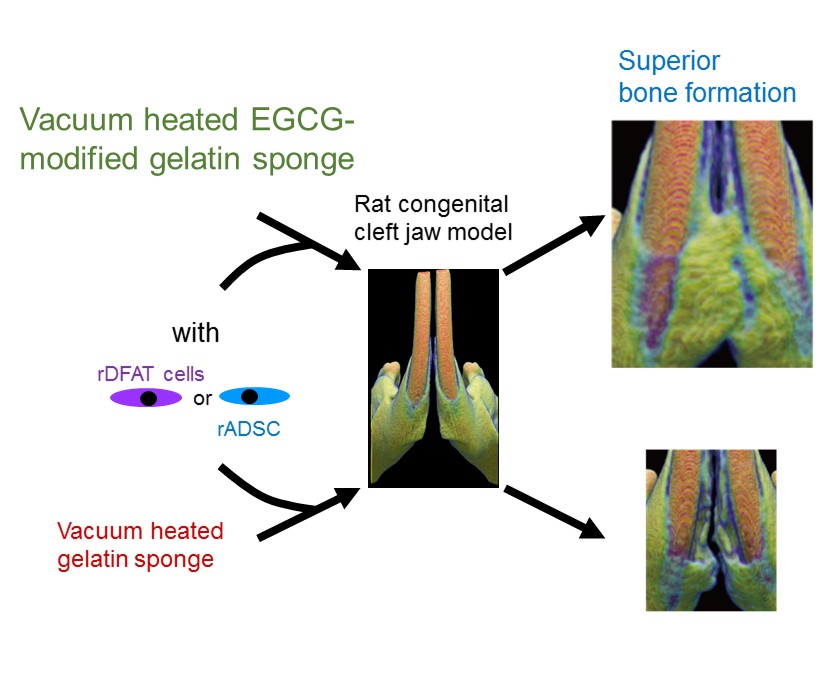
Cost-effective and functionalized scaffolds are in high demand for stem-cell-based regenerative medicine to treat refractory bone defects in craniofacial abnormalities and injuries. One potential strategy is to utilize pharmacological and cost-effective plant polyphenols and biocompatible proteins, such as gelatin. Nevertheless, the use of chemically modified proteins with plant polyphenols in this strategy has not been standardized. Here, we demonstrated that gelatin chemically modified with epigallocatechin gallate (EGCG), the major catechin isolated from green tea, can be a useful material for dedifferentiated fat cells and adipose-derived stem cells and can induce bone regeneration in a rat congenial cleft-jaw model in vivo. Vacuum-heated gelatin sponge modified with EGCG (vhEGCG-GS) induced superior osteogenesis from these two cell types compared with vacuum-heated gelatin sponge (vhGS). The EGCG-modification converted the water wettability of vhGS to a hydrophilic property (contact angle: 110° to 3.8°) and the zeta potential to a negative surface charge; the modification enhanced the cell adhesion property and promoted calcium phosphate precipitation. These results suggest that the EGCG-modification with chemical synthesis can be a useful platform to modify the physicochemical property of gelatin. This alteration is likely to provide a preferable microenvironment for multipotent progenitor cells, inducing superior bone formation in vivo.