1. Introduction
The quality of drinking water plays a significant role in the quality of health of a populace. Therefore, water that is meant for consumption should undergo various treatment stages (from source to consumer) to qualify as safe for humans and animals. With an increase in anthropogenic activities around the globe, many source-water bodies have been exposed to one level of contaminant or another [
1]. These contaminants could be naturally occurring compounds discovered during exploratory activities for diverse applications in our environment and introduced to source-water bodies via runoff or groundwater infiltration [
2]. In other cases, groundwater contamination may occur from underground man-made structures or naturally occurring elements and gases in the earth’s core [
3]. Regardless of the primary water source, it is likely that there is one form of contamination or another that makes treatment a necessity to make water fit for human consumption[
4,
5,
6].
In West African countries like Nigeria and Ghana, water is packaged in sachets as a low-cost option compared to bottled water [
7,
8]. Sachet water (SW) also serves as a source of drinking water for on-the-go commuters and other consumers. It is widely used in households with no access to clean drinking water sources. Producers of packaged water obtain water from surface water bodies and groundwater wells [
9,
10], hence it is important to investigate the quality of water before packaging and after sales to retailers or direct consumers to mitigate health risks posed by the consumption of untreated or poorly treated water. Packaged water is generally perceived to be treated water that is safe for drinking, however, studies have shown that public perception and water quality are not always directly related [
7,
11,
12,
13]. In a 2017 study conducted to determine and compare the quality of packaged water in different regions, it was discovered that 65 percent of the representative samples tested were unimproved i.e., contaminated. This result applied to both forms of packaged drinking water available – sachets and bottles [
7]. Other studies have attempted to determine contamination levels of packaged drinking water in different parts of Nigeria and Ghana and have found the presence of heavy metals, and microbial contamination [
10,
14,
15,
16].
To reduce the exposure of humans to water-borne illnesses, it is essential to monitor the source quality of drinking water. In the case of civilizations with decentralized municipal water supply, regular monitoring of water bodies would be beneficial to preventing outbreaks and health hazards [
17,
18,
19]. The detection of pathogens and contaminants in source water is not sufficient to prevent outbreaks, hence proper treatment must be ensured. In many regions where SW is consumed, there is a lack of regulatory enforcement of drinking water quality. Hence, private companies producing SW are in control of deciding the source of water used in production processes and the extent of water treatment before packaging [
9]. In many cases, pretreatment tests are not conducted to determine the nature of pollutants in the water. Often, chlorination is applied before water it is packaged and as such, many contaminants go untreated, and very low levels of residual chlorine remaining in the packaged water. Another challenge with the quality of SW is storage and method of sale. Once the SW has been packaged in the factory, it is often transported in open trucks that allow exposure to sunlight, some vendors store the bags of water in outdoor cages that allow sunlight exposure while others hawk the SW in bowls containing ice and allowing sunlight exposure. It is a known fact that chlorine concentration in water can reduce by 90% in two hours when exposed to sunlight. Sunlight exposure to SW has also been linked to increased contaminant concentrations post-packaging [
20,
21,
22].
A recent study by [
22] exposed 12 SW packs to sunlight over a period of 14, 28, and 48 days and analyzed the samples for changes in physico-chemical properties – hardness, TDS, pH, EC and turbidity, heavy metal concentration, microbial growth, and antibiotic susceptibility. The study found that all physico-chemical parameters increased after sunlight exposure except pH which reduced. Also, metal concentrations and bacterial coliform increased. The studies presented above showed that the presence of bacterial contaminants in SW can exist due to poor quality source water, inadequate treatment processes, or improper handling after packaging. Considering the above, this study aims to investigate the physico-chemical and microbial quality of SW sold in three busy neighborhoods in Ota, Ogun State, and to determine if the public perception of brand quality matches the results obtained from laboratory tests. The study also includes statistical and communality tests to identify the specific sources of bacteriological contamination. This information will add to the body of knowledge in Nigeria regarding sachet water and will also inform and/or reiterate to the various agencies the seeming plights surrounding SW consumption in Nigeria.
2. Materials and Methods
2.1. Study Area and Sampling Procedure
The sampling of SW took place in the Ado-Odo community (6.6117° N, 3.0576° E) of Ota in Ogun State, Nigeria. As of the 2006 population census, the community has about 526,565 residents living in the region. While we expect this number to have increased beyond this due to heavy rural-urban migration, this value represents an estimate as of the time of sampling. It consists of both rural and urban communities and has a large concentration of industries. The presence of industries in the area poses a challenge to water quality due to illicit effluent dumping in surface water bodies and near groundwater recharge zones [
23].
The study was centered on the bacteriological quality of SW sold and consumed in the community. SW samples were collected between November and December 2017. The sampling took place in Ado-Odo, Ota in Ogun State because the region receives about 3 million people yearly concentrating around Canaan-land for numerous Christian activities. During this period, it is envisaged that there will be an increased need for SW. Therefore, SW producers may decide to compromise on water treatment quality to meet up the demand. Canaan-land and its environs constitute low to medium-income households, however irrespective of the household income category, almost everyone depends on SW to meet drinking water demands. Generally, SW-producing companies source water from underground wells (usually termed boreholes) having very low water tables which can easily allow for pollutants if not properly protected and situated [
2,
3,
4]. Previous studies investigating groundwater quality in the region showed that the water posed no major health risk to consumers based on physico-chemical characteristics. However, bacteriological tests were not performed thus resulting in a partial view of the quality of groundwater as source water for SW producers in the region [
24]. Major rivers in the region have been sampled and have shown high levels of impairment by pathogens and heavy metals [
23,
25,
26,
27]
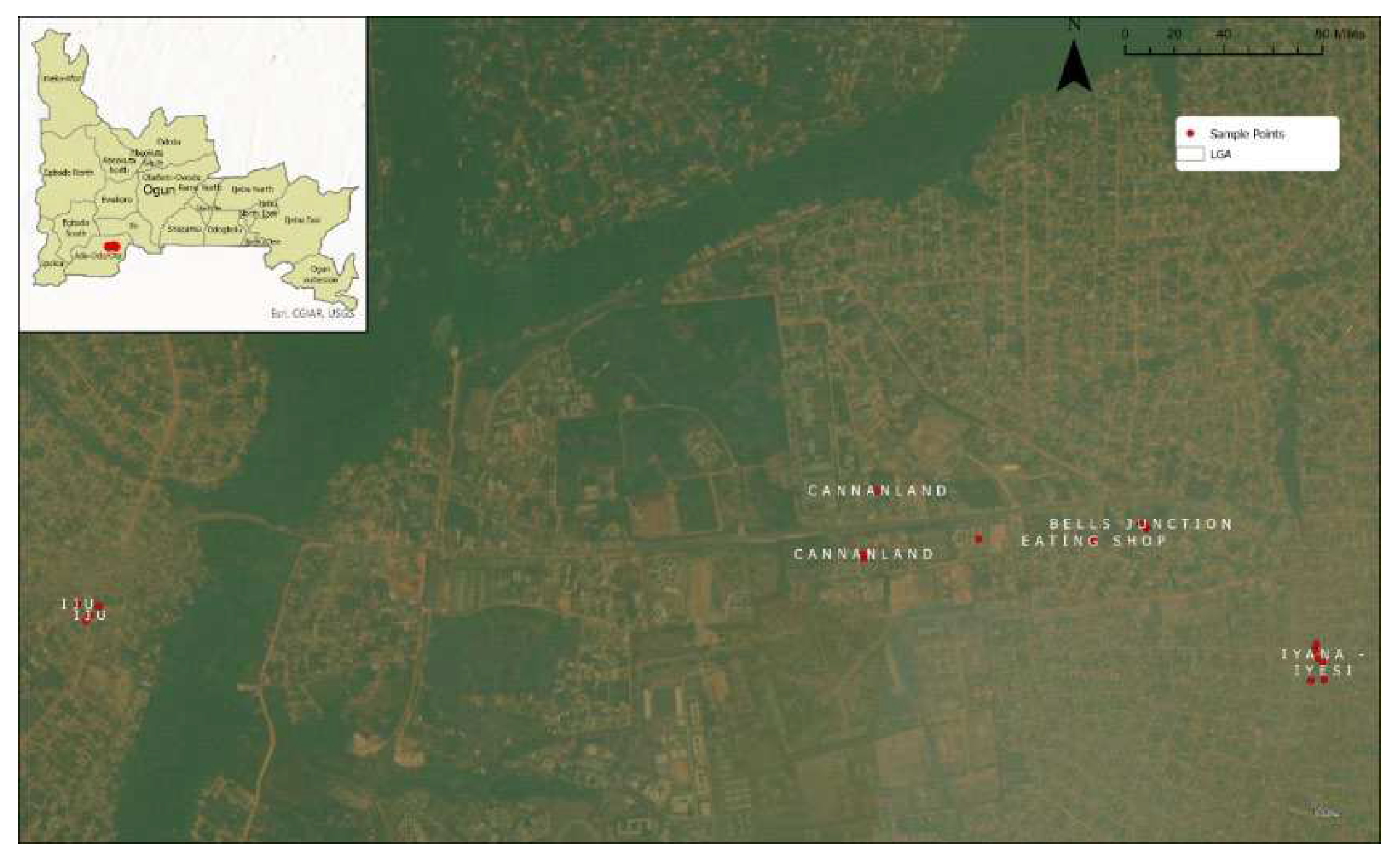
In this study, eighty (80) SW of different brands were purchased around Canaan land, the meeting place for most Christians during the November - December period. The areas from which these brands were purchased were Iyana Iyesi, Elegushi, and Igbogbo area (
Figure 1). During the sampling periods, these SW samples were purchased from either a store or a kiosk. According to [
15], eliminating samples from hawkers i.e., those who move from one point to another, was required to eliminate weather variability such as sunlight, that can impact the water quality. In this study area, we observed two distinct sets of SW producers. The first conforms to producers who have large orders and thus produce more SW while the second set of producers have fewer orders or are less preferred by SW consumers. This a priori knowledge aided our sampling design. With this information, we ensured that we sampled a mixture of SW not exceeding 1 week old. We also ensured that during the sampling period, SW produced on the day of sampling was also purchased. Similarly, we categorized SW based on the preference of consumers, storage duration, and packing quality to gain more insight into how significantly these categories can affect consumers. This further informed the study of two similar SW groupings (good or poor SW brands). Irrespective of the income level of the consumers, we noticed that cheaper and larger packets of SW gain more patronage, thus assuming to the consumers that all SW is the same by constantly reciting the parlance that “water is water” as “water has no enemy”.
Furthermore, SW collected were immediately kept in an iced cooler and transported to the laboratory for some physico-chemical and bacteriological testing. Generally, it was ensured that all tests were conducted within the first 24 hours of purchase and samples were kept in ice at a constant temperature of 4 °C before bacteriological testing.
2.2. Physico-Chemical Analysis
The physico-chemical parameters measured include pH, Total Dissolved Solids (TDS), Salinity, and Electrical Conductivity (EC). It is worth mentioning that all physico-chemical tests were carried out using Hanna Edge Multi-parameter Dissolved Oxygen Meter (HI2040) and were calibrated based on the manufacturers' description using appropriate calibration solutions. The results obtained from these tests were useful in assessing the physical characteristics of the samples collected and the impact on the growth of organisms as well as giving more insight as to the likely sources of contaminants. The results were also useful in determining the suitability of the samples collected for drinking and other forms of consumption per WHO guidelines for drinking water quality.
2.3. Bacteriological Analysis
Bacteriological testing was conducted for the detection of
E. coli, Total Heterotrophic bacteria, Total Coliform, and Staphylococcus Aureus. The Multiple Tube Fermentation Test was used to evaluate the number of active bacteria specie present in all samples collected. The total coliform count in SW samples was determined using the Most Probable Number (MPN) assay. This test is also known as the presence or absence test since it focuses on detecting the presence or absence of coliform bacteria in a sample. This test is based on the premise that for every 100 mL of drinking water, there should be no coliform bacteria present. Also, if one viable cell of coliform bacteria is present in the sample being tested, it will reproduce to give a population of lactose fermenting cells that produce acid and gas [
28,
29].
2.3.1. Most Probable Number Assay
The MPN Assay is widely used for the estimation of viable microbes in a known quantity of samples (food or water) by creating replicates of liquid broth growth in ten-fold dilutions [
30,
31]. This process involves filling the replicates into tubes and is inoculated them with varying amounts of sample. A change in color, cloudy appearance, and or gas production during incubation indicates the growth of microbes. Based on statistical probabilities, the number of viable microbes in the original sample is approximated using an MPN table [
32,
33].
The first step of the MPN tests is the presumptive test which serves as preliminary screening to determine the presence of microorganisms in the sample. A negative presumptive test indicates that the sample being tested is microbiologically fit for consumption and as such no other screening is required. A presumptive test is considered positive when there is an appearance of gas, color change, or cloudy appearance in the test tubes post-incubation. This requires further screening to identify specific microbial contaminants to inform appropriate treatment processes [
30]. In this study, all samples were considered untreated and the presumptive test for untreated water was applied.
The MacConkey broth was prepared in single and double-strength concentrations following standard procedures [
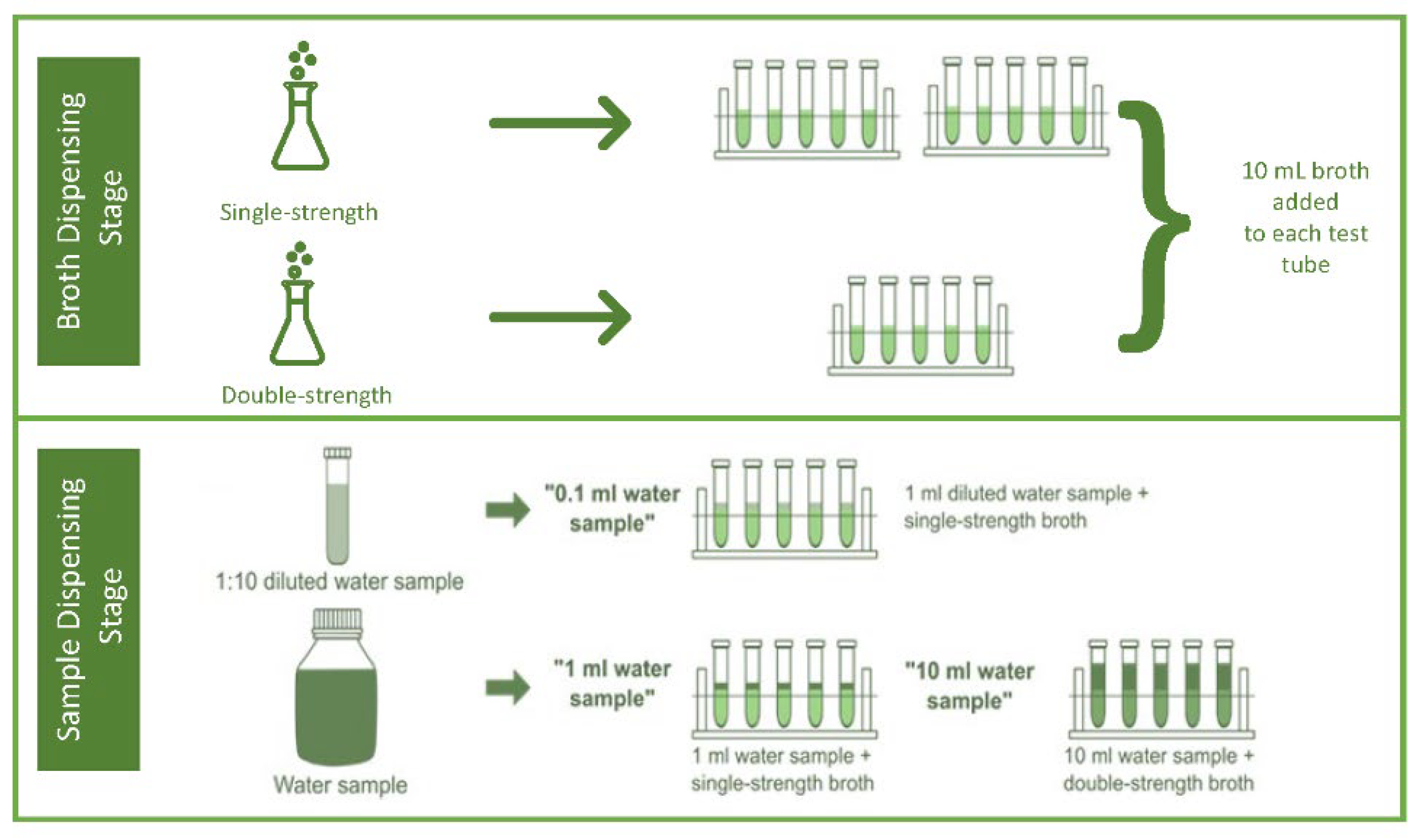
31]. Following this, the prepared double-strength broth was dispensed using a sterile pipette into five test tubes containing 10 ml in each tube and ten fermentation test tubes containing 10 mL of single-strength broth. A Durham tube was then added to each test tube in an inverted position while ensuring that no air bubbles were present in the Durham tubes before sterilizing in an autoclave for 15 mins at 121 °C. Following this process, with the aid of a sterile pipette, 10 mL of each sample was added to the five fermentation test tubes containing the double-strength concentration broth, 1 mL of the sample to five fermentation test tubes with single-strength concentration broth, and 0.1 mL of sample to the other five fermentation test tubes with single strength concentration broth (
Figure 2). The resulting solution was incubated at 37°C for 24 hrs. The fermentation tubes were inspected for microbial growth after 24 hours. Fermentation tubes with no indication of microbial growth were incubated for an additional 48 hrs. [
34]. Once the reaction time was completed, the number of fermentation tubes indicating microbial growth per sample was compared to the standard MPN charts and the number of microbes present was recorded. The results give the estimated number of coliforms present per 100 mL of sample.
Following the presumptive tests, the fermentation tubes that produced gas were inoculated on a variety of agar mediums. A loopful of a sample from each positive inoculum was transferred to selective media. The pour-plate method was used for these tests. The streak-plate method was used to isolate pure cultures on eosin methylene blue agar plates, nutritional agar plates, and MacConkey agar, which were incubated at 37 ºC for 24 hrs. For the detection of thermotolerant
E. coli, high-temperature incubation at 44.5 ±0.2 ºC was used. The colonies that grew on each agar were then counted and recorded. Most of the results for the first set of samples yielded colonies too numerous to count. This prompted the use of serial dilution in the second and third tests of the samples before plating and incubation. After incubation was complete, the plates were physically and microscopically examined for the formation of colonies [
35].
3. Results
3.1. Contamination Dominance and Brand Reputation
The 80 samples tested in this study were classified into brands of good and poor reputation. Of the 80 samples, sixty-nine (69) were placed in the category of SW brand with poor reputation while eleven (11) samples were placed under good reputation SW brand. The quantity of SW classified as good or bad based on public perception as well as the quantity of SW based on the time of purchase after production is recorded in
Table 1. 9 samples (100%) of SW containing THB were of good reputation and 67 samples (94.39%) were brands of the poor reputation and these THB were above the threshold (χ2 = 0.5337; p = 0.465) statistical significance. None of the perceived good brands of SW contained E. coli while 51(73.91%) of the perceived poor brands had E. coli present and were statistically significant. (χ2 = 5.766; p = 0.016). In addition, no SW from good reputation brands had TC present while 29 SW samples of poor reputation brands recorded TC values with statistical significance (χ2 = 2.773; p = 0.096).
Table 1 highlights the values of THB, E. coli, TC, and Staph based on the above-mentioned classifications.
Only one brand from a poor reputation SW brand reported staphylococcus, which overall was not statistically significant (χ2= 0.1284; p = 0.720). Surprisingly, some samples collected on the day of production reported the presence of THB, TC, and E. coli. As seen in
Table 1, THB values were present from 31 (93.84%) SW made on the same day while 45 (95.74%) were seen from SW samples stored over time with no significant statistical difference between them (χ2 = 0.1330; p = 0.713). this simply suggests that storing them over time was not a reason for the presence of THB, rather these bacteria were inherent probably in the distribution system of water supply. Similarly, 17 (51.2%) SW had both TC and E. coli and were produced on the same day while more samples 34 (72.24%) SW stored over time had a significantly high value of (χ2= 5.664; p = 0.77) for TC.
From the three (3) sampling cluster locations, 23 (100%) SW samples from Iyana Iyesi, 41 SW samples (53.85%) from Elegushi, and 7 SW (53.85%) from Igbogbo were classified as the brand of perceived good reputation. Of these SW of good reputation, 6 SW were purchased from Iyana Iyesi (26.09%), 21 SW (47.73%) from Elegushi, and 6 (46.15%) from Igbogbo were purchased on the day of production with a statistically significant difference between sets of SW purchased (
p < 0.05). This suggests that perceived brands of SW of poor reputation were sold in Iyana Iyesi and Elegushi combined. Fifty-two SW samples generally had
E. coli. Specifically, 9 SW from Iyana Iyesi (34.78%), 30 SW (68.18%) from Elegushi, and all 13 (100%) samples from Igbogbo with a strong statistical difference (
p = 0.001) with mean count values of
,
and
respectively for these locations with no statistically significant difference (
p>0.05). These values exceeded the world health organization's (WHO) limits of
E. coli of zero tolerance. THB values significantly higher than 500 cfu/ml were recorded from the three locations. Specifically, 21 SW (91.38) from Iyana Iyesi, 42 SW (95.45%) from Elegushi, and all 23 (100%) samples from Igbogbo with no statistically significant difference (χ2= 1.365,
p = 0.505). In addition, mean E. coli, TC, THB, and physicochemical values were estimated and recorded in
Table 2.
The mean values recorded revealed that THB values from these locations exceeded the threshold for THB having , and for each location respectively but showing a significant statistical difference (p = 0.034). TC on the other hand was not different as a significant number of SW samples had TC present. To mention, 13 SW (56.52%) from Iyana Iyesi, 15 SW (34.09%) from Elegushi, and 1 SW (7.69%) from Igbogbo with a significant statistical difference (χ2 = 8.7665, p = 0.012) between TC present in SW in the three locations. Staphylococcus was present in only one sample and was purchased from Igbogbo which may have been due to chance (χ2 = 5.219, p > 0.05). In addition, physio-chemical properties measured for all SW samples from the three locations revealed no statistical difference between them except for pH which was majorly recorded. Electrical conductivity which can be a measure of the ionic content in water showed no statistical difference between SW obtained at different locations (p = 0.8978) with mean values of 48µs/cm, 51µs/cm, and 51µs/cm for Iyana Iyesi, Elegushi, and Igbogbo respectively. The pH values measured from SW suggest that they are acidic with mean values of 5.57, 6.05, and 5.79 for Iyana Iyesi, Elegushi, and Igbogbo respectively while TDS values were also at 25mg/l, 33mg/l, and 24 mg/l with no significant difference (p = 0.7030).
During the bacteriological investigation of the SW samples, it was observed that two SW brands had no
E. coli and Total coliform (TC) present. To mention, one of the brands was already classified as a brand with a good reputation while the other was a brand perceived to have a poor reputation by the consumers living within the vicinity where the SW sample was sold. Therefore, for this study, we stratified these SW into two groups comprised of both SW brands (Hebron and Medicx) on one hand and other brands investigated on the other hand to determine whether a significant difference in their contamination levels existed. The findings of his process have been captured in
Table 3. The results showed that SW samples yield on TC (χ2 = 12.28;
p < 0.05),
E. coli (χ2 = 37.96;
p < 0.05), and Staph (χ2 = 0.273;
p > 0.05) over time with these values showing statistical significance.
3.2. Contamination Dominance and Explanatory Analysis of Bacteriological and Physico-Chemical Stressors
In this study, a correlation was carried out on 80 SW samples to determine the relationship between bacteriological and physio-chemical variables called stressors. Spearman’s correlation coefficient was used in place of Pearson’s correlation coefficient based on the normality test conducted using Shapiro-wilk’s test for normality. The test revealed a non-normal distribution of stressors measured in SW samples (p < 0.05).
Table 4 shows the relationship and the statistical significance of the 2-tailed test conducted. Statistical significance but negative correlations were observed between conductivity with [TC (r = -0.247;
p < 0.01); E.coli (r = -0.602;
p < 0.01); THB (r = -0.453;
p < 0.01)], suggesting that bacteria in water would do well and better at lower conductivity levels and that fecal matter present is not the source for high conductivity recorded from SW samples but occurring mutually exclusively mainly due to presence of salts and metalloids. For TDS, a strong statistical positive correlation was observed between TDS with [EC (r = 0.992;
p < 0.01); Salinity (r = 0.676;
p < 0.01)] while strong negative statistical significance was observed with all bacteriological stressors (
p < 0.001).
With the latter already described with the same perception as EC i.e., TDS to EC (r = 0.992; p < 0.01), the former suggests that Salinity to TDS relationship emanates from the same source. Therefore, this is likely to be from either the extraction process of water used for making SW containing rock salts or salt compounds added to reduce pH. Interestingly, the strong statistical correlation between pH and salinity (r = 0.5025; p < 0.01) could suggest that increased salinity was due to buffering pH as both increased directly. Additionally, an attempt was made to identify major pollution in the SW samples by conducting an exploratory statistical method using PCA/FA and communalities. PCA/FA is a dimension reduction that can be useful in whittling down variables to important factors depending on the subject at hand. It whittles this by grouping variables into factors or component loadings. The output of these component loading in a factor shows the relationship of the variables on each component. On the other hand, commonalities show how well these variables explain the variability in a component and it is very useful as a performance evaluator of measured components extracted during PCA/FA procedures. However, both techniques are sensitive to the size of the dataset and produce spurious output when the variables used do not follow a normal curve distribution.
Before conducting the PCA/FA analysis, the variables were first tested for sampling adequacy using the KMO and Bartlett test. These tests were conducted for two main reasons. First, the KMO test revealed whether the data set to be used for PCA/FA statistical operations was suitable. Literature has suggested that KMO values greater than 0.5 are usually suitable but most will agree that higher values of KMO i.e., > 0.5 are even better. Second, Bartlett’s test hypothesizes that the data set to be extracted are typically uncorrelated which can be determined by the
p-value. For our study as seen in
Table 7, KMO was 0.653 which suggests that our dataset is suitable for extraction while Bartlett’s test also showed that the dataset is uncorrelated (
p < 0.05). Thereafter, normalization or transformation was carried out using the CLR method discussed elsewhere. The transformed dataset was then subjected to exploratory analysis and reported in
Table 6.
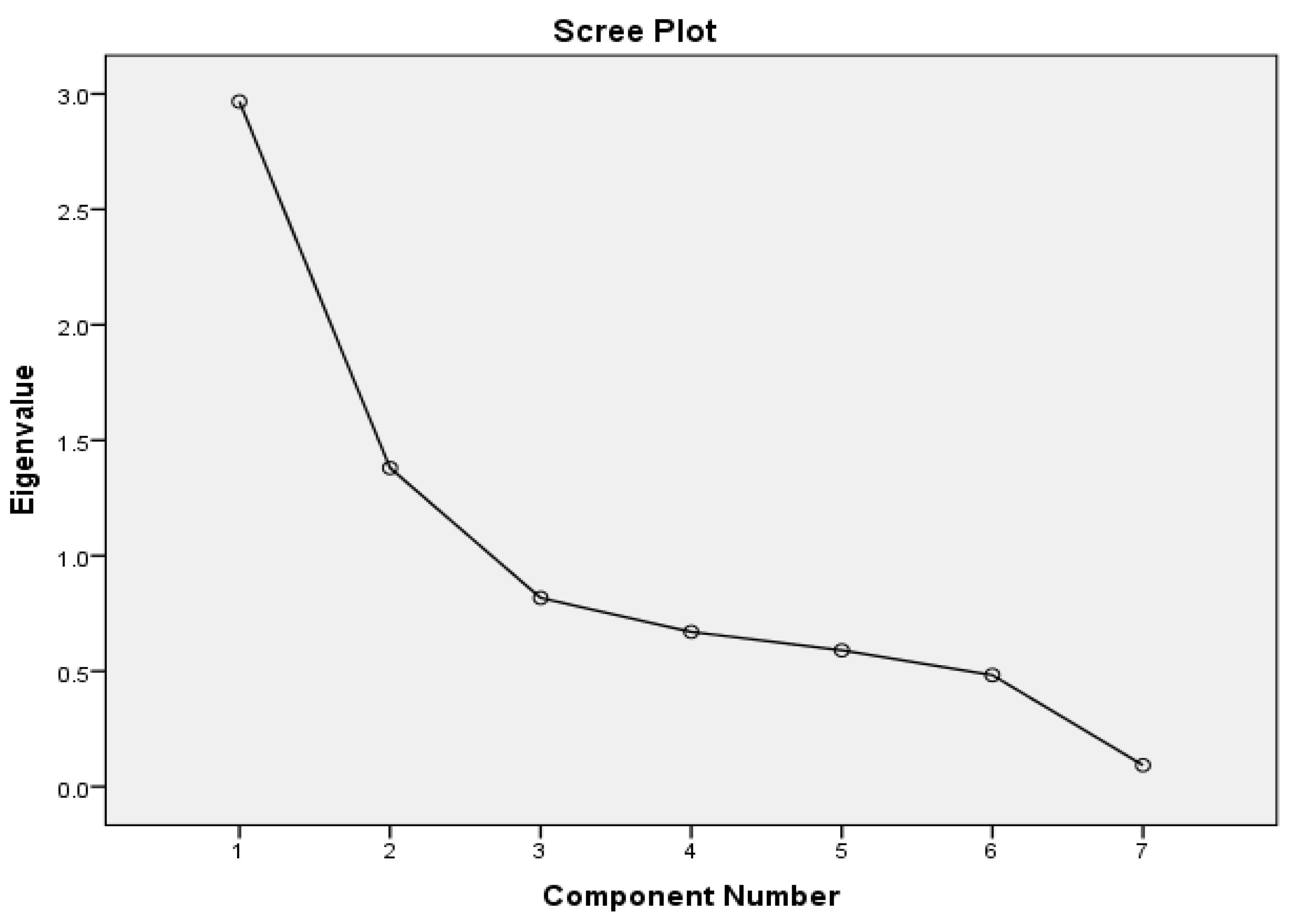
The result from the PCA/FA was determined using two main rules. The first rule is the Kaiser-Meyer-Olkin rule which suggests that meaningful components are usually extracted with Eigenvalue greater than 1. Next was the Elbow or Point of Inflexion shown on a Scree plot (
Figure 3). From both rules, components were factored as 2. The major advantage of FA is that it helps in the grouping, which can be used in this context for source allocation. For example, in this study, component 1 had strong loadings on Salinity and Conductivity. While component 2 is correlated by
E. coli and Total Coliform suggesting the presence of fecal contamination. This could only be possible as the distribution system accumulates bacterial films and sediments that do so well in retaining organisms in the water. Therefore, showing multiple lines of evidence that infrequent cleaning and lack of advanced treatment mechanisms are so lacking in the SW industry and require both the attention of consumers and relevant government agencies to be properly addressed. The absence of these would only increase the level of water insecurity as new and old packaged water companies would show increased lackadaisical behavior towards producing packaged drinking water of better quality.
Communality analysis shown in
Table 6 revealed that ionic contamination was well explained when compared to bacteriological contamination in SW studied. Specifically, it revealed that 87.6% of salinity and 76.7% of conductivity were explained by factor analysis while 69.3% and 63.3% explained the variability of TC and
E. coli in its Factor Component 2. An indication that shows or depicts a satisfactory performance as it suggests that lesser grouping was more effective than extended groupings.
4. Discussion
From this study, we investigated the presence of bacteriological contamination. Seemingly, the presence of fecal contamination was detected in 51 (≥ 81%) of SW investigated in the region, from SW with a perceived poor reputation. The contamination levels recorded were in amounts that require urgent attention. This is concerning because, before 7-fold dilution, the results initially recorded from the MPN test of SW samples revealed TNC observations of bacteriological contamination. In the past, a study [
36] investigated the presence of bacteria in 92 SW in Lagos, Nigeria. The findings from that study showed elevated
E. coli values which were significantly higher than the threshold set by WHO. The values in this study revealed a much higher presence of bacterial contamination than those reported in earlier studies. Amongst all samples tested, only one sample was positive for the presence of staph. Although this study reports the presence of staph in the SW sample as statistically insignificant, it calls for a more proactive public health check to avoid any occurrences.
Staph infections have been reported to cause extremely serious or fatal infections in humans including bacteremia, sepsis (when bacteria spread to the bloodstream), pneumonia, endocarditis (infection of the heart valves), and osteomyelitis (bone infection) [
37]. [
38] also investigated SW consumed by university students in Kumasi Ghana and found 50% of SW investigated contained TC & EC of similar contamination levels as this current study. However, their finding did not agree with findings from [
14] that found no
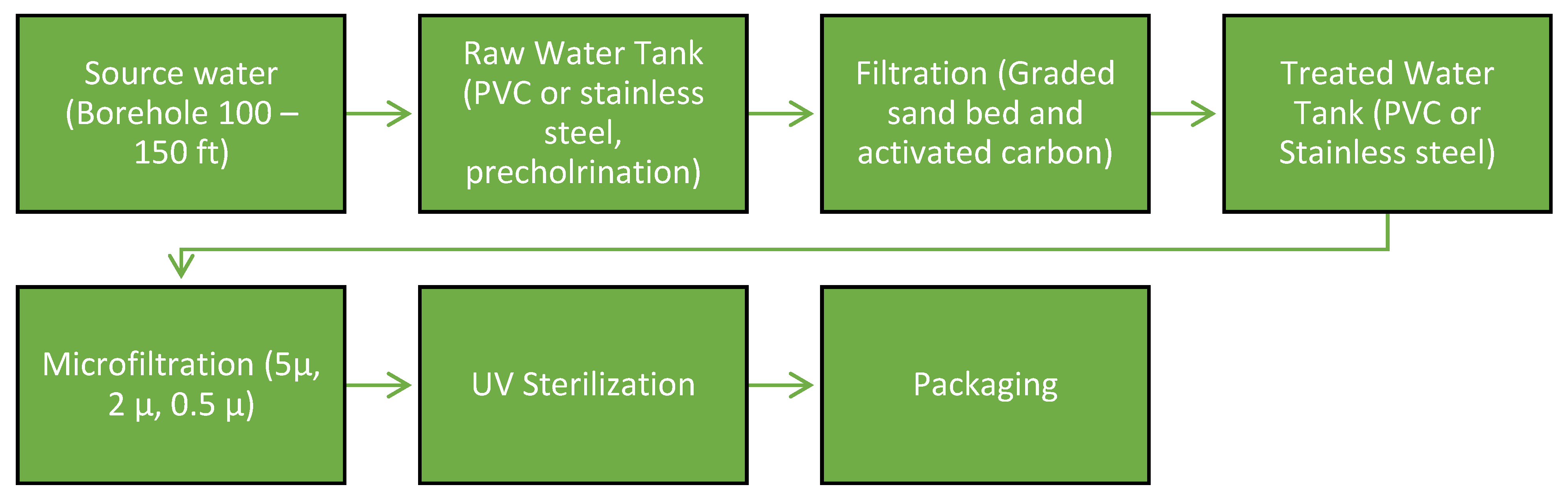
E. coli presence in SW samples investigated in Ghana. These studies suggest the need for a location-based continuous monitoring scheme to create more public awareness is required. Two SW brands in this study showed no presence of TC & EC. The first brand (Hebron) is a brand every SW consumer around the studied area would prefer to purchase but they see it beyond their reach as SW produced by this brand are bought off quickly before it spreads to the neighborhood, thus making it expensive to purchase compared to other brands in the region. The other brand is not well known and does not feature attractive and quality packaging. Despite the good performance of the two SW brands, it is opined that best management practices as stipulated by National Agency for Food and Drug Administration and Control (NAFDAC) should be unrelentingly followed. NAFDAC reported that persistent presence of pathogenic bacteria in SW is due to the use of untreated raw water as well as SW companies non-compliance for treatment of raw water. NAFDAC (2018) recommends that the treatment process in all SW production factories should follow the stated order (
Figure 4). Previous studies have shown that microfiltration has the capacity to remove all bacteria from the water being treated. Hence, it can be deduced from the results obtained that SW manufacturers do not adhere to the recommended treatment guidelines prescribed by NAFDAC. Another reason for bacteria presence in packaged SW could be linked to the fouling of membranes in facilities where microfiltration systems are used [
39,
40,
41,
42]. Hence, a need for proper cleaning of the microfiltration membranes is also essential for maintaining water quality.
Poor hygiene practices in SW production facilities can be linked to the presence of staph in packaged water. Most SW factories visited during this study were in poor environmental conditions with waste polyethylene bags and papers littered all over. It is believed that this may lead to heightened microbial contamination due to the proximity of SW production facilities to waste disposal facilities including septic tanks and small to medium dumpsters [
43]. A deformation in the physical component of these facilities can lead to the escape of substances of public health concern into SW processes. [
44,
45] reported that
E. coli and other microbes from fecal contamination can enter raw water sources when open defecation is practiced close to water bodies or when a leak exists in a septic tank. These contaminants can be transferred in packaged water in the case where proper treatment is not ensured, thus causing a public health crisis. This study, therefore, has revealed that there is a higher possibility of a range of non-treatment to inadequate treatment of water used for SW purposes even though many of the SW companies claim to involve in reverse osmosis, microfiltration process, and chlorination. This study embarked gives new insight to the contamination level years after a similar investigation was conducted in a different region of West Africa. Our findings corroborate with the conditions of [
46] that in high demand there is a possibility of the high occurrence of contamination levels because of limited or no maintenance of water supply distribution lines, as well as a non-compliance of SW manufacturers to good sanitation and hygiene measures. It is a great concern to note the high number of fecal indicators consistently found in SW across the country. Even though this is consistent with our study, being that it was conducted in a busy period where Christians from over the world gather en masse, the authors of this study see that the results might be the same irrespective of seasonality [
11]. The reason for this conclusion was due to the environmental conditions of the SW offices during our study.
5. Conclusions
The bacteriological quality of 80 SW samples collected from three neighborhoods in Ota, Nigeria was investigated. The results obtained were used to conduct statistical analysis to determine the level of significance of the contamination. Additionally, bacterial dominance was correlated with a brand reputation to give an informed re-education of public perception as to what brands they should be consuming for the safety of their health. All the results obtained for 69 samples fall below the WHO limits for the bacteriological quality of drinking water. Although THB has no associated health risk, the WHO standard requires a maximum of 500 CFU/mL in water. Additionally, a high concentration of THB is an indication of favorable conditions for the growth of more dangerous microbes such as E. Coli, Legionella, and S. Typhi and these can cause serious harm to human health and in some cases, death. The results obtained in this study show THB counts over the recommended WHO limit which is a cause for concern.
Fecal contamination in SW has become a norm in SW at an alarming rate such that entrusting the monitoring of the SW production process to NAFDAC which appears to have done less in enforcing regulations in this regard will be a grave mistake for the public. Frequent periodic monitoring programs and collaborations between SW manufacturers and researchers are encouraged to find a lasting solution to this crisis. It is also important to note that the problem of bacterial dominance in packaged water is location specific, and as such requires location-specific water treatment as opposed to a general treatment approach. Testing of source water quality should be mandated for all SW manufacturers and specific measures should be undertaken to remove the contaminants found in the source water. It is also encouraged that the public pays more attention to the SW they consume as it can impair their health negatively. Finally, properly cleaning water treatment facilities and distribution systems to remove sediments and clogged bacterial films can to a large extent help reduce the re-occurrence of bacteria into packaged SW after treatment.
Supplementary Materials
All relevant information for this study is contained in the manuscript.
Author Contributions
All authors contributed to the conception and design of the study. Material preparation, data collection, and spatial and statistical analysis were performed by Imokhai T. Tenebe, Eunice O. Babatunde, Chinonso C. Eddy-Ugorji, Nkpa M. Ogarekpe, Chikodinaka V. Ekeanyanwu, Oluwarotimi. S. Oladele, and Obiora B. Ezeudu. The first draft of the manuscript was written by Imokhai Theophilus Tenebe, and all the authors commented on the previous versions of the manuscript. All authors have read and approved the final manuscript.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Data Availability Statement
The authors have no affiliations with or involvement in any organization or entity with any financial or nonfinancial interests in the subject matter or material discussed in this manuscript. All relevant data and supporting information are within the paper.
Acknowledgments
In this section, you can acknowledge any support given which is not covered by the author contribution or funding sections. This may include administrative and technical support, or donations in kind (e.g., materials used for experiments).
Conflicts of Interest
The authors declare that they have no competing financial interests or personal relationships that could influence the work reported in this study.
References
- Tenebe, I.T. MULTIVARIATE ANALYSES OF THE EFFECTS OF LAND USE CHANGE ON RIVER WATER QUALITY: CASE STUDY OF MANAWATU RIVER WATERSHED, NEW ZEALAND. Master of Science in Engineering, Texas State University, 2020.
- Hensen, B.; Lange, J.; Jackisch, N.; Zieger, F.; Olsson, O.; Kümmerer, K. Entry of Biocides and Their Transformation Products into Groundwater via Urban Stormwater Infiltration Systems. Water Research 2018, 144, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Kresic, N. Groundwater Contamination. Hydrogeology and Groundwater Modeling 2020, 425–468. [Google Scholar] [CrossRef]
- Chinedu, S.; Nwinyi, O. Assessment of Water Quality in Canaanland, Ota, Southwest Nigeria. Agriculture and Biology Journal of North America 2011, 2, 577–583. [Google Scholar] [CrossRef]
- Mukhopadhyay, C.; Vishwanath, S.; Eshwara, V.K.; Shankaranarayana, S.A.; Sagir, A. Microbial Quality of Well Water from Rural and Urban Households in Karnataka, India: A Cross-Sectional Study. Journal of Infection and Public Health 2012, 5, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Ogamba, E.N.; Charles, E.E.; Izah, S.C. Distributions, Pollution Evaluation and Health Risk of Selected Heavy Metal in Surface Water of Taylor Creek, Bayelsa State, Nigeria. Toxicology and Environmental Health Sciences 2021, 13, 109–121. [Google Scholar] [CrossRef]
- Vedachalam, S.; MacDonald, L.H.; Omoluabi, E.; Olaolorun, F.; Otupiri, E.; Schwab, K.J. The Role of Packaged Water in Meeting Global Targets on Improved Water Access. Journal of Water Sanitation and Hygiene for Development 2017, 7, 369–377. [Google Scholar] [CrossRef]
- Tetteh, J.D.; Templeton, M.R.; Cavanaugh, A.; Bixby, H.; Owusu, G.; Yidana, S.M.; Moulds, S.; Robinson, B.; Baumgartner, J.; Annim, S.K.; et al. Spatial Heterogeneity in Drinking Water Sources in the Greater Accra Metropolitan Area (GAMA), Ghana. Popul Environ 2022, 44, 46–76. [Google Scholar] [CrossRef] [PubMed]
- Ajala, O.J.; Ighalo, J.O.; Adeniyi, A.G.; Ogunniyi, S.; Adeyanju, C.A. Contamination Issues in Sachet and Bottled Water in Nigeria: A Mini-Review. Sustain. Water Resour. Manag. 2020, 6, 112. [Google Scholar] [CrossRef]
- Angnunavuri, P.N.; Attiogbe, F.; Dansie, A.; Mensah, B. Evaluation of Plastic Packaged Water Quality Using Health Risk Indices: A Case Study of Sachet and Bottled Water in Accra, Ghana. Science of The Total Environment 2022, 832, 155073. [Google Scholar] [CrossRef]
- Emenike, P.G.C.; Tenebe, T.I.; Omeje, M.; Osinubi, D.S. Health Risk Assessment of Heavy Metal Variability in Sachet Water Sold in Ado-Odo Ota, South-Western Nigeria. Environmental Monitoring and Assessment 2017, 189, 1–16. [Google Scholar] [CrossRef]
- Mosi, L.; Adadey, S.M.; Sowah, S.A.; Yeboah, C. Microbiological Assessment of Sachet Water “Pure Water” from Five Regions in Ghana. AAS Open Research 2019, 1, 12. [Google Scholar] [CrossRef] [PubMed]
- Stoler, J. Sachet Water Quality in Ghana: The Jury Remains Out. Food Control 2014, 37, 417–418. [Google Scholar] [CrossRef]
- Stoler, J.; Tutu, R.A.; Ahmed, H.; Frimpong, L.A.; Bello, M. Sachet Water Quality and Brand Reputation in Two Low-Income Urban Communities in Greater Accra, Ghana. American Journal of Tropical Medicine and Hygiene 2014, 90, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Stoler, J.; Ahmed, H.; Asantewa Frimpong, Lady; Bello, M. Presence of Pseudomonas Aeruginosa in Coliform-Free Sachet Drinking Water in Ghana. Food Control 2015, 55, 242–247. [Google Scholar] [CrossRef]
- Omonigho, D. Bacteriological Analysis of Sachet Water Vended In Ugbor, Benin. SAU Sci-Tech. J 2017, 1, 88–100. [Google Scholar]
- Omoigberale, M.N.O.; Isibor, J.O.; Izegaegbe, J.I.; Iyamu, M.I. Seasonal Variation in the Bacteriological Quality of Ebutte River in Ehor Community EDO State, Nigeria of Microbiology, Faculty of Natural Sciences, Ambrose Alli University, Department of Zoology, Faculty of Natural Sciences, Ambrose Alli University. 2013, 1, 59–69.
- Tenebe, I.T.; Emenike, P.C.; Nnaji, C.C.; Babatunde, E.O.; Ogarekpe, N.M.; Dede-Bamfo, N.; Omole, D.O. Bacterial Characterization and Quantification of Rainwater Harvested in a Rural Community in Nigeria. Environmental Nanotechnology, Monitoring and Management 2020, 14, 100370. [Google Scholar] [CrossRef]
- Tenebe, I.; Emenike, P.C.; Babatunde, E.O.; Neris, J.B.; Fred-Ahmadu, O.H.; Dede-Bamfo, N.; Etu, E.E.; Ogarekpe, N.M.; Emakhu, J.; Nsikak, B.U. Assessing the State of Rainwater for Consumption in a Community in Dire Need of Clean Water: Human and Health Risk Using HERisk. Water Practice and Technology 2022, (pt2022109), wpt2022109. [Google Scholar] [CrossRef]
- Chinenye, I.J.; Amos, O.O. Effect of Storage and Exposure to Sunlight on the Quality of Sachet Water Sold in Ibadan Metropolis. Science Journal of Public Health 2017, 5, 321. [Google Scholar] [CrossRef]
- Abubakar, M.; Usman, K.; Yusuf, A.; Hayatu, M.M.; Sani, B.Y.; Oderinde, O.; Saadu, B. Effect of Natural Sunlight on Quality of Sachet Water Sold within Sokoto Metropolis (Sidi Mam Man Table Water). International Journal of Science for Global Sustainability 2020, 6, 151–156. [Google Scholar]
- Umoafia, N.E.; Edet, U.O.; Joseph, A.P.; Henshaw, O.U.; Nkagafel, M.E.; Obeten, H.; Edet, B.O.; Edet, A.; Richard, I.; Nwaokorie, F.; et al. Impact of Exposure to Sunlight before Consumption of Packaged Potable Water (Sachet Water) in Calabar Metropolis, Cross River State, Southeastern Nigeria 2022.
- Samuel, O.A.; Tenebe, I.T.; Emenike, P.C.; Daniel, D.I.; Omole, D.O.; Maxwell, O.; Ben, N.U.; Kelechi, O.O.; Osagie, I. Preliminary Assessment of the Current Pollution Status of the River Atuwara, Nigeria, within an Industrial Site: A Bivariate Approach. WIT Transactions on Ecology and the Environment 2019, 234, 209–219. [Google Scholar] [CrossRef]
- Enyoh, C.E.; Enyoh, E.C.; Ohiagu, F.O. Evaluation of Some Groundwater Sources in Ota, Ogun State, Southwestern Nigeria. 2021.
- Omole, D.O.; Ndambuki, J.M.; Balogun, K. Consumption of Sachet Water in Nigeria: Quality, Public Health, and Economic Perspectives. African Journal of Science, Technology, Innovation, and Development 2015, 7, 45–51. [Google Scholar] [CrossRef]
- Omole, D.O.; Ogbiye, A.S.; Longe, E.O.; Adewumi, I.K.; Elemile, O.O.; Tenebe, T.I. WATER QUALITY CHECKS ON RIVER ATUWARA, SOUTH-WEST NIGERIA.; A Coruña, Spain, May 22, 2018; pp. 165–173.
- Emenike, P.C.; Neris, J.B.; Tenebe, I.T.; Nnaji, C.C.; Jarvis, P. Estimation of Some Trace Metal Pollutants in River Atuwara Southwestern Nigeria and Spatio-Temporal Human Health Risks Assessment. Chemosphere 2020, 239, 124770. [Google Scholar] [CrossRef] [PubMed]
- Rice, E.W.; Geldreich, E.E.; Read, E.J. The Presence-Absence Coliform Test for Monitoring Drinking Water Quality. Public Health Rep 1989, 104, 54–58. [Google Scholar] [PubMed]
- MacLeod, C.; Peletz, R.; Kere, F.; Baye, A.M.; Onyango, M.; Aw, S.; El Hadj Issabre, M.; Tung, R.; Khush, R. Are Presence/Absence Microbial Tests Appropriate for Monitoring Large Urban Water Supplies in Sub-Saharan Africa? Water (Basel) 2019, 11, 491. [Google Scholar] [CrossRef] [PubMed]
- Nutrition, C. for F.S. and A. BAM Appendix 2: Most Probable Number from Serial Dilutions. FDA, 2020. [Google Scholar]
- Wright, K.M.; Wright, P.J.; Holden, N.J. MacConkey Broth Purple Provides an Efficient MPN Estimation Method for Shigatoxigenic Escherichia Coli. Journal of Microbiological Methods 2021, 181, 106132. [Google Scholar] [CrossRef] [PubMed]
- Bolton, F.J.; Hinchliffe, P.M.; Coates, D.; Robertson, L. A Most Probable Number Method for Estimating Small Numbers of Campylobacters in Water. J Hyg (Lond) 1982, 89, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Zhang, Y.; Zhang, Y. Evaluation of a Most Probable Number Method for Detection and Quantification of Legionella pneumophila. Pathogens 2022, 11, 789. [Google Scholar] [CrossRef] [PubMed]
- Thani, T.S.; Symekher, S.M.L.; Boga, H.; Oundo, J. Isolation and Characterization of Escherichia Coli Pathotypes and Factors Associated with Well and Boreholes Water Contamination in Mombasa County. The Pan African Medical Journal 2016, 23. [Google Scholar] [CrossRef] [PubMed]
- 9221 MULTIPLE-TUBE FERMENTATION TECHNIQUE FOR MEMBERS OF THE COLIFORM GROUP. In Standard Methods For the Examination of Water and Wastewater; Standard Methods for the Examination of Water and Wastewater; American Public Health Association, 2017.
- Olaoye, O.A.; Onilude, A.A. Assessment of Microbiological Quality of Sachet-Packaged Drinking Water in Western Nigeria and Its Public Health Significance. Public Health 2009, 123, 729–734. [Google Scholar] [CrossRef]
- CDC Staphylococcus Aureus in Healthcare Settings | HAI | CDC Available online:. Available online: https://www.cdc.gov/hai/organisms/staph.html (accessed on 17 January 2023).
- Addo, B.E.; Amankwaa, G.; Gyasi, R.M. Physicochemical and Bacteriological Quality of Sachet Water Used by Ghanaian University Students: Implications for Public Health. Journal of Water Sanitation and Hygiene for Development 2019, 9, 56–63. [Google Scholar] [CrossRef]
- Lay, H.T.; Wang, R.; Chew, J.W. Influence of Foulant Particle Shape on Membrane Fouling in Dead-End Microfiltration. Journal of Membrane Science 2022, 647, 120265. [Google Scholar] [CrossRef]
- 4Poerio, T.; Denisi, T.; Mazzei, R.; Bazzarelli, F.; Piacentini, E.; Giorno, L.; Curcio, E. Identification of Fouling Mechanisms in Cross-Flow Microfiltration of Olive-Mills Wastewater. Journal of Water Process Engineering 2022, 49, 103058. [Google Scholar] [CrossRef]
- Tanudjaja, H.J.; Anantharaman, A.; Ng, A.Q.Q.; Ma, Y.; Tanis-Kanbur, M.B.; Zydney, A.L.; Chew, J.W. A Review of Membrane Fouling by Proteins in Ultrafiltration and Microfiltration. Journal of Water Process Engineering 2022, 50, 103294. [Google Scholar] [CrossRef]
- Yu, R.; Wang, H.; Wang, R.; Zhao, P.; Chen, Y.; Liu, G.; Liao, X. Polyphenol Modified Natural Collagen Fibrous Network towards Sustainable and Antibacterial Microfiltration Membrane for Efficient Water Disinfection. Water Research 2022, 218, 118469. [Google Scholar] [CrossRef] [PubMed]
- Nnaji, C.C.; Onuigbo, K.; Nnam, J.P. Assessment of Seasonal Variation of Nsukka Phreatic Aquifer Groundwater Quality. 2019.
- Kabwama, S.N.; Bulage, L.; Nsubuga, F.; Pande, G.; Oguttu, D.W.; Mafigiri, R.; Kihembo, C.; Kwesiga, B.; Masiira, B.; Okullo, A.E.; et al. A Large and Persistent Outbreak of Typhoid Fever Caused by Consuming Contaminated Water and Street-Vended Beverages: Kampala, Uganda, January – June 2015. BMC Public Health 2017, 17, 23. [Google Scholar] [CrossRef]
- Okullo, J.O.; Moturi, W.N.; Ogendi, G.M. Open Defaecation and Its Effects on the Bacteriological Quality of Drinking Water Sources in Isiolo County, Kenya. 2017. [CrossRef]
- Stoler, J.; Tutu, R.A.; Winslow, K. Piped Water Flows but Sachet Consumption Grows: The Paradoxical Drinking Water Landscape of an Urban Slum in Ashaiman, Ghana. Habitat International 2015, 47, 52–60. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).