Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
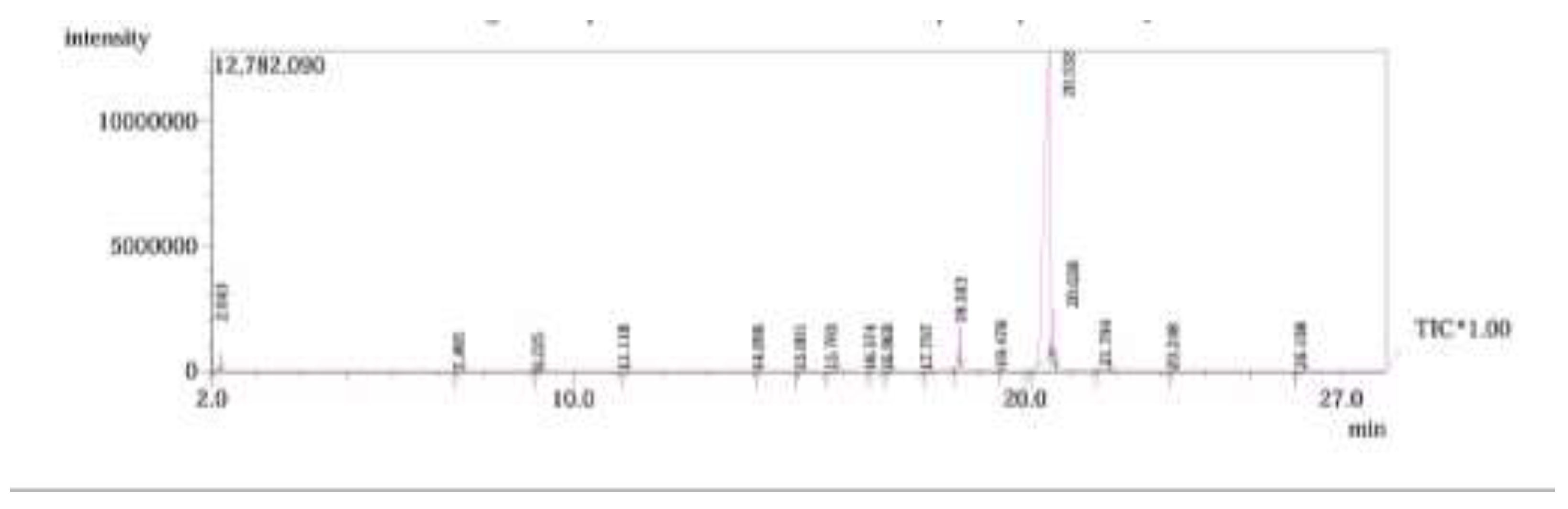
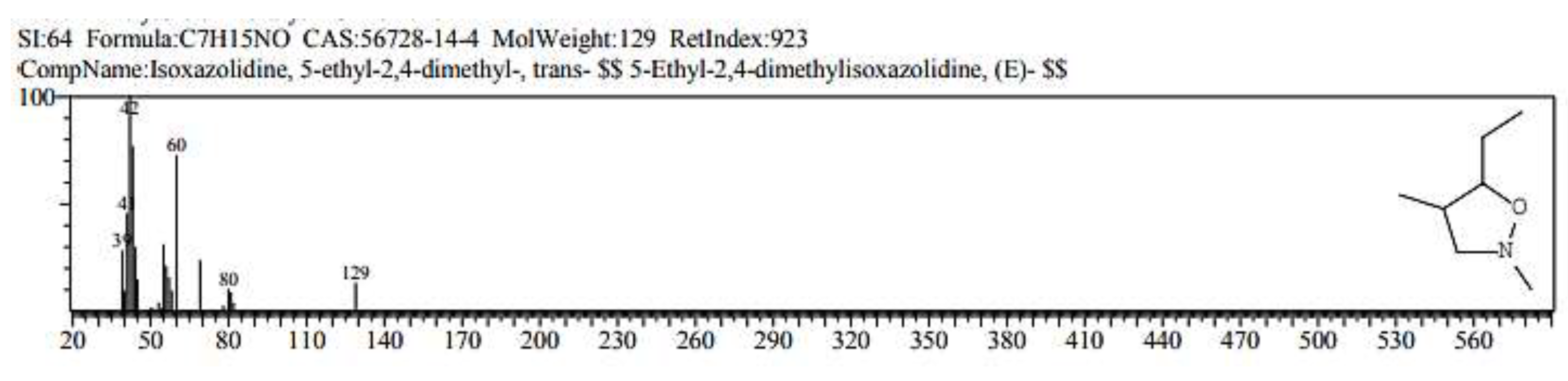
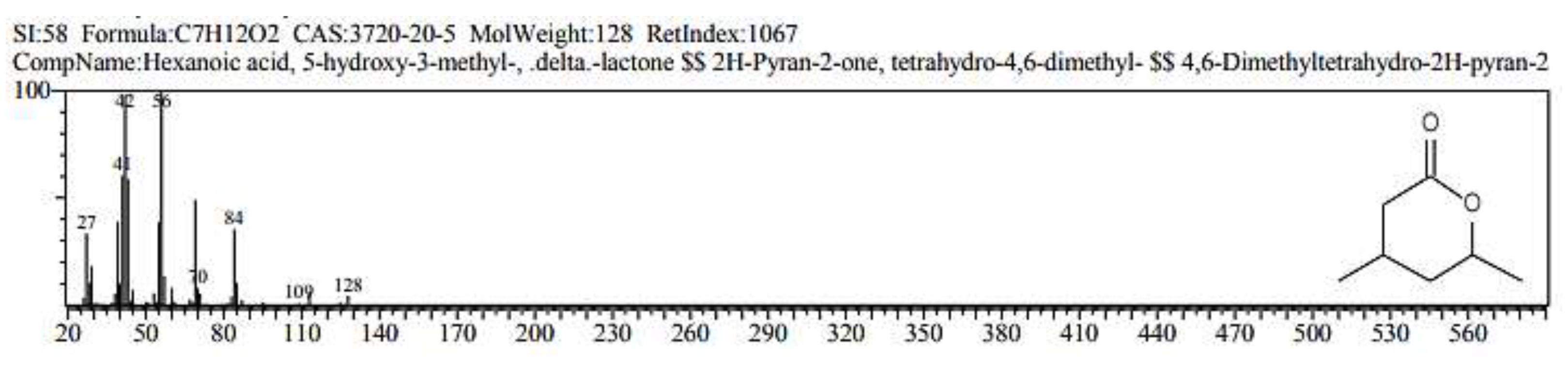
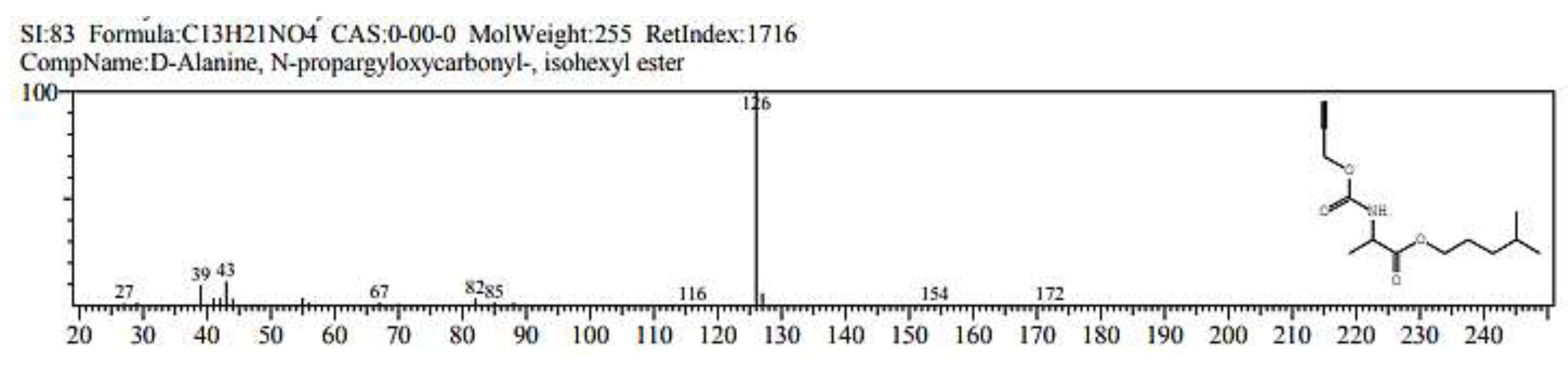
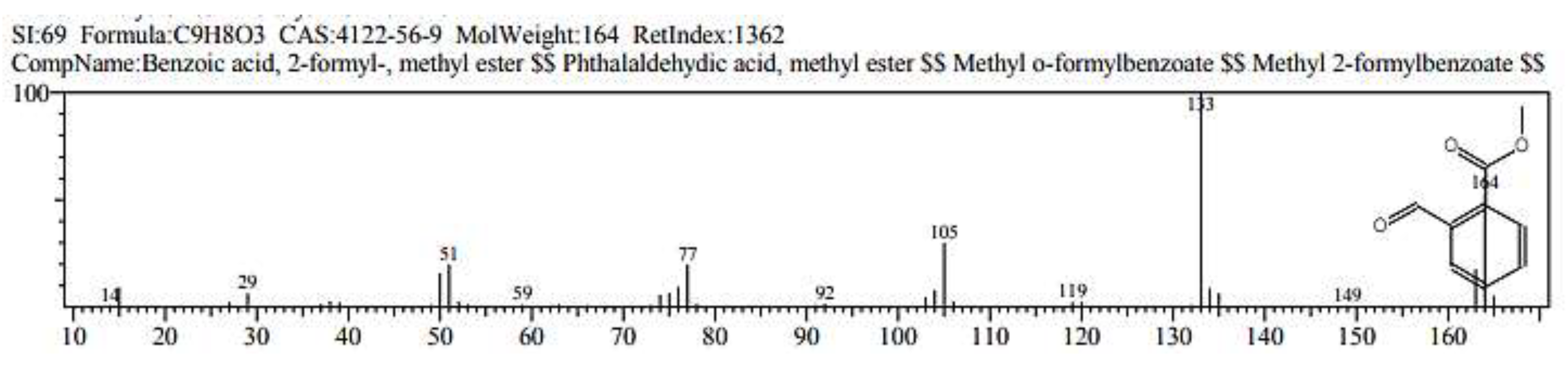
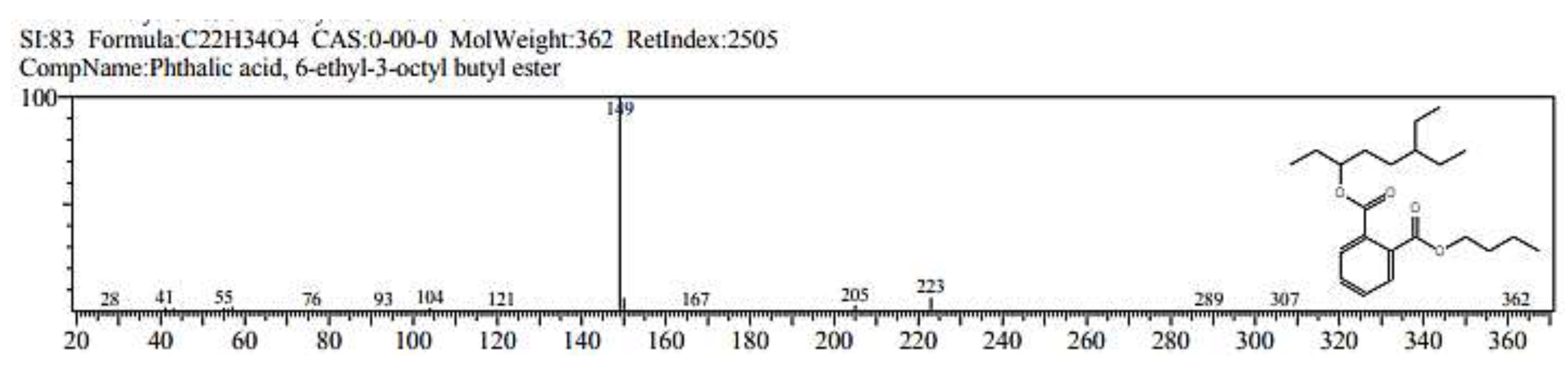
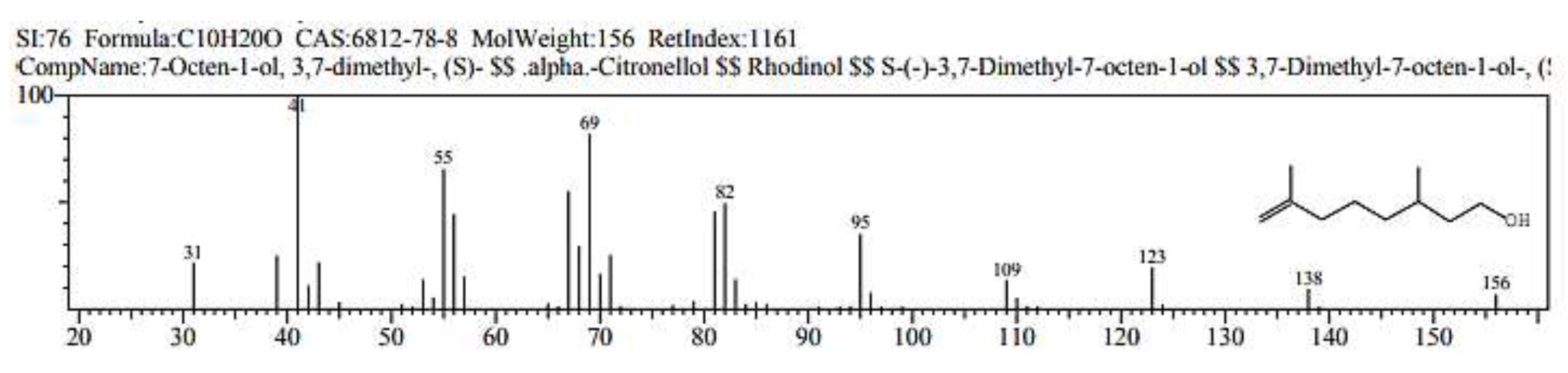
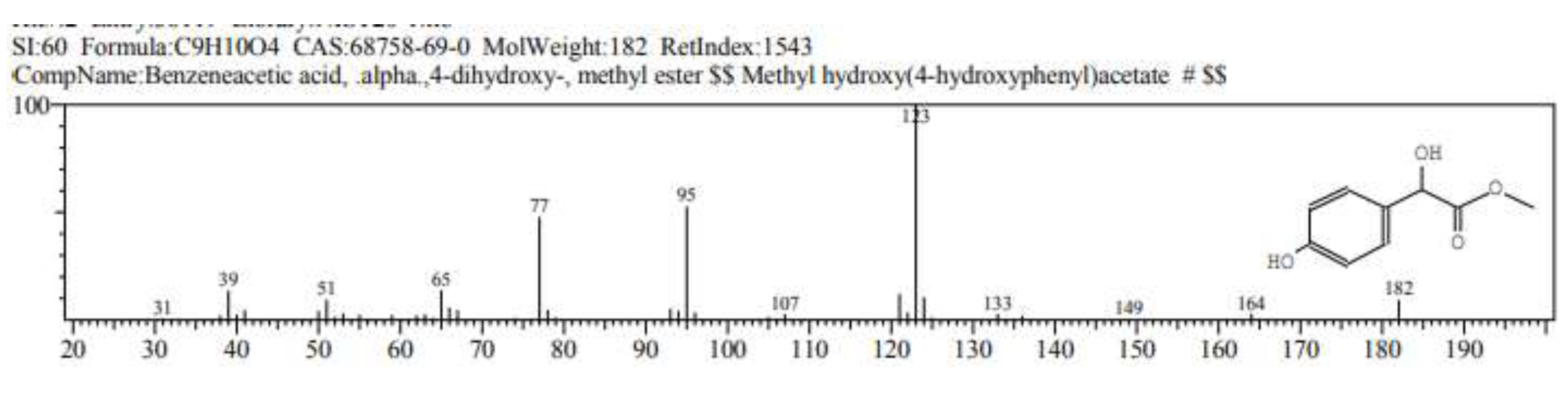
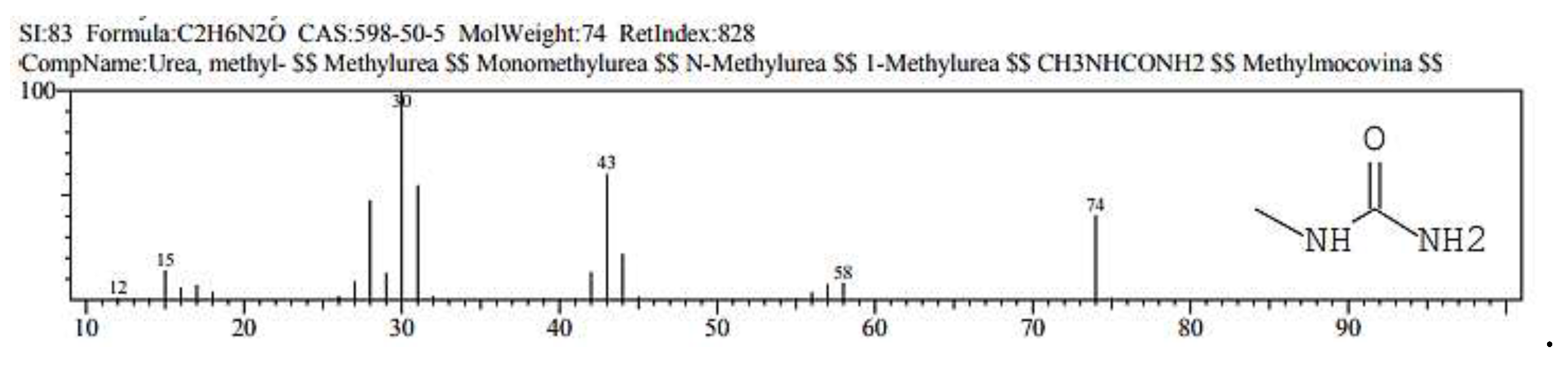
2.1. GC/ MS analysis of Olea europaea
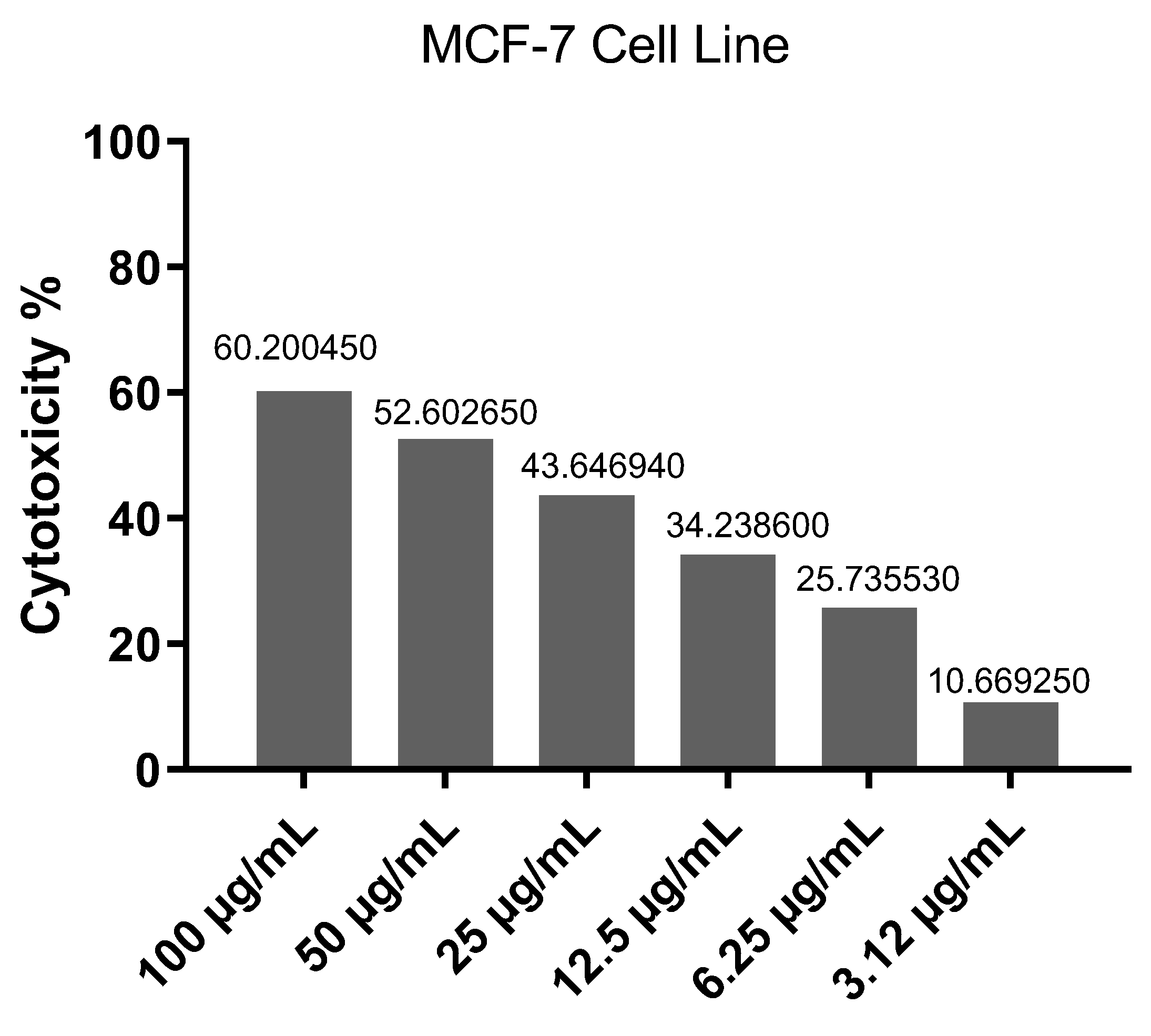
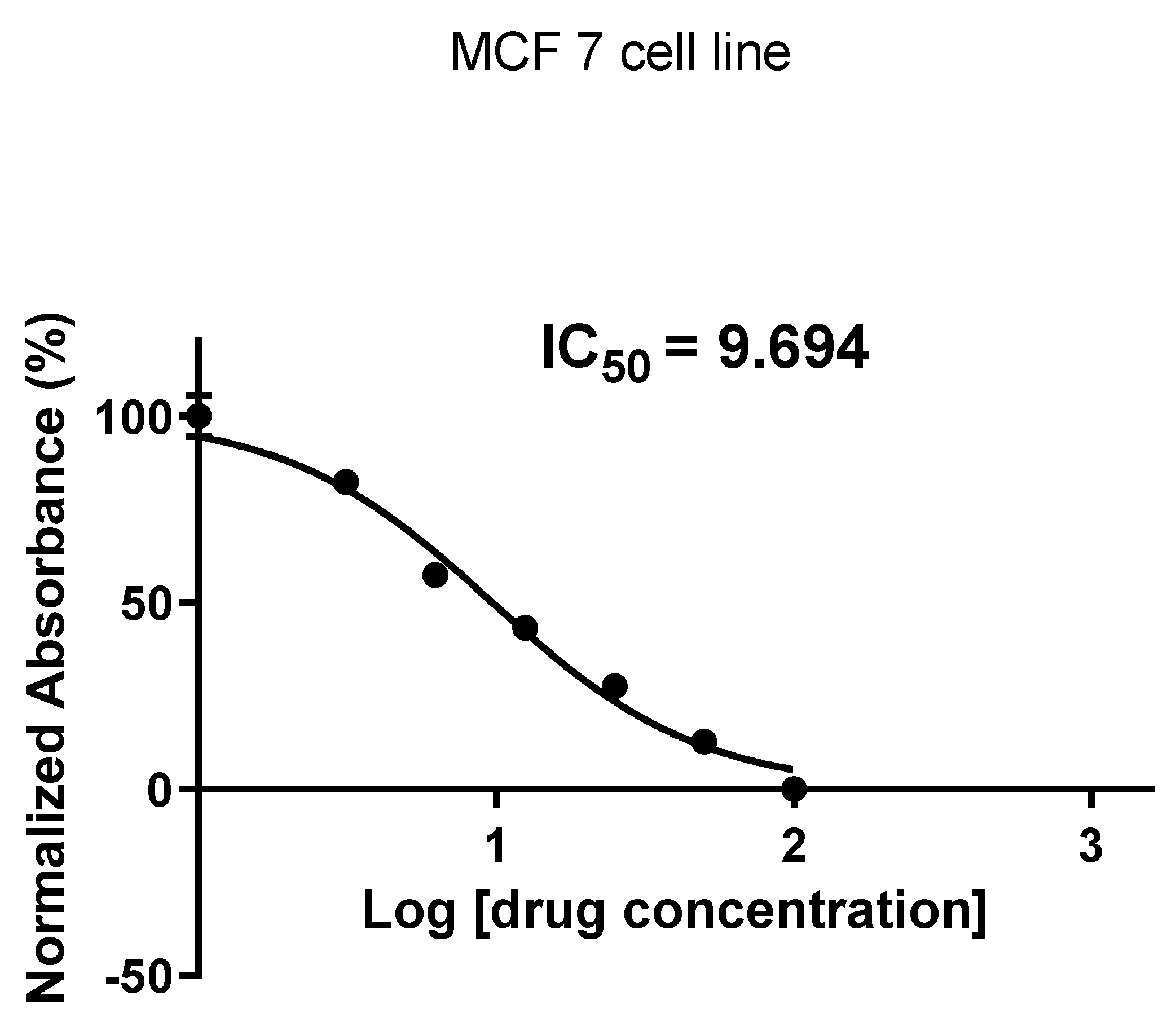
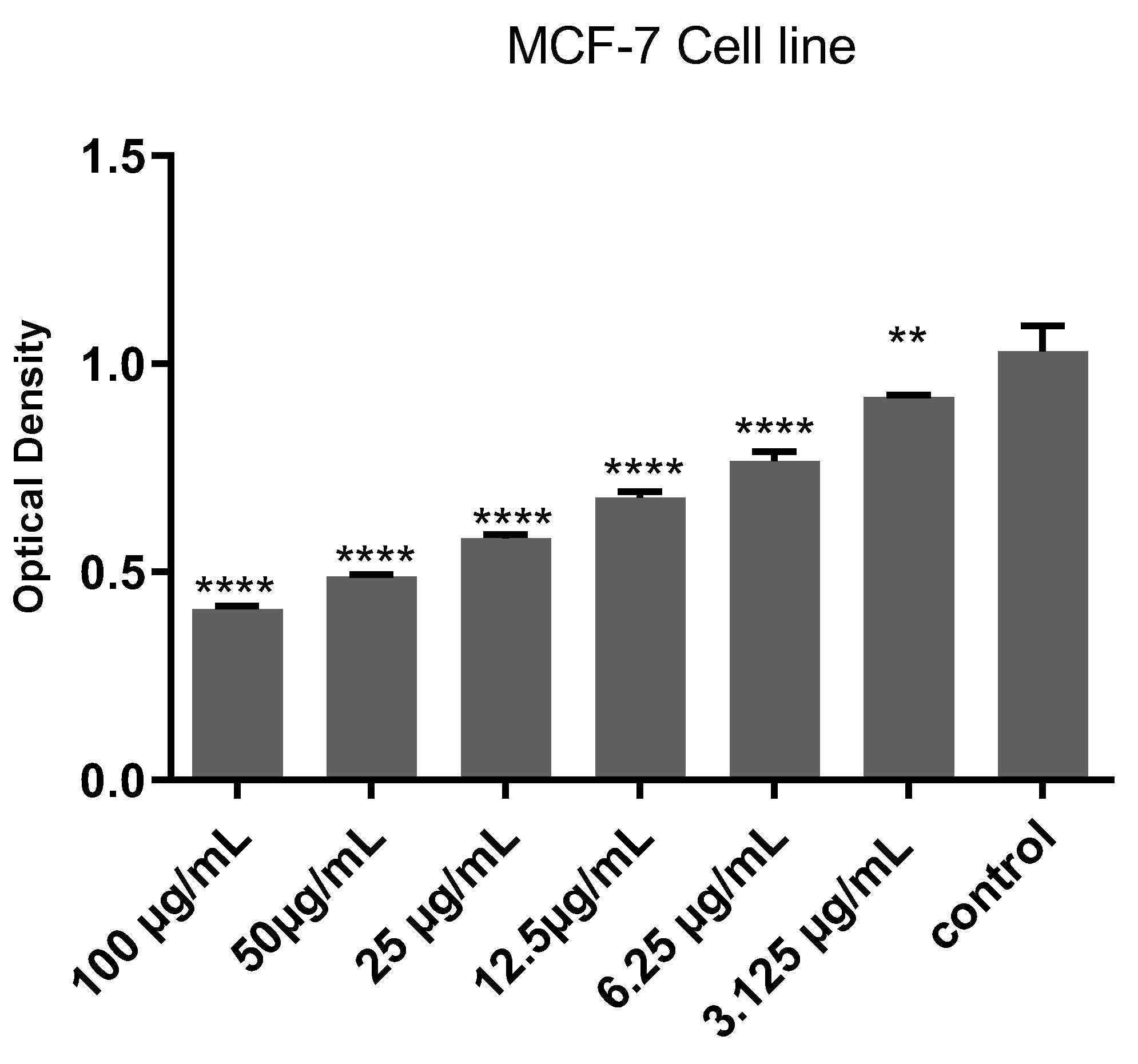
2.2. Cytotoxicity Evaluation
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extraction Procedure
4.3. GC/MS of Olea Europaea Exctract
4.4. Cytotoxicity Assay
4.5. MTT Assay
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pessoa, H. R.; Zago, L.; Difonzo, G.; Pasqualone, A.; Caponio, F.; Ferraz da Costa, D. C. Olive Leaves as a Source of Anticancer Compounds: In Vitro Evidence and Mechanisms. Molecules (Basel, Switzerland), 2024,29(17), 4249. [CrossRef]
- Hussain, S.Z.; Naseer, B.; Qadri, T.; Fatima, T.; Bhat, T.A. Olive (Olea europaea L.)—Morphology, Taxonomy, Composition and Health Benefits. In: Fruits Grown in Highland Regions of the Himalayas. Springer, Cham, 2021. [CrossRef]
- Shafiq, M.; Iqbal, M. Z.; Athar, M. The beneficial effects of olive tree (Olea europaea L.) in the nutritional, pharmaceutical and industrial application: a review. J. Plant Develop. 2024,31: 247-266. [CrossRef]
- Bavaro, A.R.; Tarantini, A.; Bruno, A.; Logrieco, A.F.; Gallo, A.; Mita, G.; Valerio, F.; Bleve, G.; Cardinali, A. Functional foods in Mediterranean diet: exploring the functional features of vegetable case-studies obtained also by biotechnological approaches. Aging Clin Exp Res. 2024 Oct 16;36(1):208. [CrossRef]
- Magyari-Pavel, I. Z.; Moacă, E.-A.; Avram, Ș.; Diaconeasa, Z.; Haidu, D.; Ștefănuț, M. N.; Rostas, A. M.; Muntean, D.; Bora, L.; Badescu, B.; Iuhas, C.; Dehelean, C. A.; Danciu, C. Antioxidant Extracts from Greek and Spanish Olive Leaves: Antimicrobial, Anticancer and Antiangiogenic Effects. Antioxidants, 2024,13(7), 774. [CrossRef]
- Rishmawi, S.; Haddad, F.; Dokmak, G.; Karaman, R. A Comprehensive Review on the Anti-Cancer Effects of Oleuropein. Life (Basel, Switzerland), 2022,12(8), 1140. [CrossRef]
- Antoniou, C.; Hull, J. The Anti-cancer Effect of Olea europaea L. Products: a Review. Curr. Nutr. Rep., 2021,10(1), 99–124. [CrossRef]
- Maleki Dana, P.; Sadoughi, F.; Asemi, Z. Yousefi, B. The role of polyphenols in overcoming cancer drug resistance: a comprehensive review. Cell Mol Biol Lett. 2022 Jan 3;27(1):1. [CrossRef]
- Albogami, S.; Hassan, A. M. Assessment of the Efficacy of Olive Leaf (Olea europaea L.) Extracts in the Treatment of Colorectal Cancer and Prostate Cancer Using In Vitro Cell Models. Molecules (Basel, Switzerland), 2021,26(13), 4069. [CrossRef]
- Uğuz, A. C.; Rocha-Pimienta, J.; Martillanes, S.; Garrido, M.; Espino, J.; Delgado-Adámez, J. Chlorophyll Pigments of Olive Leaves and Green Tea Extracts Differentially Affect Their Antioxidant and Anticancer Properties. Molecules (Basel, Switzerland), 2023,28(6), 2779. [CrossRef]
- Shah, Z.A.; Mujawah, A.A.H.; Ullah, I.; Rauf, A.; Rashid, U.; Khalil, A.A.; Shah, S.M.M.; Pervaiz, A.; Shaheen, F.; Al-Awthan, Y.S.; Qureshi, M.N.; Al-Duais, M.A.; Bahattab, O.; Almarhoon, Z.M.; Mabkhot, Y.N.; Mubarak, M.S. Antioxidant and Cytotoxic Activity of a New Ferruginan A from Olea ferruginea: In Vitro and In Silico Studies. Oxid Med Cell Longev. 2022 Jan 20;2022:8519250. [CrossRef]
- De Cicco, P.; Ercolano, G.; Tenore, G. C.; Ianaro, A. Olive leaf extract inhibits metastatic melanoma spread through suppression of epithelial to mesenchymal transition. Phytother Res, 2022,36(10), 4002–4013. [CrossRef]
- Spagnuolo, C.; Moccia, S.; Tedesco, I.; Crescente, G.; Volpe, M. G.; Russo, M.; Russo, G. L. Phenolic Extract from Extra Virgin Olive Oil Induces Different Anti-Proliferative Pathways in Human Bladder Cancer Cell Lines. Nutrients, 2022,15(1), 182. [CrossRef]
- Wed Fathi Ibrahim, Ahmed Chaloob Saddam, Terezie Tolar-Peterson. Study of Chemical Composition and Phytochemical Compounds Of Local Olive (Olea Europaea L.) Leaves. Iraqi J. Agric. Sci., 2025,56(4), 1482-1491. [CrossRef]
- Zaïri, A.; Nouir, S.; Zarrouk, A.; Haddad, H.; Khelifa, A.; Achour, L. Phytochemical profile, cytotoxic, antioxidant, and allelopathic potentials of aqueous leaf extracts of Olea europaea. Food Science & Nutrition, 2020,8(9), 4805-4813. [CrossRef]
- Ahmad, R.; Alqathama, A.; Alam, M.M.; Riza M.; AbdallaA. N.; Aldholmi M.; Al− Said, H.M.; Aljishi, F.S.; Althomali, E.H.; Alabdullah, M.M.; Altaweel, N.H.; Almubarak, A.F.; Asghar, S.S.. Biological quality and phytochemical profiling of olive fruits using gas chromatography–mass spectrometry (GCMS) analysis. Chem. Biol. Technol. Agric. 2023,10, 45. [CrossRef]
- Büker, E.; Kiran, F.; Taliboglu, S.; Casoni, D.; Ipekel, A. In Vitro Evaluation of Olive Leaf (Olea europaea L.) Extract as a Functional Food Component in Combination with Chemotherapeutics in MCF-7 Breast Cancer Cells. Pharmaceuticals (Basel, Switzerland), 2025,18(7), 965. [CrossRef]
- Jaafar, N.S.; Alshammaa, D.A.; Abdul-lalil, T.Z.; Ibrahem, N.M. Quantitative Determination and Cytotoxic Effect of Oleanolic Acid from Olea europaea Leaves Extract Cultivated in Iraq. Iraqi J. Pharm. Sci. 2022,31(2), 244-250. [CrossRef]
- Liu, L.; Ahn, K.S.; Shanmugam, M.K.; Wang, H.; Shen, H.; Arfuso, F.; Chinnathambi, A.; Alharbi, S.A.; Chang, Y.; Sethi, G.; Tang, F.R. Oleuropein induces apoptosis via abrogating NF-κB activation cascade in estrogen receptor–negative breast cancer cells. J. Cell. Biochem., 2019,120(3), 4504-4513. [CrossRef]
- Calahorra, J.; Martínez-Lara, E.; Granadino-Roldán, J.M.; Cañuelo, A.; Blanco, S.; Oliver, F.J.; Siles, E. Crosstalk between hydroxytyrosol, a major olive oil phenol, and HIF-1 in MCF-7 breast cancer cells. Sci Rep. 2020 Apr 14;10(1):6361. [CrossRef]
- Alharbi, R.M.; Gomah, N.H.; Ragab, W.S.M.; Awad, N.S.; Abdein, M.A. Antiproliferative Activity of Antioxidants-Rich Extracts from Olea europaea L. Leaves and Oil Against Six Cancerous Human Cell Lines. Int. J. Fruit Sci., 2024,24(1), 349–362. [CrossRef]
- Soule, H.D.; Vazguez, J.; Long, A.; Albert, S.; Brennan, M. A human cell line from a pleural effusion derived from a breast carcinoma. J Natl Cancer Inst. 1973 Nov;51(5):1409-16. [CrossRef]
- Salman, M.I.; Al-Shammari, A.M.; Emran, M.A. 3-Dimensional coculture of breast cancer cell lines with adipose tissue-Derived stem cells reveals the efficiency of oncolytic Newcastle disease virus infection via labeling technology. Front. Mol. Biosci. 2022,9, 754100. [CrossRef]
- Al-Shammari, A. M.; Salman, M. I. Antimetastatic and antitumor activities of oncolytic NDV AMHA1 in a 3D culture model of breast cancer. Front. Mol. Biosci., 2024,11, 1331369. [CrossRef]
- Salman, M.I.; Emran, M.A.; Al-Shammari, A.M. Spheroid-formation 3D engineering model assay for in vitro assessment and expansion of cancer cells. In Proceedings of the AIP Conference Proceedings, Yogyakarta, Indonesia, 10–11 November 2021; AIP Publishing: Melville, NY, USA, 2021; Volume 2372, No. 1 . [CrossRef]
- Freshney, R. Ian. Culture of animal cells: a manual of basic technique and specialized applications. John Wiley & Sons, 2015. [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



