1. Introduction
Renal cell carcinoma (RCC) is the most common kidney cancer and a global health concern [
1], with over 400,000 new cases reported in 2020 [
2]. Although surgery is the standard treatment for localized RCC, the recurrence rate is high [
3]. Advanced RCC treatment options such as immunotherapy and targeted therapies are promising but have limited efficacy [
4,
5,
6]. Therefore, safer and more effective treatments for metastatic RCC are urgently needed.
Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), a key member of the tumor necrosis factor (TNF) superfamily, selectively induces apoptosis in cancer cells [
7,
8] by binding to death receptors 4 and 5 (DR4/5) receptors [
9], triggering intracellular signaling, and leading to programmed cell death [
10]. However, cancer cells often resist TRAIL-induced apoptosis because of altered receptor expression and apoptotic protein dysregulation [
11]. Combining TRAIL-based therapies with agents that sensitize cancer cells to apoptosis holds promise in overcoming drug resistance and advancing therapeutic strategies.
Ginseng, a revered herb in Asia, has potential health benefits due to its adaptogenic properties [
12]. Recent studies have identified gintonin, a novel glycolipoprotein distinct from traditional ginsenosides, which activates G protein-coupled receptors, particularly LPA receptors [
13,
14]. A gintonin-enriched
Panax ginseng extract fraction (GEF) was prepared to harness the potency of gintonin through ethanol extraction and water fractionation [
15]. GEF and gintonin exhibit diverse pharmacological effects, including neuronal protection, anti-inflammatory action, and the suppression of cancer cell metastasis [
16,
17,
18,
19].
Mu-2-related death-inducing gene (MUDENG, MuD), a novel gene structurally similar to the Mu-2 adaptin subunit, is involved in intracellular trafficking mechanisms [
20,
21] and apoptotic signaling pathways [
21,
22,
23,
24]. It plays a role in TRAIL-induced apoptosis by interacting with key molecules, such as caspase-3, BH3 interacting-domain death agonist (Bid), and B-cell lymphoma (Bcl)-2 [
24].
In this study, we explored the effects of GEF on TRAIL-induced apoptosis in human RCC cells. Our findings revealed that GEF augmented DR4/5 expression and potentiated caspase-dependent TRAIL-induced apoptosis in these cells.
2. Results
2.1. GEF Enhances TRAIL-induced Cell Death and Inhibits Proliferation in Human RCC Cells
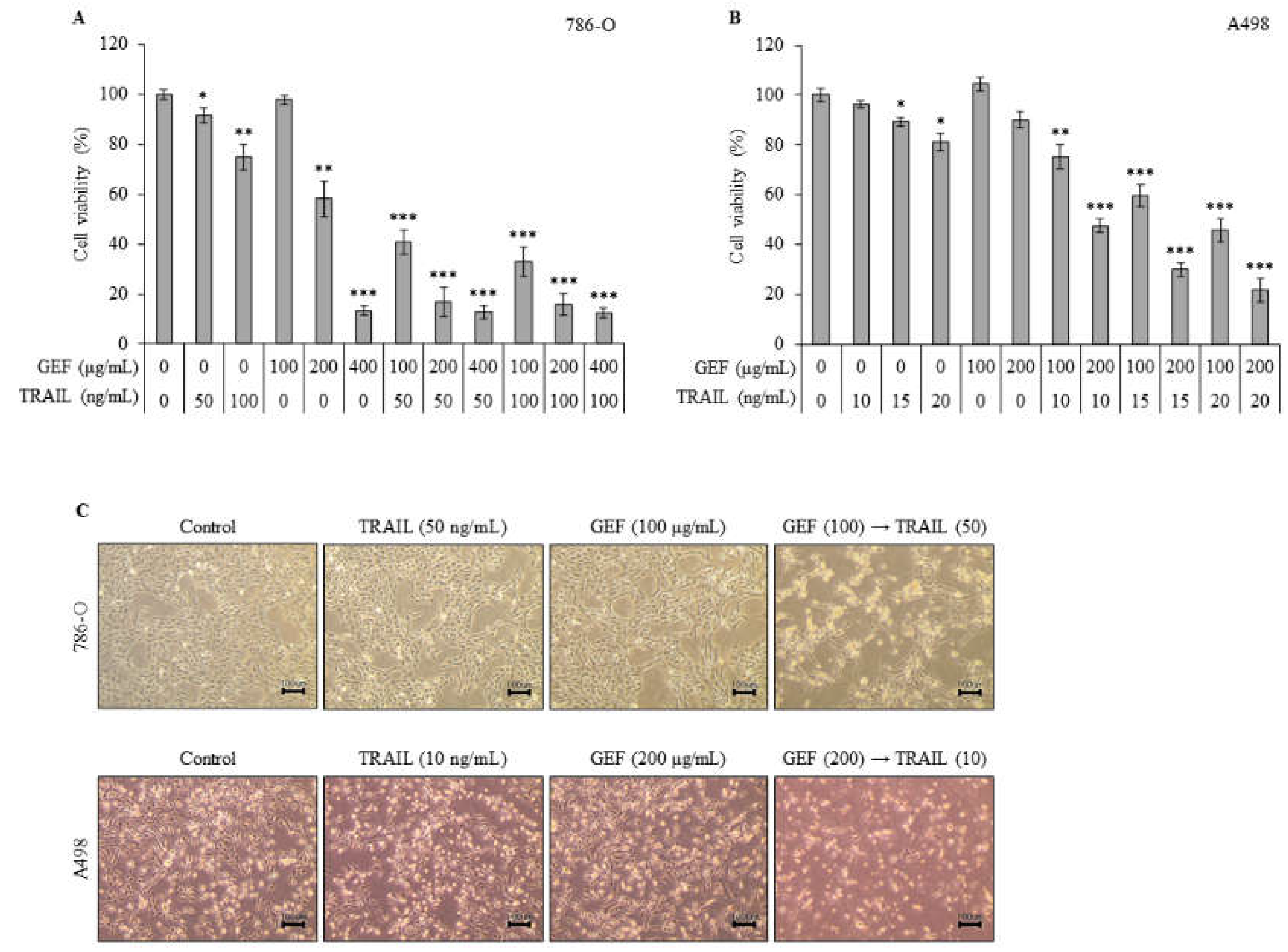
We initially evaluated the effects of GEF and TRAIL on RCC cell viability using WST-1 assay. Treatment of 786-O cells with 100, 200, and 400 μg/mL GEF reduced cell viability dose-dependently (
Figure 1A). Pre-treatment with 100 μg/mL GEF followed by 50 or 100 ng/mL TRAIL treatment significantly reduced cell viability compared with GEF pre-treatment alone (
Figure 1A). Notably, the combination of 100 μg/mL GEF and 50 ng/mL TRAIL synergistically affected cell viability, suggesting potential cooperation between the two treatments (
Figure 1A). This concentration combination was selected for subsequent experiments.
A498 cells were more sensitive to TRAIL than 786-O cells. Thus, they were treated with 10, 15, or 20 ng/mL TRAIL for 12 h after pre-treatment with 100 or 200 μg/mL GEF for the same duration. Treatment with 200 μg/mL GEF followed by 10, 15, or 20 ng/mL TRAIL treatment decreased A498 cell viability to 47.55±2.57%, 30.08±2.78%, or 21.77±4.68%, respectively (
Figure 1B). Even after the 100-μg/mL GEF pre-treatment, A498 cell viability significantly reduced upon subsequent TRAIL treatment (
Figure 1B). Therefore, for further investigations, A498 cells were pre-treated with 200 μg/mL GEF for 12 h followed by treatment with 10 ng/mL TRAIL for another 12 h.
The morphology of both 786-O and A498 cells altered after 12 h of pre-treatment with GEF, followed by TRAIL treatment for another 12 h, whereas no significant changes were observed when treated individually (
Figure 1C). Moreover, combined TRAIL treatment following GEF pre-treatment decreased both 786-O and A498 cell counts (
Figure 1C). These findings highlight the ability of GEFs to sensitize RCC cells to TRAIL, indicating a synergistic effect in suppressing RCC cell proliferation.
2.2. GEF Synergistically Potentiates TRAIL-induced Inhibition of Colony Formation and Cell Migration in Human RCC Cells.
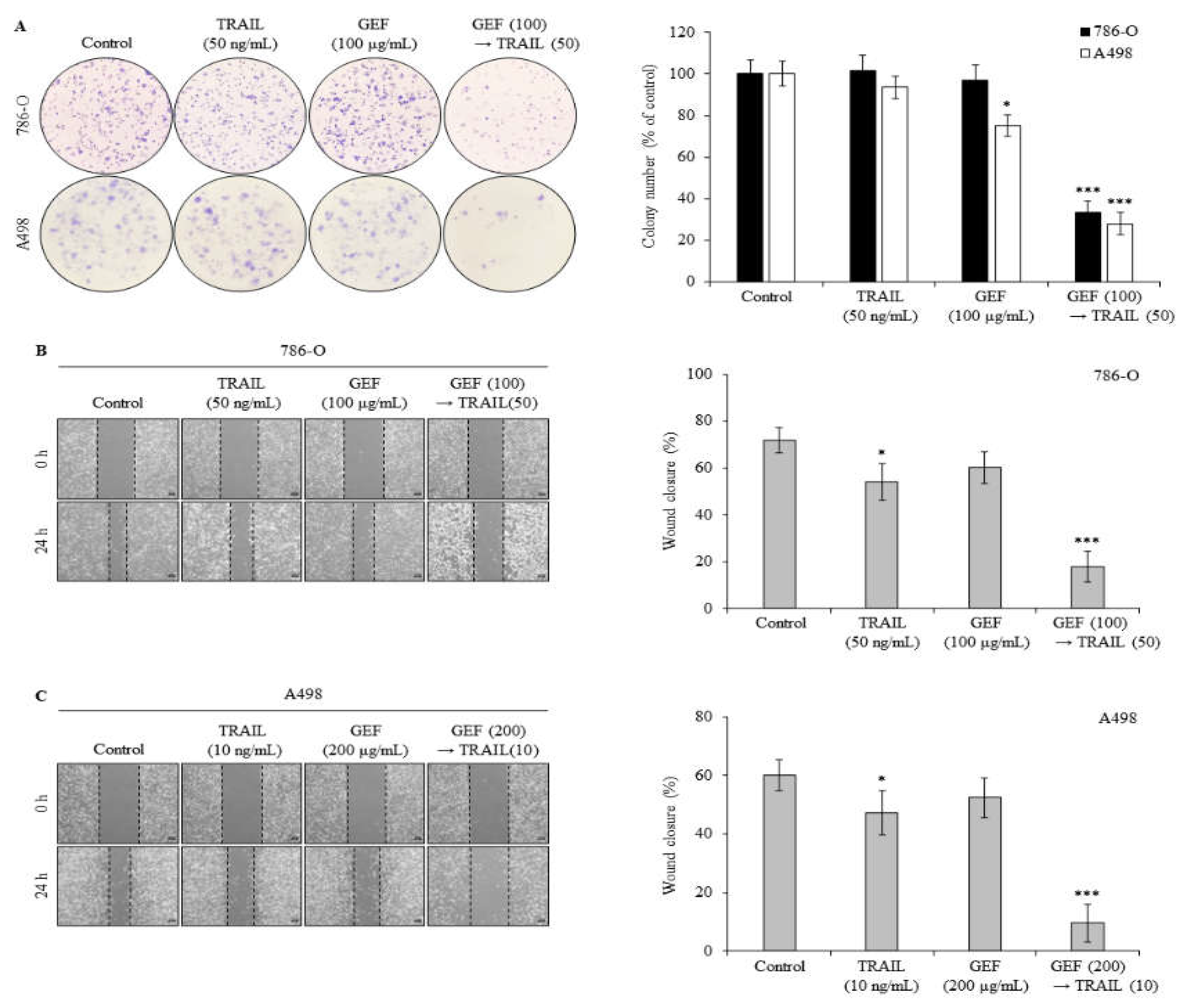
To assess the effect of GEF and TRAIL on colony formation and migration in 786-O and A498 RCC cells, we performed colony formation and wound healing assays. In both assays, pre-treatment with GEF followed by TRAIL treatment markedly suppressed colony formation (
Figure 2A). While individual GEF or TRAIL administration did not significantly alter colony formation, the colony count reduced to 33.54±5.32% and 27.89±5.33% in 786-O and A498 cells, respectively, upon TRAIL treatment following GEF pre-treatment.
Wound healing assays showed that individual GEF or TRAIL treatments modestly reduced wound closure rates in 786-O (
Figure 2B) and A498 (
Figure 2C) cells compared to controls. However, the wound closure rates significantly decreased in both cell lines following GEF pre-treatment followed by TRAIL treatment (
Figure 2B and 2C), highlighting the potent inhibitory effects of the combination of GEF and TRAIL on cell growth and migration.
2.3. GEF Augments TRAIL-induced Apoptosis and DNA Fragmentation in Human RCC Cells
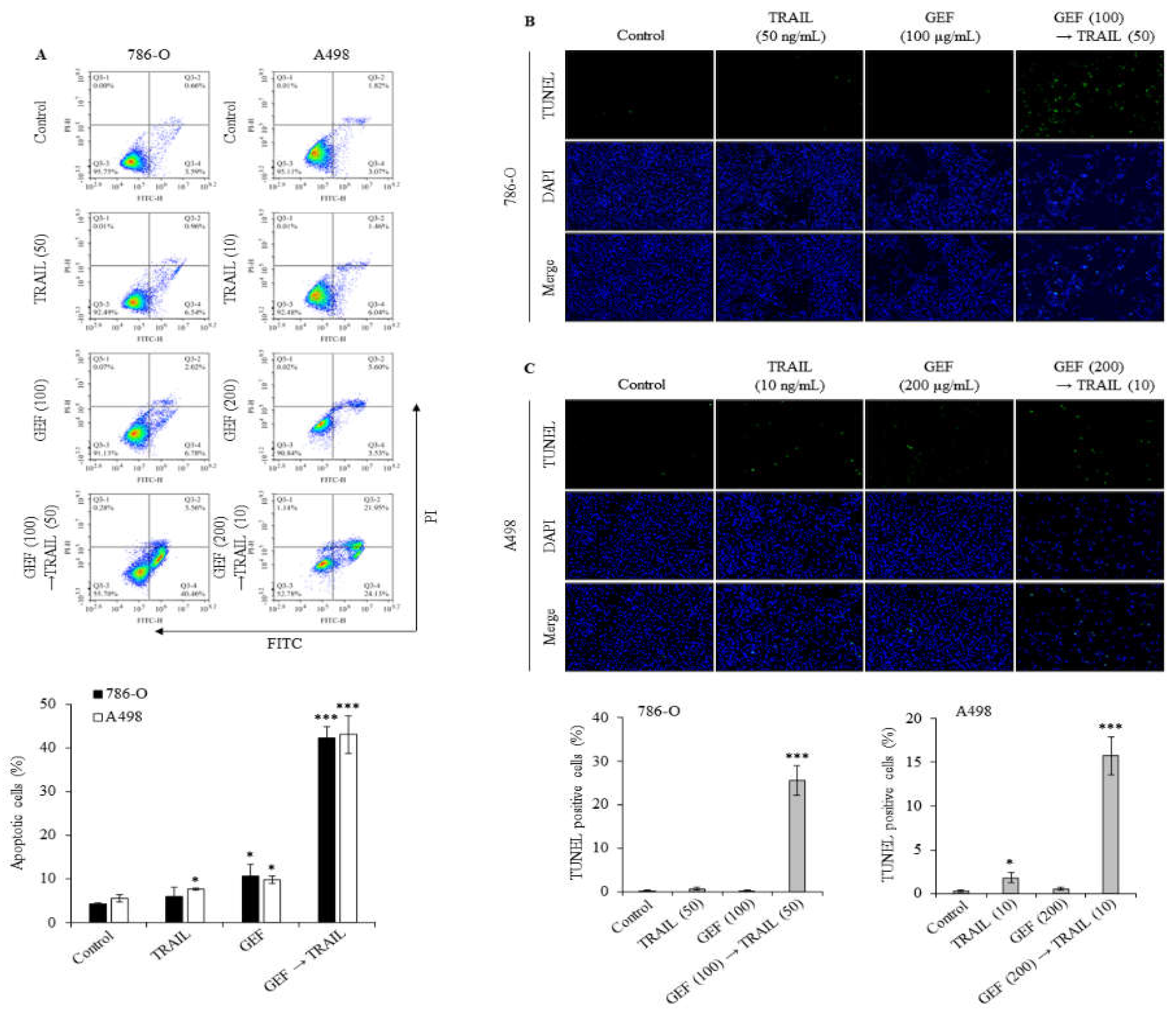
Apoptosis assays using Annexin V/PI staining revealed a significant increase in the apoptotic cell population in both 786-O and A498 cells following pre-treatment with GEF and subsequent TRAIL administration (
Figure 3A). Treatment with GEF or TRAIL alone marginally increased apoptosis. Notably, the distribution of apoptotic cell populations differed between the two cell lines: 786-O cells exhibited a higher and lower proportion of early and late apoptotic cells than A498 cells, respectively.
The terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay confirmed that GEF amplified TRAIL-induced DNA fragmentation in 786-O and A498 cells. Treatment with TRAIL following GEF pre-treatment significantly increased the number of TUNEL-positive cells in both cell lines (
Figure 3B and 3C). Notably, TRAIL treatment alone noticeably increased the number of TUNEL-positive A498 cells, indicating sensitivity to TRAIL-induced apoptosis. These findings highlight the potential of GEF to enhance TRAIL-induced apoptosis and DNA fragmentation in RCC cells.
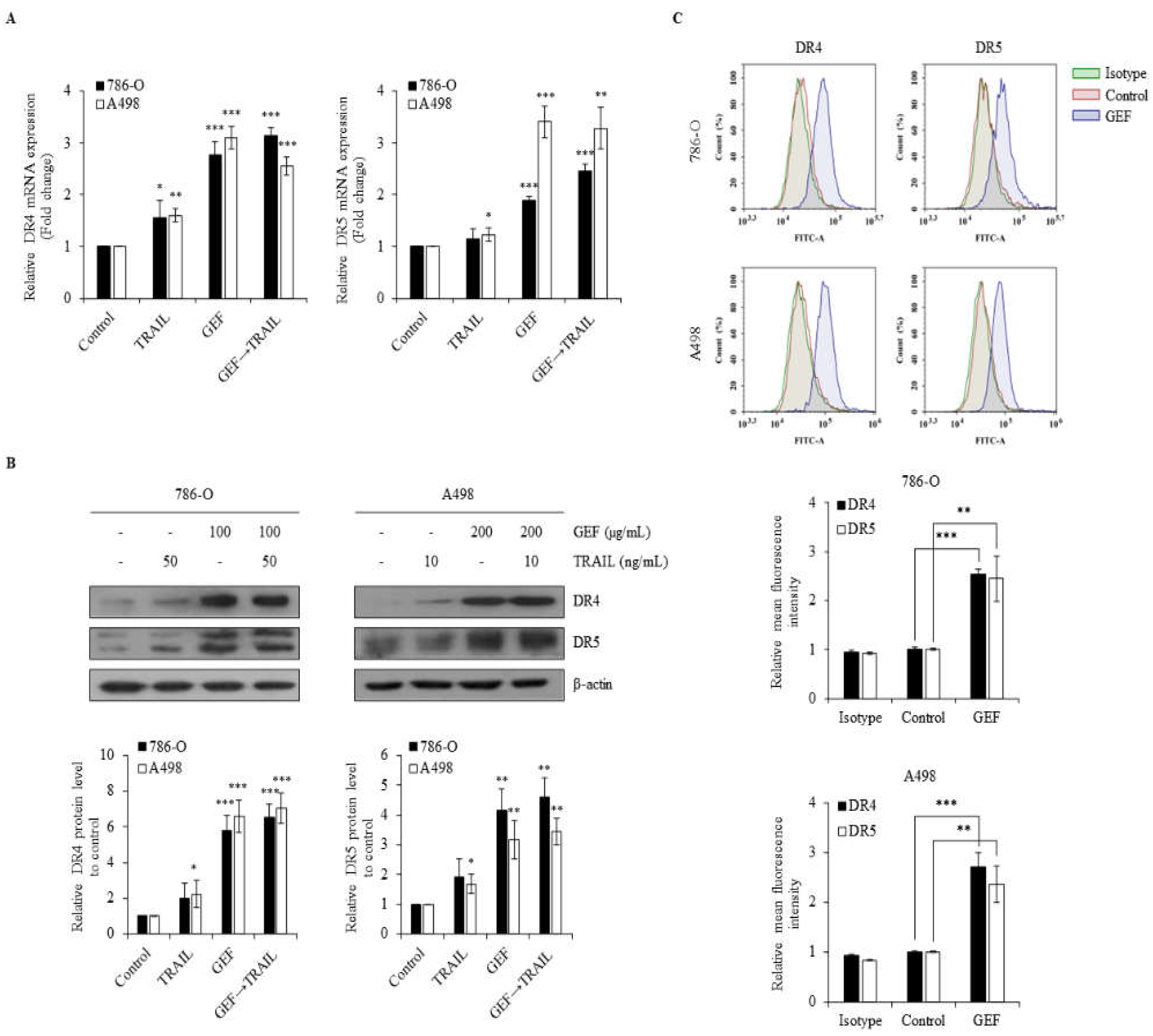
2.4. GEF Upregulates DR4/5 Expression in Human RCC Cells
GEF treatment significantly upregulated DR4/5 mRNA and protein levels in 786-O and A498 cells (
Figure 4A and 4B). Furthermore, cell surface staining revealed a >two-fold increase in DR4 and DR5 expression following GEF treatment compared to that in the control groups (
Figure 4C). These findings confirm that GEF enhances DR4/5 expression in RCC cells.
2.5. Combination of GEF and TRAIL Modulates Apoptotic Protein Expression in Human RCC Cells
Western blot analysis confirmed that the combination of GEF and TRAIL increased cleaved PARP, cleaved caspase-3, and Bax expression and decreased MuD, Bcl-2, and full-length Bid expression in both 786-O and A498 cells (
Figure 5A). Notably, GEF treatment alone decreased MuD expression but increased Bax expression (
Figure 5A).
Immunoprecipitation assays targeting DR4 and DR5 were performed to elucidate their interactions with Fas-associated death domain (FADD) and caspase-8 in 786-O and A498 cells. GEF pre-treatment followed by TRAIL treatment significantly enhanced the interactions of FADD/caspase-8 with DR4 and DR5 (
Figure 5B and 5C). These results suggest that the combination of GEF and TRAIL promotes apoptosis by facilitating the binding of TRAIL to DR4/5, leading to the recruitment of FADD and caspase-8, which are key components of the extrinsic apoptosis pathway in RCC cells. Overall, these findings highlight the potential of GEF and TRAIL combination therapy to induce apoptosis by modulating apoptotic proteins involved in both extrinsic and intrinsic pathways.
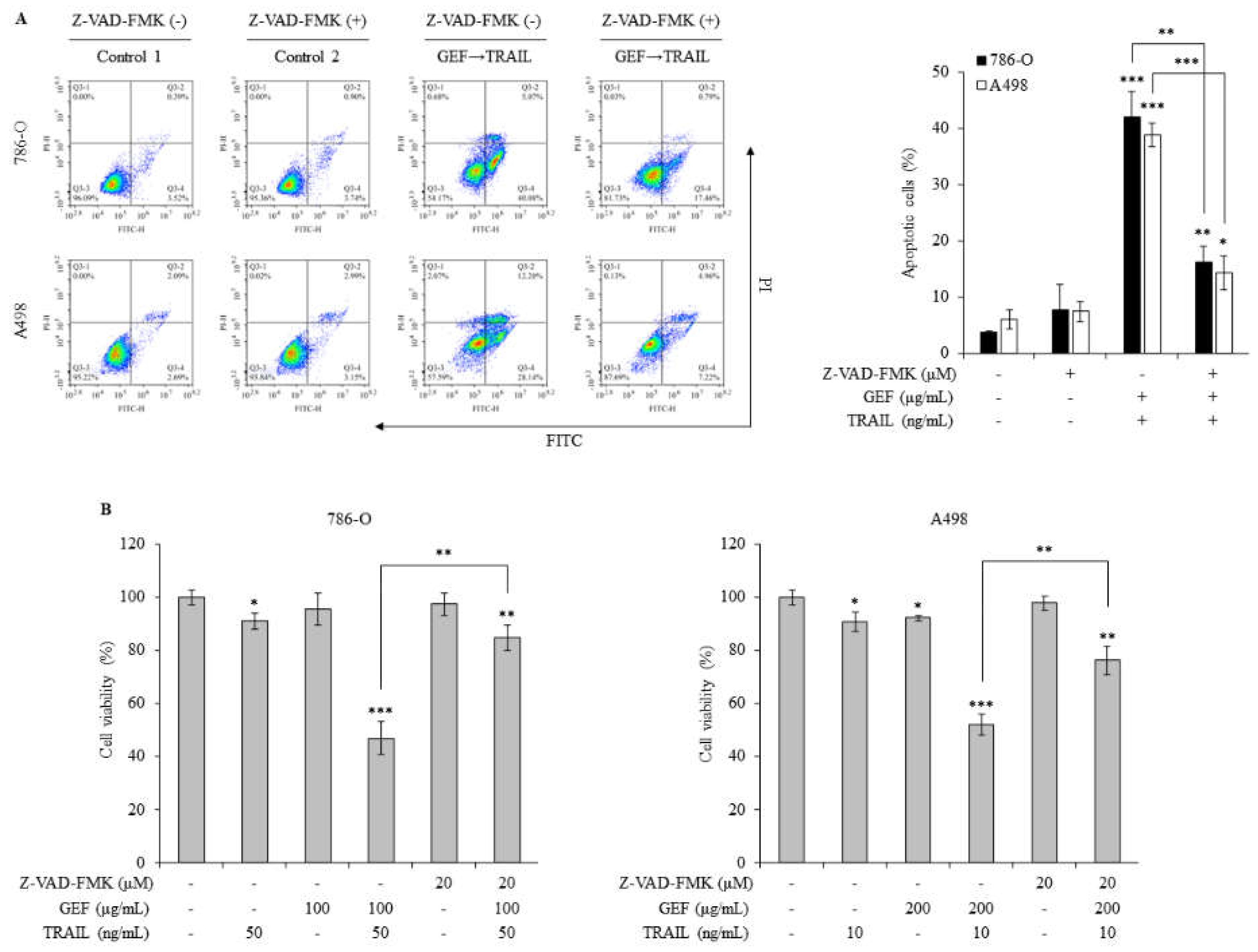
2.6. Apoptosis Induced by GEF/TRAIL in Human RCC Cells Relies on Caspase Activity
Pan-caspase inhibitors were used to explore the mechanism of GEF/TRAIL-induced apoptosis in 786-O and A498 cells. The combination treatment significantly increased the apoptotic cell population compared to that in the controls in both cell lines. However, pre-treatment with Z-VAD-FMK, a pan-caspase inhibitor, neutralized this effect (
Figure 6A), indicating the involvement of caspase activation in apoptosis.
The WST-1 assay demonstrated a significant reduction in cell viability following GEF pre-treatment and TRAIL treatment, which was reversed by Z-VAD-FMK pre-treatment in both cell lines (
Figure 6B). These results highlight the caspase-dependent mechanism underlying GEF/TRAIL-induced apoptosis in human RCC cells.
3. Discussion
We have previously detailed the extraction method and characteristics of the extracted GEF [
15,
25]. To summarize briefly, GEF comprises approximately 7.5% linoleic acid (C18:2), 2.8% palmitic acid (C16:0), and 1.5% oleic acid (C18:1). In addition, GEF contains around 0.2% lysophosphatidic acid (LPA) C18:2, 0.06% LPA C16:0, and 0.02% LPA C18:1. It also includes 0.13% lysophosphatidylinositol, 0.08% lysophosphatidylcholine, and 0.03% lysophosphatidylethanolamine. Furthermore, GEF consists of approximately 1% phosphatidic acid (PA) 16:0-18:2, 0.5% PA 18:2-18:2, and 0.2% PA 16:0-18:1. Our research indicated that LPA C18:2 is a significant active component in gintonin. We have shown that GEF affects neuronal cells via LPA receptor activation [
26] and stimulates insulin secretion through mechanisms that do not involve LPA receptors, implying the presence of other bioactive lipids in GEF besides LPAs [
25].
In this study, we investigated how GEF affects TRAIL-induced apoptosis in human RCC cells. Our results demonstrate that GEF enhances DR4/5 expression and boosts the decreased MuD expression as well as TRAIL-triggered caspase-dependent apoptosis in these cells. This highlights the potential of GEFs in sensitizing RCC cells to TRAIL-induced apoptosis and offers a promising therapeutic strategy. Considering that TRAIL selectively induces apoptosis in cancer cells and the observed resistance in various cancer types, the synergistic effect of GEF and TRAIL holds significant therapeutic promise.
Ginseng-derived GEF exhibits diverse biological effects, including anti-inflammatory and anticancer properties [
17,
19]. Our study revealed the novel role of GEF as a sensitizer to TRAIL-induced apoptosis by upregulating DR4/5, which are essential for the extrinsic apoptosis pathway and TRAIL targeting [
27,
28,
29,
30]. Consistent with previous findings [
27,
28], GEF-induced DR4/5 upregulation enhanced TRAIL-induced apoptosis (
Figure 4). Additionally, combined GEF and TRAIL treatment not only inhibited cell proliferation, colony formation, and migration but also intensified DNA fragmentation (
Figure 1‒3), highlighting its potent apoptotic impact. Notably, the early apoptotic 786-O cell population was larger than the early apoptotic A498 cell population (
Figure 3A), indicating a delayed apoptotic signaling response, possibly due to higher TRAIL resistance in 786-O cells. The involvement of caspase activity, as shown by the effects of the pan-caspase inhibitor, Z-VAD-FMK, further confirmed the caspase-dependent nature of GEF/TRAIL-induced apoptosis (
Figure 6).
Our investigation revealed a decrease in the levels of the anti-apoptotic protein MuD after GEF and TRAIL treatment (
Figure 5), suggesting its potential role in facilitating apoptosis. This interplay between GEF and MuD may offer novel avenues for enhancing TRAIL-based therapies for RCC. Owing to the implications of MuD in apoptotic signaling pathways [
24,
31] and its increased expression in kidney cancer, understanding its regulation may reveal promising therapeutic targets for RCC [
32]. GEF-mediated MuD modulation presents a mechanism through which ginseng-derived compounds influence cell fate determinants, particularly in RCC, where resistance to apoptosis is a significant challenge in treatment strategies.
Our findings extend beyond RCC and highlight the potential use of GEFs in cancer therapy. GEF has diverse pharmacological effects, including anti-inflammatory activity and cancer cell metastasis inhibition. Thus, combining GEF with TRAIL is a promising strategy for TRAIL-resistant cancers [
16,
17,
18,
19]. However, further research should aim to understand the molecular mechanisms underlying the GEF-mediated DR4/5 expression modulation and their impact on MuD function. In addition, a follow-up study is needed to determine the active ingredient in GEF involved in TRAIL-mediated apoptosis. Assessing the in vivo therapeutic efficacy and potential side effects of the GEF/TRAIL combination therapy is crucial. Exploring this approach for other TRAIL-resistant cancers is promising for transformative advancements in oncology.
Our study emphasizes the importance of investigating the effect of natural compounds such as GEF in cancer research. Identifying bioactive constituents from sources, such as ginseng opens new avenues for developing adjunctive cancer therapies as incorporating natural products into conventional treatments holds promise for overcoming drug resistance and improving patient outcomes.
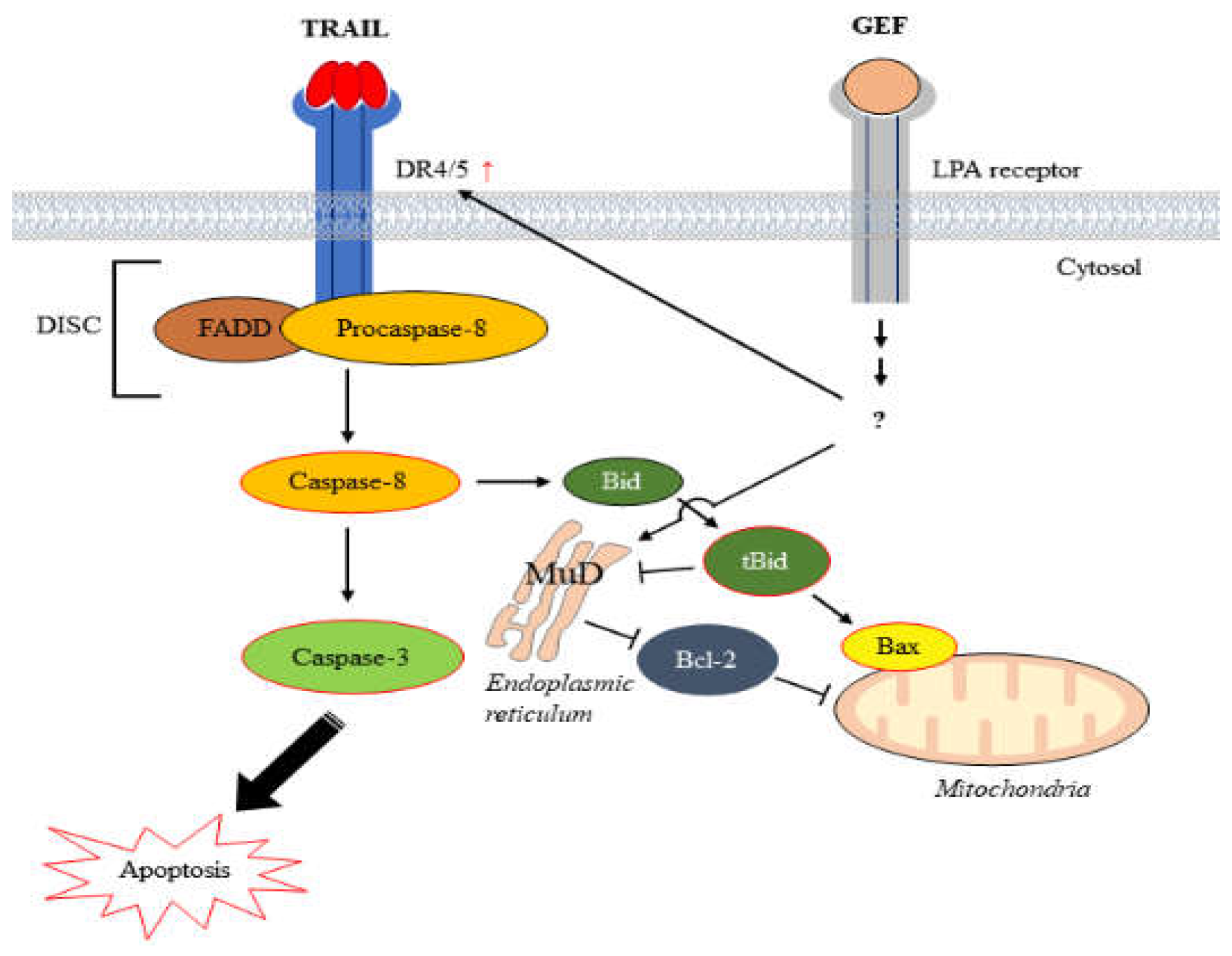
In summary, our study demonstrates that GEF sensitizes human RCC cells to TRAIL-induced apoptosis by upregulating DR4/5 and modulating apoptotic protein expression (
Figure 7). These findings suggest a promising strategy for overcoming TRAIL resistance in cancer therapy and highlight the potential of GEF as a valuable adjunct to TRAIL-based treatments.
4. Materials and Methods
4.1. GEF Preparation
GEF was extracted and lyophilized from
Panax ginseng as described previously [
15]. The dried GEF powder was stored at −80 °C and dissolved in dimethyl sulfoxide (DMSO) prior to cell treatment.
4.2. Cell Culture
Human RCC cells (786-O, A498) from ATCC (Manassas, Virginia, USA) were cultured in RPMI 1640 and DMEM supplemented with 10% FBS, 10,000 U/mL penicillin, and 10,000 μg/mL streptomycin (Welgene, Gyeongsan, Korea) at 37 °C with 5% CO2.
4.3. Cell Viability Assay
The cells (4.0 × 103 cells/well) were seeded in 96-well plates and cultured overnight. After serum deprivation, the cells were treated with various GEF concentrations or DMSO (control) for 12 h. Subsequently, they were exposed to different sTRAIL/Apo2L concentrations (PeproTech, Cranbury, NJ, USA) for another 12 h. Cell viability was assessed with 4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate (WST-1) assay using the EZ-cytox kit (Dogen, Seoul, Korea); the absorbance was measured at 450 nm with a reference wavelength of 600 nm using a SpectraMax M2e microplate reader (Molecular Devices, San Diego, CA, USA).
4.4. Colony Formation Assay
The cells (1.5×105 cells/well) were seeded in 6-well plates overnight, pre-treated with GEF or DMSO for 12 h in serum-free medium, and exposed to TRAIL for another 12 h. The cells were harvested, reseeded (6×102 cells/well) in 6-well plates, and cultured for 10 days. The colonies were fixed with 100% methanol, stained with 0.5% crystal violet, and counted using the ImageJ software (Version 1.8.0, National Institute of Health, Bethesda, MD, USA).
4.5. Wound Healing Assay
The cells (2.5×105 cells/well) were seeded in 6-well plates and allowed to adhere overnight. A scratch was made in the center of each well, followed by washing with a serum-free medium. The cells were then pre-treated with GEF or DMSO for 12 h before treatment with or without TRAIL for another 12 h. Wound images were captured using a light microscope (OLYMPUS CKX41, Tokyo, Japan), and wound size was quantified using ImageJ.
4.6. Apoptosis Assay using FACs
The cells (1.5×105) seeded in 6-well plates were pre-treated with 20 µM Z-VAD-FMK (R&D Systems, Minneapolis, MN, USA) for 1 h before exposure to GEF or DMSO for 12 h. Subsequently, the cells were treated with or without TRAIL for another 12 h. Apoptosis was assessed using the fluorescein isothiocyanate (FITC)–Annexin V Apoptosis Detection Kit (BD Biosciences, San Diego, CA, USA), and flow cytometric analysis was conducted using NovoCyte 1000 with data visualization using the NovoExpress software (version 1.2.5, ACEA Biosciences, San Diego, CA, USA).
4.7. Western Blot Analysis
The cells (2.5×10
5 cells/well in 60-mm dishes) were seeded and allowed to adhere overnight before pre-treating with GEF or DMSO for 12 h in a serum-free medium. Subsequently, the cells were treated with TRAIL for 12 h. After treatment, cell lysates (30 μg) were prepared and subjected to 10–12% SDS–PAGE. Polyvinylidene difluoride (PVDF) membranes (Bio-Rad, Hercules, CA, USA) were used for protein transfer and blocked with 5% skim milk before incubation with primary antibodies against DR4, DR5, β-actin (Santa Cruz Biotechnology, Dallas, TX, USA), Bax, Bcl-2 (Cambridge, MA, USA), PARP, caspase-3, Bid (Cell Signaling Technology, Danvers, MA, USA), and MuD proteins [
33]. After washing, the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies, and the proteins were visualized using an enhanced chemiluminescence kit (Bio-Rad).
4.8. TUNEL Staining
The cells (4.0×103 cells/well) were seeded in 96-well plates and cultured overnight. After incubation with GEF or DMSO for 12 h, the cells were treated with or without TRAIL for another 12 h. After fixation with 4% paraformaldehyde and permeabilization with PBST, the TUNEL assay was performed according to the manufacturer’s instructions (Promega, Madison, WI, USA). The cells were then stained with 4′,6-diamidino-2-phenylindole, visualized using an ECLIPSE Ts2R inverted microscope (Nikon, Melville, NY, USA), and imaged using the NIS-Elements BR software (Nikon).
4.9. Cell Surface Receptor Staining for FACS Analysis
The cells (1.5×105 cells/well) were seeded in 6-well plates and incubated overnight, pre-treated with GEF or DMSO in serum-free medium for 12 h, and then treated with or without TRAIL for another 12 h. After harvesting and washing with DPBS, the cells were incubated with antibodies against DR4 and DR5 for 1 h on ice, incubated with FITC-conjugated secondary antibodies, washed, and analyzed via flow cytometry using a NovoCyte 1000. The data were analyzed using the NovoExpress software (version 1.2.5).
4.10. Real-time Quantitative PCR
Total RNA was extracted using TRIzol reagent (Sigma-Aldrich, St. Louis, MO, USA), and cDNA was synthesized using the AccuPower® RT Premix (Bioneer, Daejeon, Korea). SYBR qPCR Mix (CellSafe, Yongin, Korea) and primers for DR4, DR5, and GAPDH: DR4 (forward) 5′-TGTGACTTTGGTTGTTCCGTTGC-3′ and (reverse) 5′-ACCTGAGCCGATGCAACAACAG-3′; DR5 (forward) 5′-AGACCCTTGTGCTCGTTGTC-3′ and (reverse) 5′-TTGTTGGGTGATCAGAGCAG-3′; GAPDH (forward) 5′-CCCTCAACGACCACTTTGTC-3′ and (reverse) 5′-CCACCACCCTGTTGCTGTA-3′. Real-time PCR was performed on a CFX96 Real-Time System (Bio-Rad).
4.11. Co-immunoprecipitation (Co-IP)
The cell lysates were co-immunoprecipitated with antibodies against DR4 and DR5 using the Dynabeads™ Protein G Immunoprecipitation Kit (Thermo Fisher Scientific, Waltham, MA, USA). The immunoprecipitated proteins were separated by SDS–PAGE, transferred to PVDF membranes, and probed with antibodies against FADD, caspase-8 (Santa Cruz Biotechnology), DR4, and DR5. Immunoreactive bands were visualized using an enhanced chemiluminescence kit (Bio-Rad).
4.12. Statistical Analysis
Each experiment was performed at least three times independently, and results were presented as mean ± SD. Statistical significance was assessed using the Student’s t-test, with P-<0.05, P<0.01, or P<0.001 considered significant.
Author Contributions
Seongwoo Hong: Methodology, Visualization. Formal analysis, Investigation, Data Curation, Writing- Original draft. Rami Lee: Resources, Methodology, Formal analysis. Gyun Seok Park: Formal analysis, Visualization. Sumin Han: Formal analysis, Visualization. Juhyun Shin: Investigation, Validation, Supervision. Yoon-Mi Lee: Formal analysis, Visualization. Seung-Yeol Nah: Conceptualization, Resources, Validation. Jae-Wook Oh: Conceptualization, Methodology, Writing-Review and Editing, Supervision, Project administration, Funding acquisition.
Funding
This paper was supported by Konkuk University in 2024.
Data Availability Statement
Data will be made available on request.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Moch, H.; Cubilla, A.L.; Humphrey, P.A.; Reuter, V.E.; Ulbright, T.M. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part A: Renal, Penile, and Testicular Tumours. Eur Urol 2016, 70, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Shinder, B.M.; Rhee, K.; Farrell, D.; Farber, N.J.; Stein, M.N.; Jang, T.L.; Singer, E.A. Surgical Management of Advanced and Metastatic Renal Cell Carcinoma: A Multidisciplinary Approach. Front Oncol 2017, 7, 107. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O. , et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med 2020, 382, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Li, P.X.; Wong, Y.N.; Armstrong, K.; Haas, N.; Subedi, P.; Davis-Cerone, M.; Doshi, J.A. Survival among patients with advanced renal cell carcinoma in the pretargeted versus targeted therapy eras. Cancer Med-Us 2016, 5, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J Clin 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Johnstone, R.W.; Frew, A.J.; Smyth, M.J. The TRAIL apoptotic pathway in cancer onset, progression and therapy. Nat Rev Cancer 2008, 8, 782–798. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, A. Targeting the extrinsic apoptotic pathway in cancer: lessons learned and future directions. J Clin Invest 2015, 125, 487–489. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, H.N.; Ashkenazi, A. Apo2L/TRAIL and its death and decoy receptors. Cell Death Differ 2003, 10, 66–75. [Google Scholar] [CrossRef]
- Huang, Y.; Sheikh, M.S. TRAIL death receptors and cancer therapeutics. Toxicol Appl Pharmacol 2007, 224, 284–289. [Google Scholar] [CrossRef]
- Smyth, M.J.; Takeda, K.; Hayakawa, Y.; Peschon, J.J.; van den Brink, M.R.; Yagita, H. Nature’s TRAIL--on a path to cancer immunotherapy. Immunity 2003, 18, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Abid, S.; Ahn, J.C.; Mathiyalagan, R.; Kim, Y.J.; Yang, D.C.; Wang, Y. Characteristics of Panax ginseng Cultivars in Korea and China. Molecules 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Nah, S.Y. Gintonin: a novel ginseng-derived ligand that targets G protein- coupled lysophosphatidic acid receptors. Curr Drug Targets 2012, 13, 1659–1664. [Google Scholar] [CrossRef] [PubMed]
- Nah, S.Y.; Kim, D.H.; Rhim, H. Ginsenosides: Are any of them candidates for drugs acting on the central nervous system? Cns Drug Rev 2007, 13, 381–404. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Jung, S.W.; Kim, H.S.; Kim, H.J.; Lee, B.H.; Kim, J.Y.; Kim, J.H.; Hwang, S.H.; Rhim, H.; Kim, H.C. , et al. A brief method for preparation of gintonin-enriched fraction from ginseng. J Ginseng Res 2015, 39, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Choi, S.H.; Lee, N.E.; Cho, H.J.; Rhim, H.; Kim, H.C.; Hwang, S.H.; Nah, S.Y. Effects of Gintonin-Enriched Fraction on Methylmercury-Induced Neurotoxicity and Organ Methylmercury Elimination. Int J Env Res Pub He 2020, 17. [Google Scholar] [CrossRef]
- Chei, S.; Song, J.H.; Oh, H.J.; Lee, K.; Jin, H.; Choi, S.H.; Nah, S.Y.; Lee, B.Y. Gintonin-Enriched Fraction Suppresses Heat Stress-Induced Inflammation through LPA Receptor. Molecules 2020, 25. [Google Scholar] [CrossRef]
- Kim, H.; Lee, B.H.; Choi, S.H.; Kim, H.J.; Jung, S.W.; Hwang, S.H.; Rhim, H.; Kim, H.C.; Cho, I.H.; Nah, S.Y. Gintonin stimulates gliotransmitter release in cortical primary astrocytes. Neurosci Lett 2015, 603, 19–24. [Google Scholar] [CrossRef]
- Hwang, S.H.; Lee, B.H.; Kim, H.J.; Cho, H.J.; Shin, H.C.; Im, K.S.; Choi, S.H.; Shin, T.J.; Lee, S.M.; Nam, S.W. , et al. Suppression of metastasis of intravenously-inoculated B16/F10 melanoma cells by the novel ginseng-derived ingredient, gintonin: Involvement of autotaxin inhibition. Int J Oncol 2013, 42, 317–326. [Google Scholar] [CrossRef]
- Hirst, J.; Barlow, L.D.; Francisco, G.C.; Sahlender, D.A.; Seaman, M.N.J.; Dacks, J.B.; Robinson, M.S. The Fifth Adaptor Protein Complex. Plos Biol 2011, 9. [Google Scholar] [CrossRef]
- Kawasaki, H.; Taira, K. A functional gene discovery in the Fas-mediated pathway to apoptosis by analysis of transiently expressed randomized hybrid-ribozyme libraries. Nucleic Acids Res 2002, 30, 3609–3614. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.R.; Shin, J.N.; Moon, A.R.; Park, S.Y.; Hong, G.; Lee, M.J.; Yun, C.W.; Seol, D.W.; Piya, S.; Bae, J. , et al. A novel protein, MUDENG, induces cell death in cytotoxic T cells. Biochem Biophys Res Commun 2008, 370, 504–508. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.N.; Han, J.H.; Kim, J.Y.; Moon, A.R.; Kim, J.E.; Chang, J.H.; Bae, J.; Oh, J.W.; Kim, T.H. MUDENG is cleaved by caspase-3 during TRAIL-induced cell death. Biochem Bioph Res Co 2013, 435, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Lim, J.B.; Wickramanayake, D.D.; Wagley, Y.; Kim, J.; Lee, H.C.; Seo, H.G.; Kim, T.H.; Oh, J.W. Characterization of MUDENG, a novel anti-apoptotic protein. Oncogenesis 2016, 5. [Google Scholar] [CrossRef]
- Cho, H.J.; Choi, S.H.; Kim, H.J.; Lee, B.H.; Rhim, H.; Kim, H.C.; Hwang, S.H.; Nah, S.Y. Bioactive lipids in gintonin-enriched fraction from ginseng. J Ginseng Res 2019, 43, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Jung, S.W.; Lee, B.H.; Kim, H.J.; Hwang, S.H.; Kim, H.K.; Nah, S.Y. Ginseng pharmacology: a new paradigm based on gintonin-lysophosphatidic acid receptor interactions. Front Pharmacol 2015, 6, 245. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Bi, R.; Guo, H.; Yang, J.; Du, Y.; Wang, C.; Wei, W. Andrographolide Enhances TRAIL-Induced Apoptosis via p53-Mediated Death Receptors Up-Regulation and Suppression of the NF-small ka, CyrillicB Pathway in Bladder Cancer Cells. Int J Biol Sci 2019, 15, 688–700. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.R.; Park, S.H.; Jeong, Y.A.; Na, Y.J.; Kim, J.L.; Jo, M.J.; Jeong, S.; Yun, H.K.; Oh, S.C.; Lee, D.H. RUNX3 enhances TRAIL-induced apoptosis by upregulating DR5 in colorectal cancer. Oncogene 2019, 38, 3903–3918. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Wang, X.; Huang, S.; Xu, Y.; Weng, G.; Yu, R. Alternol Sensitizes Renal Carcinoma Cells to TRAIL-Induced Apoptosis. Front Pharmacol 2021, 12, 560903. [Google Scholar] [CrossRef]
- Kim, B.; Seo, J.H.; Lee, K.Y.; Park, B. Icariin sensitizes human colon cancer cells to TRAIL-induced apoptosis via ERK-mediated upregulation of death receptors. Int J Oncol 2020, 56, 821–834. [Google Scholar] [CrossRef]
- Muthu, M.; Chun, S.; Gopal, J.; Park, G.S.; Nile, A.; Shin, J.; Shin, J.; Kim, T.H.; Oh, J.W. The MUDENG Augmentation: A Genesis in Anti-Cancer Therapy? Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Lv, Y.; Lin, S.Y.; Hu, F.F.; Ye, Z.; Zhang, Q.; Wang, Y.; Guo, A.Y. Landscape of cancer diagnostic biomarkers from specifically expressed genes. Brief Bioinform 2020, 21, 2175–2184. [Google Scholar] [CrossRef]
- Wagley, Y.; Choi, J.H.; Wickramanayake, D.D.; Choi, G.Y.; Kim, C.K.; Kim, T.H.; Oh, J.W. A monoclonal antibody against human MUDENG protein. Monoclon Antib Immunodiagn Immunother 2013, 32, 277–282. [Google Scholar] [CrossRef]
Figure 1.
Gintonin-enriched Panax ginseng extract fraction (GEF) enhances the sensitivity of human renal cell carcinoma (RCC) cells to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). (a, b) 786-O and A498 cells were pre-treated with varying GEF concentrations or DMSO for 12 h, followed by TRAIL treatment with or without GEF for another 12 h. Cell viability was assessed using the WST-1 assay. (c) Morphological changes in cells after 12 h of pre-treatment with GEF followed by TRAIL treatment (40× magnification). Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 1.
Gintonin-enriched Panax ginseng extract fraction (GEF) enhances the sensitivity of human renal cell carcinoma (RCC) cells to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). (a, b) 786-O and A498 cells were pre-treated with varying GEF concentrations or DMSO for 12 h, followed by TRAIL treatment with or without GEF for another 12 h. Cell viability was assessed using the WST-1 assay. (c) Morphological changes in cells after 12 h of pre-treatment with GEF followed by TRAIL treatment (40× magnification). Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 2.
Combination of gintonin-enriched Panax ginseng extract fraction (GEF) and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) suppresses clonogenic formation and migration of human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells were pre-treated with or without GEF for 12 h, followed by TRAIL treatment with or without GEF for an additional 12 h before reseeding. Colony formation results are shown on the left, with the percentage of stained colonies displayed in the graph on the right. (b) and (c) The cells were scratched and pre-treated similarly before wound size measurement. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 2.
Combination of gintonin-enriched Panax ginseng extract fraction (GEF) and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) suppresses clonogenic formation and migration of human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells were pre-treated with or without GEF for 12 h, followed by TRAIL treatment with or without GEF for an additional 12 h before reseeding. Colony formation results are shown on the left, with the percentage of stained colonies displayed in the graph on the right. (b) and (c) The cells were scratched and pre-treated similarly before wound size measurement. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 3.
Combination of GEF and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) triggers apoptosis and DNA fragmentation in human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells were pre-treated with or without GEF for 12 h, followed by TRAIL treatment with or without GEF for another 12 h. Flow cytometry analysis was conducted to assess cell apoptosis. (b, c) Cells underwent similar pre-treatment before terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) staining to observe DNA fragmentation. The proportion of TUNEL-positive cells was quantified. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 3.
Combination of GEF and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) triggers apoptosis and DNA fragmentation in human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells were pre-treated with or without GEF for 12 h, followed by TRAIL treatment with or without GEF for another 12 h. Flow cytometry analysis was conducted to assess cell apoptosis. (b, c) Cells underwent similar pre-treatment before terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) staining to observe DNA fragmentation. The proportion of TUNEL-positive cells was quantified. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 4.
Gintonin-enriched Panax ginseng extract fraction (GEF) elevates death receptors 4 and 5 (DR4/5) expression in human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells underwent 12 h pre-treatment with GEF, followed by 12 h treatment with tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). RT-qPCR was used to analyze DR4 and DR5 mRNA levels. (b) Cells received pre-treatment with GEF for 12 h before exposure to various TRAIL concentrations for another 12 h. Western blot analysis quantified DR4 and DR5 protein levels relative to that in controls. (c) After 24-h treatment with GEF or DMSO, flow cytometry assessed cell surface DR4 and DR5 expression. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 4.
Gintonin-enriched Panax ginseng extract fraction (GEF) elevates death receptors 4 and 5 (DR4/5) expression in human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells underwent 12 h pre-treatment with GEF, followed by 12 h treatment with tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). RT-qPCR was used to analyze DR4 and DR5 mRNA levels. (b) Cells received pre-treatment with GEF for 12 h before exposure to various TRAIL concentrations for another 12 h. Western blot analysis quantified DR4 and DR5 protein levels relative to that in controls. (c) After 24-h treatment with GEF or DMSO, flow cytometry assessed cell surface DR4 and DR5 expression. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 5.
Combination of gintonin-enriched Panax ginseng extract fraction (GEF) and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) regulates the expression of apoptotic proteins. (a) 786-O and A498 cells underwent 12 h pre-treatment with specified GEF concentrations, followed by 12 h treatment with specified TRAIL concentrations. Western blot analysis assessed apoptotic protein expression levels relative to controls. (b, c) Cells received 12 h pre-treatment with specified GEF concentrations before exposure to specified TRAIL concentrations for an additional 12 h. Immunoprecipitation using death receptor 4 (DR4) and DR5 Abs was followed by western blot analysis of Caspase-8 and Fas-associated death domain (FADD). Relative expression levels were compared to those in the untreated control group. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 5.
Combination of gintonin-enriched Panax ginseng extract fraction (GEF) and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) regulates the expression of apoptotic proteins. (a) 786-O and A498 cells underwent 12 h pre-treatment with specified GEF concentrations, followed by 12 h treatment with specified TRAIL concentrations. Western blot analysis assessed apoptotic protein expression levels relative to controls. (b, c) Cells received 12 h pre-treatment with specified GEF concentrations before exposure to specified TRAIL concentrations for an additional 12 h. Immunoprecipitation using death receptor 4 (DR4) and DR5 Abs was followed by western blot analysis of Caspase-8 and Fas-associated death domain (FADD). Relative expression levels were compared to those in the untreated control group. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
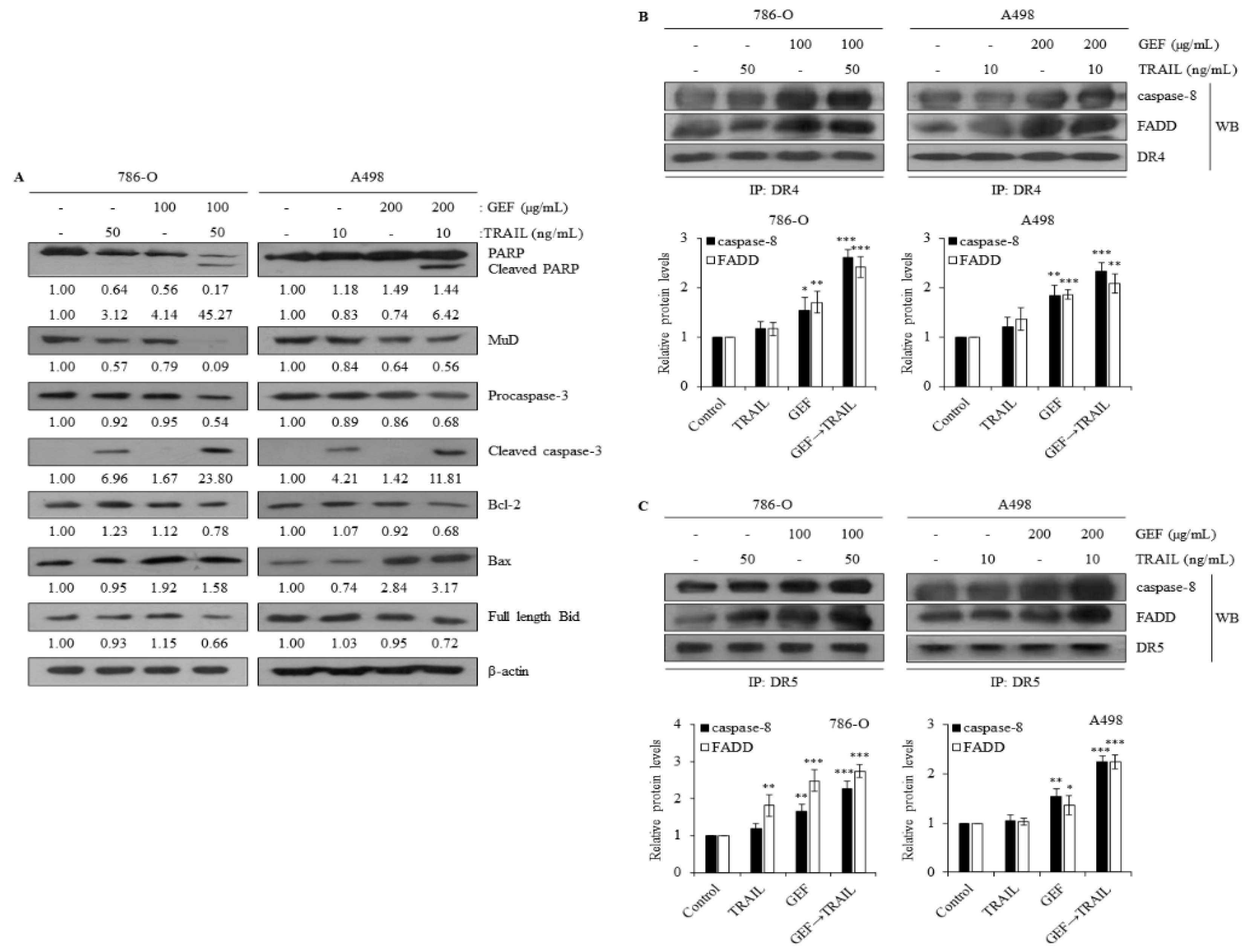
Figure 6.
Gintonin-enriched Panax ginseng extract fraction (GEF)/ tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis relies on caspase activity in human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells were pre-treated with 20 µM Z-VAD-FMK for 1 h, followed by 12 h treatment with or without GEF. Subsequently, the cells received 12 h treatment with or without TRAIL. Cell apoptosis was quantified using flow cytometry. (b) 786-O and A498 cells underwent 1 h pre-treatment with 20 µM Z-VAD-FMK, followed by 12 h treatment with specified GEF concentrations. Subsequent 12 h treatment with specified TRAIL concentrations was performed, and the cell viability was assessed using the WST-1 assay. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
Figure 6.
Gintonin-enriched Panax ginseng extract fraction (GEF)/ tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis relies on caspase activity in human renal cell carcinoma (RCC) cells. (a) 786-O and A498 cells were pre-treated with 20 µM Z-VAD-FMK for 1 h, followed by 12 h treatment with or without GEF. Subsequently, the cells received 12 h treatment with or without TRAIL. Cell apoptosis was quantified using flow cytometry. (b) 786-O and A498 cells underwent 1 h pre-treatment with 20 µM Z-VAD-FMK, followed by 12 h treatment with specified GEF concentrations. Subsequent 12 h treatment with specified TRAIL concentrations was performed, and the cell viability was assessed using the WST-1 assay. Data represent mean ± standard deviation (SD) of three independent experiments, with statistical significance denoted as *P<0.05, **P<0.01, ***P<0.001 compared to the untreated control group.
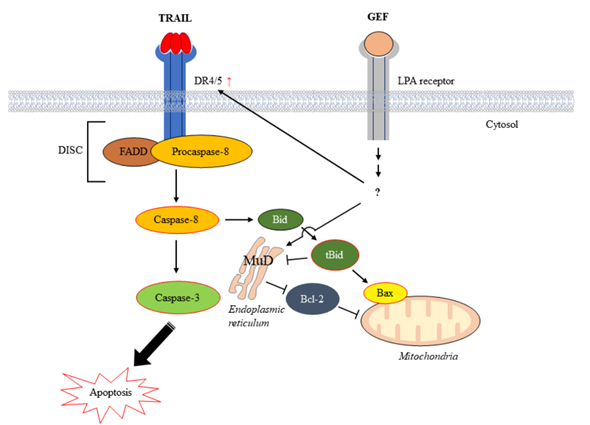
Figure 7.
Schematic representation illustrating the induction of apoptosis by the combined action of gintonin-enriched Panax ginseng extract fraction (GEF) and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL).
Figure 7.
Schematic representation illustrating the induction of apoptosis by the combined action of gintonin-enriched Panax ginseng extract fraction (GEF) and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).