Preprint
Article
Antimicrobial Resistance and Prevalence of Methicillin-resistant Staphylococcus aureus in Bovine Mastitis Milk from Conventional and Organic Dairy Farms in South Korea
Altmetrics
Downloads
137
Views
88
Comments
0
A peer-reviewed article of this preprint also exists.
† These authors contributed equally to this work as co-1st authors.
This version is not peer-reviewed
Submitted:
26 June 2023
Posted:
27 June 2023
You are already at the latest version
Alerts
Abstract
Bovine mastitis (BM) has been causing great economic and financial losses in the dairy industry worldwide and one of its major pathogenic agents is S. aureus. BM treatment still relies on antibiotics. The extensive use of antimicrobials gives rise to bacterial strains such as methicillin-resistant S. aureus (MRSA) and mupirocin-resistant S. aureus (MuRSA). This study aimed to investigate and compare the prevalence of MRSA and MuRSA and antimicrobial susceptibility in S. aureus isolates from bovine mastitis milk in conventional and organic dairy farms. Milk samples were taken from mastitis-infected cattle in 6 conventional and 7 organic dairy farms from South Korea. According to the Food Codex, 163 suspected S. aureus colonies were isolated from the pooled milk samples from each farm. Further rapid coagulase test confirmed 11 out of 74 isolates from 4 conventional farms (CF1, CF2, CF3, CF4) while 17 out of 89 isolates from 3 organic farms (OF1, OF2, OF3) exhibited coagulase activity. Multiplex PCR amplification showed that the nuc gene marker for S. aureus was found in all coagulase-positive isolates from OF 1 and at least one isolate from CF1, CF2, CF3, and CF4. Conversely, only 2 isolates from CF2 contained mecA gene for MRSA while none had the mupA gene for MuRSA. Antimicrobial susceptibility testing revealed that conventional farm isolates were more resistant to antibiotics particularly, against ampicillin and tetracycline, and, in turn, could imply a potential to develop multidrug resistance if stringent measures to control antimicrobial use in dairy farms is not implemented properly.
Keywords:
Subject: Biology and Life Sciences - Food Science and Technology
1. Introduction
Bovine mastitis (BM) is one of the major concerns in dairy industry, causing considerable economic and financial losses worldwide [1]. BM is the inflammation of mammary gland caused mainly by bacteria, fungi, some microscopic algal species, viruses, and occasionally by physical trauma [2]. BM’s pathogenic agents consist of different of gram-positive and gram-negative bacteria, and are classified as contagious (e.g., Staphylococcus aureus, Streptococcus agalactiae, Mycoplasma spp., Corynebacterium bovis) or environmental (e.g., Escherichia coli, Enterococcus spp., coagulase-negative Staphylococcus, Streptococcus uberis) [1,2]. Contagious pathogens live in the cow’s udder and teat skin and grow and colonize at the teat canal. Mastitis caused by contagious pathogens are often characterized by sub-clinical intramammary infections with no visible signs, increased somatic cell count and can be transmitted from cow-to-cow, particularly during milking. On the other hand, environmental mastitis pathogens are found in the cow’s environment and can enter the teat during milking or when the cow’s inherent or natural immunity becomes weak. Environmental pathogens are opportunistic microorganisms that cause clinical mastitis which can be easily noticed and detected, as characterized by evident swelling of the teat and with occasional pus included in the milk [3,4]. And among these pathogenic agents, the most related to both clinical and sub-clinical mastitis is S. aureus [5] which has also been noted to cause several human infections and, in turn, raises the concern of cross-infection between humans and animals through contaminated food and other factors. Although the risk of getting MRSA through pasteurized milk consumption is low, raw milk and milk products may still pose a threat to the public when exposed to viable bacteria [5,6].
The general measures applied to reduce infection and prevent spread of mastitis include improving sanitation (e.g., enhanced milking hygiene), teat disinfection after milking, and maintenance of milking machines [1,2]. However, these protocols can only reduce the degree of contamination and do not necessarily eliminate the existing infection. Treatment of active mastitis still relies greatly on antibiotics [5,6]. The continuous and extensive use of different antibiotics causes some bacterial strains like S. aureus to become resistant against antimicrobial agents, particularly on β-lactams. Hence, the prevalence and rise of S. aureus strains, coagulase-negative staphylococci (CoNS) species, and other bacterial strains which have exhibited resistance to different antibiotics such as, penicillin, clindamycin, tetracycline, methicillin, mupirocin, and vancomycin over the years [6,7,8], particularly in conventional farms. Consequently, organic farming has been introduced to combat the emergence of antibiotic-resistant mastitis pathogens. Organic livestock production practices ban the use of non-therapeutic antibiotics and growth promoters. Only when animal safety is endangered that antibiotics use is allowed, which is very seldom [9]. However, there are still some instances where these antibiotic-resistant microorganisms are found in organic farms as well [10]. In Korea, sales of antibiotics used in livestock doubled from 55 tons in 2011 to 97 tons in 2020 according to the 2020 National Antibiotics Use and Resistance and Monitoring, with the resistance level of livestock-isolated S. aureus for penicillin, ampicillin, and oxacillin to be 69.4%, 57.7%, and 2.3%, respectively [11].
Consumers are becoming more health-conscious and are leaning towards “organic” or naturally sourced food commodities. Increased awareness and concern for food safety and health issues drive the consumers to choose high quality products often labeled as organic, antibiotic-free, and animal welfare-approved milk products. Organic milk sales in Korea increased significantly from 5 billion won in 2008 to 104 billion won in 2020 [12]. With the increasing demand for organic livestock products market, there is a need to check the safety of the said products.
As far as the authors know, only a few studies had been conducted that focus on the comparative antimicrobial resistance of bacterial isolates from conventional and organic dairy farms, and most of the milk samples used in these studies were taken from bulk tank milk samples, especially in South Korea. In this manuscript we tried to isolate S. aureus from pooled milk samples of cattle already infected with mastitis (clinical and sub-clinical) from conventional and organic farms in South Korea and checked for presence for MRSA and MuRSA, as well as their corresponding antimicrobial susceptibility.
2. Materials and Methods
2.1. Materials and Reagents
Tryptic soy broth (TSB) and Baird-Parker agar (BPA) were purchased from BD Difco™ (NJ, USA). Egg-yolk tellurite emulsion was bought from Oxoid (New Hampshire, England). Muller-Hinton agar (MHA), nutrient agar (NA), and nutrient broth (NB) were distributed by MBCell (Seoul, Korea). Antibiotics used, ampicillin (AMP) was obtained from GA BioChem (GA, USA) while oxacillin (OX), gentamicin (CN), tetracycline (TE), erythromycin (E), chloramphenicol (C), ciprofloxacin (CIP), vancomycin (VA), and teicoplanin, (TEC) were supplied by LiofilChem® (Roseto degli Abruzzi, Italy). Pastorex Staph-plus latex agglutination test kit was from Bio-Rad (CA, USA). PCR premix and oligonucleotide primers were secured from Bioneer (Daejeon, Korea). Bacterial DNA extraction kit was provided by Takara Bio (Shiga, Japan). Sodium chloride (NaCl) was ordered from Sigma-Aldrich (St. Loius, MO, USA). All other reagents, media, and materials used were HPLC- or analytical-grade.
2.2. Methods
2.2.1. Sample collection and isolation of S. aureus from bovine mastitis milk samples
Pooled raw milk samples from mastitis-infected (clinical and sub-clinical) cattle were aseptically collected from 6 conventional dairy farms and 7 organic-certified dairy farms from southern area of Gyeonggido and Chungcheongnamdo, South Korea in late 2021 to the first quarter of 2022. For each infected cattle, teat surface was disinfected with 70% ethanol and after a few streams of milk were discarded, 10 ml of milk was collected in sterile polyethylene screw-capped bottles from each teat. Milk samples from individual cattle were pooled per farm and kept in icebox (at 4℃) and immediately transported to laboratory for analysis.
S. aureus was isolated according to the Korean Food Standards Codex [13]. For each sample, 25 g of milk was transferred in sample filter bag and mixed with 225 ml of TSB containing 10% NaCl using stomacher (BagMixer 400CC, InterScience, France). The homogenized mixture was then incubated at 36±1 °C for 18-24 h. The enriched media was plated onto BPA supplemented with 5% egg-yolk tellurite emulsion and incubated at 36±1°C for another 18-24 h. Each black, glossy colony surrounded by transparent bands was separated and cultured to NA for further isolation. Gram-positive cocci with staphylococcal arrangement were verified using gram-staining. Further confirmation was done by rapid coagulase test using Pastorex™ Staph-plus latex agglutination test kit (Bio-Rad, CA, USA). Confirmed S. aureus cultures were stocked in sterile glycerol solution and stored at -20°C until further analysis. Re-testing and subculture onto NA were done whenever storage period exceeded 7 days.
2.2.2. Antimicrobial susceptibility testing of isolated S. aureus cultures using disc diffusion method
Antimicrobial susceptibility testing was done following the guidelines of CLSI (Clinical and Laboratory Standards Institute) [14] using disc diffusion test against nine antibiotics (ampicillin, oxacillin, gentamicin, teicoplanin, vancomycin, erythromycin, chloramphenicol, ciprofloxacin, and tetracycline) as described in Table 1 with their corresponding concentration and inhibition zones.
Bacterial suspension was made following the density of McFarland 0.5 turbidity standard (1.5×108 CFU/ml) and swabbed evenly onto the entire surface of MHA plates. Antibiotic discs were applied firmly onto the inoculated MHA plate with enough space for the inhibition zones. The agar plates were then incubated at 36±1°C for 18-24 h and inhibition zones measured using vernier caliper. Isolates were tested for each antibiotic on duplicate plates and classified (resistant, intermediate, susceptible) according to the average of their corresponding inhibition zones.
2.2.3. Bacterial DNA extraction, PCR amplification, and gel electrophoresis
Glycerol stock of isolated S. aureus samples were plated on NA and incubated at 36±1 °C for 18-24 h. Bacterial DNA was obtained following Takara MiniBEST bacteria genomic DNA extraction kit (Takara Bio, Shiga, Japan). First, lysis was done by making 1 ml of bacterial suspension (0.5-2.0×109 CFU/ml) and centrifugation at 12,000 rpm for 2 min. Supernatant was discarded and 50 µl buffer BS and 50 µl lysozyme were added to the bacteria pellet. The mixture was homogenized and incubated in a 37°C-waterbath for 1 h, with occasional inversion mixing every 20 min. The incubated solution was centrifuged at 12,000 rpm for 5 min and supernatant was discarded. Then, 180 µl buffer GL, 20 µl proteinase K, and 10 µl RNase A were added, mixed thoroughly, and incubated at 56°C-waterbath for 10-30 min, with pipet mixing every 5 min until solution becomes clear. Buffer GB (200 µl) and 100% ethanol (200 µl) were added and mixed well. The solution was then transferred to the spin column with a collection tube and centrifuged at 12,000 rpm for 2 min. Flow-through was discarded and buffer WA (500 µl) was added to the spin column and centrifuged (12,000 rpm, 1 min). Flow-through was removed and spin column was washed with 700 µl buffer WB and centrifuged (12,000 rpm, 1 min). Flow-through was discarded and spin column was washed with buffer WB once more and centrifuged (12,000 rpm, 2 min). After, spin column was transferred to a new 1.5 ml centrifuge tube and 50 µl elution buffer and let stand at room temperature for 5 min before centrifugation (12,000 rpm, 2 min) to elute the DNA.
Extracted DNA samples were then subjected to PCR amplification using the following primers (Table 2):
Multiplex PCR (M-PCR) assay was performed according to [7] with slight modifications. An aliquot of 1 µl bacterial DNA template was added to 20-µl PCR premix containing the reaction buffer with 1.5 mM MgCl2, 250 µM each of deoxynucleoside triphosphate (dATP, dUTP, dGTP, and dCTP), 1.0 U of Top DNA polymerase (Bioneer, Daejeon, Korea), and 1 µL of each primer for mecA (1.2 µM), nuc (0.4 µM), and mupA (0.5 µM). Amplification was done using Q-Cycler II gradient thermal cycler (QuantaBiotech, Surrey, UK) and carried out as follows: initial denaturation step at 94°C for 5 min; 10 cycles of 94°C for 40 s, 58°C for 40 s, and 72°C for 1 min; 25 cycles of 94°C for 1 min, 50°C for 1 min, and 72°C for 2 min; and final extension step at 72°C for 10 min. Amplified samples were stored at 4°C prior to gel electrophoresis. PCR amplicons were loaded on 2.0% agarose gel (Bioneer, Daejeon, Korea) containing 0.5 µg/ml mango blue dye and visualized under UV light box using digital image system (GDS-200D, MDM, Korea).
2.2.4. Statistical Analysis
The connection between type of farm (conventional or organic) and isolate activity (% resistance) for each antibiotic were analyzed using Chi-Square analysis for independent variance with Statistical Packages for the Social Sciences (IBM SPSS Statistics 23.0) software (SPSS Inc., USA). Significant difference among data were identified with p < 0.05 values. The same treatment was done for the relationship between farm type and resistant categories.
3. Results
3.1. Isolation of S. aureus from bovine mastitis milk samples in conventional and organic dairy farms
Following the Korea Food Codex [13], 163 suspected S. aureus colonies were isolated from all the bovine mastitis milk samples, wherein, 74 colonies were from 6 conventional dairy farms while 89 colonies were from 7 organic dairy farms. Additional rapid coagulase test confirmed that only 11 out of 74 (15%) of the separated colonies from 4 conventional dairy farm samples while 17 out of 89 (19%) of the isolates from 3 organic dairy farm samples exhibited coagulase activity which is a characteristic of S. aureus.
3.2. Antimicrobial susceptibility of isolated S. aureus cultures
Coagulase-positive Staphylococci (CoPS) isolates in BM milk samples from conventional and organic farms were further tested for antibiotic susceptibility against 9 antimicrobial substances (see Table 3).
Overall, the CoPS isolates exhibited less than 50% resistance against the following antibiotics used in this study: gentamicin (46.4%), tetracycline (46.4%), ampicillin (39.3%), erythromycin (21.4%), oxacillin (14.3%), and chloramphenicol (14.3%).
Figure 1 showed that conventional farm isolates demonstrated more noticeable antibiotic resistance than the organic farm isolates. Resistance against ampicillin and tetracycline was detected in 81.8% of the conventional isolates while 45.5% were resistant to erythromycin, 36.4% were resistant to oxacillin, and 18.2% were resistant to chloramphenicol. Conversely, organic farm isolates exhibited lower resistance against ampicillin (11.8%), erythromycin (5.9%), chloramphenicol (11.8%), and tetracycline (23.5%). However, higher resistance against gentamicin was noted in organic farm isolates at 52.9% compared to conventional farm isolates at 36.4%. Additionally, the isolates from both dairy farms were discovered to be sensitive to the quinoline antibiotic, ciprofloxacin at 85.7% (data not shown). Furthermore, organic farm milk isolates also showed susceptibility against the β-lactam antibiotics, ampicillin, and oxacillin at 76.5% and 82.4% (data not shown).
All CoPS isolates were grouped and categorized according to the number of antimicrobial substances/s they were resistant against (shown in Figure 2). CoPS isolates from conventional farm samples were all resistant to at least one antibiotic while 23.5% of the organic farm isolates were classified as not resistant to any of the antimicrobials tested in this study. Most of the CoPS isolates from conventional farms (63.6%) were resistant to three or more substances, followed by both 18.2% of isolates resistant to one and two antimicrobial substances, respectively. Organic farm CoPS isolates were mainly composed of 52.9% resistant to one antibiotic while 17.6% were resistant to two antimicrobials. Lastly, there was a small percentage (5.9%) of organic farm isolates that were resistant to three or more substances.
3.3. DNA extraction and PCR amplification of isolated S. aureus isolates
DNA of S. aureus isolates were extracted using a bacterial DNA extraction kit and subjected to PCR amplification. The corresponding agarose gel visualization confirmed which isolates from organic (Figure 3) and conventional (Figure 4) farms contained the nuc, mecA, and mupA genes.
Of the 3 organics farm samples, 12 isolates that were all from the organic farm 1, OF1, contained the nuc gene which is specific gene marker for S. aureus [7,15]. Meanwhile, at least one isolate from conventional farms, CF1, CF2, CF3, and CF4, contained the nuc gene. Additionally, only two isolates from conventional farm 2 (CF2) were confirmed to have the mecA gene which is a determinant of methicillin resistance [7,16] while none of the organic farm isolates was noted with the mecA gene. On the other hand, the mupA gene, which is a determinant of mupirocin resistance [7,16], was not detected in any of the isolates from organic and conventional farms.
4. Discussion
In this study, higher counts of suspected S. aureus were isolated from BM milk in organic farms (89 colonies) than from the conventional farm (74 colonies) samples. Tikofsky et al. [9] also found more S. aureus isolated from milk samples from organic farms (144) than conventional farms (117) in US. Roesch et al. [17] observed a similar trend with more S. aureus and other mastitis pathogens isolated from organic farms (93) than conventional farms (65) in Switzerland. Sato et al. [18] also discovered more S. aureus from organic farms than conventional farms in Wisconsin (73 and 64, respectively). However, dairy herds in Denmark showed an opposite trend with slightly higher S. aureus isolate count in conventional farms than organic farms at 77 and 75, correspondingly [18]. The higher S. aureus isolate count found in organic farms could be due to the fact these farms have very limited use of antibiotics, which is only for emergency cases when the cattle’s life is in danger [9,10]. Use of non-therapeutic antibiotics and growth promoters are prohibited in organic farms, except for critical situations wherein animal safety is compromised that antibiotics use is allowed [9,10,19]. Even so, CoPS isolates were present in more than half of the conventional farm samples (4 out of 6) compared to organic farm samples (3 out of 7). This result was confirmed by the M-PCR amplification and shown through the agarose gel electrophoresis wherein some the organic farm milk isolates did not contain the nuc gene marker of S. aureus but were positive for the coagulase activity. It is also because of the restricted use of antimicrobials in organic farms that more varied species of microorganisms thrive in organic farms [9,17,18,19].
For antimicrobial susceptibility testing, conventional CoPS isolates were found to be significantly more resistant against ampicillin, oxacillin, erythromycin, and tetracycline than the organic CoPS isolates. Similar observations were noted in a study on MRSA from organic and conventional herds in Germany and their corresponding antimicrobial resistance wherein conventional farm isolates exhibited higher resistance against the penicillin (100%), cefoxitin (100%), tetracycline (97.2%), erythromycin (44.4%), gentamicin (11.1%), and chloramphenicol (8.3%) [20]. Ray et al. [21] checked the antimicrobial susceptibility of Salmonella from milk samples and discovered that conventional farm isolates were more resistant against streptomycin and sulfamethoxazole.
Antibiotic use had long been linked to the subsequent rise of antimicrobial resistance, in which the resistance strengthens or compounds with each exposure to the antimicrobial substance due to selective pressure [22,23,24]. The most widely used antimicrobials for the treatment of mastitis are β-lactam compounds such as ampicillin and oxacillin [2,25,26] and Korea is no exception to this practice [11]. β-lactams function as an antimicrobial agent by inactivating the enzymes called penicillin-binding proteins (PBPs) through covalent bonding. PBPs are critical to the cell cycle-related, morphogenetic synthesis of cell wall peptidoglycan. Accordingly, β-lactams can effectively inhibit the transpeptidation of PBPs, disturb the cross-linking of the bacterial peptidoglycan and, in turn, result to weakening of the cell wall, freeing the cytoplasmic contents, and eventual cell death when exposed to an appropriate concentration [27,28,29,30]. However, this antimicrobial activity can be bypassed due to the expression of a unique PBP, known as PBP2a, which has low affinity for all β-lactam antibiotics and can be found in MRSA [26,29,30]. PBP2a does not easily attach to the β-lactams and cannot be inactivated. And since it has similar structural motifs as the innate PBPs 1-4 in S. aureus, it can keep transpeptidation, support the cross-linking of peptidoglycan and allow the survival of MRSA [29,30]. On the other hand, erythromycin and tetracycline were not allowed for consumption in cattle livestock industry in Korea but similar compounds as erythromycin (spiramycin and tylosin, which belonged to macrolides antibiotics), and tetracycline (chlortetracycline and oxytetracycline) were used instead [11]. The topmost sold antibiotic classes for use in livestock and fisheries in Korea are the following: β-lactams > tetracyclines > phenicols > macrolides > sulfonamides > aminoglycosides [11].
Although resistance levels were less than 50% for most antimicrobials, the implication of multidrug resistance was evident in both types of farm isolates. A considerably higher count of multi-drug resistant CoPS isolates was found in conventional farms than in organic farm samples. Microbial isolates resistant to three or more classes of antimicrobials were classified as “multidrug-resistant” [31]. This could also be attributed to the regular exposure of infected cattle in conventional farms to antibiotics as one of their treatment methods for BM. However, the exposure parameters like dose, administration route, and dose frequency, were not specified as antibiotic usage was difficult to monitor and quantify in especially in conventional farms [18]. Grobbel et al. [31] investigated and compared the antimicrobial resistance of Escherichia coli from organic and conventional poultry farms in Germany where they found that conventional farm isolates were resistant to three or more antibiotics. Meissner et al. [10], on the other hand, noted an almost similar percentage of multidrug-resistant isolates of extended-spectrum β-lactamase-producing E. coli from conventional and organic pig fattening farms.
The isolates from organic farms 2 and 3 (OF2 and OF3) that did not contain the nuc gene could belong to other CoPS species but further tests are needed to done to properly identify them. Aside from S. aureus, the other Staphylococcus spp. that exhibit coagulase activity are the following: S. intermedius, S. schleiferi subsp. coagulans, S. hyicus, S. lutrae, S. delhini, and S. pseudintermedius, which also demonstrate antibiotic resistance and biofilm formation [32,33]. CoPS are opportunistic pathogens, meaning they can easily adapt accordingly to their surroundings or hosts, causing a lot of potent virulence factors [32].
Two isolates from conventional farm 2 were confirmed to have the mecA gene which is a known determining factors of methicillin resistance [7,16]. MRSA contains the mecA gene which is responsible for encoding PBP 2a [27,29]. This result was further confirmed with the antimicrobial resistance. On the other hand, the mupA gene, which is a determinant of mupirocin resistance [7], was not detected in any of the conventional and organic farm isolates. Mupirocin is an antibiotic that could effectively inhibit bacterial synthesis and has high antimicrobial activity against S. aureus [8,34]. Mupirocin acts as an antimicrobial by binding to the enzyme isoleucyl-transfer RNA synthetase preventing isoleucine incorporation during bacterial protein synthesis, which makes it effective against MRSA and some methicillin-resistant CoNS [7,8,34].
The presence of mupirocin-resistant isolate was investigated to check for any possibility of inter-host-adapted (human-to-cattle and vice versa) variants of S. aureus. Different serovars of host-adapted S. aureus could develop through host-switching and gene acquisition, loss and diversification [5]. Nonetheless, this non-detection of mupA gene in any of the isolates could imply that mupirocin and other mupirocin-related compounds could still be effective against S. aureus that would be isolated from conventional and organic dairy farms in South Korea.
5. Conclusions
This study showed that there is variation in S. aureus isolated from mastitis-infected cow’s milk from organic and conventional dairy farms in South Korea. Although a higher isolate count was found from organic farms, these isolates have low antimicrobial resistance and did not contain the mecA gene. On the other hand, conventional farm isolates were more resistant to antimicrobials, particularly against ampicillin and tetracycline. Moreover, mecA gene was found in conventional farm isolates through multiplex PCR. Consequently, these findings could imply a potential to develop multidrug resistance in BM- and livestock-related bacterial isolates if stringent measures to control antimicrobial use in dairy farms are not implemented properly. Additional analyses are needed to further confirm the other Staphylococcus species of the other isolates that did not contain mecA, mupA, or even nuc gene.
Supplementary Materials
Not applicable.
Author Contributions
Conceptualization, H.J.P., T.A.N., and I.S.N.; methodology, H.J.P. and T.A.N.; validation, T.A.N., H.J.P., S.H.B. and I.S.N.; formal analysis, H.J.P., T.A.N., S.J.K.; investigation, H.J.P., T.A.N., S.J.K.; resources, S.H.B. and I.S.N.; data curation, H.J.P. and T.A.N.; writing—original draft preparation, H.J.P. and T.A.N.; writing—review and editing, T.A.N.; visualization, T.A.N.; supervision, I.S.N. and S.H.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to acknowledge the farm owners that participated in this research study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bradley, A.J. Bovine Mastitis: An Evolving Disease. The Vet. J. 2002, 164(2), 116–128. [CrossRef]
- Cheng, W.N.; Han, S.G. Bovine mastitis: risk factors, therapeutic strategies, and alternative treatments - A review. Asian-Australas. J. Anim. Sci. 2020, 33(11), 1699–1713. [CrossRef]
- Barkema, H.W.; Green, M.J.; Bradley, A.J.; Zadoks, R.N. Invited review: The role of contagious disease in udder health. J. Dairy Sci. 2009, 92, 4717–4729. [CrossRef]
- Bobbo, T.; Ruegg, P.L.; Stocco, G.; Fiore, E.; Gianesella, M.; Morgante, M.; Pasotto, D.; Bittante, G.; Cecchinato, A. Associations between pathogen-specific cases of subclinical mastitis and milk yield, quality, protein composition, and cheese-making traits in dairy cows. J. Dairy Sci. 2017, 100, 4868–4883. [CrossRef]
- Campos, B.; Pickering, A.C.; Rocha, L.S.; Aguilar, A.P.; Fabres-Klein, M.H.; Mendes, T.A.; Fitzgerald, J.R.; Ribon, A. Diversity and pathogenesis of Staphylococcus aureus from bovine mastitis: current understanding and future perspectives. BMC Vet. Res. 2022, 18,115. [CrossRef]
- Annamanedi, M.; Sheela, P.; Sundareshan, S.; Isloor, S.; Gupta, P.; Jasmeen, P.; Gargi, M.; Mallick, S.; Hegde, N. Molecular fingerprinting of bovine mastitis-associated Staphylococcus aureus isolates from India. Sci. Rep.-UK 2021, 11, 15228. [CrossRef]
- Zhang, K.; Sparling, J.; Chow, B.L.; Elsayed, S.; Hussain, Z.; Church, D.L.; Gregson, D.B.; Louie, T.; Conly, J.M. New Quadriplex PCR Assay for Detection of Methicillin and Mupirocin Resistance and Simultaneous Discrimination of Staphylococcus aureus from Coagulase-Negative Staphylococci. J. Clin. Microbiol. 2004, 42(11), 4947–4955. [CrossRef]
- Seiberling, K.A.; Aruni, W.; Kim, S.; Scapa, V.I.; Fletcher, H.; Church, C.A. The effect of intraoperative mupirocin irrigation on Staphylococcus aureus within the maxillary sinus. Int. Forum Allergy Rhinol. 2012, 3, 94- 98. [CrossRef]
- Tikofsky, L.L.; Barlow, J.W.; Santisteban, C.; Schukken, Y.H.A. Comparison of Antimicrobial Susceptibility Patterns for Staphylococcus aureus in Organic and Conventional Dairy Herds. Microb. Drug Resist. 2003, 9, 39–45. http://doi.org/10.1089/107662903322541883.
- Meissner, K.; Sauter-Louis, C.; Heiden, S.E.; Schaufler, K.; Tomaso, H.; Conraths, F.J.; Homeier-Bachmann, T. Extended-Spectrum ß-Lactamase-Producing Escherichia coli in Conventional and Organic Pig Fattening Farms. Microorganisms 2022, 10(3), 603. [CrossRef]
- National Institute of Food and Drug Safety Evaluation. National Antibiotics Use and Resistance and Monitoring Report 2020: Animals, Livestock, and Marine Products, final version; Korea Ministry of Food and Drug Safety, joint publication with Korea Ministry of Agriculture, Food and Korea Rural Affairs, the Agriculture, Forestry and Livestock Quarantine: Seoul, South Korea, 2021; pp.3-4. Available online: https://www.mfds.go.kr/brd/m_231/view.do?seq=33051&srchFr=&srchTo=&srchWord=&srchTp=&itm_seq_1=0&itm_seq_2=0&multi_itm_seq=0&company_cd=&company_nm=&page=1 (accesses 19 December 2022).
- USDA Global Agricultural Information Network (GAIN) 2022 Attaché report – South Korea: Dairy and Products Annual. Available online: https://apps.fas.usda.gov/newgainapi/api/Report/DownloadReportByFileName?fileName=Dairy%20and%20Products%20Annual_Seoul%20ATO_Korea%20-%20Republic%20of_KS2022-0022.pdf Accessed December 19, 2022.
- Korean Food Standards Codex - KFS Codex, Ministry of Food and Drug Safety (in Korean). Available Online: https://foodsafetykorea.go.kr/foodcode/01_03.jsp?idx=380 (accessed 01 December 2021).
- Clinical and Laboratory Standards Institute (CLSI). CLSI Supplement M100S. In Performance Standards for Antimicrobial Susceptibility Testing, 32nd ed.; CLSI: Wayne, PA, US, 2022.
- Shortle, D. A genetic system for analysis of staphylococcal nuclease. Gene. 1983, 22, 181–189. [CrossRef]
- Pérez-Roth, E.; Claverie-Martín, F.; Villar, J.; Méndez-Alvarez, S. Multiplex PCR for simultaneous identification of Staphylococcus aureus and detection of methicillin and mupirocin resistance. J. Clin. Microbiol. 2001, 39(11), 4037–4041.
- Roesch, M.; Perreten, V.; Doherr, M.G.; Schaeren, W., Schällibaum, M., Blum, J.W. Comparison of Antibiotic Resistance of Udder Pathogens in Dairy Cows Kept on Organic and on Conventional Farms. J. Dairy. Sci. 2006, 89(3), 989–997. [CrossRef]
- Sato, K.; Bennedsgaard, T.W.; Bartlett, P.C.; Erskine, R.J.; Kaneene, J.B. Comparison of Antimicrobial Susceptibility of Staphylococcus aureus Isolated from Bulk Tank Milk in Organic and Conventional Dairy Herds in the Midwestern United States and Denmark. J. Food Protection 2004, 67(6), 1104–1110. [CrossRef]
- Álvarez-Fernández, E; Cancelo, A.; Díaz-Vega, C.; Capita, R.; Alonso-Calleja, C. Antimicrobial resistance in E. coli isolates from conventionally and organically reared poultry: A comparison of agar disc diffusion and Sensi Test Gram-negative methods. Food Control. 2013, 30 (1), 227–234. [CrossRef]
- Tenhangen, B.A.; Alt, K.; Pfefferkorn, B.; Wiehle, L.; Kasbohrer, A.; Fetsch, A. Short Communication: Methicillin-resistant Staphylococcus aureus in conventional and organic dairy herds in Germany. J. Dairy Sci. 2018, 101(4), 3380–3386. [CrossRef]
- Ray, K.A.; Warnick, L.D.; Mitchell, R.M.; Kaneene, J.B.; Ruegg, P.L.; Wells, S.J.; Fossler, C.P.; Halbert, L.W.; May, K. Anti-microbial susceptibility of Salmonella from organic and conventional dairy farms. J. Dairy Sci. 2018, 89, 2038–2050. [CrossRef]
- Chantziaras, I.; Boyen, F.; Callens, B.; Dewulf, J. Correlation between veterinary antimicrobial use and antimicrobial resistance in food-producing animals: a report on seven countries. J. Antimicrob. Chemother. 2014, 69(3),827–834. [CrossRef]
- Lam, T.; Scherpenzeel, C.G.M.; den Uijl, I.E.; van Schaik, G. Dry cow therapy: Does it still deserve a blanked recommendation? In Proceedings of the National Mastitis Council 53rd Annual Meeting, Fort Worth, TX, US, 26-28 January 2014, pp. 64–72.
- Schnitt, A.; Tenhagen, B.A. Risk Factors for the Occurrence of Methicillin-Resistant Staphylococcus aureus in Dairy Herds: An Update. Foodborne Pathog. Dis. 2020, 17(10), 585–596. http://doi.org/10.1089/fpd.2019.2638.
- Hakenbeck, R.; Coyette, J. Resistant penicillin-binding proteins. Cell. Mol. Life Sci. 1998, 54, 332–340. [CrossRef]
- Zapun, A.; Contreras-Martel, C.; Vernet, T. Penicillin-binding proteins and β-lactam resistance. FEMS Microbiol. Rev. 2008, 32(2), 361–385. [CrossRef]
- Tipper, D.J. Mode of action of beta-lactam antibiotics. Pharmacol. Ther. 1985, 27(1), 1–35. [CrossRef]
- Tang, S.S.; Apisarnthanarak, A.; Hsu, L.Y. Mechanisms of β-lactam antimicrobial resistance and epidemiology of major community- and healthcare-associated multidrug-resistant bacteria. Adv. Drug Deliv. Rev. 2014, 78, 3–13. http://dx.doi.org/10.1016/j.addr.2014.08.003.
- Stapleton, P.D.; Taylor, P.W. Methicillin resistance in Staphylococcus aureus: mechanisms and modulation. Sci. Prog. 2002, 85(1), 57–72. [CrossRef]
- Hartman, A.; Tomasz, B. Altered penicillin-binding proteins in methicillin-resistant strains of Staphylococcus aureus. Antimicrob. Agents Chemother. 1991, 19, 726–735. [CrossRef]
- Grobbel, M.; Hammerl, J.A.; Alt, K.; Irrgang, A.; Kaesbohrer, A.; Tenhagen, B.-A. Comparison of Antimicrobial Resistances in Escherichia coli from Conventionally and Organic Farmed Poultry from Germany. Antibiotics 2022, 11, 1282. [CrossRef]
- Velázquez-Guadarrama, N.; Olivares-Cervantes, A.; Salinas, E.; Martinez, L.; Escorcia, M.; Oropeza, R.; Rosas, I. Presence of environmental coagulase-positive staphylococci, their clonal relationship, resistance factors and ability to form biofilm. Rev. Argent. Microbiol. 2017, 49(1), 15–23. [CrossRef]
- Freney, J.; Kloos, W.E.; Hajek, V.; Webster, J.A.; Bes, M.; Brun, Y.; Vernozy-Rozand, C. Recommended minimal standards for description of new staphylococcal species. Int. J. Syst. Evol. Micr. 1999, 49(2), 489–502. [CrossRef]
- Sutherland, R.; Boon, R.J.; Griffin, K.E.; Masters, P.J.; Slocombe, B.; White, A.R. Antibacterial activity of mupirocin (pseudomonic acid), a new antibiotic for topical use. Antimicrob. Agents Chemother. 1985, 27, 495–498.
Figure 1.
Antibiotic resistance (%) of S. aureus isolated from bovine mastitis (BM) milk in conventional and organic dairy farms (n = number of coagulase-positive Staphylococcus isolates tested). Antimicrobial substances used were ampicillin (AMP), oxacillin (OX), gentamicin (CN), teicoplanin (TEC), vancomycin (VA), erythromycin (E), chloramphenicol (C), ciprofloxacin (CIP), and tetracycline (TE). Asterisk (*) denoting significant difference (p < 0.05) between conventional and organic farm samples.
Figure 1.
Antibiotic resistance (%) of S. aureus isolated from bovine mastitis (BM) milk in conventional and organic dairy farms (n = number of coagulase-positive Staphylococcus isolates tested). Antimicrobial substances used were ampicillin (AMP), oxacillin (OX), gentamicin (CN), teicoplanin (TEC), vancomycin (VA), erythromycin (E), chloramphenicol (C), ciprofloxacin (CIP), and tetracycline (TE). Asterisk (*) denoting significant difference (p < 0.05) between conventional and organic farm samples.
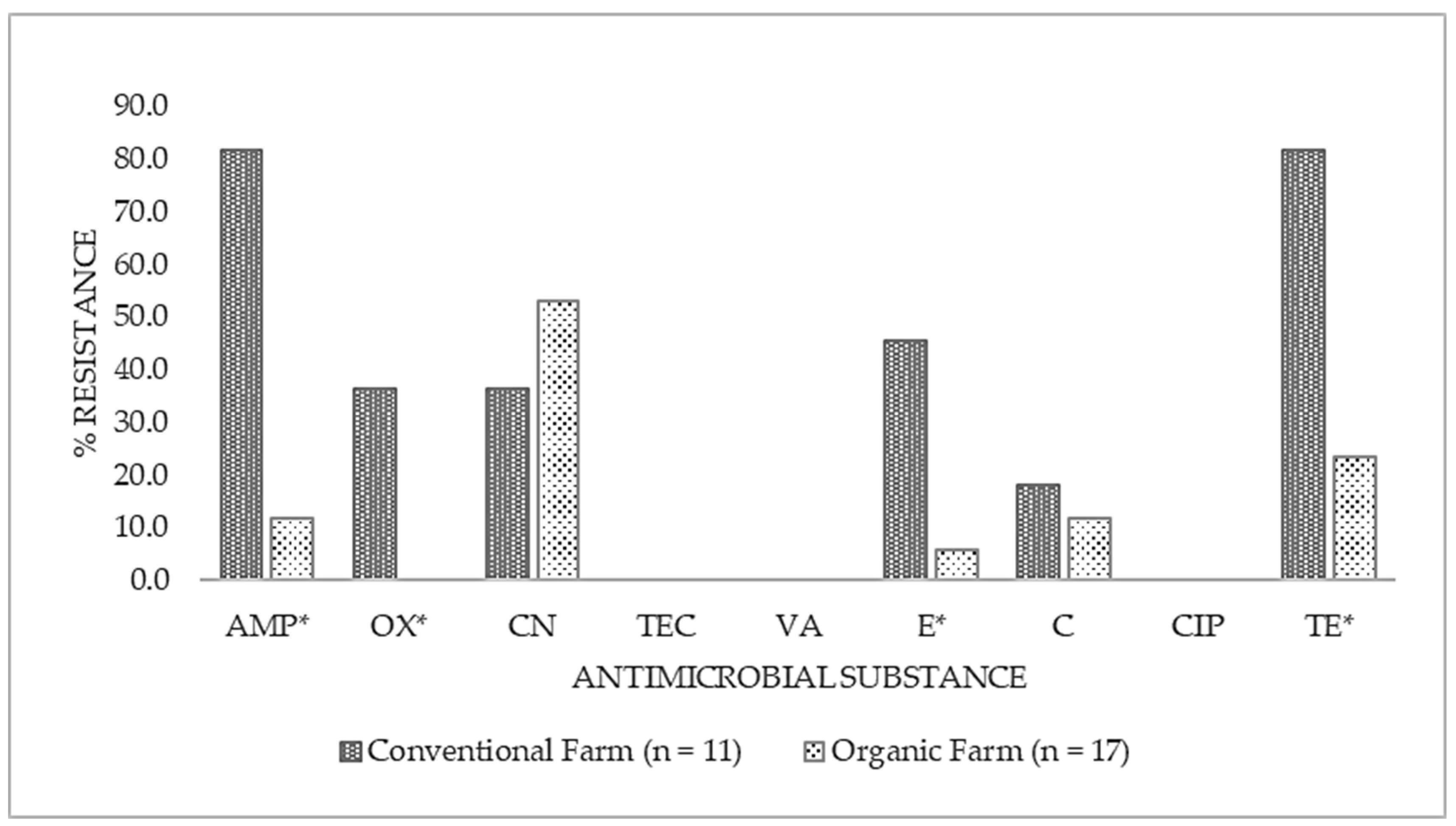
Figure 2.
Percent isolate (%) classification of coagulase-positive Staphylococcus (CoPS) isolates from bovine mastitis milk in conventional and organic dairy farms according to the number of antimicrobial substances resistant against (n = number of CoPS isolates tested). Antimcrobial agents tested were ampicillin, oxacillin, gentamicin, teicoplanin, vancomycin, erythromycin, chloramphenicol, ciprofloxacin, and tetracycline. Asterisk (*) denoting significant difference (p < 0.05) between conventional and organic farm samples.
Figure 2.
Percent isolate (%) classification of coagulase-positive Staphylococcus (CoPS) isolates from bovine mastitis milk in conventional and organic dairy farms according to the number of antimicrobial substances resistant against (n = number of CoPS isolates tested). Antimcrobial agents tested were ampicillin, oxacillin, gentamicin, teicoplanin, vancomycin, erythromycin, chloramphenicol, ciprofloxacin, and tetracycline. Asterisk (*) denoting significant difference (p < 0.05) between conventional and organic farm samples.
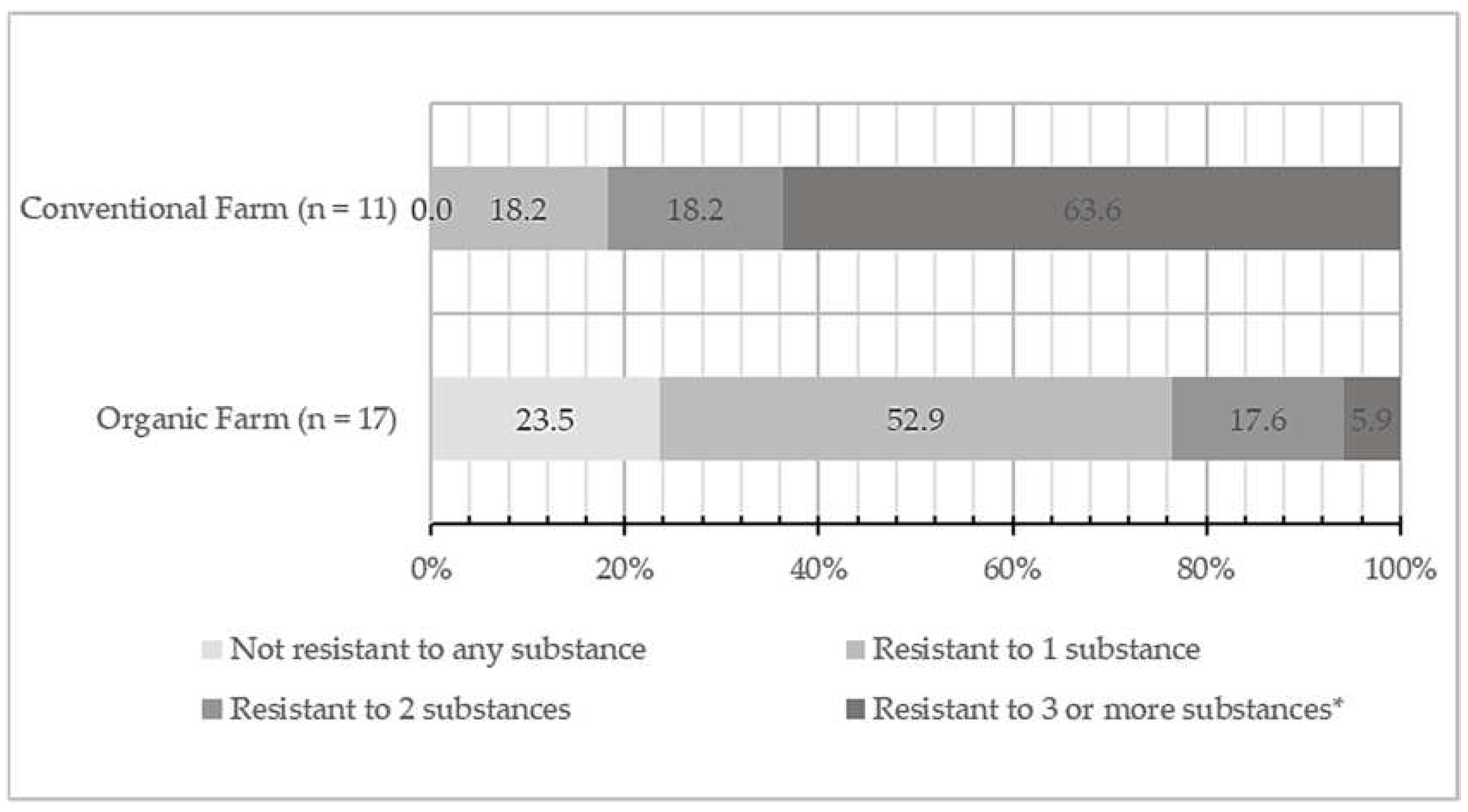
Figure 3.
Agarose gel visualization of M-PCR detecting mecA, mupA, and nuc genes in bovine mastitis milk S. aureus isolates from organic farms (Lanes 2-18; OF1, OF2, OF3 are organic farms 1, 2, and 3, respectively) with negative template control (Lane 19; no dna template added), and nuc gene control Staphylococcus aureus (Lane 1).
Figure 3.
Agarose gel visualization of M-PCR detecting mecA, mupA, and nuc genes in bovine mastitis milk S. aureus isolates from organic farms (Lanes 2-18; OF1, OF2, OF3 are organic farms 1, 2, and 3, respectively) with negative template control (Lane 19; no dna template added), and nuc gene control Staphylococcus aureus (Lane 1).
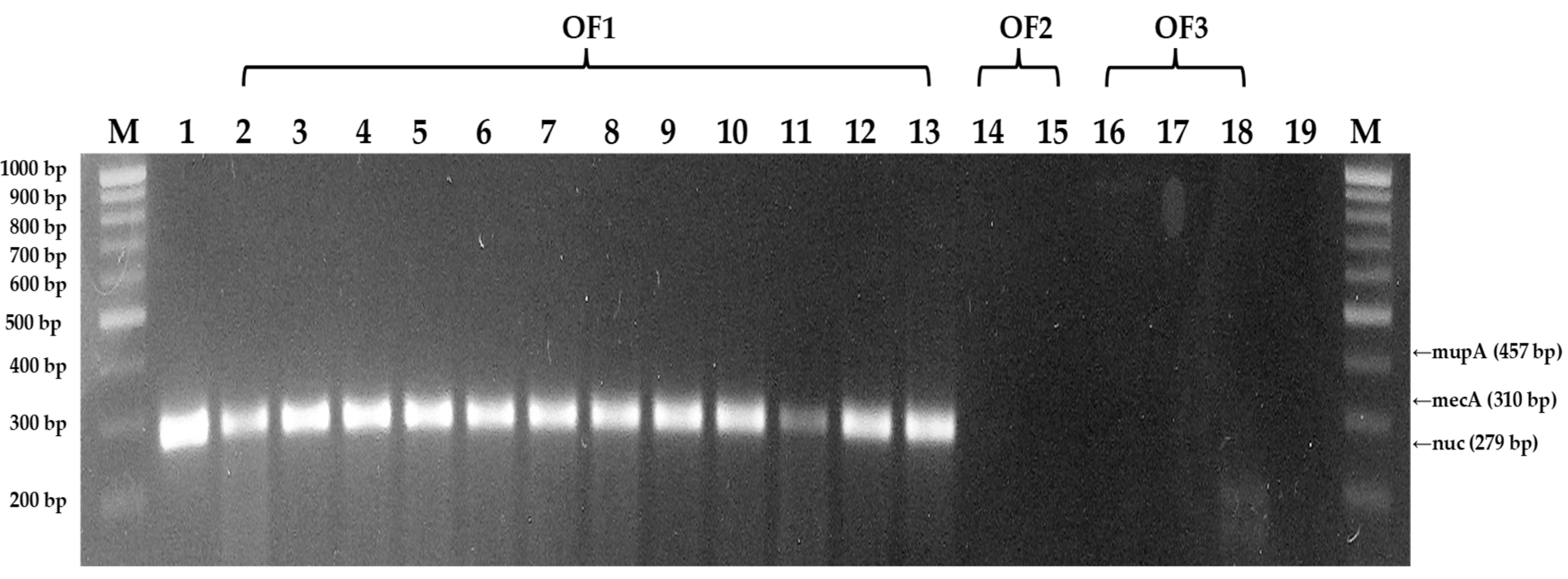
Figure 4.
Agarose gel visualization of M-PCR detecting mecA, mupA, and nuc genes in bovine mastitis milk S. aureus isolates from conventional farms (Lanes 2-12; CF1, CF2, CF3, CF4 are conventional farms 1, 2, 3, and 4, respectively) with negative template control (Lane 13; no dna template added), and nuc gene control Staphylococcus aureus (Lane 1).
Figure 4.
Agarose gel visualization of M-PCR detecting mecA, mupA, and nuc genes in bovine mastitis milk S. aureus isolates from conventional farms (Lanes 2-12; CF1, CF2, CF3, CF4 are conventional farms 1, 2, 3, and 4, respectively) with negative template control (Lane 13; no dna template added), and nuc gene control Staphylococcus aureus (Lane 1).
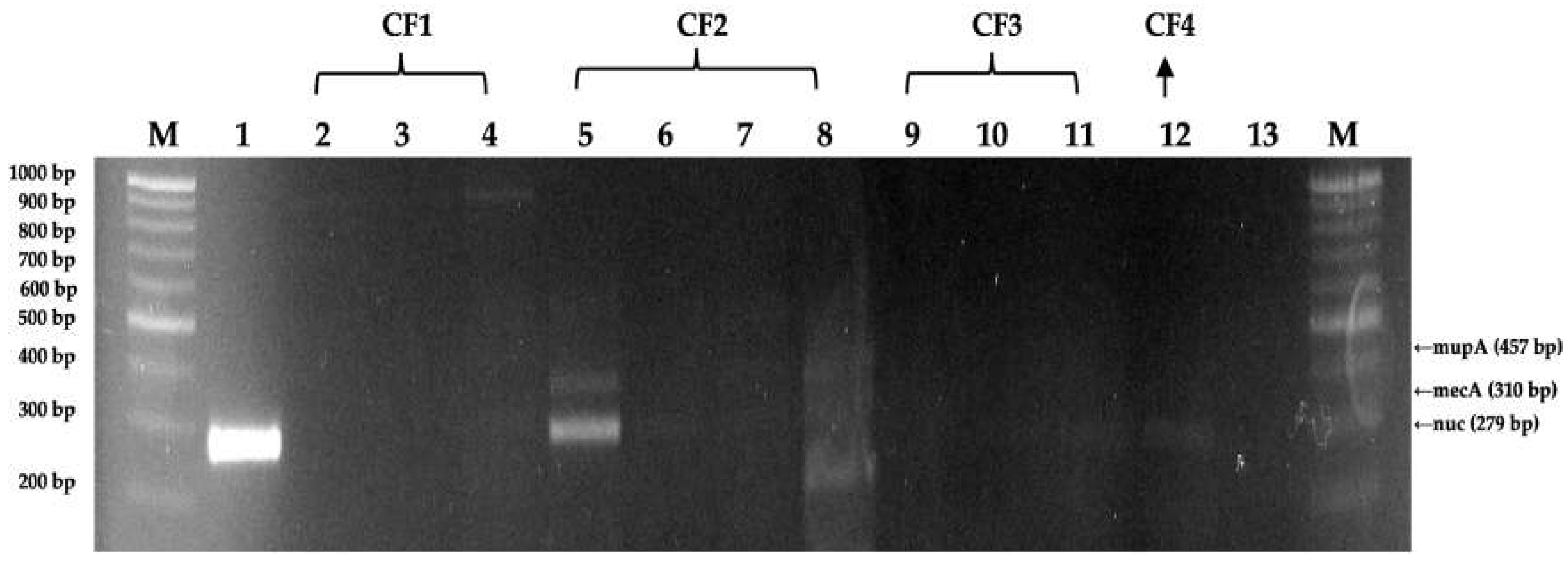
Table 1.
Class, concentrations, and inhibition zones of antibiotics that were used in this study.
| Compound Class | Antibiotic Name | Code | Disc concentration (ug) | Zone diameter (mm) | ||
|---|---|---|---|---|---|---|
| R† | I† | S† | ||||
| β-lactams | Ampicillin | AMP | 10 | 15 | 16-21 | 22 |
| Oxacillin | OX | 10 | 17 | 18-24 | 25 | |
| Aminoglycosides | Gentamicin | CN | 10 | 18 | 19-27 | 28 |
| Glycopeptides | Teicoplanin | TEC | 30 | 14 | 15-21 | 22 |
| Vancomycin | VA | 30 | 16 | 17-21 | 22 | |
| Macrolides | Erythromycin | E | 15 | 21 | 22-30 | 31 |
| Phenicols | Chloramphenicol | C | 10 | 18 | 19-26 | 27 |
| Quinolones | Ciprofloxacin | CIP | 5 | 21 | 22-30 | 31 |
| Tetracyclines | Tetracycline | TE | 30 | 23 | 24-30 | 31 |
† R, I, S means resistant, intermediate, and susceptible, respectively.
Table 2.
Primers used in PCR amplification of S. aureus isolates from bovine mastitis milk in conventional and organic farms.
Table 2.
Primers used in PCR amplification of S. aureus isolates from bovine mastitis milk in conventional and organic farms.
| Target gene | Primer | Oligonucleotide Sequence (5' - 3') | Amplicon Size (bp) |
|---|---|---|---|
| mecA | mecA 1 | GTAGAAATGACTGAACGTCCGATAA | 310 |
| mecA 2 | CCAATTCCACATTGTTTCGGTCTAA | ||
| nuc | nuc 1 | GCGATTGATGGTGATACGGTT | 279 |
| nuc 2 | AGCCAAGCCTTGACGAACTAAAGC | ||
| mupA | mupA 1 | TATATTATGCGATGGAAGGTTGG | 457 |
| mupA 2 | AATAAAATCAGCTGGAAAGTGTTG |
Table 3.
Antimicrobial resistance (number and % of resistant isolate) of coagulase-positive Staphylococcus from conventional and organic farms.
Table 3.
Antimicrobial resistance (number and % of resistant isolate) of coagulase-positive Staphylococcus from conventional and organic farms.
| Antibiotic Name | Code | Conventional Farm (n=11) | Organic Farm (n=17) | Total (n=28) | |||
|---|---|---|---|---|---|---|---|
| Number | % | Number | % | Number | % | ||
| Ampicillin | AMP | 9 | 81.8 | 2 | 11.8 | 11 | 39.3 |
| Oxacillin | OX | 4 | 36.4 | 0 | 0.0 | 4 | 14.3 |
| Gentamicin | CN | 4 | 36.4 | 9 | 52.9 | 13 | 46.4 |
| Teicoplanin | TEC | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| Vancomycin | VA | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| Erythromycin | E | 5 | 45.5 | 1 | 5.9 | 6 | 21.4 |
| Chloramphenicol | C | 2 | 18.2 | 2 | 11.8 | 4 | 14.3 |
| Ciprofloxacin | CIP | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| Tetracycline | TE | 9 | 81.8 | 4 | 23.5 | 13 | 46.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Copyright: This open access article is published under a Creative Commons CC BY 4.0 license, which permit the free download, distribution, and reuse, provided that the author and preprint are cited in any reuse.
MDPI Initiatives
Important Links
© 2024 MDPI (Basel, Switzerland) unless otherwise stated






