1. Introduction
The intestinal mucosa is responsible for nutrient absorption and barrier function in the gut and is essential for survival [
1]. Nutrients, particularly fiber, have been shown to influence the growth of the intestinal mucosa and improve intestinal functioning. Previous studies employing different fiber sources supplemented to diets in multiple animal species have shown a positive influence on intestinal function by increasing intestinal mass [2-4], mucosal weight [
5], crypt depth [
2,
3], and the expression of tight junction proteins [
6]. In agreement, we have previously shown that mice fed a fiber-deficient diet for 21 days had a loss of intestinal mass, decreased levels of glucagon-like peptide-1 and -2 (GLP-1 and GLP-2) in the colon, and increased intestinal permeability after an extended feeding period of 112 days [
7]. Other studies have reported that fiber-deficient diets are associated with degradation of the colonic mucus barrier and increased pathogen susceptibility [
8,
9]. Taken together, these results suggest that dietary fiber might also be an important factor in the treatment of intestinal diseases characterized by mucosal injury.
Short-chain fatty acids (SCFAs) are a subset of saturated fatty acids with aliphatic chains of less than six carbons. They are predominantly produced by the fermentation of dietary fiber under anaerobic conditions in the human colon [
10,
11]. Acetate (C2), propionate (C3) and butyrate (C4) are the most abundant intestinal SCFAs, with concentrations ranging from 50 to 100 mM in the colonic lumen [
12,
13]. SCFAs are capable of crossing the epithelium [
14,
15] and may enter the systemic circulation via monocarboxylate transporters, resulting in plasma concentrations in the micromolar range [
12]. Butyrate is a preferred nutrient source for colonocytes [
16], and most is thought to be consumed locally. Propionate is metabolized in the liver (although there is great variation between species), decreasing its concentration and making acetate the most abundant SCFA in the circulation [
12]. All SCFAs can trigger downstream signaling cascades by activating G protein-coupled receptors (GPCRs), namely, free fatty acid receptor (FFAR) 2 and FFAR3 [17-19]. These receptors share 40% amino acid sequence similarity and are highly conserved in several species of mammals, including mice [
18,
19]. Both receptors can be activated by two- to five-carbon SCFAs [
17], and several groups have confirmed their expression on GLP-1-secreting L-cells at the mRNA [
20] and protein levels [
21]
SCFAs, particularly propionate, have been shown to induce GLP-1 release in primary cultured colonocytes from humans [
22] and mouse tissue [
20,
23]. Accumulating evidence indicates that the FFAR2 receptor is involved [20,23-25], but the downstream signaling mechanisms of GLP-1 release remain disputed [
23]. Treatment with GLP-1 and GLP-2 analogues has been shown to ameliorate acute small intestinal injury in rodents while the secretion of both hormones is increased during recovery [
26]. Mice with deletion of L-cells fail to recover fully from intestinal damage induced by chemotherapy [
26].
Here, we investigated how the removal of dietary fiber affects the colonic luminal SCFA content, which in turn might alter the activation of the SCFA receptors FFAR2 and FFAR3 and subsequently cause GLP-1 secretion. Furthermore, we hypothesize that reduced GLP-1 secretion, resulting from the loss of SCFA signaling, increases the severity and prolongs recovery from intestinal injury.
2. Materials and Methods
Animals. All the experiments were conducted with permission from the Danish Animal Experiments Inspectorate (licence no. 2018-15-0201-01397). Female C57BL/6JRj mice were purchased from Janvier Laboratories (Saint-Berthevin Cedex, France). The FFAR2/3 double deficient mouse line (Ffar2-/-;Ffar3-/-) (FFAR2/3 KO) was bred in-house and has been described previously [
27]. Heterozygous breeding pairs were used to generate wild-type (Ffar2+/+;Ffar3+/+) (WT) littermates as controls. All mice were housed in individually ventilated cages with 12:12 h light:dark cycles. All mice were acclimatized for one week in the animal facility before experimentation began, and during that time, all mice had ad libitum access to standard chow (Altromin, Lage, Germany, cat.no. 1310) and water.
Fiber-free diet. Before diet assignment, the mice were randomly allocated to feeding groups and placed in new housing cages, with approx. four mice per cage. Mice were fed a standard chow diet (Altromin, Lage, Germany, cat.no. 1310) or were switched to a fiber-deficient diet (Altromin, cat.no. 1013), henceforth referred to as “fiber-free”, for 21 days (
Table 1; for the full nutritional composition, see Supplementary Table 1).
Luminal SCFA measurement. Eight-week-old female mice fed either a fiber-free diet or a chow diet (n=8) for 21 days were euthanized by cervical dislocation. The colon was resected and flushed with 0.5 ml of saline, and the contents were collected and stored at -80°C until further analysis. The luminal contents were manually homogenized, and 50 mg of the homogenate was mixed with 10 μl of 200 mM 3-nitrophenylhydrazine in 50% ethanol and 120 μl of N-(3-dimethylaminopropyl)-N’-ethylcarbodiimide and 6% pyridine in 50% ethanol and incubated for 30 minutes at room temperature. The solution was then diluted to a volume of 100 μl with 10% ethanol, and 100 μl of internal standard was added. The internal standard was prepared beforehand by derivatizing a 50 μl solution containing 20 mM acetic acid, 10 mM propionic acid, 5 mM butyric acid and 1 mg 13C6-3-nitrophenylhydrazine hydrochloride in 50% ethanol with 25 μl 120 mM N-(3-dimethylaminopropyl)-N’-ethylcarbodiimide and 25 μl of 6% pyridine in 50% ethanol. A standard curve of serially diluted external standards was prepared from 0.195 μM to 50 μM, and an assay blank (50% ethanol) was treated the same as the luminal samples. The analytes were randomized, and 5 μl was injected into an ACQUITY UPLC system coupled to a quadrupole time-of-flight mass spectrometer (Waters). The concentrations were determined using QuanLynx software. The analytes were separated on an ACQUITY UPLC BEH C18 VanGuard Precolumn (5 mm length, 2.1 mm internal diameter and 1.7 μm particle size) and an ACQUITY UPLC BEH C18 column (100 mm length, 2.1 mm internal diameter and 1.7 μm particle size) (Waters) at 40°C. The mobile phase was composed of a gradient of acetonitrile ranging from 20-100% containing 0.01% formic acid. The relative standard deviation was 15%, and the LLOQ was defined as the lowest measured concentration of 0.0955 μM after extrapolation from the standard curve.
Mouse colon perfusion. Male and female 20- to 30-week-old FFAR2/3 knockout (KO) (n=4) and WT mice (n=4) were used for colon perfusions. After anaesthesia, a midline incision was made in the abdominal cavity. The arterial supply to the small intestine, cecum, spleen, stomach, and kidneys was ligated. The colon was divided immediately after the caecum and before the entry of the inferior mesenteric artery, leaving a segment for perfusion of an average of 5.3 ± 0.3 cm. Tubing was inserted into the proximal lumen, and prewarmed saline (0.9% NaCl solution) was used to flush the colon. A catheter was placed into the abdominal aorta, and perfusion was started. A drainage catheter was then placed in the vena portae. The mouse was then euthanized by bilateral incisions of the diaphragm. The colon was subsequently perfused in situ using dedicated rodent organ perfusion equipment (Hugo Sachs Elektronik, March-Hugstetten, Germany). The perfusion buffer used was modified Krebs-Ringer bicarbonate buffer (5% dextran T-70, 0.1% bovine serum albumin, 10 µM 3-isobutyl-1-methylxanthine (IBMX), 3.5 mM glucose, and 5 mM pyruvate, fumarate and glutamate). The buffer was heated to 37°C and gassed for the duration of the experiment with 95% O2 and 5% CO2. The experiments were preceded by a 30-minute washing period to stabilize hormone secretion. The vascular perfusion flow rate was maintained at 1.5 ml/min, and the luminal flow rate was 0.025 ml/min. A 100 mM SCFA mixture (52 mM acetate, 30 mM butyrate and 18 mM propionate) was infused luminally for 15 minutes at an initial flow rate of 0.1 ml/min for the first 3 minutes and then at 0.025 ml/min thereafter. Following stimulation, saline was infused at an initial flow rate of 0.1 ml/min for the first 3 minutes, and then 0.025 ml/min for the next 12 minutes. Then, 1 mM SCFA mixture was infused via the vasculature for 10 minutes at a flow rate of 0.075 ml/min. At the end of the experiment, 10 mM bombesin (BSS) was infused as a positive control. Effluent samples were collected by a fraction collector each minute, and the samples were stored on ice and subsequently stored at -20°C until analysis. Total GLP-1 concentrations were measured in colon perfusion samples by radioimmunoassay using antiserum (code name 89390).
DSS-induced colitis. Dextran sulfate sodium (DSS) (molecular weight 40,000) was prepared at a concentration of 3% in drinking water. Eight-week-old female mice fed either a fiber-free diet or a chow diet for 21 days received DSS ad libitum for 4 days and were then provided with regular drinking water until the mice were either euthanized on day 6 (n=8) or day 13 (n=8). Female and male FFAR2/3-/- mice and wild-type littermates, 8 to 12 weeks of age, received 3% DSS ad libitum for 7 days and were then provided with regular drinking water until the mice were either euthanized on day 7 (n=9-12) or day 14 (n=9-14). The control groups (not receiving DSS) received regular drinking water from the same source throughout. Water and food consumption was recorded daily. The general health of the mice was assessed daily by monitoring body weight (BW) and observing clinical progression. To alleviate suffering, mice with severe BW loss were euthanized. The daily disease activity index (DAI) was calculated based on a system scoring BW loss (0 points, <1%; 1 point, 1–5%; 2 points, 5–10%; 3 points, 10–20%; and 4 points, >20%) and stool consistency (0 points, well-formed feces; 2 points, semiformed stools; 4 points, diarrhea; and 6 points, bloody diarrhea).At the end of experiment mice were euthanized by cervical dislocation. The colon was resected, measured, flushed with saline and weighed. A two cm piece of the distal colon was removed, fixed in 10% neutral formalin buffer for 24 h and subsequently transferred to 70% ethanol until further processing for histology.
Histological scoring. Fixed colons were longitudinally embedded in paraffin blocks. The block was trimmed to expose the lumen and subsequently the colonic crypts and sectioned (4 µm). Sections were stained with hematoxylin and eosin. Histological scoring was performed by an experienced histologist using high-quality virtual slides created using Zeiss Axio Scan Z1 slide scanner. The observer was blinded to the origin of the sections. Scoring ranged from 0-6 by adding the tissue damage score (0, no mucosal damage; 1, lymphoepithelial lesions; 2, surface mucosal erosion; and 3, extensive mucosal damage, extension into deeper structure) to the inflammatory cell infiltration score (0, occasional cell infiltrate; 1, increased number of infiltrating cells; 2, inflammatory cells extending to the submucosa; and 3, transmural extension of the inflammatory cells).
Statistics. All the statistical analyses were performed using GraphPad Prism 9 (GraphPad, La Jolla, CA). All the data are expressed as the mean ± SEM. The concentrations of SCFAs were compared by an unpaired t test. The GLP-1 secretion from colon perfusions is presented as the total output (fmol/min; effluent concentration × perfusion flow). In the colon perfusions, to compare treatments, baseline subtracted outputs are presented. Statistical analysis of the treatments was performed by comparing the mean basal output with the mean output during the stimulation using a paired t test. For the basal values, 5 minutes prior to the administration of SCFAs luminally or vascularly were used. For the luminal stimulation, 10 min observations starting 5 min into the infusion were used, for vascular infusions the whole 10 min observation was used. Repeated measures with missing values were analyzed using a mixed-effects analysis followed by a multiple comparisons test. Otherwise, data were compared using a two-way analysis of variance (ANOVA) followed by a multiple comparisons test. Survival curve comparison was created using the Kaplan-Meier method. Differences were considered significant at p<0.05. Number of included animals in each experiments were based on power calculations using endpoints standard deviations known from previous experiments. A power of 80% an alpha of 0.05 was chosen.
3. Results
3.1. The Removal of Dietary Fiber Decreased Colonic Luminal SCFA levels And Was Shown To Stimulate Colonic GLP-1 Secretion
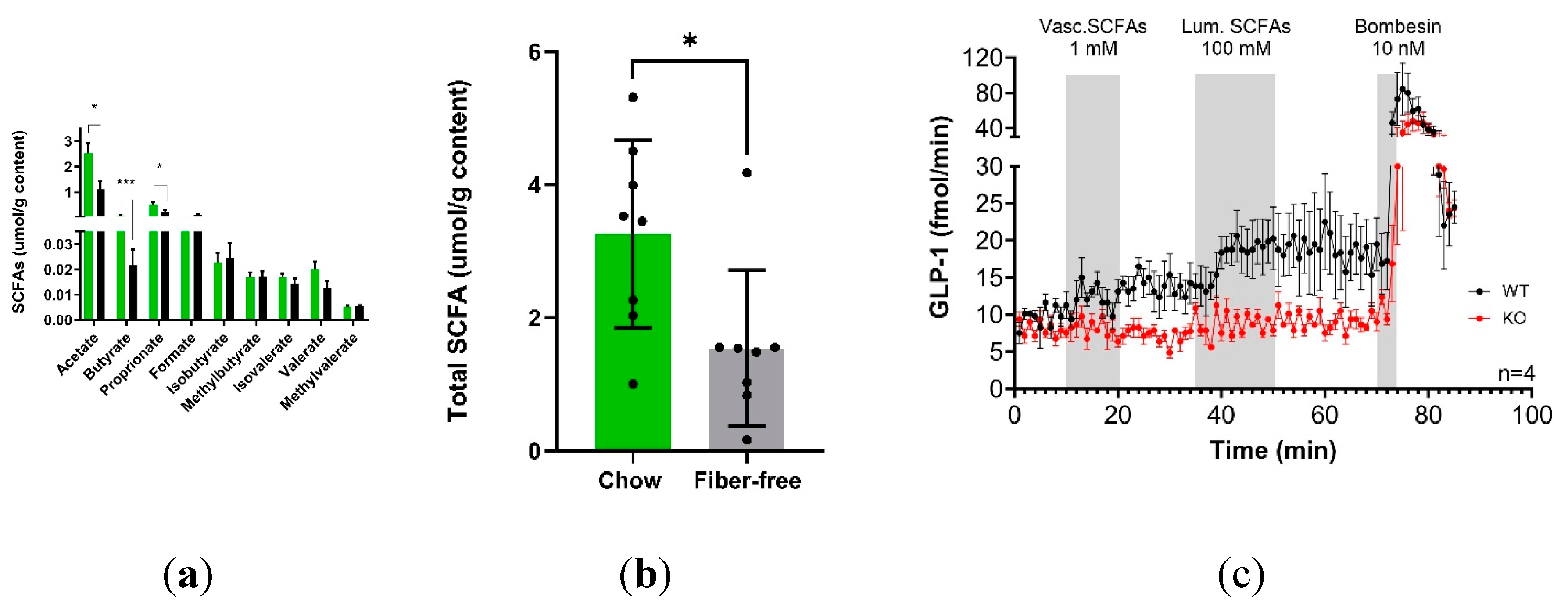
21 days of fiber-free feeding decreased the concentrations of acetate (p<0.05), butyrate (p<0.001) and propionate (p<0.05) in the colonic luminal content (
Figure 1A). The other measured SCFAs, including branch chained SCFA varieties, remained unchanged. Overall, the total measured SCFAs decreased (p<0.05) after fiber-free feeding (
Figure 1B).
The ability of SCFAs and the involvement of FFAR 2 and 3 in the pathway to stimulate colonic GLP-1 secretion was investigated using a mouse perfused colon model and an SCFA mixture (acetate, butyrate and propionate). Luminal SCFA administration (total concentration 100 mM) resulted in increased GLP-1 secretion in WT mice but not in FFAR2/3 KO mice compared to baseline. Vascular infusions of 1 mM did not increase GLP-1 secretion in either WT or KO mice (
Figure 1C).
3.2. The Removal of Dietary Fiber Increased DSS-Induced Colitis Severity
To investigate whether the removal of dietary fiber would impact the severity of DSS induced colitis, mice fed either a fiber-deficient diet or a standard chow diet for 21 days were given 3% DSS in their drinking water for four days. Consistently, throughout the four days of DSS treatment, the chow-fed mice consumed more water than did the fiber-free mice. The chow + DSS mice consumed more drinking water (and therefore more DSS) than did the fiber-free + DSS mice on each of the days (p<0.01) (Supplementary Figure 1A). Compared with chow + water treatment, DSS treatment in the chow + DSS group reduced food intake on days 5 and 6 (p<0.05 and p<0.01) (Supplementary Figure 1B). Similarly, food intake was lower on days 4, 5 and 6 in the fiber-free + DSS group than in the fiber-free + water group (p<0.01, p<0.01 and p<0.01, respectively) (Supplementary Figure 1C).
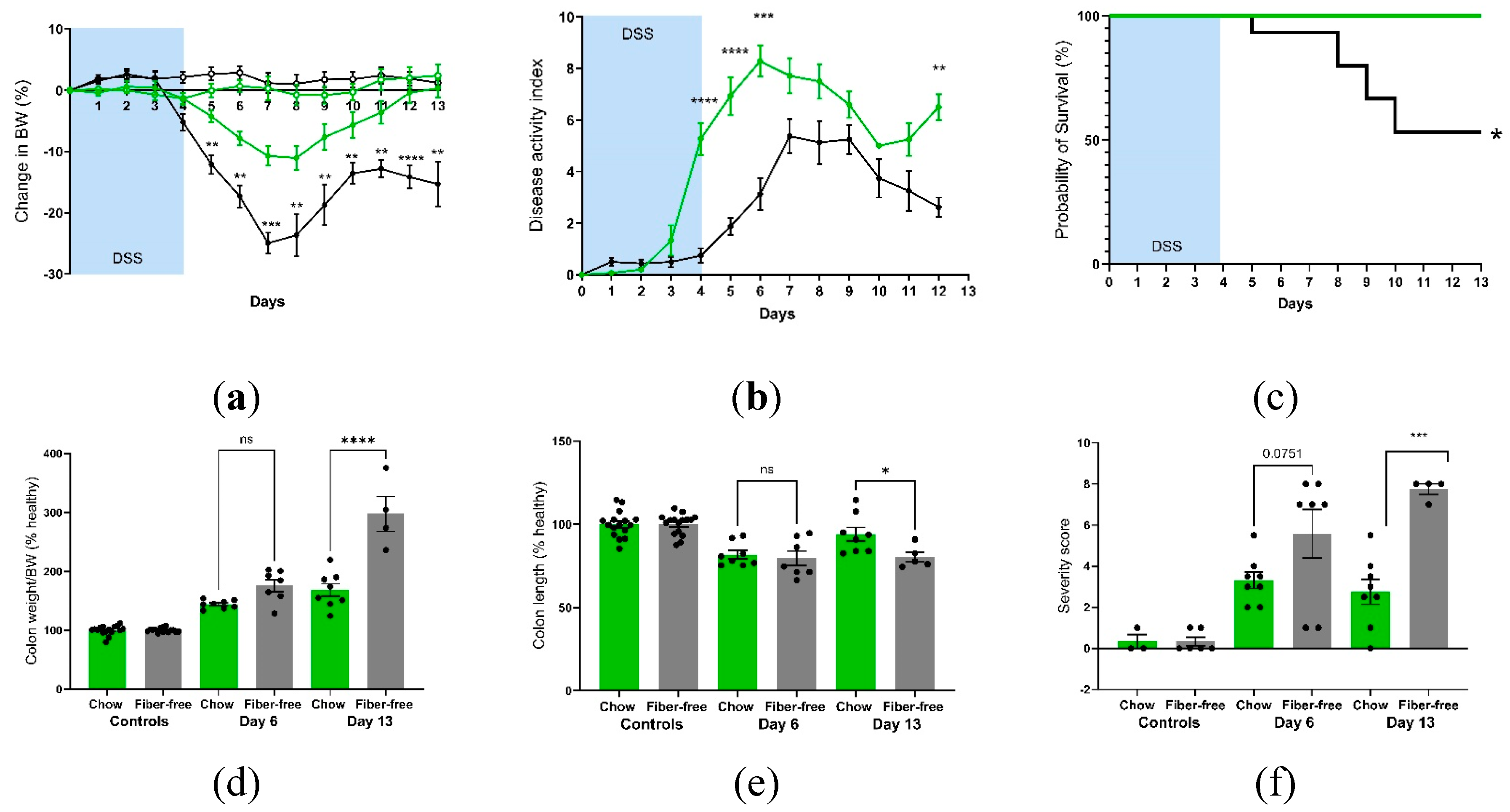
Daily monitoring of BW showed greater decreases immediately after DSS treatment in the fiber-free group than in the chow group; furthermore, these mice were incapable of recovering their BW on day 13, unlike the chow-fed mice (
Figure 2A). The DAI was greater in the fiber-free + DSS group than in the chow + DSS group (
Figure 2B). There were significant differences between day 4-6 (p<0.0001, p<0.0001, p<0.001) and day 12 (p<0.01). Furthermore, four mice from the fiber-free + DSS group had to be sacrificed on days 5, 8, 9 and 10, respectively, due to excessive BW loss. Consequently, mice receiving DSS and the fiber-free diet had a lower percentage of survival than did the chow-fed mice (p<0.05) (
Figure 2C). Fiber-free feeding decreased the colon weight in the healthy control groups (p<0.0001 in the six-day study and p<0.05 in the 13-day study); therefore, the relative colon weight and colon length are shown as percentages of the control. DSS treatment increased the colonic weight in both feeding groups on day six, with no differences between the DSS groups. On day 13, compared with chow-fed mice, fiber-free diet-fed mice continued to have increased colon weights (p<0.0001) (
Figure 2D). DSS treatment shortened the colony length on day six, with no differences between the feeding groups. On day 13, the fiber-free diet-fed mice had shorter colons than did the chow-fed animals (p<0.05) (
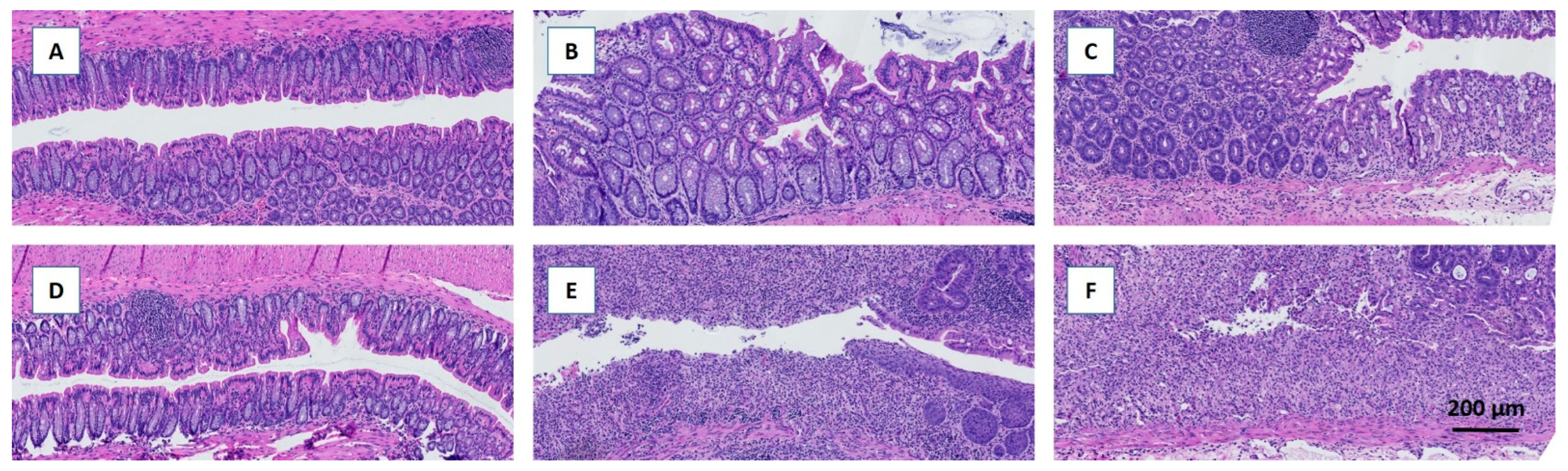
Figure 2E). Colitis severity was also assessed by scoring distal colonic tissue from histological longitudinal sections. Histopathologic analysis indicated severe gut pathology in mice receiving DSS, with a visible increase in inflammatory cell infiltration and surface mucosal erosion in both feeding groups. In agreement with the DAI and BW results, the removal of fiber increased the severity score (p<0.05) on day six and tended to increase the severity score on day 13 (p=0.07) (
Figure 2F and
Figure 3 A-F)
3.3. Attenuation of FFAR2/3 Signaling Did not Affect DSS-Induced Colitis Severity
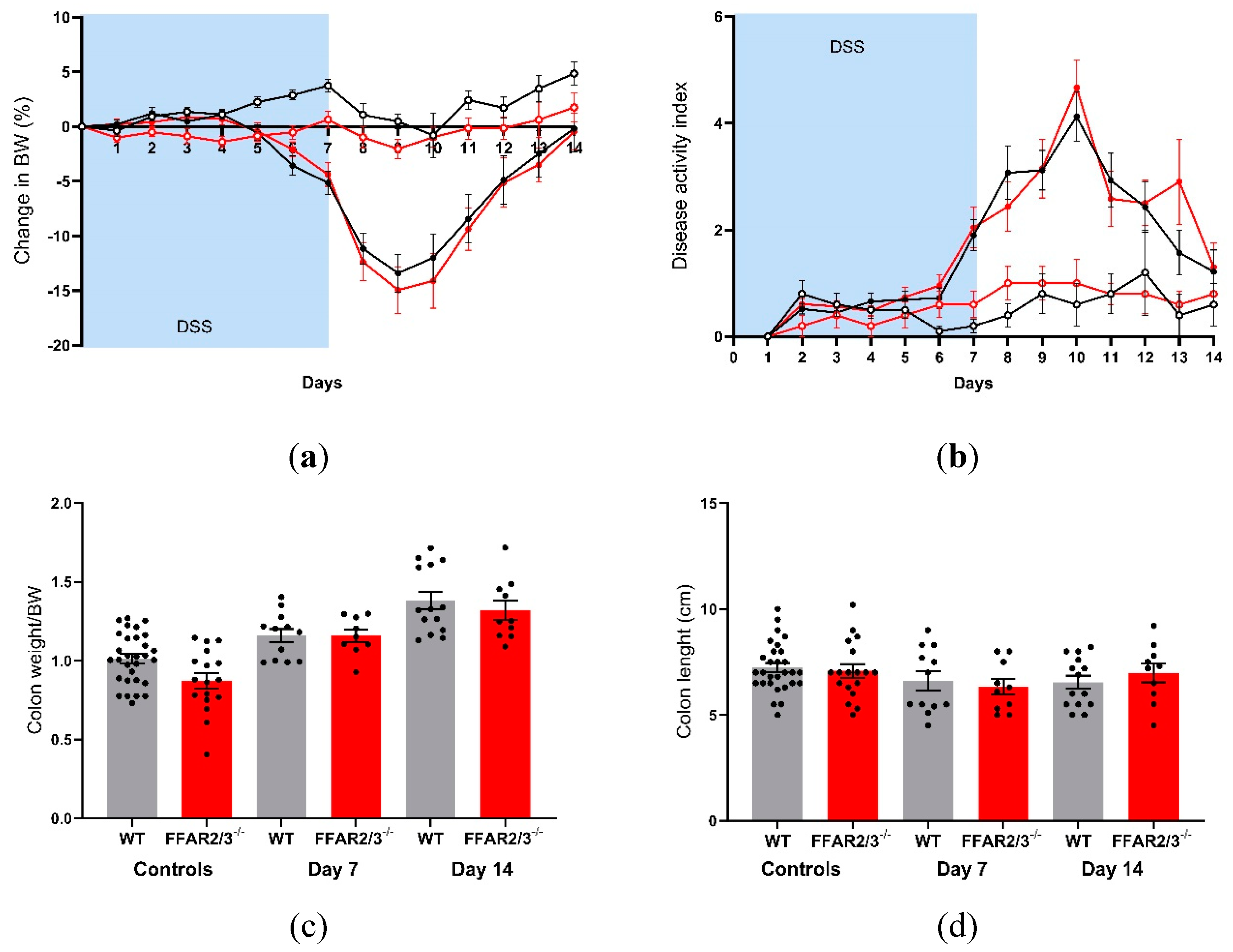
To elucidate the involvement of the SCFA receptors FFAR2 and FFAR3 in DSS-induced colitis, we repeated the DSS experiments in dual FFAR2/3 knockout mice. All animals receiving DSS exhibited a decrease in body weight, but there were no significant differences between the genotypes (
Figure 4A). Furthermore, disease activity did not differ between genotypes (
Figure 4B), and there was no difference in survival (data not shown). Colon weight increased in all DSS groups on day 7 and day 14, but there were no differences between the genotypes (
Figure 4C). Colon length decreased in all DSS groups, again with no differences between genotypes (
Figure 4D).
4. Discussion
In this study, we showed that feeding mice a fiber-deficient diet for 21 days resulted in a significant decrease in the colonic luminal SCFA concentration. Furthermore, the delivery of SCFAs to the colonic lumen at physiological concentrations stimulated GLP-1 secretion. Given these observed outcomes, we hypothesized that a fiber-deficient diet might predispose individuals to increased severity and delayed recovery from intestinal diseases characterized by mucosal injury, where glucagon-like peptides play a pivotal role. We investigated this possibility using a mouse model of DSS-induced colitis, a model that mimics human ulcerative colitis-like pathologies.
SCFAs are recognized as secretagogues of glucagon-like peptides based on multiple experimental approaches, including primary cell culture [
20,
22,
23], isolated colonic perfusion [
28] and human trials [
29]. Given these observations and our own previous research showing that fiber-free diets decrease colonic GLP-1 and GLP-2 content [
7], we speculated that the lowered colonic glucagon-like peptide levels were a consequence of decreased SCFA production. In this study, we observed a decrease in total colonic luminal SCFAs in mice fed a fiber-deficient diet for 21 days. These results support previous observations that low-fiber diets decrease cecal SCFA levels in mice, which corresponds with decreased circulating SCFA levels [
30]. Despite the fermentation of fibers being the largest source of SCFAs, SCFAs can also be produced from amino acid metabolism [
31]. Branched chained SCFAs are the major products of branched-chain amino acid (leucine, isoleucine and valine) fermentation and are increasingly being investigated for their influence on microbiota composition and human health [
32]. Following the removal of fiber, it has been speculated that there could be an increase in proteolytic fermentation [
33]. Here, the removal of fiber did not significantly affect the concentration of the measured branched chained amino acids (isovalerate, methylbutyrate and isobutyrate), suggesting that branched-chain amino acid fermentation was unlikely to increase.
The demonstrated decrease in SCFAs following the removal of fibers may have consequences for the colonic luminal environment, such as decreasing the availability of ligands for the SCFA receptors FFAR2 and FFAR3 and a subsequent decrease in glucagon-like peptide levels. Using an isolated colon perfusion model in mice, we investigated GLP-1 secretion upon the infusion of both luminal and vascular SCFAs. The SCFA mixture infused luminally reflected a physiological dose, while the dose infused vascularly was supraphysiological [
12]. Luminal infusions (100 mM) produced a rapid increase in GLP-1 secretion, but vascular infusions (1 mM) did not elicit a response. These results support previous studies of the isolated perfused colon in rats, where 100 mM luminal infusions of an acetate and butyrate mixture stimulated GLP-1 secretion; however, in these studies, 1 mM vascular fluid was also capable of stimulating GLP-1, unlike in our experiments [
28]. The differences in vascular outcomes may be due to the different ratios of SCFAs in the mixtures or the presence of propionate; however, propionate has been shown to be a strong GLP-1 secretagogue in primary mouse cultures [
20,
23]. The importance of FFAR2 and FFAR3 for SCFA-induced colonic GLP-1 secretion in the rat colon was also evaluated using FFAR2- and FFAR3-specific agonists and an FFAR3 antagonist [
28]. They found that the former did not stimulate GLP-1 secretion and that the latter did not prevent secretion during SCFA infusion, therefore challenging the involvement of these receptors in the GLP-1 response.
Next, after showing how a fiber-free diet can decrease the luminal SCFAs demonstrated to be luminal secretagogues of GLP-1, we investigated how pretreatment with a fiber-deficient diet impacts both the acute and recovery stages of colonic colitis using a robust DSS-induced colitis mouse model. We used a protocol with a low-fiber diet for 21 days before the induction of colitis because previous experiments have shown that there are no differences in intestinal markers such as intestinal weight or morphometry [
7]. As hypothesized, the removal of dietary fiber increased the severity and prolonged the recovery of DSS-induced colitis. In particular, the removal of fiber acutely increased body weight loss and increased daily disease activity scores. In the recovery stage, the removal of fiber also affected colon-specific endpoints, including body weight and disease activity, suggesting the increased importance of fiber in the recovery stage. These results support the observations that supplemented or high-fiber diets ameliorate ulcerative colitis [
34,
35]. The protective effect of dietary fiber in these studies is thought to be at least partly attributable to the increased production of SCFAs. Similarly, butyrate has been shown to promote epithelial barrier function by exerting anti-inflammatory effects [
36], upregulating tight junction proteins [
37] and inducing the production of antimicrobial peptides [
38]. Furthermore, SCFAs play a role in priming the inflammasome response to promote epithelial integrity [
39]. The modulation of cell function by SCFAs may involve inhibiting histone deacetylase activity, thereby altering gene transcription or by binding to the G protein-coupled receptors FFAR 2 and FFAR 3. Therefore, it could be speculated that the increased severity of colitis in the fiber-free mice could be a consequence of decreased SCFA production. This is further supported by the observed lower concentration of fecal SCFAs in inflammatory bowel disease patients than in healthy subjects [
40] and the clinical benefits of SCFA treatment for colitis [41-43].
To investigate the role of FFAR2 and FFAR3 in colitis, we treated dual-receptor FFAR2/3 knockout mice with DSS for 7 days. Dual receptor knockout was used instead of two single knockout mice since both receptors are encoded at a single chromosomal locus in both mice and humans; therefore, the disruption of one receptor may also affect the other, which has been proposed to explain the inconsistencies in the literature regarding the roles of these FFARs [
27]. However, DSS treatment decreased body weight, increased colonic weight and DAI and decreased colonic length independent of FFAR signaling.
Taken together: these results show that the removal of dietary fiber is sufficient to decrease the colonic concentration of SCFAs. This, however, might have consequences for colonic L-cell secretion since in these studies, SCFAs acted as secretagogues of colonic GLP-1. This may have consequences for those consuming low-fiber diets, such as the globally expanding Western diet, who, as a result, may have decreased colonic L-cell secretion, which may be linked to poor metabolic outcomes. Additionally, these results showed that a low-fiber diet predisposes the colon to increased intestinal injury, but this effect was independent of FFAR2 and FFAR3 signaling. We established that SCFA-mediated GLP-1 secretion depends on FFAR 2/3 signaling; therefore, it is unlikely that the low fiber-induced decrease in luminal SCFAs and sensitivity to intestinal disease involves the SCFA-FFAR2/3–GLP-1 pathway. Our results contribute to the understanding of the impact of dietary habits and specific food components on the endocrine function of the colon and may highlight the importance of dietary fiber for the pathobiology of ulcerative colitis, which may help improve clinical outcomes.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org., Table S1: Nutritional comparison of fiber-free diet and chow. Figure S1: Water and diet consumption.
Author Contributions
Conceptualization, Jenna Hunt and Hannelouise Kissow; Methodology, Jenna Hunt, Charlotte Christiansen, Mohammad Yassin, Bolette Hartmann, Stefan Offermanns, Lars Dragsted and Hannelouise Kissow; Supervision, Hannelouise Kissow; Writing – original draft, Jenna Hunt and Hannelouise Kissow; Writing – review & editing, Charlotte Christiansen and Jens Holst.
Funding
This work was supported by the Lundbeck Foundation (Grant No. R263-2017-3740), AP Møller Foundation, Danielsen Foundation, Agnes and Poul Friis Foundation, Dagmar Marshall Foundation, Novo Nordisk Foundation (PRIMA grant, NNF19OC0056246) and Novo Nordisk Foundation Center for Basic Metabolic Research (Grant No. NNF18CC0034900).
Institutional Review Board Statement
All the experiments were conducted with permission from the Danish Animal Experiments Inspectorate (license no. 2018-15-0201-01397 and 2023-15-0201-01408 approved 14th of April 2023). All experiments was carried out in accordance with national and institutional regulations (University of Copenhagen). All animal experiment was carried out to ensure that the experiments complied with commonly accepted 3Rs.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to acknowledge Cecilie Fryland Appeldorff, Department of Nutrition, Exercise and Sports, University of Copenhagen, for her great contribution in measuring the SCFAs.
Conflicts of Interest
MY is affiliated with AbbVie A/S, however this work was performed prior to his employment and is not related to AbbVie A/S. MY is still affiliated with UCPH. The rest of authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Shaw, D.; Gohil, K.; Basson, M.D. Intestinal mucosal atrophy and adaptation. World J Gastroenterol 2012, 18, 6357–6375. [Google Scholar] [CrossRef] [PubMed]
- Adam, C.L.; Williams, P.A.; Garden, K.E.; Thomson, L.M.; Ross, A.W. Dose-dependent effects of a soluble dietary fibre (pectin) on food intake, adiposity, gut hypertrophy and gut satiety hormone secretion in rats. PLoS One 2015, 10, e0115438. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.C.; Kelleher, J.; Losowsky, M.S. The effect of pectin on the structure and function of the rat small intestine. Br J Nutr 1979, 42, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Chassaing, B.; Miles-Brown, J.; Pellizzon, M.; Ulman, E.; Ricci, M.; Zhang, L.; Patterson, A.D.; Vijay-Kumar, M.; Gewirtz, A.T. Lack of soluble fiber drives diet-induced adiposity in mice. Am J Physiol Gastrointest Liver Physiol 2015, 309, G528–G541. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, L.R. Effects of dietary fiber on mucosal growth and cell proliferation in the small intestine of the rat: a comparison of oat bran, pectin, and guar with total fiber deprivation. Am J Clin Nutr 1983, 37, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Mao, X.; He, J.; Yu, B.; Huang, Z.; Yu, J.; Zheng, P.; Chen, D. Dietary fibre affects intestinal mucosal barrier function and regulates intestinal bacteria in weaning piglets. Br J Nutr 2013, 110, 1837–1848. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.E.; Hartmann, B.; Schoonjans, K.; Holst, J.J.; Kissow, H. Dietary Fiber Is Essential to Maintain Intestinal Size, L-Cell Secretion, and Intestinal Integrity in Mice. Front Endocrinol (Lausanne) 2021, 12, 640602. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.S.; Seekatz, A.M.; Koropatkin, N.M.; Kamada, N.; Hickey, C.A.; Wolter, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; et al. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 2016, 167, 1339–1353. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, B.O.; Birchenough, G.M.H.; Stahlman, M.; Arike, L.; Johansson, M.E.V.; Hansson, G.C.; Backhed, F. Bifidobacteria or Fiber Protects against Diet-Induced Microbiota-Mediated Colonic Mucus Deterioration. Cell Host Microbe 2018, 23, 27–40. [Google Scholar] [CrossRef]
- Cummings, J.H.; Macfarlane, G.T. The control and consequences of bacterial fermentation in the human colon. J Appl Bacteriol 1991, 70, 443–459. [Google Scholar] [CrossRef]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Backhed, F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Schwiertz, A.; Taras, D.; Schafer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity (Silver Spring) 2010, 18, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Kaji, I.; Iwanaga, T.; Watanabe, M.; Guth, P.H.; Engel, E.; Kaunitz, J.D.; Akiba, Y. SCFA transport in rat duodenum. Am J Physiol Gastrointest Liver Physiol 2015, 308, G188–G197. [Google Scholar] [CrossRef] [PubMed]
- Ruppin, H.; Bar-Meir, S.; Soergel, K.H.; Wood, C.M.; Schmitt, M.G., Jr. Absorption of short-chain fatty acids by the colon. Gastroenterology 1980, 78, 1500–1507. [Google Scholar] [CrossRef] [PubMed]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.J.; Brummer, R.J. Review article: the role of butyrate on colonic function. Aliment Pharmacol Ther 2008, 27, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem 2003, 278, 11312–11319. [Google Scholar] [CrossRef] [PubMed]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.Y.; Lannoy, V.; Decobecq, M.E.; Brezillon, S.; Dupriez, V.; Vassart, G.; Van Damme, J.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J Biol Chem 2003, 278, 25481–25489. [Google Scholar] [CrossRef]
- Nilsson, N.E.; Kotarsky, K.; Owman, C.; Olde, B. Identification of a free fatty acid receptor, FFA2R, expressed on leukocytes and activated by short-chain fatty acids. Biochem Biophys Res Commun 2003, 303, 1047–1052. [Google Scholar] [CrossRef]
- Tolhurst, G.; Heffron, H.; Lam, Y.S.; Parker, H.E.; Habib, A.M.; Diakogiannaki, E.; Cameron, J.; Grosse, J.; Reimann, F.; Gribble, F.M. Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes 2012, 61, 364–371. [Google Scholar] [CrossRef]
- Nohr, M.K.; Pedersen, M.H.; Gille, A.; Egerod, K.L.; Engelstoft, M.S.; Husted, A.S.; Sichlau, R.M.; Grunddal, K.V.; Poulsen, S.S.; Han, S.; et al. GPR41/FFAR3 and GPR43/FFAR2 as cosensors for short-chain fatty acids in enteroendocrine cells vs FFAR3 in enteric neurons and FFAR2 in enteric leukocytes. Endocrinology 2013, 154, 3552–3564. [Google Scholar] [CrossRef]
- Chambers, E.S.; Viardot, A.; Psichas, A.; Morrison, D.J.; Murphy, K.G.; Zac-Varghese, S.E.; MacDougall, K.; Preston, T.; Tedford, C.; Finlayson, G.S.; et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 2015, 64, 1744–1754. [Google Scholar] [CrossRef] [PubMed]
- Psichas, A.; Sleeth, M.L.; Murphy, K.G.; Brooks, L.; Bewick, G.A.; Hanyaloglu, A.C.; Ghatei, M.A.; Bloom, S.R.; Frost, G. The short chain fatty acid propionate stimulates GLP-1 and PYY secretion via free fatty acid receptor 2 in rodents. Int J Obes (Lond) 2015, 39, 424–429. [Google Scholar] [CrossRef]
- Bolognini, D.; Moss, C.E.; Nilsson, K.; Petersson, A.U.; Donnelly, I.; Sergeev, E.; Konig, G.M.; Kostenis, E.; Kurowska-Stolarska, M.; Miller, A.; et al. A Novel Allosteric Activator of Free Fatty Acid 2 Receptor Displays Unique Gi-functional Bias. J Biol Chem 2016, 291, 18915–18931. [Google Scholar] [CrossRef]
- Brooks, L.; Viardot, A.; Tsakmaki, A.; Stolarczyk, E.; Howard, J.K.; Cani, P.D.; Everard, A.; Sleeth, M.L.; Psichas, A.; Anastasovskaj, J.; et al. Fermentable carbohydrate stimulates FFAR2-dependent colonic PYY cell expansion to increase satiety. Mol Metab 2017, 6, 48–60. [Google Scholar] [CrossRef]
- Hytting-Andreasen, R.; Balk-Møller, E.; Hartmann, B.; Pedersen, J.; Windeløv, J.A.; Holst, J.J.; Kissow, H. Endogenous glucagon-like peptide- 1 and 2 are essential for regeneration after acute intestinal injury in mice. PLoS One 2018, 13, e0198046. [Google Scholar] [CrossRef]
- Tang, C.; Ahmed, K.; Gille, A.; Lu, S.; Grone, H.J.; Tunaru, S.; Offermanns, S. Loss of FFA2 and FFA3 increases insulin secretion and improves glucose tolerance in type 2 diabetes. Nat Med 2015, 21, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, C.B.; Gabe, M.B.N.; Svendsen, B.; Dragsted, L.O.; Rosenkilde, M.M.; Holst, J.J. The impact of short-chain fatty acids on GLP-1 and PYY secretion from the isolated perfused rat colon. Am J Physiol Gastrointest Liver Physiol 2018, 315, G53–G65. [Google Scholar] [CrossRef] [PubMed]
- Freeland, K.R.; Wolever, T.M. Acute effects of intravenous and rectal acetate on glucagon-like peptide-1, peptide YY, ghrelin, adiponectin and tumour necrosis factor-alpha. Br J Nutr 2010, 103, 460–466. [Google Scholar] [CrossRef]
- Trompette, A.; Gollwitzer, E.S.; Yadava, K.; Sichelstiel, A.K.; Sprenger, N.; Ngom-Bru, C.; Blanchard, C.; Junt, T.; Nicod, L.P.; Harris, N.L.; et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med 2014, 20, 159–166. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J. Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 2017, 19, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Rios-Covian, D.; Gonzalez, S.; Nogacka, A.M.; Arboleya, S.; Salazar, N.; Gueimonde, M.; de Los Reyes-Gavilan, C.G. An Overview on Fecal Branched Short-Chain Fatty Acids Along Human Life and as Related With Body Mass Index: Associated Dietary and Anthropometric Factors. Front Microbiol 2020, 11, 973. [Google Scholar] [CrossRef] [PubMed]
- Diether, N.E.; Willing, B.P. Microbial Fermentation of Dietary Protein: An Important Factor in Diet(-)Microbe(-)Host Interaction. Microorganisms 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Panasevich, M.R.; Allen, J.M.; Wallig, M.A.; Woods, J.A.; Dilger, R.N. Moderately Fermentable Potato Fiber Attenuates Signs and Inflammation Associated with Experimental Colitis in Mice. J Nutr 2015, 145, 2781–2788. [Google Scholar] [CrossRef] [PubMed]
- Silveira, A.L.M.; Ferreira, A.V.M.; de Oliveira, M.C.; Rachid, M.A.; da Cunha Sousa, L.F.; Dos Santos Martins, F.; Gomes-Santos, A.C.; Vieira, A.T.; Teixeira, M.M. Preventive rather than therapeutic treatment with high fiber diet attenuates clinical and inflammatory markers of acute and chronic DSS-induced colitis in mice. Eur J Nutr 2017, 56, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Xia, B.; Li, J. Effects of topical treatment of sodium butyrate and 5-aminosalicylic acid on expression of trefoil factor 3, interleukin 1beta, and nuclear factor kappaB in trinitrobenzene sulphonic acid induced colitis in rats. Postgrad Med J 2006, 82, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, B.J.; Kao, D.J.; Kitzenberg, D.A.; Dobrinskikh, E.; Schwisow, K.D.; Masterson, J.C.; Kendrick, A.A.; Kelly, C.J.; Bayless, A.J.; Kominsky, D.J.; et al. HIF-dependent regulation of claudin-1 is central to intestinal epithelial tight junction integrity. Mol Biol Cell 2015, 26, 2252–2262. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, F.; Wu, W.; Sun, M.; Bilotta, A.J.; Yao, S.; Xiao, Y.; Huang, X.; Eaves-Pyles, T.D.; Golovko, G.; et al. GPR43 mediates microbiota metabolite SCFA regulation of antimicrobial peptide expression in intestinal epithelial cells via activation of mTOR and STAT3. Mucosal Immunol 2018, 11, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Macia, L.; Tan, J.; Vieira, A.T.; Leach, K.; Stanley, D.; Luong, S.; Maruya, M.; Ian McKenzie, C.; Hijikata, A.; Wong, C.; et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat Commun 2015, 6, 6734. [Google Scholar] [CrossRef]
- Huda-Faujan, N.; Abdulamir, A.S.; Fatimah, A.B.; Anas, O.M.; Shuhaimi, M.; Yazid, A.M.; Loong, Y.Y. The impact of the level of the intestinal short chain Fatty acids in inflammatory bowel disease patients versus healthy subjects. Open Biochem J 2010, 4, 53–58. [Google Scholar] [CrossRef]
- Breuer, R.I.; Buto, S.K.; Christ, M.L.; Bean, J.; Vernia, P.; Paoluzi, P.; Di Paolo, M.C.; Caprilli, R. Rectal irrigation with short-chain fatty acids for distal ulcerative colitis. Preliminary report. Dig Dis Sci 1991, 36, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Harig, J.M.; Soergel, K.H.; Komorowski, R.A.; Wood, C.M. Treatment of diversion colitis with short-chain-fatty acid irrigation. N Engl J Med 1989, 320, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Kanauchi, O.; Suga, T.; Tochihara, M.; Hibi, T.; Naganuma, M.; Homma, T.; Asakura, H.; Nakano, H.; Takahama, K.; Fujiyama, Y.; et al. Treatment of ulcerative colitis by feeding with germinated barley foodstuff: first report of a multicenter open control trial. J Gastroenterol 2002, 37 Suppl 14, 67–72. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).