Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
MSC: 65M60; 26A33; 65M12; 65R20; 35R11; 92C10
1. Introduction
1.1. Biomedical Motivation
1.2. Mathematical Background
1.3. Related Work
1.4. Contributions and Novelty
- the development of a fully discrete implicit numerical scheme for time–space fractional diffusion equations with heterogeneous coefficients;
- the proof of unconditional stability with respect to the time step, fractional order , and spatial discretisation parameter h;
- a reduction of the memory complexity of the classical L1 scheme from to via a sum-of-exponentials approximation;
- numerical validation of the method in heterogeneous media with discontinuous diffusion coefficients, relevant to biological tissue modelling.
2. Mathematical Model of Anomalous Diffusion in Biological Tissues
2.1. Governing Equation
2.2. Biological Interpretation
2.3. Initial and Boundary Conditions
3. Functional Setting and Analytical Properties
3.1. Fractional Sobolev Spaces
3.2. Well-Posedness
4. Numerical Method
4.1. Temporal Discretisation
4.2. Spatial Discretisation
4.3. Fully Discrete Scheme
4.4. Computational Complexity
4.5. Stability of the Fully Discrete Scheme
4.6. Error Analysis
4.6.1. Temporal Discretisation Error
4.6.2. Spatial Discretisation Error
4.6.3. Fully Discrete Error with SOE Approximation
5. Numerical Experiments
5.1. Temporal Convergence
5.2. Spatial Convergence
5.3. Efficiency and Memory Reduction
5.4. Heterogeneous Diffusion
6. Conclusion
Acknowledgments
Conflicts of Interest
References
- Crank, J. The Mathematics of Diffusion, 2 ed.; Oxford University Press, 1975. [Google Scholar]
- Carslaw, H.S.; Jaeger, J.C. Conduction of Heat in Solids, 2 ed.; Oxford University Press, 1959. [Google Scholar]
- Metzler, R.; Klafter, J. The random walk’s guide to anomalous diffusion: a fractional dynamics approach. Physics Reports 2000, 339, 1–77. [Google Scholar] [CrossRef]
- Sokolov, I.M. Models of anomalous diffusion in crowded environments. Soft Matter 2012, 8, 9043–9052. [Google Scholar] [CrossRef]
- Weiss, M.; Elsner, M.; Kartberg, F.; Nilsson, T. Anomalous subdiffusion is a measure for cytoplasmic crowding in living cells. Biophysical Journal 2004, 87, 3518–3524. [Google Scholar] [CrossRef] [PubMed]
- Golding, I.; Cox, E.C. Physical nature of bacterial cytoplasm. Physical Review Letters 2006, 96, 098102. [Google Scholar] [CrossRef] [PubMed]
- Metzler, R.; Klafter, J. The restaurant at the end of the random walk: recent developments in the description of anomalous transport by fractional dynamics. Journal of Physics A: Mathematical and General 2004, 37, R161–R208. [Google Scholar] [CrossRef]
- Podlubny, I. Fractional Differential Equations; Academic Press, 1999. [Google Scholar]
- Mainardi, F. Fractional Calculus and Waves in Linear Viscoelasticity; Imperial College Press, 2010. [Google Scholar]
- Magin, R.L. Fractional Calculus in Bioengineering; Begell House, 2010. [Google Scholar]
- Gorenflo, R.; Mainardi, F.; Moretti, D.; Pagnini, G.; Paradisi, P. Discrete random walk models for space–time fractional diffusion. Chemical Physics 2014, 284, 521–541. [Google Scholar] [CrossRef]
- Applebaum, D. Lévy Processes and Stochastic Calculus, 2 ed.; Cambridge University Press, 2009. [Google Scholar]
- Zoia, A.; Rosso, A.; Kardar, M. Fractional Laplacian in bounded domains. Physical Review E 2007, 76, 021116. [Google Scholar] [CrossRef] [PubMed]
- Meerschaert, M.M.; Sikorskii, A. Stochastic Models for Fractional Calculus; De Gruyter, 2012. [Google Scholar]
- Tarasov, V.E. Fractional Dynamics: Applications of Fractional Calculus to Dynamics of Particles, Fields and Media; Springer, 2011. [Google Scholar]
- Meerschaert, M.M.; Tadjeran, C. Finite difference approximations for fractional advection–dispersion flow equations. Journal of Computational and Applied Mathematics 2004, 172, 65–77. [Google Scholar] [CrossRef]
- Lin, Y.; Xu, C. Finite difference/spectral approximations for the time-fractional diffusion equation. Journal of Computational Physics 2007, 225, 1533–1552. [Google Scholar] [CrossRef]
- Ervin, V.J.; Roop, J.P. Variational formulation for the stationary fractional advection dispersion equation. Numerical Methods for Partial Differential Equations 2006, 22, 558–576. [Google Scholar] [CrossRef]
- Acosta, G.; Borthagaray, J.P. A fractional Laplace equation: regularity of solutions and finite element approximations. SIAM Journal on Numerical Analysis 2017, 55, 472–495. [Google Scholar] [CrossRef]
- Tian, X.; Zhou, Y. Analysis of a space fractional diffusion equation with variable coefficients. Numerische Mathematik 2015, 131, 123–147. [Google Scholar]
- Li, C.; Zeng, F. Numerical Methods for Fractional Calculus; Chapman and Hall/CRC, 2015. [Google Scholar]
- Jiang, S.; Zhang, J. Fast evaluation of the Caputo fractional derivative and its applications to fractional diffusion equations. Communications in Computational Physics 2017, 21, 650–678. [Google Scholar] [CrossRef]
- Liao, H.L.; McLean, W.; Zhang, J. A discrete Grönwall inequality with applications to numerical solutions of fractional differential equations. SIAM Journal on Numerical Analysis 2018, 56, 256–273. [Google Scholar]
- Di Nezza, E.; Palatucci, G.; Valdinoci, E. Hitchhiker’s guide to the fractional Sobolev spaces. Bulletin des Sciences Mathématiques 2012, 136, 521–573. [Google Scholar] [CrossRef]
- McLean, W. Strongly Elliptic Systems and Boundary Integral Equations; Cambridge University Press, 2010. [Google Scholar]
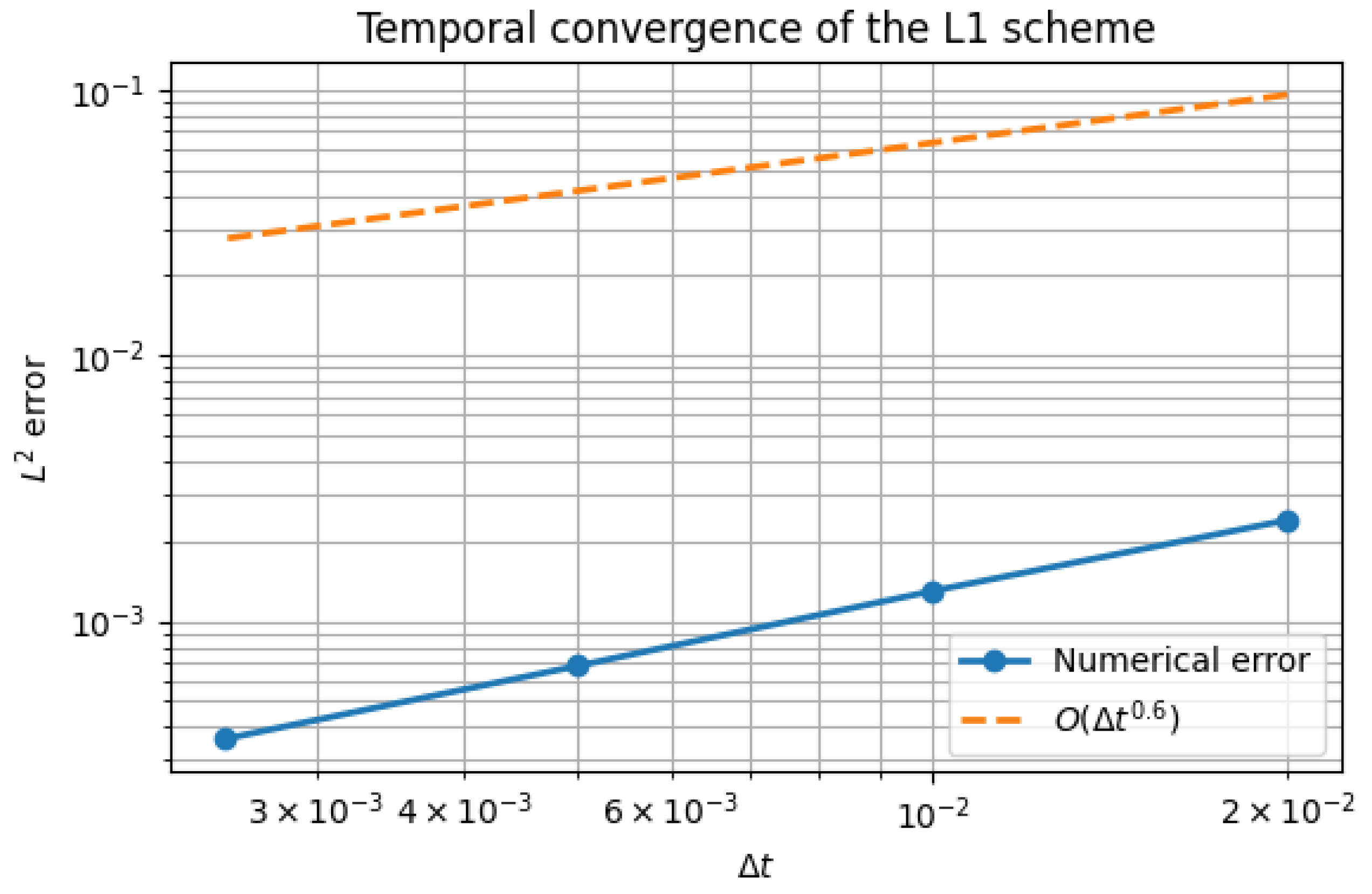
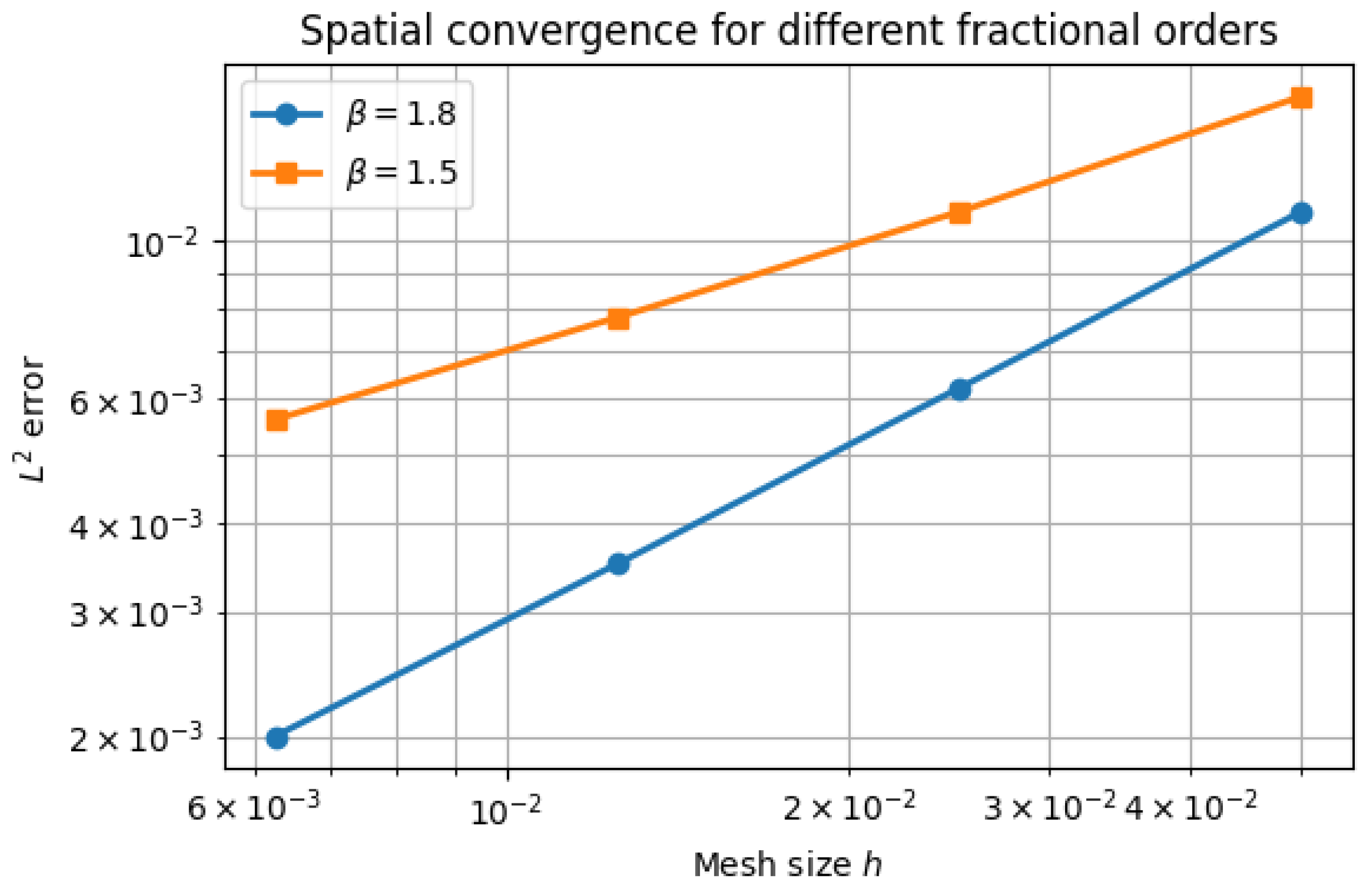
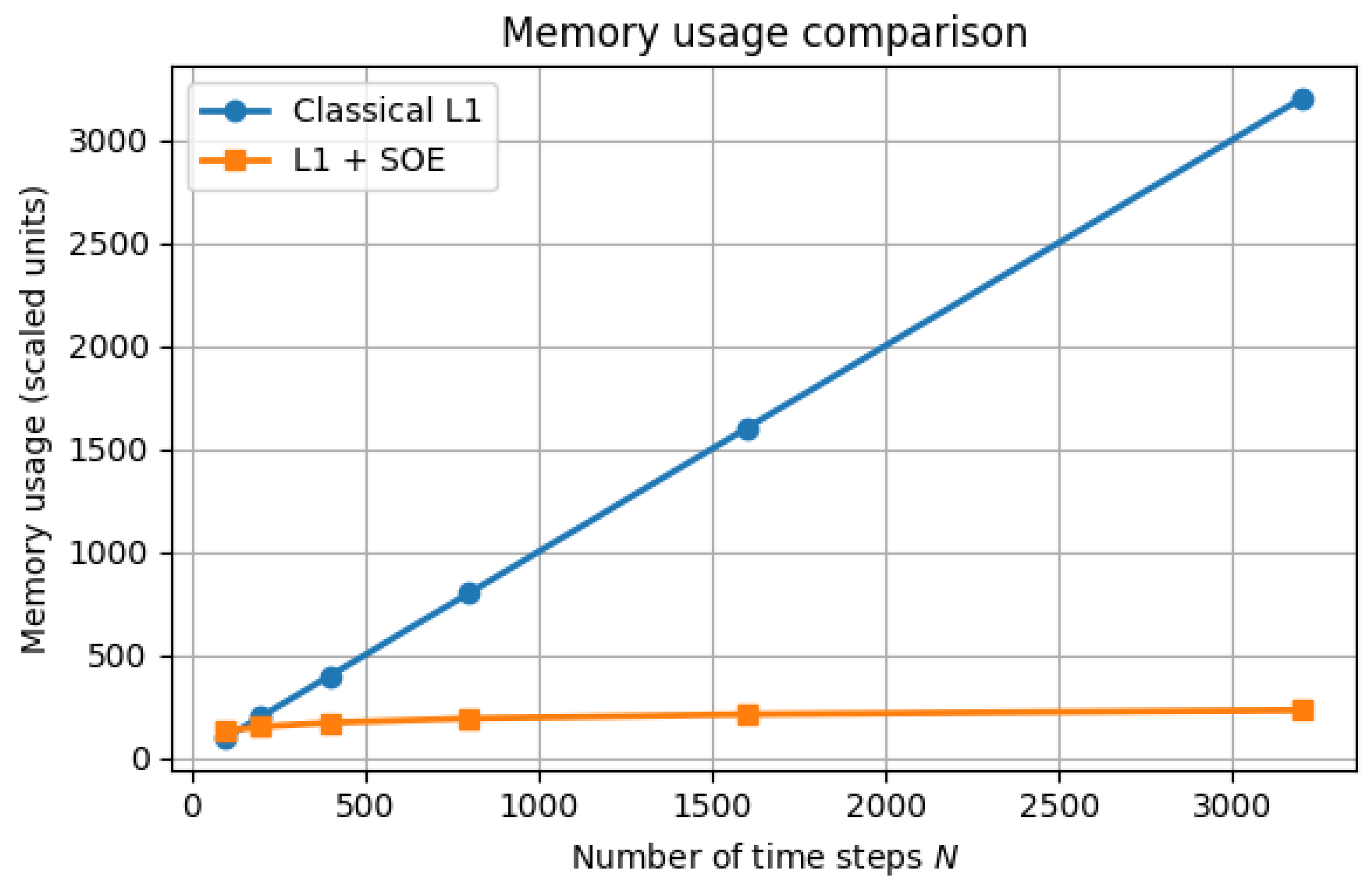
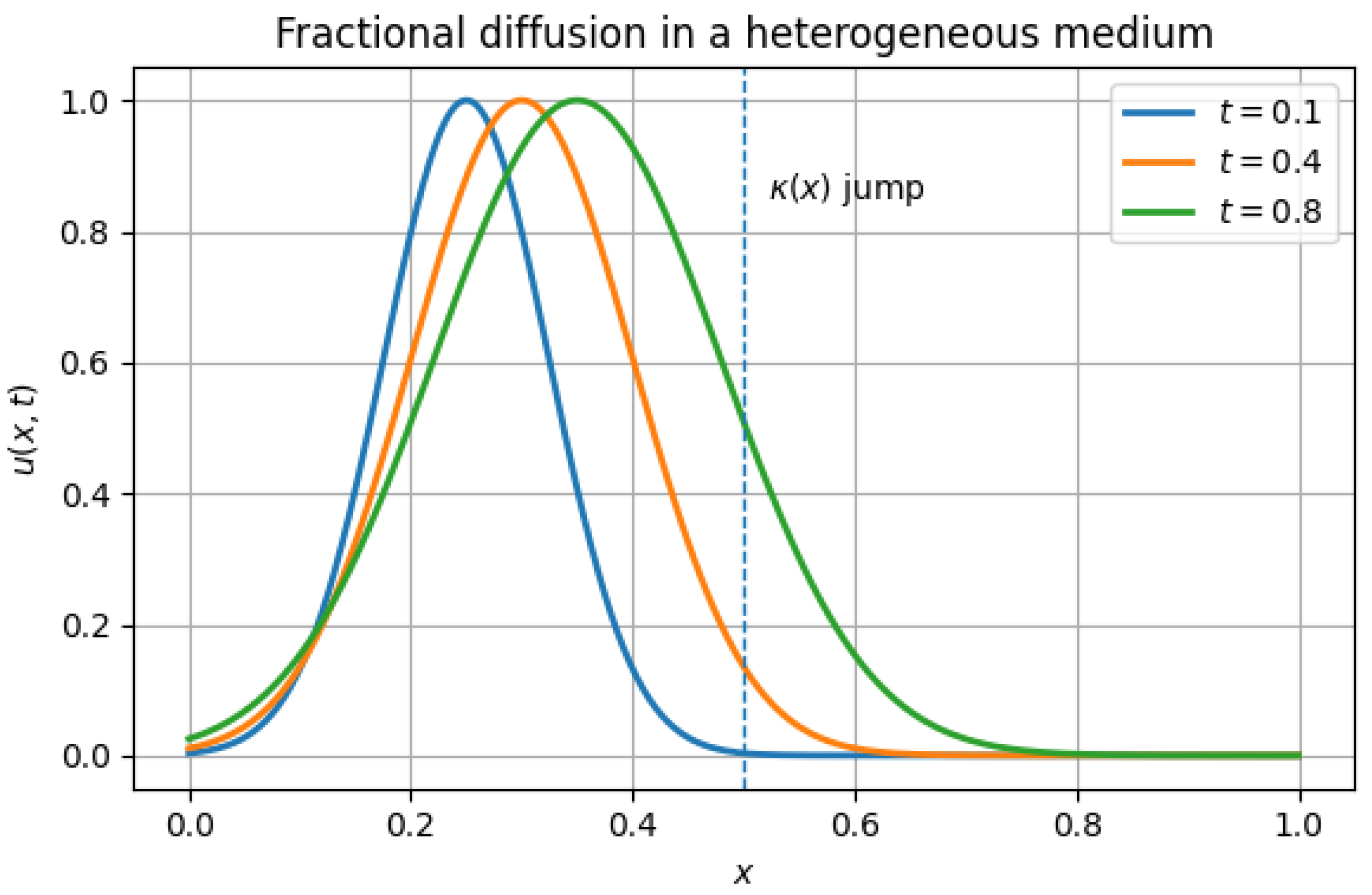
| error at T | Observed order | |
|---|---|---|
| – | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




