Submitted:
26 January 2024
Posted:
29 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental study
- methanol, ethanol, n-propanol and iso-propanol at ϕ = 0.3 – 0.7;
- mixtures of alcohols and water, such that the volumetric content of the latter ranged from 10 and 60 vol%.
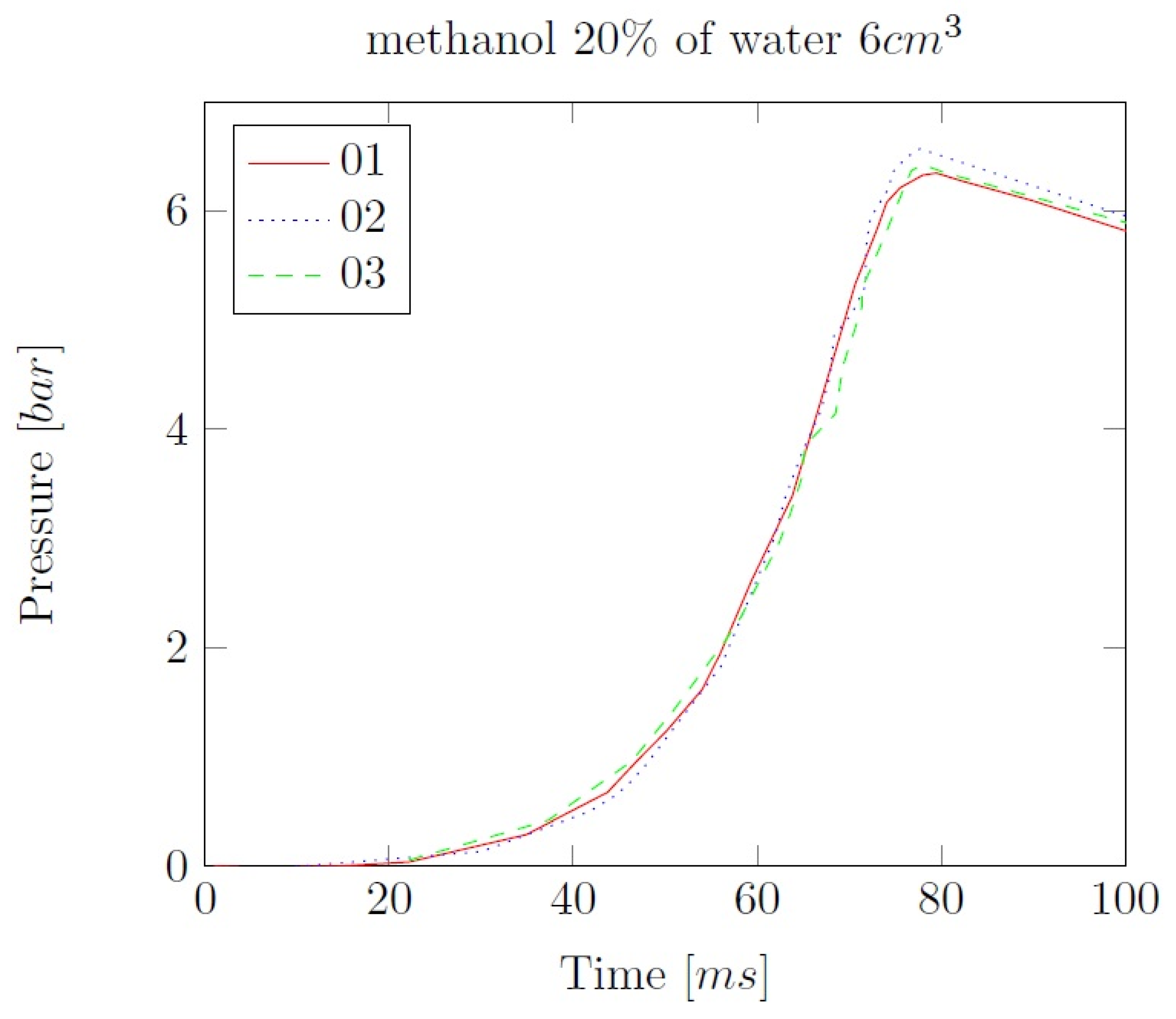
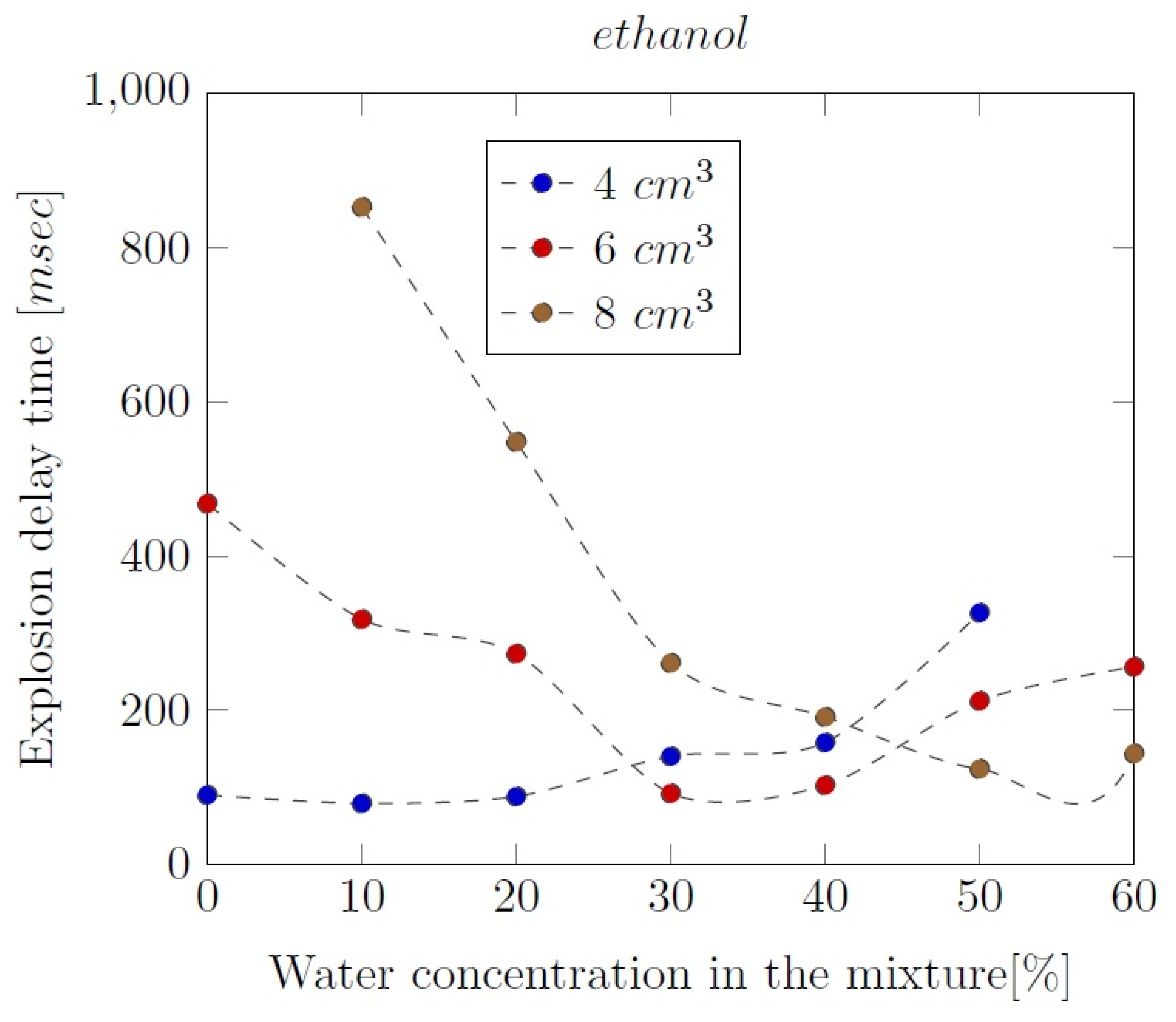
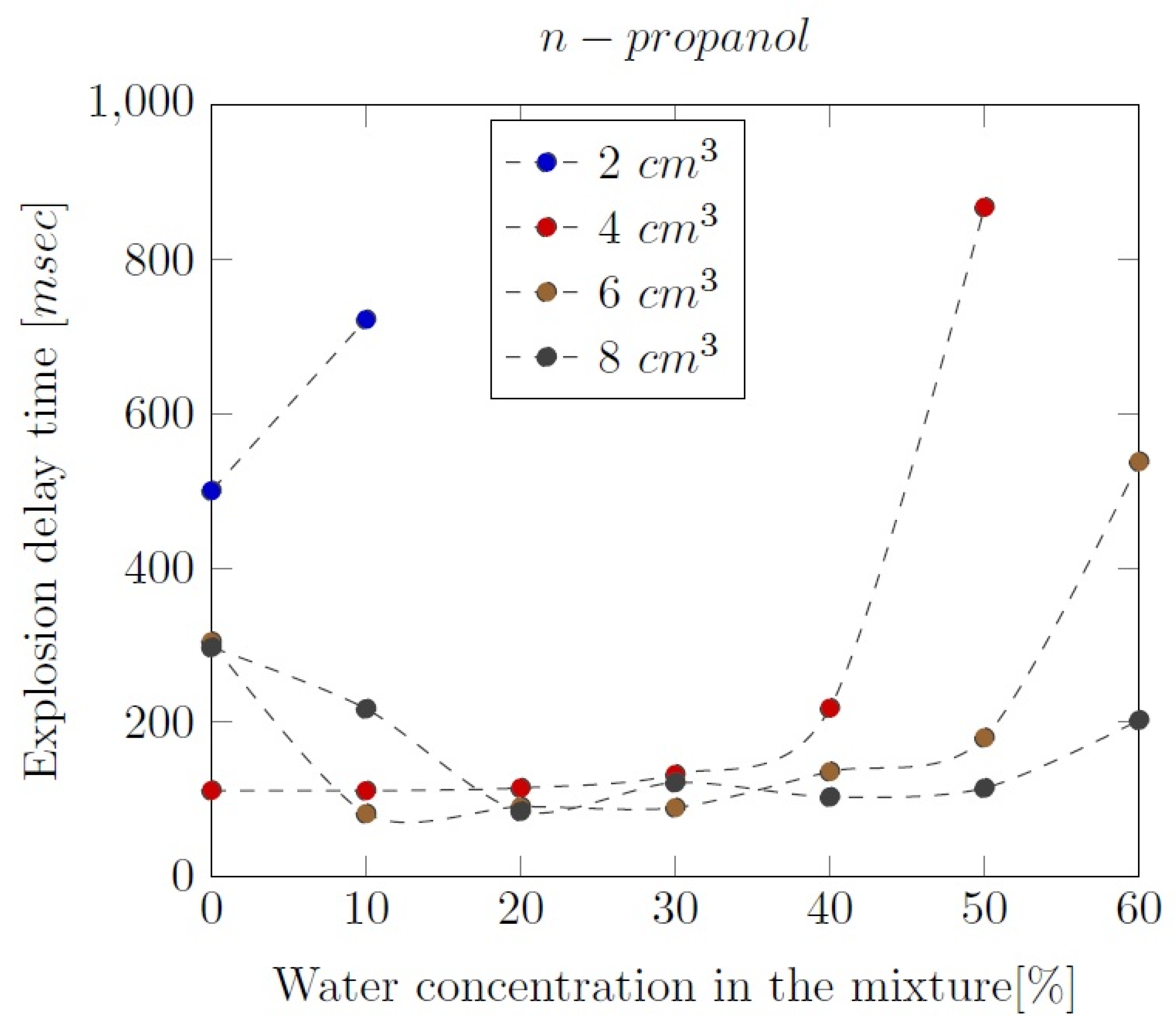
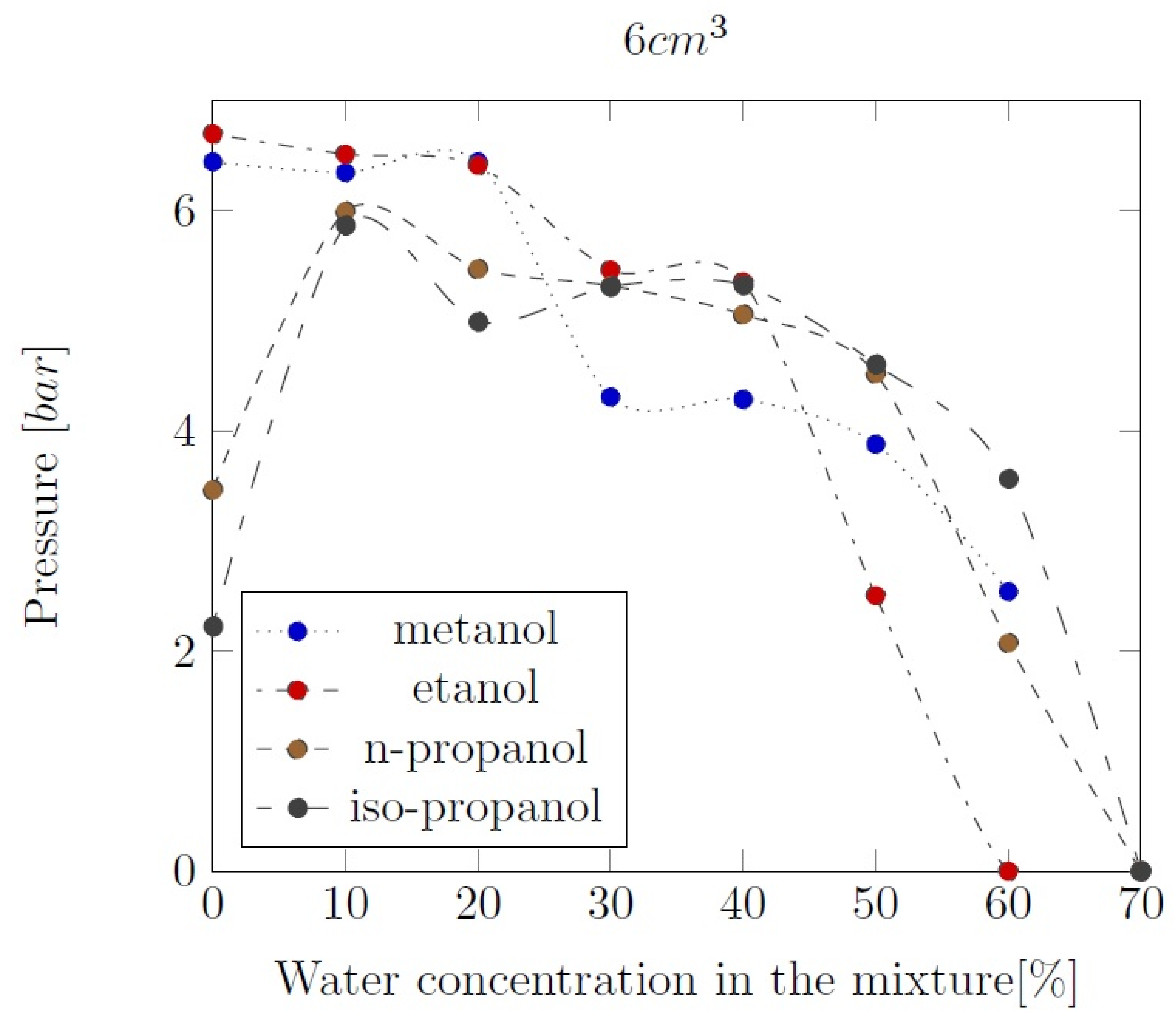
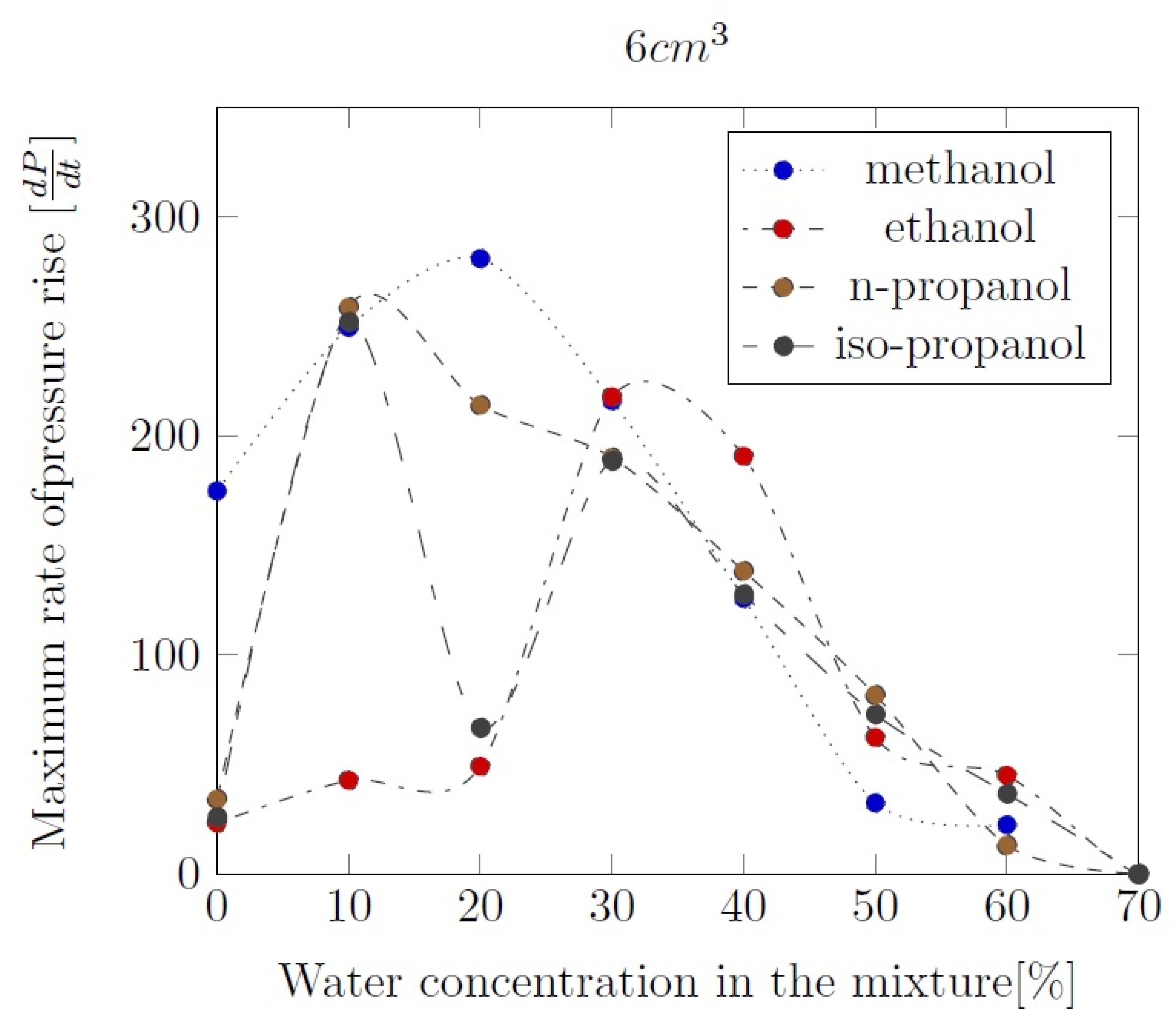
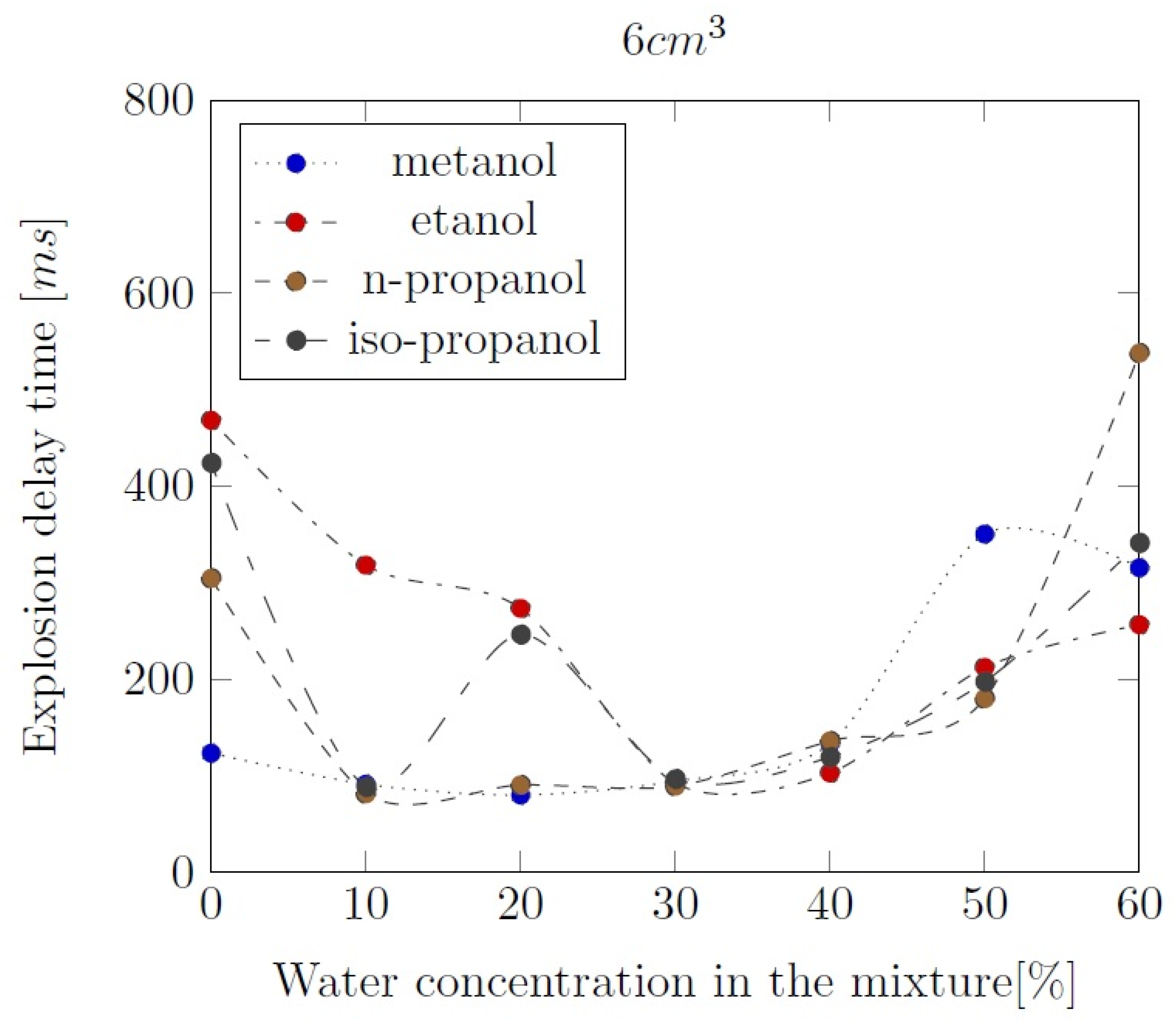
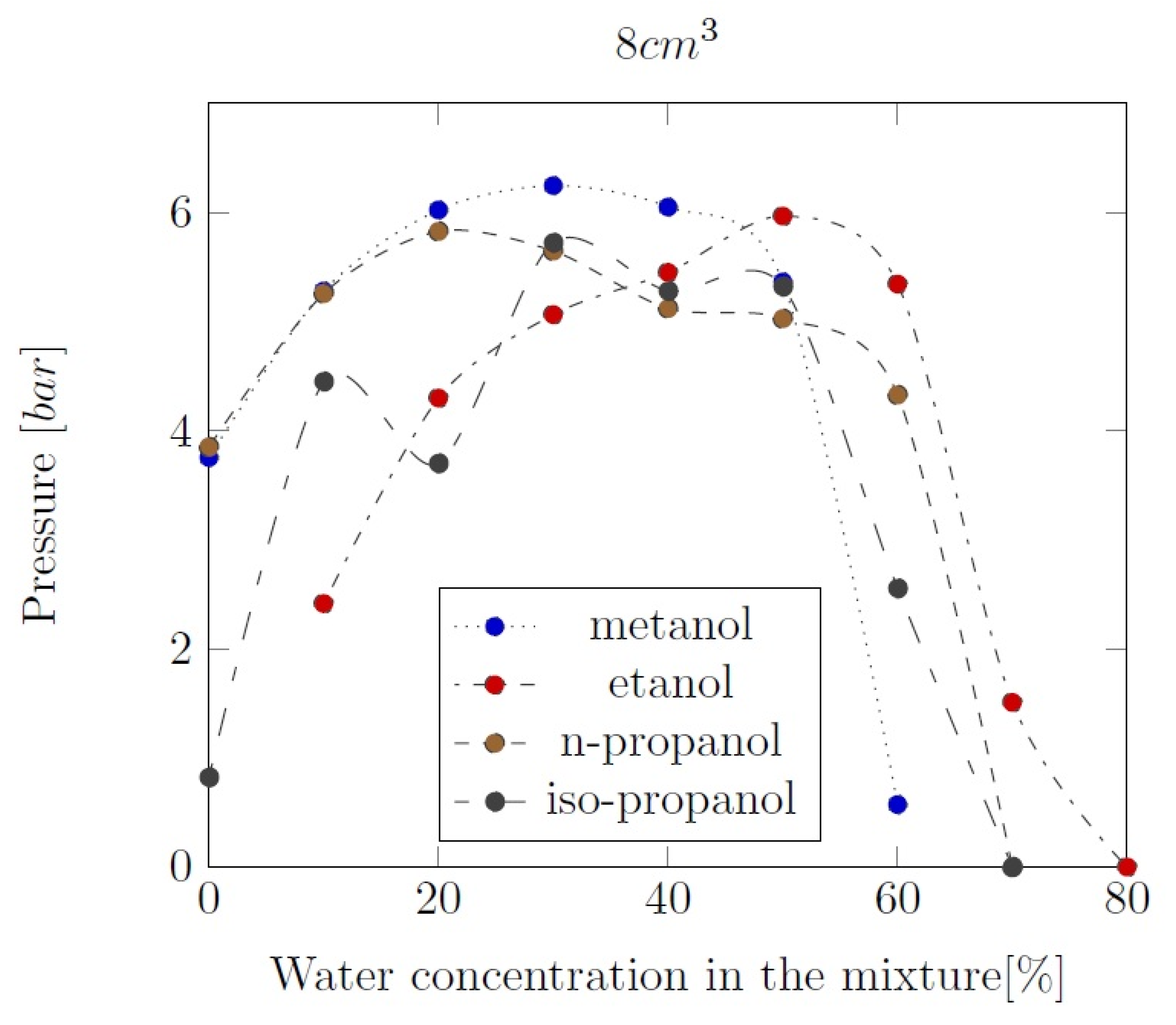
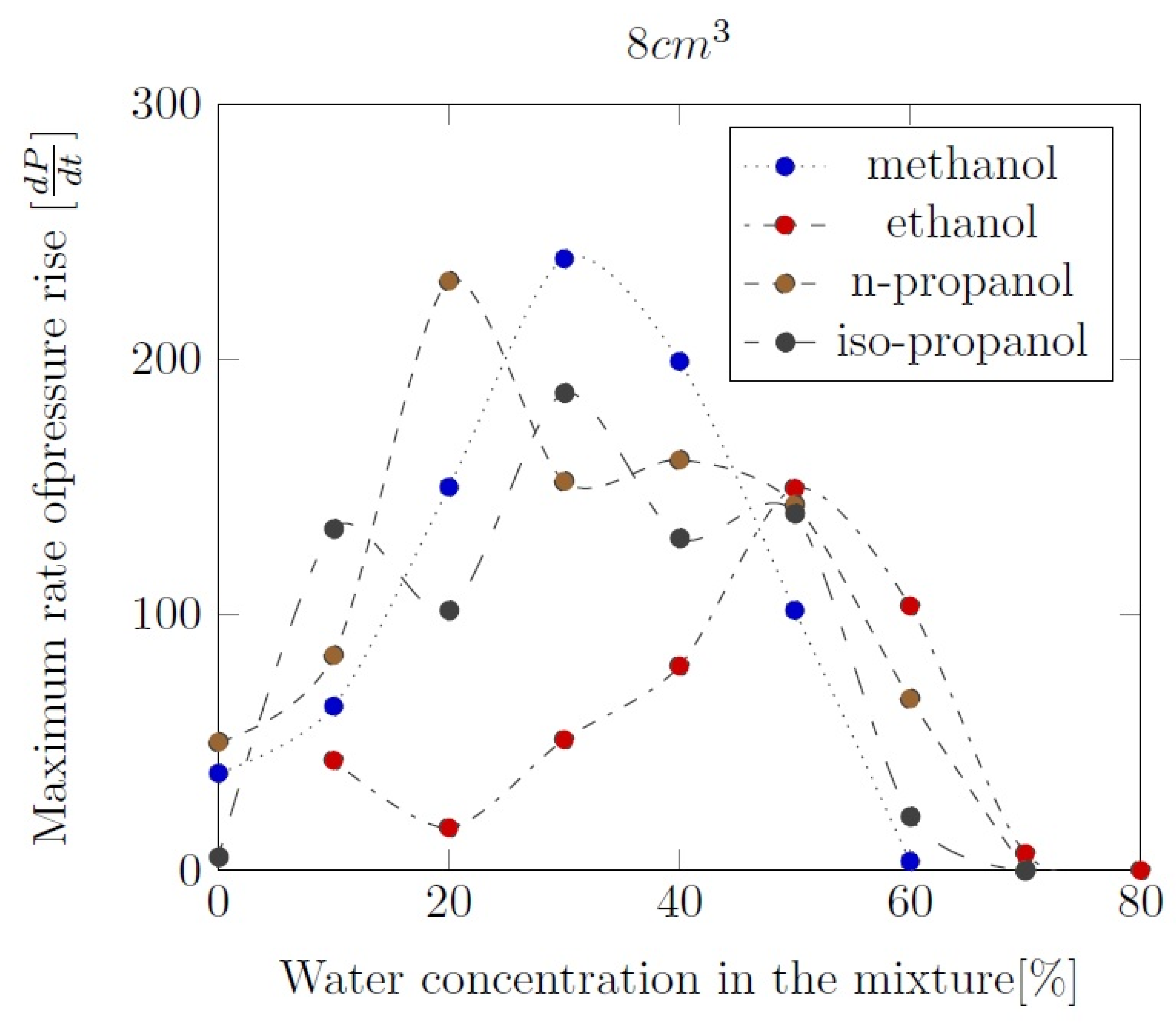
3. Liquid phase composition of the alcohol-water mixtures
| Substance | M (kg mol−1) | ρ(†) (kg m−3) |
Tc(⊕) (K) |
Pc(⊕) (bar) |
Vc(⊕) (cm3 mol−1) |
ω (-) |
Tb(‡) (K) |
PSat (∗) (bar) |
Fl. lim.(y) (vol%) |
|---|---|---|---|---|---|---|---|---|---|
| CH3OH | 32.04·10−3 | 787.2 | 512.16 | 80.92 | 117.88 | 0.565 | 338.15 | 0.17 | 6 – 37 |
| C2H5OH | 46.07·10−3 | 787.3 | 513.9 | 61.37 | 167.10 | 0.649 | 351.15 | 0.079 | 3 – 19 |
| n-C3H7OH | 60.10·10−3 | 802.0 | 536.8 | 51.70 | 218.41 | 0.629 | 370.15 | 0.028 | 2 – 14 |
| i-C3H7OH | 60.10·10−3 | 782.7 | 508.3 | 47.62 | 220.10 | 0.665 | 355.15 | 0.061 | 2 – 12 |
| H2O | 18.015·10−3 | (a)997.05 | 647.10 | 220.64 | 56.02 | 0.344 | (a)373.13 | (a)0.0317 | |
| Air | (b)28.964·10−3 | 1.161 | (c)132.63 | (c)37.858 | 92.35 |
- ➢
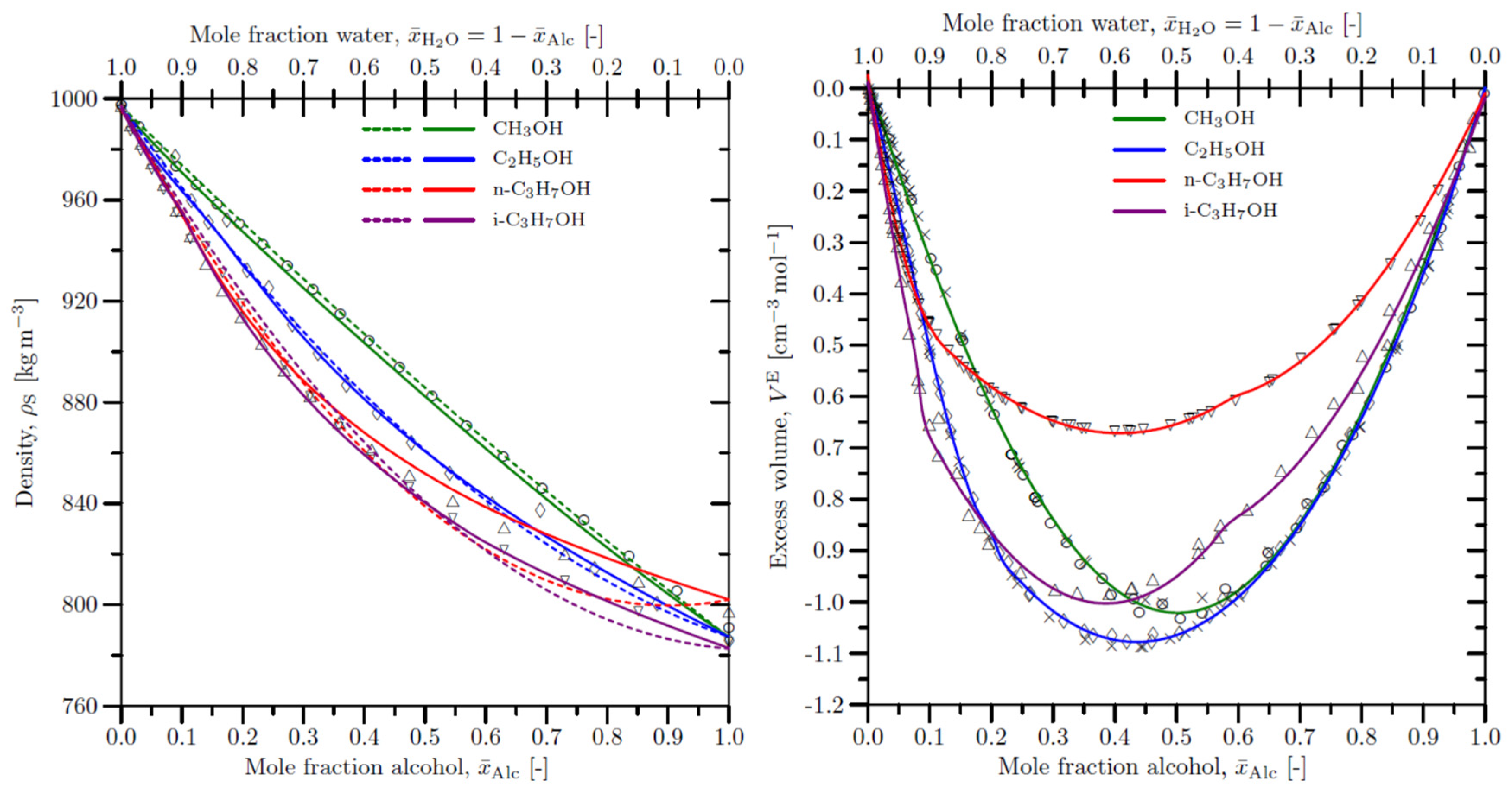
- Equations (1) to (3) become invalid for calculating mixture properties such as the liquid sample density and specific volume. Instead, equation (5) should be applied, provided that VE is known on an a priori basis from experiments or theoretical predictions.
- ➢
- The partial molar volumes of the species in the mixture are no longer equal to the molar volumes of the pure species: ≠ VAlc and ≠ . Furthermore, for real mixtures and become nonlinear functions of and . Equation (12) further on, establishes a thermodynamic relationship between the partial molar volume of a species, , and the total volume V of a mixture as a function of varying composition. Hence, although equation (4) remains valid for both ideal and real mixtures, its application to the latter requires precise a priori knowledge of the nonlinear dependence of ), i.e. ), in equation (12), on the mixture composition.
- ➢
- The application of models that predict ρS directly.
- ➢
- The deployment of equation (5) in conjunction with models that predict VE.
4. Density of the liquid samples and the excess volume of liquid mixtures
| Polynomial: VE = ρAlc Alc + ρH2O(1 − Alc) + a0 Alc(1 − Alc) (13) | |||||
| Mixture | Range | a0 | |||
| CH3OH – H2O | 0.0 ≤ Alc ≤ 1.0 | -25.2·100 | |||
| C2H5OH – H2O | 0.0 ≤ Alc ≤ 1.0 | -124.7·100 | |||
| n-C3H7OH – H2O | 0.0 ≤ Alc ≤ 1.0 | -242.1·100 | |||
| i-C3H7OH – H2O | 0.0 ≤ Alc ≤ 1.0 | -19.6·101 | |||
| Polynomial: VE = a0 Alc + a1(1 − Alc) + a2 Alc(1 − Alc) + a3 2 (1 − Alc)2 (14)Alc | |||||
| Mixture | Range | a0 | a1 | a2 | a3 |
| CH3OH – H2O | 0.0 ≤ Alc ≤ 1.0 | 0.3·10−3 | 17.0·10−3 | -370.1·10−2 | -16.7·10−1 |
| C2H5OH – H2O | 0.0 ≤ Alc < 0.2 0.2 ≤ Alc ≤ 1.0 |
25.2·100 1.3·10−3 |
8.0·10−3 -41.6·10−2 |
-29.5·100 -36.3·10−1 |
-46.6·100 8.1·10−1 |
| n-C3H7OH – H2O | 0.0 ≤ Alc < 0.1 0.1 ≤ Alc < 0.6 0.6 ≤ Alc ≤ 1.0 |
20.7·100 5.9·10−2 -10.1·10−3 |
25.6·10−3 -324.8·10−3 16.0·10−2 |
-28.4·100 -208.3·10−2 -27.3·10−1 |
0.0 0.0 0.0 |
| i-C3H7OH – H2O | 0.0 ≤ Alc < 0.1 0.1 ≤ Alc < 0.6 0.6 ≤ Alc ≤ 1.0 |
2.4·100 5.0·10−1 -1.9·10−2 |
12.3·10−3 -41.1·10−2 -1.4·10−1 |
-10.2·100 -39.8·10−1 -31.9·10−1 |
0.0 0.0 0.0 |
5. Vapor pressures of the pure liquids
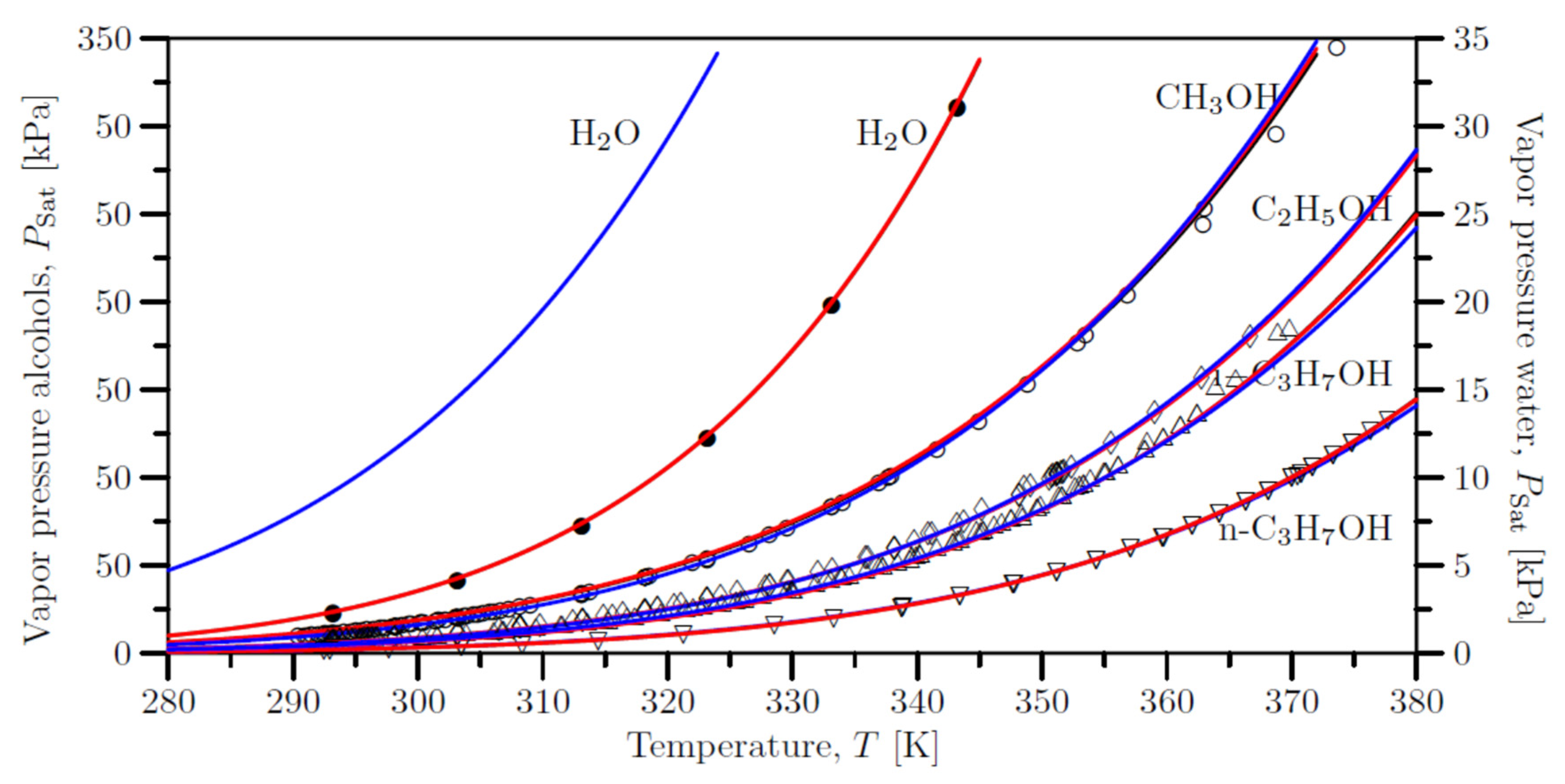
| Species | Extended Antoine equation | Wagner equation | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | E | F | t0 | n | p | q | a | b | c | d | |
| CH3OH | 5.20277 | 1580.080 | 239.500 | – | – | – | – | 8 | 12 | -8.63571 | 1.17982 | -2.4790 | -1.0240 |
| C2H5OH | 5.33675 | 1648.220 | 230.918 | – | – | – | – | 8 | 12 | -8.68587 | 1.17831 | -4.8762 | 1.5880 |
| n-C3H7OH | 4.99991 | 512.940 | 205.807 | – | – | – | – | 8 | 12 | -8.53706 | 1.96214 | -7.6918 | 2.9450 |
| i-C3H7OH | 5.24268 | 1580.920 | 219.610 | – | – | – | – | 8 | 12 | -8.73656 | 2.16240 | -8.70785 | 4.77927 |
| H2O | 5.11564 | 1687.537 | 230.17 | – | – | – | – | 3 | 6 | -7.77224 | 1.45684 | -2.71942 | -1.41336 |
6. Conclusions
Acknowledgments
Conflicts of Interest
Appendix A. Thermodynamic quantities and properties
| Methanol – water | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (cm3) | q (vol%) |
(-) |
(-) |
(-) | (-) |
(-) |
(-) |
(-) |
(-) |
(cm3 mol-1) | ρs (kg m-3) | Ignition | ||||||||||
| 4.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 10 | 0.888 | 0.112 | - | - | 0.817 | 0.183 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 20 | 0.798 | 0.202 | - | - | 0.691 | 0.309 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 30 | 0.725 | 0.275 | - | - | 0.599 | 0.401 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 40 | 0.664 | 0.336 | - | - | 0.528 | 0.472 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 50 | 0.612 | 0.388 | - | - | 0.472 | 0.528 | - | - | - | - | 2×n | ||||||||||
| 4.0 | 60 | 0.568 | 0.432 | - | - | 0.427 | 0.573 | - | - | - | - | 3×n | ||||||||||
| 6.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 10 | 0.888 | 0.112 | - | - | 0.817 | 0.183 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 20 | 0.798 | 0.202 | - | - | 0.691 | 0.309 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 30 | 0.725 | 0.275 | - | - | 0.599 | 0.401 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 40 | 0.664 | 0.336 | - | - | 0.528 | 0.472 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 50 | 0.612 | 0.388 | - | - | 0.472 | 0.528 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 60 | 0.568 | 0.432 | - | - | 0.427 | 0.573 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 10 | 0.888 | 0.112 | - | - | 0.817 | 0.183 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 20 | 0.798 | 0.202 | - | - | 0.691 | 0.309 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 30 | 0.725 | 0.275 | - | - | 0.599 | 0.401 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 40 | 0.664 | 0.336 | - | - | 0.528 | 0.472 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 50 | 0.612 | 0.388 | - | - | 0.472 | 0.528 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 60 | 0.568 | 0.432 | - | - | 0.427 | 0.573 | - | - | - | - | 3×y | ||||||||||
| Ethanol – water | ||||||||||||||||||||||
| (cm3) | q (vol%) |
(-) |
(-) |
(-) |
(-) |
Alc(-) | (-) |
(-) |
(-) | (cm3 mol-1) | ρs (kg m-3) | Ignition | ||||||||||
| 4.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 10 | 0.888 | 0.112 | - | - | 0.757 | 0.243 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 20 | 0.798 | 0.202 | - | - | 0.609 | 0.391 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 30 | 0.725 | 0.275 | - | - | 0.509 | 0.491 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 40 | 0.664 | 0.336 | - | - | 0.438 | 0.562 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 50 | 0.612 | 0.388 | - | - | 0.384 | 0.616 | - | - | - | - | 2×y | ||||||||||
| 4.0 | 60 | 0.568 | 0.432 | - | - | 0.341 | 0.659 | - | - | - | - | 3×n | ||||||||||
| 6.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 10 | 0.888 | 0.112 | - | - | 0.757 | 0.243 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 20 | 0.798 | 0.202 | - | - | 0.609 | 0.391 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 30 | 0.725 | 0.275 | - | - | 0.509 | 0.491 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 40 | 0.664 | 0.336 | - | - | 0.438 | 0.562 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 50 | 0.612 | 0.388 | - | - | 0.384 | 0.616 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 60 | 0.568 | 0.432 | - | - | 0.341 | 0.659 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 10 | 0.888 | 0.112 | - | - | 0.757 | 0.243 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 20 | 0.798 | 0.202 | - | - | 0.609 | 0.391 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 30 | 0.725 | 0.275 | - | - | 0.509 | 0.491 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 40 | 0.664 | 0.336 | - | - | 0.438 | 0.562 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 50 | 0.612 | 0.388 | - | - | 0.384 | 0.616 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 60 | 0.568 | 0.432 | - | - | 0.341 | 0.659 | - | - | - | - | 3×y | ||||||||||
| n-Propanol – water | ||||||||||||||||||||||
| (cm3) | q (vol%) |
(-) |
(-) |
(-) | (-) |
Alc (-) |
(-) |
(-) | (-) | (cm3 mol-1) | ρs (kg m-3) | Ignition | ||||||||||
| 4.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 10 | 0.888 | 0.112 | - | - | 0.708 | 0.292 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 20 | 0.798 | 0.202 | - | - | 0.548 | 0.452 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 30 | 0.725 | 0.275 | - | - | 0.447 | 0.553 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 40 | 0.664 | 0.336 | - | - | 0.378 | 0.622 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 50 | 0.612 | 0.388 | - | - | 0.327 | 0.673 | - | - | - | - | 2×n | ||||||||||
| 4.0 | 60 | 0.568 | 0.432 | - | - | 0.288 | 0.712 | - | - | - | - | 3×n | ||||||||||
| 6.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 10 | 0.888 | 0.112 | - | - | 0.708 | 0.292 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 20 | 0.798 | 0.202 | - | - | 0.548 | 0.452 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 30 | 0.725 | 0.275 | - | - | 0.447 | 0.553 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 40 | 0.664 | 0.336 | - | - | 0.378 | 0.622 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 50 | 0.612 | 0.388 | - | - | 0.327 | 0.673 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 60 | 0.568 | 0.432 | - | - | 0.288 | 0.712 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 10 | 0.888 | 0.112 | - | - | 0.708 | 0.292 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 20 | 0.798 | 0.202 | - | - | 0.548 | 0.452 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 30 | 0.725 | 0.275 | - | - | 0.447 | 0.553 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 40 | 0.664 | 0.336 | - | - | 0.378 | 0.622 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 50 | 0.612 | 0.388 | - | - | 0.327 | 0.673 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 60 | 0.568 | 0.432 | - | - | 0.288 | 0.712 | - | - | - | - | 3×y | ||||||||||
| i-Propanol – water | ||||||||||||||||||||||
| (cm3) | q (vol%) |
(-) |
(-) |
(-) | (-) |
Alc (-) |
(-) |
(-) |
(-) | (cm3 mol-1) | ρs (kg m-3) | Ignition | ||||||||||
| 4.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 10 | 0.888 | 0.112 | - | - | 0.703 | 0.297 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 20 | 0.798 | 0.202 | - | - | 0.542 | 0.458 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 30 | 0.725 | 0.275 | - | - | 0.441 | 0.559 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 40 | 0.664 | 0.336 | - | - | 0.372 | 0.628 | - | - | - | - | 3×y | ||||||||||
| 4.0 | 50 | 0.612 | 0.388 | - | - | 0.322 | 0.678 | - | - | - | - | 2×n | ||||||||||
| 4.0 | 60 | 0.568 | 0.432 | - | - | 0.283 | 0.717 | - | - | - | - | 3×n | ||||||||||
| 6.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 10 | 0.888 | 0.112 | - | - | 0.703 | 0.297 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 20 | 0.798 | 0.202 | - | - | 0.542 | 0.458 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 30 | 0.725 | 0.275 | - | - | 0.441 | 0.559 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 40 | 0.664 | 0.336 | - | - | 0.372 | 0.628 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 50 | 0.612 | 0.388 | - | - | 0.322 | 0.678 | - | - | - | - | 3×y | ||||||||||
| 6.0 | 60 | 0.568 | 0.432 | - | - | 0.283 | 0.717 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 0 | 1.000 | 0.000 | - | - | 1.000 | 0.000 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 10 | 0.888 | 0.112 | - | - | 0.703 | 0.297 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 20 | 0.798 | 0.202 | - | - | 0.542 | 0.458 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 30 | 0.725 | 0.275 | - | - | 0.441 | 0.559 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 40 | 0.664 | 0.336 | - | - | 0.372 | 0.628 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 50 | 0.612 | 0.388 | - | - | 0.322 | 0.678 | - | - | - | - | 3×y | ||||||||||
| 8.0 | 60 | 0.568 | 0.432 | - | - | 0.283 | 0.717 | - | - | - | - | 3×y | ||||||||||
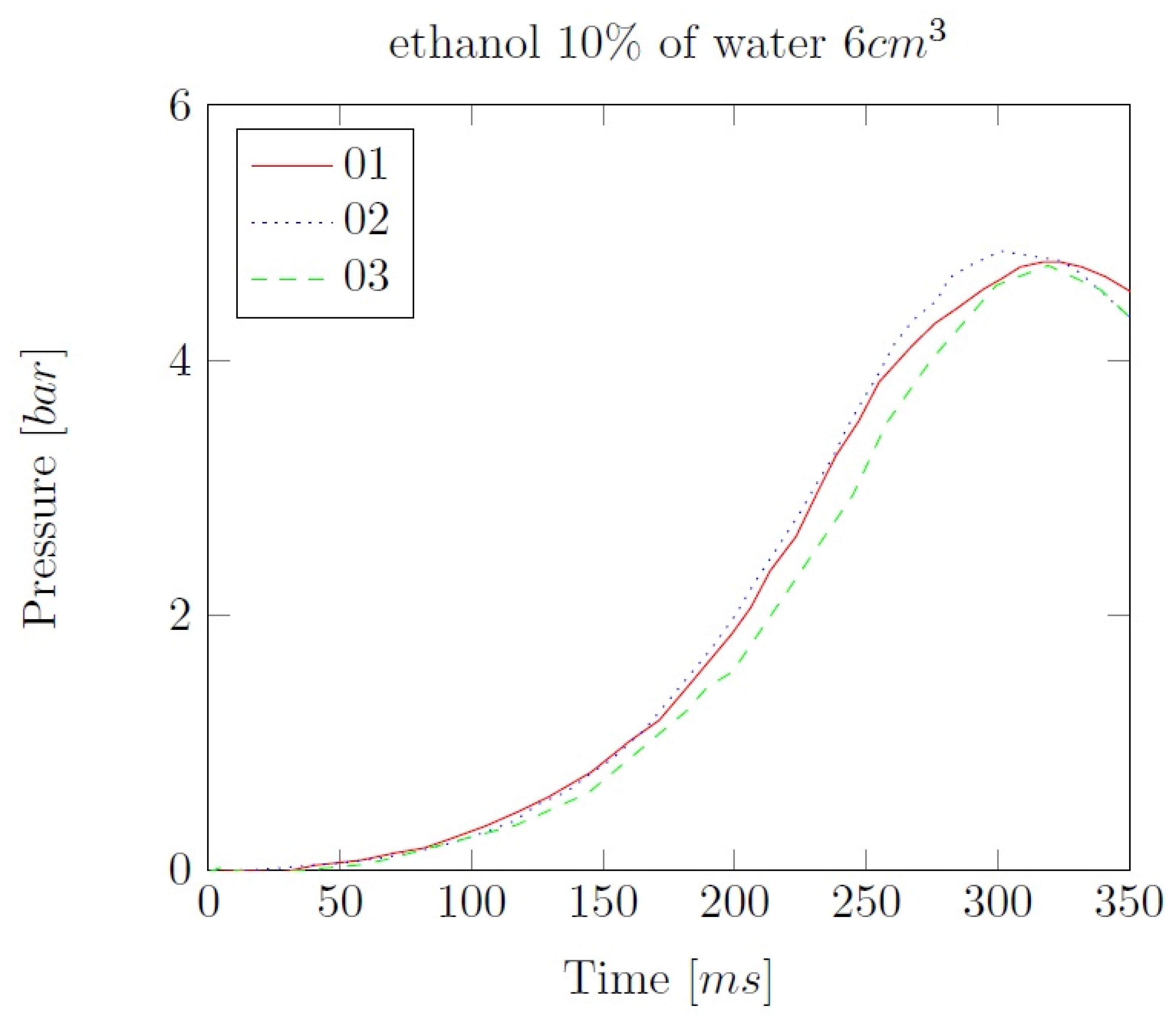
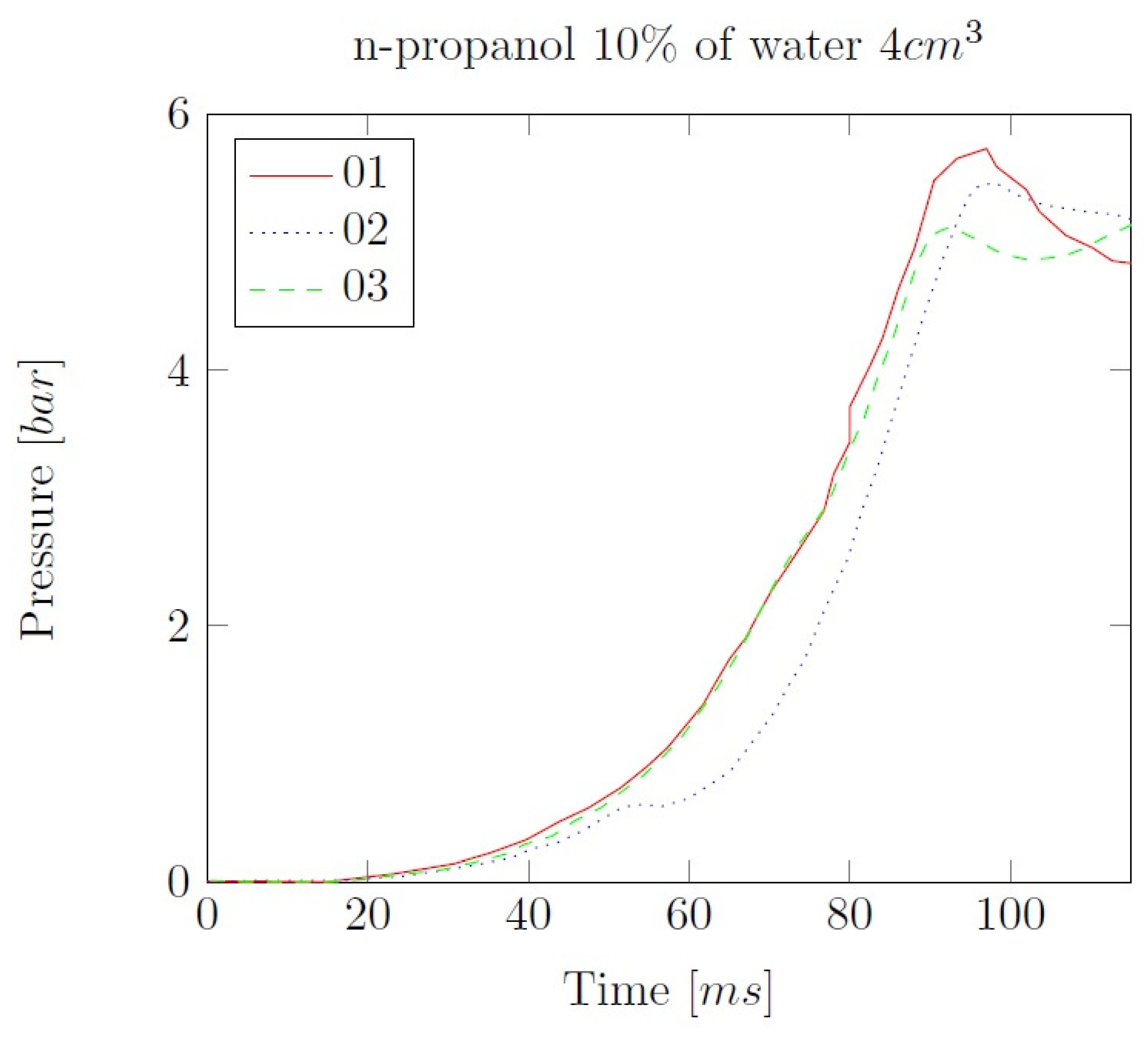
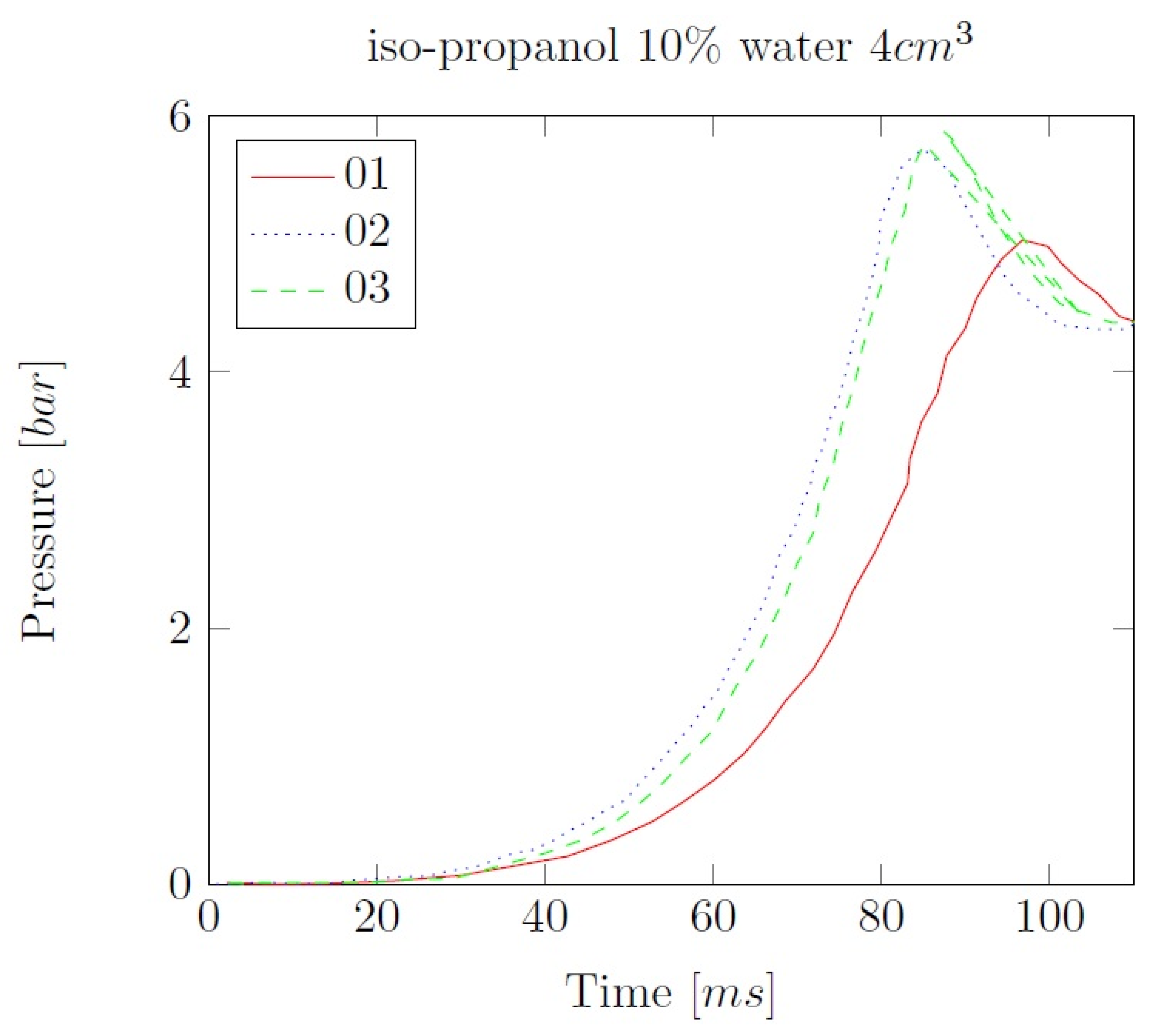
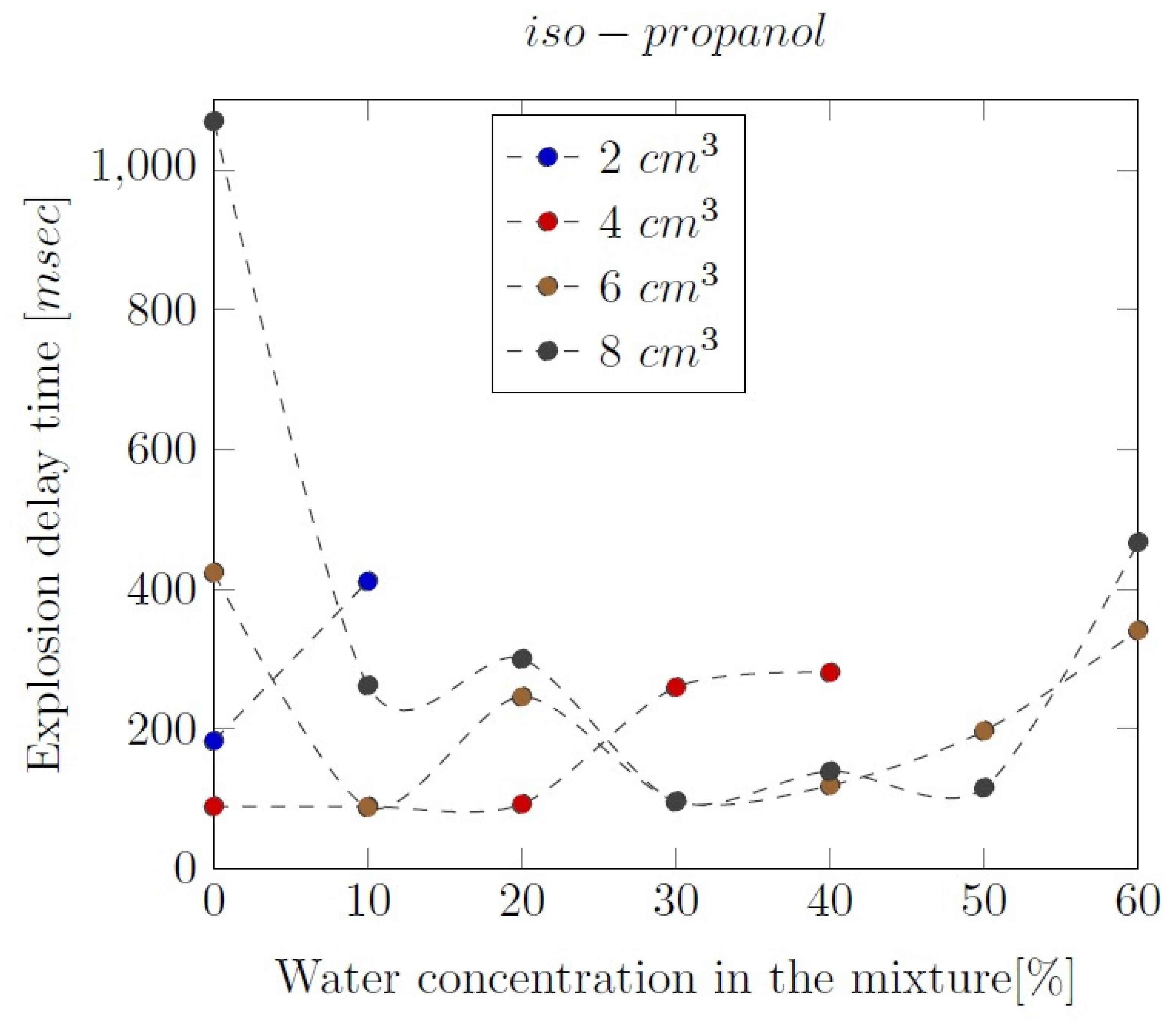
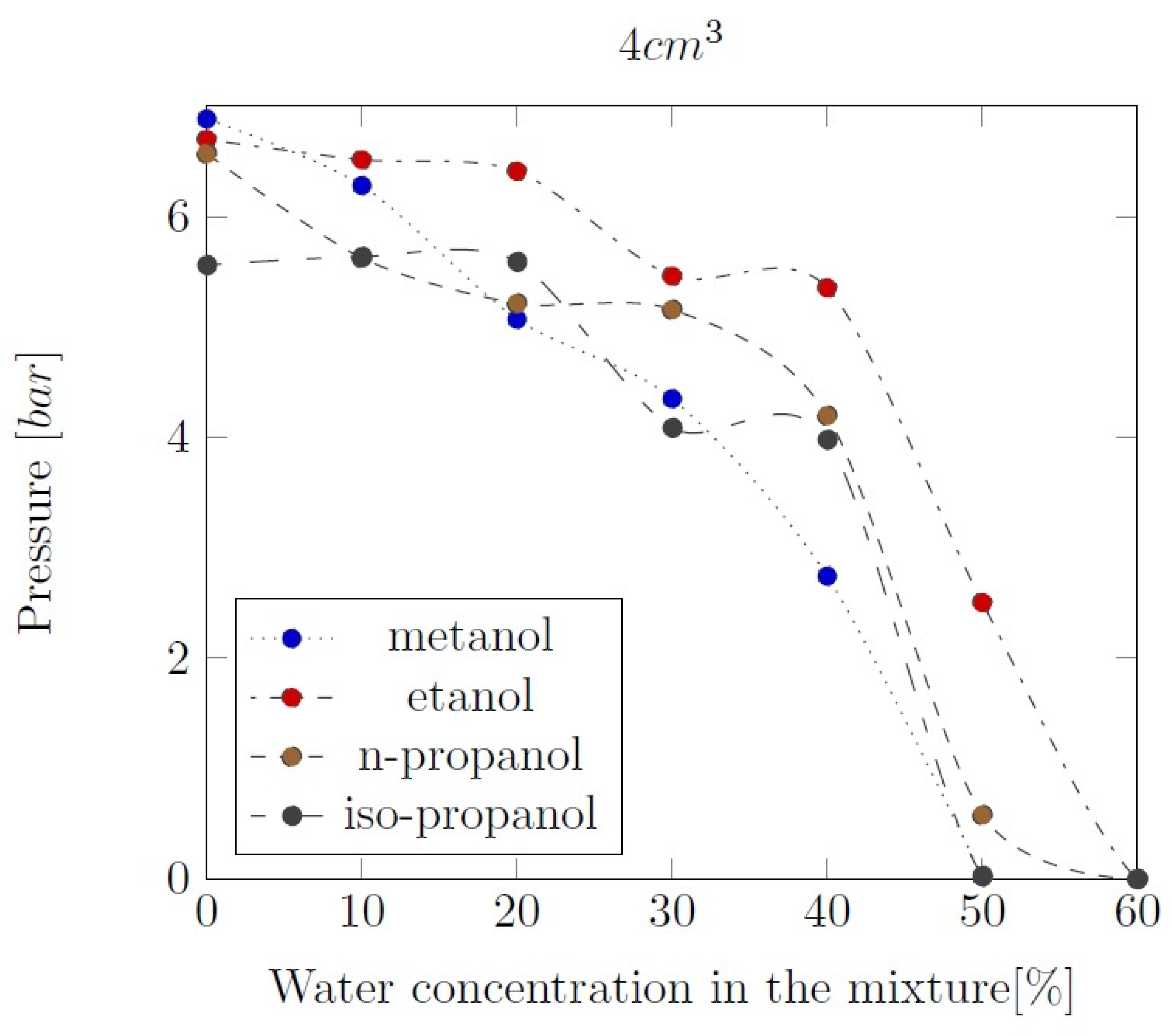
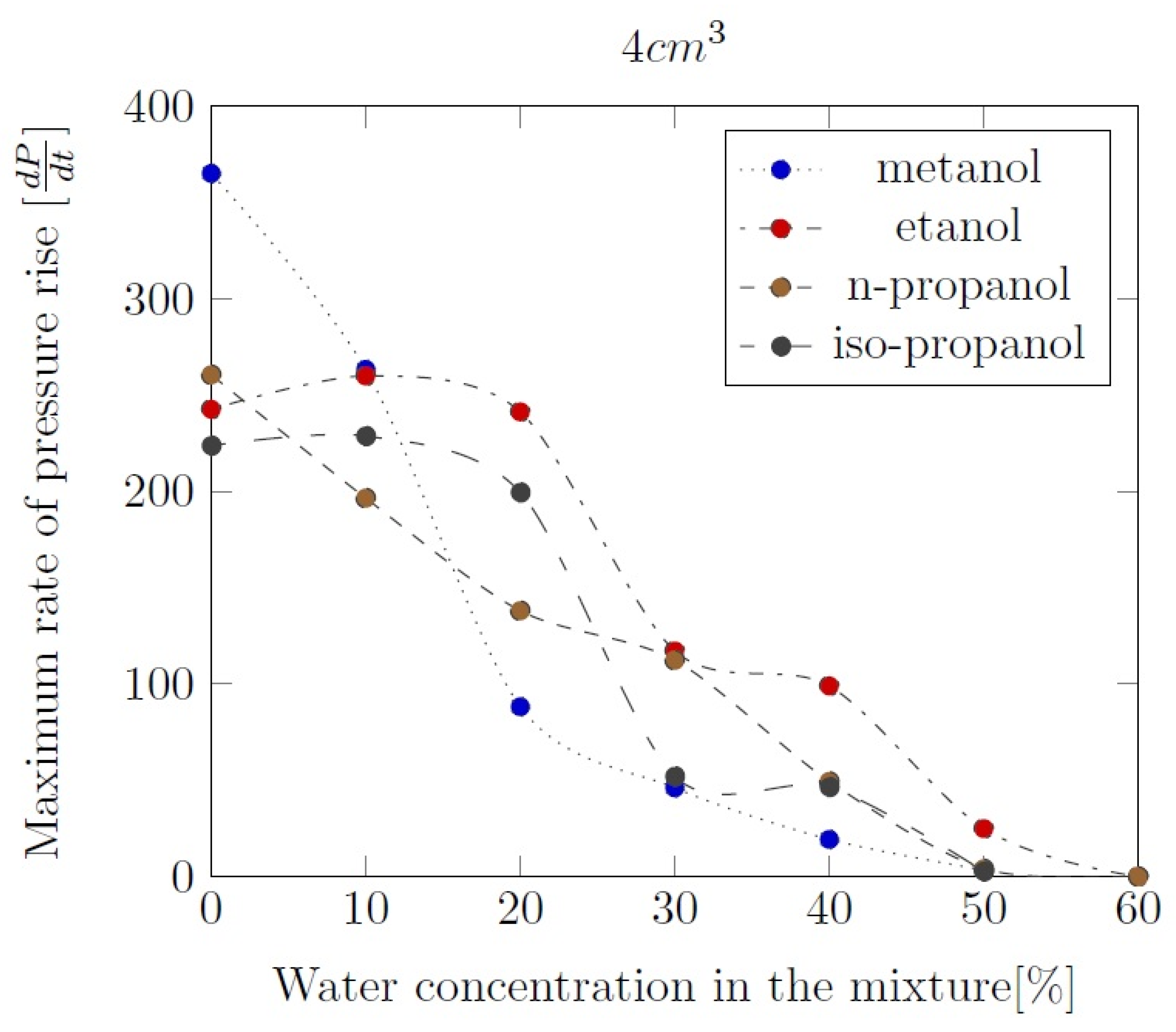
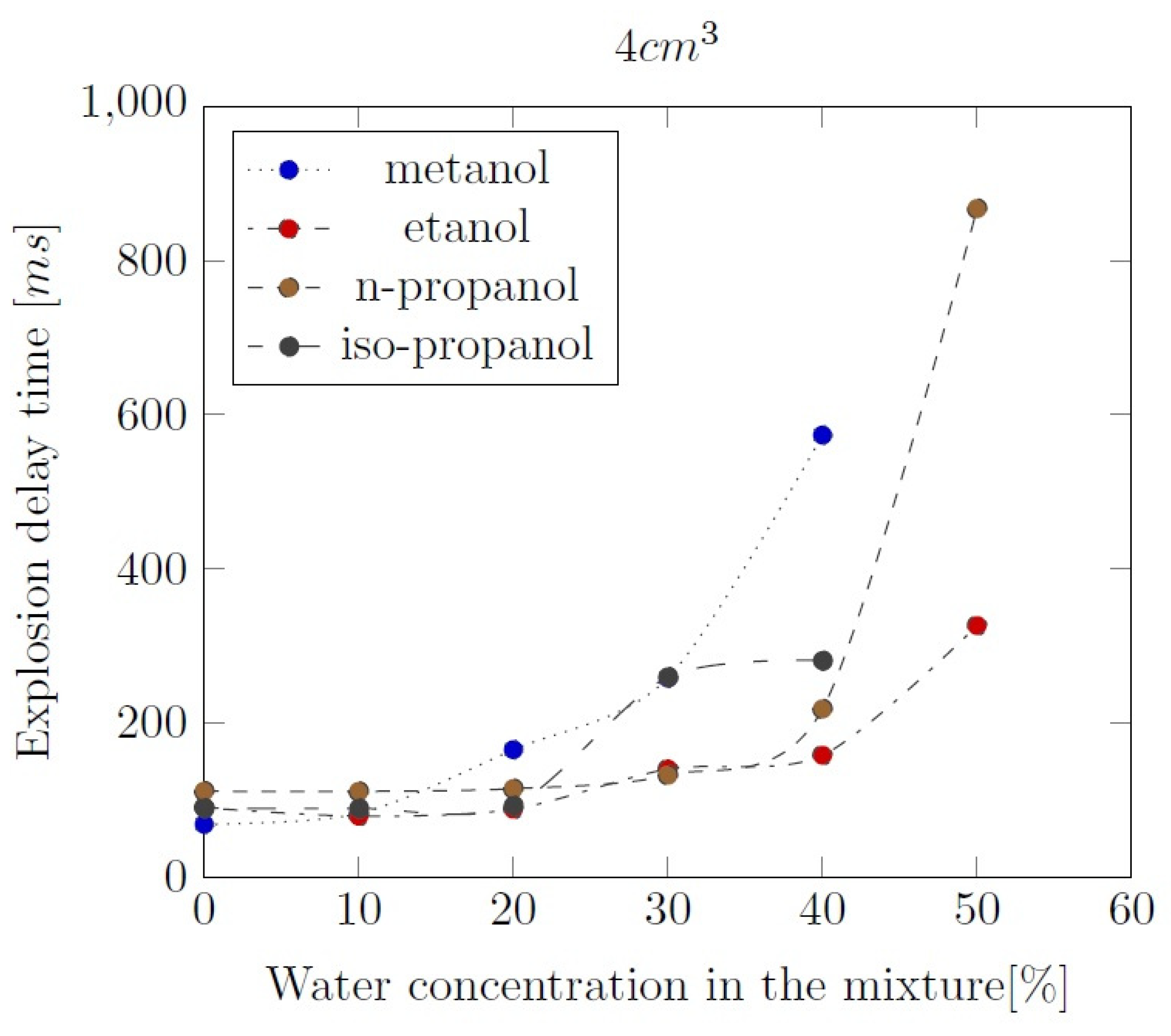
Appendix B. Experimental explosion pressure curves of alcohol-air and alcohol-water-air mixtures.
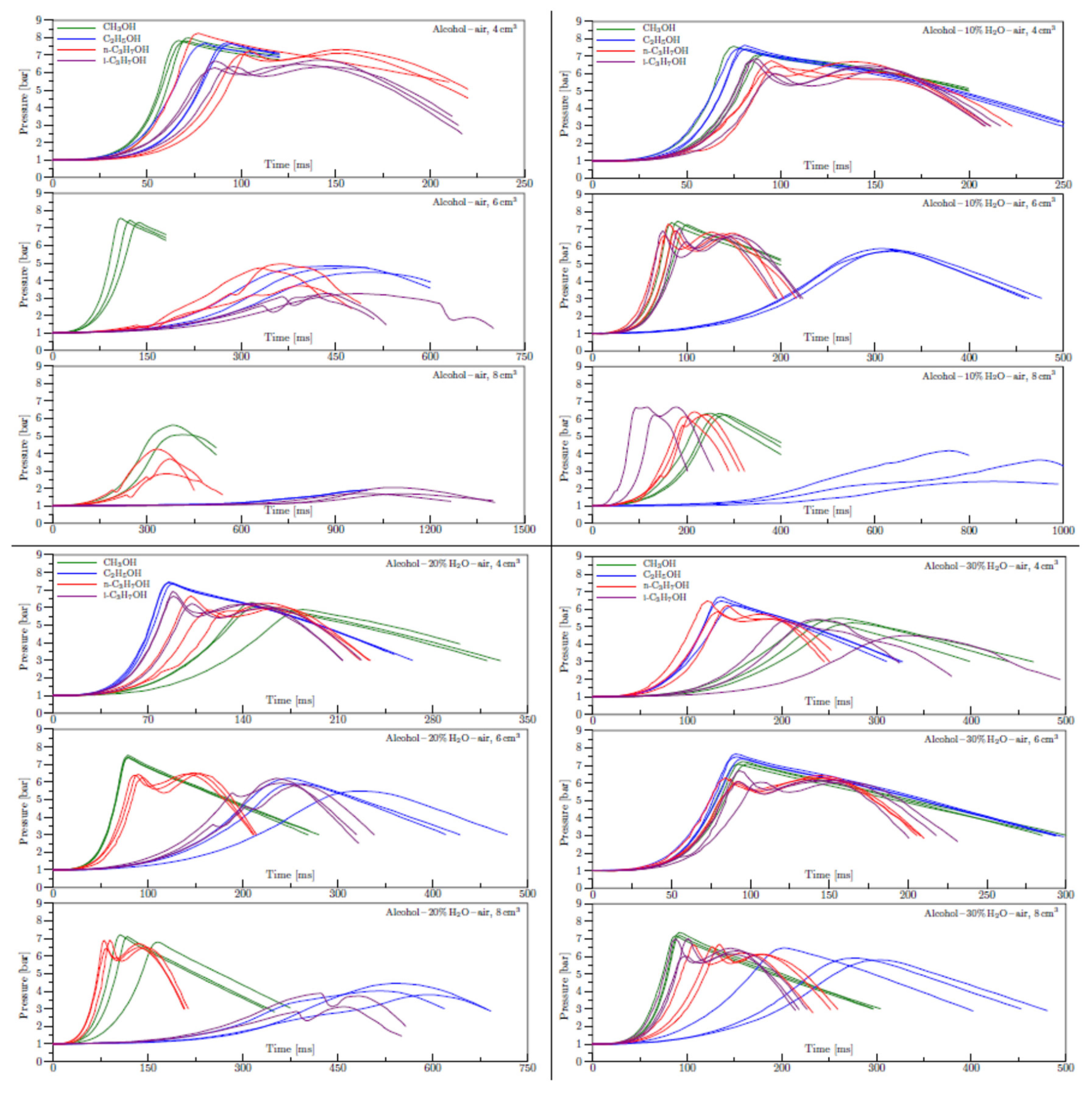
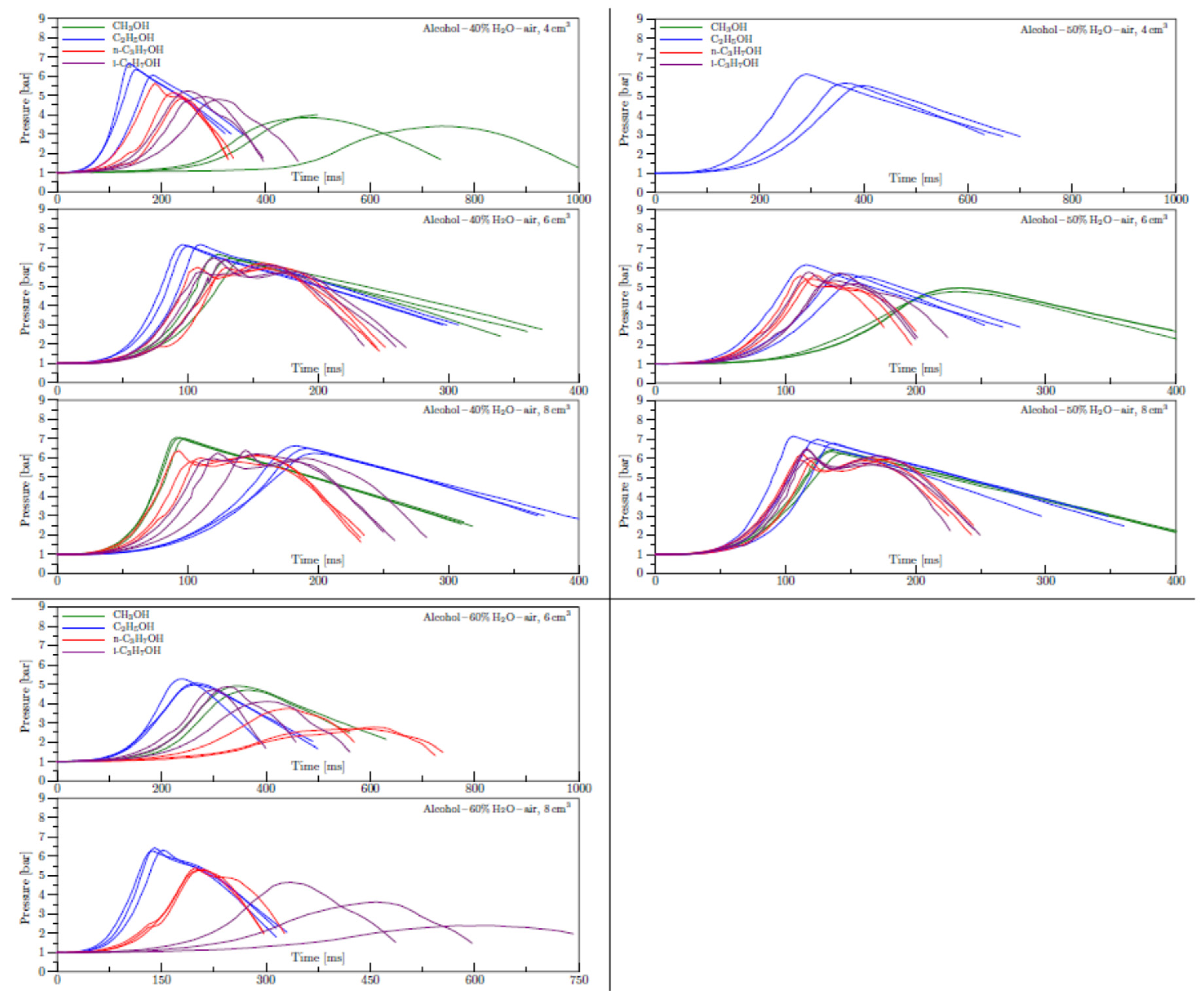
References
- Lide D.R., editor. CRC Handbook of Chemistry and Physics. CRC Press, 84th edition, 2004.
- Poling, B.E., Prausnitz J.M., and O’Connell J.P. The Properties of Gases and Liquids. McGraw-Hill, fifth edition, 2001.
- Perry, R.H., Green D.W., and Maloney J.O. Perry’s Chemical Engineers’ Handbook. McGraw-Hill, New York, seventh edition, 1999.
- The Dortmund Data Bank. Thermophysical properties of pure components and their mixtures. http://www.ddbst.com/, 2018. Publicly available online resource.
- Arce, A., Blanco A., Soto A., and Vidal I. Densities, refractive indices, and excess molar volumes of the ternary systems water + methanol + 1-octanol and water + ethanol + 1-octanol and their binary mixtures at 298.15 K. Journal of Chemical Engineering Data 1993, 38, 336–340. [CrossRef]
- Vilcu, R. and Simion A. Grandeurs d’exces par des Mesures Acoustiques, dans les Systemes Binaires de Liquides. Revue Roumaine de Chimie 1976, 21, 177–186.
- S. Mani Sarathy, Patrick Oßwald, Nils Hansen, and Katharina Kohse-Hoinghaus. Alcohol combustion chemistry. Progress in Energy and Combustion Science, 2014.
- Qianqian Li, Yu Cheng, and Zuohua Huang. Comparative assessment of the explosion characteristics of alcohol-air mixtures. Journal of Loss Prevention in the Process Industries, 2015.
- Bryan, W. Weber, Kamal Kumar, Yu Zhang, and Chih-Jen Sung. Autoignition of n-butanol at elevated pressure and low to intermediate temperature. Combustion and Flame, 2011.
- Zhu Yangye, David Frank Davidson, and Ronald K. Hanson. 1-butanol ignition delay times at low temperatures: An application of the constrained-reaction-volume strategy. Combustion and Flames, 2013.
- Douheret, G., Khadir A, and Pal A. Thermodynamic characterization of the water + methanol system, at 298.15 K. Thermochimica Acta 1989, 142, 219–243. [CrossRef]
- Grolier, J.-P.E. and Wilhelm E. Excess volumes and excess heat capacities of water + ethanol at 298.15 K. Fluid Phase Equilibria 1981, 6, 283–287. [CrossRef]
- Ott, J.B., Sipowska J.T., Gruszkiewicz M.S., and Woolley A.T. Excess volumes for (ethanol + water) at the temperatures (298.15 and 348.15) K and pressures (0.4, 5, and 15) MPa and at the temperature 323.15 K and pressures (5 and 15) MPa. The Journal of Chemical Thermodynamics 1993, 25, 307–318. [CrossRef]
- Davis, M.I. and Ham E.S. Part 2. Comparisons of the propanol isomers in their aqueous mixtures. Thermochimica Acta 1991, 190, 251–258.
- Langdon, W.M. and Keyes D.B. Isopropyl alcohol-water system. Industrial & Engineering Chemistry 1943, 35, 459–464.
- Yamamoto, H., Ichikawa K., and Tokunaga J. Solubility of helium in methanol + water, ethanol + water, 1-propanol + water, and 2-propanol + water solutions at 25°C. Journal of Chemical Engineering Data 1994, 39, 155–157. [CrossRef]
- Benson, G.C. and Kiyohara O. Thermodynamics of aqueous mixtures of nonelectrolytes. I. Excess volumes of water – n-alcohol mixtures at several temperatures. Journal of Solution Chemistry 1980, 9, 791–804. [CrossRef]
- Smith, J.M., Van Ness H.C., and Abbott M.M. Introduction to Chemical Engineering Thermodynamics. McGraw-Hill, New York, seventh edition, 2007.
- Battino, R. Volume changes on mixing for binary mixtures of liquids. Chemical Reviews 1971, 71, 5–45. [CrossRef]
- Abbott, M.M. and Van Ness H.C. Theory and problems of thermodynamics. Schaum’s outline series. McGraw-Hill, New York, 1972.
- Press, W.H., Teukolsky S.A., Vetterling W.T., and Flannery B.P. Numerical Recipes, The Art of Scientific Computing. Cambridge University Press, Cambridge, third edition, 2007.
- Marquardt, D.W. An algorithm for least-squares estimation of nonlinear parameters. SIAM Journal on Applied Mathematics 1963, 11, 431–441. [Google Scholar] [CrossRef]
- Dahoe, A.E., Skjold T, Roekaerts D.J.E.M., Pasman H.J., Eckho R.K., Hanjalic K., and Donze M. On the application of the Levenberg-Marquardt method in conjunction with an explicit Runge-Kutta and an implicit Rosenbrock method to assess burning velocities from confined deflagrations. Flow, Turbulence and Combustion 2013, 91, 281–317.
- Rackett, H.G. Equation of state for saturated liquids. Journal of Chemical & Engineering Data 1970, 15, 514–517. [Google Scholar]
- Lydersen, A.L., Greenkorn R.A., and Hougen O.A. Generalized thermodynamic properties of pure fluids. Engineering Experiment Station Report 4, 1955. College of Engineering, University of Wisconsin, Madison, Wisconsin.
- Gunn, R.D. and Yamada T. A corresponding states correlation of saturated liquid volumes. AIChE Journal 1971, 17, 1341–1345. [CrossRef]
- Yen, L.C. and Woods S.S. A generalized equation for computer calculation of liquid densities. AIChE Journal 1966, 12, 95–99. [CrossRef]
- Yamada T. and Gunn R. Saturated liquid molar volumes. The Rackett equation. Journal of Chemical & Engineering Data 1973, 18, 234–236.
- The NIST ChemistryWebBook. Data compiled under the Standard Reference Data Program. https://webbook.nist.gov/, 2018. The National Institute of Standards and Technology (NIST). Publicly available online resource.
- Kotsarenko, A.A. and Yarym-Agaev N.L. Sättigungsdampfdruck von Methanol im Temperaturbereich von 251.9 bis 298.15 k. Izvestiya Vysshikh Uchebnykh Zavedenii: Neft i gaz, pages 59–61, 1990.
- Gibbard H.F. and Creek J.L. Vapor pressure of methanol from 288.15 to 337.65 k. Journal of Chemical & Engineering Data 1974, 19, 308–310.
- Dever, D.F. Dever D.F., Finch A., and Grunwald E. Vapor pressure of methanol from 288.15 to 337.65 k. The Journal of Physical Chemistry 1974, 59, 668–669. [Google Scholar] [CrossRef]
- Chun K.W. and Davison R.R. Thermodynamic properties of binary mixtures of triethylamine with methyl and ethyl alcohol. Journal of Chemical & Engineering Data 1972, 17, 307–310.
- Ambrose, D. and Sprake C.H.S. Thermodynamic properties of organic oxygen compounds. XXV. Vapour pressures and normal boiling temperatures of aliphatic alcohols. The Journal of Chemical Thermodynamics 1970, 2, 631–645. [CrossRef]
- Ambrose, D., Sprake C.H.S., and Townsend R. Thermodynamic properties of organic oxygen compounds. XXXVII. Vapour pressures of methanol, ethanol, pentan-1-ol, and octan-1-ol from the normal boiling temperature to the critical temperature. The Journal of Chemical Thermodynamics 1975, 7, 185–190. [CrossRef]
- Hirata M. and Suda S. Vapor pressure on methanol in high pressure regions. The Journal of Chemical Thermodynamics 1967, 31, 339–342.
- Mishchenko K.P. and Subbotina V.V. Dampfdruck von Ethanol bei Temperaturen von 4 bis 46°c. Zhurnal Pikladnoi Khimii 1967, 40, 1156–1159.
- Kahlbaum, G.W.A. and von Wirkner C.G. Studien uber Dampfspannkraftsmessungen. Monograph, 1897. Reprint in 2012 under ISBN-number 9785883729552.
- Scatchard G. and Raymond C.L. II. Chloroform-ethanol mixtures at 35, 45 and 55°C. Journal of the American Chemical Society 1938, 60, 1278–1287. [CrossRef]
- Scatchard G. and Satkiewicz F.G. XII. The system ethanol-cyclohexane from 5 to 65°C. Journal of the American Chemical Society 1964, 86, 130–133. [CrossRef]
- Kretschmer C.B. and Wiebe R. Liquid-vapor equilibrium of ethanol-toluene solutions. Journal of the American Chemical Society 1949, 71, 1793–1797. [CrossRef]
- Dejoz A., Gonzalez-Alfaro V., Miguel P.J., and Vazquez M.I. Isobaric vapor-liquid equilibria of tetrachloroethylene + 1-propanol and + 2-propanol at 20 and 100 kpa. Journal of Chemical & Engineering Data 1996, 41, 1361–1365.
- Shulgin I.L., Belousov V.P., and Baglai A.K. Eine tensimetrische Untersuchungsmethode des Dampf-flussig-Gleichgewichtes in binaren Systemen. Termodinamika Organicheskoi Soedinenii 1989, 32, 32–35.
- Barr-David F. and Dodge B.F. The systems ethanol–water and 2-propanol–water. Journal of Chemical & Engineering Data 1959, 4, 107–121.
- Daubert T.E., Jalowka J.W., and Goren V. Vapor pressure of 22 pure industrial chemicals. AIChE Symposium Series 1987, 32, 128–156.
- Ambrose D. and Townsend R. Thermodynamic properties of organic oxygen compounds. the critical properties and vapour pressures, above five atmospheres, of six aliphatic alcohols. Part IX. Journal of the Chemical Society 1963, 54, 3614–3625.
- Biddiscombe D.P., Collerson R.R., Handley R., Herington E.F.G., Martin J.F., and Sprake C.H.S. Thermodynamic properties of organic oxygen compounds. Part XIII. Journal of the Chemical Society 1963, 54, 1954–1957.
- Kemme H.R. and Kreps S.I. Vapor pressure of primary n-alkyl chlorides and alcohols. Journal of Chemical & Engineering Data 1969, 14, 98–102.
- Gudkov A.N., Fermor N.A., and Smirnov N.I. Communication I. Zurnal Prikladnoj Chimii 1964, 37, 2204–2210.
- Bridgeman O.C. and Aldrich E.W. Vapor pressure tables for water. Journal of Heat Transfer 1964, 86, 279–286. [CrossRef]
- Stull, D.R. Vapor pressure of pure substances. Organic and inorganic compounds. Industrial & Engineering Chemistry 1947, 39, 517–540. [Google Scholar]
- Clapeyron, E. M´emoire sur la Puissance Motrice de la Chaleur. Journal de l’Ecole Royale Polytechnique 1834, 14, 153–191. [Google Scholar]
- Wisniak, J. Historical development of the vapor pressure equation from Dalton to Antoine. Journal of Phase Equilibria 2001, 22, 622–630. [Google Scholar] [CrossRef]
- Wagner, W. New vapour pressure measurements for argon and nitrogen and a new method for establishing rational vapour pressure equations. Cyrogenics 1973, 13, 470–482. [Google Scholar] [CrossRef]
- Wagner, W. A new correlation method for thermodynamic data applied to the vapor-pressure curve of argon, nitrogen, and water. In: Watson J.T.R. (editor). IUPACThermodynamic Tables Project Centre, London, 1977.
- Ambrose, D. The corelation and estimation of vapour pressures IV. Observations on Wagner’s method of fitting equations to vapour pressures. The Journal of Chemical Thermodynamics 1986, 18, 45–51. [Google Scholar] [CrossRef]
- Ambrose, D. Vapour pressures and critical temperatures and critical pressures of some alkanoic acids: C1 to C10. The Journal of Chemical Thermodynamics 1987, 19, 505–519. [Google Scholar] [CrossRef]
- Lee, B.I. Lee B.I. and Kesler M.G. A generalized thermodynamic correlation based on three-parameter corresponding states. AIChE Journal 1975, 21, 510–527. [Google Scholar] [CrossRef]
- Alduchov O.A. and Eskridge R.E. Improved Magnus form approximation of saturation vapor pressure. Journal of Applied Meteorology 1996, 35, 601–619. [CrossRef]
- Alduchov O.A. and Eskridge R.E. U¨ ber einige meteorologische Begriffe. Zeitschrift fur Geophysik 1930, 6, 207–309.
- Buck, A.L. New equations for computing vapor pressure and enhancement factor. Journal of Applied Meteorology 1981, 20, 1527–1532. [Google Scholar] [CrossRef]
- Murphy D.M. and Koop T. Review of the vapour pressures of ice and supercooled water for atmospheric applications. Quarterly Journal of the Royal Meteorological Society 2005, 131, 1539–1565. [CrossRef]
- Dethlefsen C., Sørensen P.G., and Hvidt A. Excess volumes of propanol-water mixtures at 5, 15, and 25°C. Journal of Solution Chemistry 1984, 13, 191–202. [CrossRef]
- Soetens J.C. and Bopp P.A. Water-methanol mixtures: simulations of mixing properties over the entire range of mole fractions. The Journal of Physical Chemistry B 2015, 119, 8593–8599. [CrossRef]
- McGlashan M.L. and Williamson A. Isothermal liquidvapor equilibria for system methanol-water. Journal of Chemical Engineering Data 1976, 21, 196–199. [CrossRef]
- Van Ness, H.C. Thermodynamics in the treatment of vapor/liquid equilibrium (VLE) data. Pure and Applied Chemistry 1995, 67, 859–872. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





