1. Introduction
Total laryngectomy with loss of natural voice and permanent tracheostomy is one of the most mutilating surgical procedures in the head and neck region, leaving patients with significant physical impairments and social stigmatization [
1]. Therefore, the development of non-surgical treatment strategies that provide comparable survival outcomes and allow preservation of the larynx in almost 60% of cases is an important alternative for patients with locally advanced laryngeal and hypopharyngeal cancer [
2]. In this context, it is important to emphasize the importance of preserving the larynx functionally and not just anatomically, allowing natural speech, swallowing and breathing without the need for permanent tube feeding and/or tracheostomy. According to the RTOG 91-11 trial, patients treated with concurrent chemoradiotherapy (cCRT) with cisplatin have the best chance of laryngeal preservation, albeit with no improvement in survival compared to induction chemotherapy (ICT) followed by radiotherapy (RT) or RT alone [
3,
4]. However, the impact of the primary tumor stage, including the mobility status of the vocal cords, and the location of the primary tumor (larynx vs. hypopharynx) must be considered when assessing the prospects for a favorable treatment outcome.
Since not all tumors are equally sensitive to chemotherapeutic agents and radiation, the ability to identify patients with resistant tumors early in the course of non-surgical therapy who should be treated with laryngectomy is crucial. Several studies have confirmed that ICT is a reliable in vivo assay for determining the chemo/bio/radiosensitivity of tumors [
5,
6]. In routine clinical practice, after a favorable response to ICT, which is used as a biomarker for patient selection, patients are subsequently treated with RT; the use of a sensitizer concurrently with radiation should be considered investigational [
7]. The latter is due to the lack of survival benefit observed in phase 3 clinical trials comparing taxane-based ICT followed by aggressive chemoradiation with chemoradiation alone [
8]. In addition, the toxicity of three cycles of taxane-based induction chemotherapy is significant and may seriously compromise the administration of subsequent concurrent chemo/bio-RT [
9].
To mitigate the negative effects of aggressive ICT therapies on subsequent cCRT, the response to a single test cycle of ICT has been proposed as a criterion for selecting patients for non-surgical treatment [
10]. Excellent survival results were achieved with this approach, with the larynx being preserved in 66% of patients [
11]. In the present study, we retrospectively analyzed the results of larynx-preserving treatment in a prospective cohort of patients with laryngeal and hypopharyngeal cancer who received a limited number of ICT cycles as a chemotherapy response selection strategy prior to platinum-based cCRT. The hypothesis tested was whether the clinical response of the tumor after a single cycle of ICT can be used to differentiate between chemo-/radiosensitive and resistant tumors.
2. Materials and Methods
The study was conducted in accordance with the Declaration of Helsinki and approved by the Protocol Review Committee of the Institute of Oncology Ljubljana, Slovenia.
2.1. Patients
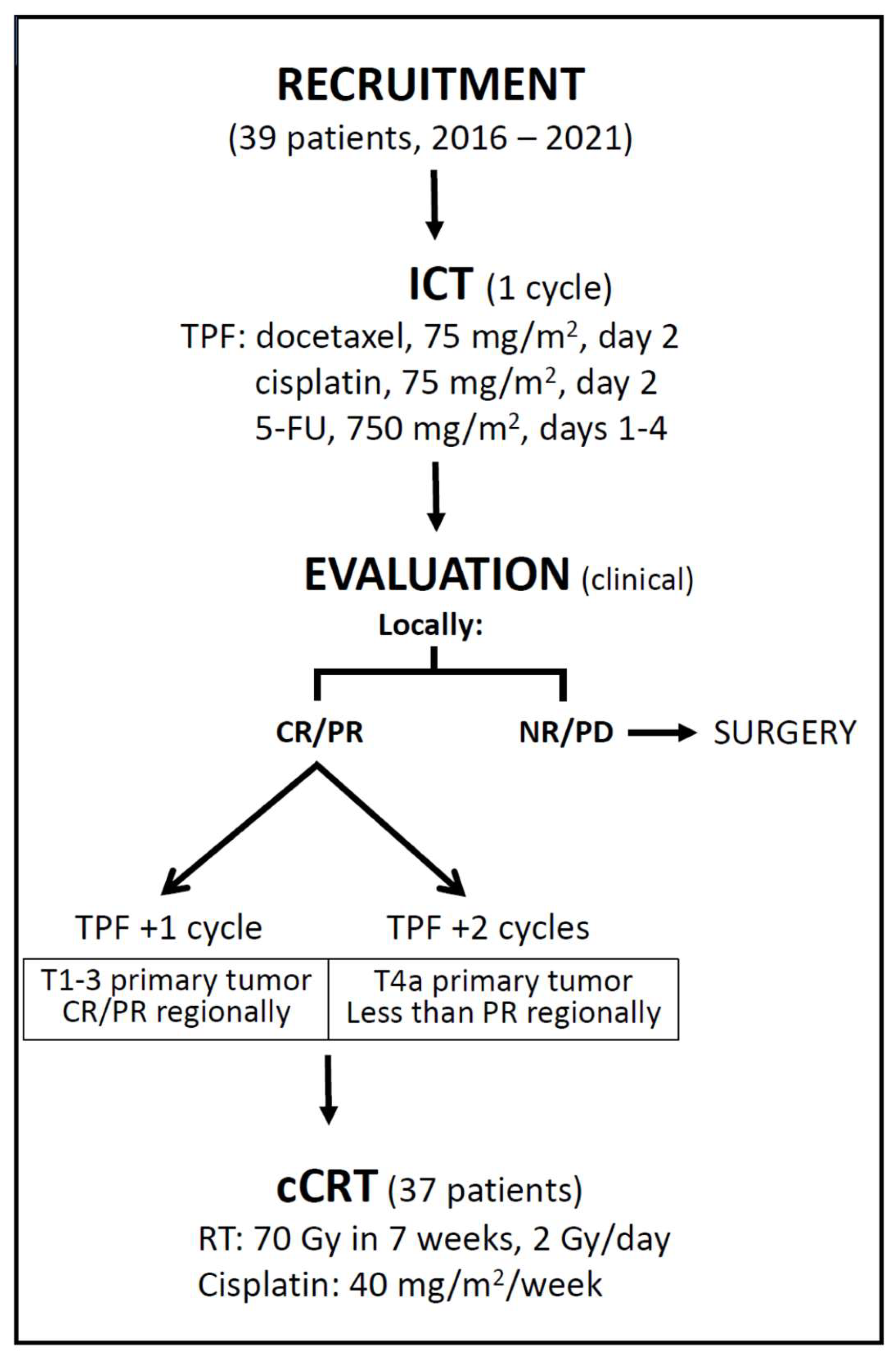
In 2016, a chemoselection program was introduced based on clinical assessment of tumor response after a single cycle of ICT for patients with laryngeal and hypopharyngeal cancer. At the Multidisciplinary Tumor Board, all consecutive patients with newly diagnosed squamous cell carcinoma of the larynx or hypopharynx (cT2-4a, cN0-3resectable, M0) who were suitable for total laryngectomy (with or without partial hypopharyngectomy) as the only possible surgical option were offered this treatment option. Further inclusion criteria were a WHO performance status of 0-1 and adequate laboratory tests. Patients with tracheostomy, feeding tube, recurrent pneumonia (requiring hospitalization within the last 12 months) suggestive of laryngeal dysfunction, or patients with a medical condition that would prevent safe delivery of the planned therapies were considered ineligible.
2.2. Treatment Protocol and Response Evaluation
Prior to treatment, the stage of disease was determined by physical, endoscopic and radiologic (CT and/or chest X-ray and/or abdominal US) examination of the upper aerodigestive tract, neck, chest and abdomen. Other tests (PET-CT, bone scan) were performed when clinically indicated.
ICT consisted of two cycles of docetaxel (75 mg/m2, day 2), cisplatin (75 mg/m2, day 2) and 5-fluorouracil, 750 mg/m2/day, days 1–4 in 96-hour infusion). The substitution of cisplatin with carboplatin (AUC 5) and the dosing recommendations in case of toxicity have been as described elsewhere [
12].
Tumor response was assessed clinically and was performed 3 weeks after the start of ICT by transnasal endoscopy (local assessment) and palpation (regional assessment). A complete response (CR) was defined as the disappearance of all evaluable disease within the treatment field, and a partial response (PR) was defined as a decrease of no less than 50% and at least partial recovery of laryngeal mobility (in the case of initial immobility). Patients with CR or PR of the primary tumor received a second cycle of ICT followed by cCRT, and non-responders were referred for surgery (
Figure 1). Patients with responding primary tumors who were diagnosed with either T4a or less than PR in the neck after the first ICT cycle were planned for the 3rd cycle of ICT.
RT was scheduled to begin 3-4 weeks after the start of the last cycle of ICT. All patients had CT-based planning and were irradiated with a 6MV photon beam from a linear accelerator and a concomitant boost intensity modulated radiotherapy/volumetric modulated arc therapy technique. A dose of 70 Gy was delivered in 35 fractions over 7 weeks (1 fraction/day, 5 days/week) to the original (i.e., pre-ICT) high-risk clinical target volumes (CTV70), taking into account the anatomical barriers to tumor spread. The low-risk CTV56 included areas considered at risk for microscopic disease. The primary tumor and nodal levels were delineated according to the guidelines [
13,
14]. An isotropic margin of 5 mm was added around the corresponding CTVs to create the planning target volumes (PTVs). During RT, cisplatin was administered intravenously weekly at a dose of 40 mg/m2 and replaced by carboplatin (1.5 AUC) when creatinine clearance decreased to < 60 mL/min and/or peripheral polyneuropathy or grade > 1 hearing impairment was detected. Tumor response was assessed clinically and radiologically 8-14 weeks after cCRT according to RECIST criteria: partial responders and non-responders were referred for surgery if the residual tumor was considered operable.
The acute toxicity of ICT was monitored every 3 weeks and weekly during cCRT. In the first two years, patients were examined at 3-month intervals and later every 4-6 months for toxicity and possible recurrence of the disease or new primary tumors. Toxicities were graded according to the National Cancer Institute’s Common Terminology Criteria or Adverse Events v5.0.
2.3. Treatment Delivered
The majority, 27 (69%) patients, received 2 cycles of ICT. Five (13%) patients received only one cycle of ICT. The reasons for this were an unresponsive primary tumor (2), febrile neutropenia with sepsis (2) and the patient’s refusal to undergo further ICT after complete tumor disappearance (1). Three ICT cycles were administered in 7 (18%) patients, as 3 patients had a cT4a tumor and 4 patients had less than PR of neck metastases (Table 1).
The median interval between the first day of the last ICT cycle and the start of cCRT was 26 days (range 12-48, IQR 21-28 and the median duration of RT course was 51 days (range 47-55, IQR 50-51). Overall, depending on the number of administered ICT cycles, the duration of treatment was between 11 and 25 weeks (median 14, IQR 13-15).
2.4. Statistics
To ensure a potential minimum follow-up period of 2 years, only patients treated between 2016 and 2021 were analyzed. Descriptive statistics were reported with medians, ranges and interquartile ranges (IQR) for numeric variables and as percentages for categorical variables. Survival curves were calculated using the Kaplan-Meier method and the log-rank test was used to assess the differences between survival curves. All statistical tests were two-sided and a p-value of < 0.05 was considered statistically significant.
The primary study objective was 2-year laryngectomy-free survival (LFS, event: laryngectomy or death from any cause). Additional endpoints were locoregional control (LRC, event: local and/or regional recurrence or death from any cause except distant metastases); disease-free survival (DFS, event: cancer recurrence or death from any cause) overall survival (OS, event: death from any cause); laryngo-esophageal dysfunction-free survival (leDFS, event: local recurrence, total or partial laryngectomy, tracheostomy or feeding tube at 2 years or death) at 2 and 5 years; acute and late toxicities. Survival times were calculated from the first day of treatment. For patients who did not respond to ICT or did not achieve local and/or regional CR after completion of all therapies, the time to event was set to zero months.
3. Results
3.1. Response to Therapy and Survival
3.1.1. Response to Induction Chemotherapy
After the first cycle of ICT, transnasal endoscopy revealed CR and PR locally in 5 and 32 patients, respectively (95 % of patients in total) and two patients (5 %) were identified as local non-responders and sent for salvage surgery (
Table 2). There was no statistically significant difference in the proportion of CR and/or PR between patients with tumors of the larynx and hypopharynx. In 14 patients with initial vocal cord fixation, only partial restoration of hemilarynx mobility was observed in two patients and no change was observed in the patient with transglottic (cT3N0) tumor. The latter and the patient with a non-responding hypopharyngeal cT3N2b tumor were directed to surgery. Regionally, 19 of 23 patients (83 %) with cN+ disease responded to the first ICT cycle with at least PR.
3.1.2. Response to Concurrent Chemotherapy
Tumor response after cCRT was assessed in 37 patients with CT (26), PET-CT (8) or both (3). The median time to assessment after completion of cCRT was 13 weeks (range 7-26, IQR 11-17). Local CR was achieved in all (100 %) patients and regional in 19 of 22 N+ patients (86 %), resulting in a 92 % (34/37) CR rate above the clavicles. (
Table 2)
3.1.3. Salvage Therapies and Survival
The three patients with residual disease in the neck underwent unilateral neck dissection. Histopathologic report confirmed residual carcinoma in all three cases. Two of them remained tumor-free and the third patient was diagnosed with local recurrence and lung metastases. Of the 34 patients with CR after cCRT, the disease progressed in three patients (
Table 3). In seven patients (18 %), a new primary tumor developed 6 months to 5 years (median 2.2 years) after treatment (6 non-small cell lung carcinomas and uterine cervix carcinoma), which was the cause of death in 5 of them.
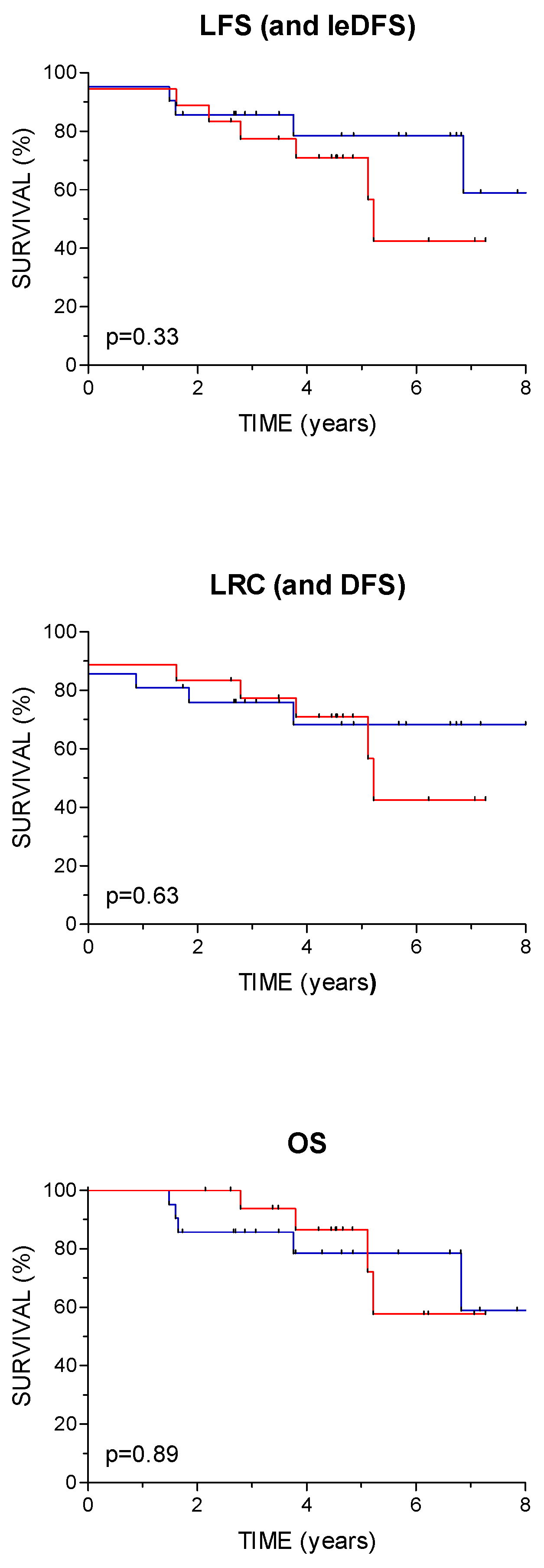
On the study close-out date (February 29, 2024), the median follow-up time was 3.8 years (range 1.5–8 years, IQR 3-6.2) and 4.5 years for patients still alive (range 2-8 years, IQR 3.2-6.4). At the last clinical visit, 29 patients were free of treated cancer and one had lung metastases. Two patients died from the treated malignancy (with locoregional recurrence and distant metastases) and seven patients died from intercurrent disease without evidence of index malignancy (second cancer – 5, pulmonary embolism, cardiac arrest). LFS at 2 and 5 years was 87 % (95 % CI 77-98) and 75 % (95 % CI 60-89), respectively, and the same rates apply to leDFS. The corresponding 2- and 5-year LRC rates (and also DFS) were 79 % (95 % CI 67-92) and 70 % (95 % CI 54-85) and for OS 92 % (95 % CI 84-100) and 82 % (95 % CI 69- 95), respectively. There were no statistically significant differences in LFS or other survival outcomes between patients with primary tumors of the larynx and hypopharynx (
Figure 2).
3.2. Toxicity
In three patients with a history of hearing impairment and impaired renal function, ICT was modified from the outset by replacing cisplatin with carboplatin, and in the second cycle two further patients were switched to carboplatin due to worsening of renal function. In 10 out of 34 patients, the dose of chemotherapeutic agent(s) had to be reduced during the second cycle, and in 2 out of 7 patients during the third cycle. The median change in body weight during ICT was positive (2.5% increase, range -10.2-25.8, IQR 0-5.5). The acute toxicities of the treatment are listed in
Table 4.
During cCRT, 5 (14 %) patients received less than 5 cycles of chemotherapy due to hematotoxicity (4) or patient refusal (1). Three patients (8 %) started concurrent carboplatin treatment (impaired hearing or renal function, chest pain during ICT) and nine (24 %) patients were switched to carboplatin during the course of cCRT due to renal toxicity (6), paresthesia (2) and persistent nausea (1). During the course of RT, eight patients (22 %) developed grade 3 radiomucositis and 2 patients (8 %; in the 5th and 6th week of RT) had a nasogastric tube placed; one of them remained tube-dependent. The median weight loss during cCRT was 7.3% (range 1.1-16.5, IQR 4.1-11.6) (
Table 4).
Late treatment-related toxicities are listed in
Table 5. Of 32 patients who survived more than 6 months after therapy and had no active disease, four (13 %) experienced no treatment-related adverse events and 16 patients (50 %) experienced grade ≥2 toxicity. No grade 4 or 5 adverse event was recorded. Twelve of these patients (38 %) continued to smoke after stopping therapy.
4. Discussion
The results of the present study confirmed the hypothesis that treatment selection based on response to chemotherapy with clinical evaluation of tumor response after a single cycle of TPF ICT is a valid discriminator between chemo-/radiosensitive and resistant tumors. The proportion of patients in whom laryngectomy was successfully avoided in the long term (i.e. 5-year LFS) was encouraging and apparently higher than in the most favorable cCRT arm of the pivotal RTOG 91-11 trial, suggesting the superiority of the combination of TPF ICT and platinum-based cCRT over cCRT alone in the larynx preservation setting [
4].
When considering non-surgical treatment options for locally advanced laryngeal and hypopharyngeal tumors, the imperative is to preserve the functional organ without jeopardizing survival. For this reason, the correct selection of candidates is crucial. Any baseline dysfunction of the larynx or major involvement of the laryngeal skeleton by a tumor reduces the possibility of satisfactory restoration of laryngeal function after treatment. Therefore, the selection of patients for non-surgical treatment is usually limited to patients with T2 and T3 tumors who are not suitable for partial surgical procedures, although some patients with T4a tumors can also breathe adequately and swallow solid and liquid food safely and have a usable voice [
7,
15]. Our patients were only offered larynx preservation after a thorough discussion of all the advantages and disadvantages in the multidisciplinary tumor board. None of them were dependent on a tracheostomy or feeding tube prior to treatment, and 92 % of the primary tumors were stage T2 or T3. In addition, the relatively low median age and low comorbidity burden assured the best possible adherence to the planned oncological treatment.
Although a meta-analysis has confirmed the association between poor response to ICT and subsequent (cC)RT in larynx preservation trials, the optimal number of ICT cycles to reliably identify tumors suitable for non-surgical treatment scenarios is not known. According to the Larynx Preservation Consensus Panel, assessment should be performed after two cycles of ICT [
16]. However, Urba et al. showed in their phase II study that after a single cycle of cisplatin/5FU ICT, 75 % of laryngeal cancer patients achieved at least a partial response at the primary site; after cCRT in responders or surgery in non-responders, DFS was 80 % and 78 % after 2 and 3 years, respectively [
10]. These results compare favorably to the larynx preservation trials, which used more than one cycle of ICT, and underscore the prognostic importance of tumor shrinkage dynamics during ICT [
5]. Several other authors later confirmed the prognostic value of tumor shrinkage after the first cycle of ICT [
17,
18]. Furthermore, tumor assessment by flexible endoscopy appears to be reliable in view of survival outcomes, as it correlates well with volumetric CT measurements but has a higher interobserver reliability [
19,
20]; moreover, it is easy to perform even in the context of a busy clinical routine. Alternative methods of assessing tumor response to ICT using modern imaging techniques, blood biomarkers or a specific gene signature are either much more complex or less validated, but do not appear to be more reliable [
21,
22,
23].
Since we used the TPF ICT regimen, the proportion of responders in our series was, as expected, higher (95 %) than in the previously mentioned study by Urba et al. (75 %) or in other studies using a platinum-based/5FU combination [
3,
10]. However, it was comparable to the reported experience with TPF ICT, although tumor response assessment in these studies occurred later in the course of ICT [
9,
24]. Another observation in our study is that both primary laryngeal and hypopharyngeal tumors responded to TCF ICT, with no statistically significant differences in the proportion of CRs and PRs between the two groups. The reliability of early assessment of tumor response during the ICT phase of treatment, as applied in our study, was confirmed by the low rate of local and/or regional recurrence (in 4 patients, 10%), half of which were cured after timely salvage surgery. Only two patients died of the disease and the third is still alive with the lung metastases 5.8 years after the treatment. In addition, the 2- and especially the 5-year LFS (and leDFS) of 87% and 75%, respectively, in our patients compares favorably with the results of the RTOG 91-11 study, despite a significant proportion of prognostically less favorable hypopharyngeal primary tumors in our group [
3,
4]. Exactly the same applies to the comparison with other prospective randomized trials and non-randomized cohort studies that included patients with both laryngeal and hypopharyngeal cancer [
9,
17,
24,
25]. The encouraging results suggest the efficacy of the approach tested and emphasize the importance of treating such patients in an experienced multidisciplinary team that provides a balanced assessment of each case, therapeutic expertise, follow-up and timely surgical salvage [
26]. In addition, the number of systemic progressions was low (2/39) and most deaths were due to smoking-related metachronous cancers (5/9 deaths). The latter reflects the high proportion of smokers (97 %) among our patients, 62 % of whom were active smokers at the time of diagnosis and half of whom did not stop smoking after treatment.
The majority of our patients received only two cycles of ICT and the cumulative doses of chemotherapeutic agents were significantly lower than in the pivotal TAX 323 and TAX 324 trials, in which TPF was compared with the PF ICT regimen [275,
28]. Still, toxicity was significant: the median interval between the start of the last ICT cycle and the start of cCRT was 26 days, and grade 3 and 4 adverse events were observed in 21 % and 13 % of patients, respectively, so that a second cycle could not be performed in two (5 %) patients. Furthermore, it is questionable whether the third ICT cycle contributed to the efficacy of the treatment, as the disease recurred in 2 of these 7 patients. On the other hand, all three responders who had received only one ICT cycle were free of malignant disease at the last follow-up. Considering the aggressiveness of subsequent cCRT, it seems reasonable to limit ICT to the smallest number of cycles that is still safe. In fact, the proportion of patients with cCRT-related toxicities of grade ≥3 was 57 %. Although this is within the expected range, it requires optimization of the induction phase of treatment [
12]. The role of immune checkpoint inhibitors in the induction phase of larynx preservation therapy is currently the subject of intensive clinical research [
29].
Despite the significant proportion (54 %) of prognostically unfavorable hypopharyngeal cancers in our group, the calculated DFS and OS rates were high, suggesting that treatment intensification by combining ICT and cCRT is important in larynx preservation setting. This has also been noted by other authors [
10,
11]. However, the results of randomized trials investigating the role of the addition of TPF ICT to cCRT in head and neck cancers contradict our observation of a positive contribution of ICT to treatment efficacy [
8]. The reason for this could be the difference in tumor burden, which is generally lower in the larynx preservation setting than in the reported studies.
Our study has limitations primarily related to the small sample size, the retrospective design, and the inclusion of laryngeal and hypopharyngeal cancers, which differ significantly in terms of prognosis [
30]. However, in our group, no difference in survival was found between patients with primary laryngeal and hypopharyngeal tumors, and the same was found in the TREMPLIN trial [
31]. Furthermore, the study was not designed to answer the question of the optimal number of ICT cycles required for reliable identification of chemo-/radiosensitive tumors, nor to test the contribution of ICT to the efficacy of a particular treatment scenario. However, given the toxicity of TPF ICT and the lack of a clear benefit of a third ICT cycle for prognosis, two applications of ICT appear to be sufficient. Whether a single “chemoselector” cycle of TPF ICT followed by platinum-based cCRT would lead to an equally favorable outcome remains to be determined.
5. Conclusions
We conclude that clinical assessment of tumor response to one cycle of TPF ICT serves as a valid and easy-to-use predictor of tumor sensitivity to platinum-based cCRT. The tested combination of two cycles of ICT and cCRT resulted in a favorable larynx preservation rate and survival outcomes, with no difference between laryngeal and hypopharyngeal primary tumors. However, due to the toxicity of combination therapy, further de-intensification of the ICT component to a single chemoselector cycle with possible optimization of the drug combination appears to be justified.
Author Contributions
Conceptualization, Primož Strojan and Cvetka Grašič Kuhar; Data curation, Primož Strojan; Formal analysis, Primož Strojan and Gaber Plavc; Funding acquisition, Primož Strojan; Investigation, Primož Strojan, Gaber Plavc and Marko Kokalj; Methodology, Primož Strojan; Project administration, Primož Strojan; Supervision, Primož Strojan; Validation, Primož Strojan, Gaber Plavc, Robert Šifrer, Simona Jereb, Boštjan Lanišnik, Marko Kokalj, Ales Grošelj and Cvetka Grašič Kuhar; Writing – original draft, Primož Strojan and Gaber Plavc; Writing – review & editing, Primož Strojan, Gaber Plavc, Robert Šifrer, Simona Jereb, Boštjan Lanišnik, Marko Kokalj, Ales Grošelj and Cvetka Grašič Kuhar.Funding: This research was funded by Slovenian Research and Innovation (ARIS), grant number P3-0307.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and the study protocol was approved by the Committee for Medical Ethics and the Protocol Review Board of the Institute of Oncology Ljubljana (ERIDNPVO-0036/2020, 19.11.2020).
Informed Consent Statement
For retrospective studies, a written patient consent is deemed unnecessary according to national and institutional regulations.
Data Availability Statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
This research was funded by Slovenian Research and Innovation (ARIS), grant number P3-0307.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Singer, S.; Danker, H.; Guntinas-Lichius, O.; Oeken, J.; Pabst, F.; Schock, J.; Vogel, H.J.; Meister, E.F.; Wulke, C.; Dietz, A. Quality of life before and after total laryngectomy: results of a multicenter prospective cohort study. Head Neck 2014, 36, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Argiris, A.; Lefebvre, J.L. Laryngeal preservation strategies in locally advanced laryngeal and hypopharyngeal cancers. Front Oncol. 2019, 9, 419. [Google Scholar] [CrossRef] [PubMed]
- Forastiere, A.A.; Goepfert, H.; Maor, M.; Pajak, T.F.; Weber, R.; Morrison, W.; Glisson, B.; Trotti, A.; Ridge, J.A.; Chao, C.; et al. Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med. 2003, 349, 2091–2098. [Google Scholar] [CrossRef] [PubMed]
- Forastiere, A.A.; Zhang, Q.; Weber, R.S.; Maor, M.H.; Goepfert, H.; Pajak, T.F.; Morrison, W.; Glisson, B.; Trotti, A.; Ridge, J.A.; et al. Long-term results of RTOG 91-11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J Clin Oncol. 2013, 31, 845–852. [Google Scholar] [CrossRef]
- Strojan, P.; Haigentz, M. Jr.; Bradford, C.R.; Wolf, G.T.; Hartl, D.M.; Langendijk, J.A.; Rinaldo, A.; Eisbruch, A.; Mendenhall, W.M.; Forastiere, A.A.; et al. Chemoradiotherapy vs. total laryngectomy for primary treatment of advanced laryngeal squamous cell carcinoma. Oral Oncol. 2013, 49, 283–286. [Google Scholar] [CrossRef]
- Kiong, K.L.; de Souza, N.N.; Sultana, R.; Iyer, N.G. Meta-analysis of induction chemotherapy as a selection marker for chemoradiation in the head and neck. Laryngoscope 2018, 128, 1594–1601. [Google Scholar] [CrossRef]
- Forastiere, A.A.; Ismaila, N.; Lewin, J.S.; Nathan, C.A.; Adelstein, D.J.; Eisbruch, A.; Fass, G.; Fisher, S.G.; Laurie, S.A.; Le, Q.T.; et al. Use of larynx-preservation strategies in the treatment of laryngeal cancer: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2018, 36, 1143–1169. [Google Scholar] [CrossRef]
- Vidal, L.; Ben Aharon, I.; Limon, D.; Cohen, E.; Popovtzer, A. Role of induction chemotherapy prior to chemoradiation in head and neck squamous cell cancer - systematic review and meta-analysis. Cancer J. 2017, 23, 79–83. [Google Scholar] [CrossRef]
- Lefebvre, J.L.; Pointreau, Y.; Rolland, F.; Alfonsi, M.; Baudoux, A.; Sire, C.; de Raucourt, D.; Malard, O.; Degardin, M.; Tuchais, C.; et al. Induction chemotherapy followed by either chemoradiotherapy or bioradiotherapy for larynx preservation: the TREMPLIN randomized phase II study. J Clin Oncol. 2013, 31, 853–859. [Google Scholar] [CrossRef]
- Urba, S.; Wolf, G.; Eisbruch, A.; Worden, F.; Lee, J.; Bradford, C.; Teknos, T.; Chepeha, D.; Prince, M.; Hogikyan, N.; et al. Single-cycle induction chemotherapy selects patients with advanced laryngeal cancer for combined chemoradiation: a new treatment paradigm. J Clin Oncol. 2006, 24, 593–598. [Google Scholar] [CrossRef]
- Wolf, G.T.; Bellile, E.; Eisbruch, A.; Urba, S.; Bradford, C.R.; Peterson, L.; Prince, M.E.; Teknos, T.N.; Chepeha, D.B.; Hogikyan, N.D.; et al. Survival rates using individualized bioselection treatment methods in patients with advanced laryngeal cancer. JAMA Otolaryngol Head Neck Surg. 2017, 143, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Strojan, P.; Grašič Kuhar, C.; Žumer, B.; Kadivec, M.; Karner, K.; Fajdiga, I.; Jančar, B.; Gale, N.; Poljak, M.; Kocjan, B.J.; et al. TPF induction chemotherapy and concomitant irradiation with cisplatin and cetuximab in unresectable squamous cell carcinoma of the head and neck. Head Neck 2014, 36, 1555–1561. [Google Scholar] [CrossRef] [PubMed]
- Grégoire, V.; Evans, M.; Le, Q.T.; Bourhis, J.; Budach, V.; Chen, A.; Eisbruch, A.; Feng, M.; Giralt, J.; Gupta, T.; et al. Delineation of the primary tumour Clinical Target Volumes (CTV-P) in laryngeal, hypopharyngeal, oropharyngeal and oral cavity squamous cell carcinoma: AIRO, CACA, DAHANCA, EORTC, GEORCC, GORTEC, HKNPCSG, HNCIG, IAG-KHT, LPRHHT, NCIC CTG, NCRI, NRG Oncology, PHNS, SBRT, SOMERA, SRO, SSHNO, TROG consensus guidelines. Radiother Oncol. 2018, 126, 3–24. [Google Scholar]
- Grégoire, V.; Ang, K.; Budach, W.; Grau, C.; Hamoir, M.; Langendijk, J.A.; Lee, A.; Le, Q.T.; Maingon, P.; Nutting, C.; et al. Delineation of the neck node levels for head and neck tumors: a 2013 update. DAHANCA, EORTC, HKNPCSG, NCIC CTG, NCRI, RTOG, TROG consensus guidelines. Radiother Oncol. 2015, 117, 83-90.
- Beitler, J.J.; Ridge, J.A.; Vermorken, J.B.; Bradford, C.R.; Strojan, P.; Saba, N.F.; Suárez, C.; Rodrigo, J.P.; Rinaldo, A.; Chen, A.Y.; et al. T4 laryngeal cancer with good function: Should we be reluctant to treat without surgery? Int J Radiat Oncol Biol Phys. 2018, 102, 1400–1403. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, J.L.; Ang, K.K.; Larynx Preservation Consensus Panel. Larynx preservation clinical trial design: key issues and recommendations--a consensus panel summary. Head Neck 2009, 31, 429-441.
- Wichmann, G.; Krüger, A.; Boehm, A.; Kolb, M.; Hofer, M.; Fischer, M.; Müller, S.; Purz, S.; Stumpp, P.; Sabri, O.; et al. Induction chemotherapy followed by radiotherapy for larynx preservation in advanced laryngeal and hypopharyngeal cancer: Outcome prediction after one cycle induction chemotherapy by a score based on clinical evaluation, computed tomography-based volumetry and 18F-FDG-PET/CT. Eur J Cancer 2017, 72, 144–155. [Google Scholar] [PubMed]
- Popovtzer, A.; Burnstein, H.; Stemmer, S.; Limon, D.; Hili, O.; Bachar, G.; Sopov, V.; Feinmesser, R.; Groshar, D.; Shvero, J. Phase II organ-preservation trial: Concurrent cisplatin and radiotherapy for advanced laryngeal cancer after response to docetaxel, cisplatin, and 5-fluorouracil-based induction chemotherapy. Head Neck 2017, 39, 227–233. [Google Scholar] [CrossRef]
- Garcia GCTE, Gorphe P, Hartl D, Ammari S, Even C, Tao Y, Balleyguier C, Varoquaux A, Bidault F. Computed tomography evaluation after induction chemotherapy for T3 laryngeal cancer: Does response correlate with vocal cord mobility? Oral Oncol 2019, 90, 13-16.
- Ferrari M, Mularoni F, Taboni S, Crosetti E, Pessina C, Carobbio ALC, Montalto N, Marchi F, Vural A, Paderno A, et al. How reliable is assessment of true vocal cord-arytenoid unit mobility in patients affected by laryngeal cancer? a multi-institutional study on 366 patients from the ARYFIX collaborative group. Oral Oncol 2024, 152: 106744.
- de Bree, R.; Wolf, G.T.; de Keizer, B.; Nixon, I.J.; Hartl, D.M.; Forastiere, A.A.; Haigentz, M. Jr.; Rinaldo, A.; Rodrigo, J.P.; Saba, N.F. Response assessment after induction chemotherapy for head and neck squamous cell carcinoma: From physical examination to modern imaging techniques and beyond. Head Neck 2017, 39, 2329–2349. [Google Scholar] [CrossRef]
- Xu, J.; Yang, Y.; Zhong, Q.; Hou, L.; Ma, H.; Zhang, Y.; Feng, L.; He, S.; Lian, M.; Fang, J. A study of peripheral blood parameters to predict response to induction chemotherapy and overall survival in advanced laryngeal squamous cell carcinoma. Curr Oncol. 2022, 29, 6472–6484. [Google Scholar] [CrossRef]
- Tan, C.; Wang, L.; Yang, Y.; He, S.; Chen, G.G.; Chan, J.Y.; Tong, M.C.; van Hasselt, C.A.; Xu, W., Feng, L.; et al. Construction of a novel six-gene signature to predict tumour response to induction chemotherapy and overall survival in locoregionally advanced laryngeal and hypopharyngeal carcinoma. Genes Dis. 2023, 11, 101022.
- Pointreau, Y.; Garaud, P.; Chapet, S.; Sire, C.; Tuchais, C.; Tortochaux, J.; Faivre, S.; Guerrif, S.; Alfonsi, M.; Calais, G. Randomized trial of induction chemotherapy with cisplatin and 5-fluorouracil with or without docetaxel for larynx preservation. J Natl Cancer Inst. 2009, 101, 498–506. [Google Scholar] [CrossRef]
- Dietz A, Wichmann G, Kuhnt T, Pfreundner L, Hagen R, Scheich M, Kölbl O, Hautmann MG, Strutz J, Schreiber F, et al. Induction chemotherapy (IC) followed by radiotherapy (RT) versus cetuximab plus IC and RT in advanced laryngeal/hypopharyngeal cancer resectable only by total laryngectomy-final results of the larynx organ preservation trial DeLOS-II. Ann Oncol 2018, 29, 2105-2114.
- Sanabria, A.; Chaves, A.L.F.; Kowalski, L.P.; Wolf, G.T.; Saba, N.F.; Forastiere, A.A.; Beitler, J.J.; Nibu, K.I.; Bradford, C.R.; Suárez, C.; et al. Organ preservation with chemoradiation in advanced laryngeal cancer: The problem of generalizing results from randomized controlled trials. Auris Nasus Larynx 2017, 44, 18–25. [Google Scholar] [CrossRef]
- Vermorken, J.B.; Remenar, E.; van Herpen, C.; Gorlia, T.; Mesia, R.; Degardin, M.; Stewart, J.S.; Jelic, S.; Betka, J.; Preiss, J.H.; et al; EORTC 24971/TAX 323 Study Group. Cisplatin, fluorouracil, and docetaxel in unresectable head and neck cancer. N Engl J Med. 2007, 357, 1695-704.
- Posner, M.R.; Hershock, D.M.; Blajman, C.R.; Mickiewicz, E.; Winquist, E.; Gorbounova, V.; Tjulandin, S.; Shin, D.M.; Cullen, K.; Ervin, T.J.; et al; TAX 324 Study Group. Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. N Engl J Med. 2007, 357, 1705-1715.
- Peng, J.; Luo, G.; Yu, Y.; Ning, K.; Liu, X. Retrospective assessment of neoadjuvant camrelizumab combined with induction chemotherapy: efficacy in laryngeal preservation for advanced hypopharyngeal and laryngeal squamous cell carcinoma. Cancer Immunol Immunother. 2024, 73, 54. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.Q.M. Head and neck cancer. N Engl J Med. 2020, 382, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Janoray, G.; Pointreau, Y.; Alfonsi, M.; Sire, C.; Geoffrois, L.; de Raucourt, D.; Bardet, E.; Calais, M.H.; Garaud, P.; Calais, G. Induction chemotherapy followed by cisplatin or cetuximab concomitant to radiotherapy for laryngeal/hypopharyngeal cancer: Long-term results of the TREMPLIN randomised GORTEC trial. Eur J Cancer. 2020, 133, 86–93. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).