Submitted:
01 September 2023
Posted:
04 September 2023
You are already at the latest version
Abstract
Keywords:
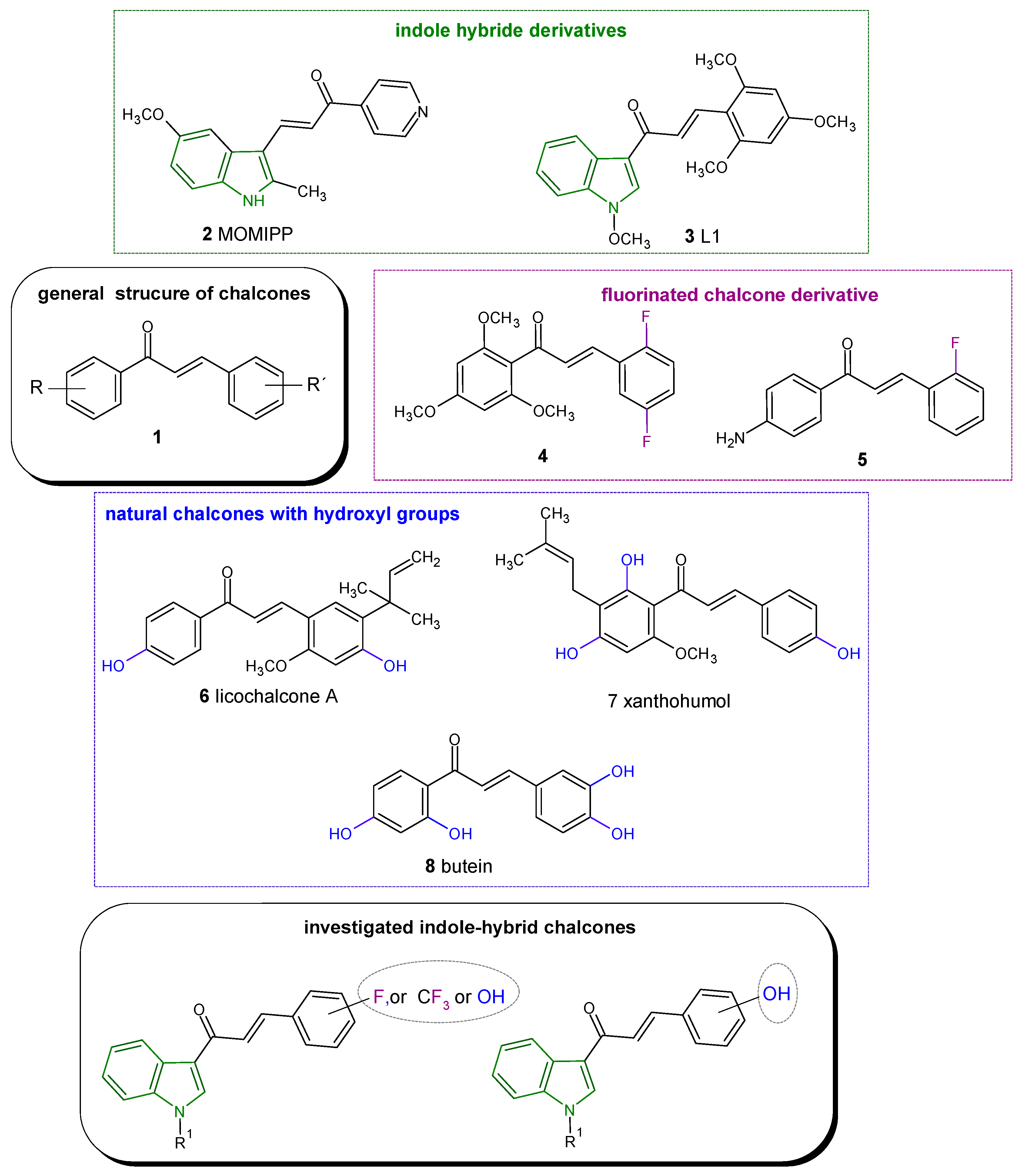
1. Introduction
2. Results and Discussion
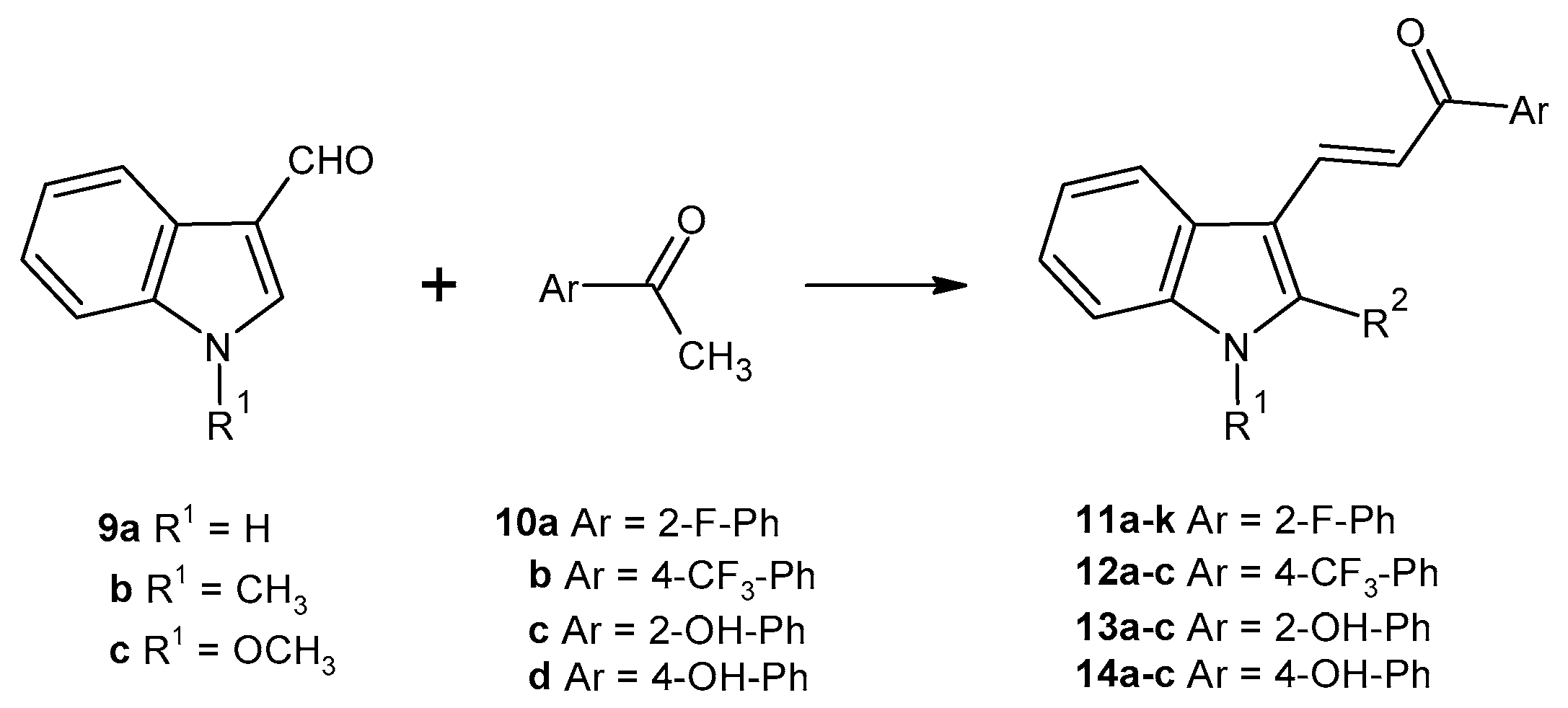
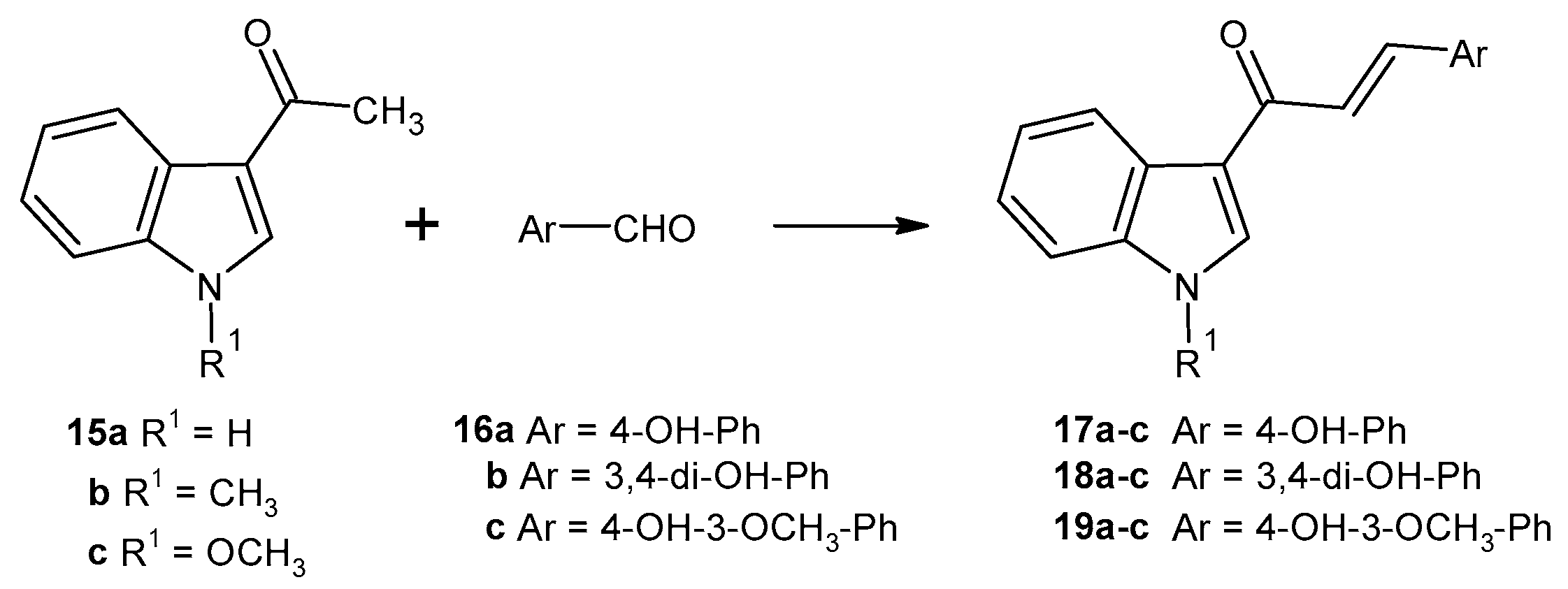
2.1. Chemistry
2.2. Antiproliferative Activity
2.3. Antioxidant Activity
3. Materials and Methods
3.1. Chemistry
3.1.1. General Method and Materials
3.1.2. General Procedure A) Acid-Catalyzed Claisen-Schmidt Condensation
3.1.3. General Procedure B1) Base-Catalyzed Claisen-Schmidt Condensation (50% aq. KOH)
3.1.4. General Procedure B2) Base-Catalyzed Claisen-Schmidt Condensation (Piperidine)
3.1.5. Synthesis and Characterisation of Compounds 11 - 14, 17 - 19
3.2. Antiproliferative Activity Studies
3.2.1. Cell Cultures
3.2.2. MTT Assay
3.3 Antiproliferative Activity Studies
3.3.1. DPPH Radical Scavenging Activity
3.3.2. ABTS Radical Scavenging Activity
3.3.3. Ferric Reducing Antioxidant Power (FRAP)
3.3.4. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gomes, M.N.; Muratov, E.N.; Pereira, M.; Peixoto, J.C.; Rosseto, L.P.; Cravo, P.V.L.; Andrade, C.H.; Neves, B.J. Chalcone Derivatives: Promising Starting Points for Drug Design. Molecules 2017, 22, 1210. [CrossRef]
- McCluskey, A.; Russell, C. Chalcones: Potential Anticancer Agents. In Translational Research in Cancer; Http://dx.doi.org/10.5772/intechopen.9144, D., Ed.; 2021; pp. 1–25 ISBN 0000957720.
- Gao, F.; Huang, G.; Xiao, J. Chalcone Hybrids as Potential Anticancer Agents: Current Development, Mechanism of Action, and Structure-Activity Relationship. Med. Res. Rev. 2020, 40, 2049–2084. [CrossRef]
- Sravanthi, T. V.; Manju, S.L. Indoles - A Promising Scaffold for Drug Development. Eur. J. Pharm. Sci. 2016, 91, 1–10. [CrossRef]
- Robinson, M.W.; Overmeyer, J.H.; Young, A.M.; Erhardt, P.W.; Maltese, W.A. Synthesis and Evaluation of Indole-Based Chalcones as Inducers of Methuosis, a Novel Type of Non-Apoptotic Cell Death. J Med Chem. 2012, 55, 1940–1956. [CrossRef]
- Cho, H.; Geno, E.; Patoor, M.; Reid, A.; Mcdonald, R.; Hild, M.; Jenkins, J.L. Indolyl-Pyridinyl-Propenone-Induced Methuosis through the Inhibition of PIKFYVE. ACS Omega 2018, 3, 6097–6103. [CrossRef]
- Kudličková, Z.; Takáč, P.; Sabolová, D.; Vilková, M.; Baláž, M.; Béres, T.; Mojžiš, J. Novel 1-Methoxyindole- and 2-Alkoxyindole-Based Chalcones: Design, Synthesis, Characterization, Antiproliferative Activity and DNA, BSA Binding Interactions. Med. Chem. Res. 2021, 30, 897–912. [CrossRef]
- Kuruc, T.; Kello, M.; Petrova, K.; Kudlickova, Z.; Kubatka, P.; Mojzis, J. The Newly Synthetized Chalcone L1 Is Involved in the Cell Growth Inhibition, Induction of Apoptosis and Suppression of Epithelial-to-Mesenchymal Transition of HeLa Cells. Molecules 2021, 26, 1–27. [CrossRef]
- Inoue, M.; Sumii, Y.; Shibata, N. Contribution of Organofluorine Compounds to Pharmaceuticals. ACS Omega 2020, 5, 10633–10640. [CrossRef]
- Yu, Y.; Liu, A.; Dhawan, G.; Mei, H.; Zhang, W.; Izawa, K.; Soloshonok, V.A.; Han, J. Fluorine-Containing Pharmaceuticals Approved by the FDA in 2020: Synthesis and Biological Activity. Chinese Chem. Lett. 2021, 32, 3342–3354. [CrossRef]
- Mei, H.; Han, J.; Fustero, S.; Medio-Simon, M.; Sedgwick, D.M.; Santi, C.; Ruzziconi, R.; Soloshonok, V.A. Fluorine-Containing Drugs Approved by the FDA in 2018. Chem. - A Eur. J. 2019, 25, 11797–11819. [CrossRef]
- Burmaoglu, S.; Algul, O.; Anil, D.A.; Gobek, A.; Duran, G.G.; Ersan, R.H.; Duran, N. Synthesis and Anti-Proliferative Activity of Fluoro-Substituted Chalcones. Bioorganic Med. Chem. Lett. 2016, 26, 3172–3176. [CrossRef]
- dos Santos, M.B.; Bertholin Anselmo, D.; de Oliveira, J.G.; Jardim-Perassi, B. V.; Alves Monteiro, D.; Silva, G.; Gomes, E.; Lucia Fachin, A.; Marins, M.; de Campos Zuccari, D.A.P.; et al. Antiproliferative Activity and P53 Upregulation Effects of Chalcones on Human Breast Cancer Cells. J. Enzyme Inhib. Med. Chem. 2019, 34, 1093–1099. [CrossRef]
- Rozmer, Z.; Perjési, P. Naturally Occurring Chalcones and Their Biological Activities. Phytochem. Rev. 2016, 15, 87–120. [CrossRef]
- Cramer, J.; Sager, C.P.; Ernst, B. Hydroxyl Groups in Synthetic and Natural-Product-Derived Therapeutics: A Perspective on a Common Functional Group. J. Med. Chem. 2019, 62, 8915–8930. [CrossRef]
- Constantinescu, T.; Lungu, C.N. Anticancer Activity of Natural and Synthetic Chalcones. Int. J. Mol. Sci. 2021, 22, 1–33. [CrossRef]
- Deng, N.; Qiao, M.; Li, Y.; Liang, F.; Li, J.; Liu, Y. Anticancer Effects of Licochalcones: A Review of the Mechanisms. Front. Pharmacol. 2023, 14, 1–10. [CrossRef]
- Yang, L.H.; Ho, Y.J.; Lin, J.F.; Yeh, C.W.; Kao, S.H.; Hsu, L.S. Butein Inhibits the Proliferation of Breast Cancer Cells through Generation of Reactive Oxygen Species and Modulation of ERK and P38 Activities. Mol. Med. Rep. 2012, 6, 1126–1132. [CrossRef]
- Cho, S.-G.; Woo, S.-M.; Ko, S.-G. Butein Suppresses Breast Cancer Growth by Reducing a Production of Intracellular Reactive Oxygen Species. J. Exp. Clin. Cancer Res. 2014, 33, 51. [CrossRef]
- Chen, W.J.; Song, J.R.; Guo, P.; Wen, Z.Y. Butein, a More Effective Antioxidant than α-Tocopherol. J. Mol. Struct. THEOCHEM 2006, 763, 161–164. [CrossRef]
- Miranda, C.L.; Stevens, J.F.; Ivanov, V.; Mccall, M.; Frei, B.; Deinzer, M.L.; Buhler, D.R. Antioxidant and Prooxidant Actions of Prenylated and Nonprenylated Chalcones and Flavanones in Vitro. J. Agric. Food Chem. 2000, 2000, 3876–3884.
- Cai, Y.Z.; Mei Sun; Jie Xing; Luo, Q.; Corke, H. Structure-Radical Scavenging Activity Relationships of Phenolic Compounds from Traditional Chinese Medicinal Plants. Life Sci. 2006, 78, 2872–2888. [CrossRef]
- Kudličková, Z.; Stahorský, M.; Michalková, R.; Vilková, M.; Baláž, M. Mechanochemical Synthesis of Indolyl Chalcones with Antiproliferative Activity. Green Chem. Lett. Rev. 2022, 15, 474–482. [CrossRef]
- Suzdalev, K.F.; Babakova, M.N. Synthesis of Analogues of Indole Alkaloids from Sea Sponges – Aplysinopsins by the Reaction of Amines with (4Z)-4-[(1H-Indol-3-Yl)- Methylene]-1,3-Oxazol-5(4H)-Ones. J. Heterocycl. Chem. 2015, 46, 1200–1206. [CrossRef]
- Somei, M.; Kawasaki, T.; Kodama, A.; Nishida, T.; Shimizu, K. Preparation of 1-Hydroxyindole Derivatives and a New Route to 2-Substituted Indoles. Heterocycles 1991, 32, 221. [CrossRef]
- Acheson, B.R.M.; Hunt, P.G.; Littlewood, D.M.; Murrer, B.A.; Rosenberg, H.E. The Synthesis, Reactions, and Spectra of 1-Acetoxy-, 1-Hydroxy-, and 1-Methoxy-Indoles. J. Chem. Soc., Perkin Trans. 1 1978, 1117–1125. [CrossRef]
- Somei, M.; Nakajou, M.; Teramoto, T.; Tanimoto, A.; Yamada, F. [NUCLEOPHILIC SUBSTITUTION REACTION OF 3-ACETYL-I-METHOXY- INWLE AND ITS APPLICATION FOR THE SYNTHESIS OF NOVEL 2-SUBSTITUTED METHYL 2,3-DIHYDRO-l-METHYL3-0X05H-PYRIDO (4,3-b)INDOLE-4-CARBOXYLATES. 1999, 51, 1949–1956.
- Venkatanarayana, M.; K. Dubey, P. Novel and Simple Methodology for the Synthesis of 3-Acetylindoles and Their N-Alkyl Derivatives Using TBAB as Phase Transfer Catalyst. Lett. Org. Chem. 2011, 8, 656–662. [CrossRef]
- Somei, M.; Nakajou, M.; Teramoto, T.; Tanimoto, A.; Yamada, F. Nucleophilic Substitution Reaction of 3-Acetyl-1-Methoxyindole and Its Application for the Synthesis of Novel 2-Substituted Methyl 2,3-Dihydro-1-Methyl-3-Oxo-5H-Pyrido-[4,3-b]Indole-4-Carboxylates. Heterocycles 1999, 51, 1949–1956. [CrossRef]
- Jacobsen, N.E. NMR Data Interpretation Explained : Understanding 1D and 2D NMR Spectra of Organic Compounds and Natural Products; John Wiley & Sons, Inc., Hoboken, New Jersey, 2016; ISBN 9781118370223.
- Kudličková, Z.; Stahorský, M.; Michalková, R.; Vilková, M.; Baláž, M. Mechanochemical Synthesis of Indolyl Chalcones with Antiproliferative Activity. Green Chem. Lett. Rev. 2022, 15, 474–482. [CrossRef]
- Michalkova, R.; Kello, M.; Kudlickova, Z.; Gazdova, M.; Mirossay, L.; Mojzisova, G.; Mojzis, J. Programmed Cell Death Alterations Mediated by Synthetic Indole Chalcone Resulted in Cell Cycle Arrest , DNA Damage , Apoptosis and Signaling Pathway Modulations in Breast Cancer Model. Pharmaceutics 2022, 14, 503. [CrossRef]
- Chripkova, M.; Zigo, F.; Mojzis, J. Antiproliferative Effect of Indole Phytoalexins. Molecules 2016, 21, 6–8. [CrossRef]
- Gacche, R.N.; Dhole, N.A.; Kamble, S.G.; Bandgar, B.P. In-Vitro Evaluation of Selected Chalcones for Antioxidant Activity. J. Enzyme Inhib. Med. Chem. 2008, 23, 28–31. [CrossRef]
- Chu, J.; Guo, C.L. Design and Discovery of Some Novel Chalcones as Antioxidant and Anti-Inflammatory Agents via Attenuating NF-ΚB. Arch. Pharm. (Weinheim). 2016, 349, 63–70. [CrossRef]
- Przybylski, P.; Konopko, A.; Łętowski, P.; Jodko-Piórecka, K.; Litwinienko, G. Concentration-Dependent HAT/ET Mechanism of the Reaction of Phenols with 2,2-Diphenyl-1-Picrylhydrazyl (Dpph˙) in Methanol. RSC Adv. 2022, 12, 8131–8136. [CrossRef]
- Wang, G.; Xue, Y.; An, L.; Zheng, Y.; Dou, Y.; Zhang, L.; Liu, Y. Theoretical Study on the Structural and Antioxidant Properties of Some Recently Synthesised 2,4,5-Trimethoxy Chalcones. Food Chem. 2015, 171, 89–97. [CrossRef]
- Munteanu, I.G.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int. J. Mol. Sci. 2021, 22. [CrossRef]
- Jung, J.C.; Lee, Y.; Min, D.; Jung, M.; Oh, S. Practical Synthesis of Chalcone Derivatives and Their Biological Activities. Molecules 2017, 22, 1–11. [CrossRef]
- Kalinowska, M.; Płońska, A.; Trusiak, M.; Gołębiewska, E.; Gorlewska-Pietluszenko, A. Comparing the Extraction Methods, Chemical Composition, Phenolic Contents and Antioxidant Activity of Edible Oils from Cannabis Sativa and Silybum Marianu Seeds. Sci. Rep. 2022, 12, 1–16. [CrossRef]
- Cheng, Z.J.; Lin, C.N.; Hwang, T.L.; Teng, C.M. Broussochalcone A, a Potent Antioxidant and Effective Suppressor of Inducible Nitric Oxide Synthase in Lipopolysaccharide-Activated Macrophages. Biochem. Pharmacol. 2001, 61, 939–946. [CrossRef]
- Sivakumar, P.M.; Prabhakar, P.K.; Doble, M. Synthesis, Antioxidant Evaluation, and Quantitative Structure-Activity Relationship Studies of Chalcones. Med. Chem. Res. 2011, 20, 482–492. [CrossRef]
- Garberová, M.; Potočňák, I.; Tvrdoňová, M.; Bago-Pilátová, M.; Bekešová, S.; Kudličková, Z.; Samoľová, E.; Kešeľáková, A.; Elečko, J.; Vilková, M. Spectral, Structural, and Pharmacological Studies of Perillaldehyde and Myrtenal Based Benzohydrazides. J. Mol. Struct. 2023, 1271. [CrossRef]
- Lagu, S.B.; Yejella, R.P.; Bhandare, R.R.; Shaik, A.B. Design, Synthesis, and Antibacterial and Antifungal Activities of Novel Trifluoromethyl and Trifluoromethoxy Substituted Chalcone Derivatives. Pharmaceuticals 2020, 13, 1–16. [CrossRef]
- Yao, Y.; Huang, T.; Wang, Y.; Wang, L.; Feng, S.; Cheng, W.; Yang, L.; Duan, Y. Angiogenesis and Anti-Leukaemia Activity of Novel Indole Derivatives as Potent Colchicine Binding Site Inhibitors. J. Enzyme Inhib. Med. Chem. 2022, 37, 652–665. [CrossRef]
- Venturella, P.; Bellino, A.; Piozzi, F. Synthesis of Indolylchalcones and Indolylchrom Indolylchromonols. Farm. Ed. Sci. 1971, 26, 591–596.
- Yesuthangam, Y.; Pandian, S.; Venkatesan, K.; Gandhidasan, R.; Murugesan, R. Photogeneration of Reactive Oxygen Species and Photoinduced Plasmid DNA Cleavage by Novel Synthetic Chalcones. J. Photochem. Photobiol. B Biol. 2011, 102, 200–208. [CrossRef]
- Chang, M.Y.; Chen, K.T.; Tsai, Y.L.; Chen, H.Y. One-Pot Access to 2-Aryl-3-(Arylmethyl)Chromones. Synth. 2020, 52, 861–872. [CrossRef]
- Kumar, D.; Kumar, N.M.; Akamatsu, K.; Kusaka, E.; Harada, H.; Ito, T. Synthesis and Biological Evaluation of Indolyl Chalcones as Antitumor Agents. Bioorganic Med. Chem. Lett. 2010, 20, 3916–3919. [CrossRef]
- Rani, P.; Srivastava, V.K.; Kumar, A. Synthesis and Antiinflammatory Activity of Heterocyclic Indole Derivatives. Eur. J. Med. Chem. 2004, 39, 449–452. [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT - Food Sci. Technol. 1995, 28, 25–30. [CrossRef]
- Tvrdoňová, M.; Borovská, B.; Salayová, A.; Rončák, R.; Michalčin, P.; Bednáriková, Z.; Gažová, Z. Design and Synthesis of Novel Carbohydrate-Amino Acid Hybrids and Their Antioxidant and Anti-β-Amyloid Aggregation Activity. Bioorg. Chem. 2023, 137, 106636. [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [CrossRef]
| comp. | Ar | R1 | R2 | H-2 (J) | H-3 (J) | C-1 | C-2 | C-3 |
|---|---|---|---|---|---|---|---|---|
| 11a | 2-F-Ph | H | H | 7.29 (15.7, 2.3) | 7.91 (15.7, 1.2) | 188.2 | 119.5 | 140.0 |
| 11b | CH3 | 7.25 (15.7, 2.3) | 7.88 (15.7, 1.4) | 188.1 | 119.4 | 139.3 | ||
| 11c | OCH3 | 7.33 (15.8, 2.2) | 7.84 (15.7, 0.9) | 188.3 | 120.9 | 138.5 | ||
| 11d | H | OCH3 | 7.02 (15.4, 2.2) | 7.84 (15.4, 1.5) | 187.4 | 115.7 | 136.8 | |
| 11e | OCH2CH3 | 7.07 (15.4, 2.3) | 7.88 (15.4, 1.5) | 187.1 | 115.6 | 136.8 | ||
| 11f | OCH2CH2CH3 | 7.06 (15.4, 2.2) | 7.88 (15.4, 1.5) | 187.3 | 115.7 | 136.9 | ||
| 11g | OCH(CH3)2 | 7.09 (15.6, 2.3) | 7.87 (15.6, 1.5) | 187.2 | 115.8 | 136.9 | ||
| 11h | OCH2CH2 CH2CH3 | 7.05 (15.4, 2.2) | 7.86 (15.4, 1.5) | 187.4 | 115.8 | 136.9 | ||
| 11i | OCH2CH(CH3)2 | 7.06 (15.4, 2.1) | 7.88 (15.4, 1.4) | 187.4 | 115.8 | 137.0 | ||
| 11j | OCHCH3CH2 OCH3 | 7.09 (15.5, 2.2) | 7.87 (15.5, 1.4) | 187.7 | 116.4 | 137.4 | ||
| 11k | OCH2CH2OH | 7.08 (15.5, 2.3) | 7.94 (15.5, 1.3) | 187.0 | 115.5 | 136.8 | ||
| 12a | 4-CF3-Ph | H | H | 7.63 (15.4) | 8.11 (15.4) | 188.2 | 115.1 | 140.3 |
| 12b | CH3 | 7.63 (15.4) | 8.06 (15.4) | 188.0 | 114.9 | 139.7 | ||
| 12c | OCH3 | 7.69 (15.4) | 8.02 (15.4) | 188.6 | 117.0 | 139.2 | ||
| 13a | 2-OH-Ph | H | H | 7.77 (15.2) | 8.21 (15.2) | 193.1 | 113.9 | 140.4 |
| 13b | CH3 | 7.75 (15.2) | 8.16 (15.2) | 193.0 | 113.9 | 139.7 | ||
| 13c | OCH3 | 7.82 (15.3) | 8.11 (15.3) | 193.1 | 115.7 | 138.7 | ||
| 14a | 4-OH-Ph | H | H | 7.64 (15.5) | 7.99 (15.5) | 187.0 | 115.4 | 137.7 |
| 14b | CH3 | 7.62 (15.5) | 7.94 (15.5) | 187.0 | 115.4 | 137.0 | ||
| 14c | OCH3 | 7.69 (15.5) | 7.91 (15.5) | 186.9 | 117.0 | 136.1 | ||
| 17a | 4-OH-Ph | H | 7.55 (15.4 | 7.63 (15.4) | 183.8 | 121.3 | 139.8 | |
| 17b | CH3 | 7.56 (15.8 | 7.56 (15.8) | 183.3 | 121.2 | 139.8 | ||
| 17c | OCH3 | 7.57 (15.5 | 7.59 (15.5) | 183.4 | 120.8 | 140.4 | ||
| 18a | 3,4-diOH-Ph | H | 7.47 (15.5) | 7.54 (15.5) | 183.8 | 121.2 | 140.3 | |
| 18b | CH3 | 7.47 | 7.47 | 183.3 | 121.1 | 140.4 | ||
| 18c | OCH3 | 7.50 (15.5) | 7.51 (15.5) | 183.4 | 120.8 | 140.9 | ||
| 19a | 3-OCH3-4-OH-Ph | H | 7.64 (15.4) | 7.55 (15.4) | 183.1 | 121.4 | 140.2 | |
| 19b | CH3 | 7.56 (15.6) | 7.55 (15.6) | 183.3 | 121.3 | 140.3 | ||
| 19c | OCH3 | 7.59 (15.5) | 7.58 (15.5) | 183.3 | 120.9 | 140.9 |
| comp. | Ar | R1 | R2 | Cell line, IC50 (µM) ± SD | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| MDA-MB-231 | HCT116 | Jurkat | Hela | MCF-7 | MCF-10A | Bj-5ta | ||||
| 11a | 2-F-Ph | H | H | 51.4 ± 3.2 | 31.8 ± 2.1 | 7.6 ± 0.4 | 41.7 ± 0.2 | 37.1 ± 0.4 | 89.1 ± 2.4 | NT |
| 11b[31] | CH3 | > 100 | 41.5 ± 1.7 | 32.4 ± 1.2 | 57.9 ± 0.4 | 47.5 ± 4.3 | > 100 | NT | ||
| 11c[7] | OCH3 | 21.1 ± 3.9 | 37.9 ± 2.7 | 5.9 ± 1.0 | 19.2 ± 5.7 | 20.0 ± 7.9 | 67 ± 2.9 | NT | ||
| 11d | H | OCH3 | 95.5 ± 2.7 | 45.5 ± 2.4 | 11.4 ± 1.9 | > 100 | 32 ± 1.2 | 98 ± 1.3 | NT | |
| 11e | OCH2CH3 | 72.2 ± 5.4 | 40.1 ± 0.6 | 8.3 ± 0.2 | 75.2 ± 4.1 | 30.3 ± 2.2 | 94 ± 4.6 | NT | ||
| 11f[32] | OCH2CH2CH3 | 34.0 ± 3.0 | 34.4 ± 1.1 | 37.7 ± 1.1 | 45.3 ± 0.7 | 37.3 ± 1.5 | > 100 | NT | ||
| 11g | OCH(CH3)2 | 33.3 ± 0.3 | 31.7 ± 1.7 | 36.7 ± 4.9 | 34.2 ± 0.1 | 17.1 ± 2.3 | 41.8 ± 1.5 | NT | ||
| 11h | OCH2CH2 CH2CH3 | 36.3 ± 2.1 | 31 ± 2.9 | 35.9 ± 1.8 | 48.2 ± 1.0 | 13.8 ± 3.5 | 65.5 ± 3.7 | NT | ||
| 11i | OCH2CH(CH3)2 | 42.9 ± 0.4 | 33.8 ± 0.1 | 40.1 ± 3.2 | 54.6 ± 3.6 | 30.7 ± 0.6 | 93.3 ± 3.4 | NT | ||
| 11j | OCHCH3CH2 OCH3 | 51.2 ± 4.1 | 32.4 ± 1.3 | 40.4 ± 2.1 | 50.0 ± 1.8 | 26.2 ± 0.5 | 76.5 ± 1.1 | NT | ||
| 11k | OCH2CH2OH | 64.5 ± 4.7 | 39.4 ± 1.5 | 37.8 ± 0.9 | 52.9 ± 1.6 | 38.1 ± 3.3 | 94.7 ± 2.6 | NT | ||
| 12a | 4-CF3-Ph | H | H | > 100 | 59.1 ± 1.1 | > 100 | 68.1 ± 3.3 | 43.3 ± 0.6 | > 100 | > 100 |
| 12b[31] | CH3 | > 100 | 12.3 ± 0.2 | > 100 | 32.8 ± 0.4 | 20.5 ± 5.1 | > 100 | > 100 | ||
| 12c | OCH3 | 81.3 ± 1.2 | 55.3 ± 1.7 | 39.3 ± 1.2 | 98 ± 2.8 | 86 ± 3.5 | 38.4 ± 3.1 | > 100 | ||
| 13a | 2-OH-Ph | H | H | 92.7 ± 1.8 | 40.5 ± 3.1 | 25.9 ± 2.4 | 39.6 ± 3.5 | 44.7 ± 2.6 | 55.4 ± 3.6 | 81.5 ± 0.2 |
| 13b | CH3 | 82 ± 3.2 | 34.7 ± 0.2 | > 100 | 42.8 ± 2.5 | 29.7 ± 2.3 | > 100 | > 100 | ||
| 13c | OCH3 | > 100 | 54.0 ± 2.2 | 42 ± 1.8 | 73.9 ± 3.4 | 53.4 ± 2.5 | > 100 | > 100 | ||
| 14a | 4-OH-Ph | H | H | 66 ± 2.5 | 36.8 ± 0.5 | 33.9 ± 2.5 | 39.9 ± 2.6 | 42.2 ± 0.7 | 51.5 ± 1.7 | 61 ± 0.6 |
| 14b | CH3 | > 100 | 49.3 ± 1.3 | > 100 | 93 ± 2.6 | 82.3 ± 1.8 | 54 ± 2.1 | > 100 | ||
| 14c | OCH3 | 82.2 ± 3.1 | 34.5 ± 0.7 | 7.3 ± 0.1 | 34.6 ± 0.3 | 52.7 ± 2.6 | 59 ± 0.2 | 76.4 ± 1.7 | ||
| 17a | 4-OH-Ph | H | 39.6 ± 2.1 | NT | NT | 31.4 ± 2.0 | 47.9 ± 0.2 | 28.6 ± 0.5 | NT | |
| 17b | CH3 | NT | > 100 | NT | NT | 50.3 ± 0.6 | 22.8 ± 0.4 | NT | ||
| 17c | OCH3 | > 100 | NT | NT | 39.8 ± 1.6 | > 100 | > 100 | NT | ||
| 18a | 3,4-diOH-Ph | H | 39.3 ± 1.9 | 29.8 ± 0.7 | NT | NT | 42.3 ± 3.3 | 11.5 ± 1.4 | NT | |
| 18b | CH3 | NT | 7.1 ± 0.7 | NT | 6.7 ± 0.8 | 37.4 ± 2.2 | 7.8 ± 0.3 | NT | ||
| 18c | OCH3 | > 100 | 18.2 ± 2.9 | 8.0 ± 1.4 | > 100 | > 100 | > 100 | > 100 | ||
| 19a | 3-OCH3-4-OH-Ph | H | NT | 62.7 ± 2.9 | NT | 25.5 ± 1.6 | NT | 24.7 ± 2.8 | NT | |
| 19b | CH3 | NT | NT | NT | 34.9 ± 1.3 | 69.8 ± 1.1 | 28.9 ± 0.8 | NT | ||
| 19a | OCH3 | 45.3 ± 0.5 | 35.3 ± 0.6 | 34.0 ± 0.3 | 41.4 ± 2.5 | 43.1 ± 1.6 | 40.2 ± 1.2 | 66.6 ± 0.1 | ||
|
Compond |
DPPH µmol GAE/mmol |
ABTS µmol GAE/mmol |
FRAP µmol GAE/mmol |
|---|---|---|---|
| 11a | 20.5 (±4.0) | 150.6 (±4.7) | 62.2 (±9.9) |
| 11b | 18.9 (±2.3) | 5.7 (±2.7) | 47.2 (±1.0) |
| 11c | 23.2 (±11.9) | 41.4 (±6.7) | 27.4 (±0.9) |
| 11f | 227.3 (±11.0) | 175.1 (±10.4) | 183.5 (±0.9) |
| 13a | 14.2 (±0.4) | 292.4 (±12.4) | 125.2 (±9.4) |
| 13b | 7.6 (±3.8) | 116.8 (±5.8) | 41.2 (±0.7) |
| 13c | 30.3 (±15.4) | 214.6 (±28.4) | 43.4 (±1.8) |
| 14a | 12.3 (±2.3) | 194.8 (±21.4) | 105.5 (±3.4) |
| 14b | 7.3 (±3.1) | 84.6 (±2.7) | 114.3 (±4.2) |
| 14c | 21.1 (±5.8) | 75.9 (±5.3) | 42.0 (±2.0) |
| 17a | 16.0 (±1.1) | 227.6 (±5.4) | 161.6 (±5.7) |
| 17b | 20.7 (±6.6) | 218.9 (±5.6) | 156.2 (±1.1) |
| 17c | 13.6 (±0.9) | 209.0 (±6.6) | 134.1 (±6.7) |
| 18a | 520.1 (±6.9) | 401.2 (±39.2) | 480.9 (±28.8) |
| 18b | 589.1 (±8.9) | 487.3 (±43.9) | 564.8 (±2.2) |
| 18c | 551.1 (±14.9) | 298.1 (±21.0) | 432.0 (±21.4) |
| 19a | 223.5 (±11.2) | 564.7 (±74.5) | 307.2 (±11.9) |
| 19b | 225.6 (±14.7) | 410.7 (±28.9) | 262.2 (±14.2) |
| 19c | 207.7 (±8.6) | 456.0 (±24.6) | 296.2 (±10.4) |
| p-coumaric acid | 23.0 (±3.1) | 278.8 (±11.1) | 105.2 (±4.2) |
| caffeic acid | 690.7 (±36.6) | 586.0 (±32.8) | 500.0 (±33.5) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





