Submitted:
13 January 2026
Posted:
13 January 2026
You are already at the latest version
Abstract
Keywords:
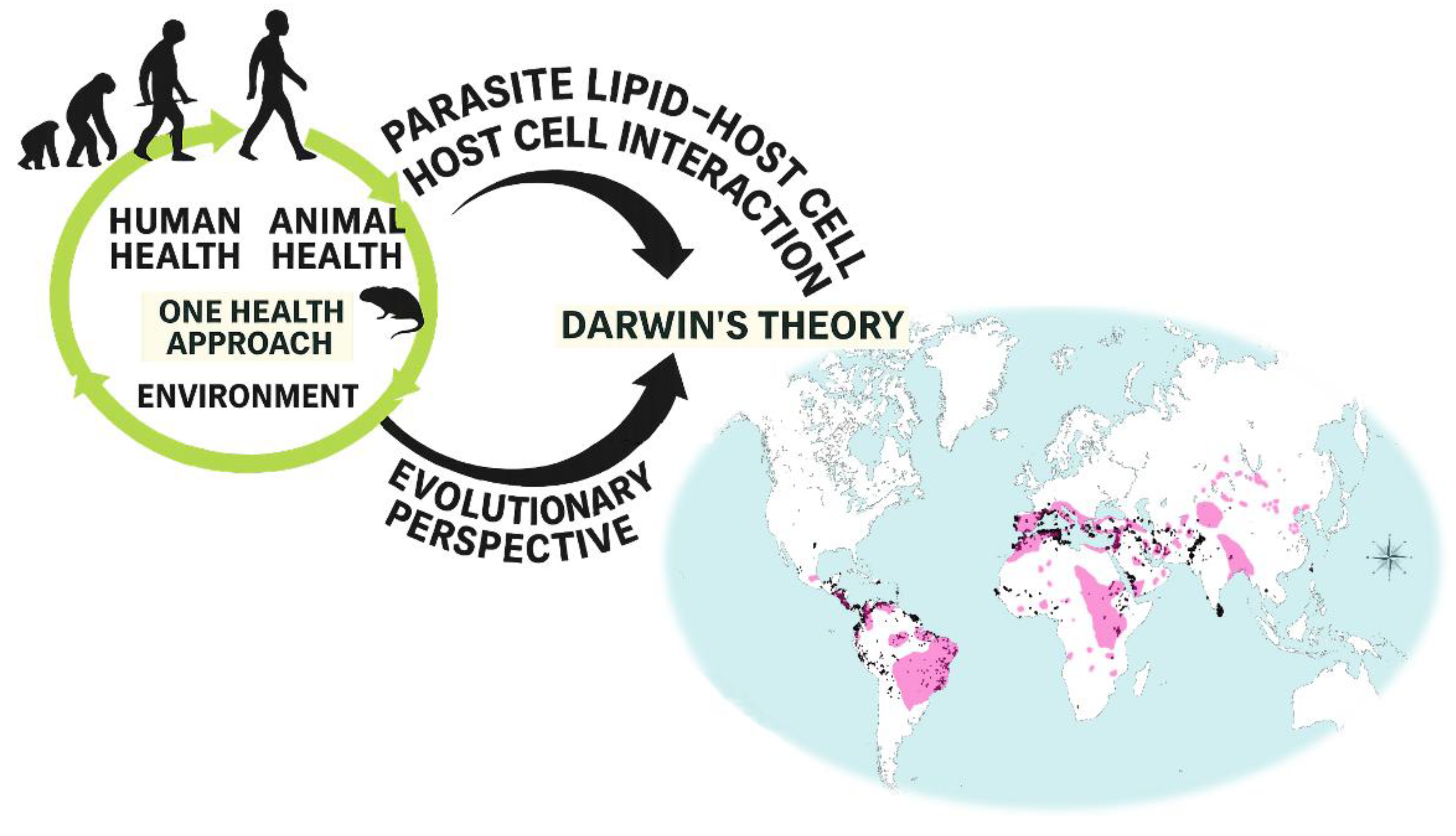
1. Introduction
2. Materials and Methods
2.1. Parasite Culture
2.2. Animals and Peritoneal Macrophage Isolation and Culture
2.3. Infection of Macrophages
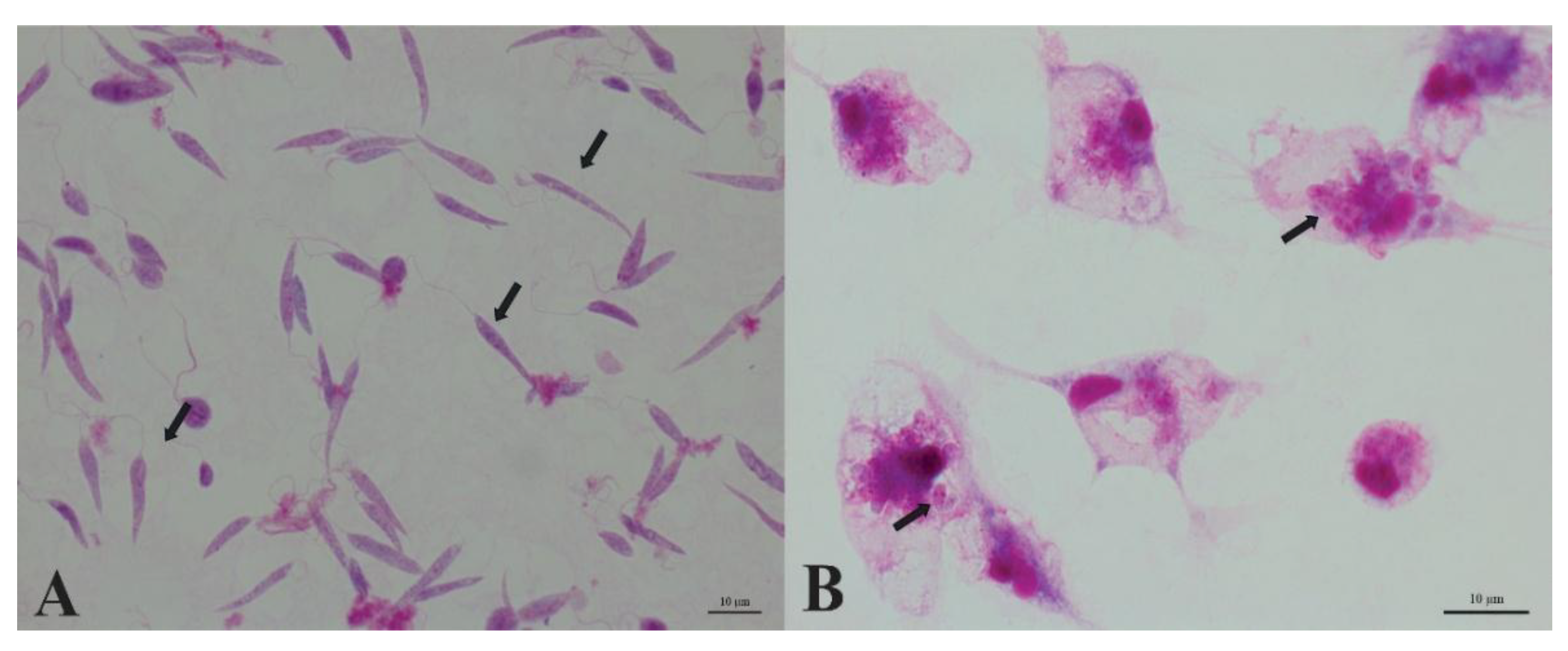
2.4. Light Microscopy and Giemsa Staining
2.4.1. Giemsa Staining – Promastigotes
2.4.2. Giemsa Staining – Intracellular Amastigotes
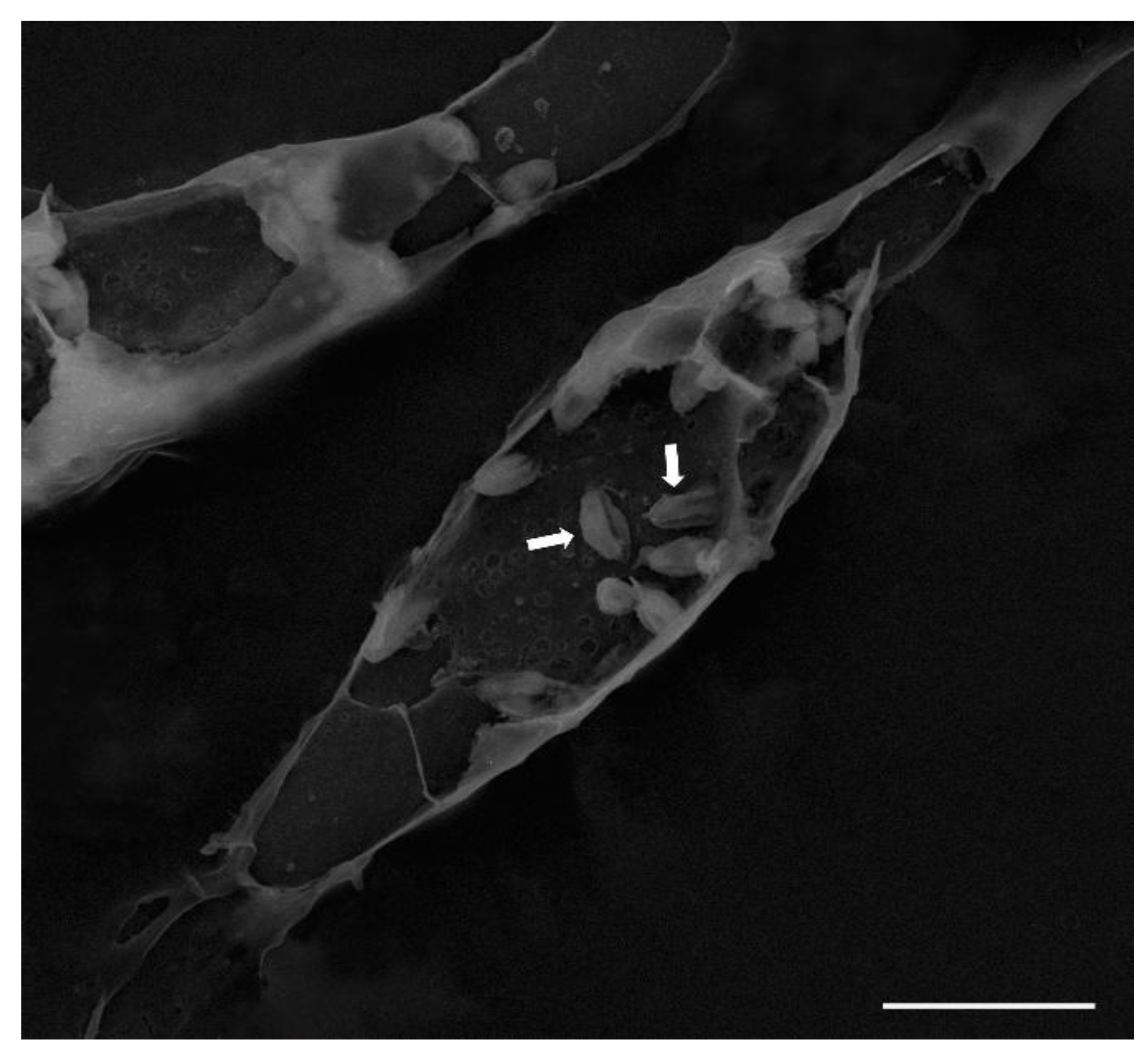
2.5. Scanning Electron Microscopy (SEM)
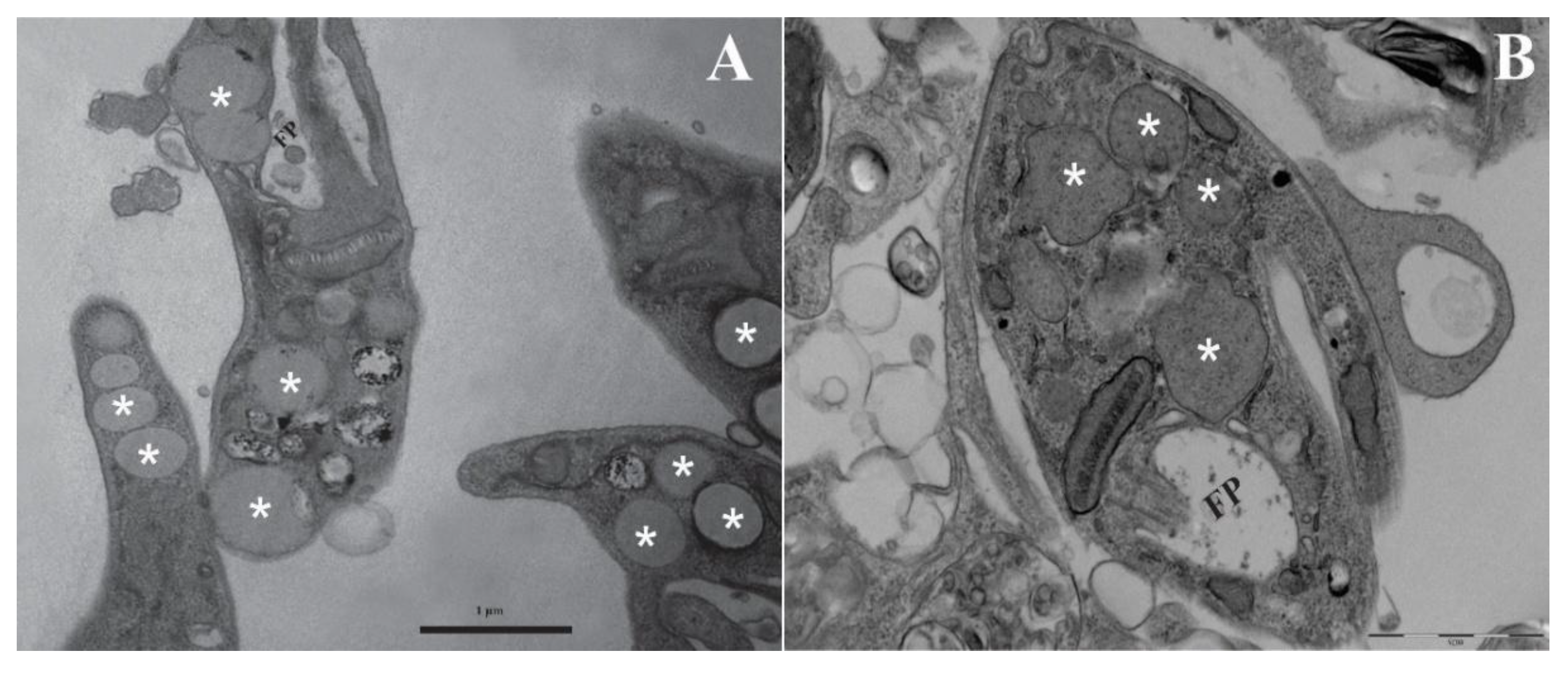
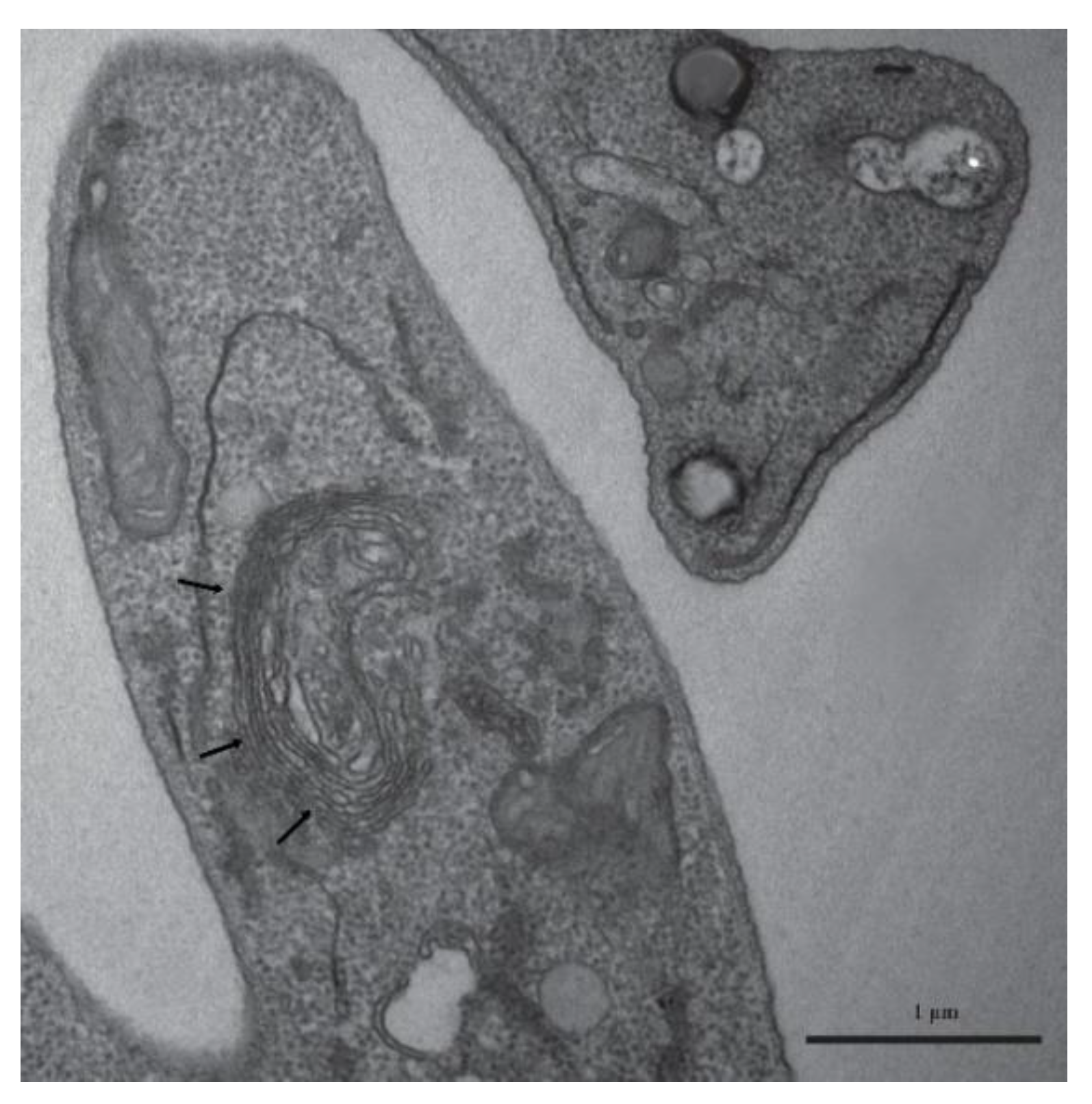
2.6. Transmission Electron Microscopy (TEM)
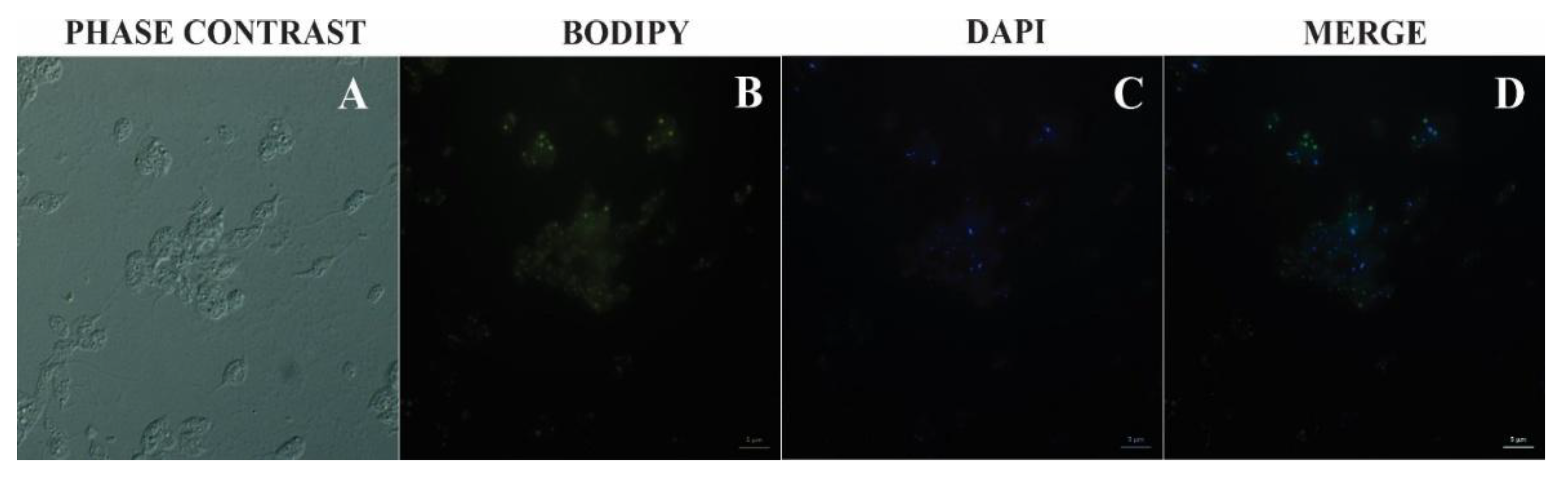
2.7. Bodipy Lipid Staining
3. Results
3.1. Promastigote Morphology
3.2. Intracellular Amastigotes
3.3. Lipid-Body–Like Structures
3.4. Bodipy Confirmation
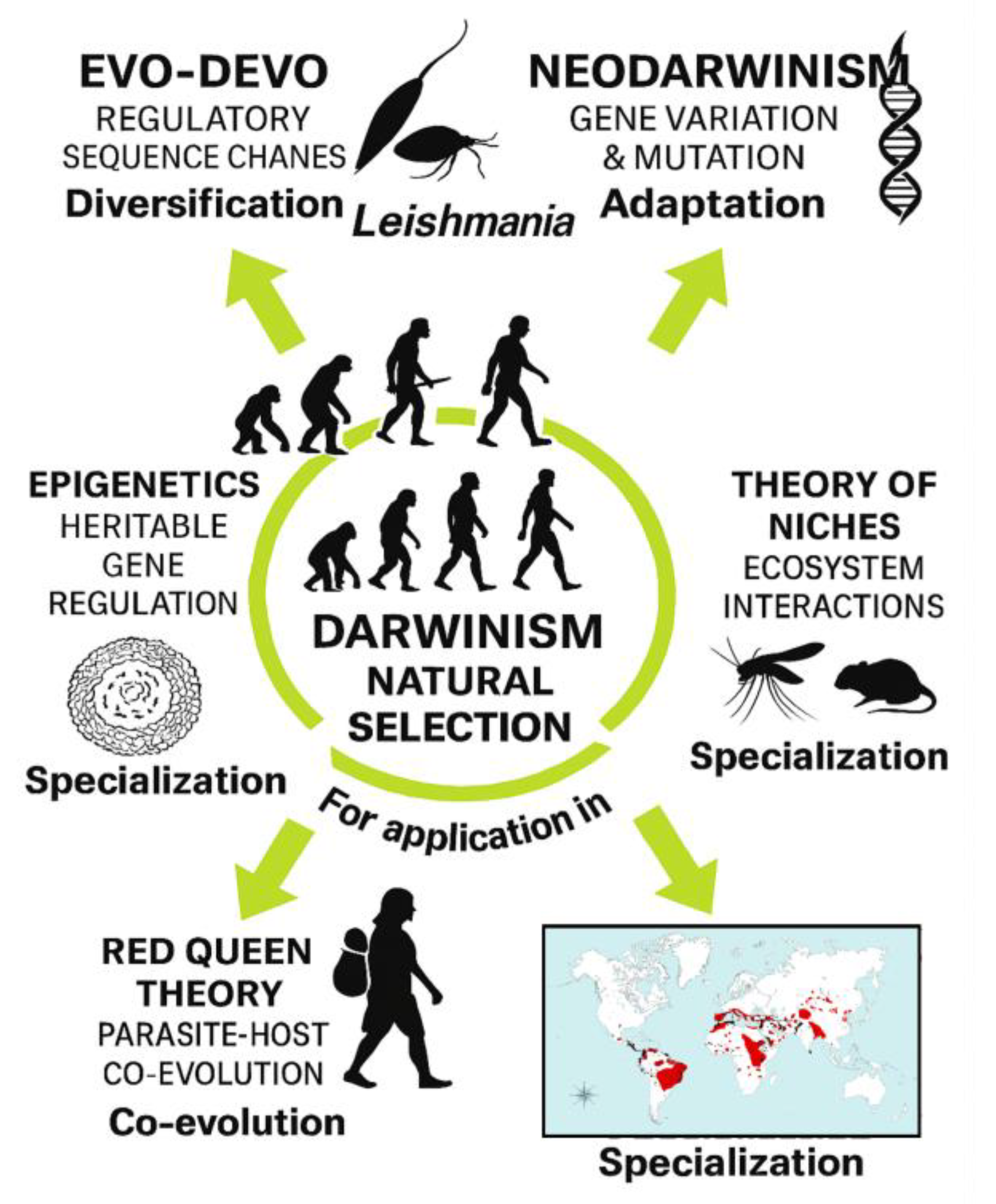
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
| AG | Fatty acids |
| BODIPY | Boron-dipyrromethene |
| CRISPR | Clustered regularly interspaced short palindromic repeats |
| DNA | Desoxyribonucleic acid |
| EM | Electron microscopy |
| EV | Extracellular vesicle |
| FA | Fatty acid |
| GPI | Glycosylphosphatidylinositol |
| LD | Lipid droplet |
| LEV | Leishmania extracellular vesicle |
| LC-MS/MS | Liquid chromatography–tandem mass spectrometry |
| MS | Mass spectrometry |
| PBS | Phosphate-buffered saline |
| RNA | Ribonucleic acid |
| TEM | Transmission electron microscopy |
| Evo-Devo | Evolutionary developmental biology |
| RQT | Red Queen Theory |
| PHCE | Parasite–Host Co-Evolution |
| PHCED | Parasite–Host Co-Evolutionary Dynamics |
Appendix A. Terminology and Conceptual Foundations
Appendix A.1 Terminology Used to Describe Lipid-Related Processes
Appendix B. Integrated Conceptual and Methodological Frameworks
Appendix B.1 Integrated Frameworks, Resources, and Methodological Approaches for Lipid and Extracellular Vesicle Research in Leishmania
Appendix C — Comparative Landscape of Lipid-Centered EV Research
Appendix C.1 Overview of Lipid-Centered Studies and Extracellular Vesicle (EV/LEV) Research in Trypanosomatids
References
- Atayde, V.D.; Hassani, K.; da Silva Lira Filho, A.; et al. Extracellular Vesicles in Leishmania Infection: Modulators of Host–Parasite Interaction. PLoS Pathog. 2023, 19, e1012636. [Google Scholar] [CrossRef]
- Silverman, J.M.; Clos, J.; de’Oliveira, C.C.; et al. Leishmania Exosomes Modulate Innate Immunity and Promote Infection. Proc. Natl. Acad. Sci. USA 2010, 107, 21635–21640. [Google Scholar] [CrossRef]
- Parreira de Aquino, G.; et al. Lipid Metabolism in Leishmania: Implications for Pathogenesis and Drug Resistance. Microb. Cell 2021, 8, 88–104. [Google Scholar] [CrossRef]
- Santos, L.C.; et al. Metabolomic Profiling of Leishmania Species Reveals Lipid Remodeling During Infection. Metabolites 2024, 14, 658. [Google Scholar] [CrossRef]
- Pal, P.; Das, S.; Chatterjee, N.; Bose, D.; Saha, K.D. Studies on the Anti-Inflammatory Effect of Leishmanial Lipid In Vitro and In Vivo. Prajnan O Sadhona 2015, 2, 1–XX. [Google Scholar]
- Sacks, D.; et al. Lipid Bodies in Leishmania: Structure, Function, and Role in Host Interaction. Mol. Biochem. Parasitol. 2009, 165, 1–9. [Google Scholar] [CrossRef]
- Gonçalves, R.; et al. Extracellular Vesicles from Protozoan Parasites: Biogenesis, Composition, and Function. Front. Microbiol. 2016, 7, 427. [Google Scholar] [CrossRef]
- Kumar, A.; et al. Lipid-Mediated Modulation of Host Immunity by Leishmania spp. Exp. Parasitol. 2021, 223, 107999. [Google Scholar] [CrossRef]
- Chowdhury, S.; et al. Host–Parasite Lipid Interactions in Leishmaniasis: Mechanisms and Therapeutic Perspectives. Int. J. Mol. Sci. 2022, 23, 2414. [Google Scholar] [CrossRef]
- Macedo, A.M.; et al. Lipidomic Signatures of Leishmania Extracellular Vesicles Reveal Species-Specific Patterns. Int. J. Mol. Sci. 2023, 24, 10637. [Google Scholar] [CrossRef]
- Rodrigues, J.C.F.; et al. Metabolic Adaptations of Leishmania Parasites: Lipid Remodeling and Survival Strategies. Microorganisms 2023, 13, 531. [Google Scholar] [CrossRef]
- Serrano, A.; et al. Lipid Droplets and Vesicular Trafficking in Leishmania: Ultrastructural Insights. Micron 2021, 142, 103089. [Google Scholar] [CrossRef]
- Parreira de Aquino, G.; et al. Lipid Remodeling in Leishmania: A Microbial Cell Perspective. Microb. Cell 2021, 8, 1–12. [Google Scholar] [CrossRef]
- Costa, D.L.; et al. Lipid-Rich Extracellular Vesicles in Leishmania Infection: Implications for Pathogenesis. Int. J. Parasitol. 2024, 54, 1–18. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Leishmaniasis; WHO Fact Sheet, April 2017. Available online: https://www.who.int/mediacentre/factsheets/fs375/en/ (accessed on 24 July 2017).
- Carroll, S.B. Evo-Devo and the Evolution of Animal Diversity. Cell 2005, 120, 201–207. [Google Scholar] [CrossRef]
- Hutchinson, G.E. Concluding Remarks. Cold Spring Harb. Symp. Quant. Biol. 1957, 22, 415–427. [Google Scholar] [CrossRef]
- Bird, A. Perceptions of Epigenetics. Nature 2007, 447, 396–398. [Google Scholar] [CrossRef]
- Van Valen, L. A New Evolutionary Law. Evolutionary Theory 1973, 1, 1–30. [Google Scholar]
- Schmid-Hempel, P. Evolutionary Parasitology; Oxford University Press: Oxford, UK, 2011; ISBN 978-0199229482. [Google Scholar]
- Fisher Scientific. Lipid-related reagents and analytical products. Available online: https://www.fishersci.pt/pt/en/catalog/search/products?keyword=lipid (accessed on 7 January 2026).
- International Society for Extracellular Vesicles (ISEV). MISEV2023: Minimal Information for Studies of Extracellular Vesicles. J. Extracell. Vesicles 2023. Available online: https://isevjournals.onlinelibrary.wiley.com/doi/10.1002/jev2.12404 (accessed on 7 January 2026).
- Gabriel, Á.M.; Galué-Parra, A.; Pereira, W.L.A.; Pedersen, K.W.; da Silva, E.O. Leishmania 360°: Guidelines for Exosomal Research. Microorganisms 2021, 9, 2081. [Google Scholar] [CrossRef]
- Gabriel, Á.M.; Galvão, G.R.; Galué-Parra, A.; Casseb, L.M.N.; Pereira, W.L.A.; Pedersen, K.W.; Aguiar, D.C.F.; Gonçalves, E.C.; da Silva, E.O. Pathogenesis of canine leishmaniasis: diagnostic accuracy and experimental models targeting Leishmania lipid-bound vesicles. Acad. Biol. 2025, 3(1), 1–XX. [Google Scholar] [CrossRef]
- Booth, L.-A.; Smith, T.K. Lipid Metabolism in Trypanosoma cruzi: A Review. Mol. Biochem. Parasitol. 2020, 240, 111324. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



