Submitted:
18 February 2026
Posted:
23 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- What are the drivers of successful implementation of TRE in patients with IBS, and how do they affect the feasibility of the treatment?
- What are the barriers to the implementation of TRE in patients with IBS, and how do they affect the feasibility of the treatment?
2. Materials and Methods
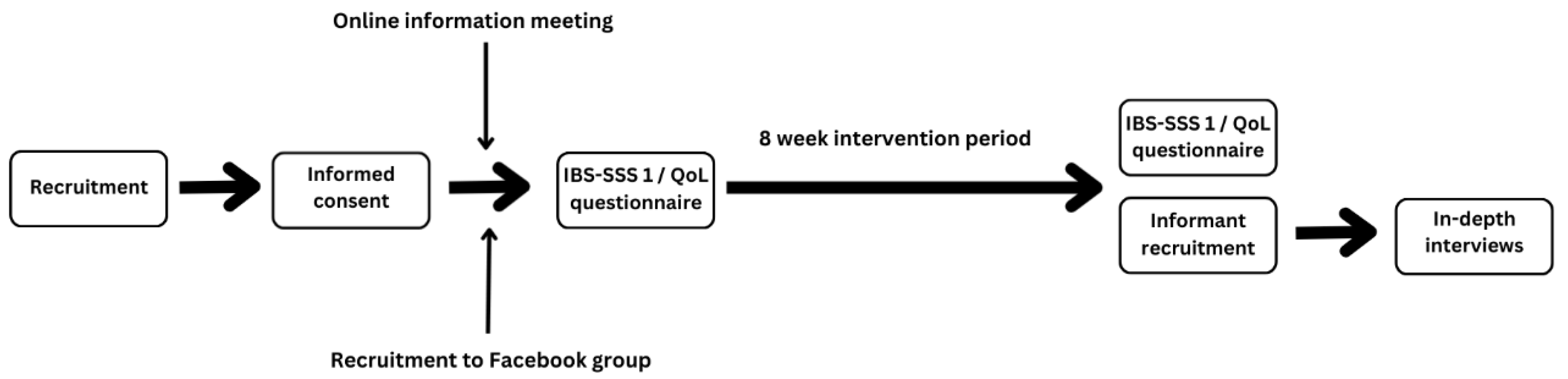
2.1. Intervention
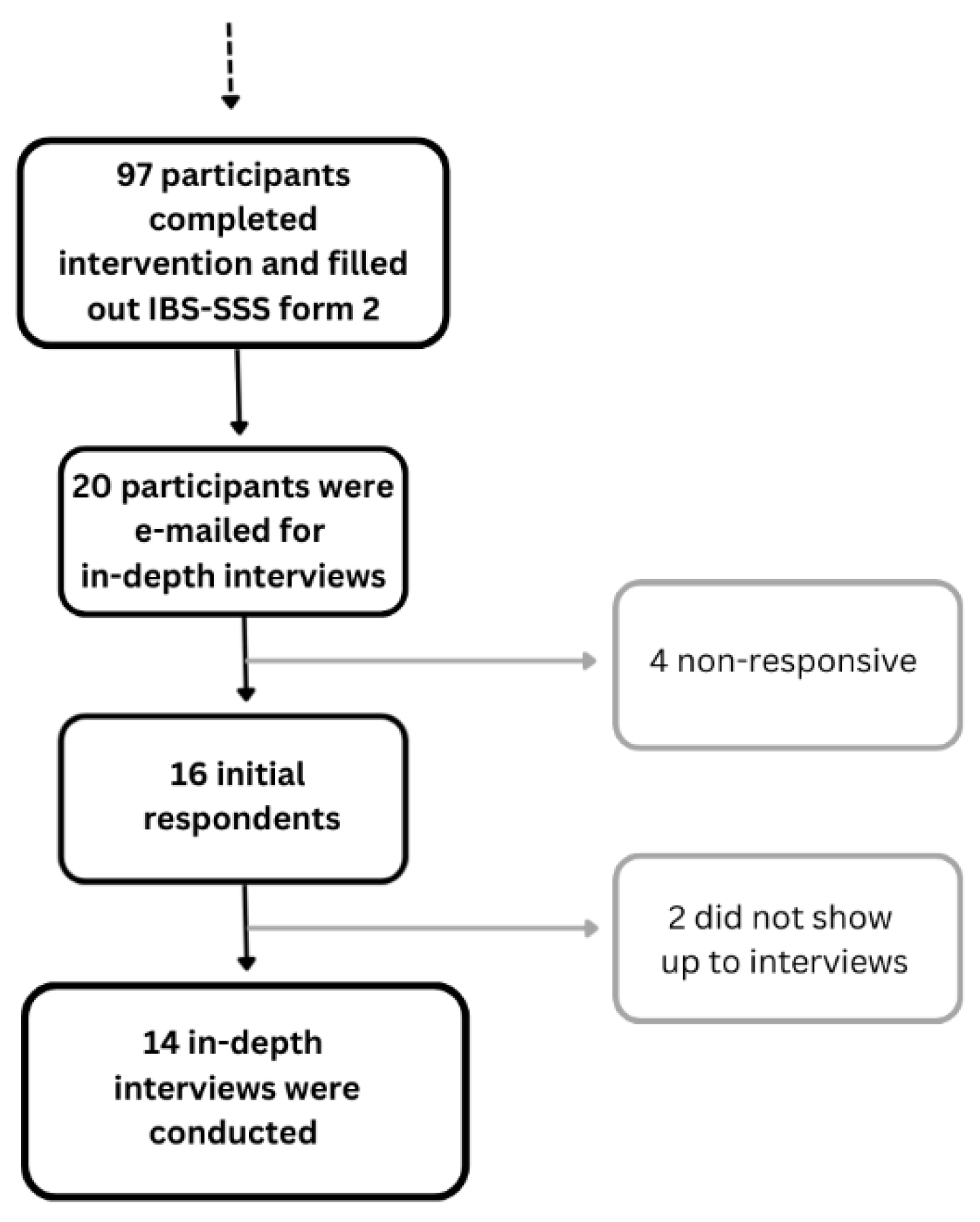
2.2. Informant Recruitment
2.3. Data Collection
2.4. Data Analysis
| Main theme | Sub-theme | Code | Key information element |
|---|---|---|---|
| Barriers | Renunciation | Not being able to partake | “I very much missed taking part in family breakfasts in the weekends. I would just sit there with a dull cup of tea.” |
2.5. Reflexivity
2.6. Ethics Statement
3. Results
3.1. Informant Characteristics
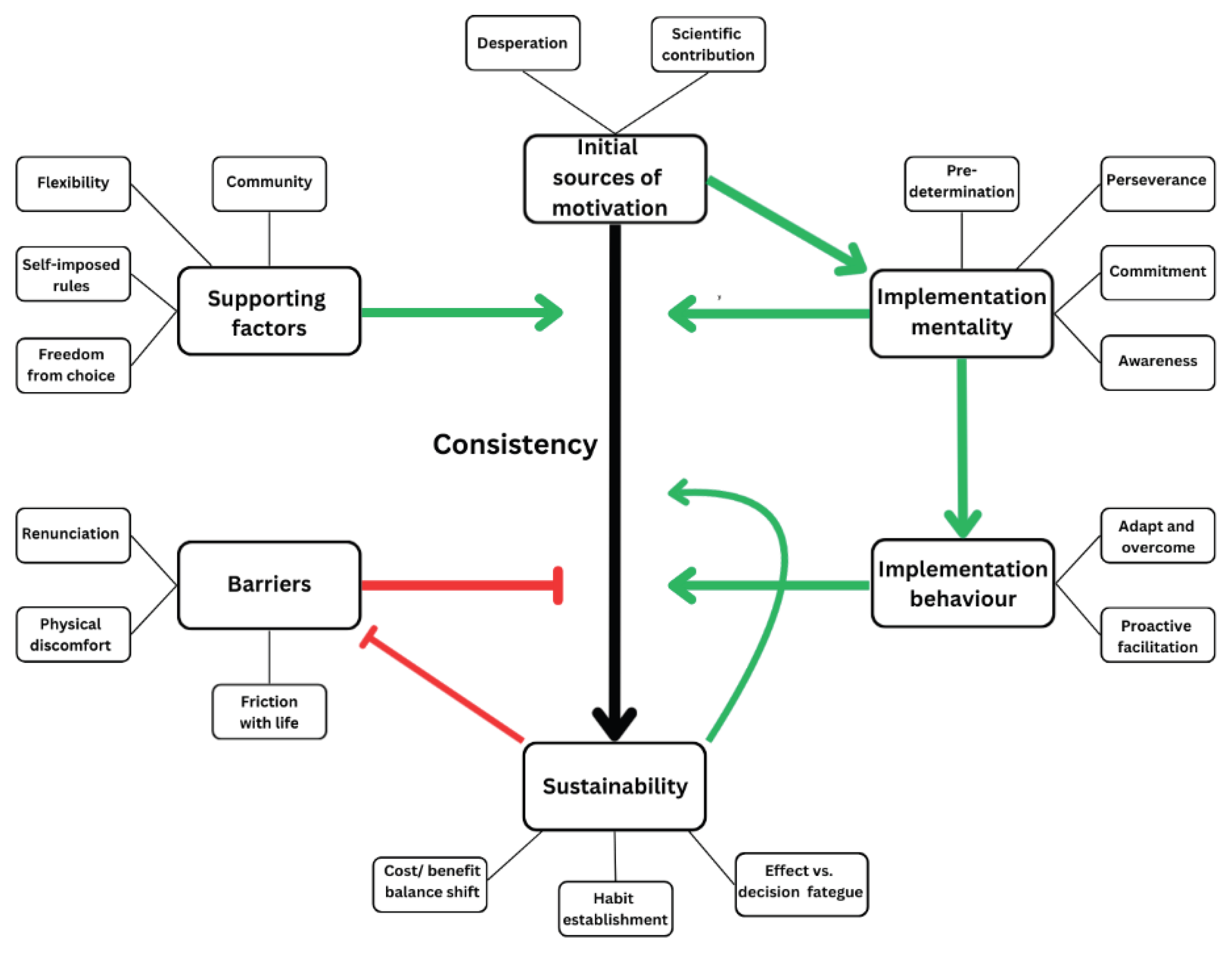
3.2. Themes
3.2.1. Initial Sources of Motivation
3.2.2. Barriers
3.2.3. Implementation Mentality
3.2.4. Supporting Factors
3.2.5. Implementation Behaviour
3.2.6. Sustainability
3.2.7. The Process of Change
4. Discussion
4.1. In Light of Previous Research
4.2. Methodical Considerations
4.3. Limitations
4.4. Focus Points for Future TRE Implementation in IBS Patients
- Allow a sufficient intervention period. Many informants reported that TRE only became sustainable after 4–6 weeks, once physical adaptation and symptom relief occurred. Future studies should ensure the intervention is long enough for this transition phase to unfold.
- Ensure mental readiness before enrollment. Require participants to engage in pre-intervention reflection or orientation sessions that emphasize realistic expectations, potential discomforts, and personal commitment. Encourage participants to visualize common barriers and decide in advance how they will maintain adherence.
- Promote a single, sustained commitment. Introduce the idea of making one firm decision to adhere to TRE throughout the study in order to reduce decision fatigue. Reinforce this by asking participants to communicate their commitment to family and peers for accountability and practical support.
- Normalize temporary challenges. Explicitly inform participants that physical, social, and psychological challenges are common but typically transient. Providing real testimonials or case examples can improve perseverance during the adaptation period.
- Facilitate peer connection and researcher presence. Create structured communities where participants can share progress and experiences. Active researcher engagement through these channels helps maintain motivation and a sense of belonging, shared purpose and accountability.
- Encourage strategic flexibility. Allow flexibility in meal timing within the 16:8 framework to reduce social strain and improve adherence. Provide practical tools and examples for adjusting eating windows without compromising intervention integrity.
- Support proactive adaptation and problem-solving. Guide participants to anticipate and manage barriers through concrete actions: meal prepping, communicating dietary routines to others, planning for social events, and avoiding high-risk situations for nonadherence. Emphasize that adaptation is gradual and iterative.
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TRE | Time-restricted eating |
| IBS | Irritable Bowel Syndrome |
References
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional Bowel Disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef]
- Sayuk, G.S.; Wolf, R.; Chang, L. Comparison of Symptoms, Healthcare Utilization, and Treatment in Diagnosed and Undiagnosed Individuals With Diarrhea-Predominant Irritable Bowel Syndrome. Am. J. Gastroenterol. 2017, 112, 892–899. [Google Scholar] [CrossRef]
- Ballou, S.; Keefer, L. The Impact of Irritable Bowel Syndrome on Daily Functioning: Characterizing and Understanding Daily Consequences of IBS. Neurogastroenterol. Motil. 2017, 29. [Google Scholar] [CrossRef]
- Österberg, E.; Blomquist, L.; Krak, I. A Population Study on Irritable Bowel Syndrome and Mental Health. Scand. J. Gastroenterol. 2000, 35, 264–268. [Google Scholar] [CrossRef]
- El-Salhy, M.; Johansson, M.; Klevstul, M.; Hatlebakk, J.G. Quality of Life, Functional Impairment and Healthcare Experiences of Patients with Irritable Bowel Syndrome in Norway: An Online Survey. BMC Gastroenterol. 2025, 25, 143. [Google Scholar] [CrossRef] [PubMed]
- Tarar, Z.I.; Farooq, U.; Zafar, Y.; Gandhi, M.; Raza, S.; Kamal, F.; Tarar, M.F.; Ghouri, Y.A. Burden of Anxiety and Depression among Hospitalized Patients with Irritable Bowel Syndrome: A Nationwide Analysis. Ir. J. Med. Sci. 1971 - 2023, 192, 2159–2166. [Google Scholar] [CrossRef]
- Black, C.J.; Ford, A.C. Global Burden of Irritable Bowel Syndrome: Trends, Predictions and Risk Factors. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Lovell, R.M.; Ford, A.C. Global Prevalence of and Risk Factors for Irritable Bowel Syndrome: A Meta-Analysis. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2012, 10, 712–721.e4. [Google Scholar] [CrossRef]
- Oka, P.; Parr, H.; Barberio, B.; Black, C.J.; Savarino, E.V.; Ford, A.C. Global Prevalence of Irritable Bowel Syndrome According to Rome III or IV Criteria: A Systematic Review and Meta-Analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 908–917. [Google Scholar] [CrossRef]
- Gibson, P.R. The Evidence Base for Efficacy of the Low FODMAP Diet in Irritable Bowel Syndrome: Is It Ready for Prime Time as a First-Line Therapy? J. Gastroenterol. Hepatol. 2017, 32 Suppl 1, 32–35. [Google Scholar] [CrossRef]
- Bardacke, J.A.; Yarrow, L.; Rosenkranz, S.K. The Long-Term Effects of a Low-Fermentable Oligosaccharides, Disaccharides, Monosaccharides, and Polyols Diet for Irritable Bowel Syndrome Management. Curr. Dev. Nutr. 2023, 7, 101997. [Google Scholar] [CrossRef]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef]
- Nybacka, S.; Törnblom, H.; Josefsson, A.; Hreinsson, J.P.; Böhn, L.; Frändemark, Å.; Weznaver, C.; Störsrud, S.; Simrén, M. A Low FODMAP Diet plus Traditional Dietary Advice versus a Low-Carbohydrate Diet versus Pharmacological Treatment in Irritable Bowel Syndrome (CARIBS): A Single-Centre, Single-Blind, Randomised Controlled Trial. Lancet Gastroenterol. Hepatol. 2024, 9, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Varjú, P.; Farkas, N.; Hegyi, P.; Garami, A.; Szabó, I.; Illés, A.; Solymár, M.; Vincze, Á.; Balaskó, M.; Pár, G.; et al. Low Fermentable Oligosaccharides, Disaccharides, Monosaccharides and Polyols (FODMAP) Diet Improves Symptoms in Adults Suffering from Irritable Bowel Syndrome (IBS) Compared to Standard IBS Diet: A Meta-Analysis of Clinical Studies. PLOS ONE 2017, 12, e0182942. [Google Scholar] [CrossRef]
- Fiocchi, C. Inflammatory Bowel Disease: Etiology and Pathogenesis. Gastroenterology 1998, 115, 182–205. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M.; Lomer, M.C. Who Should Deliver the Low FODMAP Diet and What Educational Methods Are Optimal: A Review. J. Gastroenterol. Hepatol. 2017, 32 Suppl 1, 23–26. [Google Scholar] [CrossRef]
- Koppold, D.A.; Breinlinger, C.; Hanslian, E.; Kessler, C.; Cramer, H.; Khokhar, A.R.; Peterson, C.M.; Tinsley, G.; Vernieri, C.; Bloomer, R.J.; et al. International Consensus on Fasting Terminology. Cell Metab. 2024, 36, 1779–1794.e4. [Google Scholar] [CrossRef]
- Ezzati, A.; McLaren, C.; Bohlman, C.; Tamargo, J.A.; Lin, Y.; Anton, S.D. Does Time-Restricted Eating Add Benefits to Calorie Restriction? A Systematic Review. Obes. Silver Spring Md 2024, 32, 640–654. [Google Scholar] [CrossRef]
- Moro, T.; Tinsley, G.; Bianco, A.; Marcolin, G.; Pacelli, Q.F.; Battaglia, G.; Palma, A.; Gentil, P.; Neri, M.; Paoli, A. Effects of Eight Weeks of Time-Restricted Feeding (16/8) on Basal Metabolism, Maximal Strength, Body Composition, Inflammation, and Cardiovascular Risk Factors in Resistance-Trained Males. J. Transl. Med. 2016, 14, 290. [Google Scholar] [CrossRef]
- Wang, R.; Lv, X.; Xu, W.; Li, X.; Tang, X.; Huang, H.; Yang, M.; Ma, S.; Wang, N.; Niu, Y. Effects of the Periodic Fasting-Mimicking Diet on Health, Lifespan, and Multiple Diseases: A Narrative Review and Clinical Implications. Nutr. Rev. 2025, 83, e412–e426. [Google Scholar] [CrossRef] [PubMed]
- Baxt, L.A.; Xavier, R.J. Role of Autophagy in the Maintenance of Intestinal Homeostasis. Gastroenterology 2015, 149, 553–562. [Google Scholar] [CrossRef]
- Berg, L.K.; Goll, R.; Fagerli, E.; Ludviksen, J.K.; Fure, H.; Moen, O.S.; Sørbye, S.W.; Mollnes, T.E.; Florholmen, J. Intestinal Inflammatory Profile Shows Increase in a Diversity of Biomarkers in Irritable Bowel Syndrome. Scand. J. Gastroenterol. 2020, 55, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Deloose, E.; Janssen, P.; Depoortere, I.; Tack, J. The Migrating Motor Complex: Control Mechanisms and Its Role in Health and Disease. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 271–285. [Google Scholar] [CrossRef]
- Ng, Q.X.; Soh, A.Y.S.; Loke, W.; Lim, D.Y.; Yeo, W.-S. The Role of Inflammation in Irritable Bowel Syndrome (IBS). J. Inflamm. Res. 2018, 11, 345–349. [Google Scholar] [CrossRef]
- Khan, M.N.; Khan, S.I.; Rana, M.I.; Ayyaz, A.; Khan, M.Y.; Imran, M. Intermittent Fasting Positively Modulates Human Gut Microbial Diversity and Ameliorates Blood Lipid Profile. Front. Microbiol. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Gerdel, T.; Camargo, M.; Alvarado, M.; Ramírez, J.D. Impact of Intermittent Fasting on the Gut Microbiota: A Systematic Review. Adv. Biol. 2023, 7, e2200337. [Google Scholar] [CrossRef]
- Pimentel, M.; Chow, E.J.; Lin, H.C. Eradication of Small Intestinal Bacterial Overgrowth Reduces Symptoms of Irritable Bowel Syndrome. Am. J. Gastroenterol. 2000, 95, 3503–3506. [Google Scholar] [CrossRef]
- Pimentel, M.; Soffer, E.E.; Chow, E.J.; Kong, Y.; Lin, H.C. Lower Frequency of MMC Is Found in IBS Subjects with Abnormal Lactulose Breath Test, Suggesting Bacterial Overgrowth. Dig. Dis. Sci. 2002, 47, 2639–2643. [Google Scholar] [CrossRef]
- Tap, J.; Derrien, M.; Törnblom, H.; Brazeilles, R.; Cools-Portier, S.; Doré, J.; Störsrud, S.; Le Nevé, B.; Öhman, L.; Simrén, M. Identification of an Intestinal Microbiota Signature Associated With Severity of Irritable Bowel Syndrome. Gastroenterology 2017, 152, 111–123.e8. [Google Scholar] [CrossRef]
- Teker, H.T.; Ceylani, T. Intermittent Fasting Supports the Balance of the Gut Microbiota Composition. Int. Microbiol. 2023, 26, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Kolby, M.; Brevik, A.; Dale, H.F.; Valeur, J. Intermittent Fasting as a Potential Therapeutic Approach for Irritable Bowel Syndrome: A Hypothesis and Mechanistic Perspective 2025.
- Bjerre, N.; Holm, L.; Quist, J.S.; Færch, K.; Hempler, N.F. Watching, Keeping and Squeezing Time to Lose Weight: Implications of Time-Restricted Eating in Daily Life. Appetite 2021, 161, 105138. [Google Scholar] [CrossRef]
- Bjerre, N.; Holm, L.; Veje, N.; Quist, J.S.; Færch, K.; Hempler, N.F. What Happens after a Weight Loss Intervention? A Qualitative Study of Drivers and Challenges of Maintaining Time-Restricted Eating among People with Overweight at High Risk of Type 2 Diabetes. Appetite 2022, 174, 106034. [Google Scholar] [CrossRef]
- Jan, Y.C.; Chiang, S.-W.; Hsieh, T.C. Exploring the Successful Experience of Time-Restricted Eating in Overweight Adults: A Qualitative Study. Appetite 2023, 188, 106979. [Google Scholar] [CrossRef]
- Jefcoate, P.W.; Robertson, M.D.; Ogden, J.; Johnston, J.D. Exploring Rates of Adherence and Barriers to Time-Restricted Eating. Nutrients 2023, 15, 2336. [Google Scholar] [CrossRef]
- Kesztyüs, D.; Cermak, P.; Gulich, M.; Kesztyüs, T. Adherence to Time-Restricted Feeding and Impact on Abdominal Obesity in Primary Care Patients: Results of a Pilot Study in a Pre-Post Design. Nutrients 2019, 11, 2854. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A.; Sypniewski, C.; Bensadon, B.A.; McLaren, C.; Donahoo, W.T.; Sibille, K.T.; Anton, S. Determinants of Adherence in Time-Restricted Feeding in Older Adults: Lessons from a Pilot Study. Nutrients 2020, 12, 874. [Google Scholar] [CrossRef]
- O’Connor, S.G.; Boyd, P.; Bailey, C.P.; Nebeling, L.; Reedy, J.; Czajkowski, S.M.; Shams-White, M.M. A Qualitative Exploration of Facilitators and Barriers of Adherence to Time-Restricted Eating. Appetite 2022, 178, 106266. [Google Scholar] [CrossRef] [PubMed]
- Parr, E.B.; Devlin, B.L.; Lim, K.H.C.; Moresi, L.N.Z.; Geils, C.; Brennan, L.; Hawley, J.A. Time-Restricted Eating as a Nutrition Strategy for Individuals with Type 2 Diabetes: A Feasibility Study. Nutrients 2020, 12, 3228. [Google Scholar] [CrossRef]
- Skarstad, H.M.S.; Haganes, K.L.; Sujan, M.A.J.; Gellein, T.M.; Johansen, M.K.; Salvesen, K.Å.; Hawley, J.A.; Moholdt, T. A Randomized Feasibility Trial of Time-Restricted Eating during Pregnancy in People with Increased Risk of Gestational Diabetes. Sci. Rep. 2024, 14, 22476. [Google Scholar] [CrossRef]
- Termannsen, A.-D.; Varming, A.; van Elst, C.; Bjerre, N.; Nørgaard, O.; Hempler, N.F.; Faerch, K.; Quist, J.S. Feasibility of Time-Restricted Eating in Individuals with Overweight, Obesity, Prediabetes, or Type 2 Diabetes: A Systematic Scoping Review. Obes. Silver Spring Md 2023, 31, 1463–1485. [Google Scholar] [CrossRef]
- Termannsen, A.-D.; Varming, A.; Hansen, G.S.; Bjerre, N.; Persson, F.; Bagger, J.I.; Hansen, D.L.; Ewers, B.; Jørgensen, N.B.; Blond, M.B.; et al. Time-Restricted Eating Is a Feasible Dietary Strategy in the Treatment of Complicated Type 2 Diabetes: The RESET2 Pilot Study. J. Nutr. Educ. Behav. 2025, 57, 767–777. [Google Scholar] [CrossRef]
- Thompson Clausen, M.; Sverdrup, H.; Brevik, A.; Molin, M.; Kolby, M. Time-Restricted Eating and Symptom Severity in Irritable Bowel Syndrome: Results from a Pilot Study. Preprints 2026, 2026012043. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V. To Saturate or Not to Saturate? Questioning Data Saturation as a Useful Concept for Thematic Analysis and Sample-Size Rationales. Qual. Res. Sport Exerc. Health 2021, 13, 201–216. [Google Scholar] [CrossRef]
- Malterud, K.; Siersma, V.D.; Guassora, A.D. Sample Size in Qualitative Interview Studies: Guided by Information Power. Qual. Health Res. 2016, 26, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Braun, V.; Clarke, V. Using Thematic Analysis in Psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Tong, A.; Sainsbury, P.; Craig, J. Consolidated Criteria for Reporting Qualitative Research (COREQ): A 32-Item Checklist for Interviews and Focus Groups. Int. J. Qual. Health Care J. Int. Soc. Qual. Health Care 2007, 19, 349–357. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V. Supporting Best Practice in Reflexive Thematic Analysis Reporting in Palliative Medicine: A Review of Published Research and Introduction to the Reflexive Thematic Analysis Reporting Guidelines (RTARG). Palliat. Med. 2024, 38, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Brownell, K.D.; Wadden, T.A. The Influence of the Stigma of Obesity on Overweight Individuals. Int. J. Obes. 2004, 28, 1333–1337. [Google Scholar] [CrossRef] [PubMed]
- Montali, L.; Zulato, E.; Frigerio, A.; Frangi, E.; Camussi, E. Mirroring, Monitoring, Modelling, Belonging, and Distancing: Psychosocial Processes in an Online Support Group of Breast Cancer Patients. J. Community Psychol. 2022, 50, 992–1007. [Google Scholar] [CrossRef]
- Onwuegbuzie, A.J.; Leech, N.L. A Call for Qualitative Power Analyses. Qual. Quant. 2007, 41, 105–121. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V. Reflecting on Reflexive Thematic Analysis. Qual. Res. Sport Exerc. Health 2019, 11, 589–597. [Google Scholar] [CrossRef]
- Hennink, M.; Kaiser, B.N. Sample Sizes for Saturation in Qualitative Research: A Systematic Review of Empirical Tests. Soc. Sci. Med. 2022, 292, 114523. [Google Scholar] [CrossRef] [PubMed]
- Bird, C.M. How I Stopped Dreading and Learned to Love Transcription. Qual. Inq. 2005, 11, 226–248. [Google Scholar] [CrossRef]
- Lapadat, J.C.; Lindsay, A.C. Transcription in Research and Practice: From Standardization of Technique to Interpretive Positionings. Qual. Inq. 1999, 5, 64–86. [Google Scholar] [CrossRef]
- Bispo Júnior, J.P. Social Desirability Bias in Qualitative Health Research. Rev. Saude Publica 2022, 56, 101. [Google Scholar] [CrossRef]
- Adams-Quackenbush, N.M.; Horselenberg, R.; Hubert, J.; Vrij, A.; van Koppen, P. Interview Expectancies: Awareness of Potential Biases Influences Behaviour in Interviewees. Psychiatry Psychol. Law 2019, 26, 150–166. [Google Scholar] [CrossRef]
- Sedgwick, P.; Greenwood, N. Understanding the Hawthorne Effect. BMJ 2015, 351, h4672. [Google Scholar] [CrossRef] [PubMed]
- Manoogian, E.N.C.; Chow, L.S.; Taub, P.R.; Laferrère, B.; Panda, S. Time-Restricted Eating for the Prevention and Management of Metabolic Diseases. Endocr. Rev. 2022, 43, 405–436. [Google Scholar] [CrossRef]
| Main themes | Sub-themes |
|---|---|
| Initial sources of motivation | Desperation |
| Scientific contribution | |
| Barriers |
Renunciation |
| Physical discomfort | |
| Friction with life | |
| Implementation mentality |
Predetermination |
| Perseverance | |
| Commitment | |
| Awareness | |
| Supporting factors |
Community |
| Flexibility | |
| Self-imposed rules | |
| Freedom from choice | |
| Implementation behaviour | Proactive facilitation |
| Adapt and overcome | |
| Sustainability |
Cost/benefit balance shift |
| Effect VS. decision fatigue | |
| Habit establishment |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




