1. Introduction
IgE-mediated peanut allergy poses a significant and growing public health challenge in the United States and internationally. Peanut allergy has an estimated prevalence of 2.9 percent self-reported in adults between 2015-2016 in the United States [
1]. Further, the prevalence of disease is increasing over time in the United States with survey estimates from 1997 and 2015-16 demonstrating a nearly five-fold increase in peanut allergy over the same period [
2]. Among food allergies, peanut allergy is the food most frequently associated with a history of severe reactions (around 68 percent), according to a survey of US adults [
3]. Presently, peanut allergy is primarily managed through peanut avoidance and by carrying autoinjectable epinephrine to treat symptoms arising from unintended ingestion leading to an increased stress burden for patients and families [
4]. However, there are emerging treatments such as immunotherapy and biologics becoming increasingly common to manage, treat, and potentially cure peanut allergy.
Peanut allergy is associated with poorer quality of life for both patients and caregivers, due to significant financial and psychological burden on families [
2]. A cross-sectional survey conducted on a sample of 153 adult patients, 102 adolescents, and 382 caregivers of children with peanut allergies revealed key insights into treatment satisfaction and the emotional impact of peanut allergies [
2]. The study found that 24.8% of participants expressed being somewhat dissatisfied with current methods of avoiding or preventing peanut allergy reactions [
2]. The emotional well-being of adolescents was significantly impacted, with 79.4% reporting that fear of experiencing a reaction affected them at least somewhat [
2]. The survey further highlighted that peanut allergy interfered with daily life, with 31.4% of adult patients, 26.5% of adolescents, and 34.3% of caregivers indicating that it disrupted their daily activities either very much or completely [
2].
From a clinical standpoint, it is important to provide clarity regarding the diagnosis of peanut allergy to optimize health outcomes and limit health anxiety. In this paper, we will explore various diagnostic methods for peanut allergy, including both traditional approaches such as skin prick testing and serum IgE testing, as well as emerging diagnostics like component-resolved diagnostics, oral food challenges, basophil activation tests, bead-based epitope assays, genetic testing, microarray technology, novel imaging techniques, and the use of machine learning and artificial intelligence. Given that the current approach to diagnosing peanut allergy is actively evolving in the field of Allergy and Immunology, this paper seeks to describe the range of existing modalities of diagnosing peanut allergy alongside their limitations.
2. Diagnostic Methods for Peanut Allergy
When diagnosing food allergies, including peanut allergy, clinical history remains the most critical component, as it provides context for interpreting diagnostic tests like skin prick tests (SPT) or specific IgE testing [
5,
6]. These tests are prone to both false positives and false negatives, meaning that positive results do not always indicate a true allergy, and negative results may miss an allergy. Thus, diagnostic test results must always be correlated with the patient’s clinical history to ensure accurate diagnosis and management
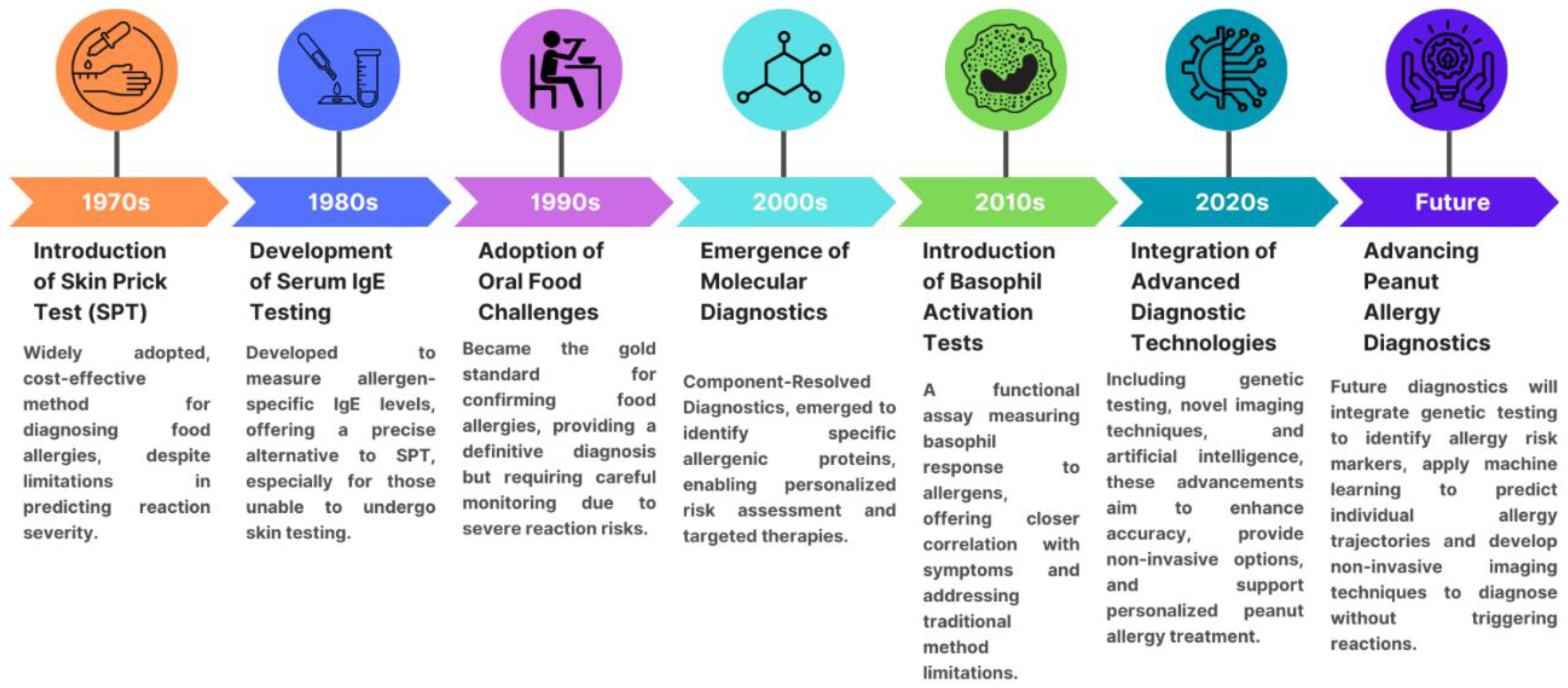
Allergy diagnostic methods can be categorized into traditional and emerging approaches. Allergen diagnostics have experienced significant advancements over the decades, starting with the introduction of skin prick testing in the 1970s, followed by the development of serum IgE testing in the 1980s, the adoption of oral food challenges in the 1990s, and the emergence of molecular diagnostics in the 2000s. In the 2010s, basophil activation tests were introduced, and more recently, the integration of advanced diagnostic technologies such as genetic testing and artificial intelligence in the 2020s has revolutionized the field, with further innovations expected in the future, see
Figure 1.
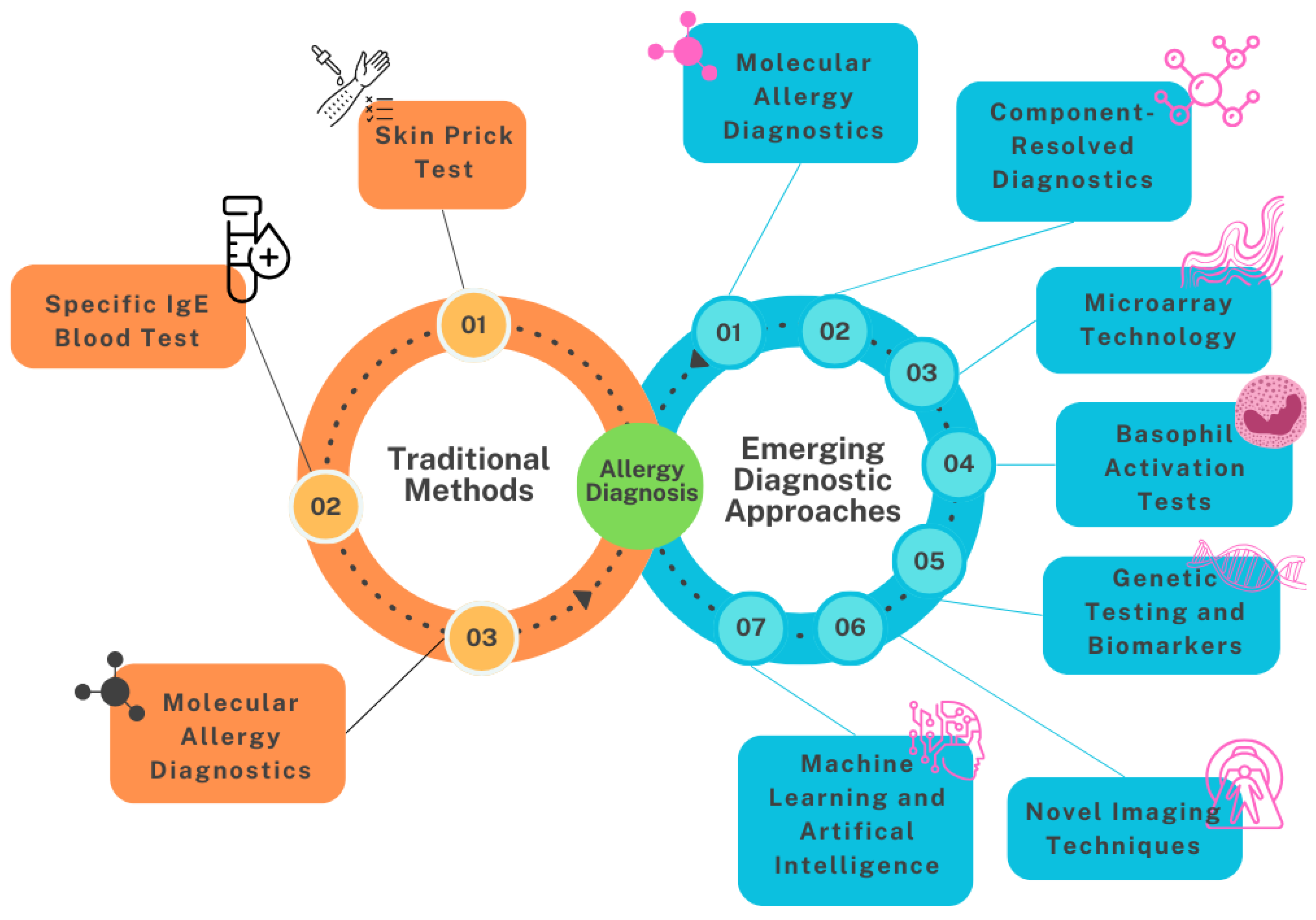
Traditional methods include the skin prick test, serum IgE blood testing with component-resolved diagnostics and oral food challenges. Emerging diagnostic approaches encompass molecular allergy diagnostics, microarray technology, basophil activation tests, genetic testing, machine learning and artificial intelligence, and novel imaging techniques, see
Figure 2.
3. Current Diagnostic Methods
3.1. Skin Prick Testing
Skin prick testing (SPT) is a commonly used initial method for diagnosing food allergies because it is sensitive, reproducible, cost-effective, safe, minimally uncomfortable, quick, and easy to perform during a routine clinic visit. It involves a small, bloodless puncture on the skin typically with a sharp plastic prick to introduce commercial allergen extracts, alongside positive (histamine) and negative (saline) controls. If the patient is sensitive to the peanut allergen, the mast cells in the skin attached to peanut IgE will degranulate, resulting in a wheal with surrounding erythematous flare [
7]. While a positive test indicates sensitization, it may not necessarily be associated with adverse reactions to that food [
8]. A positive SPT is a wheal ≥ 3 mm based on American and European recommendations, though interpretation also requires comparison to the positive and negative controls.
Many studies have been performed in different populations to determine the accuracy of peanut SPT in predicting IgE mediated peanut allergy. A wheal size of ≥ 3 mm for peanut allergy provides high sensitivity (>90%) but low specificity and positive predictive value, see
Table 1. This means the test is a great way to rule out a peanut allergy but not a great way to confirm a diagnosis without a convincing clinical history and/or other IgE testing [
8,
9,
10,
11]. However, increasing the wheal size cutoff to ≥ 8 mm improves positive predictive value (PPV) to 95% in patients over two years old, while in children under two, a wheal size of ≥ 4 mm also has a 95% PPV [
9,
12]. This adjustment in threshold values enhances the test’s reliability, making it more effective for confirming peanut allergies, particularly when combined with a relevant clinical history [
8].
Despite its usefulness, SPT has several limitations that can impact its accuracy and feasibility. Many medications, most notably antihistamines, can interfere with the rest results and should be withheld prior [
13]. This restriction can be challenging for individuals who rely on these medications for their health management. Skin conditions like atopic dermatitis or dermatographism can also interfere with test results, potentially causing false positives. Variations in the quality of allergen extracts and measurement inaccuracies can also lead to erroneous results. SPT results can be user-dependent, with variability influenced by the specific device used and the operator’s technique. Studies have shown that devices applied using the puncture method result in lower variability and more reliable measurements compared to those applied by the prick method. Moreover, SPT results tend to be more consistent when performed on the back compared to the arm, suggesting that both the device and the anatomical location play crucial roles in ensuring reproducibility in multi-operator settings [
14]. Historically, it has been suggested that recent IgE-mediated reactions might lead to falsely low results due to the consumption of IgE during the reaction. However, the findings of a prospective study indicate that false-negative SPT shortly after food-induced anaphylaxis has never been documented in cases of food allergy. In this study, all 24 subjects maintained a reactive skin test to the food allergen within 2 weeks of their anaphylactic reaction, challenging the recommendation to delay testing [
15]. Although SPT is generally safe, there are reports of anaphylaxis from SPT and caution should be taken for patients with uncontrolled asthma, reduced lung function, or a history of severe reactions to minimal allergen exposure [
13,
16].
3.2. Serum IgE Testing
Serum IgE testing, including both food-specific and food-component assays, is a widely used commonly commercially available diagnostic method for IGE-mediated peanut allergy. In component testing, specific proteins such as Ara h1-9 are analyzed, with Ara h2 being strongly associated with diagnosing peanut allergy in pediatric populations [
17,
18,
19]. Unlike SPT, serum IgE (sIgE) tests are not dependent on operator technique and allow patients to continue taking antihistamines. Results can vary however depending on laboratory technique used by different laboratories [
20]. Furthermore, patients with atopic dermatitis or other atopic conditions may have elevated total IgE and false positive specific IgE values as well. The process involves incubating the patient’s serum with a surface-fixed allergen; food-specific IgE binds to this allergen, and a fluorescently labeled anti-IgE antibody is used to detect and measure the bound IgE, indicating allergen-specific IgE levels [
12,
21].
A positive sIgE result is defined as IgE levels ≥0.35 kU/L, which provides high sensitivity (~95%) but generally low positive predictive value (PPV) and specificity for peanut-specific IgE. Increasing the cutoff to ≥14-15 kU/L improves the PPV to ≥95% [
22,
23,
24,
25]. For Ara h2, a sIgE level of 0.35 kU/L shows a sensitivity of 91%, but the PPV is low. Raising the cutoff to ≥5 kU/L increases the PPV to 96%, though it reduces sensitivity [
22]. Recommended cutoffs for Ara h2 sIgE vary widely by study, with values from 0.35 to 42.2 kU/L achieving 90%-95% PPV [
9].
Similar to SPT, serum IgE testing is most effective when combined with a detailed clinical history of IgE-mediated food allergies [
22]. However, interpreting sIgE results can be challenging since a positive result does not always confirm a true food allergy, and results can be influenced by the quality of the allergen used in the test. Additionally, sIgE testing typically requires venipuncture and takes days to weeks for results, and some insurance plans may favor SPT [
13,
23,
26].
3.3. Molecular Allergy Diagnosis
Molecular allergy diagnosis involves the use of purified allergen molecules, primarily recombinant allergens, to determine the IgE sensitization profile of allergic patients [
27,
28]. This advanced approach allows for the identification of specific allergens at the molecular level, offering greater precision compared to traditional allergy tests that typically identify whole allergen sources [
28]. By pinpointing individual allergenic components, molecular allergy diagnostics enhance the accuracy of diagnosis, improve risk assessment, and enable personalized treatment plans [
29].
Molecular peanut allergy diagnosis utilizes component-resolved diagnostics (CRD) to identify specific peanut proteins. Currently, 17 peanut (Arachis hypogaea) allergens, designated as Ara h 1 through Ara h 18, have been identified. However, Ara h 4 was determined to be an isoform of Ara h 3, according to a review by the Systematic Allergen Nomenclature in 2021 (4-). These identified peanut allergens (Ara h 1 through Ara h 18) belong to various protein families [
30]. These include cupins (Ara h 1, Ara h 3), conglutins (Ara h 2, Ara h 6, Ara h 7), profilins (Ara h 5), Bet v 1-like proteins (Ara h 8), nonspecific lipid-transfer proteins (nsLTP) (Ara h 9, Ara h 16, Ara h 17), oleosins (Ara h 10, Ara h 11, Ara h 14, Ara h 15), defensins (Ara h 12, Ara h 13), and cyclophilins (Ara h 18) [
30].
Geographic differences in peanut-specific IgE responses are likely influenced by varying pollen exposures and local dietary practices, leading to diverse clinical and immunologic phenotypes of peanut allergy in different regions [
28,
31]. In the United States, Central, and Northern Europe, Ara h 1 and Ara h 2 are recognized as major peanut allergens [
32]. A study in the United States involving 12,123 patients with reported peanut allergies found that Ara h 2 was the most frequently recognized allergen, though IgE reactivity to Ara h 1 and Ara h 3 was also common [
31]. A study involving 100 children with peanut allergies revealed that Ara h 2-specific IgE titers and specific activity are the primary factors determining the mast cell response to Ara h 2 [
19].
CRD can help distinguish between high-risk and low-risk peanut allergies by analyzing the specific IgE sensitization profile to these components [
30]. This precise identification improves risk stratification and, by identifying the exact peanut proteins a patient is sensitized, clinicians can better assess the severity and potential risk of an allergic reaction. In addition, understanding the molecular profile of a peanut allergy allows for more tailored management strategies, including dietary advice, emergency preparedness, and consideration for immunotherapy [
30,
33].
3.4. Oral Food Challenge
The double-blind placebo-controlled food challenge (DBPCFC) is considered the gold standard for diagnosing peanut allergy, offering the most definitive assessment of allergic reactions [
34]. However, it is time- and resource-intensive, not widely available, and carries potential risks, including life-threatening allergic reactions [
35]. Of the rare fatalities associated with oral food challenges (OFCs), one case was linked to peanut allergen, highlighting the potential dangers involved [
36]. Non-blinded open food challenges are more commonly used in clinical practice due to their practicality and efficiency. In these challenges, patients are given increasing amounts of peanut-containing food while being closely monitored for reactions [
18,
34]. Protocols can widely vary amongst practitioners but one example would be four doses (1/2 teaspoon, 1 teaspoon, 1/2 tablespoon, 1 tablespoon) of peanut butter given every 15 minutes with a 2 hour observation period after [
37]. 1 tablespoon of peanut butter is approximately 3 grams of protein with 1 peanut being approximately 250 mg of protein [
37].
As such, OFCs are typically used when other diagnostic tests, such as skin prick tests or specific IgE measurements, provide ambiguous results or when a clear diagnosis is essential for guiding treatment decisions [
6]. Given the risk of severe reactions, including fatal outcomes, OFCs are conducted with caution, especially considering factors like concomitant asthma, delayed epinephrine administration, and other medical conditions that could complicate the procedure. OFCs should only be conducted in facilities that are prepared and equipped to manage severe allergic reactions [
37]. It is crucial that patients undergoing OFCs are in good health and are not on medications or have conditions that could affect the challenge’s outcome [
38]. The psychosocial impact of OFCs is also significant; while a successful challenge can improve quality of life by reintroducing a common food into the diet, the stress and anxiety associated with consuming a previously "forbidden" food can be considerable [
38]. While highly effective, the logistical and safety considerations of OFCs make them a more complex and resource-intensive option compared to other diagnostic methods.
3.5. Bead-Based Epitope Assay
The bead-based epitope assay (BBEA) is a new cutting-edge tool commercially available as of 2023 in the United States that is increasingly used to diagnose peanut allergy. This assay works by coupling microbeads with specific peptide sequences, or epitopes, derived from peanut proteins. When a patient’s serum is incubated with these beads, IgE antibodies in the serum bind to the epitopes if present. By focusing on individual epitopes, Ara h 2_008 and Ara h 2_019, associated with severe allergic reactions to peanuts, this assay provides a more precise risk assessment than traditional total whole peanut IgE tests. In clinical evaluations, the peanut BBEA diagnostic test correctly identified 93% of subjects (166 patients, aged 7 to 55 years, both male and female) as compared to DBPCFC, with a sensitivity of 92% and specificity of 94%. [
39]. The test’s positive predictive value was 91%, while the negative predictive value was 95%, indicating its high reliability in diagnosing true peanut allergy cases and minimizing false negatives, [
39]. The BBEA requires only 0.1 ml of plasma or serum, making it highly practical and easily adaptable to standard clinical labs [
39]. It also offers the benefit of potentially replacing the DBPCFC in selecting appropriate patients for immunotherapy, streamlining the process of identifying candidates for treatment [
39]. These attributes make the BBEA a revolutionary tool for the personalized diagnosis and management of peanut allergy. A comparison of current diagnostic methods is presented in
Table 2.
4. Emerging Diagnostic Approaches
Emerging diagnostic approaches for peanut allergy can be divided into molecular allergy diagnostics, microarray technology, basophil activation tests, genetic testing, novel imaging techniques, and machine learning and AI, each with distinct advantages and limitations, see
Table 3.
4.1. Use of Microarrays for Allergen Components
In 2002, it was shown that microarray technology enables the assembly of large numbers of allergen molecules on chips, allowing for rapid serological testing of IgE sensitizations with minimal serum volumes [
40]. Since then, microarrayed allergens have significantly transformed allergy research and diagnosis; however, several unmet needs persist.
Microarray testing for peanut allergy is a sophisticated diagnostic tool that utilizes a panel of allergen components to detect specific IgE antibodies to multiple peanut proteins simultaneously [
41]. This approach provides a detailed profile of sensitization to individual peanut allergens, such as Ara h 1, Ara h 2, and Ara h 3. By identifying which specific allergens a patient is sensitized to, microarray testing helps determine the risk of severe allergic reactions [
42]. Additionally, it reduces the likelihood of cross-reactivity by focusing on specific components rather than whole allergen extracts, leading to a more accurate diagnosis. This method is primarily used for precise diagnosis and to develop personalized management plans for patients with peanut allergies.
The costs of IgE assays using individual allergens are about 10% higher compared to extract-based tests; microarrayed multiple allergenic molecules will produce even higher costs [
40,
43]. It is therefore conceivable that the use of molecular assays in allergology will be balanced by a general need of carefully stewarding the scarce resources of public health systems.
4.2. Basophil Activation Test (BAT)
The BAT is an in vitro diagnostic tool emerging for food, drug and venom allergy. Since its early development, which involved allergen-induced histamine release from basophils, BAT has evolved significantly [
44]. It can distinguish between IgE-reactive antigens that have minimal or no ability to induce IgE-mediated receptor aggregation and potent allergens that trigger basophil activation even at low doses [
45]. This makes BAT valuable for addressing the issue of false-positive results in allergy diagnostics, often caused by cross-reactive carbohydrate determinants (CCDs) found in plants, particularly in N-glycans [
46]. Such false positives can occur in tests like ImmunoCap assays, but BAT can overcome these by using non-glycosylated recombinant allergens or CCD inhibitors [
46].
BAT’s utility extends to predicting clinical reactions in peanut-allergic patients, with studies showing a strong correlation between negative basophil allergen threshold sensitivity and negative double-blind, placebo-controlled food challenges (DBPCFC) in children with peanut allergy [
44]. Additionally, 92% of patients with positive DBPCFC results had positive threshold sensitivity and elevated IgE levels to major peanut allergens Ara h 1, Ara h 2, and Ara h 3 [
47]. BAT has demonstrated high accuracy in diagnosing peanut allergy [
48].
Moreover, BAT is a valuable tool for assessing the efficacy of allergen immunotherapy (AIT) for peanut allergy by showing how allergen-specific IgG antibodies can block IgE-mediated allergic reactions [
49]. Flow cytometry is often used to analyze basophil activation in BAT, typically by measuring the upregulation of markers like CD203c and CD63 [
50]. The percentage of CD63+ basophils after stimulation with peanut and after stimulation with anti-IgE was found to be correlated with disease severity as well [
51].
The Basophil Activation Test (BAT) faces several limitations, including the absence of a standardized protocol across laboratories, the need for time-sensitive processing within 8-24 hours after blood collection, variability in the sensitivity of activation markers like CD63 and CD203c depending on allergens and patient groups, the issue of basophil non-responsiveness in about 10% of patients, high complexity and cost due to the requirement for specialized flow cytometry equipment and expertise, and its limited ability to account for changes in allergenicity caused by food processing, making it less practical for routine clinical use without further refinement and validation [
52].
4.3. Genetic Testing
Genetic testing can help identify individuals who are genetically predisposed to developing peanut allergies, allowing for early intervention and personalized management strategies [
53]. Variants in genes such as FLG (filaggrin), which is associated with skin barrier function, and HLA (human leukocyte antigen) genes, which are involved in immune response, have been linked to a higher risk of peanut allergies [
53,
54,
55].
In a case-control study by Brown et al., FLG loss-of-function (LOF) mutations were found to have a strong and significant link to peanut allergy in 71 English, Dutch and Irish patients who tested positive in food challenges [
53]. This association was confirmed in a Canadian study of 290 white patients with peanut allergy, further supporting the connection between these mutations and peanut allergy [
53]. Importantly, this relationship remained significant even when accounting for the presence of atopic dermatitis [
53,
56].
4.4. Novel Imaging Techniques
The gold standard double blind placebo-controlled oral food challenge (DBPCFC) presents several limitations including being time-consuming with risk of severe allergic reactions, and reliance on the subjective opinion of a third-party observer [
57]. Different imaging techniques such as thermographic imaging, surface plasmon resonance imaging (SPRi) and laser endomicroscopy are being explored to address standardization of these challenges and ameliorate the risks associated with food allergy diagnosis [
57,
58,
59]. In patients with IgE mediated peanut allergies, specific levels of IgE against the Arachis hypogogea (Ara-h2), the most potent peanut glycoprotein antigen, have been shown to be reliable biomarkers of peanut allergy severity [
58]. SPRi quantitatively detects IgE antibodies against Ara-h2 using anti-IgE antibodies after magnetic bead amplification [
58]. This has diagnostic potential to detect IgE antibodies in pure serum while maintaining high diagnostic sensitivity [
58].
Another imaging modality being explored in both murine models and in a clinical trial of 16 peanut-allergic children, detailed below, is thermographic imaging [
57,
60,
61]. Body temperature changes are correlated significantly with allergic anaphylaxis in which allergen crosslinks cause simultaneous mast cell degranulation and release of vasodilatory mediators such as histamine.( One method that has been trialed in patients, is nasal instillation of peanut allergen and measurement of small increases in skin surface temperature using infrared thermographic imaging [
57]. As the nose is highly vascularized and occupied by many mast cells causing a significant amount of local inflammation in a clinically allergic patient, an allergic reaction triggers a significant rise in temperature compared to a placebo [
57]. This method presents several potential advantages including rapid performance, and lower risk of severe and delayed reactions, though not as of yet there have been no studies comparing nasal thermography to DBPCFC [
57].
Most recently, a 2021 study published in Nature by Rath et al. explored cross-sectional imaging using a confocal laser in endomicroscopy in vivo as a means to identify patients with food allergies [
59]. The primary endpoint of the study was to assess the diagnostic performance of barrier dysfunction for food allergy diagnosis [
59]. In patients with food allergies, there was a significant increase in fluorescein leakage compared to those without [
59]. This indication of barrier dysfunction was 96% sensitive with a negative prediction of 96% for the diagnosis of food allergy and the test does not require an exclusion diet prior executing [
59]. This could potentially influence the work-up of food allergy (including peanut), as the patients with low barrier dysfunction would have a low-probability of a food allergy and would not necessitate further evaluation.
4.5. Machine Learning and AI in Allergy Diagnostics
In food allergy, artificial intelligence (AI) has become a new frontier in diagnostics and treatment via machine and deep learning. Essentially, machine learning (ML) uses inputted data through an algorithm to produce an output prediction, while deep learning (DL) can take unstructured or abstracted data and analyze/process it [
62]. A main advantage of AI is capitalizing on its data processing to correlate endotypes with phenotypes [
63]. One study used ML to diagnose food allergy by training networks to find blood epigenetic markers associated with peanut allergy and link those to gene profiles associated with an immune response [
64]. This approach showed high accuracy in differentiating between food sensitization and clinical reactivity, and flagged 13 genes associated with the immune system and food allergy [
64]. ML has also been applied to analyze patterns in Mast Cell Activation Test responses in patients sensitized to peanuts [
65]. This information can contribute to a data set facilitating the quantification of the peanut tolerance threshold based on the test, to solidify the clinical diagnostic application [
63,
66].
AI is also being explored as a tool to predict which patients will develop/ have persistent food allergies [
67]. It is known that discrepancies between got microbiome composition is linked with food sensitization and/ or IgE and non-IgE mediated reactions [
68]. DL frameworks have been applied to infants from birth to three years old in Russia, Finland and Estonia to predict clinical allergy status using a data set analyzing food allergen-specific IgE levels and participant stool sample composition [
68]. The receiver operating characteristic area under the curve (ROC AUC) for this test was 0.69 in predicting clinical food allergies [
68]. The high-risk infants selected under these parameters were then evaluated for peanut-specific and epitope specific IgE and IgG4, total IgE, skin prick test, and clinical history to expand the data set [
68]. Similarly, this antibody repertoire in infancy can then be applied to ML systems to analyze IgE and IgG4 binding, to predict allergy development at 4+ years with an AUC ranging from 0.84 to 0.87. [
69].
Still, difficulty in application of these techniques hinges on several factors including the need for a reliable data set. AI mechanisms are only as good as the data inputted into their system [
62]. Skepticism surrounding patient privacy violation in creating the data sets poses a challenge in healthcare [
62]. Moreover, the data that is collected for input, should be accurate in prediction to outweigh the benefit of established accurate tests such as the oral food challenge or the basophil activation test [
70]. Additionally, integrating AI into allergy diagnostics can enhance precision, yet it is crucial to address the current limitations in data reliability and privacy to truly benefit from its potential [
71].
4.6. Challenges and Limitatons in Emerging Diagnostic Approaches
The field of allergy and immunology has advanced significantly in food allergy diagnostics, but emerging techniques face challenges that impact their safety, efficacy, accessibility, and patient acceptance. Safety concerns arise with methods like Skin Prick Testing (SPT) and Oral Food Challenges (OFCs), which, though effective, carry risks, particularly for those with severe allergic conditions. Newer diagnostics, such as bead-based epitope assays, show promise but require further validation. Cost and accessibility also hinder widespread adoption, as advanced tests are expensive and less available compared to traditional methods. Regulatory hurdles, including lengthy approval processes and a lack of standardized protocols, further delay integration into clinical practice. Patient adherence and acceptance are additional obstacles, with stress and inconvenience deterring individuals from undergoing procedures or following necessary preparations. These factors collectively underscore the need for balance between innovation and practical implementation in allergy diagnostics.
5. Conclusions
Peanut allergy diagnosis is evolving with advanced technologies like molecular diagnostics, microarray technology, basophil activation tests, genetic testing, and machine learning, which promise greater accuracy and personalized care compared to traditional methods such as Skin Prick Testing, serum IgE testing, and Oral Food Challenges. Despite these advancements, challenges including safety, cost, accessibility, patient acceptance, and regulatory barriers hinder widespread adoption. Future developments, such as enhanced molecular profiling, AI integration, non-invasive imaging, rapid point-of-care tools, and telemedicine, hold potential to revolutionize diagnostics by offering safer, more accessible, and effective solutions. Balancing innovation with practical application is key to advancing peanut allergy management and improving patient outcomes.
Author Contributions
T.S. led the conceptualization and writing of the manuscript and coordinated contributions from all authors. N.M., V.P., A.V., and P.S. contributed to the research, drafting, and refinement of the manuscript. D.W. assisted with editing, formatting, and ensuring the accuracy of references. G.K. and M.G. provided expert guidance, critical revisions, and final approval of the manuscript. All authors reviewed the manuscript and approved it for submission.
Funding
This research received no external funding.
References
- C. Warren, D. Lei, S. Sicherer, R. Schleimer, and R. Gupta, “Prevalence and characteristics of peanut allergy in US adults,” J. Allergy Clin. Immunol., vol. 147, no. 6, pp. 2263-2270.e5, Jun. 2021. [CrossRef]
- N.-W. A et al., “The Peanut Allergy Burden Study: Impact on the quality of life of patients and caregivers,” World Allergy Organ. J., vol. 14, no. 2, Feb. 2021. [CrossRef]
- R. S. Gupta et al., “Prevalence and Severity of Food Allergies Among US Adults,” JAMA Netw. Open, vol. 2, no. 1, p. e185630, Jan. 2019. [CrossRef]
- E. Midun, S. Radulovic, H. Brough, and J.-C. Caubet, “Recent advances in the management of nut allergy,” World Allergy Organ. J., vol. 14, no. 1, p. 100491, Jan. 2021. [CrossRef]
- J. Eckman, S. S. Saini, and R. G. Hamilton, “Diagnostic evaluation of food-related allergic diseases,” Allergy Asthma Clin. Immunol., vol. 5, no. 1, p. 2, Oct. 2009. [CrossRef]
- S. H. Sicherer and H. A. Sampson, “Food allergy,” J. Allergy Clin. Immunol., vol. 125, no. 2 Suppl 2, pp. S116-125, Feb. 2010. [CrossRef]
- A.Anyane-Yeboa, W. Wang, and R. T. Kavitt, “The Role of Allergy Testing in Eosinophilic Esophagitis,” Gastroenterol. Hepatol., vol. 14, no. 8, pp. 463–469, Aug. 2018.
- R. Sporik, D. J. Hill, and C. S. Hosking, “Specificity of allergen skin testing in predicting positive open food challenges to milk, egg and peanut in children,” Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol., vol. 30, no. 11, pp. 1540–1546, Nov. 2000. [CrossRef]
- R.-X. Foong, J. A. Dantzer, R. A. Wood, and A. F. Santos, “Improving Diagnostic Accuracy in Food Allergy,” J. Allergy Clin. Immunol. Pract., vol. 9, no. 1, pp. 71–80, Jan. 2021. [CrossRef]
- D. J. Hill, R. G. Heine, and C. S. Hosking, “The diagnostic value of skin prick testing in children with food allergy,” Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol., vol. 15, no. 5, pp. 435–441, Oct. 2004. [CrossRef]
- A.D. Toews, R. B. Ray, N. D. Goines, and T. W. Bouldin, “Increased synthesis of membrane macromolecules is an early response of retinal neurons to trimethyltin intoxication,” Brain Res., vol. 398, no. 2, pp. 298–304, Nov. 1986. [CrossRef]
- G. Roberts and G. Lack, “Diagnosing peanut allergy with skin prick and specific IgE testing,” J. Allergy Clin. Immunol., vol. 115, no. 6, pp. 1291–1296, Jun. 2005. [CrossRef]
- K. Tourlas and D. Burman, “Allergy Testing,” Prim. Care, vol. 43, no. 3, pp. 363–374, Sep. 2016. [CrossRef]
- R. L. Werther, S. Choo, K. J. Lee, D. Poole, K. J. Allen, and M. L. Tang, “Variability in Skin Prick Test Results Performed by Multiple Operators Depends on the Device Used,” World Allergy Organ. J., vol. 5, no. 12, p. 200, Dec. 2012. [CrossRef]
- M. T. Kraft et al., “The sensitivity of food allergy skin testing soon after food-induced anaphylaxis,” J. Allergy Clin. Immunol. Pract., vol. 12, no. 5, pp. 1384-1386.e2, May 2024. [CrossRef]
- M. Alnæs, “Anaphylaxis following prick-by-prick testing with peanut,” Clin. Case Rep., vol. 8, no. 12, p. 2366, Jul. 2020. [CrossRef]
- H. M. Kansen et al., “Diagnostic accuracy of Ara h 2 for detecting peanut allergy in children,” Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol., vol. 51, no. 8, pp. 1069–1079, Aug. 2021. [CrossRef]
- O. Hemmings, G. D. Toit, S. Radulovic, G. Lack, and A. F. Santos, “Ara h 2 is the dominant peanut allergen despite similarities with Ara h 6,” J. Allergy Clin. Immunol., vol. 146, no. 3, pp. 621-630.e5, Sep. 2020. [CrossRef]
- A. Ji et al., “Ara h 2-Specific IgE Presence Rather Than Its Function Is the Best Predictor of Mast Cell Activation in Children,” J. Allergy Clin. Immunol. Pract., vol. 11, no. 4, pp. 1154-1161.e3, Apr. 2023. [CrossRef]
- R. A. Wood, N. Segall, S. Ahlstedt, and P. B. Williams, “Accuracy of IgE antibody laboratory results,” Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol., vol. 99, no. 1, pp. 34–41, Jul. 2007. [CrossRef]
- S. Anvari, J. Miller, C.-Y. Yeh, and C. M. Davis, “IgE-Mediated Food Allergy,” Clin. Rev. Allergy Immunol., vol. 57, no. 2, pp. 244–260, Oct. 2019. [CrossRef]
- R. J. B. Klemans et al., “The diagnostic value of specific IgE to Ara h 2 to predict peanut allergy in children is comparable to a validated and updated diagnostic prediction model,” J. Allergy Clin. Immunol., vol. 131, no. 1, pp. 157–163, Jan. 2013. [CrossRef]
- N. A. LaHood and S. U. Patil, “Food Allergy Testing,” Clin. Lab. Med., vol. 39, no. 4, pp. 625–642, Dec. 2019. [CrossRef]
- M. Kulis, B. L. Wright, S. M. Jones, and A. W. Burks, “Diagnosis, management, and investigational therapies for food allergies,” Gastroenterology, vol. 148, no. 6, pp. 1132–1142, May 2015. [CrossRef]
- H. A. Sampson, “Update on food allergy,” J. Allergy Clin. Immunol., vol. 113, no. 5, pp. 805–819; quiz 820, May 2004. [CrossRef]
- K. Y. Kwong and Y. Z. Lu, “Cost of Serum Versus Skin Allergy Testing Among Medicare Fee-for-Service Beneficiaries in the United States,” J. Health Econ. Outcomes Res., vol. 10, no. 2, pp. 14–21, 2023. [CrossRef]
- V. Fuhrmann et al., “From Allergen Molecules to Molecular Immunotherapy of Nut Allergy: A Hard Nut to Crack,” Front. Immunol., vol. 12, p. 742732, 2021. [CrossRef]
- P. M. Matricardi et al., “EAACI Molecular Allergology User’s Guide,” Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol., vol. 27 Suppl 23, pp. 1–250, May 2016. [CrossRef]
- R. S. Chinthrajah, J. D. Hernandez, S. D. Boyd, S. J. Galli, and K. C. Nadeau, “Molecular and cellular mechanisms of food allergy and food tolerance,” J. Allergy Clin. Immunol., vol. 137, no. 4, pp. 984–997, Apr. 2016. [CrossRef]
- J. Połomska, P. Dydak, B. Sozańska, and H. Sikorska-Szaflik, “Peanut Allergy and Component-Resolved Diagnostics Possibilities-What Are the Benefits?,” Nutrients, vol. 15, no. 24, p. 5132, Dec. 2023. [CrossRef]
- A. Valcour, J. E. Jones, J. Lidholm, M. P. Borres, and R. G. Hamilton, “Sensitization profiles to peanut allergens across the United States,” Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol., vol. 119, no. 3, pp. 262-266.e1, Sep. 2017. [CrossRef]
- Vereda et al., “Peanut allergy: Clinical and immunologic differences among patients from 3 different geographic regions,” J. Allergy Clin. Immunol., vol. 127, no. 3, pp. 603–607, Mar. 2011. [CrossRef]
- F. C. van Erp, R. J. B. Klemans, Y. Meijer, C. K. van der Ent, and A. C. Knulst, “Using Component-Resolved Diagnostics in the Management of Peanut-Allergic Patients,” Curr. Treat. Options Allergy, vol. 3, pp. 169–180, 2016. [CrossRef]
- H. A. Sampson et al., “Standardizing double-blind, placebo-controlled oral food challenges: American Academy of Allergy, Asthma & Immunology-European Academy of Allergy and Clinical Immunology PRACTALL consensus report,” J. Allergy Clin. Immunol., vol. 130, no. 6, pp. 1260–1274, Dec. 2012. [CrossRef]
- T. T. Perry, E. C. Matsui, M. K. Conover-Walker, and R. A. Wood, “Risk of oral food challenges,” J. Allergy Clin. Immunol., vol. 114, no. 5, pp. 1164–1168, Nov. 2004. [CrossRef]
- J. Upton, M. Alvaro, and K. Nadeau, “A perspective on the pediatric death from oral food challenge reported from the Allergy Vigilance Network,” Allergy, vol. 74, no. 6, pp. 1035–1036, Jun. 2019. [CrossRef]
- J. A. Bird et al., “Conducting an Oral Food Challenge: An Update to the 2009 Adverse Reactions to Foods Committee Work Group Report,” J. Allergy Clin. Immunol. Pract., vol. 8, no. 1, pp. 75-90.e17, Jan. 2020. [CrossRef]
- P. J. Turner, E. Jerschow, T. Umasunthar, R. Lin, D. E. Campbell, and R. J. Boyle, “Fatal Anaphylaxis: Mortality Rate and Risk Factors,” J. Allergy Clin. Immunol. Pract., vol. 5, no. 5, pp. 1169–1178, 2017. [CrossRef]
- M. Suárez-Fariñas et al., “Accurate and reproducible diagnosis of peanut allergy using epitope mapping,” Allergy, vol. 76, no. 12, pp. 3789–3797, Dec. 2021. [CrossRef]
- H.-J. Huang et al., “Microarray-Based Allergy Diagnosis: Quo Vadis?,” Front. Immunol., vol. 11, p. 594978, 2020. [CrossRef]
- T. Jakob, P. Forstenlechner, P. Matricardi, and J. Kleine-Tebbe, “Molecular allergy diagnostics using multiplex assays: methodological and practical considerations for use in research and clinical routine: Part 21 of the Series Molecular Allergology,” Allergo J. Int., vol. 24, pp. 320–332, 2015. [CrossRef]
- R. G. Hamilton, “Microarray Technology Applied to Human Allergic Disease,” Microarrays Basel Switz., vol. 6, no. 1, p. 3, Jan. 2017. [CrossRef]
- J.-P. Borges et al., “Allergenarrays: low-cost food allergy diagnosis using allergen extracts-based microarrays,” Clin. Transl. Allergy, vol. 4, no. Suppl 2, p. P37, Mar. 2014. [CrossRef]
- O. Hemmings, M. Kwok, R. McKendry, and A. F. Santos, “Basophil Activation Test: Old and New Applications in Allergy,” Curr. Allergy Asthma Rep., vol. 18, no. 12, p. 77, Nov. 2018. [CrossRef]
- H. Jaumdally et al., “Basophil activation test has high reproducibility and is feasible in the clinical setting,” Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol., vol. 33, no. 11, p. e13870, Oct. 2022. [CrossRef]
- A. Mari et al., “Specific IgE to cross-reactive carbohydrate determinants strongly affect the in vitro diagnosis of allergic diseases,” J. Allergy Clin. Immunol., vol. 103, no. 6, pp. 1005–1011, Jun. 1999. [CrossRef]
- S. Glaumann, A. Nopp, S. G. O. Johansson, M. Rudengren, M. P. Borres, and C. Nilsson, “Basophil allergen threshold sensitivity, CD-sens, IgE-sensitization and DBPCFC in peanut-sensitized children,” Allergy, vol. 67, no. 2, pp. 242–247, Feb. 2012. [CrossRef]
- L. Duan et al., “Basophil activation test shows high accuracy in the diagnosis of peanut and tree nut allergy: The Markers of Nut Allergy Study,” Allergy, vol. 76, no. 6, pp. 1800–1812, Jun. 2021. [CrossRef]
- S. Chirumbolo, “Immunotherapy in allergy and cellular tests: state of art,” Hum. Vaccines Immunother., vol. 10, no. 6, pp. 1595–1610, 2014. [CrossRef]
- H. J. Hoffmann et al., “The clinical utility of basophil activation testing in diagnosis and monitoring of allergic disease,” Allergy, vol. 70, no. 11, pp. 1393–1405, Nov. 2015. [CrossRef]
- A. F. Santos et al., “Distinct parameters of the basophil activation test reflect the severity and threshold of allergic reactions to peanut,” J. Allergy Clin. Immunol., vol. 135, no. 1, pp. 179–186, Jan. 2015. [CrossRef]
- D. B. Noriega, M. Teodorowicz, H. Savelkoul, and J. Ruinemans-Koerts, “The Basophil Activation Test for Clinical Management of Food Allergies: Recent Advances and Future Directions,” J. Asthma Allergy, vol. 14, p. 1335, Nov. 2021. [CrossRef]
- S. J. Brown et al., “Loss-of-function variants in the filaggrin gene are a significant risk factor for peanut allergy,” J. Allergy Clin. Immunol., vol. 127, no. 3, pp. 661–667, Mar. 2011. [CrossRef]
- G. R. Donovan et al., “A family study of allergy: segregation with HLA but not with T-cell receptor genes,” J. Allergy Clin. Immunol., vol. 97, no. 2, pp. 712–713, Feb. 1996. [CrossRef]
- M. Kostara, V. Chondrou, A. Sgourou, K. Douros, and S. Tsabouri, “HLA Polymorphisms and Food Allergy Predisposition,” J. Pediatr. Genet., vol. 9, no. 2, pp. 77–86, Jun. 2020. [CrossRef]
- E. Rodríguez et al., “Meta-analysis of filaggrin polymorphisms in eczema and asthma: robust risk factors in atopic disease,” J. Allergy Clin. Immunol., vol. 123, no. 6, pp. 1361-1370.e7, Jun. 2009. [CrossRef]
- A. Clark et al., “Thermographic imaging during nasal peanut challenge may be useful in the diagnosis of peanut allergy,” Allergy, vol. 67, no. 4, pp. 574–576, Apr. 2012. [CrossRef]
- A. Joshi, M. W. Peczuh, C. V. Kumar, and J. F. Rusling, “Ultrasensitive carbohydrate-peptide SPR imaging microarray for diagnosing IgE mediated peanut allergy,” The Analyst, vol. 139, no. 22, pp. 5728–5733, Nov. 2014. [CrossRef]
- T. Rath, W. Dieterich, C. Kätscher-Murad, M. F. Neurath, and Y. Zopf, “Cross-sectional imaging of intestinal barrier dysfunction by confocal laser endomicroscopy can identify patients with food allergy in vivo with high sensitivity,” Sci. Rep., vol. 11, no. 1, p. 12777, Jun. 2021. [CrossRef]
- P. Peppers et al., “Stratification of peanut allergic murine model into anaphylaxis severity risk groups using thermography,” J. Immunol. Methods, vol. 459, pp. 29–34, Aug. 2018. [CrossRef]
- K. Manzano-Szalai, I. Pali-Schöll, D. Krishnamurthy, C. Stremnitzer, I. Flaschberger, and E. Jensen-Jarolim, “Anaphylaxis Imaging: Non-Invasive Measurement of Surface Body Temperature and Physical Activity in Small Animals,” PloS One, vol. 11, no. 3, p. e0150819, 2016. [CrossRef]
- M. van Breugel et al., “Current state and prospects of artificial intelligence in allergy,” Allergy, vol. 78, no. 10, pp. 2623–2643, Oct. 2023. [CrossRef]
- G. Ferrante, A. Licari, S. Fasola, G. L. Marseglia, and S. La Grutta, “Artificial intelligence in the diagnosis of pediatric allergic diseases,” Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol., vol. 32, no. 3, pp. 405–413, Apr. 2021. [CrossRef]
- A. Alag, “Machine learning approach yields epigenetic biomarkers of food allergy: A novel 13-gene signature to diagnose clinical reactivity,” PloS One, vol. 14, no. 6, p. e0218253, 2019. [CrossRef]
- R. Bahri et al., “Mast cell activation test in the diagnosis of allergic disease and anaphylaxis,” J. Allergy Clin. Immunol., vol. 142, no. 2, pp. 485-496.e16, Aug. 2018. [CrossRef]
- K. W. Chong, M. Ruiz-Garcia, N. Patel, R. J. Boyle, and P. J. Turner, “Reaction phenotypes in IgE-mediated food allergy and anaphylaxis,” Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol., vol. 124, no. 5, pp. 473–478, May 2020. [CrossRef]
- MacMath, M. Chen, and P. Khoury, “Artificial Intelligence: Exploring the Future of Innovation in Allergy Immunology,” Curr. Allergy Asthma Rep., vol. 23, no. 6, pp. 351–362, Jun. 2023. [CrossRef]
- A. A. Metwally, P. S. Yu, D. Reiman, Y. Dai, P. W. Finn, and D. L. Perkins, “Utilizing longitudinal microbiome taxonomic profiles to predict food allergy via Long Short-Term Memory networks,” PLoS Comput. Biol., vol. 15, no. 2, p. e1006693, Feb. 2019. [CrossRef]
- M. Suprun et al., “Early epitope-specific IgE antibodies are predictive of childhood peanut allergy,” J. Allergy Clin. Immunol., vol. 146, no. 5, pp. 1080–1088, Nov. 2020. [CrossRef]
- S. K. Y. Tang, N. Castaño, K. C. Nadeau, and S. J. Galli, “Can artificial intelligence (AI) replace oral food challenge?,” J. Allergy Clin. Immunol., vol. 153, no. 3, pp. 666–668, Mar. 2024. [CrossRef]
- M. Khan et al., “The Impact of Artificial Intelligence on Allergy Diagnosis and Treatment,” Curr. Allergy Asthma Rep., vol. 24, no. 7, pp. 361–372, Jul. 2024. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).