Submitted:
05 February 2026
Posted:
20 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Results:
Discussion:
Summary of Main Results:
Results in the Context of Published Literature:
Strength and Weakness:
Implications for Practice:
Author Contributions
Funding
Clinical trial number
Ethical statement
Financial disclosure
References
- Bokhman, J.V., Two pathogenetic types of endometrial carcinoma. Gynecol Oncol, 1983. 15(1): p. 10-7. [CrossRef]
- Talhouk, A., et al., A clinically applicable molecular-based classification for endometrial cancers. Br J Cancer, 2015. 113(2): p. 299-310. [CrossRef]
- Hoang, L.N., et al., Interobserver Agreement in Endometrial Carcinoma Histotype Diagnosis Varies Depending on The Cancer Genome Atlas (TCGA)-based Molecular Subgroup. Am J Surg Pathol, 2017. 41(2): p. 245-252. [CrossRef]
- Kandoth, C., et al., Integrated genomic characterization of endometrial carcinoma. Nature, 2013. 497(7447): p. 67-73.
- Helpman, L., et al., Disparities in the survival of endometrial cancer patients in a public healthcare system: A population-based cohort study. Gynecol Oncol, 2022. 167(3): p. 532-539. [CrossRef]
- Helpman, L., et al., Endometrial cancer presentation is associated with social determinants of health in a public healthcare system: A population-based cohort study. Gynecol Oncol, 2020. 158(1): p. 130-136. [CrossRef]
- Zhang, P., et al., Differences in Prevalence of Pregnancy Complications and Placental Pathology by Race and Ethnicity in a New York Community Hospital. JAMA Netw Open, 2022. 5(5): p. e2210719. [CrossRef]
- Zhang, P., et al., Potential association between marital status and maternal and neonatal complications and placental pathology in singleton pregnancy. Reproductive Medicine, 2023. 4: p. 28 - 33. [CrossRef]
- Martinez, C.A., et al., Epigenetic modifications appear in the human placenta following anxiety and depression during pregnancy. Placenta, 2023. 140: p. 72-79. [CrossRef]
- Liu, L., et al., Differential trends in rising endometrial cancer incidence by age, race, and ethnicity. JNCI Cancer Spectr, 2023. 7(1). [CrossRef]
- Njoku, K., et al., Impact of socio-economic deprivation on endometrial cancer survival in the North West of England: a prospective database analysis. BJOG, 2021. 128(7): p. 1215-1224. [CrossRef]
- Rota, M., et al., Modelling body mass index and endometrial cancer risk in a pooled-analysis of three case-control studies. BJOG, 2016. 123(2): p. 285-92. [CrossRef]
- Van Arsdale, A., et al., Association of obesity with survival in patients with endometrial cancer. Gynecol Oncol, 2019. 154(1): p. 156-162.
- Sarink, D., et al., Racial/ethnic differences in anthropometric and hormone-related factors and endometrial cancer risk: the Multiethnic Cohort Study. Br J Cancer, 2021. 124(10): p. 1724-1733. [CrossRef]
- Kumarasinghe, A.P., et al., DNA mismatch repair enzyme immunohistochemistry in colorectal cancer: a comparison of biopsy and resection material. Pathology, 2010. 42(5): p. 414-20. [CrossRef]
- Cunningham, J.M., et al., The frequency of hereditary defective mismatch repair in a prospective series of unselected colorectal carcinomas. Am J Hum Genet, 2001. 69(4): p. 780-90. [CrossRef]
- Chapel, D.B., et al., Immunohistochemistry for mismatch repair protein deficiency in endometrioid endometrial carcinoma yields equivalent results when performed on endometrial biopsy/curettage or hysterectomy specimens. Gynecol Oncol, 2018. 149(3): p. 570-574. [CrossRef]
- Chapel, D.B., et al., Interpretation of Mismatch Repair Protein Immunohistochemistry in Endometrial Carcinoma Should Consider Both Lynch Syndrome Screening and Immunotherapy Susceptibility: An Illustrative Case Report. Int J Gynecol Pathol, 2020. 39(3): p. 233-237. [CrossRef]
- de Freitas, D., et al., Clinicopathological characteristics of endometrial carcinomas according to DNA mismatch repair protein status. Heliyon, 2023. 9(6): p. e17495.
- Konstantinopoulos, P.A., et al., Phase II Study of Avelumab in Patients With Mismatch Repair Deficient and Mismatch Repair Proficient Recurrent/Persistent Endometrial Cancer. J Clin Oncol, 2019. 37(30): p. 2786-2794. [CrossRef]
- Nagar, H., et al., Sentinel node biopsy for diagnosis of lymph node involvement in endometrial cancer. Cochrane Database Syst Rev, 2021. 6(6): p. CD013021.
- Lindemann, K., et al., Body mass, diabetes and smoking, and endometrial cancer risk: a follow-up study. Br J Cancer, 2008. 98(9): p. 1582-5. [CrossRef]
- Ryan, N.A.J., et al., The proportion of endometrial tumours associated with Lynch syndrome (PETALS): A prospective cross-sectional study. PLoS Med, 2020. 17(9): p. e1003263. [CrossRef]
- Ramchander, N.C., et al., Distinct Immunological Landscapes Characterize Inherited and Sporadic Mismatch Repair Deficient Endometrial Cancer. Front Immunol, 2019. 10: p. 3023. [CrossRef]
- Clarke, M.A., et al., Racial and Ethnic Differences in Hysterectomy-Corrected Uterine Corpus Cancer Mortality by Stage and Histologic Subtype. JAMA Oncol, 2022. 8(6): p. 895-903. [CrossRef]
- Baskovic, M., et al., Racial disparities in outcomes for high-grade uterine cancer: A California cancer registry study. Cancer Med, 2018. 7(9): p. 4485-4495. [CrossRef]
- Cote, M.L., et al., The Growing Burden of Endometrial Cancer: A Major Racial Disparity Affecting Black Women. Cancer Epidemiol Biomarkers Prev, 2015. 24(9): p. 1407-15.
- Karia, P.S., et al., Racial and ethnic differences in type II endometrial cancer mortality outcomes: The contribution of sociodemographic, clinicopathologic, and treatment factors. Gynecol Oncol, 2023. 168: p. 119-126. [CrossRef]
- Raimondo, D., et al., Race and ethnicity reporting in endometrial cancer literature. Int J Gynecol Cancer, 2023. 33(9): p. 1402-1407. [CrossRef]
- Kim, J.H., et al., Clinicopathologic significance of DNA mismatch repair protein status in endometrial cancer. Taiwan J Obstet Gynecol, 2022. 61(3): p. 415-421. [CrossRef]
- Kim, M.K., et al., Clinicopathologic significance of mismatch repair protein expression in endometrioid endometrial cancer. Taiwan J Obstet Gynecol, 2023. 62(5): p. 724-728. [CrossRef]
- Zhang, P., et al., Differences in Prevalence of Colorectal Carcinoma by Gender and Marital Status and Expression of DNA Mismatch Repair Proteins. International Journal of Translational Medicine, 2024. 4(3): p. 584-594. [CrossRef]
- Pasanen, A., M. Loukovaara, and R. Bützow, Clinicopathological significance of deficient DNA mismatch repair and MLH1 promoter methylation in endometrioid endometrial carcinoma. Mod Pathol, 2020. 33(7): p. 1443-1452. [CrossRef]
- Uribe, Y., et al., Intersectionality Between Epigenetics and Cancer Health Disparities Stemming from Social Determinants of Health (SDoH) Through a Gynecologic Oncology Lens: A Narrative Review. Clin Obstet Gynecol, 2023. 66(1): p. 53-62. [CrossRef]
- Nowakowski, R., et al., Variances in the Expression Profile of the EMT-Related Genes in Endometrial Cancer Lines In Vitro Study. Curr Pharm Biotechnol, 2022. 23(4): p. 594-608. [CrossRef]
- Loukovaara, M., A. Pasanen, and R. Bützow, Mismatch Repair Deficiency as a Predictive and Prognostic Biomarker in Molecularly Classified Endometrial Carcinoma. Cancers (Basel), 2021. 13(13). [CrossRef]
- Addante, F., et al., Mismatch Repair Deficiency as a Predictive and Prognostic Biomarker in Endometrial Cancer: A Review on Immunohistochemistry Staining Patterns and Clinical Implications. Int J Mol Sci, 2024. 25(2). [CrossRef]
- Alexa, M., A. Hasenburg, and M.J. Battista, The TCGA Molecular Classification of Endometrial Cancer and Its Possible Impact on Adjuvant Treatment Decisions. Cancers (Basel), 2021. 13(6). [CrossRef]
- Travaglino, A., et al., Prognostic value of the TCGA molecular classification in uterine carcinosarcoma. Int J Gynaecol Obstet, 2022. 158(1): p. 13-20. [CrossRef]
- Raffone, A., et al., Tumor-infiltrating lymphocytes and POLE mutation in endometrial carcinoma. Gynecol Oncol, 2021. 161(2): p. 621-628. [CrossRef]
- Restaino, S., et al., Management of Patients Diagnosed with Endometrial Cancer: Comparison of Guidelines. Cancers (Basel), 2023. 15(4). [CrossRef]
- Ferrari, F.A., et al., Performance of molecular classification in predicting oncologic outcomes of fertility-sparing treatment for atypical endometrial hyperplasia and endometrial cancer. Int J Gynecol Cancer, 2025. 35(1): p. 100016. [CrossRef]
- Loukovaara, M., et al., Clinicopathologic stratification demonstrates survival differences between endometrial carcinomas with mismatch repair deficiency and no specific molecular profile: a cohort study. Int J Gynecol Cancer, 2025. 35(1): p. 100048. [CrossRef]
- Aguilar, M., et al., Reliable Identification of Endometrial Precancers Through Combined Pax2, β-Catenin, and Pten Immunohistochemistry. Am J Surg Pathol, 2022. 46(3): p. 404-414. [CrossRef]
- Lucas, E., et al., Utility of a PAX2, PTEN, and β-catenin Panel in the Diagnosis of Atypical Hyperplasia/Endometrioid Intraepithelial Neoplasia in Endometrial Polyps. Am J Surg Pathol, 2023. 47(9): p. 1019-1026. [CrossRef]
- Sahoo, S.S., et al., PAX2 is Transcriptionally Silenced by a Distinct Mechanism of Epigenetic Reprogramming to Initiate Endometrial Carcinogenesis. bioRxiv, 2024.
| Race/ethnicity | Asian | Black | Hispanic | Other | White | Total | P value |
|---|---|---|---|---|---|---|---|
| (N=99)(15.3%) | (N=21)(3.3%) | (N=129)(20%) | (N=53)(8.2%) | (N=343)(53.2%) | (N=645) | ||
| Marital status | 0.13 | ||||||
| - Divorced | 9 ( 9.1%) | 3 (14.3%) | 13 (10.1%) | 6 (11.3%) | 47 (13.7%) | 78 (12.1%) | |
| - Married | 63 (63.6%) | 12 (57.1%) | 66 (51.2%) | 24 (45.3%) | 184 (53.6%) | 349 (54.1%) | |
| - Single | 20 (20.2%) | 4 (19.0%) | 33 (25.6%) | 19 (35.8%) | 62 (18.1%) | 138 (21.4%) | |
| - Widowed | 7 ( 7.1%) | 2 ( 9.5%) | 17 (13.2%) | 4 ( 7.5%) | 50 (14.6%) | 80 (12.4%) | |
| Religion | 71 (71.7%) | 16 (76.2%) | 113 (87.6%) | 36 (67.9%) | 237 (69.1%) | 473 (73.3%) | <0.01 |
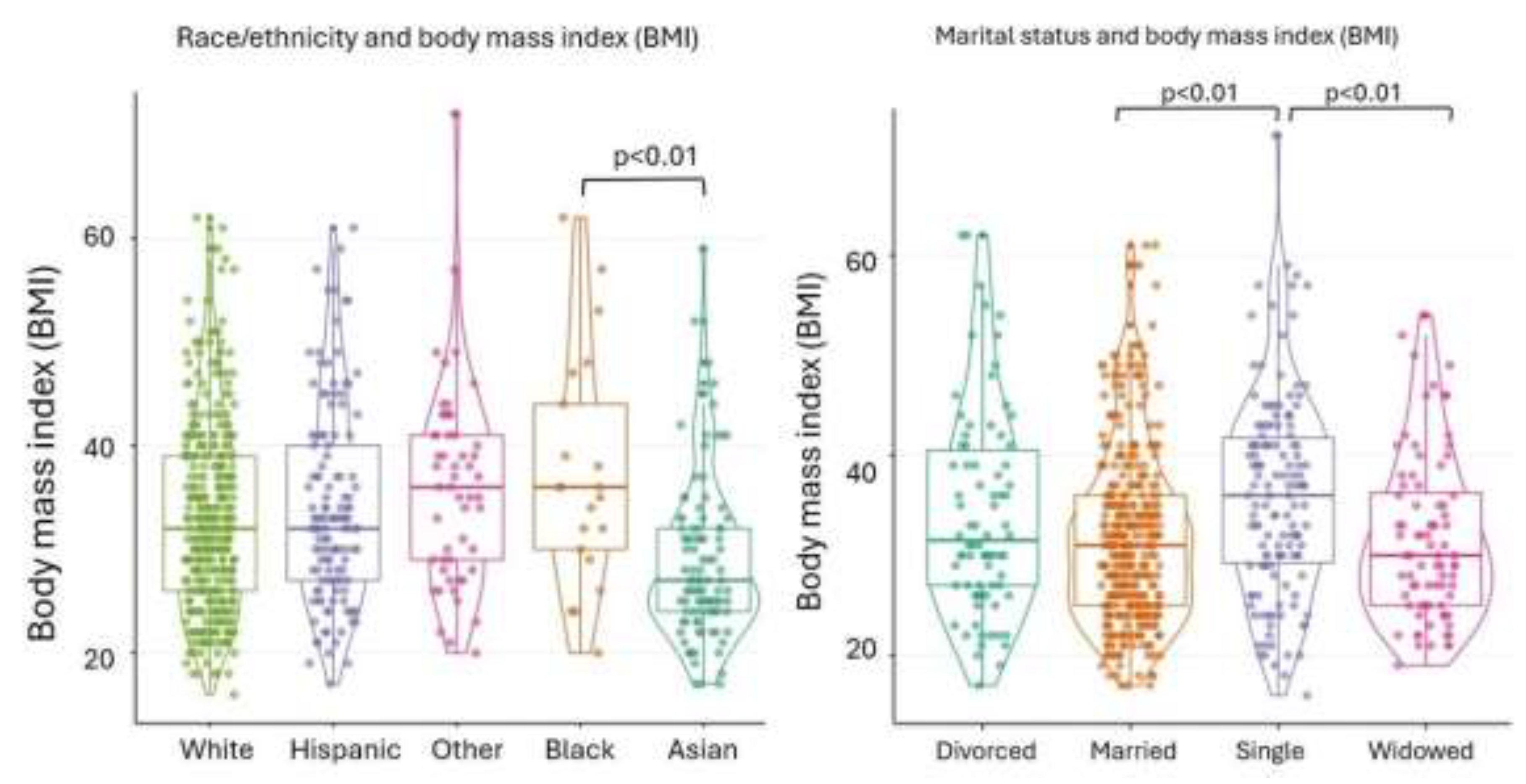
| Body mass index (BMI) | 27.0 [24.0;32.0] | 36.0 [30.0;44.0] | 32.0 [27.0;40.0] | 36.0 [29.0;41.0] | 32.0 [26.0;39.0] | 32.0 [26.0;39.0] | <0.01 |
| Obesity | <0.01 | ||||||
| - Non-obese (BMI <25) | 30 (30.3%) | 3 (14.3%) | 19 (14.7%) | 4 ( 7.5%) | 67 (19.5%) | 123 (19.1%) | |
| - Overweight (BMI 25-29) | 31 (31.3%) | 2 ( 9.5%) | 24 (18.6%) | 12 (22.6%) | 65 (19.0%) | 134 (20.8%) | |
| - Class I (BMI 30-34) | 21 (21.2%) | 4 (19.0%) | 39 (30.2%) | 6 (11.3%) | 77 (22.4%) | 147 (22.8%) | |
| - Class II (BMI 35-39) | 5 ( 5.1%) | 6 (28.6%) | 14 (10.9%) | 14 (26.4%) | 57 (16.6%) | 96 (14.9%) | |
| - Class III (BMI 40/over) | 12 (12.1%) | 6 (28.6%) | 33 (25.6%) | 17 (32.1%) | 77 (22.4%) | 145 (22.5%) | |
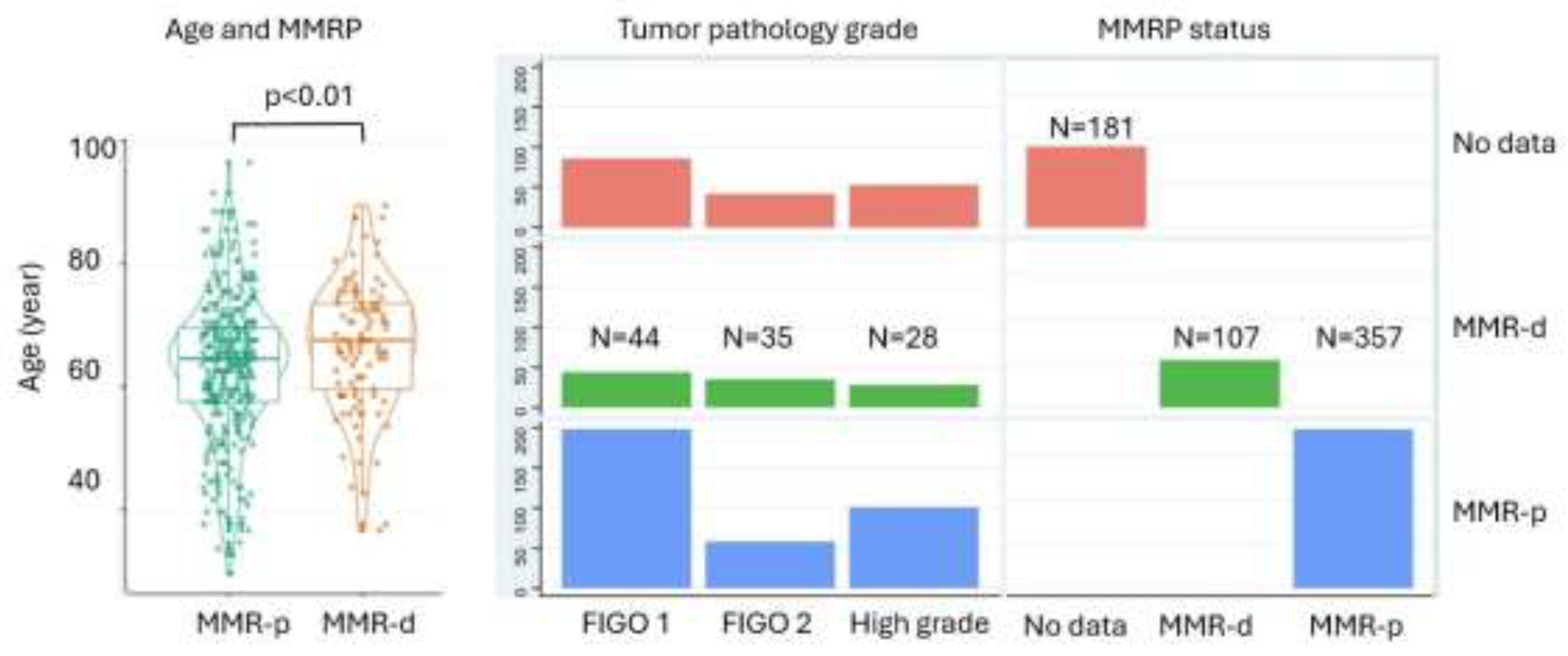
| Patient age (year) | 61.0 [55.0;69.5] | 60.0 [58.0;68.0] | 62.0 [54.0;70.0] | 60.0 [55.0;68.0] | 68.0 [63.0;74.5] | 66.0 [59.0;72.0] | <0.01 |
| Positive washing cytology | 4 ( 4.7%) | 3 (14.3%) | 11 ( 9.2%) | 3 ( 6.0%) | 22 ( 6.8%) | 43 ( 7.2%) | 0.51 |
| Tumor grade | 0.08 | ||||||
| - High grade | 26 (26.3%) | 9 (42.9%) | 47 (36.4%) | 13 (24.5%) | 87 (25.4%) | 182 (28.2%) | |
| - Low grade | 73 (73.7%) | 12 (57.1%) | 82 (63.6%) | 40 (75.5%) | 256 (74.6%) | 463 (71.8%) | |
| MMRP status | 0.34 | ||||||
| - Proficient | 54 (84.4%) | 11 (84.6%) | 80 (80.0%) | 27 (73.0%) | 185 (74.0%) | 357 (76.9%) | |
| - Deficient | 10 (15.6%) | 2 (15.4%) | 20 (20.0%) | 10 (27.0%) | 65 (26.0%) | 107 (23.1%) |
| Marital status | Divorced | Married | Single | Widowed | Total | P value |
|---|---|---|---|---|---|---|
| (N=78)(12.1%) | (N=349)(54.1%) | (N=138)(21.4%) | (N=80)(12.4%) | (N=645) | ||
| Race/ethnicity | 0.13 | |||||
| - Asian | 9 (11.5%) | 63 (18.1%) | 20 (14.5%) | 7 ( 8.8%) | 99 (15.3%) | |
| - Black | 3 ( 3.8%) | 12 ( 3.4%) | 4 ( 2.9%) | 2 ( 2.5%) | 21 ( 3.3%) | |
| - Hispanic | 13 (16.7%) | 66 (18.9%) | 33 (23.9%) | 17 (21.2%) | 129 (20.0%) | |
| - Other | 6 ( 7.7%) | 24 ( 6.9%) | 19 (13.8%) | 4 ( 5.0%) | 53 ( 8.2%) | |
| - White | 47 (60.3%) | 184 (52.7%) | 62 (44.9%) | 50 (62.5%) | 343 (53.2%) | |
| Religious belief | 61 (78.2%) | 250 (71.6%) | 94 (68.1%) | 68 (85.0%) | 473 (73.3%) | 0.03 |
| Body mass index (BMI) | 31.5 [27.0;41.0] | 31.0 [25.0;36.0] | 36.0 [29.0;42.0] | 30.0 [25.0;36.5] | 32.0 [26.0;39.0] | <0.01 |
| Obesity | <0.01 | |||||
| - Non-obese (BMI <25) | 12 (15.4%) | 75 (21.5%) | 20 (14.5%) | 16 (20.0%) | 123 (19.1%) | |
| - Overweight (BMI 25-29) | 15 (19.2%) | 82 (23.5%) | 15 (10.9%) | 22 (27.5%) | 134 (20.8%) | |
| - Class I (BMI 30-34) | 18 (23.1%) | 84 (24.1%) | 28 (20.3%) | 17 (21.2%) | 147 (22.8%) | |
| - Class II (BMI 35-39) | 13 (16.7%) | 47 (13.5%) | 26 (18.8%) | 10 (12.5%) | 96 (14.9%) | |
| - Class III (BMI 40/over) | 20 (25.6%) | 61 (17.5%) | 49 (35.5%) | 15 (18.8%) | 145 (22.5%) | |
| Patient age (year) | 68.0 [62.0;73.0] | 65.0 [59.0;71.0] | 62.0 [56.0;68.0] | 74.5 [68.0;81.5] | 66.0 [59.0;72.0] | <0.01 |
| Positive washing cytology | 4 ( 5.5%) | 24 ( 7.4%) | 10 ( 7.9%) | 5 ( 6.8%) | 43 ( 7.2%) | 0.93 |
| Tumor grade | 0.01 | |||||
| - High grade | 19 (24.4%) | 84 (24.1%) | 45 (32.6%) | 34 (42.5%) | 182 (28.2%) | |
| - Low grade | 59 (75.6%) | 265 (75.9%) | 93 (67.4%) | 46 (57.5%) | 463 (71.8%) | |
| MMRP status | 0.02 | |||||
| - Proficient | 37 (61.7%) | 189 (77.5%) | 81 (81.0%) | 50 (83.3%) | 357 (76.9%) | |
| - Deficient | 23 (38.3%) | 55 (22.5%) | 19 (19.0%) | 10 (16.7%) | 107 (23.1%) |
| Obesity | Non-obese | Overweight | Class I | Class II | Class III | p | |
|---|---|---|---|---|---|---|---|
| (N=123)(19.1%) | (N=134)(20.8%) | (N=147)(22.8%) | (N=96)(14.9%) | (N=145)(22.5%) | |||
| Race/ethnicity | <0.01 | ||||||
| - Asian | 30 (24.4%) | 31 (23.1%) | 21 (14.3%) | 5 ( 5.2%) | 12 ( 8.3%) | ||
| - Black | 3 ( 2.4%) | 2 ( 1.5%) | 4 ( 2.7%) | 6 ( 6.2%) | 6 ( 4.1%) | ||
| - Hispanic | 19 (15.4%) | 24 (17.9%) | 39 (26.5%) | 14 (14.6%) | 33 (22.8%) | ||
| - Other | 4 ( 3.3%) | 12 ( 9.0%) | 6 ( 4.1%) | 14 (14.6%) | 17 (11.7%) | ||
| - White | 67 (54.5%) | 65 (48.5%) | 77 (52.4%) | 57 (59.4%) | 77 (53.1%) | ||
| Marital status | <0.01 | ||||||
| - Divorced | 12 ( 9.8%) | 15 (11.2%) | 18 (12.2%) | 13 (13.5%) | 20 (13.8%) | ||
| - Married | 75 (61.0%) | 82 (61.2%) | 84 (57.1%) | 47 (49.0%) | 61 (42.1%) | ||
| - Single | 20 (16.3%) | 15 (11.2%) | 28 (19.0%) | 26 (27.1%) | 49 (33.8%) | ||
| - Widowed | 16 (13.0%) | 22 (16.4%) | 17 (11.6%) | 10 (10.4%) | 15 (10.3%) | ||
| Body mass index (BMI) | 22.0 [21.0;24.0] | 27.0 [26.0;28.0] | 32.0 [31.0;33.0] | 37.0 [35.0;38.0] | 45.0 [41.0;49.0] | <0.01 | |
| Patient age (year) | 69.0 [60.5;75.5] | 67.0 [62.0;74.0] | 66.0 [60.0;71.5] | 64.0 [58.0;69.5] | 62.0 [56.0;69.0] | <0.01 | |
| Tumor grade | <0.01 | ||||||
| - High grade | 52 (42.3%) | 36 (26.9%) | 47 (32.0%) | 26 (27.1%) | 21 (14.5%) | ||
| - Low grade | 71 (57.7%) | 98 (73.1%) | 100 (68.0%) | 70 (72.9%) | 124 (85.5%) | ||
| Tumor stage (TNM-T) | 0.11 | ||||||
| - T1 | 92 (76.0%) | 107 (80.5%) | 115 (78.2%) | 75 (78.9%) | 129 (89.6%) | ||
| - T2 | 8 ( 6.6%) | 11 ( 8.3%) | 10 ( 6.8%) | 11 (11.6%) | 7 ( 4.9%) | ||
| - T3 | 21 (17.4%) | 15 (11.3%) | 21 (14.3%) | 9 ( 9.5%) | 8 ( 5.6%) | ||
| - T4 | 0 ( 0.0%) | 0 ( 0.0%) | 1 ( 0.7%) | 0 ( 0.0%) | 0 ( 0.0%) | ||
| Nodal metastasis (TNM-N) | 21 (18.9%) | 15 (12.6%) | 31 (23.1%) | 18 (20.0%) | 13 (10.7%) | 0.05 |
| Tumor grade | High grade | Low grade | Total | p value |
|---|---|---|---|---|
| (N=182)(28.2%) | (N=463)(71.8%) | (N=645) | ||
| Marital status | 0.01 | |||
| - Divorced | 19 (10.4%) | 59 (12.7%) | 78 (12.1%) | |
| - Married | 84 (46.2%) | 265 (57.2%) | 349 (54.1%) | |
| - Single | 45 (24.7%) | 93 (20.1%) | 138 (21.4%) | |
| - Widowed | 34 (18.7%) | 46 ( 9.9%) | 80 (12.4%) | |
| Body mass index (BMI) | 30.0 [24.0;35.0] | 33.0 [27.0;41.0] | 32.0 [26.0;39.0] | <0.01 |
| Obesity | <0.01 | |||
| - Non-obese (BMI <25) | 52 (28.6%) | 71 (15.3%) | 123 (19.1%) | |
| - Overweight (BMI 25-29) | 36 (19.8%) | 98 (21.2%) | 134 (20.8%) | |
| - Class I (BMI 30-34) | 47 (25.8%) | 100 (21.6%) | 147 (22.8%) | |
| - Class II (BMI 35-39) | 26 (14.3%) | 70 (15.1%) | 96 (14.9%) | |
| - Class III (BMI 40/over) | 21 (11.5%) | 124 (26.8%) | 145 (22.5%) | |
| Patient age (year) | 68.0 [63.0;76.0] | 64.0 [58.0;71.0] | 66.0 [59.0;72.0] | <0.01 |
| Positive washing cytology | 30 (17.9%) | 13 ( 3.0%) | 43 ( 7.2%) | <0.01 |
| T stage (TNM-T) | <0.01 | |||
| - T1 | 104 (58.1%) | 414 (89.8%) | 518 (80.9%) | |
| - T2 | 21 (11.7%) | 26 ( 5.6%) | 47 ( 7.3%) | |
| - T3 | 53 (29.6%) | 21 ( 4.6%) | 74 (11.6%) | |
| - T4 | 1 ( 0.6%) | 0 ( 0.0%) | 1 ( 0.2%) | |
| Nodal metastasis (TNM-N) | 48 (31.2%) | 50 (11.8%) | 98 (17.0%) | <0.01 |
| Distant metastasis (TNM-M) | 25 (83.3%) | 5 (22.7%) | 30 (57.7%) | <0.01 |
| FIGO stage (2009) | <0.01 | |||
| - No data | 15 ( 8.2%) | 17 ( 3.7%) | 32 ( 5.0%) | |
| - I | 81 (44.5%) | 384 (82.9%) | 465 (72.1%) | |
| - II | 12 ( 6.6%) | 17 ( 3.7%) | 29 ( 4.5%) | |
| - III | 46 (25.3%) | 40 ( 8.6%) | 86 (13.3%) | |
| - IV | 28 (15.4%) | 5 ( 1.1%) | 33 ( 5.1%) | |
| MMRP status | 0.76 | |||
| - Proficient | 101 (78.3%) | 256 (76.4%) | 357 (76.9%) | |
| - Deficient | 28 (21.7%) | 79 (23.6%) | 107 (23.1%) |
| Total Endometrial Carcinoma Low Grade Endometrial Carcinoma | ||||||||
|---|---|---|---|---|---|---|---|---|
| MMRP status | Proficient | Deficient | Total | p | Proficient | Deficient | Total | p |
| (N=357)(76.9%) | (N=107)(23.1%) | (N=464) | (N=256)(76.4%) | (N=79)(23.6%) | (N=335) | |||
| Marital status | 0.02 | 0.02 | ||||||
| - Divorced | 37 (10.4%) | 23 (21.5%) | 60 (12.9%) | 27(10.5%) | 19 (24.1%) | 46 (13.7%) | ||
| - Married | 189 (52.9%) | 55 (51.4%) | 244 (52.6%) | 144(56.2%) | 41 (51.9%) | 185(55.2%) | ||
| - Single | 81 (22.7%) | 19 (17.8%) | 100 (21.6%) | 55(21.5%) | 13 (16.5%) | 68 (20.3%) | ||
| - Widowed | 50 (14.0%) | 10 ( 9.3%) | 60 (12.9%) | 30 (11.7%) | 6 ( 7.6%) | 36 (10.7%) | ||
| Patient age (year) | 65.0 [58.0;70.0] | 68.0 [60.0;74.0] | 66.0 [59.0;72.0] | <0.01 | 63.0 [56.0;69.0] | 68.0 [59.5;73.0] | 64.0 [58.0;71.0] | <0.01 |
| Positive washing cytology | 24 ( 7.3%) | 8 ( 8.0%) | 32 ( 7.4%) | 0.97 | 7 ( 3.0%) | 2 ( 2.7%) | 9 ( 2.9%) | 1.00 |
| Low grade | <0.01 | |||||||
| - FIGO 1 | -- | -- | -- | 198(77.3%) | 44 (55.7%) | 242(72.2%) | ||
| - FIGO 2 | -- | -- | -- | 58 (22.7%) | 35 (44.3%) | 93 (27.8%) | ||
| Nodal metastasis (TNM-N) | 47 (14.6%) | 24 (23.5%) | 71 (16.7%) | 0.05 | 22 ( 9.4%) | 18 (23.7%) | 40 (12.9%) | <0.01 |
| Distant metastasis (TNM-M) | 15 (51.7%) | 6 (50.0%) | 21 (51.2%) | 1.00 | 1 ( 8.3%) | 3 (42.9%) | 4 (21.1%) | 0.23 |
| FIGO stage (2009) | 0.55 | 0.02 | ||||||
| - No data | 16 ( 4.5%) | 3 ( 2.8%) | 19 ( 4.1%) | 11 ( 4.3%) | 1 ( 1.3%) | 12 ( 3.6%) | ||
| - I | 260 (72.8%) | 73 (68.2%) | 333(71.8%) | 214(83.6%) | 59 (74.7%) | 273(81.5%) | ||
| - II | 16 ( 4.5%) | 6 ( 5.6%) | 22 ( 4.7%) | 8 ( 3.1%) | 3 ( 3.8%) | 11 ( 3.3%) | ||
| - III | 46 (12.9%) | 20 (18.7%) | 66(14.2%) | 22 ( 8.6%) | 13 (16.5%) | 35 (10.4%) | ||
| - IV | 19 ( 5.3%) | 5 ( 4.7%) | 24 ( 5.2%) | 1 ( 0.4%) | 3 ( 3.8%) | 4 ( 1.2%) | ||
| Estimate | STD error | t-value | p value | |
| Race/ethnicity | ||||
| - Black | 0.210 | 0.193 | 1.086 | 0.28 |
| - Hispanic | 0.086 | 0.092 | 0.939 | 0.35 |
| - Others | 0.125 | 0.113 | 1.105 | 0.27 |
| - White | 0.072 | 0.080 | 0.889 | 0.38 |
| Marital status | ||||
| -Married | -0.100 | 0.075 | -1.33 | 0.18 |
| - Single | -0.114 | 0.087 | -1.307 | 0.19 |
| - Widowed | -0.239 | 0.101 | -2.375 | 0.02* |
| BMI/Obesity | ||||
| BMI | -0.006 | 0.007 | -0.828 | 0.41 |
| Patient’s age | 0.007 | 0.003 | 2.885 | 0.004* |
| T-stage | 0.009 | 0.114 | 0.077 | 0.94 |
| Nodal stage | 0.117 | 0.116 | 1.004 | 0.32 |
| FIGO stage (2009) | ||||
| - FIGO I | 0.013 | 0.066 | 0.200 | 0.84 |
| - FIGO II | 0.040 | 0.173 | 0.228 | 0.82 |
| - FIGO III | 0.089 | 0.270 | -0.330 | 0.74 |
| - FIGO IV | 0.400 | 0.245 | 1.632 | 0.10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




