Submitted:
20 June 2024
Posted:
21 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plant material
2.2. Crude extracts preparation
2.3. Chemical compounds analysis
2.4. Total phenolic compounds determination
2.5. Gel Filtration Chromatography
2.6. Total Flavonoid content (TFC) determination
2.7. Antioxidant activity determination
2.7.1. Ferric Reducing-Antioxidant Power FRAP
2.7.2. Total antioxidant capacity (TAC)
2.8. Antimicrobial activity
2.9. Statistical analysis
3. Results and discussion
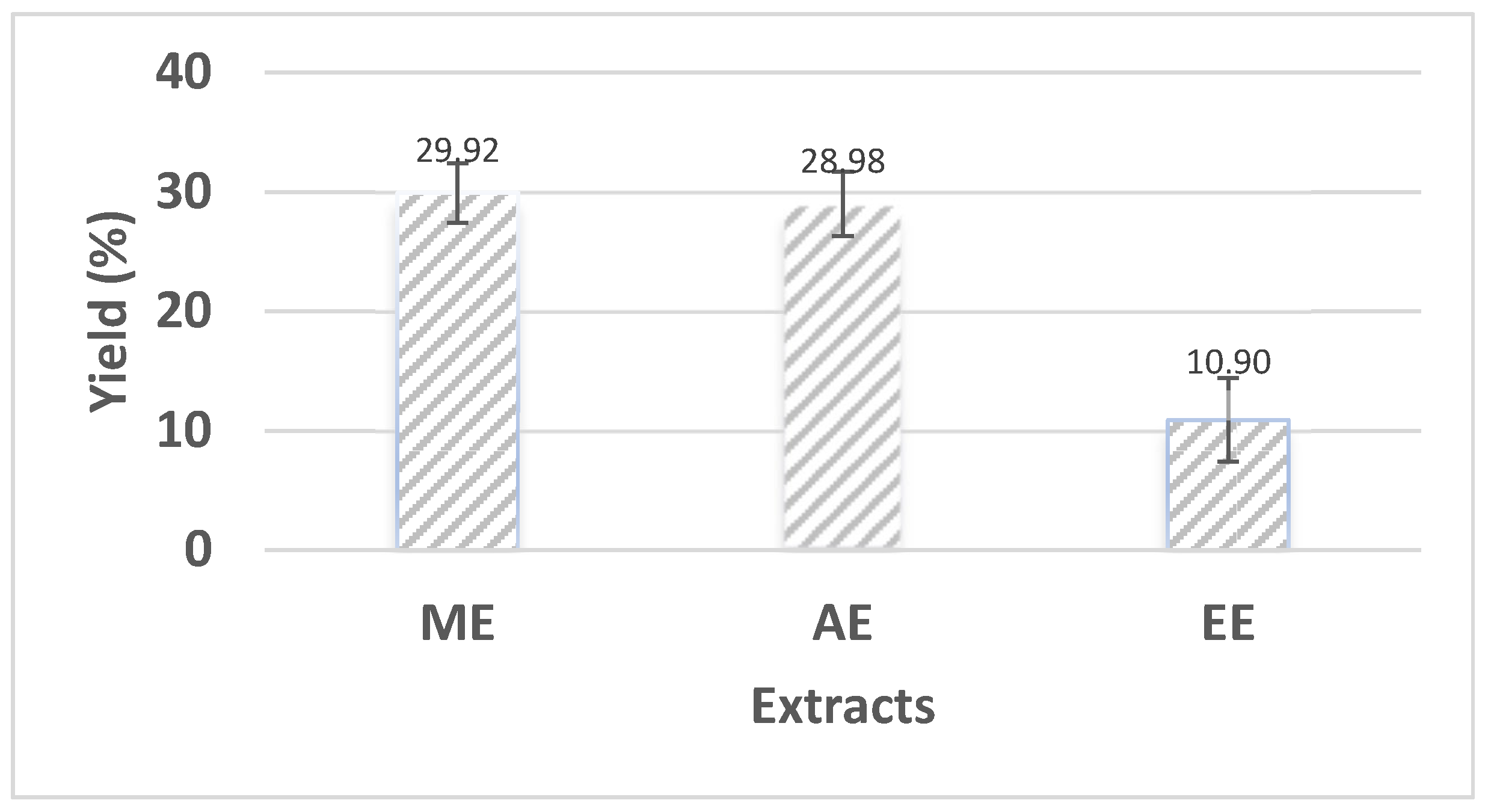
3.1. Yield of extracts
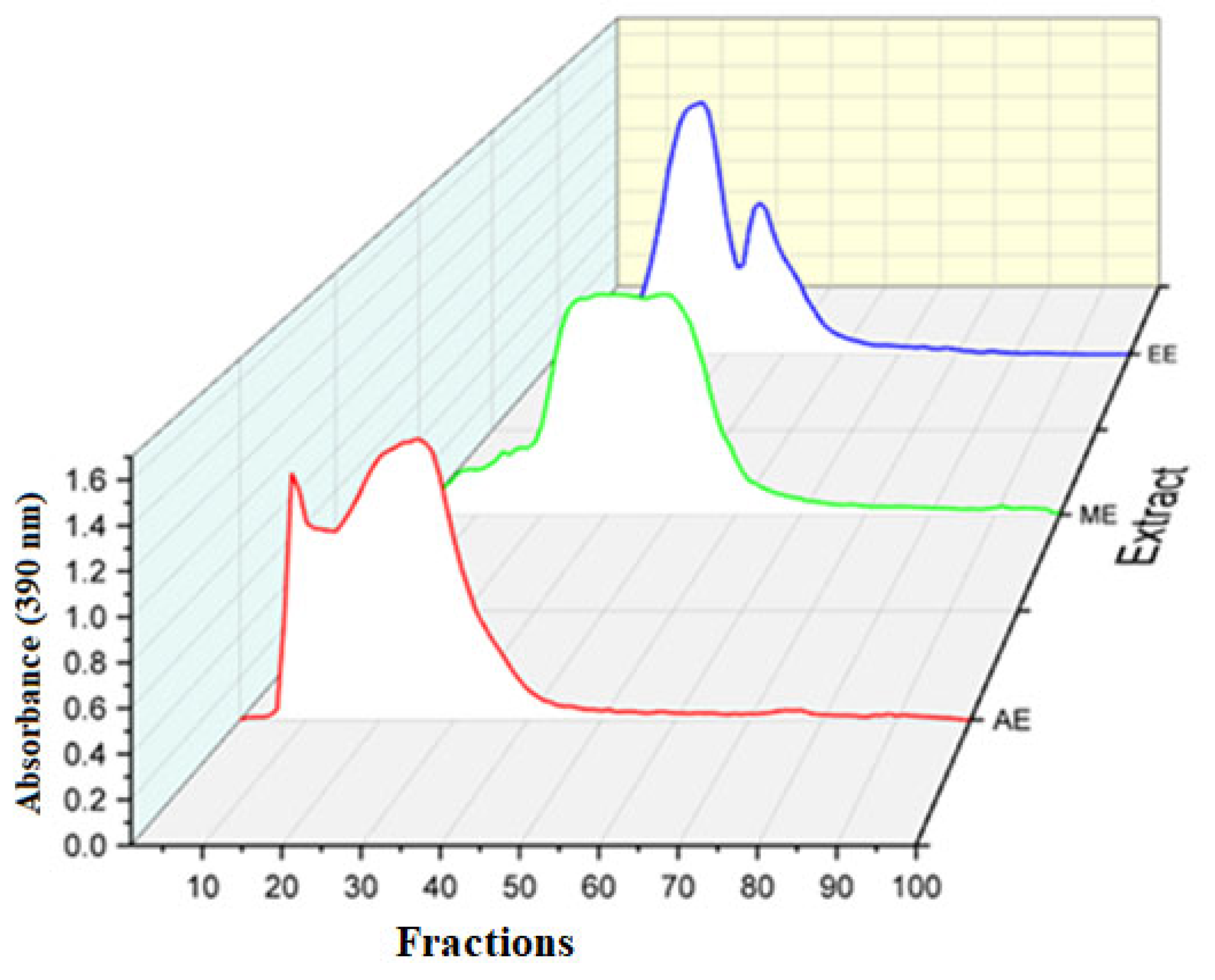
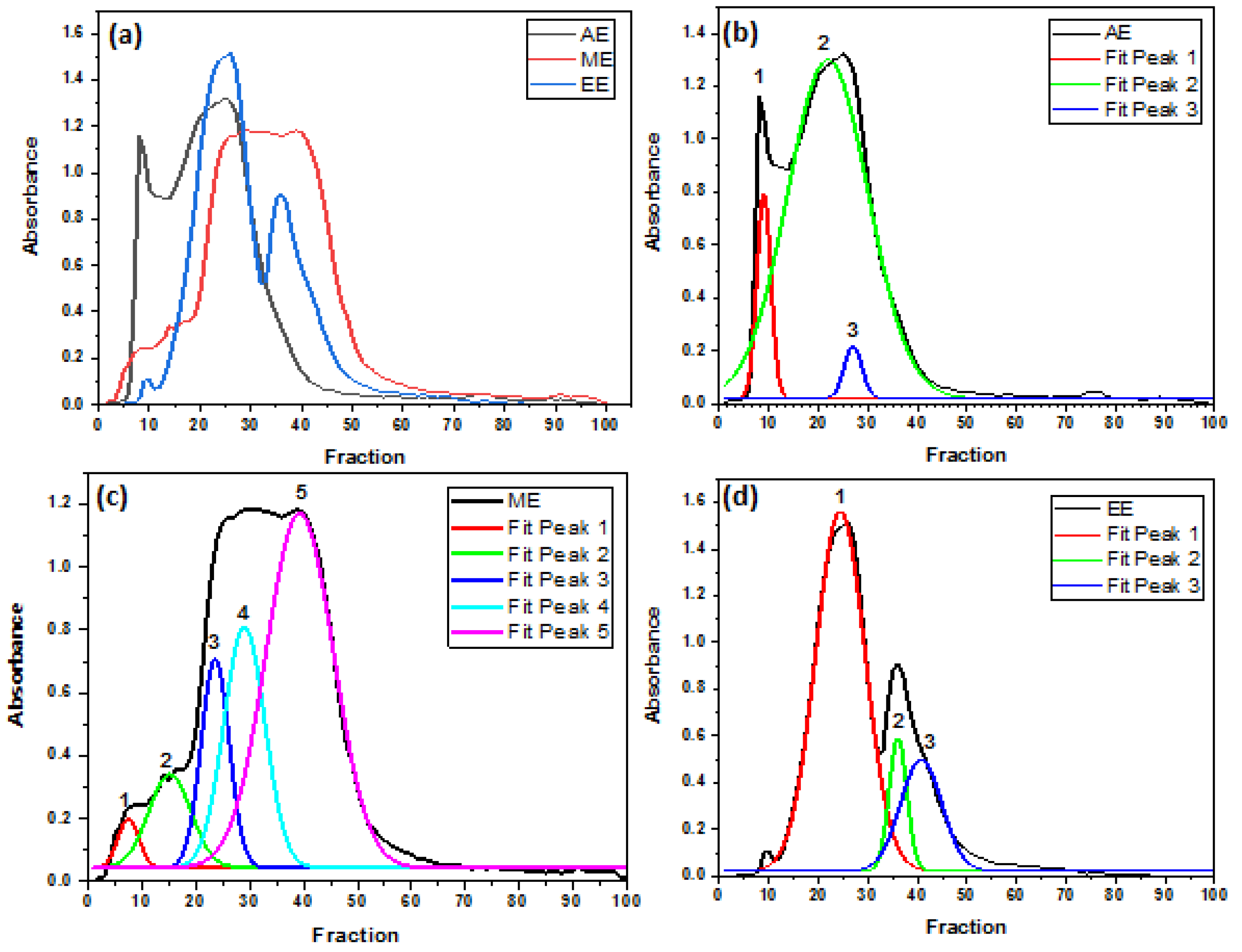
3.2. The polymerization profiles of crude extracts
3.3. Chemical compounds
| Extract | compounds | Component Name | RT | m/ z |
|---|---|---|---|---|
| Methanol | 1 | Kaempferol-3-O-rhamnoside | 9.95 | 417.21 |
| 2 | 11beta,17alpha,21-Trihydroxy-4-pregnene-3,20-dione 21-caprylate | 5.70 | 487.31 | |
| 3 | Quercétine-3-O-arabinoside | 11.12 | 379.16 | |
| 4 | Luteoline-7-O-rutinoside | 11.19 | 393.22 | |
| 5 | digalactosyl diglyceride | 11.03 | 457.19 | |
| 6 | Quercétine-3-O-glucuronide | 10.61 | 431.23 | |
| 7 | Apigénine-7-O-rutinoside | 5.70 | 585.20 | |
| 8 | Myricitine-3-O-rutinoside | 10.71 | 565.21 | |
| Water | 1 | Kaempferol-3-O-glucuronide | 5.70 | 487.30 |
| 2 | Quercétine-3-O-galactoside | 0.27 | 477.08 | |
| 3 | 4-pregnene-11beta,17alpha,21-triol-3,20-dione 21-caprylate | 6.06 | 487.30 | |
| 4 | Kaempferol 3-O-rutinoside | 11.03 | 457.19 | |
| 5 | Myricetine | 0.27 | 377.08 | |
| Ethanol | 1 | Apigenine 7-O-glucuronide | 3.06 | 545.13 |
| 2 | Quercetine-3-O-rutinoside | 11.03 | 665.17 | |
| 3 | Myricetine-3-O-glucuronide | 6.16 | 497.33 |
3.4. Total phenolic compounds determination
3.5. Total Flavonoid content (TFC) determination
3.6. Antioxidant activity determination
3.6.1. Free Radical FRAP Scavenging
3.6.2. Total antioxidant capacity (TAC)
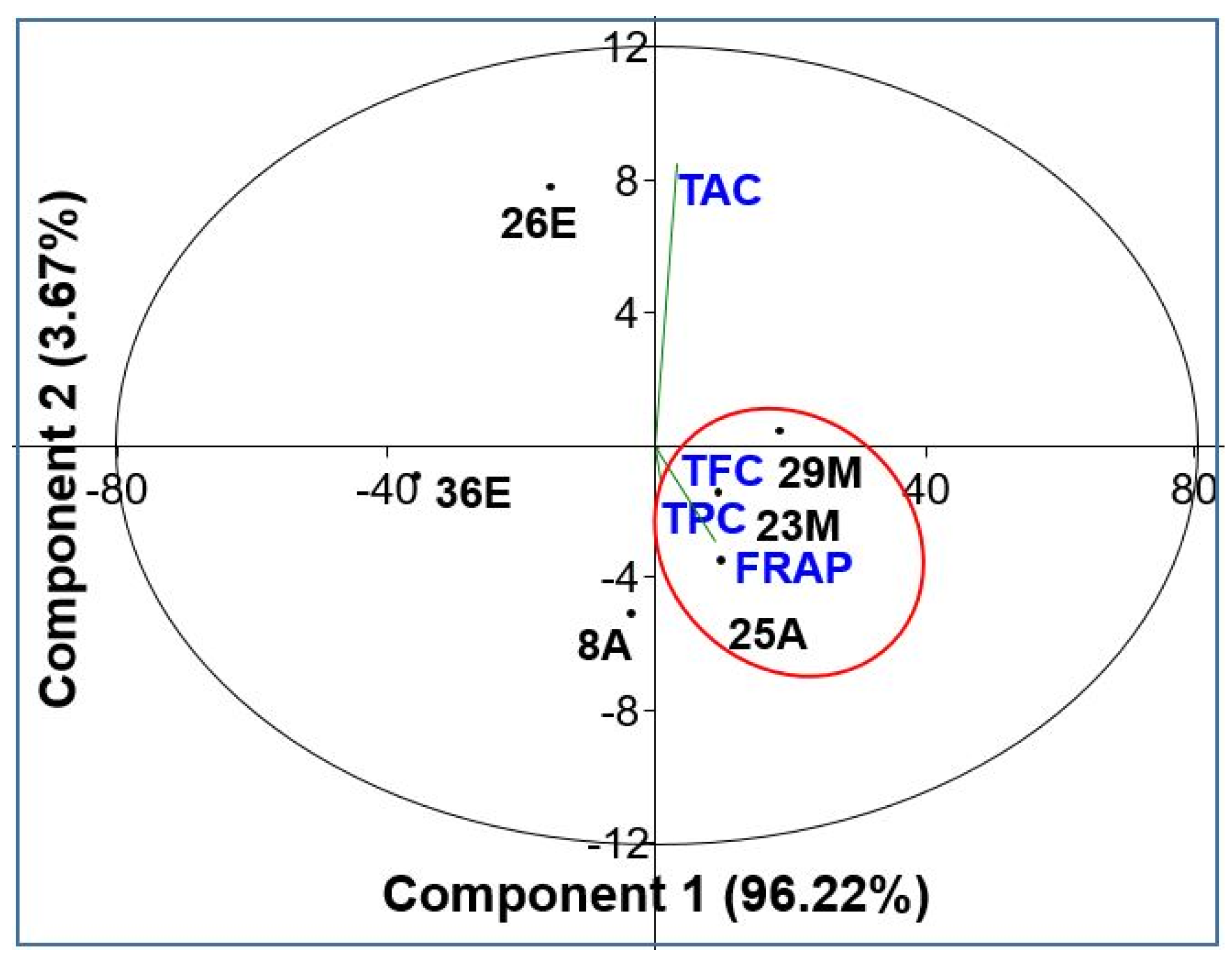
3.6.3. Dimensional analysis of Antioxidant activities and fractions content
3.7. Antimicrobial Activity
| Taxon | Tested strains | AE | EE | ME | Cefoxitin | Fluconazol | ||||||
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MFC | |||
| Bacteria | Gram - | Escherichia Coli | 6.25 | 12.25 | 6.25 | 12.25 | 3.12 | 12.25 | 1.56 | 3.12 | ||
| Pseudomonas aeruginosa | 6.25 | 12.25 | 6.25 | 12.25 | 12.25 | 12.25 | R | R | ||||
| Gram + | Bacillus cereus | 6.25 | 12.25 | 6.25 | 12.25 | 12.25 | 12.25 | 3.12 | 3.12 | |||
| Staphylococcus aureus | 6.25 | 12.25 | 6.25 | 12.25 | 6.25 | 12.25 | 1.56 | 3.12 | ||||
| Fungi | Candida tropicalis | 12.25 | 12.25 | 12.25 | 12.25 | 12.25 | 12.25 | 1.56 | 3.12 | |||
| Saccharomyces cerevisiae | 6.25 | 12.25 | 6.25 | 12.25 | 6.25 | 12.25 | R | R | ||||
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Umar, M.; Mohammed, I.; Oko, J.; TAFINTA, I.; Aliko, A.; Jobbi, Y. Phytochemical Analysis and Antimicrobial Effect of Lemon Grass (Cymbopogon Citratus) Obtained From Zaria, Kaduna State, Nigeria. Journal of Complementary and Alternative Medical Research 2016, 1, 1–8. [CrossRef]
- Süntar, I. Importance of Ethnopharmacological Studies in Drug Discovery: Role of Medicinal Plants. Phytochem Rev 2020, 19, 1199–1209. [CrossRef]
- Chandran, R.; Abrahamse, H. Identifying Plant-Based Natural Medicine against Oxidative Stress and Neurodegenerative Disorders. Oxidative Medicine and Cellular Longevity 2020, 2020, 1–9. [CrossRef]
- Chandra, H.; Singh, C.; Kumari, P.; Yadav, S.; Mishra, A.P.; Laishevtcev, A.; Brisc, C.; Brisc, M.C.; Munteanu, M.A.; Bungau, S. Promising Roles of Alternative Medicine and Plant-Based Nanotechnology as Remedies for Urinary Tract Infections. Molecules 2020, 25, 5593. [CrossRef]
- Ahmed, H.M.; Nabavi, S.; Behzad, S. Herbal Drugs and Natural Products in the Light of Nanotechnology and Nanomedicine for Developing Drug Formulations. Mini Reviews in Medicinal Chemistry 2021, 21, 302–313. [CrossRef]
- Laird, S.A.; Laird, S.; Burningham, M. The Botanical Medicine Industry. In The Commercial Use of Biodiversity; Routledge, 1999 ISBN 978-0-429-34154-0.
- Ahmad Khan, M.S.; Ahmad, I. Chapter 1 - Herbal Medicine: Current Trends and Future Prospects. In New Look to Phytomedicine; Ahmad Khan, M.S., Ahmad, I., Chattopadhyay, D., Eds.; Academic Press, 2019; pp. 3–13 ISBN 978-0-12-814619-4.
- Beri, A.; Pisulkar, S.G.; Bansod, A.V.; Dahihandekar, C. Alternative Prosthodontic Therapies: A Multifaceted Approach. Cureus 2022. [CrossRef]
- da Silva, A.P.G.; Sganzerla, W.G.; Jacomino, A.P.; da Silva, E.P.; Xiao, J.; Simal-Gandara, J. Chemical Composition, Bioactive Compounds, and Perspectives for the Industrial Formulation of Health Products from Uvaia (Eugenia Pyriformis Cambess – Myrtaceae): A Comprehensive Review. Journal of Food Composition and Analysis 2022, 109, 104500. [CrossRef]
- Gorlenko, C.L.; Kiselev, H.Y.; Budanova, E.V.; Zamyatnin, A.A.; Ikryannikova, L.N. Plant Secondary Metabolites in the Battle of Drugs and Drug-Resistant Bacteria: New Heroes or Worse Clones of Antibiotics? Antibiotics 2020, 9, 170. [CrossRef]
- Jan, R.; Asaf, S.; Numan, M.; Lubna; Kim, K.-M. Plant Secondary Metabolite Biosynthesis and Transcriptional Regulation in Response to Biotic and Abiotic Stress Conditions. Agronomy 2021, 11, 968. [CrossRef]
- Mikail, H.G.; Mohammed, M.; Umar, H.D.; Suleiman, M.M. Secondary Metabolites: The Natural Remedies. 2022.
- Wawrosch, C.; Zotchev, S.B. Production of Bioactive Plant Secondary Metabolites through in Vitro Technologies—Status and Outlook. Appl Microbiol Biotechnol 2021, 105, 6649–6668. [CrossRef]
- Abdulhafiz, F.; Mohammed, A.; Reduan, M.F.H.; Kari, Z.A.; Wei, L.S.; Goh, K.W. Plant Cell Culture Technologies: A Promising Alternatives to Produce High-Value Secondary Metabolites. Arabian Journal of Chemistry 2022, 15, 104161. [CrossRef]
- Raut, J.S.; Karuppayil, S.M. A Status Review on the Medicinal Properties of Essential Oils. Industrial Crops and Products 2014, 62, 250–264. [CrossRef]
- Manousi, N.; Sarakatsianos, I.; Samanidou, V. 10 - Extraction Techniques of Phenolic Compounds and Other Bioactive Compounds From Medicinal and Aromatic Plants. In Engineering Tools in the Beverage Industry; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing, 2019; pp. 283–314 ISBN 978-0-12-815258-4.
- Soussi, M.; Fadil, M.; Yaagoubi, W.A.; Benjelloun, M.; El Ghadraoui, L. Simultaneous Optimization of Phenolic Compounds and Antioxidant Abilities of Moroccan Pimpinella Anisum Extracts Using Mixture Design Methodology. Processes 2022, 10, 2580. [CrossRef]
- Jha, A.K.; Sit, N. Extraction of Bioactive Compounds from Plant Materials Using Combination of Various Novel Methods: A Review. Trends in Food Science & Technology 2022, 119, 579–591. [CrossRef]
- Sarikurkcu, C.; Locatelli, M.; Tartaglia, A.; Ferrone, V.; Juszczak, A.M.; Ozer, M.S.; Tepe, B.; Tomczyk, M. Enzyme and Biological Activities of the Water Extracts from the Plants Aesculus Hippocastanum, Olea Europaea and Hypericum Perforatum That Are Used as Folk Remedies in Turkey. Molecules 2020, 25, 1202. [CrossRef]
- Afqir, H.; Belmalha, S.; Ouhssine, M. Growth of Hypericum Perforatum under Moroccan (North African) Climatic and Soil Characteristics. Journal of the Saudi Society of Agricultural Sciences 2024, 23, 219–226. [CrossRef]
- Santos, M.B.; Sillero, L.; Gatto, D.A.; Labidi, J. Bioactive Molecules in Wood Extractives: Methods of Extraction and Separation, a Review. Industrial Crops and Products 2022, 186, 115231. [CrossRef]
- Sadeghi, A.; Rajabiyan, A.; Nabizade, N.; Meygoli Nezhad, N.; Zarei-Ahmady, A. Seaweed-Derived Phenolic Compounds as Diverse Bioactive Molecules: A Review on Identification, Application, Extraction and Purification Strategies. International Journal of Biological Macromolecules 2024, 266, 131147. [CrossRef]
- Lefebvre, T.; Destandau, E.; Lesellier, E. Selective Extraction of Bioactive Compounds from Plants Using Recent Extraction Techniques: A Review. Journal of Chromatography A 2021, 1635, 461770. [CrossRef]
- Manvitha, Karkala; Bidya Bhushan Review on Pharmacological Activity of Cymbopogon Citratus. IJFMR 2022, 4, 1015. [CrossRef]
- Gupta, P.K.; Rithu, B.S.; A, S.; Lokur, A.V.; M, R. Phytochemical Screening and Qualitative Analysis of Cymbopogon Citratus. J Pharmacogn Phytochem 2019, 8, 3338–3343.
- Hassan, H.M.; Aboel-Ainin, M.A.; Ali, S.K.; Darwish, A.G.G. Antioxidant and Antimicrobial Activities of MEOH Extract of Lemongrass (Cymbopogon Citratus). Journal of Agricultural Chemistry and Biotechnology 2021, 12, 25–28. [CrossRef]
- Soares, M.O.; Alves, R.C.; Pires, P.C.; Oliveira, M.B.P.P.; Vinha, A.F. Angolan Cymbopogon Citratus Used for Therapeutic Benefits: Nutritional Composition and Influence of Solvents in Phytochemicals Content and Antioxidant Activity of Leaf Extracts. Food and Chemical Toxicology 2013, 60, 413–418. [CrossRef]
- Oladeji, O.S.; Adelowo, F.E.; Ayodele, D.T.; Odelade, K.A. Phytochemistry and Pharmacological Activities of Cymbopogon Citratus: A Review. Scientific African 2019, 6, e00137. [CrossRef]
- Gaba, J.; Bhardwaj, G.; Sharma, A. Lemongrass. In Antioxidants in Vegetables and Nuts - Properties and Health Benefits; Nayik, G.A., Gull, A., Eds.; Springer: Singapore, 2020; pp. 75–103 ISBN 9789811574702.
- Rahimi, G.; Yousefnia, S.; Angnes, L.; Negahdary, M. Design a PEGylated Nanocarrier Containing Lemongrass Essential Oil (LEO), a Drug Delivery System: Application as a Cytotoxic Agent against Breast Cancer Cells. Journal of Drug Delivery Science and Technology 2023, 80, 104183. [CrossRef]
- Sharmin, D.D.; Revathi, K.; Mahendra, J.; Anandhi, D.; Arun, M.; Vigila, J. Antibacterial Efficacy of Lemon Grass Oil (Cymbopogon Citratus) on the Type of Bacteria and Its Count in Dental Aerosols. Research Journal of Pharmacy and Technology 2022, 15, 4024–4028.
- Nambiar, V.; Matela, H. Potential Functions of Lemon Grass (Cymbopogon Citratus) in Health and Disease. International Journal of Pharmaceutical & Biological Archives 0976 – 3333 2012, 2012; 3(5):1035-1043.
- Gao, S.; Liu, G.; Li, J.; Chen, J.; Li, L.; Li, Z.; Zhang, X.; Zhang, S.; Thorne, R.F.; Zhang, S. Antimicrobial Activity of Lemongrass Essential Oil (Cymbopogon Flexuosus) and Its Active Component Citral Against Dual-Species Biofilms of Staphylococcus Aureus and Candida Species. Frontiers in Cellular and Infection Microbiology 2020, 10.
- Moore-Neibel, K.; Gerber, C.; Patel, J.; Friedman, M.; Ravishankar, S. Antimicrobial Activity of Lemongrass Oil against Salmonella Enterica on Organic Leafy Greens. Journal of Applied Microbiology 2012, 112, 485–492. [CrossRef]
- Olorunnisola, K.; Asiyanbi-Hammed, T.; Hammed, A.; Simsek, S. Biological Properties of Lemongrass: An Overview. International Food Research Journal 2014, 21, 455–462.
- Gbenou, J.D.; Ahounou, J.F.; Akakpo, H.B.; Laleye, A.; Yayi, E.; Gbaguidi, F.; Baba-Moussa, L.; Darboux, R.; Dansou, P.; Moudachirou, M.; et al. Phytochemical Composition of Cymbopogon Citratus and Eucalyptus Citriodora Essential Oils and Their Anti-Inflammatory and Analgesic Properties on Wistar Rats. Mol Biol Rep 2013, 40, 1127–1134. [CrossRef]
- Tazi, A.; Zinedine, A.; Rocha, J.M.; Errachidi, F. Review on the Pharmacological Properties of Lemongrass (Cymbopogon Citratus) as a Promising Source of Bioactive Compounds. Pharmacological Research - Natural Products 2024, 100046. [CrossRef]
- Bassolé, I.H.N.; Lamien-Meda, A.; Bayala, B.; Obame, L.C.; Ilboudo, A.J.; Franz, C.; Novak, J.; Nebié, R.C.; Dicko, M.H. Chemical Composition and Antimicrobial Activity of Cymbopogon Citratus and Cymbopogon Giganteus Essential Oils Alone and in Combination. Phytomedicine 2011, 18, 1070–1074. [CrossRef]
- Mirghani, M.E.S.; Liyana, Y.; Parveen, J. Bioactivity Analysis of Lemongrass (Cymbopogan Citratus) Essential Oil. 2012.
- Valková, V.; Ďúranová, H.; Galovičová, L.; Borotová, P.; Vukovic, N.L.; Vukic, M.; Kačániová, M. Cymbopogon Citratus Essential Oil: Its Application as an Antimicrobial Agent in Food Preservation. Agronomy 2022, 12, 155. [CrossRef]
- Sousa, R.; Figueirinha, A.; Batista, M.T.; Pina, M.E. Formulation Effects in the Antioxidant Activity of Extract from the Leaves of Cymbopogon Citratus (DC) Stapf. Molecules 2021, 26, 4518. [CrossRef]
- Irfan, S.; Ranjha, M.M.A.N.; Nadeem, M.; Safdar, M.N.; Jabbar, S.; Mahmood, S.; Murtaza, M.A.; Ameer, K.; Ibrahim, S.A. Antioxidant Activity and Phenolic Content of Sonication- and Maceration-Assisted Ethanol and Acetone Extracts of Cymbopogon Citratus Leaves. Separations 2022, 9, 244. [CrossRef]
- Lakhdar, L.; Farah, A.; Bajjou, T.; Rida, S.; Bouziane, A.; Ennibi, O. In Vitro Antibacterial Activity of Essentials Oils from Mentha Pulegium, Citrus Aurantium and Cymbopogon Citratus on Virulent Strains of Aggregatibacter Actinomycetemcomitans. Int. J. Pharmacog. Phytochem. Res 2014, 6, 1035–1042.
- Fouad, R.; Bousta, D.; Lalami, A.E.O.; Chahdi, F.O.; Amri, I.; Jamoussi, B.; Greche, H. Chemical Composition and Herbicidal Effects of Essential Oils of Cymbopogon Citratus (DC) Stapf, Eucalyptus Cladocalyx, Origanum Vulgare L and Artemisia Absinthium L. Cultivated in Morocco. Journal of Essential Oil Bearing Plants 2015, 18, 112–123. [CrossRef]
- Kasmi, M.; Aourach, M.; El Boukari, M.; Barrijal, S.; Essalmani, H. [Effectiveness of aqueous extracts of aromatic and medicinal plants against tomato grey mould in Morocco]. C R Biol 2017, 340, 386–393. [CrossRef]
- Lamuela-ravents, R.M. 2,6-Di-Tert-Butyl-4-Hydroxytoluene. 1999, 299, 152–178.
- El Massoudi, S.; Zinedine, A.; Rocha, J.M.; Benidir, M.; Najjari, I.; El Ghadraoui, L.; Benjelloun, M.; Errachidi, F. Phenolic Composition and Wound Healing Potential Assessment of Moroccan Henna (Lawsonia Inermis) Aqueous Extracts. Cosmetics 2023, 10, 92. [CrossRef]
- Ettayebi, K.; Errachidi, F.; Jamai, L.; Tahri-Jouti, M.A.; Sendide, K.; Ettayebi, M. Biodegradation of Polyphenols with Immobilized Candida Tropicalis under Metabolic Induction. FEMS Microbiology Letters 2003, 223, 215–219. [CrossRef]
- Woisky, R.G.; Salatino, A. Analysis of Propolis: Some Parameters and Procedures for Chemical Quality Control. Journal of Apicultural Research 1998, 37, 99–105. [CrossRef]
- Aazza, S.; Lyoussi, B.; Miguel, M.G. Antioxidant and Antiacetylcholinesterase Activities of Some Commercial Essential Oils and Their Major Compounds. Molecules 2011, 16, 7672–7690. [CrossRef]
- Libbey, L.M.; Walradt, J.P. 3,5-Di-Tert-Butyl-4-Hydroxytoluene (BHT) as an Artifact from Diethyl Ether. Lipids 1968, 3, 561–561. [CrossRef]
- El Moujahed, S.; Dinica, R.M.; Abou Oualid, H.; Cudalbeanu, M.; Botezatu-Dediu, A.-V.; Cazanevscaia Busuioc, A.; Ouazzani Chahdi, F.; Kandri Rodi, Y.; Errachidi, F. Sustainable Biomass as Green and Efficient Crosslinkers of Collagen: Case of by-Products from Six Pomegranate Varieties with Global Commercial Interest in Morocco. Journal of Environmental Management 2023, 335, 117613. [CrossRef]
- Olukunle, O.; Adenola, O. Comparative Antimicrobial Activity of Lemon Grass (Cymbopogon Citratus) and Garlic (Allium Sativum) Extracts on Salmonella Typhi. Journal of Advances in Medical and Pharmaceutical Sciences 2019, 1–9. [CrossRef]
- Enriquez-Ochoa, D.; Sánchez-Trasviña, C.; Hernández-Sedas, B.; Mayolo-Deloisa, K.; Zavala, J.; Rito-Palomares, M.; Valdez-García, J.E. Aqueous Two-Phase Extraction of Phenolic Compounds from Sedum Dendroideum with Antioxidant Activity and Anti-Proliferative Properties against Breast Cancer Cells. Separation and Purification Technology 2020, 251, 117341. [CrossRef]
- Alara, O.R.; Abdurahman, N.H.; Ukaegbu, C.I. Extraction of Phenolic Compounds: A Review. Current Research in Food Science 2021, 4, 200–214. [CrossRef]
- El Moujahed, S.; Dinica, R.-M.; Cudalbeanu, M.; Avramescu, S.M.; Msegued Ayam, I.; Ouazzani Chahdi, F.; Kandri Rodi, Y.; Errachidi, F. Characterizations of Six Pomegranate (Punica Granatum L.) Varieties of Global Commercial Interest in Morocco: Pomological, Organoleptic, Chemical and Biochemical Studies. Molecules 2022, 27, 3847. [CrossRef]
- El moujahed, S.; Chahdi, F.O.; Rodi, Y.K.; El ghadraoui, L.; Lemjallad, L.; Errachidi, F. The Moroccan Pomegranate: An Underrated Source of Tannins Extracts and Natural Antimicrobials from Juice Processing Byproducts. Waste Biomass Valor 2021, 12, 5383–5399. [CrossRef]
- Weissmann, G. Studies on Pine Bark Extracts. International Journal of Adhesion and Adhesives 1983, 3, 31–35. [CrossRef]
- Çam, M.; Hışıl, Y. Pressurised Water Extraction of Polyphenols from Pomegranate Peels. Food Chemistry 2010, 123, 878–885. [CrossRef]
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.-H. Effect of Extraction Solvent on Total Phenol Content, Total Flavonoid Content, and Antioxidant Activity of Limnophila Aromatica. Journal of Food and Drug Analysis 2014, 22, 296–302. [CrossRef]
- Hossain, M.A.; Disha, N.K.; Shourove, J.H.; Dey, P. Determination of Antioxidant Activity and Total Tannin from Drumstick (Moringa Oleifera Lam.) Leaves Using Different Solvent Extraction Methods. Turkish Journal of Agriculture-Food Science and Technology 2020, 8, 2749–2755.
- Roriz, C.L.; Barros, L.; Carvalho, A.M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Pterospartum Tridentatum, Gomphrena Globosa and Cymbopogon Citratus: A Phytochemical Study Focused on Antioxidant Compounds. Food Research International 2014, 62, 684–693. [CrossRef]
- Widyawati, P.S.; Budianta, T.D.W.; Kusuma, F.A.; Wijaya, E.L. Difference of Solvent Polarity to Phytochemical Content and Antioxidant Activity of Pluchea Indicia Less Leaves Extracts. International Journal of Pharmacognosy and Phytochemical Research 2014, 6, 850–855.
- Nawaz, H.; Shad, M.A.; Rehman, N.; Andaleeb, H.; Ullah, N. Effect of Solvent Polarity on Extraction Yield and Antioxidant Properties of Phytochemicals from Bean (Phaseolus Vulgaris) Seeds. Braz. J. Pharm. Sci. 2020, 56, e17129. [CrossRef]
- Lezoul, N.E.H.; Belkadi, M.; Habibi, F.; Guillén, F. Extraction Processes with Several Solvents on Total Bioactive Compounds in Different Organs of Three Medicinal Plants. Molecules 2020, 25, 4672. [CrossRef]
- Cruz, M.; Wang, M.; Frisch-Daiello, J.; Han, X. Improved Butanol–Methanol (BUME) Method by Replacing Acetic Acid for Lipid Extraction of Biological Samples. Lipids 2016, 51, 887–896. [CrossRef]
- Chaves, J.O.; de Souza, M.C.; da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; Machado, A.P. da F.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of Flavonoids From Natural Sources Using Modern Techniques. Frontiers in Chemistry 2020, 8.
- Jaradat, N.A.; Zai̇d, A.N.; Hussen, F.; Ali̇, İ. The Effects of Preservation Methods of Grapevine Leaves on Total Phenols, Total Flavonoids and Antioxidant Activity. Marmara Pharm J 2017, 21, 291–297. [CrossRef]
- Gbashi, S.; Njobeh, P.; Steenkamp, P.; Tutu, H.; Madala, N. The Effect of Temperature and Methanol–Water Mixture on Pressurized Hot Water Extraction (PHWE) of Anti-HIV Analogoues from Bidens Pilosa. Chemistry Central Journal 2016, 10, 37. [CrossRef]
- Yusoff, I.M.; Mat Taher, Z.; Rahmat, Z.; Chua, L.S. A Review of Ultrasound-Assisted Extraction for Plant Bioactive Compounds: Phenolics, Flavonoids, Thymols, Saponins and Proteins. Food Research International 2022, 157, 111268. [CrossRef]
- Zhao, J.; Yang, J.; Xie, Y. Improvement Strategies for the Oral Bioavailability of Poorly Water-Soluble Flavonoids: An Overview. International Journal of Pharmaceutics 2019, 570, 118642. [CrossRef]
- Balakrishnan, B.; Paramasivam, S.; Arulkumar, A. Evaluation of the Lemongrass Plant (Cymbopogon Citratus) Extracted in Different Solvents for Antioxidant and Antibacterial Activity against Human Pathogens. Asian Pacific Journal of Tropical Disease 2014, 4, S134–S139. [CrossRef]
- Fokom, R.; Adamou, S.; Essono, D.; Ngwasiri, D.P.; Eke, P.; Teugwa Mofor, C.; Tchoumbougnang, F.; Fekam, B.F.; Amvam Zollo, P.H.; Nwaga, D.; et al. Growth, Essential Oil Content, Chemical Composition and Antioxidant Properties of Lemongrass as Affected by Harvest Period and Arbuscular Mycorrhizal Fungi in Field Conditions. Industrial Crops and Products 2019, 138, 111477. [CrossRef]
- Mukarram, M.; Choudhary, S.; Khan, M.A.; Poltronieri, P.; Khan, M.M.A.; Ali, J.; Kurjak, D.; Shahid, M. Lemongrass Essential Oil Components with Antimicrobial and Anticancer Activities. Antioxidants 2022, 11, 20. [CrossRef]
- Kiani, H.S.; Ali, A.; Zahra, S.; Hassan, Z.U.; Kubra, K.T.; Azam, M.; Zahid, H.F. Phytochemical Composition and Pharmacological Potential of Lemongrass (Cymbopogon) and Impact on Gut Microbiota. AppliedChem 2022, 2, 229–246. [CrossRef]
- Ajayi, E.O.; Sadimenko, A.P.; Afolayan, A.J. GC–MS Evaluation of Cymbopogon Citratus (DC) Stapf Oil Obtained Using Modified Hydrodistillation and Microwave Extraction Methods. Food Chemistry 2016, 209, 262–266. [CrossRef]
- Kabotso, D.E.K.; Neglo, D.; Kwashie, P.; Agbo, I.A.; Abaye, D.A. GC/MS Composition and Resistance Modulatory Inhibitory Activities of Three Extracts of Lemongrass: Citral Modulates the Activities of Five Antibiotics at Sub-Inhibitory Concentrations on Methicillin-Resistant Staphylococcus Aureus. Chemistry & Biodiversity 2022, 19, e202200296. [CrossRef]
- Bhambhani, S.; Kondhare, K.R.; Giri, A.P. Diversity in Chemical Structures and Biological Properties of Plant Alkaloids. Molecules 2021, 26, 3374. [CrossRef]
- Dhama, K.; Sharun, K.; Gugjoo, M.B.; Tiwari, R.; Alagawany, M.; Iqbal Yatoo, Mohd.; Thakur, P.; Iqbal, H.M.N.; Chaicumpa, W.; Michalak, I.; et al. A Comprehensive Review on Chemical Profile and Pharmacological Activities of Ocimum Basilicum. Food Reviews International 2023, 39, 119–147. [CrossRef]
- Sitarek, P.; Merecz-Sadowska, A.; Kowalczyk, T.; Wieczfinska, J.; Zajdel, R.; Śliwiński, T. Potential Synergistic Action of Bioactive Compounds from Plant Extracts against Skin Infecting Microorganisms. International Journal of Molecular Sciences 2020, 21, 5105. [CrossRef]
- Hosseini-Zare, M.S.; Sarhadi, M.; Zarei, M.; Thilagavathi, R.; Selvam, C. Synergistic Effects of Curcumin and Its Analogs with Other Bioactive Compounds: A Comprehensive Review. European Journal of Medicinal Chemistry 2021, 210, 113072. [CrossRef]
- Sepahpour, S.; Selamat, J.; Abdul Manap, M.Y.; Khatib, A.; Abdull Razis, A.F. Comparative Analysis of Chemical Composition, Antioxidant Activity and Quantitative Characterization of Some Phenolic Compounds in Selected Herbs and Spices in Different Solvent Extraction Systems. Molecules 2018, 23, 402. [CrossRef]
- Unuigbe, C.; Enahoro, J.; Erharuyi, O.; Okeri, H.A. Phytochemical Analysis and Antioxidant Evaluation of Lemon Grass (Cymbopogon Citratus DC.) Stapf Leaves. Journal of Applied Sciences and Environmental Management 2019, 23, 223–228. [CrossRef]
- Godwin, A.; Daniel, G.A.; Shadrack, D.; Elom, S.A.; Afua, N.; Ab, K.; Godsway, B.; Joseph, K.G.; Sackitey, N.O.; Isaak, K.B. Determination of Elemental, Phenolic, Antioxidant and Flavonoid Properties of Lemon Grass (Cymbopogon Citratus Stapf). International Food Research Journal 2014, 21.
- Sah, S.Y.; Sia, C.M.; Chang, S.K.; Ang, Y.K.; Yim, H.S. Antioxidant Capacity and Total Phenolic Content of Lemon Grass (Cymbopogon Citratus) Leave. Annals Food Science and Technology 2012, 13, 150–155.
- Oracz, J.; Nebesny, E.; Żyżelewicz, D. Identification and Quantification of Free and Bound Phenolic Compounds Contained in the High-Molecular Weight Melanoidin Fractions Derived from Two Different Types of Cocoa Beans by UHPLC-DAD-ESI-HR-MSn. Food Research International 2019, 115, 135–149. [CrossRef]
- Oracz, J.; Zyzelewicz, D. In Vitro Antioxidant Activity and FTIR Characterization of High-Molecular Weight Melanoidin Fractions from Different Types of Cocoa Beans. Antioxidants 2019, 8, 560. [CrossRef]
- Mirzaei, M.; Ladan Moghadam, A.; Hakimi, L.; Danaee, E. Plant Growth Promoting Rhizobacteria (PGPR) Improve Plant Growth, Antioxidant Capacity, and Essential Oil Properties of Lemongrass (Cymbopogon Citratus) under Water Stress. Iranian Journal of Plant Physiology 2020, 10, 3155–3166. [CrossRef]
- El Gharras, H. Polyphenols: Food Sources, Properties and Applications – a Review. International Journal of Food Science & Technology 2009, 44, 2512–2518. [CrossRef]
- Aourach, M.; Barbero, G.F.; González de Peredo, A.V.; Diakite, A.; El Boukari, M.; Essalmani, H. Composition and Antifungal Effects of Aqueous Extracts of Cymbopogon Citratus, Laurus Nobilis and Santolina Chamaecyparissus on the Growth of Fusarium Oxysporum f. Sp. Lentis. Archives of Phytopathology and Plant Protection 2021, 54, 2141–2159. [CrossRef]
- Alagawany, M.; El-Saadony, M.T.; Elnesr, S.S.; Farahat, M.; Attia, G.; Madkour, M.; Reda, F.M. Use of Lemongrass Essential Oil as a Feed Additive in Quail’s Nutrition: Its Effect on Growth, Carcass, Blood Biochemistry, Antioxidant and Immunological Indices, Digestive Enzymes and Intestinal Microbiota. Poultry Science 2021, 100, 101172. [CrossRef]
- Khosravi Bakhtiari, M.; Sharifiyazdi, H.; Nazifi, S.; Ghaemi, M.; Hadadipour Zarandi, M. Effects of Citral on Serum Antioxidant Status and Liver Genes Expressions of Paraoxonase 1 and Nitric Oxide Synthase in a Rat Model of Streptozotocin-Induced Diabetes Mellitus. Iran J Vet Res 2021, 22, 195–202. [CrossRef]
- Pisoschi, A.M.; Negulescu, G.P. Methods for Total Antioxidant Activity Determination: A Review. Biochem Anal Biochem 2011, 1, 106.
- Wang, B.; Huang, Q.; Venkitasamy, C.; Chai, H.; Gao, H.; Cheng, N.; Cao, W.; Lv, X.; Pan, Z. Changes in Phenolic Compounds and Their Antioxidant Capacities in Jujube (Ziziphus Jujuba Miller) during Three Edible Maturity Stages. LWT - Food Science and Technology 2016, 66, 56–62. [CrossRef]
- Addai, Z.R.; Abdullah, A.; Mutalib, S.A. Effect of Extraction Solvents on the Phenolic Content and Antioxidant Properties of Two Papaya Cultivars. J. Med. Plants Res. 2013, 7, 3354–3359.
- El Moujahed, S.; Errachidi, F.; Abou Oualid, H.; Botezatu-Dediu, A.-V.; Ouazzani Chahdi, F.; Kandri Rodi, Y.; Dinica, R.M. Extraction of Insoluble Fibrous Collagen for Characterization and Crosslinking with Phenolic Compounds from Pomegranate Byproducts for Leather Tanning Applications. RSC Adv. 2022, 12, 4175–4186. [CrossRef]
- Cardinali, A.; Cicco, N.; Linsalata, V.; Minervini, F.; Pati, S.; Pieralice, M.; Tursi, N.; Lattanzio, V. Biological Activity of High Molecular Weight Phenolics from Olive Mill Wastewater. J. Agric. Food Chem. 2010, 58, 8585–8590. [CrossRef]
- Stasiuk, M.; Kozubek, A. Biological Activity of Phenolic Lipids. Cell. Mol. Life Sci. 2010, 67, 841–860. [CrossRef]
- Bouchama, C.; Zinedine, A.; Rocha, J.M.; Chadli, N.; El Ghadraoui, L.; Chabir, R.; Raoui, S.M.; Errachidi, F. Effect of Phenolic Compounds Extracted from Turmeric (Curcuma Longa L.) and Ginger (Zingiber Officinale) on Cutaneous Wound Healing in Wistar Rats. Cosmetics 2023, 10, 137. [CrossRef]
- Matos, M.; Claro, F.C.; Lima, T.A.M.; Avelino, F.; Hansel, F.A.; Maciel, G.M.; Lomonaco, D.; Magalhães, W.L.E. Acetone:Water Fractionation of Pyrolytic Lignin Improves Its Antioxidant and Antibacterial Activity. Journal of Analytical and Applied Pyrolysis 2021, 156, 105175. [CrossRef]
- Kgang, I.E.; Mathabe, P.M.K.; Klein, A.; Kalombo, L.; Belay, Z.A.; Caleb, O.J. Effects of Lemon (Citrus Limon L.), Lemongrass (Cymbopogon Citratus) and Peppermint (Mentha Piperita L.) Essential Oils against of Botrytis Cinerea and Penicillium Expansum. JSFA reports 2022, 2, 405–414. [CrossRef]
- Schweitzer, B.; Balázs, V.L.; Molnár, S.; Szögi-Tatár, B.; Böszörményi, A.; Palkovics, T.; Horváth, G.; Schneider, G. Antibacterial Effect of Lemongrass (Cymbopogon Citratus) against the Aetiological Agents of Pitted Keratolyis. Molecules 2022, 27, 1423. [CrossRef]
- Boukhatem, M.N.; Ferhat, M.A.; Kameli, A.; Saidi, F.; Kebir, H.T. Lemon Grass (Cymbopogon Citratus) Essential Oil as a Potent Anti-Inflammatory and Antifungal Drugs. Libyan Journal of Medicine 2014, 9. [CrossRef]
- Jain, N.; Sharma, M. Phytochemical Screening and Antidermatophytic Activity of Cymbopogon Citratus Leaves Essential Oil and Their Fractions. Journal of Essential Oil Bearing Plants 2017, 20, 1107–1116. [CrossRef]
- Özkütük, A.S. Antimicrobial Effects of Carnosic Acid, Kaempferol and Luteolin on Biogenic Amine Production by Spoilage and Food-Borne Pathogenic Bacteria. Food Bioscience 2022, 46, 101588. [CrossRef]
- Dilbar, S.; Sher, H.; Ali, A.; Ullah, Z.; Ali, I. Biological Synthesis of Ag-Nanoparticles Using Stachys Parviflora and Its Inhibitory Potential against Xanthomonas Campestris. South African Journal of Botany 2023, 157, 409–422. [CrossRef]
- Abuga, I.; Sulaiman, S.F.; Abdul Wahab, R.; Ooi, K.L.; Abdull Rasad, M.S.B. In Vitro Antibacterial Effect of the Leaf Extract of Murraya Koenigii on Cell Membrane Destruction against Pathogenic Bacteria and Phenolic Compounds Identification. European Journal of Integrative Medicine 2020, 33, 101010. [CrossRef]
- Lan, J.-E.; Li, X.-J.; Zhu, X.-F.; Sun, Z.-L.; He, J.-M.; Zloh, M.; Gibbons, S.; Mu, Q. Flavonoids from Artemisia Rupestris and Their Synergistic Antibacterial Effects on Drug-Resistant Staphylococcus Aureus. Natural Product Research 2021, 35, 1881–1886. [CrossRef]
- Embaby, M.A.; El-Raey, M.A.; Zaineldain, M.; Almaghrabi, O.; Marrez, D.A. Synergistic Effect and Efflux Pump Inhibitory Activity of Ficus Nitida Phenolic Extract with Tetracycline against Some Pathogenic Bacteria. Toxin Reviews 2021, 40, 1187–1197. [CrossRef]
| Extracts | Fractions | TPC [mg/g] |
|---|---|---|
| Aqueous extract (AE) | 8 | 2.04±0.11** |
| 16 | 2.77±0.12*** | |
| 25 | 4.60±0.29**** | |
| 58 | 0.33±0.05** | |
| 75 | 0.11±0.03* | |
| Ethanolic extract (EE) | 26 | 0.89±0.09** |
| 36 | 0.11±0.08* | |
| Methanolic extract (ME) | 23 | 2.83±0.80* |
| 29 | 3.26±0.44** |
| Extracts | Fractions | TFC [mg/g] |
|---|---|---|
| Aqueous extract (AE) | 8 | 0.60±0.07*** |
| 16 | 0.46±0.07* | |
| 25 | 0.57±0.07** | |
| Ethanolic extract (EE) | 26 | 0.26±0.03** |
| 36 | 0.19±0.05* | |
| Methanolic extract (ME) | 23 | 0.48±0.12* |
| 29 | 0.70±0.08** |
| Extracts | Fractions | FRAP (%) |
|---|---|---|
| Aqueous extract (AE) | 8 | 63,91 |
| 25 | 75,86 | |
| Ethanolic extract (EE) | 26 | 48,53 |
| 36 | 32.67 | |
| Methanolic extract (ME) | 23 | 75,00 |
| 29 | 97,89 |
| Extracts | Fractions | TAC |
|---|---|---|
| Aqueous extract (AE) | 8 | 68.98% |
| 25 | 75.18% | |
| Ethanolic extract (EE) | 26 | 77.21% |
| 36 | 62.35% | |
| Methanolic extract (ME) | 23 | 76.81% |
| 29 | 89.89% |
| Antioxidant Activity | Predictors | SS | Df | MS | F-Ratio | P-Value |
|---|---|---|---|---|---|---|
| FRAP | TPC | 9.39 | 1 | 9.39 | 9.33 | 0.03 |
| Residual | 4.03 | 4 | 1.01 | |||
| TFC | 0.16 | 1 | 0.16 | 19.23 | 0.01 | |
| Residual | 0.03 | 4 | 0.01 | |||
| TAC | TPC | 4.14 | 1 | 4.14 | 1.78 | 0.25 |
| Residual | 9.28 | 4 | 2.32 | |||
| TFC | 0.07 | 1 | 0.07 | 2.30 | 0.20 | |
| Residual | 0.13 | 4 | 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





