1. Introduction
The first plastic was invented in 1860 (celluloid) and the beginning of the global plastic industry was marked in 1907 (bakelite). After 1945, plastic production increased exponentially and the problem of plastic solid waste (PSW) has reached alarming proportions. By the end of 2015, a staggering 6300 million metric tons (Mt) of PSW had been reached of which only 9% was recycled, leaving more than 80% accumulating in landfills or in the natural environment [
1]. Worldwide plastic production is estimated to be around 1.1 billion tons by 2050 [
2]. This pollution, both on land and at sea waters, has peaked in a new era of environmental crisis, as plastics defy the natural processes of degradation, persisting for centuries and beyond [
3]. The plastic degradation rates range from 100 to 1000 years. Consequently, the first invented plastics may still exist in nature [
4].
There are many solutions to the problem of plastic waste pollution. Increasing the use of bioplastics that can be mineralized through compost could be a likely solution [
4]. Unfortunately, most plastics continue to be non-biodegradable. The European Union is targeting of 100% recycling and reutilization of these plastics by 2030 [
5].
Recycling involves the transformation of plastic waste into secondary materials that can be reintegrated into the system either by its direct use or by obtaining new compounds (organic intermediates or fuels). Through this approach, one can "close the loop" in the circular economy framework. An essential step before applying recycling processes is the efficient sorting of plastic waste. Mechanical recycling of plastics undergoes several stages including separating, grinding, washing, drying, regranulating, and compounding, and then the recyclates are converted into new product [
4]. Mechanical recycling is the cheapest process, but it needs very well sorted and cleaned waste.
Chemical recycling involves depolymerization (hydrolysis and glycolysis) or thermolysis (gasification or pyrolysis) to recover monomers and oligomers that can be used again in the synthesis of plastics, resins, adhesives, etc. [
6]. Sometimes new functional groups can be introduced by crosslinking, grafting and copolymerization to obtain secondary materials with high impact resistance, heat resistance, aging resistance, storage stability and rheological properties [
7].
Pyrolysis (thermal cracking of polymer waste in inert atmosphere) can produce valuable fuels. In particular, gaseous fuels can be used to power engines connected to electric generators or in boilers without special treatments of the gases obtained [
8].
In the case of thermal recycling, the aim is to burn the waste for its conversion into thermal or electrical energy [
9] The process takes place at a temperature of approx. 850
0C and effective methods of reducing CO or NOx emissions can be applied [
10].
In terms of sustainability, converting plastic trash into fuels or any other better option, individual monomers, is a far more environmentally friendly waste management option than landfill dumping [
11]. It is difficult to determine which of these processes is the most effective, a better approach is to select the method depending on the type of plastic waste that can be sorted. The most important aspect is to avoid the proliferation of plastic waste in the environment to the greatest extent.
1.1. Plastics
Plastics can be classified according to several criteria: origin, structure, temperature behavior, etc. Natural polymers (lignin, chitin, starch) have been known since prehistoric times. Synthetic polymers (polyethylene, polystyrene, nylon, etc.) are obtained from petrochemical sources. Semi-synthetic plastics are modified natural polymers (like cellulose based plastics) [
4].
Apart from the above traditional classifications, the Society of the Plastic Industry (SPI) introduced a number-coding system for plastics. In this classification, numbers from 1 to 7 have been assigned for different plastic types in the following order: 1 – for polyethylene terephthalate (PET), 2 – for high-density polyethylene (HDPE), 3 – for polyvinyl chloride (PVC), 4 – for low-density polyethylene (LDPE), 5 – for polypropylene (PP), 6 – for polystyrene (PS), and 7 – for other types of plastics [
4].
Of these plastics, Low-Density Polyethylene (LDPE) and High-Density Polyethylene (HDPE) forms most of the plastic waste [
4]. For this reason, in this paper, LDPE was selected as the type of plastic subjected to pyrolysis.
LDPE is a type of low density plastic, around 0.910 - 0.940 g/ cm³ [
12]. LDPE also has a relatively low melting temperature, around 110 – 130 °C, and a softening point between 85 - 120 °C, which makes it easy to process and shape [
13].
1.2. Pyrolysis Mechanism of LDPE
1.2.1. Non-Catalytic Pyrolysis of LDPE
During the pyrolysis of LDPE, a process by which long chain polymers are decomposed into smaller molecules occurs. LDPE, like other plastics, undergoes thermal degradation when subjected to high temperatures in the absence of oxygen. This degradation process, known as pyrolysis, results in the formation of valuable products such as oils and gases, which serve as precursors for fuels and chemicals.
In the pyrolysis of Polyethylene (PE), the random scission of C-C bonds in the molecular chains formed free radicals, which was followed by the intermolecular or intramolecular hydrogen transfer and β-scission to produce secondary radicals and low molecular weight hydrocarbons [
14]. The main compounds that are obtained from the non-catalytic pyrolysis of PE are n-alkanes and 1-alkenes. At low temperatures, large hydrocarbons (C
20+) predominate, the higher the temperature, the higher the proportion of smaller hydrocarbons [
14]. Additionally, operating at higher temperatures may slightly enhance the formation of aromatics.
1.2.2. Catalytic Pyrolysis of LDPE
On the other hand, catalytic degradation of plastics, referred to as catalytic pyrolysis, has become a prominent approach for improving the quality of liquid oil derived from plastic degradation. Catalysts provide numerous benefits, including lowering the reaction temperature, increasing the proportion of light fractions, conversion of linear hydrocarbons to obtain isoalkanes, cycloalkanes and aromatic hydrocarbons [
15].
The pyrolysis of plastic waste primarily uses different kinds of catalysts, i.e., silica−alumina, zeolites, and fluid catalytic cracking (FCC) catalysts [
16]. The acidity of these catalysts has an impact on plastic pyrolysis to produce liquid oil. The generation of liquid oil will decrease as acidity increases [
16]. The mechanism of catalytic pyrolysis of various plastics is well described in the literature [
16,
17].
The products obtained from catalytic cracking possess distinctive characteristics compared to those from thermal conversion, with a higher aromatic content. Proper control of reaction conditions, particularly temperature, determines the extent to which thermal and catalytic cracking occur concurrently. In some cases, both thermal and catalytic cracking may occur simultaneously, making it challenging to delineate the boundary between the two processes under specific reaction conditions. Despite this complexity, catalytic cracking offers significant potential for producing hydrocarbons suitable for diesel and gasoline production, provided legal restrictions, particularly regarding aromatics [
16].
The solid catalyst can be applied either directly with feedstock in the reactor or on organic vapors produced in a separate catalyst chamber [
18].
Catalysts that treat the gaseous phase from the pyrolysis reactor are most often used, in this case the catalyst has a longer life, but the installation is more complicated because it contains two distinct reactors: the pyrolysis reactor and the catalytic reactor.
When the catalyst is used in direct contact with the melted plastic waste, some difficulties arise since the catalyst pores are blocked due to the sticky nature of the plastic feedstock [
19]. Also, the separation of the catalyst from the solid residue and its regeneration is quite difficult [
20,
21]. However, if a cheap catalyst, of the natural zeolite type, is used, this option can be used considering that the installation is much simplified, which will contain only one reactor where the catalytic pyrolysis of the product takes place.
The aim of this study is to investigate polyethylene pyrolysis using the catalyst into melted plastic. Such an approach allows for the utilization of a relatively simple setup, employing a single reactor. The system used is a batch-type installation with a column for separating the obtained liquid fractions. Initially, the effect of various catalysts on the pyrolysis process of clean PE granules was examined, including bentonite and treated natural zeolites. Subsequently, the pyrolysis of plastic waste obtained through household waste sorting was investigated, highlighting the catalytic effect.
2. Materials and Methods
All substances used were of the highest quality suitable for analytical, chromatographic, or spectroscopic purposes.
In these experiments, clean polyethylene granules (LDPE MI 2 from Romcolor Romania) and polyethylene foil waste from IRIDEX company (Bucharest), were used.
3 types of catalysts were used in the pyrolysis process: bentonite catalyst (Bentonite), with chemical formula Al2O34SiO2H2O, and two zeolite clinoptilolite catalyst (AMD1 and AMD4) with chemical formula (Na,K,Ca)2–3Al3(Al,Si)2Si13O36•12H2O.
The zeolite catalysts used, AMD1 and AMD4, were modified using the following procedures:
1. Physical treatments; 2. Thermal treatments; 3. Chemical treatments; 4. Treatments for selection and enrichment in the crystalline phase.
Thus, in the case of the AMD4 catalyst, compared to AMD1, the specific surface area increased by about 3 times, the acidity by about 60 times, and the porosity by about 200%. In
Table 1 are presented a few properties of the catalyst used.
2.1. Pyrolysis Installation
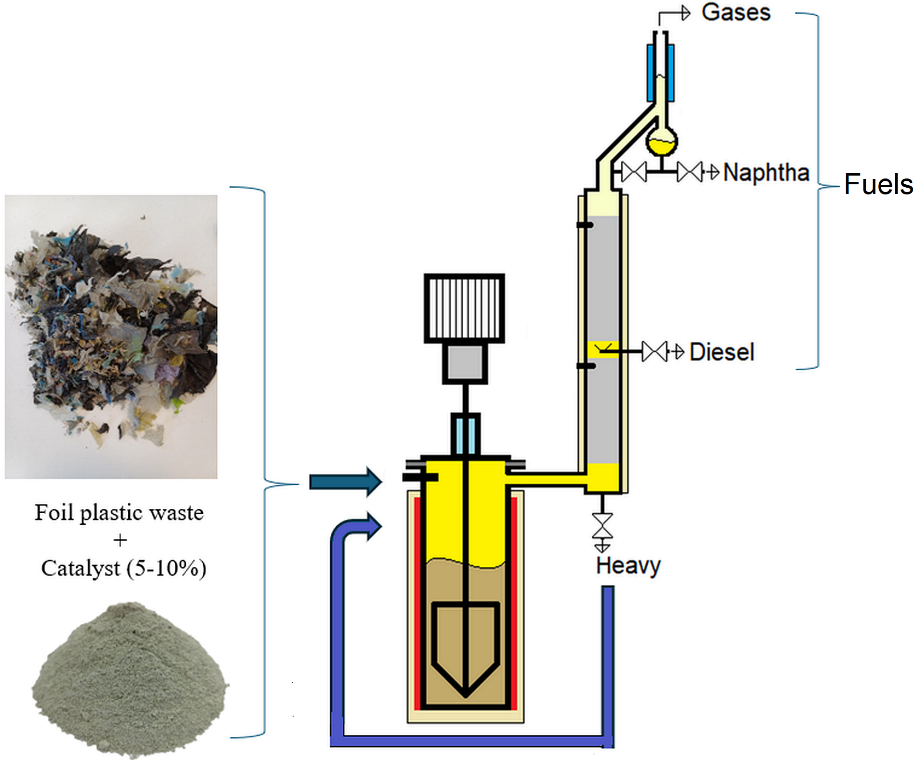
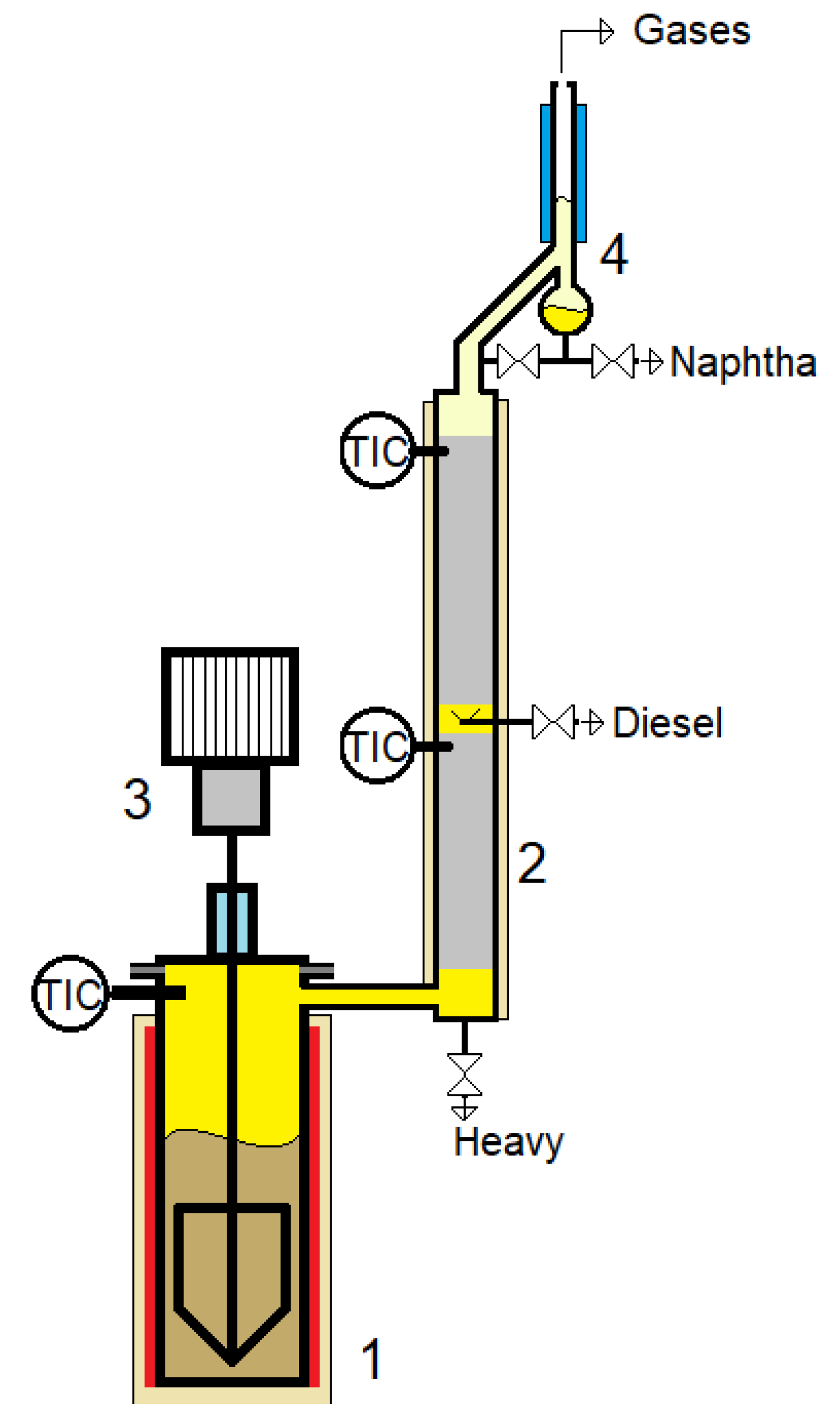
The equipment used was a prototype laboratory plant coupled with a distillation column. The experimental set-up was described in
Figure 1.
As shown in
Figure 1, a pyrolysis reactor with a supply flange and temperature control is coupled to a distillation column to ensure the separation of the fractions obtained. The stirring system, consisting of an electric engine with variable speed, speed reducer, sealing box, and stirrer, ensures proper mixing. The column, which features two sections, includes a distillation head equipped with a reflux divider to provide an appropriate reflux ratio. Heating elements are mounted on the column to simulate adiabatic operation. Three fractions – naphtha, diesel, and heavy fraction – are collected from the column. The collection rates of these fractions are manually set using fine taps. The maximum capacity of the pyrolysis reactor is 3000 g of polymer waste.
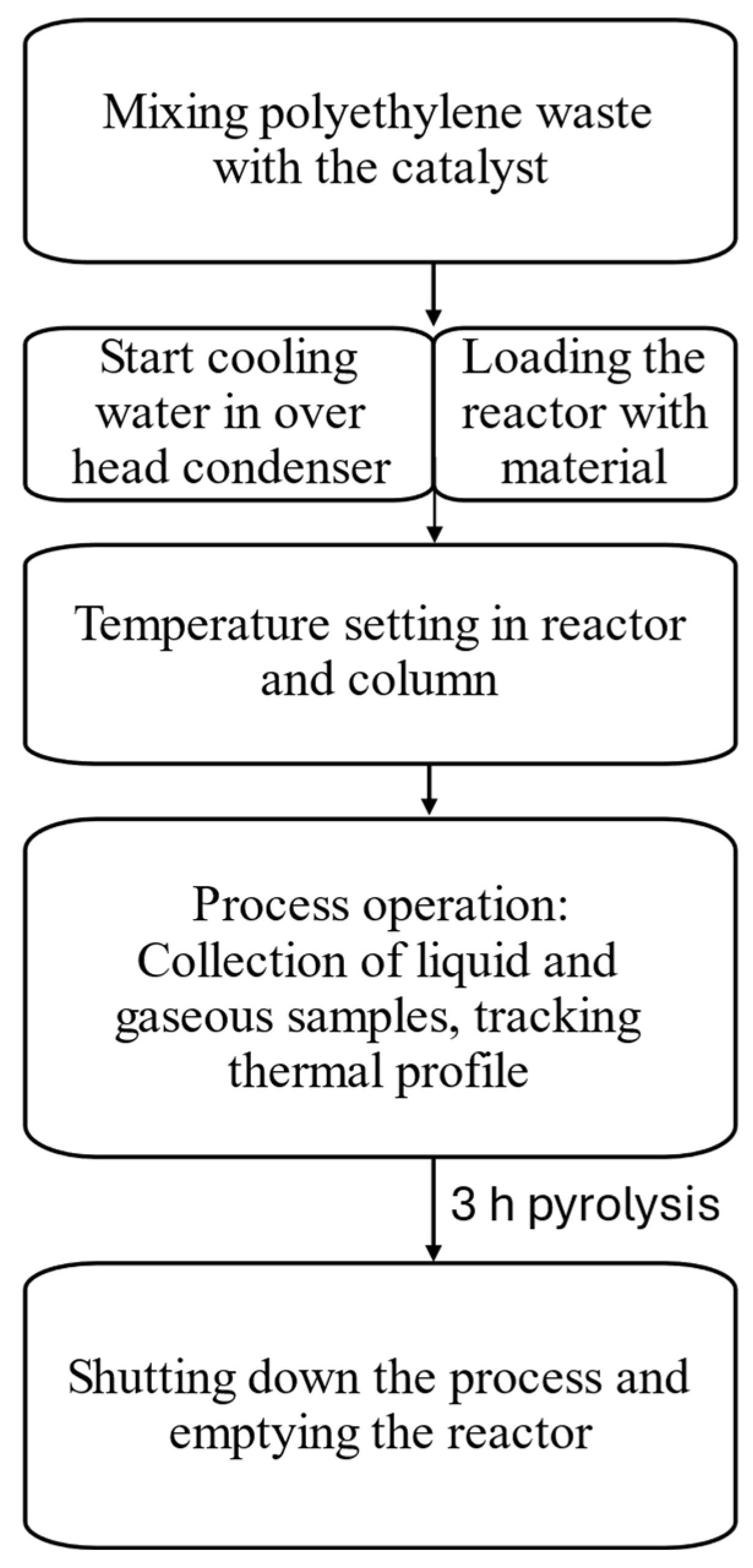
2.2. Pyrolysis Process
During the experiments, the reactor was loaded with 1000 g of polyethylene and different amounts (5/10/20 wt.%) of catalyst. The initial set temperature was 250 ℃. When the set temperature was reached, it was increased in steps of 50 ℃ degrees to a maximum of 400 ℃ degrees (see the diagram in
Figure 2). This method of operation was carried out to allow an adequate increase of the temperature to control the flow of liquid fractions in the distillation column.
2.3. Analysis of Gas and Liquid Samples
Gas samples were collected in Tedlar gas sampling bag during the process. Liquid samples were collected at the end of the pyrolysis process in glass flasks.
For the analysis of gaseous samples a Buck Scientific 910 gas chromatograph (GC) (Buck Scientific Instruments, Norwalk, USA) was used, with the following configuration: Two columns: (1) – molecular sieves 13X for gas separation of atmospheric gases – nitrogen, oxygen, CO, CO2, 6”x 0.53 mm and (2) – Silicagel 6” x 0.53 mm for separation of light hydrocarbons <C6; carrier gas - helium: pressure - 84 psi, helium flowrate – 30 mL/min (constant); multiport valve : loop – 1 mL, 40 ºC; temperature program: level 1 – 4 min, 40 ºC, ramp 1 – 20 ºC/min until 220 ºC, level 2 – 10 min, 220 ºC; detectors: FID – with methanizer at 350 ºC, hydrogen flowrate – 25 mL/min, air flowrate – 250 mL/min and TCD at 150 ºC, current – 255 mA.
Liquid fraction was analyzed on BUCK Scientific 910 set for liquid fraction analysis with configuration: Capillary column MTX-1, 60 m, 0.53 mm ID; temperature program: level 1 – 2.5 min, 40 ºC, ramp 1 – 5 ºC/min until 315 ºC, level 2 – 30 min, 315 °C; detectors: FID și DLCD.
GC-MS analysis was developed using an Agilent GC/MS system (Agilent Technologies, Santa Clara, USA) with the following configuration: Agilent 6890N chromatograph, coupled with MSD 5975 Inert XL detector, HP5–MS capillary column (30 m x 0.25 mm × 0.25 μm), constant temperature 17 °C, carrier gas He 5.5, pressure 0.11 psi, adsorption from gas bag on SPME fiber for 1 minute, desorption in chromatograph inlet at 150 °C for 30 seconds, inlet in splitless mode.
3. Results and Discussions
The experimental results from the pyrolysis of polyethylene (PE) waste, along with their subsequent fractionation using a distillation column, provide valuable insights into the feasibility and efficiency of this waste-to-energy conversion process.
3.1. Pyrolysis of LDPE Granules
3.1.1. Pyrolysis Production Yield
Pyrolysis product yield (oil, char, and gas) was estimated based on the weight obtained for the char and oil at the end of the pyrolysis process. The pyrolysis production yield of LDPE granules (1000 g) is shown in
Table 2.
The process yielded three distinct liquid fractions - naphtha, diesel and heavy paraffinic residue, alongside solid and gaseous fraction. The quantitative analysis yields for non-catalytic pyrolysis on each fraction: liquid fraction 43.4 % solid residue 18.3 % and gaseous 38.3 %. based on the initial 1000 g of PE foil. Pyrolysis of PE granules using bentonite catalyst resulted in gas production of 48.1 % and 51.9 % of liquid fraction. In this case, the solid residue contains only the catalyst (200 g). indicating a complete pyrolysis process of the LDPE. The obtained results are similar to the literature information that indicates the role of the catalyst: “The catalyst's acidity and microporous crystalline structure enhance the process of cracking and isomerization, promoting the breakdown of larger molecules into smaller ones, resulting in higher yields of gaseous and liquid products”[
22].
Utilizing 200 g of AMD1 catalyst per 1000 g of LDPE granules led to 55.9 % gas. 44.1 % liquid oil, and no carbon residue (only catalyst was obtained as solid residue). Reducing the quantity of AMD1 catalyst to 100 g per 1000 g of PE granules decreased gas production to 40.6 %, while liquid oil increased to 59.4 % and no carbon residue. Further reduction of the AMD1 catalyst quantity to 50 g resulted in gas, liquid oil and solid residue percentages of 43.7 %, 55.5 %, and 0.8 % respectively. Similarly, employing 50 g of AMD4 catalyst per 1000 g of PE granules led to 50.4 % gas, 49.6 % liquid oil, and no carbon residue.
Establishing the optimal amount of catalyst will depend on its efficiency, its cost and the possibility to reuse it (at least partially).
3.1.2. Product Distribution in Liquid Fraction
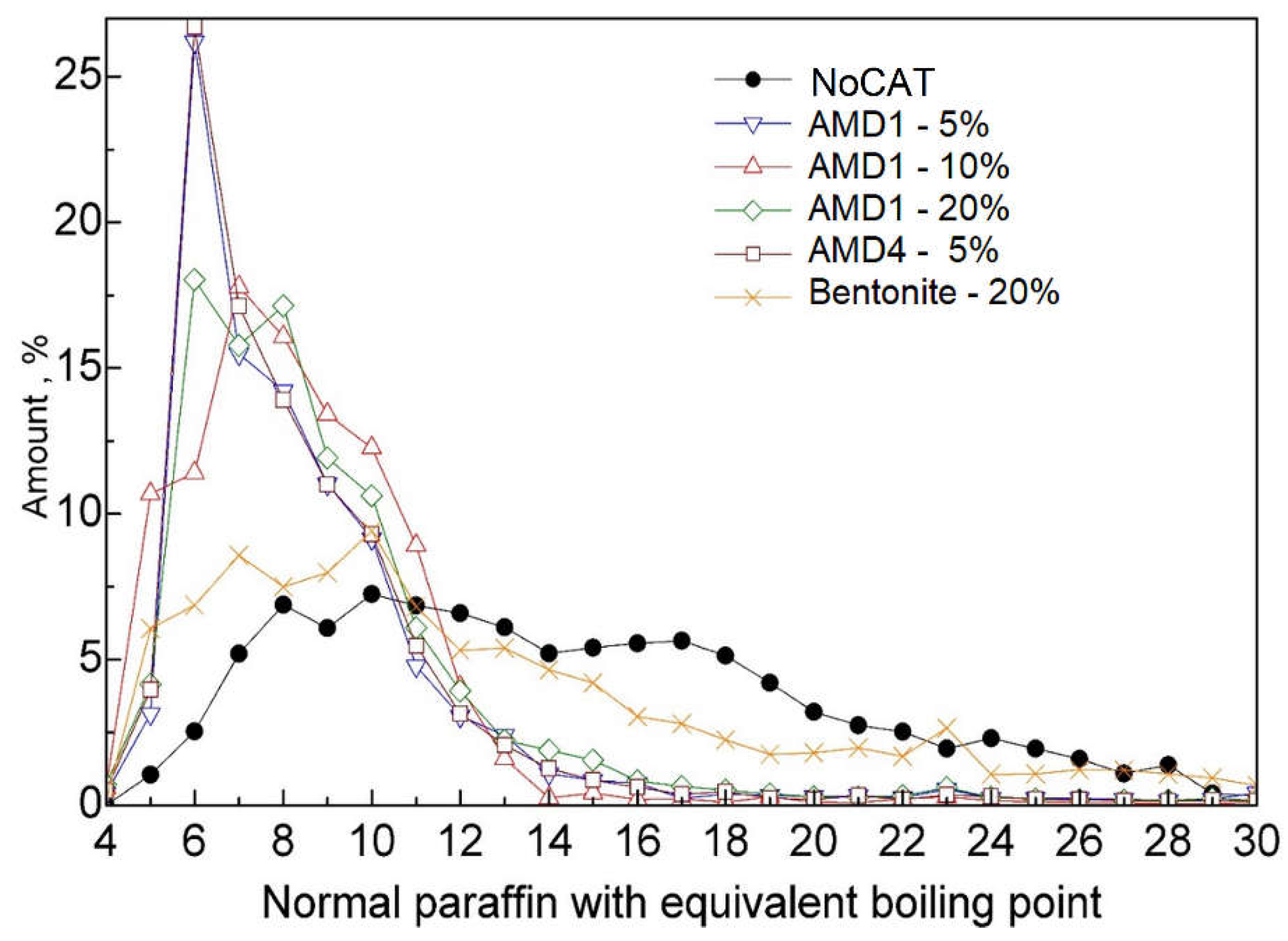
To increase the clarity of the presentation, the results obtained during pyrolysis for the combined liquid fractions are reproduced below (
Figure 3).
The thermal cracking of polyethylene involves the random breaking of the polymer structure producing numerous hydrocarbon radicals in wide range of carbon numbers [
23]. It can be observed that in non-catalytic pyrolysis (NoCAT), carbon numbers are spread from C5 to C28. A reason of this wide range is the random scission of PE as primary reaction occurred during the pyrolysis process, mostly, random scissions, generating of free radicals which are stabilized by extracting a hydrogen atom from the substrate producing alkanes or by intramolecular hydrogen transfer and β-scission producing alkenes.
In the case of using AMD catalysts, it shows a narrow range distribution number of carbon. Primary, most distribution of carbon number is in C5-C12, which was highly effective in both breaking down the polymer backbone through cracking and converting light olefins into aromatic compounds, due to secondary reactions occurring in pyrolysis process. The optimal outcomes can be seen in the case of using 50 g of AMD1 catalyst and 50 g of AMD4 catalyst in which peak amount of carbon number distribution is C6.
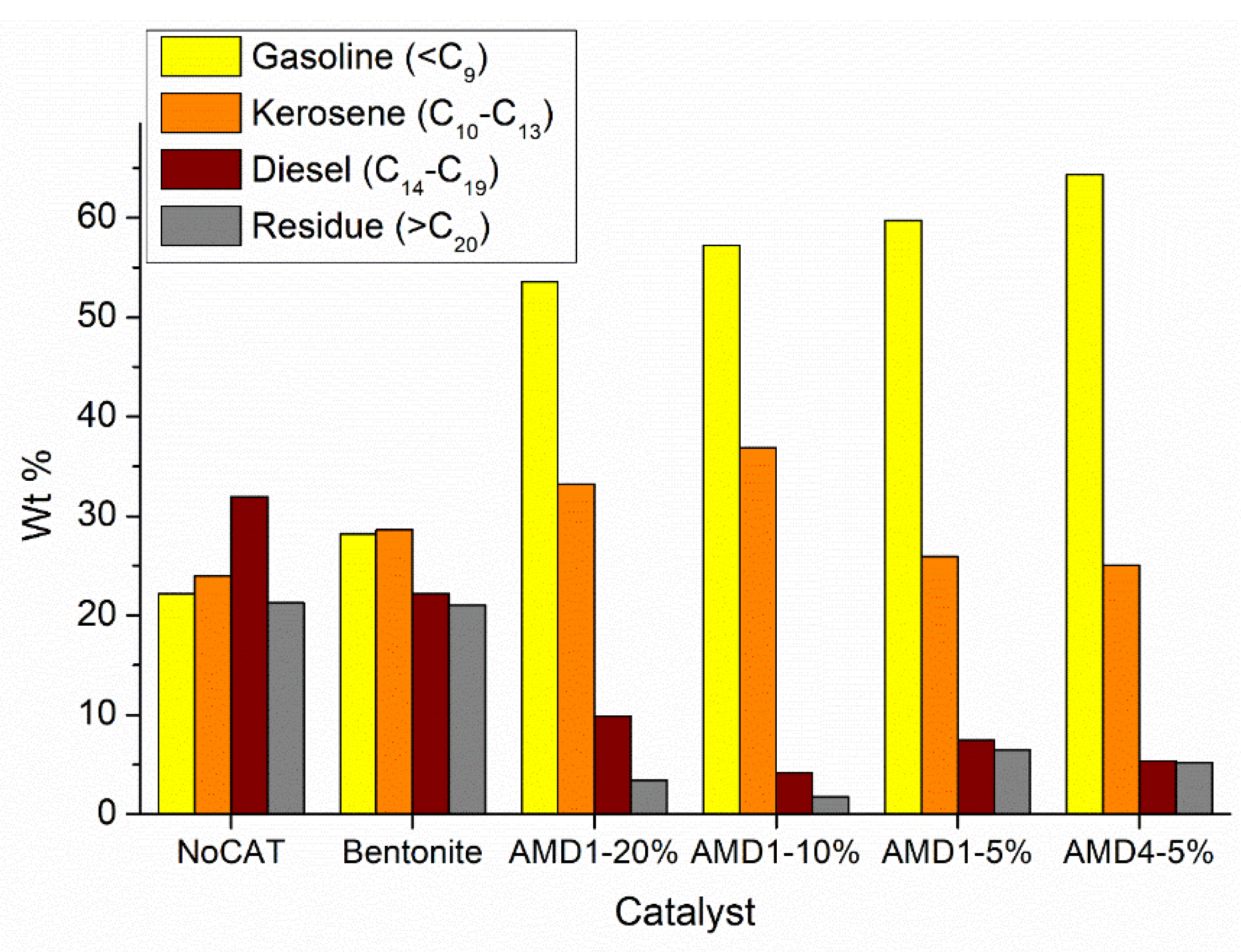
Figure 4 shows the same results regarding the composition of the liquid fraction obtained during the pyrolysis of polyethylene grains, but this time the hydrocarbons were grouped according to the number of carbon atoms: gasoline (<C9); kerosene (C10-C13), diesel (C14-C19) and residues (>C20). The analysis of the presented data confirms that in the case of using AMD type catalysts, the composition of the liquid fraction is very rich in hydrocarbons <C13, respectively from the gasoline or kerosene classes (more than 80 %).
3.1.3. Composition by Classes of Compounds in Liquid Fraction
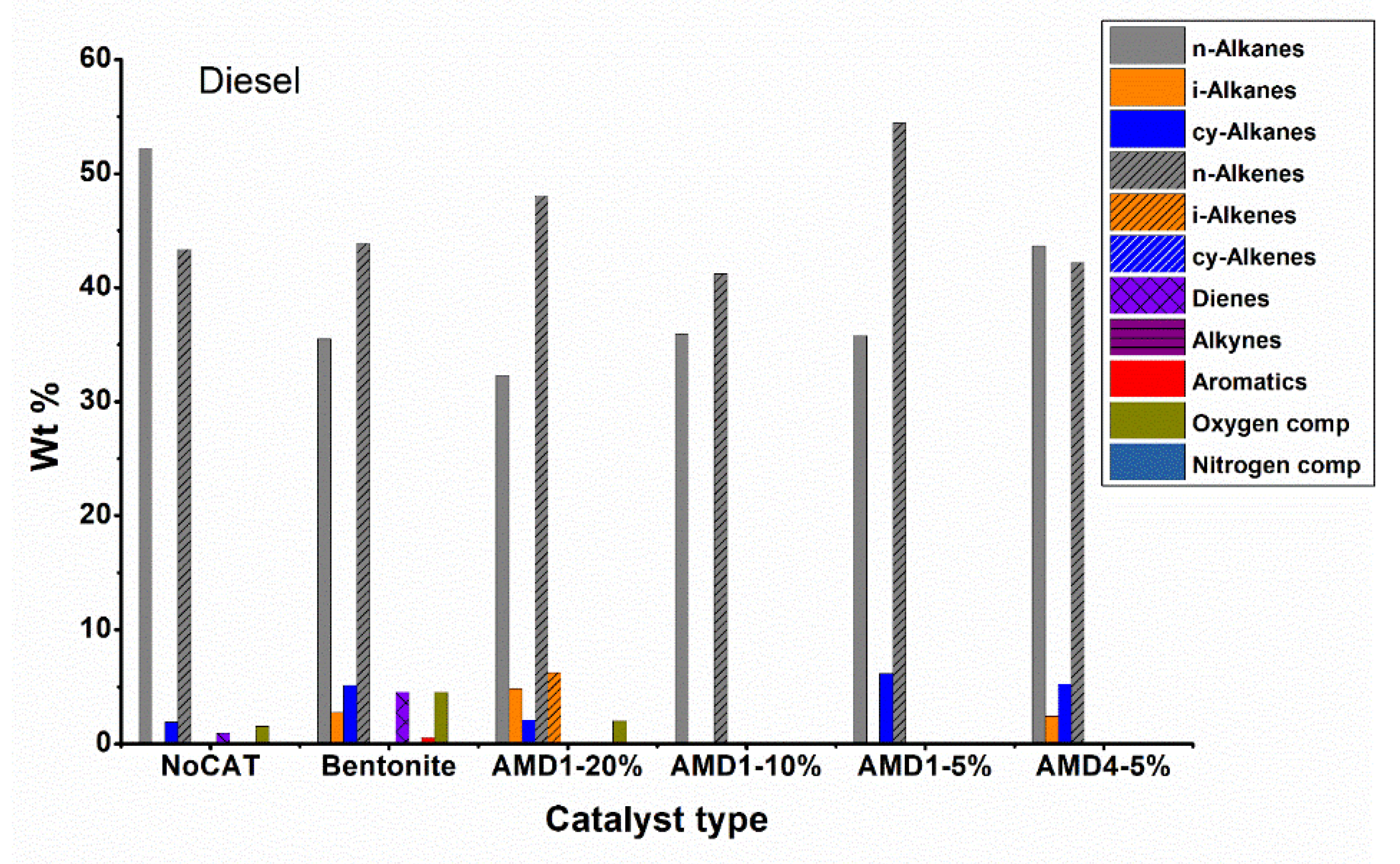
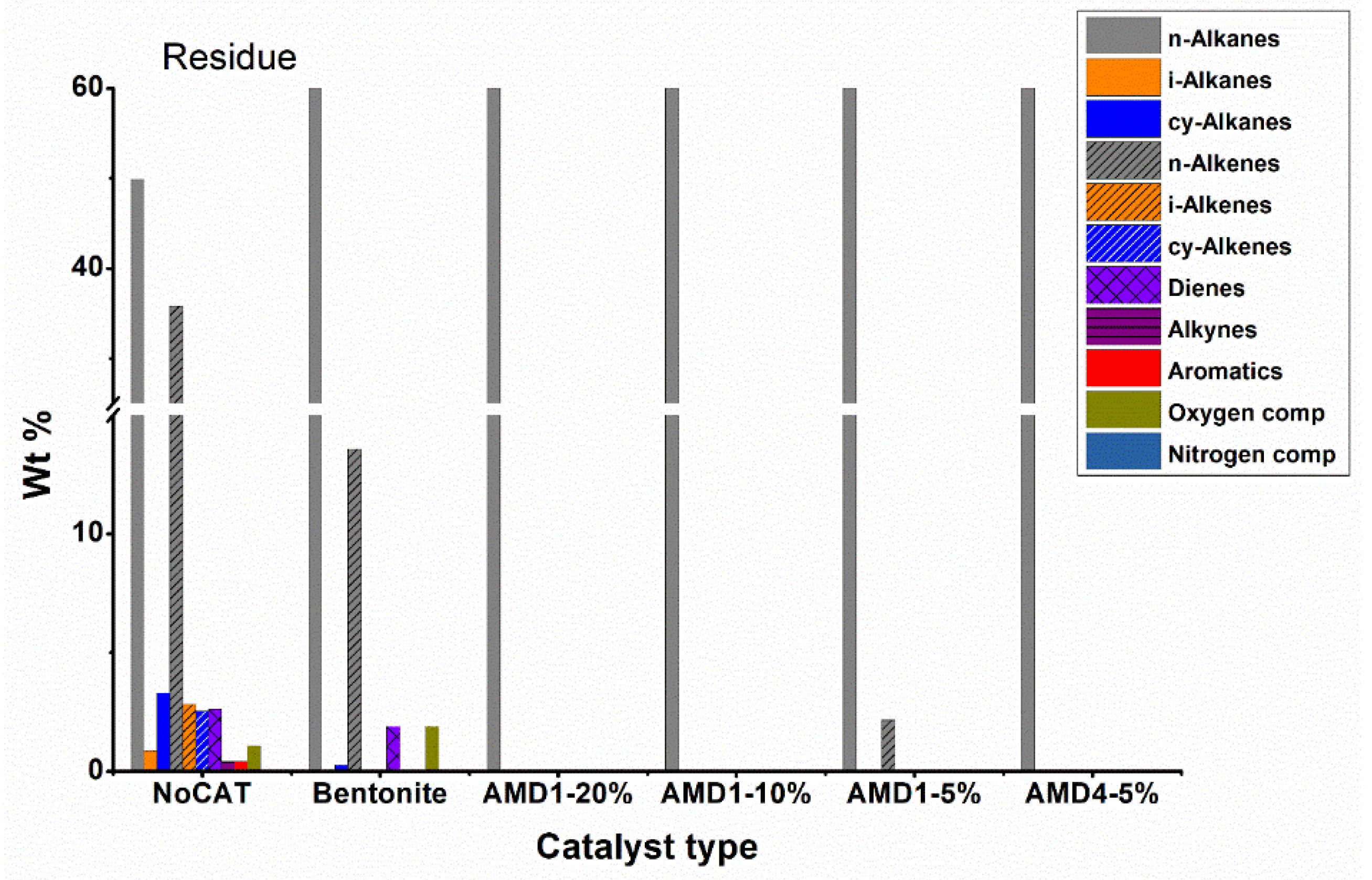
Hydrocarbons obtained from the batch plant with liquid fraction separation column were classified into compound fractions, which are gasoline C5-C9, kerosene C10-C13, C14-C19, residue C20+. For each liquid fraction, elemental analysis and GC analysis were carried out, then the identified compounds were categorized into groups: n-alkanes, i-alkanes, cy-alkanes, n-alkenes, i-alkenes, cy-alkenes, dienes, alkynes, aromatics, oxygen compounds, nitrogen compounds, that are shown in
Figure 5,
Figure 6,
Figure 7,
Figure 8 and
Figure 9.
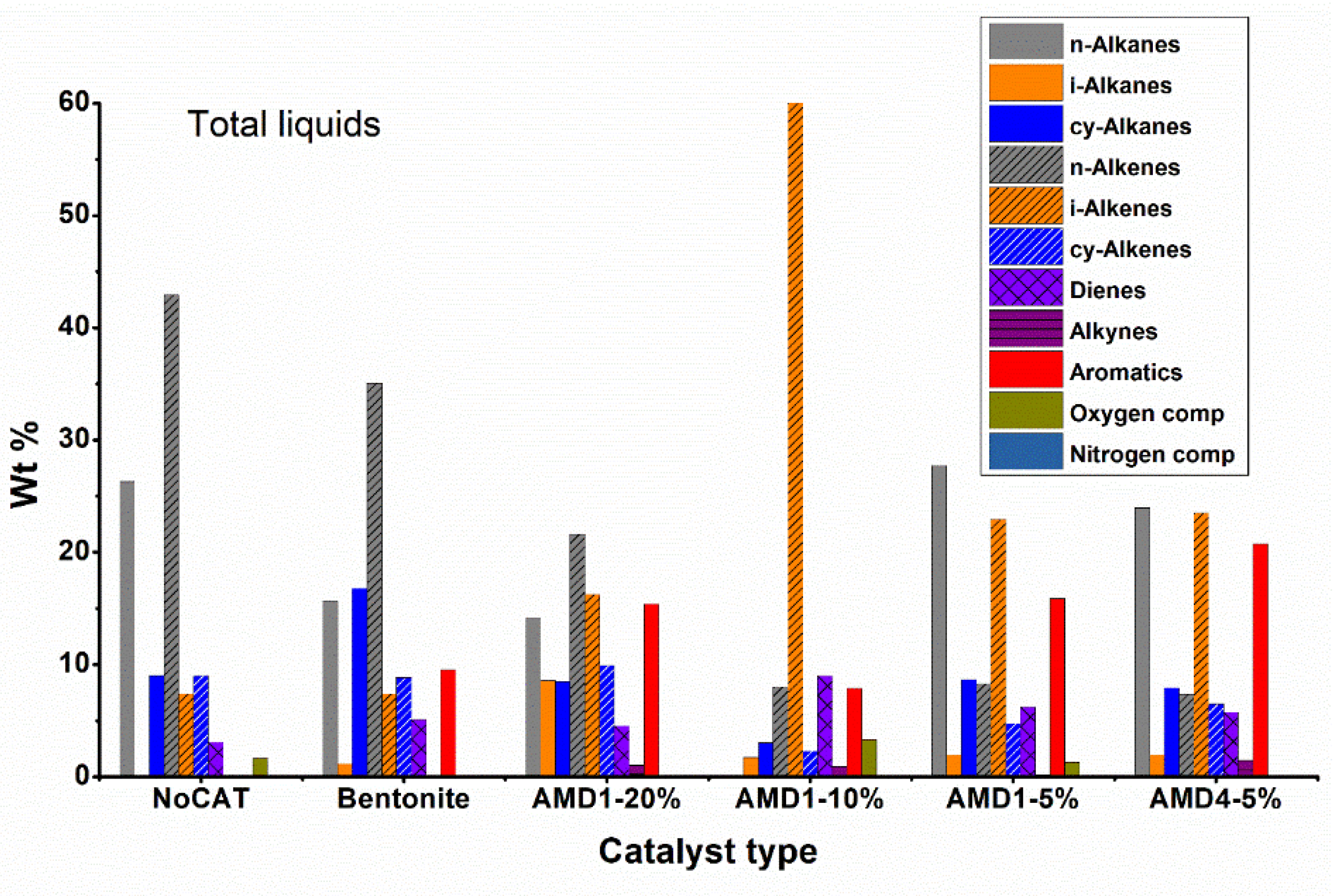
The analysis of the composition for the combined liquid fractions (
Figure 5) allows us to highlight again the important role of the catalyst. In the non-catalytic pyrolysis, most obtained compounds were n-alkanes 49.6 % and n-alkenes 35.6 %, hydrocarbons with a branched or cyclic chain being present in very low concentrations. The presence of compounds with oxygen (~1 %) is because it is difficult to completely purge the air and replace it with nitrogen before the pyrolysis process. These results are in agreement with the literature data regarding the non-catalytic pyrolysis of polyethylene [
3].
For catalytic processes (especially with AMD type catalysts), the concentration of linear chain hydrocarbons decreases, they are being converted into branched chain hydrocarbons, cycloalkanes or even aromatic hydrocarbons. The AMD4 catalyst shows a good aromatization activity of the obtained hydrocarbons, the concentration of aromatic hydrocarbons reaching 21.2 %. Regarding branched chain hydrocarbons, iso-alkenes predominate, their concentration being 3-4 times higher than that of iso-alkanes, the best results being obtained with AMD1 10 % catalyst. The concentration of cycloalkanes is usually significantly lower than the concentration of aromatic hydrocarbons (about half of their concentration). Bentonite is a less active catalyst than AMD type catalysts, the concentration of linear chain hydrocarbons dropped to only 67 % compared to 85 % in the non-catalytic process.
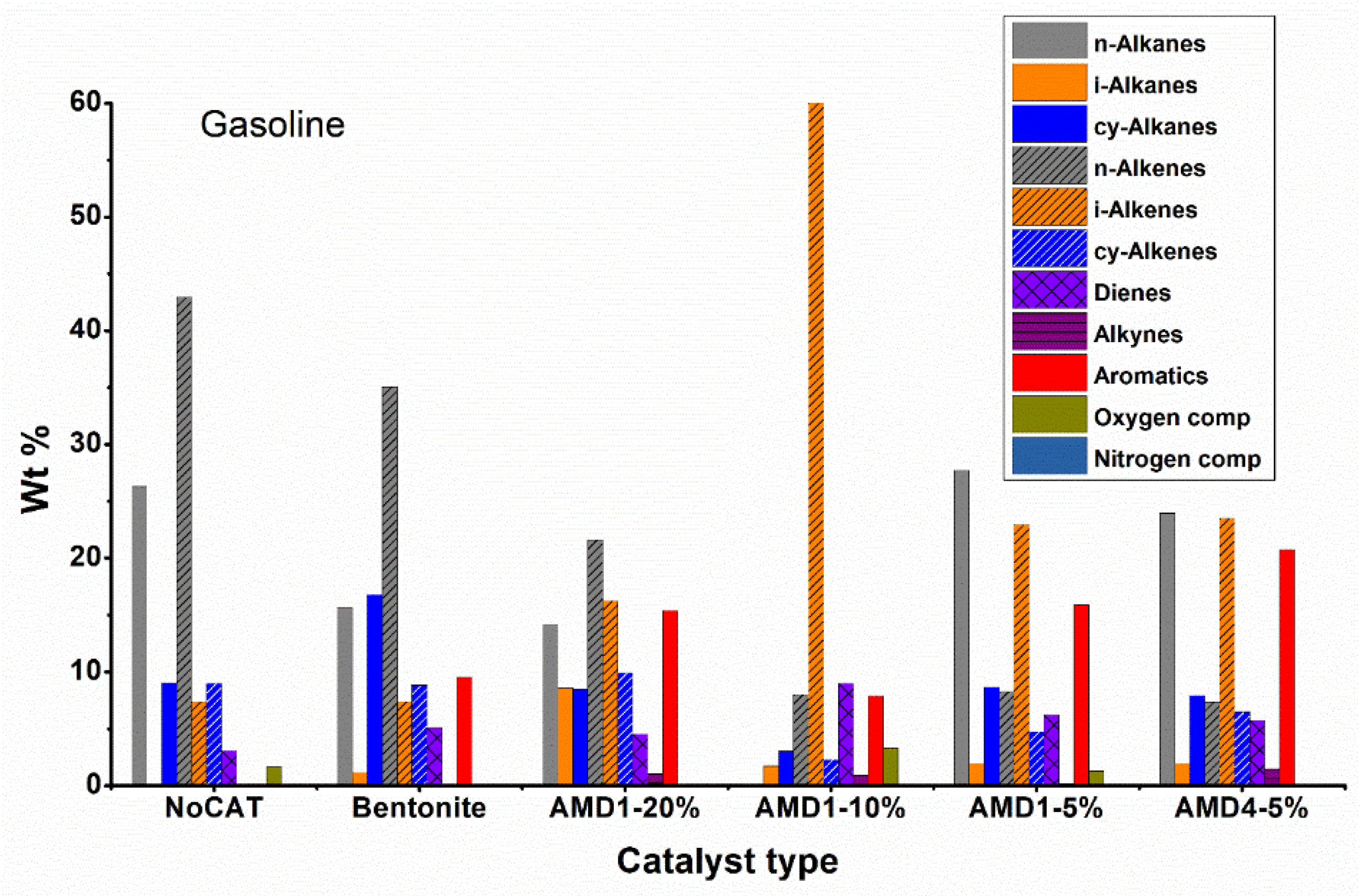
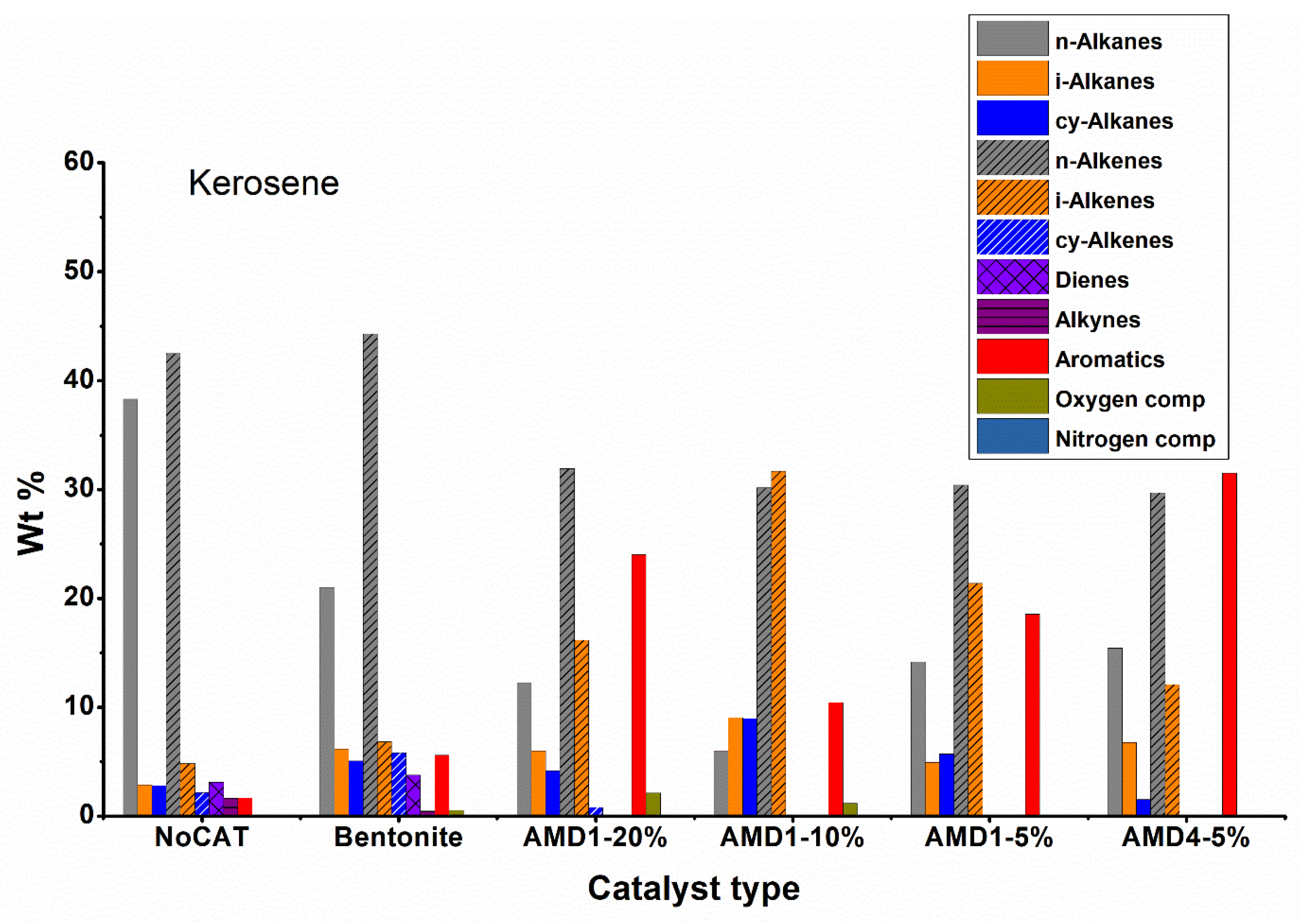
Analyzing the data regarding the composition by fractions (
Figure 6,
Figure 7,
Figure 8 and
Figure 9) the high proportion of branched chain hydrocarbons (especially iso-alkenes) and aromatics are found in the gasoline and kerosene fractions, while in the diesel and residue fractions hydrocarbons still predominate with linear chain. However, it should be noted that the diesel fraction represents only 4 – 10 % of the total liquid fraction obtained during pyrolysis in the presence of AMD type catalysts and the residue fraction is obtained in smaller quantities.
3.2. Pyrolysis of Plastic Foil Waste
3.2.1. Pyrolysis Production Yield
Pyrolysis experiments were conducted on plastic film waste sourced from household waste sorting (IRIDEX). The waste contains not only plastic but also some paper, which absorbs a lot of water, the humidity being 23 %; part of this water is collected with the liquid fraction, and another part leaves together with the gases.
The yields of pyrolysis products. including liquid fractions, gases, and solid residues were examined under varying conditions. The results obtained from pyrolyzing 900 grams of this waste material are presented in
Table 3.
The non-catalytic pyrolysis yielded 325.9 g (47 %) of liquid fractions 252 g (36.4 %) of gases and 115.2 g (16.6 %) of solid residue (the percentage values refer to the dry weight of the introduced waste). The presence of water in amount of 57.3 g in the liquid fraction is due to the moisture of the waste introduced into the reactor. These values are quite close to the values obtained in the case of non-catalytic pyrolysis of LDPE granules
However, when 90 g of catalyst was introduced, the distribution shifted significantly. This condition decreased liquid fraction from 325.9 g to 115.1 g. The decrease in liquid fractions under catalytic conditions can be attributed to the efficient conversion of the heavier hydrocarbons into gaseous products, which increased from 252 g to 507.9 g. This change is due to extensive hydrogenolysis, a process where the oligomeric primary products are further converted into lighter hydrocarbons. Over hydrogenolysis becomes the main process only after most or all the long-chain polyethylene has been used up [
22]. The presence of a catalyst also influences the formation of solid residue, which decreased from 115.2 g to 70 g,
3.2.2. Product Distribution in Liquid Phase
The pyrolysis of plastic film waste sourced from household waste sorting was investigated and the carbon number distribution in the liquid fraction both in non-catalytic and catalytic conditions were determined. In non-catalytic pyrolysis process resulted a wide range of carbon numbers, spanning from C4 to C20+. This broad distribution reflects the random scission of polymer chains within the plastic film waste during thermal cracking, leading to the formation of diverse hydrocarbon radicals across various carbon numbers.
Table 4.
Fractions obtained (% by weight) from the pyrolysis of plastic foil waste.
Table 4.
Fractions obtained (% by weight) from the pyrolysis of plastic foil waste.
| Fraction |
Carbon Range |
NoCAT (%) |
Catalytic (%) |
| Gasoline |
C4 - C9 inclusive |
24 |
36.7 |
| Kerosene |
C10 - C13 |
38 |
31.3 |
| Diesel |
C14 - C19 |
26.5 |
22.1 |
| Residue |
C20+ |
11.6 |
10.1 |
Catalytic pyrolysis exhibited a significant increase in the gasoline fraction, with a higher percentage of C4 to C9 hydrocarbons produced (36.7 %), indicating improved conversion efficiency compared to non-catalytic conditions. Also, catalytic conditions resulted in a slight reduction in the kerosene and diesel fractions, from 38 % to 31.3 % and 26.5 % to 22.1% respectively, compared to non-catalytic conditions, suggesting a shift towards the production of lighter hydrocarbons. Both non-catalytic and catalytic pyrolysis yielded similar percentages of residue, indicating that catalysts did not significantly affect the formation of high carbon number compounds, 11.6 % and 10.1 %.
3.3. Comparison of Catalyst Efficiency in Pyrolysis of Plastic Granules and Plastic Waste
In experiments conducted on LDPE granules, catalysts demonstrated a remarkable increase in the yield of liquid oil fractions while decreasing gas production. For instance, the utilization of 100 g (10 % wt.) of AMD1 catalyst per 1000 g of PE granules led to a significant shift, with liquid oil production increasing from 40 % to 59.4 % and gas fraction decreasing from 60 % to 40.6 %. This indicates the catalyst's effectiveness in promoting the conversion of polymer chains into valuable liquid products. Conversely, in the pyrolysis of plastic film waste, catalysts showed a different impact on product distribution. Introducing catalysts led to a notable decrease in the liquid fraction, attributed to efficient conversion of polymer chains into gaseous products. For instance, with 90 g (10 % wt.) of catalyst, the liquid fraction decreased significantly from 36.2 % to 12.8 %, while gas production increased from 28 % to 56.4 %. This suggests a distinct reaction pathway influenced by catalyst presence in the pyrolysis of plastic waste compared to PE granules.
Catalysts demonstrated varying effects on solid residue formation depending on the feedstock used. With LDPE granules, catalysts facilitated complete pyrolysis, resulting in the absence of solid residue. On the other hand, in the case of plastic film waste, while catalysts reduced solid residue formation, they did not eliminate it. For example, with catalyst, solid residue decreased from 12.8 % to 7.8 %, indicating a partial conversion of plastic material into gaseous and liquid products, furthermore, resulting impurities that are in initial feedstock.
Catalytic pyrolysis of LDPE granules resulted in a narrower range of carbon number distribution, particularly with catalysts like AMD1, leading to a peak distribution around C6. This suggests effective cracking of the polymer backbone and conversion into smaller hydrocarbons. In contrast, catalytic pyrolysis of plastic waste exhibited a significant increase in the gasoline fraction (C4 to C9 hydrocarbons), indicating improved conversion efficiency. Both cases of LDPE granules and plastic waste got significant improvement of gasoline fraction, which means a better selectivity of compounds in liquid fraction.
3.4. Gas Composition
The gas composition resulting from the non-catalytic pyrolysis process, referred to as “NoCAT” and various catalytic pyrolysis processes using different catalysts, namely bentonite, AMD1 with different concentrations (20 %, 10 %, 5 %), and AMD4 (5 %), containing different compounds, such as oxygen, nitrogen, carbon monoxide, methane, ethane, carbon dioxide, ethylene, propane, butane, C5+, and others are presented in the
Table 5.
The significant concentration of C6+ hydrocarbons is because the condensation system of the distillation column was undersized, sometimes the gas flow leaving the column is high and liquid hydrocarbons cannot be condensed effectively.
Catalytic sites likely play a crucial role in promoting specific reaction pathways leading to the formation of lighter hydrocarbons Thus, in the presence of AMD catalysts the concentration of propylene and 1-butene is much higher than that obtained in the absence of catalysts (propylene: 8.62 % NoCAT vs. 10.2 - 16.34 % with catalysts; 1-butene: 8.3 % NoCAT vs. 15.04 - 21.76 % with catalysts). The bentonite catalyst is less active, the composition of the gas is quite close to that obtained in the non-catalytic process.
The gas composition of plastic film waste sourced from household waste resulting from the non-catalytic pyrolysis process, referred to as “NoCAT” and the catalytic pyrolysis process, referred to as “Cat.” consisting of various compounds such as oxygen, nitrogen, carbon monoxide, methane, ethane, carbon dioxide, ethylene, propane, propylene, butane, and heavier hydrocarbons (C5, C6+), are presented in
Table 6.
Comparing the results obtained during the pyrolysis of plastic film waste with those obtained during the pyrolysis of PE granules, several important differences can be observed due to the impurities in the waste and its moisture content (23 %):
- the percentage of CO2 in the gases obtained during the pyrolysis of plastic film waste is much higher than that obtained during the pyrolysis of PE granules.
- the concentrations of ethylene, propene and 1-butene are somewhat lower in the pyrolysis of plastic film waste than in the pyrolysis of PE granules.
These observations indicate that the catalytic pyrolysis process significantly alters the distribution of hydrocarbons compared to non-catalytic pyrolysis, promoting the formation of specific valuable products such as ethane, propylene, and C6+ hydrocarbons while reducing the formation of CO2. The catalyst enhances cracking and selective reactions, leading to a more efficient and targeted conversion of the pyrolyzed material.
3.4.1. Calorific Power
The gas composition resulting from the non-catalytic pyrolysis process (NoCAT) and various catalytic pyrolysis processes using different catalysts, namely bentonite, AMD1 with different concentrations (20 %, 10 %, 5 %), and AMD4 (5 %), along with the lower heating value (LHV) of the compounds, is presented in
Table 7.
The analysis of the data on the calorific value of the gases obtained during the pyrolysis of LDPE granules (presented in
Table 6) shows that these gases have a very good calorific value of 72 - 80 % of the calorific value of methane.
Table 8.
Calorific power of gases obtained in pyrolysis of plastic film waste.
Table 8.
Calorific power of gases obtained in pyrolysis of plastic film waste.
| Compound |
LHV
MJ/kg |
noCAT
%wt |
AMD1-10 %
% wt |
| CO |
10.16 |
1.47 |
1.66 |
| CH4
|
50.00 |
0.00 |
0.00 |
| C2H6
|
47.20 |
5.20 |
7.68 |
| CO2
|
0.00 |
0.00 |
0.00 |
| C2H4
|
47.70 |
2.75 |
2.40 |
| C3H8
|
46.13 |
1.72 |
1.64 |
| C3H6
|
45.78 |
3.58 |
4.56 |
| n-C4H10
|
46.46 |
1.30 |
1.05 |
| 1-C4H8
|
45.17 |
2.24 |
3.00 |
| 2-C4H8
|
45.17 |
2.68 |
3.01 |
| C5H12
|
44.94 |
1.25 |
1.03 |
| +C6H14
|
43.40 |
4.40 |
5.68 |
| Total |
|
26.57 |
31.71 |
Table 7 shows the calorific values of the gaseous fraction obtained during the pyrolysis of plastic film waste. It can be observed that the values obtained both in the non-catalytic and in the catalytic process are significantly lower than in the case of the pyrolysis of LDPE granules, representing 53 % and 63.4 % respectively of the calorific value of methane gas. The catalyst causes an increase in calorific power by 19.3 %, which is very important for using this gas to power an engine connected to an electric generator.
4. Conclusions
Converting plastic waste into fuel is a sustainable way to solve the problems generated by this waste. The catalytic pyrolysis tests of LDPE granules and then of a waste containing LDPE film were carried out on a discontinuous pyrolysis installation equipped with a distillation column to collect and separate the gaseous and liquid fractions that were subjected to GC/FID and GC/MS. The catalysts were used directly in the pyrolysis reactor, a solution that greatly simplifies the process, requiring only one reactor. Inexpensive catalysts such as bentonite or treated clinoptilolite were used. The clinoptilolite catalysts were active and determined the important change in the composition of the liquid fractions and their distribution. The catalysts were first tested on LDPE granules, their effect being visible even at concentrations of 5 % when they determined the increase in the proportion of the light gasoline fraction from 22 % to over 60 %. Regarding the composition of the fractions obtained during the pyrolysis of LDPE granules, if, in the absence of catalysts linear chain hydrocarbons predominate (n-alkanes 49.6 % and n-alkenes 35.6 %), in the presence of catalysts, the composition changes a lot, among chain hydrocarbons branched, the concentration of iso-alkenes is usually 3 - 4 times higher than that of iso-alkanes (the best results on the AMD1 catalyst – 10 %). The concentration of cycloalkanes is usually significantly lower than the concentration of aromatic hydrocarbons (about half of their concentration). The AMD4 catalyst shows a good aromatization activity of the obtained hydrocarbons, the concentration of aromatic hydrocarbons reaching 21.2 %.
When plastic film waste was subjected to pyrolysis, the effect of the catalysts was especially in the increase of the gaseous fraction, from 252 to 507.9 g for a sample of 900 g of waste. Considering that these gases have a good calorific power (approximately 63.4 % of the calorific power of methane) and the catalyst causes an increase in calorific power by 19.3 %, it can be feasible to use this gas to power an engine connected to an electric generator.
Author Contributions
Conceptualization, I.C. and C.A.; methodology, I.C., C.A. and P.C.; software, A.T.and MA.B.; validation, I.C., P.C. and C.A.; formal analysis, GC.C-N. and MA.B; investigation, G.P., M.C.,C.A., A.T. and P.C.; resources, ND.I. and MA.B.; data curation, A.T.; writing—original draft preparation, G.P.; writing—review and editing, I.C. and P.C.; visualization, G.P.,A.T. and P.C.; supervision, I.C.; project administration, I.C.; funding acquisition, ND.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
The authors acknowledge the support received from the Competitiveness Operational Program 2014-2020, Action: Stimulating the demand of companies for innovation through CDI projects carried out by companies individually or in partnership with CD institutes and universities, to innovate processes and products in economic sectors that show growth potential, MySMIS Code 122990, “INODES” financed by contract: 365/390041/27.09.2021.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kibria, M.G.; Masuk, N.I.; Safayet, R.; Nguyen, H.Q.; Mourshed, M. Plastic Waste: Challenges and Opportunities to Mitigate Pollution and Effective Management. Int J Environ Res 2023, 17, 20. [Google Scholar] [CrossRef] [PubMed]
- An, L.; Kou, Z.; Li, R.; Zhao, Z. Research Progress in Fuel Oil Production by Catalytic Pyrolysis Technologies of Waste Plastics. Catalysts 2024, 14, 212. [Google Scholar] [CrossRef]
- Dai, L.; Zhou, N.; Lv, Y.; Cheng, Y.; Wang, Y.; Liu, Y.; Cobb, K.; Chen, P.; Lei, H.; Ruan, R. Pyrolysis technology for plastic waste recycling: A state-of-the-art review. Progress in Energy and Combustion Science 2022, 93, 101021. [Google Scholar] [CrossRef]
- Pilapitiya, P.G.C.N.T.; Ratnayake, A.S. The world of plastic waste: A review. Cleaner Materials 2024, 11. [Google Scholar] [CrossRef]
- Haider, T.P.; Völker, C.; Kramm, J.; Landfester, K.; Wurm, F.R. Plastics of the future? The impact of biodegradable polymers on the environment and on society. Angewandte Chemie International Edition 2019, 58, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Chandran, M.; Tamilkolundu, S.; Murugesan, C. Conversion of plastic waste to fuel. in Plastic waste and recycling. 2020, Elsevier. p. [CrossRef]
- RPadhan, K.; Sreeram, A. Enhancement of storage stability and rheological properties of polyethylene (PE) modified asphalt using cross linking and reactive polymer based additives. Construction and Building Materials 2018, 188, 772–780. [Google Scholar] [CrossRef]
- Tsuchimoto, I.; Kajikawa, Y. Recycling of Plastic Waste: A Systematic Review Using Bibliometric Analysis. Sustainability 2022, 14, 16340. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; Van Geem, K. Mechanical and chemical recycling of solid plastic waste. Waste management 2017, 6, 24–58. [Google Scholar] [CrossRef] [PubMed]
- Zia, K.M.; Bhatti, H.N.; Bhatti, I.A. Methods for polyurethane and polyurethane composites, recycling and recovery: A review. Reactive and functional polymers 2007, 67, 675–692. [Google Scholar] [CrossRef]
- Radhakrishnan, K.; Kumar, P.S.; Rangasamy, G.; Perumal, L.P.; Sanaulla, S.; Nilavendhan, S.; Manivasagan, V.; Saranya, K. A critical review on pyrolysis method as sustainable conversion of waste plastics into fuels. Fuel 2023, 337, 126890. [Google Scholar] [CrossRef]
- Shivasharana, C.; Kesti, S.S. Physical and chemical characterization of low density polyethylene and high density polyethylene. Journal of Advanced Scientific Research 2019, 10, 30–34. [Google Scholar]
- Olabisi, O. Polyolefins. in Handbook of Thermoplastics, O. Olabisi, Adewale, K., Editor. 2016, CRC Press, Taylor&Francis Group: Boca Raton, FL. p. 1-53.
- Zhang, Y.; Fu, Z.; Wang, W.; Ji, G.; Zhao, M.; Li, A. Kinetics, Product Evolution, and Mechanism for the Pyrolysis of Typical Plastic Waste. ACS Sustainable Chemistry & Engineering 2021, 10, 91–103. [Google Scholar] [CrossRef]
- Vollmer, I.; Jenks, M.J.; Roelands, M.C.; White, R.J.; Van Harmelen, T.; De Wild, P.; van Der Laan, G.P.; Meirer, F.; Keurentjes, J.T.; Weckhuysen, B.M. Beyond mechanical recycling: Giving new life to plastic waste. Angewandte Chemie International Edition 2020, 59, 15402–15423. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Kumar, A.; Singh, E.; Kumar, S. Recent research advancements in catalytic pyrolysis of plastic waste. ACS Sustainable Chemistry & Engineering 2023, 11, 2033–2049. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, K.; Wei, B.; Yang, H.; Jin, L.; Hu, H. Pyrolysis behavior of low-density polyethylene over HZSM-5 via rapid infrared heating. Sci Total Environ 2022, 806, 151287. [Google Scholar] [CrossRef] [PubMed]
- Miandad, R.; Barakat, M.A.; Aburiazaiza, A.S.; Rehan, M.; Nizami, A.S. Catalytic pyrolysis of plastic waste: A review. Process Safety and Environmental Protection 2016, 102, 822–838. [Google Scholar] [CrossRef]
- Syamsiro, M.; Saptoadi, H.; Norsujianto, T.; Noviasri, P.; Cheng, S.; Alimuddin, Z.; Yoshikawa, K. Fuel Oil Production from Municipal Plastic Wastes in Sequential Pyrolysis and Catalytic Reforming Reactors. Energy Procedia 2014, 47, 180–188. [Google Scholar] [CrossRef]
- Lopez, A.; de Marco, I.; Caballero, B.M.; Adrados, A.; Laresgoiti, M.F. Deactivation and regeneration of ZSM-5 zeolite in catalytic pyrolysis of plastic wastes. Waste Manag 2011, 31, 1852–8. [Google Scholar] [CrossRef]
- Aguado, J.; Serrano, D.P.; Miguel, G.S. European trends in the feedstock recycling of plastic wastes. Global NEST Journal 2007, 9, 12–19. [Google Scholar]
- Sivagami, K.; Kumar, K.V.; Tamizhdurai, P.; Govindarajan, D.; Kumar, M.; Nambi, I. Conversion of plastic waste into fuel oil using zeolite catalysts in a bench-scale pyrolysis reactor. RSC advances 2022, 12, 7612–7620. [Google Scholar] [CrossRef]
- Fan, L.; Liu, L.; Xiao, Z.; Su, Z.; Huang, P.; Peng, H.; Lv, S.; Jiang, H.; Ruan, R.; Chen, P. Comparative study of continuous-stirred and batch microwave pyrolysis of linear low-density polyethylene in the presence/absence of HZSM-5. Energy 2021, 228, 120612. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).