Submitted:
25 May 2023
Posted:
26 May 2023
You are already at the latest version
Abstract
Keywords:
Introduction
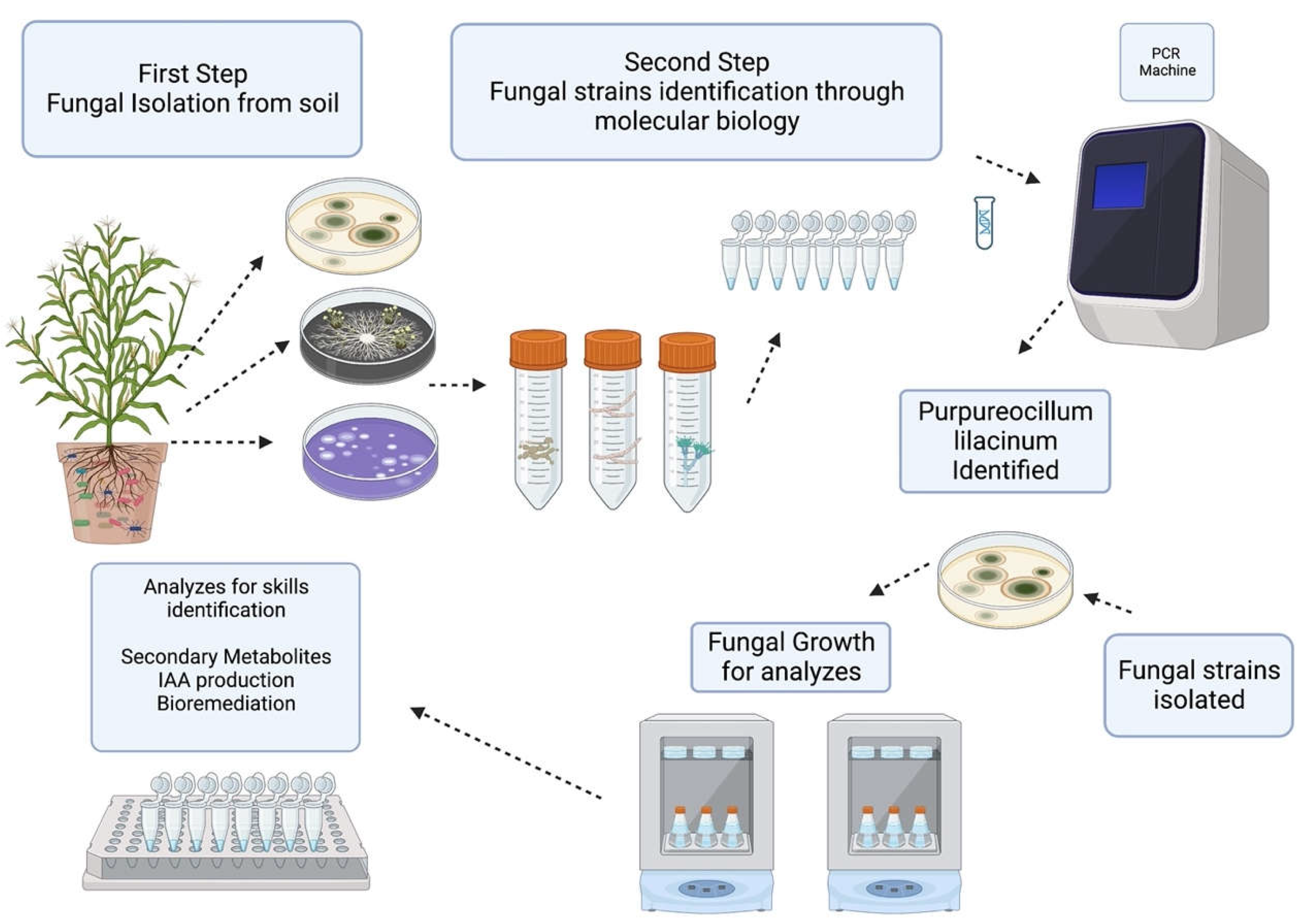
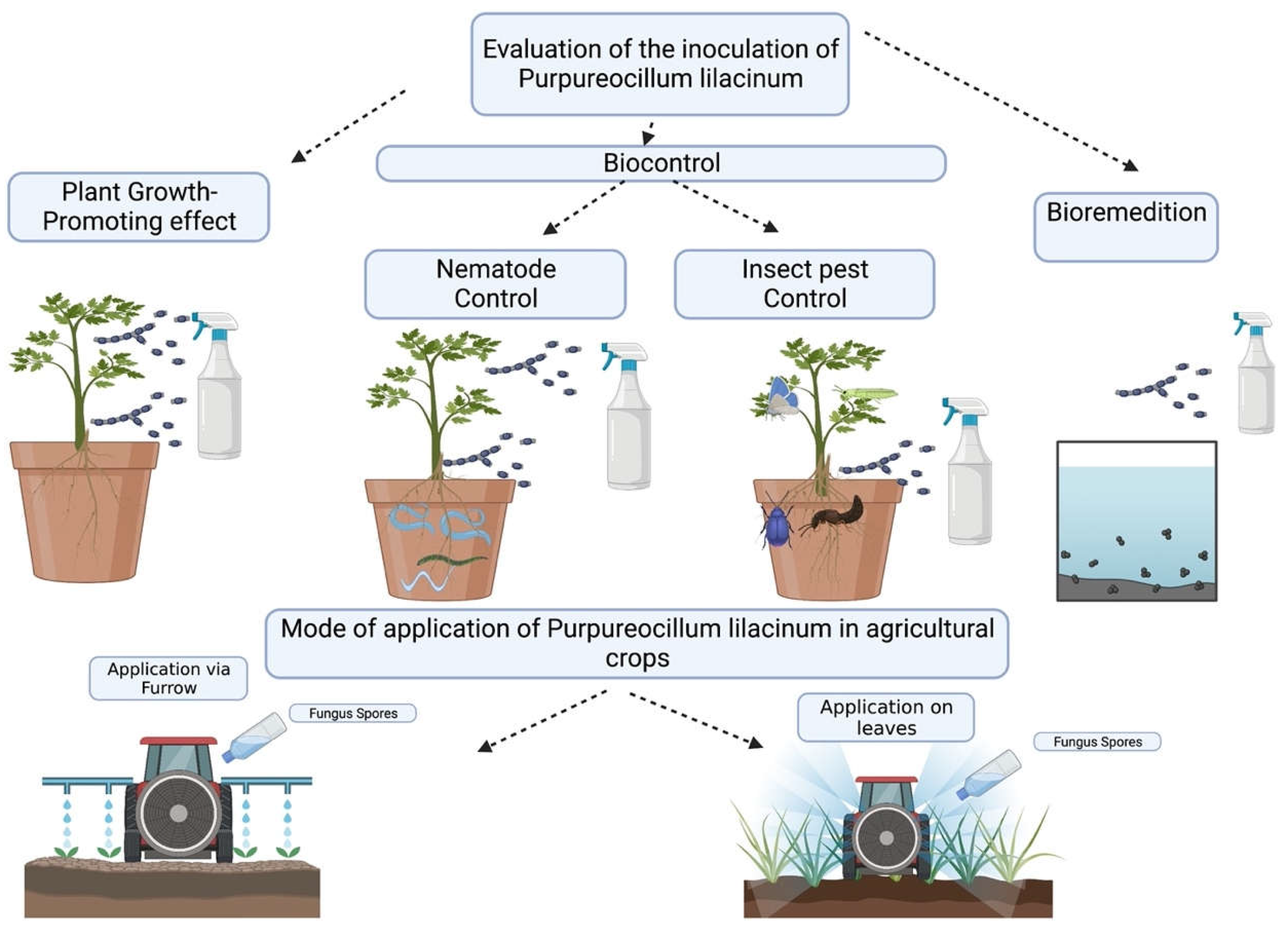
Purpureocillium lilacinum for biocontrol
Purpureocillium lilacinum as biofertilizer
Purpureocillum lilacinum for bioremediation
Mechanisms of action
Conclusions
Future Perspectives
Funding
References
- Backer, R.; Rokem, J.S.; Ilangumaran, G.; Lamont, J.; Praslickova, D.; Ricci, E.; Subramanian, S.; Smith, D.L. Plant growth-promoting rhizobacteria: context, mechanisms of action, and roadmap to commercialization of biostimulants for sustainable agriculture. Frontiers in plant science 2018, 9, 1473. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Pataczek, L.; Hilger, T.H.; Zahir, Z.A.; Hussain, A.; Rasche, F.; Schafleitner, R.; Solberg, S.Ø. Perspectives of microbial inoculation for sustainable development and environmental management. Frontiers in microbiology 2018, 9, 2992. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Salazar, R.; Sánchez-García, I.; Chan-Cupul, W.; Ruiz-Sánchez, E.; Hernández-Ortega, H.A.; Pineda-Lucatero, J.; Figueroa-Chávez, D. Plant growth, foliar nutritional content and fruit yield of Capsicum chinense biofertilized with Purpureocillium lilacinum under greenhouse conditions. Scientia Horticulturae 2020, 261, 108950. [Google Scholar] [CrossRef]
- Alves, G.S.; Bertini, S.C.B.; Barbosa, B.B.; Pimentel, J.P.; Junior, V.A.R.; de Oliveira Mendes, G.; Azevedo, L.C.B. Fungal endophytes inoculation improves soil nutrient availability, arbuscular mycorrhizal colonization and common bean growth. Rhizosphere 2021, 18, 100330. [Google Scholar] [CrossRef]
- Dahlin, P.; Eder, R.; Consoli, E.; Krauss, J.; Kiewnick, S. Integrated control of Meloidogyne incognita in tomatoes using fluopyram and Purpureocillium lilacinum strain 251. Crop Protection 2019, 124, 104874. [Google Scholar] [CrossRef]
- Lopez, D.C.; Sword, G.A. The endophytic fungal entomopathogens Beauveria bassiana and Purpureocillium lilacinum enhance the growth of cultivated cotton (Gossypium hirsutum) and negatively affect survival of the cotton bollworm (Helicoverpa zea). Biological Control 2015, 89, 53–60. [Google Scholar] [CrossRef]
- Chen, W.; Hu, Q. Secondary metabolites of Purpureocillium lilacinum. Molecules 2021, 27, 18. [Google Scholar] [CrossRef]
- Spina, F.; Tummino, M.L.; Poli, A.; Prigione, V.; Ilieva, V.; Cocconcelli, P.; Puglisi, E.; Bracco, P.; Zanetti, M.; Varese, G.C. Low density polyethylene degradation by filamentous fungi. Environmental Pollution 2021, 274, 116548. [Google Scholar] [CrossRef]
- Baron, N.C.; de Souza Pollo, A.; Rigobelo, E.C. Purpureocillium lilacinum and Metarhizium marquandii as plant growth-promoting fungi. PeerJ 2020, 8, e9005. [Google Scholar] [CrossRef]
- Messa, V.R.; da Costa, A.C.T.; Kuhn, O.J.; Stroze, C.T. Nematophagous and endomycorrhizal fungi in the control of Meloidogyne incognita in soybean. Rhizosphere 2020, 15, 100222. [Google Scholar] [CrossRef]
- Nagachandrabose, S. Management of potato cyst nematodes using liquid bioformulations of Pseudomonas fluorescens, Purpureocillium lilacinum and Trichoderma viride. Potato Research 2020, 63, 479–496. [Google Scholar] [CrossRef]
- Barbosa, B.B.; Pimentel, J.P.; Rodovalho, N.S.; Bertini, S.C.B.; Kumar, A.; Ferreira, L.F.R.; Azevedo, L.C.B. Ascomycetous isolates promote soil biological and nutritional attributes in corn and soybeans in sandy and clayey soils. Rhizosphere 2022, 24, 100625. [Google Scholar] [CrossRef]
- El-Ashry, R.; Ali, M.A.; Elsobki, A.E.; Aioub, A.A. Integrated management of Meloidogyne incognita on tomato using combinations of abamectin, Purpureocillium lilacinum, rhizobacteria, and botanicals compared with nematicide. Egyptian Journal of Biological Pest Control 2021, 31, 1–10. [Google Scholar] [CrossRef]
- Giné, A.; Sorribas, F.J. Effect of plant resistance and BioAct WG (Purpureocillium lilacinum strain 251) on Meloidogyne incognita in a tomato–cucumber rotation in a greenhouse. Pest management science 2017, 73, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Girardi, N.S.; Sosa, A.L.; Etcheverry, M.G.; Passone, M.A. In vitro characterization bioassays of the nematophagous fungus Purpureocillium lilacinum: Evaluation on growth, extracellular enzymes, mycotoxins and survival in the surrounding agroecosystem of tomato. Fungal Biology 2022, 126, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Sharma, S.; Sabir, N.; El-Sheikh, M.A.; Alyemeni, M. Impact assessment of Karanja deoiled cake and sundried biogas slurry as a mixed substrate on the nematicidal potential of Purpureocillium lilacinum. Journal of King Saud University-Science 2021, 33, 101399. [Google Scholar] [CrossRef]
- Silva, D.M.; de Souza, V.H.M.; Moral, R.d.A.; Delalibera Júnior, I.; Mascarin, G.M. Production of Purpureocillium lilacinum and Pochonia chlamydosporia by Submerged Liquid Fermentation and Bioactivity against Tetranychus urticae and Heterodera glycines through Seed Inoculation. Journal of Fungi 2022, 8, 511. [Google Scholar] [CrossRef]
- Chapman, M.A. Introduction: The importance of eggplant. The Eggplant Genome 2019, 1–10. [Google Scholar]
- Khan, M.; Tanaka, K. Purpureocillium lilacinum for plant growth promotion and biocontrol against root-knot nematodes infecting eggplant. Plos one 2023, 18, e0283550. [Google Scholar] [CrossRef]
- Hossain, M. World pineapple production: An overview. African Journal of Food, Agriculture, Nutrition and Development 2016, 16, 11443–11456. [Google Scholar] [CrossRef]
- Kiriga, A.W.; Haukeland, S.; Kariuki, G.M.; Coyne, D.L.; Beek, N.V. Effect of Trichoderma spp. and Purpureocillium lilacinum on Meloidogyne javanica in commercial pineapple production in Kenya. Biological Control 2018, 119, 27–32. [Google Scholar] [CrossRef]
- Pagano, M.C.; Miransari, M. The importance of soybean production worldwide. In Abiotic and biotic stresses in soybean production; Elsevier: 2016; pp. 1-26.
- Khush, G. Productivity improvements in rice. Nutrition reviews 2003, 61, S114–S116. [Google Scholar] [CrossRef] [PubMed]
- Haque, Z.; Khan, M.R.; Ahamad, F. Relative antagonistic potential of some rhizosphere biocontrol agents for the management of rice root-knot nematode, Meloidogyne graminicola. Biological control 2018, 126, 109–116. [Google Scholar] [CrossRef]
- Hernandez-Trejo, A.; Estrada-Drouaillet, B.; Lopez-Santillan, J.A.; Rios-Velasco, C.; Rodríguez-Herrera, R.; Osorio-Hernández, E. Effects of native entomopathogenic fungal strains and neem extract on Spodoptera frugiperda on Maize. Southwestern entomologist 2019, 44, 117–124. [Google Scholar] [CrossRef]
- Castillo Lopez, D.; Zhu-Salzman, K.; Ek-Ramos, M.J.; Sword, G.A. The entomopathogenic fungal endophytes Purpureocillium lilacinum (formerly Paecilomyces lilacinus) and Beauveria bassiana negatively affect cotton aphid reproduction under both greenhouse and field conditions. PloS one 2014, 9, e103891. [Google Scholar] [CrossRef] [PubMed]
- Hotaka, D.; Amnuaykanjanasin, A.; Maketon, C.; Siritutsoontorn, S.; Maketon, M. Efficacy of Purpureocillium lilacinum CKPL-053 in controlling Thrips palmi (Thysanoptera: Thripidae) in orchid farms in Thailand. Applied Entomology and Zoology 2015, 50, 317–329. [Google Scholar] [CrossRef]
- Vitale, J. Economic importance of cotton in Burkina Faso. Food and Agriculture Organisation, Rome, Italy. Retrieved April 2018, 20, 2018. [Google Scholar]
- Rodrigues, J.; Bergamini, C.; Montalva, C.; Humber, R.A.; Luz, C. Simple method to detect and to isolate entomopathogenic fungi (Hypocreales) from mosquito larvae. Journal of Invertebrate Pathology 2021, 182, 107581. [Google Scholar] [CrossRef]
- Goffré, D.; Folgarait, P.J. Purpureocillium lilacinum, potential agent for biological control of the leaf-cutting ant Acromyrmex lundii. Journal of Invertebrate Pathology 2015, 130, 107–115. [Google Scholar] [CrossRef]
- Desoky, S.M.A.; Abdelall, M.F.; Ahmed, Y. Isolation, Identification, Evaluation of Purpureocillium Lilacinum Egyptian Isolate Toxicity Test in Vitro and Analysis Its Bioactive Products. Journal of the Advances in Agricultural Researches 2022, 27, 602–617. [Google Scholar] [CrossRef]
- Bali, G.K.; Singh, S.K.; Maurya, D.K.; Wani, F.J.; Pandit, R.S. Morphological and molecular identification of the entomopathogenic fungus Purpureocillium lilacinum and its virulence against Tuta absoluta (Meyrick)(Lepidoptera: Gelechiidae) larvae and pupae. Egyptian Journal of Biological Pest Control 2022, 32, 86. [Google Scholar] [CrossRef]
- Rodriguez, R.; White Jr, J.; Arnold, A.; Redman, a.R.a. Fungal endophytes: diversity and functional roles. New phytologist 2009, 182, 314–330. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Liu, Z.; Lin, R.; Li, E.; Mao, Z.; Ling, J.; Yang, Y.; Yin, W.-B.; Xie, B. Biosynthesis of antibiotic leucinostatins in bio-control fungus Purpureocillium lilacinum and their inhibition on Phytophthora revealed by genome mining. PLoS pathogens 2016, 12, e1005685. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wu, H.; Zhou, W.; Yuan, Z.; Hao, J.; Liu, X.; Han, L. Effects of indole derivatives from Purpureocillium lilacinum in controlling tobacco mosaic virus. Pesticide Biochemistry and Physiology 2022, 183, 105077. [Google Scholar] [CrossRef] [PubMed]
- Barra, P.; Nesci, A.; Etcheverry, M. In vitro compatibility of natural and food grade fungicide and insecticide substances with Purpureocillium lilacinum and their effect against Aspergillus flavus. Journal of stored products research 2013, 54, 67–73. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, J.; Liu, Y.; Feng, W. Biodegradation of hydrocarbons by Purpureocillium lilacinum and Penicillium chrysogenum from heavy oil sludge and their potential for bioremediation of contaminated soils. International Biodeterioration & Biodegradation 2023, 178, 105566. [Google Scholar]
- Gong, B.; Liu, G.; Liao, R.; Song, J.; Zhang, H. Endophytic fungus Purpureocillium sp. A5 protect mangrove plant Kandelia candel under copper stress. Brazilian journal of microbiology 2017, 48, 530–536. [Google Scholar] [CrossRef]
- Zhan, J.; Qin, Y.; Gao, K.; Fan, Z.; Wang, L.; Xing, R.; Liu, S.; Li, P. Efficacy of a chitin-based water-soluble derivative in inducing Purpureocillium lilacinum against nematode disease (Meloidogyne incognita). International Journal of Molecular Sciences 2021, 22, 6870. [Google Scholar] [CrossRef]
- Elsherbiny, E.; Taher, M.; Abd El-Aziz, M.; Mohamed, S. Action mechanisms and biocontrol of Purpureocillium lilacinum against green mould caused by Penicillium digitatum in orange fruit. Journal of Applied Microbiology 2021, 131, 1378–1390. [Google Scholar] [CrossRef]
| Kind of Effect | Results | References |
|---|---|---|
|
Nematode Control |
Control of root-knot nematodes in tomatoes | Kepenekci et al., 2018 |
| Control of Meloidogyne javanica in commercial pineapple |
Kiriga et al., 2018 | |
| The fungus showed maximum egg mass inhibition of Meloidogyne incognita | Sharma et al., 2021 | |
| Reduction of Meloidogyne incognita in tomatoes | Dahlin et al., 2019 | |
| The fungus was effectively applied as biocontrol agents of phytoparasitic nematodes in tomatoes under variable agroecological conditions. |
Girardi et al., 2022 | |
| Suppressed nematode Meloidogyne incognita population in a tomato–cucumber rotation in a greenhouse |
Giné and Sorribas, 2017 | |
| The fungus controlled root-knot nematodes infecting eggplant | Khan and Tanaka, 2023 | |
|
Secondary Metabolites |
Production of indole-3-carboxaldehyde (A1) andindole-3-carboxylic acid for tobacco mosaic virus control | Sun et al., 2022 |
| Production of metabolites for biological control | Chen and Hu, 2021 | |
| Proteases production | Chen and Hu, 2021 | |
|
Biological Control |
Controlled the insect Thrips palmi in orchid farms | Hotaka et al., 2015 |
| Potential control against cotton aphids | Castillo Lopez et al., 2014 | |
|
Plant-Growth |
In soybean stimulated root growth and nutrient absorption | Barbosa et al., 2022 |
| Increased the availability of P and N and promoted the growth of maize, beans and soybean | Baron et al., 2020 | |
| Improved soil nutrient availability in common bean growth | Alves et al., 2021 | |
| Promoted significant increases in plant dry biomass in cotton crop | Lopez and Sword, 2015 | |
|
Bioremediation |
Reduced the amount of total petroleum hydrocarbons in contaminated soils | Yang et al., 2023 |
| Removed 44.55% of crude oil from contaminated soil | Benguenab and Chibani, 2021 | |
| Protected mangrove plant Kandelia candel under copper stress | Gong et al., 2017 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





