Submitted:
28 March 2023
Posted:
30 March 2023
You are already at the latest version
Abstract
Keywords:
Introduction
2. Experimental Section
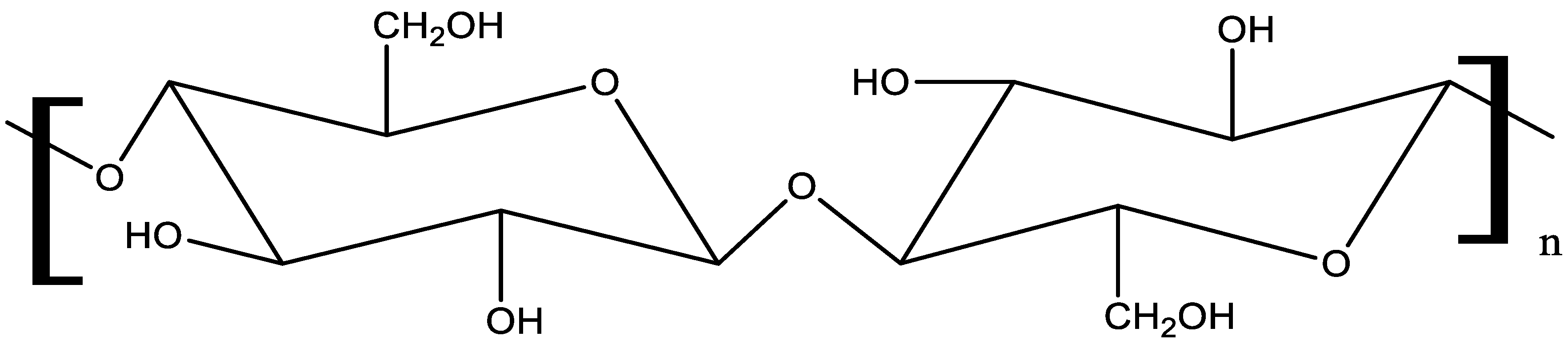
2.1. Materials
2.2. Composite fabrication
2.2. Characterization
3. Results and discussion
Conclusion
References
- Cao, Y.; Wu, Y.Q. Evaluation of statistical strength of bamboo fiber and mechanical properties of fiber reinforced green composites. J. Cent. South Univ. Technol. 2008, 15, 564–567. [Google Scholar] [CrossRef]
- Li, X.; et al. Chemical treatments of natural fiber for use in natural fiber-reinforced composites: a review. J. Polym. Environ. 2007, 15, 25–33. [Google Scholar] [CrossRef]
- Lee, B.-H.; et al. Fabrication of long and discontinuous natural fiber reinforced polypropylene biocomposites and their mechanical properties. Fibers Polym. 2009, 10, 83–90. [Google Scholar] [CrossRef]
- Mehta, G.; et al. Novel biocomposites sheet molding compounds for low-cost housing panel applications. J. Polym. Environ. 2005, 13, 169–175. [Google Scholar] [CrossRef]
- Ioelovich, M.; Figovsky, O. Advanced environment-friendly polymer materials. Polym. Adv. Technol. 2002, 13, 1112–1115. [Google Scholar] [CrossRef]
- Lee, S.-Y.; et al. Creep behavior and manufacturing parameters of wood flour filled polypropylene composites. Compos. Struct. 2004, 65, 459–469. [Google Scholar] [CrossRef]
- Kim, H.-S.; et al. Thermogravimetric analysis of rice husk flour filled thermoplastic polymer composites. J. Therm. Anal. Calorim. 2004, 76, 395–404. [Google Scholar]
- Dorée. The Methods of Cellulose Chemistry, Chapman & Hall, London, UK, 1947.
- The Merck Index, Merck & Co, Rahway, NJ, USA, 8th edition, 1968.
- Chand, N.; Fahim, M. Natural fibers and their composites, in Tribology of Natural Fiber Polymer Composites (Second Edition), N. Chand and M. Fahim, Editors. 2021, Woodhead Publishing. p. 1-59.
- Joseph, B.; et al. Cellulose nanocomposites: Fabrication and biomedical applications. J. Bioresour. Bioprod. 2020, 5, 223–237. [Google Scholar] [CrossRef]
- Kalka, S.; et al. Biodegradability of all-cellulose composite laminates. Compos. Part A Appl. Sci. Manuf. 2014, 59, 37–44. [Google Scholar] [CrossRef]
- Samir, A.; et al. Recent advances in biodegradable polymers for sustainable applications. NPJ Mater. Degrad. 2022, 6, 68. [Google Scholar] [CrossRef]
- Immonen, K.; et al. Thermoplastic cellulose-based compound for additive manufacturing. Molecules 2021, 26, 1701. [Google Scholar] [CrossRef] [PubMed]
- Norizan, M.N.; et al. Nanocellulose-based nanocomposites for sustainable applications: A Review. Nanomaterials 2022, 12, 3483. [Google Scholar] [CrossRef] [PubMed]
- Rashid, S.; Dutta, H. Characterization of nanocellulose extracted from short, medium and long grain rice husks. Ind. Crops Prod. 2020, 154, 112627. [Google Scholar] [CrossRef]
- Babu, N.K.; et al. Self-reinforced polymer composites: An opportunity to recycle plastic wastes and their future trends. J. Appl. Polym. Sci. 2022, 139, e53143. [Google Scholar] [CrossRef]
- Park, K.; et al. Recyclable and mendable cellulose-reinforced composites crosslinked with Diels–Alder adducts. Polymers 2019, 11, 117. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P.; et al. Effect of processing variables on the mechanical properties of sisal-fiber-reinforced polypropylene composites. Compos. Sci. Technol. 1999, 59, 1625–1640. [Google Scholar] [CrossRef]
- Rana, A.; et al. Short jute fiber reinforced polypropylene composites: Effect of compatibiliser, impact modifier and fiber loading. Compos. Sci. Technol. 2003, 63, 801–806. [Google Scholar] [CrossRef]
- Lu, J.Z.; et al. Chemical coupling in wood fiber and polymer composites: A review of coupling agents and treatments. Wood Fiber Sci. 2007, 32, 88–104. [Google Scholar]
- Amash, A.; Zugenmaier, P.J.P. Morphology and properties of isotropic and oriented samples of cellulose fibre–polypropylene composites. Polymer 2000, 41, 1589–1596. [Google Scholar] [CrossRef]
- Pothan, L.A.; et al. Dynamic mechanical analysis of banana fiber reinforced polyester composites. Compos. Sci. Technol. 2003, 63, 283–293. [Google Scholar] [CrossRef]
- George, J.; et al. A review on interface modification and characterization of natural fiber reinforced plastic composites. Polym. Eng. Sci. 2001, 41, 1471–1485. [Google Scholar] [CrossRef]
- Sue, H.-J.; et al. Epoxy nanocomposites based on the synthetic α-zirconium phosphate layer structure. Chem. Mater. 2004, 16, 242–249. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, S.; Qi, Z. Synthesis and characterization of elastomeric polyurethane/clay nanocomposites. J. Appl. Polym. Sci. 2001, 82, 1444–1448. [Google Scholar] [CrossRef]
- Lee, K.Y.; Goettler, L.A. Structure-property relationships in polymer blend nanocomposites. Polym. Eng. Sci. 2004, 44, 1103–1111. [Google Scholar] [CrossRef]
- Dan, C.H.; et al. Effect of clay modifiers on the morphology and physical properties of thermoplastic polyurethane/clay nanocomposites. Polymer 2006, 47, 6718–6730. [Google Scholar] [CrossRef]
- Morgan, A.B.; Gilman, J.W. Characterization of polymer-layered silicate (clay) nanocomposites by transmission electron microscopy and X-ray diffraction: A comparative study. J. Appl. Polym. Sci. 2003, 87, 1329–1338. [Google Scholar] [CrossRef]
- Chesworth, W.; et al. Clay-organic interactions, in Encyclopedia of Soil Science, W. Chesworth, Editor. 2008, Springer Netherlands: Dordrecht. p. 144-150.
- Vasudeo Rane, A.; et al. Chapter 4 - Clay–polymer composites: Design of clay polymer nanocomposite by mixing, in clay-polymer nanocomposites, K. Jlassi, M.M. Chehimi, and S. Thomas, Editors. 2017, Elsevier. p. 113-144.
- Guo, F.; et al. A review of the synthesis and applications of polymer–nanoclay composites. Appl. Sci. 2018, 8, 1696. [Google Scholar] [CrossRef]
- Poletto, M.; Ornaghi, H.L.; Zattera, A.J.J.M. Native cellulose: Structure, characterization and thermal properties. Materials 2014, 7, 6105–6119. [Google Scholar] [CrossRef]
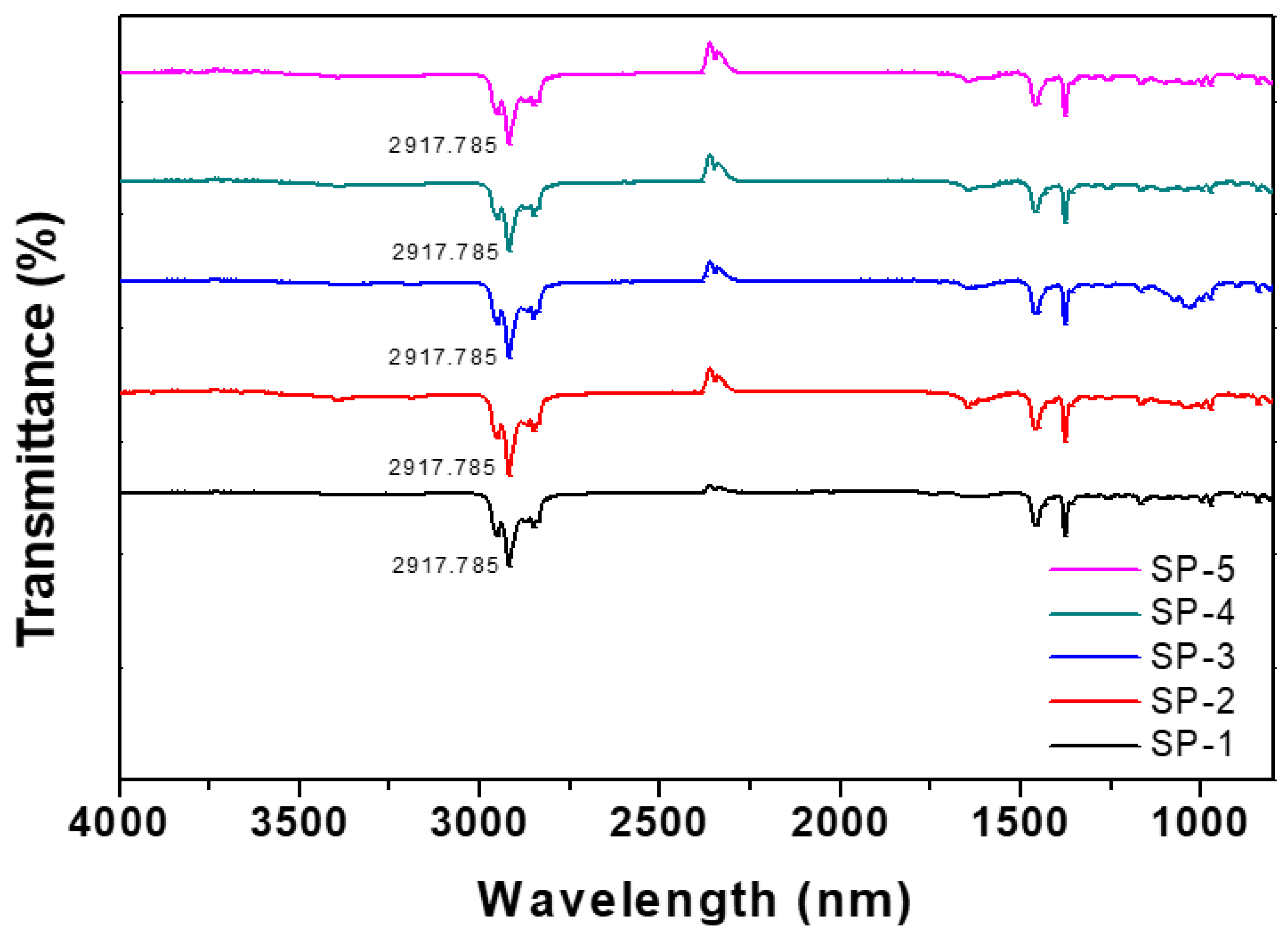
- Fan, M.; Dai, D.; Huang, B. Fourier transform infrared spectroscopy for natural fibres. Fourier Transform.-Mater. Anal. 2012, 3, 45–68. [Google Scholar]
- Ellis, T.S.; D’Angelo, J.S. Thermal and mechanical properties of a polypropylene nanocomposite. Mater. Des. 2003, 90, 1639–1647. [Google Scholar] [CrossRef]
- Botev, M.; et al. Mechanical properties and viscoelastic behavior of basalt fiber-reinforced polypropylene. J. Appl. Polym. Sci. 1999, 74, 523–531. [Google Scholar] [CrossRef]
- Bondeson, D.; et al. All cellulose nanocomposites produced by extrusion. J. Biobased Mater. Bioenergy 2007, 1, 367–371. [Google Scholar] [CrossRef]
- Jonoobi, M.; et al. Mechanical properties of cellulose nanofiber (CNF) reinforced polylactic acid (PLA) prepared by twin screw extrusion. Compos. Sci. Technol. 2010, 70, 1742–1747. [Google Scholar] [CrossRef]
- Lee, S.-Y.; et al. Thermal behavior of hwangto and wood flour reinforced high density polyethylene (HDPE) composites. J. Korean Wood Sci. Technol. 2006, 34, 59–66. [Google Scholar]
- Lee, S.; et al. Thermal, mechanical and morphological properties of polypropylene/clay/wood flour nanocomposites. eXPRESS Polym. Lett. 2008, 2, 78–87. [Google Scholar] [CrossRef]
- Wang, K.H.; et al. Morphology and physical properties of polyethylene/silicate nanocomposite prepared by melt intercalation. J. Polym. Sci. Part B Polym. Phys. 2002, 40, 1454–1463. [Google Scholar] [CrossRef]

| Sample name | PP% |
TPO EG8407 |
Ethanox 310 | Ethanox-368 | EV-91 | EV-12 | Clay% | Cellulose powder% |
| SP-1 | 79.96 | 20 | 0.01 | 0.01 | 0.01 | 0.01 | 0 | 0 |
| SP-2 | 79.46 | 20 | 0.01 | 0.01 | 0.01 | 0.01 | 0 | 5.5 |
| SP-3 | 74.46 | 20 | 0.01 | 0.01 | 0.01 | 0.01 | 5.5 | 0 |
| SP-4 | 72.46 | 20 | 0.01 | 0.01 | 0.01 | 0.01 | 2.0 | 5.5 |
| SP-5 | 68.46 | 20 | 0.01 | 0.01 | 0.01 | 0.01 | 2.0 | 9.5 |
| SP-6 | 67.4.6 | 20 | 0.01 | 0.01 | 0.01 | 0.01 | 3.0 | 9.5 |
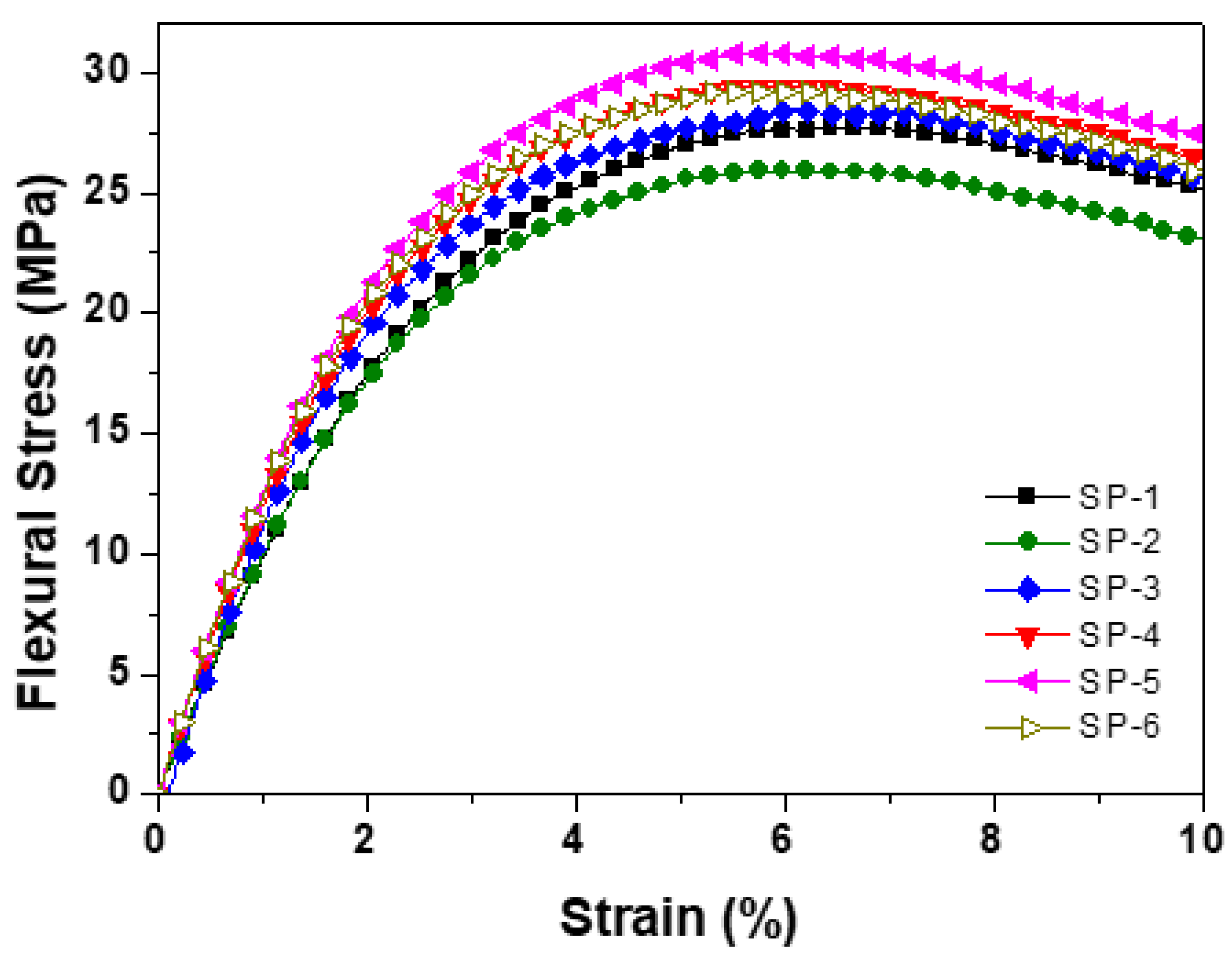
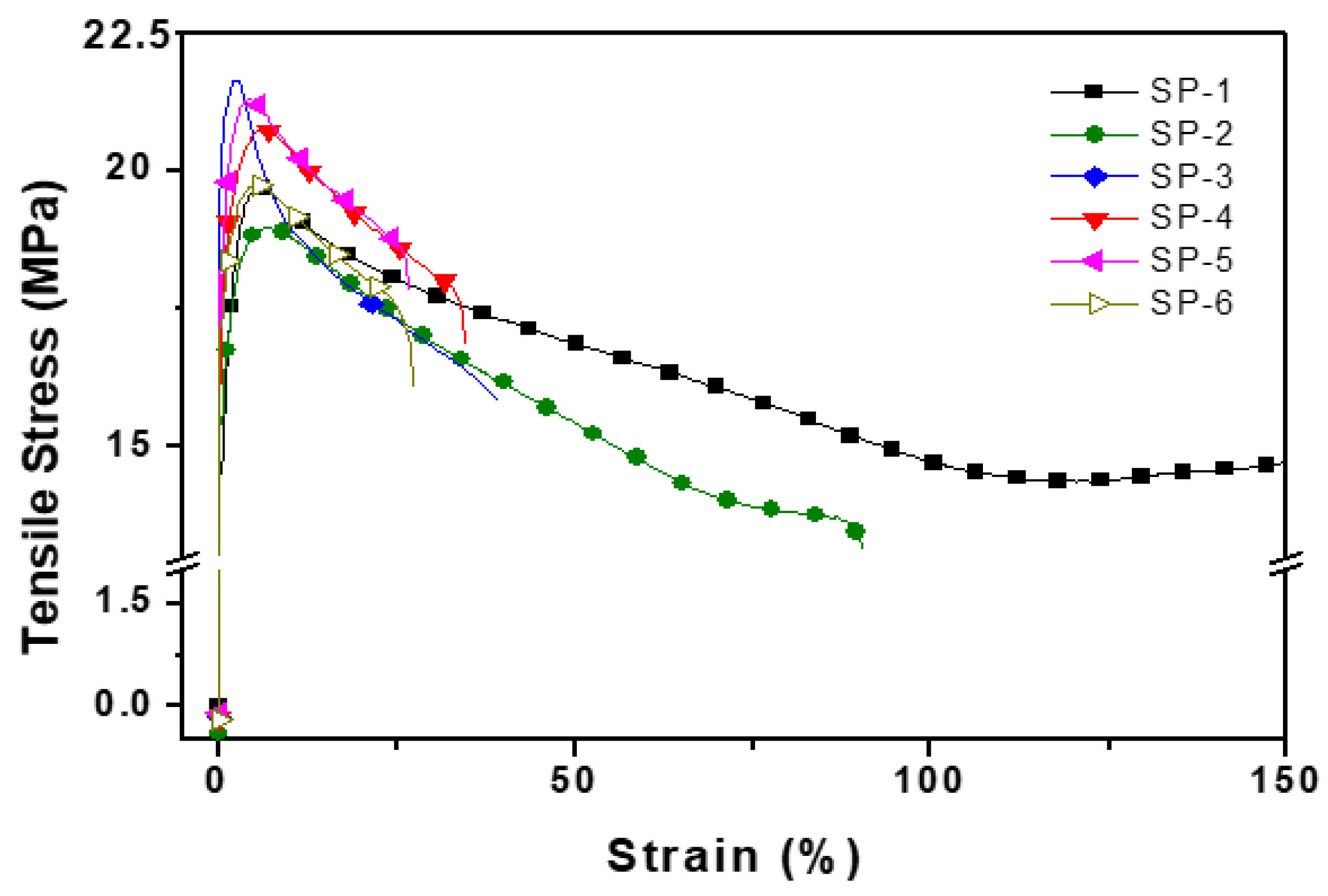
| Sample name |
Flexural Strength MPa |
Flexural Modulus MPa |
Tensile strength MPa |
Tensile Modulus MPa |
Yield strain | Elongation % | Notched Izod strength kg. cm/cm2 |
| SP-1 | 27.5 | 134.8 | 18.5 | 110 | 0.168182 | 82 | 42.5 |
| SP-2 | 26.0 | 112.8 | 18.7 | 115 | 0.162609 | 102 | 30 |
| SP-3 | 28.0 | 130 | 21.75 | 120 | 0.18125 | 93 | 38 |
| SP-4 | 29.0 | 145 | 21.0 | 125 | 0.168 | 86.2 | 27 |
| SP-5 | 31.0 | 155 | 21.85 | 122 | 0.179098 | 79 | 25 |
| SP-6 | 28.8 | 135 | 19.5 | 120 | 0.1625 | 89 | 22 |
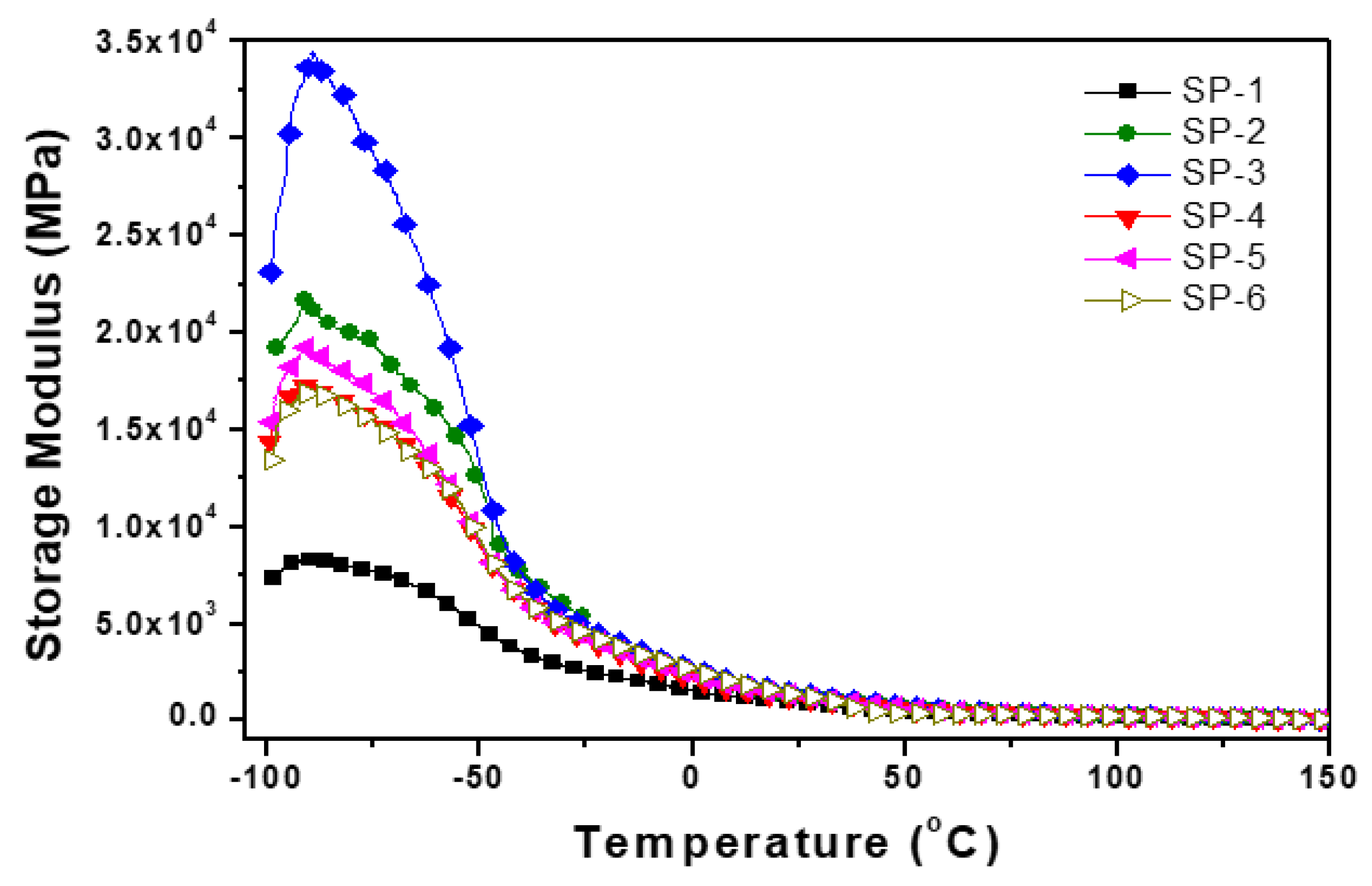
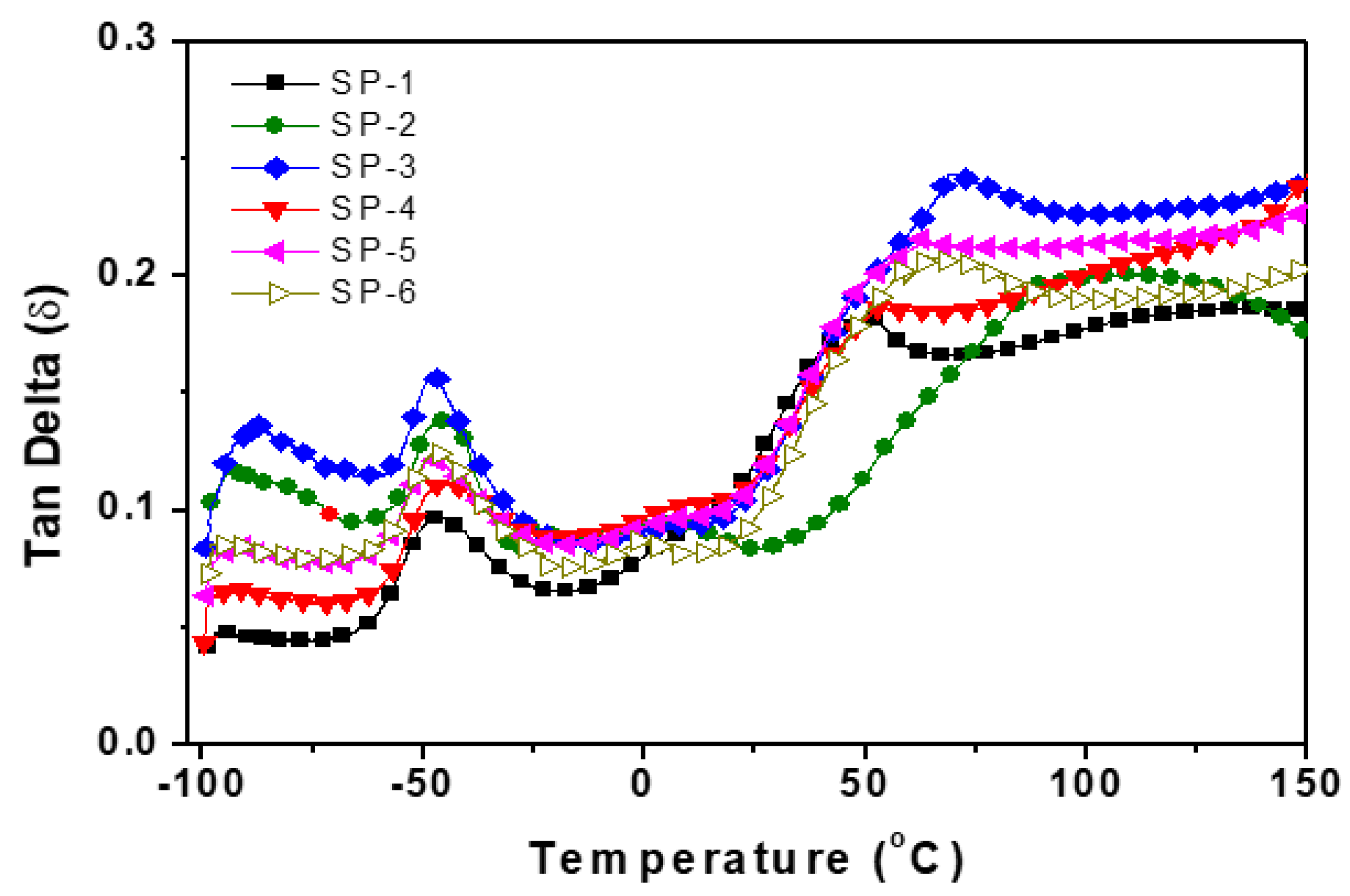
| Sample name |
Storage modulus E’(MPa) −100°C |
Storage modulus E’(MPa) −80°C |
Tg (°C) |
tanδ1 | Loss modulus E’’ T°C | tanδ2 |
Loss modulus E’’(MPa) |
| SP-1 | 7500 | 8000 | 52 | 0.08 | 55 | 0.19 | 1425 |
| SP-2 | 18000 | 22000 | 57 | 0.14 | 100 | 0.22 | 3960 |
| SP-3 | 26000 | 33000 | 55 | 0.16 | 75 | 0.24 | 6240 |
| SP-4 | 14000 | 17500 | 52 | 0.12 | 57 | 0.2 | 2800 |
| SP-5 | 16000 | 19000 | 52 | 0.13 | 65 | 0.18 | 2880 |
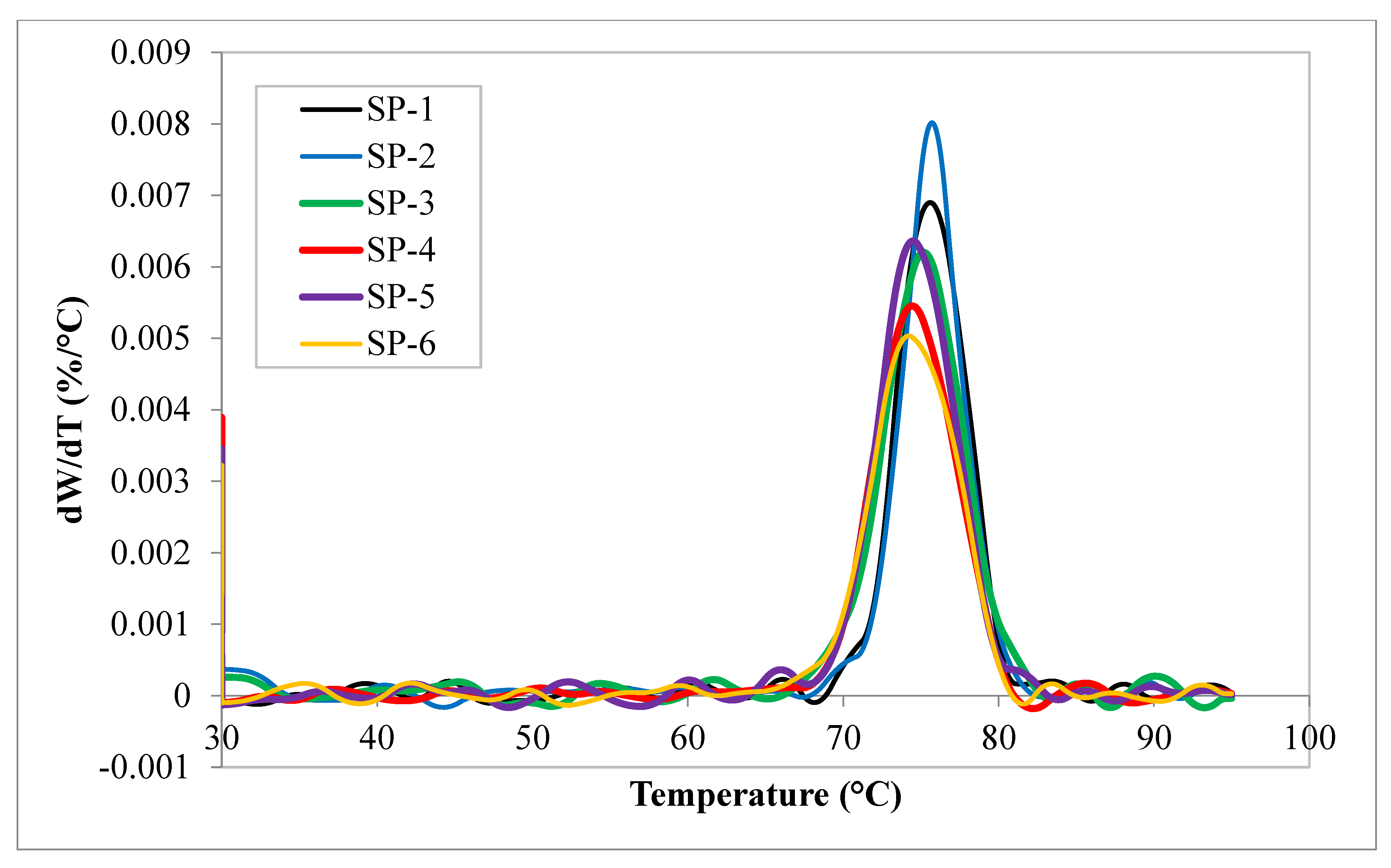
| Composites |
Melting point °C |
Enthalpy of melting (J/g) ΔH | Crystallization exotherm maximum(°C) |
Enthalpy of crystallization (J/g) ΔH |
Crystallinity % |
| SP-1 | 157.35 | 62.37 | 111.34 | 68.92 | 36 |
| SP-2 | 161.15 | 60.58 | 121.25 | 69.51 | 36.6 |
| SP-3 | 163.15 | 63.48 | 113.25 | 72.86 | 38.4 |
| SP-4 | 163.85 | 65.58 | 113.00 | 76.45 | 40.2 |
| SP-5 | 163.89 | 55.08 | 113.55 | 66.68 | 35.1 |
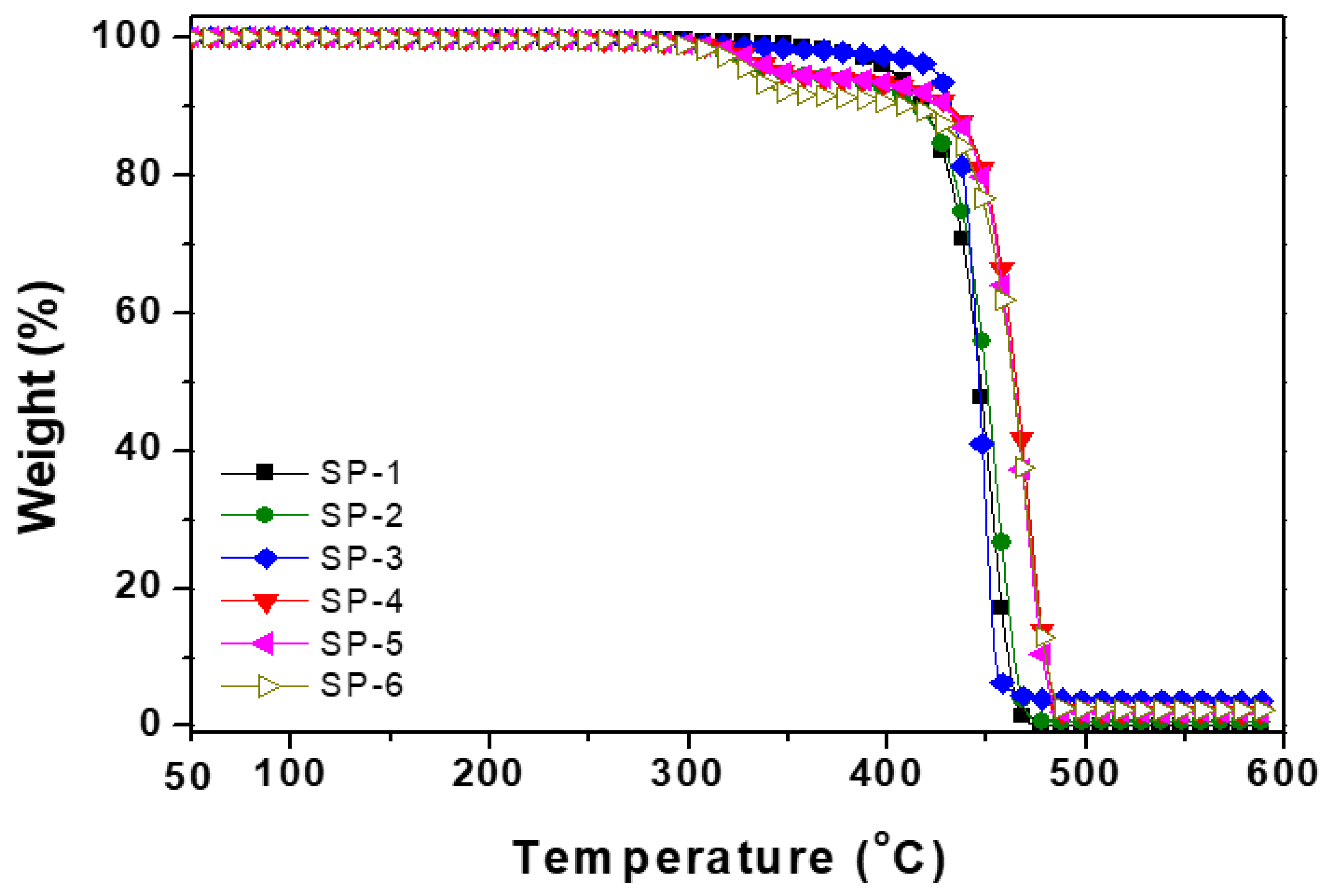
| Composites | Onset 1 temperature °C | Onset 2 temperature °C | Residue wt.% |
| SP-1 | 400 | 425.25 | 0.76 |
| SP-2 | 410 | 435.00 | 3.782 |
| SP-3 | 290.0 | 434.65 | 0.580 |
| SP-4 | 313.6 | 448.00 | 1.926 |
| SP-5 | 309.0 | 446.00 | 2.35 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





