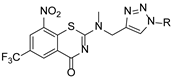
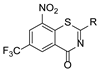
2.2. Characterization Data for the Compounds
BTZ:
8-nitro-6-(trifluoromethyl)-4H-benzo[e][
1,
3]
thiazin-4-one
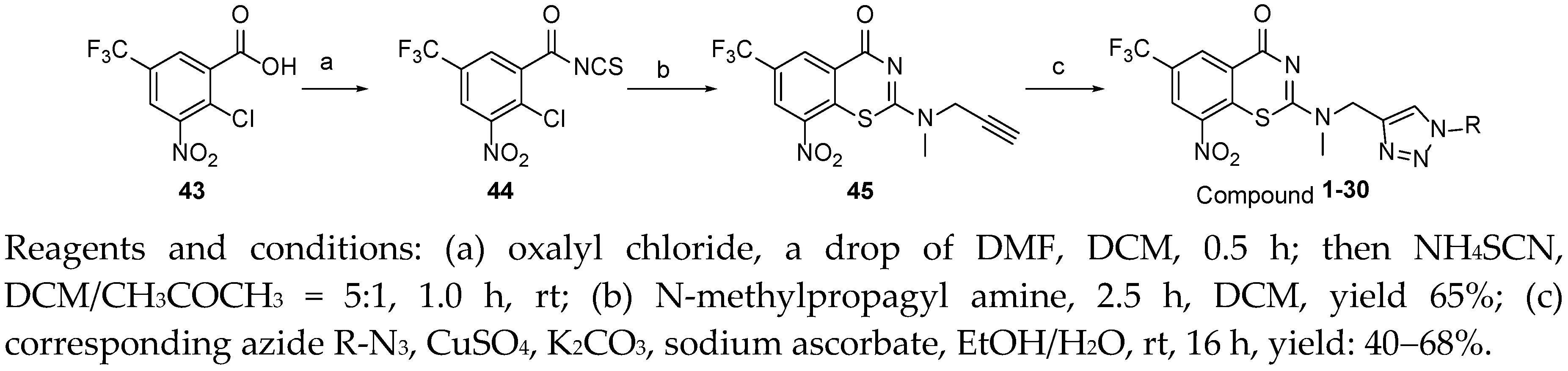
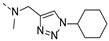
2-(((1-cyclohexyl-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (1). yield 53%; Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 163.3, 166.1, 141.4, 143.9, 133.7 (d, J = 2.2 Hz), 134.4, 130.0 (q, J = 35.6 Hz), 126.0 (d, J = 2.6 Hz), 126.7, 122.5 (q, J = 273.3 Hz), 121.6, 46.7, 60.5, 37.0, 25.3, 33.6, 25.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.77 (s, 1H), 7.77 (s, 1H), 5.12 (s, 2H), 4.43 – 4.37 (m, 1H), 3.51 (s, 3H), 2.16 (d, J = 8.0 Hz, 2H), 1.90 (d, J = 8.0 Hz, 2H), 1.76 – 1.70 (m, 2H), 1.47 – 1.38 (m, 2H), 1.30 – 1.25 (m, 2H); HRMS-ESI (m/z) calcd [M+H]+ for C19H20F3N6O3S+ 469.1264, found 469.1259.
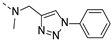
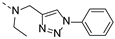
2-(methyl((1-phenyl-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (2). yield 43%; Solid, white; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.4, 144.1, 142.5, 136.8, 134.2, 133.6 (d, J = 3.0 Hz), 130.0 (q, J = 36.1 Hz), 123.0, 129.0, 126.7, 126.1 (d, J = 3.0 Hz), 122.4 (q, J = 273.2 Hz), 122.3, 120.6, 46.7, 37.0; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.76 (s, 1H), 8.24 (s, 1H), 7.70 (d, J = 7.60 Hz, 2H), 7.51 (t, J = 7.2 Hz, 2H), 7.44 (d, J = 7.2 Hz, 1H), 5.20 (s, 2H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H14F3N6O3S+ 463.0795, found 463.0797.
2-(ethyl((1-phenyl-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (3). yield 43%; Solid, white; 13C NMR (151 MHz, CDCl3) δ: 166.2, 162.6, 144.1, 142.9, 137.0, 134.5, 133.6, 129.9, 129.9 (q, J = 34.7 Hz), 129.1, 126.8, 126.2 (d, J = 3.0 Hz), 122.9, 122.5 (q, J = 273.3 Hz), 120.7, 45.1, 44.7, 13.1; 1H NMR (400 MHz, CDCl3) δ: 9.12 (s, 1H), 8.76 (s, 1H), 8.33 (s, 1H), 7.71 (d, J = 8.0 Hz, 2H), 7.50 (t, J = 7.4 Hz, 2H), 7.43 (d, J = 6.8 Hz, 1H), 5.13 (s, 2H), 3.98 (q, J = 7.0 Hz, 2H), 1.50 (t, J = 3.8 Hz, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H16F3N6O3S+ 477.0951, found 477.0944.
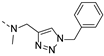
2-(((1-benzyl-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (4). yield 40%; A white solid; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.4, 144.0, 142.3, 134.4, 134.4, 133.6 (d, J = 3.1 Hz), 130.0 (q, J = 36.1 Hz), 129.3, 129.0, 128.3, 126.7, 126.1 (d, J = 3.0 Hz), 123.9, 122.5 (q, J = 273.3 Hz), 54.3, 46.3, 36.8; 1H NMR (400 MHz, CDCl3) δ: 9.12 (s, 1H), 8.78 (s, 1H), 7.72 (s, 1H), 7.36 (br, 3H), 7.28 (br, 2H), 5.50 (s, 2H), 5.11 (s, 2H), 3.51 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H16F3N6O3S+ 477.0951, found 477.0944.
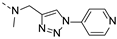
2-(methyl((1-(pyridin-4-yl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (5). yield 49%; A yellow solid; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.6, 151.7, 144.0, 143.4, 142.6, 134.3, 133.7 (d, J = 2.9 Hz), 130.1 (q, J = 35.9 Hz), 126.7 (d, J = 3.3 Hz), 126.3 (d, J = 3.3 Hz), 122.5 (q, J = 273.2 Hz), 121.9, 46.8, 37.2; 1H NMR (400 MHz, DMSO-d6) δ: 9.00 (s, 1H), 8.88 (s, 2H), 8.82 (br, 2H), 7.98 (s, 2H), 5.21 (s, 2H), 3.44 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C18H13F3N7O3S+ calculated 464.0747, found 464.0748.
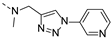
2-(methyl((1-(pyridin-3-yl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (6). yield 57%; A yellow solid; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 150.3, 150.2, 144.0, 143.2, 141.9, 134.3, 133.5 (d, J = 2.6 Hz), 130.1 (q, J = 35.5 Hz), 128.1, 126.6, 126.2 (d, J = 2.9 Hz), 122.5, 122.5 (q, J = 273.3 Hz), 121.6, 46.7, 37.3; 1H NMR (400 MHz, CDCl3) δ: 9.12 (s, 1H), 9.04 (s, 1H), 8.77 (s, 1H), 8.72 (brs, 1H), 8.35 (brs, 1H), 8.08 (d, J = 7.4 Hz, 1H), 7.50 (brs, 1H), 5.20 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+Na]+ for C18H12F3N7NaO3S+ 486.0567, found 486.0547.
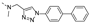
2-(((1-([1,1’-biphenyl]-4-yl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (7). yield 64%; A yellow solid; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.5, 144.0, 142.6, 142.1, 139.6, 136.0, 134.4, 133.7, 130.0 (q, J = 35.49 Hz), 129.1, 128.5, 128.2, 127.2, 126.7, 126.2 (d, J = 2.6 Hz), 122.6 (q, J = 273.1 Hz), 122.2, 120.9, 46.8, 37.0; 1H NMR (400 MHz, CDCl3) δ: 9.14 (s, 1H), 8.77 (s, 1H), 8.30 (s, 1H), 7.79 (d, J = 8.0 Hz, 2H), 7.72 (d, J = 8.0 Hz, 2H), 7.60 (d, J = 7.2 Hz, 2H), 7.47 (t, J = 7.4 Hz, 2H), 7.39 (t, J = 7.2 Hz, 1H), 5.21 (s, 2H), 3.59 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C25H18F3N6O3S+ 539.1108, found 539.1108.
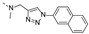
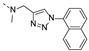
2-(methyl((1-(naphthalen-2-yl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (8). yield 67%; yellow solid; 13C NMR (151 MHz, CDCl3) δ: 166.2 163.6, 134.4, 134.3, 133.6 (d, J = 3.4 Hz), 133.3, 133.1, 130.2, 129.9 (q, J = 35.48 Hz), 128.4, 128.1, 127.7, 127.3, 126.6, 126.2 (d, J = 2.6 Hz), 122.5 (q, J = 273.1 Hz), 122.4, 118.9, 118.7, 46.7, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.14 (d, J = 1.6 Hz, 1H), 8.77 (d, J = 1.6 Hz, 1H), 8.39 (s, 1H), 8.16 (d, J = 1.6 Hz, 1H), 7.99 (d, J = 8.8 Hz, 1H), 7.93 – 7.86 (m, 3H), 7.60 – 7.54 (m, 2H), 5.24 (s, 2H), 3.60 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C23H16F3N6O3S+ 513.0951, found 513.0949.
2-(methyl((1-(naphthalen-1-yl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (9). yield 60%; Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.5, 144.0, 141.9, 134.3, 133.7, 133.5, 130.7, 130.0 (q, J = 35.4 Hz), 128.5, 128.4, 128.1, 127.3, 126.7, 126.2, 125.1, 123.6, 122.5 (q, J = 273.1 Hz), 122.3, 46.8, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.10 (s, 1H), 8.77 (d, J = 1.6 Hz, 1H), 8.19 (s, 1H), 8.02 (d, J = 8.2 Hz, 1H), 7.95 (d, J = 8.0 Hz, 1H), 7.60 – 7.51 (m, 5H), 5.27 (s, 2H), 3.65 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C23H16F3N6O3S+ 513.0951, found 513.0951.
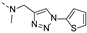
2-(methyl((1-(thiophen-2-ylmethyl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (10). yield 71%; Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.5, 144.0, 142.2, 135.8, 134.4, 133.6 (d, J = 2.6 Hz), 129.9 (q, J = 35.5 Hz), 128.5, 127.5, 127.4, 126.7, 126.1 (d, J = 2.8 Hz), 123.6, 122.5 (q, J = 273.3 Hz), 48.7, 46.5, 36.9; 1H NMR (400 MHz, CDCl3) δ: 9.11 (d, J = 1.6 Hz, 1H), 8.76 (d, J = 1.6 Hz, 1H), 7.77 (s, 1H), 7.31 (d, J = 4.8 Hz, 1H), 7.10 (d, J = 2.8 Hz, 1H), 7.03 – 6.93 (m, 1H), 5.67 (s, 2H), 5.10 (s, 2H), 3.49 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C18H14F3N6O3S2+ 483.0515, found 483.0512.
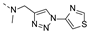
2-(methyl((1-(thiazol-4-ylmethyl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (11). yield 43%; Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.5, 154.3, 150.2, 144.0, 142.2, 134.3, 133.6 (d, J = 3.3 Hz), 129.9 (q, J = 35.6 Hz), 126.7, 126.1 (d, J = 3.3 Hz), 124.3, 122.5 (d, J = 273.1 Hz), 118.1, 49.8, 46.5, 36.9; 1H NMR (400 MHz, CDCl3) δ: 9.11 (d, J = 2.0 Hz, 1H), 8.80 (s, 1H), 8.76 (d, J = 2.0 Hz, 1H), 7.91 (s, 1H), 7.32 (s, 1H), 5.68 (s, 2H), 5.12 (s, 2H), 3.49 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C17H13F3N7O3S2+ 484.0468, found 484.0467.
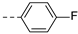
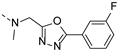
2-(((1-(4-fluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (12). yield 66%; Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 162.8 (d, J = 250.0 Hz), 143.9, 142.7, 134.3, 133.6 (d, J = 2.4 Hz), 133.1, 130.0 (q, J = 35.1 Hz), 126.7, 126.3 (d, J = 3.0 Hz), 122.7 (d, J = 8.4 Hz), 122.6, 122.5 (q, J = 273.1 Hz), 116.9 (d, J = 23.3 Hz), 46.6, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.12 (s, 1H), 8.78 (s, 1H), 8.26 (s, 1H), 7.69 (dd, J = 7.6, 4.4 Hz, 2H), 7.19 (t, J = 8.0 Hz, 2H), 5.20 (s, 2H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13F4N6O3S+ 481.0700, found 481.0692.
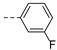
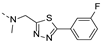
2-(((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (13). yield 59%; Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 163.2 (d, J = 249.0 Hz), 143.9, 142.8, 138.0 (d, J = 9.5 Hz), 134.3, 133.7 (d, J = 2.4 Hz), 131.4 (d, J = 8.5 Hz), 132.9 (q, J = 35.5 Hz), 126.6, 126.2 (d, J = 2.8 Hz), 122.5 (q, J = 273.0 Hz), 122.4, 116.1, 116.0, 108.5 (d, J = 26.3 Hz), 46.7, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.78 (s, 1H), 8.30 (s, 1H), 7.56 – 7.47 (m, 3H), 7.14 (s, 1H), 5.18 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13F4N6O3S+ 481.0700, found:481.0701.
2-(((1-(2-fluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (14). yield 53%; Solid,yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.6, 153.5 (d, J = 251.8 Hz), 143.9, 142.3, 134.2, 133.8 (d, J = 2.9 Hz), 130.7 (d, J = 6.8 Hz), 130.0 (q, J = 35.4 Hz), 126.7, 126.1, 125.4, 125.3, 125.1, 125.0, 122.5 (q, J = 273.2 Hz), 117.3 (d, J = 19.9 Hz), 46.5, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.12 (s, 1H), 8.76 (s, 1H), 8.29 (s, 1H), 7.88 (br, 1H), 7.44 (d, J = 5.2 Hz, 1H), 7.31 (t, J = 8.2 Hz, 2H), 5.23 (s, 2H), 3.56 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13F4N6O3S+ 481.0700, found 481.0696.
2-(((1-(3-chlorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (15). yield 49%; Solid, white color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 144.0, 142.9, 137.7, 135.8, 134.3, 133.7, 131.0, 130.0 (q, J = 35.9 Hz), 129.2, 126.7, 126.2, 122.6, 122.5 (q, J = 273.4 Hz), 121.0, 118.7, 46.8, 37.4; 1H NMR (400 MHz, CDCl3) δ: 9.14 (s, 1H), 8.78 (s, 1H), 8.25 (s, 1H), 7.77 (s, 1H), 7.60 (d, J = 7.3 Hz, 1H), 7.47–7.42 (m, 2H), 5.20 (s, 2H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for: C19H13ClF3N6O3S+ 497.0405, found:497.0407.
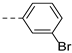
2-(((1-(3-bromophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (16). yield 57%; Solid, white color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.4, 144.0, 142.9, 137.8, 134.2, 133.6 (d, J = 2.4 Hz), 132.2, 131.2, 130.0 (q, J = 35.8 Hz), 126.5, 126.1 (d, J = 2.7 Hz), 123.8, 123.5, 122.5 (q, J = 273.0 Hz), 122.2, 119.1, 46.6, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.77 (s,1H), 8.26 (s, 1H), 7.93 (s, 1H), 7.65 (d, J = 7.6 Hz, 1H), 7.56 (d, J = 7.6 Hz, 1H), 7.38 (t, J = 7.8 Hz, 1H), 5.19 (s, 2H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13BrF3N6O3S+ 540.9900; 542.9879, found 540.9899; 542.9880.
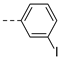
2-(((1-(3-iodophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (17). yield 70%; Solid,white color. 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.6, 144.1, 142.8, 138.2, 137.6, 134.3, 133.7, 131.3, 130.0 (q, J = 35.2 Hz), 129.5, 126.6, 126.2 (d, J = 2.6 Hz), 122.5 (q, J = 273.2 Hz), 122.2, 119.8, 94.6, 46.6, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.77 (s, 1H), 8.24 (s, 1H), 8.10 (s, 1H), 7.76 (d, J = 7.6 Hz, 1H), 7.68 (d, J = 7.6 Hz, 1H), 7.22 (t, J = 7.6 Hz, 1H), 5.19 (s, 2H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13F3IN6O3S+ 588.9761, found: 588.9760.
2-(((1-(3-methoxyphenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (18). yield 57%; Solid, white color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.5, 160.8, 144.1, 142.5, 137.9, 134.3, 133.7, 130.7, 130.0 (q, J = 35.6 Hz), 126.7, 126.2 (d, J = 2.6 Hz), 122.5 (q, J = 273.2 Hz), 122.3, 115.0, 112.6, 106.5, 55.8, 46.6, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.12 (s, 1H), 8.77 (s, 1H), 8.24 (s, 1H), 7.39 (t, J = 8.0 Hz, 1H), 7.30 (s, 1H), 7.23 (d, J = 7.8 Hz, 1H), 6.95 (d, J = 7.8 Hz, 1H), 5.20 (s, 2H), 3.86 (s, 3H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H16F3N6O4S+ 493.0900, found 493.0900.
2-(methyl((1-(m-tolyl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (19). yield 66%; Solid,white color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.5, 144.0, 142.5, 140.2, 136.8, 134.4, 133.7 (d, J = 2.5 Hz), 130.0 (q, J = 35.6 Hz), 129.9, 129.7, 126.7, 126.2 (d, J = 2.7 Hz), 122.5 (q, J = 273.2 Hz), 122.3, 121.3, 117.8, 46.6, 37.1, 21.5; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.77 (s, 1H), 8.22 (s, 1H), 7.53 (s, 1H), 7.48 (d, J = 8.0 Hz, 1H), 7.37 (t, J = 7.6 Hz, 1H), 7.23 (d, J = 7.6 Hz, 1H), 5.20 (s, 2H), 3.57 (s, 3H), 2.43 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H16F3N6O3S+ 477.0951, found 477.0948.
2-(methyl((1-(3-(trifluoromethyl)phenyl)-1H-1,2,3-triazol-4-yl)methyl)amino)-BTZ (20). yield 65%; Solid, white color, 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.7, 144.0, 143.1, 137.2, 134.3, 132.7 (q, J = 33.5 Hz), 130.7, 130.0 (q, J = 35.6 Hz), 129.6 (d, J = 4.0 Hz), 126.6, 126.2 (d, J = 3.3 Hz), 125.8, 123.8 (q, J = 272.8 Hz), 123.7, 123.4 (q, J = 272.7 Hz), 122.4, 117.8 (d, J = 3.7 Hz), 46.6, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.79 (s, 1H), 8.34 (s, 1H), 8.04 (br, 1H), 7.91 (br, 1H), 7.68 (d, J = 12.0 Hz, 2H), 5.20 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H13F6N6O3S+ 531.0669, found 531.0669.
3-(4-((methyl(8-nitro-4-oxo-6-(trifluoromethyl)-4H-benzo[e][
1,
3]thiazin-2-yl)amino)methyl)-1H-1,2,3-triazol-1-yl)benzonitrile (21). yield 63%; Solid, white color.
13C NMR (151 MHz, CDCl
3) δ: 166.1, 163.5, 143.9, 143.3, 137.5, 134.3, 133.7 (d, J = 2.30 Hz), 132.4, 131.2, 130.1 (q, J = 35.6 Hz), 126.6, 126.3 (d, J = 3.1 Hz), 124.5, 123.9, 122.5 (q, J = 273.3 Hz), 122.3, 117.4, 114.5, 46.7, 37.3;
1H NMR (400 MHz, CDCl
3) δ: 9.13 (s, 1H), 8.77 (s, 1H), 8.36 (s, 1H), 8.10 (s, 1H), 7.98 (d, J = 7.60 Hz, 1H), 7.73 (d, J = 7.40 Hz, 1H), 7.66 (t, J = 7.80 Hz, 1H), 5.19 (s, 2H), 3.59 (s, 3H); HRMS-ESI (m/z) calcd [M+H]
+ for C
20H
13F
3N
7O
3S
+ 488.0747, found 488.074.
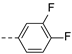
2-(((1-(3,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (22). yield 61%; Solid,white color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 150.8 (dd, J = 252.2, 13.60 Hz), 150.5 (dd, J = 253.70, 13.6 Hz), 144.0, 143.0, 134.3 133.7 (d, J = 1.50 Hz),133.2, 130.1 (q, J = 36.24 Hz), 126.6, 126.2 (d, J = 3.0 Hz), 122.5 (q, J = 273.3 Hz), 122.5, 118.6 (d, J = 19.4 Hz), 116.7, 110.7 (d, J = 22.7 Hz), 46.7, 37.3; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.78 (s, 1H), 8.26 (s, 1H), 7.68 – 7.64 (m, 1H), 7.44 (brs, 1H), 7.35 – 7.28 (m, 1H), 5.17 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F5N6O3S+ 499.0606, found 499.0606.
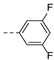
2-(((1-(3,5-difluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (23). yield 43%; Solid, white color. 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 163.55 (dd, J = 251.10, 13.7 Hz), 144.0, 143.1, 138.4 (d, J = 8.90 Hz), 134.3, 133.7 (d, J = 2.60 Hz), 130.1 (q, J = 35.40 Hz), 126.6, 126.3 (d, J = 3.0 Hz), 122.5 (q, J = 273.30 Hz), 122.4, 104.3 (d, J = 40.10 Hz), 104.2 (d, J = 30.50 Hz), 46.7, 37.3; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.78 (s, 1H), 8.30 (s, 1H), 7.34 (d, J = 5.20 Hz, 2H), 6.89 (brs, 1H), 5.18 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F5N6O3S+ 499.0606, found 499.0603.
2-(((1-(2,6-difluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (24). yield 57%; Solid, white color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.6, 156.9 (d, J = 256.70 Hz), 144.0, 141.8, 134.4, 133.7 (d, J = 2.10 Hz), 131.71 (t, J = 9.10 Hz), 130.0 (q, J = 35.50 Hz), 127.0, 126.7,126.2 (d, J = 3.00 Hz), 122.5 (q, J = 273.30 Hz), 115.1 (t, J = 15.10 Hz), 112.7 (d, J = 19.80 Hz), 46.6, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.11 (s, 1H), 8.77 (s, 1H), 8.10 (s, 1H), 7.50 – 7.47 (m, 1H), 7.12 (t, J = 8.4 Hz, 2H), 5.22 (s, 2H), 3.59 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F5N6O3S+ 499.0606, found 499.0602.
2-(((1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (25). yield 85%; Solid, white color. 13C NMR (151 MHz, CDCl3) δ: 166.0, 163.6, 162.7 (dd, J = 253.40, 10.9 Hz), 154.1 (dd, J = 254.70, 11.8 Hz), 144.0, 142.4, 134.3, 133.7 (d, J = 2.80 Hz), 130.0 (q, J = 35.50 Hz), 126.7,126.4 (d, J = 9.90 Hz), 126.1 (d, J = 2.60 Hz), 125.3 (d, J = 4.10 Hz), 122.5 (q, J = 273.60 Hz), 121.9 (d, J = 7.20 Hz), 112.7 (d, J = 22.40 Hz), 105.6 (dd, J = 78.5, 2.30 Hz), 46.5, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.78 (s, 1H), 8.26 (s, 1H), 7.88 – 7.82 (m, 1H), 7.08– 7.03 (m, 2H), 5.21 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F5N6O3S+ 499.0606, found 499.0607.
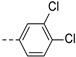
2-(((1-(3,4-dichlorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (26). yield 65%; Solid, white color. 13C NMR (151 MHz, CDCl3) δ: 166.3, 163.6, 144.1, 143.1, 135.9, 134.2, 133.6 (d, J = 2.000 Hz), 133.4, 131.6, 130.1 (q, J = 35.3 Hz), 126.5, 126.3 (d, J = 3.0 Hz), 122.5 (q, J = 273.30 Hz), 122.5, 122.4, 122.3, 119.6, 46.7, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.14 (s, 1H), 8.78 (s, 1H), 8.29 (s, 1H), 7.91 (s, 1H), 7.59 (br, 2H),5.18 (s, 2H), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12Cl2F3N6O3S+ 531.0015, found 531.0012.
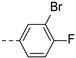
2-(((1-(3-bromo-4-fluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (27). yield 74%; solid, white color. Poor solubility to obtain a 13C NMR. 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.78 (s, 1H), 8.25 (s, 1H), 7.98 (d, J = 3.2 Hz, 1H), 7.65 (d, J = 8.4 Hz, 1H), 7.29 (br, 1H), 5.18 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12BrF4N6O3S+ 558.9806, 560.9785, found 558.9794,560.9766.
2-(((1-(3-chloro-4-fluorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (28). 87%; Solid, white color. 13C NMR (151 MHz, CDCl3) δ: 166.3, 163.5, 158.2 (d, J = 252.2 Hz), 144.0, 143.0, 134.4, 133.6, 133.5, 130.1 (q, J = 34.7 Hz), 126.5, 126.3 (d, J = 3.0 Hz), 123.3, 122.8, 122.5, 122.5 (q, J = 273.3 Hz), 120.4 (d, J = 7.6 Hz), 117.8 (d, J = 22.7 Hz), 46.8, 37.3; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.78 (s, 1H), 8.24 (s, 1H), 7.85 (d, J = 4.41 Hz, 1H), 7.61 (d, J = 7.19 Hz, 1H), 7.31 – 7.29 (m, 1H), 5.17 (s, 2H), 3.56 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12ClF4N6O3S+ 515.0311, found 515.0303.
2-(((1-(2,4-dichlorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (29). yield 72%; Solid, white color; 13C NMR (151 MHz, CDCl3) δ: 166.0, 163.4, 143.9, 141.9, 136.6, 134.4, 133.8 (d, J = 2.6 Hz), 133.4, 130.8, 130.0 (q, J = 35.6 Hz), 129.6, 128.5, 128.4, 126.6, 126.1, 122.5 (q, J = 273.0 Hz), 46.6, 37.3; 1H NMR (400 MHz, CDCl3) δ: 9.11 (d, J = 1.62 Hz, 1H), 8.77 (d, J = 1.62 Hz, 1H), 8.22 (s, 1H), 7.58 (d, J = 2.0 Hz, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.41 (dd, J = 8.4, 1.6 Hz, 1H), 5.21 (s, 2H), 3.58 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12Cl2F3N6O3S+ 531.0015, found 531.0015.
2-(((1-(3,5-dichlorophenyl)-1H-1,2,3-triazol-4-yl)methyl)(methyl)amino)-BTZ (30). yield 75%; Solid, white color. 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.7, 144.0, 143.1, 138.1, 136.5, 134.2, 133.7 (d, J = 2.8 Hz), 130.1 (q, J = 35.2 Hz), 129.1, 126.6, 126.2 (d, J = 2.7 Hz), 122.6 (q, J = 273.0 Hz), 122.2, 119.1, 46.6, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (d, J = 1.6 Hz, 1H), 8.78 (d, J = 1.2 Hz, 1H), 8.28 (s,1H), 7.68 (d, J = 1.6 Hz, 2H), 7.42 (s, 1H), 5.20 (s, 2H), 3.56 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12Cl2F3N6O3S+ 531.0015, found 531.0016.
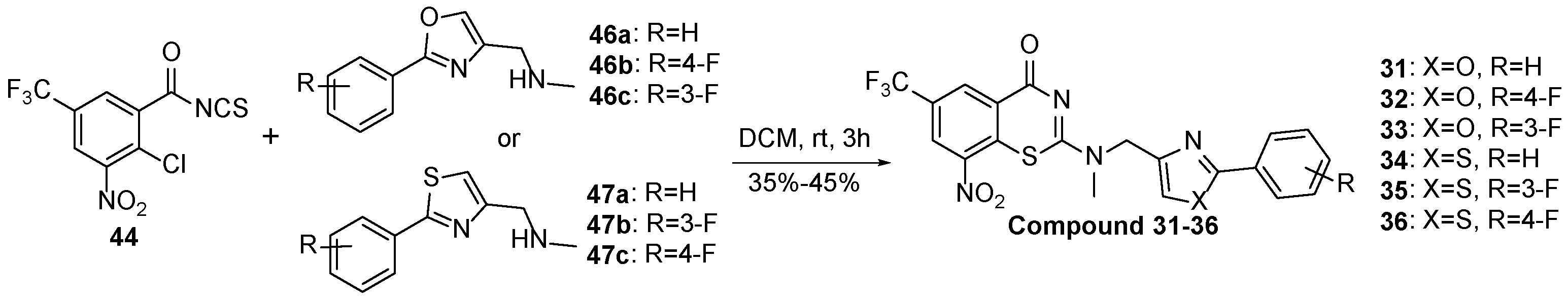
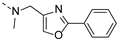
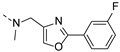
2-(methyl((2-phenyloxazol-4-yl)methyl)amino)-BTZ (31). Solid, white color; 1H NMR indicates 3:1 atropisomeric ratio through the integral value of −CH2 protons and the oxazole – CH protons; 13C NMR (151 MHz, CDCl3) δ: 166.2, 166.1, 162.1, 144.0, 137.6, 136.2, 134.4, 133.7, 130.8, 129.9 (q, J = 35.40 Hz), 129.0, 126.8, 126.7, 126.1, 122.5 (q, J = 273.3 Hz), 116.3, 46.9, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.15 (s, 1H), 8.76 (s, 1H), 8.01 (br, 2H), 7.85 (s, 0.75H, major), 7.77 (s, 0.25H, minor), 7.45 (br, 3H), 5.06 (s, 1.5H, major), 4.88 (s, 0.5H, minor), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H14F3N4O4S+ 463.0682, found 463.0683.
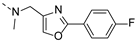
2-(((2-(4-fluorophenyl)oxazol-4-yl)methyl)(methyl)amino)-BTZ (32). Solid, white; 1H NMR indicates 3:1 atropisomeric ratio through the integral value of −CH2 protons and the oxazole −CH protons; 13C NMR (151 MHz, CDCl3) δ: 166.1, 164.3 (d, J = 252.4 Hz), 163.5, 161.2, 144.0, 137.6, 136.2, 134.4, 133.7, 129.9 (d, J = 35.6 Hz), 128.7 (d, J = 7.9 Hz), 126.7, 126.1, 123.6, 122.5 (d, J = 273.0 Hz), 116.0 (d, J = 22.1 Hz), 46.9, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.09 (s, 1H), 8.72 (s, 1H), 7.96 (br, 2H), 7.78 (s, 0.75H, major), 7.72 (s, 0.25H, minor), 7.09 (t, J = 7.5 Hz, 2H), 4.99 (s, 1.5H, major), 4.81 (s, 0.5H, minor), 3.51 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H13F4N4O4S+ 481.0588, found 481.0586.
2-(((2-(3-fluorophenyl)oxazol-4-yl)methyl)(methyl)amino)-BTZ (33). A white solid; 1H NMR indicates 3:1 atropisomeric ratio through the integral value of −CH2 protons and the oxazole −CH protons; 13C NMR (151 MHz, CDCl3) δ: 166.1, 163.5, 160.1 (d, J = 256.0 Hz), 158.4, 138.0, 137.6, 136.2, 134.4, 133.7, 132.4 (d, J = 7.2 Hz), 129.9 (d, J = 35.6 Hz), 129.7, 128.9, 126.7, 126.1, 124.5, 122.5 (q, J = 273.2 Hz), 117.0 (d, J = 21.2 Hz), 46.8, 37.1; 1H NMR (400 MHz, CDCl3) δ: 9.09 (s, 1H), 8.73 (s, 1H), 7.95 (s, 1H), 7.86 (s, 0.75H, major), 7.79 (s, 0.25H, minor), 7.41 (br, 1H), 7.21 – 7.10 (m, 2H), 5.04 (s, 1.5H, major), 4.85 (s, 0.5H, minor), 3.52 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H13F4N4O4S+ 481.0588, found 481.0587.
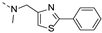
2-(methyl((2-phenylthiazol-4-yl)methyl)amino)-BTZ (34). Solid, white color; 1H NMR indicates 3:1 atropisomeric ratio through the integral value of −CH2 protons; 13C NMR (151 MHz, CDCl3) δ: 168.7, 166.2, 163.5, 151.1, 144.0, 134.5, 133.7, 130.4, 129.8 (q, J = 35.4 Hz), 129.1, 126.9, 126.6, 126.1, 122.5 (q, J = 273.3 Hz), 118.2, 116.8, 50.7, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.76 (d, J = 1.9 Hz, 1H), 7.96 – 7.88 (m, 2H), 7.44 – 7.41 (m, 4H), 5.22 (s, 1.5H , major), 5.03 (s, 0.5H , minor), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H14F3N4O3S2+ 479.0454, found 479.0451.
2-(((2-(3-fluorophenyl)thiazol-4-yl)methyl)(methyl)amino)-BTZ (35). Solid, white color; 1H NMR indicates 3:1 atropisomeric ratio through the integral value of −CH2 protons and the thiazole −CH protons; 13C NMR (151 MHz, CDCl3) δ: 166.2, 163.6, 160.1 (d, J = 253.5 Hz), 150.1, 144.0, 134.5, 133.7, 131.4 (d, J = 7.2 Hz), 129.8 (q, J = 34.6 Hz), 129.1, 128.8, 126.8, 126.6, 126.1, 124.7, 122.5 (q, J = 272.8 Hz), 119.6 (d, J = 7.3 Hz), 116.3 (d, J = 21.8 Hz), 50.7, 37.2; 1H NMR (400 MHz, CDCl3) δ: 9.13 (s, 1H), 8.76 (s, 1H), 8.24 (br, 1H), 7.91 (s, 0.75H,major), 7.52 (s, 0.25H,minor), 7.42 (br, 2H), 7.23 – 7.16(m, 1H), 5.24 (s, 1.5H, major), 5.06 (s, 0.5H, minor), 3.57 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H13F4N4O3S2+ 497.0360, found 497.0358.
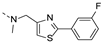
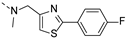
2-(((2-(4-fluorophenyl)thiazol-4-yl)methyl)(methyl)amino)-BTZ (36). Solid, white color; 1H NMR indicates 2:3 atropisomeric ratio through the integral value of −CH2 protons and the thiazole −CH protons; 13C NMR (151 MHz, CDCl3) δ: 166.3, 163.5, 162.3 (d, J = 279.0 Hz), 151.1, 144.0, 134.5, 133.7, 130.4, 129.8 (q, J = 35.4 Hz), 129.1, 126.8, 126.7, 126.1, 122.5 (d, J = 273.1 Hz), 118.2, 116.8, 50.7, 37.2; 1H NMR (400 MHz, DMSO-d6) δ: 8.86 (s, 1H), 8.82 (s, 1H), 7.92 (br, 2H), 7.80 (s, 0.4 H, minor), 7.63 (s, 0.6 H, major), 7.49 (br, 2H), 5.18 (s, 1.2H, major), 5.14 (s, 0.8H, minor), 3.47 – 3.41 (m, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C20H13F4N4O3S2+ 497.0360, found 497.0358.
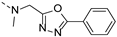
2-(methyl((5-phenyl-1,3,4-oxadiazol-2-yl)methyl)amino)-BTZ (37). Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.2, 166.1, 164.8, 160.8, 144.0, 133.9 (d, J = 2.8 Hz), 132.2, 130.2 (q, J = 35.49 Hz), 129.2, 127.1, 126.5, 126.2 (d, J = 3.2 Hz), 123.2, 122.3 (q, J = 274.0 Hz), 45.2, 36.7; 1H NMR (400 MHz, CDCl3) δ: 9.15 (s, 1H), 8.82 (s, 1H), 8.03 (d, J = 7.60 Hz, 2H), 7.56 – 7.46 (m, 3H), 5.43 (s, 2H), 3.54 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13F3N5O4S+ 464.0635, found 464.0632.
2-(((5-(3-fluorophenyl)-1,3,4-oxadiazol-2-yl)methyl)(methyl)amino)-BTZ (38). Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.1,165.6, 164.9, 162.8, 160.2 (d, J = 259.0 Hz), 144.1, 134.2 (d, J = 5.4 Hz), 133.8, 130.3 (q, J = 35.8 Hz), 130.0, 126.6, 126.4 (d, J = 3.2 Hz), 124.9 (d, J = 3.1 Hz), 122.4 (q, J = 273.6 Hz), 117.2 (d, J = 20.8 Hz), 111.9 (d, J = 14.2 Hz), 110.2, 45.3, 37.0; 1H NMR (400 MHz, CDCl3) δ: 9.14 (s, 1H), 8.81 (s, 1H), 8.03 (t, J = 7.0 Hz, 1H), 7.55 (dd, J = 6.8 Hz , J = 12.4 Hz , 1H), 7.31 – 7.21 (m, 2H), 5.44 (s, 2H), 3.56 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F4N5O4S+ 482.0541, found 482.0541.
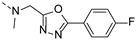
2-(((5-(4-fluorophenyl)-1,3,4-oxadiazol-2-yl)methyl)(methyl)amino)-BTZ (39). Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 166.1, 165.3, 165.2 (d, J = 254.1 Hz), 165.0, 161.0, 144.2, 134.0 (d, J = 2.9 Hz), 130.3 (q, J = 35.5 Hz), 129.6 (d, J = 8.9 Hz), 126.6, 126.4 (d, J = 3.2 Hz), 122.4 (q, J = 273.6 Hz), 119.7, 116.7 (d, J = 22.3 Hz), 45.2, 37.0; 1H NMR (400 MHz, CDCl3) δ: 9.14 (s, 1H), 8.82 (s, 1H), 8.04 – 8.01 (m, 2H), 7.18 (t, J = 8.0 Hz, 2H), 5.41 (s, 2H), 3.55 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F4N5O4S+ 482.0541, found 482.0541.
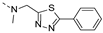
2-(methyl((5-phenyl-1,3,4-thiadiazol-2-yl)methyl)amino)-BTZ (40). Solid, white color; 13C NMR (151 MHz, CDCl3) δ: 171.2, 165.8, 164.0, 162.1, 134.0, 133.9 (d, J = 3.2 Hz), 131.6, 130.3 (q, J = 35.6 Hz), 129.8, 129.4, 128.1, 126.6, 126.4 (d, J = 3.5 Hz), 122.4 (q, J = 273.2 Hz), 110.0, 49.8, 36.9; 1H NMR (400 MHz, CDCl3) δ: 9.17 (s, 1H), 8.82 (s, 1H), 7.93 (d, J = 6.8 Hz, 2H), 7.49 – 7.45 (m, 3H), 5.44 (s, 2H), 3.54 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H13F3N5O3S2+ 480.0406, found 480.0405.
2-(((5-(3-fluorophenyl)-1,3,4-thiadiazol-2-yl)methyl)(methyl)amino)-BTZ (41). Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 170.0, 165.8, 164.1, 163.7, 159.7 (d, J = 253.0 Hz), 144.1, 133.9 (d, J = 3.0 Hz), 133.1 (d, J = 8.4 Hz), 130.3 (q, J = 35.6 Hz), 129.2, 126.7, 126.3 (d, J = 3.0 Hz), 125.1, 122.3 (q, J = 273.1 Hz), 118.1 (d, J = 7.7 Hz), 116.6 (d, J = 21.7 Hz), 49.5, 36.8; 1H NMR (400 MHz, CDCl3) δ: 9.17 (s, 1H), 8.81 (s, 1H), 8.35 (t, J = 6.8 Hz, 1H), 7.50 (dd, J = 5.6 Hz, J = 11.2 Hz, 1H), 7.30 (t, J = 7.19 Hz, 1H), 7.20 (t, J = 9.6 Hz, 1H), 5.50 (s, 2H), 3.54 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F4N5O3S2+ 498.0312, found 498.0314.
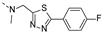
2-(((5-(4-fluorophenyl)-1,3,4-thiadiazol-2-yl)methyl)(methyl)amino)-BTZ (42). Solid, yellow color; 13C NMR (151 MHz, CDCl3) δ: 170.1, 165.7, 164.6 (d, J = 253.1 Hz), 163.9, 162.1, 144.1, 134.0 (d, J = 3.0 Hz), 130.3, 130.1 (d, J = 8.7 Hz), 123.0 (q, J = 35.4 Hz), 126.5, 126.3 (d, J = 3.2 Hz), 126.2, 122.3 (q, J = 273.2 Hz), 116.5 (d, J = 22.1 Hz), 49.9, 37.0; 1H NMR (400 MHz, CDCl3) δ: 9.17 (s, 1H), 8.82 (s, 1H), 8.02 – 7.86 (m, 2H), 7.14 (t, J = 8.0 Hz, 2H), 5.42 (s, 2H), 3.53 (s, 3H); HRMS-ESI (m/z) calcd [M+H]+ for C19H12F4N5O3S2+ 498.0312, found 498.0317.