrons make excellent molecular scaffolds [
108,
109]. Moreover, DNA tetrahedron have multiple sites for targeting decoration [
110,
111], and functional nucleic acids can be designed and constructed in DNA tetrahedron with superior properties for APE1 imaging. Su’s group used tumor aptamer-modified and AP site-contained DNA tetrahedron to functionalize with fluorescent probes (DTFP) for imaging of APE1 activity (shown in
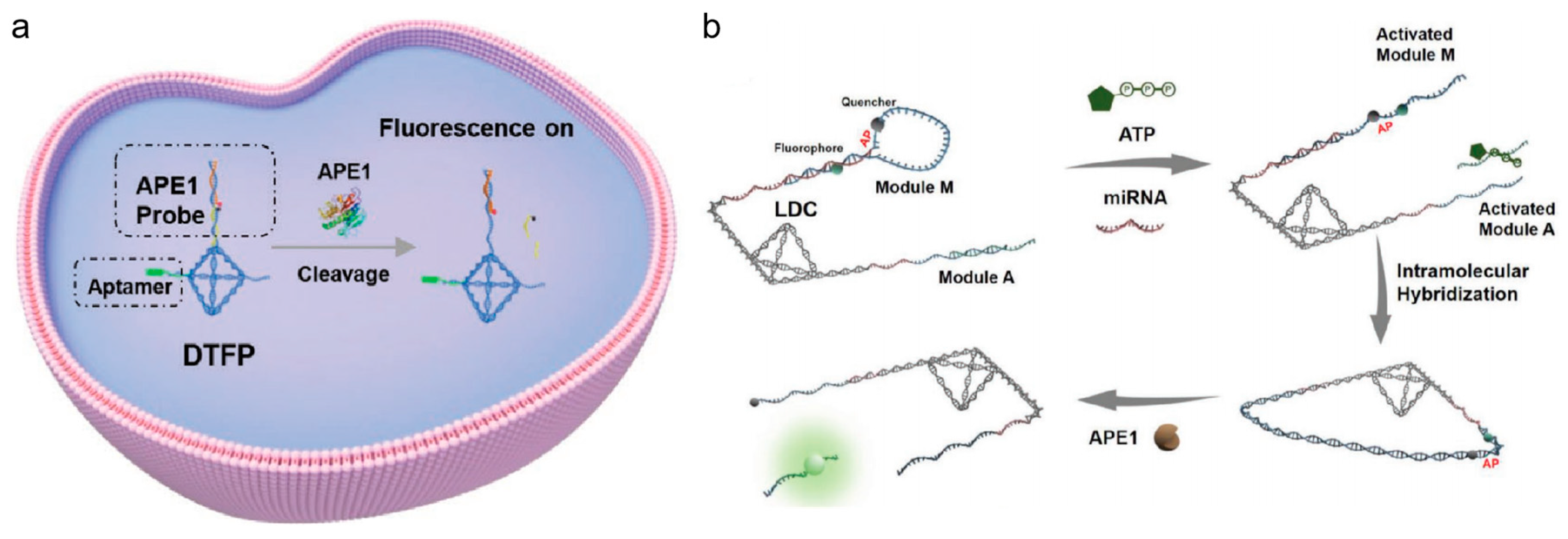
Figure 9a) [
112]. The AP site of the DNA could be cleaved by APE1 and short chains containing fluorophores could be released. As platinum-resistant cell lines with down-regulated APE1 expression were highly sensitive to platinum treatment, the potential to overcome this drug-resistance was demonstrated. Compared to traditional single-molecule biomarker assays, the use of multi-target detection can effectively enhance targeting accuracy. Moreover, the four overhangs of the tetrahedral structure make the design more flexible and programmable. Besides, the compact tetrahedral structure can link a large number of recognition chains with a large local concentration, thus greatly increasing the reaction rate. In Su’s group, Kou et al. created the intramolecular hybridization of localized DNA circuits (LDCs) with excellent sensitivity and quick reaction times that could concurrently detect intracellular miRNAs, APE1 and ATP (shown in
Figure 9b) [
113]. To carry out AND logic gate operation, three response elements were positioned on the DNA tetrahedron nanostructure. The release of fluorescence signal was dependent on the coexistence of miRNA and APE1 with a significant amount of ATP. This discovery offered a potent tool for accurate diagnostics and substantially enhanced the performance of DNA-based nanodevices.
Besides, the high-efficiency amplification technology provides a robust tool for the sensitive detection of APE1, which can improve the accurate diagnosis. Meanwhile, as a disease biomarker, APE1 can be used to release small interfering RNAs (siRNAs) for precise treatment. Two kinds of functionalized tetrahedral DNA nanostructures (f-TND1 and f-TND2) were constructed to form a DNA nanostructure by Zhou and co-workers [
114]. f-TND1 contained tetrahedral DNA and two hairpin DNA: H1 and H2, while f-TND2 contained tetrahedral DNA, H1 and H3. As there were aptamer Sgc8 and AP sites in H1, Cy5, BHQ2 and siRNA in H2, APE1 could cause the AP sites to split, releasing S1 to start CHA reactions on two tetrahedral DNA nanostructures, creating massive network nanostructures that significantly increased the production of fluorescent signals, enabling sensitive detection and intracellular imaging of APE1. To fulfill the goal of targeted tumor therapy, siRNA produced during the CHA response may inhibit cell survival. This work offered a superb and efficient nanoplatform for accurate gene treatment of cancers, sensitive biomarker detection, and disease-specific diagnostics.
APE1 is a desirable target for the development of anticancer drugs. Various small-molecule APE1 inhibitors have been studied in recent years to improve anticancer treatments. Small molecule drugs, however, are typically linked to numerous medication resistance and adverse effects [
115]. DNA tetrahedrons have been widely used in delivery of siRNA and small molecules drugs, such as platinum drug and doxorubicin. Interestingly, DNA tetrahedron can regulate the APE1 activity. Using DNA tetrahedrons, Zhang et al. reported a unique method for examining and controlling cellular APE1 activity (shown in
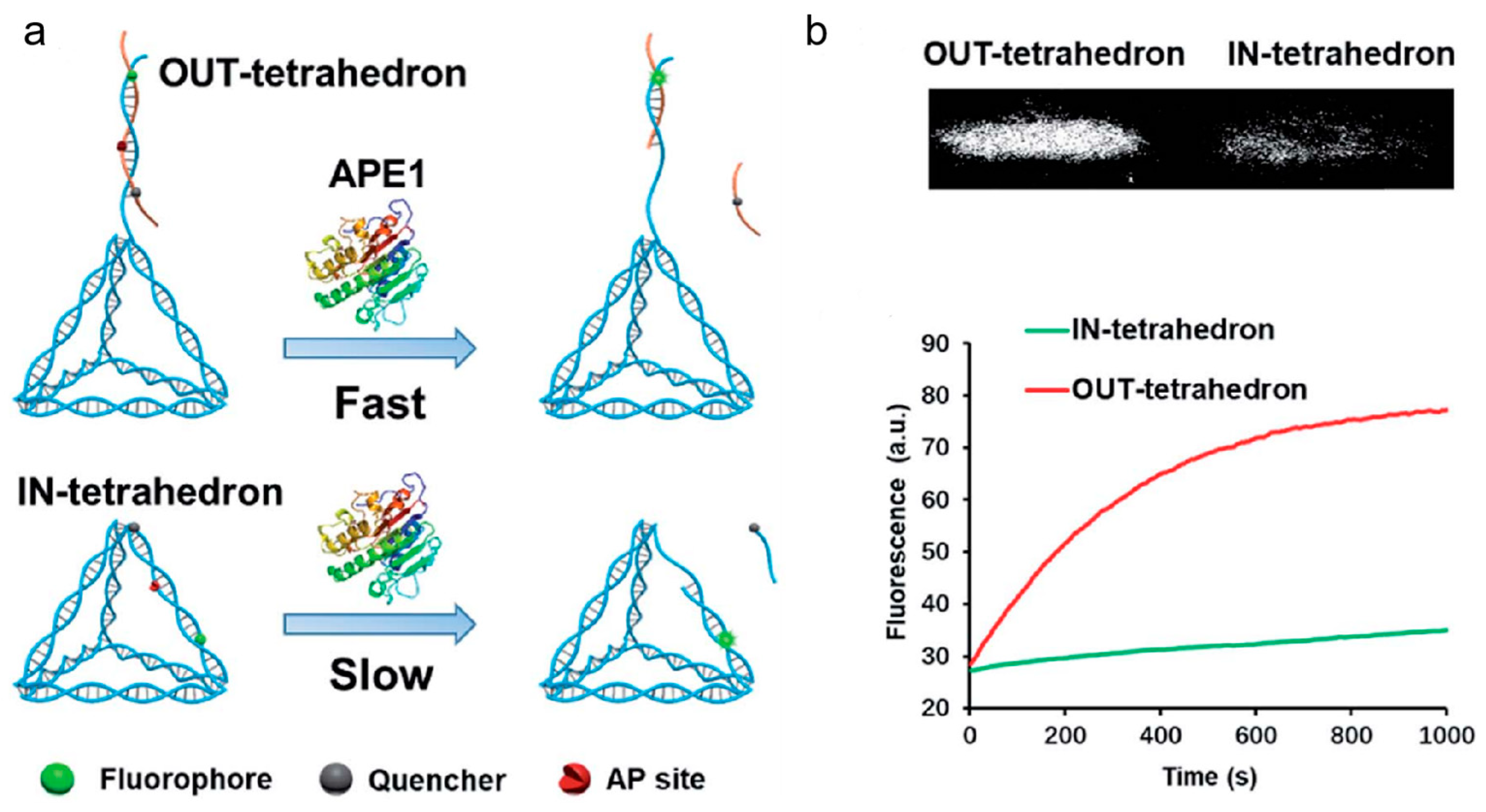
Figure 10) [
116]. Contrary to typical molecular probes and inhibitors, the DNA tetrahedrons function as both a probe and an inhibitor, and their functions may be changed by modifying the AP site. The tetrahedron with an antenna that contains an AP site demonstrated good APE1 sensitivity and specificity. On the other hand, the tetrahedron with an AP site on its scaffold dis-played a substantial affinity for APE1 binding. This research offered a unique strategy for controlling enzyme activity as well as fresh understanding of how enzymes and substrates interact. It had potential implications in the inhibition of APE1 activity and the treatment of cancer.
All three of the above probes have been reviewed for imaging of APE1 activity. In fact, linear DNA probes, composite DNA nanomaterials as well as 3D DNA nanostructures can also be combined for APE1 imaging. Lv and co-workers described a stochastic bipedal DNA walker that traveled on GNPs by itself via the CHA reaction [
117]. They designed a hairpin chain containing the AP site to serve as a substrate for APE1 and a stochastic bipedal DNA walker that autonomously traversed on GNPs. To improve the internalization efficiency, lipofectamine 3000 encapsulated probes were incubated with cells. This allowed for very sensitive, activatable fluorescence imaging of intracellular BER activity. Besides, to precisely monitor intracellular APE1 activity, Zhou et al. created the DNAzyme-modified tetrahedral DNA nanostructure (DZ-TDN) and molecular beacon (MB) integrated biosensing system [
118]. Thanks to liposomes, the device can be successfully internalized into live cells to monitor changes in intracellular APE1 activity. They developed an effective technique for tumor cells imaging and APE1 detection. In 2022, APE1 was successfully detected and imaged intracellularly by Zhou and co-workers. They used a tetrahedral DNA walker with four arms that moved autonomously on the GNPs via CHA. With the help of lipofectamine 3000, the DNA nanomaterials were transfected into tumor cells [
119]. This design offered a unique DNA walker for sensitive detection of APE1.
4. Discussion and Perspectives
In this review, we summarized recent advances of DNA nanotechnology-empowered fluorescence imaging of APE1 activity. APE1, which is important in BER pathway and regulation of redox signaling in cells, is an essential multifunctional protein in mammals. The abnormal expression and disruptions in the biological functions of APE1 are linked to a variety of diseases. Since it can specifically recognize and cleave AP sites in DNA strands, DNA nanotechnology is commonly used for APE1 imaging. Linear DNA-based nucleic acid probes are the most basic type of nucleic acid probes used for APE1 imaging. However, since naked linear DNA probes are small and have a negatively charged backbone, they exhibit ineffective cellular internalization. Also, they are quickly broken down once within cells, preventing them from getting to the target areas. In order to transport DNA probes intracellularly and achieve a desired performance in bioanalysis and cancer therapies, nanocarriers have been widely employed. Despite the unique properties of these nanomaterials, the fabrication process of nanomaterials is complex and time-consuming, which severely restricts their practical applicability. With the rapid development of DNA nanotechnology, the advantages of programmability and high biocompatibility of DNA have spurred extensive research on the design and assembly of a variety of 3D nanostructures with well-defined structures for the delivery of AP sites in recent years.
Despite current achievements, there is still plenty of room in DNA nanotechnology-empowered fluorescence imaging of APE1 activity. Firstly, complex biological matrices may seriously interfere with the selectivity of probes for imaging APE1 in living cells and
in vivo, causing false-positive results. Thus, the specificity of APE1 detection can be improved through the following strategies: (1) introducing a small loop structure to reduce non-specific cleavage by other nucleases in the cell or replacing traditional dsDNA with DNA/RNA probes; (2) using more complex DNA nanostructures. Secondly, the use of DNA nanotechnology for imaging APE1 is mainly based on its endonuclease activity. The measurement of APE1 expression in cells is also important for understanding the disease progression. APE1 aptamer may be an excellent tool for APE1 assay [
120]. For example, Qin et al. used the APE1 aptamer to inhibit APE1, and they designed a partial complementary strand to enable APE1 release and activity restoration [
121].
Author Contributions
Conceptualization, investigation, writing original draft, H.H.; conceptualization, investigation, X.L. and Y.W.; investigation, L.Q., Y.Z. and J.Z.; general guidance, project directing, and manuscript revisions, K.W. and X. H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Natural Science Foundation of China (21735002, 21874035 and 22174044).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Izumi, T.; Mitra, S. Deletion analysis of human AP-endonuclease: minimum sequence required for the endonuclease activity. Carcinogenesis 1998, 19, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Lopez, D.J.; Rodriguez, J.A.; Banuelos, S. Molecular mechanisms regulating the DNA repair protein APE1: a focus on its flexible N-terminal tail domain. Int. J. Mol. Sci. 2021, 22, 6308. [Google Scholar] [CrossRef]
- Demple, B.; Herman, T.; Chen, D.S. Cloning and expression of APE, the cDNA encoding the major human apurinic endonuclease: definition of a family of DNA repair enzymes. Proc. Natl. Acad. Sci. 1991, 88, 11450–11454. [Google Scholar] [CrossRef]
- Oliveira, T.T.; Coutinho, L.G.; de Oliveira, L.O.A.; de Souza Timoteo, A.R.; Farias, G.C.; Agnez-Lima, L.F. APE1/Ref-1 role in inflammation and immune response. Front. Immunol. 2022, 13, 793096. [Google Scholar] [CrossRef] [PubMed]
- Barnes, T.; Kim, W.C.; Mantha, A.K.; Kim, S.E.; Izumi, T.; Mitra, S.; Lee, C.H. Identification of Apurinic/apyrimidinic endonuclease 1 (APE1) as the endoribonuclease that cleaves c-myc mRNA. Nucleic Acids Res. 2009, 37, 3946–3958. [Google Scholar] [CrossRef]
- Tell, G.; Pellizzari, L.; Cimarosti, D.; Pucillo, C.; Damante, G. Ref-1 controls pax-8 DNA-binding activity. Biochem. Biophys. Res. Commun. 1998, 252, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Berquist, B.R.; McNeill, D.R.; Wilson III, D.M. Characterization of abasic endonuclease activity of human Ape1 on alternative substrates, as well as effects of ATP and sequence context on AP site incision. J. Mol. Biol. 2008, 379, 17–27. [Google Scholar] [CrossRef]
- Cohen, I. DNA damage talks to inflammation. Cytokine Growth Factor Rev. 2017, 33, 35–39. [Google Scholar] [CrossRef]
- Zou, G.M.; Luo, M.H.; Reed, A.; Kelley, M.R.; Yoder, M.C. Ape1 regulates hematopoietic differentiation of embryonic stem cells through its redox functional domain. Blood 2007, 109, 1917–1922. [Google Scholar] [CrossRef]
- Fishel, M.L.; He, Y.; Reed, A.M.; Chin-Sinex, H.; Hutchins, G.D.; Mendonca, M.S.; Kelley, M.R. Knockdown of the DNA repair and redox signaling protein Ape1/Ref-1 blocks ovarian cancer cell and tumor growth. DNA Repair 2008, 7, 177–186. [Google Scholar] [CrossRef]
- Fishel, M.L.; Jiang, Y.; Rajeshkumar, N.V.; Scandura, G.; Sinn, A.L.; He, Y.; Shen, C.; Jones, D.R.; Pollok, K.E.; Ivan, M.; Maitra, A.; Kelley, M.R. Impact of APE1/Ref-1 redox inhibition on pancreatic tumor growth. Mol. Cancer Ther. 2011, 10, 1698–1708. [Google Scholar] [CrossRef] [PubMed]
- Dianov, G.; Lindahl, T. Reconstitution of the DNA base excision-repair pathway. Curr. Bio. 1994, 4, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Schermerhorn, K.M.; Delaney, S. A chemical and kinetic perspective on base excision repair of DNA. Acc. Chem. Res. 2014, 47, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Zou, G.M.; Karikari, C.; Kabe, Y.; Handa, H.; Anders, R.A.; Maitra, A. The Ape-1/Ref-1 redox antagonist E3330 inhibits the growth of tumor endothelium and endothelial progenitor cells: therapeutic implications in tumor angiogenesis. J. Cell. Physiol. 2009, 219, 209–218. [Google Scholar] [CrossRef]
- Zhong, C.; Xu, M.; Wang, Y.; Xu, J.; Yuan, Y. An APE1 inhibitor reveals critical roles of the redox function of APE1 in KSHV replication and pathogenic phenotypes. PLoS Pathog 2017, 13, e1006289. [Google Scholar] [CrossRef]
- Pines, A.; Perrone, L.; Bivi, N.; Romanello, M.; Damante, G.; Gulisano, M.; Kelley, M.R.; Quadrifoglio, F.; Tell, G. Activation of APE1/Ref-1 is dependent on reactive oxygen species generated after purinergic receptor stimulation by ATP. Nucleic Acids Res. 2005, 33, 4379–4394. [Google Scholar] [CrossRef]
- Mohni, K. N; Wessel, S.R.; Zhao, R.; Wojciechowski, A.C.; Luzwick, J.W.; Layden, H.; Eichman, B.F.; Thompson, P.S.; Mehta, K.P.M.; Cortez, D. HMCES maintains genome integrity by shielding abasic sites in single-strand DNA. Cell 2019, 176, 144–153. [Google Scholar] [CrossRef]
- Lindahl, T. Instability and decay of the primary structure of DNA. Nature 1993, 362, 709–715. [Google Scholar] [CrossRef]
- Lindahl, T.; Nyberg, B. Rate of depurination of native deoxyribonucleic acid. Biochemistry 1972, 11, 3610–3618. [Google Scholar] [CrossRef]
- Sczepanski, J.T.; Wong, R.S.; McKnight, J.N.; Bowman, G.D.; Greenberg, M.M. Rapid DNA-protein cross-linking and strand scission by an abasic site in a nucleosome core particle. Proc. Natl. Acad. Sci. 2010, 107, 22475–22480. [Google Scholar] [CrossRef]
- Hoitsma, N.M.; Whitaker, A.M.; Beckwitt, E.C.; Jang, S.; Agarwal, P.K.; Houten, B.V.; Freudenthal, B.D. AP-endonuclease 1 sculpts DNA through an anchoring tyrosine residue on the DNA intercalating loop. Nucleic Acids Res. 2020, 48, 7345–7355. [Google Scholar] [CrossRef] [PubMed]
- Drohat, A.C.; Coey, C.T. Role of base excision "repair" enzymes in erasing epigenetic marks from DNA. Chem. Rev. 2016, 116, 12711–12729. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, T. An N-glycosidase from Escherichia coli that releases free uracil from DNA containing deaminated cytosine residues. Proc. Natl. Acad. Sci. 1974, 71, 3649–3653. [Google Scholar] [CrossRef] [PubMed]
- Kubota, Y.; Nash, R.A.; Klungland, A.; Schar, P.; Barnes, D.E.; Lindahl, T. Reconstitution of DNA base excision-repair with purified human proteins: interaction between DNA polymerase β and the XRCC1 protein. The EMBO Journal 1996, 15, 6662–6670. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Demple, B. Knockout and inhibition of Ape1: roles of Ape1 in base excision DNA repair and modulation of gene expression. Antioxidants 2022, 11, 1817. [Google Scholar] [CrossRef]
- Chen, W.; Wang, S.; Xing, D. New horizons for the roles and association of APE1/Ref-1 and ABCA1 in atherosclerosis. J. Inflamm. Res. 2021, 14, 5251–5271. [Google Scholar] [CrossRef]
- Weaver, T.M.; Hoitsma, N.M.; Spencer, J.J.; Gakhar, L.; Schnicker, N.J.; Freudenthal, B.D. Structural basis for APE1 processing DNA damage in the nucleosome. Nat. Commun. 2022, 13, 5390. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, A.M.; Freudenthal, B.D. APE1: A skilled nucleic acid surgeon. DNA Repair 2018, 71, 93–100. [Google Scholar] [CrossRef]
- Fan, Z.; Beresford, P.J.; Zhang, D.; Xu, Z.; Novina, C.D.; Yoshida, A.; Pommier, Y.; Lieberman, J. Cleaving the oxidative repair protein Ape1 enhances cell death mediated by granzyme A. Nat. Immunol. 2003, 4, 145–153. [Google Scholar] [CrossRef]
- Lu, H.; Hong, J.; Peng, D.; Bhat, A.A.; Chen, Z.; Zaika, A.; El-Rifai, W.M. A new function of APE1 in barrett’s esophagus and esophageal adenocarcinoma: APE1 upregulates MMP2 and MMP14 to promote invasion. Gastroenterology 2017, 152, S237. [Google Scholar] [CrossRef]
- Antoniali, G.; Serra, F.; Lirussi, L.; Tanaka, M.; D’Ambrosio, C.; Zhang, S.; Radovic, S.; Dalla, E.; Ciani, Y.; Scaloni, Y.; Li, M.; Piazza, S.; Tell, G. Mammalian APE1 controls miRNA processing and its interactome is linked to cancer RNA metabolism. Nat. Commun. 2017, 8, 797. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yang, X.; Lu, X.; Dai, N.; Zhang, S.; Cheng, Y.; Zhang, L.; Yang, Y.; Liu, Y.; Yang, Z.; Wang, D.; Wilson III, D.M. APE1 deficiency promotes cellular senescence and premature aging features. Nucleic Acids Res. 2018, 46, 5664–5677. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Willner, B.; Willner, I. DNA nanotechnology: from sensing and DNA machines to drug-delivery systems. ACS Nano 2013, 7, 8320–8332. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.; Huang, J.; Yang, X.; Yang, Y.; Quan, K.; Ou, M.; Fang, H.; Wang, K. Competition-mediated FRET-switching DNA tetrahedron molecular beacon for intracellular molecular detection. ACS Sensors 2016, 1, 1445–1452. [Google Scholar] [CrossRef]
- Lei, Y.; Qian, Z.; Tang, J.; He, X.; Shi, H.; Ye, X.; Yan, L.; He, D.; Wang, K. DNA nanotriangle-scaffolded activatable aptamer probe with ultralow background and robust stability for cancer theranostics. Theranostics 2018, 8, 4062–4071. [Google Scholar] [CrossRef]
- Zou, S.; Lei, Y.; Ma, W.; Chen, B.; Cheng, H.; Jia, R.; Li, Z.; He, X.; Wang, K. Extracellular pH-manipulated in situ reconfiguration of aptamer functionalized DNA monomer enables specifically improved affinity, detection and drug delivery. Analyst 2020, 145, 2562–2569. [Google Scholar] [CrossRef]
- Huang, J.; Wu, Y.; He, H.; Ma, W.; Liu, J.; Cheng, H.; Sun, H.; He, X.; Wang, K. Acidic microenvironment triggered in situ assembly of activatable three-arm aptamer nanoclaw for contrast-enhanced imaging and tumor growth inhibition in vivo. Theranostics 2022, 12, 3474–3487. [Google Scholar] [CrossRef]
- Wang, F.; Willner, B.; Willner, I. DNA nanotechnology with one-dimensional self-assembled nanostructures. Curr. Opin. Biotechnol. 2013, 24, 562–574. [Google Scholar] [CrossRef]
- Atamna, H.; Cheung, I.; Ames, B.N. A method for detecting abasic sites in living cells: age-dependent changes in base excision repair. Proc. Natl. Acad. Sci. 2000, 97, 686–691. [Google Scholar] [CrossRef]
- Khodyreva, S.; Lavrik, O. Non-canonical interaction of DNA repair proteins with intact and cleaved AP sites. DNA Repair 2020, 90, 102847. [Google Scholar] [CrossRef]
- Abe, Y.S.; Sasaki, S. DNA cleavage at the AP site via β-elimination mediated by the AP site-binding ligands. Bioorg. Med. Chem. 2016, 24, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Boiteux, S.; Guillet, M. Abasic sites in DNA: repair and biological consequences in Saccharomyces cerevisiae. DNA Repair 2004, 3, 1–12. [Google Scholar] [CrossRef]
- Abe, Y.S.; Sasaki, S. The adduct formation between the thioguanine-polyamine ligands and DNA with the AP site under UVA irradiated and non-irradiated conditions. Bioorg. Med. Chem. 2019, 27, 115160. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, A.A.; Fedorova, O.S.; Kuznetsov, N.A. Kinetic features of 3'-5' exonuclease activity of human AP-endonuclease APE1. Molecules 2018, 23, 2101. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, S.B.; Samanta, D.; Mirkin, C.A. DNA-based nanostructures for live-cell analysis. J. Am. Chem. Soc. 2020, 142, 11343–11356. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, N.; Fu, S.; Deng, Y.; Yu, C.; Su, X. Base excision repair-inspired DNA motor powered by intracellular apurinic/apyrimidinic endonuclease. Nanoscale 2019, 11, 1343–1350. [Google Scholar] [CrossRef]
- Strohsahl, C.M.; Krauss, T.D.; Miller, B.L. Identification of high-stringency DNA hairpin probes by partial gene folding. Biosens. Bioelectron. 2007, 23, 233–240. [Google Scholar] [CrossRef]
- Bidar, N.; Amini, M.; Oroojalian, F.; Baradaran, B.; Hosseini, S.S.; Shahbazi, M.A.; Hashemzaei, M.; Mokhtarzadeh, A.; Hamblin, M.R.; de la Guardia, M. Molecular beacon strategies for sensing purpose. TrAC Trends in Analytical Chemistry 2021, 134, 116143. [Google Scholar] [CrossRef]
- Zhang, Y.; Ren, W.; Luo, H.Q.; Li, N.B. Label-free cascade amplification strategy for sensitive visual detection of thrombin based on target-triggered hybridization chain reaction-mediated in situ generation of DNAzymes and Pt nanochains. Biosens. Bioelectron. 2016, 80, 463–470. [Google Scholar] [CrossRef]
- Yang, D.; Ning, L.; Gao, T.; Ye, Z.; Li, G. Enzyme-free dual amplification strategy for protein assay by coupling toehold-mediated DNA strand displacement reaction with hybridization chain reaction. Electrochemistry Communications 2015, 58, 33–36. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, X.; Yang, X.; Liu, P.; Wang, K.; Huang, J.; Liu, J.; Song, C.; Wang, J. Colorimetric detection of mercury ion based on unmodified gold nanoparticles and target-triggered hybridization chain reaction amplification. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 136, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Lin, Y.; Lin, Y.; Wei, Q.; Chen, G.; Tang, D. In situ amplified electrochemical aptasensing for sensitive detection of adenosine triphosphate by coupling target-induced hybridization chain reaction with the assembly of silver nanotags. Talanta 2016, 146, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Qiu, Z.; Lu, M.; Shu, J.; Tang, D. Hybridization chain reaction-based colorimetric aptasensor of adenosine 5'-triphosphate on unmodified gold nanoparticles and two label-free hairpin probes. Biosens. Bioelectron. 2017, 89, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Miao, X.; Zhu, A.; Ling, L. Hybridization chain reaction and gold nanoparticles dual signal amplification for sensitive glucose detection. Biochemical Engineering Journal 2015, 103, 205–210. [Google Scholar] [CrossRef]
- Dai, J.; Xing, C.; Lin, Y.; Huang, Y.; Yang, Y.; Chen, Z.; Lu, C.; Yang, H. Localized DNA catalytic hairpin assembly reaction on DNA origami for tumor-associated microRNA detection and imaging in live cells. Sensors and Actuators B: Chemical 2021, 344, 130195. [Google Scholar] [CrossRef]
- Miao, P. Magnetic multipedal DNA walking nanomachine driven by catalytic hairpin assembly. Anal. Chem. 2023, 95, 6760–6764. [Google Scholar] [CrossRef] [PubMed]
- Okabe, K.; Harada, Y.; Zhang, J.; Tadakuma, H.; Tani, T.; Funatsu, T. Real time monitoring of endogenous cytoplasmic mRNA using linear antisense 2'-O-methyl RNA probes in living cells. Nucleic Acids Res. 2011, 39, e20. [Google Scholar] [CrossRef]
- Li, L.; Guan, R.; Guo, M.; Ning, P.; Shao, R.; Meng, X. A FRET based two-photon fluorescent probe for ratiometric detection of Pd2+ in living cells and in vivo. Sensors and Actuators B: Chemical 2018, 254, 949–955. [Google Scholar] [CrossRef]
- He, J.H.; Cheng, Y.Y.; Zhang, Q.Q.; Liu, H.; Huang, C.Z. Carbon dots-based fluorescence resonance energy transfer for the prostate specific antigen (PSA) with high sensitivity. Talanta 2020, 219, 121276. [Google Scholar] [CrossRef]
- Lu, X.; Li, D.; Luo, Z.; Duan, Y. A dual-functional fluorescent biosensor based on enzyme-involved catalytic hairpin assembly for the detection of APE1 and miRNA-21. Analyst 2022, 147, 2834–2842. [Google Scholar] [CrossRef]
- Fang, S.; Chen, L.; Zhao, M. Unimolecular chemically modified DNA fluorescent probe for one-step quantitative measurement of the activity of human apurinic/apyrimidinic endonuclease 1 in biological samples. Anal. Chem. 2015, 87, 11952–11956. [Google Scholar] [CrossRef] [PubMed]
- Chai, Q.; Chen, J.; Zeng, S.; Zhu, T.; Chen, J.; Qi, C.; Mao, G.; Liu, Y. Closed cyclic DNA machine for sensitive logic operation and APE1 detection. Small 2023, 19, e2207736. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ma, Y.; Li, Y.; Xiong, M.; Li, X.; Zhang, L.; Zhao, S. Sensitive and label-free fluorescence detection of apurinic/apyrimidinic endonuclease 1 activity based on isothermal amplified-generation of G-quadruplex. New Journal of Chemistry 2017, 41, 1893–1896. [Google Scholar] [CrossRef]
- Liu, B.; Yang, Z.; Huang, T.; Li, M.M.; Duan, W.; Xie, B.; Chen, J.X.; Dai, Z.; Chen, J. Label-free and highly sensitive APE1 detection based on rolling circle amplification combined with G-quadruplex. Talanta 2022, 244, 123404. [Google Scholar] [CrossRef]
- Simeonov, A.; Kulkarni, A.; Dorjsuren, D.; Jadhav, A.; Shen, M.; McNeill, D.R.; Austin, C.P.; Wilson III, D.M. Identification and characterization of inhibitors of human apurinic/apyrimidinic endonuclease APE1. PLoS One 2009, 4, e5740. [Google Scholar] [CrossRef]
- Seiple, L.A.; Cardellina II, J.H.; Akee, R.; Stivers, J.T. Potent inhibition of human apurinic/apyrimidinic endonuclease 1 by arylstibonic acids. Mol. Pharmacol. 2008, 73, 669–677. [Google Scholar] [CrossRef]
- Li, F.; Xie, Q.; Qin, Y.; Tong, C.; Liu, B.; Wang, W. Real-time monitoring and effector screening of APE1 based on rGO assisted DNA nanoprobe. Anal. Biochem. 2021, 633, 114394. [Google Scholar] [CrossRef]
- Lu, P.; Cao, X.; Zheng, J.; Zhu, C.; Zhang, R.; Sun, Y.; Yang, Z.; Tang, Z.; Wang, J.; Zhao, M. A DNA/RNA hybrid fluorescent probe for high-throughput quantification of the activity of human apurinic/apyrimidinic endonuclease 1 in subcellular extracts. Biosensors and Bioelectronics: X 2023, 14, 100329. [Google Scholar] [CrossRef]
- Li, X.; Xiong, M.; Huang, Y.; Zhang, L.; Zhao, S. Simple label-free fluorescence detection of apurinic/apyrimidinic endonuclease 1 activity and its inhibitor using the abasic site-binding fluorophore. Analytical Methods 2019, 11, 739–743. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Tang, H.; Yang, B.; Zhao, Y.; Wu, P. Evaluation of the sequence-dependent relative activity of APE1 for optimal biosensing design. Biosens. Bioelectron. 2022, 214, 114539. [Google Scholar] [CrossRef]
- Moon, W.J.; Liu, J. Interfacing catalytic DNA with nanomaterials. Advanced Materials Interfaces 2020, 7, 2001017. [Google Scholar] [CrossRef]
- Lv, Z.; Zhu, Y.; Li, F. DNA Functional nanomaterials for controlled delivery of nucleic acid-based drugs. Front. Bioeng. Biotechnol. 2021, 9, 720291. [Google Scholar] [CrossRef] [PubMed]
- Baron, R.; Willner, B.; Willner, I. Biomolecule-nanoparticle hybrids as functional units for nanobiotechnology. Chem. Commun. 2007, 4, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Samanta, A.; Medintz, I.L. Nanoparticles and DNA-a powerful and growing functional combination in bionanotechnology. Nanoscale 2016, 8, 9037–9095. [Google Scholar] [CrossRef]
- Heuer-Jungemann, A.; Linko, V. Engineering inorganic materials with DNA nanostructures. ACS Cent. Sci. 2021, 7, 1969–1979. [Google Scholar] [CrossRef]
- Wang, J.; Li, Z.; Willner, I. Dynamic reconfigurable DNA nanostructures, networks and materials. Angew. Chem. Int. Ed. 2023, 62, e202215332. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Lin, L.; Chao, Z.; Shao, C.; Chen, Z.; Wei, Z.; Lu, J.; Huang, Y.; Li, L.; Liu, Q.; Liang, Y.; Tian, L. Organic spherical nucleic acids for the transport of a NIR-II-emitting dye across the blood-brain barrier. Angew. Chem. Int. Ed. 2020, 59, 9702–9710. [Google Scholar] [CrossRef]
- Lombardo, D.; Calandra, P.; Pasqua, L.; Magazu, S. Self-assembly of organic nanomaterials and biomaterials: the bottom-up approach for functional nanostructures formation and advanced applications. Materials 2020, 13, 1048. [Google Scholar] [CrossRef]
- Ahirwar, S.; Mallick, S.; Bahadur, D. Electrochemical method to prepare graphene quantum dots and graphene oxide quantum dots. ACS Omega 2017, 2, 8343–8353. [Google Scholar] [CrossRef]
- Zhang, H.; Ba, S.; Yang, Z.; Wang, T.; Lee, J.Y.; Li, T.; Shao, F. Graphene quantum dot-based nanocomposites for diagnosing cancer biomarker APE1 in living cells. ACS Appl. Mater. Interfaces. 2020, 12, 13634–13643. [Google Scholar] [CrossRef]
- Wong, A.C.; Wright, D.W. Size-dependent cellular uptake of DNA functionalized gold nanoparticles. Small 2016, 12, 5592–5600. [Google Scholar] [CrossRef] [PubMed]
- Jans, H.; Huo, Q. Gold nanoparticle-enabled biological and chemical detection and analysis. Chem. Soc. Rev. 2012, 41, 2849–2866. [Google Scholar] [CrossRef]
- Cai, Z.; Wang, A.; Wang, Y.; Qiu, Z.; Li, Y.; Yang, H.; Fu, M.; Liu, M.; Yu, Y.; Gao, F. Smart programmable scalable dual-mode diagnostic logic nanoflare strategy for dual-tumor marker detection. Anal. Chem. 2022, 94, 9715–9723. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, Y.; Wang, W.; Min, Q.; Zhang, J.R.; Zhu, J.J. A telomerase-assisted strategy for regeneration of DNA nanomachines in living cells. Angew. Chem. Int. Ed. 2023, 62, e202213884. [Google Scholar] [CrossRef]
- Du, K.; Feng, J.; Gao, X.; Zhang, H. Nanocomposites based on lanthanide-doped upconversion nanoparticles: diverse designs and applications. Light Sci. Appl. 2022, 11, 222. [Google Scholar] [CrossRef]
- Liang, G.; Wang, H.; Shi, H.; Wang, H.; Zhu, M.; Jing, A.; Li, J.; Li, G. Recent progress in the development of upconversion nanomaterials in bioimaging and disease treatment. J Nanobiotechnology 2020, 18, 154. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Zhao, J.; Yuan, J.; Zhao, Y.; Li, L. Organelle-specific photoactivation of DNA nanosensors for precise profiling of subcellular enzymatic activity. Angew Chem Int Ed 2021, 60, 8923–8931. [Google Scholar] [CrossRef]
- Yu, F.; Shao, Y.; Chai, X.; Zhao, Y.; Li, L. Spatially selective monitoring of subcellular enzyme dynamics in response to mitochondria-targeted photodynamic therapy. Angew Chem Int Ed 2022, 61, e202203238. [Google Scholar] [CrossRef]
- Qian, R.; Ding, L.; Ju, H. Switchable fluorescent imaging of intracellular telomerase activity using telomerase-responsive mesoporous silica nanoparticle. J. Am. Chem. Soc. 2013, 135, 13282–13285. [Google Scholar] [CrossRef]
- Torney, F.; Trewyn, B.G.; Lin, V.S.; Wang, K. Mesoporous silica nanoparticles deliver DNA and chemicals into plants. Nat. Nanotechnol. 2007, 2, 295–300. [Google Scholar] [CrossRef]
- He, X.X.; Wang, K.; Tan, W.; Liu, B.; Lin, X.; He, C.; Li, D.; Huang, S.; Li, J. Bioconjugated nanoparticles for DNA protection from cleavage. J. Am. Chem. Soc. 2003, 125, 7168–7169. [Google Scholar] [CrossRef]
- Zhai, J.; Liu, Y.; Huang, S.; Fang, S.; Zhao, M. A specific DNA-nanoprobe for tracking the activities of human apurinic/apyrimidinic endonuclease 1 in living cells. Nucleic Acids Res. 2017, 45, e45. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Feng, X.; Hu, W.; Li, L. An activatable DNA nanodevice for correlated imaging of apoptosis-related dual proteins. Nanoscale 2022, 14, 6465–6470. [Google Scholar] [CrossRef]
- Li, J.J.; Du, W.F.; Liu, Y.N.; Wang, F.; Tang, L.J.; Jiang, J.H. Protein-scaffolded DNA nanostructures for imaging of apurinic/apyrimidinic endonuclease 1 activity in live cells. Anal. Chem. 2023, 95, 3551–3555. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Tian, T.; Zhou, R.; Li, S.; Ma, W.; Zhang, Y.; Liu, N.; Shi, S.; Li, Q.; Xie, X.; Ge, Y.; Liu, M.; Zhang, Q.; Lin, S.; Cai, X.; Lin, Y. Design, fabrication and applications of tetrahedral DNA nanostructure-based multifunctional complexes in drug delivery and biomedical treatment. Nat. Protoc. 2020, 15, 2728–2757. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; He, W.; Du, Z.; Zhu, L.; Huang, K.; Lu, Y.; Luo, Y. Functional nucleic acid nanomaterials: development, properties, and applications. Angew. Chem. Int. Ed. 2021, 60, 6890–6918. [Google Scholar] [CrossRef]
- Mou, Q.; Ma, Y.; Pan, G.; Xue, B.; Yan, D.; Zhang, C.; Zhu, X. DNA trojan horses: self-assembled floxuridine-containing DNA polyhedra for cancer therapy. Angew. Chem. Int. Ed. 2017, 56, 12528–12532. [Google Scholar] [CrossRef]
- Zhang, Y.; Ptacin, J.L.; Fischer, E.C.; Aerni, H.R.; Caffaro, C.E.; Jose, K.S.; Feldman, A.W.; Turner, C.R.; Romesberg, F.E. A semi-synthetic organism that stores and retrieves increased genetic information. Nature 2017, 551, 644–647. [Google Scholar] [CrossRef]
- Tian, T.; Zhang, T.; Shi, S.; Gao, Y.; Cai, X.; Lin, Y. A dynamic DNA tetrahedron framework for active targeting. Nat. Protoc. 2023, 18, 1028–1055. [Google Scholar] [CrossRef]
- Xie, N.; Liu, S.; Fang, H.; Yang, Y.; Quan, K.; Li, J.; Yang, X.; Wang, K.; Huang, J. Three-dimensional molecular transfer from DNA nanocages to inner gold nanoparticle surfaces. ACS Nano 2019, 13, 4174–4182. [Google Scholar] [CrossRef]
- Du, R.R.; Cedrone, E.; Romanov, A.; Falkovich, R.; Dobrovolskaia, M.A.; Bathe, M. Innate immune stimulation using 3D wireframe DNA origami. ACS Nano 2022, 16, 20340–20352. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Zhang, S.; Chen, L.; Li, M.; Zhang, Y.; Zhou, N. Self-assembled DNA nanoflowers triggered by a DNA walker for highly sensitive electrochemical detection of staphylococcus aureus. ACS Appl. Mater. Interfaces 2021, 13, 4905–4914. [Google Scholar] [CrossRef]
- Zhang, L.; Abdullah, R.; Hu, X.; Bai, H.; Fan, H.; He, L.; Liang, H.; Zou, J.; Liu, Y.; Sun, Y.; Zhang, X.; Tan, W. Engineering of bioinspired, size-controllable, self-degradable cancer-targeting DNA nanoflowers via the incorporation of an artificial sandwich base. J. Am. Chem. Soc. 2019, 141, 4282–4290. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.X.; Wang, J.; Wang, Y.X.; Du, Y.C.; Huang, Y.; Tang, A.N.; Cui, Y.X.; Kong, D.M. DNA nanostructure-based nucleic acid probes: construction and biological applications. Chem. Sci. 2021, 12, 7602–7622. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Pei, H.; Ge, Z.; Simmons, C.R.; Yan, H.; Fan, C. Charge transport within a three-dimensional DNA nanostructure framework. J. Am. Chem. Soc. 2012, 134, 13148–13151. [Google Scholar] [CrossRef]
- Liang, M.; Li, N.; Liu, F.; Zeng, N.; Yu, C.; Li, S. Apurinic/apyrimidinic endonuclease triggered doxorubicin-releasing DNA nanoprism for target therapy. Cell Cycle 2022, 21, 2627–2634. [Google Scholar] [CrossRef] [PubMed]
- Goodman, R.P.; Schaap, I.A.T.; Tardin, C.F.; Erben, C.M.; Berry, R.M.; Schmidt, C.F.; Turberfield, A.J. Rapid chiral assembly of rigid DNA building blocks for molecular nanofabrication. Science 2005, 310, 1661–1665. [Google Scholar] [CrossRef]
- Walsh, A.S.; Yin, H.; Erben, C.M.; Wood, M.J.; Turberfield, A.J. DNA cage delivery to mammalian cells. ACS Nano 2011, 5, 5427–5432. [Google Scholar] [CrossRef]
- Liang, L.; Li, J.; Li, Q.; Huang, Q.; Shi, J.; Yan, H.; Fan, C. Single-particle tracking and modulation of cell entry pathways of a tetrahedral DNA nanostructure in live cells. Angew. Chem. Int. Ed. 2014, 53, 7745–7750. [Google Scholar] [CrossRef]
- Keum, J.W.; Bermudez, H. Enhanced resistance of DNA nanostructures to enzymatic digestion. Chem. Commun. 2009, 7036–7038. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, G.; Wang, T.; Fu, J.; Li, R.; Song, L.; Wang, Z.G.; Ding, B.; Chen, H. Enhanced stability of DNA nanostructures by incorporation of unnatural base pairs. Chemphyschem 2017, 18, 2977–2980. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, L.; Qin, Z.; Karges, J.; Xiao, H.; Su, X. Unraveling and overcoming platinum drug-resistant cancer tumors with DNA nanostructures. Advanced Functional Materials 2022, 33, 2208797. [Google Scholar] [CrossRef]
- Kou, Q.; Wang, L.; Zhang, L.; Ma, L.; Fu, S.; Su, X. Simulation-assisted localized DNA logical circuits for cancer biomarkers detection and imaging. Small 2022, 18, e2205191. [Google Scholar] [CrossRef]
- Tang, Y.; Lu, B.; Zhu, Z.; Li, B. Establishment of a universal and rational gene detection strategy through three-way junction-based remote transduction. Chem. Sci. 2018, 9, 760–769. [Google Scholar] [CrossRef]
- Dorjsuren, D.; Kim, D.; Vyjayanti, V.N.; Maloney, D.J.; Jadhav, A.; Wilson III, D.M.; Simeonov, A. Diverse small molecule inhibitors of human apurinic/apyrimidinic endonuclease APE1 identified from a screen of a large public collection. PLoS One 2012, 7, e47974. [Google Scholar] [CrossRef]
- Zhang, Y.; Deng, Y.; Wang, C.; Li, L.; Xu, L.; Yu, Y.; Su, X. Probing and regulating the activity of cellular enzymes by using DNA tetrahedron nanostructures. Chem. Sci. 2019, 10, 5959–5966. [Google Scholar] [CrossRef]
- Lv, M.M.; Liu, J.W.; Yu, R.Q.; Jiang, J.H. A bipedal DNA nanowalker fueled by catalytic assembly for imaging of base-excision repairing in living cells. Chem. Sci. 2020, 11, 10361–10366. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Luo, R.; Li, Y.; Fan, J.; Hu, Y.; Tong, C.; Liu, B.; Li, D. Activity assay and intracellular imaging of APE1 assisted with tetrahedral DNA nanostructure modified-dnazyme and molecular beacon. Sensors and Actuators B: Chemical 2020, 317, 128203. [Google Scholar] [CrossRef]
- Zhou, X.M.; Zhuo, Y.; Tu, T.T.; Yuan, R.; Chai, Y.Q. Construction of fast-walking tetrahedral DNA walker with four arms for sensitive detection and intracellular imaging of apurinic/apyrimidinic endonuclease 1. Anal. Chem. 2022, 94, 8732–8739. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, L.; Sun, H.; Xu, S.; Li, K.; Su, X. Screening and application of inhibitory aptamers for DNA repair protein apurinic/apyrimidinic endonuclease 1. Int. J. Biol. Macromol. 2023, 242, 124918. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Liu, Y.; Zhang, L.; Liu, J.; Su, X. Programming dissipation systems by DNA timer for temporally regulating enzyme catalysis and nanostructure assembly. ACS Nano 2022, 16, 14274–14283. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).