1. Introduction
Colorectal cancer (CRC) is the most common neoplasm in both sexes in Spain and the second leading cause of specific death, only behind lung cancer. It is estimated that by 2024, 44,294 new cases will be diagnosed, of which 29,648 will be colon and 14,646 will be rectal, with the average age for CRC diagnosis being around 70 years [
1]. Hereditary factors have little impact on the population’s incidence (less than 5% of CRC cases), and most are sporadic tumors with multifactorial etiology. Throughout the disease, 20-25% of patients will develop metastasis, and the overall mortality is around 40-45% [
2].
Figure 1.
Graphical abstract.
Figure 1.
Graphical abstract.
The TNM classification at diagnosis is the most important prognostic factor [
3]. Other factors related to survival include the number of lymph nodes resected after surgery (a minimum of 12 [
4]), response to neoadjuvant treatment, involvement of the radial resection margin, venous and perineural invasion [
5,
6], obstructive and/or perforated tumors, grade GIII [
7,
8], age, carcinoembryonic antigen (CEA) above 20 ng/ml [
9], and the patient’s status performance [
10,
11].
The classic treatment scheme includes preoperative chemoradiotherapy as it reduces pelvic recurrences, controls micrometastases, increases complete response rates to treatment, and improves overall survival by around 10% [
12,
13,
14,
15,
16,
17]. The standard scheme of RT is conventional fractionation (1.8-2 Gy/fraction/day) up to 50-50.4 Gy combined with capecitabine (CPC) or 5-fluorouracil (5-FU). Complete pathological responses with this treatment scheme are around 20%, and tumor regression is achieved in 40-80% of cases [
18,
19,
20].
Surgery is the main curative treatment, and the technique of choice is low or ultralow anterior resection with total mesorectal excision (TME), as it preserves the function of the sphincter complex. It is usually performed between 6 and 8 weeks after neoadjuvant treatment [
21].
Adjuvant chemotherapy is administered in high-risk stage II or stage III, despite having achieved a complete clinical response, and has shown benefits in DFS. The standard scheme is based on fluoropyrimidines (with or without oxaliplatin) by extrapolation from the data of the ADORE study [
22,
23], however, it should be individualized assessing the risk/benefit.
Since 2020, following the publication of the RAPIDO study results [
24], there are new strategies for neoadjuvant treatment in rectal cancer.
In the RAPIDO trial, patients received 9 cycles of FOLFOX (5-fluorouracil+Oxaliplatin) or 6 of CAPOX (Capecitabine+Oxaliplatin) in a neoadjuvant setting and a short cycle of RT (5 x 5 Gy) before surgery. An ypCR-rate was observed in favor of the experimental arm compared to the arm with 50.4 Gy conventional fractionation + CPC (28% vs. 14%; p˂ 0.0001). The improvement in local recurrence (LR) and disease-free-survival (DFS) was not statistically significant: LR (7.2% vs. 4.3%; p = 0.20) and DFS (28.5% vs. 24.4%; p = 0.34).
The total neoadjuvant treatment strategy has been explored in another 4 prospective trials: Polish-II [
25], TIMING (the only non-randomized one) [
26], FOWARC [
27], GCR-3 [
28], and in a meta-analysis [
29]. This strategy improves the rates of complete pathological responses and has proven to be a surrogate factor for DFS [
30].
“Watch and wait” or observation after a complete clinical response is not a standard strategy. The decision should arise within a clinical trial or be agreed upon by the multidisciplinary committee in the selected cases. The recommended follow-up in these cases, according to the Habr-Gama et al. protocol [
31], should be carried out with anamnesis every 6-10 weeks during the first two years, every 3 months during the third year, and every 6 months from the fourth year, including in all of them: rectal examination, rigid colonoscopy, and determination of CEA levels. In the OPRA study, it was established that patients will undergo rectal examination and flexible proctosigmoidoscopy every 4 months during the first 2 years from the time of response evaluation with magnetic resonance image (MRI) every 6 months in those first 2 years. During the following 3 years, rectal examination and flexible sigmoidoscopy will be repeated every 6 months [
32].
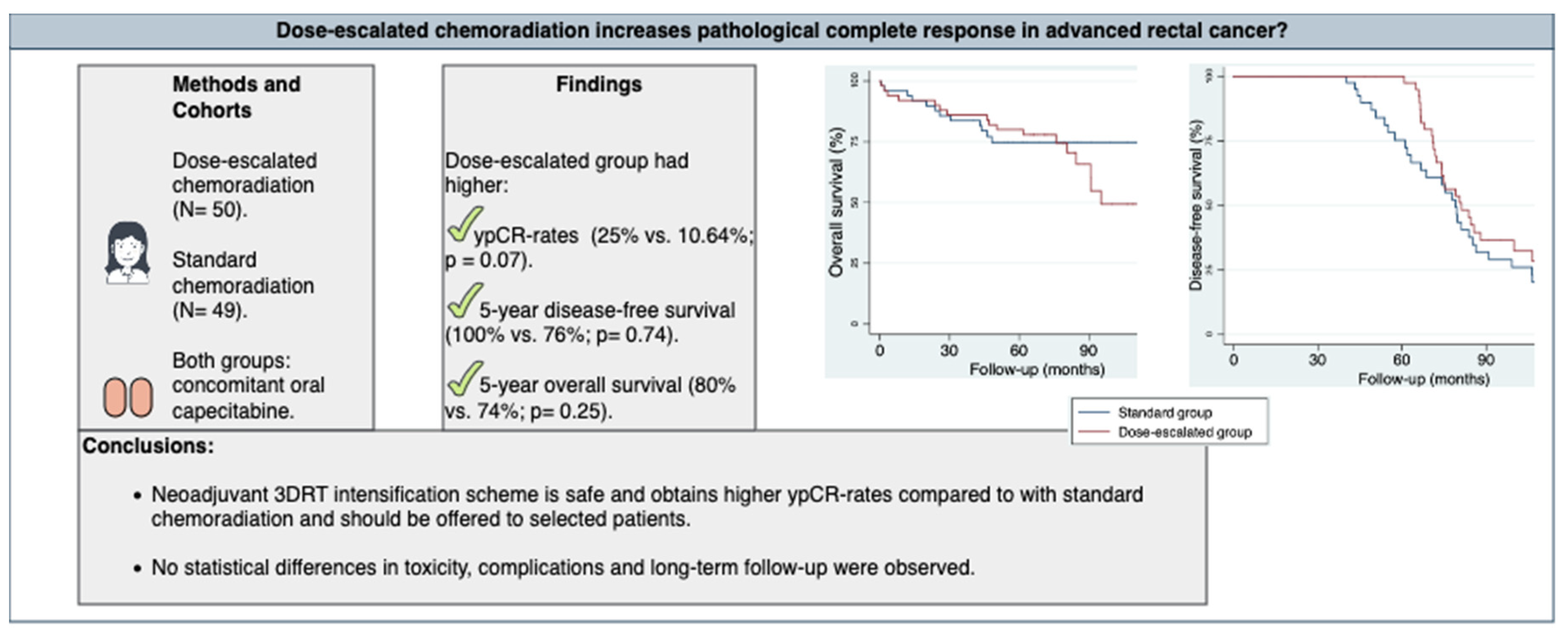
Our objective was to retrospectively assesss the impact of clinical, histological and outcomes parameters from dose intensification concomitantly with oral capecitabina, and compare rates of pathological complete responses between two differents arms of treatment dose.
2. Materials and Methods
Based on a prospective database, a retrospective study was conducted on all patients treated consecutively between June 2012 and December 2019 with neoadjuvant radiotherapy for locally advanced rectal cancer.
Since our study is single-institutional, our series are treated homogeneously with consistent and uniform pathological specimen analysis. Clinicians made the decision on dosimetry jointly with physicians in a daily meeting session based on organ-at-risk limitations.
2.1. Characteristics of the Patients
All patients were evaluated by the multidisciplinary committee for digestive tumors, including the pre-treatment study which consisted of a physical examination, rectal examination, colonoscopy, biopsy, thoracoabdominal-pelvic computerized tomography (CT) scan, abdominopelvic MRI, chest X-ray, complete blood count, and blood tests with tumor markers.
The inclusion criteria were: patients over 18 years old diagnosed with American Joint Committee on Cancer stage II-III rectal adenocarcinoma (classified according to the 6th edition) [
33] who had received at least one dose greater than 50.4Gy (considered standard in classic neoadjuvant chemoradiation schemes).
The exclusion criteria were: patients who ultimately did not undergo surgery (due to refusal or high surgical risk), synchronous tumors at diagnosis, and those with a history of other neoplasms.
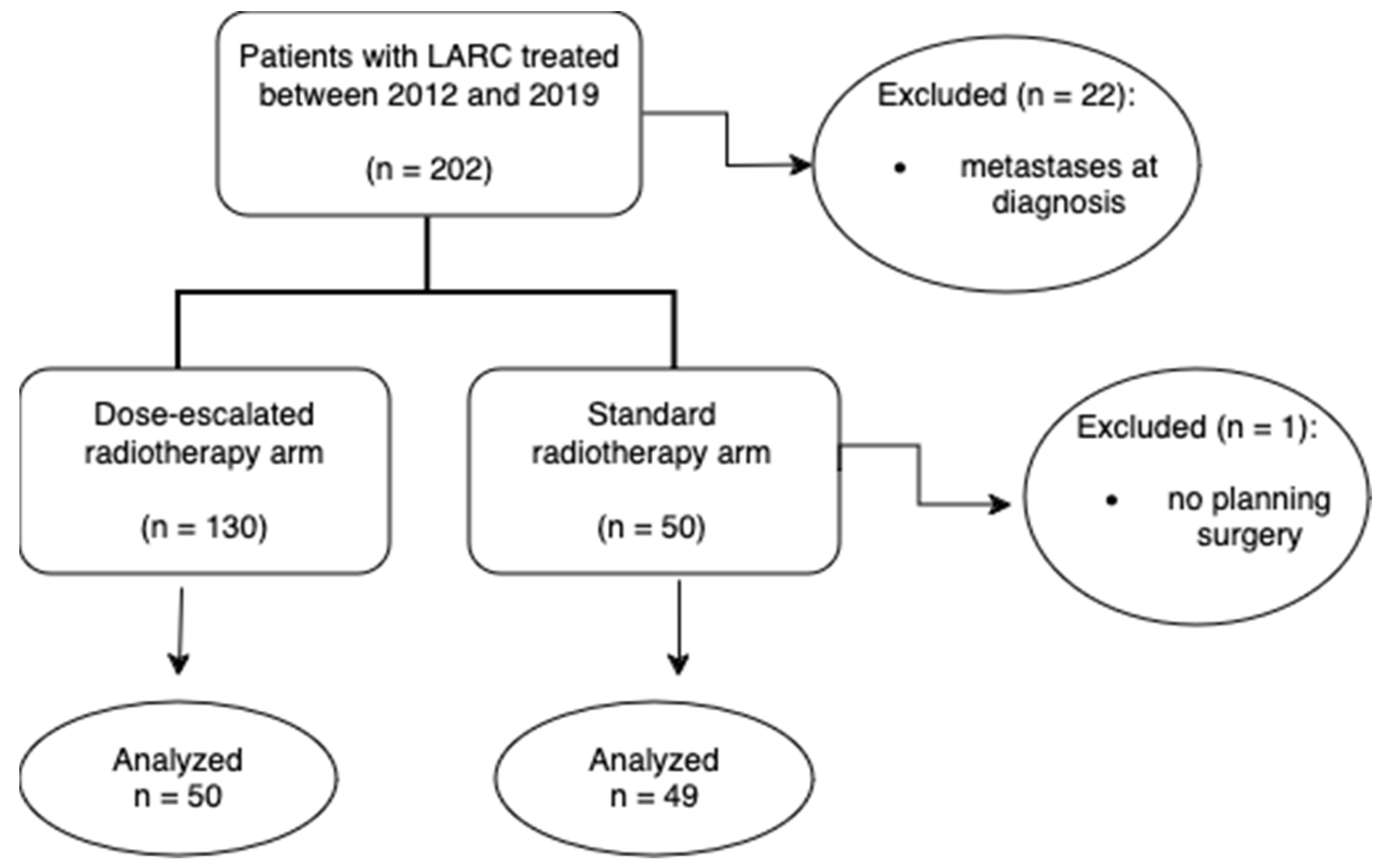
Patients with LARC were analyzed 1:1 to either a standard treatment arm consisting in preoperative chemoradiation (mean 47.5Gy) or to dose-escalated radiotherapy arm (mean 54.3Gy) as a CONSORT diagram shows in
Figure 2.
2.2. Treatment Protocol
2.2.1. Neoadjuvant Chemotherapy
Only 4 patients, those with stage cT4 and/or cN2 at diagnosis and positive extramural invasion, received neoadjuvant chemotherapy. The most commonly used regimens were 1 to 4 cycles of CAPOX. In all cases, the chemotherapy concomitant with radiotherapy was oral CPC at 825 mg/m² every 12 hours on all days that radiotherapy was administered.
2.2.2. Radiotherapy
The patients were immobilized in the supine position with an indexed vacuum mattress, knee support, and arms on the chest [
34].
The treatment volumes were defined according to the consensus guidelines for the contouring of gross, clinical and planning tumor volumes structures in anorectal cancer [
35].
The upper limit for tumors in the middle-upper third of the rectum was L5-S1, depending on the location of the peritoneal reflection. For tumors in the lower third, the upper limit of the irradiation volume was S1-S2.
The organ-at-risk and the dose limits used are: bladder (V40 < 50%, Dmax < 65Gy), femoral heads (V50 < 5-10%, Dmax < 42Gy), and small intestine (defined in our center as the abdominal cavity 1 cm above the PTV; V40 < 150cc or as close as possible).
The technique used was the “box” type with 3D conformal radiotherapy (0°, 90°, 180° and 270° fields) with a multileaf collimator when the dose went up to 50.4Gy (standard arm). In addition to the “box” technique, lateral fields were modified and oblique fields were added in the boost phase to spare the small bowel when the dose went up to 54 Gy (dose-escalated arm). The planning system used for dose calculation was Pinnacle® from Oracle®. The treatment was performed on a linear accelerator (2100 CD MLC from Varian®) using 6 and 18 MV photons as required by the patient’s anatomy. Daily positioning verifications were carried out for all patients using MV radiographic imaging.
We differentiated two treatment groups: the dose escalation group with a mean of 54.3Gy (IQR 43.4-60.76Gy) in 25 fractions over the tumor and the group that received standard radiotherapy doses with a mean of 47.5Gy (IQR 43.2-50.4Gy) with the same number of total fractions.
2.2.3. Surgery
The response was evaluated using CT or MRI after a minimum interval of about 6 weeks. Only in 8 patients neither of these tests could be performed. In all patients TME was performed. For lower rectal tumors close to the sphincter complex, an abdominoperineal resection (APR) was performed; however, for those with sufficient margin, a low anterior resection was carried out. Hartmann’s procedure was performed in patients with high surgical risk or as a temporary treatment to later perform reconstruction in patients with prolonged surgical time.
2.2.4. Adjuvant chemotherapy
Postoperative chemotherapy was optional based on the pathological features, postoperative and on distant risk factors dissemination. The most commonly used regimens were XELOX and CPC in monotherapy, or both, when oxaliplatin was withdrawn from XELOX due to poor tolerance/toxicity.
2.2.5. Response
During chemoradiotherapy, patients were evaluated weekly; a complete blood count and basic biochemistry were requested biweekly.
As for acute radiation-induced toxicity, data on diarrhea, cystitis, radiodermatitis, abdominal pain, and proctitis were collected and classified according to the CTCAE version 4.0. Likewise, chronic GI and GU toxicity were recorded starting from the first year after completing treatment, with serial records at the third and fifth years.
The usual follow-up for patients was every 3-4 months during the first two years, every 6 months until the fifth year, and then annually until discharge, which could be up to 10 years. For complementary tests, the recommendations of the ESMO guidelines [
11] were followed.
To evaluate the tumor response to chemoradiotherapy, the four-category tumor regression grading (TRG) system developed by the American Joint Committee on Cancer and the College of American Pathologists (AJCC/CAP) was used: TRG 0 (complete response, no tumor cells), TRG 1 (near complete response, isolated cells or small groups of isolated cells), TRG 2 (partial response, residual tumor with evident regression but more groups of cells), TRG 3 (poor or no response, extensive residual tumor with no evidence of regression). The following variables were also collected: lymphovascular, perineural, and extramural vascular invasion and the status of the resection margins (proximal, distal, and circumferential) according to AJCC/TNM R0, R1, and R2, based on whether the resection is clear, microscopically involved, or macroscopically involved, respectively.
Postoperative complications were defined by surgical, infectious, cardiovascular, or other causes, and the need for surgical re-intervention in these patients.
Surgical mortality was defined as that occurring within the first 30 days after the intervention. For the survival analysis, the following variables were defined:
Overall survival (OS). Calculated as the interval of time from the date of surgery, considered the definitive curative intent treatment, to the patient’s death from any cause. Patients who were alive at the end of the study were censored at the time of the last follow-up visit.
Disease-free survival (DFS). Calculated as the time from the date of surgery to the date of detection of the first disease relapse and/or death, whichever event occurred first. Patients who were alive and disease-free were censored at the time of the last follow-up visit.
2.3. Statistical Analysis
As measures of central tendency and dispersion for quantitative variables, the median and interquartile range were described, respectively. For categorical variables, frequency statistics such as absolute and relative percentages were used.
We evaluated the association between two categorical variables using the Chi-square method, and if an expected frequency was less than 5, Fisher’s exact test was used. In contrast, the association between quantitative variables was evaluated using t-Student tests or analysis of variance (ANOVA), and continuous variables were analyzed with the Mann-Whitney U test. The precision and statistical significance in all cases were at a 95% confidence interval (CI) and a significance level of 5% (p-value < 0.05), respectively.
For the analysis of overall survival and disease-free survival, estimations were made for the entire series and comparisons between patients with different responses to neoadjuvant treatment using the Kaplan-Meier method. The precision and statistical significance in all cases were at a 95% confidence interval (CI) and a significance level of 5% (p-value < 0.05), respectively.
To build the models, a univariate analysis will be conducted where variables with a p-value > 0.10 will be selected for inclusion in the multivariate model. In the multivariate analysis, only variables with a p-value < 0.05 will be considered significant. Data were analyzed using the Stata® statistical software version 14.2 (StataCorp LP, College Station, TX, USA).
3. Results
3.1. Characteristics of the Patients
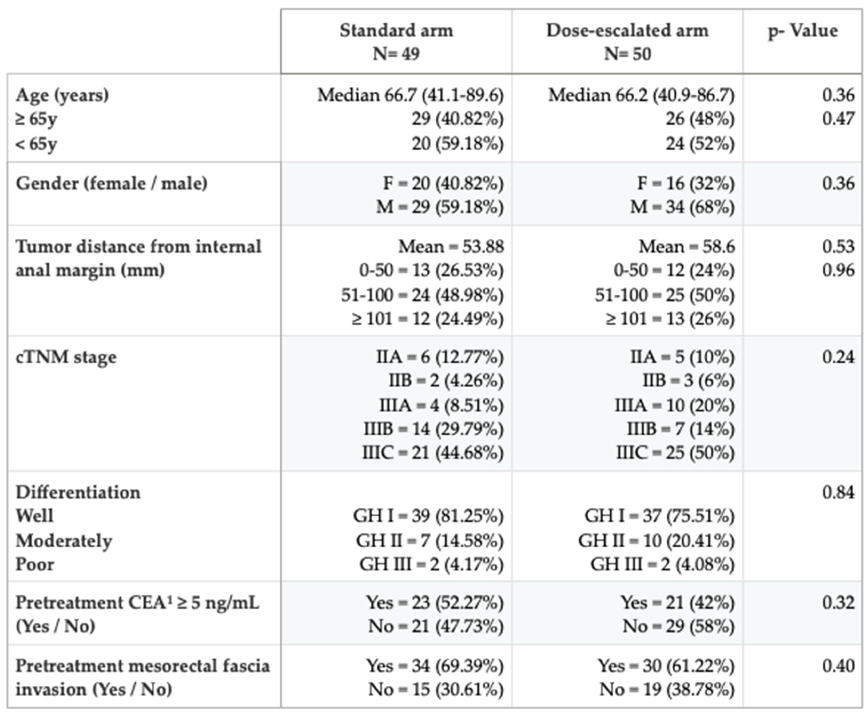
From 2012 to 2019, 99 patients were retrospectively analyzed (standard group: 49, and dose escalation group: 50) with a median follow-up of 5.93 years (IQR 0.03-11.33). The characteristics of the patients by group of treatment are shown in
Table 1. The proportions between the treatment arms are similar, indicating that both arms are well-balanced. The variables that influence the achievement of pathological complete responses are analyzed in the present study.
3.2. Surgery
The surgical procedure par excellence for preserving the sphincter complex is the low anterior resection, which was performed on the majority of patients. The second most performed intervention was the abdominoperineal resection. In only one patient from the dose escalation group, a curative surgery was not performed; instead, due to an ypCR and the advanced age of the patient, a local excision was decided upon. This patient was not included in the disease control and survival analyses as they did not meet the criteria for definitive treatment.
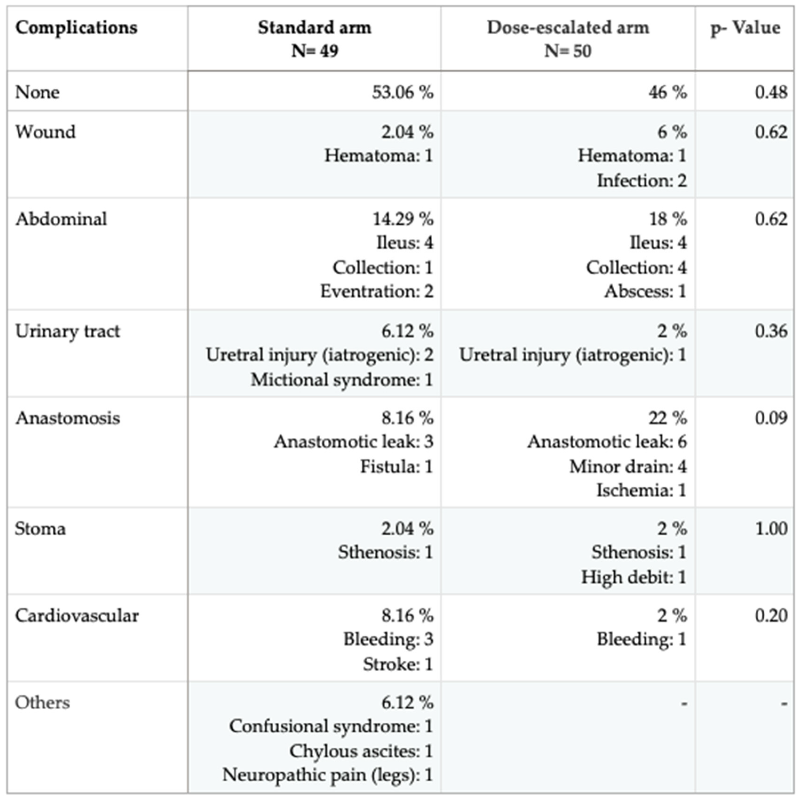
There were no differences between the groups in terms of all postoperative complications (p = 0.15), second surgeries (p = 0.62), or deaths during postoperative recovery (p = 0.62). The most common complication was anastomotic dehiscence or leakage in the dose escalation group, which involved 2 more re-interventions in this group than in the standard dose group
(p = 0.09
). This was followed by paralytic ileus and intra-abdominal collection as the second most common complication in the dose escalation group and the first in the standard group. All paralytic ileus cases, except for one death in the dose escalation group, were resolved with conservative treatment, and the collections were resolved by drainage from interventional radiology. Two patients in the standard group and one in the dose escalation group died within 30 days following the intervention. The causes of death in the standard group were a stroke and persistent anastomotic failure. All complications experienced by the patients can be found in
Table 2.
3.3. Pathological Response and Downstaging
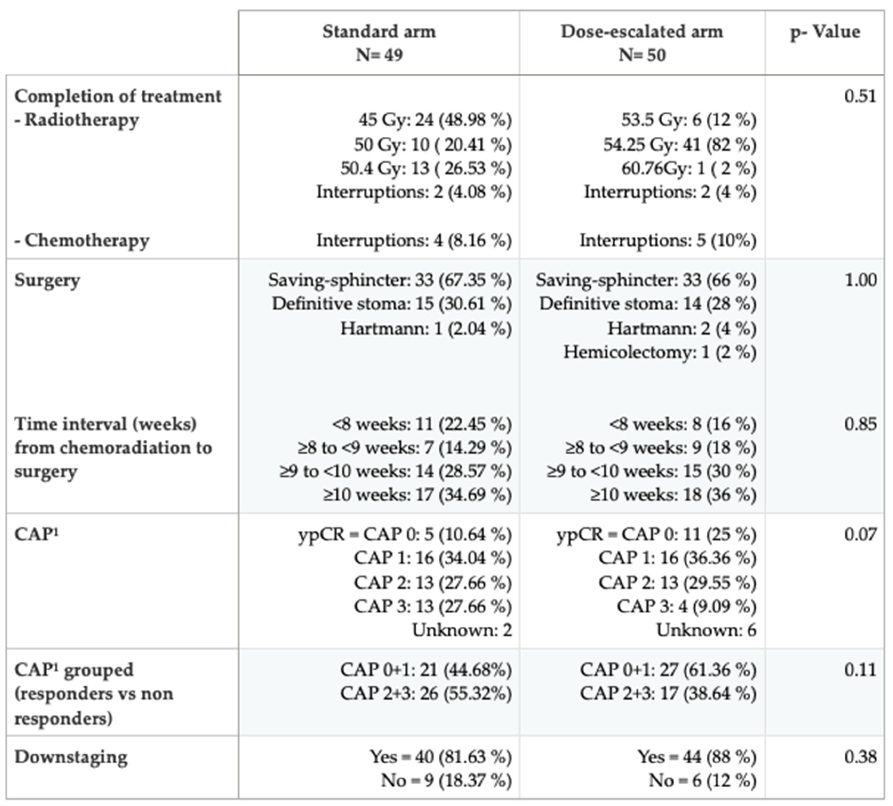
The dose-escalated group obtained more than double the number of pathological complete responses vs. standard group (25% vs. 10.64%; p = 0.07), showing a trend towards significance; and a lower rate of no response to treatment (38.64% vs. 61.36%; p = 0.11
). In contrast, the rate of responders and non-responders in the standard group is similar. Downstaging was similar in both groups. Variables related to our treatment protocol and the response by groups are shown in
Table 3.
3.4. Acute and Chronic Toxicity
Completion of neoadjuvant treatment in both groups was satisfactory and a similar number of patients had interruptions in both groups. The most common cause of interruption was grade III diarrhea, followed by proctitis. There were twice as many interruptions associated with chemotherapy since these patients continued radiotherapy without CPC until resolution. The cases of radiotherapy interruption (2 patients in each arm) corresponded to the non-administration of the last sessions due to grade III diarrhea.
Grade II acute-GI toxicity (according to CTCAE version 4.0) was lower in the standard group (16.33% vs. 24%; p = 0.06). Only 4 patients (8%) in the dose escalation group experienced grade III acute gastrointestinal toxicity, and none in the standard group. Grade II acute GU toxicity was lower in the standard group than in the dose escalation group (6.12% vs. 8%; p = 0.39). No patient experienced a higher grade of toxicity. Grade I GI toxicity at 3 years was lower in the standard group (12% vs. 18%; p = 0.25). Only 2 patients (4.55%) had grade II, and 1 patient (2.27%) had grade III in the dose escalation group. Most patients in both groups were asymptomatic at 3 years in terms of GU toxicity (97.62% vs. 95.45%; p = 1.00).
3.5. Bivariable and Multivariate Analysis
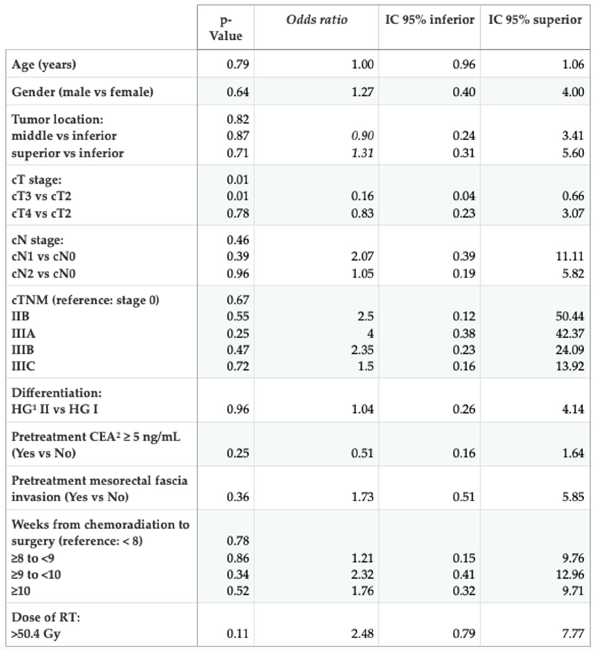
The variables analyzed were: age, sex, tumor distance from the anal margin, tumor size (cT) and number of positive lymph nodes (cN), histological grade and CEA at diagnosis, tumor distance to the mesorectal fascia (measured by MRI at diagnosis), and the interval of time from neoadjuvant therapy to surgery.
In the bivariable analysis, we found that for achieving a complete pathological response, smaller tumor size (cT2) is the most favorable factor
(p = 0.01). Although the RT dose did not reach statistical significance, its OR (
Table 4) shows that with dose escalation, more than double the complete pathological responses can be obtained compared to the standard dose (25% vs. 10.64%), not being statistically significant due to the sample size limitation (p = 0.07). Since only one variable reached statistical significance, no further multivariate analysis was performed.
3.6. Control and Survival
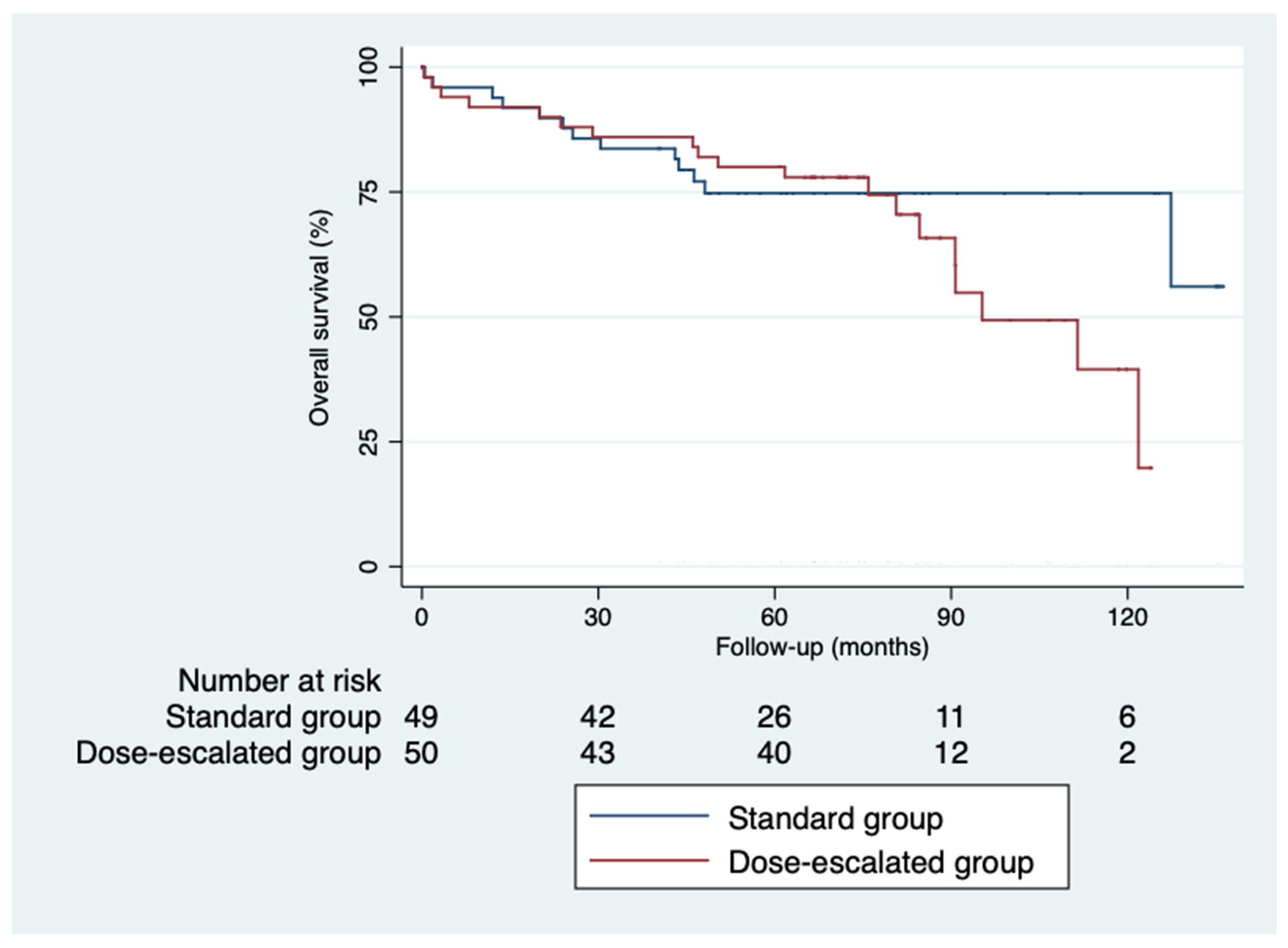
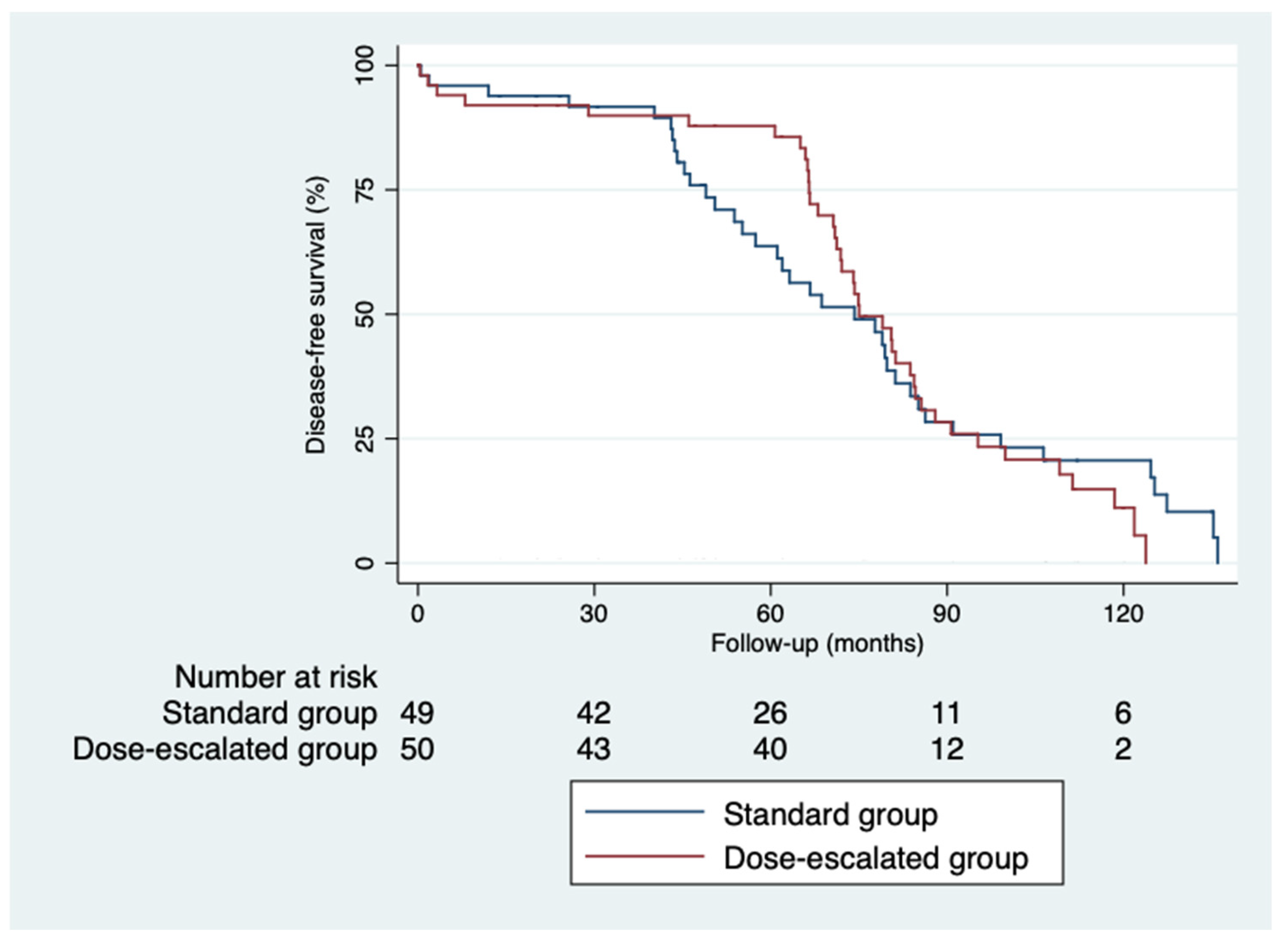
The 5-year disease-free survival was higher in the dose escalation group than in the standard group (87.6% vs. 64%; p = 0.82), as was the 5-year overall survival (81.6% vs. 74.6%; p = 0.32) [
Figure 1 and
Figure 2]. However, we did not find a benefit in terms of outcomes despite achieving higher rates of pathological complete response with a median follow-up of 5.93 years (IQR 0.03-11.33). Local control was similar in both groups (
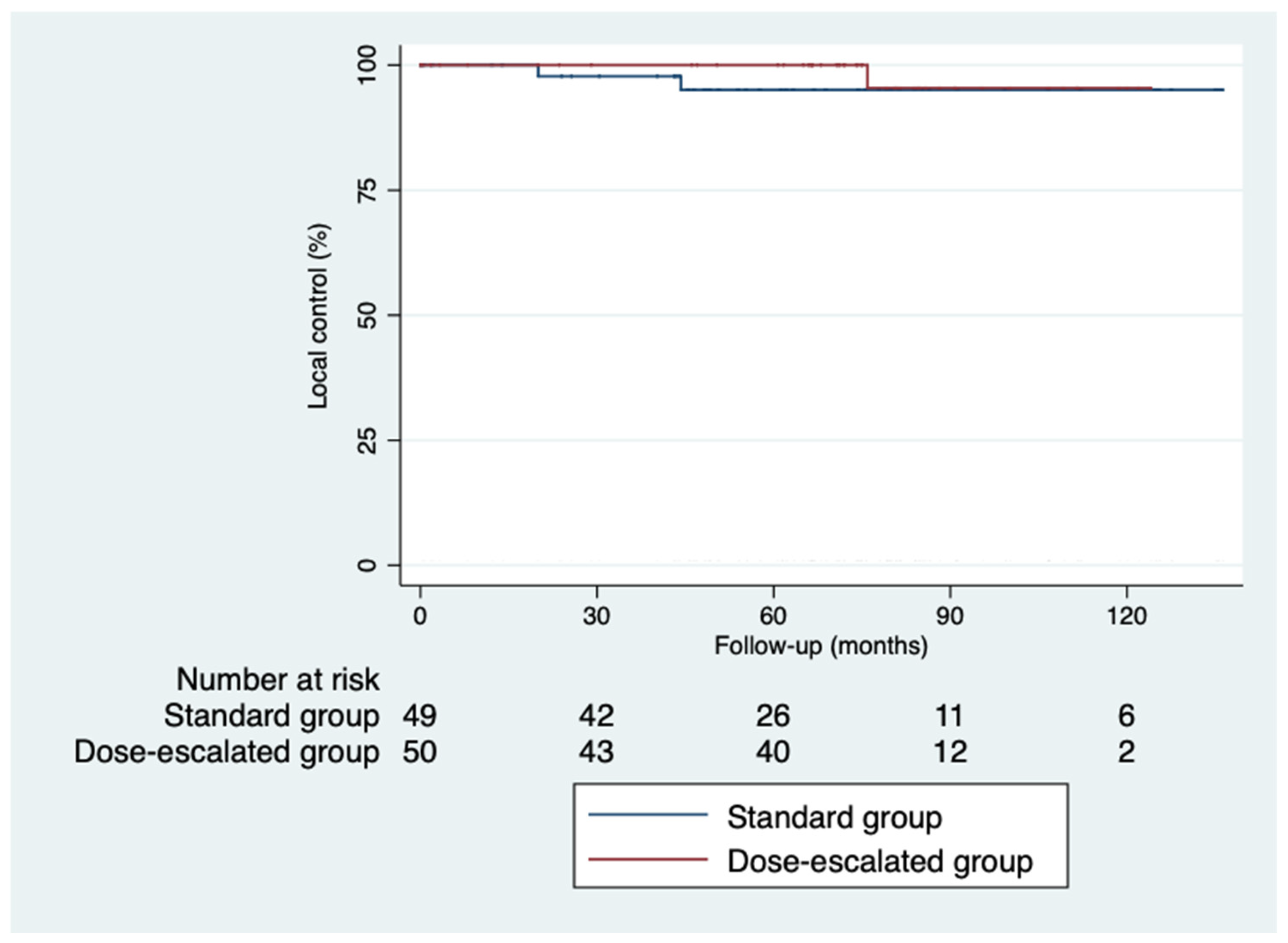
Figure 3). Only 3 local recurrences were documented in the entire series: 1 in the dose escalation group founded outside the irradiation field, and 2 recurrences in the standard group located within the irradiation field. Nine distant recurrences were reported in each group. Regarding patients who achieved an ypCR, only 2 of them died from rectal cancer. Concerning recurrences in patients with ypCR, only 2 patients had a distant recurrence, and none had a locoregional recurrence.
Figure 3.
Kaplan-Meier curves for overall survival.
Figure 3.
Kaplan-Meier curves for overall survival.
Figure 4.
Kaplan-Meier curves for disease-free survival.
Figure 4.
Kaplan-Meier curves for disease-free survival.
Figure 5.
Kaplan-Meier curves for local control.
Figure 5.
Kaplan-Meier curves for local control.
4. Discussion
Intensifying the neoadjuvant treatment by increasing the radiotherapy dose in patients diagnosed with locally advanced rectal cancer (LARC) achieved ypCR rates of 25% in our series, similar to those reported in the literature in dose escalation studies [
36,
37]. These results are higher than expected with the administration of the standard radiotherapy regimen [
18,
19,
20], without a relevant increase in postoperative complications or toxicity.
Preoperative treatment of locally advanced rectal cancer with chemoradiotherapy was established as the standard to reduce tumor volume and achieve R0 surgeries, as well as lower rates of complications and toxicity compared to administering radiotherapy postoperatively [
21]. Since then, new neoadjuvant treatment regimens have been sought to increase disease-free survival and overall survival. The ypCR is considered an independent factor for increasing local control (LC) and disease-free survival (DFS) [
38,
39,
40,
41]. Our treatment regimen is based on the Xelac arm of the phase III Interact Trial [
42], which establishes concurrent boost with capecitabine as the reference regimen for neoadjuvant therapy. Our primary objective was to compare ypCR in a group of patients treated with integrated dose escalation of the tumor versus standard doses of radiotherapy. Unlike the Interact Trial, where the boost was administered twice a week for 5 weeks, in our case, it was administered from the beginning at a dose of 2.16 Gy/day reaching 54 Gy instead of the 55 Gy of the Interact Trial, although this represents an equivalent biological dose (BED). The goal of increasing the number of ypCR with dose escalation versus the standard regimen was achieved (25% vs 10.64%; p = 0.07). Both short, and long-term toxicity were acceptable, considering our regimen safe.
To our knowledge, the first study that had evaluated the pathological response rate by increasing the RT dose in American patients to 52.5 Gy in conjunction with intravenous 5 FU was published in 2000 [
43]. Subsequently, in 2006, the same group published long-term results for 54 patients, reporting the same RT dose, but concurrent with oral capecitabine, demonstrating a safety profile. They achieved an 18% ypCR rate and a 59% downstaging rate. The first meta-analysis published in 2014 gathered studies conducted to date with 3D irradiation technique using dose escalation via linear accelerator or, in some cases, high-dose-rate brachytherapy; none used IMRT technique. It concluded that increasing the dose to ≥60 Gy in LARC increases the ypCR rate with acceptable acute toxicity, although randomized controlled trials with larger patient samples are needed [
36]. The second, more recent meta-analysis from 2021 includes studies using intensity-modulated radiation therapy (IMRT) with accelerated boost (between 54-60 Gy) that were not included in previous studies. It concludes that a dose of ≥54 Gy is associated with high ypCR rates close to 25%, achieved with a dose boost of between 54 and 60 Gy with IMRT, all without increasing the risk of grade III acute toxicity. However, a clear dose-response relationship was not identified in the regression analysis. The optimal boost dose has not been determined, and data on late toxicity and long-term oncological outcomes are lacking in the literature [
37]. Other studies using integrated boost with IMRT are those by Hernando-Requejo [
44] and Jin-luan Li [
45], reporting ypCR rates close to 31%. Except for these two studies, we do not know any others that report higher ypCR rates.
From the bivariate analyses, we found that the variable influencing the achievement of ypCR is clinical tumor size (cT). Patients with a favorable profile for better tumor regression are those with tumors not presenting deep invasion. A recently published retrospective study similar to ours by Qi Zhang [
46] found that CEA and surgical interval >8 weeks are variables influencing higher pathological response rates. In our case, we did not obtain sufficient statistical significance due to the smaller sample size. However, we obtained a higher ypCR rate (25% in our series vs. 20.8% reported in Qi Zhang’s study). Since the introduction of advanced techniques (IMRT) no more patients are treated with 3DRT planning in our institution and that explains the sample of this study. The p-value of 0.09 for suture failures in anastomoses is not negligible. Confounding factors such as the surgical team with practicing surgeons, the type of suture material used, or intraoperative complications that might explain this higher rate have not been considered. A review is planned in the near future with an IMRT dose-escalated boost using the same scheme that is reported in the present study.
5. Conclusions
In our study we found a trend towards a higher ypCR-rates with neoadjuvant 3DRT intensification compared to the standard scheme with no differences in terms of postoperative complications and acceptable early and late toxicity rates. Long-term follow-up shows no differences in disease-free and overall survival. Well-designed randomized and controlled trials are needed to obtain conclusive data.
Author Contributions
Conceptualization M.S., D.D., C.D.-B.; methodology D.D., T.T.-B., C.D.-B.; validation M.S., T.T.-B., C.D.-B.; formal analysis C.D.-B., D.D.; investigation T.T.-B., C.D.-B., F.L.-C.; resources M.S., T.T.-B.; data curation C.D.-B., M.S.; writing—original draft preparation C.D.-B., D.D., T.T.-B; writing—review and editing C.D.-B., D.D., T.T.-B., M.S., F.L.-C.; visualization C.D.-B., F.L.-C.; supervision C.D.-B., F.L.-C.; project administration C.D.-B. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sociedad Española de Oncología Médica. Available online: https://seom.org/images/publicaciones/informes-seom-de-evaluacion-de-farmacos/LAS_CIFRAS_2024.pdf (accessed on 8th April 2024).
- Siegel, R.L.; Miller, K.D.; et al. Colorectal cancer statistics. Cancer J Clin 2017, 67, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Bown, E.J.; Lloyd, G.M.; et al. Rectal cancer: Prognostic indicators of long-term outcome in patients considered for surgery. Int J Colorectal Dis 2014, 29, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Compton, C.C.; Fielding, L.P.; et al. Prognostic factors in colorectal cancer. Arch Pathol Lab Med 2000, 124. [Google Scholar] [CrossRef] [PubMed]
- Betge, J.; Pollheimer, M.J.; et al. Intramural and extramural vascular invasion in colorectal cancer. Cancer 2012, 118, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Liebig, C.; Ayala, G.; et al. Perineural invasion is an independent predictor of outcome in colorectal cancer. J Clin Oncol 2009, 27, 5131–5137. [Google Scholar] [CrossRef] [PubMed]
- Dukes, C.E.; Bussey, H.J.R. The spread of rectal cancer and its effect on prognosis. Br J Cancer 1958, 12, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Blenkinsopp, W.K.; Stewart-Brown, S; et al. Histopathology reporting in large bowel cancer. J Clin Pathol 1981, 3, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Revista Ciencia y Salud. Available online: http://revistacienciaysalud.ac.cr/ojs/index.php/cienciaysalud/article/view/25 (accessed on 23th November 2023).
- Van Cutsem, E.; Nordlinger, B.; et al. Advanced colorectal cancer: ESMO Clinical Practice Guidelines for treatment. Ann Oncol 2010, 21, v93–v97. [Google Scholar] [CrossRef]
- Schmoll, H.J.; Van Cutsem, E.; et al. ESMO Consensus Guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann Oncol 2012, 23, 2479–2516. [Google Scholar] [CrossRef]
- Krook, J.E.; Moertel, C.G.; et al. Effective surgical adjuvant therapy for high-risk rectal carcinoma. N Engl J Med 1991, 324, 709–715. [Google Scholar] [CrossRef]
- Gastrointestinal Tumor Study Group. Radiation therapy and fluorouracil with or without semustine for the treatment of patients with surgical adjuvant adenocarcinoma of the rectum. J Clin Oncol 1992, 10, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Hofheinz, R.; Wenz, F.K.; et al. Capecitabine (Cape) versus 5-fluorouracil (5-FU)–based (neo)adjuvant chemoradiotherapy (CRT) for locally advanced rectal cancer (LARC): Long-term results of a randomized, phase III trial. J Clin Oncol 2011, 29, 3504–3504. [Google Scholar] [CrossRef]
- Swedish Rectal Cancer Group. Improved survival with preoperative radiotherapy in resectable rectal cancer. N Engl J Med 1997, 336, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Cammà, C.; Giunta, M.; et al. Preoperative radiotherapy for resectable rectal cancera meta-analysis. JAMA 2000, 284, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Colorectal Cancer Collaborative Group. Adjuvant radiotherapy for rectal cancer: A systematic overview of 8507 patients from 22 randomised trials. The Lancet 2001, 358, 1291–1304. [Google Scholar] [CrossRef] [PubMed]
- Bosset, J.F.; Collette, L.; et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med 2006, 355, 1114–1123. [Google Scholar] [CrossRef] [PubMed]
- Saif, M.W.; Hashmi, S.; et al. Capecitabine vs continuous infusion 5-FU in neoadjuvant treatment of rectal cancer. A retrospective review. Int J Colorectal Dis 2008, 23, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Gérard, J.P.; Azria, D.; et al. Comparison of two neoadjuvant chemoradiotherapy regimens for locally advanced rectal cancer: Results of the Phase III Trial ACCORD 12/0405-prodige 2. J Clin Oncol 2012, 28, 1638–1644. [Google Scholar] [CrossRef] [PubMed]
- Sauer, R.; Becker, H.; et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004, 351, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Glynne-Jones, R.; Wyrwicz, L.; et al. Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 2017, 28, iv22–iv40. [Google Scholar] [CrossRef]
- Hong, Y.S.; Kim, S.Y.; et al. Oxaliplatin-based adjuvant chemotherapy for rectal cancer after preoperative chemoradiotherapy (ADORE): Long-term results of a randomized controlled trial. J Clin Oncol 2019, 37, 3111–3123. [Google Scholar] [CrossRef] [PubMed]
- Bahadoer, R.R.; Dijkstra, E.A.; et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): A randomised, open-label, phase 3 trial. Lancet Oncol 2021, 22, 29–42. [Google Scholar] [CrossRef]
- Ciseł, B.; Pietrzak, L.; et al. Long-course preoperative chemoradiation versus 5 × 5 Gy and consolidation chemotherapy for clinical T4 and fixed clinical T3 rectal cancer: Long-term results of the randomized Polish II study. Ann Oncol 2019, 30, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aguilar, J.; Smith, D.D.; et al. Optimal timing of surgery after chemoradiation for advanced rectal cancer: Preliminary results of a multicenter, nonrandomized phase II prospective trial. Ann Surg 2011, 254, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Chi, P.; et al. Neoadjuvant modified FOLFOX6 with or without radiation versus fluorouracil plus radiation for locally advanced rectal cancer: Final results of the chinese FOWARC trial. J Clin Oncol 2019, 37, 3223–3233. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Martos, C.; Garcia-Albeniz, X.; et al. Chemoradiation, surgery and adjuvant chemotherapy versus induction chemotherapy followed by chemoradiation and surgery: Long-term results of the Spanish GCR-3 phase II randomized trial. Ann Oncol 2015, 26, 1722–1728. [Google Scholar] [CrossRef] [PubMed]
- Petrelli, F.; Trevisan, F.; et al. Total neoadjuvant therapy in rectal cancer: A systematic review and meta-analysis of treatment outcomes. Ann Surg 2020, 271, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Vecchio, F.M.; Valentini, V.; et al. The relationship of pathologic tumor regression grade (TRG) and outcomes after preoperative therapy in rectal cancer. Int J Radiat Oncol Biol Phys 2005, 62, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Habr-Gama, A.; Perez, R.O.; et al. Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: Long-term results. Ann Surg 2004, 240, 711–718. [Google Scholar] [CrossRef]
- Garcia-Aguilar, J.; Patil, S.; et al. Organ preservation in patients with rectal adenocarcinoma treated with total neoadjuvant therapy. J Clin Oncol 2022, 40, 2546–2556. [Google Scholar] [CrossRef]
- Greene, F.L. American Joint Committee on Cancer staging manual, 6th ed. Springer-Verlag: New York, EEUU, 2002; pp 421.
- Nijkamp, J.; de Jong, R.; et al. Target volume shape variation during irradiation of rectal cancer patients in supine position: Comparison with prone position. Radiother Oncol 2009, 93, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.D.; Nijkamp, J.; et al. Prospective randomized double-blind pilot study of site-specific consensus atlas implementation for rectal cancer target volume delineation in the cooperative group setting. Int J Radiat Oncol Biol Phys 2011, 79, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Burbach, J.P.M.; den Harder, A.M.; et al. Impact of radiotherapy boost on pathological complete response in patients with locally advanced rectal cancer: A systematic review and meta-analysis. Radiother Oncol 2014, 113, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hearn, N.; Atwell, D.; et al. Neoadjuvant radiotherapy dose escalation in locally advanced rectal cancer: A systematic review and meta-analysis of modern treatment approaches and outcomes. Clin Oncol 2021, 33, e1–e14. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aguilar, J.; Hernandez De Anda, E.; et al. A pathologic complete response to preoperative chemoradiation is associated with lower local recurrence and improved survival in rectal cancer patients treated by mesorectal excision. Dis Colon Rectum 2003, 46, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Capirci, C.; Valentini, V.; et al. Prognostic value of pathologic complete response after neoadjuvant therapy in locally advanced rectal cancer: Long-term analysis of 566 ypCR patients. Int J Radiat Oncol 2008, 72, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Vecchio, F.M.; Valentini, V.; et al. The relationship of pathologic tumor regression grade (TRG) and outcomes after preoperative therapy in rectal cancer. Int J Radiat Oncol Biol Phys 2005, 62, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.T.; Heneghan, H.M.; Winter, D.C. Systematic review and meta-analysis of outcomes following pathological complete response to neoadjuvant chemoradiotherapy for rectal cancer. Br J Surg 2012, 99, 918–928. [Google Scholar] [CrossRef] [PubMed]
- Valentini, V.; Gambacorta, M.A.; et al. The INTERACT Trial: Long-term results of a randomised trial on preoperative capecitabine-based radiochemotherapy intensified by concomitant boost or oxaliplatin, for cT2 (distal)–cT3 rectal cancer. Radiother Oncol 2019, 134, 110–118. [Google Scholar] [CrossRef]
- Janjan, N.A.; Crane, C.N.; et al. Prospective trial of preoperative concomitant boost radiotherapy with continuous infusion 5-fluorouracil for locally advanced rectal cancer. Int J Radiat Oncol 2000, 47, 713–718. [Google Scholar] [CrossRef]
- Hernando-Requejo, O.; López, M.; et al. Complete pathological responses in locally advanced rectal cancer after preoperative IMRT and integrated-boost chemoradiation. Strahlenther Onkol 2014, 190, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Jin-luan, L.; Jia- fu, J.; et al. Preoperative concomitant boost intensity-modulated radiotherapy with oral capecitabine in locally advanced mid-low rectal cancer: A phase II trial. Radiother Oncol 2012, 102, 4–9. [Google Scholar] [CrossRef]
- Zhang, Q.; Liang, J.; et al. Predictive Factors for Pathologic Complete Response Following Neoadjuvant Chemoradiotherapy for Rectal Cancer. Asian Pac J Cancer Prev 2021, 22, 1607–1611. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).