Submitted:
19 February 2026
Posted:
20 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
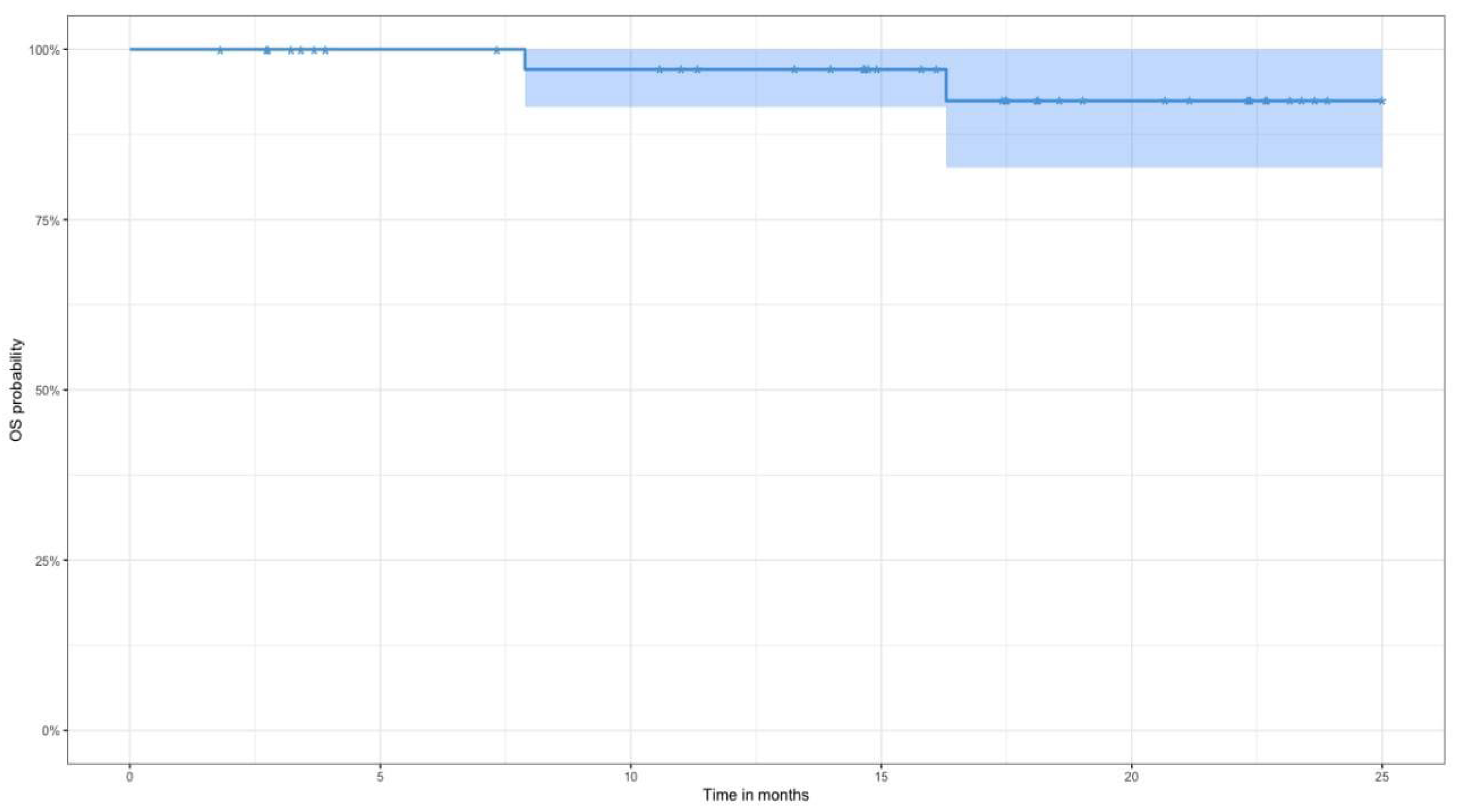
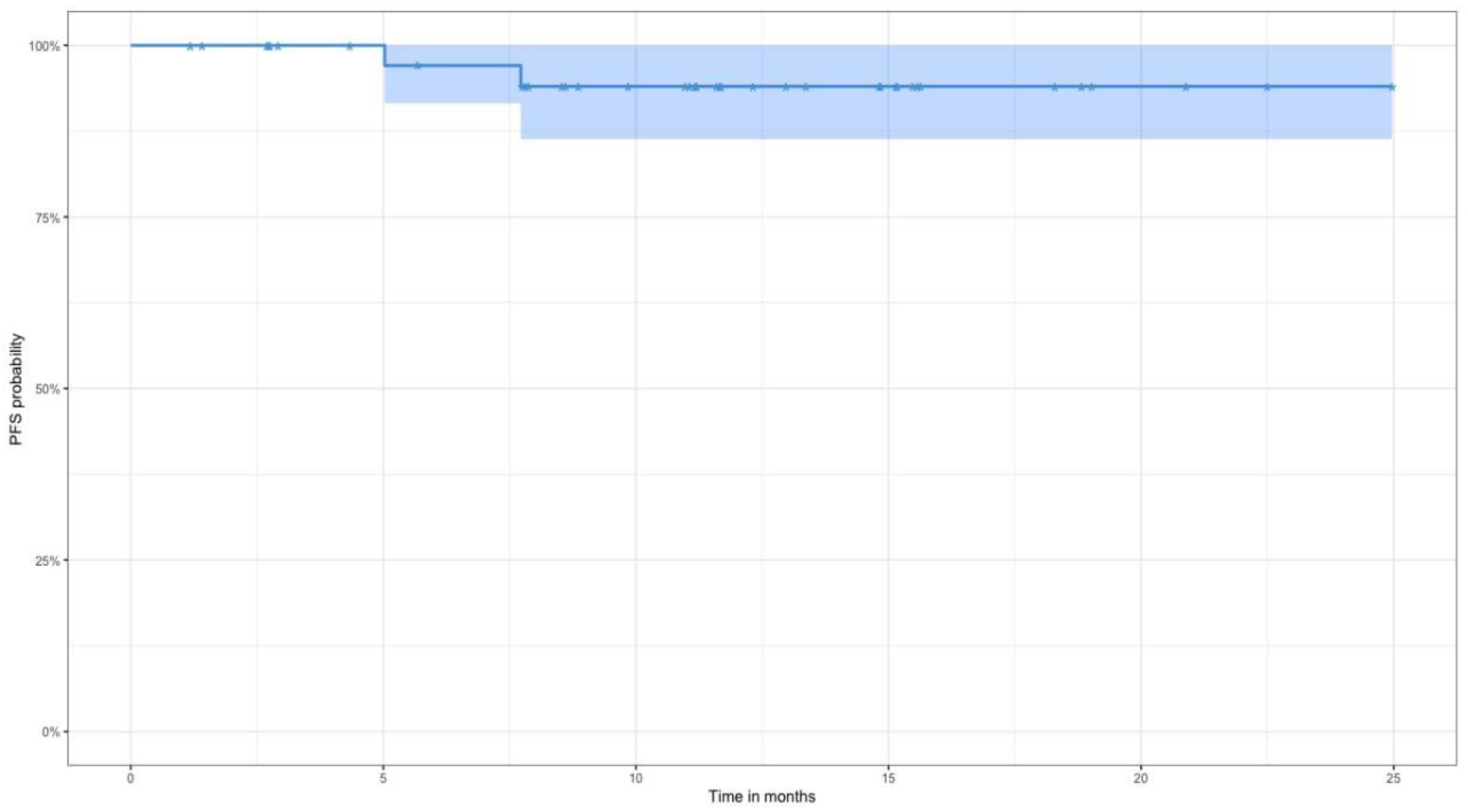
Discussion
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
References
- Hilton, L.K.; Scott, D.W.; Morin, R.D. Biological heterogeneity in diffuse large B-cell lymphoma. Semin. Hematol. 2023, 60, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, AD.; Araujo, IB.; De, O.; Berti, E.; Borges, A,M.; Boyer, D.; Calaminici, M. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36(7), 1720–48. [Google Scholar] [CrossRef] [PubMed]
- Campo, E.; Jaffe, E.S.; Cook, J.R.; Quintanilla-Martinez, L.; Swerdlow, S.H.; Anderson, K.C.; Brousset, P.; Cerroni, L.; de Leval, L.; Dirnhofer, S.; et al. The International Consensus Classification of Mature Lymphoid Neoplasms: a report from the Clinical Advisory Committee. Blood 2022, 140, 1229–1253. [Google Scholar] [CrossRef] [PubMed]
- Lacy, S.E.; Barrans, S.L.; Beer, P.A.; Painter, D.; Smith, A.G.; Roman, E.; Cooke, S.L.; Ruiz, C.; Glover, P.; Van Hoppe, S.J.L.; et al. Targeted sequencing in DLBCL, molecular subtypes, and outcomes: a Haematological Malignancy Research Network report. Blood 2020, 135, 1759–1771. [Google Scholar] [CrossRef] [PubMed]
- Wright, G.W.; Huang, D.W.; Phelan, J.D.; Coulibaly, Z.A.; Roulland, S.; Young, R.M.; Wang, J.Q.; Schmitz, R.; Morin, R.D.; Tang, J.; et al. A Probabilistic Classification Tool for Genetic Subtypes of Diffuse Large B Cell Lymphoma with Therapeutic Implications. Cancer Cell 2020, 37, 551–568.e14. [Google Scholar] [CrossRef]
- Chapuy, B.; Wood, T.; Stewart, C.; Dunford, A.; Wienand, K.; Khan, S.J.; Serin, N.; Wang, M.; Calabretta, E.; Shimono, J.; et al. DLBclass: a probabilistic molecular classifier to guide clinical investigation and practice in diffuse large B-cell lymphoma. Blood 2025, 145, 2041–2055. [Google Scholar] [CrossRef]
- Shi, Y.; Xu, Y.; Shen, H.; Jin, J.; Tong, H.; Xie, W. Advances in biology, diagnosis and treatment of DLBCL. Ann. Hematol. 2024, 103, 3315–3334. [Google Scholar] [CrossRef]
- Vodicka, P.; Klener, P.; Trneny, M. Diffuse Large B-Cell Lymphoma (DLBCL): Early Patient Management and Emerging Treatment Options. OncoTargets Ther. 2022, ume 15, 1481–1501. [Google Scholar] [CrossRef]
- Dabrowska-Iwanicka, A.P.; Nowakowski, G.S. DLBCL: who is high risk and how should treatment be optimized? Blood 2024, 144, 2573–2582. [Google Scholar] [CrossRef]
- Thieblemont, C.; Da Silva, M.G.; Leppä, S.; Lenz, G.; Cottereau, A.; Fox, C.; Lopez-Guillermo, A.; Illidge, T.; Jurczak, W.; Eich, H.; et al. Large B-cell lymphoma (LBCL): EHA Clinical Practice Guidelines for diagnosis, treatment, and follow-up. HemaSphere 2025, 9, e70207. [Google Scholar] [CrossRef]
- Fox, C.P.; Chaganti, S.; McIlroy, G.; Barrington, S.F.; Burton, C.; Cwynarski, K.; Eyre, T.A.; Illidge, T.; Kalakonda, N.; Kuhnl, A.; et al. The management of newly diagnosed large B-cell lymphoma: A British Society for Haematology Guideline. Br. J. Haematol. 2024, 204, 1178–1192. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health of Russia. Clinical guidelines for the diagnosis and treatment of aggressive B cell lymphomas (КР129_3). Available online: https://cr.minzdrav.gov.ru/view_cr/129_3 (accessed on 12.04.2025).
- Mingalimov, M.A.; Baryakh, E.A.; Misyurin, A.V.; Kesaeva, L.A.; Mkrtchyan, A.S.; Misyurina, E.N.; Tolstykh, T.N.; Orlova, M.S.; Akhmadeeva, A.A.; Chudnova, T.S.; et al. Personalized genotype-directed antitumor therapy for newly diagnosed diffuse large B-cell lymphoma: efficacy and toxicity of the R-CHOP-X protocol in a single-center, non-randomized, prospective clinical trial (first results). Oncohematology 2024, 19, 84–92. [Google Scholar] [CrossRef]
- Mingalimov, M.A.; Baryakh, E.A.; Tolstykh, T.N.; Misyurin, A.V.; Kesaeva, L.A.; Mkrtchyan, A.S.; Kovrigina, A.M.; Misyurina, E.N.; Orlova, M.S.; Chudnova, T.S.; et al. Personalized Approach to Treatment of Newly Diagnosed Diffuse Large B-Cell Lymphoma Patients with Respect to Molecular Landscape: A Case Report and Literature Review. Clin. oncohematology 2025, 18, 177–183. [Google Scholar] [CrossRef]
- Zhang, M.-C.; Tian, S.; Fu, D.; Wang, L.; Cheng, S.; Yi, H.-M.; Jiang, X.-F.; Song, Q.; Zhao, Y.; He, Y.; et al. Genetic subtype-guided immunochemotherapy in diffuse large B cell lymphoma: The randomized GUIDANCE-01 trial. Cancer Cell 2023, 41, 1705–1716.e5. [Google Scholar] [CrossRef]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A. Recommendations for initial evaluation, stag-ing, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef]
- Yoo, K.H. Staging and response assessment of lymphoma: a brief review of the Lugano classification and the role of FDG-PET/CT. BLOOD Res. 2022, 57, S75–S78. [Google Scholar] [CrossRef]
- Freites-Martinez, A.; Santana, N.; Arias-Santiago, S.; Viera, A. CTCAE versión 5.0. Evaluación de la gravedad de los eventos adversos dermatológicos de las terapias antineoplásicas. Actas Dermo-Sifiliograficas 2021, 112, 90–92. [Google Scholar] [CrossRef]
- Babicheva, LG; Poddubnaya, IV. Challenges and perspectives of first-line therapy in patients with diffuse B-cell lymphoma: A review. Journal of Modern Oncology (in Russian). 2023, 25(2), 178–184. [Google Scholar]
- Falchi, L.; Clausen, M.; Offner, F.; de Vos, S.; Brody, J.; Linton, K.M.; Snauwaert, S.; Cordoba, R.; Wu, J.; Bykhovski, I.; et al. Metabolic response rates of epcoritamab + R-CHOP in patients with previously untreated (1L) high-risk diffuse large B-cell lymphoma, including double-hit/triple-hit lymphoma: Updated EPCORE NHL-2 data. J. Clin. Oncol. 2023, 41, 7519–7519. [Google Scholar] [CrossRef]
- A study to evaluate change in disease activity in subcutaneous (sc) epcoritamab combined with intravenous and oral rituximab, cyclophosphamide, doxorubicin hydrochloride, vincristine and prednisone (R-CHOP) or R-CHOP in adult participants with newly diagnosed diffuse large B-cell lymphoma (DLBCL) (EPCORE DLBCL-2). ClinicalTrials.gov identifier: NCT05578976. Updated 14 Nov 2023. Accessed 17 Nov 2023. LINK (https://clinicaltrials.gov/study/NCT05578976).
- A phase II study of glofitamab + polatuzumab-R-CHP for patients with high-risk diffuse large B-cell lymphoma. ClinicalTrials.gov identifier: NCT05800366. 13 Sep 2023. Available online: https://clinicaltrials.gov/ct2/show/NCT05800366 (accessed on 17 Nov 2023).
- Dührsen, U.; Müller, S.; Hertenstein, B.; Thomssen, H.; Kotzerke, J.; Mesters, R.; Berdel, W.E.; Franzius, C.; Kroschinsky, F.; Weckesser, M.; et al. Positron Emission Tomography–Guided Therapy of Aggressive Non-Hodgkin Lymphomas (PETAL): A Multicenter, Randomized Phase III Trial; Annual Meeting of the German, Austrian, and Swiss Societies of Hematology and Medical Oncology. LOCATION OF CONFERENCE, COUNTRYDATE OF CONFERENCE; pp. 2024–2034.
- Assessing a ctDNA and PET-oriented therapy in patients with DLBCL. A multicenter, open-label, phase 2 trial. ClinicalTrials.gov identifier: NCT04604067. 18 Apr 2023. Available online: https://clinicaltrials.gov/ct2/show/NCT04604067 (accessed on 17 Nov 2023).
- Neelapu, S.S.; Dickinson, M.; Munoz, J.; Ulrickson, M.L.; Thieblemont, C.; Oluwole, O.O.; Herrera, A.F.; Ujjani, C.S.; Lin, Y.; Riedell, P.A.; et al. Axicabtagene ciloleucel as first-line therapy in high-risk large B-cell lymphoma: the phase 2 ZUMA-12 trial. Nat. Med. 2022, 28, 735–742. [Google Scholar] [CrossRef]
- Davies, A.; E Cummin, T.; Barrans, S.; Maishman, T.; Mamot, C.; Novak, U.; Caddy, J.; Stanton, L.; Kazmi-Stokes, S.; McMillan, A.; et al. Gene-expression profiling of bortezomib added to standard chemoimmunotherapy for diffuse large B-cell lymphoma (REMoDL-B): an open-label, randomised, phase 3 trial. Lancet Oncol. 2019, 20, 649–662. [Google Scholar] [CrossRef]
- Davies, A.J.; Barrans, S.; Stanton, L.; Caddy, J.; Wilding, S.; Saunders, G.; Mamot, C.; Novak, U.; McMillan, A.; Fields, P.; et al. Differential Efficacy From the Addition of Bortezomib to R-CHOP in Diffuse Large B-Cell Lymphoma According to the Molecular Subgroup in the REMoDL-B Study With a 5-Year Follow-Up. J. Clin. Oncol. 2023, 41, 2718–2723. [Google Scholar] [CrossRef]
- Younes, A.; Sehn, L.H.; Johnson, P.; Zinzani, P.L.; Hong, X.; Zhu, J.; Patti, C.; Belada, D.; Samoilova, O.; Suh, C.; et al. Randomized Phase III Trial of Ibrutinib and Rituximab Plus Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone in Non–Germinal Center B-Cell Diffuse Large B-Cell Lymphoma. 60th Annual Meeting of the American-Society-of-Hematology (ASH); LOCATION OF CONFERENCE, COUNTRYDATE OF CONFERENCE; pp. 1285–1295.
- Wilson, W.H.; Wright, G.W.; Huang, D.W.; Hodkinson, B.; Balasubramanian, S.; Fan, Y.; Vermeulen, J.; Shreeve, M.; Staudt, L.M. Effect of ibrutinib with R-CHOP chemotherapy in genetic subtypes of DLBCL. Cancer Cell 2021, 39, 1643–1653.e3. [Google Scholar] [CrossRef]
| Parameters | Number of patients, n (%) |
| Age, median, range (years) | 63 (38-78) |
| ECOG performance status ≥ 2 | 24 (54) |
| Sex - male | 15 (43) |
| IPI 3-5 | 28 (80) |
| Stage by Ann Arbor (Lugano modification, 2014) II III-IV |
14 (40) 21 (60) |
| Immunophenotype GCB nonGCB |
11 (32) 24 (68) |
| Co-expression of c-MYC/BCL2 | 1 (3) |
| «Single-hit» | 2 (7) |
| LymphGen: MCD N1 BN2 EZB A53 ST2 NOS |
2 (6) 11 (31) 2 (6) 5 (14) 0 (0) 3 (9) 12 (34) |
| Parameters | Number of patients, n (%) |
|---|---|
| Age, median, range (years) | 58 (60-78) |
| ECOG performance status ≥ 2 |
1 (12) |
| Sex - male |
4 (50) |
| IPI 3-5 |
3 (38) |
| Stage by Ann Arbor (Lugano modification, 2014) II III-IV |
4 (50) 4 (50) |
| Immunophenotype GCB nonGCB |
3 (38) 5 (62) |
| Co-expression of c-MYC/BCL2 | 1 (12) |
| «Single-hit» | 0 |
| LymphGen: MCD N1 BN2 EZB A53 ST2 NOS |
1 (12) 1 (12) 0 (0) 2 (26) 1 (12) 2 (26) 1 (12) |
| Drug | Dosage and rout of adminisrtation | Days of administration | LymphGen genotype |
| Rituximab | 375 mg/m2, intravenously | 0 | - |
| Doxorubicin | 50 mg/m2, intravenously | 1 | - |
| Vincristine | 1.4 mg/m2, intravenously | 1 | - |
| Cyclophosphamide | 750 mg/m2, intravenously | 1 | - |
| Prednisone | 60 mg/m2, intravenously | 1-5 | - |
| Acalabrutinib | 100 mg twice daily, orally | 1-21 | MCD, N1, BN2 |
| Lenalidomide | 25 mg once daily, orally | 1-10 | ST2, NOS |
| Vorinostat | 400 mg once daily, orally | 1-9 | EZB |
| Decitabine | 10 mg/m2, intravenously | 1-5 | A53 |
| Drug | Dosage and rout of adminisrtation | Days of administration | LymphGen genotype |
|---|---|---|---|
| Rituximab | 375 mg/m2, intravenously | 0 | - |
| Doxorubicin | 50 mg/m2, intravenously | 1 | - |
| Vincristine | 1.4 mg/m2, intravenously | 1 | - |
| Cyclophosphamide | 750 mg/m2, intravenously | 1 | - |
| Prednisone | 60 mg/m2, intravenously | 1-5 | - |
| Acalabrutinib | 100 mg twice daily, orally | 1-21 | MCD, N1, BN2 |
| Lenalidomide | 25 mg once daily, orally | 1-10 | ST2, NOS |
| Decitabine | 10 mg/m2, intravenously | 1-5 | A53, EZB |
| Outcome | Completed therapy (n = 35) | Ongoing therapy (n = 8) |
| Overall response | 35 (100) | 8 (100) |
| Complete response | 35 (100) | 8 (100) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




