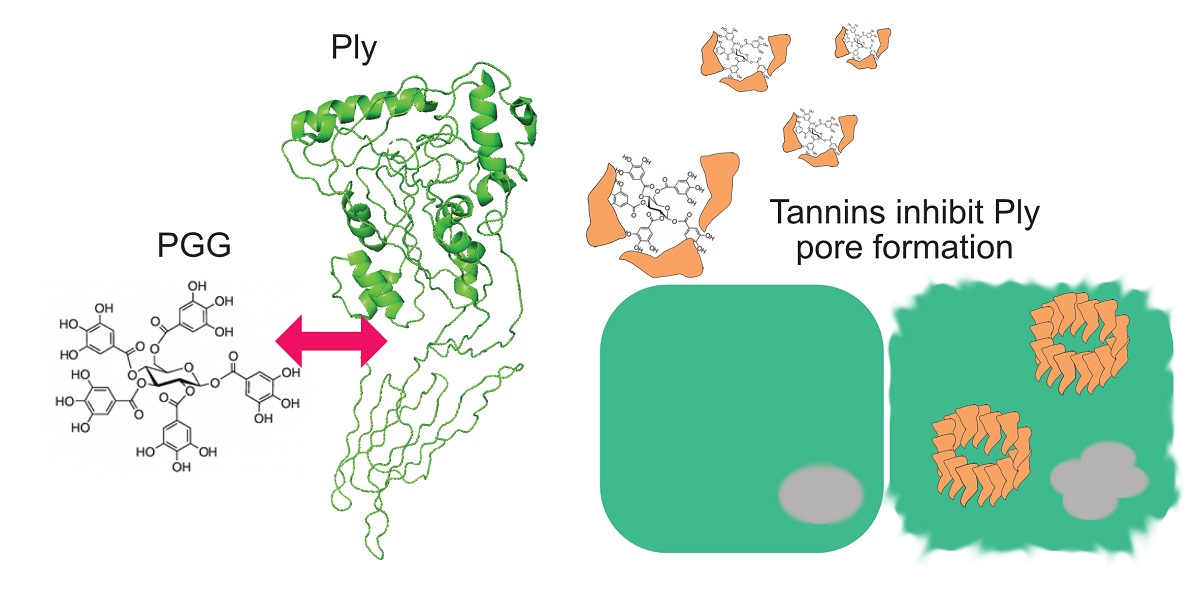
Streptococcus pneumoniae causes invasive infections such as otitis media, pneumonia and meningitis. It produces pneumolysin (Ply) toxin, which forms a pore to host cell membrane and has multiple functions in S. pneumoniae pathogenesis. The Ply C-terminal domain 4 mediates binding to membrane cholesterol and induces the formation of pores composed of up to 40 Ply monomers. Ply has a key role in the establishment of nasal colonization, pneumococcal transmission from host to host and pathogenicity. Altogether, 27 hydrolyzable tannins were tested for Ply inhibition in hemolysis assay and tannin-protein precipitation assay. Pentagalloylglucose (PGG) and gemin A showed nanomolar inhibitory activity. Ply oligomerization on the erythrocyte surface was inhibited with PGG. PGG also inhibited Ply cytotoxicity to A549 human lung epithelial cells. Molecular modelling of Ply interaction with PGG suggested that it binds to the pocket formed by domains 2, 3 and 4. In this study, we reveal the structural features of hydrolyzable tannins that are required for interaction with Ply. Monomeric hydrolysable tannins containing a sufficient number of polyvalent and flexible galloyl groups have highest inhibitory power to Ply cytotoxicity, and are followed by oligomers. Of the oligomers, macrocyclic and C-glycosidic structures were weaker in their inhibition than the glucopyranose-based oligomers. Accordingly, PGG-type monomers and certain oligomers might have therapeutic value in the targeting of S. pneumoniae infections.