Submitted:
23 September 2024
Posted:
23 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
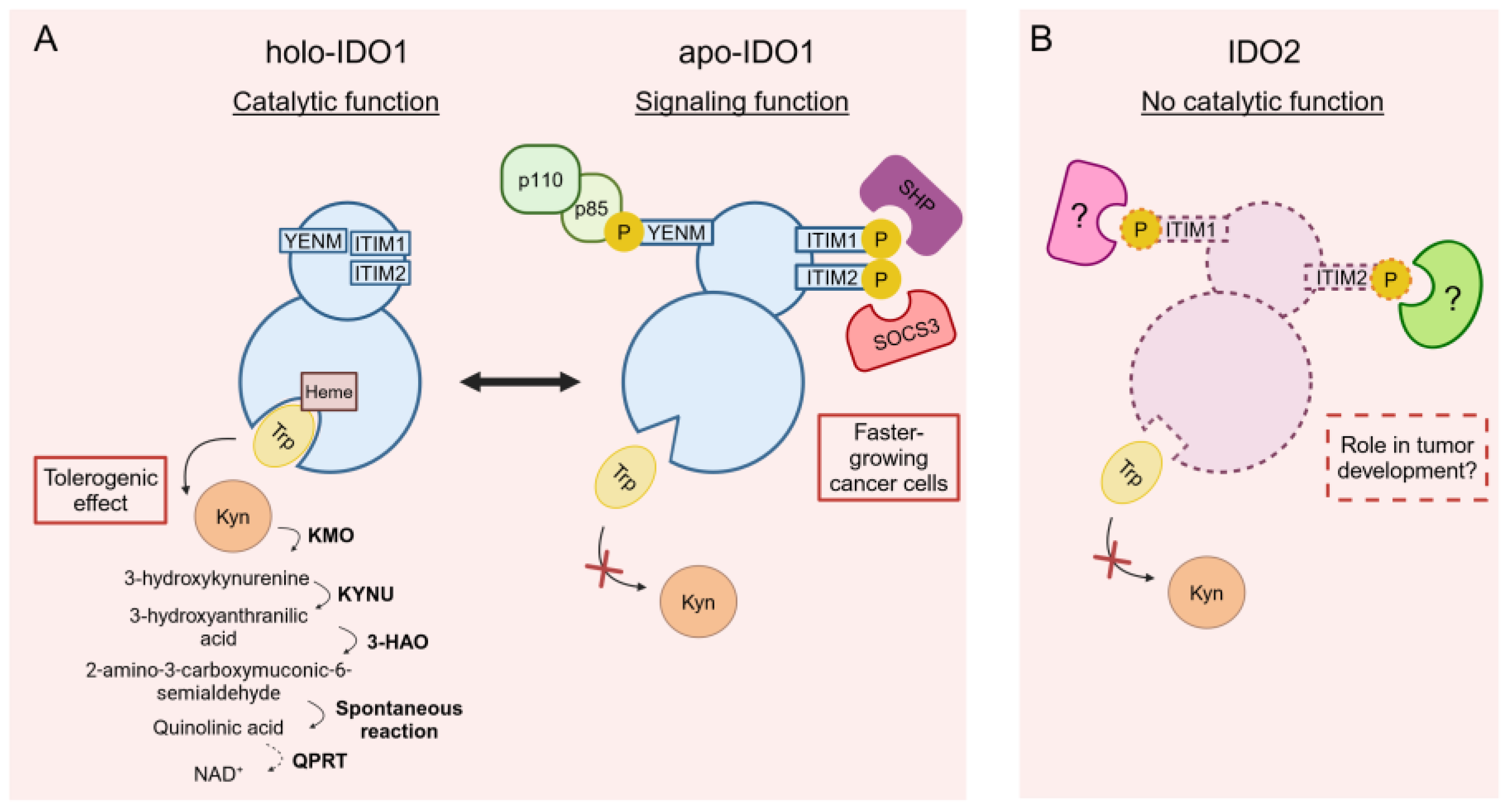
2. The First Life of IDO2: Living as an Enzyme in the IDO1's Shadow
3. Enigmatic IDO2 and Where to Find It
4. One, None and Hundred Thousand IDO2
5. The Second Life of IDO2: New Light on IDO2 Biology
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lynch, M.; Conery, J.S. The evolutionary fate and consequences of duplicate genes. Science 2000, 290, 1151–1155. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Ohta, T. On some principles governing molecular evolution. Proc Natl Acad Sci U S A 1974, 71, 2848–2852. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J. Evolution by gene duplication: an update. Trends in Ecology & Evolution 2003, 18, 292–298. [Google Scholar] [CrossRef]
- Yuasa, H.J.; Mizuno, K.; Ball, H.J. Low efficiency IDO2 enzymes are conserved in lower vertebrates, whereas higher efficiency IDO1 enzymes are dispensable. FEBS J 2015, 282, 2735–2745. [Google Scholar] [CrossRef]
- Mellor, A.L.; Lemos, H.; Huang, L. Indoleamine 2,3-Dioxygenase and Tolerance: Where Are We Now? Front Immunol 2017, 8, 1360. [Google Scholar] [CrossRef]
- Munn, D.H.; Mellor, A.L. Indoleamine 2,3-dioxygenase and tumor-induced tolerance. J Clin Invest 2007, 117, 1147–1154. [Google Scholar] [CrossRef]
- Tang, K.; Wu, Y.H.; Song, Y.; Yu, B. Indoleamine 2,3-dioxygenase 1 (IDO1) inhibitors in clinical trials for cancer immunotherapy. J Hematol Oncol 2021, 14, 68. [Google Scholar] [CrossRef]
- Peyraud, F.; Guegan, J.P.; Bodet, D.; Cousin, S.; Bessede, A.; Italiano, A. Targeting Tryptophan Catabolism in Cancer Immunotherapy Era: Challenges and Perspectives. Front Immunol 2022, 13, 807271. [Google Scholar] [CrossRef]
- Li, P.; Xu, W.; Liu, F.; Zhu, H.; Zhang, L.; Ding, Z.; Liang, H.; Song, J. The emerging roles of IDO2 in cancer and its potential as a therapeutic target. Biomed Pharmacother 2021, 137, 111295. [Google Scholar] [CrossRef] [PubMed]
- Van den Eynde, B.J.; van Baren, N.; Baurain, J.-F. Is There a Clinical Future for IDO1 Inhibitors After the Failure of Epacadostat in Melanoma? Annual Review of Cancer Biology 2020, 4, 241–256. [Google Scholar] [CrossRef]
- Yao, Y.; Liang, H.; Fang, X.; Zhang, S.; Xing, Z.; Shi, L.; Kuang, C.; Seliger, B.; Yang, Q. What is the prospect of indoleamine 2,3-dioxygenase 1 inhibition in cancer? Extrapolation from the past. J Exp Clin Cancer Res 2021, 40, 60. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.W.; Hajjar, J.; Hwu, P.; Naing, A. Targeting the indoleamine 2,3-dioxygenase pathway in cancer. J Immunother Cancer 2015, 3, 51. [Google Scholar] [CrossRef] [PubMed]
- Jusof, F.F.; Bakmiwewa, S.M.; Weiser, S.; Too, L.K.; Metz, R.; Prendergast, G.C.; Fraser, S.T.; Hunt, N.H.; Ball, H.J. Investigation of the Tissue Distribution and Physiological Roles of Indoleamine 2,3-Dioxygenase-2. Int J Tryptophan Res 2017, 10, 1178646917735098. [Google Scholar] [CrossRef] [PubMed]
- Ball, H.J.; Sanchez-Perez, A.; Weiser, S.; Austin, C.J.; Astelbauer, F.; Miu, J.; McQuillan, J.A.; Stocker, R.; Jermiin, L.S.; Hunt, N.H. Characterization of an indoleamine 2,3-dioxygenase-like protein found in humans and mice. Gene 2007, 396, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, H.J.; Takubo, M.; Takahashi, A.; Hasegawa, T.; Noma, H.; Suzuki, T. Evolution of vertebrate indoleamine 2,3-dioxygenases. J Mol Evol 2007, 65, 705–714. [Google Scholar] [CrossRef]
- Metz, R.; Duhadaway, J.B.; Kamasani, U.; Laury-Kleintop, L.; Muller, A.J.; Prendergast, G.C. Novel tryptophan catabolic enzyme IDO2 is the preferred biochemical target of the antitumor indoleamine 2,3-dioxygenase inhibitory compound D-1-methyl-tryptophan. Cancer Res 2007, 67, 7082–7087. [Google Scholar] [CrossRef] [PubMed]
- Rossini, S.; Ambrosino, S.; Volpi, C.; Belladonna, M.L.; Pallotta, M.T.; Panfili, E.; Suvieri, C.; Macchiarulo, A.; Mondanelli, G.; Orabona, C. Epacadostat stabilizes the apo-form of IDO1 and signals a pro-tumorigenic pathway in human ovarian cancer cells. Front Immunol 2024, 15, 1346686. [Google Scholar] [CrossRef]
- Theate, I.; van Baren, N.; Pilotte, L.; Moulin, P.; Larrieu, P.; Renauld, J.C.; Herve, C.; Gutierrez-Roelens, I.; Marbaix, E.; Sempoux, C.; et al. Extensive profiling of the expression of the indoleamine 2,3-dioxygenase 1 protein in normal and tumoral human tissues. Cancer Immunol Res 2015, 3, 161–172. [Google Scholar] [CrossRef]
- Meireson, A.; Devos, M.; Brochez, L. IDO Expression in Cancer: Different Compartment, Different Functionality? Front Immunol 2020, 11, 531491. [Google Scholar] [CrossRef]
- Platten, M.; Nollen, E.A.A.; Rohrig, U.F.; Fallarino, F.; Opitz, C.A. Tryptophan metabolism as a common therapeutic target in cancer, neurodegeneration and beyond. Nat Rev Drug Discov 2019, 18, 379–401. [Google Scholar] [CrossRef]
- Pallotta, M.T.; Orabona, C.; Volpi, C.; Vacca, C.; Belladonna, M.L.; Bianchi, R.; Servillo, G.; Brunacci, C.; Calvitti, M.; Bicciato, S.; et al. Indoleamine 2,3-dioxygenase is a signaling protein in long-term tolerance by dendritic cells. Nat Immunol 2011, 12, 870–878. [Google Scholar] [CrossRef] [PubMed]
- Iacono, A.; Pompa, A.; De Marchis, F.; Panfili, E.; Greco, F.A.; Coletti, A.; Orabona, C.; Volpi, C.; Belladonna, M.L.; Mondanelli, G.; et al. Class IA PI3Ks regulate subcellular and functional dynamics of IDO1. EMBO Rep 2020, 21, e49756. [Google Scholar] [CrossRef]
- Orabona, C.; Pallotta, M.T.; Volpi, C.; Fallarino, F.; Vacca, C.; Bianchi, R.; Belladonna, M.L.; Fioretti, M.C.; Grohmann, U.; Puccetti, P. SOCS3 drives proteasomal degradation of indoleamine 2,3-dioxygenase (IDO) and antagonizes IDO-dependent tolerogenesis. Proc Natl Acad Sci U S A 2008, 105, 20828–20833. [Google Scholar] [CrossRef]
- Albini, E.; Rosini, V.; Gargaro, M.; Mondanelli, G.; Belladonna, M.L.; Pallotta, M.T.; Volpi, C.; Fallarino, F.; Macchiarulo, A.; Antognelli, C.; et al. Distinct roles of immunoreceptor tyrosine-based motifs in immunosuppressive indoleamine 2,3-dioxygenase 1. J Cell Mol Med 2017, 21, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Fatokun, A.A.; Hunt, N.H.; Ball, H.J. Indoleamine 2,3-dioxygenase 2 (IDO2) and the kynurenine pathway: characteristics and potential roles in health and disease. Amino Acids 2013, 45, 1319–1329. [Google Scholar] [CrossRef] [PubMed]
- Qian, F.; Villella, J.; Wallace, P.K.; Mhawech-Fauceglia, P.; Tario, J.D., Jr.; Andrews, C.; Matsuzaki, J.; Valmori, D.; Ayyoub, M.; Frederick, P.J.; et al. Efficacy of levo-1-methyl tryptophan and dextro-1-methyl tryptophan in reversing indoleamine-2,3-dioxygenase-mediated arrest of T-cell proliferation in human epithelial ovarian cancer. Cancer Res 2009, 69, 5498–5504. [Google Scholar] [CrossRef]
- Yuasa, H.J.; Ball, H.J.; Austin, C.J.; Hunt, N.H. 1-L-methyltryptophan is a more effective inhibitor of vertebrate IDO2 enzymes than 1-D-methyltryptophan. Comp Biochem Physiol B Biochem Mol Biol 2010, 157, 10–15. [Google Scholar] [CrossRef]
- Pantouris, G.; Serys, M.; Yuasa, H.J.; Ball, H.J.; Mowat, C.G. Human indoleamine 2,3-dioxygenase-2 has substrate specificity and inhibition characteristics distinct from those of indoleamine 2,3-dioxygenase-1. Amino Acids 2014, 46, 2155–2163. [Google Scholar] [CrossRef]
- Li, J.; Li, Y.; Yang, D.; Hu, N.; Guo, Z.; Kuang, C.; Yang, Q. Establishment of a human indoleamine 2, 3-dioxygenase 2 (hIDO2) bioassay system and discovery of tryptanthrin derivatives as potent hIDO2 inhibitors. Eur J Med Chem 2016, 123, 171–179. [Google Scholar] [CrossRef]
- Prendergast, G.C.; Metz, R.; Muller, A.J.; Merlo, L.M.; Mandik-Nayak, L. IDO2 in Immunomodulation and Autoimmune Disease. Front Immunol 2014, 5, 585. [Google Scholar] [CrossRef]
- Lob, S.; Konigsrainer, A.; Schafer, R.; Rammensee, H.G.; Opelz, G.; Terness, P. Levo- but not dextro-1-methyl tryptophan abrogates the IDO activity of human dendritic cells. Blood 2008, 111, 2152–2154. [Google Scholar] [CrossRef] [PubMed]
- Lob, S.; Konigsrainer, A.; Zieker, D.; Brucher, B.L.; Rammensee, H.G.; Opelz, G.; Terness, P. IDO1 and IDO2 are expressed in human tumors: levo- but not dextro-1-methyl tryptophan inhibits tryptophan catabolism. Cancer Immunol Immunother 2009, 58, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Austin, C.J.; Mailu, B.M.; Maghzal, G.J.; Sanchez-Perez, A.; Rahlfs, S.; Zocher, K.; Yuasa, H.J.; Arthur, J.W.; Becker, K.; Stocker, R.; et al. Biochemical characteristics and inhibitor selectivity of mouse indoleamine 2,3-dioxygenase-2. Amino Acids 2010, 39, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, H.; Oda, S.; Otsuki, T.; Hino, T.; Yoshida, T.; Shiro, Y. Crystal structure of human indoleamine 2,3-dioxygenase: catalytic mechanism of O2 incorporation by a heme-containing dioxygenase. Proc Natl Acad Sci U S A 2006, 103, 2611–2616. [Google Scholar] [CrossRef]
- Yuasa, H.J.; Ball, H.J. Indoleamine 2,3-dioxygenases with very low catalytic activity are well conserved across kingdoms: IDOs of Basidiomycota. Fungal Genet Biol 2013, 56, 98–106. [Google Scholar] [CrossRef]
- Meininger, D.; Zalameda, L.; Liu, Y.; Stepan, L.P.; Borges, L.; McCarter, J.D.; Sutherland, C.L. Purification and kinetic characterization of human indoleamine 2,3-dioxygenases 1 and 2 (IDO1 and IDO2) and discovery of selective IDO1 inhibitors. Biochim Biophys Acta 2011, 1814, 1947–1954. [Google Scholar] [CrossRef]
- Kudo, Y.; Koh, I.; Sugimoto, J. Localization of Indoleamine 2,3-Dioxygenase-1 and Indoleamine 2,3-Dioxygenase-2 at the Human Maternal-Fetal Interface. Int J Tryptophan Res 2020, 13, 1178646920984163. [Google Scholar] [CrossRef]
- Broekhuizen, M.; Danser, A.H.J.; Reiss, I.K.M.; Merkus, D. The Function of the Kynurenine Pathway in the Placenta: A Novel Pharmacotherapeutic Target? Int J Environ Res Public Health 2021, 18. [Google Scholar] [CrossRef]
- Mandarano, M.; Bellezza, G.; Belladonna, M.L.; Vannucci, J.; Gili, A.; Ferri, I.; Lupi, C.; Ludovini, V.; Falabella, G.; Metro, G.; et al. Indoleamine 2,3-Dioxygenase 2 Immunohistochemical Expression in Resected Human Non-small Cell Lung Cancer: A Potential New Prognostic Tool. Front Immunol 2020, 11, 839. [Google Scholar] [CrossRef]
- Adams, S.; Teo, C.; McDonald, K.L.; Zinger, A.; Bustamante, S.; Lim, C.K.; Sundaram, G.; Braidy, N.; Brew, B.J.; Guillemin, G.J. Involvement of the kynurenine pathway in human glioma pathophysiology. PLoS One 2014, 9, e112945. [Google Scholar] [CrossRef]
- Gu, P.; Ling, B.; Ma, W.; Zhang, J.; Zhang, W.; Zeng, Y.; Liu, Y.; Chi, J.; Ruan, X.; Zheng, X.; et al. Indoleamine 2,3-dioxygenase 2 immunohistochemical expression in medullary thyroid carcinoma: implications in prognosis and immunomodulatory effects. BMC Cancer 2022, 22, 1116. [Google Scholar] [CrossRef] [PubMed]
- Panda, A.; Ganesan, S. Genomic and Immunologic Correlates of Indoleamine 2,3-Dioxygenase Pathway Expression in Cancer. Front Genet 2021, 12, 706435. [Google Scholar] [CrossRef]
- Miyashita, H.; Bevins, N.J.; Thangathurai, K.; Lee, S.; Pabla, S.; Nesline, M.K.; Glenn, S.T.; Conroy, J.M.; DePietro, P.; Rubin, E.; et al. The transcriptomic expression pattern of immune checkpoints shows heterogeneity between and within cancer types. Am J Cancer Res 2024, 14, 2240–2252. [Google Scholar] [CrossRef] [PubMed]
- Suvieri, C.; De Marchis, F.; Mandarano, M.; Ambrosino, S.; Rossini, S.; Mondanelli, G.; Gargaro, M.; Panfili, E.; Orabona, C.; Pallotta, M.T.; et al. Membrane Localization and Phosphorylation of Indoleamine 2,3-Dioxygenase 2 (IDO2) in A549 Human Lung Adenocarcinoma Cells: First Steps in Exploring Its Signaling Function. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Kado, S.Y.; Bein, K.; Castaneda, A.R.; Pouraryan, A.A.; Garrity, N.; Ishihara, Y.; Rossi, A.; Haarmann-Stemmann, T.; Sweeney, C.A.; Vogel, C.F.A. Regulation of IDO2 by the Aryl Hydrocarbon Receptor (AhR) in Breast Cancer. Cells 2023, 12. [Google Scholar] [CrossRef]
- Mo, B.; Zhao, X.; Wang, Y.; Jiang, X.; Liu, D.; Cai, H. Pan-cancer analysis, providing a reliable basis for IDO2 as a prognostic biomarker and target for immunotherapy. Oncologie 2023, 25, 17–35. [Google Scholar] [CrossRef]
- Guo, D.; Wang, Y.; Wu, X.; Gao, Y.; Wang, A.; Zhang, Z.; Zhao, K.; Wang, X.; Liu, M.; Zhang, Y.; et al. Expression of Tryptophan Metabolism Enzymes in Patients with Diffuse Large B-cell Lymphoma and NK/T-cell Lymphoma. Cancer Med 2023, 12, 12139–12148. [Google Scholar] [CrossRef]
- Zhang, S.; Gao, Y.; Wang, P.; Wang, S.; Wang, Y.; Li, M.; Wang, A.; Zhao, K.; Zhang, Z.; Sun, J.; et al. Tryptophan metabolism enzymes are potential targets in ovarian clear cell carcinoma. Cancer Med 2023, 12, 21996–22005. [Google Scholar] [CrossRef]
- Jacquerie, A.; Hoeben, A.; Eekers, D.B.P.; Postma, A.A.; Vanmechelen, M.; de Smet, F.; Ackermans, L.; Anten, M.; Severens, K.; Zur Hausen, A.; et al. Prognostic relevance of high expression of kynurenine pathway markers in glioblastoma. Sci Rep 2024, 14, 14975. [Google Scholar] [CrossRef]
- Merlo, L.M.F.; Peng, W.; Mandik-Nayak, L. Impact of IDO1 and IDO2 on the B Cell Immune Response. Front Immunol 2022, 13, 886225. [Google Scholar] [CrossRef]
- Mondanelli, G.; Mandarano, M.; Belladonna, M.L.; Suvieri, C.; Pelliccia, C.; Bellezza, G.; Sidoni, A.; Carvalho, A.; Grohmann, U.; Volpi, C. Current Challenges for IDO2 as Target in Cancer Immunotherapy. Front Immunol 2021, 12, 679953. [Google Scholar] [CrossRef] [PubMed]
- Merlo, L.M.; Mandik-Nayak, L. IDO2: A Pathogenic Mediator of Inflammatory Autoimmunity. Clin Med Insights Pathol 2016, 9, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Yamasuge, W.; Yamamoto, Y.; Fujigaki, H.; Hoshi, M.; Nakamoto, K.; Kunisawa, K.; Mouri, A.; Nabeshima, T.; Saito, K. Indoleamine 2,3-dioxygenase 2 depletion suppresses tumor growth in a mouse model of Lewis lung carcinoma. Cancer Sci 2019, 110, 3061–3067. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Zheng, X.; Zhang, X.; Wang, H.; Li, Q.; Yuan, K.; Zhou, N.; Yu, Y.; Song, N.; et al. Gene silencing of indoleamine 2,3-dioxygenase 2 in melanoma cells induces apoptosis through the suppression of NAD+ and inhibits in vivo tumor growth. Oncotarget 2016, 7, 32329–32340. [Google Scholar] [CrossRef]
- Sorensen, R.B.; Kollgaard, T.; Andersen, R.S.; van den Berg, J.H.; Svane, I.M.; Straten, P.; Andersen, M.H. Spontaneous cytotoxic T-Cell reactivity against indoleamine 2,3-dioxygenase-2. Cancer Res 2011, 71, 2038–2044. [Google Scholar] [CrossRef]
- Merlo, L.M.F.; DuHadaway, J.B.; Montgomery, J.D.; Peng, W.D.; Murray, P.J.; Prendergast, G.C.; Caton, A.J.; Muller, A.J.; Mandik-Nayak, L. Differential Roles of IDO1 and IDO2 in T and B Cell Inflammatory Immune Responses. Front Immunol 2020, 11, 1861. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xu, P.; Liu, H.; Fang, C.; Guo, H.; Chen, X.; Tan, M.; Zhang, Y.; Min, W. Silencing IDO2 in dendritic cells: A novel strategy to strengthen cancer immunotherapy in a murine lung cancer model. Int J Oncol 2020, 57, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Trabanelli, S.; Ocadlikova, D.; Ciciarello, M.; Salvestrini, V.; Lecciso, M.; Jandus, C.; Metz, R.; Evangelisti, C.; Laury-Kleintop, L.; Romero, P.; et al. The SOCS3-independent expression of IDO2 supports the homeostatic generation of T regulatory cells by human dendritic cells. J Immunol 2014, 192, 1231–1240. [Google Scholar] [CrossRef]
- Lee, Y.K.; Lee, H.B.; Shin, D.M.; Kang, M.J.; Yi, E.C.; Noh, S.; Lee, J.; Lee, C.; Min, C.K.; Choi, E.Y. Heme-binding-mediated negative regulation of the tryptophan metabolic enzyme indoleamine 2,3-dioxygenase 1 (IDO1) by IDO2. Exp Mol Med 2014, 46, e121. [Google Scholar] [CrossRef]
- Pallotta, M.T.; Rossini, S.; Suvieri, C.; Coletti, A.; Orabona, C.; Macchiarulo, A.; Volpi, C.; Grohmann, U. Indoleamine 2,3-dioxygenase 1 (IDO1): an up-to-date overview of an eclectic immunoregulatory enzyme. FEBS J 2022, 289, 6099–6118. [Google Scholar] [CrossRef]
- Konan, K.V.; Taylor, M.W. Importance of the two interferon-stimulated response element (ISRE) sequences in the regulation of the human indoleamine 2,3-dioxygenase gene. J Biol Chem 1996, 271, 19140–19145. [Google Scholar] [CrossRef] [PubMed]
- Croitoru-Lamoury, J.; Lamoury, F.M.; Caristo, M.; Suzuki, K.; Walker, D.; Takikawa, O.; Taylor, R.; Brew, B.J. Interferon-gamma regulates the proliferation and differentiation of mesenchymal stem cells via activation of indoleamine 2,3 dioxygenase (IDO). PLoS One 2011, 6, e14698. [Google Scholar] [CrossRef] [PubMed]
- Witkiewicz, A.K.; Costantino, C.L.; Metz, R.; Muller, A.J.; Prendergast, G.C.; Yeo, C.J.; Brody, J.R. Genotyping and expression analysis of IDO2 in human pancreatic cancer: a novel, active target. J Am Coll Surg 2009, 208, 781–787; discussion 787–789. [Google Scholar] [CrossRef] [PubMed]
- Vogel, C.F.; Goth, S.R.; Dong, B.; Pessah, I.N.; Matsumura, F. Aryl hydrocarbon receptor signaling mediates expression of indoleamine 2,3-dioxygenase. Biochem Biophys Res Commun 2008, 375, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Lo, B.K.; Jalili, R.B.; Zloty, D.; Ghahary, A.; Cowan, B.; Dutz, J.P.; Carr, N.; Shapiro, J.; McElwee, K.J. CXCR3 ligands promote expression of functional indoleamine 2,3-dioxygenase in basal cell carcinoma keratinocytes. Br J Dermatol 2011, 165, 1030–1036. [Google Scholar] [CrossRef]
- Simones, T.; Shepherd, D.M. Consequences of AhR activation in steady-state dendritic cells. Toxicol Sci 2011, 119, 293–307. [Google Scholar] [CrossRef]
- Bankoti, J.; Rase, B.; Simones, T.; Shepherd, D.M. Functional and phenotypic effects of AhR activation in inflammatory dendritic cells. Toxicol Appl Pharmacol 2010, 246, 18–28. [Google Scholar] [CrossRef]
- Nevler, A.; Muller, A.J.; Sutanto-Ward, E.; DuHadaway, J.B.; Nagatomo, K.; Londin, E.; O'Hayer, K.; Cozzitorto, J.A.; Lavu, H.; Yeo, T.P.; et al. Host IDO2 Gene Status Influences Tumor Progression and Radiotherapy Response in KRAS-Driven Sporadic Pancreatic Cancers. Clin Cancer Res 2019, 25, 724–734. [Google Scholar] [CrossRef]
- Kollgaard, T.; Klausen, T.W.; Idorn, M.; Holmgaard, R.B.; Straten, P.T.; Andersen, M.H. Association of a functional Indoleamine 2,3-dioxygenase 2 genotype with specific immune responses. Oncoimmunology 2012, 1, 441–447. [Google Scholar] [CrossRef]
- Eldredge, H.B.; Denittis, A.; Duhadaway, J.B.; Chernick, M.; Metz, R.; Prendergast, G.C. Concurrent Whole Brain Radiotherapy and Short-Course Chloroquine in Patients with Brain Metastases: A Pilot Trial. J Radiat Oncol 2013, 2. [Google Scholar] [CrossRef]
- Negrotto, L.; Correale, J. Amino Acid Catabolism in Multiple Sclerosis Affects Immune Homeostasis. J Immunol 2017, 198, 1900–1909. [Google Scholar] [CrossRef] [PubMed]
- Agliardi, C.; Guerini, F.R.; Zanzottera, M.; Rovaris, M.; Caputo, D.; Clerici, M. Indoleamine-2,3-dioxygenase(IDO)2 polymorphisms are not associated with multiple sclerosis in Italians. J Neurol Sci 2017, 377, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Kanuri, N.; Zhang, Y.; Sayuk, G.S.; Li, E.; Ciorba, M.A. IDO1 and IDO2 non-synonymous gene variants: correlation with crohn's disease risk and clinical phenotype. PLoS One 2014, 9, e115848. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Schurink, B.; Roos, E.; Nossent, E.J.; Duitman, J.W.; Vlaar, A.P.; van der Valk, P.; Vaz, F.M.; Yeh, S.R.; Geeraerts, Z.; et al. Indoleamine 2,3-dioxygenase (IDO)-1 and IDO-2 activity and severe course of COVID-19. J Pathol 2022, 256, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Napolioni, V.; Pariano, M.; Borghi, M.; Oikonomou, V.; Galosi, C.; De Luca, A.; Stincardini, C.; Vacca, C.; Renga, G.; Lucidi, V.; et al. Genetic Polymorphisms Affecting IDO1 or IDO2 Activity Differently Associate With Aspergillosis in Humans. Front Immunol 2019, 10, 890. [Google Scholar] [CrossRef]
- Cao, T.; Dai, G.; Chu, H.; Kong, C.; Duan, H.; Tian, N.; Sun, Z. Single-nucleotide polymorphisms and activities of indoleamine 2,3-dioxygenase isoforms, IDO1 and IDO2, in tuberculosis patients. Hereditas 2022, 159, 5. [Google Scholar] [CrossRef]
- Mondanelli, G.; Bianchi, R.; Pallotta, M.T.; Orabona, C.; Albini, E.; Iacono, A.; Belladonna, M.L.; Vacca, C.; Fallarino, F.; Macchiarulo, A.; et al. A Relay Pathway between Arginine and Tryptophan Metabolism Confers Immunosuppressive Properties on Dendritic Cells. Immunity 2017, 46, 233–244. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




