Submitted:
22 February 2026
Posted:
23 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Equipment and Chromatographic Analysis Conditions
2.3. Preparation SiO2@NiO
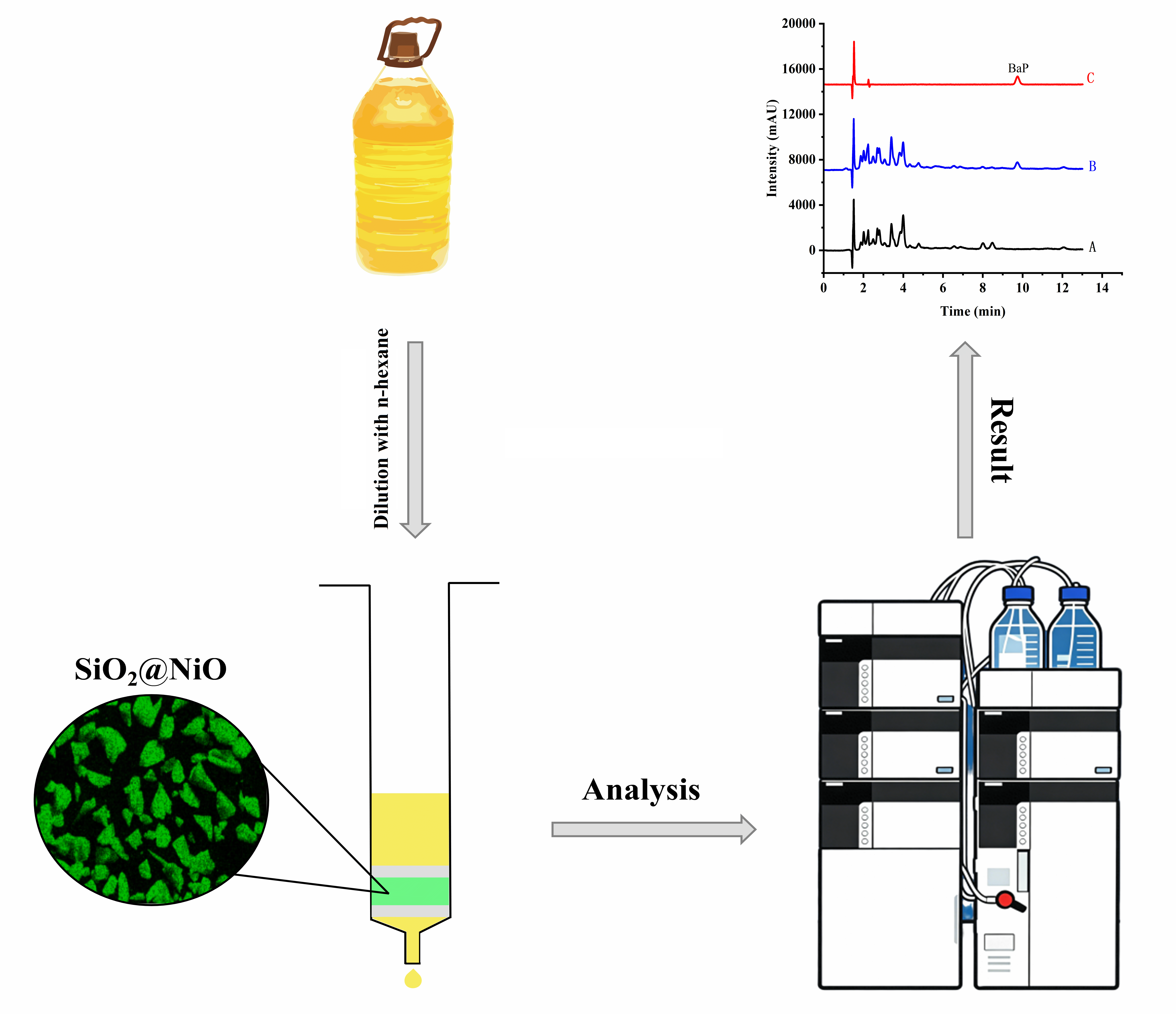
2.4. Sample Preparation
3. Results and Discussion
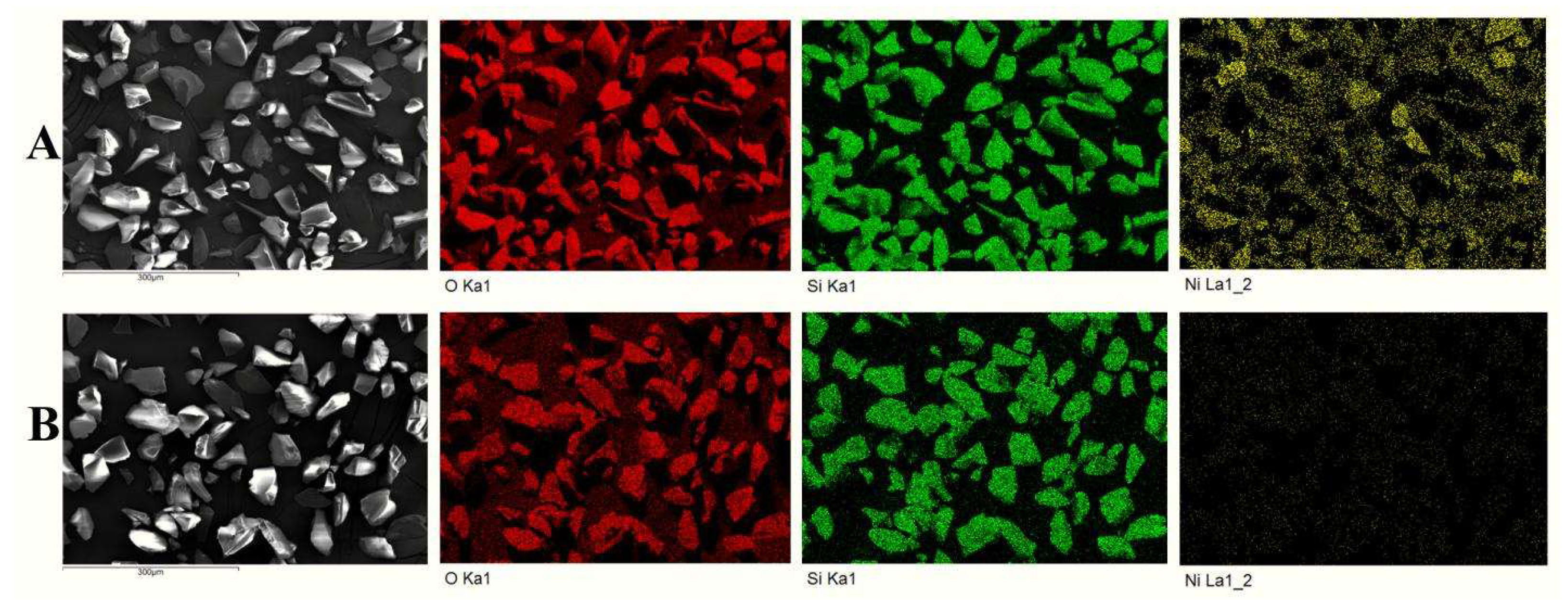
3.1. Characterization of SiO2@NiO
3.2. Optimization of SPE
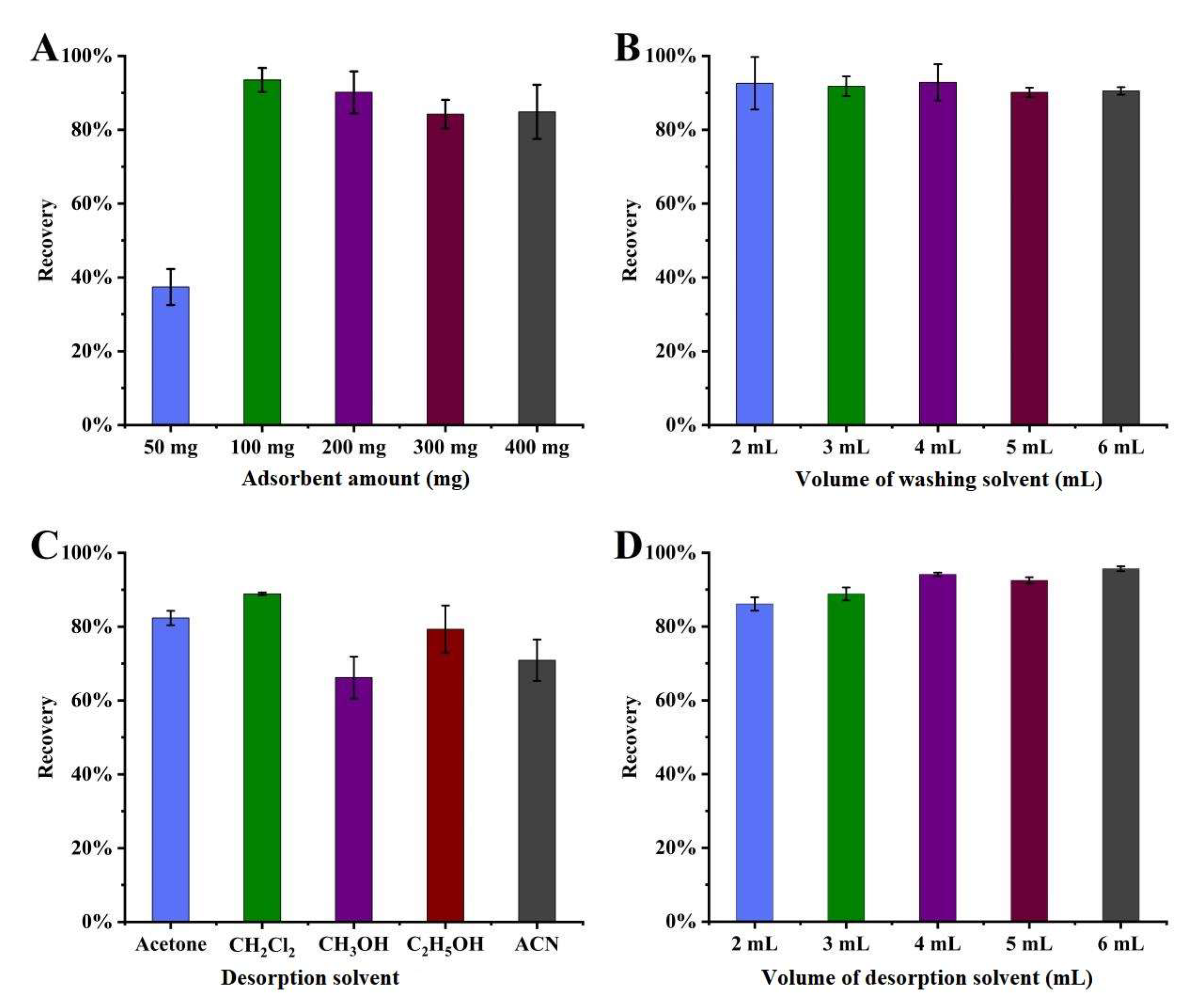
3.2.1. Influence of Adsorbent Amount
3.2.2. Influence of Washing Solvent Volume
3.2.3. Influence of Desorption Solvent
3.3. Comparison of BaP SPE on SiO2@NiO and SiO2
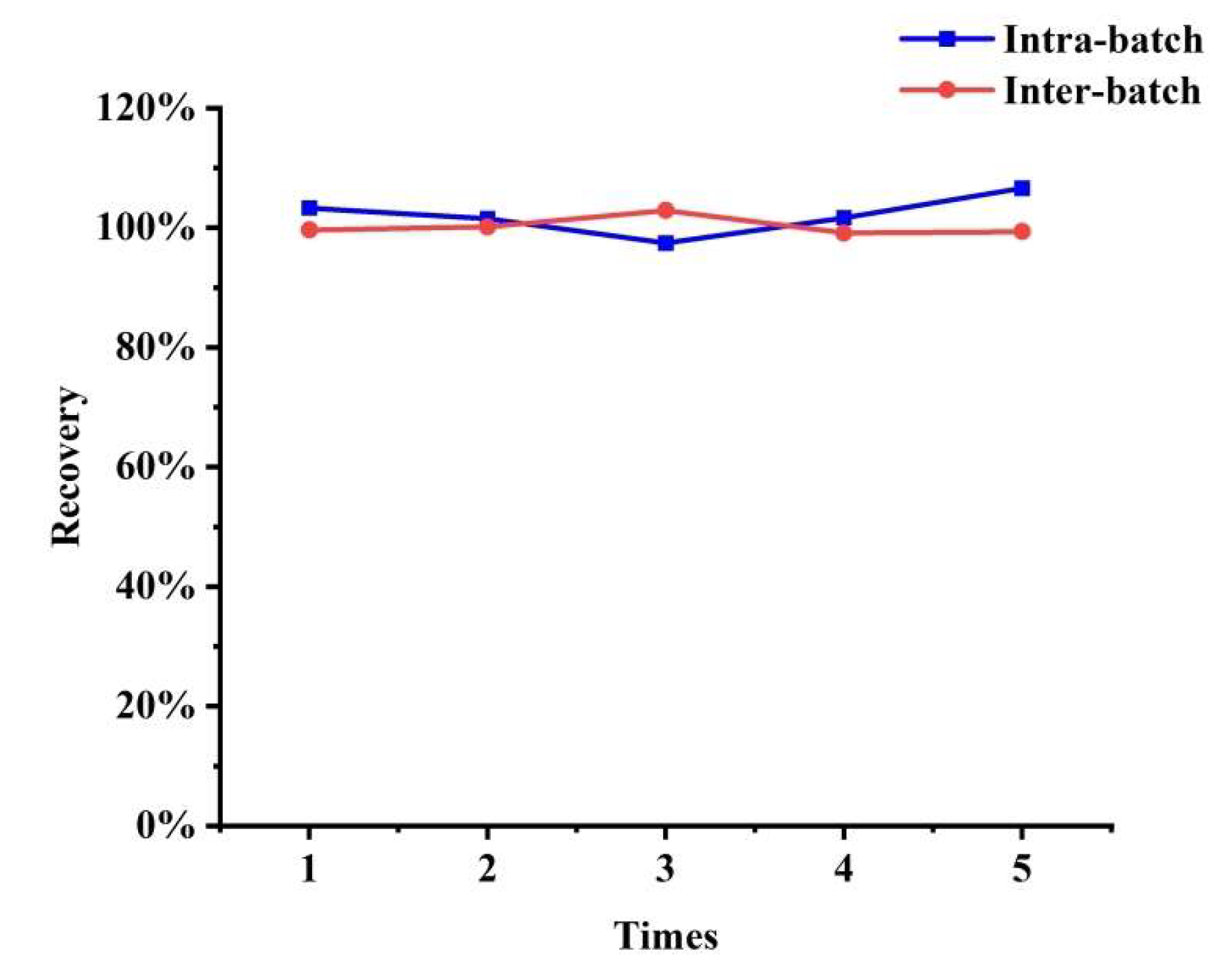
3.4. Reproducibility of SiO2@NiO
3.5. Method Validation
3.7. Comparison with Reported Methods
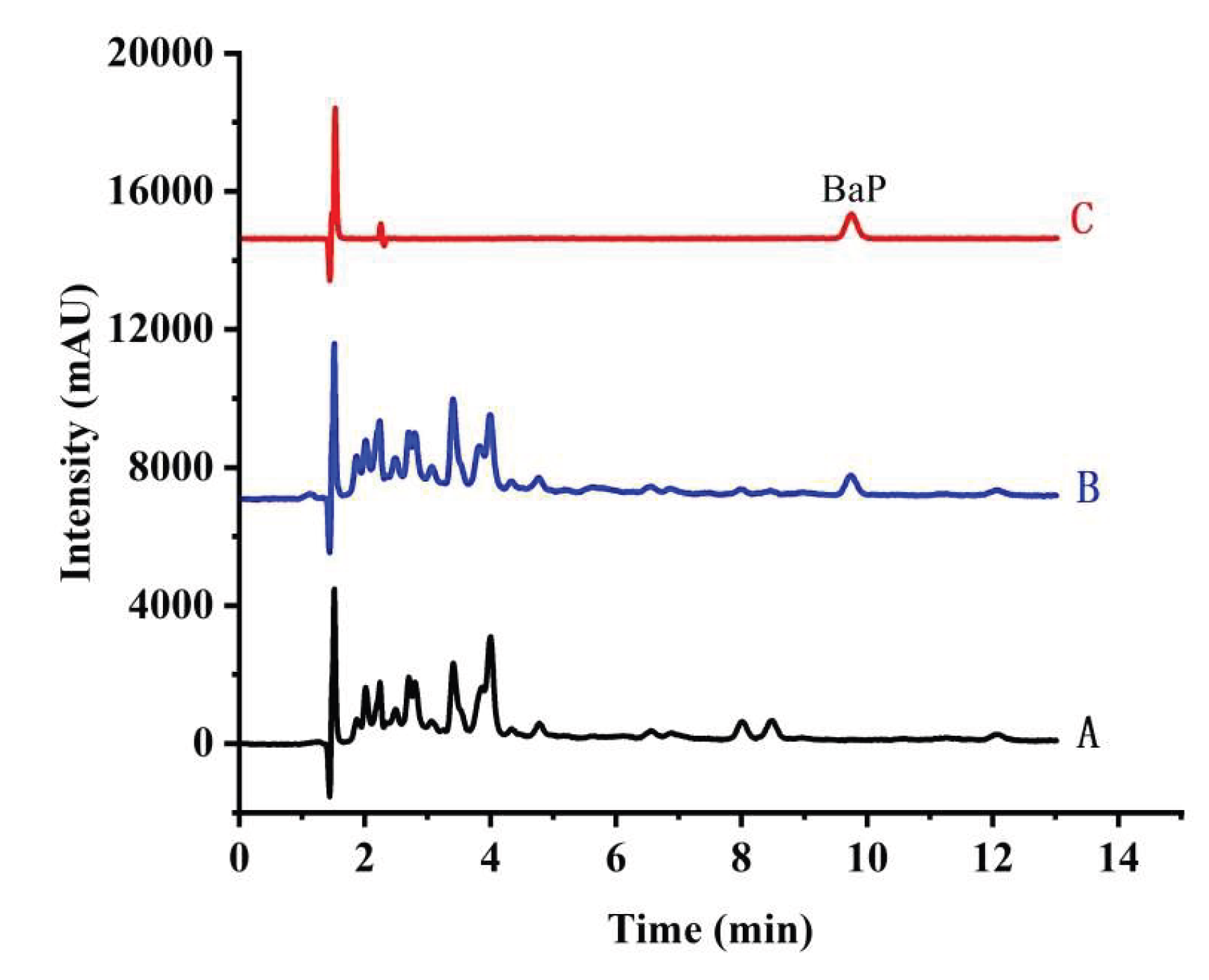
3.8. Real Samples Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jiang, Y.; Chen, X.; Yang, G.; Wang, Q.; Wang, J.; Xiong, W.; Yuan, J. BaP-induced DNA damage initiated p53-independent necroptosis via the mitochondrial pathway involving Bax and Bcl-2. Hum. Exp. Toxicol. 2013, 32, 1245–1257. [Google Scholar] [CrossRef] [PubMed]
- Hajiahmadi, Z.; Tavangar, Z.; Behzadi, H. A DFT study of the reaction between benzopyrene epoxide and C60 derivatives as possible anticancer activity. Polycycl. Aromat. Compd. 2019, 41, 593–603. [Google Scholar] [CrossRef]
- Li, Z.; Mu, L.; Peng, L.; Bai, H.L.; Liu, X.F.; Du, B. Correlation between the concentration of aromatic hydrocarbons and BaP from coke oven fugitive emissions in Shanxi, China. Aerosol Air Qual. Res. 2012, 12, 1373–1378. [Google Scholar] [CrossRef]
- Maier, M.L.V.; Siddens, L.K.; Pennington, J.M.; Uesugi, S.L.; Anderson, K.A.; Tidwell, L.G.; Tilton, S.C.; Ognibene, T; Turteltaub, J; Smith, K.W.; Williams, J.N. Benzo[a]pyrene (BaP) metabolites predominant in human plasma following escalating oral micro-dosing with 14C-BaP. Environ. Int. 2022, 159, 107045. [Google Scholar] [CrossRef]
- Wu, M.; Luo, J.M.; Huang, T.; Lian, L.L.; Chen, T.L.; Song, S.J.; Wang, Z.X.; Ma, S.X.; Xie, C.R.; Zhao, Y.; Mao, X.X.; Gao, H.; Ma, J.M. Effects of african BaP emission from wildfire biomass burning on regional and global environment and human health. Environ. Int. 2022, 162, 107162. [Google Scholar] [CrossRef]
- Yousefi, M.; Shemshadi, G.; Khorshidian, N.; Mohammadi, V.G.; Fakgri, Y.; Hosseini, H.; Khaneghah, A.M. Polycyclic aromatic hydrocarbons (PAHs) content of edible vegetable oils in Iran: A risk assessment study. Food Chem. Toxicol. 2018, 118, 480–489. [Google Scholar] [CrossRef]
- Chiou, A.; Kalogeropoulos, N. Virgin olive oil as frying oil. Compr. Rev. Food Sci. Food Saf. 2017, 16, 632–646. [Google Scholar] [CrossRef] [PubMed]
- Narayanankutty, A.; Illam, S.P.; Raghavamenon, A.C. Health impacts of different edible oils prepared from coconut (Cocos nucifera): A comprehensive review. Trends Food Sci. Technol. 2018, 80, 1–7. [Google Scholar] [CrossRef]
- Sekeroglu, G.; Gogus, F.; Fadiloglu, S. Determination of benzo(a)pyrene in vegetable oils by high performance liquid chromatography. J. Food Qual. 2007, 30, 300–308. [Google Scholar]
- Liu, X.F.; Zhang, Z.F.; Shen, M.Y.; Wu, Y.Y.; He, X.D.; Liang, L.; Zhang, J.X.; Xu, X.; Liu, G.Y. Optimization of the refining process for removing benzo(a)pyrene and improving the quality of tea seed oil. Eur. J. Lipid Sci. Technol. 2021, 124, 2100143. [Google Scholar] [CrossRef]
- Gao, P.; Zheng, Y.L.; Liu, H.; Yang, W.; Hu, C.R.; He, D.P. Effects of roasting and deodorisation on 3-monochloropropane-1, 2-diol esters, 3, 4-benzopyrene and trans fatty acids in peanut oil. Food Addit. Contam. Part A 2022, 39, 451–461. [Google Scholar] [CrossRef]
- Badger, G.M.; Novotny, J. Mode of formation of 3,4-benzopyrene at high temperatures. Nature 1963, 198, 1086. [Google Scholar] [CrossRef]
- Cheng, W.W.; Liu, G.Q.; Wang, X.D.; Liu, X.Q.; Liu, B.G. Formation of benzo(a)pyrene in sesame seeds during the roasting process for production of sesame seed oil. J. Am. Oil Chem. Soc. 2015, 92, 1725–1733. [Google Scholar] [CrossRef]
- Xu, Y.X.; Yang, J.J.; Liu, M.P.; Li, J.M.; Zhao, G.; Wang, X.H.; Li, J.; Peng, L.X.; Xiao, D. Sensitive determination of benzo(a)pyrene in vegetable oils based on the electrochemiluminescence quenching of ruthenium (II) dipyrido[3,2-a:2’,3’-c]phenazine complex. Microchem. J. 2023, 189, 108579. [Google Scholar] [CrossRef]
- Li, X.; Sun, C.L.; Xu, Y.; Shan, S.H.; Zheng, H.; Guo, X.L.; Hu, J.N. Construction of novel magnetic nanoparticles for enrichment of benzo(alpha)pyrene from edible oils followed by HPLC determination. Food Chem. 2022, 386, 132838. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.M.; Jiang, M.M.; Zhang, Y.X.; Hou, J.; Sun, S.D. Polycyclic aromatic hydrocarbons contamination in edible oils: A review. Food Rev. Int. 2022, 1, 1–27. [Google Scholar] [CrossRef]
- Liu, Y.H.; Wu, P.P.; Liu, Q.; Luo, H.D.; Cao, S.H.; Lin, G.C.; Fu, D.S.; Zhong, X.D.; Li, Y.Q. A simple fluorescence spectroscopic approach for simultaneous and rapid detection of four polycyclic aromatic hydrocarbons (PAH4) in vegetable oils. Food Anal. Methods 2016, 9, 3209–3217. [Google Scholar] [CrossRef]
- Peng, Y.; He, S.Y.; Wang, F.H.; Zheng, H.B.; Meng, Z. Determination of polycyclic aromatic hydrocarbons in edible oil by magnetic solid phase extraction based on a mesoporous molybdenum disulfide/graphite prior to gas chromatography-mass spectrometry. Microchem. J. 2022, 183, 108146. [Google Scholar] [CrossRef]
- Abdar, A.; Amiri, A.; Mirzaei, M. Semi-automated solid-phase extraction of polycyclic aromatic hydrocarbons based on stainless steel meshes coated with metal–organic framework/graphene oxide. Microchem. J. 2022, 177, 107269. [Google Scholar] [CrossRef]
- Payanan, T.; Leepipatpiboon, N.; Varanusupakul, P. Low-temperature cleanup with solid-phase extraction for the determination of polycyclic aromatic hydrocarbons in edible oils by reversed phase liquid chromatography with fluorescence detection. Food Chem. 2013, 141, 2720–2726. [Google Scholar] [CrossRef]
- Belo, R.F.C.; Nunes, C.M.; Santos, E.V.D.; Augusti, D.V. Single laboratory validation of a SPE method for the determination of PAHs in edible oils by GC-MS. Anal. Methods 2012, 4, 4068–4076. [Google Scholar] [CrossRef]
- Shi, L.K.; Zhang, D.D.; Liu, Y.L. Survey of Polycyclic Aromatic Hydrocarbons of Vegetable Oils and Oilseeds by GC-MS in China. Food Addit. Contam. Part A 2016, 33, 603–11. [Google Scholar] [CrossRef]
- Wang, S.L.; Hu, X.F.; Wu, W.Q.; Wang, D.; Li; Zhang, P.W. Dual-template magnetic molecularly imprinted polymers for selective extraction and sensitive detection of aflatoxin B1 and benzo(α)pyrene in environmental water and edible oil. Food Chem. 2024, 459, 140234. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.C.; Fung, L.C.; Hong, T.S.; Hong, T.H.; Sarah, L.; Niang, C.K. Acetic acid liquid-liquid extraction and UHPLC-DAD detection of polycyclic aromatic hydrocarbons in toasted and fried foods. J. Serb. Chem. Soc. 2025, 90, 943–956. [Google Scholar]
- Luo, D.; Yu, Q.W.; Yin, H.R.; Feng, Y.Q. Humic acid-bonded silica as a novel sorbent for solid-phase extraction of benzo[a]pyrene in edible oils. Anal. Chim. Acta 2007, 588, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhao, W.J.; Deng, Z.F.; Wang, H.B.; Peng, B.; Ma, X.; Lan, C.; Zhang, S.S. Determination of benzo[alpha]pyrene in edible oil using tetraoxocalix[2]arene[2]triazine bonded silica SPE sorbent. Food Addit. Contam. Part A 2018, 35, 1356–1365. [Google Scholar] [CrossRef]
- Yu, Q. W.; Feng, Y.Q. Application of liquid-phase deposition in analytical chemistry. Progress in chemistry. 2011, 23(6), 1211–1223. [Google Scholar]
- Yu, Q.W.; Ma, Q.; Feng, Y. Q. Temperature-response polymer coating for in-tube solid-phase microextraction coupled to high-performance liquid chromatography. Talanta 2011, 84(4), 1019–1025. [Google Scholar] [CrossRef]
- Sun, H.; Yu, Q.W.; He, H.B.; Lu, Q.; Shi, Z.G.; Feng, Y.Q. Nickel oxide nanoparticle-deposited silica composite solid-phase extraction for benzimidazole residue analysis in milk and eggs by liquid chromatography-mass spectrometry. J. Agric. Food Chem. 2016, 64, 356–363. [Google Scholar] [CrossRef]
- Yu, Q.W.; Liu, S.J.; Zheng, F.; Xiao, H.M.; Guan, H.Y.; Feng, Y.Q. Identification and quantification of benzimidazole metabolites of thiophonate-methyl sprayed on celery cabbage using SiO2@NiO solid-phase extraction in combination with HPLC-MS/MS. Chinese Chemical Letters 2020, 31, 482–486. [Google Scholar] [CrossRef]
- Zheng, D.; Hu, X.Z.; Fu, X.F.; Xia, Z.Z.; Zhou, Y.X.; Peng, L.J.; Yu, Q.W.; Peng, X.T. Flowerlike Ni-NiO composite as magnetic solid-phase extraction sorbent for analysis of carbendazim and thiabendazole in edible vegetable oils by liquid chromatography-mass spectrometry. Food Chem. 2022, 374, 131761. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.S.; Lee, N.; Park, J.; Kim, B.H.; Yi, Y.W.; Kim, T.; Kim, T.K.; Lee, I.H.; Paik, S.R.; Hyeon, T. Ni/NiO core/shell nanoparticles for selective binding and magnetic separation of histidine-tagged proteins. J. Am. Chem. Soc. 2006, 128, 10658–10659. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W. J.; Chen, X.B.; Fang, L.; Zhao, C.L. Determination of light–medium–heavy polycyclic aromatic hydrocarbons in vegetable oils by solid-phase extraction and high-performance liquid chromatography with diode array and fluorescence detection. J. Agric. Food Chem. 2013, 61, 1804–1809. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, H.; Zhang, Z.H.; Wu, X.L.; Chen, W.G.; Zhu, Y.; Fang, C.F.; Zhao, Y.G. Three-dimensional ionic liquid functionalized magnetic graphene oxide nanocomposite for the magnetic dispersive solid phase extraction of 16 polycyclic aromatic hydrocarbons in vegetable oils. J. Chromatogr. A 2017, 1489, 29–38. [Google Scholar] [CrossRef]
- Liu, X.F.; Jiang, Y.; Zhang, F.F.; Li, Z.Y.; Yang, B.C. Preparation and evaluation of a polymer-based sulfobetaine zwitterionic stationary phase. J. Chromatogr. A 2021, 1649, 462229. [Google Scholar] [CrossRef]
| Adsorbents | Specific Surface Area (m2/g) | Pore Volume (cm3/g) | Pore Size (nm) |
| SiO2 | 391.9 | 0.9 | 9.8 |
| SiO2@NiO | 396.4 | 0.9 | 9.6 |
| Analyte | Linear Range(ng/g) | Regression linear | LOD(ng/g) | LOQ(ng/g) | |
| Linear equation | R2 | ||||
| BaP | 6-1875 | Y =227.3147X-38.9316 | 0.9999 | 1.3 | 4.4 |
| Adsorbent | Detector | Recoveries(RSDs) | Advantages and drawbacks | Ref. |
| SPE (SiO2-OCA) |
HPLC-FLD | 88.9-122.3% (9.2%) |
Low LOD and effective, but tedious and demanding sorbent preparation | [26] |
| SPE (ProElut C18) |
HPLC-DAD-FD | 62.6-65.7% (4.9%) |
Wide application and practical but low recoveries, complex procedures, and expensive |
[33] |
| MSPE (3D-IL@mGO) |
GC-MS | 84.4-96.6% (7.9%) |
Fast and reusable, but expensive raw materials and tedious production | [34] |
| SPE (SiO2@NiO) |
HPLC-DAD | 97.4-105.1% (3.0%) |
Simple, economic adsorbent, effective, stable recoveries, and low cost | This work |
| Sample | Soybean oil |
Olive oil (1) |
Corn oil |
Olive oil (2) |
Linseed oil | Walnut oil | Sunflower oil | Peanut oil |
Unrefined oil (1) |
Unrefined oil (2) |
Fried oil |
|
Real concentration (ng/g, RSD%) |
N.D. | N.D | N.D | N.D | N.D | N.D | N.D | N.D | 13.1 (1.8) |
8.3 (14.5) |
8.6 (7.1) |
|
Found concentration* (ng/g, RSD%) |
11.1 (8.4) |
10.5 (5.1) |
9.9 (10.7) |
9.9 (2.0) |
10.8 (5.4) |
10.9 (1.7) |
9.7 (9.3) |
9.5 (3.3) |
22.9 (0.6) |
18.4 (6.4) |
17.2 (1.8) |
| Recovery (%) | 111.0 | 104.9 | 99.3 | 99.1 | 108.9 | 109.6 | 97.6 | 95.0 | 99.2 | 103.0 | 87.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




