1. Introduction
Helicobacter pylori infection has a high prevalence, an overall prevalence of 44% worldwide. This rate ranged from 50.8% in developing countries compared with 34.7% in developed countries [
1].
H. pylori is a microaerophilic Gram-negative bacterium that can survive in highly acidic environments and is generally acquired in the first 5 years of life by oral-oral or fecal-oral transmission, linked to of low socioeconomic status, poor hygiene, and overcrowding [
2,
3].
H. pylori causes persistent inflammation in the gastric mucosa that increases the risk of clinical symptoms associated with peptic ulcers or chronic gastritis that may later progress to chronic forms leading to intestinal metaplasia, dysplasia and ultimately, gastric adenocarcinoma [
2,
4,
5,
6].
H. pylori infection is consistently recognised as the most important risk factor for sporadic gastric cancer [
7]. Since 1994,
H. pylori has been identified as a class-1 carcinogen by the International Agency for Research on Cancer [IARC] [
8].
H. pylori eradication may rapidly decrease active inflammation in the gastric mucosa, prevent progression toward precancerous lesions and reverse gastric atrophy before the development of intestinal metaplasia. Undoubtedly, the earliest possible eradication of
H. pylori is highly beneficial [
9].
H. pylori gastritis as an infectious disease is now included as a nosological entity in itself in the new International Classification of Disease 11th Revision (ICD 11), which implies treatment of all
H. pylori-infected patients. Eradication of
H. pylori is recommended even in the absence of symptoms. Empirical
H. pylori eradication including triple standard therapy. It consists of a proton pump inhibitor [PPI] in standard doses accompanied by two antibiotics such as clarithromycin plus amoxicillin for 14 days [
10]. Resistance of
H. pylori to antibiotics has reached alarming levels worldwide, which has been figured out as one of the main causes of therapeutic failure [
11,
12]. Amoxicillin, metronidazole, clarithromycin, tetracycline and levofloxacin are the most frequent antibiotics used in different combinations in eradication regimens [
13].
The current study aims to first, isolate H. pylori in cultures from gastric biopsy samples from patients coming of the western central region of Colombia and test their susceptibility to amoxicillin, clarithromycin, metronidazole, tetracycline, rifampicin and levofloxacin. Secondly, we aim to assess the efficacy of the standard triple therapy in the population from two specialized Health Centers located in the cities Pereira and Manizales. In this part of the country, like different regions, positive infected patients are treated in an empirical way, without previous analysis of antimicrobial drug resistance, which could increase the risk of apparition of multidrug resistant strains.
2. Results
2.1. Patient Population
The sociodemographic variables of this population were told in our previous publication (2). Between February and October 2018, 740 gastroenterology consultations were registered in two centers in Pereira and Manizales, of which 94 patients met the selection criteria (646 individuals did not meet the selection criteria: did not sign the informed consent N=20 (3.09%) or did not meet the inclusion criteria N=580 (89.7%)). The females were 76.5% and the mean age was 46 years (SD 10.43). Most patients were housewives (48.9%) and 59.5% of them were from Pereira.
Regarding the diagnostic tests, 67 (71.2%) patients had at least one positive test (histology or culture), 77.7% being women and 22.2% men. Of the positive, 56 (59.5%) patients were culture positive and 37 (39.3%) were histology positive. Finally, 37 (39.3%) patients were positive for both tests while 27 (28.7%) patients were negative. Antibiotic susceptibility test report was obtained in 42 (64.1%) of the patients. The missing data were due to no growth or contamination. Histologically, 24 (25.5%) patients had malignancy precursors, being 9 (37.5%) H. pylori positive while in the other 70 (74.46%) patients without malignancy precursors, 43 (61.4%) were positive. In the endoscopy fifty (53.19%) patients with chronic antral gastritis were H. pylori positive, while for chronic antral atrophic gastritis, chronic corporal atrophic gastritis and chronic corporal gastritis, the positivity was 10 (10.6%), 9 (9.57%) and 8 (8.51%) patients, respectively.
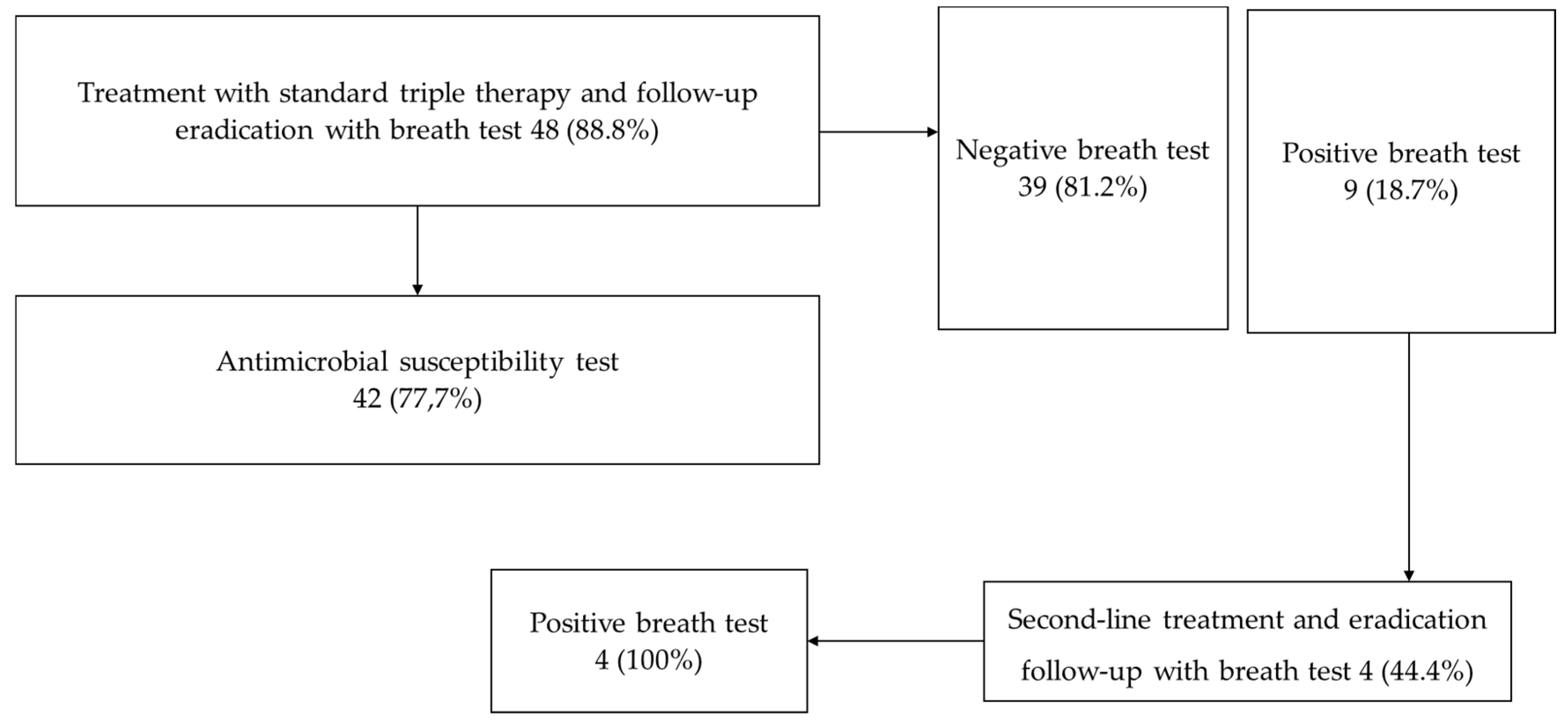
Of all patients, 54 received triple therapy, of them 48 patients were treated and followed up with 13C UBT, constituting the group for analysis of H. pylori eradication. Drug compliance was excellent. The most common side effects included bitter taste, diarrhea, dizziness, and malaise, but no serious signs and symptoms were documented.
2.2. The E-Test and the Eradication
Of the 54 patients with a positive report for
H. pylori who received antibiotic management, 48 (88.8%) underwent follow-up with a breath test after finishing the pharmacological management (
Figure 1), where only 9 patients (18.7%) had a positive result, finding an eradication percentage with standard triple therapy of 81.2%. 8 of the 9 positive patients accepted the sign for pharmacological medical management with second-line therapy; however, then, four breath tests were collected as a follow-up, which persisted positive over time. The remaining 4 patients refused to take them.
2.3. Antimicrobial Susceptibility
Of the 67 patients positive for
H. pylori, antibiotic susceptibility profiles were figured out for 42 patients. All isolates were susceptible to tetracycline. Metronidazole, levofloxacin, rifampicin, clarithromycin and amoxicillin and dual, triple, quadruple, resistance was found (
Table 1). The missing data of the 25 strains was due to non-growing or contaminated samples.
Table 2 gives an overview of the resistance rates, the number of resistant isolates and distribution of the MICs (mg/L) found in the 42 isolates. Briefly, according to the minimal inhibitory concentrations, 66% (23/35) and 38% (3/8) of the isolates showed MIC values over 256 mg/L for metronidazole and clarithromycin, respectively.
When comparing the results obtained in the studies conducted previously in the Western Central Region of Colombia [
16,
17] and the present report, a stable rate of resistance to metronidazole could be seen; in the case of clarithromycin there was an increase of more than double, finding a statistically significant difference, between the years 2009 and the present study (p = 0.004). In the 2019 study, no resistance to amoxicillin was found, however, in the 2009 study and in this work the associated percentage was 1.9% and 9.5%, the difference found between 2019 and the recent year being significant (p = 0.02). In this report, no resistance to tetracycline was found, which is consistent with what was told in previous studies. There are no earlier data in the region on the presence of resistance related to levofloxacin and rifampicin, so it was not possible to make a comparison. In the rest of the comparisons, no significant statistical difference was found. Resistance to two or more antibiotics had a significant increase for the antibiotics evaluated in the previous studies (
Table 3).
2.4. Efficacy of Triple Therapy and Effects of Drug Resistance
Of the 67 H. pylori-positive patients, 54 received triple standard therapy in their entirety. Of these, 48 patients underwent follow-up breath testing and 30 underwent susceptibility testing. Missing data refers to samples with lack of growth or contamination.
Triple standard therapy failed in 5 of the 30 patients with complete breath testing and antimicrobial susceptibility: 2 with strains resistant to metronidazole/clarithromycin, one was resistant to clarithromycin/levofloxacin, and 2 had combined resistance to 3 or more antibiotics. Of the group, 4 patients accepted the administration of the second line therapy and only one attended the follow-up with UBT which remained positive over time for the metronidazole/clarithromycin/amoxicillin resistant strain. Eradication rates are shown in
Table 4.
No significant differences were found in the mean eradication rates between patients with metronidazole resistant and susceptible strains (84 and 80% respectively), however, eradication was significantly higher in patients with clarithromycin susceptible than in resistant strains (95.6% vs. 42.8% p= 0.001). Finally, of the 5 strains that did not respond to triple standard therapy, 4 had combined resistances to clarithromycin and metronidazole with MICs of 1.5, 64, 256, 64 mg/l and 256, 12, 16, 24 mg/l, respectively.
3. Discussion
In this study, triple standard therapy for 14 days was effective for the eradication of
H. pylori in 81.2% of the patients with a positive histology or culture, which is like the results in other studies in Colombia that reported the eradication success in about 80% of the cases [
18]. The goal of any antimicrobial therapy is to cure reliably
H. pylori infection in the majority (eg, ≥90%) of patients [
10]. It is important to highlight that, of the positives, 59.5% of the patients were detected by culture and 39.3% of them by histological study, finding that the culture detection rate was the same to or lower than that reported by others (50-95%) [
19].
Multiple studies worldwide have confirmed the increase in therapeutic failures in patients with strains resistant to clarithromycin, finding eradication rates of only 18% compared to 88% in sensitive strains [
20]. According to Sierra et.al. (2013) [
21], when the first line fails, this antibiotic should not be included in the second intervention since secondary resistance would drastically increase. For this reason, is recommends separating the regions, considering the prevalence of resistance to this antimicrobial and using ITT as the first line only in those with a low prevalence of resistance (<15%) [10, 22]. The increasing
H. pylori resistance to previously effective antibiotic treatments has become of great concern and requires careful choice of therapies and revision of therapeutic strategies. Maastricht VI a new focus is set on molecular testing for
H. pylori detection and antibiotic susceptibility with support for the role of antibiotic stewardship. The most effective empirical regimens are revised if individual antibiotic resistance is not available [
10]. In this study, the clarithromycin resistance rate was 19%, being 10.8% higher than that reported in the 2019 study [
16], without statistically significant variation. However, it is the first report in the Western Central Region of Colombia of resistance to clarithromycin greater than 15%, after more than 10 years of resistance surveillance. It is important to avoid the use of clarithromycin based triple regimens in countries/regions with high (>15%) clarithromycin resistance in
H. pylori without susceptibility testing [
10,
22]. This is like the results reported in Tumaco, where the value is 19.8% [
23] or Bogotá, whose rates range between 13.6 and 60% [
24,
25,
26] however, it is important to mention that although clarithromycin is the antibiotic associated with the most resistance studies in Colombia, the differences between the reports have been variable [16,17, 23-26]. In general, in Latin America, the highest rates of resistance to clarithromycin were seen in Mexico, Colombia and Brazil [
27,
28]. European countries report primary resistance rates between 14 and 40% and in the United States it is higher than 16% [
27,
29,
30,
31].
According to previously published studies on
H. pylori resistance in Colombia, the antibiotic with the highest percentage of resistance is metronidazole, which reached 72 to 93% in Bogotá and around 88% in the Western Central Region of Colombia [
17,
24,
25,
29] however this has been considered minor clinically important because it does not significantly influence eradication rates [
32,
33]. Resistance rate to metronidazole (81%) remained similar to the results previously published by the group [
16,
17]. This is like the rates reported in developing countries due to the frequent use of the drug for the treatment of common infections, mainly dental, gastrointestinal or genital origin. The prevalence of resistance to metronidazole in developed countries was 20% in Europe, 40% in the USA, and 12% in Japan [
27].
Amoxicillin resistance in Colombia has been evaluated in three studies showing rates in the city of Bogotá in the years 2008 and 2010 of 7% [
25] and 3.8% [
31] respectively, while in the case of Tumaco the percentage in 2012 was 20.5% [
23]. In this study, the existence of resistant strains in the Western Central Region of Colombia, resistance to amoxicillin was 9.5%, which is like the values reported in the city of Bogotá, the difference found in the evaluation of the results obtained from the previous study being statistically significant (p = 0.02). However, Martínez et. al. (2014) [
27] told that no resistant strains have been found in 9 studies in Latin America, making it difficult to figure out a possible increase in resistance to amoxicillin in the region.
Levofloxacin resistance in this study was 26.2%, which is like the values reported in Colombia in the studies in the city of Bogotá (11.8% and 27.3%) in 2009 and 2014 [
29]. On the other hand, no isolates with tetracycline resistant, similarly to previous results (2009 and 2019) [
16,
17] in the Western Central Region of Colombia, as well as of other authors in other parts of the world; however, there are exceptional cases where resistances of up to 85.7% have been reported [
20,
34,
35]. Additionally of the second line regimen was limited, so it is not possible to extrapolate relevant data to the research. It should be noted that 15 of the 30 patients studied with UBT follow-up and antimicrobial susceptibility presented combined resistance to two or more antibiotics, the main combination being metronidazole and clarithromycin (13.3%). In Colombia, only two studies have reported multidrug-resistant strains that, and in the study by Arévalo et.al. (2019) [
33] were found in patients with three or more failed treatments. While many factors are associated with the failure of
H. pylori eradication, the main contributors are patient noncompliance and increased antimicrobial resistance, especially to quinolones and macrolides [
35]. In this study we observed that in patients with clarithromycin resistance, the eradication rate decreased to less than 50% (
Table 4), corroborating what Roberts et al (2022) reported [
35]. Therefore, optimizing the first-line regimen based on local antibiotic resistance patterns is critical to prevent repeated courses of treatment and the spread of secondary antibiotic resistance [
36]. The increasing
H. pylori resistance to previously effective antibiotic treatments has become of great concern and requires careful choice of therapies and revision of therapeutic strategies [
10]. Growing antibiotic resistance and previous unsuccessful treatment attempts impede eradication success and ease emergence and spread of multidrug (MDR “resistance to ≥3 antibiotics of different class”) resistant strains [
37] and this is the first time that the region reports isolates with MDR.
4. Materials and Methods
4.1. Patient Population
Adult patients with gastric disease who underwent gastroduodenoscopy and biopsy sampling in two specialized centers in the Western Central Region of Colombia (Pereira and Manizales cities) between February and October 2018, were included (those patients who gave their informed consent to participate in the study were enrolled). Histological and culture studies for the diagnosis of H. pylori infection were performed, excluding patients with comorbidities, immunosuppression due to risk of infection, previous gastric surgery, and those who had used proton pump inhibitors, anti-H2 inhibitors, or antibiotics four weeks prior to the study. The research was approved by the Bioethics Committee of the Universidad Tecnológica de Pereira and the patients signed informed written consents prior to the start of the project.
4.2. H. pylori Culture
H. pylori was cultured from gastric biopsy samples of the antrum, body, and fundus. Samples were stored in BHI broth with 20% glycerol, supplemented with antibiotics (vancomycin 10 mg/l, polymyxin B 0.33 mg/l, bacitracin 1.07 mg/l, and amphotericin B 5 mg/l). The biopsy specimens were mixed with sterile saline solution and macerated with a homogenizer (Deltaware Pellet Pestle). We kept the maceration tissues at -80°C in BHI broth with glycerol. Then 100 µl of each mash solution were plated onto culture media (Tryptic soy agar (TSA) (Oxoid or Merck), supplemented with sheep blood (7%), isovitalex (0.5%), and the same antibiotics/concentrations used for the BHI broth media during transportation and were incubated under microaerophilic conditions (5% O2, 10% CO2 and 85% N2) at 37°C for 5 to 7 days. Colonies were confirmed with Gram staining and biochemical tests (positive urease, catalase and oxidase test).
4.3. E-Test and Definition of Susceptibility Testing
H. pylori isolates obtained by the primary culture were subcultured on non-selective Tryptic soy agar (TSA) (Oxoid or Merck) with 7 % sheep blood, 0.5 % isovitalex (BBL). The isolates were subcultured to figure out the profile of antimicrobial resistance by E-test (AB BIODISK North American Inc., Piscataway, NJ, USA) for metronidazole, clarithromycin, amoxicillin, levofloxacin, rifampicin, and tetracycline. Suspensions from pure 48-h subcultures were prepared in Brucella broth supplemented with 0.5 % Isovitalex and inoculum turbidity was adjusted to McFarland 3.0 standard. Thereafter they were inoculated onto TSA plates supplemented with sheep blood (7%), isovitalex (0.5%) and without antibiotics. E-test strips were placed and incubated under microaerophilic conditions at 37°C for 72 hours. Antimicrobial activity was detected as a minimum inhibitory concentration (MICs).
H. pylori strain ATCC 43504 was used as a control. Clarithromycin MICs were interpreted based on CLSI breakpoints (≥1.0 mg/l resistant) (CLSI. 2015) [
14]; we also used EUCAST breakpoints for all the antibiotics [
15]: amoxicillin ≥ 0.125 mg/l, levofloxacin ≥ 1 mg/l, tetracycline ≥ 1 mg/l, rifampin ≥ 1 mg/l, and metronidazole ≥ 8 mg/l.
4.4. Treatment and Follow-Up by 13 C-Urea Breath Test (13C UBT)
H. pylori positive patients received triple therapy with PPI (omeprazole 40 mg b.i.d.), clarithromycin (500 mg b.i.d.), amoxicillin (1 g b.i.d.) for 14 days. Forty-five days after the end of therapy, UBT 13C-urea (TAU-KIT, Isomed S.L., Madrid, Spain) was performed with citric acid and 100 mg of 13C-urea. Patients who evaluated positive after completion of the first treatment were re-treated with the second-line regimen with PPI (40 mg b.i.d.), amoxicillin (1 g b.i.d.), tetracycline (500 mg t.i.d.) and bismuth subsalicylate (524 mg b.i.d.) for 14 days.
4.5. Statistical Analysis
For eradication analysis, the population included all H. pylori positive patients who received the full treatment doses and had 13C UBT for follow-up, while for the interpretation of data related to antimicrobials, the population included all H. pylori positive patients who received or not pharmacological treatment and had an antibiogram. Eradication rates between the groups were compared by the Chi 2 test, or Fisher’s exact test, and odds ratios (OR) and 95% confidence intervals (CI) were estimated. All p values <0.05 were significant.
5. Conclusions
The high rate of resistance to metronidazole, clarithromycin and levofloxacin found in this study proves the risk of therapeutic failure when using these antimicrobials in the treatment of H. pylori in the population from Western Central Region of Colombia. The ideal success rate in eradicating H. pylori infection (≥90%) was not achieved in this study. H. pylori MDR is the biggest challenge in the management, and this is the first time that the region reports isolates with MDR. Additionally, the updated percentages of resistance to clarithromycin in this geographical area have increased.
The main results of this study emphasize that patients should be treated according to local rates of antimicrobial resistance, promoting the establishment of tests not only in these cities but throughout the country. Future studies with a larger number of patients are desirable to confirm these results. The results imply that inadequate eradication therapy not only increases healthcare costs due to the need for more studies or treatments, but also has a negative impact on the quantity and quality of life of affected patients.
Author Contributions
Conceptualization, A.A. and J.M.; methodology, A.A., Y.G., J.M., J.S., L.C., T.M, P.F., L.L., R.P. and B.A; software, P.F and R.P.; validation, A.A., T.M., B.A. and J.M.; formal analysis, A.A and P.F; investigation, A.A., J.M., B.A., and J.S.; data curation, A.A., T.M. and P.F.; resources, A.A., T.M. and P.F.; writing—original draft preparation, A.A and P.F.; writing—review and editing, A.A., Y.G., J.M., J.S., L.C., T.M, P.F., L.L., R.P. and B.A.; visualization, A.A., L.L.,T.M., B.A. and J.M.; supervision, A.A., T.M., Y.G. and Y.G.; project administration, A.A. and J.M; funding acquisition, A.A., J.S and J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Universidad Libre (Convocatoria interna 07) and Universidad Tecnológica de Pereira (Cod: CIE 5-17-4).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Universidad Tecnológica de Pereira [protocol code CBE-SYR-162016, September 12th of 2016] for studies involving humans.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patient[s] to publish this paper”.
Data Availability Statement
The data presented in this study are available on request from the corresponding author of the project A.A.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zamani M, Ebrahimtabar F, Zamani V, Miller WH, Alizadeh-Navaei R, Shokri-Shirvani J, et al. Systematic review with meta-analysis: the worldwide prevalence of Helicobacter pylori infection. Vol. 47, Alimentary Pharmacology and Therapeutics. Blackwell Publishing Ltd; 2018. p. 868–76. [CrossRef]
- Mejía Valencia T, Lucía Arturo Arias B, Álvarez Aldana A, Ignacio Moncayo Ortiz J, Javier Santacruz Ibarra J, Marcela Guaca Gonzales Y, et al. Prevalence, Clinical and Demographic Characteristics of Helicobacter pylori in Patients Undergoing Esophagogastroduodenoscopy in Specialized Health Centers in Two Departments of the Colombian Coffee Growing Axis. Revista de Investigación de la Universidad Norbert Wiener [Internet]. 2023;12(1). Available from: . [CrossRef]
- Cervantes García E. Diagnóstico y tratamiento de infecciones causadas por Helicobacter pylori. Rev Latinoam Patol Clin Med Lab [Internet]. 2016;63(4):179–89. Available from: www.medigraphic.com/patologiaclinicawww.medigraphic.org.mx.
- Gracía Yilán LM, Méndez Martínez M del C, García Arias T, Hernández Hernández T, Regueria Betancourt S. Caracterización de infección por Helicobacter pylori en pacientes asistidos en la consulta de gastroenterología. Revista Electrónica Dr Zoilo E Marinello Vidaurreta,. 2014;39(11).
- Guevara T AA, Criollo R Á, Suarez O JJ, Bohórquez L ME, Echeverry de Polanco MM. Coevolución genética Homo sapiens-Helicobacter pylori y sus implicaciones en el desarrollo del cáncer gástrico: una revisión sistemática. Rev Colomb Gastroenterol. 2016;31(4). [CrossRef]
- Hernández Hernández L, Lazcano Ponce E, López Vidal Y, Aguilar Gutierrez G. Relevance of Helicobacter pylori virulence factors for vaccine development. Salud Pública México. 2009;51. 10.1590/s0036-36342009000900010.
- Sugano K, Tack J, Kuipers EJ, Graham DY, El-Omar EM, Miura S, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015 Sep 1;64(9):1353–67. [CrossRef]
- Vainio H. Schistosomes, liver flukes, and Helicobacter pylori. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Lyon, 7–14 June 1994. IARC monographs on the evaluation of carcinogenic risks to humans 1994; 61:1–241.
- Hu Y, Zhu Y, Lu NH. Novel and effective therapeutic regimens for helicobacter pylori in an era of increasing antibiotic resistance. Front Cell Infect Microbiol. 2017 May 5;7(168). [CrossRef]
- Malfertheiner P, Megraud F, Rokkas T, Gisbert JP, Liou JM, Schulz C, et al. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut. 2022 Sep 1;71(9):1724–62. [CrossRef]
- Gu Q, Xia HHX, Wang JD, Wong WM, Chan AOO, Lai KC, et al. Update on clarithromycin resistance in Helicobacter pylori in Hong Kong and its effect on clarithromycin-based triple therapy. Digestion. 2006 Jul;73(2–3):101–6. [CrossRef]
- Savoldi A, Carrara E, Graham DY, Conti M, Tacconelli E. Prevalence of antibiotic resistance in Helicobacter pylori: A systematic review and meta-analysis in world health organization regions. Gastroenterology. 2018 Nov 1;155(5):1372-1382.e17. [CrossRef]
- Boyanova L, Hadzhiyski P, Gergova R, Markovska R. Evolution of Helicobacter pylori resistance to antibiotics: a topic of increasing concern. Vol. 12, Antibiotics. MDPI; 2023. [CrossRef]
-
CLSI. M45 Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria. 3rd edition Wayne. PA: Clinica.
-
ESCMID - European Society of Clinical Microbiology and Infectious Diseases 2008. (2023, 2 enero). eucast: Clinical breakpoints and dosing of antibiotics. https://www.eucast.org/clinical_breakpoints.
- Bedoya Gómez IJ, Alvarez Aldana A, Moncayo Ortiz JI, Guaca González YM, Santacruz Ibarra JJ, Arturo Arias BL, et al. Surveillance of the antimicrobial resistance rates of Helicobacter pylori ten years later in the western central region, Colombia. Digestive Diseases. 2020 May 1;38(3):196–203. [CrossRef]
- Álvarez A, Moncayo JI, Santacruz JJ, Santacoloma M, Corredor LF, Reinosa E. Antimicrobial susceptibility and mutations involved in clarithromycin resistance in Helicobacter pylori isolates from patients in the western central region of Colombia. Antimicrob Agents Chemother. 2009;53(9):4022–4. [CrossRef]
- Zuluaga Arbelaez N, Sierra Vargas EC, Guevara Casallas LG, Pérez Viana S. Estrategias terapéuticas para Helicobacter pylori en Colombia. CES Medicina. 2021 Oct 29;35(3):244–56. [CrossRef]
- Talebi Bezmin Abadi A. Diagnosis of Helicobacter pylori using invasive and noninvasive approaches. J Pathog. 2018;2018:9064952. [CrossRef]
- Pajares García JM, Pajares Villaroya R, Gisbert JP. Helicobacte rpylori: resistencia a los antibióticos. Rev Esp Enferm Dig (Madrid). 2007;99(2):67–70.
- Sierra F, Forero JD, Rey M. Tratamiento ideal del Helicobacter pylori: una revisión sistemática. Rev Gastroenterol Mex. 2014;79(1):28–49. [CrossRef]
- Malfertheiner P, Megraud F, O’Morain C, Gisbert JP, Kuipers EJ, Axon A, et al. Management of helicobacter pylori infection-the Maastricht V/Florence consensus report. Gut. 2017 Oct 1;66(1):6–30. [CrossRef]
- Figueroa M, Cortés A, Pazos Á, Bravo LE. Sensibilidad in vitro a amoxicilina y claritromicina de Helicobacter pylori obtenido de biopsias gástricas de pacientes en zona de bajo riesgo para cáncer gástrico. Biomédica. 2012;32:32–42. [CrossRef]
- Yepes CA, Rodríguez A, Ruiz Á, Ariza B. Resistencia antibiótica del Helicobacter pylori en el Hospital Universitario San Ignacio de Bogotá. Acta Médica Colombiana. 2008;33(1).
- Trespalacios AA, Otero Regino W, Mercado Reyes M. Helicobacter pylori resistance to metronidazole, clarithromycin and amoxicillin in Colombian patients. Rev Colomb Gastroenterol. 2010;
- Trespalacios AA, Otero W, Caminos JE, Mercado MM, Ávila J, Rosero LE, et al. Phenotypic and genotypic analysis of clarithromycin-resistant Helicobacter pylori from Bogotá D.C., Colombia. Journal of Microbiology. 2013 Aug;51(4):448–52. [CrossRef]
- Martínez M JD, Henao R SC, Lizarazo R JI. Resistencia antibiótica del Helicobacter pylori en América Latina y el Caribe. Rev Colomb Gastroenterol. 2014;
- Scaletsky ICA, Aranda KRS, Garcia GT, Gonç Alves MEP, Cardoso SR, Iriya K, et al. Application of real-time PCR stool assay for Helicobacter pylori detection and clarithromycin susceptibility testing in brazilian children. Blackwell Publishing Ltd, Helicobacter. 2011;16:311–5. [CrossRef]
- Atehortúa Rendón JD, Martínez A, Pérez Cala TL. Helicobacter pylori susceptibility to six commonly used antibiotics in Colombia. Rev Colomb Gastroenterol. 2020 Jul 1;35(3):351–61. [CrossRef]
- Papastergiou V, Georgopoulos SD, Stylianos K. Treatment of Helicobacter pylori infection: meeting the challenge of antimicrobial resistance. Wordl Journal of Gastroenterology. 2014;20(29). [CrossRef]
- Otero Regino W, Trespalacios AA, Otero E. Helicobacter pylori: current treatment an important challenge for gastroenterology. Rev Colomb Gastroenterol. 2009;24(3):279–92.
- Trespalacios Rangél AA, Otero W, Arévalo Galvis A, Poutou Piñales RA, Rimbara E, Graham DY. Surveillance of levofloxacin resistance in helicobacter pylori isolates in Bogotá-Colombia (2009-2014). PLoS One. 2016 Jul 1;11(7). [CrossRef]
- Arévalo A, Otero W, Trespalacios AA. Helicobacter pylori: multiple resistance in patients from Bogotá-Colombia. Biomedica. 2019;39(1):125–34. [CrossRef]
- Henao R SC, Otero R W, Ángel A LA, Martínez M JM. Resistencia primaria a metronidazol en aislamientos de Helicobacter pylori en pacientes adultos de Bogotá, Colombia. Rev Colomb Gastroenterol. 2009;
- Roberts LT, Issa PP, Sinnathamby ES, Granier M, Mayeux H, Eubanks TN, et al. Helicobacter pylori: a review of current treatment options in clinical practice. Vol. 12, Life. MDPI; 2022. [CrossRef]
- Miftahussurur M, Yamaoka Y. Appropriate first-line regimens to combat Helicobacter pylori antibiotic resistance: an asian perspective. Vol. 20, Molecules. MDPI AG; 2015. p. 6068–92. [CrossRef]
- Boyanova L, Hadzhiyski P, Kandilarov N, Markovska R, Mitov I. Multidrug resistance in Helicobacter pylori: current state and future directions. Expert Rev Clin Pharmacol. 2019 Sep 2;12(9):909–15. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).