Submitted:
20 June 2023
Posted:
20 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
2.1. Traditional uses
2.2. Botanical description
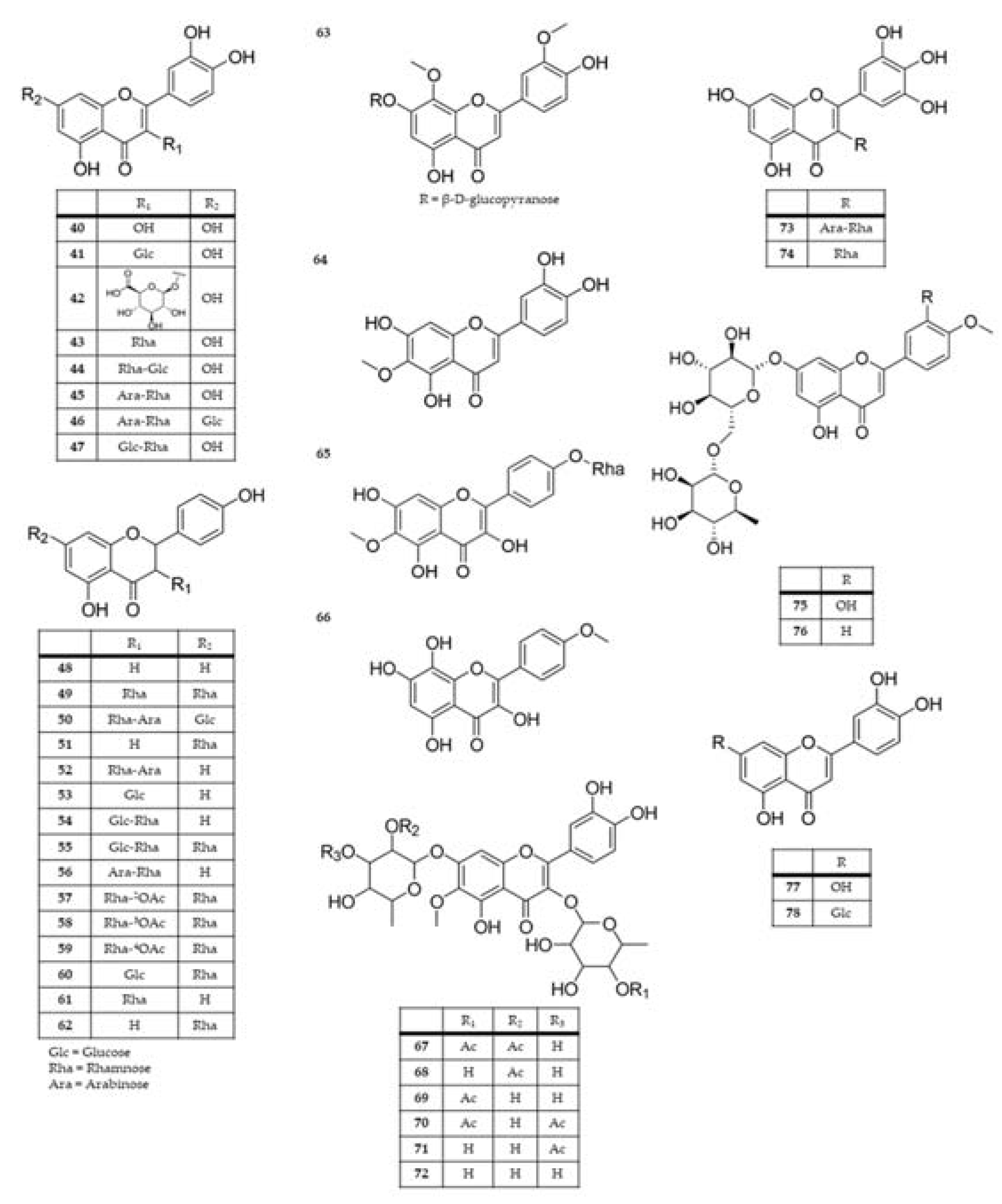
2.3. Chemical composition
2.4. Pharmacological activities
3. Methodology
4. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- POWO (2023). Crassulaceae. Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Published on the Internet; https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:30000887-2. Retrieved April 2023.
- Fernandes JM, Cunha LM, Azevedo EP, Lourenco EMG et al. (2019) Kalanchoe laciniata and Bryophyllum pinnatum: an updated review about ethnopharmacology, phytochemistry, pharmacology and toxicology. Rev Bras Farmacogn 29(4):529-558. [CrossRef]
- Smith GF, Figueiredo E, Wyk AE. (2019) Chapter 4 - The Genus Kalanchoe (Crassulaceae) in Southern Africa, Academic Press 23-28. [CrossRef]
- GBIF (2023). Crassulaceae. The Global Biodiversity Information Facility. Published on the Internet; https://www.gbif.org/species/2406. Retrieved June 2023.
- POWO (2023). Kalanchoe. Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Published on the Internet; https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:30060186-2. Retrieved April 2023.
- Akulova-Barlow, Z. Kalanchoe. Cactus and Succulent Journal 2009; 81(6):268-276. [CrossRef]
- GBIF (2023). Kalanchoe. The Global Biodiversity Information Facility. Published on the Internet; https://www.gbif.org/species/2985928. Retrieved June 2023.
- Ferreira RT, Coutinho MA, Malvar DC, Costa EA et al. (2014) Mechanisms Underlying the Antinociceptive, Antiedematogenic, and Anti-Inflammatory Activity of the Main Flavonoid from Kalanchoe pinnata. Evid Based Complementary Altern Med. [CrossRef]
- Mawla F, Khatoon S, Rehana F, Jahan S et al. (2012) Ethnomedicinal plants of folk medicinal practitioners in four villages of Natore and Rajshahi districts, Bangladesh. Am-Eurasian J Sustain Agric 6(4): 406-416.
- Molina GA, Esparzab R, López-Mirandab JL, Hernández-Martínez AR et al. (2019) Green synthesis of Ag nanoflowers using Kalanchoe daigremontiana extract for enhanced photocatalytic and antibacterial activities. Colloids Surf B. 180:141-149. [CrossRef]
- Stefanowicz-Hajduk J, Asztemborska M, Krauze-Baranowska M, Godlewska S et al. (2020) Identification of Flavonoids and Bufadienolides and Cytotoxic Effects of Kalanchoe daigremontiana Extracts on Human Cancer Cell Lines. Planta Med 86(4): 239-246. [CrossRef]
- Zawirska-Wojtasiak R, Jankowska B, Piechowska P, Szkudlarz SM. (2019) Vitamin C and aroma composition of fresh leaves from Kalanchoe pinnata and Kalanchoe daigremontiana. Sci Rep 9:19786. [CrossRef]
- Okwu DE; Josiah C (2006) Evaluation of the chemical composition of two Nigerian medicinal plants. Afr J Biotechnol 5(4):357-361.
- Kołodziejczyk-Czepas J, Nowak P, Wachowicz B, Piechocka J et al. (2016) Antioxidant efficacy of Kalanchoe daigremontiana bufadienolide-rich fraction in blood plasma in vitro. Pharm Biol 54(12):3182-3188. [CrossRef]
- Kołodziejczyk-Czepas J, Stochmal A (2017) Bufadienolides of Kalanchoe species: an overview of chemical structure, biological activity and prospects for pharmacological use. Phytochem Rev 16:1155-1171. [CrossRef]
- Lai ZR, Peng WH, Ho YL, Huang SC et al. (2010) Analgesic and anti-inflammatory activities of the methanol extract of Kalanchoe gracilis (L.) DC stem in mice. Am J Chinese Med 38(3):529-546. [CrossRef]
- Lai ZR, Ho YL, Huang SC, Huang TH et al. (2011) Antioxidant, Anti-Inflammatory, and Antiproliferative Activities of Kalanchoe gracilis (L.) DC Stem. Am J Chinese Med 39(6):1275–1290. [CrossRef]
- Asiedu-Gyekye IJ, Antwi DA, Awortwe C, N'guessan BB et al. Short-term administration of an aqueous extract of Kalanchoe integra var. crenata (Andr.) Cuf leaves produce no major organ damage in Sprague-Dawley rats. J Ethnopharmacol 2014; 151(2):891-6. [CrossRef]
- Asiedu-Gyekye IJ, Arhin E, Arthur SA, N'guessan BB et al. Genotoxicity, nitric oxide level modulation and cardio-protective potential of Kalanchoe integra var. crenata (Andr.) cuf leaves in murine models. J Ethnopharmacol 2022; 283:114640. [CrossRef]
- Awortwe C, Manda KV, Avonto C, Khan IS et al. In vitro evaluation of reversible and time-dependent inhibitory effects of Kalanchoe crenata on CYP2C19 and CYP3A4 activities. Drug Metab Lett 2015; 9(1):48-62. [CrossRef]
- Garcia-Perez P, Lozano-Milo E, Landin M, Gallego PP (2020) From Ethnomedicine to Plant Biotechnology and Machine Learning: The Valorization of the Medicinal Plant Bryophyllum sp. Pharmaceuticals 13(12):444. [CrossRef]
- Kamgang R, Foyet AF, Essame JL, Ngogang JY (2012) Effect of methanolic fraction of Kalanchoe crenata on metabolic parameters in adriamycin-induced renal impairment in rats. Indian J Pharmacol 44(5):566-570. [CrossRef]
- Kuete V, Fokou FW, Karaosmanoğlu O, Beng VP et al. (2017) Cytotoxicity of the methanol extracts of Elephantopus mollis, Kalanchoe crenata and 4 other Cameroonian medicinal plants towards human carcinoma cells. BMC Complement Altern Med 17(1):280. [CrossRef]
- Mutie, F.M.; Mbuni, Y.M.; Rono, P.C.; Mkala, E.M.; Nzei, J.M.; Phumthum, M.; Hu, G.-W.; Wang, Q.-F. Important Medicinal and Food Taxa (Orders and Families) in Kenya, Based on Three Quantitative Approaches. Plants 2023, 12, 1145. [Google Scholar] [CrossRef] [PubMed]
- Ngezahayo J, Havyarimana F, Hari L, Stévigny C et al. (2015) Medicinal plants used by Burundian traditional healers for the treatment of microbial diseases. J Ethnopharmacol 173:338-351. [CrossRef]
- Odukoya, J.O.; Odukoya, J.O.; Mmutlane, E.M.; Ndinteh, D.T. Ethnopharmacological Study of Medicinal Plants Used for the Treatment of Cardiovascular Diseases and Their Associated Risk Factors insub-Saharan Africa. Plants 2022, 11, 1387. [Google Scholar] [CrossRef] [PubMed]
- Raadts E (1977) The Genus Kalanchoe (Crassulaceae) in Tropical East Africa. Willdenowia 8 (1):101-157.
- Akentieva NP, Shushanov SS, Gizatullin AR, Prikhodchenko TR et al. The Effect of Plant Extracts Kalanchoe daigremontiana and Aloe arborescens on the Metabolism of Human Multiple Myeloma Cells. Biointerface Res Appl Chem 2021; 11(5):13171-13186. [CrossRef]
- Chernetskyy M, Woźniak A, Skalska-Kamińska A, Żuraw B et al. Structure Of Leaves And Phenolic Acids In Kalanchoe daigremontiana Raym.-Hamet & H. Perrier. Acta Sci Pol Hortorum Cultus 2018, 17(4):137-155. [CrossRef]
- Moniuszko-Szajwaj B, Pecio L, Kowalczyk M, Stochmal A (2016) New Bufadienolides Isolated from the Roots of Kalanchoe daigremontiana (Crassulaceae). Molecules 21(243):1-13. [CrossRef]
- Hsieh YJ, Yang MY, Leu IY, Chen C et al. (2012) Kalanchoe tubiflora extract inhibits cell proliferation by affecting the mitotic apparatus. BMC Complement Altern Med 12(59). http://www.biomedcentral.com/1472-6882/12/149.
- Huang HC, Huang GJ, Liaw CC, Yanget CS al. (2013a) A new megastigmane from Kalanchoe tubiflora (Harvey) Hamet. Phytochem Lett 6(3):379-382. [CrossRef]
- Huang HC, Lin MK, Yang HL, Hseu YC et al. (2013b) Cardenolides and bufadienolide glycosides from Kalanchoe tubiflora and evaluation of cytotoxicity. Planta Med 79:1362-1369. [CrossRef]
- Schmidt C, Fronza M, Goetter M, Geller F et al. (2009) Biological studies on Brazilian plants used in wound healing. J Ethnopharmacol 122(3):523-532. [CrossRef]
- Getachew M, Belayneh A, Kebede B et al. Medicinal plants used for management of hemorrhoids in Ethiopia: A systematic review. Heliyon. 2022 Aug 13;8(8): e10211. [CrossRef]
- Arias-González I, García-Carrancá AM, Cornejo-Garrido J, Ordaz-Pichardo C. Cytotoxic effect of Kalanchoe flammea and induction of intrinsic mitochondrial apoptotic signaling in prostate cancer cells. J Ethnopharmacol 2018; 222:133-147. [CrossRef]
- Richwagen N, James TL, Brandon LFD, Quave CL (2019) Antibacterial activity of Kalanchoe mortagei and K. fedtschenkoi against ESKAPE pathogens. Front Pharmacol 10. [CrossRef]
- Palumbo A, Casanova LM, Corrêa MFP, Da Costa NM et al. (2019) Potential therapeutic effects of underground parts of Kalanchoe gastonis-bonnieri on benign prostatic hyperplasia. Evid Based Complementary Altern Med. [CrossRef]
- Frimpong-Manso S, Asiedu-Gyekye IJ, Naadu JP, Magnus-Aryitey GT et al. (2015) Micro and macro element composition of Kalanchoe integra leaves: an adjuvant treatment for hypertension in Ghana. Int J Hypertens. [CrossRef]
- Costa ACO, Fernandes JM, Negreiros TS, Mendoça JN et al. Quantification of Chemical Marker of Kalanchoe brasiliensis (Crassulaceae) Leaves by HPLC-DAD. J Liq Chromatogr Relat Technol 2015, 38(7):795-800. [CrossRef]
- Cruz BP, Chedier LM, Fabri RL, Pimenta DS. Chemical and agronomic development of Kalanchoe brasiliensis Camb. and Kalanchoe pinnata (Lamk.) Pers under light and temperature levels. Anais da Academia Brasileira de Ciências 2011, 83(4):1435-1441.
- Cruz BP, Chedier LM, Peixoto PHP, Fabri RL et al. Effects of light intensity on the distribution of anthocyanins in Kalanchoe brasiliensis Camb. and Kalanchoe pinnata (Lamk.) Pers. Anais da Academia Brasileira de Ciências 2012, 84(1):211-217.
- de Araujo ERD, Guerra GCB, Araújo DFS, de Araújo AA et al. (2018) Gastroprotective and antioxidant activity of Kalanchoe brasiliensis and Kalanchoe pinnata leaf juices against indomethacin and ethanol- 45 induced gastric lesions in rats. Int J Mol Sci. [CrossRef]
- Fernandes JM, Félix-Silva J, da Cunha LM, Gomes JA et al. (2016) Inhibitory Effects of Hydroethanolic Leaf Extracts of Kalanchoe brasiliensis and Kalanchoe pinnata (Crassulaceae) against Local Effects Induced by Bothrops jararaca Snake Venom. Plos One 11(12):e0168658. [CrossRef]
- Fonseca AG, Ribeiro Dantas LLSF, Fernandes JM, Zucolotto SM et al. (2018) In Vivo and In Vitro Toxicity Evaluation of Hydroethanolic Extract of Kalanchoe brasiliensis (Crassulaceae) Leaves. J Toxicol. [CrossRef]
- Haile AA, Tsegay BA, Seid A, Adnew W, Moges A. A Review on Medicinal Plants Used in the Management of Respiratory Problems in Ethiopia over a Twenty-Year Period (2000-2021). Evid Based Complement Alternat Med. 2022 Jun 27; 2022:2935015. [CrossRef]
- Manan M, Hussain L, Ijaz H, Qadir MI (2015) Phytochemical screening of different extracts of Kalanchoe laciniata. Pak J Pharm Sci 29(4):1321-1324. [CrossRef]
- Sharif A, Akhtar MF, Akhtar B, Saleem A et al. (2017) Genotoxic and Cytotoxic Potential Of Whole Plant Extracts Of Kalanchoe Laciniata By Ames And Mtt Assay. EXCLI Journal 16:593-601. [CrossRef]
- Stefanowicz-Hajduk J, Heringa A, Gucwaa M, Hałasa R et al. (2020) Biological activities of leaf extracts from selected Kalanchoe species and their relationship with bufadienolides content. Pharm Biol 58(1):732-740. [CrossRef]
- Bogale M, Sasikumar JM, Egigu MC. An ethnomedicinal study in tulo district, west hararghe zone, oromia region, Ethiopia, Heliyon, Volume 9, Issue 4, 2023, e15361, ISSN 2405-8440. [CrossRef]
- Singab AB, El-Ahmady SH, Labib RM, Fekry SS (2011) Phenolics from Kalanchoe marmorata Baker, Family Crassulaceae. Bull Fac Pharm Cairo Univ 49:1-5. [CrossRef]
- Tahir M, Asnake H, Beyene T, Van Damme P, Mohammed A. Ethnobotanical study of medicinal plants in Asagirt District, Northeastern Ethiopia. Trop Med Health. 2023 Jan 9;51(1):1. PMID: 36617576; PMCID: PMC9827656. [CrossRef]
- Abebe, W. An overview of Ethiopian traditional medicinal plants used for cancer treatment. European J Med Plants 2016; 14(4):1-16. [CrossRef]
- Agize M, Asfaw Z, Nemomissa S. et al. Ethnobotany of traditional medicinal plants and associated indigenous knowledge in Dawuro Zone of Southwestern Ethiopia. J Ethnobiology Ethnomedicine 18, 48 (2022). [CrossRef]
- Mekonnen AB, Mohammed AS, Tefera AK. Ethnobotanical Study of Traditional Medicinal Plants Used to Treat Human and Animal Diseases in Sedie Muja District, South Gondar, Ethiopia. Evidence-Based Complementary and Alternative Medicine, vol. 2022, Article ID 7328613, 22 pages, 2022. [CrossRef]
- Tadesse TY, Zeleke MM, Dagnew SB. Review of Ethnobotanical and Ethnopharmacological Evidence of Some Ethiopian Medicinal Plants Traditionally Used for Peptic Ulcer Disease Treatment. Clin Exp Gastroenterol. 2022; 15:171-187. [CrossRef]
- Ahmed MN, Hughes K. Role of ethno-phytomedicine knowledge in healthcare of COVID-19: advances in traditional phytomedicine perspective. Beni-Suef Univ J Basic Appl Sci 11, 96 (2022). [CrossRef]
- Ahmed M, Wazir ZU, Khan RA, Khan MI et al. Pharmacological evaluation of crude methanolic extract of Kalanchoe pinnata leaves. Environ Toxicol Chem 2013; 95(9):1539-1545. [CrossRef]
- Anadozie SO, Akinyemi JA, Agunbiade S, Ajiboyeet BO et al. Bryophyllum pinnatum inhibits arginase II activity and prevents oxidative damage occasioned by carbon tetrachloride (CCl4) in rats. Biomed Pharmacother 2018; 101:8-13. [CrossRef]
- Bachmann S, Betschart C, Gerber J, Fürer K et al. Potential of Bryophyllum pinnatum as a detrusor relaxant: an in vitro exploratory study. Planta Med 2017; 83: 1274-1280. [CrossRef]
- Barbhuiya PA, Laskar AM, Mazumdar H et al. Ethnomedicinal Practices and Traditional Medicinal Plants of Barak Valley, Assam: a systematic review. J Pharmacopuncture 2022; 25 (3): 149-185. [CrossRef]
- Bhandari R, Sabina G, Nisha A, Devika G et al. Evaluation of phytochemical, antioxidant, and memory-enhancing activity of Garuga pinnata Roxb. Bark and Bryophyllum pinnatum (Lam) Oken. leaves. Sci World J 2021. 1155.
- Bhatti M, Kamboj A, Saluja AK, Jain UK. In vitro evaluation and comparison of antioxidant activities of various extracts of leaves and stems of Kalanchoe pinnatum. Int J Green Pharm 2012; 6(4)340-347. [CrossRef]
- Bhavsar S, Chandel D. Cytotoxic and genotoxic effects of Kalanchoe pinnata (Lam.) Pers. fresh leaf juice in the cultured human blood lymphocytes. Drug Chem Toxicol 2019; 45:1:360-366. [CrossRef]
- Bopda OSM, Longo F, Bella TN, Edzah PMO et al. Antihypertensive activities of the aqueous extract of Kalanchoe pinnata (Crassulaceae) in high salt-loaded rats. J Ethnopharmacol 2014; 153(2):400-407. [CrossRef]
- Clemen-Pascual LM, Macahig RAS, Rojas NRL. Comparative toxicity, phytochemistry, and use of 53 Philippine medicinal plants. Toxicol Rep. 2021 Dec 10; 9:22-35. [CrossRef]
- Coutinho MAS, Casanova LM, Nascimento LBDS, Leal D et al. Wound healing cream formulated with Kalanchoe pinnata major flavonoid is as effective as the aqueous leaf extract cream in a rat model of excisional wound. Nat Prod Res 2020, 35(24):6034-6039. [CrossRef]
- Cruz EA, Reuter H, Martin N, Dehzad N et al. Kalanchoe pinnata inhibits mast cell activation and prevents allergic airway disease. Phytomedicine 2012, 19(2):115-121. [CrossRef]
- Cryer M, Lane K, Greer M, Cates R et al. Isolation and identification of compounds from Kalanchoe pinnata having human alphaherpesvirus and vaccinia virus antiviral activity. Pharm Biol 2017, 55(1)1586-1591. [CrossRef]
- El Abdellaoui S, Destandau E, Toribio A, Elfakir C et al. (2010) Bioactive molecules in Kalanchoe pinnata leaves: extraction, purification, and identification. Anal Bioanal Chem 398:1329–1338. [CrossRef]
- Fernandes JM, Ortiz S, Padilha M, Tavares RPM et al. (2021) Bryophyllum pinnatum markers: CPC isolation, simultaneous quantification by a validated UPLC-DAD method and biological evaluations. J Pharm Biomed Anal 193. [CrossRef]
- Furer K, Simões-Wüst AP, von Mandach U, Hamburger M et al. (2016) Bryophyllum pinnatum and Related Species Used in Anthroposophic Medicine: Constituents, Pharmacological Activities, and Clinical Efficacy. Planta Med 82:930-941. [CrossRef]
- Gomes DCO, Muzitano MF, Costa SS, Rossi-Bergmann B et al. (2010) Effectiveness of the immunomodulatory extract of Kalanchoe pinnata against murine visceral leishmaniasis. Parasitology 137(4):613-618. [CrossRef]
- Kadam S, Madhusoodhanan V, Bandgar A, Kaushik KS. (2020) From Treatise to Test: Evaluating Traditional Remedies for Anti-Biofilm Potential. Front Pharmacol 11. [CrossRef]
- Kukuia KEK, Asiedu-Gyekye IJ, Woode E, Biney RP et al. (2015) Phytotherapy of experimental depression: Kalanchoe integra var. crenata (Andr.) Cuf leaf extract. J Pharm Bioallied Sci 7(1):26-31. [CrossRef]
- Leal-Costa MV, Nascimento LBS, Moreira NS, Reinert F et al. (2010) Influence of Blue Light on the Leaf Morphoanatomy of In Vitro Kalanchoe pinnata (Lamarck) Persoon (Crassulaceae). Microsc Microanal 16:576-582. [CrossRef]
- Lebedeva AA, Zakharchenko NS, Trubnikova EV, Medvedeva OA et al. (2017) Bactericide, Immunomodulating, and Wound Healing Properties of Transgenic Kalanchoe pinnata Synergize with Antimicrobial Peptide Cecropin P1 In Vivo. J Immunol Res. 1155. [CrossRef]
- Majaz AQ; Khurshid M; Nazim S (2011a) The Miracle Plant (Kalanchoe pinnata): A Phytochemical and Pharmacological Review. Int J Res Ayurveda Pharm 2(5):1478-1482.
- Majaz AQ; Khurshid M; Nazim S; Rahil K et al. (2011b) Evaluation of Antimicrobial activity of Roots of K. pinnata. Int J Pharmacol Bio Sci 5(1):93-96.
- Mawla F, Khatoon S, Rehana F, Jahan S et al. (2012) Ethnomedicinal plants of folk medicinal practitioners in four villages of Natore and Rajshahi districts, Bangladesh. Am-Eurasian J Sustain Agric 6(4): 406-416.
- Mora-Pérez A; Hernández-Medel MDR (2016) Anticonvulsant activity of methanolic extract from Kalanchoe pinnata (Lam.) stems and roots in mice: A comparison to diazepam. Neurología 31(3):161-168. [CrossRef]
- Mourão RHV, Santos FO, Franzotti EM, Moreno MPN et al. (1999) Antiinflammatory Activity and Acute Toxicity (LD50) of the Juice of Kalanchoe brasiliensis (Comb.) Leaves Picked Before and During Blooming. Phytother Res 13: 352-354. [CrossRef]
- Muzitano MF, Bergonzi MC, De Melo GO, Lage CLS et al. (2011) Influence of cultivation conditions, season of collection and extraction method on the content of antileishmanial flavonoids from Kalanchoe pinnata. J Ethnopharmacol 133:132-137. [CrossRef]
- Nascimento LBS, Leal-Costa MV, Coutinho MAS, Moreira NS et al. (2013) Increased antioxidant activity and changes in phenolic profile of Kalanchoe pinnata (Lamarck) Persoon (Crassulaceae) specimens grown under supplemental blue light. Photochem Photobiol 89:391-399. [CrossRef]
- Nascimento LBS, Leal-Costa MV, Menezes EA, Lopes VR et al. Ultraviolet-B radiation effects on phenolic profile and flavonoid content of Kalanchoe pinnata, Journal of Photochemistry and Photobiology B: Biology, Vol. 148, 2015, Pages 73-81.
- Nascimento LBS, Aguiar PF, Leal-Costa MV, Coutinho MAS et al. (2018) Optimization of Aqueous Extraction from Kalanchoe pinnata Leaves to Obtain the Highest Content of an Anti-inflammatory Flavonoid Using a Response Surface Model. Phytochem Anal 29(3):308-315. [CrossRef]
- Oufir M, Seiler C, Gerodetti M, Gerber J et al. (2015) Quantification of bufadienolides in Bryophyllum pinnatum leaves and manufactured products by UHPLC-ESIMS/MS. Planta Med 81:1190-1197. [CrossRef]
- Patil SB, Dongare VR, Kulkarni CR, Joglekar MM et al. (2013) Antidiabetic activity of Kalanchoe pinnata in streptozotocin-induced diabetic rats by glucose independent insulin secretagogue action. Pharm Biol 51(11):1411-1418. [CrossRef]
- Paul A, Chakraborty N, Sarkar A, Acharya K, Ranjan A, Chauhan A, Srivastava S, Singh AK, Rai AK, Mubeen I, Prasad R. Ethnopharmacological Potential of Phytochemicals and Phytogenic Products against Human RNA Viral Diseases as Preventive Therapeutics. BioMed Research International, vol. 2023, Article ID 1977602, 21 pages, 2023. [CrossRef]
- Santos S, Zurfluh L, Mennet M, Potterat O et al. (2021) Bryophyllum pinnatum Compounds Inhibit Oxytocin-Induced Signaling Pathways in Human Myometrial Cells. Front Pharmacol 12:1-10. [CrossRef]
- Yuliani T, Dewijanti ID, Banjarnahor SDS (2016) Antidiabetic activity of ethanolic extracts of Kalanchoe pinnata leaves in alloxan induced hyperglycemic rats. Indones J Pharm 27(3):139-144. [CrossRef]
- Zakharchenko NS, Lebedeva AA, Furs OV, Rukavtsova EB et al. (2016) Producing Marker-Free Kalanchoe Plants Expressing Antimicrobial Peptide Cecropin P1 Gene. Russ J Plant Physiol 63(2):273–282. [CrossRef]
- Zakharchenko NS, Rukavtsova EB, Shevchuk TV, Furs OV et al. (2016) The Obtainment and Characteristics of Kalanchoe pinnata L. Plants Expressing the Artificial Gene of the Cecropin P1 Antimicrobial Peptide. Appl Biochem Microbiol 52(4):421–428. [CrossRef]
- Zakharchenko NS, Belous AS, Biryukova,YK, Medvedeva OA et al. (2017) Immunomodulating and Revascularizing Activity of Kalanchoe pinnata Synergize with Fungicide Activity of Biogenic Peptide Cecropin P1. J Immunol Res. [CrossRef]
- Kamboj A; Saluja AK (2009) Bryophyllum pinnatum (Lam.) Kurz: phytochemical and pharmacological profile: A review. Pharmacogn Rev 3(6):364-375.
- Lorenzi H; Matos FJA (2008) Plantas medicinais no Brasil: nativas exóticas cultivadas. 2.ed. Nova Odessa: Instituto Plantarum de Estudos da Flora, p. 223.
- Milad R, El-Ahmady S, Singab AN (2014) Genus Kalanchoe (Crassulaceae): A Review of Its Ethnomedicinal, Botanical, Chemical and Pharmacological Properties. J Medicinal Plants 4:86-104.
- Herrera I, Hernandez MJ, Lampo M, Nassar JM (2011) Plantlet recruitment is the key demographic transition in invasion by Kalanchoe daigremontiana. Popul Ecol 54:225-237. [CrossRef]
- Moreira NS, Nascimento LBS, Leal-Costa MV, Tavares ES (2012) Comparative anatomy of leaves of Kalanchoe pinnata and K. crenata in sun and shade conditions, as a support for their identification. Rev Bras Farmacogn 22(5):929-936. [CrossRef]
- Wild B, Wanek W, Postl W, Richter A (2010) Contribution of carbon fixed by Rubisco and PEPC to phloem export in the Crassulacean acid metabolism plant Kalanchoe daigremontiana. J Exp Bot 61(5):1375–1383. [CrossRef]
- Yang X, Hu R, Yin H et al. (2017) The Kalanchoë genome provides insights into convergent evolution and building blocks of crassulacean acid metabolism. Nat Commun 8(1899). [CrossRef]
- Gotoh E, Oiwamoto K, Inoue SI, Shimazaki KI et al. (2019) Stomatal response to blue light in crassulacean acid metabolism plants Kalanchoe pinnata and Kalanchoe daigremontiana. J Exp Bot 70(4):1367-1374. [CrossRef]
- Herrando-Moraira S, Vitales D, Nualart N, Gómez-Bellver C et al. (2020) Global distribution patterns and niche modelling of the invasive Kalanchoe × houghtonii (Crassulaceae). Sci Rep 10, 3143. [CrossRef]
- Al-Khayri JM, Mahdy EMB, Taha HSA et al. Genetic and Morphological Diversity Assessment of Five Kalanchoe Genotypes by SCoT, ISSR and RAPD-PCR Markers. Plants 2022, 11, 1722. [CrossRef]
- García-Sogo B, Pineda B, Castelblanque L, Antón T et al. (2009) Efficient transformation of Kalanchoe blossfeldiana and production of male-sterile plants by engineered anther ablation. Plant Cell Rep 29(1):61-77. [CrossRef]
- Liu C, Zhu C, Zeng HM (2016) Key KdSOC1 gene expression profiles during plantlet morphogenesis under hormone, photoperiod, and drought treatments. Genet Mol Res 15(1):1-14. [CrossRef]
- Quintero EJ, De León EG, Morán-Pinzón J, Mero A et al. (2021) Evaluation of the Leaf Extracts of Kalanchoe Pinnata and Kalanchoe Daigremontiana Chemistry, Antioxidant and Anti-inflammatory Activity. European J Med Plants 32(5):45-54. [CrossRef]
- Casanova JM, Nascimento LBS, Casanova LM, Leal-Costa MV et al. Differential Distribution of Flavonoids and Phenolic Acids in Leaves of Kalanchoe delagoensis Ecklon & Zeyher (Crassulaceae). Microsc 2020, 26(5):1061-1068. [CrossRef]
- Brzezicka E, Karwowska K, Kozieradzka-Kiszkurno M, Chernetskyy M et al. Leaf micromorphology of Kalanchoë laciniata (Crassulaceae). Modern Phytomorphology 2015; 8:49-52.
- Bhavsar S, Bhavita D, Maitreyi Z, Divya C. A comparative pharmacognostical and phytochemical analysis of Kalanchoe pinnata (Lam.) Pers. leaf extracts. J Pharmacogn Phytochem 2018; 7(5):1519-1527.
- Chernetskyy M; Weryszko-Chmielewska E. Structure of Kalanchoë pumila Bak. leaves (Crassulaceae DC.). Acta Agrobot 2008, 61(2).
- McKenzie RA, Franke FP, Dunster PJ (1987) The toxicity to cattle and bufadienolide content of six Bryophyllum species. Aust Vet J 64(10):298-301. [CrossRef]
- Supratman U, Fujita T, Akiyama K, Hayashi H. New insecticidal bufadienolide, bryophyllin C, from Kalanchoe pinnata. Biosci Biotechnol Biochem. 2000 Jun;64(6):1310-2. [CrossRef]
- Wu PL, Hsu YL, Wu TS, Bastow KF et al. (2006) Kalanchosides A–C, new cytotoxic bufadienolides from the aerial parts of Kalanchoe gracilis. Org Lett 8(23):5207-10. [CrossRef]
- Yamagishi T, Haruna M, Yan XZ, Chang JJ, Lee KH. Antitumor agents, 110. Bryophyllin B, a novel potent cytotoxic bufadienolide from Bryophyllum pinnatum. J Nat Prod. 1989 Sep-Oct;52(5):1071-9. [CrossRef]
- Capon RJ, MacLeod JK, Oelrichs PB. Bryotoxins B and C, toxic bufadienolide orthoacetates from the flowers of Bryophyllum tubiflorum (Crassulaceae). Australian Journal of Chemisty, Vol 39, Issue 10, Pages 1711-15, 1986. [CrossRef]
- Kołodziejczyk-Czepas J, Pasiński B, Ponczek MB, Moniuszko-Szajwaj B et al. (2018) Bufadienolides from Kalanchoe daigremontiana modulate the enzymatic activity of plasmin - In vitro and in silico analyses. Int J Biol Macromol 120:1591-1600. [CrossRef]
- Wagner H, Fischer M, Lotter H. Isolation and Structure Determination of Daigremontianin, a Novel Bufadienolide from Kalanchoe daigremontiana. Planta Med 1985; 51(2): 169-170. [CrossRef]
- Ürményi FG, Saraiva GD, Casanova LM, Matos AD et al. (2016) Anti-HSV-1 and HSV-2 flavonoids and a new kaempferol triglycoside from the medicinal plant Kalanchoe daigremontiana. Chem Biodivers 13(12):1707-1714. [CrossRef]
- Anderson LAP, Steyn PS, Heerden FR. The characterization of two novel bufadienolides, lanceotoxins A and B from Kalanchoe lanceolata [Forssk.] Pers. J Chem Soc, Perkin trans 1 15 1984; 1573-1575. [CrossRef]
- Kuo PC, Kuo TH, Su CR, Liou MJ et al. (2008) Cytotoxic principles and α-pyrone ring-opening derivatives of bufadienolides from Kalanchoe hybrida. Tetrahedron 64(15):3392-3396. [CrossRef]
- Chibli, L. A. et al. Anti-inflammatory effects of Bryophyllum pinnatum (Lam.) Oken ethanol extract in acute and chronic cutaneous inflammation. J Ethnopharmacol. 11, 330–338 (2014).
- Coutinho MA, Muzitano MF, Cruz EA, Bergonzi MC, Kaiser CR, Tinoco LW, Bilia AR, Vincieric FF, Rossi-Bergmann B, Costa SS. Flowers from Kalanchoe pinnata are a rich source of T cell-suppressive flavonoids. Nat Prod Commun. 2012 Feb;7(2):175-8. PMID: 22474947.
- Elansary HO, Abdelgaleil SAM, Mahmoud EA, Yessoufou K et al. (2018) Effective antioxidant, antimicrobial and anticancer activities of essential oils of horticultural aromatic crops in northern Egypt. BMC Complement Altern Med.
- Elizondo-Luevano JH, Pérez-Narváez OA, Sánchez-García E, Castro-Ríos R et al. (2021) In Vitro Effect of Kalanchoe daigremontiana and Its Main Component, Quercetin against Entamoeba histolytica and Trichomonas vaginalis. Iran J Parasitol 16(3):394-401.
- Hegazy MM, Metwaly AM, Mostafa AE, Radwan MM et al. (2021) Biological and chemical evaluation of some African plants belonging to Kalanchoe species: Antitrypanosomal, cytotoxic, antitopoisomerase I activities and chemical profiling using ultra-performance liquid chromatography/quadrupole-time-of-flight mass spectrometer. Pharmacog Mag 17:6-15.
- Liu KCS, Yang SL, Roberts MF, Phillipson JD (1989) Eupafolin Rhamnosides from Kalanchoe gracilis. J Nat Prod 52(5):970-974.
- Nielsen AH, Olsen CE, Møller BL. Flavonoids in flowers of 16 Kalanchoë blossfeldiana varieties. Phytochemistry. 2005 Dec;66(24):2829-35. Epub 2005 Nov 16. [CrossRef]
- Sarkar R, Mondal C, Bera R, Chakraborty S et al. (2015) Antimicrobial properties of Kalanchoe blossfeldiana: a focus on drug resistance with reference to quorum sensing-mediated bacterial biofilm formation. J Pharm Pharmacol 67(7):951-62. [CrossRef]
- Fürer K, Raith M, Brenneisen R, Mennet M et al. (2013) Two new flavonol glycosides and a metabolite profile of Bryophyllum pinnatum, a phytotherapeutic used in obstetrics and gynaecology. Planta Med 79(16):1565-71. [CrossRef]
- Muzitano MF, Cruz EA, de Almeida AP, Da Silva SA, Kaiser CR, Guette C, Rossi-Bergmann B, Costa SS. Quercitrin: an antileishmanial flavonoid glycoside from Kalanchoe pinnata. Planta Med. 2006 Jan;72(1):81-3. [CrossRef]
- de Araujo ERD, Félix-Silva J, Xavier-Santos JB, Fernandes JM et al. (2019) Local anti-inflammatory activity: Topical formulation containing Kalanchoe brasiliensis and Kalanchoe pinnata leaf aqueous extract. Biomed Pharmacother. [CrossRef]
- Tatsimo, S., Tamokou, J., Havyarimana, L., Csupor, D., Forgo, P., Hohmann, J., Kuiate,J.-R., Tane, P., 2012. Antimicrobial and antioxidant activity of kaempferol rhamnoside derivatives from Bryophyllum pinnatum. BMC Res. Notes. [CrossRef]
- Costa SS, Jossang A, Bodo B, Souza ML, Moraes VL. Patuletin acetylrhamnosides from Kalanchoe brasiliensis as inhibitors of human lymphocyte proliferative activity. J Nat Prod. 1994 Nov; 57(11):1503-10. [CrossRef]
- García-Pérez P, Lozano-Milo E, Zhang L, Miras-Moreno B, Landin M, Lucini L, Gallego PP. Neurofuzzy logic predicts a fine-tuning metabolic reprogramming on elicited Bryophyllum PCSCs guided by salicylic acid. Front Plant Sci. 2022 Sep 23; 13:991557. [CrossRef]
- Bogucka-Kocka A, Zidorn C, Kasprzycka M, Szymczak G et al. Phenolic acid content, antioxidant and cytotoxic activities of four Kalanchoe species. Saudi J Biol Sci 2018; 25(4):622-630. [CrossRef]
- Gaind KN, Gupta RL. Phenolic components from the leaves of Kalanchoe pinnata. Planta Med. 1973 Mar;23(2):149-53. [CrossRef]
- Siddiqui S, Faizi S, Siddiqui BS, Sultana N (1989) Triterpenoids and phenanthrenes from leaves of Bryophyllum pinnatum. Phytochemistry 28(9):2433-2438. [CrossRef]
- Kruk J, Pisarski A, Szymanska R (2011) Novel vitamin E forms in leaves of Kalanchoe daigremontiana and Phaseolus coccineus. J Plant Physiol 168:2021-2027. [CrossRef]
- Siems K, Jas G, Arriaga-Ginerb FJ, Wollenweber E et al. (1995) On the chemical nature of epicuticular waxes in some succulent Kalanchoe and Senecio species. Zeitschrift für Naturforschung C 50:451-454.
- Van Maarseveen C, Jetter R (2009) Composition of the Epicuticular and Intracuticular Wax Layers On Kalanchoe daigremontiana (Hamet Et Perr. De La Bathie) Leaves. Phytochemistry 70(7):899-906. [CrossRef]
- Sharker SM, Hossain KM, Haque MR et al. Phytochemical and pharmacological studies of Bryophyllum daigremontianum (Raym.). American Journal of PharmTech Research. Vol 3, Issue 3, Pages 484-492, 2013.
- Aldalbahi A, Alterary S, Almoghim RAA, Awad MA et al. Greener Synthesis of Zinc Oxide Nanoparticles: Characterization and Multifaceted Applications. Molecules 2020; 25(18):4198. [CrossRef]
- Wang CY, Huang SC, Zhang Y et al. Antiviral Ability of Kalanchoe gracilis Leaf Extract against Enterovirus 71 and Coxsackievirus A16. Evid Based Complement Alternat Med. 2012; 2012:503165. [CrossRef]
- Nguelefack TB, Nana P, Atsamo AD, Dimo T, Watcho P, Dongmo AB, Tapondjou LA, Njamen D, Wansi SL, Kamanyi A. Analgesic and anticonvulsant effects of extracts from the leaves of Kalanchoe crenata (Andrews) Haworth (Crassulaceae). J Ethnopharmacol. 2006 Jun 15;106(1):70-5. Epub 2006 Jan 19. PMID: 16423479. [CrossRef]
- Sánchez-Martínez JD, Valdés A, Gallego R, Suárez-Montenegro ZJ, Alarcón M, Ibañez E, Alvarez-Rivera G, Cifuentes A. Blood-Brain Barrier Permeability Study of Potential Neuroprotective Compounds Recovered from Plants and Agri-Food by-Products. Front Nutr. 2022 Jun 16; 9:924596. [CrossRef]
- Viršile A, Samuoliene G, Laužike K, Šipailaite E, Balion Z, Jekabsone A. Species-Specific Plant-Derived Nanoparticle Characteristics. Plants 2022, 11, 3139. [CrossRef]
- Stefanowicz-Hajduk, J.; Hering, A.; Gucwa, M.; Sztormowska-Achranowicz, K.; Kowalczyk, M.; Soluch, A.; Ochocka, J.R. An In Vitro Anticancer, Antioxidant, and Phytochemical Study on Water Extract of Kalanchoe daigremontiana Raym.-Hamet and H. Perrier. Molecules 2022, 27, 2280. [CrossRef]
- Hsieh YJ, Huang HS, Leu YL, Peng KC et al. (2015) Anticancer activity of Kalanchoe tubiflora extract against human lung cancer cells in vitro and in vivo. Environ Toxicol 31(11):1663-1673. [CrossRef]
- Almeida AP, Da Silva SA, Souza ML, Lima LM, Rossi-Bergmann B, de Moraes VL, Costa SS. Isolation and chemical analysis of a fatty acid fraction of Kalanchoe pinnata with a potent lymphocyte suppressive activity. Planta Med. 2000 Mar;66(2):134-7. [CrossRef]
- Adam M, Elhassan GOM, Yagi S, Senol FS et al. In Vitro Antioxidant and Cytotoxic Activities of 18 Plants from the Erkowit Region, Eastern Sudan. Nat Prod Bioprospecting 2018; 8:97-105. [CrossRef]
- Ferreira AC, Rosenthal D, Carvalho DP. Thyroid peroxidase inhibition by Kalanchoe brasiliensis aqueous extract. Food Chem Toxicol. 2000 May;38(5):417-21. [CrossRef]
- Costa SS, de Souza Mde L, Ibrahim T, de Melo GO, de Almeida AP, Guette C, Férézou JP, Koatz VL. Kalanchosine dimalate, an anti-inflammatory salt from Kalanchoe brasiliensis. J Nat Prod. 2006 May;69(5):815-8. [CrossRef]
- Ibrahim T, Cunha JM, Madi K, da Fonseca LM, Costa SS, Gonçalves Koatz VL. Immunomodulatory and anti-inflammatory effects of Kalanchoe brasiliensis. Int Immunopharmacol. 2002 Jun;2(7):875-83. [CrossRef]
- Mayorga OAS, da Costa YFG, da Silva JB, Scio E et al. (2019) Kalanchoe brasiliensis Cambess., a Promising Natural Source of Antioxidant and Antibiotic Agents against Multidrug-Resistant Pathogens for the Treatment of Salmonella Gastroenteritis. Oxid Med Cell Longev. [CrossRef]
- Hewagama SP, Hewawasam RP. Antiurolithiatic Potential of Three Sri Lankan Medicinal Plants by the Inhibition of Nucleation, Growth, and Aggregation of Calcium Oxalate Crystals In Vitro. Scientific World Journal. 2022 Apr 12; 2022:8657249. [CrossRef]
- Kenderson CA, Kagoro ML, Adelakun EA (2021) Phytochemical and Pharmacological Evaluation of Nigerian Kalanchoe pinnata (Lam.) Stem-Bark. J Chem Soc Nigeria 46(4):0751-0756. [CrossRef]
- Cruz EA, Da-Silva SA, Muzitano MF, Silva PM, Costa SS, Rossi-Bergmann B. Immunomodulatory pretreatment with Kalanchoe pinnata extract and its quercitrin flavonoid effectively protects mice against fatal anaphylactic shock. Int Immunopharmacol. 2008 Dec 10;8(12):1616-21. Epub 2008 Aug 13. Epub 2008 Aug 13. PMID: 18675940. [CrossRef]
- Yadav NP, Dixit VK. Hepatoprotective activity of leaves of Kalanchoe pinnata Pers. J Ethnopharmacol. 2003 Jun;86(2-3):197-202. [CrossRef]
- de Araújo ERD, Guerra GCB, Andrade AWL, Fernandes JM, Da Silva VC, De Aragão Tavares E, De Araújo AA, de Araújo Júnior RF and Zucolotto SM (2021) Gastric Ulcer Healing Property of Bryophyllum pinnatum Leaf Extract in Chronic Model In Vivo and Gastroprotective Activity of Its Major Flavonoid. Front. Pharmacol. 12:744192. [CrossRef]
- de Araújo ERD, Xavier-Santos JB, da Silva VC, de Lima JBF, Schlamb J, Fernandes-Pedrosa MdF, da Silva Júnior AA, de Araújo Júnior RF, Rathinasabapathy T, Moncada M, Esposito D, Guerra GCB and Zucolotto SM (2023), Gel formulated with Bryophyllum pinnatum leaf extract promotes skin wound healing in vivo by increasing VEGF expression: A novel potential active ingredient for pharmaceuticals. Front. Pharmacol. 13:1104705. [CrossRef]
- Nayak BS, Marshall JR, Isitor G. Wound healing potential of ethanolic extract of Kalanchoe pinnata Lam. leaf--a preliminary study. Indian J Exp Biol. 2010 Jun;48(6):572-6. PMID: 20882759.
- Torres-Santos EC, Da Silva SA, Costa SS, Santos AP, Almeida AP, Rossi-Bergmann B. Toxicological analysis, and effectiveness of oral Kalanchoe pinnata on a human case of cutaneous leishmaniasis. Phytother Res. 2003 Aug;17(7):801-3. [CrossRef]
- Phatak RS (2016) Lack of anthelmintic activity of Kalanchoe pinnata fresh leaves. J Pharm Negat Results 7:21-24. [CrossRef]
- Ramon, P.; Bergmann, D.; Abdulla, H.; Sparks, J.; Omoruyi, F. Bioactive Ingredients in K. pinnata Extract and Synergistic Effects of Combined K. pinnata and Metformin Preparations on Antioxidant Activities in Diabetic and Non-Diabetic Skeletal Muscle Cells. Int. J. Mol. Sci. 2023, 24, 6211. 2023; 24. [Google Scholar] [CrossRef]
- Saravanan V, Murugan SS, Kumaravel TS. (2020) Genotoxicity studies with an ethanolic extract of Kalanchoe pinnata leaves. Mutat Res Gen Tox En 856-857: 503229. [CrossRef]
- Mahata, S., Maru, S., Shukla, S. et al. Anticancer property of Bryophyllum pinnata (Lam.) Oken. leaf on human cervical cancer cells. BMC Complement Altern Med 12, 15 (2012). [CrossRef]
- Hernández-Caballero, ME; Sierra-Ramírez, JA; Valencia, RV; Seseña-Méndez, E. Potential of Kalanchoe pinnata as a Cancer Treatment Adjuvant and an Epigenetic Regulator. Molecules 2022, 27, 6425. [CrossRef]
- Plangger N, Rist L, Zimmermann R, von Mandach U. Intravenous tocolysis with Bryophyllum pinnatum is better tolerated than beta-agonist application. Eur J Obstet Gynecol Reprod Biol. 2006 Feb 1;124(2):168-72. Epub 2005 Jul 26. [CrossRef]
- Gwehenberger B, Rist L, Huch R, von Mandach U. Effect of Bryophyllum pinnatum versus fenoterol on uterine contractility. Eur J Obstet Gynecol Reprod Biol. 2004 Apr 15;113(2):164-71. [CrossRef]
- Sazhina NN, Lapshin PV, Zagoskina NV, Korotkova EI et al. (2014) A Comparative Analysis of the Antioxidant Activity of Kalanchoe Juices. Russ J Bioorg Chem 40(7):771-776. [CrossRef]
- Lambrigger-Steiner C, Simões-Wüst AP, Kuck A, Fürer K, Hamburger M, von Mandach U. Sleep quality in pregnancy during treatment with Bryophyllum pinnatum: an observational study. Phytomedicine. 2014 Apr 15;21(5):753-7. Epub 2013 Dec 25. [CrossRef]
- Simões-Wüst AP, Hassani TA, Müller-Hübenthal B, Pittl S, Kuck A, Meden H, Eberhard J, Decker M, Fürer K, von Mandach U; Bryophyllum Collaborative Group. Sleep Quality Improves During Treatment with Bryophyllum pinnatum: An Observational Study on Cancer Patients. Integr Cancer Ther. 2015 Sep;14(5):452-9. Epub 2015 Apr 14. PMID: 25873294; PMCID: PMC4538317. [CrossRef]
- Pereira KMF, de Carvalho AC, Veiga TAM, Melgoza A, Hernandez RB, Grecco SS, et al. (2022) The psychoactive effects of Bryophyllum pinnatum (Lam.) Oken leaves in young zebrafish. PLoS ONE 17(3): e0264987. [CrossRef]
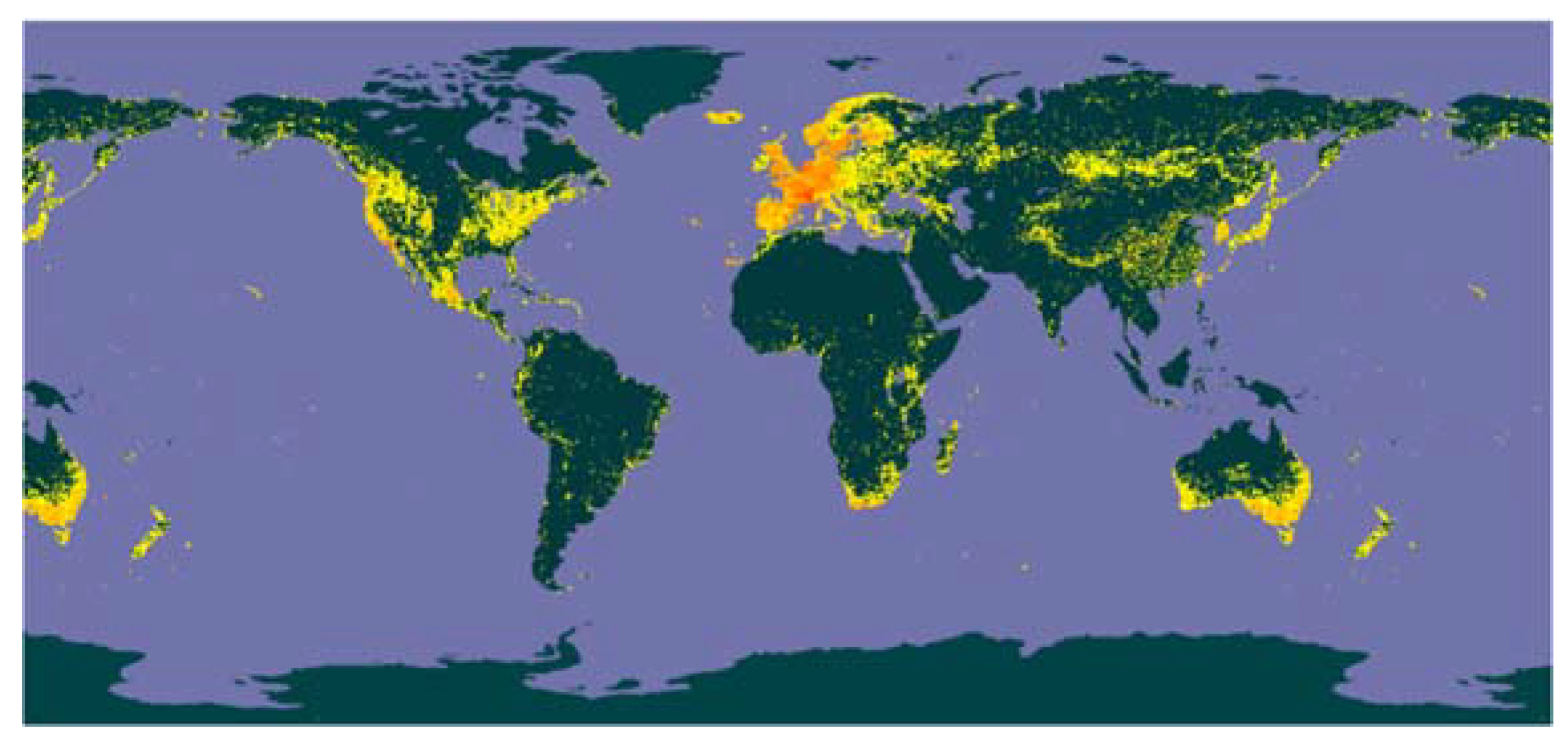
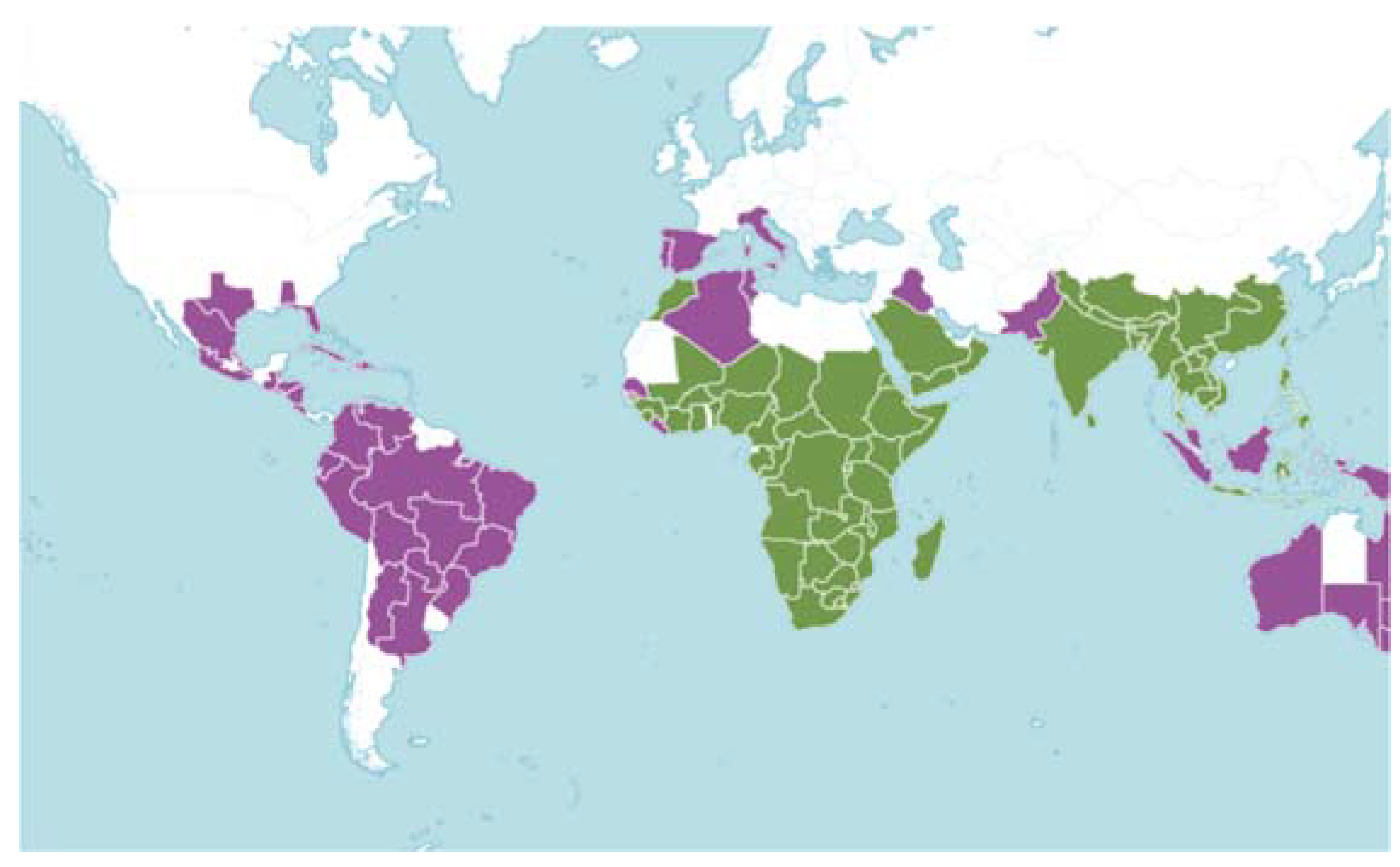
| Scientific name | Synonym | Occurrences |
|---|---|---|
| Kalanchoe adelae Raym.-Hamet | Bryophyllum adelae (Raym.-Hamet) A.Berger, Kalanchoe floribunda Tul. | 2 |
| Kalanchoe aliciae Raym.-Hamet | Bryophyllum aliciae (Raym.-Hamet) A.Berger, Kalanchoe miniata var. tsinjoarivensis H.Perrier, Kalanchoe pubescens var. brevicalyx Boiteau & Mannoni, Kalanchoe pubescens var. grandiflora Boiteau & Mannoni | 4 |
| Kalanchoe alternans (Vahl) Pers. | Cotyledon alternans Vahl, Vereia alternans (Vahl) Spreng. | 56 |
| Kalanchoe alticola Compton | - | 5 |
| Kalanchoe ambolensis Humbert | - | 10 |
| Kalanchoe angolensis N.E.Br. | - | 1 |
| Kalanchoe antennifera Desc. | - | 2 |
| Kalanchoe arborescens Humbert | - | 43 |
| Kalanchoe aromatica H.Perrier | - | 27 |
| Kalanchoe aubrevillei Raym.-Hamet ex Cufod. | - | 12 |
| Kalanchoe × auriculata (Raadts) V.V.Byalt | Kalanchoe nyikae subsp. auriculata Raadts | 47 |
| Kalanchoe ballyi Raym.-Hamet ex Cufod. | - | 27 |
| Kalanchoe beauverdii Raym.-Hamet | Bryophyllum beauverdii (Raym.-Hamet) A.Berger, Kalanchoe beauverdii var. typica Boiteau & Mannoni | 152 |
| Kalanchoe beharensis Drake | Kalanchoe vantieghemii Raym.-Hamet | 386 |
| Kalanchoe benbothae Gideon F.Sm. & N.R.Crouch | - | 6 |
| Kalanchoe bentii C.H.Wright ex Hook f. | - | 37 |
| Kalanchoe berevoensis Rebmann | - | - |
| Kalanchoe bergeri Raym.-Hamet & H.Perrier | Bryophyllum bergeri (Raym.-Hamet & H.Perrier) Govaerts, Kalanchoe bergeri var. typica Boiteau & Mannoni, Kalanchoe bergeri var. glabra Boiteau & Mannoni | 27 |
| Kalanchoe bhidei T.Cooke | - | 16 |
| Kalanchoe bipartita Chiov. | - | 14 |
| Kalanchoe blossfeldiana Poelln. | Kalanchoe coccinea (H.Perrier) Boiteau, Kalanchoe coccinea var. blossfeldiana (Poelln.) Boiteau, Kalanchoe globulifera var. coccinea H.Perrier | 493 |
| Kalanchoe bogneri Rauh | Bryophyllum bogneri (Rauh) V.V.Byalt | 10 |
| Kalanchoe boisii Raym.-Hamet & H.Perrier | - | 2 |
| Kalanchoe boranae Raadts | - | 10 |
| Kalanchoe bouvetii Raym.-Hamet & H.Perrier | Bryophyllum bouvetii (Raym.-Hamet & H.Perrier) A.Berger | 16 |
| Kalanchoe bouvetii Raym.-Hamet & H.Perrier | Kalanchoe baumii Engl. & Gilg, Kalanchoe multiflora Schinz, Kalanchoe pruinosa Dinter, Kalanchoe pyramidalis Schönland | 16 |
| Kalanchoe bracteata Scott Elliot | Kalanchoe bracteata var. aurantiaca Rauh & Hebding, Kalanchoe bracteata var. glabra Rauh & Hebding, Kalanchoe bracteata subsp. glabra Rauh & Hebding, Kalanchoe bracteata var. longisepala Boiteau ex L.Allorge, Kalanchoe bracteata var. pubescens Rauh & Hebding, Kalanchoe bracteata var. virescens Desc., Kalanchoe nadyae Raym.-Hamet | 88 |
| Kalanchoe brevicalyx (Raym.-Hamet & H.Perrier) Gideon F.Sm. & Figueiredo | Kalanchoe pinnata var. brevicalyx Raym.-Hamet & H.Perrier | 1 |
| Kalanchoe briquetii Raym.-Hamet | - | 2 |
| Kalanchoe campanulata (Baker) Baill. | Bryophyllum campanulatum (Baker) V.V.Byalt, Udalova & I.M.Vassiljeva, Kitchingia campanulata Baker, Kalanchoe amplexicaulis (Baker) Baill., Kalanchoe campanulata subsp. orthostyla Boiteau & Mannoni, Kalanchoe panduriformis (Baker) Baill., Kalanchoe parviflora (Baker) Baill., Kitchingia amplexicaulis Baker, Kitchingia panduriformis Baker, Kitchingia parviflora Baker | 80 |
| Kalanchoe ceratophylla Haw. | 111 | |
| Kalanchoe chapototii Raym.-Hamet & H.Perrier | 2 | |
| Kalanchoe cherukondensis Subba Rao & Kumari | Vereia ceratophylla (Haw.) D.Dietr. | - |
| Kalanchoe chevalieri Gagnep. | Kalanchoe integra var. chevalieri (Gagnep.) H.H.Pham | 9 |
| Kalanchoe citrina Schweinf. | Kalanchoe citrina var. ballyi Raym.-Hamet ex Wickens, Kalanchoe citrina var. erythreae Schweinf. | 102 |
| Kalanchoe costantinii Raym.-Hamet | Bryophyllum costantinii (Raym.-Hamet) A.Berger | 1 |
| Kalanchoe craibii Raym.-Hamet | - | 1 |
| Kalanchoe crenata (Andrews) Haw. | Cotyledon crenata (Andrews) Vent., Cotyledon verea Jacq., Kalanchoe afzeliana Britten, Kalanchoe crenata var. verea Cufod., Kalanchoe integra var. crenata (Andrews) Cufod., Kalanchoe integra var. varea Cufod., Kalanchoe verea Pers., Vereia crenata Andrews | 1320 |
| Kalanchoe crouchii Gideon F.Sm. & Figueiredo | - | 3 |
| Kalanchoe crundallii I.Verd. | - | 6 |
| Kalanchoe curvula Desc. | Bryophyllum curvulum (Desc.) V.V.Byalt | 16 |
| Kalanchoe cymbifolia Desc. | Bryophyllum cymbifolium (Desc.) V.V.Byalt | - |
| Kalanchoe daigremontiana Raym.-Hamet & H.Perrier | Bryophyllum daigremontianum (Raym.-Hamet & H.Perrier) A.Berger | 768 |
| Kalanchoe darainensis D.-P.Klein & Callm. | - | 22 |
| Kalanchoe decumbens Compton | - | - |
| Kalanchoe deficiens (Forssk.) Asch. & Schweinf. | Cotyledon deficiens Forssk., Kalanchoe glaucescens var. deficiens (Asch. & Schweinf.) Senni | 342 |
| Kalanchoe delagoensis Eckl. & Zeyh. | Bryophyllum delagoense (Eckl. & Zeyh.) Druce, Bryophyllum tubiflorum Harv., Kalanchoe tubiflora (Harv.) Raym.-Hamet, Bryophyllum verticillatum (Scott Elliot) A.Berger, Geaya purpurea Costantin & Poiss., Kalanchoe verticillata Scott Elliot | 5341 |
| Kalanchoe densiflora Rolfe | - | 687 |
| Kalanchoe × descoingsii Shtein, Gideon F.Sm. & J.Ikeda | - | - |
| Kalanchoe dinklagei Rauh | Kalanchoe brevisepala (Humbert) L.Allorge, Kalanchoe millotii var. brevisepala Humbert | 14 |
| Kalanchoe dyeri N.E.Br. | - | 21 |
| Kalanchoe elizae A.Berger | Cotyledon elizae (A.Berger) Raym.-Hamet, Cotyledon insignis N.E.Br., Kalanchoe insignis (N.E.Br.) N.E.Br., Kalanchoe laurensii Raym.-Hamet | 80 |
| Kalanchoe eriophylla Hils. & Bojer ex Tul. | Cotyledon pannosa Baker | 43 |
| Kalanchoe × estrelae Gideon F.Sm. | - | - |
| Kalanchoe fadeniorum Raadts | - | 8 |
| Kalanchoe farinacea Balf.f. | - | 55 |
| Kalanchoe faustii Font Quer | Kalanchoe laciniata subsp. faustii (Font Quer) Maire | 33 |
| Kalanchoe fedtschenkoi Raym.-Hamet & H.Perrier | Bryophyllum fedtschenkoi (Raym.-Hamet & H.Perrier) Lauz.-March., Kalanchoe fedtschenkoi var. isalensis Boiteau & Mannoni | 514 |
| Kalanchoe fernandesii Raym.-Hamet | - | 4 |
| Kalanchoe × flaurantia Desc. | - | - |
| Kalanchoe gastonis-bonnieri Raym.-Hamet & H.Perrier | Bryophyllum gastonis-bonnieri (Raym.-Hamet & H.Perrier) Lauz.-March., Kalanchoe adolphi-engleri Raym.-Hamet, Kalanchoe gastonis-bonnieri var. ankaizinensis Boiteau ex L.Allorge | 173 |
| Kalanchoe germanae Raym.-Hamet ex Raadts | - | 16 |
| Kalanchoe gideonsmithii N.R.Crouch & Figueiredo | - | 1 |
| Kalanchoe glaucescens Britten | Kalanchoe beniensis De Wild., Kalanchoe elliptica Raadts, Kalanchoe flammea Stapf, Kalanchoe holstii Engl., Kalanchoe magnidens N.E.Br., Kalanchoe marinellii Pamp., Kalanchoe ndorensis Schweinf. ex Engl. | 379 |
| Kalanchoe globulifera H.Perrier | - | 15 |
| Kalanchoe gracilipes (Baker) Baill. | Bryophyllum gracilipes (Baker) Eggli, Kitchingia gracilipes Baker | 89 |
| Kalanchoe grandidieri Baill. | - | 64 |
| Kalanchoe grandiflora Wight & Arn. | Vereia grandiflora (Wight & Arn.) D.Dietr. | 77 |
| Kalanchoe guignardii Raym.-Hamet & H.Perrier | Kalanchoe beauverdii var. guignardii (Raym.-Hamet & H.Perrier) Boiteau & Mannoni | 1 |
| Kalanchoe × gunniae Gideon F.Sm. & Figueiredo | - | - |
| Kalanchoe hametiorum Raym.-Hamet | - | 4 |
| Kalanchoe hauseri Werderm. | - | - |
| Kalanchoe hildebrandtii Baill. | Kalanchoe gomphophylla Baker, Kalanchoe hildebrandtii var. glabra Rauh & Hebding | 95 |
| Kalanchoe hirta Harv. | - | 13 |
| Kalanchoe × houghtonii D.B.Ward | Bryophyllum × houghtonii (D.B.Ward) P.I.Forst. | 1650 |
| Kalanchoe humifica Desc. | Bryophyllum humificum (Desc.) V.V.Byalt | 1 |
| Kalanchoe humilis Britten | - | 30 |
| Kalanchoe hypseloleuce Friis & M.G.Gilbert | - | 1 |
| Kalanchoe inaurata Desc. | Bryophyllum inauratum (Desc.) V.V.Byalt | - |
| Kalanchoe integra (Medik.) Kuntze | Cotyledon integra Medik., Bryophyllum serratum Blanco, Cotyledon acutiflora (Haw.) W.T.Aiton, Cotyledon hybrida Dum.Cours., Cotyledon spathulata (DC.) Poir., Echeveria spathulata (DC.) W.Bull ex É.Morren, Kalanchoe acutiflora (Andrews) Haw., Kalanchoe annamica Gagnep., Kalanchoe corymbosa Wall., Kalanchoe dixoniana Raym.-Hamet, Kalanchoe garambiensis Kudô, Kalanchoe hybrida Desf. ex Steud., Kalanchoe integra var. annamica (Gagnep.) H.H.Pham, Kalanchoe nudicaulis Buch.-Ham. ex C.B.Clarke, Kalanchoe schumacheri Koord., Kalanchoe spathulata DC., Kalanchoe spathulata var. annamica (Gagnep.) H.Ohba, Kalanchoe spathulata var. baguioensis H.Ohba, Kalanchoe spathulata var. ciliata, Kalanchoe spathulata var. dixoniana (Raym.-Hamet) H.Ohba, Kalanchoe spathulata var. garambiensis (Kudô) H.Ohba, Kalanchoe spathulata var. schumacheri (Koord.) H.Ohba, Kalanchoe spathulata var. simlensis H.Ohba, Kalanchoe spathulata var. staintonii H.Ohba, Kalanchoe subamplectens Wall., Kalanchoe varians Haw., Kalanchoe yunnanensis Gagnep., Vereia acutiflora Andrews, Vereia spathulata (DC.) D.Dietr. | 289 |
| Kalanchoe integrifolia Baker | Kalanchoe bitteri Raym.-Hamet & H.Perrier, Kalanchoe heckelii Raym.-Hamet & H.Perrier, Kalanchoe integrifolia var. bitteri Raym.-Hamet & H.Perrier, Kalanchoe integrifolia var. flava Boiteau | 85 |
| Kalanchoe jongmansii Raym.-Hamet & H.Perrier | Bryophyllum jongmansii (Raym.-Hamet & H.Perrier) Govaerts, Kalanchoe jongmansii subsp. ivohibensis Humbert | 54 |
| Kalanchoe klopperae Gideon F.Sm. & Figueiredo | - | - |
| Kalanchoe laciniata (L.) DC. | Cotyledon laciniata L., Vereia laciniata (L.) Willd., Kalanchoe angustifolia A.Rich., Kalanchoe biternata Wight ex Wall., Kalanchoe carnea N.E.Br., Kalanchoe gloveri Cufod., Kalanchoe lentiginosa Cufod., Kalanchoe petitiaesii Rich. ex Jacques, Kalanchoe rohlfsii Engl., Kalanchoe rosea A.Chev., Kalanchoe schweinfurthii Penz., Kalanchoe teretifolia Haw. | 430 |
| Kalanchoe laetivirens Desc. | Bryophyllum laetivirens (Desc.) V.V.Byalt | 223 |
| Kalanchoe lanceolata (Forssk.) Pers. | Cotyledon lanceolata Forssk., Vereia lanceolata (Forssk.) Spreng., Cotyledon amplexicaulis B.Heyne ex C.B.Clarke, Cotyledon corymbosa Rottler ex Wight & Arn., Cotyledon heterophylla Roxb., Cotyledon hirsuta B.Heyne ex C.B.Clarke, Cotyledon paniculata Rottler ex Wight & Arn., Kalanchoe amplexicaulis B.Heyne, Kalanchoe brachycalyx A.Rich., Kalanchoe crenata var. collina Engl., Kalanchoe ellacombei N.E.Br., Kalanchoe floribunda Wight & Arn., Kalanchoe floribunda var. glabra C.B.Clarke, Kalanchoe glandulosa Hochst. ex A.Rich., Kalanchoe glandulosa var. benguellensis Engl., Kalanchoe glandulosa var. rhodesica Baker f., Kalanchoe glandulosa var. tomentosa Keissl., Kalanchoe goetzei Engl., Kalanchoe gregaria Dinter, Kalanchoe heterophylla (Roxb.) Wight & Arn., Kalanchoe heterophylla (Roxb.) Prain, Kalanchoe homblei De Wild., Kalanchoe homblei f. reducta De Wild., Kalanchoe junodii Schinz, Kalanchoe laciniata var. brachycalyx (A.Rich.) Chiov., Kalanchoe lanceolata var. glabra (C.B.Clarke) S.R.Sriniv., Kalanchoe lanceolata var. glandulosa (Hochst. ex A.Rich.) Cufod., Kalanchoe modesta Kotschy & Peyr., Kalanchoe pentheri Schltr., Kalanchoe pilosa Baker, Kalanchoe platysepala Welw. ex Britten, Kalanchoe pubescens R.Br. ex Britten, Kalanchoe ritchieana Dalzell, Kalanchoe spathulata Wall., Kalanchoe wightianum Wall., Meristostylus macrocalyx Klotzsch, Vereia floribunda (Wight & Arn.) D.Dietr., Vereia heterophylla (Wight & Arn.) D.Dietr. | 893 |
| Kalanchoe lateritia Engl. | - | 284 |
| Kalanchoe latisepala N.E.Br. | - | 31 |
| Kalanchoe laxiflora Baker | Bryophyllum laxiflorum (Baker) Govaerts, Bryophyllum crenatum Baker, Kalanchoe crenata (Baker) Raym.-Hamet, Kalanchoe laxiflora subsp. stipitata Boiteau & Mannoni, Kalanchoe laxiflora subsp. subpeltata Boiteau & Mannoni, Kalanchoe laxiflora subsp. violacea Boiteau & Mannoni, Kalanchoe tieghemii Raym.-Hamet | 469 |
| Kalanchoe leblanciae Raym.-Hamet | - | 17 |
| Kalanchoe lindmanii Raym.-Hamet | Kalanchoe gossweileri Croizat, Kalanchoe humbertii Guillaumin, Kalanchoe pearsonii N.E.Br. | 16 |
| Kalanchoe linearifolia Drake | Kalanchoe bonnieri Raym.-Hamet | 118 |
| Kalanchoe lobata R.Fern. | - | 6 |
| Kalanchoe × lokarana Desc. | Bryophyllum × lokarana (Desc.) V.V.Byalt | 2 |
| Kalanchoe longiflora Schltr. | - | 35 |
| Kalanchoe longifolia E.T.Geddes | - | 2 |
| Kalanchoe lubangensis R.Fern. | - | 1 |
| Kalanchoe luciae Raym.-Hamet | Kalanchoe albiflora H.M.L.Forbes | 60 |
| Kalanchoe macrochlamys H.Perrier | Bryophyllum macrochlamys (H.Perrier) A.Berger | 12 |
| Kalanchoe mandrarensis Humbert | - | 7 |
| Kalanchoe manginii Raym.-Hamet & H.Perrier | Bryophyllum manginii (Raym.-Hamet & H.Perrier) Nothdurft | 67 |
| Kalanchoe marmorata Baker | Kalanchoe grandiflora A.Rich., Kalanchoe macrantha Baker ex Maire, Kalanchoe macrantha var. marmorata (Baker) Maire, Kalanchoe macrantha var. richardiana Maire | 264 |
| Kalanchoe marnieriana H.Jacobsen ex L.Allorge | Bryophyllum marnierianum (H.Jacobsen ex L.Allorge) Govaerts, Kalanchoe humbertii Mannoni & Boiteau | 46 |
| Kalanchoe maromokotrensis Desc. & Rebmann | - | 5 |
| Kalanchoe migiurtinorum Cufod. | - | 7 |
| Kalanchoe millotii Raym.-Hamet & H.Perrier | - | 82 |
| Kalanchoe miniata Hils. & Bojer ex Tul. | Bryophyllum miniatum (Hils. & Bojer ex Tul.) A.Berger, Kalanchoe miniata var. typica H.Perrier, Kitchingia miniata (Hils. & Bojer ex Tul.) Baker | 252 |
| Kalanchoe mitejea Leblanc & Raym.-Hamet | - | 29 |
| Kalanchoe montana Compton | Kalanchoe luciae subsp. montana (Compton) Toelken | 2 |
| Kalanchoe mortagei Raym.-Hamet & H.Perrier | Bryophyllum mortagei (Raym.-Hamet & H.Perrier) Wickens, Kalanchoe poincarei var. mortagei (Raym.-Hamet & H.Perrier) Boiteau | 42 |
| Kalanchoe ndotoensis L.E.Newton | - | 1 |
| Kalanchoe neglecta Toelken | Kalanchoe rotundifolia f. peltata R.Fern. | 7 |
| Kalanchoe nyikae Engl. | Kalanchoe hemsleyana Cufod. | 53 |
| Kalanchoe obtusa Engl. | - | 39 |
| Kalanchoe olivacea Dalzell | - | 10 |
| Kalanchoe orgyalis Baker | Kalanchoe antanosiana Drake | 162 |
| Kalanchoe paniculata Harv. | Sedum harveyanum Kuntze, Kalanchoe oblongifolia Harv. | 193 |
| Kalanchoe pareikiana Desc. & Lavranos | - | 2 |
| Kalanchoe peltata (Baker) Baill. | Bryophyllum peltatum (Baker) V.V.Byalt, Udalova & I.M.Vassiljeva, Kitchingia peltata Baker | 152 |
| Kalanchoe peltigera Desc. | Bryophyllum peltigerum (Desc.) V.V.Byalt | 5 |
| Kalanchoe perrieri Shtein, Gideon F.Sm. & D.-P.Klein | - | - |
| Kalanchoe peteri Werderm. | - | 35 |
| Kalanchoe petitiana A.Rich. | - | 95 |
| Kalanchoe pinnata (Lam.) Pers. | Bryophyllum pinnatum (Lam.) Oken, Cotyledon pinnata Lam., Crassula pinnata (Lam.) L.f., Kalanchoe pinnata var. genuina Raym.-Hamet, Vereia pinnata (Lam.) Spreng., Baumgartenia sobolifera Tratt., Bryophyllum calcicola (H.Perrier) V.V.Byalt, Bryophyllum calycinum Salisb., Bryophyllum germinans Blanco, Bryophyllum pinnatum simplicifolium Kuntze, Cotyledon calycina (Salisb.) B.Heyne, Cotyledon calyculata Sol. ex Sims, Cotyledon rhizophylla Roxb., Crassuvia floripendia Comm. ex Lam., Kalanchoe calcicola (H.Perrier) Boiteau, Kalanchoe floripendula Steud, Kalanchoe pinnata var. calcicola H.Perrier, Kalanchoe pinnata var. floripendula Pers. | 7288 |
| Kalanchoe × poincarei Raym.-Hamet & H.Perrier | Bryophyllum poincarei (Raym.-Hamet & H.Perrier) Govaerts | 10 |
| Kalanchoe porphyrocalyx (Baker) Baill. | Bryophyllum porphyrocalyx (Baker) A.Berger, Kalanchoe porphyrocalyx var. typica Boiteau & Mannoni, Kitchingia porphyrocalyx Baker | 187 |
| Kalanchoe prasina N.E.Br. | Kalanchoe figuereidoi Croizat | - |
| Kalanchoe prittwitzii Engl. | Kalanchoe dielsii Raym.-Hamet, Kalanchoe lugardii Bullock, Kalanchoe robynsiana Raym.-Hamet, Kalanchoe secunda Werderm. | 136 |
| Kalanchoe prolifera (Bowie ex Hook.) Raym.-Hamet | Bryophyllum proliferum Bowie ex Hook., Bryophyllum cochleatum Lem., Kalanchoe cochleatum (Lem.) B.D.Jacks. | 180 |
| Kalanchoe pseudocampanulata Mannoni & Boiteau | Bryophyllum pseudocampanulatum (Mannoni & Boiteau) Govaerts, Kalanchoe miniata var. decaryana H.Perrier | 5 |
| Kalanchoe pubescens Baker | Bryophyllum pubescens (Baker) Govaerts, Kalanchoe pubescens var. typica Boiteau & Mannoni | 162 |
| Kalanchoe pumila Baker | Kalanchoe brevicaulis Baker, Kalanchoe multiceps Baill., Kalanchoe pumila f. venustior Boiteau | 71 |
| Kalanchoe quadrangularis Desc. | - | 3 |
| Kalanchoe quartiniana A.Rich. | - | 23 |
| Kalanchoe rebmannii Desc. | - | 1 |
| Kalanchoe × rechingeri Raym.-Hamet ex Rauh & Hebding | Bryophyllum × rechingeri (Raym.-Hamet ex Rauh & Hebding) V.V.Byalt | 2 |
| Kalanchoe rhombopilosa Mannoni & Boiteau | Kalanchoe rhombopilosa var. argentea Rauh, Kalanchoe rhombopilosa var. viridifolia Rauh | 30 |
| Kalanchoe × richaudii Desc. | - | 2 |
| Kalanchoe robusta Balf.f. | Kalanchoe abrupta Balf.f. | 7 |
| Kalanchoe rolandi-bonapartei Raym.-Hamet & H.Perrier | Bryophyllum rolandi-bonapartei (Raym.-Hamet & H.Perrier) Govaerts, Bryophyllum tsaratananense (H.Perrier) A.Berger, Kalanchoe tsaratananensis H.Perrier | 16 |
| Kalanchoe rosea C.B.Clarke | - | - |
| Kalanchoe rosei Raym.-Hamet & H.Perrier | Bryophyllum rosei (Raym.-Hamet & H.Perrier) A.Berger, Kalanchoe bouvieri Raym.-Hamet & H.Perrier | 74 |
| Kalanchoe rotundifolia (Haw.) Haw. | Crassula rotundifolia Haw., Sedum subrotundifolium (Haw.) Kuntze, Vereia rotundifolia (Haw.) D.Dietr., Kalanchoe guillauminii Raym.-Hamet, Kalanchoe integerrima Lange, Kalanchoe luebbertiana Engl., Kalanchoe rotundifolia var. guillauminii (Raym.-Hamet) Raym.-Hamet, Kalanchoe rotundifolia f. tripartita R.Fern., Kalanchoe seilleana Raym.-Hamet, Kalanchoe stearnii Raym.-Hamet, Meristostylus brachycalyx Klotzsch | 623 |
| Kalanchoe rubella (Baker) Raym.-Hamet | Bryophyllum rubellum Baker | 23 |
| Kalanchoe salazarii Raym.-Hamet | - | 2 |
| Kalanchoe sanctula Desc. | Bryophyllum sanctulum (Desc.) V.V.Byalt | 2 |
| Kalanchoe scandens H.Perrier | Bryophyllum scandens (H.Perrier) A.Berger, Kalanchoe beauverdii var. parviflora Boiteau & Mannoni | 7 |
| Kalanchoe scapigera Welw. ex Britten | - | 15 |
| Kalanchoe schimperiana A.Rich. | Cotyledon deficiens Hochst. & Steud. ex A.Rich. | 78 |
| Kalanchoe schizophylla (Baker) Baill. | Bryophyllum schizophyllum (Baker) A.Berger, Kitchingia schizophylla Baker | 48 |
| Kalanchoe schliebenii Werderm. | - | 3 |
| Kalanchoe serrata Mannoni & Boiteau | Bryophyllum lauzac-marchaliae V.V.Byalt, Bryophyllum serratum (Mannoni & Boiteau) Lauz.-March. | 36 |
| Kalanchoe sexangularis N.E.Br. | - | 130 |
| Kalanchoe stenosiphon Britten | - | 9 |
| Kalanchoe streptantha Baker | Bryophyllum streptanthum (Baker) A.Berger | 28 |
| Kalanchoe suarezensis H.Perrier | Bryophyllum suarezense (H.Perrier) A.Berger, Kalanchoe poincarei var. suarezensis (H.Perrier) L.Allorge | 20 |
| Kalanchoe subrosulata Thulin | - | 4 |
| Kalanchoe synsepala Baker | Kalanchoe brachycalyx Baker, Kalanchoe gentyi Raym.-Hamet & H.Perrier, Kalanchoe trichantha Baker | 214 |
| Kalanchoe tachingshuii S.S.Ying | - | - |
| Kalanchoe tashiroi Yamam. | - | 4 |
| Kalanchoe teixeirae Raym.-Hamet ex R.Fern. | - | 3 |
| Kalanchoe tenuiflora Desc. | - | 3 |
| Kalanchoe tetramera E.T.Geddes | - | 2 |
| Kalanchoe tetraphylla H.Perrier | - | 30 |
| Kalanchoe thyrsiflora Harv. | Kalanchoe alternans Eckl. & Zeyh. ex Harv. | 251 |
| Kalanchoe tomentosa Baker | Bryophyllum triangulare Blanco | 179 |
| Kalanchoe torrejacqii Shtein & Gideon F.Sm. | - | 3 |
| Kalanchoe tuberosa H.Perrier | - | 11 |
| Kalanchoe uniflora (Stapf) Raym.-Hamet | Bryophyllum uniflorum (Stapf) A.Berger, Kitchingia uniflora Stapf, Bryophyllum ambrense (H.Perrier) A.Berger, Kalanchoe ambrensis H.Perrier, Kalanchoe uniflora var. brachycalyx Boiteau & Mannoni | 97 |
| Kalanchoe usambarensis Engl. & Raym.-Hamet | - | 16 |
| Kalanchoe variifolia (Guillaumin & Humbert) Shtein, D.-P.Klein & Gideon F.Sm. | Kalanchoe rosei var. variifolia (Guillaumin & Humbert) J.M.H.Shaw, Kalanchoe rosei subsp. variifolia Guillaumin & Humbert | 13 |
| Kalanchoe velutina Welw. ex Britten | - | 56 |
| Kalanchoe viguieri Raym.-Hamet & H.Perrier | Kalanchoe viguieri var. latisepala Raym.-Hamet & H.Perrier | 68 |
| Kalanchoe waldheimii Raym.-Hamet & H.Perrier | Bryophyllum waldheimii (Raym.-Hamet & H.Perrier) Lauz.-March. | 50 |
| Kalanchoe waterbergensis van Jaarsv. | - | 3 |
| Kalanchoe welwitschii Britten | - | 17 |
| Kalanchoe wildii Raym.-Hamet ex R.Fern. | Kalanchoe aleuroides Stearn | 2 |
| Kalanchoe winteri Gideon F.Sm., N.R.Crouch & Mich.Walters | - | 3 |
| Kalanchoe yemensis (Deflers) Schweinf. | Kalanchoe brachycalyx var. yemensis Deflers | 17 |
| Species | Traditional uses | References |
|---|---|---|
| K. ceratophylla | To treat injuries, pain, fever, and inflammation | [14,15,16,17] |
| K. crenata | Antidiabetic, anti-inflammatory, antimicrobial, vermifuge and anti-infective agent to treat wounds, abscesses, abdominal pain, asthma, headache, convulsion, smallpox, peptic ulcer, upper respiratory tract infections, coughs, otitis, palpitations, cancer (or disease states with symptoms related to cancer), diabetes, swollen areas for muscle sprain and myalgia, and to heal umbilical cord wounds in newborns | [6,14,15,16,18,19,20,21,22,23,24,25,26,27] |
| K. daigremontiana | Anticancer, anti-inflammatory, antimicrobial, antiseptic, carminative and cardioactive agent, to treat skin injuries and wounds, to staunch bleeding, to treat infections, rheumatism, earache, burns, arthritis, gastric and menstrual disorders, cough, fever, cardiovascular dysfunction, diabetes, psychic agitation, restlessness and anxiety, some cancers, and as a chemo preventive | [9,10,11,12,15,21,28,29,30] |
| K. delagoensis | To treat wounds, epilepsy, neoplastic diseases, fever, abscesses, bruises, pneumonia, coughs, stomachache and as a vermifuge | [14,15,21,32,33,34] |
| K. densiflora | To treat wounds and skin disorders, rheumatism, hemorrhoids, eye problems, joint and muscle pains, stomach and liver problems, umbilical cord, cardiac disorders, edema, poisonous, abortifacient | [15,24,27,35] |
| K. flammea | To treat fever, wounds, inflammation, and cancer | [36] |
| K. fedtschenkoi | Analgesic, cytotoxic, and antimicrobial treatments | [21,37] |
| K. gastonis-bonnieri | To treat genital-urinary and vaginal infections and as a vaginal contraceptive | [38] |
| K. germanae | After removal of ganglions the leaves are used to treat the affected area | [15] |
| K. glaucescens | To treat coughs and rheumatism | [15,24] |
| K. integra | Antihypertensive | [39] |
| K. laciniata | As an anti-inflammatory, astringent, and antiseptic. To treat wounds, inflammation, headache, diabetes, heart discomfort, gastric disorders, lithiasis, diarrhea, fever, cough, snakebites, erysipelas, boils, and human prostate cancer | [6,11,15,40,41,42,43,44,45,46,47,48,49] |
| K. lanceolata | To treat dysentery, rheumatism, hemorrhoids, splenomegaly, hepatomegaly, and pains | [15,24,27,35] |
| K. marmorata | To treat wounds, boils, bruises, periodontal disease, cracked lips, arthritis, gastric ulcers, ear diseases, eye infections, dysentery, fever, common cold, coughs, cholera, urinary diseases, stiff muscles, liver problems, and headaches. | [15,24,50,51,52] |
| K. mortagei | As an antimicrobial, to treat digestive disorders, parasites, and neoplastic diseases orally, and as a local remedy for cancer | [21,37] |
| K. obtusa | Children’s diseases and as pesticide | [24] |
| K. petitiana | To treat epilepsy, trachoma, allergies, intestinal parasites, gonorrhea, bone setting after fractures, wound healing, breast tumors, skin cancer, swelling of gland/lymph adenitis, toothache, dysentery, liver problems, stomachache, tonsillitis, gastritis, peptic ulcer disease, and foot problems (fungal nails, corns, and calluses, athlete’s foot, plantar warts). | [15,53,54,55,56] |
| K. pinnata | Antipyretic, antibacterial, antiseptic, antimalaria, anti-inflammatory, and antipsychotic agent. To treat the following: wounds; burns; cardiovascular dysfunctions; cancer; rheumatoid arthritis; digestive, menstrual and psychiatric disorders; hypertension; skin, respiratory and genitourinary infections; kidney, liver and urinary disorders; ear, head and toothache; insect, snake and scorpion bites; muscle bruises; cholera; leishmania; leprosy; lithiasis; viruses; restlessness; biostimulator during skin transplantation; to prevent premature labor and help women recover after childbirth; diabetes; cold, whooping cough, bone fracture, Chikungunya virus, and against COVID-19 symptoms | [6,8,14,15,21,41,42,43,44,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94] |
| K. prittwitzii | Stiff joints and rheumatism | [24] |
| K. serrata | To treat pain, inflammation, fever, and viruses | [21] |
| K. x houghtonii | To treat infections, rheumatism, coughs, fever, and inflammation | [30] |
| Species | Macro aspects | Micro aspects | References |
|---|---|---|---|
| K. beharensis | The largest species of the genus, with 3 m in height; unbranched stems; leaves crowded at the branch tips; lobed, covered in a dense felt; ranging from 12–35 cm in length and 7–35 cm in width. | [6] | |
| K. blossfeldiana | Dark green, succulent, and perennial plant, with scallop-edged leaves and large umbels of flower clusters held above the foliage. The fleshy, dark shiny green leaves have lobed edges and can reach 7.7 cm in length and 3.8 cm in width. Floral colors range from traditional red to yellow, orange, salmon and pink. | [105] | |
| K. daigremontiana | Perennial short-lived succulent herb; monocarpic multi-annuals. The most characteristic feature of the species is its method of asexual reproduction by auto-propagation. Flowering tends to be sporadic, in winter, and, when it occurs, the main stalk elongates vertically, developing a terminal inflorescence of small, bell-shaped, pendulous flowers with a pinkish or purple corolla. The stem is unbranched, up to 1.5 m in height. The leaves are thick, fleshy, lanceolate, tapered at the apex and serrated in the margins, dark green colored, and have purple-brown spots on the abaxial side. The apex bears hydathodes and adventitious buds, from which propagules are formed and developed. | The epidermis is single-layered, with parenchymatic cells; convex outer walls surface; wax patches in cuticles; is smooth-undulating and striated only on subsidiary cells. The leaves are amphistomatic, with anisocytic stomata. The subepidermal mesophyll consists of one or several layers of small, closely adherent cells. The central vascular bundles are surrounded by perivascular sheaths composed of mesophyll cells. Between the epidermis and mesophyll in the petioles there are 1-3 layers of compact angular collenchyma. The vascular bundles are collateral. In the central veins in the petiole and the leaf are three large bundles. The cross-sections show fine lateral vascular bundles surrounding large bundles in the petioles and leaf blades. The different tissues of the leaf contain numerous phenolic idioblasts, accumulating phenolic compounds in their vacuoles, present in epidermal cells, in the subepidermal layer, near the vascular elements, around the large vascular bundles in the leaf petioles, and surrounding the smaller vascular bundles; dispersed in the parenchyma as single cells or form multicellular aggregates. | [6,12,29,98,106,107] |
| K. delagoensis | It has dark purplish, speckled, tubular leaves, which are filled with plantlets. It typically grows to about 1 m in height before blooming. It overwinters as a terminal inflorescence bearing orange or red pendant bell-shaped flowers and then dies. | The leaves are tubular and have 6-8 apical buds. The epidermal cells are uniseriate with sinuous anticlinal walls. The leaves are amphistomatic with anisocytic stomata. The mesophyll has regular chlorenchyma. The vascular system has collateral bundles distributed in the form of an arc. Anthocyanin idioblasts occur throughout the leaf blade, in the epidermis; hypodermis; layer beneath the hypodermis; scattered in the chlorenchyma; surrounding the vascular bundles; vascular tissues; and apical buds. | [6,108] |
| K. ceratophylla | Perennial, succulent, and glabrous species. | [16] | |
| K. laciniata | Perennial or biennial herb that grows from 30 cm to 1.5 m in height. Its leaves are oval, opposite, fleshy, simple, short-petiolate, glossy, and pale green to dark green in color. They have dentate to crenate leaf margins, with a cylindrical herbaceous stem and fleshy petiole. | The secretory structures found in the stems, petioles and leaf blades consist of idioblasts that contain anthocyanins. The epidermis of K. laciniata is a single layer with adhering and oblong cells. The outer cell wall is convex and covered with cuticles. The leaves are amphistomatic and the chlorenchyma tissue is uniform. The cells of the chlorenchyma tissue have irregular, spherical-ellipsoidal shapes. The vacuoles of some mesophyll cells located near the epidermis, vascular bundles, and hydathodes contain phenolic compounds. The leaves show the presence of adaptive traits that enable them to survive in dry environments | [42,44,109] |
| K. laxiflora | Perennial species: with multicolored leaves, that are crenate, green in shady settings and pink or purple in bright sun. The flower buds are almost transparent but when they open, they turn orange. | [6] | |
| K. marmorata | The leaves are large, oval, blue-green colored, with purple markings, arranged in stacked, opposite pairs to a height of 30 cm. The brown spots become brighter during summer dormancy and in strong sunlight; and during winter they become pale or disappear altogether. | [3,6] | |
| K. orgyalis | It is a much-branched slow-growing shrub that can reach from approximately 1-2 m in height. It has spoon-shaped leaves, which are bronze to gray on the underside, and felted on the top of each leaf, with cinnamon-toned fuzz. Late winter or early spring brings bright yellow flowers in terminal clusters at the branch tips. | [6] | |
| K. pinnata | An erect, succulent, perennial and glabrous plant that grows up to 1.5 m in height. The species reproduces through seeds and from leaf bulbils. The freshly dark green leaves are large (12-18 cm and 6-8 cm in size), simple, opposite, ovate or elliptic, have serrate-crenate margins with buds, an obtuse apex, asymmetric base, reticulate venation, and long petiole. The flowers are pendulous, dark, and bell-like. The stems are tall, hollow, obtuse, and four-angled. The fruits are enclosed in the calyx and corolla. The seeds are small, smooth, oblong-ellipsoid, rarely striate, and smooth. | The leaves are broadly shallow on the adaxial side and convex on the abaxial side. The epidermal layer is thin, with small prominent cells on the adaxial side and less distinct on the abaxial side. The ground tissue of the midrib is parenchymatous and homogenous. The cells are circular or angular and compact. The vascular strand is single, collateral, small, and hemispherical; it consists of a thick horizontal band of xylem and a wide band of phloem. The lamina is uniformly flat with an even surface. The mesophyll tissue is not differentiated into palisade and spongy parenchyma. The stomata are anisocytic. The leaf petiole shows prismatic crystals of calcium oxalate embedded in parenchymatous cells, and annular and spiral vessels. In the powder, part of the vascular bundle, epidermis, annular and spiral xylem vessels were observed. The secretory structures found in the stems, petioles, and leaf blades consisted of idioblasts containing anthocyanins. | [12,42,44,63,78,79,107,110] |
| K. pumila | It is a 30 cm high shrublet with small, fleshy leaves covered with powdery deposits formed by calcium carbonate sediments. The leaves are obovate (2.8 cm long, 1.7 cm wide, and 2.5 mm thick), opposite, wedge-shaped, and have a sinuate basis and dentate-serrate margins. | The reddish-brown or purple color appears along the leaf margins after exposure to sunlight due to the presence of anthocyanins in the epidermal cells and mesophyll vacuoles. The epidermal cells are polygonal–isodiametric or slightly oblong; they are more numerous on the abaxial surface. The anticlinal walls are curved or straight and are convex on the outer walls. The walls are thickened due to the presence of wax. The cuticula is smooth or slightly undulating, elevated or with striae, with sparse white or gray irregularly shaped and sized wax structures on the surface. The leaves are amphistomatic, with anisocytic stomata. The vascular bundles are collateral and closed. The sheath cells or phloem, xylem parenchyma cells, subepidermal ground tissue, mesophyll tissue, and chlorenchyma tissue cells may contain tannin substances. | [111] |
| K. rhombopilosa | Small plant (no more than 10 cm tall), which blooms in spring. The leaves are hard and triangular, with a pale and wavy margin and green-yellow flowers with red lines. | [6] | |
| K. synsepala | One of the more unusual species of the genus because it is one of the few that produces stolon (lateral spreading stems). The leaves are arranged in rosettes and are thick, succulent, smooth, shiny, and green, with violet-red marks along the margins. This species is dormant in winter. The flowers are small, hairy, tubular, numerous, and pink. | [6] | |
| K. tetraphylla | The leaves are silvery pale green, which turn red in bright sun and revert to green in active growth. It has a large rosette of rounded or wavy leaves. The inflorescence is terminal and erect, with densely clustered panicles of greenish, waxy, narrow, urn-shaped flowers. | [6] | |
| K. tomentosa | The leaves are silvery, about 30 cm tall, reflecting the sun’s rays, lessening the chances of leaves overheating. | Its dense trichomes arise in triplets and perform a vital function in dry environments, helping to reduce the transpiration of water from the leaf surface | [6] |
| K. × houghtonii | A perennial erect herb; monocarpic; and can reach a height of up to 1.5 m. The leaves are opposite or verticillate, petiolate, with the leaf blade simple. The leaves vary from triangular to narrowly lanceolate, are serrate and mottled. The species forms corymbiform inflorescences of more than 100 pendulous, tetra or pentameric, dark-red flowers. | [103] |
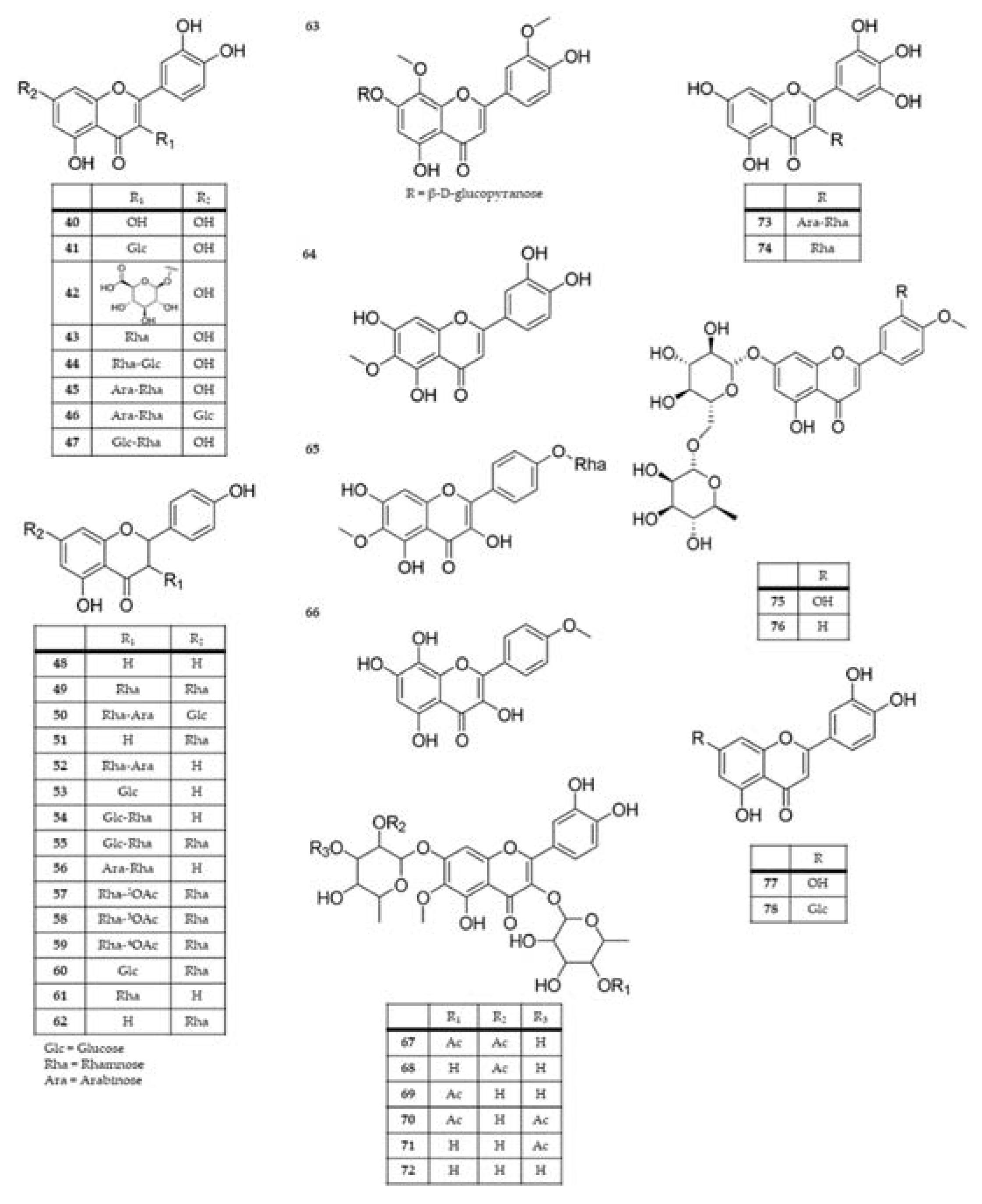
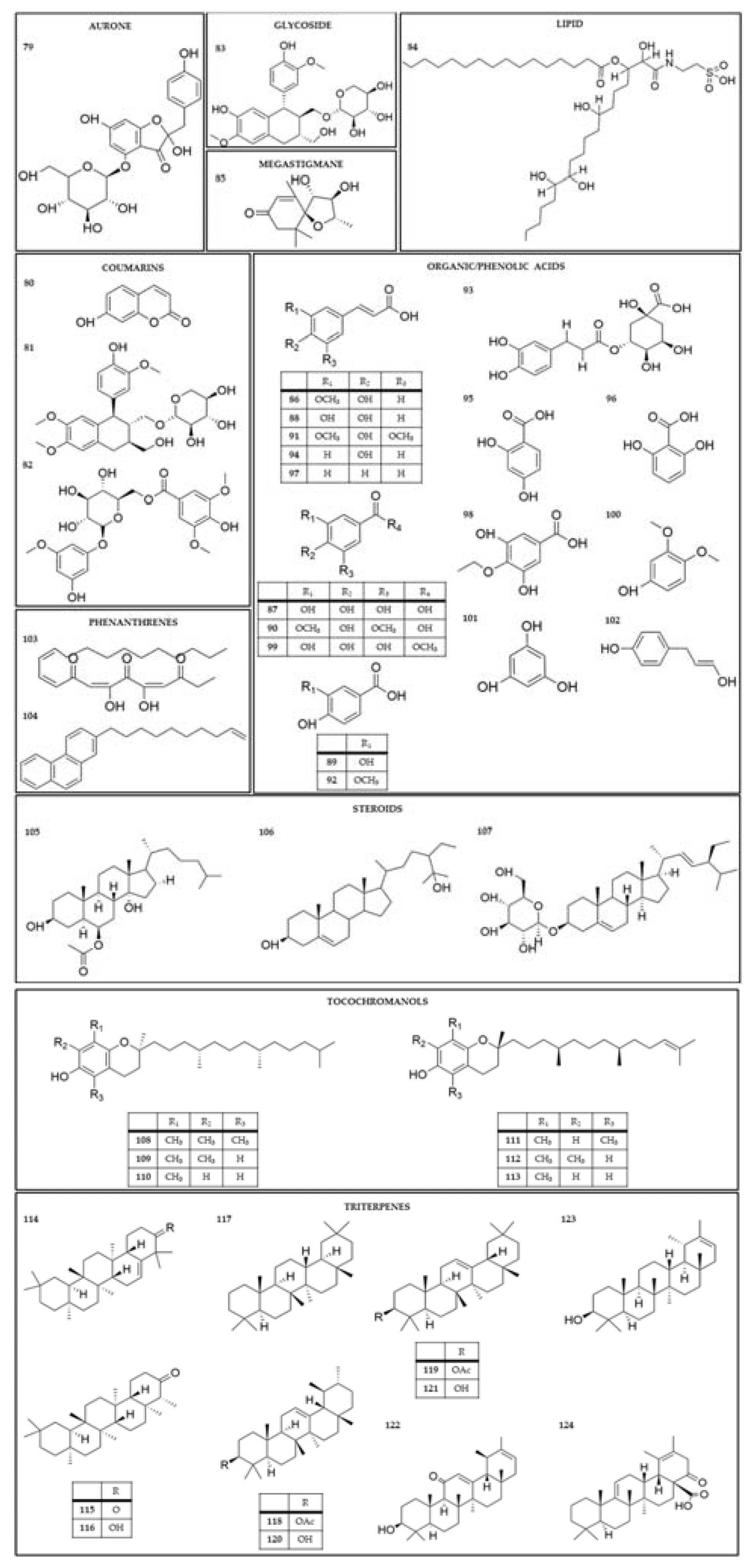
| Extract and/or plant part | Compound name | Species | References |
|---|---|---|---|
| Dichloromethane extract; methanol extractof aerial parts; flowers | bryophyllin A (bryotoxin C) (1) |
K. daigremontiana; K. pinnata; K. delagoensis; K. ceratophylla |
[11,32,87,112,113,114,115,116] |
| Aqueous extract from the roots or whole plant; methanol extractof aerial parts | bryophyllin B (2) |
K. daigremontiana; K. ceratophylla; K. delagoensis; K. pinnata |
[30,112,114,115,117] |
| Dichloromethane extract; methanol extract of the leaves; dichloromethane fraction from methanol extract | bryophyllin C (3) daigremontianin (4) methyl daigremonate (5) |
K. daigremontiana K. pinnata |
[11,113,118] |
| Dichloromethane extract; aqueous extract from the roots | bersaldegenin-1,3,5-orthoacetate (6) |
K. daigremontiana;K. pinnata; K. delagoensis |
[11,30,32,87,117] |
| Aqueous extract from the roots; ethanol and dichloromethane extracts from the leaves | bersaldegenin 1-acetate (7) |
K. daigremontiana;K. pinnata; K. delagoensis |
[11,30,32,87] |
| Ethanol and dichloromethane extracts from the leaves | bersaldegenin 3-acetate (8) | K. pinnata | [11,87,115] |
| Aqueous extract from the roots | bersaldegenin (9) | K. daigremontiana | [30] |
| Ethanol and dichloromethane extracts from the leaves | bufalin (10) | K. pinnata | [87] |
| Flower heads | bryotoxin A (11) | K. delagoensis | [112] |
| Aqueous extract from the roots; flowers | bryotoxin B (12) |
K. daigremontiana;K. delagoensis; K. pinnata |
[30,112,116,117] |
| Aqueous leaf extract | bufadienolide A (13) bufadienolide B (14) |
K. daigremontiana | [119] |
| Aqueous extract from the roots | daigredorigenin 3-acetate (15) | K. daigremontiana | [30,117] |
| 11α,19-dihydroxytelocinobufagin (16) | |||
| Methanol extract of aerial parts |
hellebrigenin (17) | K. ceratophylla | [114] |
| Methanol extract of aerial parts |
hellebrigenin-3-acetate (18) |
K. ceratophylla K. daigremontiana |
[114,117] |
| Methanol extract of aerial parts |
kalanchoside A (19) kalanchoside B (20) kalanchoside C (21) |
K. ceratophylla | [114] |
| Methanol extract of aerial parts |
thesiuside (22) | K. ceratophylla | [114] |
| Ethanol extract | kalantuboside A (23) kalantuboside B (24) |
K. delagoensis | [32] |
| Aqueous extract from the roots | 1β,3β,5β,14β,19-pentahydroxybufa-20,22-dienolide (kalandaigremoside A) (25) 19-(acetyloxy)-1β,3β,5β,14β-tetrahydroxybufa-20,22-dienolide (kalandaigremoside B) (26) 3β-(O-α-L-rhamnopyranosyl)-5β,11α,14β,19-tetrahydroxybufa-20,22-dienolide (kalandaigremoside C) (27) 19-(acetyloxy)-3β,5β,11α,14β-tetrahydroxybufa-20,22-dienolide (kalandaigremoside D) (28) 3β,5β,11α,14β,19-pentahydroxy-12-oxo-bufa-20,22-dienolide (kalandaigremoside E) (29) 19-(acetyloxy)-3β,5β,11α,14β-tetrahydroxy-12-oxo-bufa-20,22-dienolide (kalandaigremoside F) (30) 19-(acetyloxy)-1β,3β,5β,11α,14β-pentahydroxy-12-oxo-bufa-20,22-dienolide (kalandaigremoside G) (31) 1β-(acetyloxy)-3β,5β,11α,14β,19-pentahydroxy-12-oxo-bufa-20,22-dienolide (kalandaigremoside H) (32) |
K. daigremontiana | [30] |
| Ethyl acetate extract of the fresh plant | lanceotoxin A (33) lanceotoxin B (34) |
K. lanceolata | [120] |
| Methanol extract | kalanhybrin A (35) kalanhybrin B (36) kalanhybrin C (37) |
K. hybrida | [121] |
| Ethanol extract of the whole plant | kalantubolide A (38) kalantubolide B (39) |
K. delagoensis | [32] |
| Extract and/or plant part | Compound name | Species | References |
|---|---|---|---|
| Flower; ethanol leaf extract | quercetin (40) |
K. pinnata K. delagoensis K. blossfeldiana; K. mortagei; K. fedtschenkoi; K. daigremontiana;K. longiflora K. ceratophylla |
[32,37,122,123,124,125,126,127,128] |
| Flower extract | Quercetin 3-O-β-glucoside (quercetin 3-O-glucoside; isoquercetin; isoquercetrin) (41) |
K. pinnata; K. blossfeldiana; K. daigremontiana;K. delagoensis |
[49,123,124,128,129] |
| Flower extract | quercetin 3-O-β-D-glucuronopyranoside (miquelianin) (42) | K. pinnata | [123] |
| Aqueous and methanolic leaf extracts | quercetin 3-O-rhamnoside (quercitrin) (43) |
K. pinnata; K. delagoensis; K. longiflora; K. ceratophylla |
[42,83,123,124,126,127,130,131] |
| Flowers, Aqueous leaf extract | quercetin-3-O-β-D-xylopyranosyl (1→2)-α-L-rhamnopyranoside (44) |
K. blossfeldiana K. daigremontiana |
[119,128] |
| Aqueous and methanolic leaf extracts; flower extract | quercetin 3-O-α-L-arabinopyranosyl-(1 → 2)-α-L-rhamnopyranoside (45) | K. pinnata | [8,83,84,123,130,131] |
| Methanol leaf extract | quercetin 3-O-α-L-arabinopyranosyl-(1 → 2)-α-L-rhamnopyranoside-7-O-β-D-glucopyranoside (46) | K. pinnata | [130] |
| Ethanol leaf extract | quercetin 3-O-rutinoside (rutin) (47) | K. pinnata | [122] |
| Methanolic and hydroethanolic extracts from the leaves | kaempferol (48) |
K. delagoensis; K. pinnata; K. fedtschenkoi; K. longiflora; K. ceratophylla |
[2,37,43,124,126,127,130,132] |
| Water and ethanol extracts | kaempferol 3,7-O-dirhamnoside (kaempferitrin) (49) kaempferol 3-O-β-D-xylopyranosyl-(1→2)-α-L-rhamnopyranoside-7-O-β-D-glucopyranoside (daigremontrioside) (50) |
K. daigremontiana | [49] |
| Leaves | kaempferol 7-O-rhamnoside (51) |
K. delagoensis; K. longiflora |
[124,126] |
| Methanol leaf extract | kaempferol 3-O-β-D-xylopyranosyl-(1→2)-α-L-rhamnopyranoside (kaempferol 3-O-xylosyl-rhamnoside) (52) |
K. pinnata; K. daigremontiana |
[49,119,130] |
| Leaves | kaempferol 3-O-galactoside (trifolin) (53) | K. delagoensis | [124] |
| Leaves | kaempferol 3-rutinoside (nicotiflorin) (54) |
K. pinnata; K. longiflora |
[70,126] |
| Leaves | kaempferol-3-O-robinoside-7-O-rhamnoside (robinin) (55) |
K. delagoensis; K. longiflora |
[124,126] |
| Aqueous and methanolic leaf extracts | kaempferol 3-O-α-L-arabinopyranosyl (1→2)-α-L-rhamnopyranoside (kapinnatoside) (56) | K. pinnata | [84,130,131] |
| Ethyl acetate extract of the whole plant | kaempferol 3-O-α-L-(2-O-acetyl)rhamnopyranoside 7-O-α-L-rhamnopyranoside (57) kaempferol 3-O-α-L-(3-O-acetyl)rhamnopyranoside 7-O-α-L-rhamnopyranoside (58) kaempferol 3-O-α-L-(4-O-acetyl)rhamnopyranoside 7-O-α-L-rhamnopyranoside (59) kaempferol 3-O-α-D-glucopyranoside 7-O-α-L-rhamnopyranoside (60) afzelin (kaempferol 3-O-α-L-rhamnopyranoside) (61) α-rhamnoisorobin (kaempferol 7-O-α-L-rhamnopyranoside) (62) |
K. pinnata | [133] |
| Aqueous leaf extract | 4’,5-dihydroxy-3’,8-dimethoxyflavone 7-O-β-D-glucopyranoside (63) | K. pinnata | [131] |
| Aerial parts; methanol extract from the stems | eupafolin (6-methoxyluteolin) (64) | K. ceratophylla | [17,127] |
| Aerial parts | eupafolin 4’-O-rhamnoside (65) | K. ceratophylla | [127] |
| Ethanol extract of the whole plant | 4'-methoxyherbacetin (66) | K. delagoensis | [32] |
| Stems and leaves | kalambroside A (67) kalambroside B (68) kalambroside C (69) patuletin 3-O-(4'-O-acetyl-α-L-rhamnopyranosyl)-7-O-(3'-O-acetyl-α-L-rhaminopyranoside) (70) patuletin 3-O-α-L-rhamnopyranosyl-7-O-(3'-O-acetyl-α-L-rhaminopyranoside) (71) |
K. laciniata | [134] |
| Stems and leaves; hydroethanolic extract from leaves | patuletin 3-O-α-L-rhamnopyranosyl-7-O-α-L-rhamnopyranoside (72) | K. laciniata | [40,44,134] |
| Methanol leaf extract | myricetin 3-O-α-L-arabinopyranosyl-(1 → 2)-α-L-rhamnopyranoside (73) myricitrin (myricetin 3-O-α-L-rhamnopyranoside) (74) diosmine (diosmetin 7-O-α-L-rhamnopyranosyl-(1 → 6)-β-D-glucopyranoside) (75) acacetin 7-O-α-L-rhamnopyranosyl-(1 → 6)-β-D-glucopyranoside (76) |
K. pinnata | [130] |
| Ethanol leaf extract | luteolin (77) |
K. ceratophylla; K. pinnata |
[122,127] |
| Ethanol leaf extract | luteolin 7-O-β-D-glucoside (78) | K. pinnata | [122] |
| Class | Extract and/or plant part | Compound name | Species | References |
|---|---|---|---|---|
| Aurone | Aqueous root extract | hovetrichoside C (79) | K. daigremontiana | [117] |
| Coumarin | Aerial parts | 7-hydroxycoumarin (80) | K. ceratophylla | [127] |
| Roots | KPB 100 (81) KPB 200 (82) |
K. pinnata | [69] | |
| Glycoside | Aqueous root extract | schisandriside (83) | K. daigremontiana | [117] |
| Lipid | Ethanol extract of the whole plant | taurolipid C (84) | K. delagoensis | [32] |
| Megastigmane | Ethanol extract of the whole plant | (6S,7R,8R,9S)-6- oxaspiro-7,8-dihydroxymegastigman-4-en-3-one (tubiflorone) (85) |
K. delagoensis | [32] |
| Organic/ phenolic acid |
Leaves, ether leaves extract | ferulic acid (86) |
K. delagoensis; Kalanchoe sp. K.daigremontiana K. pinnata |
[29,124,136,137] |
| Ethanol extract of the whole plant; leaves | gallic acid (87) |
K. delagoensis; Kalanchoe sp.; K. daigremontiana |
[29,32,124,136] | |
| Leaves; ether leaves extract | caffeic acid (88) |
K. delagoensis; Kalanchoe sp; K. longiflora; K. daigremontiana K. pinnata |
[29,124,126,136,137] | |
| Leaves | protocatechuic acid (89) |
K. delagoensis; Kalanchoe sp.; K. daigremontiana |
[29,124,136] | |
| Ethanol extract of the whole plant; leaves; ether leaves extract | syringic acid (90) |
K. delagoensis; Kalanchoe spp. K. pinnata |
[32,124,136] | |
| Leaves | sinapic acid 91) | Kalanchoe sp. | [136] | |
| Ethanol extract of the whole plant; leaves | vanillic acid (92) |
K. delagoensis; Kalanchoe sp. |
[32,136,137] | |
| Leaves | chlorogenic acid (93) |
Kalanchoe sp.; K. longiflora |
[126,136] | |
| Leaves; ether leaves extract | p-Coumaric acid (94) |
Kalanchoe sp.; K. longiflora; K. daigremontiana K. pinnata |
[29,126,136,137] | |
| Leaves | β- resorcylic acid (95) | Kalanchoe sp. | [136] | |
| γ-resorcylic acid (96) | ||||
| Ethanol extract of the whole plant | cinnamic acid (97) | K. delagoensis | [32] | |
| 4-O-ethylgallic acid (98) methyl gallate (99) 3,4-dimethoxyphenol (100) phloroglucinol (101) 3,4-dihydroxyallylbenzene (102) |
K. delagoensis | [32] | ||
| Phenanthrene | Leaves | bryophollenone (103) 2(9-decenyl) phenanthrene (104) |
K. pinnata | [138] |
| Steroid | Leaves | bryophyllol (105) | K. pinnata | [138] |
| 24-ethyl-25-hydroxycholesterol (106) | ||||
| Ethanol extract of the whole plant | stigmasterol-O-D-glucoside (107) | K.delagoensis | [32] | |
| Tocochromanol | Hexane leaf extract | α-tocopherol (108) γ-tocopherol (109) δ-tocopherol (110) β-tocomonoenol (111) γ-tocomonoenol (112) δ-tocomonoenol (113) |
K. daigremontiana | [139] |
| iterpene | Aerial parts; petroleum ether extract from flowers;methanol extract | friedelin (114) |
K. fedtschenkoi; K. marnieriana; K. daigremontiana K. integra |
[137,140,141] |
| glutinone (115) | K. miniata | [140] | ||
| glut-5-en-3- β-ol (glutinol) (116) |
K. fedtschenkoi; K. daigremontiana K. integra |
[137,140,141,142] | ||
| Leaves | 18α-oleanane (117) | K. pinnata | [138] | |
| α-amyrin acetate (118) | ||||
| Leaves | β-amyrin acetate (119) |
K. pinnata; K. miniata |
[138,140] | |
| Leaves; methanol extract | α-amyrin (120) |
K. pinnata K. daigremontiana |
[138,142] | |
| Leaves; methanol extract | β-amyrin (121) | K. pinnata; | [138,141,142] | |
| Leaves, petroleum ether extract from flowers | bryophynol (122) Ψ-taraxasterol (123) bryophollone (124) |
K. daigremontiana K. pinnata K. integra |
[137,138] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





