Submitted:
29 June 2023
Posted:
30 June 2023
You are already at the latest version
Abstract
Keywords:
1. Endocrine disrupting chemicals (EDCs)
1.1. Phytoestrogens (PEs)
1.1.1. Signaling via nuclear receptors
1.1.2. GPER signaling
1.1.3. Signaling not mediated by ERs – a significant source of differences in bioactivity between E2 and PEs
1.2. Phytoestrogens (PEs) as Endocrine Disrupting Chemicals (EDCs)
1.2.1. Endocrine disruption and altered immune function
2. Endometriosis
2.1. General characteristics of the disease
2.2. Disruption in estrogen and P4 signaling
2.2.1. Estrogen dominance
2.2.1.1. Aromatase activity
2.2.2. The importance of epigenetic factors
2.2.2.1. Epigenetic modulation of ERs in endometriosis
2.3. Estrogen-dependent immune system interactions in endometriosis
2.3.1. Estrogen and mast cells (MCs) in endometriotic lesions
3. Dietary PEs and endometriosis
3.1. PE intake and the risk of endometriosis – interactions at the level of gut microbiota
3.2. PE oral intake and the course of endometriosis – the results in animal models
3.3. PE oral intake and the course of endometriosis – the results obtained in human studies
4. Concluding remarks
Abbreviations
| 17β-hydroxysteroid dehydrogenases type 1 and 4 (, respectively), 17β-HSD1, 17β-HSD2, 17β-HSD4 – 17β-hydroxysteroid dehydrogenase type 1, 2, and 4, respectively AKT – protein kinase B AP-1 – activator protein 1 ATP – adenosine triphosphate Bcl-2 – anti-apoptotic B-cell lymphoma-2 protein c-IAP1, c-IAP2 – cellular inhibitors of apoptosis 1 and 2, respectively CADD – computer aided drug design cAMP – cyclic adenosine monophosphate CINC-1, CINC-2, CINC-3 – cytokine-induced neutrophil chemoattractant proteins 1-3 COX-2 – cyclooxygenase 2 c-Src/ERK pathway – Src/extracellular signal-regulated kinase pathway CTLs – cytotoxic T lymphocytes, also known as killer T cells DBD – DNA binding domain (or C domain) DCs – dendritic cells DDT – dichlorodiphenyltrichloroethane E1, E2, E3 – estrone, estradiol and estriol, respectively estradiol and estriol, respectively E2 – estradiol EDCs – endocrine disrupting chemicals EGCG – polyphenol epigallocatechin-3-gallate EGFR – epidermal growth factor receptor EMT – epithelial-mesenchymal-transition EnSCs – endometrial stromal cells ERα, ERβ – estrogen receptors α and β, also known as NR3A1 and NR3A2, respectively ERE – estrogen response element ERK1, ERK2 – mitogen-activated protein-serine/threonine kinases ERs – estrogen receptors ESR1, ESR2 – genes encoding estrogen receptors ERα and ERβ, respectively FSH – follicle-stimulating hormone GDNF – glial cell line derived neurotrophic factor GFRα1 – glial cell line derived neurotrophic factor (GDNF) family receptor alpha 1 GM-CSF – granulocyte-macrophage colony stimulating factor GPCRs – G protein-coupled receptors GPER – G protein-coupled estrogen receptor, also known as G protein-coupled receptor 30 (GPR30) HB-EGF – heparin-binding epidermal growth factor (EGF)-like growth factor HDACs – histone deacetylases HLA-G – human leukocyte antigen G HLA-DR – major histocompatibility complex (MHC) II cell surface receptor HO-1 – heme oxygenase-1 HPG axis – hypothalamic-pituitary-gonadal axis HPO axis – hypothalamic-pituitary-ovarian axis HSP90 – heat shock protein 90 HUVECs – human umbilical vein endothelial cells IBD – inflammatory bowel disease IGFR1 – insulin-like growth factor receptor 1 IL-1, IL-1ß, IL-2, IL-3, IL-4, IL-5, IL-6, IL-9, IL-10, IL-12, IL-13, IL-18 – interleukins: 1, 1ß, 2, 3, 4, 5, 6, 9, 10, 12, 13, and 18 IL-18Rα – interleukin 18 receptor alpha IFN-γ – interferon gamma IKK – IκB kinase iNOS – inducible nitric oxide synthase JNK – cJun NH(2)-terminal kinase KEAP1 – Kelch-like ECH associated protein 1 LBD – ligand binding domain LH – luteinizing hormone lncRNAs – long non-coding RNAs MAP – mitogen-activated protein MAPK – mitogen-activated protein kinase MCP-1 – monocyte chemoattractant protein-1 MCs – mast cells MHCI – major histocompatibility complex class I MHCII – major histocompatibility complex class II MMP-2, MMP-9 – matrix metalloproteinases 2 and 9 MMPs – matrix metalloproteinases MNAR – modulator of non-genomic activity of estrogen receptor, also known as proline-, glutamate- and leucine-rich protein 1 (PELP1) mPRα, mPRβ, mPRγ, mPRδ, mPRε – membrane progesterone receptors MSCs – mesenchymal stem cells MW – molecular weight mTOR – mammalian target of rapamycin (a serine-threonine protein kinase) NADPH – nicotinamide adenine dinucleotide phosphate NF-κB – nuclear factor kappa-light-chain-enhancer of activated B cells NK-cell – natural killer cell NO – nitric oxide NQO1 – nicotinamide adenine dinucleotide phosphate (NADPH)-quinone oxidoreductase-1 Nrf2 – factor erythroid 2-related factor 2 NTD – N-terminal domain OT - oxytocin P4 – progesterone P450AROM – aromatase cytochrome P450 PAK1 – p21-activated kinase 1 PCB – polychlorinated biphenyls PCDD – polychlorinated dibenzo-p-dioxins PCDF – polychlorinated dibenzofurans PCNA – proliferating cell nuclear antigen PEA – palmitoylethanolamide PELP1 – proline-, glutamate- and leucine-rich protein 1, also known as modulator of non-genomic activity of estrogen receptor (MNAR) PEs – phytoestrogens PGE2 – prostaglandin E2 PGF2-ɑ – prostaglandin F2-alpha PI3K – phosphatidylinositol-3-kinase PLD – polydatin (natural precursor of resveratrol) PlGF – placental growth factor POPs – persistent organic pollutants PR-A, PR-B – progesterone receptors type A and B, respectively RNA Pol II – RNA polymerase II ROS – reactive oxygen species RTKs – receptor tyrosine kinases SCF – stem cell factor SERMS – selective estrogen receptor (ER) modulators SF-1 – steroidogenic factor 1 SIRTs – sirtuins SRA – steroid receptor RNA activator Src – non-receptor tyrosine kinase (proto-oncogene tyrosine-protein kinase Src) SRC – steroid receptor coactivator SRC-2 – steroid receptor coactivator-2, also known as transcriptional mediators/intermediary factor 2 (TIF2) T3, T4 – triiodothyronine, thyroxine (tetraiodothyronine) TAK1 – transforming growth factor β-activated kinase 1 TIF2 – transcriptional mediators/intermediary factor 2, also known as (SRC-2) TF – transcription factor Th1, Th2, Th17 cells – T helper cell subtypes Tregs – regulatory T cells TNF-α – tumor necrosis factor alpha TPO – thyroid peroxidase VCAM-1 – vascular cell adhesion molecule 1, also known as vascular cell adhesion protein 1 VEGF – vascular endothelial growth factor |
References
- Milling S. Beyond cytokines: Influences of the endocrine system on human immune homeostasis. Immunology. 2021;163(2):113-114. [CrossRef]
- Verburg-van Kemenade BML, Cohen N, Chadzinska M. Neuroendocrine-immune interaction: Evolutionarily conserved mechanisms that maintain allostasis in an ever-changing environment. Dev Comp Immunol. 2017;66:2-23. [CrossRef]
- Tanida T. Molecular dynamics of estrogen-related receptors and their regulatory proteins: roles in transcriptional control for endocrine and metabolic signaling. Anat Sci Int. 2022; 97(1):15-29. [CrossRef]
- Baumbach JL, Zovkic IB. Hormone-epigenome interactions in behavioural regulation. Horm Behav. 2020;118:104680. [CrossRef]
- Ruzzin J. Public health concern behind the exposure to persistent organic pollutants and the risk of metabolic diseases. BMC Public Health. 2012;12:298. [CrossRef]
- Li QQ, Loganath A, Chong YS, Tan J, Obbard JP. Persistent organic pollutants and adverse health effects in humans. J Toxicol Environ Health A. 2006;69(21):1987-2005. [CrossRef]
- Xie Z, Zhang P, Wu Z, Zhang S, Wei L, Mi L, Kuester A, Gandrass J, Ebinghaus R, Yang R, Wang Z, Mi W. Legacy and emerging organic contaminants in the polar regions. Sci Total Environ. 2022;835:155376. [CrossRef]
- Jayaraj R, Megha P, Sreedev P. Organochlorine pesticides, their toxic effects on living organisms and their fate in the environment. Interdiscip Toxicol. 2016;9(3-4):90-100. [CrossRef]
- Guo W, Pan B, Sakkiah S, Yavas G, Ge W, Zou W, Tong W, Hong H. Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods. Int J Environ Res Public Health. 2019;16(22):4361. [CrossRef]
- Organochlorine Pollutants in Plasma, Blood Pressure, and Hypertension in a Longitudinal Study. Hypertension. 2018;71(6):1258-1268. [CrossRef]
- Wilson J, Berntsen HF, Zimmer KE, Verhaegen S, Frizzell C, Ropstad E, Connolly L. Do persistent organic pollutants interact with the stress response? Individual compounds, and their mixtures, interaction with the glucocorticoid receptor. Toxicol Lett. 2016;241:121-32. [CrossRef]
- La Merrill MA, Vandenberg LN, Smith MT, Goodson W, Browne P, Patisaul HB, Guyton KZ, Kortenkamp A, Cogliano VJ, Woodruff TJ, Rieswijk L, Sone H, Korach KS, Gore AC, Zeise L, Zoeller RT. Consensus on the key characteristics of endocrine-disrupting chemicals as a basis for hazard identification. Nat Rev Endocrinol. 2020;16(1):45-57. [CrossRef]
- Yilmaz B, Terekeci H, Sandal S, Kelestimur F. Endocrine disrupting chemicals: exposure, effects on human health, mechanism of action, models for testing and strategies for prevention. Rev Endocr Metab Disord. 2020;21(1):127-147. [CrossRef]
- Gregoraszczuk EL, Ptak A. Endocrine-Disrupting Chemicals: Some Actions of POPs on Female Reproduction. Int J Endocrinol. 2013;2013:828532. [CrossRef]
- Kowalczyk A, Wrzecińska M, Czerniawska-Piątkowska E, Araújo JP, Cwynar P. Molecular consequences of the exposure to toxic substances for the endocrine system of females. Biomed Pharmacother. 2022;155:113730. [CrossRef]
- Thambirajah AA, Wade MG, Verreault J, Buisine N, Alves VA, Langlois VS, Helbing CC. Disruption by stealth - Interference of endocrine disrupting chemicals on hormonal crosstalk with thyroid axis function in humans and other animals. Environ Res. 2022 ;203: 111906. [CrossRef]
- Buoso E, Masi M, Racchi M, Corsini E. Endocrine-Disrupting Chemicals' (EDCs) Effects on Tumour Microenvironment and Cancer Progression: Emerging Contribution of RACK1. Int J Mol Sci. 2020;21(23):9229. [CrossRef]
- Masuo Y, Ishido M. Neurotoxicity of endocrine disruptors: possible involvement in brain development and neurodegeneration. J Toxicol Environ Health B Crit Rev. 2011;14(5-7):346-69. [CrossRef]
- Rahman MS, Pang WK, Amjad S, Ryu DY, Adegoke EO, Park YJ, Pang MG. Hepatic consequences of a mixture of endocrine-disrupting chemicals in male mice. J Hazard Mater. 2022;436:129236. [CrossRef]
- Singh RD, Koshta K, Tiwari R, Khan H, Sharma V, Srivastava V. Developmental Exposure to Endocrine Disrupting Chemicals and Its Impact on Cardio-Metabolic-Renal Health. Front Toxicol. 2021;3:663372. [CrossRef]
- Sabuz Vidal O, Deepika D, Schuhmacher M, Kumar V. EDC-induced mechanisms of immunotoxicity: a systematic review. Crit Rev Toxicol. 2021;51(7):634-652. [CrossRef]
- Meeker JD. Exposure to environmental endocrine disruptors and child development. Arch Pediatr Adolesc Med. 2012;166(6):E1-7. [CrossRef]
- Lakshmanan MD, Shaheer K. Endocrine disrupting chemicals may deregulate DNA repair through estrogen receptor mediated seizing of CBP/p300 acetylase. J Endocrinol Invest. 2020; 43(9):1189-1196. [CrossRef]
- Zoeller RT, Brown TR, Doan LL, Gore AC, Skakkebaek NE, Soto AM, Woodruff TJ, Vom Saal FS. Endocrine-disrupting chemicals and public health protection: a statement of principles from The Endocrine Society. Endocrinology. 2012;153(9):4097-110. [CrossRef]
- Lisco G, Giagulli VA, Iovino M, Guastamacchia E, Pergola G, Triggiani V. Endocrine-Disrupting Chemicals: Introduction to the Theme. Endocr Metab Immune Disord Drug Targets. 2022;22(7):677-685. [CrossRef]
- Marty MS, Borgert C, Coady K, Green R, Levine SL, Mihaich E, Ortego L, Wheeler JR, Yi KD, Zorrilla LM. Distinguishing between endocrine disruption and non-specific effects on endocrine systems. Regul Toxicol Pharmacol. 2018;99:142-158. [CrossRef]
- Keller DA, Juberg DR, Catlin N, Farland WH, Hess FG, Wolf DC, Doerrer NG. Identification and characterization of adverse effects in 21st century toxicology. Toxicol Sci. 2012;126(2):291-7. [CrossRef]
- McKone TE, Daniels JI. Estimating human exposure through multiple pathways from air, water, and soil. Regul Toxicol Pharmacol. 1991;13(1):36-61. [CrossRef]
- Anwer F, Chaurasia S, Khan AA. Hormonally active agents in the environment: a state-of-the-art review. Rev Environ Health. 2016;31(4):415-433. [CrossRef]
- Yue B, Ning S, Miao H, Fang C, Li J, Zhang L, Bao Y, Fan S, Zhao Y, Wu Y. Human exposure to a mixture of endocrine disruptors and serum levels of thyroid hormones: A cross-sectional study. J Environ Sci (China). 2023;125:641-649. [CrossRef]
- Darbre PD, Harvey PW. Paraben esters: review of recent studies of endocrine toxicity, absorption, esterase and human exposure, and discussion of potential human health risks. J Appl Toxicol. 2008;28(5):561-78. [CrossRef]
- He D, Ye X, Xiao Y, Zhao N, Long J, Zhang P, Fan Y, Ding S, Jin X, Tian C, Xu S, Ying C. Dietary exposure to endocrine disrupting chemicals in metropolitan population from China: a risk assessment based on probabilistic approach. Chemosphere. 2015;139:2-8. [CrossRef]
- Chen Y, Yang J, Yao B, Zhi D, Luo L, Zhou Y. Endocrine disrupting chemicals in the environment: Environmental sources, biological effects, remediation techniques, and perspective. Environ Pollut. 2022;310:119918. [CrossRef]
- Sakali AK, Bargiota A, Fatouros IG, Jamurtas A, Macut D, Mastorakos G, Papagianni M. Effects on Puberty of Nutrition-Mediated Endocrine Disruptors Employed in Agriculture. Nutrients. 2021;13(11):4184. [CrossRef]
- Beszterda M, Frański R. Endocrine disruptor compounds in environment: As a danger for children health. Pediatr Endocrinol Diabetes Metab. 2018;24(2):88-95. [CrossRef]
- Li N, Li J, Zhang Q, Gao S, Quan X, Liu P, Xu C. Effects of endocrine disrupting chemicals in host health: Three-way interactions between environmental exposure, host phenotypic responses, and gut microbiota. Environ Pollut. 2021;271:116387. [CrossRef]
- Shore LS, Shemesh M. Estrogen as an Environmental Pollutant. Bull Environ Contam Toxicol. 2016;97(4):447-8. [CrossRef]
- Mheidli N, Malli A, Mansour F, Al-Hindi M. Occurrence and risk assessment of pharmaceuticals in surface waters of the Middle East and North Africa: A review. Sci Total Environ. 2022;851(Pt 2):158302. [CrossRef]
- Atkinson S, Atkinson MJ, Tarrant AM. Estrogens from sewage in coastal marine environments. Environ Health Perspect. 2003;111(4):531-5. [CrossRef]
- Adeel M, Song X, Wang Y, Francis D, Yang Y. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ Int. 2017;99:107-119. [CrossRef]
- Shore LS, Bar-El CK. The environmental compartments of environmental hormones. Rev Environ Health. 2010;25(4):345-50. [CrossRef]
- Varticovski L, Stavreva DA, McGowan A, Raziuddin R, Hager GL. Endocrine disruptors of sex hormone activities. Mol Cell Endocrinol. 2022;539:111415. [CrossRef]
- Thompson LU, Robb P, Serraino M, Cheung F. Mammalian lignan production from various foods. Nutr Cancer. 1991;16(1):43-52. [CrossRef]
- Murkies AL, Wilcox G, Davis SR. Clinical review 92: Phytoestrogens. J Clin Endocrinol Metab. 1998;83(2):297-303. [CrossRef]
- Bennetts HW, Underwood EJ, Shier FL. A specific breeding problem of sheep on subterranean clover pastures in Western Australia. Aust Vet J. 1946;22(1):2-12. [CrossRef]
- Rietjens IMCM, Louisse J, Beekmann K. The potential health effects of dietary phytoestrogens. Br J Pharmacol. 2017;174(11):1263-1280. [CrossRef]
- Lecomte S, Demay F, Ferrière F, Pakdel F. Phytochemicals Targeting Estrogen Receptors: Beneficial Rather Than Adverse Effects? Int J Mol Sci. 2017;18(7):1381. [CrossRef]
- Bar-El DS, Reifen R. Soy as an endocrine disruptor: cause for caution? J Pediatr Endocrinol Metab. 2010;23(9):855-61. [CrossRef]
- Beekmann K. The potential health effects of dietary phytoestrogens. Br J Pharmacol. 2017; 174(11):1263-1280. [CrossRef]
- van Duursen MBM. Modulation of estrogen synthesis and metabolism by phytoestrogens in vitro and the implications for women's health. Toxicol Res (Camb). 2017;6(6):772-794. [CrossRef]
- Gaya P, Medina M, Sánchez-Jiménez A, Landete JM. Phytoestrogen Metabolism by Adult Human Gut Microbiota. Molecules. 2016;21(8):1034. [CrossRef]
- Kuiper GG, Lemmen JG, Carlsson B, Corton JC, Safe SH, van der Saag PT, van der Burg B, Gustafsson JA. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology. 1998;139(10):4252-63. [CrossRef]
- Kiyama R. Estrogenic flavonoids and their molecular mechanisms of action. J Nutr Biochem. 2022;114:109250. [CrossRef]
- Han DH, Denison MS, Tachibana H, Yamada K. Relationship between estrogen receptor-binding and estrogenic activities of environmental estrogens and suppression by flavonoids. Biosci Biotechnol Biochem. 2002;66(7):1479-87. [CrossRef]
- Rietjens IM, Sotoca AM, Vervoort J, Louisse J. Mechanisms underlying the dualistic mode of action of major soy isoflavones in relation to cell proliferation and cancer risks. Mol Nutr Food Res. 2013;57(1):100-13. [CrossRef]
- Paterni I, Granchi C, Katzenellenbogen JA, Minutolo F. Estrogen receptors alpha (ERα) and beta (ERβ): subtype-selective ligands and clinical potential. Steroids. 2014;90:13-29. [CrossRef]
- Thomas P, Dong J. Binding and activation of the seven-transmembrane estrogen receptor GPR30 by environmental estrogens: a potential novel mechanism of endocrine disruption. J Steroid Biochem Mol Biol. 2006;102(1-5):175-9. [CrossRef]
- Liu EYL, Xu ML, Jin Y, Wu Q, Dong TTX, Tsim KWK. Genistein, a Phytoestrogen in Soybean, Induces the Expression of Acetylcholinesterase via G Protein-Coupled Receptor 30 in PC12 Cells. Front Mol Neurosci. 2018;11:59. [CrossRef]
- Sundermann EE, Maki PM, Bishop JR. A review of estrogen receptor alpha gene (ESR1) polymorphisms, mood, and cognition. Menopause. 2010;17(4):874-86. [CrossRef]
- Li J, Liu Q, Jiang C. Signal Crosstalk and the Role of Estrogen Receptor beta (ERβ) in Prostate Cancer. Med Sci Monit. 2022;28:e935599. [CrossRef]
- Arao Y, Korach KS. The physiological role of estrogen receptor functional domains. Essays Biochem. 2021;65(6):867-875. [CrossRef]
- Fuentes N, Silveyra P. Estrogen receptor signaling mechanisms. Adv Protein Chem Struct Biol. 2019;116:135-170. [CrossRef]
- Yaşar P, Ayaz G, User SD, Güpür G, Muyan M. Molecular mechanism of estrogen-estrogen receptor signaling. Reprod Med Biol. 2016;16(1):4-20. [CrossRef]
- George AJ, Hannan RD, Thomas WG. Unravelling the molecular complexity of GPCR-mediated EGFR transactivation using functional genomics approaches. FEBS J. 2013; 280(21):5258-68. [CrossRef]
- Yu X, Stallone JN, Heaps CL, Han G. The activation of G protein-coupled estrogen receptor induces relaxation via cAMP as well as potentiates contraction via EGFR transactivation in porcine coronary arteries. PLoS One. 2018;13(1):e0191418. [CrossRef]
- Kato S, Endoh H, Masuhiro Y, Kitamoto T, Uchiyama S, Sasaki H, Masushige S, Gotoh Y, Nishida E, Kawashima H, Metzger D, Chambon P. Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase. Science. 1995;270(5241):1491-4. [CrossRef]
- Coleman KM, Smith CL. Intracellular signaling pathways: nongenomic actions of estrogens and ligand-independent activation of estrogen receptors. Front Biosci. 2001;6: D1379-91. [CrossRef]
- Atanaskova N, Keshamouni VG, Krueger JS, Schwartz JA, Miller F, Reddy KB. MAP kinase/estrogen receptor cross-talk enhances estrogen-mediated signaling and tumor growth but does not confer tamoxifen resistance. Oncogene. 2002;21(25):4000-8. [CrossRef]
- Visser K, Mortimer M, Louw A. Cyclopia extracts act as ERα antagonists and ERβ agonists, in vitro and in vivo. PLoS One. 2013;8(11):e79223. Erratum in: PLoS One. 2013;8(12). [CrossRef]
- Vitale DC, Piazza C, Melilli B, Drago F, Salomone S. Isoflavones: estrogenic activity, biological effect and bioavailability. Eur J Drug Metab Pharmacokinet. 2013;38(1):15-25. [CrossRef]
- Messina M, Kucuk O, Lampe JW. An overview of the health effects of isoflavones with an emphasis on prostate cancer risk and prostate-specific antigen levels. J AOAC Int. 2006; 89(4):1121-34.
- Křížová L, Dadáková K, Kašparovská J, Kašparovský T. Isoflavones. Molecules. 2019; 24(6):1076. [CrossRef]
- Bartuzi D, Kaczor AA, Targowska-Duda KM, Matosiuk D. Recent Advances and Applications of Molecular Docking to G Protein-Coupled Receptors. Molecules. 2017;22(2): 340. [CrossRef]
- Meng XY, Zhang HX, Mezei M, Cui M. Molecular docking: a powerful approach for structure-based drug discovery. Curr Comput Aided Drug Des. 2011;7(2):146-57. [CrossRef]
- Fukunishi Y, Yamashita Y, Mashimo T, Nakamura H. Prediction of Protein-compound Binding Energies from Known Activity Data: Docking-score-based Method and its Applications. Mol Inform. 2018;37(6-7):e1700120. [CrossRef]
- Sulimov VB, Kutov DC, Sulimov AV. Advances in Docking. Curr Med Chem. 2019;26(42):7555-7580. [CrossRef]
- Sahayarayan JJ, Rajan KS, Vidhyavathi R, Nachiappan M, Prabhu D, Alfarraj S, Arokiyaraj S, Daniel AN. In-silico protein-ligand docking studies against the estrogen protein of breast cancer using pharmacophore based virtual screening approaches. Saudi J Biol Sci. 2021;28(1):400-407. [CrossRef]
- Powers CN, Setzer WN. A molecular docking study of phytochemical estrogen mimics from dietary herbal supplements. In Silico Pharmacol. 2015;3:4. [CrossRef]
- Ramírez D, Caballero J. Is It Reliable to Use Common Molecular Docking Methods for Comparing the Binding Affinities of Enantiomer Pairs for Their Protein Target? Int J Mol Sci. 2016;17(4):525. [CrossRef]
- Cassidy A, Bingham S, Setchell KD. Biological effects of a diet of soy protein rich in isoflavones on the menstrual cycle of premenopausal women. Am J Clin Nutr. 1994;60333- 340.
- Jang WY, Kim MY, Cho JY. Antioxidant, Anti-Inflammatory, Anti-Menopausal, and Anti-Cancer Effects of Lignans and Their Metabolites. Int J Mol Sci. 2022;23(24):15482. [CrossRef]
- Liu Y, Hassan S, Kidd BN, Garg G, Mathesius U, Singh KB, Anderson JP. Ethylene Signaling Is Important for Isoflavonoid-Mediated Resistance to Rhizoctonia solani in Roots of Medicago truncatula. Mol Plant Microbe Interact. 2017;30(9):691-700. [CrossRef]
- Setchell KD. Phytoestrogens: the biochemistry, physiology, and implications for human health of soy isoflavones. Am J Clin Nutr. 1998;68(6 Suppl):1333S-1346S. [CrossRef]
- Zhu Y, Kawaguchi K, Kiyama R. Differential and directional estrogenic signaling pathways induced by enterolignans and their precursors. PLoS One. 2017;12(2):e0171390. [CrossRef]
- JavanMoghadam S, Weihua Z, Hunt KK, Keyomarsi K. Estrogen receptor alpha is cell cycle-regulated and regulates the cell cycle in a ligand-dependent fashion. Cell Cycle. 2016;15(12):1579-90. [CrossRef]
- Livezey M, Kim JE, Shapiro DJ. A New Role for Estrogen Receptor α in Cell Proliferation and Cancer: Activating the Anticipatory Unfolded Protein Response. Front Endocrinol (Lausanne). 2018;9:325. [CrossRef]
- Thomas C, Gustafsson JÅ. The different roles of ER subtypes in cancer biology and therapy. Nat Rev Cancer. 2011;11(8):597-608. [CrossRef]
- Yang ZM, Yang MF, Yu W, Tao HM. Molecular mechanisms of estrogen receptor β-induced apoptosis and autophagy in tumors: implication for treating osteosarcoma. J Int Med Res. 2019;47(10):4644-4655. [CrossRef]
- Zava DT, Dollbaum CM, Blen M. Estrogen and progestin bioactivity of foods, herbs, and spices. Proc Soc Exp Biol Med. 1998;217(3):369-78. [CrossRef]
- Gruber CJ, Tschugguel W, Schneeberger C, Huber JC. Production and actions of estrogens. N Engl J Med. 2002;346(5):340-52. [CrossRef]
- Cassidy A. Potential tissue selectivity of dietary phytoestrogens and estrogens. Curr Opin Lipidol. 1999;10(1):47-52. [CrossRef]
- Patel S, Homaei A, Raju AB, Meher BR. Estrogen: The necessary evil for human health, and ways to tame it. Biomed Pharmacother. 2018;102:403-411. [CrossRef]
- Manavathi B, Samanthapudi VS, Gajulapalli VN. Estrogen receptor coregulators and pioneer factors: the orchestrators of mammary gland cell fate and development. Front Cell Dev Biol. 2014;2:34. [CrossRef]
- Zhou Y, Liu X. The role of estrogen receptor beta in breast cancer. Biomark Res. 2020; 8: 39. [CrossRef]
- Bhat SS, Prasad SK, Shivamallu C, Prasad KS, Syed A, Reddy P, Cull CA, Amachawadi RG. Genistein: A Potent Anti-Breast Cancer Agent. Curr Issues Mol Biol. 2021;43(3):1502-1517. [CrossRef]
- Kushner PJ, Agard DA, Greene GL, Scanlan TS, Shiau AK, Uht RM, Webb P. Estrogen receptor pathways to AP-1. J Steroid Biochem Mol Biol. 2000;74(5):311-7. [CrossRef]
- Prossnitz ER, Barton M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat Rev Endocrinol. 2011;7(12):715-26. [CrossRef]
- Puglisi R, Mattia G, Carè A, Marano G, Malorni W, Matarrese P. Non-genomic Effects of Estrogen on Cell Homeostasis and Remodeling With Special Focus on Cardiac Ischemia/Reperfusion Injury. Front Endocrinol (Lausanne). 2019;10:733. [CrossRef]
- Yu K, Huang ZY, Xu XL, Li J, Fu XW, Deng SL. Estrogen Receptor Function: Impact on the Human Endometrium. Front Endocrinol (Lausanne). 2022;13:827724. [CrossRef]
- Filardo EJ, Quinn JA, Frackelton AR Jr, Bland KI. Estrogen action via the G protein-coupled receptor, GPR30: stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol Endocrinol. 2002;16(1):70-84. [CrossRef]
- Albanito L, Sisci D, Aquila S, Brunelli E, Vivacqua A, Madeo A, Lappano R, Pandey DP, Picard D, Mauro L, Andò S, Maggiolini M. Epidermal growth factor induces G protein-coupled receptor 30 expression in estrogen receptor-negative breast cancer cells. Endocrinology. 2008;149(8):3799-808. [CrossRef]
- Pupo M, Maggiolini M, Musti AM. GPER Mediates Non-Genomic Effects of Estrogen. Methods Mol Biol. 2016;1366:471-488. [CrossRef]
- Barton M, Filardo EJ, Lolait SJ, Thomas P, Maggiolini M, Prossnitz ER. Twenty years of the G protein-coupled estrogen receptor GPER: Historical and personal perspectives. J Steroid Biochem Mol Biol. 2018;176:4-15. [CrossRef]
- Filardo EJ, Quinn JA, Bland KI, Frackelton AR Jr. Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol Endocrinol. 2000;14(10):1649-60. [CrossRef]
- Prenzel N, Zwick E, Daub H, Leserer M, Abraham R, Wallasch C, Ullrich A. EGF receptor transactivation by G-protein-coupled receptors requires metalloproteinase cleavage of proHB-EGF. Nature. 1999;402(6764):884-8. [CrossRef]
- Edwin F, Wiepz GJ, Singh R, Peet CR, Chaturvedi D, Bertics PJ, Patel TB. A historical perspective of the EGF receptor and related systems. Methods Mol Biol. 2006;327:1-24. [CrossRef]
- Fan DX, Yang XH, Li YN, Guo L. 17β-Estradiol on the Expression of G-Protein Coupled Estrogen Receptor (GPER/GPR30) Mitophagy, and the PI3K/Akt Signaling Pathway in ATDC5 Chondrocytes In Vitro. Med Sci Monit. 2018;24:1936-1947. [CrossRef]
- Prossnitz ER, Maggiolini M. Mechanisms of estrogen signaling and gene expression via GPR30. Mol Cell Endocrinol. 2009;308(1-2):32-8. [CrossRef]
- Xu S, Yu S, Dong D, Lee LTO. G Protein-Coupled Estrogen Receptor: A Potential Therapeutic Target in Cancer. Front Endocrinol (Lausanne). 2019;10:725. [CrossRef]
- Maggiolini M, Vivacqua A, Fasanella G, Recchia AG, Sisci D, Pezzi V, Montanaro D, Musti AM, Picard D, Andò S. The G protein-coupled receptor GPR30 mediates c-fos up-regulation by 17beta-estradiol and phytoestrogens in breast cancer cells. J Biol Chem. 2004;279(26): 27008-16. [CrossRef]
- Luo J, Liu D. Does GPER Really Function as a G Protein-Coupled Estrogen Receptor in vivo? Front Endocrinol (Lausanne). 2020;11:148. [CrossRef]
- Roque C, Baltazar G. G protein-coupled estrogen receptor 1 (GPER) activation triggers different signaling pathways on neurons and astrocytes. Neural Regen Res. 2019;14(12): 2069-2070. [CrossRef]
- Conn PM, Ulloa-Aguirre A. Trafficking of G-protein-coupled receptors to the plasma membrane: insights for pharmacoperone drugs. Trends Endocrinol Metab. 2010;21(3):190-7. [CrossRef]
- Acconcia F, Bocedi A, Ascenzi P, Marino M. Does palmitoylation target estrogen receptors to plasma membrane caveolae? IUBMB Life. 2003;55(1):33-5. [CrossRef]
- Mendoza MC, Er EE, Blenis J. The Ras-ERK and PI3K-mTOR pathways: cross-talk and compensation. Trends Biochem Sci. 2011;36(6):320-8. [CrossRef]
- Moriarty K, Kim KH, Bender JR. Minireview: estrogen receptor-mediated rapid signaling. Endocrinology. 2006;147(12):5557-63. [CrossRef]
- Sirotkin AV, Harrath AH. Phytoestrogens and their effects. Eur J Pharmacol. 2014; 741: 230-6. [CrossRef]
- Cederroth CR, Zimmermann C, Nef S. Soy, phytoestrogens and their impact on reproductive health. Mol Cell Endocrinol. 2012;355(2):192-200. [CrossRef]
- Moreira AC, Silva AM, Santos MS, Sardão VA. Phytoestrogens as alternative hormone replacement therapy in menopause: What is real, what is unknown. J Steroid Biochem Mol Biol. 2014;143:61-71. [CrossRef]
- Domańska A, Orzechowski A, Litwiniuk A, Kalisz M, Bik W, Baranowska-Bik A. The Beneficial Role of Natural Endocrine Disruptors: Phytoestrogens in Alzheimer's Disease. Oxid Med Cell Longev. 2021;2021:3961445. [CrossRef]
- Nguyen M, Osipo C. Targeting Breast Cancer Stem Cells Using Naturally Occurring Phytoestrogens. Int J Mol Sci. 2022;23(12):6813. [CrossRef]
- Scherbakov AM, Andreeva OE. Apigenin Inhibits Growth of Breast Cancer Cells: The Role of ERα and HER2/neu. Acta Naturae. 2015;7(3):133-9.
- Petrakis NL, Barnes S, King EB, Lowenstein J, Wiencke J, Lee MM, Miike R, Kirk M, Coward L. Stimulatory influence of soy protein isolate on breast secretion in pre- and postmenopausal women. Cancer Epidemiol Biomarkers Prev. 1996;5(10):785-94.
- Hargreaves DF, Potten CS, Harding C, Shaw LE, Morton MS, Roberts SA, Howell A, Bundred NJ. Two-week dietary soy supplementation has an estrogenic effect on normal premenopausal breast. J Clin Endocrinol Metab. 1999;84(11):4017-24. [CrossRef]
- Lee AW, Poynor V, McEligot AJ. Urinary Phytoestrogen Levels Are Associated with Female Hormonal Cancers: An Analysis of NHANES Data From 1999 to 2010. Nutr Cancer. 2022;74(8):2748-2756. [CrossRef]
- Talaei M, Pan A. Role of phytoestrogens in prevention and management of type 2 diabetes. World J Diabetes. 2015;6(2):271-83. [CrossRef]
- Jefferson WN, Patisaul HB, Williams CJ. Reproductive consequences of developmental phytoestrogen exposure. Reproduction. 2012;143(3):247-60. [CrossRef]
- Swathi Krishna S, Kuriakose BB, Lakshmi PK. Effects of phytoestrogens on reproductive organ health. Arch Pharm Res. 2022;45(12):849-864. [CrossRef]
- Solano F, Hernández E, Juárez-Rojas L, Rojas-Maya S, López G, Romero C, Casillas F, Betancourt M, López A, Heidari R, Ommati MM, Retana-Márquez S. Reproductive disruption in adult female and male rats prenatally exposed to mesquite pod extract or daidzein. Reprod Biol. 2022;22(3):100683. [CrossRef]
- Patisaul HB, Jefferson W. The pros and cons of phytoestrogens. Front Neuroendocrinol. 2010;31(4):400-19. [CrossRef]
- Frankenfeld CL. Cardiometabolic risk and gut microbial phytoestrogen metabolite phenotypes. Mol Nutr Food Res. 2017;61(1). [CrossRef]
- Gore AC, Chappell VA, Fenton SE, Flaws JA, Nadal A, Prins GS, Toppari J, Zoeller RT. EDC-2: The Endocrine Society's Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr Rev. 2015;36(6):E1-E150. [CrossRef]
- Frye CA, Bo E, Calamandrei G, Calzà L, Dessì-Fulgheri F, Fernández M, Fusani L, Kah O, Kajta M, Le Page Y, Patisaul HB, Venerosi A, Wojtowicz AK, Panzica GC. Endocrine disrupters: a review of some sources, effects, and mechanisms of actions on behaviour and neuroendocrine systems. J Neuroendocrinol. 2012;24(1):144-59. [CrossRef]
- Degen GH, Janning P, Diel P, Michna H, Bolt HM. Transplacental transfer of the phytoestrogen daidzein in DA/Han rats. Arch Toxicol. 2002;76(1):23-9. [CrossRef]
- Jarrell J, Foster WG, Kinniburgh DW. Phytoestrogens in human pregnancy. Obstet Gynecol Int. 2012;2012:850313. [CrossRef]
- Patisaul HB, Adewale HB. Long-term effects of environmental endocrine disruptors on reproductive physiology and behavior. Front Behav Neurosci. 2009;3:10. [CrossRef]
- Patisaul HB. Endocrine disruption by dietary phyto-oestrogens: impact on dimorphic sexual systems and behaviours. Proc Nutr Soc. 2017;76(2):130-144. [CrossRef]
- Akiyama T, Ishida J, Nakagawa S, Ogawara H, Watanabe S, Itoh N, Shibuya M, Fukami Y. Genistein, a specific inhibitor of tyrosine-specific protein kinases. J Biol Chem. 1987;262(12):5592-5.
- Harvey PA, Leinwand LA. Dietary phytoestrogens present in soy dramatically increase cardiotoxicity in male mice receiving a chemotherapeutic tyrosine kinase inhibitor. Mol Cell Endocrinol. 2015;399:330-5. [CrossRef]
- Divi RL, Doerge DR. Inhibition of thyroid peroxidase by dietary flavonoids. Chem Res Toxicol. 1996;9(1):16-23. [CrossRef]
- Divi RL, Chang HC, Doerge DR. Anti-thyroid isoflavones from soybean: isolation, characterization, and mechanisms of action. Biochem Pharmacol. 1997;54(10):1087-96. [CrossRef]
- Sathyapalan T, Manuchehri AM, Thatcher NJ, Rigby AS, Chapman T, Kilpatrick ES, Atkin SL. The effect of soy phytoestrogen supplementation on thyroid status and cardiovascular risk markers in patients with subclinical hypothyroidism: a randomized, double-blind, crossover study. J Clin Endocrinol Metab. 2011;96(5):1442-9. [CrossRef]
- Sathyapalan T, Dawson AJ, Rigby AS, Thatcher NJ, Kilpatrick ES, Atkin SL. The Effect of Phytoestrogen on Thyroid in Subclinical Hypothyroidism: Randomized, Double Blind, Crossover Study. Front Endocrinol (Lausanne). 2018;9:531. [CrossRef]
- Dal Forno GO, Oliveira IM, Cavallin MD, Santos TIA, Sleiman HK, Falbo MK, Romano MA, Romano RM. Peripubertal soy isoflavone consumption leads to subclinical hypothyroidism in male Wistar rats. J Dev Orig Health Dis. 2022:1-14. [CrossRef]
- Awobajo FO, Medobi EF, Abdul MW, Aminu BB, Ojimma CT, Dada OG. The effect of genistein on IGF-1, PlGF, sFLT-1 and fetoplacental development. Gen Comp Endocrinol. 2022;329:114122. [CrossRef]
- Picherit C, Dalle M, Néliat G, Lebecque P, Davicco MJ, Barlet JP, Coxam V. Genistein and daidzein modulate in vitro rat uterine contractile activity. J Steroid Biochem Mol Biol. 2000;75(2-3):201-8. [CrossRef]
- Richter DU, Mylonas I, Toth B, Scholz C, Briese V, Friese K, Jeschke U. Effects of phytoestrogens genistein and daidzein on progesterone and estrogen (estradiol) production of human term trophoblast cells in vitro. Gynecol Endocrinol. 2009;25(1):32-8. [CrossRef]
- Badger TM, Ronis MJ, Hakkak R, Rowlands JC, Korourian S. The health consequences of early soy consumption. J Nutr. 2002;132(3):559S-565S. [CrossRef]
- Franke AA, Custer LJ, Tanaka Y. Isoflavones in human breast milk and other biological fluids. Am J Clin Nutr. 1998;68(6 Suppl):1466S-1473S. [CrossRef]
- Setchell KD, Zimmer-Nechemias L, Cai J, Heubi JE. Exposure of infants to phyto-oestrogens from soy-based infant formula. Lancet. 1997;350(9070):23-7. [CrossRef]
- Setchell KD, Zimmer-Nechemias L, Cai J, Heubi JE. Isoflavone content of infant formulas and the metabolic fate of these phytoestrogens in early life. Am J Clin Nutr. 1998; 68(6 Suppl):1453S-1461S. [CrossRef]
- Suk An E, Park D, Ban YH, Jieun C, Seo DW, Bok Lee Y, Shon MY, Choi EK, Kim YB. Effects of a soybean milk product on feto-neonatal development in rats. J Biomed Res. 2017;32(1):51–7. [CrossRef]
- Guerrero-Bosagna CM, Sabat P, Valdovinos FS, Valladares LE, Clark SJ. Epigenetic and phenotypic changes result from a continuous pre and postnatal dietary exposure to phytoestrogens in an experimental population of mice. BMC Physiol. 2008;8:17. [CrossRef]
- Becker M, Hesse V. Minipuberty: Why Does it Happen? Horm Res Paediatr. 2020;93(2):76-84. [CrossRef]
- Suen AA, Kenan AC, Williams CJ. Developmental exposure to phytoestrogens found in soy: New findings and clinical implications. Biochem Pharmacol. 2022;195: 114848. [CrossRef]
- Upson K, Sathyanarayana S, Scholes D, Holt VL. Early-life factors and endometriosis risk. Fertil Steril. 2015;104(4):964-971.e5. [CrossRef]
- Ottolina J, Schimberni M, Makieva S, Bartiromo L, Fazia T, Bernardinelli L, Viganò P, Candiani M, Gentilini D. Early-life factors, in-utero exposures and endometriosis risk: a meta-analysis. Reprod Biomed Online. 2020;41(2):279-289. [CrossRef]
- Gao M, Allebeck P, Mishra GD, Koupil I. Developmental origins of endometriosis: a Swedish cohort study. J Epidemiol Community Health. 2019;73(4):353-359. [CrossRef]
- Krishnamoorthy SP, Kalimuthu V, Chandran Manimegalai S, Arulanandu AM, Thiyagarajan R, Balamuthu K. Evaluation of the potential role of diethylstilbestrol on the induction of endometriosis in a rat model - An alternative approach. Biochem Biophys Res Commun. 2022;617(Pt 2):18-24. [CrossRef]
- Golinski P, Vesonder RF, Latus-Zietkiewicz D, Perkowski J. Formation of fusarenone X, nivalenol, zearalenone, alpha-trans-zearalenol, beta-trans-zearalenol, and fusarin C by Fusarium crookwellense. Appl Environ Microbiol. 1988;54(8):2147-8. [CrossRef]
- Bryła M, Pierzgalski A, Zapaśnik A, Uwineza PA, Ksieniewicz-Woźniak E, Modrzewska M, Waśkiewicz A. Recent Research on Fusarium Mycotoxins in Maize-A Review. Foods. 2022;11(21):3465. [CrossRef]
- Ropejko K, Twarużek M. Zearalenone and Its Metabolites-General Overview, Occurrence, and Toxicity. Toxins (Basel). 2021;13(1):35. [CrossRef]
- Sondergaard TE, Hansen FT, Purup S, Nielsen AK, Bonefeld-Jørgensen EC, Giese H, Sørensen JL. Fusarin C acts like an estrogenic agonist and stimulates breast cancer cells in vitro. Toxicol Lett. 2011;205(2):116-21. [CrossRef]
- Zheng W, Wang B, Li X, Wang T, Zou H, Gu J, Yuan Y, Liu X, Bai J, Bian J, Liu Z. Zearalenone Promotes Cell Proliferation or Causes Cell Death? Toxins (Basel). 2018;10(5):184. [CrossRef]
- Belli P, Bellaton C, Durand J, Balleydier S, Milhau N, Mure M, Mornex JF, Benahmed M, Le Jan C. Fetal and neonatal exposure to the mycotoxin zearalenone induces phenotypic alterations in adult rat mammary gland. Food Chem Toxicol. 2010;48(10):2818-26. [CrossRef]
- Jing S, Liu C, Zheng J, Dong Z, Guo N. Toxicity of zearalenone and its nutritional intervention by natural products. Food Funct. 2022;13(20):10374-10400. [CrossRef]
- Kinkade CW, Rivera-Núñez Z, Gorcyzca L, Aleksunes LM, Barrett ES. Impact of Fusarium-Derived Mycoestrogens on Female Reproduction: A Systematic Review. Toxins (Basel). 2021;13(6):373. [CrossRef]
- Gao X, Sun L, Zhang N, Li C, Zhang J, Xiao Z, Qi D. Gestational Zearalenone Exposure Causes Reproductive and Developmental Toxicity in Pregnant Rats and Female Offspring. Toxins (Basel). 2017;9(1):21. [CrossRef]
- Bartiromo L, Schimberni M, Villanacci R, Ottolina J, Dolci C, Salmeri N, Viganò P, Candiani M. Endometriosis and Phytoestrogens: Friends or Foes? A Systematic Review. Nutrients. 2021;13(8):2532. [CrossRef]
- Yan WK, Liu YN, Song SS, Kang JW, Zhang Y, Lu L, Wei SW, Xu QX, Zhang WQ, Liu XZ, Wu Y, Su RW. Zearalenone affects the growth of endometriosis via estrogen signaling and inflammatory pathways. Ecotoxicol Environ Saf. 2022;241:113826. [CrossRef]
- Cai X, Liu M, Zhang B, Zhao SJ, Jiang SW. Phytoestrogens for the Management of Endometriosis: Findings and Issues. Pharmaceuticals (Basel). 2021;14(6):569. [CrossRef]
- Rosenfeld CS, Cooke PS. Endocrine disruption through membrane estrogen receptors and novel pathways leading to rapid toxicological and epigenetic effects. J Steroid Biochem Mol Biol. 2019;187:106-117. [CrossRef]
- Feng JX, Riddle NC. Epigenetics and genome stability. Mamm Genome. 2020;31(5-6): 181-195. [CrossRef]
- Zubrzycka A, Zubrzycki M, Perdas E, Zubrzycka M. Genetic, Epigenetic, and Steroidogenic Modulation Mechanisms in Endometriosis. J Clin Med. 2020;9(5):1309. [CrossRef]
- Szukiewicz D, Stangret A, Ruiz-Ruiz C, Olivares EG, Soriţău O, Suşman S, Szewczyk G. Estrogen- and Progesterone (P4)-Mediated Epigenetic Modifications of Endometrial Stromal Cells (EnSCs) and/or Mesenchymal Stem/Stromal Cells (MSCs) in the Etiopathogenesis of Endometriosis. Stem Cell Rev Rep. 2021;17(4):1174-1193. [CrossRef]
- Chen H, Malentacchi F, Fambrini M, Harrath AH, Huang H, Petraglia F. Epigenetics of Estrogen and Progesterone Receptors in Endometriosis. Reprod Sci. 2020;27(11):1967-1974. [CrossRef]
- Bansal A, Henao-Mejia J, Simmons RA. Immune System: An Emerging Player in Mediating Effects of Endocrine Disruptors on Metabolic Health. Endocrinology. 2018; 159(1):32-45. [CrossRef]
- Abramiuk M, Grywalska E, Małkowska P, Sierawska O, Hrynkiewicz R, Niedźwiedzka-Rystwej P. The Role of the Immune System in the Development of Endometriosis. Cells. 2022;11(13):2028. [CrossRef]
- Szukiewicz D. Epigenetic regulation and T-cell responses in endometriosis - something other than autoimmunity. Front Immunol. 2022;13:943839. [CrossRef]
- Li Y, Zhang JJ, Chen RJ, Chen L, Chen S, Yang XF, Min JW. Genistein mitigates oxidative stress and inflammation by regulating Nrf2/HO-1 and NF-κB signaling pathways in hypoxic-ischemic brain damage in neonatal mice. Ann Transl Med. 2022;10(2):32. [CrossRef]
- Chen Y, Peng F, Xing Z, Chen J, Peng C, Li D. Beneficial effects of natural flavonoids on neuroinflammation. Front Immunol. 2022;13:1006434. [CrossRef]
- Dąbek J, Kułach A, Gąsior Z. Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB): a new potential therapeutic target in atherosclerosis? Pharmacol Rep. 2010; 62(5):778-83. [CrossRef]
- Desmawati D, Sulastri D. Phytoestrogens and Their Health Effect. Open Access Maced J Med Sci. 2019;7(3):495-499. [CrossRef]
- Cady N, Peterson SR, Freedman SN, Mangalam AK. Beyond Metabolism: The Complex Interplay Between Dietary Phytoestrogens, Gut Bacteria, and Cells of Nervous and Immune Systems. Front Neurol. 2020;11:150. [CrossRef]
- Masilamani M, Wei J, Bhatt S, Paul M, Yakir S, Sampson HA. Soybean isoflavones regulate dendritic cell function and suppress allergic sensitization to peanut. J Allergy Clin Immunol. 2011;128(6):1242-1250.e1. [CrossRef]
- Chiang SS, Pan TM. Beneficial effects of phytoestrogens and their metabolites produced by intestinal microflora on bone health. Appl Microbiol Biotechnol. 2013;97(4):1489-500. [CrossRef]
- Mace TA, Ware MB, King SA, Loftus S, Farren MR, McMichael E, Scoville S, Geraghty C, Young G, Carson WE 3rd, Clinton SK, Lesinski GB. Soy isoflavones and their metabolites modulate cytokine-induced natural killer cell function. Sci Rep. 2019;9(1):5068. [CrossRef]
- Wei J, Bhatt S, Chang LM, Sampson HA, Masilamani M. Isoflavones, genistein and daidzein, regulate mucosal immune response by suppressing dendritic cell function. PLoS One. 2012;7(10):e47979. [CrossRef]
- Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8(12):958-69. [CrossRef]
- Dia VP, Berhow MA, Gonzalez De Mejia E. Bowman-Birk inhibitor and genistein among soy compounds that synergistically inhibit nitric oxide and prostaglandin E2 pathways in lipopolysaccharide-induced macrophages. J Agric Food Chem. 2008;56(24):11707-17. [CrossRef]
- Abron JD, Singh NP, Price RL, Nagarkatti M, Nagarkatti PS, Singh UP. Genistein induces macrophage polarization and systemic cytokine to ameliorate experimental colitis. PLoS One. 2018;13(7):e0199631. [CrossRef]
- Iyer SS, Cheng G. Role of interleukin 10 transcriptional regulation in inflammation and autoimmune disease. Crit Rev Immunol. 2012;32(1):23-63. [CrossRef]
- Csaba G. Effect of endocrine disruptor phytoestrogens on the immune system: Present and future. Acta Microbiol Immunol Hung. 2018;65(1):1-14. [CrossRef]
- Saunders PTK, Horne AW. Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell. 2021;184(11):2807-2824. [CrossRef]
- International working group of AAGL, ESGE, ESHRE and WES; Tomassetti C, Johnson NP, Petrozza J, Abrao MS, Einarsson JI, Horne AW, Lee TTM, Missmer S, Vermeulen N, Zondervan KT, Grimbizis G, De Wilde RL. An International Terminology for Endometriosis, 2021. J Minim Invasive Gynecol. 2021;28(11):1849-1859. [CrossRef]
- Sampson JA. Metastatic or Embolic Endometriosis, due to the Menstrual Dissemination of Endometrial Tissue into the Venous Circulation. Am J Pathol. 1927;3(2):93-110.43.
- Mikhaleva LM, Radzinsky VE, Orazov MR, Khovanskaya TN, Sorokina AV, Mikhalev SA, Volkova SV, Shustova VB, Sinelnikov MY. Current Knowledge on Endometriosis Etiology: A Systematic Review of Literature. Int J Womens Health. 2021;13:525-537. [CrossRef]
- Signorile PG, Viceconte R, Baldi A. New Insights in Pathogenesis of Endometriosis. Front Med (Lausanne). 2022;9:879015. [CrossRef]
- Chamié LP, Ribeiro DMFR, Tiferes DA, Macedo Neto AC, Serafini PC. Atypical Sites of Deeply Infiltrative Endometriosis: Clinical Characteristics and Imaging Findings. Radiographics. 2018;38(1):309-328. [CrossRef]
- Machairiotis N, Stylianaki A, Dryllis G, Zarogoulidis P, Kouroutou P, Tsiamis N, Katsikogiannis N, Sarika E, Courcoutsakis N, Tsiouda T, Gschwendtner A, Zarogoulidis K, Sakkas L, Baliaka A, Machairiotis C. Extrapelvic endometriosis: a rare entity or an under diagnosed condition? Diagn Pathol. 2013;8:194. [CrossRef]
- Kamergorodsky G, Ribeiro PA, Galvão MA, Abrão MS, Donadio N, Lemos NL, Aoki T. Histologic classification of specimens from women affected by superficial endometriosis, deeply infiltrating endometriosis, and ovarian endometriomas. Fertil Steril. 2009;92(6):2074-7. [CrossRef]
- Al-Jefout M, Dezarnaulds G, Cooper M, Tokushige N, Luscombe GM, Markham R, Fraser IS. Diagnosis of endometriosis by detection of nerve fibres in an endometrial biopsy: a double blind study. Hum Reprod. 2009;24(12):3019-24. [CrossRef]
- Bulun SE, Cheng YH, Pavone ME, Xue Q, Attar E, Trukhacheva E, Tokunaga H, Utsunomiya H, Yin P, Luo X, Lin Z, Imir G, Thung S, Su EJ, Kim JJ. Estrogen receptor-beta, estrogen receptor-alpha, and progesterone resistance in endometriosis. Semin Reprod Med. 2010;28(1):36-43. [CrossRef]
- Plante BJ, Lessey BA, Taylor RN, Wang W, Bagchi MK, Yuan L, Scotchie J, Fritz MA, Young SL. G protein-coupled estrogen receptor (GPER) expression in normal and abnormal endometrium. Reprod Sci. 2012;19(7):684-93. [CrossRef]
- Kim JH, Han E. Endometriosis and Female Pelvic Pain. Semin Reprod Med. 2018;36(2): 143-151. [CrossRef]
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511-9. [CrossRef]
- Patel BG, Lenk EE, Lebovic DI, Shu Y, Yu J, Taylor RN. Pathogenesis of endometriosis: Interaction between Endocrine and inflammatory pathways. Best Pract Res Clin Obstet Gynaecol. 2018;50:50-60. [CrossRef]
- Tomassetti C, D'Hooghe T. Endometriosis and infertility: Insights into the causal link and management strategies. Best Pract Res Clin Obstet Gynaecol. 2018;51:25-33. [CrossRef]
- Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN, Viganò P. Endometriosis. Nat Rev Dis Primers. 2018;4(1):9. [CrossRef]
- Kajiyama H, Suzuki S, Yoshihara M, Tamauchi S, Yoshikawa N, Niimi K, Shibata K, Kikkawa F. Endometriosis and cancer. Free Radic Biol Med. 2019;133:186-192. [CrossRef]
- Klemmt PAB, Starzinski-Powitz A. Molecular and Cellular Pathogenesis of Endometriosis. Curr Womens Health Rev. 2018;14(2):106-116. [CrossRef]
- Rolla E. Endometriosis: advances and controversies in classification, pathogenesis, diagnosis, and treatment. F1000Res. 2019;8:F1000 Faculty Rev-529. [CrossRef]
- Logan PC, Yango P, Tran ND. Endometrial Stromal and Epithelial Cells Exhibit Unique Aberrant Molecular Defects in Patients With Endometriosis. Reprod Sci. 2018;25(1):140-159. [CrossRef]
- Maruyama T, Yoshimura Y. Stem cell theory for the pathogenesis of endometriosis. Front Biosci (Elite Ed). 2012;4(8):2754-63. [CrossRef]
- Gargett CE, Schwab KE, Brosens JJ, Puttemans P, Benagiano G, Brosens I. Potential role of endometrial stem/progenitor cells in the pathogenesis of early-onset endometriosis. Mol Hum Reprod. 2014;20(7):591-8. [CrossRef]
- Dinsdale N, Nepomnaschy P, Crespi B. The evolutionary biology of endometriosis. Evol Med Public Health. 2021;9(1):174-191. [CrossRef]
- Ng SW, Norwitz GA, Pavlicev M, Tilburgs T, Simón C, Norwitz ER. Endometrial Decidualization: The Primary Driver of Pregnancy Health. Int J Mol Sci. 2020;21(11):4092. [CrossRef]
- Xu Y, Zhu H, Zhao D, Tan J. Endometrial stem cells: clinical application and pathological roles. Int J Clin Exp Med. 2015;8(12):22039-44.
- da Costa e Silva Rde C, Moura KK, Ribeiro Júnior CL, Guillo LA. Estrogen signaling in the proliferative endometrium: implications in endometriosis. Rev Assoc Med Bras. (1992). 2016;62(1):72-7. [CrossRef]
- Ballaré C, Uhrig M, Bechtold T, Sancho E, Di Domenico M, Migliaccio A, Auricchio F, Beato M. Two domains of the progesterone receptor interact with the estrogen receptor and are required for progesterone activation of the c-Src/Erk pathway in mammalian cells. Mol Cell Biol. 2003;23(6):1994-2008. [CrossRef]
- Kasubuchi M, Watanabe K, Hirano K, Inoue D, Li X, Terasawa K, Konishi M, Itoh N, Kimura I. Membrane progesterone receptor beta (mPRβ/Paqr8) promotes progesterone-dependent neurite outgrowth in PC12 neuronal cells via non-G protein-coupled receptor (GPCR) signaling. Sci Rep. 2017;7(1):5168. [CrossRef]
- Appleyard CB, Flores I, Torres-Reverón A. The Link Between Stress and Endometriosis: from Animal Models to the Clinical Scenario. Reprod Sci. 2020;27(9):1675-1686. [CrossRef]
- Huhtinen K, Desai R, Ståhle M, Salminen A, Handelsman DJ, Perheentupa A, Poutanen M. Endometrial and endometriotic concentrations of estrone and estradiol are determined by local metabolism rather than circulating levels. J Clin Endocrinol Metab. 2012;97(11):4228-35. [CrossRef]
- Streuli I, Gaitzsch H, Wenger JM, Petignat P. Endometriosis after menopause: physiopathology and management of an uncommon condition. Climacteric. 2017;20(2):138-143. [CrossRef]
- Leone Roberti Maggiore U, Ferrero S, Mangili G, Bergamini A, Inversetti A, Giorgione V, Viganò P, Candiani M. A systematic review on endometriosis during pregnancy: diagnosis, misdiagnosis, complications and outcomes. Hum Reprod Update. 2016;22(1):70-103. [CrossRef]
- Jeng CJ, Chuang L, Shen J. A comparison of progestogens or oral contraceptives and gonadotropin-releasing hormone agonists for the treatment of endometriosis: a systematic review. Expert Opin Pharmacother. 2014;15(6):767-73. [CrossRef]
- Stocco C. Tissue physiology and pathology of aromatase. Steroids. 2012;77(1-2):27-35. [CrossRef]
- Bulun SE, Fang Z, Imir G, Gurates B, Tamura M, Yilmaz B, Langoi D, Amin S, Yang S, Deb S. Aromatase and endometriosis. Semin Reprod Med. 2004;22(1):45-50. [CrossRef]
- Noble LS, Takayama K, Zeitoun KM, Putman JM, Johns DA, Hinshelwood MM, Agarwal VR, Zhao Y, Carr BR, Bulun SE. Prostaglandin E2 stimulates aromatase expression in endometriosis-derived stromal cells. J Clin Endocrinol Metab. 1997;82(2):600-6. [CrossRef]
- Bulun SE, Takayama K, Suzuki T, Sasano H, Yilmaz B, Sebastian S. Organization of the human aromatase p450 (CYP19) gene. Semin Reprod Med. 2004;22(1):5-9. [CrossRef]
- Izawa M, Harada T, Taniguchi F, Ohama Y, Takenaka Y, Terakawa N. An epigenetic disorder may cause aberrant expression of aromatase gene in endometriotic stromal cells. Fertil Steril. 2008;89(5 Suppl):1390-6. [CrossRef]
- Izawa M, Taniguchi F, Uegaki T, Takai E, Iwabe T, Terakawa N, Harada T. Demethylation of a nonpromoter cytosine-phosphate-guanine island in the aromatase gene may cause the aberrant up-regulation in endometriotic tissues. Fertil Steril. 2011;95(1):33-9. [CrossRef]
- Moore LD, Le T, Fan G. DNA methylation and its basic function. Neuropsychopharmacology. 2013;38(1):23-38. [CrossRef]
- Xue Q, Zhou YF, Zhu SN, Bulun SE. Hypermethylation of the CpG island spanning from exon II to intron III is associated with steroidogenic factor 1 expression in stromal cells of endometriosis. Reprod Sci. 2011;18(11):1080-4. [CrossRef]
- Koukoura O, Sifakis S, Spandidos DA. DNA methylation in endometriosis (Review). Mol Med Rep. 2016;13(4):2939-48. [CrossRef]
- Zeitoun K, Takayama K, Sasano H, Suzuki T, Moghrabi N, Andersson S, Johns A, Meng L, Putman M, Carr B, Bulun SE. Deficient 17beta-hydroxysteroid dehydrogenase type 2 expression in endometriosis: failure to metabolize 17beta-estradiol. J Clin Endocrinol Metab. 1998;83(12):4474-80. [CrossRef]
- Yamagata Y, Nishino K, Takaki E, Sato S, Maekawa R, Nakai A, Sugino N. Genome-wide DNA methylation profiling in cultured eutopic and ectopic endometrial stromal cells. PLoS One. 2014;9(1):e83612. [CrossRef]
- Husen B, Psonka N, Jacob-Meisel M, Keil C, Rune GM. Differential expression of 17beta-hydroxysteroid dehydrogenases types 2 and 4 in human endometrial epithelial cell lines. J Mol Endocrinol. 2000;24(1):135-44. [CrossRef]
- He W, Gauri M, Li T, Wang R, Lin SX. Current knowledge of the multifunctional 17β-hydroxysteroid dehydrogenase type 1 (HSD17B1). Gene. 2016;588(1):54-61. [CrossRef]
- Allis CD, Jenuwein T. The molecular hallmarks of epigenetic control. Nat Rev Genet. 2016;17(8):487-500. [CrossRef]
- Loganathan T, Doss C GP. Non-coding RNAs in human health and disease: potential function as biomarkers and therapeutic targets. Funct Integr Genomics. 2023;23(1):33. [CrossRef]
- Trerotola M, Relli V, Simeone P, Alberti S. Epigenetic inheritance and the missing heritability. Hum Genomics. 2015;9(1):17. [CrossRef]
- Wątroba M, Dudek I, Skoda M, Stangret A, Rzodkiewicz P, Szukiewicz D. Sirtuins, epigenetics and longevity. Ageing Res Rev. 2017;40:11-19. [CrossRef]
- Cousins FL, Filby CE, Gargett CE. Endometrial Stem/Progenitor Cells-Their Role in Endometrial Repair and Regeneration. Front Reprod Health. 2022;3:811537. [CrossRef]
- Gurusamy N, Alsayari A, Rajasingh S, Rajasingh J. Adult Stem Cells for Regenerative Therapy. Prog Mol Biol Transl Sci. 2018;160:1-22. [CrossRef]
- Papatsenko D, Waghray A, Lemischka IR. Feedback control of pluripotency in embryonic stem cells: Signaling, transcription and epigenetics. Stem Cell Res. 2018;29:180-188. [CrossRef]
- Edmunds KM, Holloway AC, Crankshaw DJ, Agarwal SK, Foster WG. The effects of dietary phytoestrogens on aromatase activity in human endometrial stromal cells. Reprod Nutr Dev. 2005;45(6):709-20. [CrossRef]
- Koninckx PR, Ussia A, Adamyan L, Wattiez A, Gomel V, Martin DC. Pathogenesis of endometriosis: the genetic/epigenetic theory. Fertil Steril. 2019;111(2):327-340. [CrossRef]
- Retis-Resendiz AM, González-García IN, León-Juárez M, Camacho-Arroyo I, Cerbón M, Vázquez-Martínez ER. The role of epigenetic mechanisms in the regulation of gene expression in the cyclical endometrium. Clin Epigenetics. 2021;13(1):116. [CrossRef]
- Szukiewicz D. Aberrant epigenetic regulation of estrogen and progesterone signaling at the level of endometrial/endometriotic tissue in the pathomechanism of endometriosis. Vitam Horm. 2023;122:193-235. [CrossRef]
- Vrtačnik P, Ostanek B, Mencej-Bedrač S, Marc J. The many faces of estrogen signaling. Biochem Med (Zagreb). 2014;24(3):329-42. [CrossRef]
- Zhou Q, Shaw PG, Davidson NE. Epigenetics meets estrogen receptor: regulation of estrogen receptor by direct lysine methylation. Endocr Relat Cancer. 2009;16(2):319-23. [CrossRef]
- Brandenberger AW, Lebovic DI, Tee MK, Ryan IP, Tseng JF, Jaffe RB, Taylor RN. Oestrogen receptor (ER)-alpha and ER-beta isoforms in normal endometrial and endometriosis-derived stromal cells. Mol Hum Reprod. 1999;5(7):651-5. [CrossRef]
- Simmen RC, Kelley AS. Reversal of fortune: estrogen receptor-β in endometriosis. J Mol Endocrinol. 2016;57(2):F23-7. [CrossRef]
- Xue Q, Lin Z, Cheng YH, Huang CC, Marsh E, Yin P, Milad MP, Confino E, Reierstad S, Innes J, Bulun SE. Promoter methylation regulates estrogen receptor 2 in human endometrium and endometriosis. Biol Reprod. 2007;77(4):681-7. [CrossRef]
- Bulun SE, Zeitoun KM, Takayama K, Simpson E, Sasano H. Aromatase as a therapeutic target in endometriosis. Trends Endocrinol Metab. 2000;11(1):22-7. [CrossRef]
- Patel BG, Rudnicki M, Yu J, Shu Y, Taylor RN. Progesterone resistance in endometriosis: origins, consequences and interventions. Acta Obstet Gynecol Scand. 2017;96(6):623-632. [CrossRef]
- Trukhacheva E, Lin Z, Reierstad S, Cheng YH, Milad M, Bulun SE. Estrogen receptor (ER) beta regulates ERalpha expression in stromal cells derived from ovarian endometriosis. J Clin Endocrinol Metab. 2009;94(2):615-22. [CrossRef]
- Ferlita A, Battaglia R, Andronico F, Caruso S, Cianci A, Purrello M, Pietro CD. Non-Coding RNAs in Endometrial Physiopathology. Int J Mol Sci. 2018;19(7):2120. [CrossRef]
- Vidal-Gómez X, Pérez-Cremades D, Mompeón A, Dantas AP, Novella S, Hermenegildo C. MicroRNA as Crucial Regulators of Gene Expression in Estradiol-Treated Human Endothelial Cells. Cell Physiol Biochem. 2018;45(5):1878-1892. [CrossRef]
- Cai H, Zhu XX, Li ZF, Zhu YP, Lang JH. MicroRNA Dysregulation and Steroid Hormone Receptor Expression in Uterine Tissues of Rats with Endometriosis during the Implantation Window. Chin Med J (Engl). 2018;131(18):2193-2204. [CrossRef]
- Klinge CM. miRNAs and estrogen action. Trends Endocrinol Metab. 2012;23(5):223-33. [CrossRef]
- Pandey DP, Picard D. miR-22 inhibits estrogen signaling by directly targeting the estrogen receptor alpha mRNA. Mol Cell Biol. 2009;29(13):3783-90. [CrossRef]
- Lin Y, Xiao L, Zhang Y, Li P, Wu Y, Lin Y. MiR-26b-3p regulates osteoblast differentiation via targeting estrogen receptor α. Genomics. 2019;111(5):1089-1096. [CrossRef]
- Al-Nakhle H, Burns PA, Cummings M, Hanby AM, Hughes TA, Satheesha S, Shaaban AM, Smith L, Speirs V. Estrogen receptor {beta}1 expression is regulated by miR-92 in breast cancer. Cancer Res. 2010;70(11):4778-84. [CrossRef]
- He SZ, Li J, Bao HC, Wang MM, Wang XR, Huang X, Li FH, Zhang W, Xu AL, Fang HC, Sheng YX. G protein-coupled estrogen receptor/miR-148a/human leukocyte antigen-G signaling pathway mediates cell apoptosis of ovarian endometriosis. Mol Med Rep. 2018; 18(1):1141-1148. [CrossRef]
- Knoll M, Lodish HF, Sun L. Long non-coding RNAs as regulators of the endocrine system. Nat Rev Endocrinol. 2015;11(3):151-60. [CrossRef]
- Yan W, Hu H, Tang B. Progress in understanding the relationship between long noncoding RNA and endometriosis. Eur J Obstet Gynecol Reprod Biol X. 2019;5:100067. [CrossRef]
- Bhan A, Hussain I, Ansari KI, Kasiri S, Bashyal A, Mandal SS. Antisense transcript long noncoding RNA (lncRNA) HOTAIR is transcriptionally induced by estradiol. J Mol Biol. 2013;425(19):3707-22. [CrossRef]
- Trisciuoglio D, Di Martile M, Del Bufalo D. Emerging Role of Histone Acetyltransferase in Stem Cells and Cancer. Stem Cells Int. 2018;2018:8908751. [CrossRef]
- Zhang J, Jing L, Li M, He L, Guo Z. Regulation of histone arginine methylation/demethylation by methylase and demethylase (Review). Mol Med Rep. 2019; 19(5):3963-3971. [CrossRef]
- Yokoyama A, Fujiki R, Ohtake F, Kato S. Regulated histone methyltransferase and demethylase complexes in the control of genes by nuclear receptors. Cold Spring Harb Symp Quant Biol. 2011;76:165-73. [CrossRef]
- Liu C, Wu HT, Zhu N, Shi YN, Liu Z, Ao BX, Liao DF, Zheng XL, Qin L. Steroid receptor RNA activator: Biologic function and role in disease. Clin Chim Acta. 2016;459:137-146. [CrossRef]
- Lin K, Zhan H, Ma J, Xu K, Wu R, Zhou C, Lin J. Silencing of SRA1 Regulates ER Expression and Attenuates the Growth of Stromal Cells in Ovarian Endometriosis. Reprod Sci. 2017;24(6):836-843. [CrossRef]
- Moore RL, Dai Y, Faller DV. Sirtuin 1 (SIRT1) and steroid hormone receptor activity in cancer. J Endocrinol. 2012;213(1):37-48. [CrossRef]
- Yoo JY, Kim TH, Fazleabas AT, Palomino WA, Ahn SH, Tayade C, Schammel DP, Young SL, Jeong JW, Lessey BA. KRAS Activation and over-expression of SIRT1/BCL6 Contributes to the Pathogenesis of Endometriosis and Progesterone Resistance. Sci Rep. 2017; 7(1):6765. [CrossRef]
- Xiaomeng X, Ming Z, Jiezhi M, Xiaoling F. Aberrant histone acetylation and methylation levels in woman with endometriosis. Arch Gynecol Obstet. 2013;287(3):487-94. [CrossRef]
- Han SJ, O'Malley BW. The dynamics of nuclear receptors and nuclear receptor coregulators in the pathogenesis of endometriosis. Hum Reprod Update. 2014;20(4):467-84. [CrossRef]
- Mahajan V, Farquhar C, Ponnampalam AP. Could DNA hydroxymethylation be crucial in influencing steroid hormone signaling in endometrial biology and endometriosis? Mol Reprod Dev. 2020;87(1):7-16. [CrossRef]
- Klinge CM. Estrogen action: Receptors, transcripts, cell signaling, and non-coding RNAs in normal physiology and disease. Mol Cell Endocrinol. 2015;418 Pt 3:191-2. [CrossRef]
- Grimstad FW, Decherney A. A Review of the Epigenetic Contributions to Endometriosis. Clin Obstet Gynecol. 2017;60(3):467-476. [CrossRef]
- Nasu K, Nishida M, Kawano Y, Tsuno A, Abe W, Yuge A, Takai N, Narahara H. Aberrant expression of apoptosis-related molecules in endometriosis: a possible mechanism underlying the pathogenesis of endometriosis. Reprod Sci. 2011;18(3):206-18. [CrossRef]
- Chopyak VV, Koval HD, Havrylyuk AM, Lishchuk-Yakymovych KA, Potomkina HA, Kurpisz MK. Immunopathogenesis of endometriosis - a novel look at an old problem. Cent Eur J Immunol. 2022;47(1):109-116. [CrossRef]
- Vallvé-Juanico J, Houshdaran S, Giudice LC. The endometrial immune environment of women with endometriosis. Hum Reprod Update. 2019; 25(5):564-591. [CrossRef]
- Kobayashi H, Imanaka S. Understanding the molecular mechanisms of macrophage polarization and metabolic reprogramming in endometriosis: A narrative review. Reprod Med Biol. 2022;21(1):e12488. [CrossRef]
- Pernis AB. Estrogen and CD4+ T cells. Curr Opin Rheumatol. 2007;19(5):414-20. [CrossRef]
- Wan YY. Regulatory T cells: immune suppression and beyond. Cell Mol Immunol. 2010;7(3):204-10. [CrossRef]
- Bayati F, Mohammadi M, Valadi M, Jamshidi S, Foma AM, Sharif-Paghaleh E. The Therapeutic Potential of Regulatory T Cells: Challenges and Opportunities. Front Immunol. 2021;11:585819. [CrossRef]
- de Barros IBL, Malvezzi H, Gueuvoghlanian-Silva BY, Piccinato CA, Rizzo LV, Podgaec S. "What do we know about regulatory T cells and endometriosis? A systematic review". J Reprod Immunol. 2017;120:48-55. Erratum in: J Reprod Immunol. 2017;121:34. [CrossRef]
- Camboni A, Marbaix E. Ectopic Endometrium: The Pathologist's Perspective. Int J Mol Sci. 2021;22(20):10974. [CrossRef]
- McKinnon B, Mueller M, Montgomery G. Progesterone Resistance in Endometriosis: an Acquired Property? Trends Endocrinol Metab. 2018;29(8):535-548. [CrossRef]
- Chimento A, De Luca A, Avena P, De Amicis F, Casaburi I, Sirianni R, Pezzi V. Estrogen Receptors-Mediated Apoptosis in Hormone-Dependent Cancers. Int J Mol Sci. 2022;23(3):1242. [CrossRef]
- Khan D, Ansar Ahmed S. The Immune System Is a Natural Target for Estrogen Action: Opposing Effects of Estrogen in Two Prototypical Autoimmune Diseases. Front Immunol. 2016;6:635. [CrossRef]
- Hewagama A, Patel D, Yarlagadda S, Strickland FM, Richardson BC. Stronger inflammatory/cytotoxic T-cell response in women identified by microarray analysis. Genes Immun. 2009;10(5):509-16. [CrossRef]
- Priyanka HP, Krishnan HC, Singh RV, Hima L, Thyagarajan S. Estrogen modulates in vitro T cell responses in a concentration- and receptor-dependent manner: effects on intracellular molecular targets and antioxidant enzymes. Mol Immunol. 2013;56(4):328-39. [CrossRef]
- Greenbaum H, Galper BL, Decter DH, Eisenberg VH. Endometriosis and autoimmunity: Can autoantibodies be used as a non-invasive early diagnostic tool? Autoimmun Rev. 2021;20(5):102795. [CrossRef]
- Giannoni E, Guignard L, Knaup Reymond M, Perreau M, Roth-Kleiner M, Calandra T, Roger T. Estradiol and progesterone strongly inhibit the innate immune response of mononuclear cells in newborns. Infect Immun. 2011;79(7):2690-8. [CrossRef]
- Harding AT, Heaton NS. The Impact of Estrogens and Their Receptors on Immunity and Inflammation during Infection. Cancers (Basel). 2022;14(4):909. [CrossRef]
- Harding AT, Goff MA, Froggatt HM, Lim JK, Heaton NS. GPER1 is required to protect fetal health from maternal inflammation. Science. 2021;371(6526):271-276. [CrossRef]
- Fan Z, Che H, Yang S, Chen C. Estrogen and estrogen receptor signaling promotes allergic immune responses: Effects on immune cells, cytokines, and inflammatory factors involved in allergy. Allergol Immunopathol (Madr). 2019;47(5):506-512. [CrossRef]
- Hsu JT, Kan WH, Hsieh CH, Choudhry MA, Schwacha MG, Bland KI, Chaudry IH. Mechanism of estrogen-mediated attenuation of hepatic injury following trauma-hemorrhage: Akt-dependent HO-1 up-regulation. J Leukoc Biol. 2007;82(4):1019-26. [CrossRef]
- Xing D, Feng W, Miller AP, Weathington NM, Chen YF, Novak L, Blalock JE, Oparil S. Estrogen modulates TNF-alpha-induced inflammatory responses in rat aortic smooth muscle cells through estrogen receptor-beta activation. Am J Physiol Heart Circ Physiol. 2007; 292(6):H2607-12. [CrossRef]
- Dai R, Cowan C, Heid B, Khan D, Liang Z, Pham CT, Ahmed SA. Neutrophils and neutrophil serine proteases are increased in the spleens of estrogen-treated C57BL/6 mice and several strains of spontaneous lupus-prone mice. PLoS One. 2017;12(2):e0172105. [CrossRef]
- Biswas P, Delfanti F, Bernasconi S, Mengozzi M, Cota M, Polentarutti N, Mantovani A, Lazzarin A, Sozzani S, Poli G. Interleukin-6 induces monocyte chemotactic protein-1 in peripheral blood mononuclear cells and in the U937 cell line. Blood. 1998;91(1): 258-65.
- Michlewska S, Dransfield I, Megson IL, Rossi AG. Macrophage phagocytosis of apoptotic neutrophils is critically regulated by the opposing actions of pro-inflammatory and anti-inflammatory agents: key role for TNF-alpha. FASEB J. 2009 ;23(3): 844-54. [CrossRef]
- Azenabor AA, Yang S, Job G, Adedokun OO. Expression of iNOS gene in macrophages stimulated with 17beta-estradiol is regulated by free intracellular Ca2+. Biochem Cell Biol. 2004;82(3):381-90. [CrossRef]
- Karpuzoglu E, Ahmed SA. Estrogen regulation of nitric oxide and inducible nitric oxide synthase (iNOS) in immune cells: implications for immunity, autoimmune diseases, and apoptosis. Nitric Oxide. 2006;15(3):177-86. [CrossRef]
- Lélu K, Laffont S, Delpy L, Paulet PE, Périnat T, Tschanz SA, Pelletier L, Engelhardt B, Guéry JC. Estrogen receptor α signaling in T lymphocytes is required for estradiol-mediated inhibition of Th1 and Th17 cell differentiation and protection against experimental autoimmune encephalomyelitis. J Immunol. 2011;187(5):2386-93. [CrossRef]
- Robinson DP, Hall OJ, Nilles TL, Bream JH, Klein SL. 17β-estradiol protects females against influenza by recruiting neutrophils and increasing virus-specific CD8 T cell responses in the lungs. J Virol. 2014;88(9):4711-20. [CrossRef]
- Karpuzoglu-Sahin E, Hissong BD, Ansar Ahmed S. Interferon-gamma levels are upregulated by 17-beta-estradiol and diethylstilbestrol. J Reprod Immunol. 2001;52(1-2):113-27. [CrossRef]
- Dragin N, Nancy P, Villegas J, Roussin R, Le Panse R, Berrih-Aknin S. Balance between Estrogens and Proinflammatory Cytokines Regulates Chemokine Production Involved in Thymic Germinal Center Formation. Sci Rep. 2017;7(1):7970. Erratum in: Sci Rep. 2018;8(1):8118. [CrossRef]
- Tang S, Han H, Bajic VB. ERGDB: Estrogen Responsive Genes Database. Nucleic Acids Res. 2004;32(Database issue):D533-6. [CrossRef]
- Polanczyk MJ, Carson BD, Subramanian S, Afentoulis M, Vandenbark AA, Ziegler SF, Offner H. Cutting edge: estrogen drives expansion of the CD4+CD25+ regulatory T cell compartment. J Immunol. 2004;173(4):2227-30. [CrossRef]
- Tai P, Wang J, Jin H, Song X, Yan J, Kang Y, Zhao L, An X, Du X, Chen X, Wang S, Xia G, Wang B. Induction of regulatory T cells by physiological level estrogen. J Cell Physiol. 2008; 214(2):456-64. [CrossRef]
- Brown MA, Su MA. An Inconvenient Variable: Sex Hormones and Their Impact on T Cell Responses. J Immunol. 2019;202(7):1927-1933. [CrossRef]
- Hosokawa H, Rothenberg EV. How transcription factors drive choice of the T cell fate. Nat Rev Immunol. 2021;21(3):162-176. [CrossRef]
- Grimaldi CM, Cleary J, Dagtas AS, Moussai D, Diamond B. Estrogen alters thresholds for B cell apoptosis and activation. J Clin Invest. 2002;109(12):1625-33. [CrossRef]
- Grimaldi CM, Jeganathan V, Diamond B. Hormonal regulation of B cell development: 17 beta-estradiol impairs negative selection of high-affinity DNA-reactive B cells at more than one developmental checkpoint. J Immunol. 2006;176(5):2703-10. [CrossRef]
- Verthelyi DI, Ahmed SA. Estrogen increases the number of plasma cells and enhances their autoantibody production in nonautoimmune C57BL/6 mice. Cell Immunol. 1998;189(2):125-34. [CrossRef]
- Hill L, Jeganathan V, Chinnasamy P, Grimaldi C, Diamond B. Differential roles of estrogen receptors α and β in control of B-cell maturation and selection. Mol Med. 2011;17(3-4):211-20. [CrossRef]
- Stice JP, Mbai FN, Chen L, Knowlton AA. Rapid activation of nuclear factor κB by 17β-estradiol and selective estrogen receptor modulators: pathways mediating cellular protection. Shock. 2012;38(2):128-36. [CrossRef]
- Xing D, Oparil S, Yu H, Gong K, Feng W, Black J, Chen YF, Nozell S. Estrogen modulates NFκB signaling by enhancing IκBα levels and blocking p65 binding at the promoters of inflammatory genes via estrogen receptor-β. PLoS One. 2012;7(6):e36890. [CrossRef]
- Stice JP, Knowlton AA. Estrogen, NFkappaB, and the heat shock response. Mol Med. 2008;14(7-8):517-27. [CrossRef]
- Monteiro R, Teixeira D, Calhau C. Estrogen signaling in metabolic inflammation. Mediators Inflamm. 2014;2014:615917. [CrossRef]
- Pelekanou V, Kampa M, Kiagiadaki F, Deli A, Theodoropoulos P, Agrogiannis G, Patsouris E, Tsapis A, Castanas E, Notas G. Estrogen anti-inflammatory activity on human monocytes is mediated through cross-talk between estrogen receptor ERα36 and GPR30/GPERJ Leukoc Biol. 2016;99(2):333-47. [CrossRef]
- Liu CJ, Lo JF, Kuo CH, Chu CH, Chen LM, Tsai FJ, Tsai CH, Tzang BS, Kuo WW, Huang CY. Akt mediates 17beta-estradiol and/or estrogen receptor-alpha inhibition of LPS-induced tumor necresis factor-alpha expression and myocardial cell apoptosis by suppressing the JNK1/2-NFkappaB pathway. J Cell Mol Med. 2009;13(9B):3655-67. [CrossRef]
- Slabe N, Meden-Vrtovec H, Verdenik I, Kosir-Pogacnik R, Ihan A. Cytotoxic T-Cells in Peripheral Blood in Women with Endometriosis. Geburtshilfe Frauenheilkd. 2013;73(10): 1042-1048. [CrossRef]
- Bolitho P, Voskoboinik I, Trapani JA, Smyth MJ. Apoptosis induced by the lymphocyte effector molecule perforin. Curr Opin Immunol. 2007;19(3):339-47. [CrossRef]
- Osińska I, Popko K, Demkow U. Perforin: an important player in immune response. Cent Eur J Immunol. 2014;39(1):109-15. [CrossRef]
- Lu Q, Wu A, Ray D, Deng C, Attwood J, Hanash S, Pipkin M, Lichtenheld M, Richardson B. DNA methylation and chromatin structure regulate T cell perforin gene expression. J Immunol. 2003;170(10):5124-32. [CrossRef]
- Zierau O, Zenclussen AC, Jensen F. Role of female sex hormones, estradiol and progesterone, in mast cell behavior. Front Immunol. 2012;3:169. [CrossRef]
- Szukiewicz D, Wojdasiewicz P, Watroba M, Szewczyk G. Mast Cell Activation Syndrome in COVID-19 and Female Reproductive Function: Theoretical Background vs. Accumulating Clinical Evidence. J Immunol Res. 2022;2022:9534163. [CrossRef]
- Cutolo M, Capellino S, Sulli A, Serioli B, Secchi ME, Villaggio B, Straub RH. Estrogens and autoimmune diseases. Ann N Y Acad Sci. 2006;1089:538-47. [CrossRef]
- Jacenik D, Krajewska WM. Significance of G Protein-Coupled Estrogen Receptor in the Pathophysiology of Irritable Bowel Syndrome, Inflammatory Bowel Diseases and Colorectal Cancer. Front Endocrinol (Lausanne). 2020;11:390. [CrossRef]
- Moon TC, Befus AD, Kulka M. Mast cell mediators: their differential release and the secretory pathways involved. Front Immunol. 2014;5:569. [CrossRef]
- Theoharides TC, Alysandratos KD, Angelidou A, Delivanis DA, Sismanopoulos N, Zhang B, Asadi S, Vasiadi M, Weng Z, Miniati A, Kalogeromitros D. Mast cells and inflammation. Biochim Biophys Acta. 2012;1822(1):21-33. [CrossRef]
- Valent P, Hartmann K, Bonadonna P, Niedoszytko M, Triggiani M, Arock M, Brockow K. Mast Cell Activation Syndromes: Collegium Internationale Allergologicum Update Int Arch Allergy Immunol. 2022;183(7):693-705. [CrossRef]
- Szewczyk G, Pyzlak M, Klimkiewicz J, Smiertka W, Miedzińska-Maciejewska M, Szukiewicz D. Mast cells and histamine: do they influence placental vascular network and development in preeclampsia? Mediators Inflamm. 2012;2012:307189. [CrossRef]
- Kempuraj D, Papadopoulou N, Stanford EJ, Christodoulou S, Madhappan B, Sant GR, Solage K, Adams T, Theoharides TC. Increased numbers of activated mast cells in endometriosis lesions positive for corticotropin-releasing hormone and urocortin. Am J Reprod Immunol. 2004;52(4):267-75. [CrossRef]
- McCallion A, Nasirzadeh Y, Lingegowda H, Miller JE, Khalaj K, Ahn S, Monsanto SP, Bidarimath M, Sisnett DJ, Craig AW, Young SL, Lessey BA, Koti M, Tayade C. Estrogen mediates inflammatory role of mast cells in endometriosis pathophysiology. Front Immunol. 2022;13:961599. [CrossRef]
- Pansrikaew P, Cheewakriangkrai C, Taweevisit M, Khunamornpong S, Siriaunkgul S. Correlation of mast cell density, tumor angiogenesis, and clinical outcomes in patients with endometrioid endometrial cancer. Asian Pac J Cancer Prev. 2010;11(3):623-6.
- Mercorio A, Giampaolino P, Romano A, Dällenbach P, Pluchino N. Is intracrinology of endometriosis relevant in clinical practice? A systematic review on estrogen metabolism. Front Endocrinol (Lausanne). 2022;13:950866. [CrossRef]
- Binda MM, Donnez J, Dolmans MM. Targeting mast cells: a new way to treat endometriosis. Expert Opin Ther Targets. 2017;21(1):67-75. [CrossRef]
- Bulfone-Paus S, Bahri R. Mast Cells as Regulators of T Cell Responses. Front Immunol. 2015; 6:394. [CrossRef]
- Krystel-Whittemore M, Dileepan KN, Wood JG. Mast Cell: A Multi-Functional Master Cell. Front Immunol. 2016;6:620. [CrossRef]
- Khan D, Ansar Ahmed S. The Immune System Is a Natural Target for Estrogen Action: Opposing Effects of Estrogen in Two Prototypical Autoimmune Diseases. Front Immunol. 2016;6:635. [CrossRef]
- Walker ME, Simpson JB, Redinbo MR. A structural metagenomics pipeline for examining the gut microbiome. Curr Opin Struct Biol. 2022;75:102416. [CrossRef]
- Putignani L, Del Chierico F, Petrucca A, Vernocchi P, Dallapiccola B. The human gut microbiota: a dynamic interplay with the host from birth to senescence settled during childhood. Pediatr Res. 2014;76(1):2-10. [CrossRef]
- Stephen AM, Cummings JH. The microbial contribution to human faecal mass. J Med Microbiol. 1980;13(1):45-56. [CrossRef]
- Kho ZY, Lal SK. The Human Gut Microbiome - A Potential Controller of Wellness and Disease. Front Microbiol. 2018;9:1835. [CrossRef]
- Valdes AM, Walter J, Segal E, Spector TD. Role of the gut microbiota in nutrition and health. BMJ. 2018;361:k2179. [CrossRef]
- Kasarello K, Cudnoch-Jedrzejewska A, Czarzasta K. Communication of gut microbiota and brain via immune and neuroendocrine signaling. Front Microbiol. 2023;14:1118529. [CrossRef]
- Nova E, Gómez-Martinez S, González-Soltero R. The Influence of Dietary Factors on the Gut Microbiota. Microorganisms. 2022;10(7):1368. [CrossRef]
- Qi X, Yun C, Pang Y, Qiao J. The impact of the gut microbiota on the reproductive and metabolic endocrine system. Gut Microbes. 2021;13(1):1-21. [CrossRef]
- Wang LQ. Mammalian phytoestrogens: enterodiol and enterolactone. J Chromatogr B Analyt Technol Biomed Life Sci. 2002;777(1-2):289-309. [CrossRef]
- Mukhija M, Joshi BC, Bairy PS, Bhargava A, Sah AN. Lignans: a versatile source of anticancer drugs. Beni Suef Univ J Basic Appl Sci. 2022;11(1):76. [CrossRef]
- Salliss ME, Farland LV, Mahnert ND, Herbst-Kralovetz MM. The role of gut and genital microbiota and the estrobolome in endometriosis, infertility and chronic pelvic pain. Hum Reprod Update. 2021;28(1):92-131. [CrossRef]
- Anderson G. Endometriosis Pathoetiology and Pathophysiology: Roles of Vitamin A, Estrogen, Immunity, Adipocytes, Gut Microbiome and Melatonergic Pathway on Mitochondria Regulation. Biomol Concepts. 2019;10(1):133-149. [CrossRef]
- Chakaroun RM, Massier L, Kovacs P. Gut Microbiome, Intestinal Permeability, and Tissue Bacteria in Metabolic Disease: Perpetrators or Bystanders? Nutrients. 2020;12(4): 1082. [CrossRef]
- Ata B, Yildiz S, Turkgeldi E, Brocal VP, Dinleyici EC, Moya A, Urman B. The Endobiota Study: Comparison of Vaginal, Cervical and Gut Microbiota Between Women with Stage 3/4 Endometriosis and Healthy Controls. Sci Rep. 2019;9(1):2204. [CrossRef]
- Jess T, Frisch M, Jørgensen KT, Pedersen BV, Nielsen NM. Increased risk of inflammatory bowel disease in women with endometriosis: a nationwide Danish cohort study. Gut. 2012; 61(9):1279-83. [CrossRef]
- Yuan M, Li D, Zhang Z, Sun H, An M, Wang G. Endometriosis induces gut microbiota alterations in mice. Hum Reprod. 2018;33(4):607-616. [CrossRef]
- Chadchan SB, Cheng M, Parnell LA, Yin Y, Schriefer A, Mysorekar IU, Kommagani R. Antibiotic therapy with metronidazole reduces endometriosis disease progression in mice: a potential role for gut microbiota. Hum Reprod. 2019;34(6):1106-1116. [CrossRef]
- Bailey MT, Coe CL. Endometriosis is associated with an altered profile of intestinal microflora in female rhesus monkeys. Hum Reprod. 2002;17(7):1704-8. [CrossRef]
- Qin R, Tian G, Liu J, Cao L. The gut microbiota and endometriosis: From pathogenesis to diagnosis and treatment. Front Cell Infect Microbiol. 2022;12:1069557. [CrossRef]
- Laganà AS, Garzon S, Franchi M, Casarin J, Gullo G, Ghezzi F. Translational animal models for endometriosis research: a long and windy road. Ann Transl Med. 2018;6(22):431. [CrossRef]
- Grümmer R. Animal models in endometriosis research. Hum Reprod Update. 2006;12(5):641-9. [CrossRef]
- Bruner-Tran KL, Osteen KG, Taylor HS, Sokalska A, Haines K, Duleba AJ. Resveratrol inhibits development of experimental endometriosis in vivo and reduces endometrial stromal cell invasiveness in vitro. Biol Reprod. 2011;84(1):106-12. [CrossRef]
- Rudzitis-Auth J, Menger MD, Laschke MW. Resveratrol is a potent inhibitor of vascularization and cell proliferation in experimental endometriosis. Hum Reprod. 2013;28 (5):1339-47. [CrossRef]
- Ricci AG, Olivares CN, Bilotas MA, Bastón JI, Singla JJ, Meresman GF, Barañao RI. Natural therapies assessment for the treatment of endometriosis. Hum Reprod. 2013;28(1):178-88. [CrossRef]
- Ozcan Cenksoy P, Oktem M, Erdem O, Karakaya C, Cenksoy C, Erdem A, Guner H, Karabacak O. A potential novel treatment strategy: inhibition of angiogenesis and inflammation by resveratrol for regression of endometriosis in an experimental rat model. Gynecol Endocrinol. 2015;31(3):219-24. [CrossRef]
- Di Paola R, Fusco R, Gugliandolo E, Crupi R, Evangelista M, Granese R, Cuzzocrea S. Co-micronized Palmitoylethanolamide/Polydatin Treatment Causes Endometriotic Lesion Regression in a Rodent Model of Surgically Induced Endometriosis. Front Pharmacol. 2016;7:382. [CrossRef]
- Cotroneo MS, Lamartiniere CA. Pharmacologic, but not dietary, genistein supports endometriosis in a rat model. Toxicol Sci. 2001;61(1):68-75. [CrossRef]
- Yavuz E, Oktem M, Esinler I, Toru SA, Zeyneloglu HB. Genistein causes regression of endometriotic implants in the rat model. Fertil Steril. 2007;88(4 Suppl):1129-34. [CrossRef]
- Takaoka O, Mori T, Ito F, Okimura H, Kataoka H, Tanaka Y, Koshiba A, Kusuki I, Shigehiro S, Amami T, Kitawaki J. Daidzein-rich isoflavone aglycones inhibit cell growth and inflammation in endometriosis. J Steroid Biochem Mol Biol. 2018;181:125-132. [CrossRef]
- Chen Y, Chen C, Shi S, Han J, Wang J, Hu J, Liu Y, Cai Z, Yu C. Endometriotic implants regress in rat models treated with puerarin by decreasing estradiol level. Reprod Sci. 2011;18 (9):886-91. [CrossRef]
- Rudzitis-Auth J, Körbel C, Scheuer C, Menger MD, Laschke MW. Xanthohumol inhibits growth and vascularization of developing endometriotic lesions. Hum Reprod. 2012 ;27(6): 1735-44. [CrossRef]
- Nahari E, Razi M. Silymarin amplifies apoptosis in ectopic endometrial tissue in rats with endometriosis; implication on growth factor GDNF, ERK1/2 and Bcl-6b expression. Acta Histochem. 2018;120(8):757-767. [CrossRef]
- Melekoglu R, Ciftci O, Eraslan S, Cetin A, Basak N. The beneficial effects of nerolidol and hesperidin on surgically induced endometriosis in a rat model. Gynecol Endocrinol. 2018; 34(11):975-980. [CrossRef]
- Hsu YW, Chen HY, Chiang YF, Chang LC, Lin PH, Hsia SM. The effects of isoliquiritigenin on endometriosis in vivo and in vitro study. Phytomedicine. 2020;77:153214. [CrossRef]
- Yang YM, Yang WX. Epithelial-to-mesenchymal transition in the development of endometriosis. Oncotarget. 2017;8(25):41679-41689. [CrossRef]
- Kapoor R, Sirohi VK, Gupta K, Dwivedi A. Naringenin ameliorates progression of endometriosis by modulating Nrf2/Keap1/HO1 axis and inducing apoptosis in rats. J Nutr Biochem. 2019;70:215-226. [CrossRef]
- Ilhan M, Ali Z, Khan IA, Taştan H, Küpeli Akkol E. Bioactivity-guided isolation of flavonoids from Urtica dioica L. and their effect on endometriosis rat model. J Ethnopharmacol. 2019;243:112100. [CrossRef]
- Demirel MA, Suntar I, Ilhan M, Keles H, Kupeli Akkol E. Experimental endometriosis remission in rats treated with Achillea biebersteinii Afan.: histopathological evaluation and determination of cytokine levels. Eur J Obstet Gynecol Reprod Biol. 2014;175:172-7. [CrossRef]
- Ferella L, Bastón JI, Bilotas MA, Singla JJ, González AM, Olivares CN, Meresman GF. Active compounds present inRosmarinus officinalis leaves andScutellaria baicalensis root evaluated as new therapeutic agents for endometriosis. Reprod Biomed Online. 2018;37(6): 769-782. [CrossRef]
- Ilhan M, Ali Z, Khan IA, Taştan H, Küpeli Akkol E. Promising activity of Anthemis austriaca Jacq. on the endometriosis rat model and isolation of its active constituents. Saudi Pharm J. 2019;27(6):889-899. [CrossRef]
- Bina F, Daglia M, Santarcangelo C, Baeeri M, Abdollahi M, Nabavi SM, Tabarrai M, Rahimi R. Phytochemical profiling and ameliorative effects of Achillea cretica L. on rat model of endometriosis. J Ethnopharmacol. 2020;254:112747. [CrossRef]
- Ilhan M, Ali Z, Khan IA, Taştan H, Küpeli Akkol E. The regression of endometriosis with glycosylated flavonoids isolated from Melilotus officinalis (L.) Pall. in an endometriosis rat model. Taiwan J Obstet Gynecol. 2020;59(2):211-219. [CrossRef]
- Machairiotis N, Vasilakaki S, Kouroutou P. Natural products: Potential lead compounds for the treatment of endometriosis. Eur J Obstet Gynecol Reprod Biol. 2020;245:7-12. [CrossRef]
- Ilhan M, Gürağaç Dereli FT, Akkol EK. Novel Drug Targets with Traditional Herbal Medicines for Overcoming Endometriosis. Curr Drug Deliv. 2019;16(5):386-399. [CrossRef]
- Bina F, Soleymani S, Toliat T, Hajimahmoodi M, Tabarrai M, Abdollahi M, Rahimi R. Plant-derived medicines for treatment of endometriosis: A comprehensive review of molecular mechanisms. Pharmacol Res. 2019;139:76-90. [CrossRef]
- Parazzini F, Viganò P, Candiani M, Fedele L. Diet and endometriosis risk: a literature review. Reprod Biomed Online. 2013;26(4):323-36. [CrossRef]
- Della Corte L, Noventa M, Ciebiera M, Magliarditi M, Sleiman Z, Karaman E, Catena U, Salvaggio C, Falzone G, Garzon S. Phytotherapy in endometriosis: an up-to-date review. J Complement Integr Med. 2020;17(3). [CrossRef]
- Kodarahmian M, Amidi F, Moini A, Kashani L, Shabani Nashtaei M, Pazhohan A, Bahramrezai M, Berenjian S, Sobhani A. The modulating effects of Resveratrol on the expression of MMP-2 and MMP-9 in endometriosis women: a randomized exploratory trial. Gynecol Endocrinol. 2019;35(8):719-726. [CrossRef]
- Maia H Jr, Haddad C, Pinheiro N, Casoy J. Advantages of the association of resveratrol with oral contraceptives for management of endometriosis-related pain. Int J Womens Health. 2012;4:543-9. [CrossRef]
- Mendes da Silva D, Gross LA, Neto EPG, Lessey BA, Savaris RF. The Use of Resveratrol as an Adjuvant Treatment of Pain in Endometriosis: A Randomized Clinical Trial. J Endocr Soc. 2017;1(4):359-369. [CrossRef]
- Nagata C, Takatsuka N, Kawakami N, Shimizu H. Soy product intake and premenopausal hysterectomy in a follow-up study of Japanese women. Eur J Clin Nutr. 2001;55(9):773-7. [CrossRef]
- Parazzini F, Chiaffarino F, Surace M, Chatenoud L, Cipriani S, Chiantera V, Benzi G, Fedele L. Selected food intake and risk of endometriosis. Hum Reprod. 2004;19(8):1755-9. [CrossRef]
- Signorile PG, Viceconte R, Baldi A. Novel dietary supplement association reduces symptoms in endometriosis patients. J Cell Physiol. 2018;233(8):5920-5925. [CrossRef]
- Trabert B, Peters U, De Roos AJ, Scholes D, Holt VL. Diet and risk of endometriosis in a population-based case-control study. Br J Nutr. 2011;105(3):459-67. [CrossRef]
- Tsuchiya M, Miura T, Hanaoka T, Iwasaki M, Sasaki H, Tanaka T, Nakao H, Katoh T, Ikenoue T, Kabuto M, Tsugane S. Effect of soy isoflavones on endometriosis: interaction with estrogen receptor 2 gene polymorphism. Epidemiology. 2007;18(3):402-8. [CrossRef]
- Youseflu S, Jahanian Sadatmahalleh SH, Mottaghi A, Kazemnejad A. Dietary Phytoestrogen Intake and The Risk of Endometriosis in Iranian Women: A Case-Control Study. Int J Fertil Steril. 2020;13(4):296-300. [CrossRef]
- Harris HR, Eke AC, Chavarro JE, Missmer SA. Fruit and vegetable consumption and risk of endometriosis. Hum Reprod. 2018;33(4):715-727. [CrossRef]
- Mirmiran P, Bahadoran Z, Gaeini Z. Common Limitations and Challenges of Dietary Clinical Trials for Translation into Clinical Practices. Int J Endocrinol Metab. 2021;19(3): e108170. [CrossRef]
- Dull AM, Moga MA, Dimienescu OG, Sechel G, Burtea V, Anastasiu CV. Therapeutic Approaches of Resveratrol on Endometriosis via Anti-Inflammatory and Anti-Angiogenic Pathways. Molecules. 2019;24(4):667. [CrossRef]
- Meresman GF, Götte M, Laschke MW. Plants as source of new therapies for endometriosis: a review of preclinical and clinical studies. Hum Reprod Update. 2021; 27(2):367-392. [CrossRef]
- Afrin S, AlAshqar A, El Sabeh M, Miyashita-Ishiwata M, Reschke L, Brennan JT, Fader A, Borahay MA. Diet and Nutrition in Gynecological Disorders: A Focus on Clinical Studies. Nutrients. 2021;13(6):1747. [CrossRef]
- Chapadgaonkar SS, Bajpai SS, Godbole MS. Gut microbiome influences incidence and outcomes of breast cancer by regulating levels and activity of steroid hormones in women. Cancer Rep (Hoboken). 2023:e1847. [CrossRef]
- Lathigara D, Kaushal D, Wilson RB. Molecular Mechanisms of Western Diet-Induced Obesity and Obesity-Related Carcinogenesis-A Narrative Review. Metabolites. 2023; 13(5):675. [CrossRef]
- Saguyod SJU, Kelley AS, Velarde MC, Simmen RC. Diet and endometriosis-revisiting the linkages to inflammation. Journal of Endometriosis and Pelvic Pain Disorders. 2018;10(2):51-58. [CrossRef]
- Arumugam K, Templeton AA. Endometriosis and race. Aust N Z J Obstet Gynaecol. 1992;32:164–165. [CrossRef]
- Yen CF, Kim MR, Lee CL. Epidemiologic Factors Associated with Endometriosis in East Asia. Gynecol Minim Invasive Ther. 2019;8(1):4-11. [CrossRef]
- Yamamoto A, Johnstone EB, Bloom MS, Huddleston HG, Fujimoto VY. A higher prevalence of endometriosis among Asian women does not contribute to poorer IVF outcomes. J Assist Reprod Genet. 2017;34(6):765-774. [CrossRef]
- Gajbhiye RK. Endometriosis and inflammatory immune responses: Indian experience. Am J Reprod Immunol. 2023;89(2):e13590. [CrossRef]
- Moradi Y, Shams-Beyranvand M, Khateri S, Gharahjeh S, Tehrani S, Varse F, Tiyuri A, Najmi Z. A systematic review on the prevalence of endometriosis in women. Indian J Med Res. 2021;154(3):446-454. [CrossRef]
- Chottanapund S, Van Duursen MB, Navasumrit P, Hunsonti P, Timtavorn S, Ruchirawat M, Van den Berg M. Anti-aromatase effect of resveratrol and melatonin on hormonal positive breast cancer cells co-cultured with breast adipose fibroblasts. Toxicol In Vitro. 2014; 28(7): 1215-21. [CrossRef]
- Klein CB, King AA. Genistein genotoxicity: critical considerations of in vitro exposure dose. Toxicol Appl Pharmacol. 2007;224(1):1-11. [CrossRef]
- Landete JM, Arqués J, Medina M, Gaya P, de Las Rivas B, Muñoz R. Bioactivation of Phytoestrogens: Intestinal Bacteria and Health. Crit Rev Food Sci Nutr. 2016;56(11):1826-43. [CrossRef]
- Ervin SM, Li H, Lim L, Roberts LR, Liang X, Mani S, Redinbo MR. Gut microbial β-glucuronidases reactivate estrogens as components of the estrobolome that reactivate estrogens. J Biol Chem. 2019;294(49):18586-18599. [CrossRef]
- Tew BY, Xu X, Wang HJ, Murphy PA, Hendrich S. A diet high in wheat fiber decreases the bioavailability of soybean isoflavones in a single meal fed to women. J Nutr. 1996; 126(4):871-7. [CrossRef]
- Baldi S, Tristán Asensi M, Pallecchi M, Sofi F, Bartolucci G, Amedei A. Interplay between Lignans and Gut Microbiota: Nutritional, Functional and Methodological Aspects. Molecules. 2023;28(1):343. [CrossRef]
- Baker JM, Al-Nakkash L, Herbst-Kralovetz MM. Estrogen-gut microbiome axis: Physiological and clinical implications. Maturitas. 2017;103:45-53. [CrossRef]
- Adlercreutz H, Höckerstedt K, Bannwart C, Bloigu S, Hämäläinen E, Fotsis T, Ollus A. Effect of dietary components, including lignans and phytoestrogens, on enterohepatic circulation and liver metabolism of estrogens and on sex hormone binding globulin (SHBG). J Steroid Biochem. 1987;27(4-6):1135-44. [CrossRef]
- Nap A, de Roos N. Endometriosis and the effects of dietary interventions: what are we looking for? Reprod Fertil. 2022;3(2):C14-C22. [CrossRef]
- Gołąbek A, Kowalska K, Olejnik A. Polyphenols as a Diet Therapy Concept for Endometriosis-Current Opinion and Future Perspectives. Nutrients. 2021;13(4):1347. [CrossRef]
- Amro B, Ramirez Aristondo ME, Alsuwaidi S, Almaamari B, Hakim Z, Tahlak M, Wattiez A, Koninckx PR. New Understanding of Diagnosis, Treatment and Prevention of Endometriosis. Int J Environ Res Public Health. 2022;19(11):6725. [CrossRef]
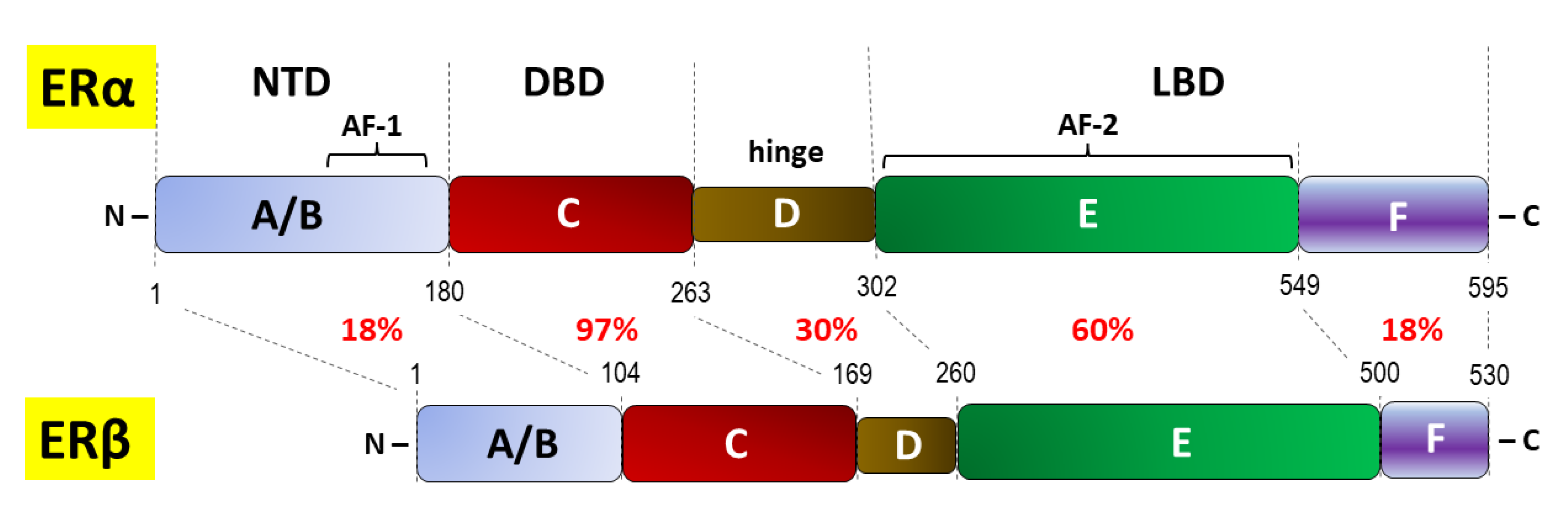
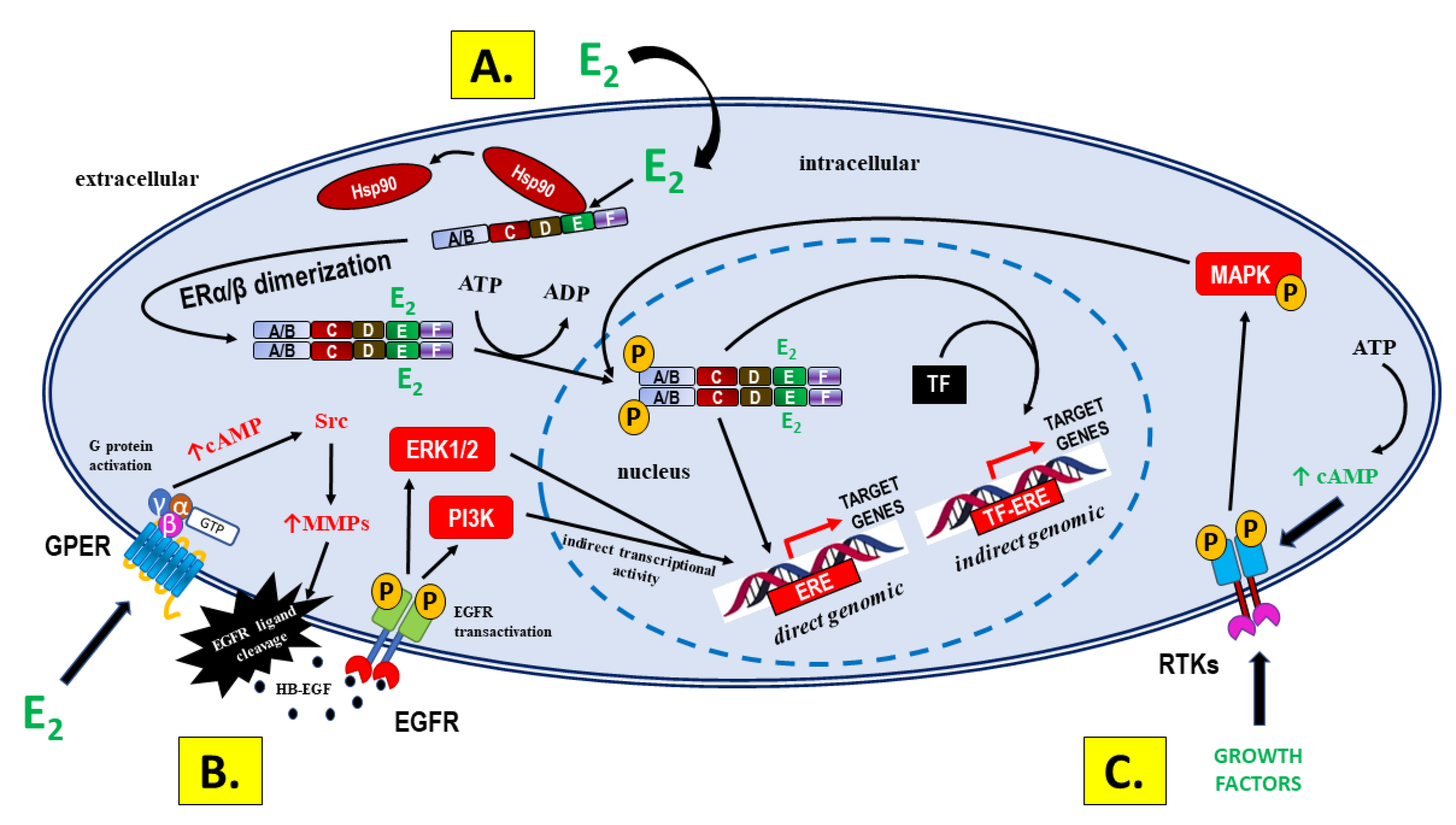
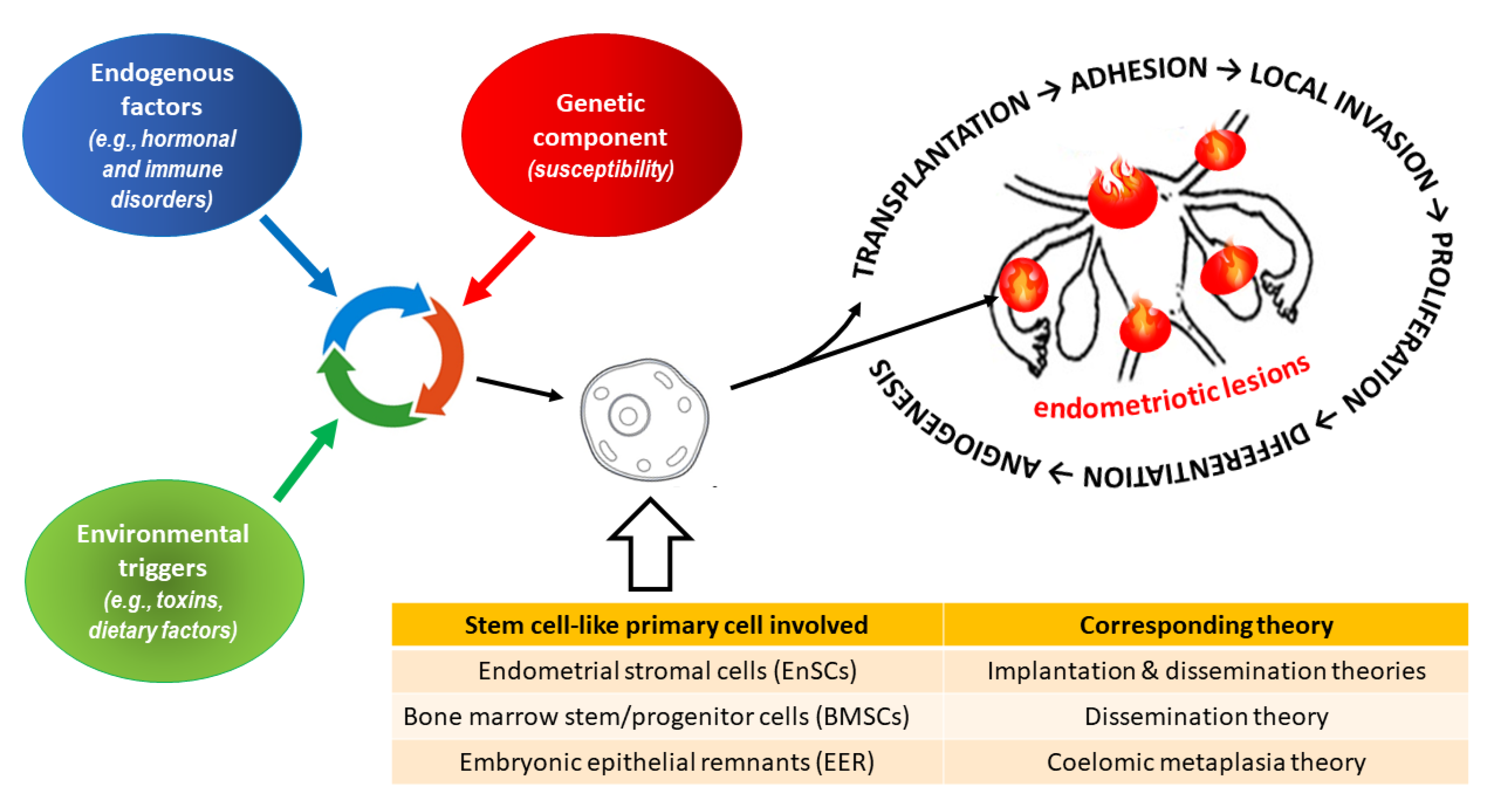
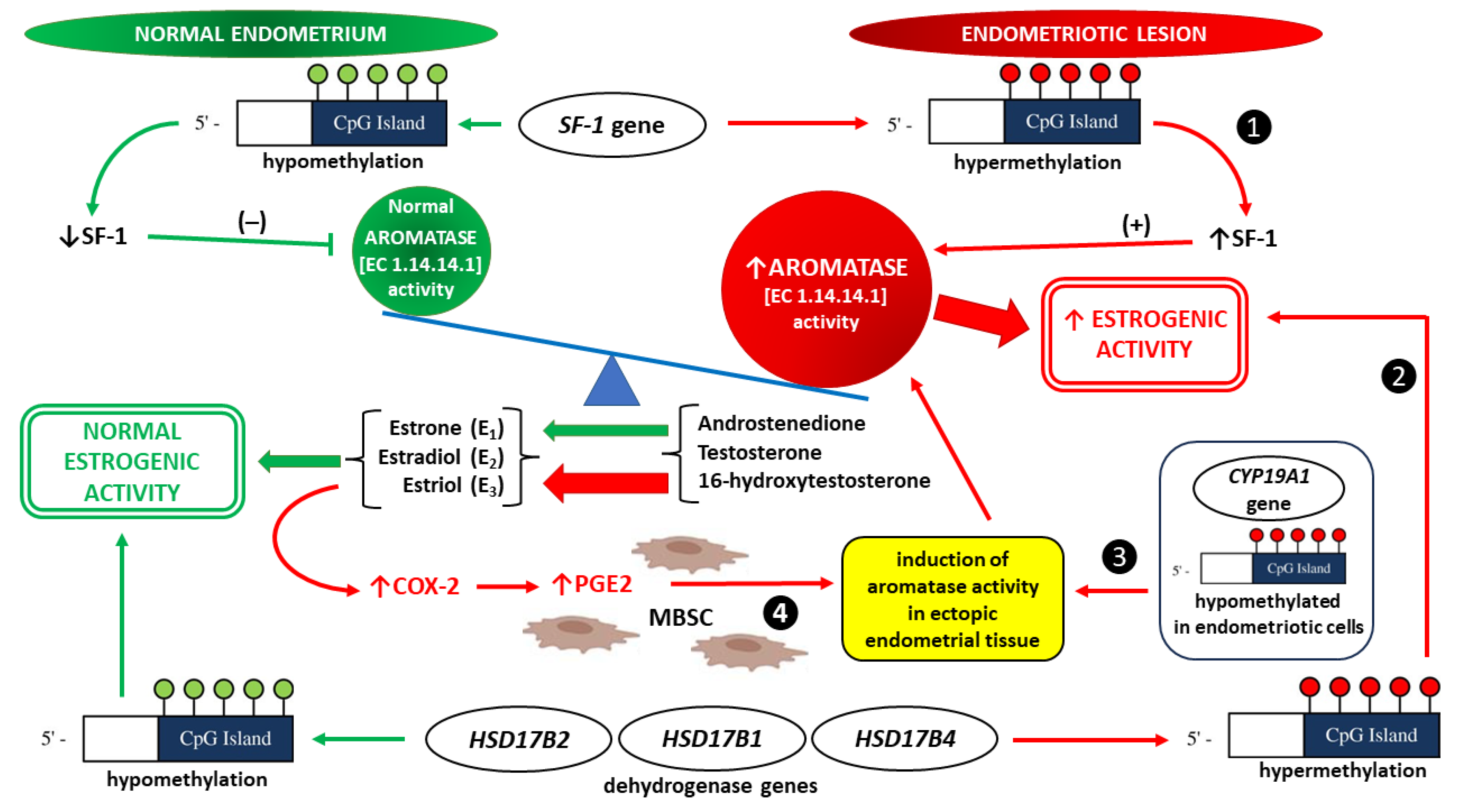
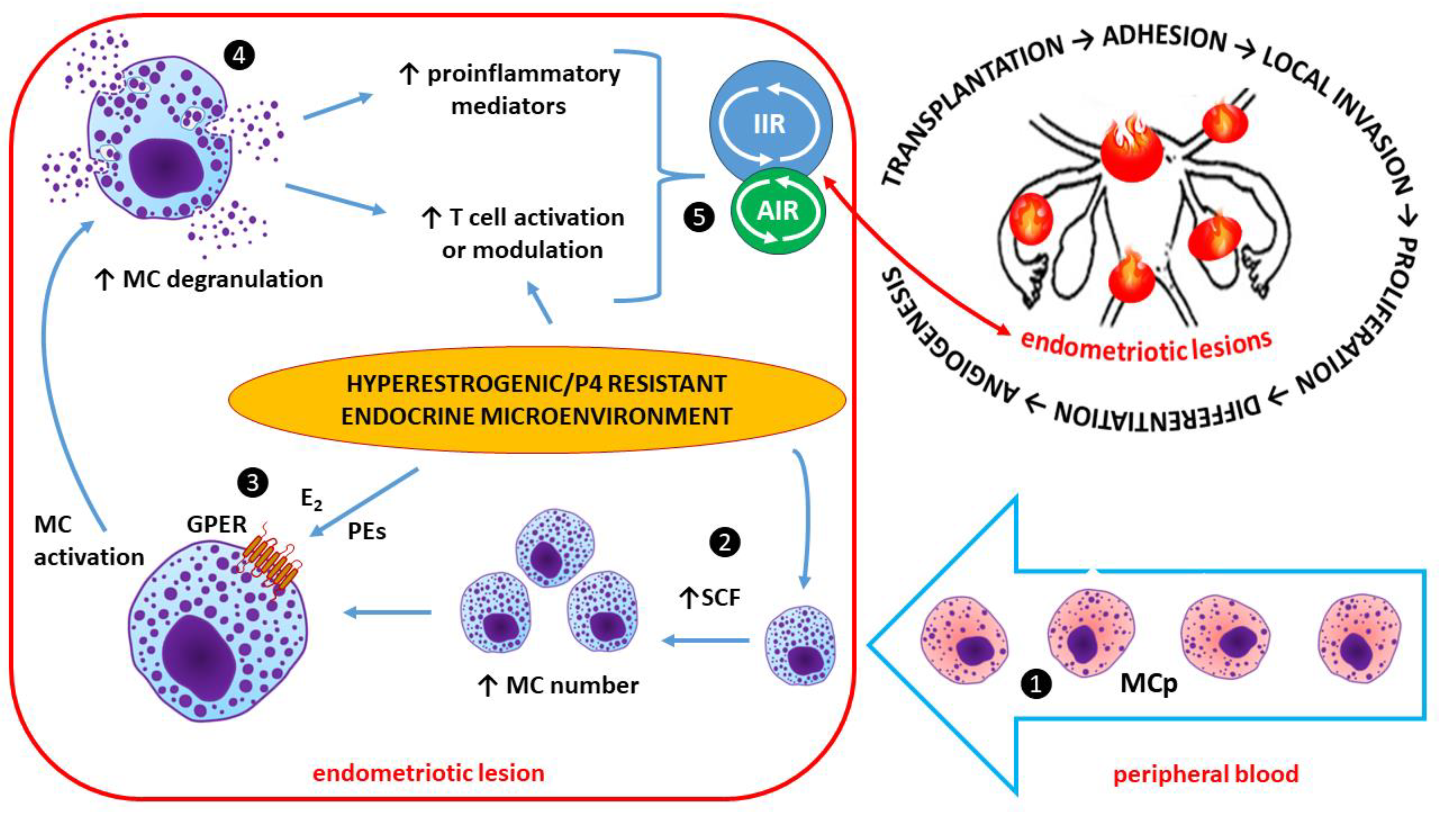
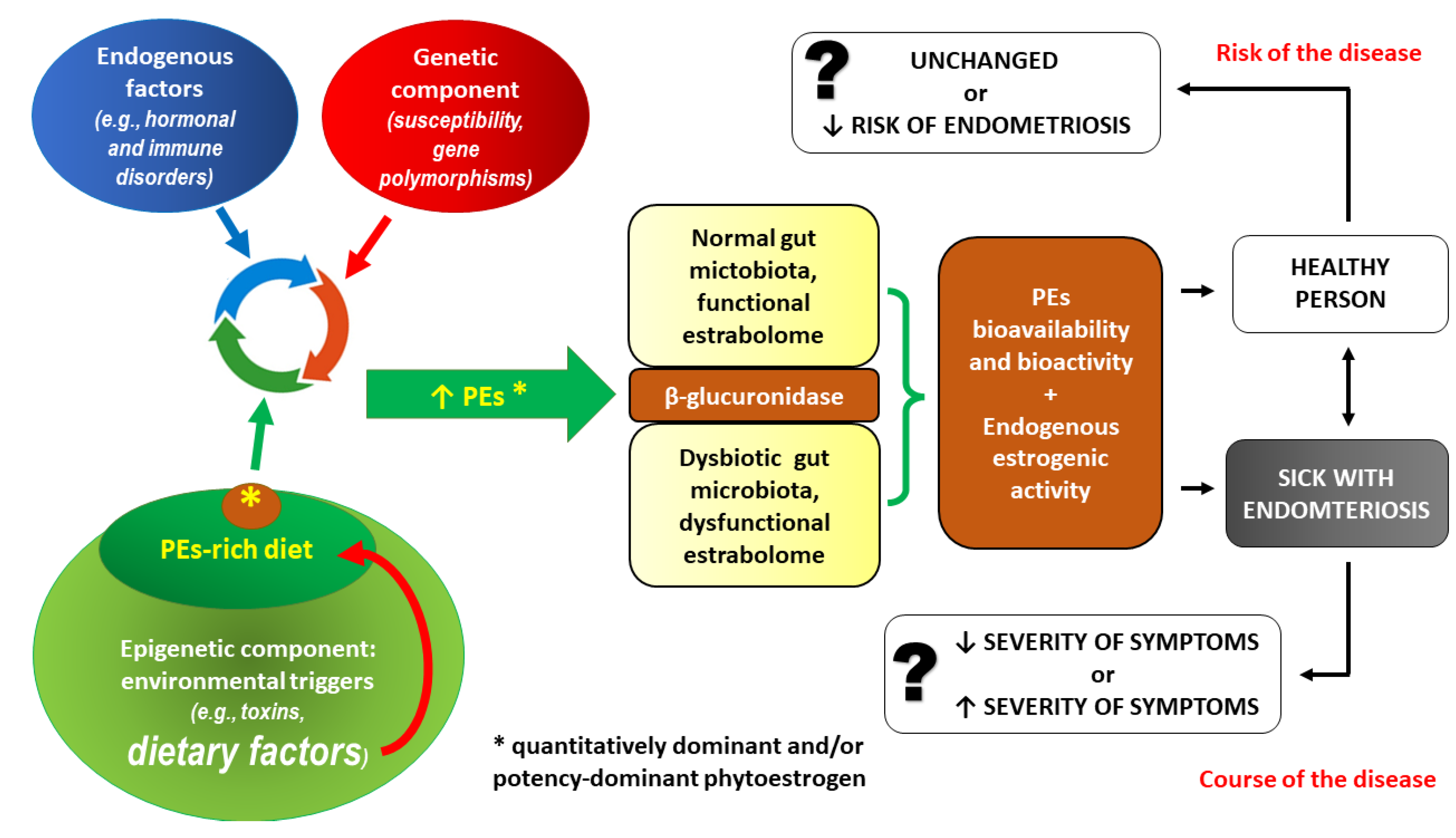
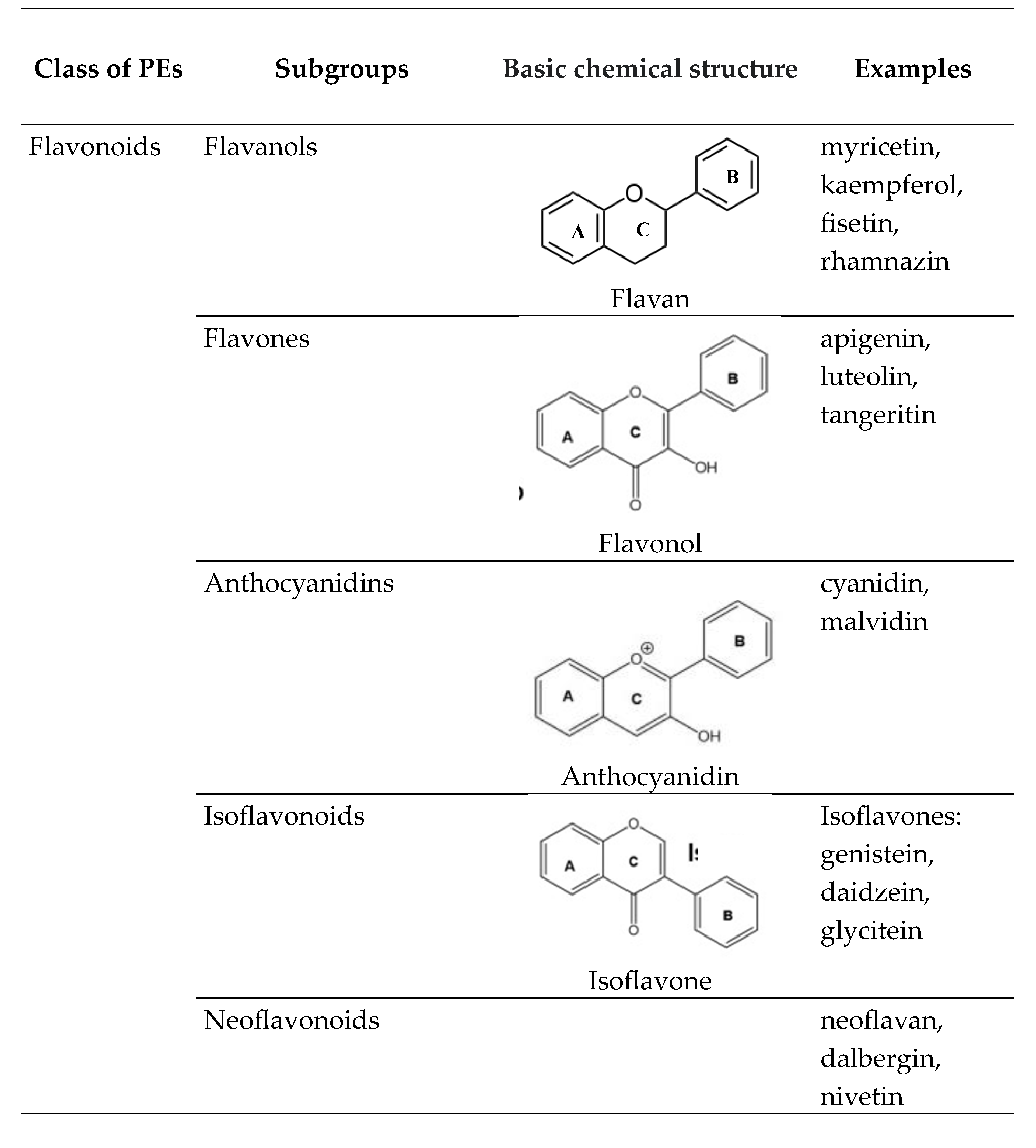
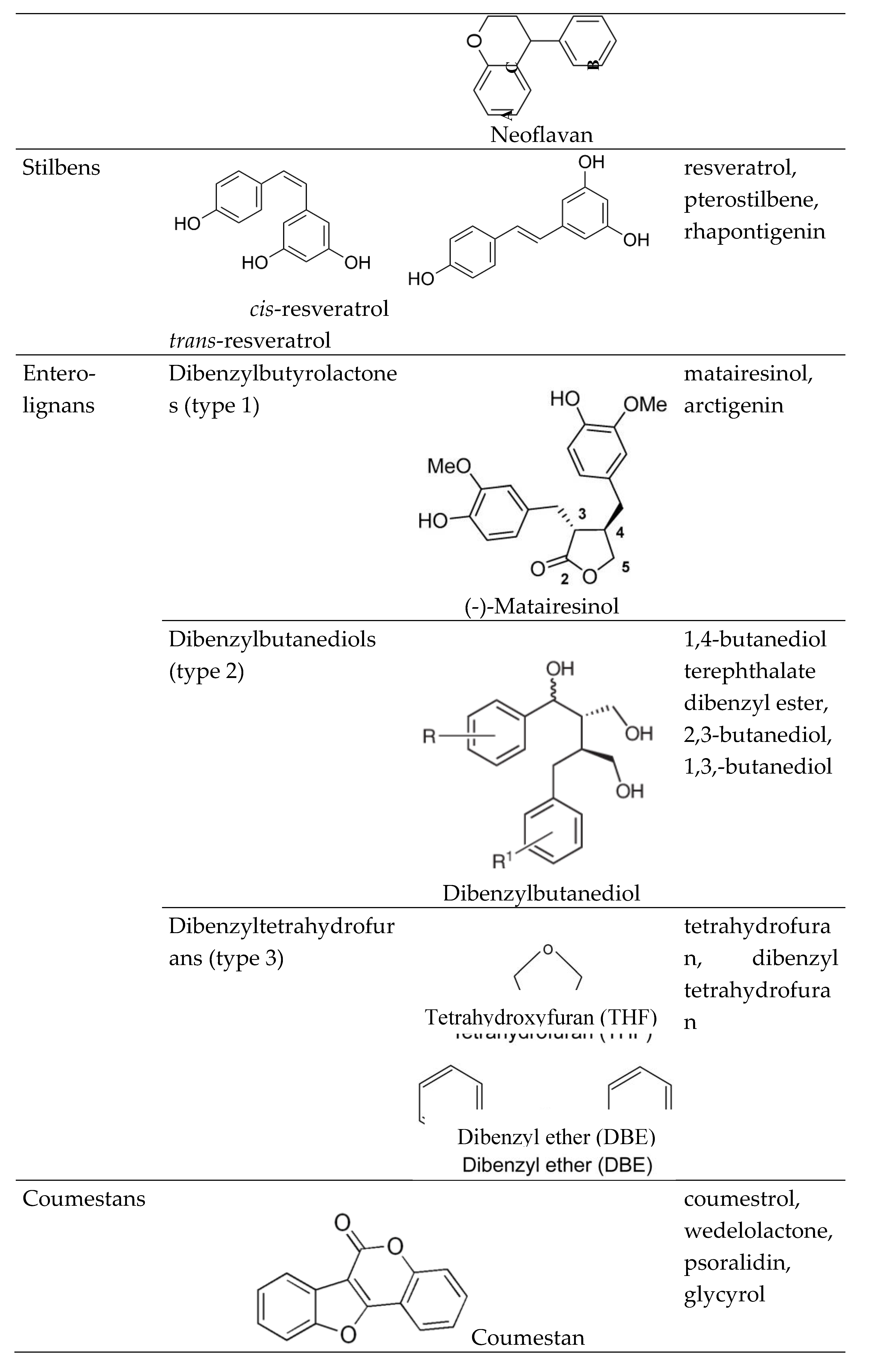
| Authors | Year | Type of the study | Compound(s), duration | Sample size (n) | Age range (years, mean) | Control (n) | Main results (p < 0.05) | LoE * |
| Kodarahmian et al. [394] | 2019 | Placebo-controlled, randomized, double-blind clinical trial | resveratrol 400 mg; 12 – 14 weeks | 17 | 18-37 (30.19 ±2.4) | 17 (placebo) | - ↓ level of mRNA and protein of both MMP-2 and MMP-9; - ↓ concentration of MMP-2 and MMP-9 in the serum and the endome-trial fluid | II |
| Maia Jr et al. [395] | 2012 | retrospective study | resveratrol 30 mg; 2 – 6 months | 26 using OC | 24-40 (31 ±4.0) | 16 using OC | - ↓ pain (82% of patients reporting complete resolution of dysmenorrhea and pelvic pain after 2 months); - ↓ expressions of both COX-2 and aromatase in eutopic endometrium | II |
| Mendes da Silva et al. [396] | 2017 | randomized clinical trial | resveratrol 40 mg; 42 days | 22 using MOC | 20-50 (35.4 ±7.1) | 22 using MOC (placebo) | - NO DIFFERENCE in median pain scores between the groups; resveratrol is not superior to placebo for treatment of pain in endometriosis | III |
| Nagata et al. [397] | 2001 | prospective cohort study | soy isoflavones: daidzein and genistein; 6 years | 1172 | 35-54 (42.9 ±4.4) | N/A | - ↓ risk of premenopausal hysterectomy: RR (95% CI) 0.35 (0.13-0.97) | II |
| Parazzini et al. [398] | 2004 | two case-control studies | PE-rich vs. low-PE diet; 15-year data | 504 | Cases: 20-65 (33 ±3.3) Controls: 20-61 (34 ± 2.9) | 504 | - ↓ risk of endometriosis for PE-rich diet (OR = 0.3 for the highest tertile of intake for green vegetables, and OR = 0.6 for fresh fruit) | III |
| Signorile et al. [399] | 2018 | Prospective, placebo-controlled, cohort study | dietary supplement containing quer-cetin (200 mg), curcumin (turmeric curcumin 20 mg), parthenium (19.5 mg); 3 months | 34 | NP | 30 (placebo) | - ↓ symptoms in endometriosis: dysmenorrhea and chronic pelvic pain (both from 62% to 18 %), dyspareunia (from 30% to 15%); - ↓ serum levels of PGE2 and CA-125 | III |
| Trabert B et al. [400] | 2011 | population-based case control study | overall intake of fruits and vegetables, dairy, vegetable, fruit (excluding fruit juice), whole grains, legumes, red meat, poultry, fatty fish, nonfatty fish and seafood; 60 months | 284 | Cases: 18-49 (NP) Controls: 18-49 (NP) |
660 (randomly selected, without a history of endometriosis) | - ↑ risk of endometriosis positively correlated with β-carotene consumption and servings/d of fruit, whereas - vegetable intake was NOT ASSOCIATED with endometriosis risk |
I |
| Tsuchiya et al. [401] | 2007 | case-control study | urinary levels of soy isoflavones: daidzein and genistein; 24 months of recruiting period | 79 (stage I-II: 31) (stage III-IV: 48) |
20-45 (stage I-II: 32.3 ±3.2) (stage III-IV: 32.6 ±3.7) | 59 | - ↑ urinary level of isoflavones was inversely associated with both the risk of advanced endometriosis (stage III-IV) and severity of endometriosis; - for advanced endometriosis, ER2 gene RsaI polymorphism significantly modifies the effects of genistein | III |
| Youseflu et al. [402] | 2020 | case-control study on dietary data | isoflavones, lignans, and coumestrol; 12 months | 78 | 15-45 (31.01 ±6.56) | 78 | - ↓ risk of endometriosis for isoflavones, lignans, and coumestrol | III |
| Harris et al. [403] | 2018 | prospective cohort study | intake of fruits and vegetables; 22-year follow-up period | 70835 | 25-42 (NP) | N/A | - ↓ risk of endometriosis for higher fruit consumption, especially for citrus fruits - ↓ risk of endometriosis was positively correlated with β-Cryptoxanthin intake - No association between total vegetable intake and endometriosis risk. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




