1. Introduction
A cell’s means of communicating with the outside world depends on the screening and specific binding to molecular recognition patterns in order to react to or impact the cellular environment. Hence, the molecular binding dynamics of receptor–ligand pairs at the interface of cells are at the very beginning of many signaling cascades leading to essential effector functions.
Receptor–ligand interactions can be discriminated into the interaction of soluble ligands with their respective membrane-associated receptors (3D environment), or the interaction of ligands attached to a solid phase, be it another cellular membrane or extracellular components (2D environment). The immune system has a variety of highly motile cells in its arsenal in order to scan for sources of antigen. One of these prominent receptor–ligand interactions are essential for the effector functions of T cells, a key player of the adaptive immune system. Pathogen-derived peptides are presented by the major histocompatibility complex (pMHC) proteins at the surfaces of antigen-presenting cells, while T cells scan for these complexes via their T cell receptors (TCRs).
The binding duration (or dwell time) of the pMHC complex to the TCR is a direct reflection of its affinity and other physicochemical parameters, and it correlates very well with quantitative parameters of the downstream signaling cascades leading to T cell activation [
1,
2]. Strong agonistic interactions often exhibit several seconds to tens of seconds length, whereas weak agonists can feature lifetimes of less than 100 ms [
1,
3,
4]. The rates of unbinding are therefore relevant read-out parameters at the state of signal initiation for a multitude of immunological assays. This is especially important since the mere measurement of 3D affinity is often not enough to predict the stimulating potency of antigenic peptides [
5,
6,
7].
The standard approach for quantification of affinity and binding kinetics utilizes surface plasmon resonance (SPR) to study the interaction of purified recombinant TCRs and pMHCs of interest [
5,
7]. This method sensitively quantitates the 3D affinity of the interaction partners, but does not provide any cellular context and hence does not encompass cellular interactions such as adhesion factors, co-stimulation, or coreceptor engagement. Especially, the impact of external forces, membrane fluctuation and protein flexibility cannot be estimated by SPR [
7]. An alternative approach for receptor–ligand interaction lifetime quantification in 2D uses fluorescence microscopy and is based on single-molecule Förster resonance energy transfer (smFRET) [
8]. In this method, a functionalized glass-supported lipid bilayer (SLB) is used as mimicry of the surface of an antigen-presenting cell. Besides adhesion and co-stimulation factors, it contains MHCs loaded with fluorescently labeled peptides that act as FRET acceptors; the corresponding TCRs of SLB-interacting T cells are fluorescently labeled with a specific antibody fragment providing the FRET donor molecule. Whenever the two binding partners engage, the donor molecule transfers its energy to the acceptor molecule and a FRET signal can be observed. From multiple single-molecule FRET signals the interaction lifetime of the receptor–ligand pair can be quantified. The 2D binding kinetics measured in such a way are in line with the downstream signaling outcome [
8,
9].
This study introduces a rigorous mathematical model for determination of exponentially distributed receptor–ligand interaction lifetimes based on the statistical framework of survival analysis. The model is verified and characterized using simulated data. Additionally, a comprehensive analysis pipeline to infer interaction lifetimes from raw microscopy image data is presented and applied to experimental data from several TCR–pMHC pairs.
The new algorithm yields accurate and robust results and allows for determination of TCR–pMHC interaction lifetimes across several orders of magnitude. Furthermore, we created a new analysis software suite [
10] which strives to rely on automated processing as much as possible to optimize throughput and to minimize subjective criteria which could lead to biased data presentation.
2. Materials and Methods
2.1. Animal Model
5c.c7 TCR-transgenic mice (Tg(Tcra5CC7,Tcrb5CC7)IWep, PMID: 1328464) bred onto the B10.A background were a kind gift from Michael Dustin (University of Oxford, UK). The mice were housed in groups of 2–5 per cage in the pathogen-free facility at the Medical University of Vienna, Austria. Spleens and lymph nodes were harvested from 12–16 weeks old gender-mixed mice.
Spleens of AND-TCR transgenic B10.BR animals (Tg(TcrAND)53Hed, PMID: 2571940) were removed and sent in DMEM/1% BSA to the Medical University of Vienna on ice. The mice were genotyped by PCR or by cytometry and housed in groups of 2-5 animals per cage in the specific pathogen-free Core Facility Animal Models at the Biomedical Center of LMU Munich, Germany.
2.2. Tissue Culture
Splenocytes or lymphocytes of 5c.c7 and AND
TCR-transgenic mice were isolated and pulsed with 2 µM C18 reverse-phase HPLC–purified MCC (88-103) peptide (sequence: ANERADLIAYLKQATK; Intavis, Germany) and 50 U ml
-1 IL-2 (eBioscience, USA) for 7 days to arrive at a transgenic T cell culture [
11]. T cells were maintained at 37°C in an atmosphere of 5 % CO2 in RPMI 1640 media (Life technologies) supplemented with 100 µg ml-1 penicillin (Life technologies, USA), 100 µg ml-1 streptomycin (Life technologies, USA), 2 mM L-glutamine (Life technologies, USA), 10% FCS (Biowest, France), 0.1 mM non-essential amino acids (Lonza, Switzerland), 1 mM sodium pyruvate (Life technologies, USA) and 50 µM
-mercaptoethanol (Life technologies, USA). After expansion, debris and dead cells were removed by Histopaque-1119 (Merck, USA) density gradient centrifugation. Antigen-experienced T cells were used for experiments from day eight to ten.
2.3. Formation of Functionalized Lipid
Bilayers
Lipids dissolved in chloroform were mixed (98 mol-% POPC (1-palmitoyl-2-oleoyl-glycero-3-phosphocholine) plus 2 mol-% DGS-NTA(Ni) (1,2-dioleoyl-sn-glycero-3-[(N-(5-amino-1-carboxypentyl) iminodiacetic acid)succinyl] (nickel salt)); Avanti Polar Lipids, Inc., USA) and subsequently dried under a nitrogen stream for 20 min in a glass test tube (Schott FIOLAX 12×75mm, Carl Roth, Germany). After resuspension in 1 ml Dulbecco’s Phosphate Buffered Saline (DPBS, Merck KGaA, Germany), they were sonicated for 10 min in an ultrasound water bath (USC500TH, VWR, England) at room temperature. The resulting small unilamellar vesicle solution was diluted to 125 µM using DPBS.
The original cover slip of an eight-well chamber (Nunc Lab-Tek, Thermo Fisher Scientific, USA) was replaced by attaching a plasma-treated (10 min; PDC-002 Plasma Cleaner, Harrick Plasma, USA) microscopy cover slip (MENZEL-Gläser Deckgläser 24×60mm #1.5) using duplicating silicone (Twinsil soft 18, picodent, Germany). 150 µl of vesicle solution was filled into each well and left to incubate for 20 min. at room temperature. Subsequent washing with DPBS removed excess vesicles.
Into each well, 30 ng of His-tagged murine ICAM-1, 50 ng of His-tagged murine B7-1, and 2 ng of His-tagged IEk-MCC-Alexa Fluor 647 (for experiments with murine T cells) were added and left for incubation for 75 min. Unbound excess proteins were washed away using DPBS.
2.4. Protein Expression and Refolding
The TCR
-reactive H57 scF
V (J0, GenBank: MH045460.1) and the fluorescently labelled H57 scF
V (J1, GenBank: MH045461.1) were produced as described [
12]. In short, scF
V constructs were expressed in Escherichia coli and inclusion bodies were extracted. H57 scF
Vs were refolded in vitro, concentrated, and purified by gel filtration. The monomeric H57 scF
V (J1) was conjugated with Alexa Fluor 555 C2 Maleimide (Thermo Fisher Scientific, USA). Protein-to-dye ratios of site-specifically decorated H57 scF
V-Alexa Fluor 555 were 1.0.
The murine MHC class II molecule I-E
k subunits (with a 12x histidine-tag) and the
subunits were expressed in E. coli as inclusion bodies and refolded in vitro with a fluorescently labelled MCC peptide as described [
12]. In short, his-tagged I-E
k/MCC was refolded and purified via Ni-NTA-based affinity chromatography followed by gel filtration. MCC peptides were site-specifically labelled via Alexa Fluor 647 C2 Maleimide (Thermo Fisher Scientific, USA) and purified as described [
12].
Murine recombinant ICAM-1-10xHis and B7-1-10xHis were purchased from Sino Biological (China).
2.5. Single-Molecule Fluorescence
Microscopy
By means of an objective with high numerical aperture ( Plan-FLUAR 100x/1.45 oil, Zeiss, Germany), objective-type total internal reflection (TIR) illumination of fluorophores was realized. Donor fluorophores (Alexa Fluor 555) were excited with a 532 nm laser (LCX-532L with L1C-AOM, Oxxius, France), acceptor fluorophores (Alexa Fluor 647) with a 640 nm laser (OBIS 640, Coherent, USA). A quad-band dichroic mirror (Di01-R405/488/532/635-25×36, Semrock Inc., USA) separated the emission beam from the excitation light. With the help of a beam splitter device (Optosplit II, Cairn Research, UK) employing a dichroic mirror (FF640-FDi01-25×36, Semrock Inc., USA) and bandpass filters (ET570/60m and ET675/50m, Chroma Technology Corp, USA), donor and acceptor fluorophore emission images were projected side by side onto the chip of an electron multiplying charge-coupled device (EM-CCD) camera (Andor iXon Ultra 897, Andor Technology Ltd, UK). The microscope and peripherals were controlled by using the SDT-control software developed in-house.
Stroboscopic illumination of fluorophores was synchronized with camera read-out. For the main illumination sequence, the following operations were repeated a pre-defined number of times: a) read out of the camera chip to remove charges from stray light, b) excitation of donor fluorophores and simultaneous recording of donor and acceptor emission, c) pause for a pre-defined amount of time to achieve the desired recording rate.
The number of repeats was typically chosen to allow monitoring a single cell for several minutes. Before and after this sequence, a single image upon acceptor excitation was recorded to assess the integrity of the lipid bilayer.
TCRs were fluorescently labeled as follows: cells in medium were centrifuged for 3 min at 350g. Consecutively, the supernatant was removed, leaving the cells in roughly 60 µl of medium. 45 ng of unlabeled and 15 ng of fluorescently labeled H57-scFV in 5 µl of DPBS were added and left for incubation on ice for 20 min. Excess scFV was washed away by adding 5 ml of Hank’s Buffered Salt Solution (HBSS; Merck KGaA, Germany) + 2% Fetal Calf Serum (FCS; Merck KGaA, Germany) at 4°C, centrifuging for 3 min at 350g and 4°C and removing the supernatant. Cells were subsequently kept on ice.
Immediately before microscopy measurements, the buffer in the wells containing SLBs was exchanged for HBSS + 2% FCS (pre-warmed to the temperature of interest). T cells were added and left to attach for 2–3 min.
2.6. Calcium Flux Measurements
T cell quality was monitored via calcium flux experiments in parallel to the single-molecule FRET experiments. Intracellular calcium levels were measured with the ratio-metric dye Fura-2-AM (Thermo Fischer Scientific, USA) as published [
13]. 1–2
T cells were incubated with 2 µM Fura-2-AM (Life technologies, USA) in T cell growth medium for 15-20 minutes at room temperature and subsequently washed with warm (room temperature) imaging buffer (1x HBSS (Life technologies, USA) supplemented with 2% FCS (Biowest, France), 2 mM CaCl
2 and 2 mM MgCl
2 (Merck, USA)). Immediately afterwards, T cells were seeded onto functionalized SLBs featuring either unlabeled B7-1 and ICAM-1 (100 µm
-2) as negative control, or additionally unlabeled I-E
k/MCC (100 µm
-2) as positive control. Calcium response was recorded at room temperature. Fura-2 was excited with a monochromatic light source (Polychrome V, TILL Photonics, Germany) coupled to a Zeiss Axiovert 200M which was equipped with a 10× objective (UPlanFL N 10x, NA = 0.3, Olympus, Japan), a 1.6× tube lens, a long-pass filter (T400lp, Chroma Technology, USA), an emission filter ET510/80 (Chroma Technology, USA) and an EM-CCD camera (Andor iXon 897, Andor, UK). Imaging was performed with excitation at 340 and 380 nm with illumination times of 50 and 10 ms, respectively. The total recording time was 10 min with one image per second. Calcium image analysis was carried out with a custom-made MATLAB (Mathworks, Inc., USA) software [
14].
2.7. Maximum Likelihood Estimation (MLE) of Apparent
Lifetimes
For the inference of apparent lifetimes from single-molecule track lengths by means of survival analysis, the
lifelines Python package [
15] was used. Maximizing the likelihood derived in
Section 3.1 is implemented in the
lifelines.ExponentialFitter class via the
fit_interval_censoring method, which yields the maximum likelihood estimate and its standard error.
To perform “conventional” MLE (i.e., no proper handling of traces continuing beyond the observation window), we calculated the mean track length shifted by
. The result was additionally shifted by half a recording interval to account for the fact that the signal vanishes sometime between two recordings. This results in
using the nomenclature from
Section 3.1.
2.8. Fitting of the Lifetime
Having inferred apparent lifetimes at different recording intervals, eq.
1 was fit by means of a non-linear least squares method using the
optimize.curve_fit function from the
SciPy Python package [
16]. Apparent lifetimes’ standard errors were used as weights for the fit. To estimate the uncertainty of the fit results, the square roots of the resulting covariance matrix’s diagonal elements was computed.
2.9. Simulation of Single-Molecule FRET Time
Traces
Generation of on- and off-state changes of the FRET signals proceeded in the same manner as simulation of state transition trajectories described previously [
17]. In short, durations of on-state and off-state were randomly drawn from exponential distributions with decay time parameters
and
. These durations were concatenated to form a trajectory.
was set according to eq.
1 with
, which is in the typical range of values determined from experiments,
, and
as indicated in
Section 3.2.
was fixed as
.
Next, the trajectories were sampled at fixed intervals
to simulate stroboscopic illumination for a duration , starting after a predefined time (
) to allow for transients to subside.
Figure 3a shows a sketch of the process. The number of consecutive samples in which a trajectory was in the on-state, corresponding the number of frames a FRET signal was visible in an experiment (termed
in
Section 3.1), was used to test our method
Section 3.2. The number of sampling points (see suppl. tables 1 and 2 for the respective values) was adjusted for each
to yield more trajectories present at the beginning or at the end of the measurement window, and fewer that fully lie within the window than in a typical experiment (see suppl. figs. 1–3) to further challenge our algorithm.
2.10. Microscopy Data Analysis
Extraction of single-molecule FRET time traces from raw microscopy images relied heavily on methods provided by the
sdt Python package [
18]. The
sdt.roi.ROI class allowed for defining the emission channels within raw images. The single-molecule localization algorithm [
19] implemented in the
sdt.loc.cg module was used to detect fluorescent beads for image registration as well as the single-molecule FRET signals. Signal intensities were measured by summing pixel intensity values in a region around their positions via the
sdt.brightness.from_raw_image function. Image registration was done using the
sdt.multicolor.Registrator class. The
trackpy Python package [
20] was utilized to perform single-molecule tracking. Each track was extended by 5 frames before the first and after the last frame by measuring the intensity at the first and last position of the track, respectively. This permitted employing a changepoint detection algorithm (
PELT [
21] as implemented in the
sdt.changepoint module) for stepwise bleaching analysis of smFRET signals.
All analysis steps are integrated into our software GUI application, which leverages functionality from the sdt.gui sub-package.
3. Results
This study presents a new approach for extraction of average lifetime estimates from single molecule FRET microscopy data. To exploit FRET for the determination of receptor–ligand interaction lifetimes, receptors need to be fluorescently labeled with one constituent of a FRET pair, while ligands need to carry the other constituent. Fluorophores as well as their attachment sites and -strategies have to be chosen such that their spatial separation is less than or roughly equal to the Förster radius
when receptor and ligand are bound.
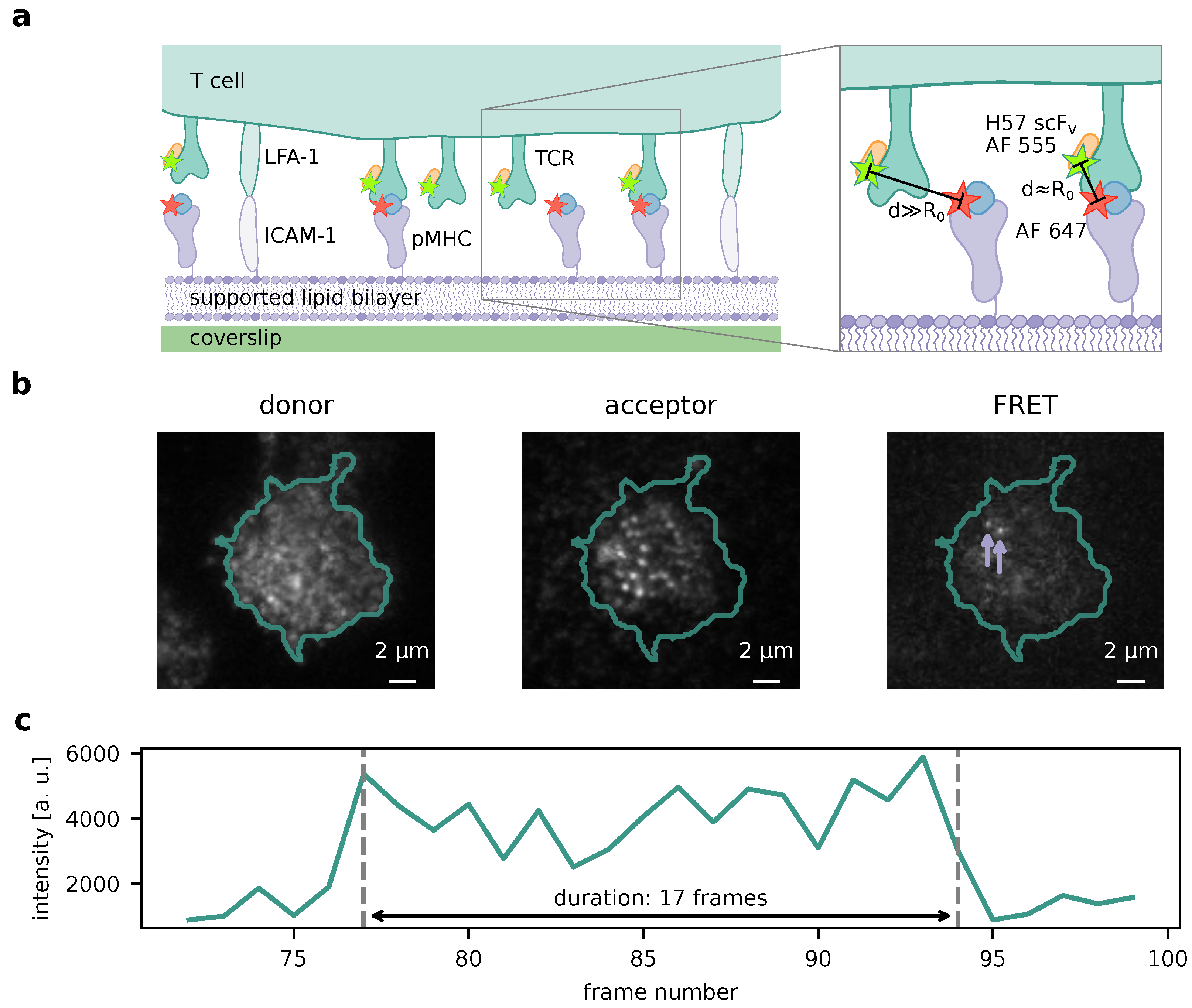
Figure 1a illustrates this for TCR and pMHC as interaction partners, for which bond lifetimes have been successfully measured [
8]. If binding events are rare enough, they appear as single-molecule FRET events (
Figure 1b), which can be traced over time and allow for measurement of their durations (
Figure 1c).
Single-molecule tracks may, however, not only end because of receptor–ligand unbinding, but also because of photobleaching of one of the fluorophores forming the FRET pair. Since labeling density is high in both the donor and the acceptor channel (
Figure 1b), it is not possible to separate the causes directly e.g. by means of alternating laser excitation [
22]. Instead, we rely on the observation that the interaction lifetime is independent of the fluorophore illumination, while the photobleaching rate is not. Hence, the true characteristic lifetime can be inferred from datasets recorded with different intervals between consecutive frames [
8].
In the following, we present
a mathematical model for accurate determination of binding lifetimes from single-molecule tracking data (
Section 3.1),
characterization of the model by evaluating simulated data and comparing the results to the known ground truth (
Section 3.2),
an efficient data analysis pipeline (
Section 3.3), which we implemented in an easy-to-use, free and open source software application, and
application of said analysis pipeline to experimental data from TCR–pMHC pairs with lifetimes spanning several orders of magnitude (
Section 3.4).
3.1. Mathematical Framework
We model the bond lifetimes
as exponentially distributed random variables,
[
8,
23]. Fluorophore survival times with respect to photobleaching
are also assumed to be exponentially distributed,
. Since these are competing processes, the apparent lifetimes
measured in the experiment are exponentially distributed as well,
.
When using stroboscopic illumination, i.e., fluorophores are only excited during the image acquisitions, but not in between, the photobleaching rate
is proportional to the acquisition rate and therefore inversely proportional to the interval
between images. Mathematically,
for some constant
. The characteristic apparent lifetime
is thus a function of
,
where
is the characteristic bond lifetime. With apparent lifetimes
inferred from tracking data recorded at various
,
(and also
) can therefore be determined by fitting eq.
1.
For fixed , the single-molecule FRET tracking experiment yields a set of frame counts , which is defined as the number of frames the i-th bound ligand’s signal is counted within the recording window. In order to accurately determine the characteristic apparent lifetime , the following aspects need to be considered:
The exact moment of unbinding is unknown. If a signal is detectable until the j-th frame (), whereafter it disappears, the unbinding / bleaching time point lies between the j-th and the -th frame. A signal can also be still present at the end of a recording.
The exact time of binding is unknown. If a signal first appears in the j-th frame (), the time of binding lies between the -th and the j-th frame. A signal can also be already present at the start of a recording.
To distinguish actual single-molecule FRET tracks from short-lived noise e.g. due to cellular background fluctuations, one may wish to introduce a minimum length for tracks to analyze.
We employ
survival analysis to take these issues into account. For a comprehensive introduction to survival analysis, refer e.g. to Klein and Moeschberger [
24]. In order to infer the
, we perform maximum likelihood estimation. To this end, the log-likelihood
is maximized with respect to
, where
is the likelihood contribution of the
i-th single-molecule track’s frame count
.
denotes the characteristic decay time (i.e., the inverse of the rate parameter) of the exponential distribution.
Considering only the first aspect in the list above, a track detected for a time
yields
if the signal ceased before the end of the observation window and
otherwise.
Receptor and ligand generally start to interact an unknown time
before the first exposure (aspect #2; see also
Figure 2), which needs to be added to
(i.e.,
) in the equations above. The fact that the receptor–ligand pair would escape detection if its binding duration was shorter than
needs to be accounted for by probabilities being conditional on
. The
i-th likelihood contribution accommodating both aspects #1 and #2 is thus given by
for tracks disappearing during the measurement and
for tracks still present at the end.
Since the exponential distribution is memory-less, i.e.,
, these expressions reduce to
respectively.
The minimum frame count
(aspect #3) is taken into account by modifying the expressions above to be conditional on
. The full expressions for the
i-th likelihood contribution are therefore as follows:
for tracks terminating before the end of the observation window and
for tracks outliving the observation window.
The exponential distribution’s cumulative distribution function (CDF)
can be used together with the identity
to arrive at the functional dependencies
Performing experiments employing a recording interval
yields a set of values
which, using the expressions above, define the (log) likelihood function
. Numerical methods (see
Section 2.7) allow for maximizing this likelihood with respect to
, i.e., finding the value
for which
is maximal.
is the maximum likelihood estimate of the characteristic apparent lifetime
. Repeating this process for multiple values of
yields pairs
, which permit inference of the interaction lifetime
by fitting eq.
1.
3.2. Characterization Using Simulated Data
In order to test and characterize our algorithm, we simulated smFRET time traces and analyzed them as described in the previous section. The traces were generated by switching molecules between dark and bright states with exponentially distributed lifetimes characterized by
and
, respectively. The traces were sampled at a predefined number of discrete time points separated by
after an initial time to simulate the image acquisition process. This process is depicted in
Figure 3a. For further information as well as the numerical parameter values used, refer to methods
Section 2.9.
Figure 3.
Proof of concept using simulated data. (a)
Simulation of time traces (green). FRET signals are switched on and off with exponentially distributed lifetimes. Microscopy image acquisitions correspond to sampling the time trace at time points separated by an interval
, illustrated by the vertical lines. Possible scenarios taken into account via survival analysis (
Section 3.1) are indicated by the annotated arrows. (b)
Inference of . The histogram depicts simulated track lengths for fixed
for three scenarios: i) signals are present at the start of the recording window, ii) at the end of the recording window, iii) they lie fully within the recording window. The probability density function (PDF) derived using survival analysis (green line) is virtually indistinguishable from the true PDF (red dotted line). Analysis utilizing conventional maximum likelihood estimation (MLE, see also
Section 2.7) yields a clear deviation in the PDF (purple line) and a value for
which is too low. (c)
Determination of the binding lifetime . Datasets as in (b) were simulated and analyzed for different
. Red crosses mark the simulated values for
, green dots indicate values determined using survival analysis, and purple dots denote values inferred via conventional MLE. Eq.
1 was fit to the resulting
, of which results are shown as dotted red line, green line, and purple line, respectively. The shaded areas indicate corresponding error margins.
Figure 3.
Proof of concept using simulated data. (a)
Simulation of time traces (green). FRET signals are switched on and off with exponentially distributed lifetimes. Microscopy image acquisitions correspond to sampling the time trace at time points separated by an interval
, illustrated by the vertical lines. Possible scenarios taken into account via survival analysis (
Section 3.1) are indicated by the annotated arrows. (b)
Inference of . The histogram depicts simulated track lengths for fixed
for three scenarios: i) signals are present at the start of the recording window, ii) at the end of the recording window, iii) they lie fully within the recording window. The probability density function (PDF) derived using survival analysis (green line) is virtually indistinguishable from the true PDF (red dotted line). Analysis utilizing conventional maximum likelihood estimation (MLE, see also
Section 2.7) yields a clear deviation in the PDF (purple line) and a value for
which is too low. (c)
Determination of the binding lifetime . Datasets as in (b) were simulated and analyzed for different
. Red crosses mark the simulated values for
, green dots indicate values determined using survival analysis, and purple dots denote values inferred via conventional MLE. Eq.
1 was fit to the resulting
, of which results are shown as dotted red line, green line, and purple line, respectively. The shaded areas indicate corresponding error margins.

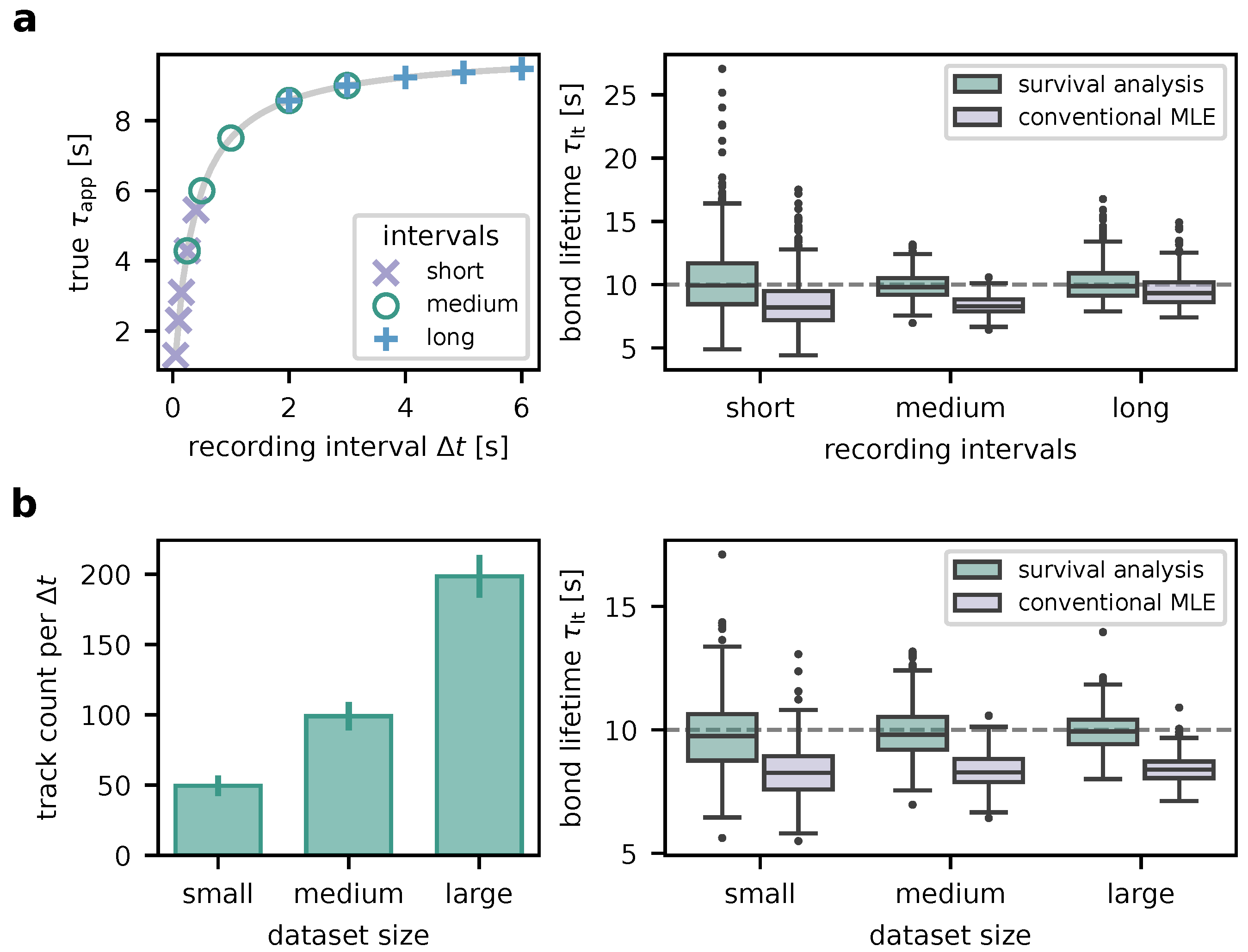
As a proof of concept, we generated a large dataset (about 2500 FRET traces per
, i.e., 25 times the size of a typical experimental dataset) to keep the influence of random fluctuations low.
Figure 3b shows a histogram of the track lengths at a single
. Employing survival analysis, we were indeed able to infer an accurate estimate
of the true value
. Notably, attempting to determine the apparent lifetime solely from frame counts—i.e., disregarding the fact that traces may live beyond the observation window (conventional MLE, see
Section 2.7 for details)—can lead to a bias towards shorter lifetimes (
). From datasets generated at several different
(
Figure 3c), an accurate estimate for the characteristic interaction lifetime
could be obtained if apparent lifetimes
were inferred using survival analysis (
). Failure of properly handling traces exceeding the observation window is cause for underestimation (
)).
Next, we wanted to explore the impact of the choice of recording intervals
with respect to the a priori unknown interaction lifetime
. To this end, we simulated 500 experiments with ground truth
for each of three different sets of
and evaluated their results (
Figure 4a). To resemble the typical experimental dataset size, around 100 FRET time traces were generated for each
. The following interval sets were investigated:
short intervals: 0.05, 0.1, 0.15, 0.25, 0.4
. These lie on the steep left part of the
vs.
curve given by eq.
1.
medium intervals: 0.25, 0.5, 1.0, 2.0, 3.0, which cover the bend of the vs. curve.
long intervals: 2, 3, 4, 5, 6, which lie on the flat right part of the vs. curve.
As shown in the right panel of
Figure 4a, medium intervals yield the most precise results (
mean ± std across the simulated experiments), followed by long intervals (
). Short intervals also permit accurate results, however with worse precision (
). Also note that the fit failed (i.e., did not converge, yielded infinite covariance or a large negative value) for short intervals in two and for long intervals in one of the simulated experiments. Of note, analysis without proper consideration of traces outliving the observation window (conventional MLE) leads to systematic bias towards lower values (
,
, and
mean ± std for the short, medium, and long interval sets, respectively).
In order to determine the effect of the size of the experimental dataset on the results (
Figure 4b), we generated 500 experiments consisting of small (about 50 smFRET traces per
), medium (around 100 traces; typical experiment), and large datasets (approximately 200 traces). Other parameters were the same as with medium intervals above. Increasing the dataset size increases the precision (small:
, medium:
, large:
). Notably, hardly any outliers occur in medium-sized and large datasets. Especially for the larger datasets yielding higher-precision results, the necessity of proper survival analysis becomes very evident. Without, the interaction lifetime is substantially underestimated (
,
, and
for the small, medium, and large datasets, respectively).
3.3. Data Analysis Pipeline
As a prerequisite to the determination of interaction lifetimes, smFRET tracking data needs to be extracted from microscopy recordings. To this end, we developed an analysis pipeline, which we implemented in an efficient and straightforward software application [
10]. The software features permits input of the excitation sequence, definition of the emission channels, optional image registration using fiducial markers, bleed-through correction, and single-molecule localization and tracking. As a novum the single-molecule inspection is assisted by automatic detection of photobleaching events and various filter settings. Fully automated single-molecule analysis can yield false tracks due to cellular background, accumulation of ligands by the cells, etc. We therefore found manual inspection in addition to parametric filtering indispensable. The final filtered datasets are subjected to the presented survival analysis. For further information and detailed instructions, please refer to the software’s manual.
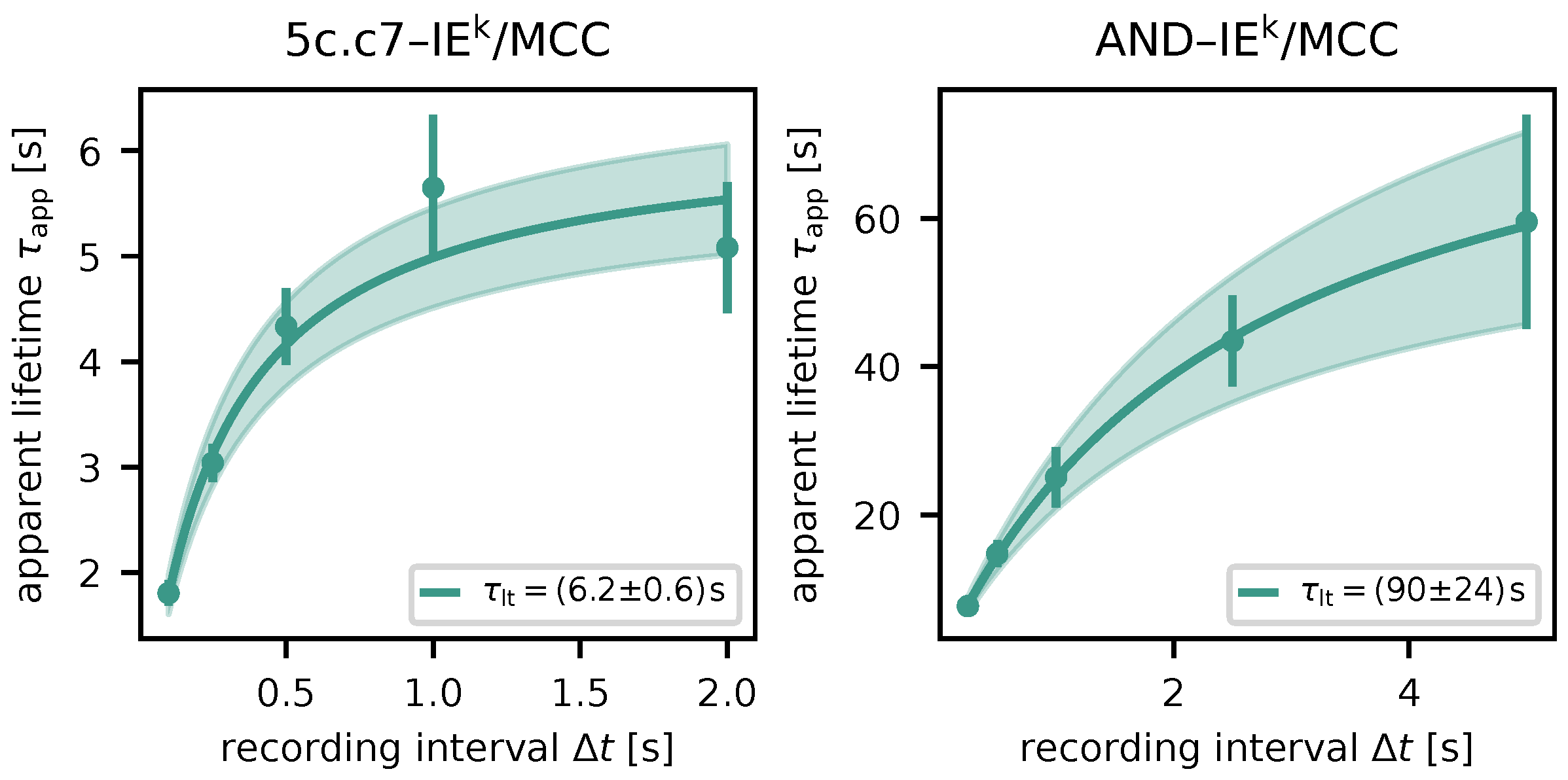
3.4. Experimental application: TCR–pMHC interaction lifetimes
To challenge our analysis pipeline experimentally, we recorded datasets of two different TCR–pMHC pairs (5c.c7–IE
k/MCC, AND–IE
k/MCC), for which previous studies [
8,
23] have shown interaction lifetimes of different orders of magnitude.
Figure 5.
Lifetime measurements for different TCR–pMHC pairs. Apparent lifetimes
for respective recording intervals
are displayed as dots (maximum likelihood estimate via survival analysis) with error bars (standard error of the estimate). The solid line shows the result of fitting eq.
1, the shaded area indicates the uncertainty. Left: 5c.c7 TCR, IE
k/MCC pMHC; right: AND TCR, IE
k/MCC pMHC.
Figure 5.
Lifetime measurements for different TCR–pMHC pairs. Apparent lifetimes
for respective recording intervals
are displayed as dots (maximum likelihood estimate via survival analysis) with error bars (standard error of the estimate). The solid line shows the result of fitting eq.
1, the shaded area indicates the uncertainty. Left: 5c.c7 TCR, IE
k/MCC pMHC; right: AND TCR, IE
k/MCC pMHC.
As in aforementioned articles, we used functionalized glass-supported lipid bilayers (SLB) in lieu of antigen-presenting cells (
Section 2.3), which allowed for precise control of protein composition and the use of total internal reflection fluorescence (TIRF) microscopy. T cells with fluorescently labeled TCR seeded onto the SLB would attach and bind to the pMHC (
Figure 1). These interactions were monitored via smFRET.
The long-lived AND–IE
k/MCC pairs entailed some experimental challenges. We found a maximum usable recording interval of about 5 seconds. For longer
, signals moved further between frames than the tracking algorithm could cope. Additionally, many signals migrated towards clusters so that they were not distinguishable until the end. Therefore, despite the insights from
Section 3.2, our measurements were restricted to the steep, left part of the
vs.
curve.
We determined characteristic lifetimes of
and
for 5c.c7–IE
k/MCC and AND–IE
k/MCC, respectively. These are moderately longer than previously published values (
and
) [
23], which were derived by measuring the duration of TCR-mediated pMHC immobilization. The difference may be explained by the fact that latter data were not analyzed by using survival analysis.
4. Discussion
Single-molecule FRET assays are a very sensitive tool for the investigation of in situ receptor-ligand interactions in 2D as they can capture fast and rare dynamics in even the most complex cellular environments. Failing to account for side effects of the image recording process and the limited observation time can lead, however, to a substantial underestimation of the average bond lifetime. We here present an improved and robust statistical analysis tool for the lifetime estimation of receptor–ligand interactions determined by smFRET assay. In order to show the accuracy of the new analysis pipeline, we provide simulated and experimental data and compare the results with the standardized algorithm currently in use by the community.
In particular, we created a new analysis algorithm which robustly estimates apparent (i.e., uncorrected for photobleaching) lifetimes at different frame rates to accurately infer the interaction lifetime. The new feature is based on the mathematical framework of survival analysis. This approach outperforms the original, conventional MLE algorithm, which utilizes solely the number of frames in which the individuals smFRET traces are visible, resulting in better accuracy and robustness.
To test the limits of our method, we used simulated smFRET traces and compared the analysis pipeline’s output to the ground truth. We show that the best results are obtained with intermediate recording delays which cover the curved part of the graph of . If no a priori estimate of the interaction lifetime is available, it is better to err on the long side. However, there are some experimental challenges in this regard: If is greater than , few events will be present in more than one consecutive frame since the track lengths are exponentially distributed. Additionally, for large smFRET signals may move by a substantial distance between frames, which can go beyond the single-molecule tracking algorithm’s capabilities. Furthermore, aggregation of molecules can also be a challenge, e.g. during formation of an immunological synapse, in which case it becomes difficult to identify the appearance and disappearance of individual signals. The latter may, to some extent, be mitigated by labeling only a fraction of the receptors or ligands; on the other hand, this reduces the overall dataset size. Comparing the results from simulated datasets of different sizes revealed that in order to obtain reliable results, one should gather at least 50 smFRET traces for each .
Our mathematical model (
Section 3.1) relies on interaction lifetimes being exponentially distributed. This implies exponential distribution of the apparent lifetimes, for whose likelihood we can specify an analytic expression due to being memory-less. For systems with non-exponentially distributed lifetimes, the situation would be more complex. First, the distribution of apparent lifetimes and a relation between
,
, and
akin to eq.
1 needed to be derived. Additionally, the survival analysis of individual
may require an iterative approach [sec. 5.3 [
24].
The survival analysis approach is not only applicable to single-molecule FRET data, but to all data based on counting the number of frames in which a single-molecule signal is visible during a limited observation time. For instance, it may be advantageous to employ our method when inferring receptor–ligand interaction lifetimes via receptor-mediated ligand immobilization [
23].
We chose to present our new approach using the TCR–pMHC interaction as an example. However, the experimental and analytical pipeline can easily be transferred to any receptor-ligand pair of interest if respective FRET binding pairs have been established. The binding partners need to be site-specifically decorated with a FRET donor and acceptor molecule within a distance of 10 nm (that means the actual molecular distance needs to be in range of the Förster radius ) in order to yield well-defined single-molecule FRET signals. The fluorescent labels can either be introduced via bio-orthogonal conjugation directly to the protein backbone or via antibody labeling (preferably with smaller antibody fragments such as Fab fragments or single-chain fragments, scFV). The distance of the fluorophores can be predicted either via utilizing existent crystal structures of the target proteins or via experimental trial. We recommend the use of newer generation fluorophores such as Alexa Fluor 647 and Alexa Fluor 555, as such FRET pairs allow the tracking of longer FRET trajectories before photobleaching. The imaging system needs to be capable of detecting and tracking single molecules on a planar surface over varying exposure intervals.
To support the experimentalist in dealing with large data amounts, we created a software suite for reproducible filtering of the trajectories as well as decreasing subjective bias in choosing single molecule FRET events. The standard procedure so far was to manually inspect the recorded video sequences for FRET signal appearance and disappearance and noting down the number of frames. The chosen trajectories could not be reinspected and shifting perspective and sensitivity of the analyst in choosing trajectories led to biased data selection. Re-analyzing the data set by a second experimentalist often led to different results. In the presented software, datasets and respective filters can now be reinspected and all chosen and rejected data points can be used for subsequent evaluation. Additionally, basic parameters (such as average intensity of the signal, and background level) associated with each trajectory can be used for automatic filtering. Intensity profiles of the automatically detected single molecules are displayed for a less biased evaluation of single arrival and departure steps of the signal (which are hallmarks of single-molecule signals), disturbance within the track by other single molecules or intensity fluctuations (which could indicate rebinding events), or the premature end of the trajectory because of receptor clustering. Furthermore, the use of automated algorithms (localization, tracking, intensity step detection, …) drastically decreases the amount of time required for analysis. The software is published under a permissive open-source license and thus freely available [
10].
The investigation of the bond lifetime of a TCR–pMHC pair is a meaningful parameter for a multitude of scientific questions and, therefore, of interest for a wide scientific community. The threshold time an interaction needs to trigger productive downstream signaling is an important optimization parameter for artificial T cell receptors and equivalents [
25,
26]. For a small subset of mainly transgenic TCRs these interaction dynamics have been characterized and are used as model systems for other biological questions, yet more and more researchers inquire the binding dynamics of the natural occurring TCR repertoire as well. From lessons from the kinetic proofreading model [
27] we know that downstream cascades initiate within a few seconds of pMHC ligation [
1,
28]. Consequently, the TCR decision process for discrimination of varying ligand potencies happens within this small time window. The direct observation of ligand binding events is, therefore, a very informative tool for understanding T cell immune surveillance (i.e., scanning parameters, triggering thresholds, as well as signaling consequences).
Author Contributions
conceptualization: J.G. and L.S.; data curation: J.G. and L.S.; formal analysis: L.S.; funding acquisition: G.S., J.G, and J.H.; investigation: J.G., L.S., and V.M.; methodology: F.K., J.G., J.W., L.S, R.P., R.O., V.M..; project administration: G.S., J.G, and J.H.; resources: F.K., J.G., J.W., R.O., R.P., and V.M.; software: L.S.; supervision: G.S., J.G., and J.H.; validation: J.G. and L.S.; visualization: L.S.; writing—original draft preparation: J.G. and L.S.; writing—review and editing: G.S., J.G., J.H., L.S., and L.T.-H. All authors have read and agreed to the published version of the manuscript.
Funding
The study received support from the Vienna Science and Technology Fund (WWTF) project LS13-030 (J.G., L.S., F.K., J.H., G.S.) and FWF project P32307-B (L.S., J.G., G.S.). Additional funding was provided by predoctoral fellowships from the Boehringer Ingelheim Fonds (R.P.).
Institutional Review Board Statement
Mouse breeding and euthanasia were evaluated by the ethics committees of the Medical University of Vienna and approved by the Federal Ministry of Science, Research and Economy, BMWFW (BMWFW-66.009/0378-WF/V/3b/2016). All procedures to isolate lymphocytes, splenocytes and bone marrow from 8-12 weeks old gender-mixed mice were performed in accordance to Austrian law (Federal Ministry for Science and Research, Vienna, Austria), the guidelines of the Federation of Laboratory Animal Science Associations (FELASA), which match those of Animal Research Reporting In Vivo Experiments (ARRIVE), and the guidelines of the ethics committees of the Medical University of Vienna. Breeding and keeping of AND-TCR transgenic mice has been approved by the Government of Upper Bavaria, protocol 55.2-2532.Vet_02-21-4.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| APC |
antigen-presenting cell |
| FRET |
Förster resonance energy transfer |
| H57-scFV
|
H57 antibody-derived single-chain fragment |
| MHC |
major histocompatibility complex |
| MLE |
maximum likelihood estimation |
| SLB |
supported lipid bilayer |
| TCR |
T cell receptor |
| pMHC |
peptide-loaded major histocompatibility complex |
| smFRET |
single-molecule Förster Resonance Energy Transfer |
References
- Govern, C.C.; Paczosa, M.K.; Chakraborty, A.K.; Huseby, E.S. Fast on-rates allow short dwell time ligands to activate T cells. Proceedings of the National Academy of Sciences 2010, 107, 8724–8729. [Google Scholar] [CrossRef] [PubMed]
- Aleksic, M.; Dushek, O.; Zhang, H.; Shenderov, E.; Chen, J.L.; Cerundolo, V.; Coombs, D.; van der Merwe, P.A. Dependence of T Cell Antigen Recognition on T Cell Receptor-Peptide MHC Confinement Time. Immunity 2010, 32, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Acuto, O. T-cell virtuosity in ‘‘knowing thyself”. Frontiers in Immunology 2024, 15. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Ge, C.; Jothikumar, P.; Yuan, Z.; Liu, B.; Bai, K.; Li, K.; Rittase, W.; Shinzawa, M.; Zhang, Y.; Palin, A.; Love, P.; Yu, X.; Salaita, K.; Evavold, B.D.; Singer, A.; Zhu, C. A TCR mechanotransduction signaling loop induces negative selection in the thymus. Nature Immunology 2018, 19, 1379–1390. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.; Narayanan, S.; Liu, B.; Birnbaum, M.; Kruse, A.; Bowerman, N.; Chen, W.; Levin, A.; Connolly, J.; Zhu, C.; Kranz, D.; Garcia, K. T Cell Receptor Signaling Is Limited by Docking Geometry to Peptide-Major Histocompatibility Complex. Immunity 2011, 35, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Klotzsch, E.; Schütz, G. Improved Ligand Discrimination by Force-Induced Unbinding of the T Cell Receptor from Peptide-MHC. Biophysical Journal 2013, 104, 1670–1675. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Zou, M.; Shao, Y.; Wu, H.; Wu, H.; Wang, X. Two-dimensional measurements of receptor-ligand interactions. Frontiers in Molecular Biosciences 2023, 10. [Google Scholar] [CrossRef] [PubMed]
- Huppa, J.B.; Axmann, M.; Mörtelmaier, M.A.; Lillemeier, B.F.; Newell, E.W.; Brameshuber, M.; Klein, L.O.; Schütz, G.J.; Davis, M.M. TCR-peptide-MHC interactions in situ show accelerated kinetics and increased affinity. Nature 2010, 463, 963–967. [Google Scholar] [CrossRef]
- Axmann, M.; Huppa, J.; Davis, M.; Schütz, G. Determination of Interaction Kinetics between the T Cell Receptor and Peptide-Loaded MHC Class II via Single-Molecule Diffusion Measurements. Biophysical Journal 2012, 103, L17–L19. [Google Scholar] [CrossRef]
- Schrangl, L. smfret-bondtime: Quantification of receptor–ligand interaction times via single-molecule FRET, 2024. [CrossRef]
- Huppa, J.B.; Gleimer, M.; Sumen, C.; Davis, M.M. Continuous T cell receptor signaling required for synapse maintenance and full effector potential. Nature Immunology 2003, 4, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Platzer, R.; Hellmeier, J.; Göhring, J.; Perez, I.D.; Schatzlmaier, P.; Bodner, C.; Focke-Tejkl, M.; Schütz, G.J.; Sevcsik, E.; Stockinger, H.; Brameshuber, M.; Huppa, J.B. Monomeric agonist peptide/MHCII complexes activate T‐cells in an autonomous fashion. EMBO reports 2023, 24, e57842. [Google Scholar] [PubMed]
- Roe, M.; Lemasters, J.; Herman, B. Assessment of Fura-2 for measurements of cytosolic free calcium. Cell Calcium 1990, 11, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Göhring, J.; Kellner, F.; Schrangl, L.; Platzer, R.; Klotzsch, E.; Stockinger, H.; Huppa, J.B.; Schütz, G.J. Temporal analysis of T-cell receptor-imposed forces via quantitative single molecule FRET measurements. Nature Communications 2021, 12, 2502. [Google Scholar] [CrossRef]
- Davidson-Pilon, C. lifelines, survival analysis in Python, 2023. [CrossRef]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; van der Walt, S.J.; Brett, M.; Wilson, J.; Millman, K.J.; Mayorov, N.; Nelson, A.R.J.; Jones, E.; Kern, R.; Larson, E.; Carey, C.J.; Feng, Y.; Moore, E.W.; VanderPlas, J.; Laxalde, D.; Perktold, J.; Cimrman, R.; Henriksen, I.; Quintero, E.A.; Harris, C.R.; Archibald, A.M.; Ribeiro, A.H.; Pedregosa, F.; van Mulbregt, P.; Vijaykumar, A.; Bardelli, A.P.; Rothberg, A.; Hilboll, A.; Kloeckner, A.; Scopatz, A.; Lee, A.; Rokem, A.; Woods, C.N.; Fulton, C.; Masson, C.; Häggström, C.; Fitzgerald, C.; Nicholson, D.A.; Hagen, D.R.; Pasechnik, D.V.; Olivetti, E.; Martin, E.; Wieser, E.; Silva, F.; Lenders, F.; Wilhelm, F.; Young, G.; Price, G.A.; Ingold, G.L.; Allen, G.E.; Lee, G.R.; Audren, H.; Probst, I.; Dietrich, J.P.; Silterra, J.; Webber, J.T.; Slavič, J.; Nothman, J.; Buchner, J.; Kulick, J.; Schönberger, J.L.; de Miranda Cardoso, J.V.; Reimer, J.; Harrington, J.; Rodríguez, J.L.C.; Nunez-Iglesias, J.; Kuczynski, J.; Tritz, K.; Thoma, M.; Newville, M.; Kümmerer, M.; Bolingbroke, M.; Tartre, M.; Pak, M.; Smith, N.J.; Nowaczyk, N.; Shebanov, N.; Pavlyk, O.; Brodtkorb, P.A.; Lee, P.; McGibbon, R.T.; Feldbauer, R.; Lewis, S.; Tygier, S.; Sievert, S.; Vigna, S.; Peterson, S.; More, S.; Pudlik, T.; Oshima, T.; Pingel, T.J.; Robitaille, T.P.; Spura, T.; Jones, T.R.; Cera, T.; Leslie, T.; Zito, T.; Krauss, T.; Upadhyay, U.; Halchenko, Y.O.; Vázquez-Baeza, Y.; Contributors, S. . SciPy 1.0: fundamental algorithms for scientific computing in Python. Nature Methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Schrangl, L.; Göhring, J.; Schütz, G.J. Kinetic analysis of single molecule FRET transitions without trajectories. The Journal of Chemical Physics 2018, 148, 123328, [https://pubs.aip.org/aip/jcp/article-pdf/doi/10.1063/1.5006038/13346113/123328_1_online.pdf]. [Google Scholar] [CrossRef] [PubMed]
- Schrangl, L. sdt-python: Python library for fluorescence microscopy data analysis, 2024.
- Gao, Y.; Kilfoil, M.L. Accurate detection and complete tracking of large populations of features in three dimensions. Opt. Express 2009, 17, 4685–4704. [Google Scholar] [CrossRef] [PubMed]
- Allan, D.B.; Caswell, T.; Keim, N.C.; van der Wel, C.M.; Verweij, R.W. soft-matter/trackpy: v0.6.2, 2024. [CrossRef]
- Killick, R.; Fearnhead, P.; Eckley, I.A. Optimal Detection of Changepoints With a Linear Computational Cost. Journal of the American Statistical Association 2012, 107, 1590–1598. [Google Scholar] [CrossRef]
- Kapanidis, A.N.; Lee, N.K.; Laurence, T.A.; Doose, S.; Margeat, E.; Weiss, S. Fluorescence-aided molecule sorting: Analysis of structure and interactions by alternating-laser excitation of single molecules. Proceedings of the National Academy of Sciences 2004, 101, 8936–8941. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, G.P.; Pielak, R.M.; Smoligovets, A.A.; Lin, J.J.; Groves, J.T. Direct single molecule measurement of TCR triggering by agonist pMHC in living primary T cells. eLife 2013, 2, e00778. [Google Scholar] [CrossRef] [PubMed]
- Klein, J.P.; Moeschberger, M.L. Survival Analysis, 2 ed.; Statistics for Biology and Health, Springer New York, NY, 2003. [CrossRef]
- Banik, D.; Hamidinia, M.; Brzostek, J.; Wu, L.; Stephens, H.M.; MacAry, P.A.; Reinherz, E.L.; Gascoigne, N.R.J.; Lang, M.J. Single Molecule Force Spectroscopy Reveals Distinctions in Key Biophysical Parameters of αβ T-Cell Receptors Compared with Chimeric Antigen Receptors Directed at the Same Ligand. The Journal of Physical Chemistry Letters 2021, 12, 7566–7573. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Kolawole, E.M.; Chan, W.; Feng, Y.; Yang, X.; Gee, M.H.; Jude, K.M.; Sibener, L.V.; Fordyce, P.M.; Germain, R.N.; Evavold, B.D.; Garcia, K.C. Tuning T cell receptor sensitivity through catch bond engineering. Science 2022, 376, eabl5282, [https://www.science.org/doi/pdf/10.1126/science.abl5282]. [Google Scholar] [CrossRef] [PubMed]
- McKeithan, T.W. Kinetic proofreading in T-cell receptor signal transduction. Proceedings of the National Academy of Sciences 1995, 92, 5042–5046, [https://www.pnas.org/doi/pdf/10.1073/pnas.92.11.5042]. [Google Scholar] [CrossRef]
- Dushek, O.; van der Merwe, P.A. An induced rebinding model of antigen discrimination. Trends in Immunology 2014, 35, 153–158. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).