Submitted:
08 November 2023
Posted:
09 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Setting and Subjects
2.2. Procedure and instruments
2.3. Samples
2.4. Assessment of Biochemical Indices and Hormones
2.5. Statistical analysis
3. Results
3.1. Clinical characteristics of the patients
3.2. Biochemical characteristics of the patient and control groups
3.3. Distribution of clinical and biochemical parameters according to the importance of their influence on the development of NSSI in patients with NPMD
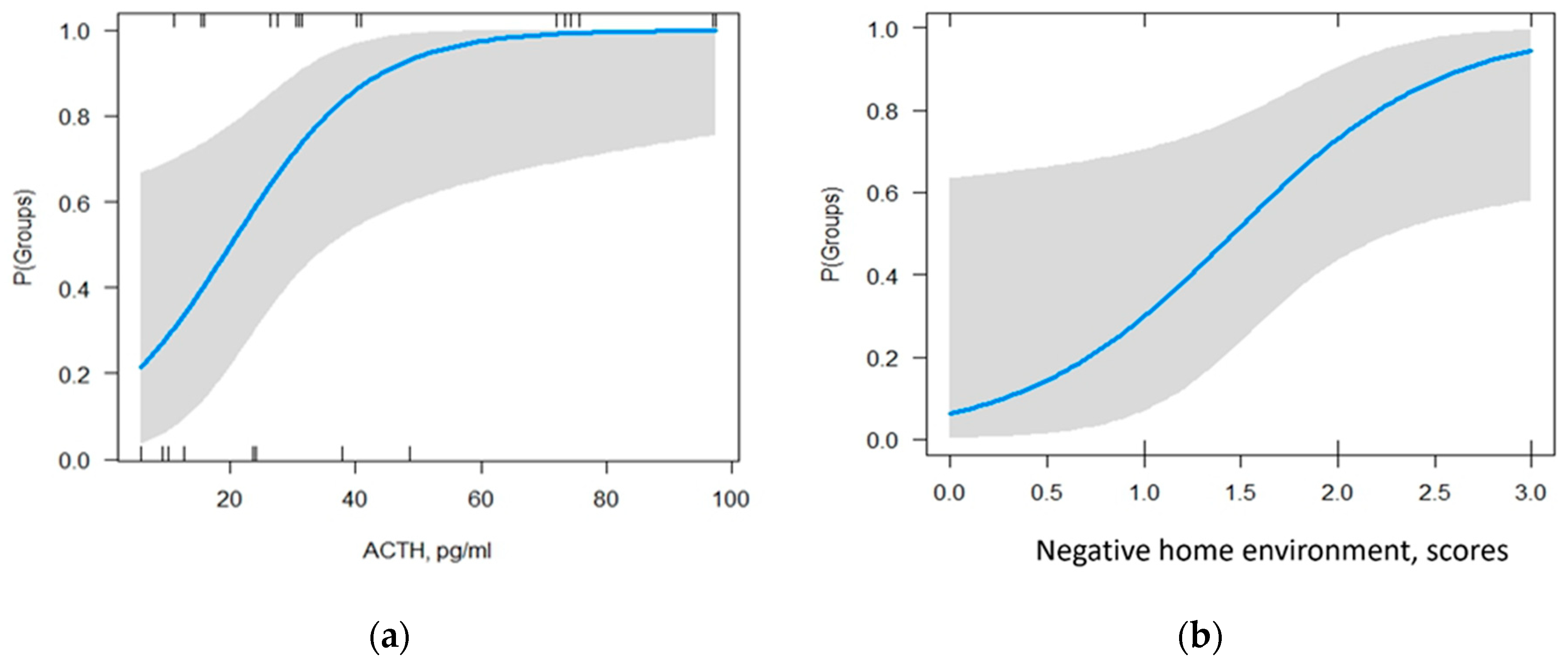
3.4. Model for predicting the probability of developing NSSI in patients with NPMD
4. Discussion
5. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbrevations
References
- Diagnostic And Statistical Manual Of Mental Disorders, Fifth Edition. American Psychiatric Association Publishing. [CrossRef]
- Wang, M.; Eaton, N.R. Linking non-suicidal self-injury to psychopathology: The utility of transdiagnostic and DSM-based models. J. Affect. Disord. 2023, 332, 55-63. [CrossRef]
- Eaton, N.R.; Krueger, R.F.; Markon, K.E.; Keyes, K.M.; Skodol, A.E.; Wall, M.; Hasin, D.S.; Grant, B.F. The structure and predictive validity of the internalizing disorders. J. Abnorm. Psychol. 2013, 122, 86-92. [CrossRef]
- Karnick, A.T.; Fergerson, A.K.; Caulfield, N.M.; Buerke, M.; Albanese, B.J.; Schmidt, N.B.; Capron, D.W. Toward an integrative model of transdiagnostic risk factors and suicide: A network comparison of psychiatric outpatients. J. Psychiatr. Res. 2022, 154,252-260. [CrossRef]
- Nitkowski, D.; Petermann, F. Selbstverletzendes Verhalten und komorbide psychische Störungen: ein Überblick [Non-suicidal self-injury and comorbid mental disorders: a review]. Fortschr. Neurol. Psychiatr.2011,79, 9-20. [CrossRef]
- Kiekens, G.; Claes, L.; Hasking, P.; Mortier, P.; Bootsma, E.; Boyes, M.; Myin-Germeys, I.; Demyttenaere, K.; Cuijpers, P.; Kessler, R.C.; Nock, M.K.; Bruffaerts, R. A longitudinal investigation of non-suicidal self-injury persistence patterns, risk factors, and clinical outcomes during the college period. Psychol. Med.2023,53, 6011-6026. [CrossRef]
- Kang, L.; Li, R.; Liu, H.; Ma, S.; Sun, S.; Zhang, N.; Yao, L.; Wang, Y.; Zong, X.; Ai, C.; Zou, Z.; Yang, B.X.; Bai, H.; Liu, Z. Nonsuicidal self-injury in undergraduate students with major depressive disorder: The role of psychosocial factors. J. Affect. Disord. 2021, 290, 102-108. [CrossRef]
- Groschwitz, R.C.; Kaess, M.; Fischer, G.; Ameis, N.; Schulze, U.M.; Brunner, R.; Koelch, M.; Plener, P.L. The association of non-suicidal self-injury and suicidal behavior according to DSM-5 in adolescent psychiatric inpatients. Psychiatry Res. 2015, 228, 454-461. [CrossRef]
- Shao, C.; Wang, X.; Ma, Q.; Zhao, Y.; Yun, X. Analysis of risk factors of non-suicidal self-harm behavior in adolescents with depression. Ann. Palliat. Med. 2021, 10, 9607-9613. [CrossRef]
- Taş Torun, Y., Gul, H.; Yaylali, F.H.; Gul, A. Intra/interpersonal Functions of Non-suicidal Self-injury in Adolescents with Major Depressive Disorder: The Role of Emotion Regulation, Alexithymia, and Childhood Traumas. Psychiatry2022, 85, 86-99. [CrossRef]
- Kiekens, G.; Hasking, P.; Boyes, M.; Claes, L.; Mortier, P.; Auerbach, R.P.; Cuijpers, P.; Demyttenaere, K.; Green, J.G.; Kessler, R.C.; Myin-Germeys, I.; Nock, M.K.; Bruffaerts, R. The associations between non-suicidal self-injury and first onset suicidal thoughts and behaviors. J. Affect. Disord. 2018, 239, 171-179. [CrossRef]
- Koenig, J., Brunner, R., Fischer-Waldschmidt, G., Parzer, P., Plener, P.L., Park, J., Wasserman, C., Carli, V., Hoven, C.W., Sarchiapone, M., Wasserman, D., Resch, F., Kaess, M. Prospective risk for suicidal thoughts and behaviour in adolescents with onset, maintenance or cessation of direct self-injurious behaviour. Eur. Child. Adolesc. Psychiatry2017, 26, 345-354. [CrossRef]
- Hamza, C.A.; Stewart, S.L.; Willoughby, T. Examining the link between nonsuicidal self-injury and suicidal behavior: a review of the literature and an integrated model. Clin. Psychol. Rev. 2012, 32, 482-495. [CrossRef]
- Ribeiro, J.D., Franklin, J.C., Fox, K.R., Bentley, K.H., Kleiman, E.M., Chang, B.P., Nock, M.K. Self-injurious thoughts and behaviors as risk factors for future suicide ideation, attempts, and death: a meta-analysis of longitudinal studies. Psychol. Med.2016, 46, 225-236. [CrossRef]
- Bryan, C.J., Rudd, M.D., Wertenberger, E., Young-McCaughon, S., Peterson, A. Nonsuicidal self-injury as a prospective predictor of suicide attempts in a clinical sample of military personnel. Compr. Psychiatry2015, 59:1-7. [CrossRef]
- Miller, G.E., Chen, E., Zhou, E.S. If it goes up, must it come down? Chronic stress and the hypothalamic-pituitary-adrenocortical axis in humans. Psychol. Bull.2007, 133, 25-45. [CrossRef]
- Reichl, C., Heyer, A., Brunner, R., Parzer, P., Völker, J.M., Resch, F., Kaess, M. Hypothalamic-pituitary-adrenal axis, childhood adversity and adolescent nonsuicidal self-injury. Psychoneuroendocrinology2016, 74, 203-211. [CrossRef]
- Lupien, S.J., McEwen, B.S., Gunnar, M.R., Heim, C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat. Rev. Neurosci.2009, 10, 434-445. [CrossRef]
- Kaess, M., Hooley, J.M., Klimes-Dougan, B., Koenig, J., Plener, P.L., Reichl, C., Robinson, K., Schmahl, C., Sicorello, M., Westlund Schreiner, M., Cullen, K.R. Advancing a temporal framework for understanding the biology of nonsuicidal self- injury: An expert review. Neurosci. Biobehav. Rev.2021, 130, 228-239. [CrossRef]
- Klimes-Dougan, B.; Begnel, E.; Almy, B.; Thai, M.; Schreiner, M.W.; Cullen, K.R. Hypothalamic-pituitary-adrenal axis dysregulation in depressed adolescents with non-suicidal self-injury. Psychoneuroendocrinology2019, 102, 216-224. [CrossRef]
- Bresin, K.; Schoenleber, M. Gender differences in the prevalence of nonsuicidal self-injury: A meta-analysis. Clin. Psychol. Rev.2015, 38, 55-64. [CrossRef]
- Gutiérrez-Rojas L.; Porras-Segovia A.; Dunne H.; Andrade-González N.; Cervilla J.A. Prevalence and correlates of major depressive disorder: a systematic review. Braz. J. Psychiatry2020, 42, 657-672. [CrossRef]
- Zinchuk, M.; Beghi, M.; Beghi, E.; Bianchi, E.; Avedisova, A.; Yakovlev, A.; Guekht, A. Non-Suicidal Self-Injury in Russian Patients with Suicidal Ideation. Arch. Suicide Res.2022, 26, 776-800. [CrossRef]
- The global health observatory. Suicide rates. https://www.who.int/data/gho/data/themes/mental-health/suicide-rates.
- World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. J.A.M.A. 2013, 310, 2191-2194. [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res.1975, 12, 189-198. [CrossRef]
- Nock, M.K.; Holmberg, E.B.; Photos, V.I.; Michel, B.D. Self-Injurious Thoughts and Behaviors Interview: development, reliability, and validity in an adolescent sample. Psychol. Assess.2007, 19, 309-317. [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Manual for the Beck Depression Inventory, 2nd ed.; (BDI-II); Psychological Corporation: San Antonio, TX, USA, 1996.
- Spielberger, C. D.; Gorsuch, R. L.; Lushene, R.; Vagg, P. R.; Jacobs, G. A. Manual for the State-Trait Anxiety Inventory. Palo Alto, CA: Consulting Psychologists Press.1983.
- Ivanoff, A.; Jang, S.J.; Smyth, N.J.;Linehan,M. M. Fewer reasons for staying alive when you are thinking of killing yourself: The brief reasons for living inventory. J. Psychopathol. Behav. Assess.1994, 16, 1–13. [CrossRef]
- Kustov, G.V.; Zinchuk, M.S.; Gersamija, A.G.; Voinova, N.I.; Yakovlev, A.A.; Avedisova, A.S.; Guekht, A.B. Psychometric properties of the Russian version of the brief «Reasons for Living Inventory». Zh. Nevrol. Psikhiatr. Im. S. S. Korsakova2021, 121, 87-94. [CrossRef]
- The Personality Inventory for DSM-5—Brief Form (PID-5-BF)—Adult https://www.psychiatry.org/getmedia/f65c4386-b2bc-44a5-9ace-d6fea2211506/APA-DSM5TR-ThePersonalityInventoryForDSM5BriefFormAdult.pdf.
- Sanders, B.; Becker-Lausen, E. The measurement of psychological maltreatment: early data on the Child Abuse and Trauma Scale. Child Abuse Negl.1995, 19, 315-323. [CrossRef]
- Maciejewski, DF, Creemers, HE, Lynskey, MT, Madden, PA, Heath, AC, Statham, DJ, Martin, NG, Verweij, KJ. Overlapping genetic and environmental influences on nonsuicidal self-injury and suicidal ideation: different outcomes, same etiology? J.A.M.A. Psychiatry2014, 71, 699-705. [CrossRef]
- James, K. M.; Brandon, E. G. The Parent-Child Dyad and Other Family Factors Associated with Youth Nonsuicidal Self-Injury.The Oxford Handbook of Nonsuicidal Self-Injury, Lloyd-Richardson, E.E.; Baetens E.I; Whitlock J.L. (eds). 2023. [CrossRef]
- Nederhof, E.; van Oort, F.V.; Bouma, E.M.; Laceulle, O.M.; Oldehinkel, A.J.; Ormel, J. Predicting mental disorders from hypothalamic-pituitary-adrenal axis functioning: a 3-year follow-up in the TRAILS study. Psychol. Med. 2015, 45, 2403-2412. [CrossRef]
- Wu, B.; Zhang, H.; Chen, G.; Chen, G.; Liu, Z.; Cheng, Y.; Yuan, N.; Peng, D. Potential mechanisms of non-suicidal self-injury (NSSI) in major depressive disorder: a systematic review. 2023, General Psychiatry 36, e100946. [CrossRef]
- Braquehais, M.D.; Picouto, M.D.; Casas, M.; Sher, L. Hypothalamic-pituitary-adrenal axis dysfunction as a neurobiological correlate of emotion dysregulation in adolescent suicide. World J. Pediatr. 2012, 8, 197-206. [CrossRef]
- Klimes-Dougan, B.; Begnel, E.; Almy, B.; Thai, M.; Schreiner, M.W.; Cullen, K.R. Hypothalamic-pituitary-adrenal axis dysregulation in depressed adolescents with non-suicidal self-injury. Psychoneuroendocrinology2019, 102, 216-224. [CrossRef]
- Wingenfeld, K.; Wolf, O.T. HPA axis alterations in mental disorders: impact on memory and its relevance for therapeutic interventions. C.N.S. Neurosci. Ther.2011, 17, 714-722. [CrossRef]
- McKlveen, J.M.; Myers, B.; Flak, J.N.; Bundzikova, J.; Solomon, M.B.; Seroogy, K.B.; Herman, J.P. Role of prefrontal cortex glucocorticoid receptors in stress and emotion. Biol. Psychiatry2013, 74, 672-679. [CrossRef]
- Lalli, E., Figueiredo, B.C. Prolactin as an adrenocorticotropic hormone: Prolactin signalling is a conserved key regulator of sexually dimorphic adrenal gland function in health and disease. Bioessays2022, 44, e2200109. [CrossRef]
- Garner, B., Pariante, C.M., Wood, S.J., Velakoulis, D., Phillips, L., Soulsby, B., Brewer, W.J., Smith, D.J., Dazzan, P., Berger, G.E., Yung, A.R., van den Buuse, M., Murray, R., McGorry, P.D., Pantelis, C. Pituitary volume predicts future transition to psychosis in individuals at ultra-high risk of developing psychosis. Biol. Psychiatry2005, 58, 417-423. [CrossRef]
- Ganella, D.E., Allen, N.B., Simmons, J.G., Schwartz, O., Kim, J.H., Sheeber, L., Whittle, S. Early life stress alters pituitary growth during adolescence-a longitudinal study. Psychoneuroendocrinology2015, 53, 185-194. [CrossRef]
- Schär, S., Mürner-Lavanchy, I., Slavova, N., Lerch, S., Reichl, C., Brunner, R., Koenig, J., Kaess, M. Pituitary volume in adolescents with non-suicidal self-injury: Preliminary evidence for alterations in pituitary maturation. Psychoneuroendocrinology2022, 138, 105662. [CrossRef]
- Dorn, L.D. Measuring puberty. J. Adolesc. Health2006, 39, 625-626. [CrossRef]
- Ordaz, S, Luna, B. Sex differences in physiological reactivity to acute psychosocial stress in adolescence. Psychoneuroendocrinology2012, 37, 1135-1157. [CrossRef]
- Morrison, K.E., Rodgers, A.B., Morgan, C.P., Bale, T.L. Epigenetic mechanisms in pubertal brain maturation. Neuroscience2014, 264, 17-24. [CrossRef]
- Handa, R.J., Weiser, M.J. Gonadal steroid hormones and the hypothalamo-pituitary- adrenal axis. Front. Neuroendocrinol.2014, 35, 197-220. [CrossRef]
- Teicher, M.H., Samson, J.A. Annual Research Review: Enduring neurobiologicaleffects of childhood abuse and neglect. J. Child. Psychol. Psychiatry2016, 57, 241-266. [CrossRef]
- Liu, R.T., Scopelliti, K.M., Pittman, S.K., Zamora, A.S. Childhood maltreatment and non-suicidal self-injury: a systematic review and meta-analysis. Lancet Psychiatry2018, 5, 51-64. [CrossRef]
- Brown, R.C., Heines, S., Witt, A., Braehler, E., Fegert, J.M., Harsch, D., Plener, P.L. The impact of child maltreatment on non-suicidal self-injury: data from a representative sample of the general population. BMC Psychiatry2018, 18, 181. [CrossRef]
- Franzke, I., Wabnitz, P., Catani, C. Dissociation as a mediator of the relationship between childhood trauma and nonsuicidal self-injury in females: a path analytic approach. J. Trauma Dissociation2015, 16, 286-302. [CrossRef]
- Chen, Y.; Zhao, X.; Liu, X.; Wu, D.; Zhan, S.; Liu, S.; Yin, H. Childhood maltreatment and non-suicidal self-injury among Chinese adolescents: a moderated mediation model of psychological suzhi and inferiority complex. Curr Psychol.2023. [CrossRef]
- Titelius, E.N.; Cook, E.; Spas J.; Orchowski, L.; Kivisto, K.; O’Brien, K.H.M.; Frazier, E.; Wolff, J.C.; Dickstein, D.P.; Kim, K.L., Seymour, K. Emotion Dysregulation Mediates the Relationship Between Child Maltreatment and Non-Suicidal Self-Injury. J. Aggress. Maltreat. Trauma2018, 27, 323-331. [CrossRef]
- Kuhlman, K.R.; Chiang, J.J.; Horn, S.; Bower, J.E. Developmental psychoneuroendocrine and psychoneuroimmune pathways from childhood adversity to disease. Neurosci. Biobehav. Rev. 2017, 80, 166-184. [CrossRef]
- Gu, W.; Zhao, Q.; Yuan, C.; Yi, Z.; Zhao, M.; Wang, Z. Impact of adverse childhood experiences on the symptom severity of different mental disorders: a cross- diagnostic study. Gen. Psychiatry2022, 35, e100741. [CrossRef]
- Carballedo, A., Lisiecka, D., Fagan, A., Saleh, K., Ferguson, Y., Connolly, G., Meaney, J., Frodl, T. Early life adversity is associated with brain changes in subjects at family risk for depression. World J. Biol. Psychiatry2012, 13, 569-578. [CrossRef]
- Farrow, P.; Simmons, J.G.; Pozzi, E.; Díaz-Arteche, C.; Richmond, S.; Bray, K.; Schwartz, O.; Whittle, S. Associations between early life stress and anterior pituitary gland volume development during late childhood. Psychoneuroendocrinology. 2020, 122. [CrossRef]
- Coffman, J. A. Chronic stress, physiological adaptation and developmental programming of the neuroendocrine stress system. Future Neurol. 2023, 15. [CrossRef]
- Stalder, T., Steudte-Schmiedgen, S., Alexander, N., Klucken, T., Vater, A., Wichmann, S., Kirschbaum, C., Miller, R. Stress-related and basic determinants of hair cortisol in humans: A meta-analysis. Psychoneuroendocrinology.2017, 77, 261-274. [CrossRef]
- Gulyaeva, N.V. Glucocorticoids Orchestrate Adult Hippocampal Plasticity: Growth Points and Translational Aspects. Biochemistry (Mosc).2023, 88, 565-589. [CrossRef]
- Miller, A.B.; Linthicum, K.P.; Helms, S.W.; Giletta, M.; Rudolph, K.D.; Hastings, P.D.; Nock, M.K.; Prinstein, M.J. Reciprocal Associations Between Adolescent Girls’ Chronic Interpersonal Stress and Nonsuicidal Self-Injury: A Multi-wave Prospective Investigation. J. Adolesc. Health. 2018, 63, 694-700. [CrossRef]
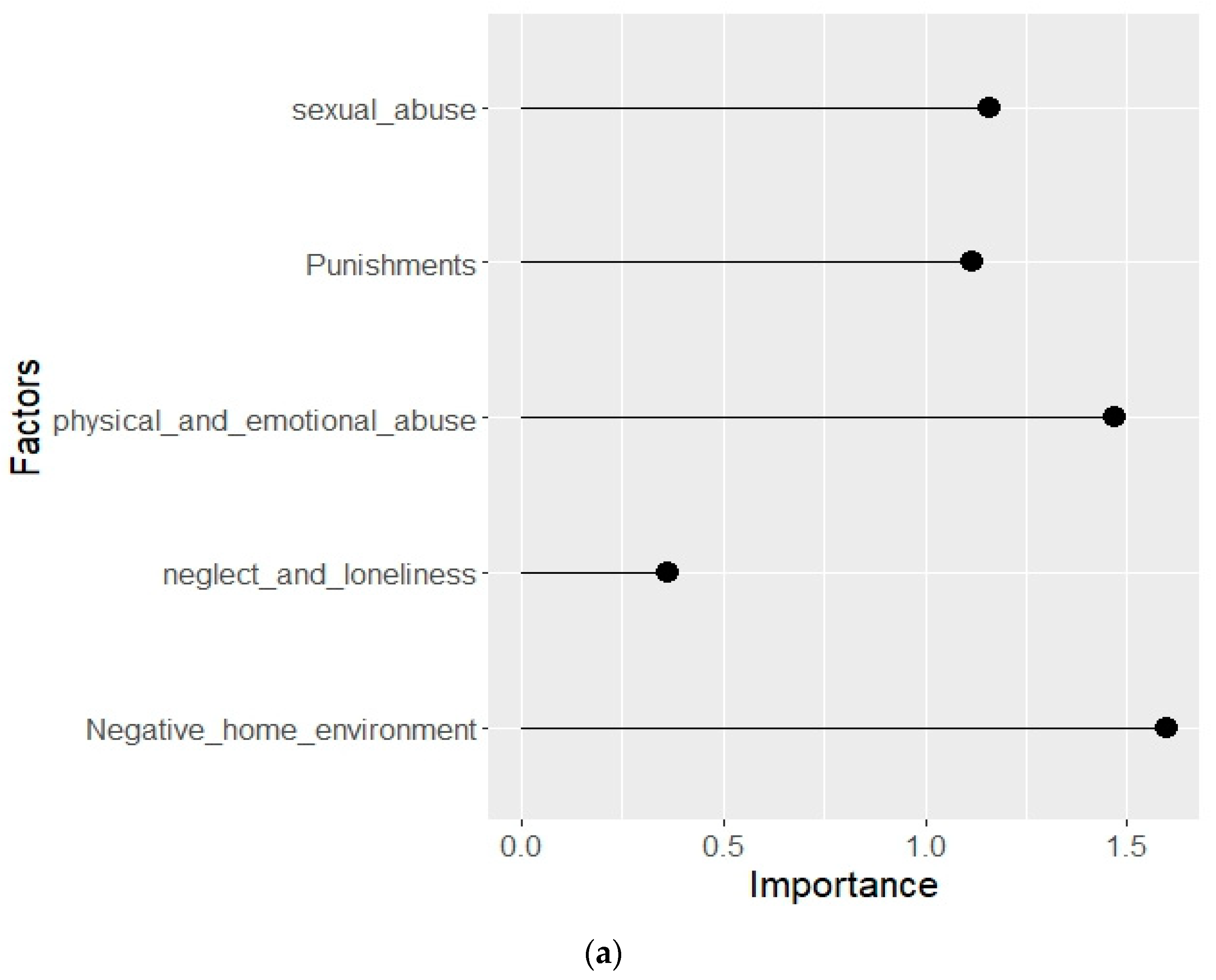
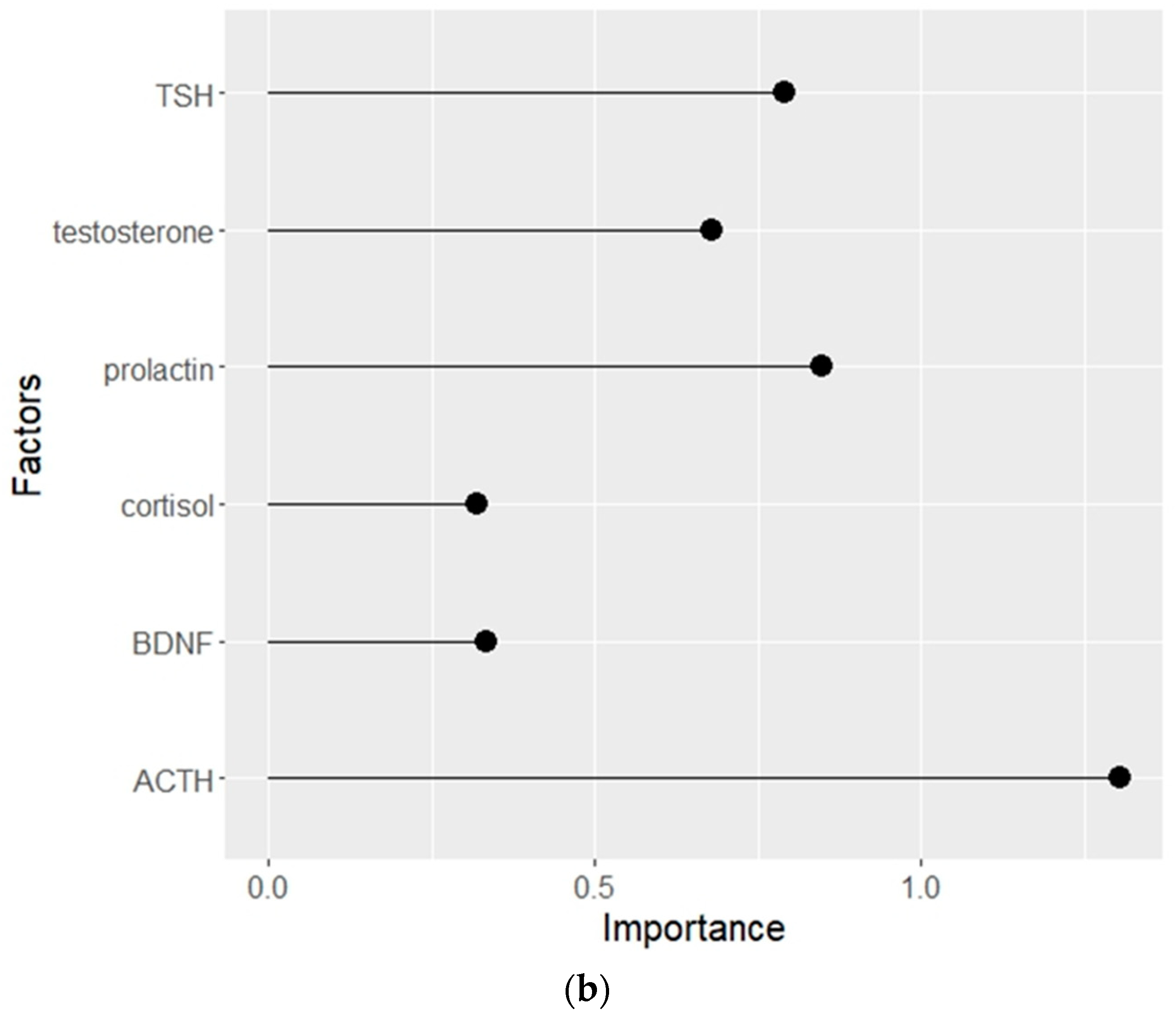
| Parameter/Group | NSSI- | NSSI+ |
|---|---|---|
| Socio-demographic data | ||
| Age (years) | 27.5(22.0;32.0) | 27.5(22.0;31.8) |
| Education (secondary/incomplete higher; higher), % | 35/65 | 28/72 |
| Employed (-/+), % | 52/48 | 50/50 |
| Marital status (single/coupled), % | 55/45 | 50/50 |
| Parent’s family structure (incomplete/complete), % | 50/50 | 45/55 |
| Current diagnosis | ||
| Bipolar affective disorders (F31), % | 12 | 18 |
| Depressive disorders (F32/33), % | 30 | 30 |
| Neurotic and stress-related disorders (F40/41/43/44/45), % | 20 | 5∗ |
| Personality disorders(F60/61), % | 31 | 37 |
| Eating disorders, % | 23 | 40 |
| Mental disorders caused by brain damage and dysfunction (F06/07), % | 5 | 8 |
| Diagnosed with 2 or more mental disorders, % | 28 | 45 |
| Clinical characteristics | ||
| Family history of mental health disorders, % | 65 | 68 |
| Family history of suicidality, % | 20 | 38 |
| Family history of NSSI, % | 5 | 23* |
| History of physical abuse, % | 65 | 83 |
| History of bullying at school, % | 65 | 80 |
| Lifetime suicide attempts, % | 38 | 38 |
| Current smoking (nicotine products), %. | 56 | 55 |
| Age at first contact with mental health services, years | 22.5 (18.5;30) | 22.0 (18.0;28.0) |
| Age of onset of suicidal ideation, years old | 18.0 (13.8;22.0) | 14. 0 (13.0;16.3) |
| Age of onset of NSSI, years old | - | 14.5 (13.0;18.0) |
| Psychological scales data | ||
| BDI, scores | 27.8±9.4 | 31.1±9.1∗ |
| STAI (State Anxiety), scores | 60.2±9.9 | 63.8±9.6 |
| STAI (Trait anxiety), scores | 59.0±8.4 | 62.8±8.6∗ |
| bRFL (total), scores | 34.4±11.7 | 39.7±15.3 |
| PID-5-BF (total), scores | 34,8±9.2 | 32.4±11.4 |
| CATS (total), scores | 7.4±3.2 | 9.0±3.4 |
| Parameter/Group | HC n=48 |
NSSI- n=40 |
NSSI+ n=40 |
|---|---|---|---|
| age | 25.0 (23.0;27.0) | 27.5 (22.0;32.0) | 27.5 (22.0;31.8) |
| TSH3, ulU/mL | 1.7 (1.2;2.3) | 1.7 (1.3;2.1) | 1.9 (1.5;2.7) |
| ACTH, pg/mL | 15.8 (9.7;22.2) | 24.3 (16.0;57.0)*** | 37.4 (26.7;56.8)* |
| Testosterone, nmol/L | 1.4 (1.0;2.1) | 2.5 (1.6;2.9)* | 2.1 (1.5;2.7)* |
| Prolactin, ng/mL | 11.9 (8.2;15.9) | 20.9 (14.8;34.3)* | 19.7 (13.1;36.1)* |
| Cortisol, nmol/L | 265.9 (192.5; 378.8) | 447.6 (381.5;558.7)* | 533.7 (427.1; 600.9)* |
| BDNF, ng/ml | 25.5±5.5 | 22.1±6.0*** | 18.8±5.7* |
| While blood cell, WBC, 103µL | 6.3±1.6 | 6.5±1.5 | 6.0±1.5 |
| Neutrophils, NEI, %/ NEI, 103µL |
56.8±10.8/3.6±1.3 | 50.4±11.3/3.3±1.2∗ | 49.6±10.0/3.0±1.2∗ |
| Lymphocytes, LY, %/LY, 103µL | 32.2±9.6/2.0±0.7 | 37.2±10.0/2.4±0.8∗ | 38.9±9.0/2.3±0.7∗ |
| Monocytes, MO, %/MO, 103µL | 7.7±2.4 | 8.0±2.2 | 8.3±2.2 |
| Eosinophils, EO, % | 2.4±1.1 | 3.5±1.9*** | 2.4±1.3 |
| Basophils, BA, % | 0.8±0.4 | 0.9±0.4** | 0.7±0.3 |
| Erythrocytes, RBC, 106µL | 4.9±0.4 | 4.4±0.3 | 4.4±0.4 |
| Hemoglobin, Hb, g/L | 146.2±13.0 | 134.2±8.0∗ | 133.1±9.6∗ |
| Platelets, PLT, 103µL | 248.6±60.3 | 230.3±52.7 | 253.1±67.5 |
| Parameter | Estimate | Std Error | Statistic | p-Value |
|---|---|---|---|---|
| ACTH, pg/ml | 1.10 | 0.04 | 221 | 0.027 |
| CATS “negative home environment” subscale, scores | 6.37 | 0.9 | 2.06 | 0.039 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





