Submitted:
14 March 2024
Posted:
17 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
- synthesis and physicochemical characterization of the samples (size, morphology, and the phase composition)
- spectral-kinetic characterization to choose optimal Eu3+ and Nd3+ concentrations
- spectral-kinetic characterization in order to understand the influence of the annealing procedure on spectral-kinetic characteristics
- The calculation of Sa and Sr.
2. Materials and Methods
3. Results and Discussion
3.1. Physicochemical Characterization of the Nanoparticles
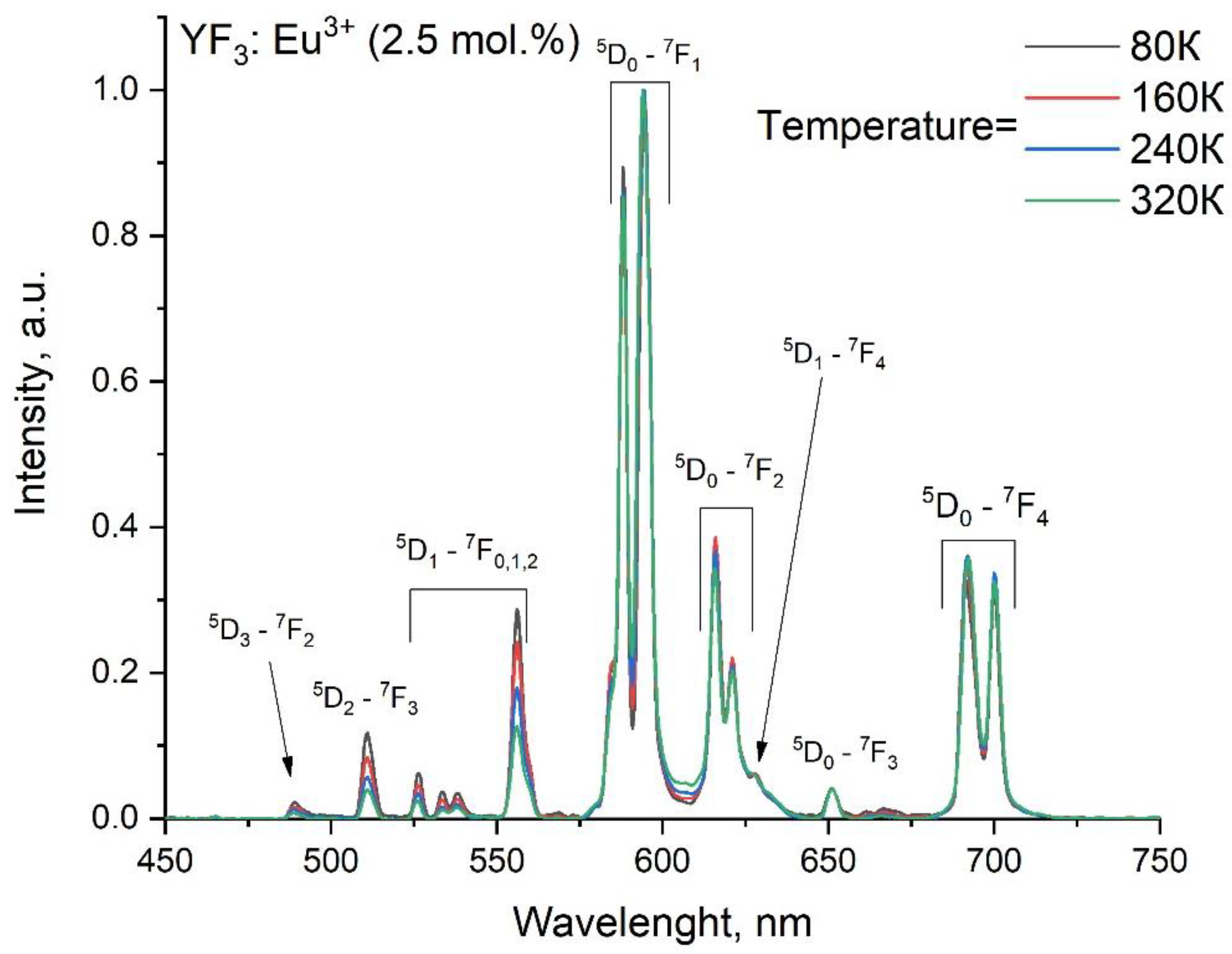
3.2. Temperature-Dependent Spectral-Kinetic Characterization of Single-Doped YF3: Eu3+
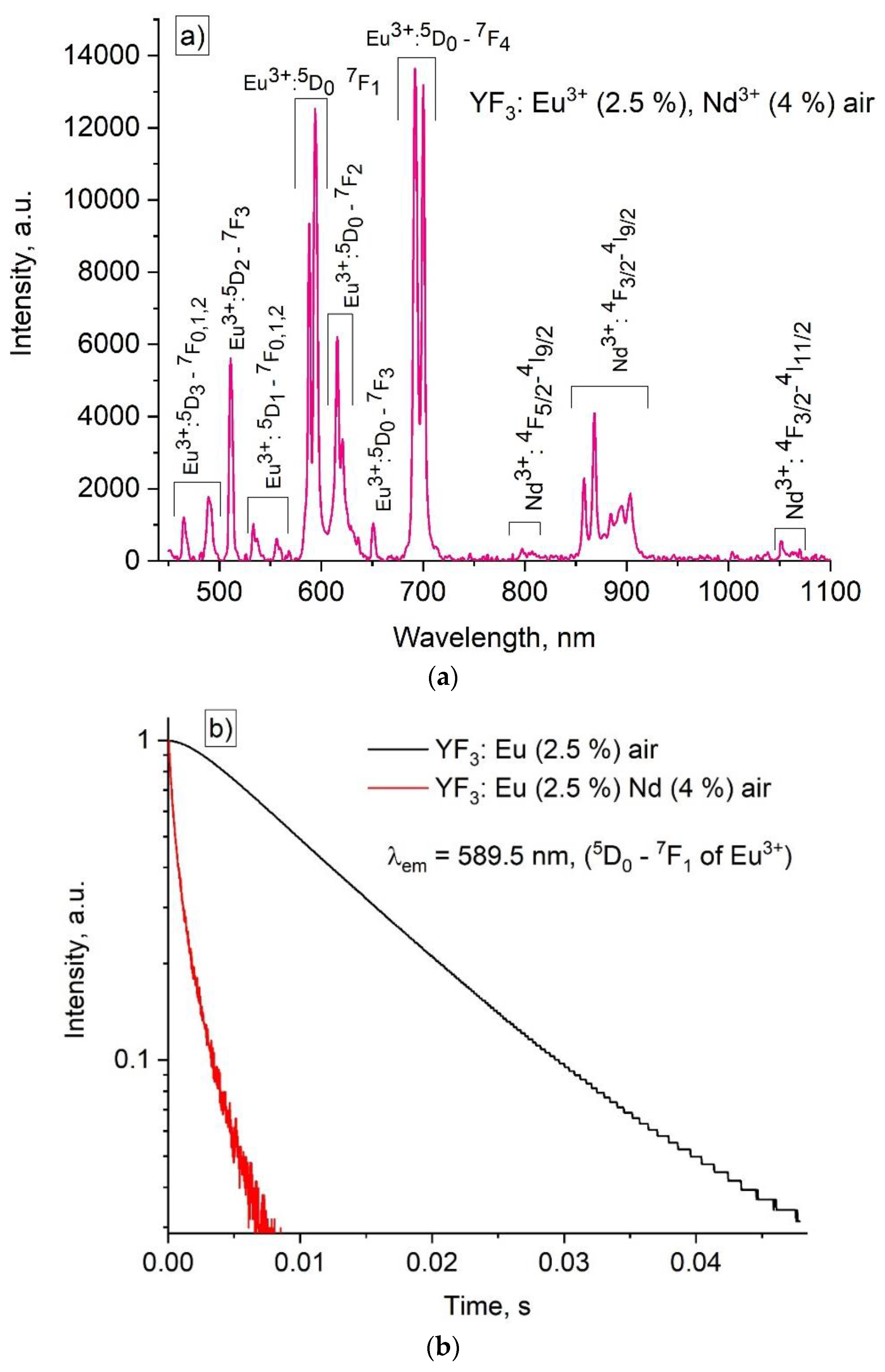
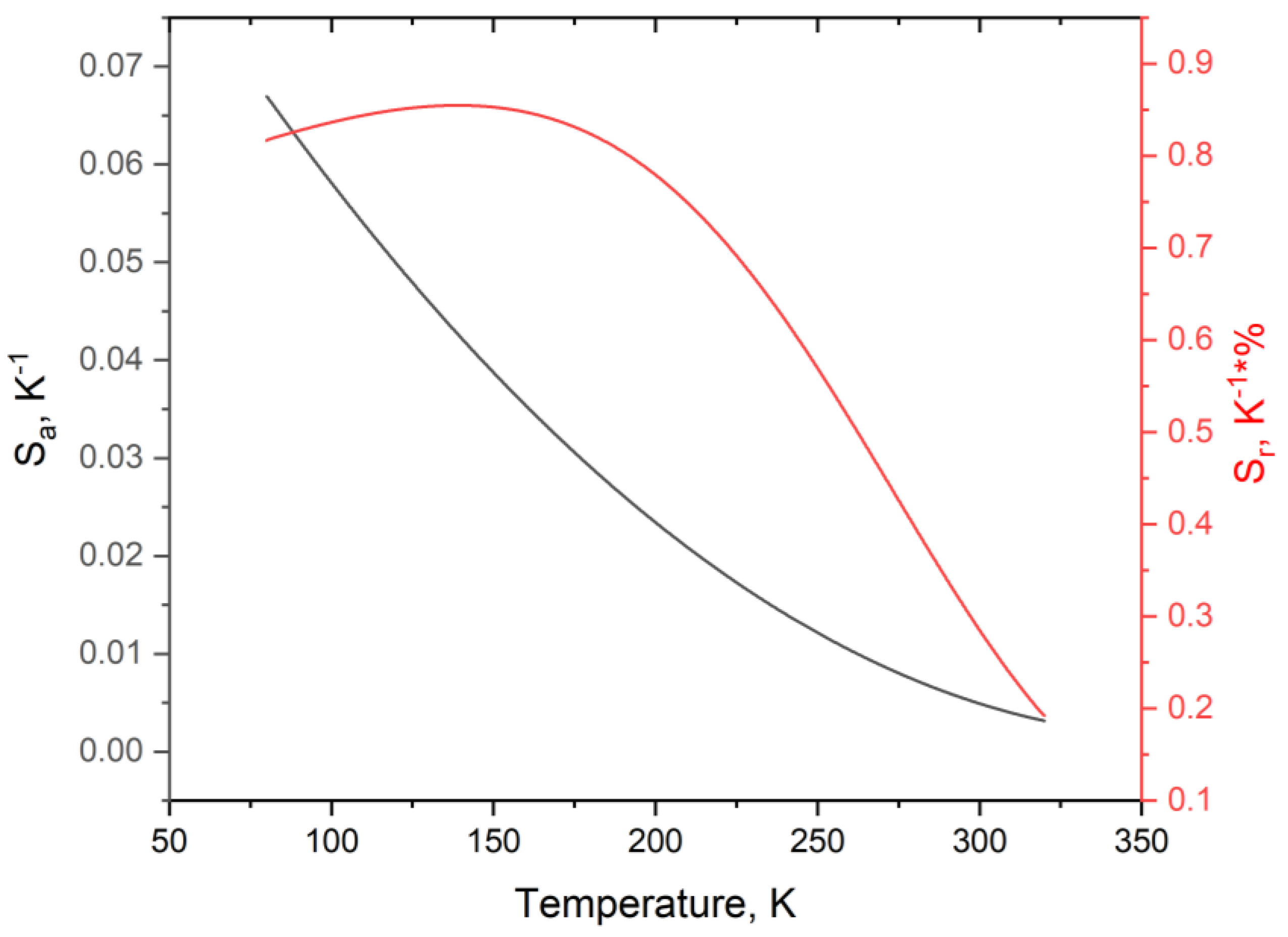
3.3. Temperature-Dependent Spectral Characterization of Double-Doped YF3:(Eu3+, Nd3+)
3.4. Temperature-Dependent Kinetic Characterization of Double-Doped YF3: Eu3+, Nd3+
| Sample | Transition, wavelength, and excitation conditions | Max Sa [μs/K] | Max Sr [%/K] in the | Ref. |
|---|---|---|---|---|
| annealed YF3: Eu3+,Nd3+ | Emission of Eu3+ (5D0 - 7F1, ~ 590 nm), λex = 394 nm (7F0 - 5L6 absorption band) | 10 - 18 in the 80 - 200 K | 0.2 - 0.3, in the 80 - 200 K | This work |
| β-NaGdF4: Nd3+,Yb3+ | Yb3+ (2F5/2 - 2F7/2, ~ 980 nm),λex = 808 nm (4I9/2 - 4F5/2 abs. of Nd3+). | Linear increase from 1.0 (300 K) to 2.8 (at 350 K) | Increases from 0.7 (300 K) to 1.6 (at 350 K) | [29] |
| Nd0.5RE0.4Yb0.1PO4 (RE = Y, Lu, La, Gd) | Yb3+ (2F5/2 - 2F7/2, ~ 980 nm), λex = 940 nm, 2F7/2 - 2F5/2 absorption band of Yb3+. | 0.4 - 1.6 at 300 K | 0.5 - 1.2 at 300 K | [30] |
| LiYXYb1-XF4: Tm3+ | λex = 688 nm, 3H6 - 3F2,3 (Tm3+) absorption band of | 1.2 | 0.36 | [31] |
| β-PbF2: Tm3+, Yb3+ | Tm3+ (1G4 - 3H6, 478 nm), (2F7/2 - 2F5/2 abs. of Yb3+) | – | 0.20 (at 300 K) | [32] |
| Gd2O2S: Eu3+ | Eu3+, 5D0 level, λex = 375 nm (the transition is not specified) | – | Linear decreas: 4.5 (at 280 K) to 3.0 (at 335 K) | [33] |
| LaGdO3: Er3+/Yb3+ | Er3+ (4S3/2 - 4I15/2, 530 nm), (4F9/2 - 4I15/2, 670 nm) (2F7/2 - 2F5/2 abs. of Yb3+) | – | 1.79 (4S3/2) and 0.94 (4F9/2) in the 290 - 350 K range. | [34] |
| TiO2: Sm3+ | Sm3+ (4G5/2 - 6H7/2, 612 nm) 438 nm (matrix excitation) | 10 %/ºC at 70 ºC | [35] | |
| NaPr(PO3)4 | Pr3+ (emission from 3P0, the wavelength is not specified), λex = 488 nm (3H4 - 3P0 absorption band of Pr3+. | Linearl increas: 44·10-4 (at 300 K) to 60·10-4 (at 365 K) | [22] | |
| LaF3: Pr3+ | Pr3+ (3P0 - 3H4, 486 nm) λex = 444 nm (3H4 - 3P2 abs. of Pr3+) | 0.7·10-3 in the 80 - 320 K. | – | [36] |
| LaPO4: Nd3+,Er3+ | Nd3+ (4F5/2 - 4F11/2 λem = 1055 nm), λex = 808 nm abs. 4I9/2 - 4F5/2) | max value 0.003 at 600 K | max value ~ 2.5 at 600 K | [37] |
| MOF: Eu3+ | Host excitation under 368 nm, λem = 525 nm | linear decrease: ~ 550 us (at 270 K) to ~460 us (at 360 K). The estimated Sa is equal to 1.0 us/K | – | [38] |
| GAG: Mn3+, Mn4+ | λex = 266 nm, λem = 610 nm (5T2 - 5E″ of Mn3+) | 2.08 at 249 K | [39] | |
| Pr3+:YAG | Pr3+ (1D2 - 3H4, 617 nm), λex = 488 nm (3H4 - 3P0 absorption band of Pr3+. | linear decrease: ~ 190 us (at 10 K) to ~ 110 us (at 1000 K). The estimated Sa is equal to 0.080 us/K | – | [40] |
| CaF2: Ho3+ | Ho3+ (5F5 - 5I8, λem = 650 nm), λex = 488 nm (5F3 - 5I8 absorption band of Ho3+. | linear decrease: ~ 100 us (at 100 K) to ~ 40 us (at 450 K). The estimated Sa is equal to 0.17 us/K | – | [40] |
| LiPr(PO3)4 | Pr3+ (emission from 3P0, the wavelength is not specified) λex = 488 nm (3H4 - 3P0 absorption band of Pr3+. | 0.0044 K-1 in the 300 - 365 K range | The Sa increases almost linearly from 0.44 %/K (at 300 K) to 0.65 %/K (at 365 K) | [22] |
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dramićanin, M.D. Trends in luminescence thermometry. Journal of Applied Physics 2020, 128. [Google Scholar] [CrossRef]
- Brites, C.D.; Balabhadra, S.; Carlos, L.D. Lanthanide-based thermometers: at the cutting-edge of luminescence thermometry. Advanced Optical Materials 2019, 7, 1801239. [Google Scholar] [CrossRef]
- Brites, C.D.S.; Millán, A.; Carlos, L.D. Lanthanides in luminescent thermometry. In Handbook on the Physics and Chemistry of Rare Earths; 2016; Volume 49, pp. 339–427. [Google Scholar] [CrossRef]
- Fedorov, P.P.; Luginina, A.A.; Kuznetsov, S.V.; Osiko, V.V. Nanofluorides. Journal of Fluorine Chemistry 2011, 132, 1012–1039. [Google Scholar] [CrossRef]
- Nizamutdinov, A.; Lukinova, E.; Shamsutdinov, N.; Zelenikhin, P.; Khusainova, A.; Gafurov, M.; Pudovkin, M. CeF3-YF3-TbF3 nanoparticle-polymer–“radachlorin” conjugates for combined photodynamic therapy: synthesis, characterization, and biological activity. Journal of Composites Science 2023, 7, 255. [Google Scholar] [CrossRef]
- Pudovkin, M.S.; Zelenikhin, P.V.; Shtyreva, V.V.; Evtugyn, V.G.; Salnikov, V.V.; Nizamutdinov, A.S.; Semashko, V.V. Cellular uptake and cytotoxicity of unmodified Pr3+: LaF3 nanoparticles. Journal of Nanoparticle Research 2019, 21, 1–13. [Google Scholar] [CrossRef]
- Meijer, J.M.; Aarts, L.; van der Ende, B.M.; Vlugt, T.J.; Meijerink, A. Downconversion for solar cells in YF3: Nd3+, Yb3+. Physical Review B 2010, 81, 035107. [Google Scholar] [CrossRef]
- Pudovkin, M.; Oleynikova, E.; Kiiamov, A.; Cherosov, M.; Gafurov, M. Nd3+, Yb3+: YF3 Optical Temperature Nanosensors Operating in the Biological Windows. Materials 2022, 16, 39. [Google Scholar] [CrossRef]
- Piñol, R.; Brites, C.D.; Silva, N.J.; Carlos, L.D.; Millán, A. Nanoscale thermometry for hyperthermia applications. In Nanomaterials for Magnetic and Optical Hyperthermia Applications; 2019; pp. 139–172. [Google Scholar] [CrossRef]
- Jaque, D.; Vetrone, F. Luminescence nanothermometry. Nanoscale 2012, 4, 4301–4326. [Google Scholar] [CrossRef]
- Chen, D.; Wang, Z.; Zhou, Y.; Huang, P.; Ji, Z. Tb3+/Eu3+: YF3 nanophase embedded glass ceramics: Structural characterization, tunable luminescence and temperature sensing behavior. Journal of Alloys and Compounds 2015, 646, 339–344. [Google Scholar] [CrossRef]
- Pudovkin, M.S.; Ginkel, A.K.; Lukinova, E.V. Temperature sensitivity of Nd3+, Yb3+:YF3 ratiometric luminescent thermometers at different Yb3+ concentration. Optical Materials 2021, 119, 111328. [Google Scholar] [CrossRef]
- Pudovkin, M.S.; Ginkel, A.K.; Morozov, O.A.; Kiiamov, A.G.; Kuznetsov, M.D. Highly-sensitive lifetime optical thermometers based on Nd3+, Yb3+: YF3 phosphors. Journal of Luminescence 2022, 249, 119037. [Google Scholar] [CrossRef]
- Kaczmarek, A.M.; Kaczmarek, M.K.; Van Deun, R. Er3+-to-Yb3+ and Pr3+-to-Yb3+ energy transfer for highly efficient near-infrared cryogenic optical temperature sensing. Nanoscale 2019, 11, 833–837. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Li, S.; Min, Q.; Lu, W.; Xu, X.; Zhou, D.; Yu, X. Optical temperature sensing properties of KLu2F7: Yb3+/Er3+/Nd3+ nanoparticles under NIR excitation. Journal of Alloys and Compounds 2018, 742, 497–503. [Google Scholar] [CrossRef]
- Alakshin, E.M.; Klochkov, A.V.; Kondratyeva, E.I.; Korableva, S.L.; Kiiamov, A.G.; Nuzhina, D.S.; Stanislavovas, A.A.; Tagirov, M.S.; Zakharov, M.Y.; Kodjikian, S. Microwave-assisted hydrothermal synthesis and annealing of DyF3 nanoparticles. Journal of Nanomaterials 2016, 2016, 7148307. [Google Scholar] [CrossRef]
- Tan, M.C.; Kumar, G.A.; Riman, R.E.; Brik, M.G.; Brown, E.; Hommerich, U. Synthesis and optical properties of infrared-emitting YF3:Nd nanoparticles. Journal of Applied Physics 2009, 106. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. Journal of applied crystallography 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Wang, X.; Sheng, T.; Fu, Z.; Li, W. Highly uniform YF3: Ln3+ (Ln = Ce3+, Tb3+) walnut-like microcrystals: Hydrothermal synthesis and luminescent properties. Materials Research Bulletin 2013, 48. [Google Scholar] [CrossRef]
- Vanetsev, A.; Kaldvee, K.; Puust, L.; Keevend, K.; Nefedova, A.; Fedorenko, S.; Orlovskii, Y. Relation of Crystallinity and Fluorescent Properties of LaF3: Nd3+ Nanoparticles Synthesized with Different Water-Based Techniques. ChemistrySelect 2017, 2, 4874–4881. [Google Scholar] [CrossRef]
- Alakshin, E.M.; Blokhin, D.S.; Sabitova, A.M.; Klochkov, A.V.; Klochkov, V.V.E.; Kono, K.; Tagirov, M.S. Experimental proof of the existence of water clusters in fullerene-like PrF3 nanoparticles. JETP letters 2012, 96, 181–183. [Google Scholar] [CrossRef]
- Gharouel, S.; Labrador-Páez, L.; Haro-González, P.; Horchani-Naifer, K.; Férid, M. Fluorescence intensity ratio and lifetime thermometry of praseodymium phosphates for temperature sensing. Journal of Luminescence 2018, 201, 372–383. [Google Scholar] [CrossRef]
- Liu, J.; Van Deun, R.; Kaczmarek, A.M. Eu3+, Tb3+-and Er3+, Yb3+-doped α-MoO3 nanosheets for optical luminescent thermometry. Nanomaterials 2019, 9, 646. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Zhao, Z.; Chi, F.; Wei, X.; Yin, M. Structural characterization and temperature-dependent luminescence of CaF2: Tb3+/Eu3+ glass ceramics. Journal of Rare Earths 2017, 35, 536–541. [Google Scholar] [CrossRef]
- Zhou, A.; Song, F.; Song, F.; Feng, M.; Adnan, K.; Ju, D.; Wang, X. Optical thermometry using fluorescence intensities multi-ratios in NaGdTiO4: Yb3+/Tm3+ phosphors. Optical Materials 2018, 78, 438–444. [Google Scholar] [CrossRef]
- Maciejewska, K.; Bednarkiewicz, A.; Marciniak, L. NIR luminescence lifetime nanothermometry based on phonon assisted Yb3+–Nd3+ energy transfer. Nanoscale Advances 2021, 3, 4918–4925. [Google Scholar] [CrossRef] [PubMed]
- Pudovkin, M.S.; Kuznetsov, S.V.; Proydakova, V.Y.; Voronov, V.V.; Semashko, V.V. Luminescent thermometry based on Ba4Y3F17: Pr3+ and Ba4Y3F17:Pr3+,Yb3+ nanoparticles. Ceramics International 2020, 46, 11658–11666. [Google Scholar] [CrossRef]
- Bednarkiewicz, A.; Stefanski, M.; Tomala, R.; Hreniak, D.; Strek, W. Near infrared absorbing near infrared emitting highly-sensitive luminescent nanothermometer based on Nd3+ to Yb3+ energy transfer. Physical Chemistry Chemical Physics 2015, 17, 24315–24321. [Google Scholar] [CrossRef]
- Ji, Z.; Cheng, Y.; Cui, X.; Lin, H.; Xu, J.; Wang, Y. Heating-induced abnormal increase in Yb3+ excited state lifetime and its potential application in lifetime luminescence nanothermometry. Inorganic Chemistry Frontiers 2019, 6, 110–116. [Google Scholar] [CrossRef]
- Maciejewska, K.; Bednarkiewicz, A.; Marciniak, L. NIR luminescence lifetime nanothermometry based on phonon assisted Yb3+–Nd3+ energy transfer. Nanoscale Advances 2021, 3, 4918–4925. [Google Scholar] [CrossRef]
- Khadiev, A.R.; Korableva, S.L.; Ginkel, A.K.; Morozov, O.A.; Nizamutdinov, A.S.; Semashko, V.V.; Pudovkin, M.S. Down-conversion based Tm3+: LiY1-XYbXF4 temperature sensors. Optical Materials 2022, 134, 113118. [Google Scholar] [CrossRef]
- Fu, Y.; Zhao, L.; Guo, Y.; Yu, H. Up-conversion luminescence lifetime thermometry based on the 1G4 state of Tm3+ modulated by cross relaxation processes. Dalton Transactions 2019, 48, 16034–16040. [Google Scholar] [CrossRef]
- Katumo, N.; Gao, G.; Laufer, F.; Richards, B.S.; Howard, I.A. Smartphone-based luminescent thermometry via temperature-sensitive delayed fluorescence from Gd2O2S: Eu3+. Advanced Optical Materials 2020, 8, 2000507. [Google Scholar] [CrossRef]
- Siaï, A.; Haro-González, P.; Naifer, K.H.; Férid, M. Optical temperature sensing of Er3+/Yb3+ doped LaGdO3 based on fluorescence intensity ratio and lifetime thermometry. Optical Materials 2018, 76, 34–41. [Google Scholar] [CrossRef]
- Dramićanin, M.D.; Antić, Ž.; Ćulubrk, S.; Ahrenkiel, S.P.; Nedeljković, J.M. Self-referenced luminescence thermometry with Sm3+ doped TiO2 nanoparticles. Nanotechnology 2014, 25, 485501. [Google Scholar] [CrossRef] [PubMed]
- Pudovkin, M.S.; Koryakovtseva, D.A.; Lukinova, E.V.; Korableva, S.L.; Khusnutdinova, R.S.; Kiiamov, A.G.; Semashko, V.V. Luminescence nanothermometry based on Pr3+: LaF3 single core and Pr3+:LaF3/LaF3 core/shell nanoparticles. Advances in Materials Science and Engineering 2019, 2019, 2618307. [Google Scholar] [CrossRef]
- Maciejewska, K.; Bednarkiewicz, A.; Marciniak, L. The influence of the Er3+ dopant concentration in LaPO4: Nd3+, Er3+ on thermometric properties of ratiometric and kinetic-based luminescent thermometers operating in NIR II and NIR III optical windows. Physica B: Condensed Matter 2021, 620, 413247. [Google Scholar] [CrossRef]
- Zhou, Y.; Yan, B. Ratiometric detection of temperature using responsive dual-emissive MOF hybrids. Journal of Materials Chemistry C 2015, 3, 9353–9358. [Google Scholar] [CrossRef]
- Marciniak, L.; Trejgis, K. Luminescence lifetime thermometry with Mn3+–Mn4+ co-doped nanocrystals. Journal of Materials Chemistry C 2018, 6, 7092–7100. [Google Scholar] [CrossRef]
- Kamma, I.; Kommidi, P.; Reddy, B.R. High temperature measurement using luminescence of Pr3+ doped YAG and Ho3+ doped CaF2. Physica Status Solidi C 2009, 6 (S1 1), S187–S190. [Google Scholar] [CrossRef]
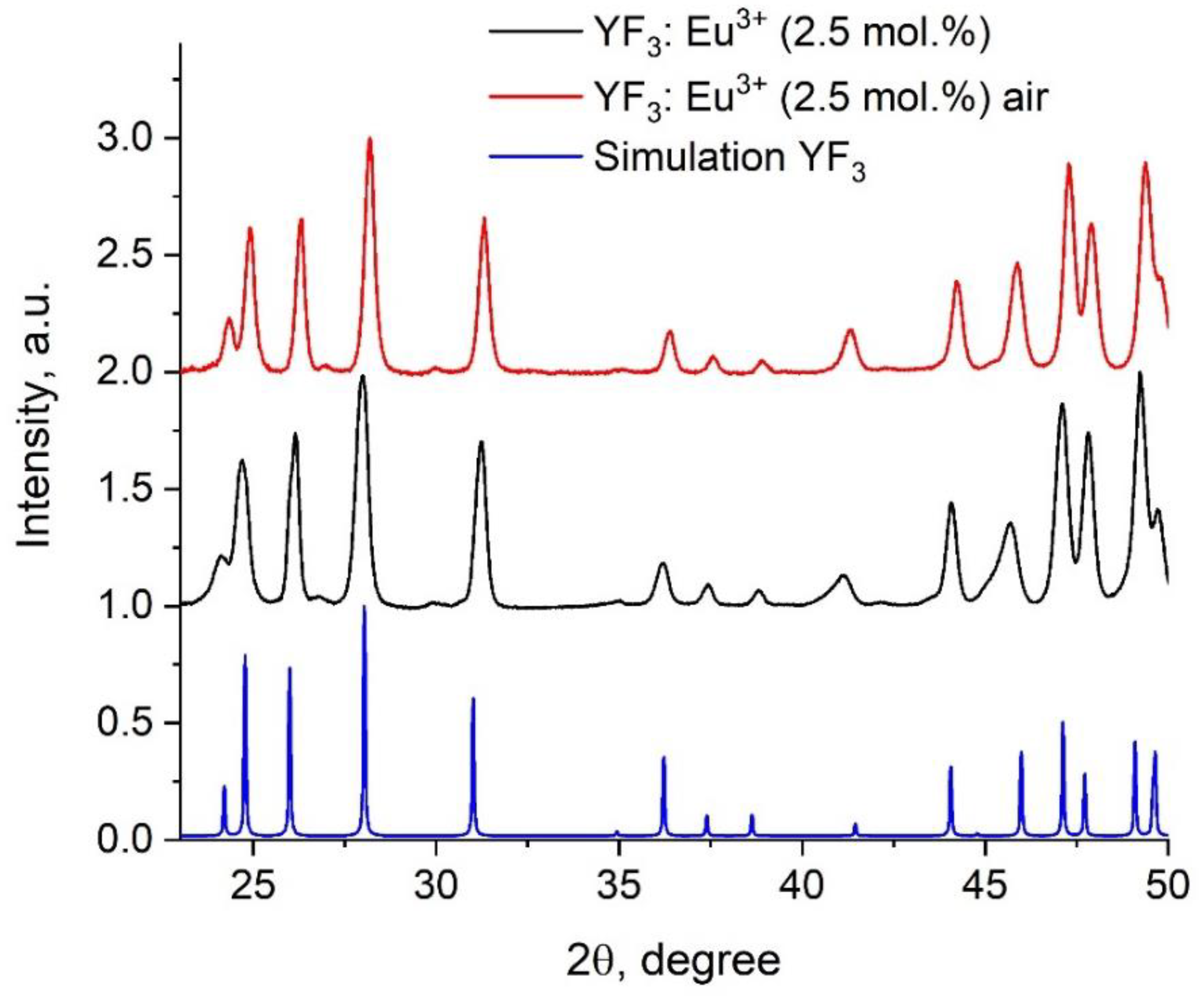

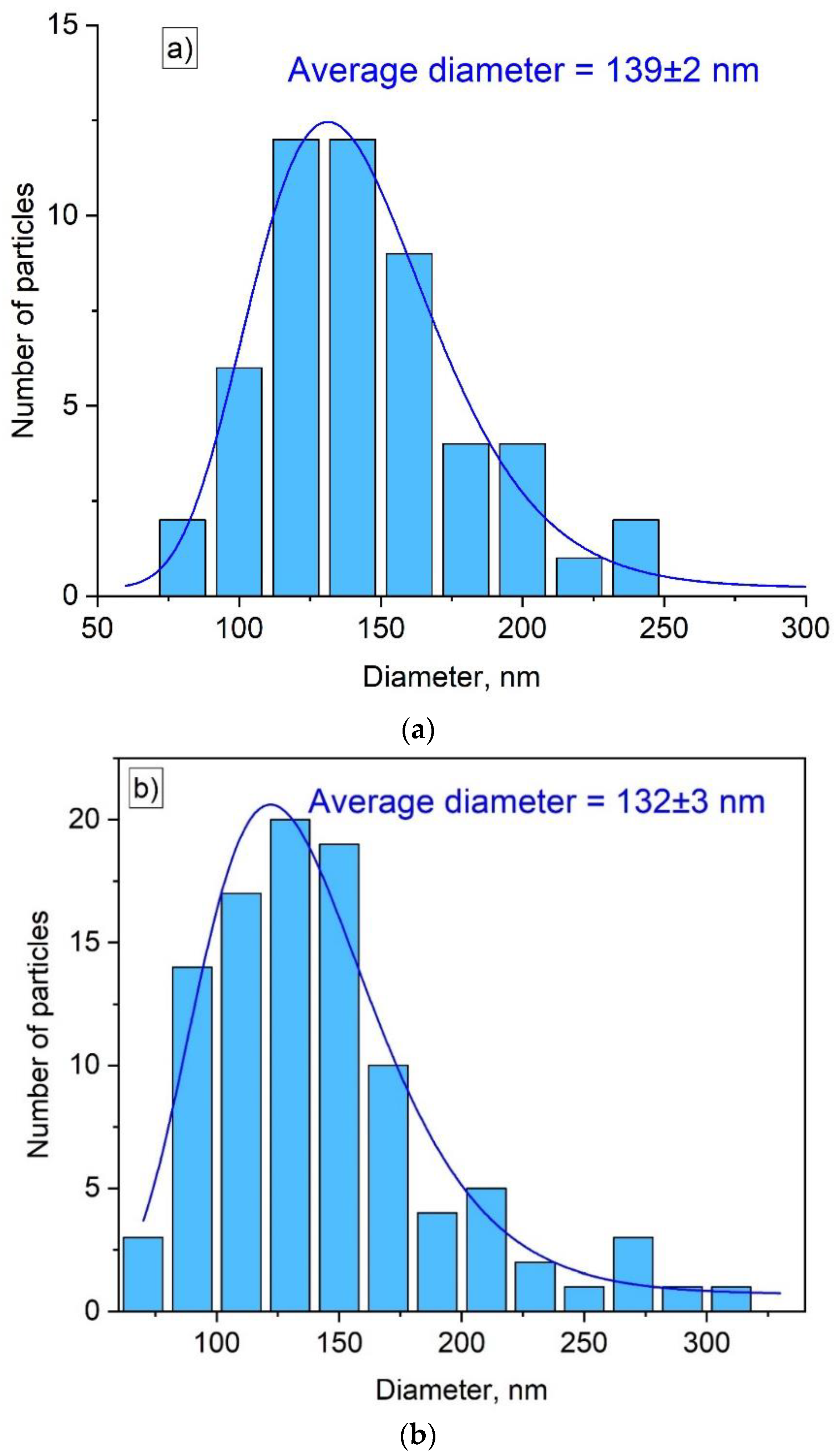
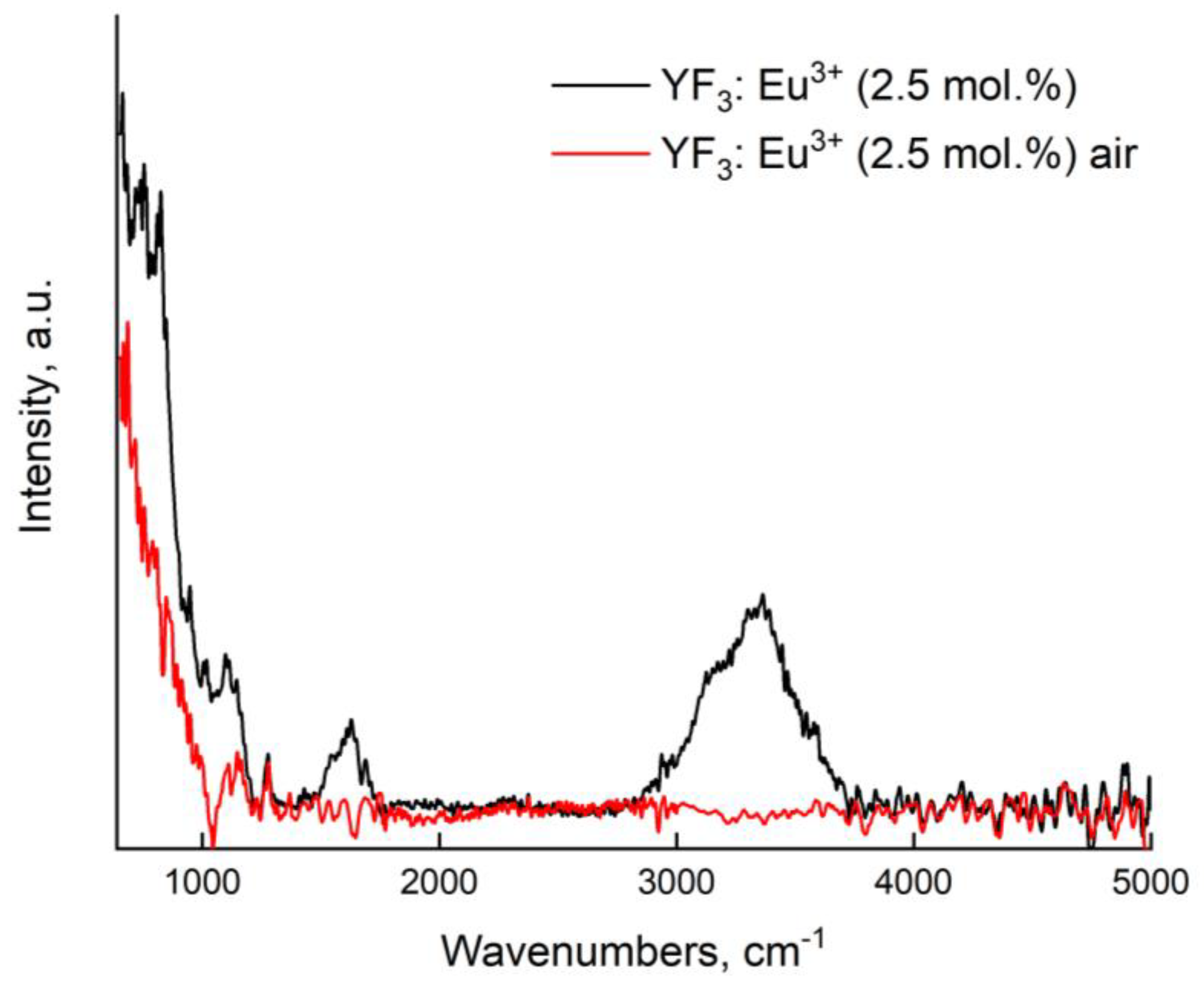

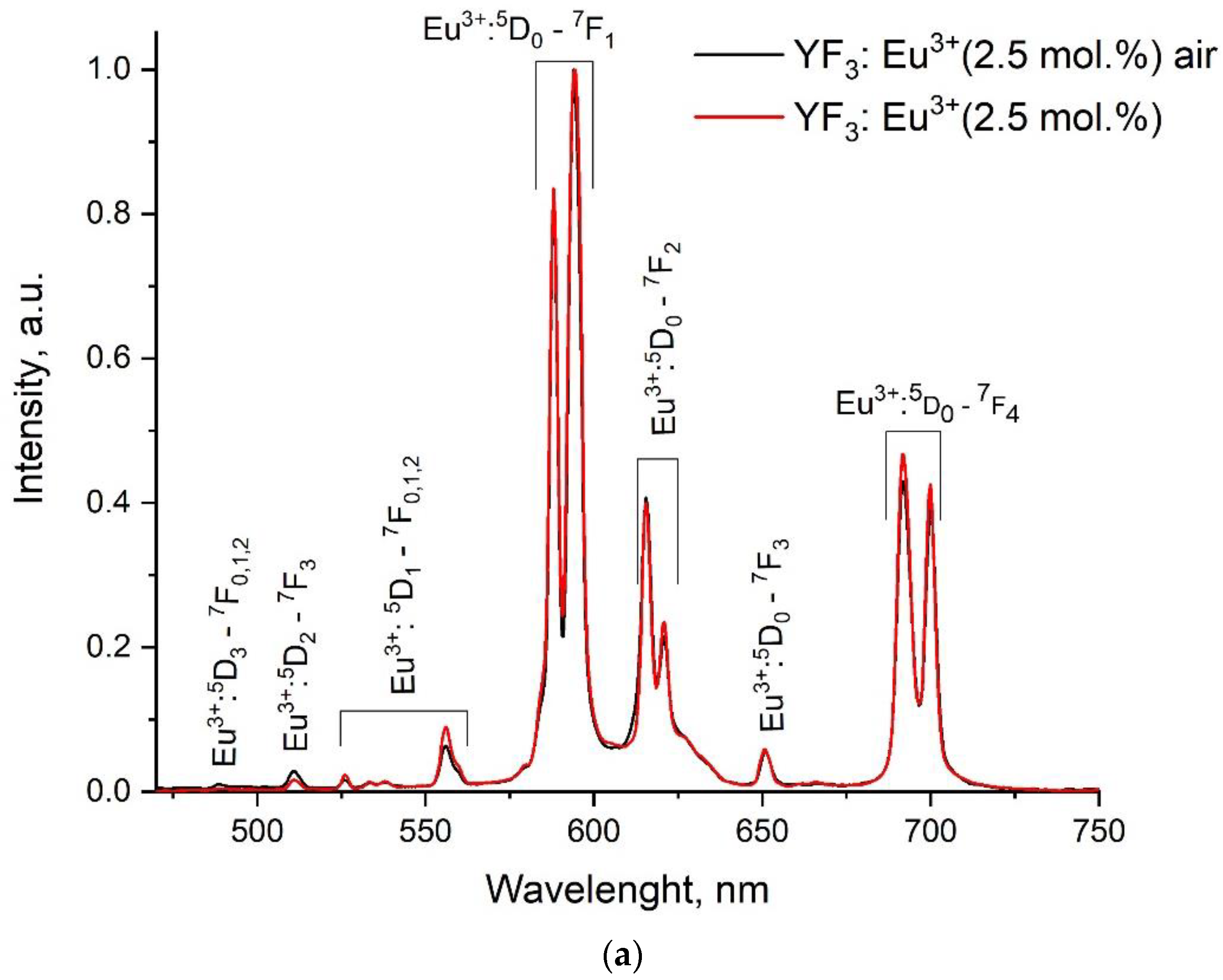
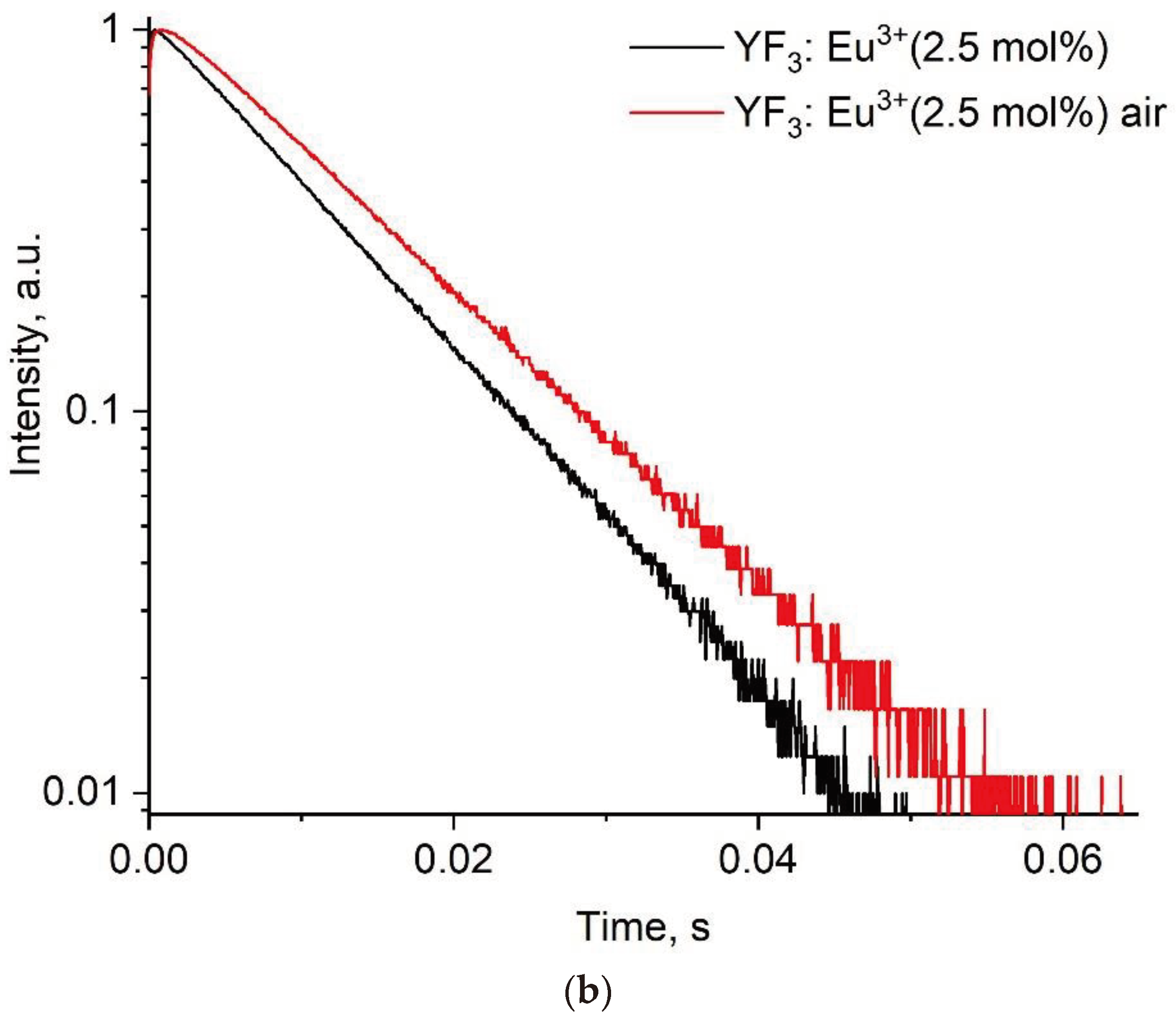
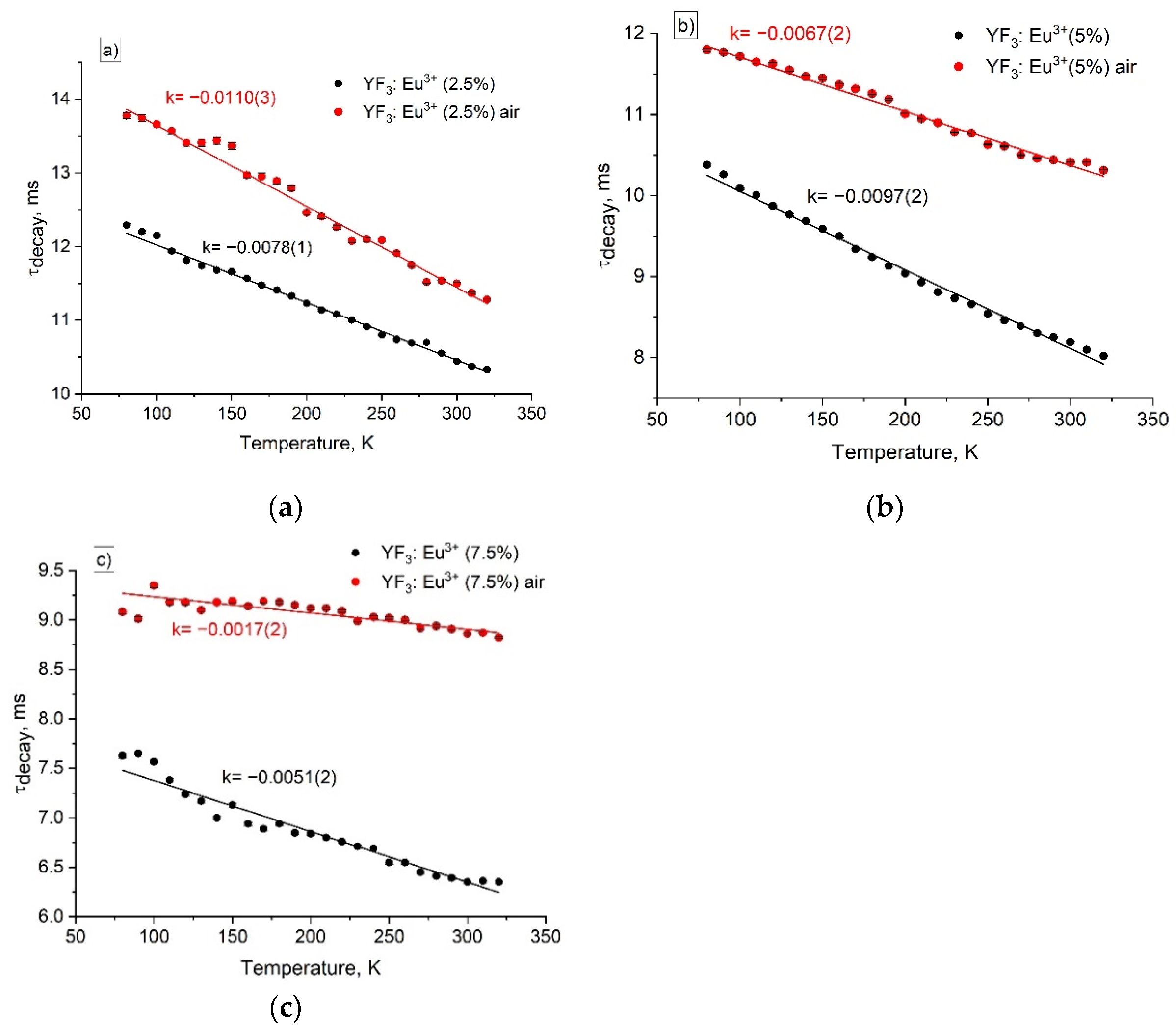
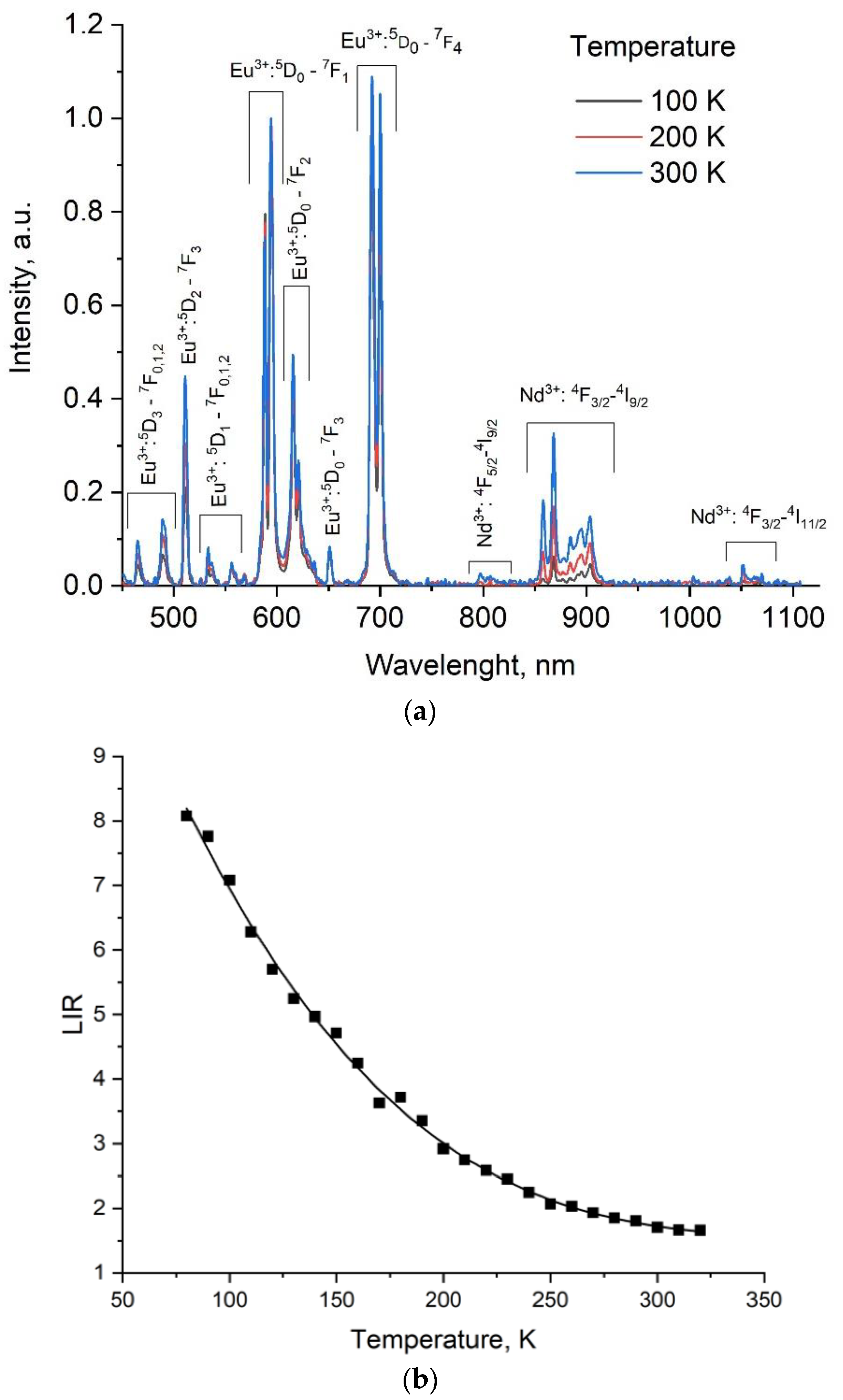
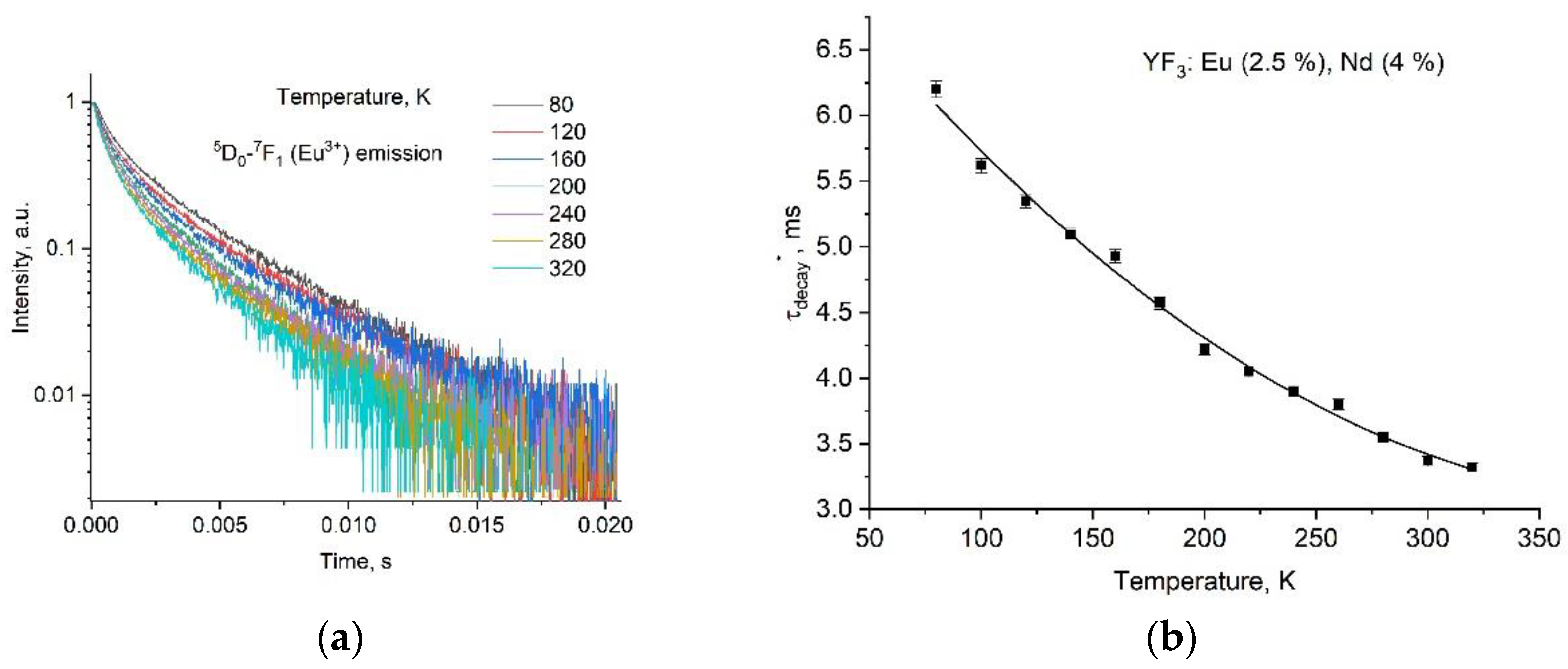
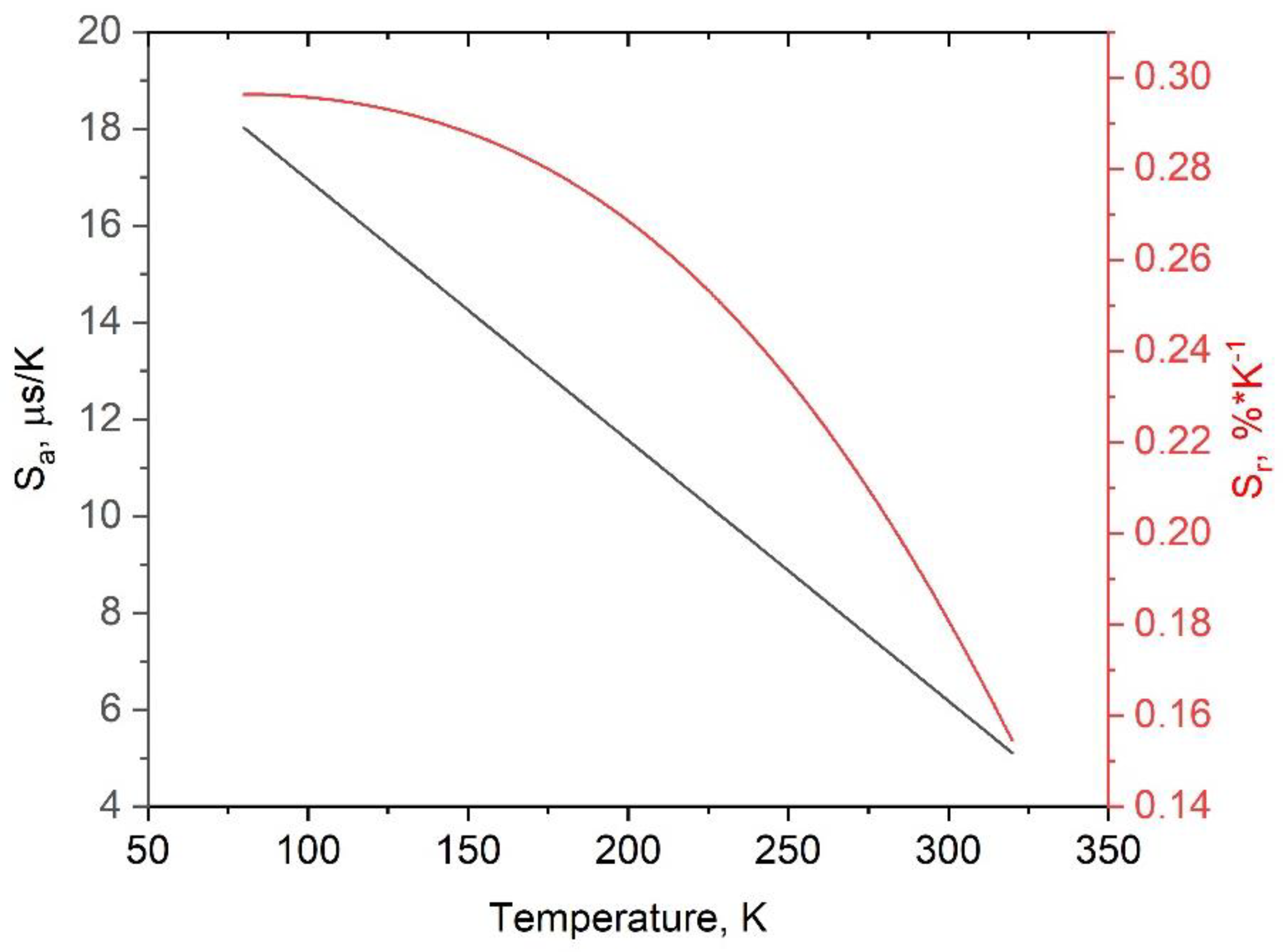
| Sample | YF3: Eu3+ 2.5% | YF3: Eu3+ 5.0% | YF3: Eu3+ 7.5% |
| Before annealing | 78·10-4 | 97·10-4 | 51·10-4 |
| After annealing | 110·10-4 | 67·10-4 | 17·10-4 |
| Sample | Transitions and wavelengths for LIR (I1/I2) and optical excitation conditions | Maximum Sa [K-1] in the 100 - 220 K range | Maximum Sr [%K-1] in the 100 - 220 K range | Ref. |
| annealed YF3: Eu3+,Nd3+ | Nd3+ (4F3/2 - 4I9/2, ~ 866 nm), Eu3+ (5D0 - 7F1, ~ 590 nm) is carried out at 394 nm (7F0 - 5L6 absorption band) | 0.065 (80 K) | 0.85 (160 K) | This work |
| α-MoO3:Eu3+, Tb3+ | ITb (5D4 - 7F5, ~ 548 nm)/IEu (5D0 - 7F2, ~ 621 nm) | ~10-3 at 105 K, not studied at higher temperatures | ~ 0.50 at 105 K, not studied at higher temperatures | [23] |
| Tb3+, Eu3+:CaF2 | ITb (5D4 - 7F5, ~ 545 nm)/IEu (5D0 - 7F2, ~ 615 nm), λex = 485 nm pulse laser | 4.0·10-3 | – | [24] |
| Tb3+(6.0%),Eu3+(8.0%):Ca5(PO4)3F | ITb (5D4 - 7F5, ~ 548 nm)/IEu (5D0 - 7F2, ~ 621 nm), λex = 299 nm, laser | 1.31·10-3 | 0.40 | [24] |
| Yb3+,Tm3+:NaGdTiO4 | ITm (3H4 (1) →3H6, 812 nm)/ ITm (3H4 (2) →3H6, 798 nm), λex = 980 nm, CW laser | 2.0·10-3 at 100 K and 1.0·10-3 at 200 K | – | [25] |
| Nd3+(1%),Yb3+(0.5-5%):LiLaP4O12 | INd (4F3/2 - 4I9/2, ~ 866 nm)/IYb (2F5/2 - 2F7/2, ~ 980 nm), λex = 808 nm, CW laser | – | From 0.05 to 0.25 (depends on the Yb3+ concentration) | [26] |
| Pr3+(0.1%),Yb3+(10.0%):Ba4Y3F17 | IPr (2P0 - 4H6)/IYb (2F5/2 - 2F7/2), λex = 442 nm, pulse laser | 1.0·10-3 | 0.20 | [27] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





