1. Introduction
Manganese (Mn) is an important element widely employed in both ferrous and non-ferrous metallurgy, mainly in its metallic form, to produce various alloys. The compounds of Mn have also numerous applications, and one particularly significant use is the incorporation of manganese IV oxide (MnO
2) in battery manufacturing. The main processing route for manganese ores involves their utilization in the production of manganese ferroalloys through a carbothermic reduction process in a submerged arc furnace (SAF) [
1]. Previous studies have investigated the thermochemistry of the SAF process and the underlying reaction mechanisms involved in the production of Mn metal [
2]. In the SAF process, the furnace charge mainly consists of Mn oxides, which are largely transformed into the Mn ferroalloy product after reduction. In addition, a smaller portion of Mn ends up in the process slag as a byproduct [
3]. Furthermore, the furnace off-gas carries fine particles (dust), which are separated from the gas using dedusting techniques [
4,
5]. When wet scrubbers are employed to capture the dust, the wet particles are separated from the gas through water spray, resulting in the formation of a sludge known as Mn sludge [
5]. This Mn sludge contains significant amounts of valuable elements, including Mn, Zn, and Pb, making its valorization crucial for sustainable development and circular economy. A study by Shen et al. has characterized manganese furnace dust regarding the charge materials and indicated significant amounts of Mn and Zn in the dust [
6]. It is worth noting that the recycling of Mn sludge into the SAF process is difficult due to the presence of volatile components such as Zn and K. Currently, this sludge is considered as an industrial hazardous waste and is landfilled, emphasizing the importance of its valorization. Pyrometallurgical processing of SAF dust/sludge to recycle it into the SAF process has technical and economic limitation. However, there is growing interest in the application of hydrometallurgical processes, which offer potential profitability through the extraction of valuable metals [
7].
Extensive research has been conducted on the utilization of hydrometallurgical processes for extracting Mn from both primary and secondary raw materials. The production of electrolytic Mn metal (EMM) and electrolytic Mn dioxide (EMD) through hydrometallurgical processing of Mn ores are mature processes. As mentioned earlier, the Mn yield in the SAF process is relatively low, with a portion of Mn lost mostly in the slag byproduct and dust. Therefore, there has been considerable interest in recovering Mn from these streams. Various studies in the literature have investigated the recovery of Mn from Mn-containing slags using different acids, such as H
2SO
4 [
8,
9,
10,
11,
12,
13]. Notably, high Mn recovery rates of 88% and 90% have been reported. Although the generation of Mn sludge is smaller compared to the slag byproduct in the SAF process, it represents a more complex material. The Mn sludge contains a significant amount of Mn, along with various highly volatile substances found in the SAF charge, such as Zn, Pb, K, Cd, etc. Sancho et al. studied the extraction of electrolytic Mn metal from Mn sludge. Their process involved primary leaching using H
2SO
4, purification steps, and an electrolysis phase for electrolytic manganese production, resulting in the production of high-purity Mn metal [
14]. Similarly, Ghafarizadeh et al. focused on the reductive leaching of SAF dust from the ferromanganese process using sulfuric acid, with additional reducing agents such as oxalic acid, hydrogen peroxide, and glucose [
15]. They observed Mn leaching rate of up to 40% using 0.5 to 3 M H
2SO
4 solutions at 70°C within 90 minutes, with a liquid to solid ratio (L/S) of 10. Furthermore, they found that the presence of oxalic acid and hydrogen peroxide significantly increased Mn leaching recovery (almost complete recovery), while glucose had a lesser effect. Sadeghi et al. conducted the reductive acid leaching of a Mn residue using 1 M H
2SO
4. They employed glucose as the reducing agent, which resulted in high leaching rates of over 90% for Mn and Zn [
16]. Additionally, they observed that microwave-assisted leaching indicates a faster leaching rate compared to ultrasound-assisted and conventional leaching techniques.
In the present study, the leaching behavior of a Mn sludge using H2SO4 solutions is investigated through experimental work, aiming to evaluate the effect of process conditions. Various materials characterization techniques are employed to gain insights into the Mn sludge. Furthermore, thermodynamic calculations are performed to further understand the acid leaching process of Mn sludge.
2. Materials and Methods
2.1. Materials and Preparation
The manganese sludge used in this study was received from ferromanganese industry and contained a significant amount of water. To minimize errors in mass measurement, the sludge was dried in an oven at approximately 90°C for a period of two days. Sulfuric acid (H2SO4) solutions with concentrations of 0.7, 1.2, and 1.7 Molar (M) were prepared using the standard methods of acid dilution by adding distilled water.
2.2. Leaching Experiments
The leaching experiments were performed in a beaker using magnetic stirrer with the rotation speed of 350 rpm on a hot plate. Once the H2SO4 solution reached the target temperature, the Mn sludge powder was gradually added to the stirred solution. In all experiments, a precise amount of 10 ± 0.05 g of dried sludge powder was used. Different volumes of leaching solutions, namely 12.5, 25, 50, and 100 mL, were employed to achieve liquid to solid ratio (L/S) of 1.25, 2.5, 5, and 10 mL/g, respectively. To investigate the effect of temperature, six experiments were conducted at 30°C and 50°C, employing three different L/S ratios under fixed leaching conditions using a 1.7 M H2SO4 solution. The remaining experiments were carried out at 50°C. To study the impact of leaching duration, leaching tests were performed for durations ranging from 10 to 120 minutes using 1.2 and 1.7 M H2SO4 solutions. After the leaching process, the leaching residue was separated by filtration using a Büchner funnel. The residue was then washed with distilled water, dried in an oven, and its mass was measured. The pH of the solution was measured for selected experiments both before and after the leaching process.
2.3. Characterization of Materials and Products
The chemical composition of the dried sludge powder and leaching residues was measured using X-ray fluorescence (XRF) with a device from Thermo Fisher, Degerfors, Sweden. For XRF testing, sample preparation was conducted using the flux fusion method. Phase analysis of these materials was performed by X-ray diffraction (XRD) using the Bruker D8 A25 DaVinciTM, Karlsruhe, Germany, with CuK radiation (wavelength of 1.54 Å). The measurement range was set from 10 to 80° with a step size of 0.03. It is worth mentioning that XRD analysis was employed to study all the leaching residues. In addition, selected leaching solutions were analyzed using inductively coupled plasma-mass spectrometry (ICP-MS). Furthermore, the leaching residue samples were characterized by scanning electron microscope (SEM) (Zeiss Ultra FESEM, National Institute of Standards and Technology, Gaithersburg, MD, USA) equipped with an XFlash® 4010 Detector supplied by the Bruker Corporation (Billerica, MA, USA) for energy-dispersive X-ray spectroscopy (EDS).
3. Results
The obtained experimental results are presented and described as follows.
3.1. The Characteristics of Mn Sludge
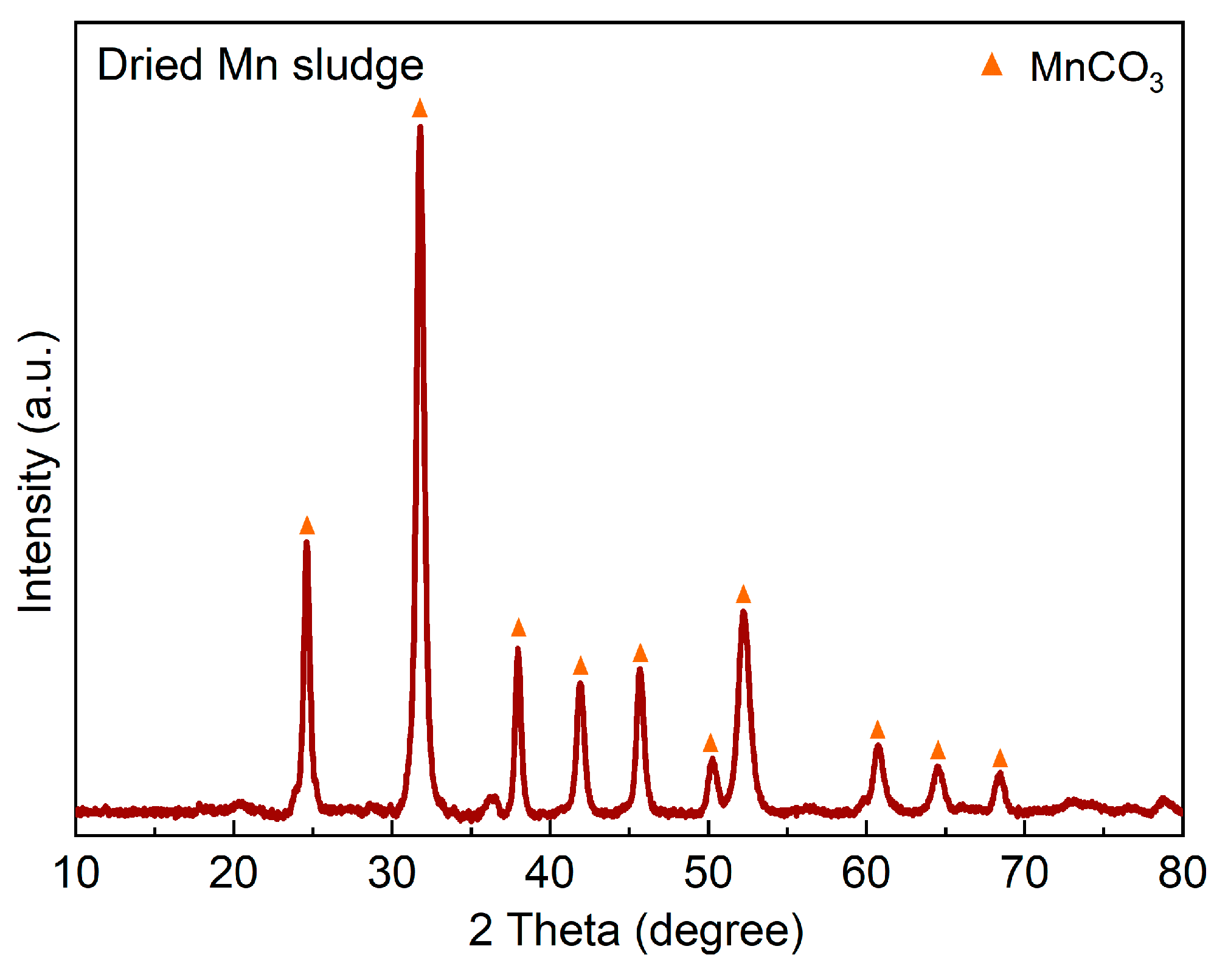
Table 1 displays the XRF analysis for the Mn sludge, showing a significant amount of Mn. Moreover,
Figure 1 illustrates the XRD spectrum for the Mn sludge, which indicates that the Mn exists in the form of a carbonate compound as rhodochrosite (MnCO
3). Previous studies have also reported the presence of MnCO
3 in this type of sludge [
5]. Therefore, in the XRF analysis presented in
Table 1, this compound was specifically considered, while the most stable oxides were considered for the other metals. It is important to note that due to the high intensity of the diffracted X-ray signal from MnCO
3, the identification of other minor phases was not possible.
3.2. Leachability of Mn Sludge
The impact of varying the leaching parameters on the dissolution of the main sludge components are described as follows.
3.2.1. Effect of Temperature
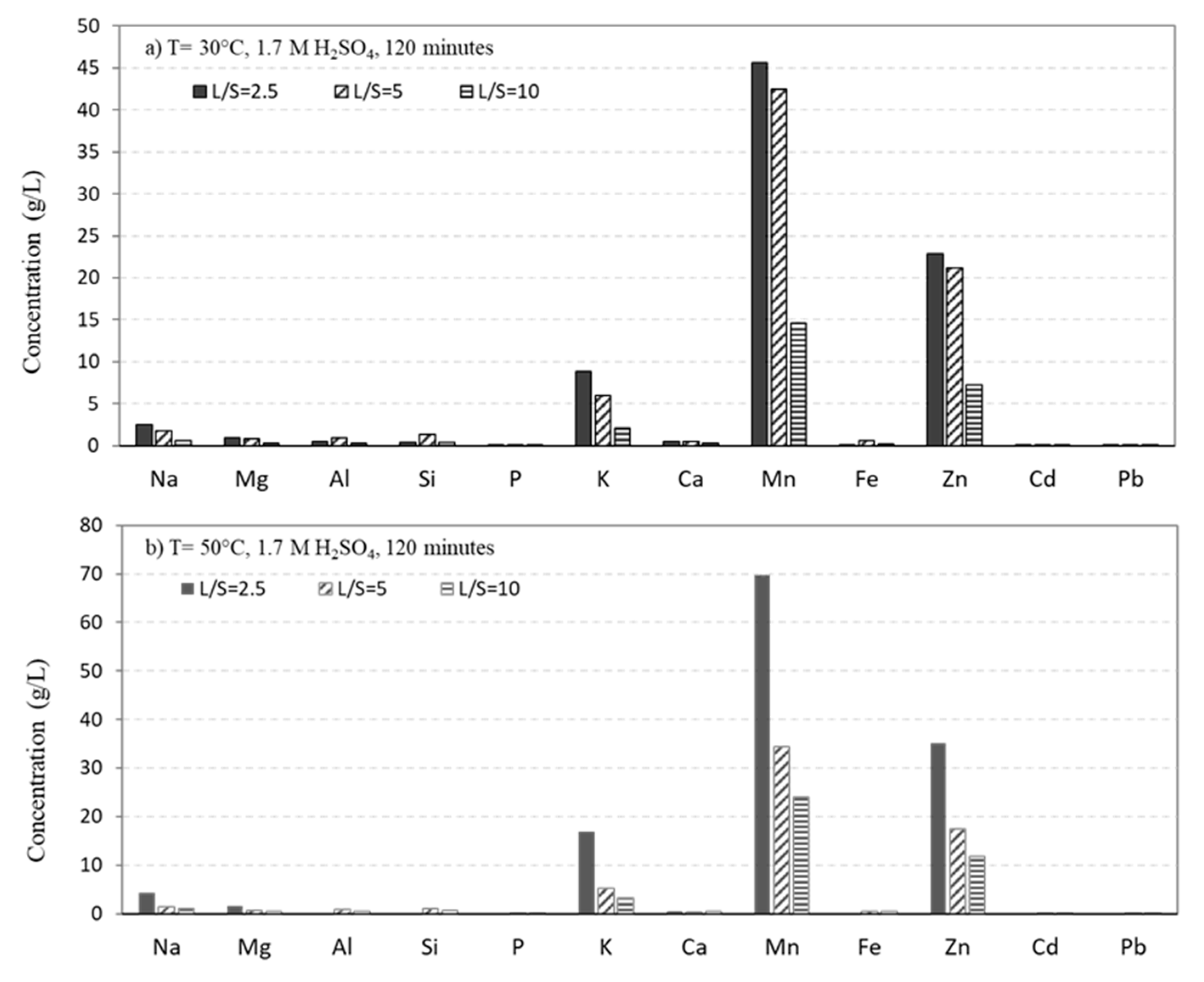
Figure 2 presents the measured chemical compositions of the leaching solutions, obtained using 1.7 M H
2SO
4 solution for three L/S ratios at 30°C and 50°C. The analysis reveals that Mn, Zn, and K are the main elements being dissolved into the solution, followed by Na, Mg, etc. Clearly, higher L/S ratios result in lower concentrations of Mn, Zn, and K in the solutions, indicating that for the lowest L/S ratio, a significant portion of these elements are leached. Moreover, increasing the leaching temperature from 30°C to 50°C generally leads to a higher leaching rate of the elements.
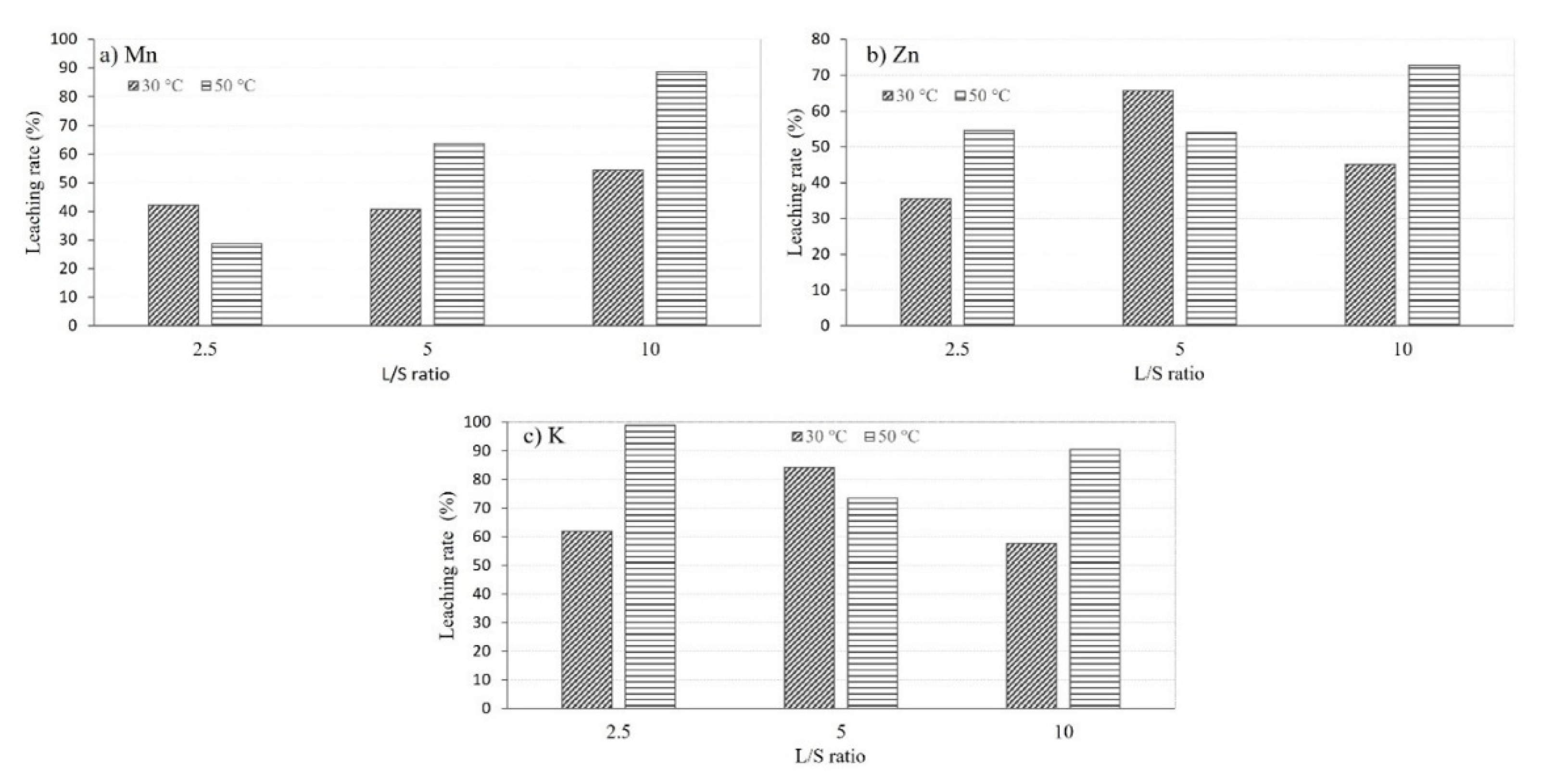
To gain a better understanding of the temperature effect, the recovery of Mn, Zn, and K was calculated for different L/S ratios, as illustrated in
Figure 3. The results demonstrate that as the temperature rises from 30°C to 50°C, the recovery of metals into the leachates increases, considering slightly lower accuracy observed for L/S=5. Therefore, the temperature was fixed at 50°C for all the other experiments.
3.2.2. Effect of Solution Volume and Concentration
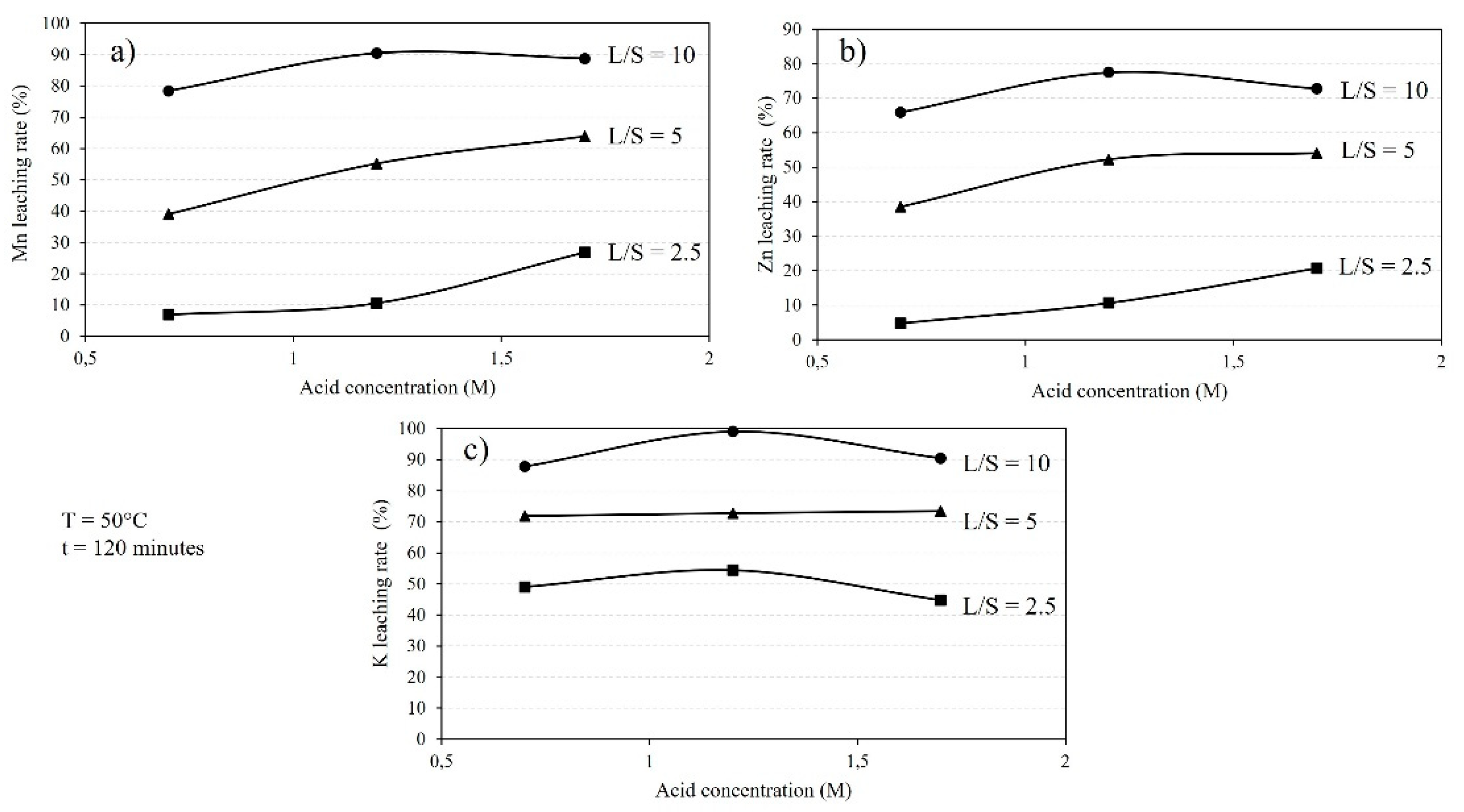
The leaching behavior of the sludge is influenced by the L/S ratio and acid concentration, as displayed in
Figure 4. It is worth to mention that the L/S ratio during the leaching process was not constant due to gas release via the involved chemical reactions and the formation of bubbles. Hence, the L/S ratio values that are presented are the numbers at initial. It is evident that increasing the L/S ratio, for a given acid concentration, leads to a higher leaching rate, with Mn and Zn recoveries reaching approximately 90% and 70%, respectively.
Figure 4a indicates that different acid concentrations result in the leaching of most of Mn. Higher acid concentrations lead to increased Mn leaching rates at fixed leaching durations for each L/S ratio. However, for higher L/S ratios, the Mn leaching rate becomes less dependent on the acid concentration. A similar trend is observed for the leaching of Zn from the sludge (
Figure 4b), where 66% to 77% of Zn is dissolved using 0.7 M to 1.7 M acid solutions.
Figure 4c shows that the dissolution of K during the leaching process is consistently higher than that of Mn and Zn, following the same trend with changes in the L/S ratio. Moreover, the differences in K leaching rates for different acid concentrations, at a given L/S ratio, are smaller compared to Mn and Zn leaching rates.
3.2.3. Effect of Leaching Duration
Figure 4 shows the effect of leaching duration on the dissolution of Mn, Zn, and K from the sludge under fixed temperature and L/S ratio. The experimental data consistently indicate a rapid initial stage of leaching, followed by a much slower stage.
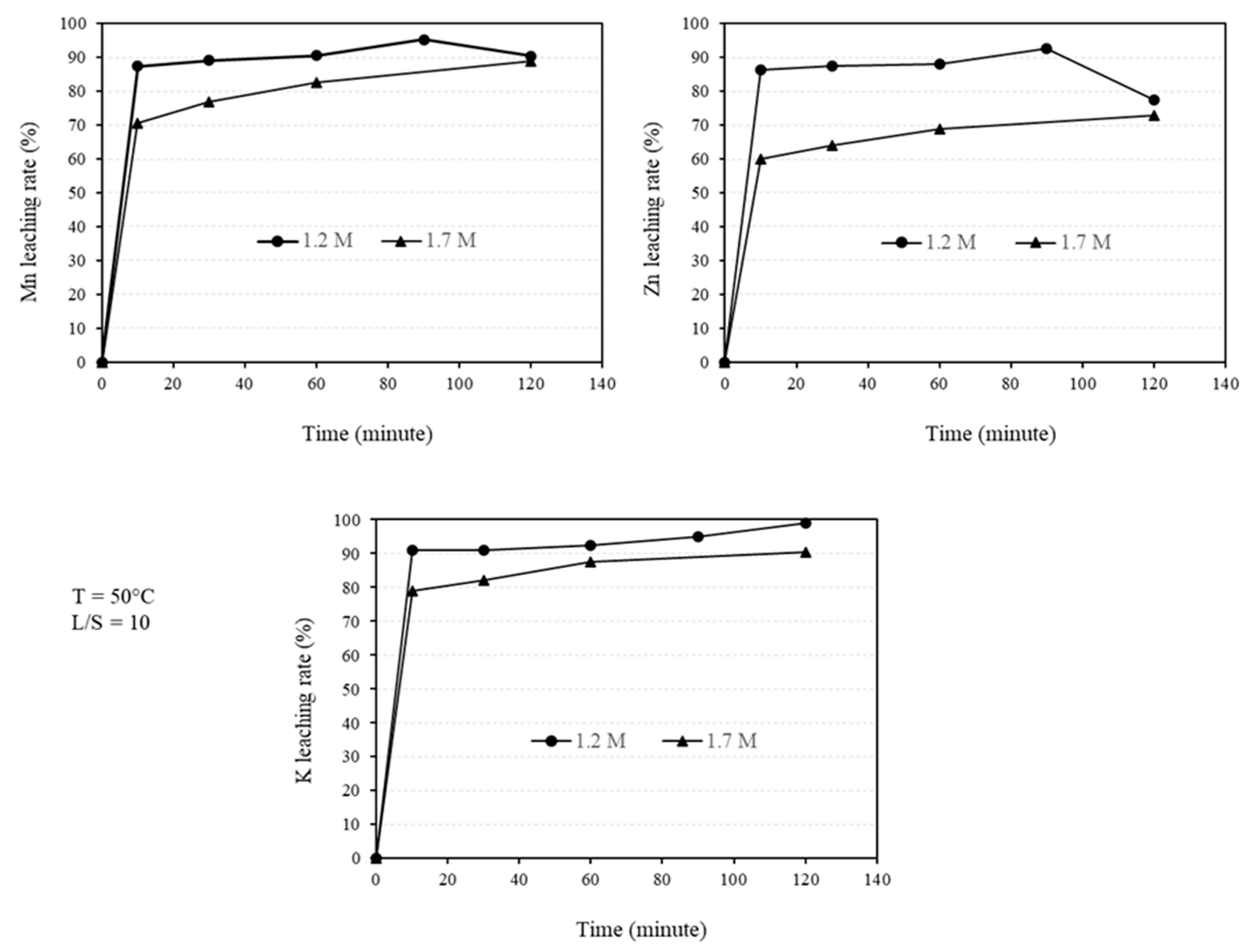
Figure 5.
The leaching rate of Mn, Zn and K using different acid concentrations, namely 1.2 M and 1.7 M H2SO4 solution at 50°C for L/S ratio of 10.
Figure 5.
The leaching rate of Mn, Zn and K using different acid concentrations, namely 1.2 M and 1.7 M H2SO4 solution at 50°C for L/S ratio of 10.
As illustrated in
Figure 4, a significant portion of Mn, Zn, and K is leached within the first 10 minutes. Furthermore, the initial leaching rate is high and not significantly dependent on the acid concentration. However, the leaching extent is higher for the 1.2 M solution compared to the 1.7 M solution. Nevertheless, this difference becomes insignificant after 120 minutes of leaching.
The pH measurements reveal an increase in solution pH during the leaching process, with greater pH changes observed when lower acid volumes are used. For example, when using a 1.7 M solution, the pH increases from approximately -0.23 to approximately 5 for L/S ratios of 1.25 and 2.5, while it reaches approximately 5 for L/S=5. In several experiments using a 1.7 M solution at L/S=10, the pH increases within a range of 0.16 to 0.44. The final pH for 0.7 and 1.2 M H2SO4 concentrations is also approximately 5. However, since the pH changes were not continuously measured, they cannot be used to study the leaching rate.
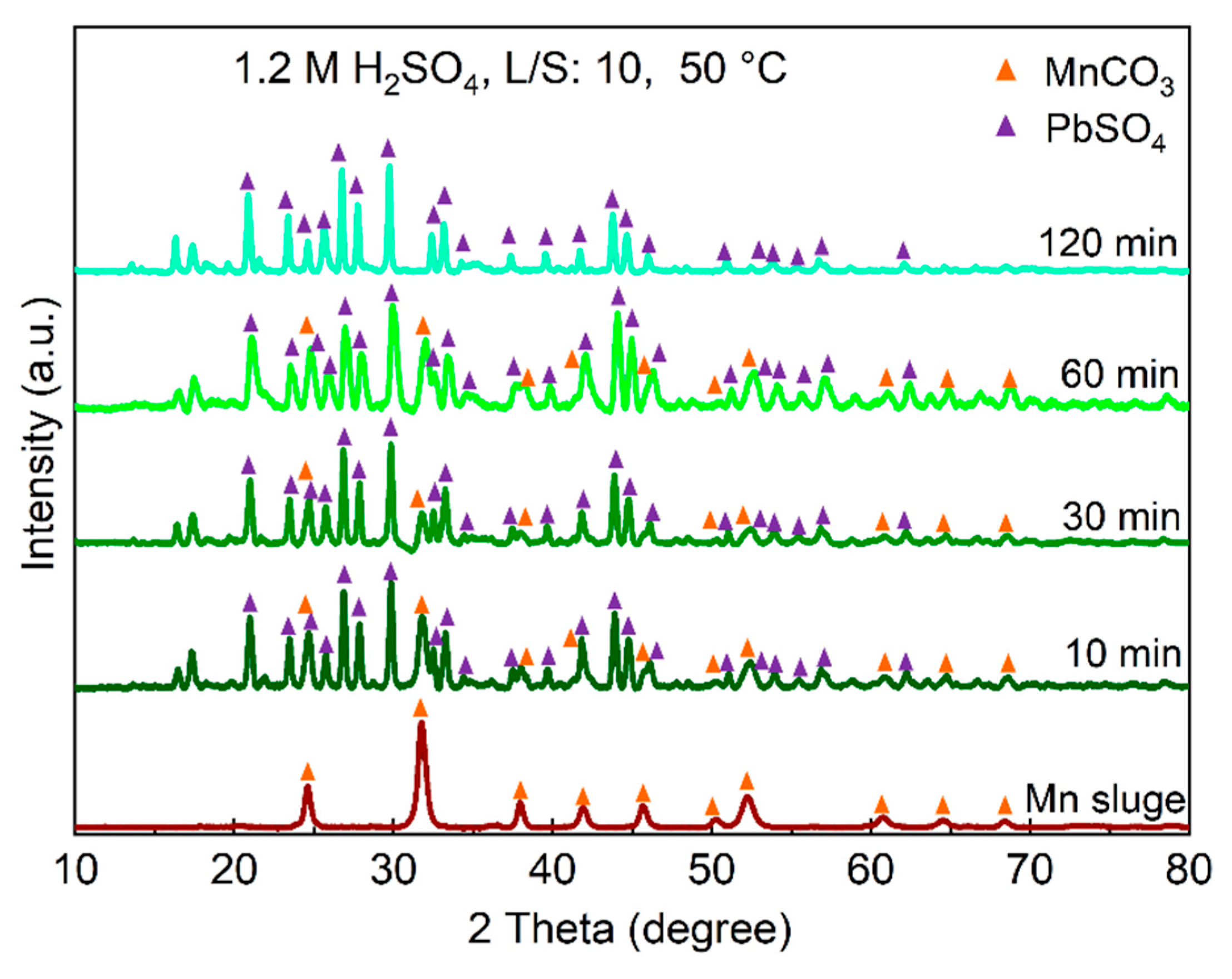
3.3. The Phase Analysis of Residue
Figure 4 displays the XRD spectra of the leaching residues obtained from experiments conducted at different leaching durations using a 1.2 M H
2SO
4 solution and L/S ratio of 10, in comparison to the Mn sludge. The results show that during the leaching process, MnCO
3 from the sludge is leached, while lead sulfate (PbSO
4) simultaneously forms. The XRD analysis data presented in
Figure 4 confirm that the presence of other crystalline residue components is not significant compared to these two phases.
Figure 6.
The XRD spectra of leaching residue samples at different leaching durations using 1.2 M H2SO4 solution and L/S of 10 at 50°C.
Figure 6.
The XRD spectra of leaching residue samples at different leaching durations using 1.2 M H2SO4 solution and L/S of 10 at 50°C.
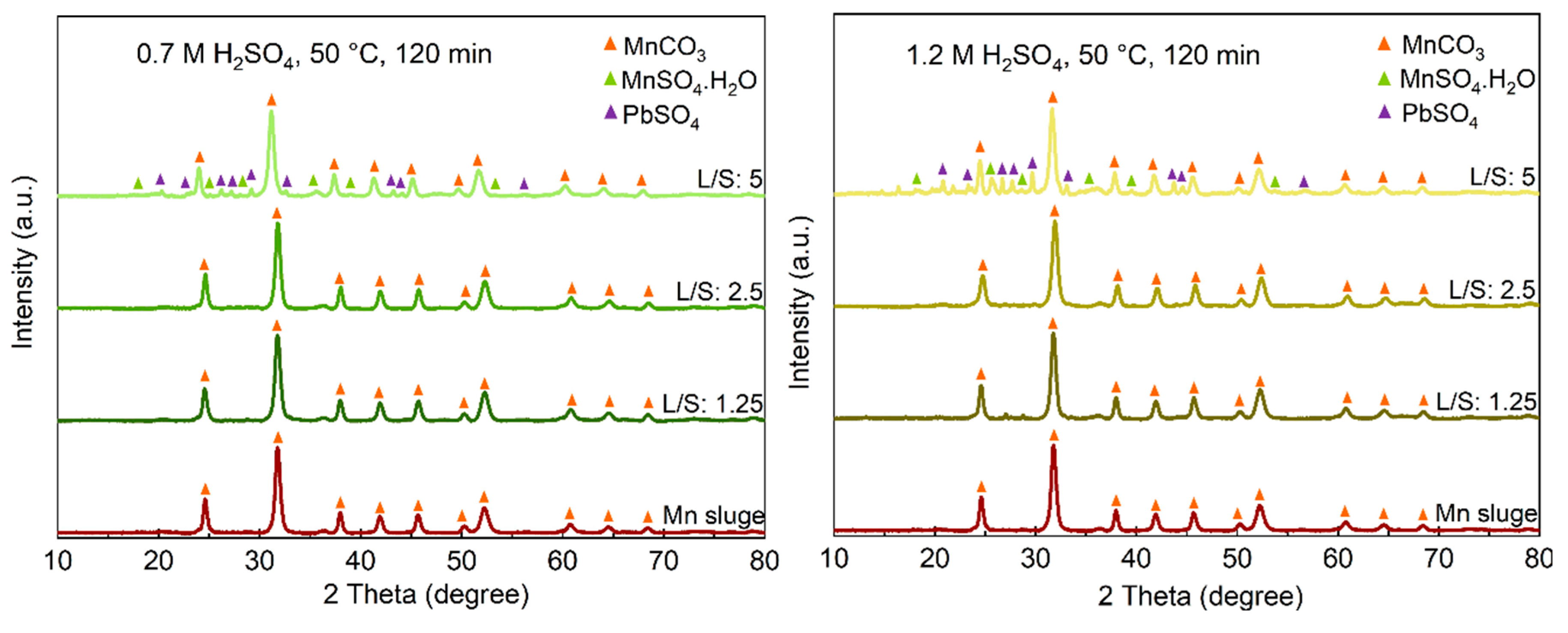
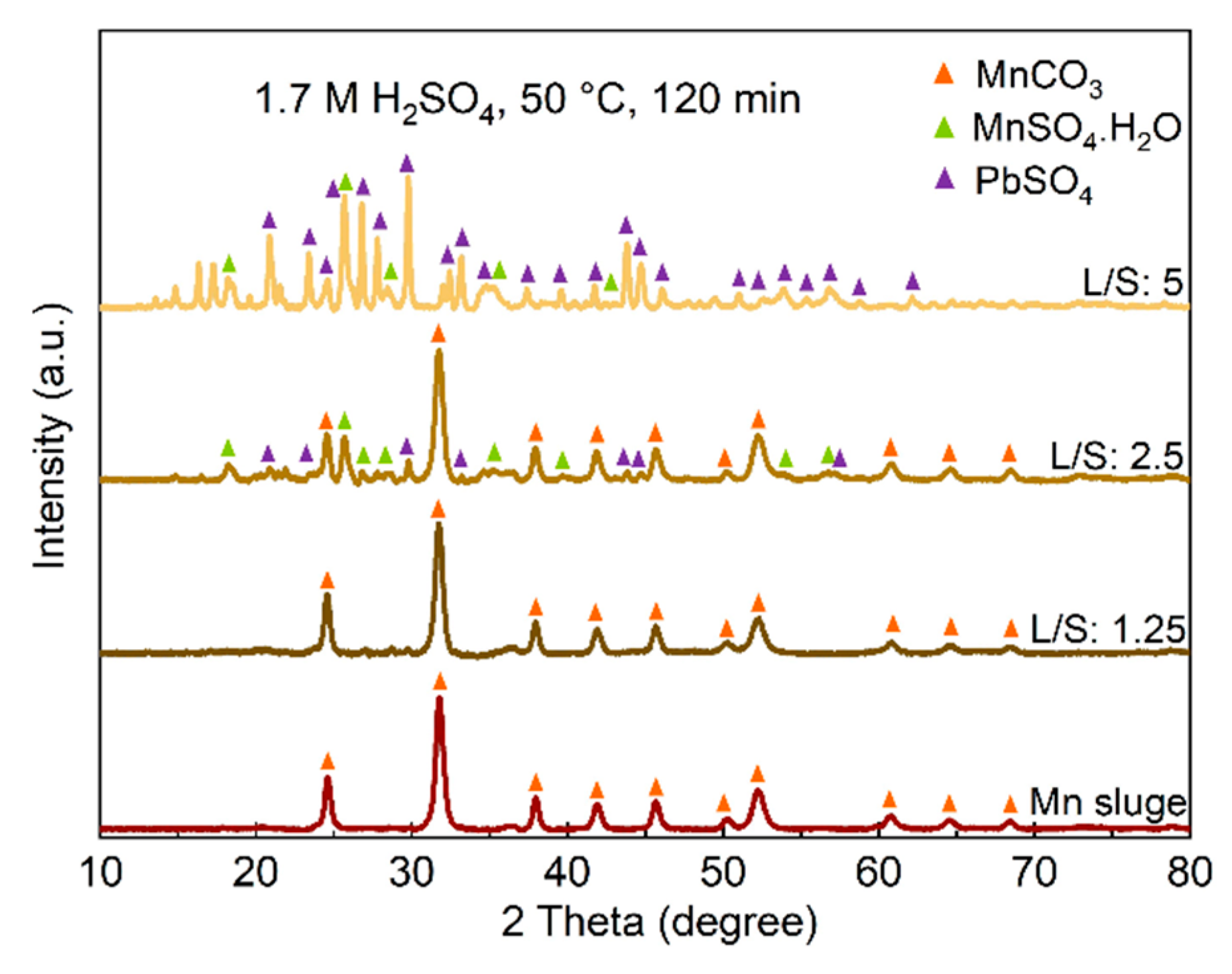
The identified phases in the leaching residues obtained using various acid concentrations and L/S ratios are shown in
Figure 4. When a 0.7 M acid solution is used, a significant amount of MnCO
3 remains unbleached, even at higher L/S ratios, which is consistent with the previously presented results regarding the recovery of Mn into the solutions. Comparing the XRD spectra in
Figure 4 indicates that the PbSO
4 phase appears in the sludge leached at L/S=2.5 when a 1.7 M acid solution is used, while for lower acid concentrations, PbSO
4 is observed at L/S=5. Moreover, for this L/S ratio, more intense PbSO
4 peaks and correspondingly weaker MnCO
3 peaks are observed when a stronger acid is used. The XRD spectra of the residue samples obtained from leaching conditions with L/S=10 and a 1.7 M acid solution were quite similar to the sample leached with a 1.2 M solution for L/S=10, presented in
Figure 4, and they exhibit PbSO
4 as the main phase.
Figure 7.
The XRD spectra of leaching residues obtained in leaching with different acid concentrations and L/S ratios.
Figure 7.
The XRD spectra of leaching residues obtained in leaching with different acid concentrations and L/S ratios.
3.4. The Microstructural Analysis of Residue
Figure 4 presents the elemental X-ray mapping of a representative leaching residue, demonstrating a high Mn recovery rate of approximately 89%. The figure illustrates that, during the leaching process, both small particles and relatively large agglomerates can be formed. The agglomerates exhibit a significant amount of Pb and S, which are closely associated, confirming the dominance of PbSO
4 phase as the main compound in the residue. Additionally, a distinct layer of PbSO
4 is observed on the surface of the agglomerates. The distribution of Mn, Fe, Al, Si, and Ca within the sample is not uniform, whereas Zn, K, and Na are observed to be distributed throughout the sample.
Figure 8.
The X-ray mapping of elements in an agglomerate of the leaching residue, and EDS point analysis for selected particles.
Figure 8.
The X-ray mapping of elements in an agglomerate of the leaching residue, and EDS point analysis for selected particles.
The distribution of Mn, Fe, and O as well as the analysis of point 1 in
Figure 4 indicates that Fe, Mn, and O are accumulated in some particles. Similarly, the distribution of Al, Si, and O, as observed in point 3, demonstrates a similar pattern for these elements. On the other hand, the point analysis 2 reveals the coexistence of all major elements, including Pb, Zn, Fe, Mn, S, and O, in certain areas of the sample.
4. Discussion
The obtained results are discussed in more detail.
4.1. Mn Sludge Composition
Obtaining an overview about the composition and formation of Mn sludge components is important to understand the leaching behavior of the sludge. The minor oxides components of sludge such as SiO
2, Al
2O
3, CaO, MgO (
Table 1) are originated from the SAF charge via the transportation of fine particles (dust) by the furnace gas. However, Mn, Zn and K components end up in the sludge via a different mechanism with chemical reactions involved. Mn in the sludge was found in the form of MnCO
3, as shown in
Figure 4, whereas Mn ores used in the ferroalloy industry are typically in the form of oxides (e.g., MnO
2, Mn
2O
3) or silicates (e.g., MnSiO
3). The presence of MnCO
3 in the submerged arc furnace off-gas can be attributed to the significant amounts of CO and CO
2 gases generated during the furnace operation. These gases are produced from the charged solid carbon reductant through the carbothermic reduction of MnO by C, the gaseous reduction of higher Mn oxides by CO gas, and the Boudouard reaction, as discussed previously [
2]. In addition, carbonate compounds present in the furnace charge, such as CaCO
3, MgCO
3, and K
2CO
3, are thermally decomposed at elevated temperatures, resulting in the generation of CO
2 gas within the furnace burden. Given the significant amount of CO in the SAF gas outlet, it is expected that Mn-containing compound leaving the furnace is mainly in the form of MnO at temperatures ranging from 200-400℃. The decrease in gas temperature causes an increase in the CO
2 content, leading to the conversion of MnO to MnCO
3. This change in gas composition results from the conversion of CO to CO
2. Consequently, MnCO
3 is formed through the following reaction:
By calculating the changes in Gibbs energy with temperature for various CO
2 partial pressures for chemical reaction (1), it was found that the formation of MnCO
3 occurs at temperatures below 340℃, 335℃, 325℃, 320℃, and 300℃ for CO
2 partial pressures of 1 atm, 0.8 atm, 0.6 atm, 0.4 atm, and 0.2 atm, respectively. Clearly, the reaction temperature is not significantly affected by the CO
2 partial pressure, and the reaction proceeds at temperatures below 300℃. Similar reactions may take place for some minor components present in the dust, such as K and Zn, leading to the formation of their carbonates. The presence of these phases was investigated previously [
5]. The studied sludge contained a high amount of Zn, which could be present in the forms of ZnO and ZnCO
3. However, these compounds were not detected in the XRD analysis, possibly because ZnO exists in an amorphous form, resulting from its formation from the gas phase.
4.2. The Kinetics of Leaching
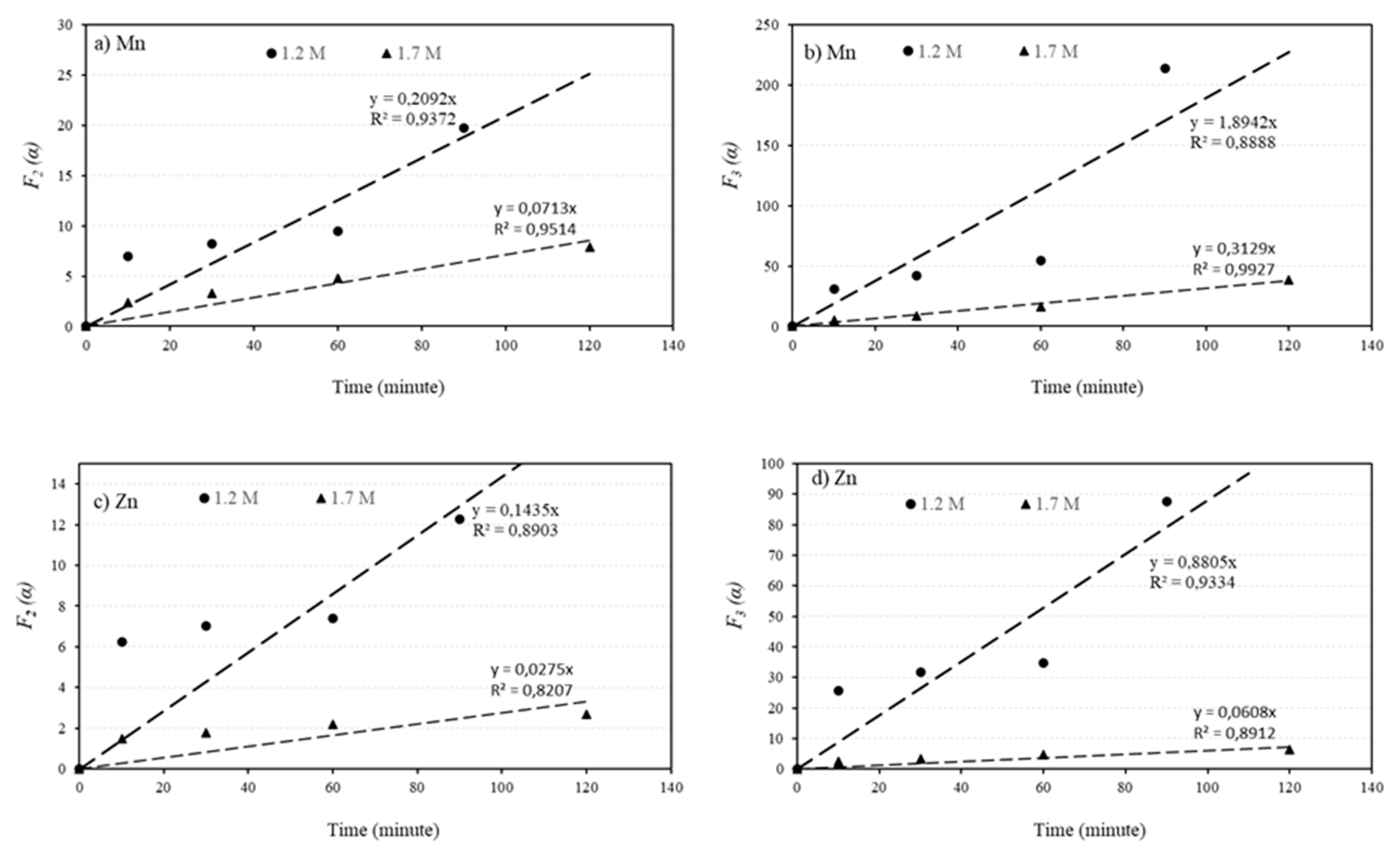
Mn and Zn are the most valuable metals in the sludge due to their abundance and leachability. The leaching process of these elements from the sludge takes place in two stages: an initial fast stage followed by a slower secondary stage, as shown in
Figure 5. To determine the reaction rate constant, the conversion fraction of MnCO
3 to Mn
2+ (α) and ZnO to Zn
2+ were calculated based on the leachability data for 1.2 and 1.7 M solutions at different durations. The α-values were tested for different chemical reaction orders, and it was found that the first-order reaction does not fit to the leaching process for Mn and Zn. However, the second and third-order reactions, described by Equations (2) and (2 respectively, provide a better fit to the data, as demonstrated in
Figure 4, with the exception of one outlier measurement for the 1.7 M solution. It is important to note that the vertical axis values in
Figure 4 represent the α functions given by the following equation:
where
k2 and
k3 are the reaction rate constants, and
t is the leaching time.
Figure 9.
The relationship between F2 (α) and F3 (α) with leaching time for two different acid concentrations of 1.2 and 1.7 M.
Figure 9.
The relationship between F2 (α) and F3 (α) with leaching time for two different acid concentrations of 1.2 and 1.7 M.
Based on the above equations, the slope of the fitted lines in
Figure 4 represent the reaction rate constants
k2 and
k3. These values were calculated and presented in
Table 2. The obtained rate constant values show that the leaching rate of Mn is 3 to 6 times higher for the 1.2 M solution compared to the 1.7 M solution. Similarly, the leaching rate of Zn is 4 to 15 times higher for the 1.2 M solution compared to the 1.7 M solution. This observation may explain the reason of the higher leaching rate of Mn in the 1.2 M solution compared to the 1.7 M solution for L/S ratio of 10, as shown in
Figure 4 and
Figure 5. Furthermore, comparing the rate constant values for Mn and Zn leaching indicates that the leaching of Mn is 2 to 5 times faster than the leaching of Zn. However, the difference in leaching rates between Mn and Zn is greater for the 1.7 M solution compared to the 1.2 M solution.
4.3. Thermochemistry of Reactions
The results showed a significant mass transport of Mn and Zn from the sludge into the solution during leaching. The leaching chemistry of these elements are discussed as follows.
4.3.1. Mn Leaching
It was observed that the majority of Mn in the sludge exists in the form of MnCO
3. During the leaching process, Mn is leached, resulting in an increase in pH and the consumption of acid. On the other hand, significant gas evolution occurs during leaching, suggesting the following reaction:
Clearly, the generation of CO
2 gas within the system is the main mechanism for bubble formation. The formation of bubbles combined with the applied magnetics stirring may explain the observed rapid leaching rates of Mn, as shown in
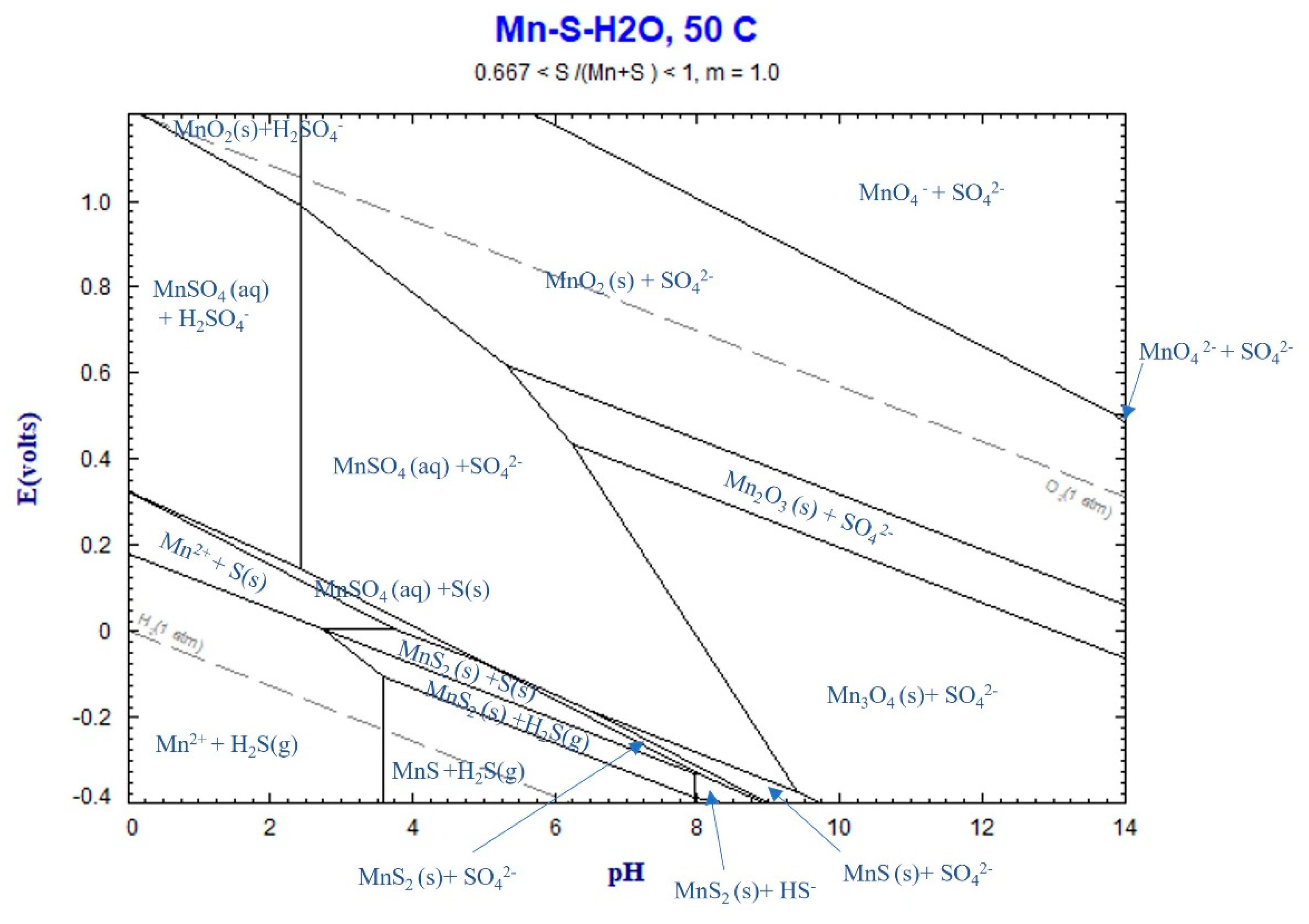
Figure 4. The Eh-pH diagram of the Mn-S-H
2O system, depicted in
Figure 4 using FactSage thermodynamic software, illustrates that Mn
2+ and MnSO
4(aq) are formed during leaching, considering the above reaction. The final pH of the solution was measured within the range of 0.16 to 5, indicating that the solution is within the Mn
2+ and MnSO
4(aq) region.
Figure 10.
Eh-pH diagram for Mn-S-H2O system at 50℃, calculated by FactSage thermodynamic software.
Figure 10.
Eh-pH diagram for Mn-S-H2O system at 50℃, calculated by FactSage thermodynamic software.
The obtained highest Mn leaching rate of about 90% (Figs. 4-5), the shape of the leaching curves in Fig. 5, and observing no MnCO3 phase in the samples leached by 1.2M and 1.7M solutions at L/S=10 indicate that the majority of MnCO3 has been leached. It is possible that the remained Mn has not had proper contact with the acid due to the formation of PbSO4 on sludge agglomerates (Fig. 8) that causes further slow Mn leaching rate, a kind of passivation. The other explanation is that a small portion of Mn (less than 10wt%) exist in other amorphous minor phases (not MnCO3) that were not observed in XRD spectra and such phases may not not leachable.
4.3.2. Zn Leaching
As mentioned earlier, Zn in the sludge exists in the form of ZnO and ZnCO
3. In the presence of H
2SO
4 solution, these compounds dissolve through the following reactions:
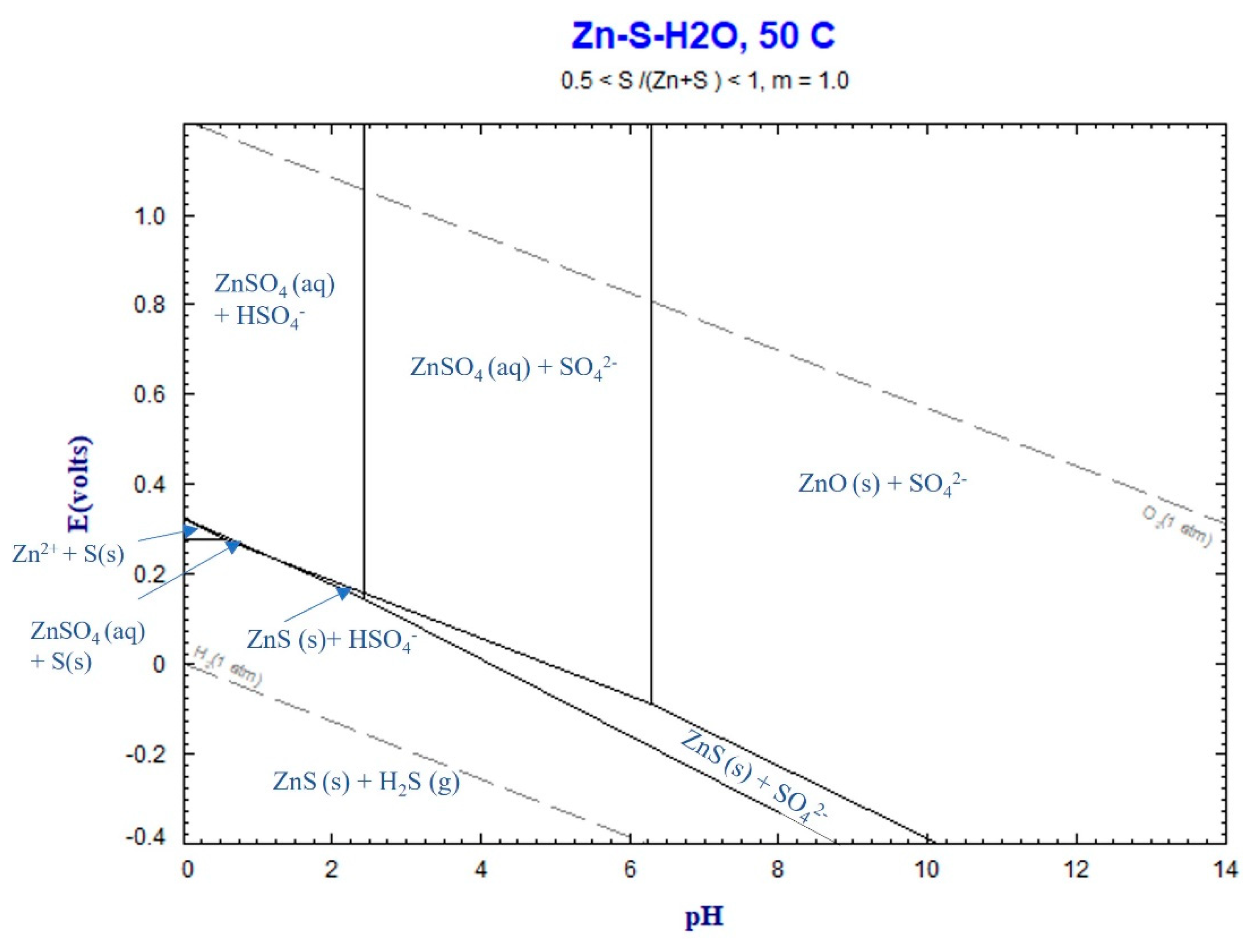
The Eh-pH diagram for the Zn-S-H
2O system in
Figure 4 illustrates that the aqueous ZnSO
4 remains a stable phase within the range of final pH values measured between 0.16 and 5. Consequently, the chemical reaction (2) takes place, leading to the dissolution of Zn in the sludge. Like chemical reaction (2), the formation of CO
2 gas through chemical reaction (2) may contribute to the formation of gas bubbles during leaching.
Figure 11.
Eh-pH diagram for Zn-S-H2O system at 50℃, calculated by FactSage thermodynamic software.
Figure 11.
Eh-pH diagram for Zn-S-H2O system at 50℃, calculated by FactSage thermodynamic software.
Chemical reactions (2) and (2) exhibit significant progress when using high concentrations of acids and higher L/S ratios. For example, when employing 1.2 M and 1.7 M acid solutions with an L/S ratio of 10, the acid content is sufficient to leach Mn and Zn significantly. However, when using a 1.2 M acid solution with L/S ratios of 1.25 and 2.5, it is theoretically possible to leach up to 30% and 60% of Mn, respectively. Nevertheless, due to the simultaneous leaching of Zn and K, the leaching of Mn is lower than the theoretical values. On the other hand, as the system approaches equilibrium, the leaching does not proceed more. This explains the existence of unbleached sludge at lower L/S ratios, as depicted in
Figure 4. Based on the obtained results, the best leaching conditions can be achieved by using acid solutions with concentrations of 1.2 M and higher, in combination with high L/S ratios.
4.3.3. Lead Sulfate Formation
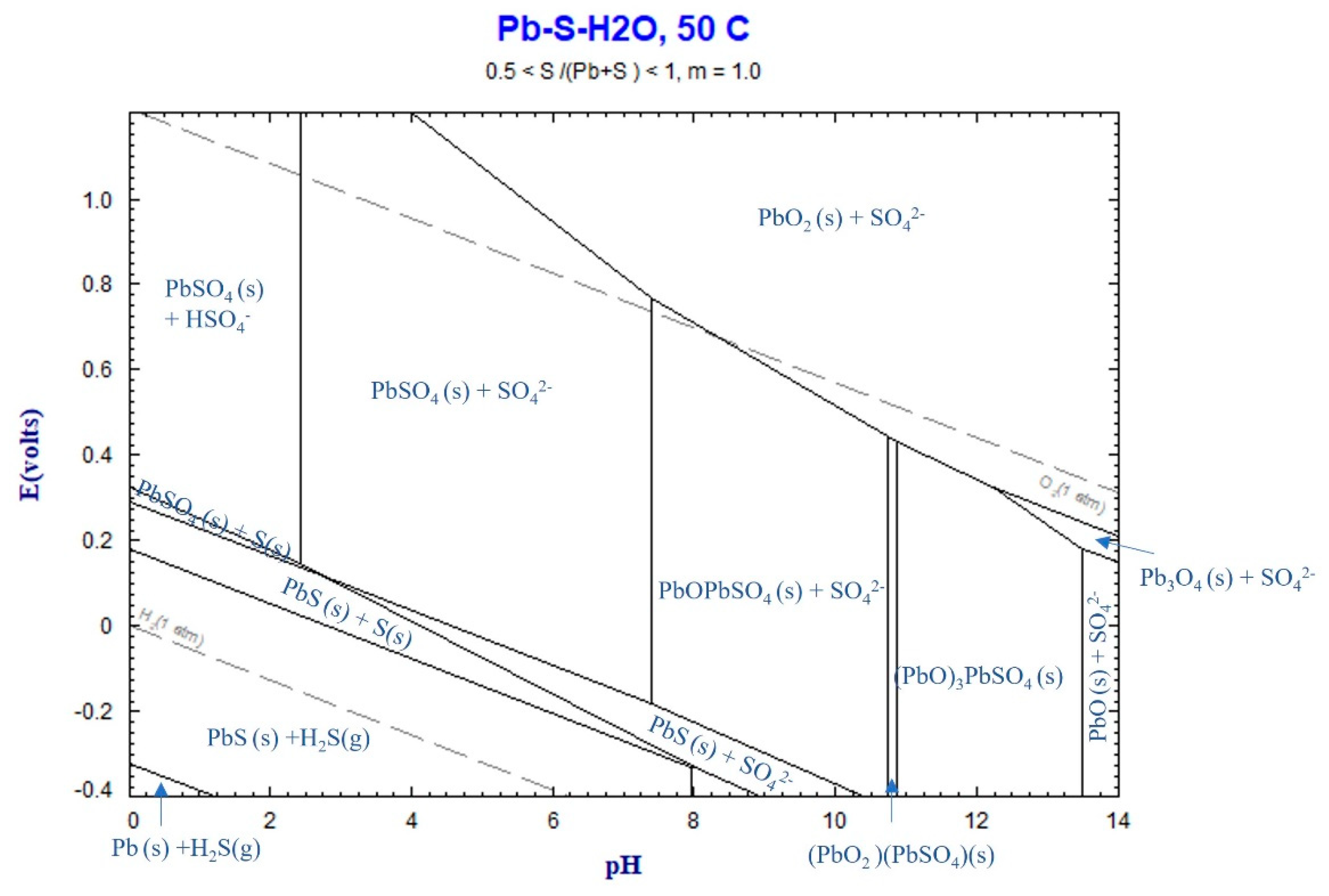
In all the leaching experiments, the formation of solid PbSO
4 and the insignificant dissolution of Pb into the solution were observed. This can be attributed to the stability of solid PbSO
4 in the system, as predicted by the Eh-pH diagram for the Pb-S-H
2O system in
Figure 4. The final pH values of all the experiments ranged from 0.16 to approximately 5, within which solid PbSO
4 remains a stable phase.
Figure 12.
Eh-pH diagram for Zn-S-H2O system at 50℃, calculated by FactSage thermodynamic software.
Figure 12.
Eh-pH diagram for Zn-S-H2O system at 50℃, calculated by FactSage thermodynamic software.
In the presence of sulfuric acid with the sludge, the formation of PbSO
4 can be explained by the following reaction:
The complete conversion of PbO to PbSO4 during leaching is evident from the intense peaks of PbSO4 observed in the XRD spectra of several residue samples. Clearly, the other components of unbleached sludge are found in significantly lower amounts in samples with high recoveries of Mn and Zn. The extensive progress of reaction (2) can be attributed to the particle size of PbO in the sludge, which is expected to be very fine. These particles likely condense from the gas phase as Pb gas exits SAF and subsequently oxidized to PbO (which has a high melting point of 888℃) before condensation.
5. Conclusions
The leaching behavior of a manganese sludge using sulfuric acid solutions was investigated, and the main conclusions are as follows:
The leaching rate of Mn sludge is fast, resulting in high leaching rates of 90% for both Mn and Zn.
The leaching rates of Mn, Zn, and K increase with increasing temperature, liquid to solid ratio, and duration. Mn exhibits a higher maximum leaching rate compared to Zn.
Comparing acid solutions, the leaching rate of Mn and Zn is slightly higher using a 1.2 M H2SO4 solution compared to a 1.7 M H2SO4 solution, while the leaching rate is much lower with a 0.7 M acid solution.
Kinetic studies of leaching with 1.2 M and 1.7 M H2SO4 acid solutions reveal that the reaction rate constant for Mn dissolution exceeds 1.2×10-3 s-1, while for Zn dissolution it is higher than 5×10-4 s-1.
During the leaching process, aqueous sulfate solutions of MnSO4 and ZnSO4 are formed from the dissolution of solid MnCO3 and ZnO (or ZnCO3) present in the sludge. This process is accompanied by the generation of CO2 gas and bubble formation.
The Pb content in the sludge is fully converted to solid PbSO4 during leaching, and samples with high Mn and Zn leaching rates yield a residue of lead sulfate.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, J.S. ; methodology, J.S., A. S.E., M. A.E.P., and S.H.; software, J.S., A. S.E., M. A.E.P., S.H.; validation, J.S., A. S.E., M. A.E.P., and S.H.; formal analysis, J.S., A. S.E., M. A.E.P., and S.H..; investigation, J.S., A. S.E., M. A.E.P., and S.H.; resources, J.S. .; data curation, J.S., A. S.E., M. A.E.P., and S.H.; writing—original draft preparation, J.S., A. S.E., M. A.E.P., and S.H.; writing—review and editing, J.S., S.H.; visualization, J.S., A. S.E., M. A.E.P., and S.H.; supervision, J.S. ; project administration, J.S.; funding acquisition, J.S. All authors have read and agreed to the published version of the manuscript.
Funding
Please add: This research was funded by SFI Metal Production (Centre for Research-based Innovation), grant number 237738. The APC was funded by SFI Metal Production.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data available in a publicly accessible repository that does not issue. DOIs. Publicly available datasets were analyzed in this study. This data can be found here:
https://www.ntnu.edu/metpro (accessed on 20 May 2023).
Acknowledgments
This publication has been funded by the SFI Metal Production (Centre for Research-based Innovation, 237738). The authors gratefully acknowledge the financial support from the Research Council of Norway and the partners of the SFI Metal Production.
Conflicts of Interest
The funders had no role in the design of the study; in the collection, analysis, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results. On behalf of all the authors, the corresponding author states that there is no conflict of interest.
References
- Olsen S. E., Tangstad M., and Lindstad T., Production of Manganese Ferroalloys. Trondheim: SINTEF and Tapir Academic Press, 2007.
- J. Safarian, “Duplex Process to Produce Ferromanganese and Direct Reduced Iron by Natural Gas,” ACS Sustainable Chem. Eng. 2021, 9, 5010–5026. [CrossRef]
- Safarian, J., Kinetics and mechanisms of reduction of MnOcontaining silicate slags by selected forms of carbonaceous materials. Trondheim, Norway: PhD Thesis, NTNU, 2007.
- S. Gaal, M. Tangstad, and B. Ravary, “Recycling of waste materials from the production of FeMn and SiMn,” The Twelfth International Ferroalloys Congress, Helsinki, Finland, pp. 81–88, Jun. 2010.
- B. Ravary and L. Hunsbedt, “Progress in recycling sludge from off-gas cleaning of manganese alloy furnaces,” 2013.
- R. Shen, G. Zhang, M. Dell’Amico, P. Brown, and O. Ostrovski, “Characterisation of Manganese Furnace Dust and Zinc Balance in Production of Manganese Alloys,” ISIJ Int. 2005, 45, 1248–1254. [CrossRef]
- K. Binnemans, P. T. Jones, Á. Manjón Fernández, and V. Masaguer Torres, “Hydrometallurgical Processes for the Recovery of Metals from Steel Industry By-Products: A Critical Review,” Journal of Sustainable Metallurgy 2020, 6, 505–540. [CrossRef]
- J. K. Mohanty, P. K. Sahoo, K. C. Nathsarma, D. Panda, and R. K. Paramguru, “Characterization and leaching of ferromanganese slag,” Mining, Metallurgy & Exploration 1998, 15, 30–33. [CrossRef]
- D. R. Groot, T. Redtmann, and J. Steenkamp, “The recovery of manganese and generation of a valuable residue from ferromanganese slags by a hydrometallurgical route,” 2013.
- Groot, D., Kazadi, D., Pollmann, H., de Villiers, J., Redtmann, T., and Steenkamp, J., “2013. Utilization of ferromanganese slags for manganese extraction and as a cement additive,” Proceedings of the Advances in Cement and Concrete Technology in Africa, Emperor’s Palace, Johannesburg, pp. 984–985, Jan. 2013.
- Kazadi, D., Groot, D., Pöllmann, H., de Villiers, J., and Redtmann, T., “Utilization of Ferromanganese slags for manganese extraction and as a cement additive,” Proceedings of the Advances in Cement and Concrete Technology in Africa conference, Emperor’s Palace, Johannesburg, pp. 983–995, Jan. 2013.
- S. Yan and Y. Qiu, “Preparation of electronic grade manganese sulfate from leaching solution of ferromanganese slag,” Transactions of Nonferrous Metals Society of China 2014, 24, 3716–3721. [CrossRef]
- D. M. Kazadi, D. R. Groot, J. D. Steenkamp, and H. Pöllmann, “Control of silica polymerisation during ferromanganese slag sulphuric acid digestion and water leaching,” Hydrometallurgy 2016, 166, 214–221. [CrossRef]
- J. Sancho et al., “Method of obtaining electrolytic manganese from ferroalloy production waste,” 2009.
- B. Ghafarizadeh and F. Rashchi, “Separation of Manganese and Iron from Reductive Leaching Liquor of Electric Arc Furnace Dust of Ferromanganese Production Units by Solvent Extraction,” vol. 45, 2011.
- S. M. Sadeghi, C. M. H. Ferreira, and H. M. V. M. Soares, “Evaluation of two-step processes for the selective recovery of Mn from a rich Mn residue,” Minerals Engineering 2019, 130, 148–155. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).