Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
Background
Methods
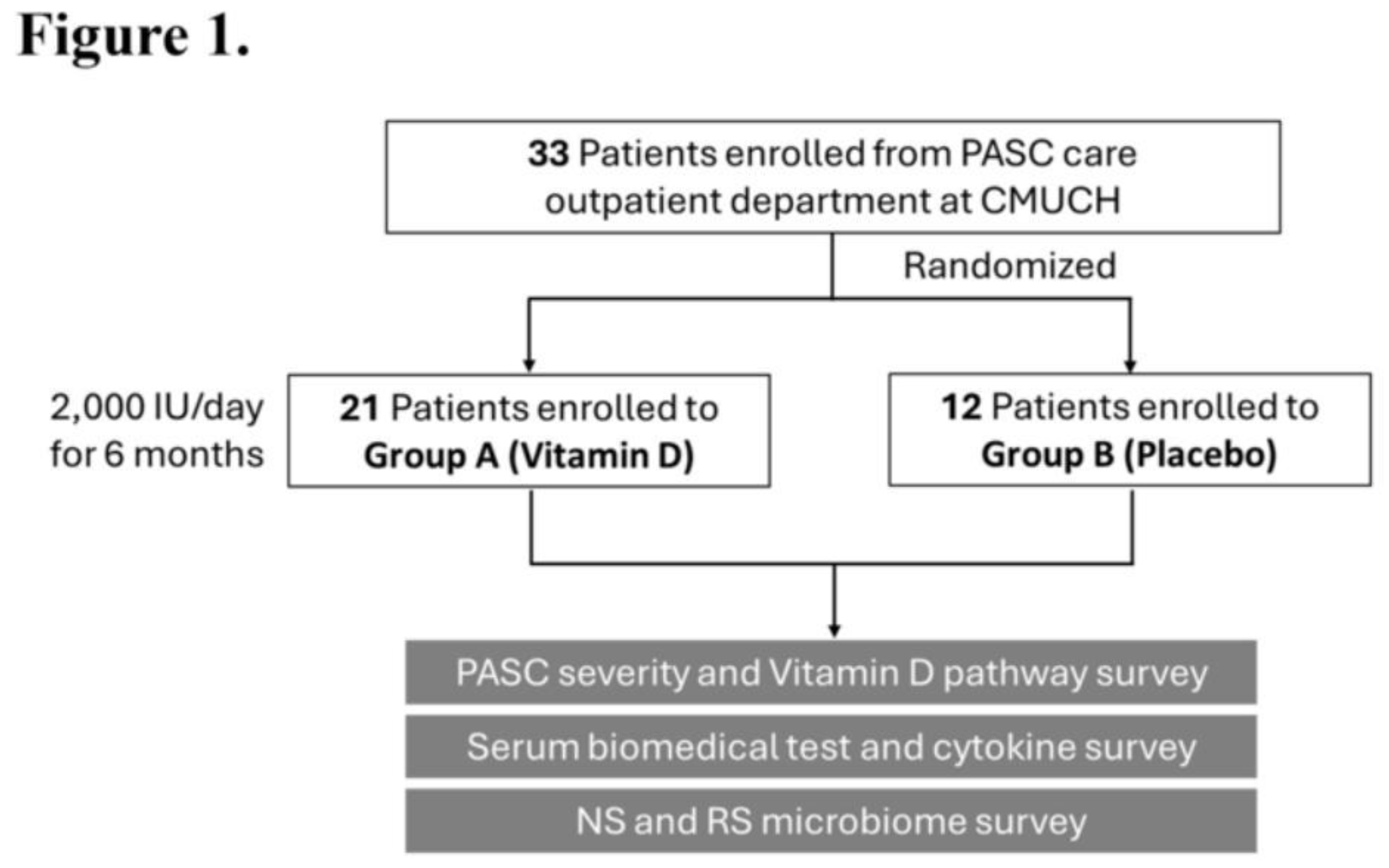
Study Design and Participant Recruitment
Clinical Assessments
Sample Collection and Processing
16S rRNA Gene Sequencing
Statistical Analysis
Results
Participant Characteristics
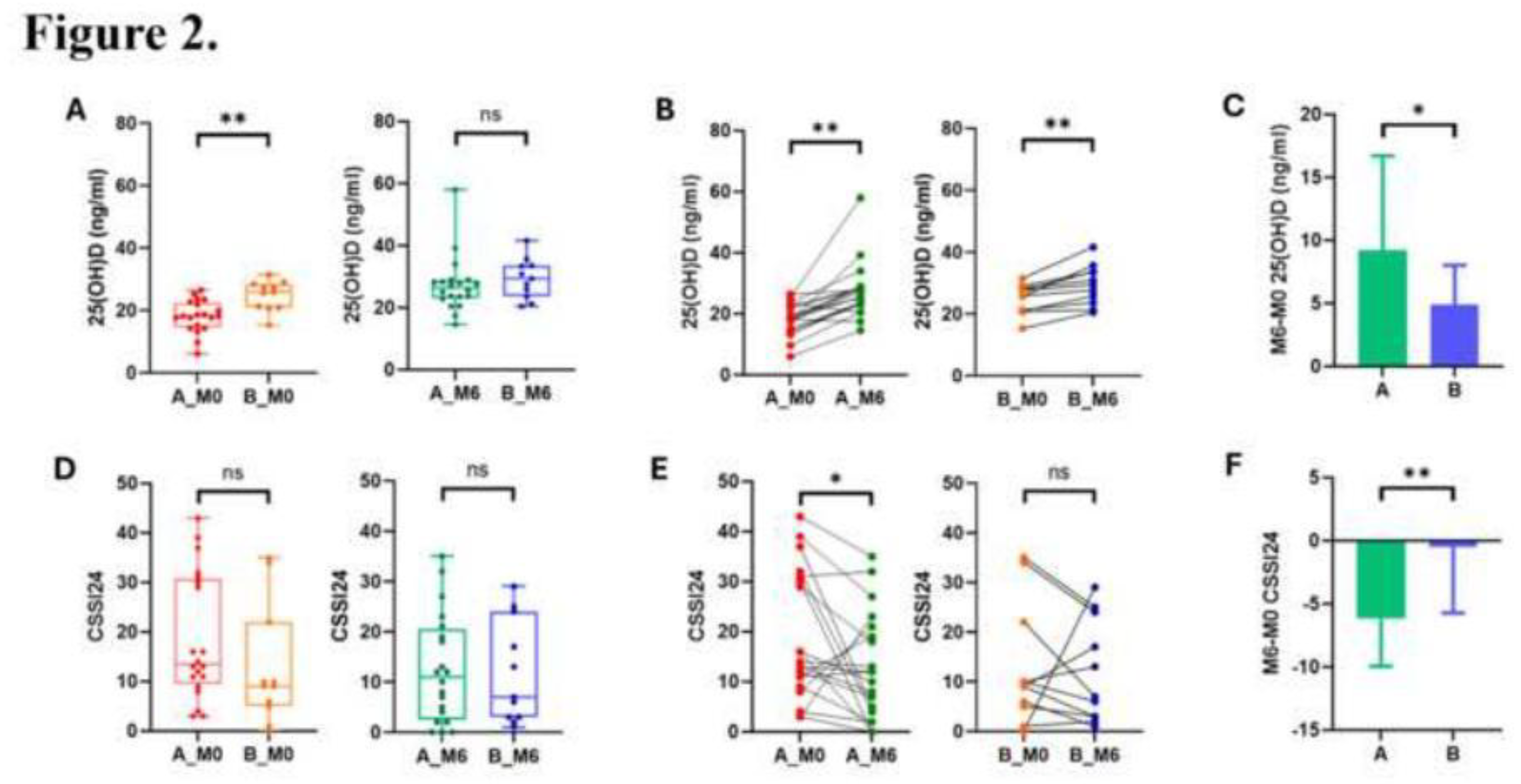
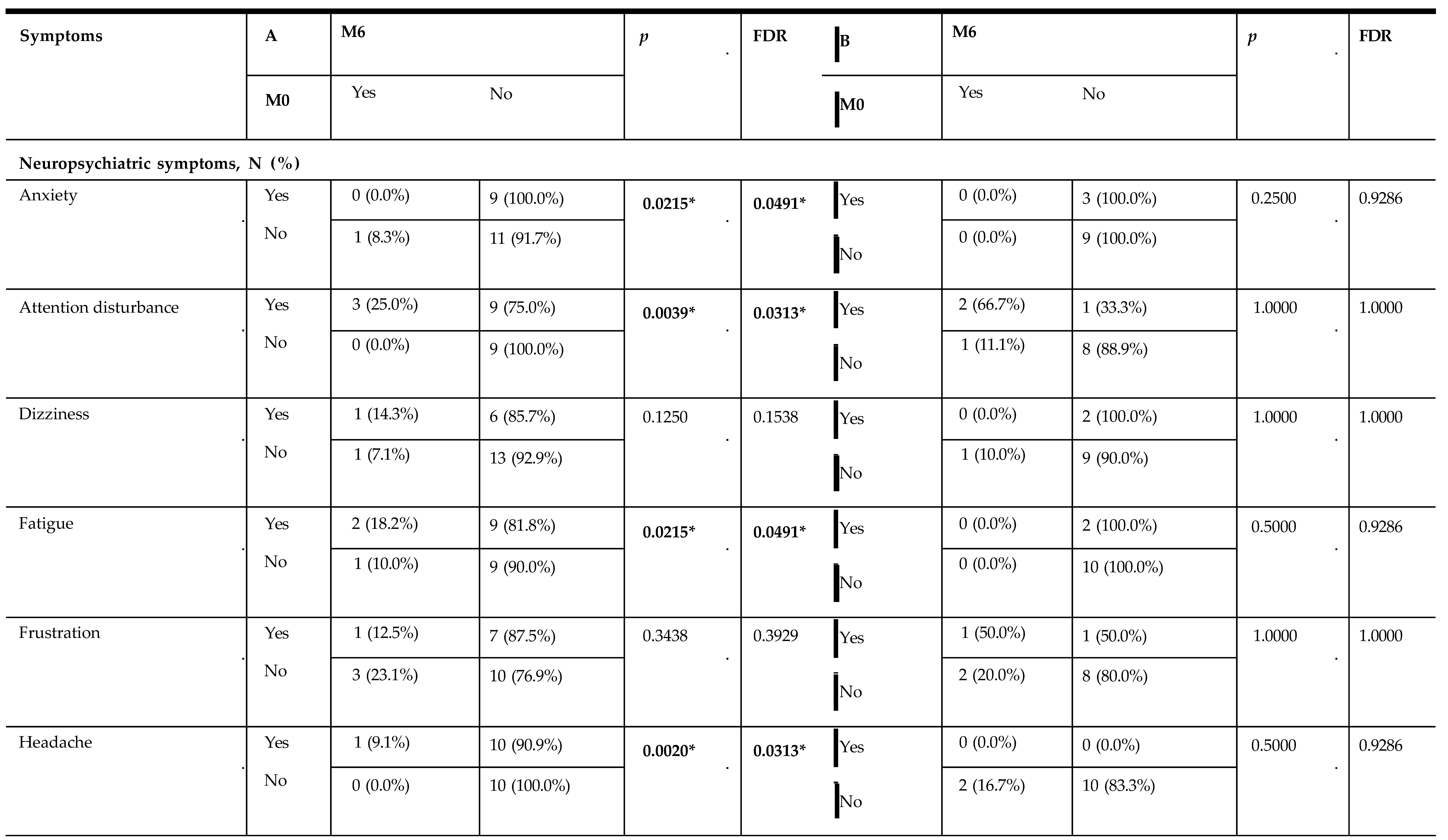
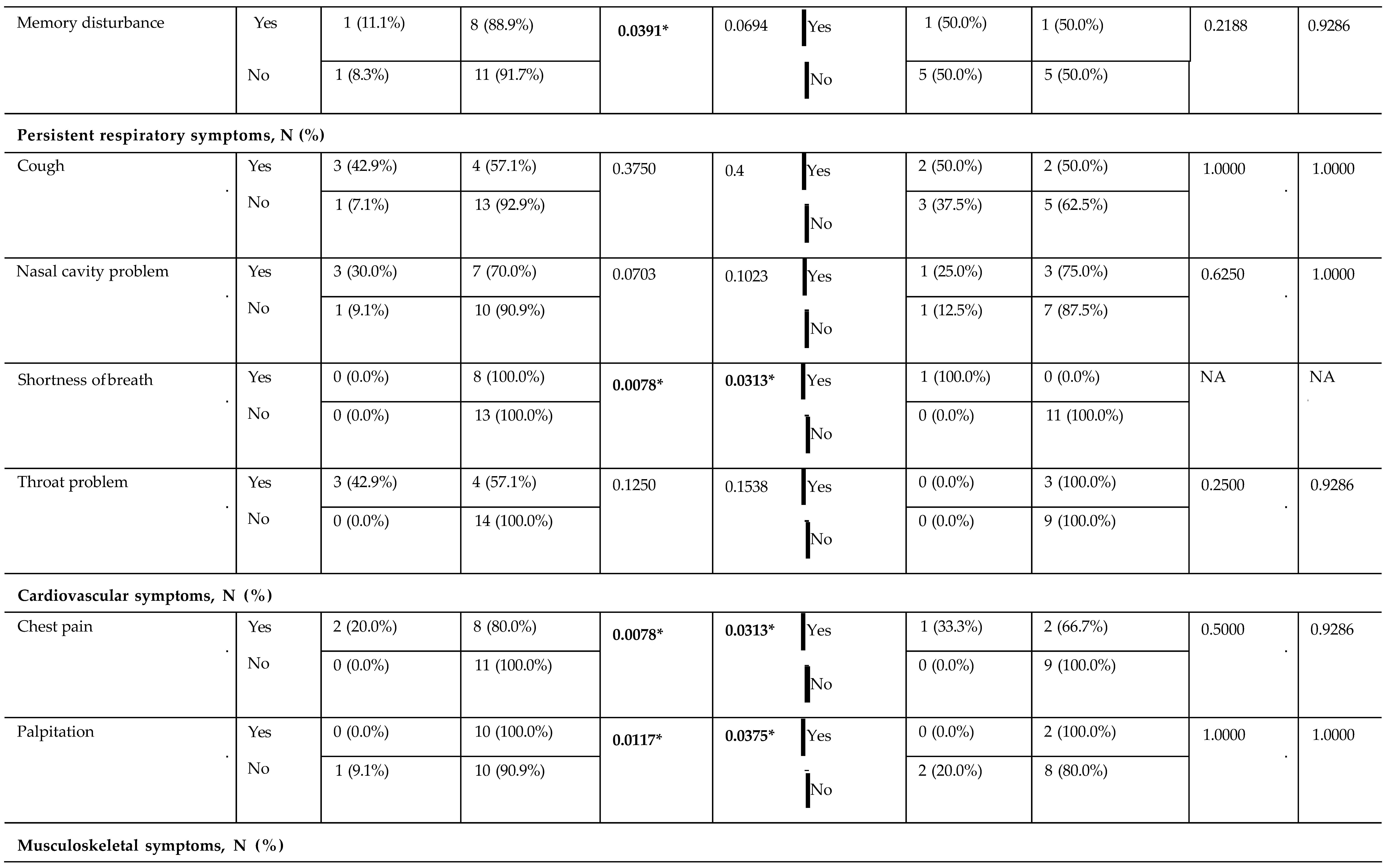
Impact of Vitamin D3 Supplementation on Serum 25(OH)D Levels and Symptom
Severity
Nasal and Rectal Microbiota Diversity and Composition
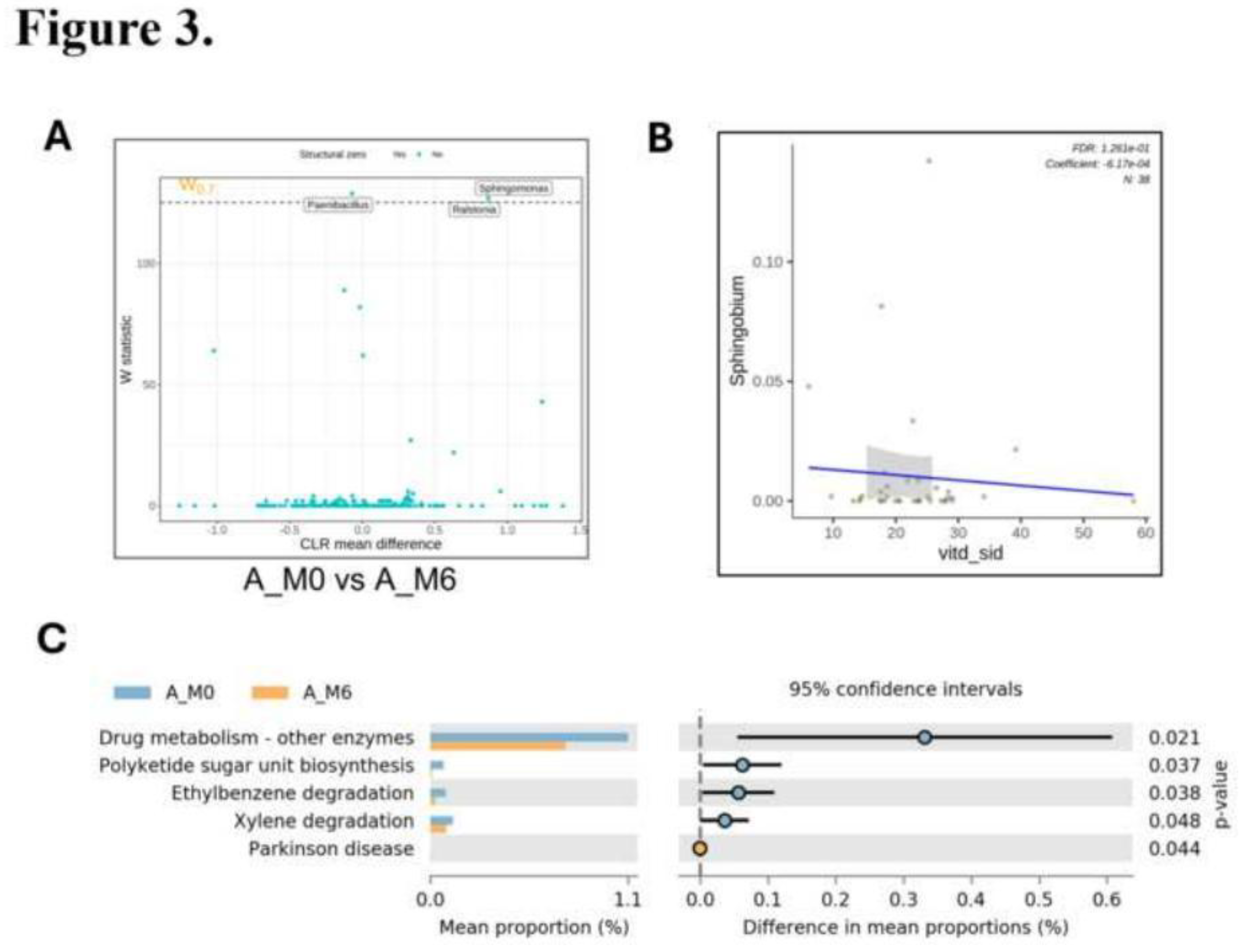
Differential Abundance and Functional Changes in Nasal Microbiota
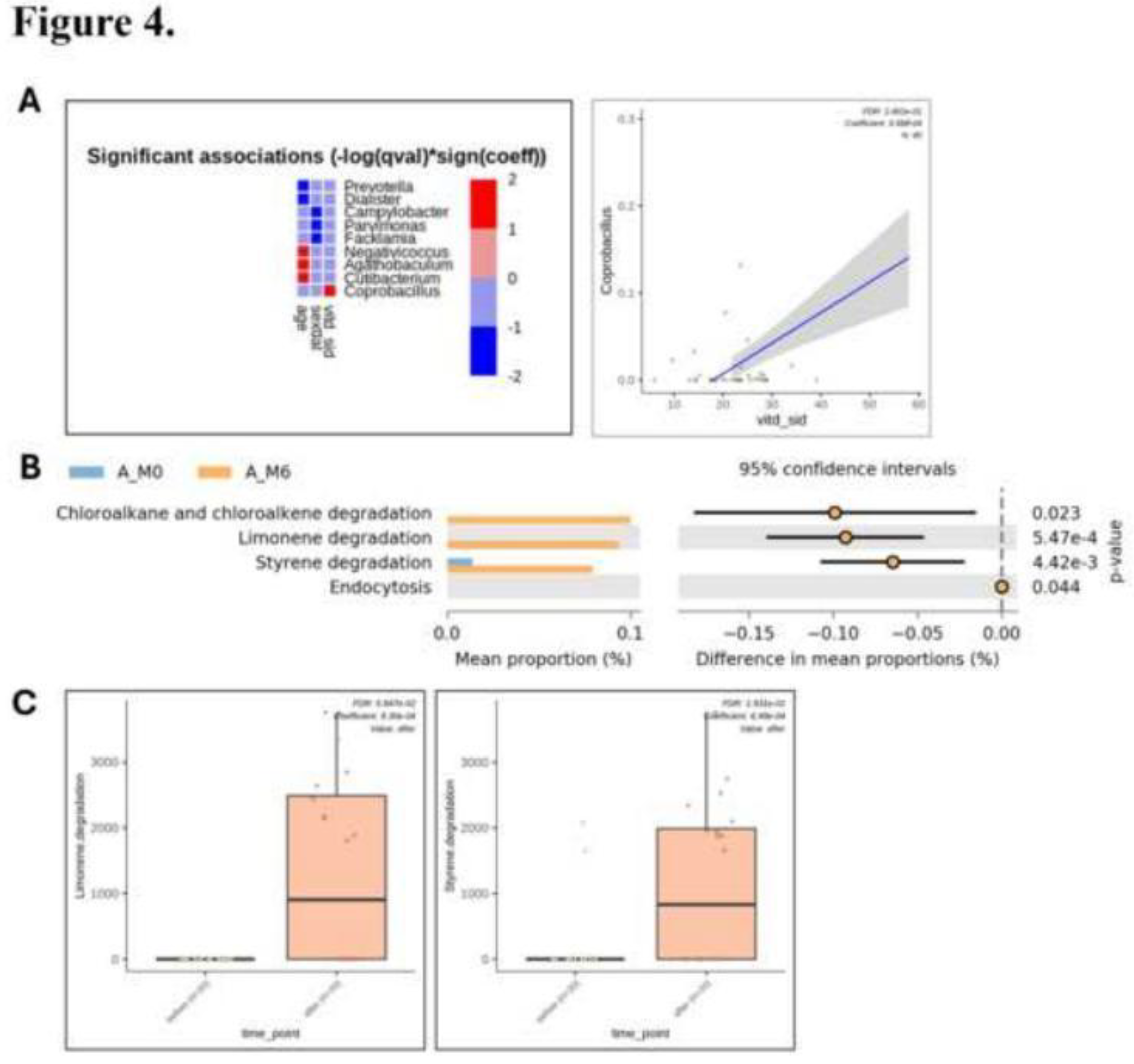
Differential Abundance and Functional Changes in Rectal Microbiota
Immunomodulatory Outcomes
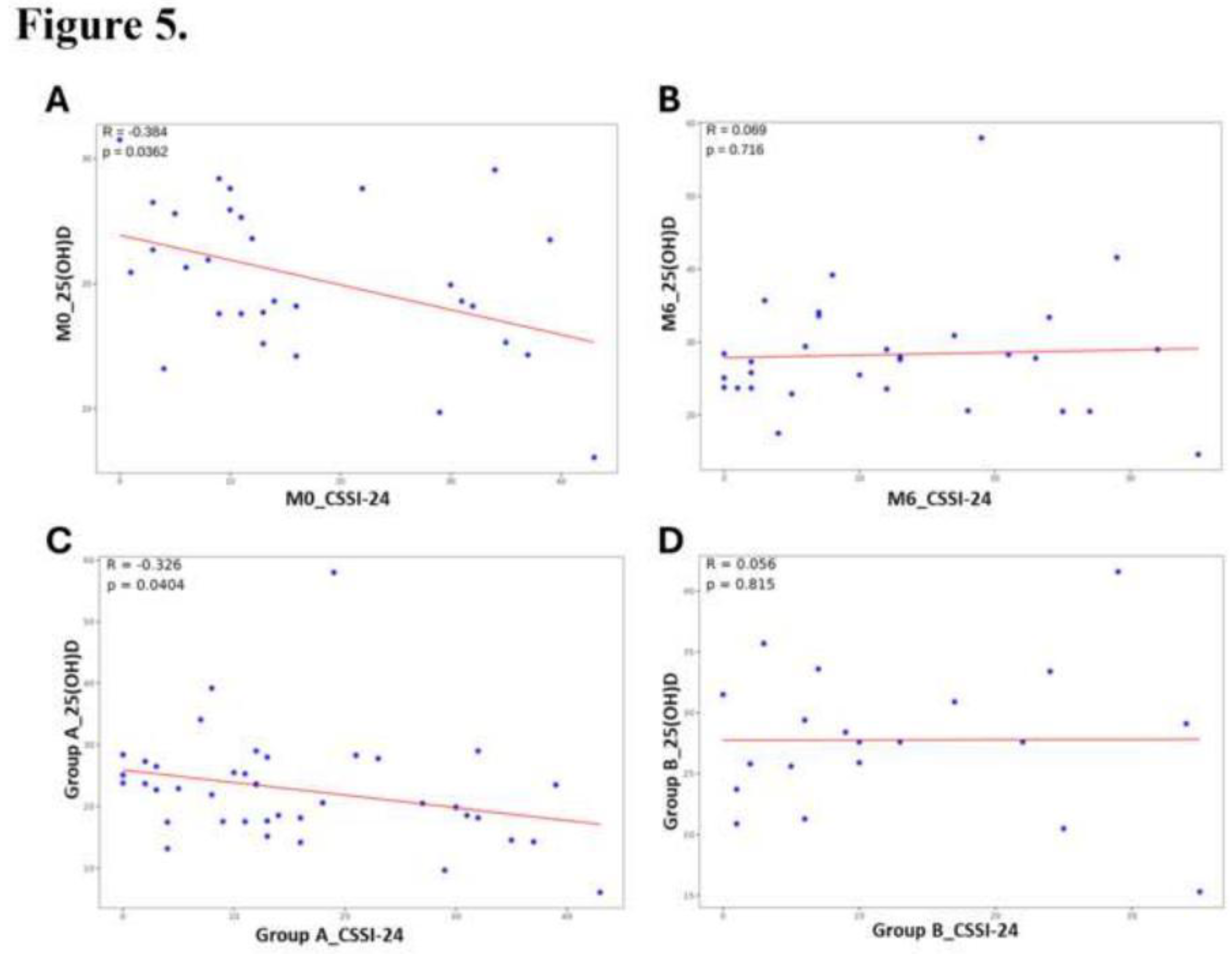
Influence of Serum 25(OH)D on Symptom Burden
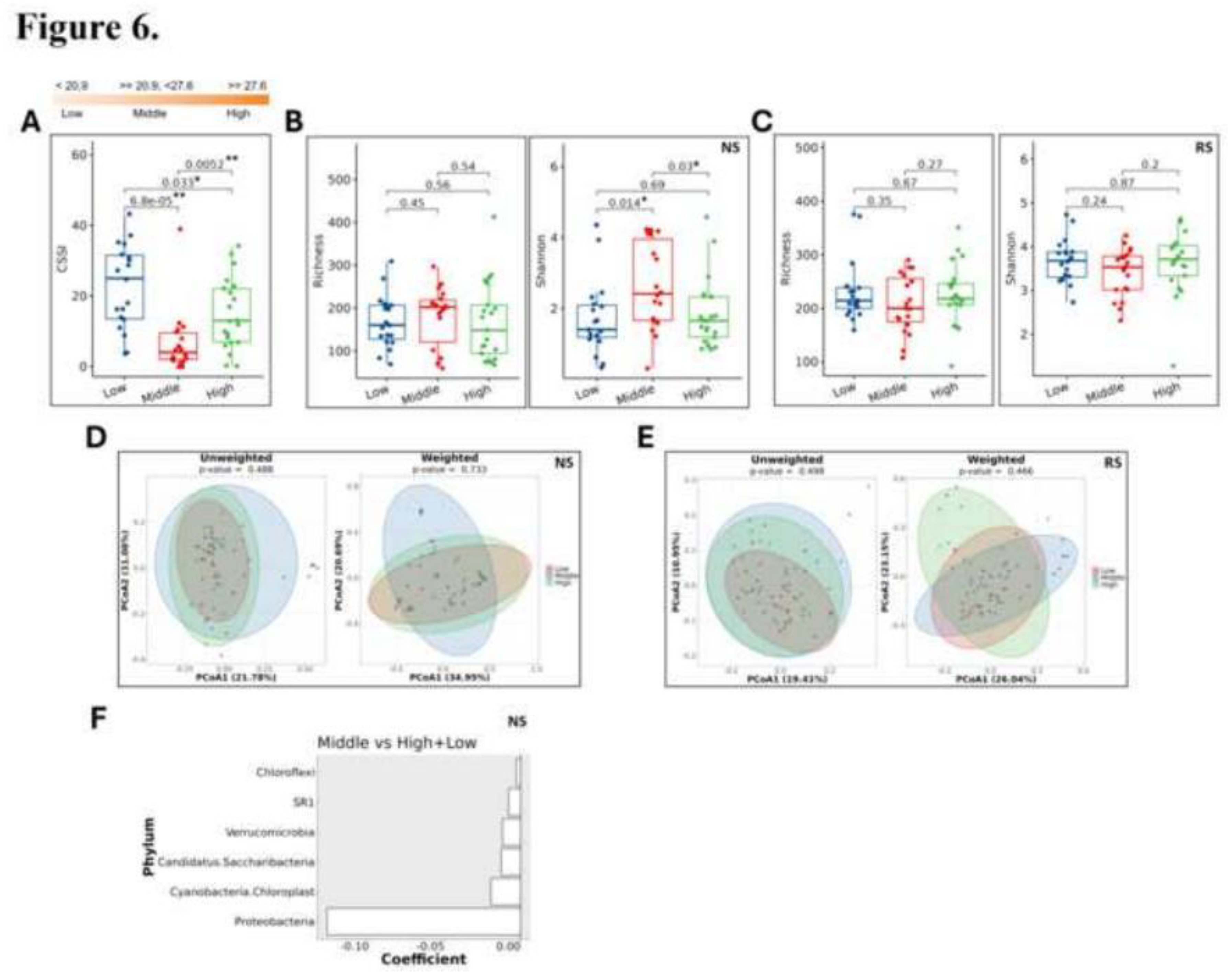
Serum 25(OH)D Categories and Microbial Composition
Discussion
Supplementary Materials
Funding
Authors’ contributions
Availability of data and materials
Ethical Approval and Consent to participate
Competing interests
Abbreviations
| 25(OH)D | 25-hydroxyvitamin D |
| CSSI-24 | Children’s Somatic Symptom Inventory-24, |
| FDR | false discovery rate |
| MCT | medium-chain triglyceride |
| NS | nasal swab |
| PASC | Post-acute sequelae ofCOVID-19 |
| RCTs | randomized controlled trials |
| RS | rectal swab |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
References
- Phetsouphanh, C; Darley, DR; Wilson, DB; Howe, A; Munier, CML; Patel, SK; et al. Immunological dysfunction persists for 8 months following initial mild-to-moderate SARS-CoV-2 infection. Nat Immunol;PubMed 2022, 23(2), 210–6. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, T; Shafran, R; Ladhani, SN. Long COVID in children and adolescents. Curr Opin Infect Dis. 2022, 35(5), 461–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Davis, HE; Assaf, GS; McCorkell, L; Wei, H; Low, RJ; Re'em, Y; et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine 2021, 38, 101019. [Google Scholar] [CrossRef] [PubMed Central]
- Meo MOS, AlHusseini N, Ibrahim DI, Meo MZS, Ahsan F, Tamim H, et al. Clinical manifestations of Long-COVID: an observational perspective. Front Med (Lausanne). 2025;12:1523817. PubMed PMID: 40177288. Pubmed Central PMCID: PMC11961443. Epub 20250319. eng.
- Holick MF. Vitamin D deficiency. N Engl J Med. 2007 Jul 19;357(3):266-81. PubMed PMID: 17634462. eng. 3).
- Martineau, AR; Jolliffe, DA; Hooper, RL; Greenberg, L; Aloia, JF; Bergman, P; et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. Bmj 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed Central]
- Martens PJ, Gysemans C, Verstuyf A, Mathieu AC. Vitamin D's Effect on Immune Function. Nutrients. 2020 Apr 28;12(5). PubMed PMID: 32353972. Pubmed Central PMCID: PMC7281985. Epub 20200428. eng.
- Cantorna, MT; McDaniel, K; Bora, S; Chen, J; James, J. Vitamin D, immune regulation, the microbiota, and inflammatory bowel disease. Exp Biol Med (Maywood) 2014, 239(11), 1524–30. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ooi, JH; Chen, J; Cantorna, MT. Vitamin D regulation of immune function in the gut: why do T cells have vitamin D receptors? Mol Aspects Med 2012, 33(1), 77–82. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bellerba F, Muzio V, Gnagnarella P, Facciotti F, Chiocca S, Bossi P, et al. The Association between Vitamin D and Gut Microbiota: A Systematic Review of Human Studies. Nutrients. 2021 Sep 26;13(10). PubMed PMID: 34684379. Pubmed Central PMCID: PMC8540279. Epub 20210926. eng.
- Raj, ST; Bruce, AW; Anbalagan, M; Srinivasan, H; Chinnappan, S; Rajagopal, M; et al. COVID-19 influenced gut dysbiosis, post-acute sequelae, immune regulation, and therapeutic regimens. Front Cell Infect Microbiol. 2024, 14, 1384939. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liu, Q; Mak, JWY; Su, Q; Yeoh, YK; Lui, GC; Ng, SSS; et al. Gut microbiota dynamics in a prospective cohort of patients with post-acute COVID-19 syndrome. Gut;PubMed 2022, 71(3), 544–52. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, A; Sehgal, K; Gupta, A; Madhavan, MV; McGroder, C; Stevens, JS; et al. Post-acute COVID-19 syndrome. Nat Med 2021, 27(4), 601–15. [Google Scholar] [CrossRef] [PubMed Central]
- Yong, SJ. Long COVID or post-COVID-19 syndrome: putative pathophysiology, risk factors, and treatments. Infect Dis (Lond) 2021, 53(10), 737–54. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Entrenas Castillo M, Entrenas Costa LM, Vaquero Barrios JM, Alcalá Díaz JF, López Miranda J, Bouillon R, et al. "Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study". J Steroid Biochem Mol Biol. 2020 Oct;203:105751. PubMed PMID: 32871238. Pubmed Central PMCID: PMC7456194. Epub 20200829. eng.
- Quesada-Gomez, JM; Entrenas-Castillo, M; Bouillon, R. Vitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with coronavirus SARS-CoV-2 infections: Revised Ms SBMB 2020_166. J Steroid Biochem Mol Biol. 2020, 202, 105719. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Baktash, V; Hosack, T; Patel, N; Shah, S; Kandiah, P; Van den Abbeele, K; et al. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med J 2021, 97(1149), 442–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kaufman, HW; Niles, JK; Kroll, MH; Bi, C; Holick, MF. SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PLoS One PubMed. 2020, 15(9), e0239252. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mercola, J; Grant, WB; Wagner, CL. Evidence Regarding Vitamin D and Risk of COVID-19 and Its Severity. Nutrients;PubMed 2020, 12(11). [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Singh P, Rawat A, Alwakeel M, Sharif E, Al Khodor S. The potential role of vitamin D supplementation as a gut microbiota modifier in healthy individuals. Sci Rep. 2020 Dec 10;10(1):21641. PubMed PMID: 33303854. Pubmed Central PMCID: PMC7729960. Epub 20201210. eng.
- Akimbekov, NS; Digel, I; Sherelkhan, DK; Lutfor, AB; Razzaque, MS. Vitamin D and the Host-Gut Microbiome: A Brief Overview. Acta Histochem Cytochem PubMed. 2020, 53(3), 33–42. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kenny G, Townsend L, Savinelli S, Mallon PWG. Long COVID: Clinical characteristics, proposed pathogenesis and potential therapeutic targets. Front Mol Biosci. 2023;10:1157651. PubMed PMID: 37179568. Pubmed Central PMCID: PMC10171433. Epub 20230426. eng.
- Giannos, P; Prokopidis, K. Gut dysbiosis and long COVID-19: Feeling gutted. J Med Virol. 2022, 94(7), 2917–8. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Álvarez-Santacruz, C; Tyrkalska, SD; Candel, S. The Microbiota in Long COVID. Int J Mol Sci.;PubMed 2024, 25(2). [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hsu, YL; Chen, PC; Tsai, YF; Wei, CH; Wu, LS; Hsieh, KS; et al. Clinical Features and Vaccination Effects among Children with Post-Acute Sequelae of COVID-19 in Taiwan. Vaccines (Basel);PubMed 2024, 12(8). [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Walker, LS; Beck, JE; Garber, J; Lambert, W. Children's Somatization Inventory: psychometric properties of the revised form (CSI-24). J Pediatr Psychol.;PubMed 2009, 34(4), 430–40. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pichler, M; Coskun, Ö K; Ortega-Arbulú, AS; Conci, N; Wörheide, G; Vargas, S; et al. A 16S rRNA gene sequencing and analysis protocol for the Illumina MiniSeq platform. Microbiologyopen 2018, 7(6), e00611. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rao, S; Gross, RS; Mohandas, S; Stein, CR; Case, A; Dreyer, B; et al. Postacute Sequelae of SARS-CoV-2 in Children. In Pediatrics;PubMed; Pubmed Central, 1 Mar 2024; 3, p. 153. [Google Scholar] [PubMed] [PubMed Central]
- Guo, C; Yi, B; Wu, J; Lu, J. The microbiome in post-acute infection syndrome (PAIS). Comput Struct Biotechnol J 2023, 21, 3904–11. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wykes MN, Lewin SR. Immune checkpoint blockade in infectious diseases. Nat Rev Immunol. 2018 Feb;18(2):91-104. PubMed PMID: 28990586. Pubmed Central PMCID: PMC5991909. Epub 20171009. eng.
- Cătălina, GR; Gheorman, V; Gheorman, V; Forțofoiu, MC. The Role of Neuroinflammation in the Comorbidity of Psychiatric Disorders and Internal Diseases. In Healthcare (Basel);PubMed; Pubmed Central, 7 Apr 2025; 7, p. 13. [Google Scholar] [PubMed] [PubMed Central]
- Durack, J; Lynch, SV; Nariya, S; Bhakta, NR; Beigelman, A; Castro, M; et al. Features of the bronchial bacterial microbiome associated with atopy, asthma, and responsiveness to inhaled corticosteroid treatment. J Allergy Clin Immunol. 2017, 140(1), 63–75. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Torres-Sánchez, A; Ruiz-Rodríguez, A; Ortiz, P; Moreno, MA; Ampatzoglou, A; Gruszecka-Kosowska, A; et al. Exploring Next Generation Probiotics for Metabolic and Microbiota Dysbiosis Linked to Xenobiotic Exposure: Holistic Approach. Int J Mol Sci;PubMed 2022, 23(21). [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Louis, P; Flint, HJ. Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 2017, 19(1), 29–41. [Google Scholar] [CrossRef] [PubMed]
- Ullah, H. Gut-vitamin D interplay: key to mitigating immunosenescence and promoting healthy ageing. Immun Ageing 2025, 22(1), 20. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Demographic characteristics | ||
| Age | A | B |
| mean ± SD (N) | 9±3.36 (21) | 9±3.53 (12) |
| Sex, N (%) | ||
| Female | 9 (42.86%) | 5 (41.67%) |
| Male | 12 (57.14%) | 7 (58.33%) |
| Vaccination, N (%) | 17 (80.95%) | 10 (83.33%) |
 |
 |
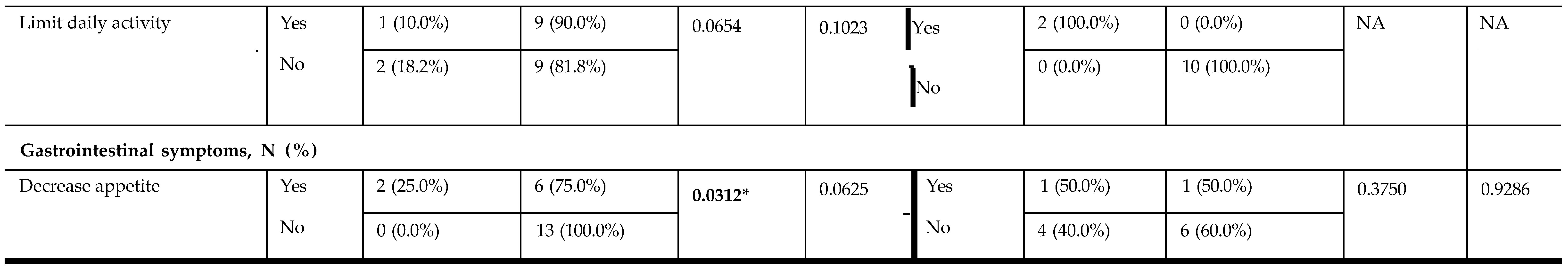 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




