Submitted:
13 July 2023
Posted:
14 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Change of Survivability and Body Weight.
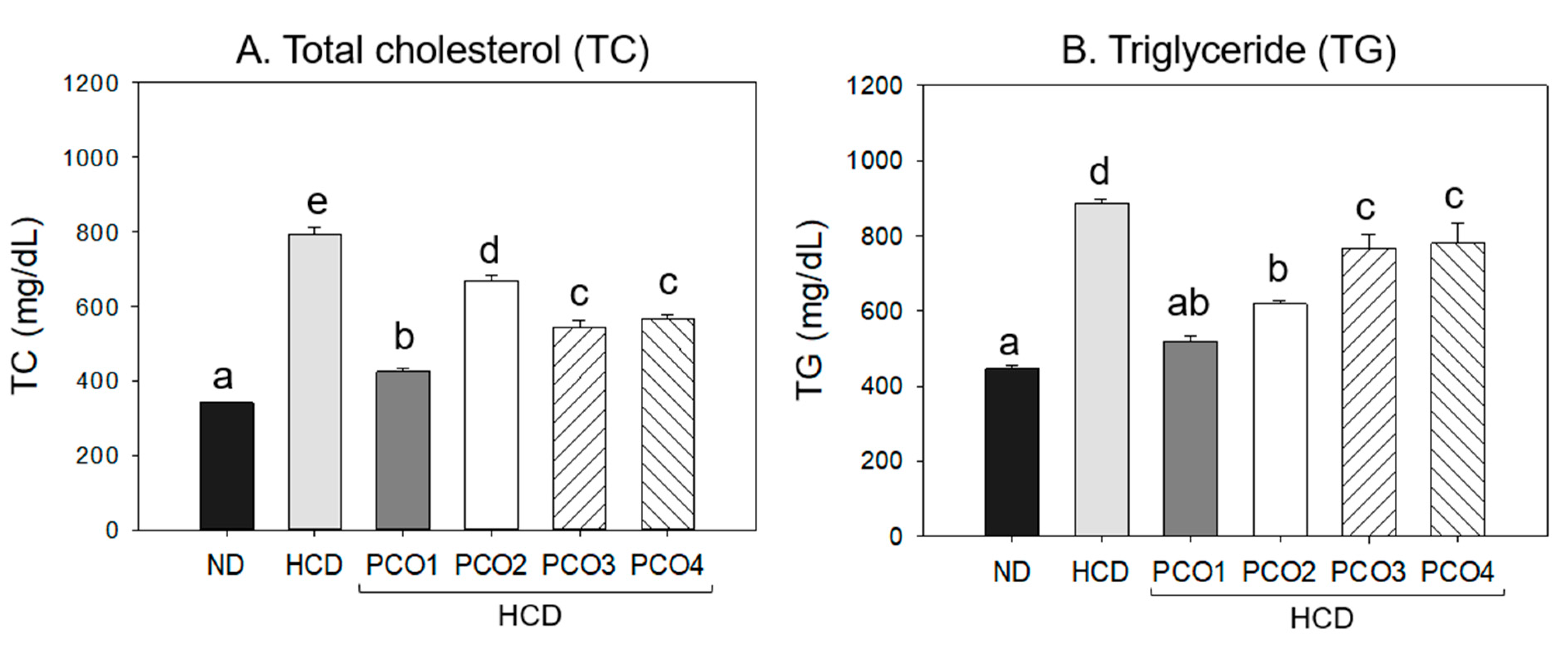
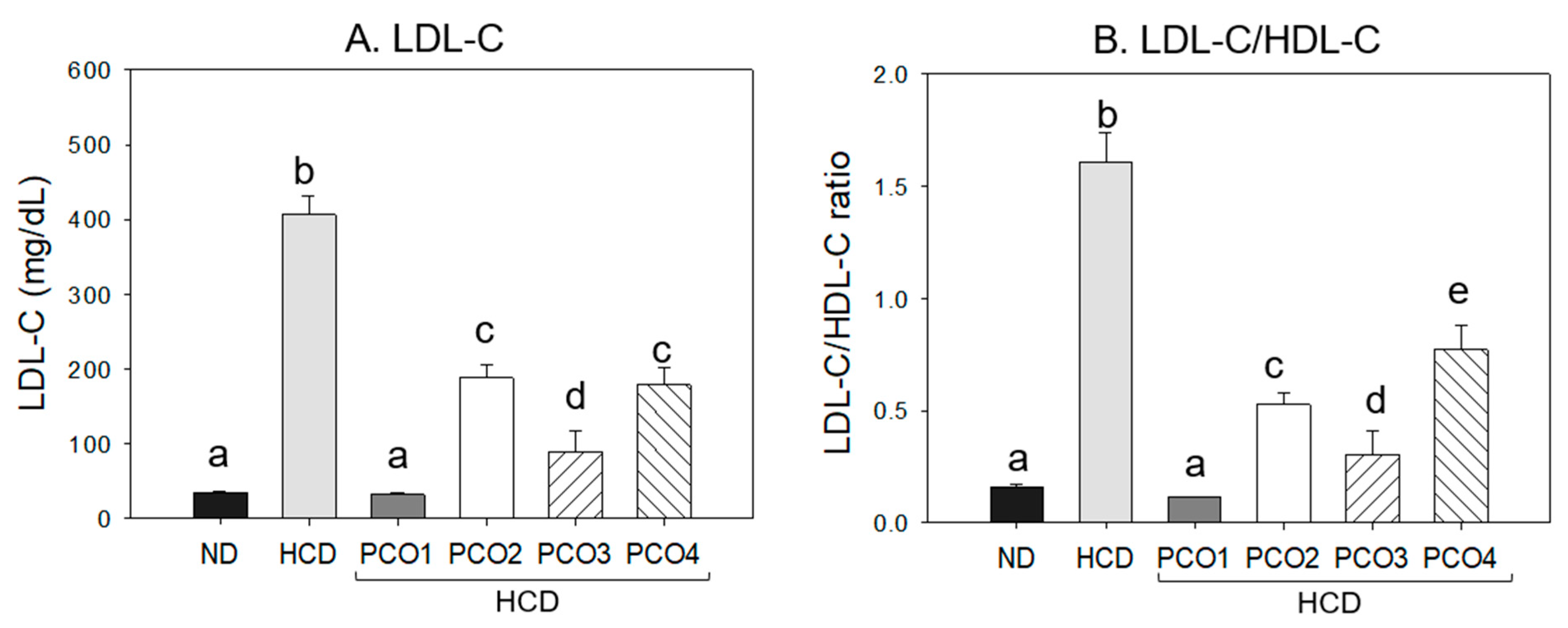
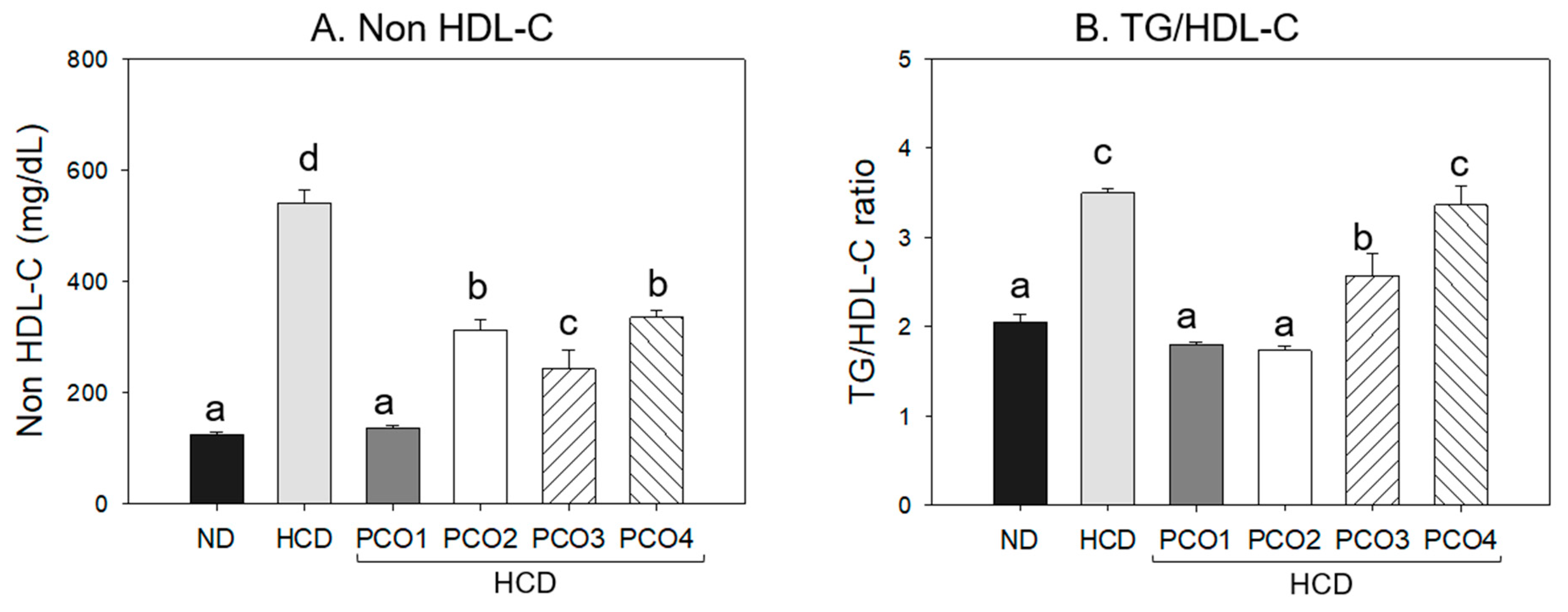
2.2. Change in Blood Lipid Levels
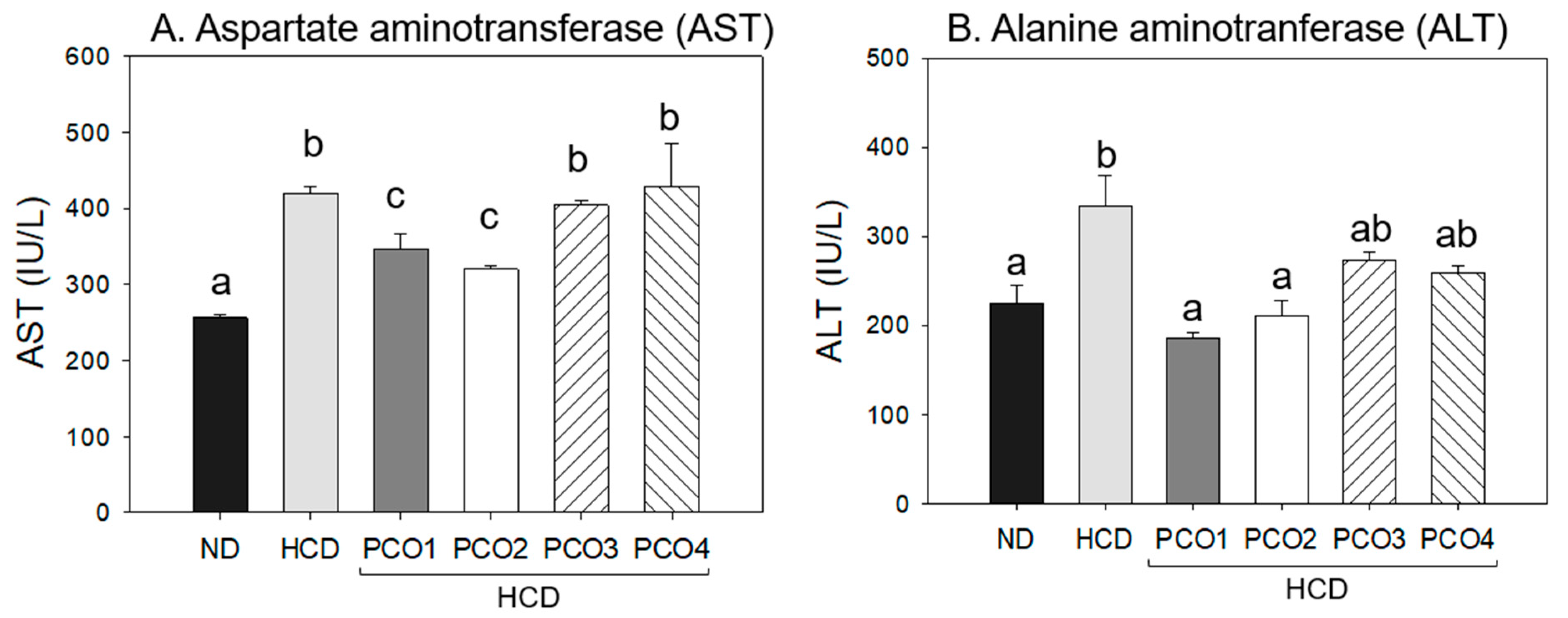
2.3. Change of Hepatic Function Parameters
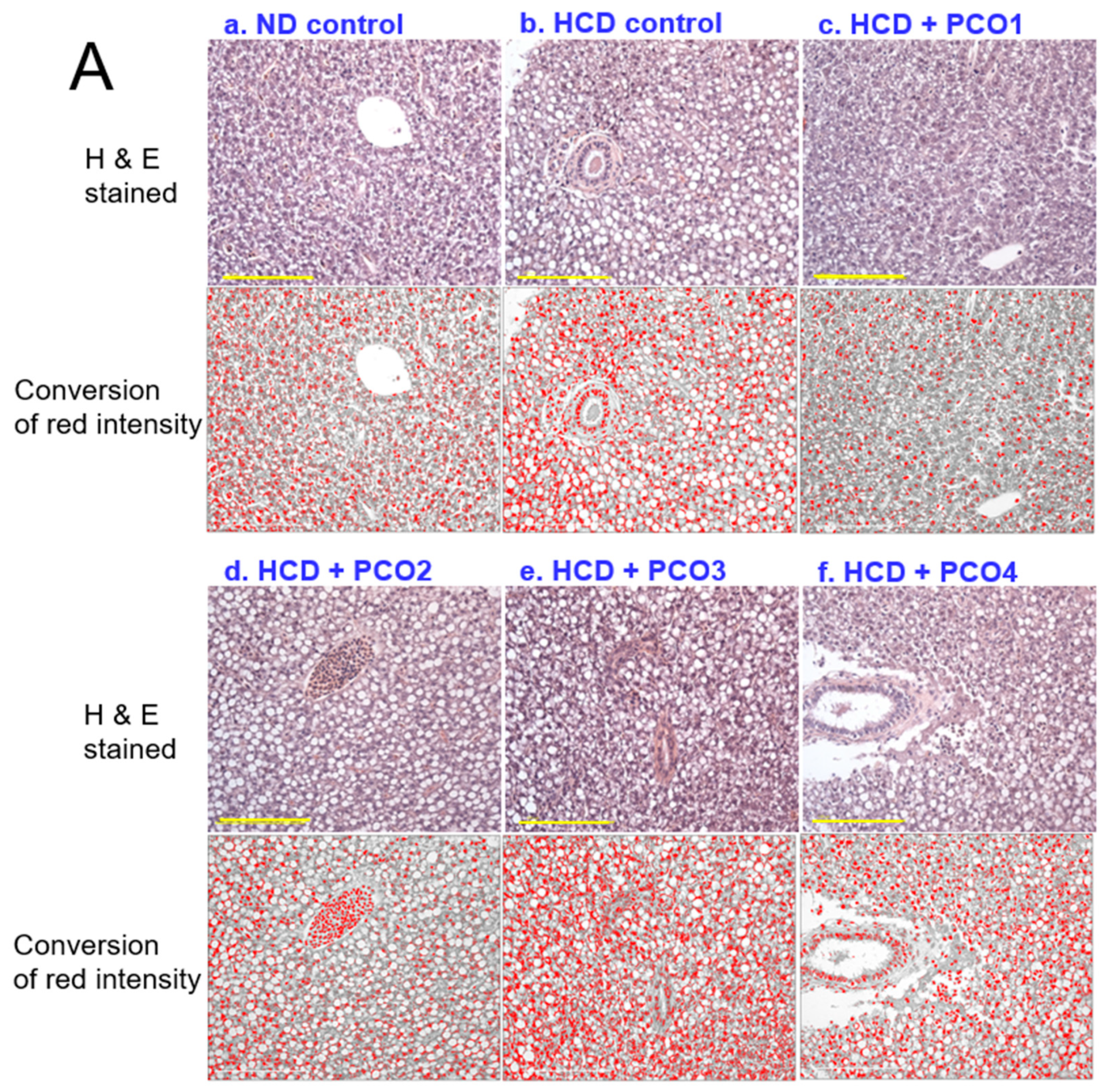
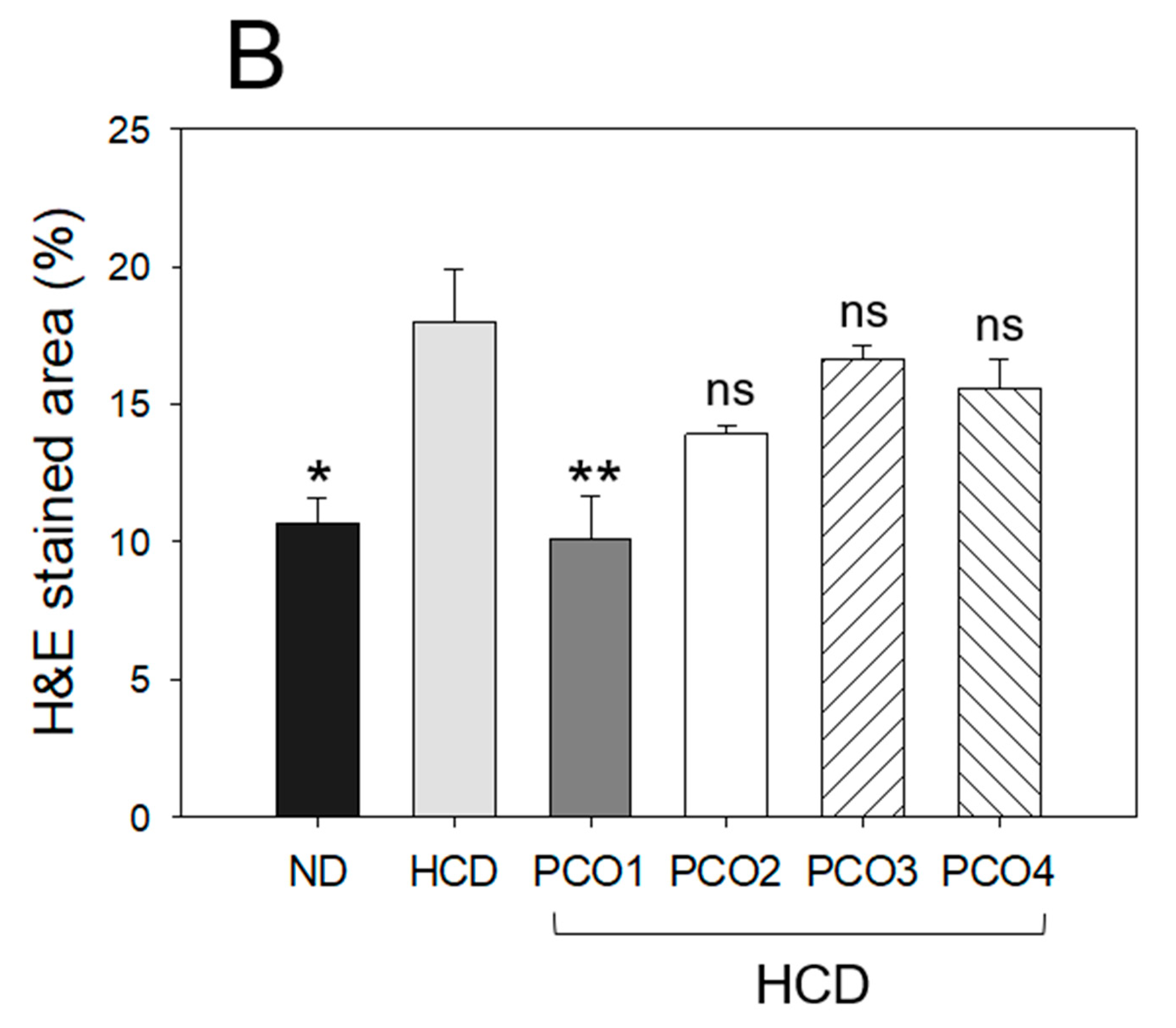
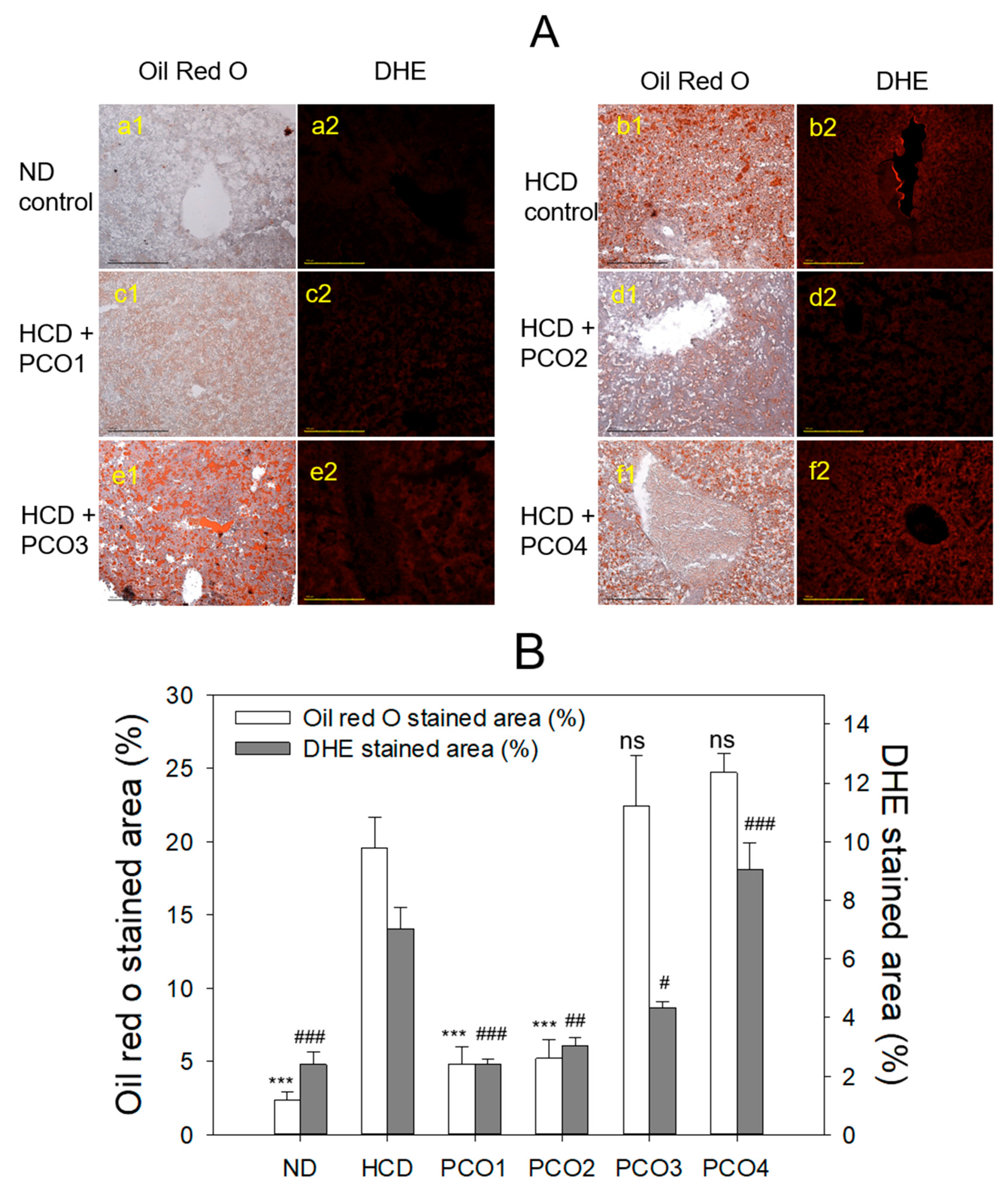
2.4. Histopathological Analysis with Liver
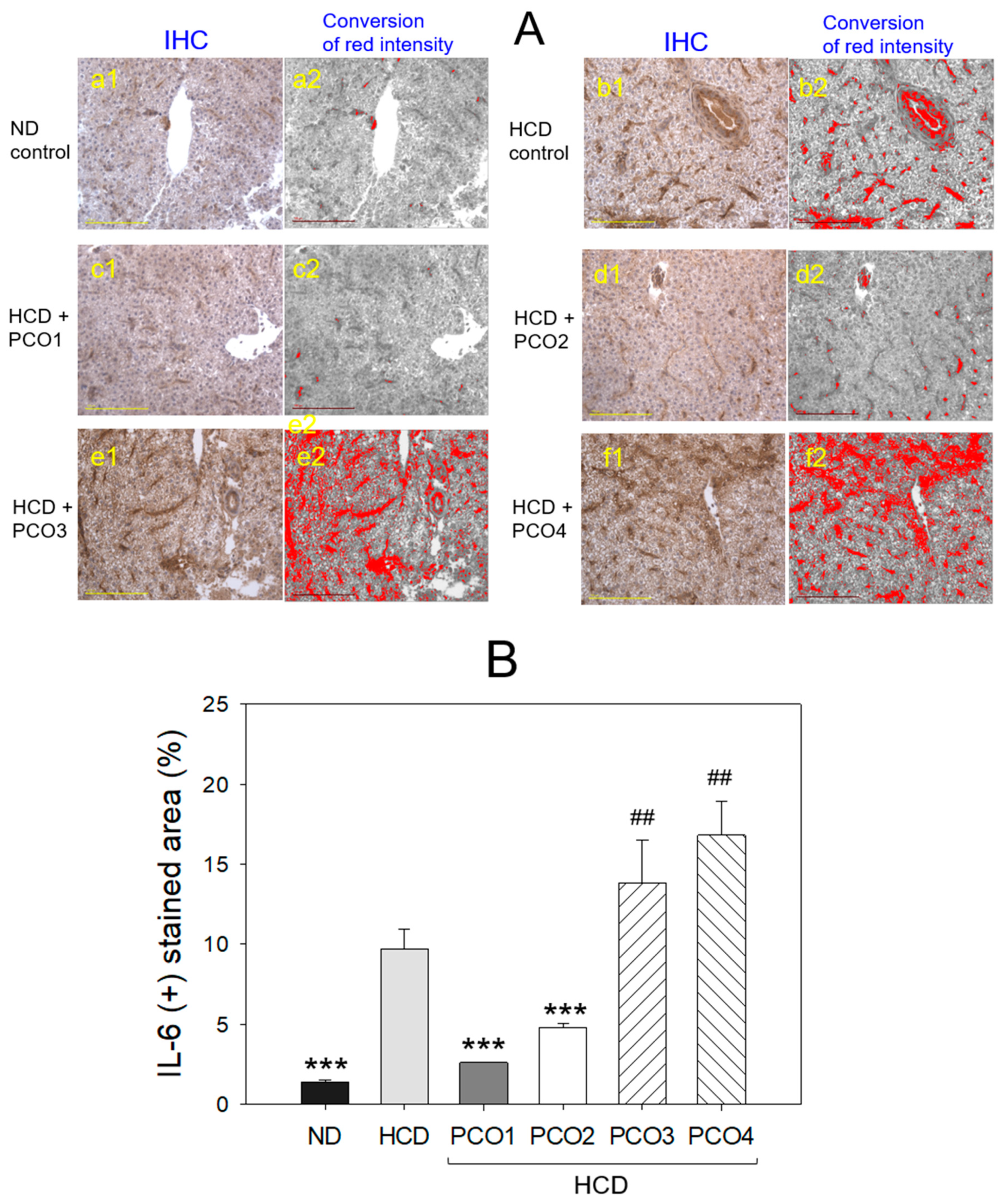
2.5. Immunohistochemical Analysis with Liver Tissue
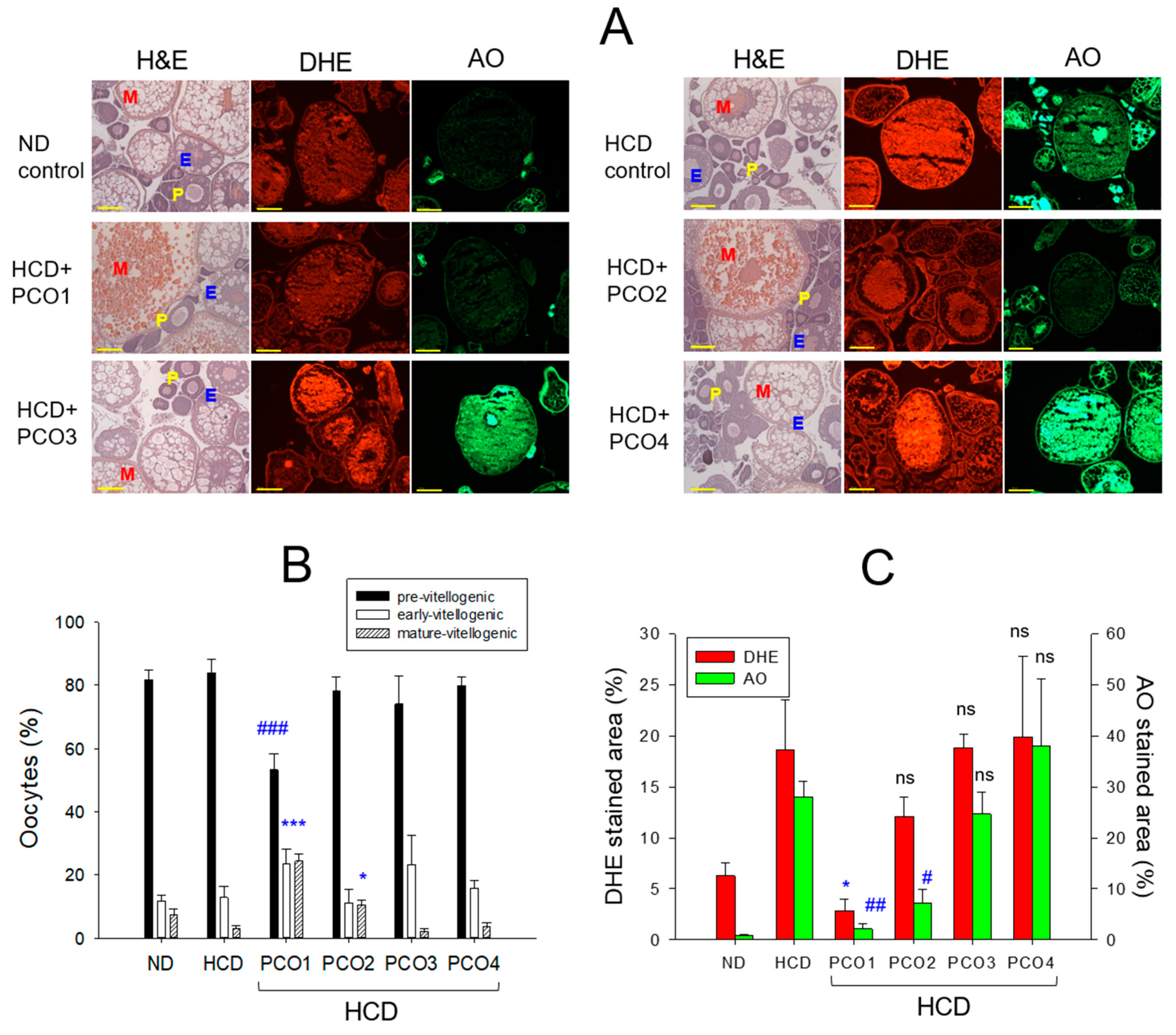
2.6. Histological Analysis with Ovary
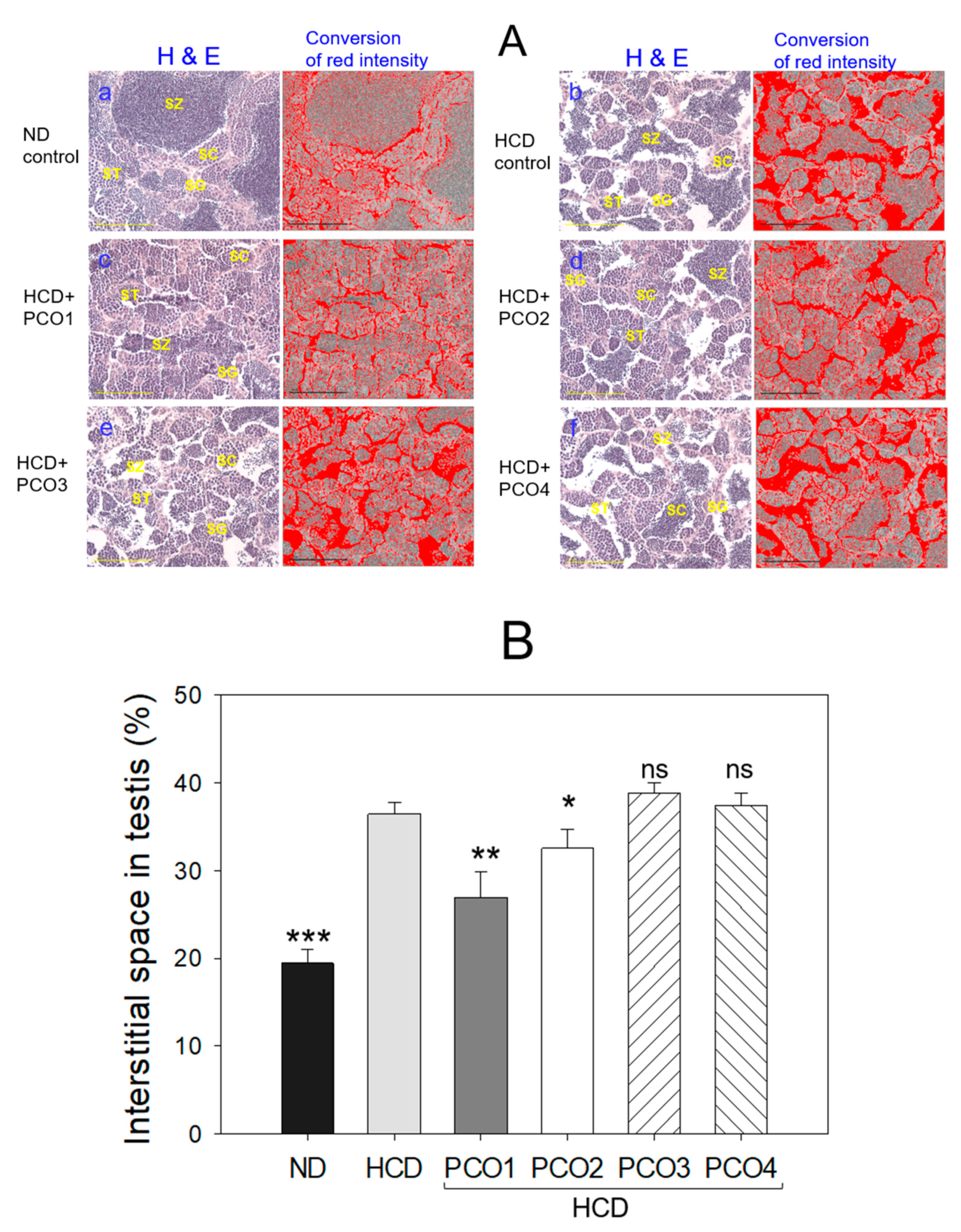
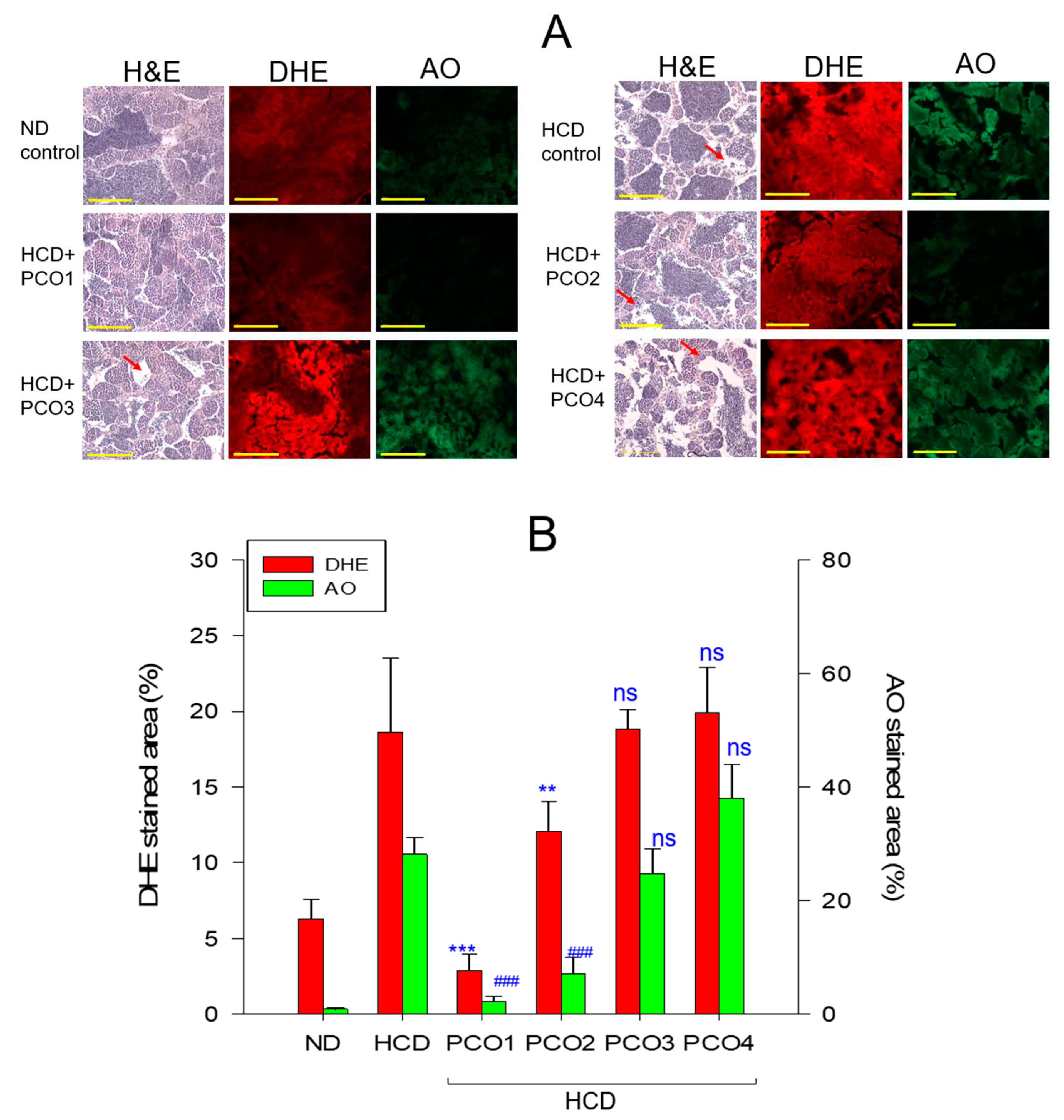
2.9. Histological Analysis with Testis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Zebrafish and Policosanol Supplementation
4.3. Blood Collection and Analysis
4.4. Histopathology Analysis
4.5. Histological Analysis with Ovarian Cell
4.6. Histological Analysis with Testicular Cell
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cho K. H. (2022). The Current Status of Research on High-Density Lipoproteins (HDL): A Paradigm Shift from HDL Quantity to HDL Quality and HDL Functionality. International journal of molecular sciences, 23(7), 3967. https://doi.org/10.3390/ijms23073967. [CrossRef]
- Wang J, Shi L, Zou Y, Tang J, Cai J, Wei Y, Qin J, Zhang Z. Positive association of familial longevity with the moderate-high HDL-C concentration in Bama Aging Study. Aging (Albany NY). 2018 Nov 28;10(11):3528-3540. https://doi.org/10.18632/aging.101663. PMID: 30485248; PMCID: PMC6286851. [CrossRef]
- Milman S, Atzmon G, Crandall J, Barzilai N. Phenotypes and genotypes of high density lipoprotein cholesterol in exceptional longevity. Curr Vasc Pharmacol. 2014;12(5):690-7. https://doi.org/10.2174/1570161111666131219101551. PMID: 24350928; PMCID: PMC4087084. [CrossRef]
- Barter, P. J., Nicholls, S., Rye, K. A., Anantharamaiah, G. M., Navab, M., & Fogelman, A. M. (2004). Antiinflammatory properties of HDL. Circulation research, 95(8), 764–772. https://doi.org/10.1161/01.RES.0000146094.59640.13. [CrossRef]
- Gotto A. M., Jr (2001). Low high-density lipoprotein cholesterol as a risk factor in coronary heart disease: a working group report. Circulation, 103(17), 2213–2218. https://doi.org/10.1161/01.cir.103.17.2213. [CrossRef]
- Cho, K. H., Park, H. J., & Kim, J. R. (2020). Decrease in Serum HDL-C Level Is Associated with Elevation of Blood Pressure: Correlation Analysis from the Korean National Health and Nutrition Examination Survey 2017. International journal of environmental research and public health, 17(3), 1101. https://doi.org/10.3390/ijerph17031101. [CrossRef]
- Denimal, D., Monier, S., Bouillet, B., Vergès, B., & Duvillard, L. (2023). High-Density Lipoprotein Alterations in Type 2 Diabetes and Obesity. Metabolites, 13(2), 253. https://doi.org/10.3390/metabo13020253. [CrossRef]
- Zimetti, F., Adorni, M. P., Marsillach, J., Marchi, C., Trentini, A., Valacchi, G., & Cervellati, C. (2021). Connection between the Altered HDL Antioxidant and Anti-Inflammatory Properties and the Risk to Develop Alzheimer's Disease: A Narrative Review. Oxidative medicine and cellular longevity, 2021, 6695796. https://doi.org/10.1155/2021/6695796. [CrossRef]
- Peppa, M., & Manta, A. (2023). Sexual Dysfunction in Diabetic Patients: Τhe Role of Advanced Glycation End Products. Current diabetes reviews, 10.2174/1573399819666230407095522. Advance online publication. https://doi.org/10.2174/1573399819666230407095522. [CrossRef]
- Tangvarasittichai S. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J Diabetes. 2015 Apr 15;6(3):456-80. https://doi.org/10.4239/wjd.v6.i3.456. PMID: 25897356; PMCID: PMC4398902. [CrossRef]
- Cipriani, S., & Simon, J. A. (2022). Sexual Dysfunction as a Harbinger of Cardiovascular Disease in Postmenopausal Women: How Far Are We?. The journal of sexual medicine, 19(9), 1321–1332. https://doi.org/10.1016/j.jsxm.2022.06.007. [CrossRef]
- Nikoobakht, M., Pourkasmaee, M., & Nasseh, H. (2005). The relationship between lipid profile and erectile dysfunction. Urology journal, 2(1), 40–44.
- Verit FF, Yildiz Zeyrek F, Zebitay AG, Akyol H. Cardiovascular risk may be increased in women with unexplained infertility. Clin Exp Reprod Med. 2017 Mar;44(1):28-32. https://doi.org/10.5653/cerm.2017.44.1.28. Epub 2017 Mar 31. PMID: 28428941; PMCID: PMC5395549. [CrossRef]
- Tall, A. R., & Rader, D. J. (2018). Trials and Tribulations of CETP Inhibitors. Circulation research, 122(1), 106–112. https://doi.org/10.1161/CIRCRESAHA.117.311978. [CrossRef]
- Luna-Castillo KP, Lin S, Muñoz-Valle JF, Vizmanos B, López-Quintero A, Márquez-Sandoval F. Functional Food and Bioactive Compounds on the Modulation of the Functionality of HDL-C: A Narrative Review. Nutrients. 2021 Apr 1;13(4):1165. https://doi.org/10.3390/nu13041165. PMID: 33916032; PMCID: PMC8066338. [CrossRef]
- Askarpour, M., Ghaedi, E., Roshanravan, N., Hadi, A., Mohammadi, H., Symonds, M. E., & Miraghajani, M. (2019). Policosanol supplementation significantly improves blood pressure among adults: A systematic review and meta-analysis of randomized controlled trials. Complementary therapies in medicine, 45, 89–97. https://doi.org/10.1016/j.ctim.2019.05.023. [CrossRef]
- Arruzazabala ML, Carbajal D, Mas R, Garcia M, Fraga V. Effects of Policosanol on platelet aggregation in rats. Thrombosis research. 1993;69(3):321-7.
- Valdes S, Arruzazabala ML, Fernandez L, Mas R, Carbajal D, Aleman C, et al. Effect of policosanol on platelet aggregation in healthy volunteers. International journal of clinical pharmacology research. 1996;16(2-3):67-72.
- Lee HG, Woo SY, Ahn HJ, Yang JY, Lee MJ, Kim HY, Song SY, Lee JH, Seo WD. Comparative Analysis of Policosanols Related to Growth Times from the Seedlings of Various Korean Oat (Avena sativa L.) Cultivars and Screening for Adenosine 5'-Monophosphate-Activated Protein Kinase (AMPK) Activation. Plants (Basel). 2022 Jul 14;11(14):1844. https://doi.org/10.3390/plants11141844. [CrossRef]
- Muthusamy M, Kim JH, Kim SH, Kim JY, Heo JW, Lee H, Lee KS, Seo WD, Park S, Kim JA, Lee SI. Changes in Beneficial C-glycosylflavones and Policosanol Content in Wheat and Barley Sprouts Subjected to Differential LED Light Conditions. Plants (Basel). 2020 Nov 6;9(11):1502. https://doi.org/10.3390/plants9111502. PMID: 33172036; PMCID: PMC7694615. [CrossRef]
- Zhang X, Ma C, Sun L, He Z, Feng Y, Li X, Gan J, Chen X. Effect of policosanol from insect wax on amyloid β-peptide-induced toxicity in a transgenic Caenorhabditis elegans model of Alzheimer's disease. BMC Complement Med Ther. 2021 Mar 30;21(1):103. https://doi.org/10.1186/s12906-021-03278-2. PMID: 33785017; PMCID: PMC8011155. [CrossRef]
- Venturelli A, Brighenti V, Mascolo D, Pellati F. A new strategy based on microwave-assisted technology for the extraction and purification of beeswax policosanols for pharmaceutical purposes and beyond. J Pharm Biomed Anal. 2019 Aug 5;172:200-205. https://doi.org/10.1016/j.jpba.2019.04.015. Epub 2019 Apr 9. PMID: 31060032. [CrossRef]
- Wong WT, Ismail M, Tohit ER, Abdullah R, Zhang YD. Attenuation of Thrombosis by Crude Rice (Oryza sativa) Bran Policosanol Extract: Ex Vivo Platelet Aggregation and Serum Levels of Arachidonic Acid Metabolites. Evid Based Complement Alternat Med. 2016;2016:7343942. https://doi.org/10.1155/2016/7343942. Epub 2016 Oct 5. PMID: 27800004; PMCID: PMC5069368. [CrossRef]
- Ishaka A, Umar Imam M, Mahamud R, Zuki AB, Maznah I. Characterization of rice bran wax policosanol and its nanoemulsion formulation. Int J Nanomedicine. 2014 May 9;9:2261-9. https://doi.org/10.2147/IJN.S56999. [CrossRef]
- Saavedra, D., Añé-Kourí, A. L., Barzilai, N., Caruso, C., Cho, K. H., Fontana, L., Franceschi, C., Frasca, D., Ledón, N., Niedernhofer, L. J., Pereira, K., Robbins, P. D., Silva, A., Suarez, G. M., Berghe, W. V., von Zglinicki, T., Pawelec, G., & Lage, A. (2023). Aging and chronic inflammation: highlights from a multidisciplinary workshop. Immunity & ageing : I & A, 20(1), 25. https://doi.org/10.1186/s12979-023-00352-w. [CrossRef]
- Cho, K. H., Kim, S. J., Yadav, D., Kim, J. Y., & Kim, J. R. (2018). Consumption of Cuban Policosanol Improves Blood Pressure and Lipid Profile via Enhancement of HDL Functionality in Healthy Women Subjects: Randomized, Double-Blinded, and Placebo-Controlled Study. Oxidative medicine and cellular longevity, 2018, 4809525. https://doi.org/10.1155/2018/4809525. [CrossRef]
- Park HJ, Yadav D, Jeong DJ, Kim SJ, Bae MA, Kim JR, Cho KH. Short-Term Consumption of Cuban Policosanol Lowers Aortic and Peripheral Blood Pressure and Ameliorates Serum Lipid Parameters in Healthy Korean Participants: Randomized, Double-Blinded, and Placebo-Controlled Study. Int J Environ Res Public Health. 2019 Mar 5;16(5):809. https://doi.org/10.3390/ijerph16050809. PMID: 30841655; PMCID: PMC6427682. [CrossRef]
- Cho, K. H., Kim, S. J., Yadav, D., Kim, J. Y., & Kim, J. R. (2018). Consumption of Cuban Policosanol Improves Blood Pressure and Lipid Profile via Enhancement of HDL Functionality in Healthy Women Subjects: Randomized, Double-Blinded, and Placebo-Controlled Study. Oxidative medicine and cellular longevity, 2018, 4809525. https://doi.org/10.1155/2018/4809525. [CrossRef]
- Kim JH, Lim DK, Suh YH, Chang KA. Long-Term Treatment of Cuban Policosanol Attenuates Abnormal Oxidative Stress and Inflammatory Response via Amyloid Plaques Reduction in 5xFAD Mice. Antioxidants (Basel). 2021 Aug 23;10(8):1321. https://doi.org/10.3390/antiox10081321. PMID: 34439569; PMCID: PMC8389325. [CrossRef]
- Cho, K. H., Baek, S. H., Nam, H. S., Kim, J. E., Kang, D. J., Na, H., & Zee, S. (2023). Cuban Sugar Cane Wax Alcohol Exhibited Enhanced Antioxidant, Anti-Glycation and Anti-Inflammatory Activity in Reconstituted High-Density Lipoprotein (rHDL) with Improved Structural and Functional Correlations: Comparison of Various Policosanols. International journal of molecular sciences, 24(4), 3186. https://doi.org/10.3390/ijms24043186. [CrossRef]
- Jin, S., & Cho, K. H. (2011). Water extracts of cinnamon and clove exhibits potent inhibition of protein glycation and anti-atherosclerotic activity in vitro and in vivo hypolipidemic activity in zebrafish. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 49(7), 1521–1529. https://doi.org/10.1016/j.fct.2011.03.043. [CrossRef]
- Yoon, J. H., & Cho, K. H. (2012). A point mutant of apolipoprotein A-I (V156K) showed enhancement of cellular insulin secretion and potent activity of facultative regeneration in zebrafish. Rejuvenation research, 15(3), 313–321. https://doi.org/10.1089/rej.2011.1246. [CrossRef]
- Stoletov, K., Fang, L., Choi, S. H., Hartvigsen, K., Hansen, L. F., Hall, C., Pattison, J., Juliano, J., Miller, E. R., Almazan, F., Crosier, P., Witztum, J. L., Klemke, R. L., & Miller, Y. I. (2009). Vascular lipid accumulation, lipoprotein oxidation, and macrophage lipid uptake in hypercholesterolemic zebrafish. Circulation research, 104(8), 952–960. https://doi.org/10.1161/CIRCRESAHA.108.189803. [CrossRef]
- Lee, E. Y., Yoo, J. A., Lim, S. M., & Cho, K. H. (2016). Anti-Aging and Tissue Regeneration Ability of Policosanol Along with Lipid-Lowering Effect in Hyperlipidemic Zebrafish via Enhancement of High-Density Lipoprotein Functionality. Rejuvenation research, 19(2), 149–158. https://doi.org/10.1089/rej.2015.1745. [CrossRef]
- Su, Y., He, L., Hu, Z., Li, Y., Zhang, Y., Fan, Z., Zhao, K., Zhang, H., & Liu, C. (2021). Obesity Causes Abrupt Changes in the Testicular Microbiota and Sperm Motility of Zebrafish. Frontiers in immunology, 12, 639239. https://doi.org/10.3389/fimmu.2021.639239. [CrossRef]
- Verit, F. F., Yildiz Zeyrek, F., Zebitay, A. G., & Akyol, H. (2017). Cardiovascular risk may be increased in women with unexplained infertility. Clinical and experimental reproductive medicine, 44(1), 28–32. https://doi.org/10.5653/cerm.2017.44.1.28. [CrossRef]
- Whitfield, M., Guiton, R., Rispal, J., Acar, N., Kocer, A., Drevet, J. R., & Saez, F. (2017). Dyslipidemia alters sperm maturation and capacitation in LXR-null mice. Reproduction (Cambridge, England), 154(6), 827–842. https://doi.org/10.1530/REP-17-0467 Reproduction (2017) 154: 827–842]. [CrossRef]
- Cho, K.-H.; Kim, J.-E.; Komatsu, T.; Uehara, Y. Protection of Liver Functions and Improvement of Kidney Functions by Twelve Weeks Consumption of Cuban Policosanol (Raydel®) with a Decrease of Glycated Hemoglobin and Blood Pressure from a Randomized, Placebo-Controlled, and Double-Blinded Study with Healthy and Middle-Aged Japanese Participants. Life 2023, 13, 1319. https://doi.org/10.3390/life13061319. [CrossRef]
- Cho, K. H., Nam, H. S., Baek, S. H., Kang, D. J., Na, H., Komatsu, T., & Uehara, Y. (2023). Beneficial Effect of Cuban Policosanol on Blood Pressure and Serum Lipoproteins Accompanied with Lowered Glycated Hemoglobin and Enhanced High-Density Lipoprotein Functionalities in a Randomized, Placebo-Controlled, and Double-Blinded Trial with Healthy Japanese. International journal of molecular sciences, 24(6), 5185. https://doi.org/10.3390/ijms24065185. [CrossRef]
- Ramiro Zardoya, Leonel Tula, Gladys Castaño, Rosa Más, Jose Illnait, Julio C. Fernández, Eduardo Díaz, Lilia Fernández, Effects of policosanol on hypercholesterolemic patients with abnormal serum biochemical indicators of hepatic function, Current Therapeutic Research, Volume 57, Issue 7, 1996, Pages 568-577, ISSN 0011-393X, https://doi.org/10.1016/S0011-393X(96)80068-3. [CrossRef]
- Yan, F., Zhao, Q., Li, Y., Zheng, Z., Kong, X., Shu, C., Liu, Y., & Shi, Y. (2022). The role of oxidative stress in ovarian aging: a review. Journal of ovarian research, 15(1), 100. https://doi.org/10.1186/s13048-022-01032-x. [CrossRef]
- Lee, E. Y., & Cho, K. H. (2016). High-dose consumption of NaCl resulted in severe degradation of lipoproteins associated with hyperlipidemia, hyperglycemia, and infertility via impairment of testicular spermatogenesis. Toxicology research, 5(2), 557–569. https://doi.org/10.1039/c5tx00059a. [CrossRef]
- Cho, K. H., Yadav, D., Kim, S. J., & Kim, J. R. (2018). Blood Pressure Lowering Effect of Cuban Policosanol is Accompanied by Improvement of Hepatic Inflammation, Lipoprotein Profile, and HDL Quality in Spontaneously Hypertensive Rats. Molecules (Basel, Switzerland), 23(5), 1080. https://doi.org/10.3390/molecules23051080. [CrossRef]
- Rodríguez, M. D., Sánchez, M., & García, H. (1997). Multigeneration reproduction study of policosanol in rats. Toxicology letters, 90(2-3), 97–106. https://doi.org/10.1016/s0378-4274(96)03844-1. [CrossRef]
- Rodríguez, M. D., & García, H. (1994). Teratogenic and reproductive studies of policosanol in the rat and rabbit. Teratogenesis, carcinogenesis, and mutagenesis, 14(3), 107–113. https://doi.org/10.1002/tcm.1770140302. [CrossRef]
- Klisic, A., Isakovic, A., Kocic, G., Kavaric, N., Jovanovic, M., Zvrko, E., Skerovic, V., & Ninic, A. (2018). Relationship between Oxidative Stress, Inflammation and Dyslipidemia with Fatty Liver Index in Patients with Type 2 Diabetes Mellitus. Experimental and clinical endocrinology & diabetes : official journal, German Society of Endocrinology [and] German Diabetes Association, 126(6), 371–378. https://doi.org/10.1055/s-0043-118667. [CrossRef]
- Gonnella, F., Konstantinidou, F., Di Berardino, C., Capacchietti, G., Peserico, A., Russo, V., Barboni, B., Stuppia, L., & Gatta, V. (2022). A Systematic Review of the Effects of High-Fat Diet Exposure on Oocyte and Follicular Quality: A Molecular Point of View. International journal of molecular sciences, 23(16), 8890. https://doi.org/10.3390/ijms23168890. [CrossRef]
- Ruebel, M. L., Cotter, M., Sims, C. R., Moutos, D. M., Badger, T. M., Cleves, M. A., Shankar, K., & Andres, A. (2017). Obesity Modulates Inflammation and Lipid Metabolism Oocyte Gene Expression: A Single-Cell Transcriptome Perspective. The Journal of clinical endocrinology and metabolism, 102(6), 2029–2038. https://doi.org/10.1210/jc.2016-3524. [CrossRef]
- Nusslein-Volhard, C.; Dahm, R. Zebrafish: A Practical Approach, 1st ed.; Oxford University Press: Oxford, UK, 2002.
- National Research Council of the National Academy of Sciences. Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA, 2010.
- Cho, K. H., Nam, H. S., Kim, J. E., Na, H. J., Del Carmen Dominguez-Horta, M., & Martinez-Donato, G. (2023). CIGB-258 Exerts Potent Anti-Inflammatory Activity against Carboxymethyllysine-Induced Acute Inflammation in Hyperlipidemic Zebrafish via the Protection of Apolipoprotein A-I. International journal of molecular sciences, 24(8), 7044. https://doi.org/10.3390/ijms24087044. [CrossRef]
- Owusu-Ansah, E.; Yavari, A.; Mandal, S.; Banerjee, U., Distinct mitochondrial retrograde signals control the G1-S cell cycle checkpoint. Nature genetics 2008, 40, (3), 356-361.
- Cho, K. H., Kim, J. E., Bahuguna, A., & Kang, D. J. (2023). Long-Term Supplementation of Ozonated Sunflower Oil Improves Dyslipidemia and Hepatic Inflammation in Hyperlipidemic Zebrafish: Suppression of Oxidative Stress and Inflammation against Carboxymethyllysine Toxicity. Antioxidants (Basel, Switzerland), 12(6), 1240. https://doi.org/10.3390/antiox12061240. [CrossRef]
- Patel, U. N., Patel, U. D., Khadayata, A. V., Vaja, R. K., Modi, C. M., & Patel, H. B. (2022). Long-term exposure of the binary mixture of cadmium and mercury damages the developed ovary of adult zebrafish. Environmental science and pollution research international, 29(29), 44928–44938. https://doi.org/10.1007/s11356-022-18988-4. [CrossRef]
- Sutha, J., Anila, P. A., Gayathri, M., & Ramesh, M. (2022). Long term exposure to tris (2-chloroethyl) phosphate (TCEP) causes alterations in reproductive hormones, vitellogenin, antioxidant enzymes, and histology of gonads in zebrafish (Danio rerio): In vivo and computational analysis. Comparative biochemistry and physiology. Toxicology & pharmacology : CBP, 254, 109263. https://doi.org/10.1016/j.cbpc.2021.109263. [CrossRef]
- Sabaliauskas, N. A., Foutz, C. A., Mest, J. R., Budgeon, L. R., Sidor, A. T., Gershenson, J. A., Joshi, S. B., & Cheng, K. C. (2006). High-throughput zebrafish histology. Methods (San Diego, Calif.), 39(3), 246–254. https://doi.org/10.1016/j.ymeth.2006.03.001. [CrossRef]
- N. D. Koc, N. Teksöz, M. Ural and C. Akbulut, Elixir Aquacult., 2012, 46, Histological structure of zebrafish (Danio rerio, Hamilton, 1822) testicles 8117–8120.

| HCD | ||||||||
|---|---|---|---|---|---|---|---|---|
| Groups | ND control |
Control | PCO1 Raydel Sugarcane wax alcohol |
PCO2 Xi’an Natural Sugarcane |
PCO3 Xi’an Realin Sugarcane |
PCO4 Shaanxi Rice bran |
||
| Diet composition (%) |
Tetrabits | 100 | 96 | 95.9 | 95.9 | 95.9 | 95.9 | |
| Cholesterol (%, wt./wt.) |
- | 4 | 4 | 4 | 4 | 4 | ||
| PCO (%, wt./wt.) |
- | - | 0.1 | 0.1 | 0.1 | 0.1 | ||
| Body weight (mg) |
0 | 276 ±13 | 275±9 | 278±12 | 265±8 | 262 ±7 | 271 ±5 | |
| 12 | 507±14a | 635 ±31 c | 547±17 ab | 605±23 bc | 602±24bc | 509±13 a | ||
| p† | - | - | < 0.001 | ns | ns | < 0.001 | ||
| Survi-vability (%) |
0 | 100 (n=70) |
100 (n=70) |
100 (n=70) |
100 (n=70) |
100 (n=70) |
100 (n=70) |
|
| 12 | 97 (n=68) |
61 (n=43) |
81 (n=53) |
81 (n=53) |
57 (n=40) |
67 (n=47) |
||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




