Submitted:
05 December 2023
Posted:
07 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Ethical Consideration
2.3. Statistical Analysis
3. Results
3.1. Characteristics of Patients
3.2. Skin and Mucosal Lesions
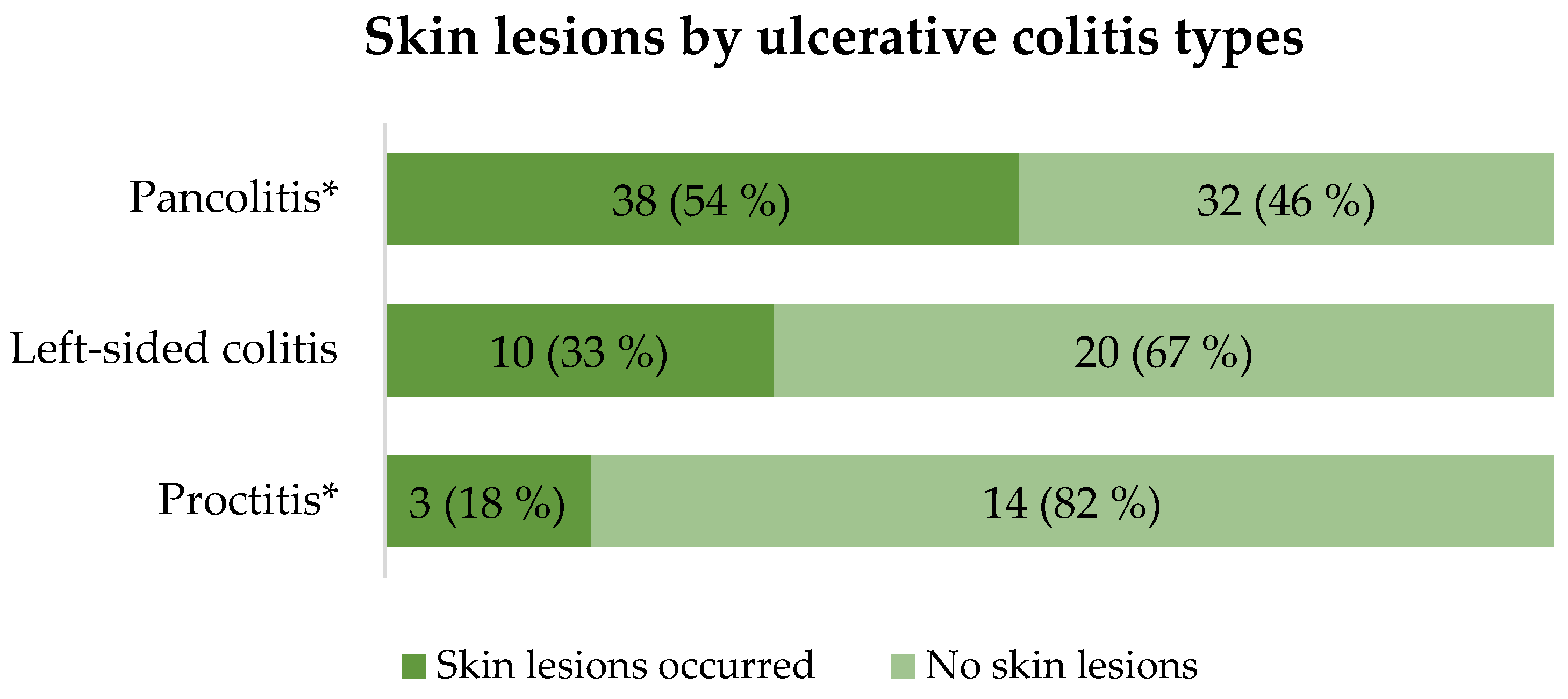
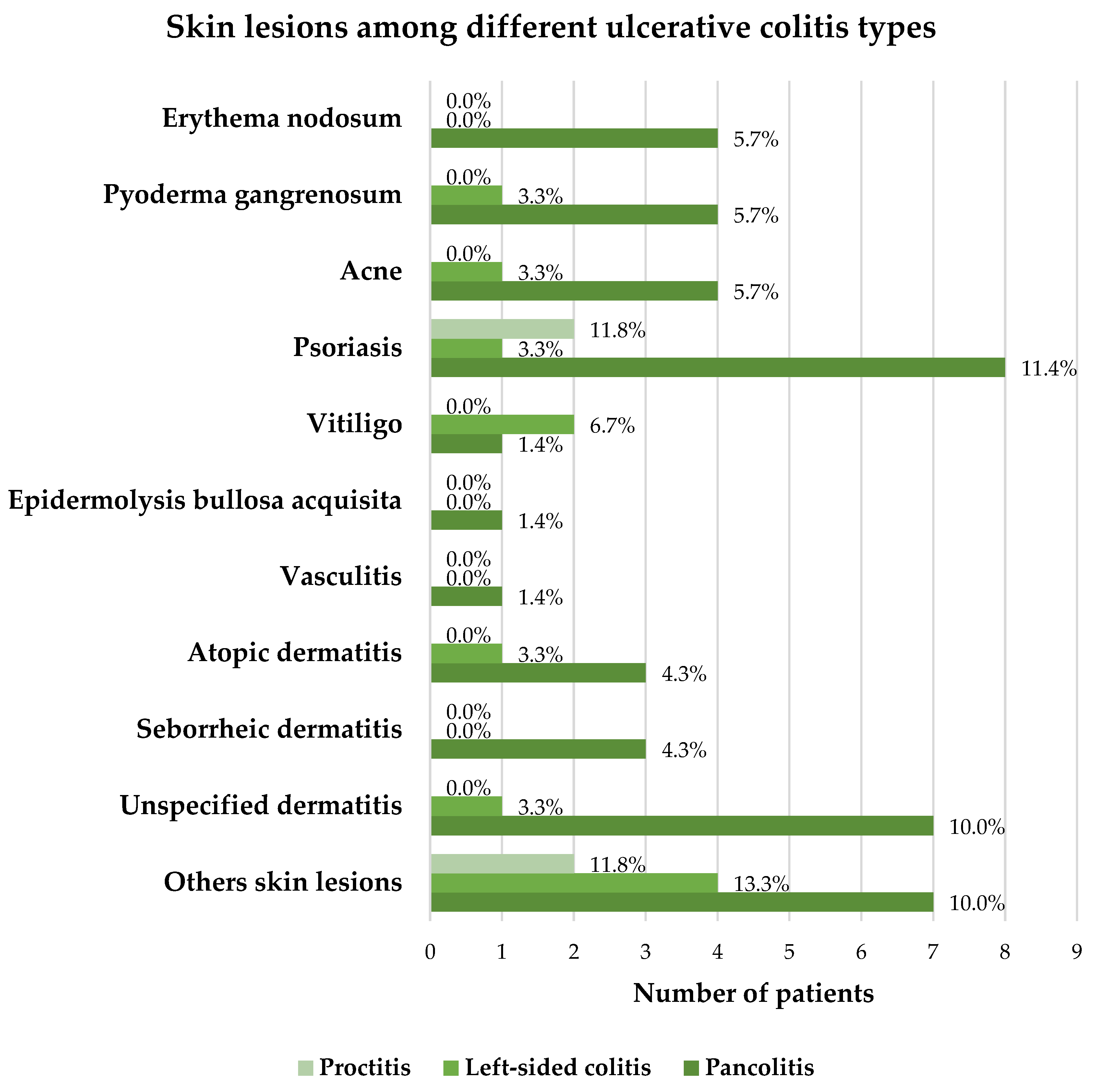
3.3. The Relationship Between Skin and Mucosal Lesions with Extension of IBD
3.4. The Relationship Between Skin and Mucosal Lesions with duration of IBD
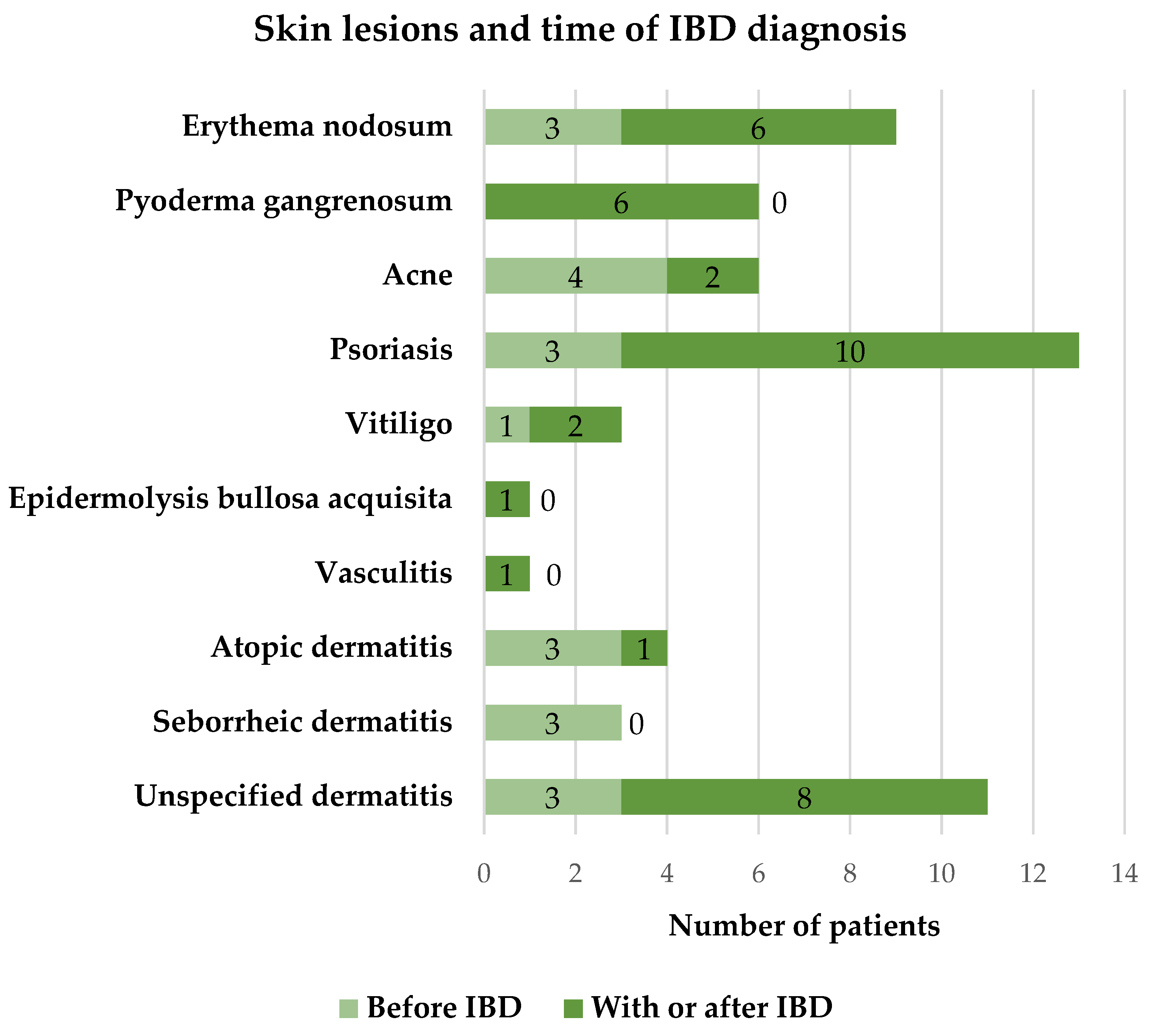
3.5. The Relationship Between Skin Lesions with the Time of IBD Diagnosis
3.6. The Relationship of Skin Lesions and Drugs Used for the Treatment of IBD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol 2020, 5, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.C.; Shi, H.Y.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Wu, J.C.Y.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. The Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- Karpavičiūtė, V.; Kiudelis, G.; Kupčinskas, J.; Kupčinskas, L. Trends in the Prevalence of Inflammatory Bowel Disease in Lithuania during 2001-2020. J Crohn‘s Colitis 2023, 17, 965–966. [Google Scholar] [CrossRef]
- Rogler, G.; Singh, A.; Kavanaugh, A.; Rubin, D.T. Extraintestinal Manifestations of Inflammatory Bowel Disease: Current Concepts, Treatment, and Implications for Disease Management. Gastroenterology 2021, 161, 1118–1132. [Google Scholar] [CrossRef] [PubMed]
- Vavricka, S.R.; Rogler, G.; Gantenbein, C.; Spoerri, M.; Vavricka, M.P.; Navarini, A.A.; French, L.E.; Safroneeva, E.; Fournier, N.; Straumann, A.; et al. Chronological Order of Appearance of Extraintestinal Manifestations Relative to the Time of IBD Diagnosis in the Swiss Inflammatory Bowel Disease Cohort. Inflamm Bowel Dis 2015, 21, 1794–800. [Google Scholar] [CrossRef] [PubMed]
- Karmiris, K.; Avgerinos, A.; Tavernaraki, A.; Zeglinas, C.; Karatzas, P.; Koukouratos, T.; Oikonomou, K.A.; Kostas, A.; Zampeli, E.; Papadopoulos, V.; et al. Prevalence and Characteristics of Extra-intestinal Manifestations in a Large Cohort of Greek Patients with Inflammatory Bowel Disease. J Crohn‘s Colitis 2016, 10, 429–436. [Google Scholar] [CrossRef]
- Demir, F.T.; Kocatürk, E.; Yorulmaz, E.; Adali, G.; Kavala, M. Mucocutaneous manifestations of inflammatory bowel disease in Turkey. J Cutan Med Surg 2014, 18, 397–404. [Google Scholar] [CrossRef]
- Zippi, M.; Corrado, C.; Pica, R.; Avallone, E.V.; Cassieri, C.; De Nitto, D. ; Paoluzi, P.; Vernia, P. Extraintestinal manifestations in a large series of Italian inflammatory bowel disease patients. World J Gastroenterol 2014, 20, 17463–17467. [Google Scholar] [CrossRef]
- Greuter, T.; Navarini, A.; Vavricka, S.R. Skin Manifestations of Inflammatory Bowel Disease. Clin Rev Allergy Immunol 2017, 53, 413–427. [Google Scholar] [CrossRef]
- Antonelli, E.; Bassotti, G.; Tramontana, M.; Hansel, K.; Stingeni, L.; Ardizzone, S.; Genovese, G.; Marzano, A.V.; Maconi, G. Dermatological Manifestations in Inflammatory Bowel Diseases. J Clin Med 2021, 10, 364. [Google Scholar] [CrossRef]
- Keyal, U.; Liu, Y.; Bhatta, A.K. Dermatologic manifestations of inflammatory bowel disease: A review. Discov Med 2018, 25, 225–233. [Google Scholar]
- Plumptre, I.; Knabel, D.; Tomecki, K. Pyoderma Gangrenosum: A Review for the Gastroenterologist. Inflamm Bowel Dis 2018, 24, 2510–2517. [Google Scholar] [CrossRef]
- Sbeit, W.; Kadah, A.; Mahamid, M.; Karayanni, H.; Mari, A.; Tali, S.; Srouji, S.; Khoury, T. Oral manifestations of inflammatory bowel disease: the neglected piece of the puzzle. Eur J Gastroenterol Hepatol 2020, 32, 1422–1431. [Google Scholar] [CrossRef] [PubMed]
- Laranjeira, N.; Fonseca, J.; Meira, T.; Freitas, J.; Valido, S.; Leitão, J. Oral mucosa lesions and oral symptoms in inflammatory bowel disease patients. Arq Gastroenterol 2015, 52, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Harbord, M.; Annese, V.; Vavricka, S.R.; Allez, M.; Acosta, M.B.-D.; Boberg, K.M.; Burisch, J.; De Vos, M.; De Vries, A.-M.; Dick, A.D.; et al. The First European Evidence-based Consensus on Extra-intestinal Manifestations in Inflammatory Bowel Disease. J Crohn’s Colitis 2016, 10, 239–254. [Google Scholar] [CrossRef] [PubMed]
- Vavricka, S.R.; Schoepfer, A.; Scharl, M.; Lakatos, P.L.; Navarini, A.; Rogler, G. Extraintestinal Manifestations of Inflammatory Bowel Disease. Inflamm Bowel Dis 2015, 21, 1982–1992. [Google Scholar] [CrossRef] [PubMed]
- Marzano, A. V.; Borghi, A.; Stadnicki, A.; Crosti, C.; Cugno, M. Cutaneous Manifestations in Patients with Inflammatory Bowel Diseases: Pathophysiology, Clinical Features, and Therapy. Inflamm Bowel Dis 2014, 20, 213–227. [Google Scholar] [CrossRef]
- Alinaghi, F.; Tekin, H.G.; Burisch, J.; Wu, J.J.; Thyssen, J.P.; Egeberg, A. Global Prevalence and Bidirectional Association Between Psoriasis and Inflammatory Bowel Disease—A Systematic Review and Meta-analysis. J Crohn’s Colitis 2020, 14, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Blegvad, C.; Egeberg, A.; Tind Nielsen, T.E.; Gislason, G.H.; Zachariae, C.; Nybo Andersen, A.M.; Skov, L. Autoimmune Disease in Children and Adolescents with Psoriasis: A Cross-sectional Study in Denmark. Acta Derm Venereol 2017, 97, 1225–1229. [Google Scholar] [CrossRef] [PubMed]
- Lolli, E.; Saraceno, R.; Calabrese, E.; Ascolani, M.; Scarozza, P.; Chiricozzi, A.; Onali, S.; Petruzziello, C.; Chimenti, S.; Pallone, F.; et al. Psoriasis Phenotype in Inflammatory Bowel Disease: A Case-Control Prospective Study. J Crohn’s Colitis 2015, 9, 699–707. [Google Scholar] [CrossRef]
- Garcovich, S.; De Simone, C.; Genovese, G.; Berti, E.; Cugno, M.; Marzano, A.V. Paradoxical skin reactions to biologics in patients with rheumatologic disorders. Front Pharmacol 2019, 10, 282. [Google Scholar] [CrossRef]
- Nigam, G.B.; Bhandare, A.P.; Antoniou, G.A.; Limdi, J.K. Systematic review and meta-analysis of dermatological reactions in patients with inflammatory bowel disease treated with anti-tumour necrosis factor therapy. Eur J Gastroenterol Hepatol 2021, 33, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Russell, M.D.; Stovin, C.; Alveyn, E.; Adeyemi, O.; Chan, C.K.D.; Patel, V.; Adas, M.A.; Atzeni, F.; Ng, K.K.H.; Rutherford, A.I.; et al. JAK inhibitors and the risk of malignancy: a meta-analysis across disease indications. Ann Rheum Dis 2023, 82, 1059–1067. [Google Scholar] [CrossRef] [PubMed]
- Curtis, J.R.; Yamaoka, K.; Chen, Y.H.; Bhatt, D.L.; Gunay, L.M.; Sugiyama, N.; Connell, C.A.; Wang, C.; Wu, J.; Menon, S.; et al. Malignancy risk with tofacitinib versus TNF inhibitors in rheumatoid arthritis: results from the open-label, randomised controlled ORAL Surveillance trial. Ann Rheum Dis 2023, 82, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Weizman, A.V.; Sharma, R.; Afzal, N.M.; Xu, W.; Walsh, S.; Stempak, J.M.; Nguyen, G.C.; Croitoru, K.; Steinhart, A.H.; Silverberg, M.S. Stricturing and Fistulizing Crohn's Disease Is Associated with Anti-tumor Necrosis Factor-Induced Psoriasis in Patients with Inflammatory Bowel Disease. Dig Dis Sci 2018, 63, 2430–2438. [Google Scholar] [CrossRef] [PubMed]
- Ampuero, J.; Rojas-Feria, M.; Castro-Fernández, M.; Cano, C.; Romero-Gómez, M. Predictive factors for erythema nodosum and pyoderma gangrenosum in inflammatory bowel disease. J Gastroenterol Hepatol 2014, 29, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Denadai, R.; Teixeira, F.V.; Steinwurz, F.; Romiti, R.; Saad-Hossne, R. Induction or exacerbation of psoriatic lesions during anti-TNF-α therapy for inflammatory bowel disease: a systematic literature review based on 222 cases. J Crohn‘s Colitis 2013, 7, 517–524. [Google Scholar] [CrossRef]
- Cleynen, I.; Van Moerkercke, W.; Billiet, T.; Vandecandelaere, P.; Vande Casteele, N.; Breynaert, C.; Ballet, V.; Ferrante, M.; Noman, M.; Assche, G.V.; et al. Characteristics of Skin Lesions Associated with Anti-Tumor Necrosis Factor Therapy in Patients with Inflammatory Bowel Disease: A Cohort Study. Ann Intern Med. 2016, 164, 10–22. [Google Scholar] [CrossRef]
- Au, M.; Heddle, G.; Young, E.; Ryan, E.; Graf, S.; Tee, D.; Philpott, H. Anti-tumour necrosis factor-induced skin rashes in inflammatory bowel disease: a systematic review and evidence-based management algorithm. Intern Med J 2023, 53, 1854–1865. [Google Scholar] [CrossRef]
| Total | UC | CD | |
|---|---|---|---|
| Patient count | 162 (100 %) | 117 (72.2%) | 45 (27.8%) |
| Male | 93 (57.4%) | 67 (57.3%) | 26 (57.8%) |
| Female | 69 (42.6%) | 50 (42.7%) | 19 (42.2%) |
| Mean age (SD) | 42.5 (14.2) | 43.1 (14) | 40.9 (14.9) |
| Illness duration in years (SD) | 9 (6.8) | 9.5 (7.0) | 7.7 (6.1) |
| <5 years | 46 (28.4%) | 31 (26.5%) | 15 (33.3%) |
| 5-10 years | 53 (32.7%) | 35 (29.9%) | 18 (40%) |
| >10 years | 63 (38.9%) | 51 (43.6%) | 12 (26.7%) |
| Used systemic treatment | 155 (95.7%) | 112 (95.7%) | 43 (95.6%) |
| Biological therapy | 82 (50,6%) | 50 (42.7%) | 32 (71.1%) |
| Infliximab/adalimumab | 46 (56.1%) | 25 (50.0%) | 21 (65.6%) |
| Vedolizumab | 13 (15.9%) | 11 (22.0%) | 2 (6.3%) |
| Ustekinumab | 9 (11.0%) | 3 (6.0%) | 6 (18.8%) |
| Mirikizumab (clinical trial) | 11 (13.4%) | 8 (16.0%) | 3 (9.4%) |
| Risankizumab (clinical trial) | 3 (3.7%) | 3 (6.0%) | 0 (0.0%) |
| JAK inhibitors | 6 (3.7%) | 5 (4.27%) | 1 (2.22%) |
| Tofacitinib | 4 (66.7%) | 4 (80.0%) | 0 (0%) |
| Upadacitinib (clinical trial) | 2 (33.3%) | 1 (20.0%) | 1 (100%) |
| Skin lesions | Total n = 162 |
Ulcerative colitis n = 117 |
Crohn’s disease n = 45 |
|
|---|---|---|---|---|
| Psoriasis | 13 (8,0%) | 11 (9,4%) | 2 (4,4%) | |
| Erythema nodosum | 9 (5,6%) | 4 (3,4%) | 5 (11,1%) | |
| Pyoderma gangrenosum | 6 (3,7%) | 5 (4,3%) | 1 (2,2%) | |
| Acne | 6 (3,7%) | 5 (4,3%) | 1 (2,2%) | |
| Vitiligo | 3 (1,9%) | 3 (2,6%) | 0 (0%) | |
| Epidermolysis bullosa acquisita | 1 (0,6%) | 1 (0,9%) | 0 (0%) | |
| Vasculitis | 1 (0,6%) | 1 (0,9%) | 0 (0%) | |
| Sweet syndrome | 0 (0%) | 0 (0%) | 0 (0%) | |
| Hidradenitis suppurativa | 0 (0%) | 0 (0%) | 0 (0%) | |
| Others: | ||||
| Seborrheic dermatitis | 3 (1,9%) | 3 (2,6 %) | 0 (0%) | |
| Atopic dermatitis | 4 (2,5%) | 4 (3,4%) | 0 (0%) | |
| Unspecified dermatitis | 11 (6,8%) | 8 (6,8%) | 3 (6,7%) | |
| Others skin lesions | 17 (10,5%) | 13 (11,1%) | 4 (8,9%) |
| Skin lesions | Ileitis | Colitis | Ileocolitis |
|---|---|---|---|
| Erythema nodosum | 1 (5,9%) | 3 (27,3%) | 1 (5,9%) |
| Pyoderma gangrenosum | 0 (0%) | 1 (9,1%) | 0 (0%) |
| Acne | 0 (0%) | 0 (0%) | 1 (5,9%) |
| Psoriasis | 0 (0%) | 0 (0%) | 2 (11,8%) |
| Vitiligo | 0 (0%) | 0 (0%) | 0 (0%) |
| Epidermolysis bullosa acquisita | 0 (0%) | 0 (0%) | 0 (0%) |
| Vasculitis | 0 (0%) | 0 (0%) | 0 (0%) |
| Others: | |||
| Atopic dermatitis | 0 (0%) | 0 (0%) | 0 (0%) |
| Seborrheic dermatitis | 0 (0%) | 0 (0%) | 0 (0%) |
| Unspecified dermatitis | 1 (5,9%) | 1 (9,1%) | 1 (5,9%) |
| Others skin lesions | 2 (11,8%) | 0 (0%) | 2 (11,8%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




