1. Introduction
In genetic research, deoxyribose nucleic acid (DNA) amplification serves as a crucial technique that empowers researchers to examine and manipulate genetic information effectively. This method involves generating multiple copies of a specific DNA sequence, enabling in-depth investigations into genetic material and the functions of particular genes. Its remarkable sensitivity to detect even minute amounts of target molecules makes it indispensable in molecular diagnostics [
1].
Among the prominent techniques for DNA amplification, the polymerase chain reaction (PCR) stands out as the gold standard. PCR plays a pivotal role in duplicating individual DNA segments through various reactions under varying temperatures, which can then be sequenced to identify genetic variants or point mutations. Point mutations encompass alterations in a single DNA base, including additions, deletions, or changes. This is hypothesized to be caused by tautomeric mutations. Tautomers are isomers that can rapidly convert in a solution often differing in proton position [
2]. Analyzing these mutations within the context of existing scientific knowledge provides insights into their implications across various disciplines related to personal health such as nutrition.
Quantum tunneling, a concept derived from quantum mechanics, holds fascinating potential for comprehending tautomeric mutations in DNA. This phenomenon allows particles to surpass energy barriers that classical physics considers insurmountable. Consequently, nucleotide bases can overcome energy obstacles and form unconventional base pairs during replication. The likelihood of quantum tunneling differs across various systems. However, in a biological context, this phenomenon leads to the creation of unconventional base pairs. These pairs play a role in genetic variation, vulnerability to illnesses, and responsiveness to therapies [
3].
This paper aims to explore the effects of temperature fluctuations in PCR on the probability of quantum tunneling during DNA amplification. The significance of this research lies in its potential to inform the development of more sensitive and accurate molecular diagnostic techniques, provide insights into how point mutations occur at the molecular level, and expand knowledge of the interplay between quantum effects and biological systems, potentially opening up new avenues for scientific exploration and technological applications.
2. Materials and Methods
In the study conducted to examine the impact of temperature on the probability of quantum tunneling in DNA, a simulation model was employed. This model utilized a Gaussian wavefunction and the finite-difference time-domain method. The entire simulation was executed on Google Colab, utilizing a system configuration consisting of 12.7 GB of System RAM and 107.7 GB of Disk space.
The simulation methodology was rooted in the 1D time-dependent Schrödinger equation, as originally proposed by Nishantsule in 2019 [
4]. However, certain adjustments were made to the model to align it with the specific research objectives of this study.
To initiate the simulations, initial parameters were chosen in accordance with the findings of [
3]. These parameters encompassed the barrier height in eV, barrier width in Angstrom, proton energy in eV, and proton wavefunction spread in Angstrom.
A noteworthy constraint in the study was the computational limitations, which led to the decision to focus solely on the median temperature recorded during each PCR step. This pragmatic approach allowed for a meaningful exploration of the temperature-dependent aspects of quantum tunneling in DNA, effectively balancing the research goals with the available computational resources. The integration of these methodologies and considerations constituted the foundation of the investigation into the intriguing phenomenon of quantum tunneling in DNA under varying temperature conditions.
3. Results
To establish uniform starting conditions, normalization was conducted through the implementation of the Boltzmann Distribution.
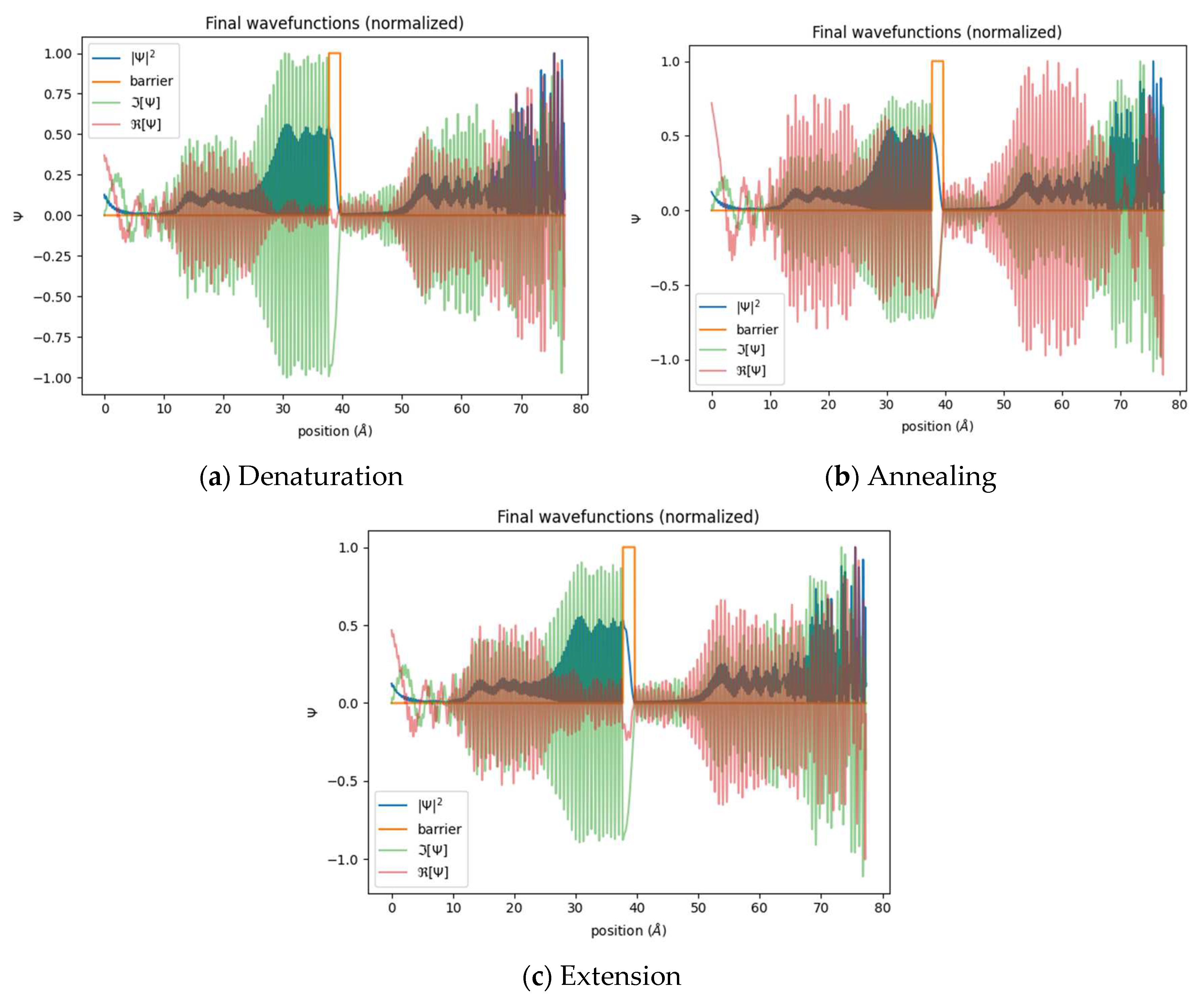
Figure 1.
Temperature dependent quantum proton tunneling simulation. (a) Denaturation at 95 °C, (b) Annealing at 60 °C, (c) Extension at 71 °C.
Figure 1.
Temperature dependent quantum proton tunneling simulation. (a) Denaturation at 95 °C, (b) Annealing at 60 °C, (c) Extension at 71 °C.
The simulation results demonstrated a consistent probability distribution for quantum tunneling, shown in blue, across a range of PCR temperatures, with probabilities spanning approximately 0.5 to 0. Interestingly, distinct oscillating patterns emerged after passing through the energy barrier. These oscillations were particularly noticeable during the annealing and extension phases compared to the denaturation phase. The increased oscillations in the probability wave function after crossing the barrier suggest complex interactions involving multiple-path interference and quantum reflection. These observed oscillations suggest the potential for resonant tunneling. These intricate oscillations arise from the interplay between energy level interactions, bounded states, and the particle’s wavelength interacting with the energy barrier, resulting in complex interference phenomena. Examining the oscillations’ imaginary (green) and real (red) components provides valuable insights into the intrinsic nature of the tunneling process. Notably, the imaginary component dominates before breaching the barrier, while the real component becomes more prominent after passing the barrier. This difference suggests potential issues with the initial conditions imposed on the model. Given the temperature-dependent changes in proton energy, temperature variations could significantly impact the overall energy distribution within the system. As a result, the initial conditions for the denaturation simulation may not align with classical principles. On the contrary, a robust simulation model should accurately depict scenarios where real oscillations take precedence before surpassing the energy barrier. This behavior is expected when the particle’s energy is lower than the barrier’s height, indicating compliance with classically allowed realms. Upon overcoming the barrier, the notable increase in imaginary oscillations marks the beginning of the quantum tunneling phenomenon. This is when the particle’s wave function enters classically forbidden areas, and its amplitude experiences exponential decay while moving through the barrier. The importance of the imaginary component increases as the amplitude decreases in the barrier zone.
4. Conclusions
In conclusion, the study’s exploration of quantum tunneling within the polymerase chain reaction (PCR) process offers intriguing insights into the interplay between quantum phenomena and DNA amplification. However, it’s important to acknowledge the limitations of the model in drawing definitive conclusions. The complex oscillations and behaviors observed during PCR, combined with the temperature-dependent nature of quantum effects, suggest intricate interactions that require further investigation. The study underscores the need for more comprehensive experimental validations and refined models to ascertain the true extent of quantum tunnelling’s influence on DNA amplification dynamics.
With these considerations in mind, several recommendations emerge:
Development of a more accurate and inclusive model: Future research should prioritize the creation of a model that can faithfully replicate biological samples from their natural environment. Such an improved model would better capture the complexities of quantum tunneling within DNA amplification.
Evaluation of alternative kinetic models: The exploration of other kinetic models that can provide a more comprehensive description of quantum proton tunneling is warranted. Diversifying the range of models may offer insights into different facets of quantum effects in biological processes.
Investigation of additional parameters: Beyond temperature, researchers should examine other parameters within PCR machines that may contribute to quantum tunneling probability. Identifying these factors could lead to a more complete understanding of the quantum mechanical aspects of DNA amplification.
Exploration of tautomeric mutations: Establishing a solid connection between tautomeric mutations and replication/translation errors is essential. Investigating this relationship can help clarify how quantum tunneling influences genetic variations and molecular processes more precisely.
These recommendations collectively represent crucial steps toward a deeper comprehension of the interplay between quantum mechanics and biology. While the study’s results provide a promising glimpse into this potential connection, additional research is necessary to uncover the full scope of its impact on genetic variations and molecular processes.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy reasons.
Conflicts of Interest
The author declare no conflict of interest.
References
- Oliveira, B.B.P.P.; Veigas, B.; Baptista, P.V. Isothermal Amplification of Nucleic Acids: The Race for the next “Gold Standard. ” Frontiers in Sensors 2021, 2. [Google Scholar] [CrossRef]
- Al-Sehemi, A.G.; El-Gogary, T.M.; Wolschann, K.P.; Koehler, G. Structure and Stability of Chemically Modified DNA Bases: Quantum Chemical Calculations on 16 Isomers of Diphosphocytosine. ISRN Physical Chemistry (Online) 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Slocombe, L.; Al-Khalili, J.; Sacchi, M. Quantum and Classical Effects in DNA Point Mutations: Watson–Crick Tautomerism in AT and GC Base Pairs. Physical Chemistry Chemical Physics 2021, 23, 4141–4150. [Google Scholar] [CrossRef] [PubMed]
- Nishantsule GitHub - Nishantsule/Quantum-Tunneling: A Wave Function Tunneling across a Barrier. Available online: https://github.com/nishantsule/Quantum-Tunneling.
- Boster Biological Technology PCR and Molecular Biology Fundamental Principles. Available online: https://www.bosterbio.com/protocol-and-troubleshooting/pcr-principle.
- Hood, L.; Rowen, L. The Human Genome Project: Big Science Transforms Biology and Medicine. Genome Medicine 2013, 5, 79. [Google Scholar] [CrossRef] [PubMed]
- Burton, E. Module 4.2: Denaturation, Annealing, and Primer Extension. Available online: https://pressbooks.umn.edu/cvdl/chapter/module-4-4-denaturation-annealing-and-primer-extension/.
- Acosta-Reyes, F.J.; Alechaga, E.; Subirana, J.A.; Campos, J.L. Structure of the DNA Duplex d(ATTAAT)2 with Hoogsteen Hydrogen Bonds. PLOS ONE 2015, 10, e0120241. [Google Scholar] [CrossRef] [PubMed]
- Erjavec, M.S. Annealing Temperature of 55°C and Specificity of Primer Binding in PCR Reactions. In Synthetic Biology; 2020.
- Porta, A.R.; Enners, E. Determining Annealing Temperatures for Polymerase Chain Reaction. American Biology Teacher 2012, 74, 256–260. [Google Scholar] [CrossRef]
- Wang, X.; Son, A. Effects of Pretreatment on the Denaturation and Fragmentation of Genomic DNA for DNA Hybridization. Environmental Science: Processes & Impacts 2013, 15, 2204. [Google Scholar] [CrossRef]
- Wolfe, M. Quantum Tunneling in DNA. Available online: https://www.semanticscholar.org/paper/Quantum-Tunneling-in-DNA-Wolfe/4d3641009241666ea53a47517c35c2651a13cf54.
- Hoque, M.E.; Rajaee, N. Encoding Information in DNA: From Basic Structure to Nanoelectronics. World Journal of Nanomaterials 2017, 5, 51–58. [Google Scholar] [CrossRef]
- Nelson, D.W.; Cox, M. Lehninger Principles of Biochemistry, 7th Ed; 2021.
- Krasilnikov, P.M. Two-Dimensional Model of a Double-Well Potential: Proton Transfer upon Hydrogen Bond Deformation. Biophysics 2014. [CrossRef]
- Gutzwiller, M.C. Chaos in Classical and Quantum Mechanics; Springer Science & Business Media, 2013.
- Hydrogen Bond (Molecular Biology). Available online: http://what-when-how.com/molecular-biology/hydrogen-bond-molecular-biology/.
- Umesaki, K.; Odai, K. A Kinetic Approach to Double Proton Transfer in Watson–Crick DNA Base Pairs. Journal of Physical Chemistry B 2020. [CrossRef] [PubMed]
- Ghasemi, F.; Tirandaz, A. Environment Assisted Quantum Model for Studying RNA-DNA-Error Correlation Created Due to the Base Tautomery. Scientific Reports 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Luo, J. Sub-Picosecond Proton Tunnelling in Deformed DNA Hydrogen Bonds under an Asymmetric Double-Oscillator Model. European Physical Journal E 2018, 41. [Google Scholar] [CrossRef] [PubMed]
- Turaeva, N.N.; Brown-Kennerly, V. Marcus Model of Spontaneous Point Mutation in DNA. Chemical Physics 2015, 461, 106–110. [Google Scholar] [CrossRef]
- Parker, B.; Van Everv, J. Quantum Tunnelling in DNA. Chemical Physics Letters 1971, 8, 94–99. [Google Scholar] [CrossRef]
- Srivastava, R. The Role of Proton Transfer on Mutations. Frontiers in Chemistry 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Löwdin, P.-O. Proton Tunneling in DNA and Its Biological Implications. Reviews of Modern Physics 1963, 35, 724–732. [Google Scholar] [CrossRef]
- Chaudhury, R. Tautomeric Mutation in DNA. A Theoretical Comparative Study and Synthesis between Various Proposed Quantum... ResearchGate 2015.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).