Submitted:
05 June 2024
Posted:
06 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
Statistical Analysis
3. Results
4. Discussion
5. Limitations
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Appendix (ACTS questionnaire)
- How much does the possibility of bleeding as a result of anti-clot treatment limit you from taking part in vigorous physical activities? (e.g. exercise, sports, dancing, etc.).
- How much does the possibility of bleeding as a result of anti-clot treatment limit you from taking part in your usual activities? (e.g. work, shopping, housework etc.).
- How bothered are you by the possibility of bruising as a result of anti-clot treatment?
- How bothered are you by having to avoid other medicines (e.g. aspirin) as a result of anti-clot treatment?
- How much does anti-clot treatment limit your diet? (e.g. food or drink, including alcohol).
- How much of a hassle (inconvenience) are the daily aspects of anti-clot treatment? (e.g. remembering to take your medicine at a certain time, taking the correct dose of your medicine, following a diet, limiting alcohol, etc.).
- How much of a hassle (inconvenience) are the occasional aspects of anti-clot treatment? (e.g. the need for blood tests, going to or contacting the clinic/doctor, making arrangements for treatment while travelling etc.).
- 8.
- How difficult is it to follow your anti-clot treatment?
- 9.
- How time-consuming is your anti-clot treatment?
- 10.
- How much do you worry about your anti-clot treatment?
- 11.
- How frustrating is your anti-clot treatment?
- 12.
- How much of a burden is your anti-clot treatment?
- 13.
- Overall, how much of a negative impact has your anti-clot treatment had on your life?
- 14.
- How confident are you that your anti-clot treatment will protect your health? (e.g. prevent blood clots, stroke, heart attack, DVT, embolism)
- 15.
- How reassured do you feel because of your anti-clot treatment?
- 16.
- How satisfied are you with your anti-clot treatment?
- 17.
- Overall, how much of a positive impact has your anti-clot treatment had on your life?
References
- Chugh SS, Havmoeller R, Narayanan K, Singh D, Rienstra M, Benjamin EJ, Gillum RF, Kim YH, McAnulty JH Jr, Zheng ZJ, Forouzanfar MH, Naghavi M, Mensah GA, Ezzati M, Murray CJ. Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation 2014;129:837–847.
- Andersson T, Magnuson A, Bryngelsson IL, Frobert O, Henriksson KM, Edvardsson N, Poci D. All-cause mortality in 272,186 patients hospitalized with incident atrial fibrillation 1995-2008: a Swedish nationwide long-term casecontrol study. Eur Heart J 2013;34:1061–1067.
- Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation as an independent risk factor.
- for stroke: the Framingham Study. Stroke 1991;22:983–988.
- Hart RG, Pearce LA, Aguilar MI. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med 2007; 146:857–867.
- Ruff CT, Giugliano RP, Braunwald E, Hoffman EB, Deenadayalu N, Ezekowitz MD,.
- Camm AJ, Weitz JI, Lewis BS, Parkhomenko A, Yamashita T, Antman EM. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet 2014;383:.
- 955–962.
- Dorian P, Jung W, Newman D, Paquette M, Wood K, Ayers GM, Camm J, Akhtar M, Luderitz B. The impairment of health-related quality of life in patients with intermittent atrial fibrillation: implications for the assessment of investigational therapy. J Am Coll Cardiol 2000;36:1303–1309.
- Thrall G, Lane D, Carroll D, Lip GY. Quality of life in patients with atrial fibrillation: a systematic review. Am J Med 2006;119:448.e1–19.
- Steg PG, Alam S, Chiang CE, Gamra H, Goethals M, Inoue H, Krapf L, Lewalter T, Merioua I, Murin J, Naditch-Brule L, Ponikowski P, Rosenqvist M, Silva-Cardoso J, Zharinov O, Brette S, Neill JO, RealiseAF investigators. Symptoms, functional status and quality of life in patients with controlled and uncontrolled atrial fibrillation: data from the RealiseAF cross-sectional international registry. Heart 2012;98: 195–201.
- Tiffany C. Randolph, DaJuanicia N. Simon, Laine Thomas, Larry A. Allen, Gregg C. Fonarow, Bernard J. Gersh, Peter R. Kowey, James A. Reiffel, Gerald V. Naccarelli, Paul S. Chan, John A. Spertus, Eric D. Peterson, Jonathan P. Piccini, and on behalf of the ORBIT AF Investigators and Patients. Patients Factors Associtatted with Quality of Life in Atrial Fibrillation Randolph-Determinants of Quality of Life in Atrial Fibrillation. Am Heart J. 2016;182:135-143.
- Zhuang J, Lu Y, Tang K, PengW, Xu Y. Influence of body mass index on recurrence and quality of life in atrial fibrillation patients after catheter ablation: a meta-analysis and systematic review. Clin Cardiol 2013;36:269–275.
- Javier García Seara, Laila González Melchor, María Vázquez Caamaño, Emilio Fernández-Obanza Windcheid, Miriam Piñeiro Portela, Eva González Babarro, Pilar Cabanas Grandío, Olga Durán Bobín, Óscar Prada Delgado , Juliana Elices Teja, Evaristo Freire, Mario Gutiérrez Feijoo, Óscar Díaz Castro, Javier Muñiz García, Carlos González Juanatey. Rationale, design and basal characteristics of REGUEIFA study (Multicenter Registry of Atrial. REC: CARDIOCLINICS, ahead of print.
- Fibrillation in Galicia).
- Rosalind Rabin and Frank de Charro. EQ-SD: a measure of health status from the EuroQol Group. Annals of Medicine. 2001; 33(5): 337-343.
- Herdman, M, Badía X and Berra S. El EuroQol-5D: una alternativa sencilla para la medición de la calidad de vida relacionada con la salud en atención primaria. Atención primaria. 2001;28(6):425-429.
- Carmen Suárez, Antonio Pose, Manuel Montero-Pérez Barquero, Jaume Roquer, Jaime Gállego, Carles Ráfols, Daniel Cazorla, José Vivanco son behalf of the Grupo de Trabajo Comité Científico Estudio ALADIN. Validation of satisfaction questionnaire ACTS in outpatients with atrial fibrillation treated with oral anticoagulants in Spain. ALADIN Study. Med Clin (Barc). 2016;147(5):192-198.
- Arribas F, Ormaetxe JM, Peinado R, Perulero N, Ramirez P, Badia X. Validation of.
- the AF-QoL, a disease-specific quality of life questionnaire for patients with atrial fibrillation. Europace 2010;12:364–370.
- Spertus J, Dorian P, Bubien R, Lewis S, Godejohn D, Reynolds MR, Lakkireddy DR, Wimmer AP, Bhandari A, Burk C. Development and validation of the Atrial Fibrillation Effect on QualiTy-of-Life (AFEQT) Questionnaire in patients with atrial fibrillation. Circ Arrhythm Electrophysiol 2011;4:15–25.
- Herdman M, Gudex C, Lloyd A, Janssen M, Kind P, Parkin D, Bonsel G, Badia X. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res 2011;20:1727–1736.
- Linn Arvidsson Strømnes, Helene Ree, Knut Gjesdal, Inger Ariansen. Sex Differences in Quality of Life in Patients With Atrial Fibrillation:A Systematic Review. J Am Heart Assoc. 2019;8:e010992.
- Kirchhof P, Auricchio A, Bax J, Crijns H, Camm J, Diener HC, Goette A, Hindricks G, Hohnloser S, Kappenberger L, Kuck KH, Lip GY, Olsson B, Meinertz T, Priori S, Ravens U, Steinbeck G, Svernhage E, Tijssen J, Vincent A, Breithardt G. Outcome parameters for trials in atrial fibrillation: executive summary. Eur Heart J 2007;28:2803–2817.
- Gareth J. Wynn, Derick M. Todd, MatthewWebber, Laura Bonnett, James McShane, Paulus Kirchhof, and Dhiraj Gupta. The European Heart Rhythm Association.
- symptom classification for atrial fibrillation: validation and improvement through a simple modification. Europace. 2014; 16, 965–972.
- Justin B. Echouffo-Tcheugui, Peter Shrader, Laine Thomas, Bernard J. Gersh, Peter R. Kowey, Kenneth W. Mahaffey, Daniel E. Singer, Elaine M. Hylek, Alan S. Go, Eric D. Peterson, Jonathan P. Piccini, Gregg C. Fonaro. Care Patterns and Outcomes inAtrial Fibrillation Patients With andWithout DiabetesORBIT-AF Registry. JAm Coll Cardiol 2017;70:1325–35.
- Informe de posicionamiento terapéutico UT_ACOD/V5/21112016. Criterios y recomendaciones generales para el uso de los anticoagulantes orales directos (ACOD) en la prevención del ictus y la embolia sistémica en pacientes con fibrilación auricular no valvular. Fecha de publicación 21 de noviembre de 2016. https://www.aemps.gob.es/medicamentosUsoHumano/informesPublicos/docs/criterios-anticoagulantes-orales.pdf.
- Coleman CI, Haas S, Turpie AG, Kuhls S, Hess S, Evers T, Amarenco P, KirchhofP, Camm AJ; XANTUS Investigators. Impact of switching from a vitamin Kantagonist to rivaroxaban on satisfaction with anticoagulation therapy: theXANTUS-ACTS substudy.Clin Cardiol. 2016;39:565–569.
- Contreras Muruaga MD, Vivancos J, Reig G, Gonzalez A, Cardona P, Ramirez-Moreno JM, Marti J, Fernandez CS; ALADIN Study Investigators. Satisfaction,quality of life and perception of patients regarding burdens and benefits ofvitamin K antagonists compared with direct oral anticoagulants in patientswith nonvalvular atrialfibrillation.J Comp Eff Res. 2017;6:303–312.
- Ute I. Schwarz, Marylyn D. Ritchie, Yuki Bradford, Chun Li, ScottM. Dudek, Amy Frye-Anderson, Richard B. Kim, Dan M. Roden, and C. Michael Stein. Genetic determinants of response to warfarin during initial anticoagulation. N Engl J Med 2008;358:999–1008.
- Kenneth A Bauer. Pros and cons of new oral anticoagulants. Hematology Am Soc Hematol Educ Program 2013;2013:464–470.
| Variables | Total (n=904) | VKA (n=532) | DOAC (n=372) | p-value |
|---|---|---|---|---|
| Age, mean ± SD | 68.36 ± 11.67 | 71.69 ± 10,35 | 63.61 ± 11.81 | <0.001 |
| Women, % | 33.96 | 37.03 | 29.57 | 0.020 |
| Renal failure, % | 6.86 | 10.34 | 1.88 | <0.001 |
| PAD, % | 2.99 | 4.14 | 1.34 | 0.016 |
| CAD, % | 12.17 | 14.29 | 9.14 | 0.020 |
| Dementia, % | 0.66 | 1.13 | 0.00 | 0.046 |
| OSAHS, % | 4.87 | 4.89 | 4.84 | 0.973 |
| COPD, % | 11.28 | 13.35 | 8.33 | 0.019 |
| Anaemia, % | 3.98 | 5.45 | 1.88 | 0.009 |
| Diabetes, % | 19.91 | 22.74 | 15.86 | 0.011 |
| Hyperlipidaemia, % | 50.00 | 53.57 | 44.89 | 0.010 |
| Hypertension, % | 66.37 | 72.93 | 56.99 | <0.001 |
| Obesity (BMI≥30), % | 42.29 | 41.24 | 43.78 | 0.448 |
| CHA2DS2-VASc score, mean±SD | 2.48 ± 1.48 | 2.87 ± 1.4 | 1.93 ± 1.43 | <0.001 |
| HAS-BLED score, mean ± SD | 0.73 ± 0.77 | 0.91 ± 0.78 | 0.47 ± 0.66 | <0.001 |
| EHRA ≥ II, % | 66.48 | 65.41 | 68.01 | 0.416 |
| AF classification | <0.001 | |||
| First diagnosed episode, % | 23.12 | 25.56 | 19.62 | |
| Paroxysmal, % | 20.24 | 17.86 | 23.66 | |
| Persistent, % | 27,43 | 21.80 | 35.48 | |
| Long standing persistent, % | 2.54 | 2.26 | 2.96 | |
| Permanent, % | 26.66 | 32.52 | 18.28 | |
| EF <50%, % | 18.30 | 19.55 | 16.57 | 0.268 |
| Valvular heart disease, % | 17.26 | 21.05 | 11.83 | <0.001 |
| Beta blockers, % | 68.47 | 65.98 | 72.04 | 0.053 |
| Calcium channel blockers, % | 5.64 | 6.02 | 5.11 | 0.561 |
| Digoxin, % | 6.64 | 7.89 | 4.84 | 0.069 |
| ACE inhibitors / ARBs, % | 54,20 | 54,89 | 53,23 | 0,622 |
| Diuretics, % | 36,06 | 42,67 | 26,61 | <0,001 |
| Antiarrhythmic drugs, % | 38,83 | 28,01 | 54,30 | <0,001 |
| Total (n=846) | VKA (n=490) | DOAC (n=356) | p-value | |
|---|---|---|---|---|
| Mobility | ||||
| Patients with some problems (%) | 31.91 | 37.55 | 24.16 | <0.001 |
| Patients with extreme problems (%) | 0.47 | 0.41 | 0.56 | 1.000 |
| Self-care | ||||
| Patients with some problems (%) | 7.92 | 8.78 | 6.74 | 0.279 |
| Patients with extreme problems (%) | 0.71 | 0.61 | 0.84 | 0.700 |
| Usual activities | ||||
| Patients with some problems (%) | 21.99 | 24.69 | 18.26 | 0.026 |
| Patients with extreme problems (%) | 1.89 | 2.04 | 1.69 | 0.802 |
| Pain / Discomfort | ||||
| Patients with some problems (%) | 28.61 | 31.84 | 24.16 | 0.015 |
| Patients with extreme problems (%) | 2.72 | 3.47 | 1.69 | 0.136 |
| Anxiety / depression | ||||
| Patients with some problems (%) | 26.12 | 26.53 | 25.56 | 0.751 |
| Patients with extreme problems (%) | 3.19 | 3.88 | 2.25 | 0.235 |
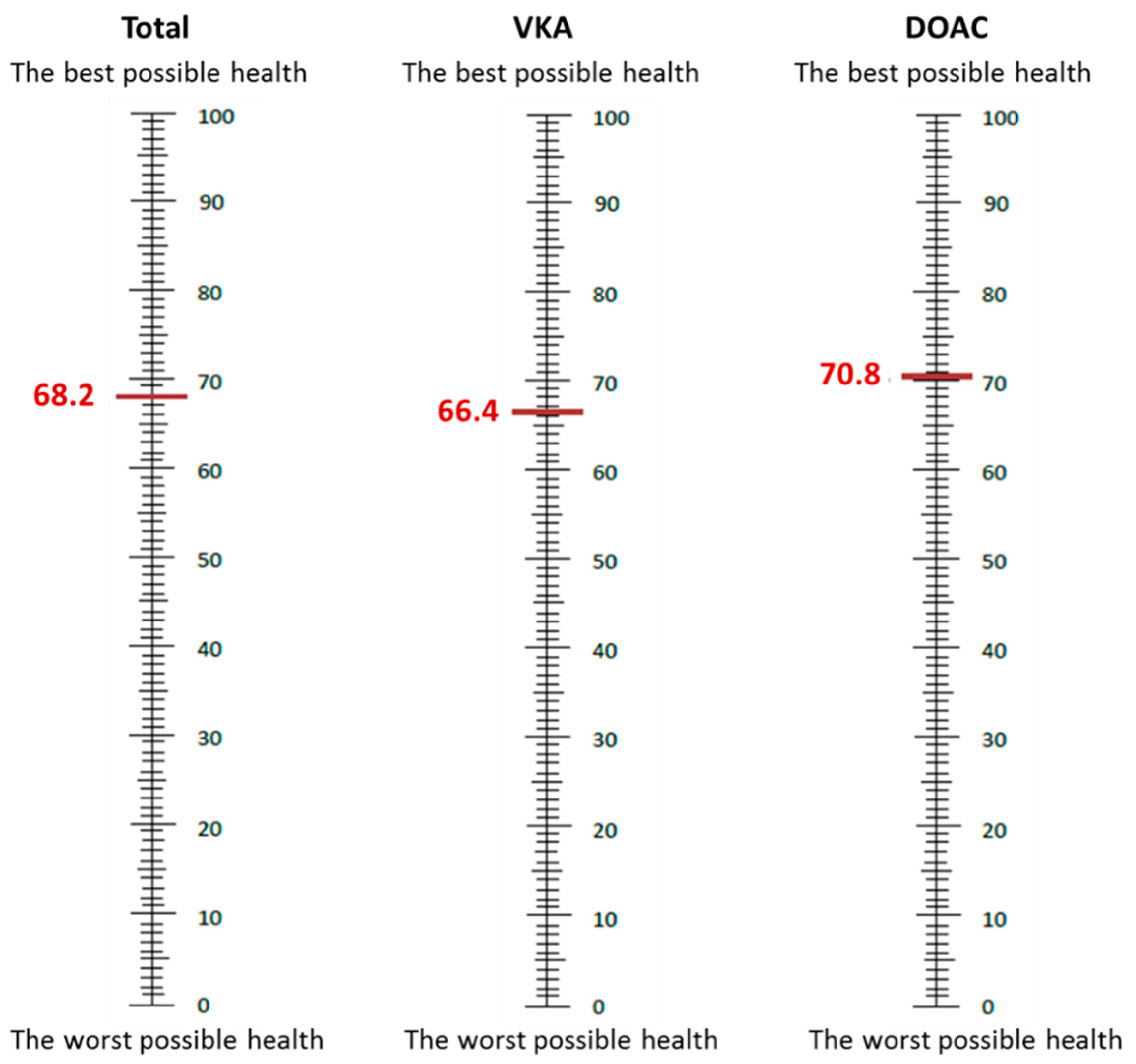
| Visual Analog Scale (VAS), mean ± SD | 68.2 ± 16.1 | 66.35 ± 16.21 | 70.75 ± 15.62 | <0.001 |
| EQ-5D index, mean ± SD | 0.82 ± 0.21 | 0.79 ± 0.21 | 0.85 ± 0.2 | <0.001 |
| Patients with health status 11111 (%) | 46.10 | 39.39 | 55.34 | <0.001 |
| Mobility | ||||
|---|---|---|---|---|
| Final model | ||||
| OR | CI 95% | P value | ||
| OAC (DOAC) | 0.99 | 0.69 | 1.42 | 0.959 |
| Age | 1.04 | 1.02 | 1.07 | <0.001 |
| Female | 2.46 | 1.71 | 3.55 | <0.001 |
| CAD | 1.84 | 1.13 | 2.98 | 0.014 |
| COPD | 1.91 | 1.15 | 3.20 | 0.013 |
| CHA2DSVASc score | 1.2 | 1 | 1.43 | 0.044 |
| HASBLED score | 1.2 | 1 | 1.43 | 0.006 |
| EHRA scale ≥2 | 1.75 | 1.20 | 2.56 | 0.004 |
| AF type | ||||
| Paroxysmal | 1.41 | 0.82 | 2.42 | 0.212 |
| Persistent | 2.42 | 1.42 | 4.14 | 0.001 |
| Long-standing persistent | 2.43 | 0.84 | 7.06 | 0.102 |
| Permanent | 1.59 | 0.96 | 2.63 | 0.069 |
| Diuretics | 1.57 | 1.09 | 2.27 | 0.015 |
| Usual Activity | ||||
| Final model | ||||
| OR | CI 95% | P value | ||
| OAC (DOAC) | 0.99 | 0.68 | 1.44 | 0.966 |
| Female | 2.01 | 1.41 | 2.88 | <0.001 |
| CAD | 2.05 | 1.27 | 2.31 | 0.003 |
| HASBLED score | 1.70 | 1.32 | 2.19 | <0.001 |
| EHRA scale 3-4 | 4.27 | 2.40 | 7.62 | <0.001 |
| AF type | ||||
| Paroxysmal | 2.70 | 1.48 | 4.93 | 0.001 |
| Persistent | 2.81 | 1.54 | 5.11 | 0.001 |
| Long-standing persistent | 2.04 | 0.63 | 6.55 | 0.233 |
| Permanent | 2.80 | 1.59 | 4.94 | <0.001 |
| Diuretics | 1.77 | 1.22 | 2.58 | 0.003 |
| Pain/Discomfort | ||||
| Final model | ||||
| OR | CI 95% | P value | ||
| OAC (DOAC) | 0.97 | 0.69 | 1.36 | 0.854 |
| Female | 1.95 | 1.39 | 2.74 | <0.001 |
| CAD | 1.57 | 0.99 | 2.48 | 0.055 |
| CHA2DSVASc score | 1.25 | 1.08 | 1.44 | 0.003 |
| HASBLED score | 1.28 | 1 | 1.68 | 0.047 |
| EHRA scale 3-4 | 2.42 | 1.43 | 4.11 | 0.001 |
| Valvular heart disease | 1.51 | 1.01 | 2.27 | 0.046 |
| EQ-5D index | ||||
|---|---|---|---|---|
| Final model | ||||
| Coefficient | CI 95% | P value | ||
| OAC (DOAC) | 0.009 | -0.019 | 0.036 | 0.534 |
| Female | -0.092 | -0.121 | -0.063 | <0.001 |
| CAD | -0.055 | -0.095 | -0.016 | 0.006 |
| COPD | -0.059 | -0.101 | -0.017 | 0.006 |
| CHA2DSVASc score | -0.014 | -0.026 | -0.001 | 0.029 |
| HASBLED score | -0.040 | -0.061 | -0.019 | <0.001 |
| EHRA scale ≥2 | -0.040 | -0.068 | -0.013 | 0.004 |
| AF type | ||||
| Paroxysmal | -0.026 | -0.065 | 0.013 | 0.185 |
| Persistent | -0.039 | -0.076 | -0.001 | 0.042 |
| Long-standing persistent | -0.041 | -0.123 | -0.042 | 0.336 |
| Permanent | -0.044 | -0.082 | -0.006 | 0.024 |
| Diuretics | -0.039 | -0.068 | -0.009 | 0.011 |
| VAS | ||||
| Final model | ||||
| Coefficient | CI 95% | P value | ||
| OAC (DOAC) | 1.47 | -0.74 | 3.68 | 0.192 |
| Female | -3.39 | -5.73 | -1.05 | 0.005 |
| COPD | -4.31 | -7.68 | -0.93 | 0.012 |
| Anaemia | -5.68 | -11.03 | -0.33 | 0.038 |
| Hyperlipidaemia | -2.91 | -5.03 | -0.79 | 0.007 |
| CHA2DSVASc score | -1.09 | -2 | -0.19 | 0.018 |
| EHRA scale ≥2 | -5.10 | -7.33 | -2.86 | <0.001 |
| Diuretics | -2.80 | -5.18 | -0.41 | 0.021 |
| Antiarrhythmic drug treatment | 2.44 | 0.16 | 4.72 | 0.036 |
| Total (n= 713= | VKA (n= 416) | DOAC (n= 297) | p-value | |
|---|---|---|---|---|
| Burden score, mean ± SD | 53.51 ± 7.96 | 52.09 ± 8.44 | 55.5 ± 6.75 | <0.001 |
| Benefit score, mean ± SD | 11.39 ± 2.51 | 11.13 ± 2.42 | 11.75 ± 2.6 | <0.001 |
| General negative impact, mean ± SD | 1.68 ± 1.01 | 1.77 ± 1.02 | 1.56 ± 0.99 | <0.001 |
| Distribution of scores, % | <0.001 | |||
| 1 | 58.20 | 51.20 | 68.01 | |
| 2 | 26.51 | 31.73 | 19.19 | |
| 3 | 6.59 | 8.65 | 3.70 | |
| 4 | 6.17 | 5.29 | 7.41 | |
| 5 | 2.52 | 3.13 | 1.68 | |
| General positive impact, mean ± SD | 3.65 ± 0.99 | 3.56 ± 0.96 | 3.78 ± 1.02 | <0.001 |
| Distribution of scores, % | <0.001 | |||
| 1 | 3.65 | 3.61 | 3.70 | |
| 2 | 9.96 | 10.34 | 9.43 | |
| 3 | 20.62 | 25.00 | 14.48 | |
| 4 | 49.51 | 49.04 | 50.17 | |
| 5 | 16.27 | 12.02 | 22.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





