1. Introduction
Plastic materials are essential to society due to their wide range of applications. However, the vast production and use of fossil-based non-biodegradable plastics (~99% of all plastics) has raised growing global concerns, including plastic littering and pollution, fossil dependence, and greenhouse gas emissions [
1]. Recently, significant attention has been paid in academia and industry to developing effective plastics recycling technologies, including mechanical, chemical, and biological methods [
2,
3,
4]. Notably, using microbes and enzymes for plastics recycling is attractive because of its milder and environmentally friendly conditions that can enhance sustainability [
5].
Polyethylene terephthalate (PET) is a major plastic material widely used in packaging, beverage bottles, and textiles. The polymer structure consists of terephthalic acid and ethylene glycol units linked by ester bonds. Despite PET's resistance to biodegradation, several cutinase-like enzymes with PET-hydrolase activity have been discovered [
3]. The enzymatic PET-hydrolysis was reported for the first time in 2005. A low crystallinity (9%) PET was depolymerized up to 50% by using a thermostable hydrolase (TfH) from
Thermobifida fusca, at 55°C for three weeks [
6]. Recently, other thermostable hydrolases have shown promising results. Without PET pre-treatment, a cutinase (HiC) from
Humicola insolens has shown equal depolymerizing activity of amorphous and crystalline regions at 45 to 55 °C [
7]. A modified form of compost metagenome-derived cutinase (LCC) depolymerized a pre-treated (amorphized and micronized) PET up to 90% in 10 hours at 72°C [
8]. Recently, a PET-hydrolase (PET2) discovered in a marine metagenomic library that showed optimal activity between 55 and 70°C and higher stability than the LCC at 90°C [
9] attracted our attention for assessing its activity in a series of different aromatic synthetic polymers (
Figure 1).
Apart from PET, many another aromatic terephthalate homo- and copolyesters are also widely used or gaining interest due to their physical and chemical properties. For instance, AkestraTM (Perstorp AB), a new terephthalate copolyester with spiroacetal structures, is an amorphous, durable polyester with potential use in hot-filling packaging [
10,
11]. Similar thermal and mechanical properties have also been achieved by using other rigid cyclic diols (e.g. 1,4-cyclohexanedimethanol, 2,2,4,4-tetramethyl-1,3-cyclobutanediol) in the production of terephthalate copolyesters (e.g. PETG, TritanTM from Eastman). Furthermore, polybutylene terephthalate (PBT), synthesized from TPA (or DMT) and 1,4-butanediol, is used in electrical plug connectors, household devices like irons, and sportswear [
12,
13]. Incorporating flexible adipic acid in the backbone of PBT resulted in a biodegradable polyester, PBAT, which has recently received considerable attention [
14,
15]. Polyhexamethylene terephthalate (PHT) has excellent potential in semiconductor materials and textiles [
16,
17,
18].
Recently, the exploration of new aromatic polyesters with other potentially sustainable aromatic building blocks has been active, such as those with furan, indole, pyrazine, or various lignin-derived aromatics [
19,
20,
21,
22,
23,
24,
25]. Particularly, sugar-based 2,5-furan-dicarboxylic acid (FDCA) has been intensively studied for the production of polyester, namely poly(ethylene furanoate) (PEF), which shows similar thermal and mechanical properties, and even better barrier properties as PET and thus has potential to replace PET in the beverage bottles [
26]. However, the relatively low thermal stability and discolouration during the polymerization of furan-based monomers have been a frequently countered challenge [
27,
28]. A lab-scale synthesis of bottle-grade PEF has been achieved using a complex 3-step procedure [
21]. Many other furan-based polyesters have also been reported on a lab scale [
29]. In parallel to the rapid development of furan-based polyesters, some authors of this paper have been exploring the possibilities of developing polyesters based on indole, a larger aromatic unit. Indole and many indole-derived aromatic molecules widely exist in nature and wastewater streams, which could also be produced by microorganisms [
30]. It makes indole a potentially attractive aromatic building block for developing sustainable polymers. So far, several series of indole-based polyesters with different structures (e.g. flexibility, substitution patterns, type of ester bonds, and others) have been successfully synthesized, and their possibility to be degraded by enzymes has also been preliminarily demonstrated [
31,
32,
33,
34].
This article focuses on fundamental depolymerization studies and not directly on recycling. Therefore, the polymers used as enzyme substrates were prepared in powder form by dissolution-precipitation [
35]. The PET2 activity on different aromatic terephthalate and indole-based polyesters was compared with the
IsPETase activity on the same substrates. PET2, characterized initially as a lipase (lipIAF5-2) [
36], has been recently suggested as another alternative for PET depolymerization, and thermostable mutants with improved activity have been developed [
37]. However, to the best of our knowledge, the PET2 activity on polyesters other than PET is evaluated for the first time in this work.
2. Results and Discussion
2.1. Production of the Recombinant PET2
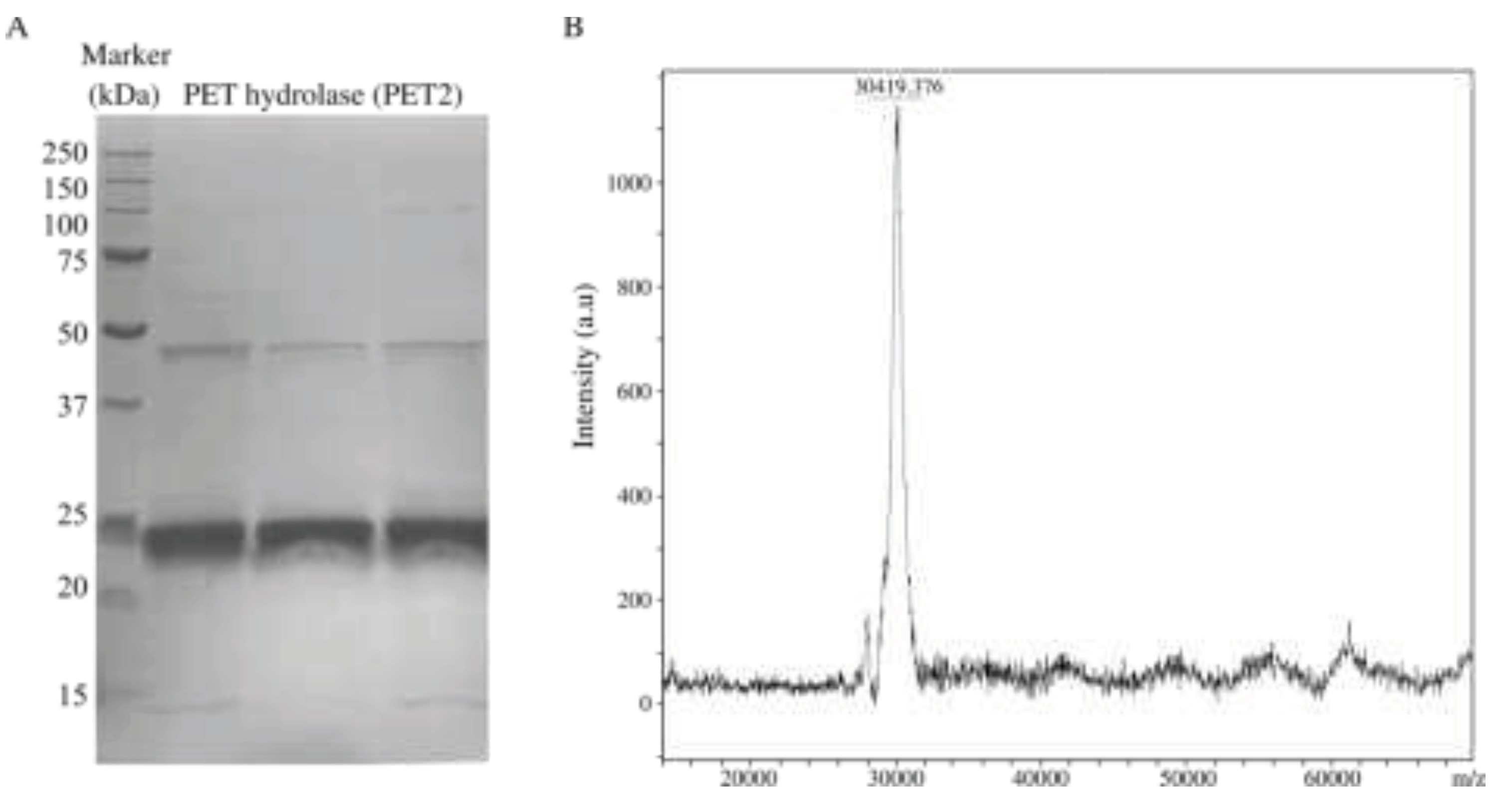
Recombinant PET2 was successfully produced in
E. coli BL21(DE3). The yield was 0.3 g/L, and the estimated purity was more than 93%, according to the SDS-PAGE analysis (
Figure 2A). However, the band observed in the gel, close to 25 kDa, did not match the molecular mass of the PET2. The average theoretical mass of the protein, including the tag in the N-terminal containing six histidines (MGSSHHHHHHSSGLVPRGSH), is 30258 Da. Therefore, the intact mass of the produced protein was analysed by linear MALDI mass spectrometry, giving 30419 Da (
Figure 2B) using external calibration. This result indicates that PET2 was produced at its full length, and the band observed in the gel could be attributed to an anomalous migration.
2.2. PET2 Activity on Terephthalate Aromatic Polyesters
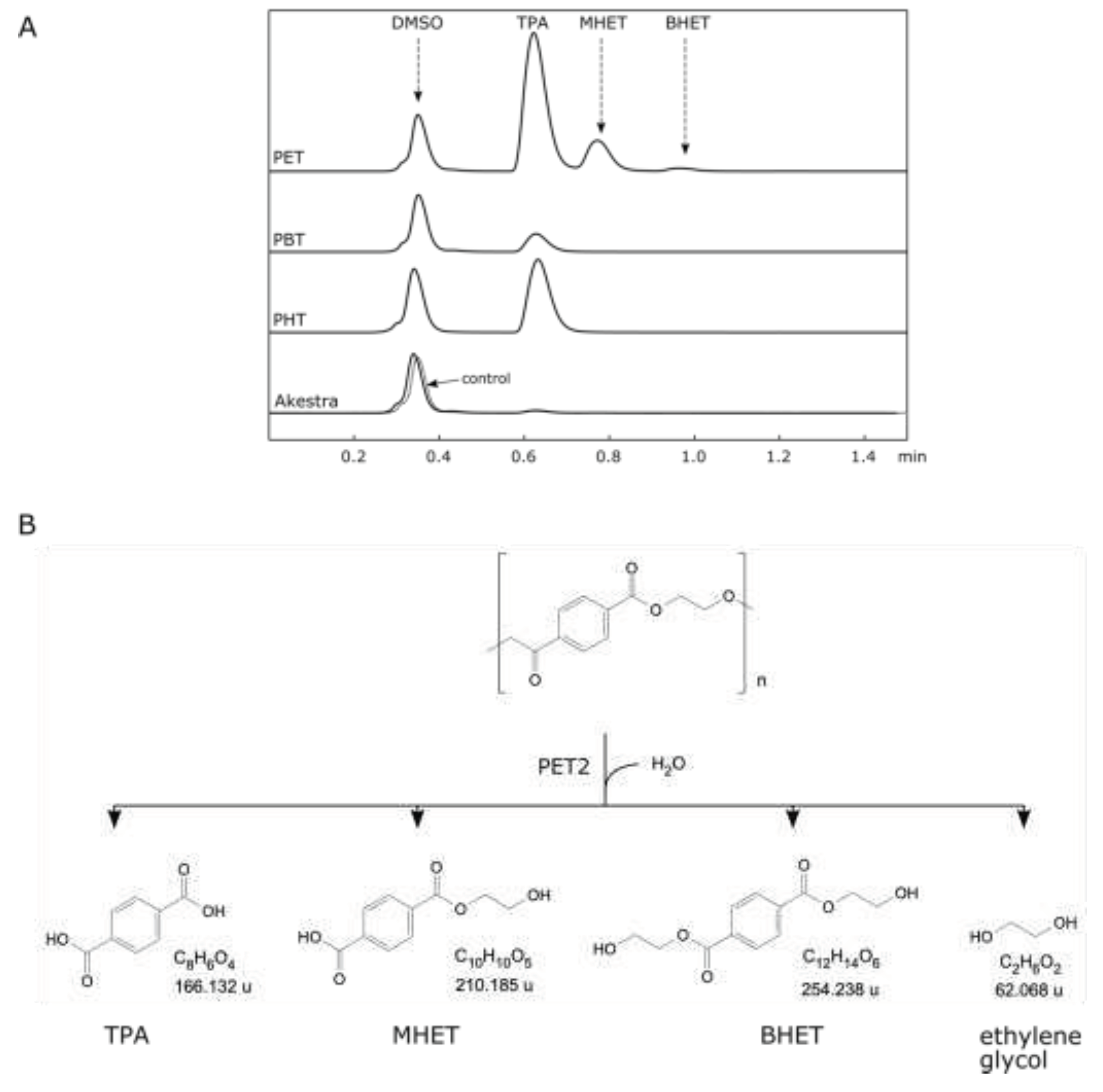
PET2 showed activity on PET, PBT, and PHT, while there was no activity on Akestra
TM. The highest activity was with PET, followed by PHT and PBT (
Figure 3A). Compared to the
IsPETase activity, both enzymes preferred PET with markedly higher
IsPETase activity than PET2 under respective suitable pH and temperature conditions (
Table 1). Both enzymes showed similar product profiles, including TPA, mono-(2-hydroxyethyl) terephthalate (MHET), bis-(2-hydroxyethyl) terephthalate (BHET), and ethylene glycol (
Figure 3B), which was also reported in other work [
37]. However, PET2 showed greater activity than
IsPETase on PBT and PHT. Regarding Akestra,
IsPETase showed low activity while no activity was detected for PET2 (
Table 1), which significatively contrasts with the remarkable activity of HiC cutinase from
Humicola insolens [
38].
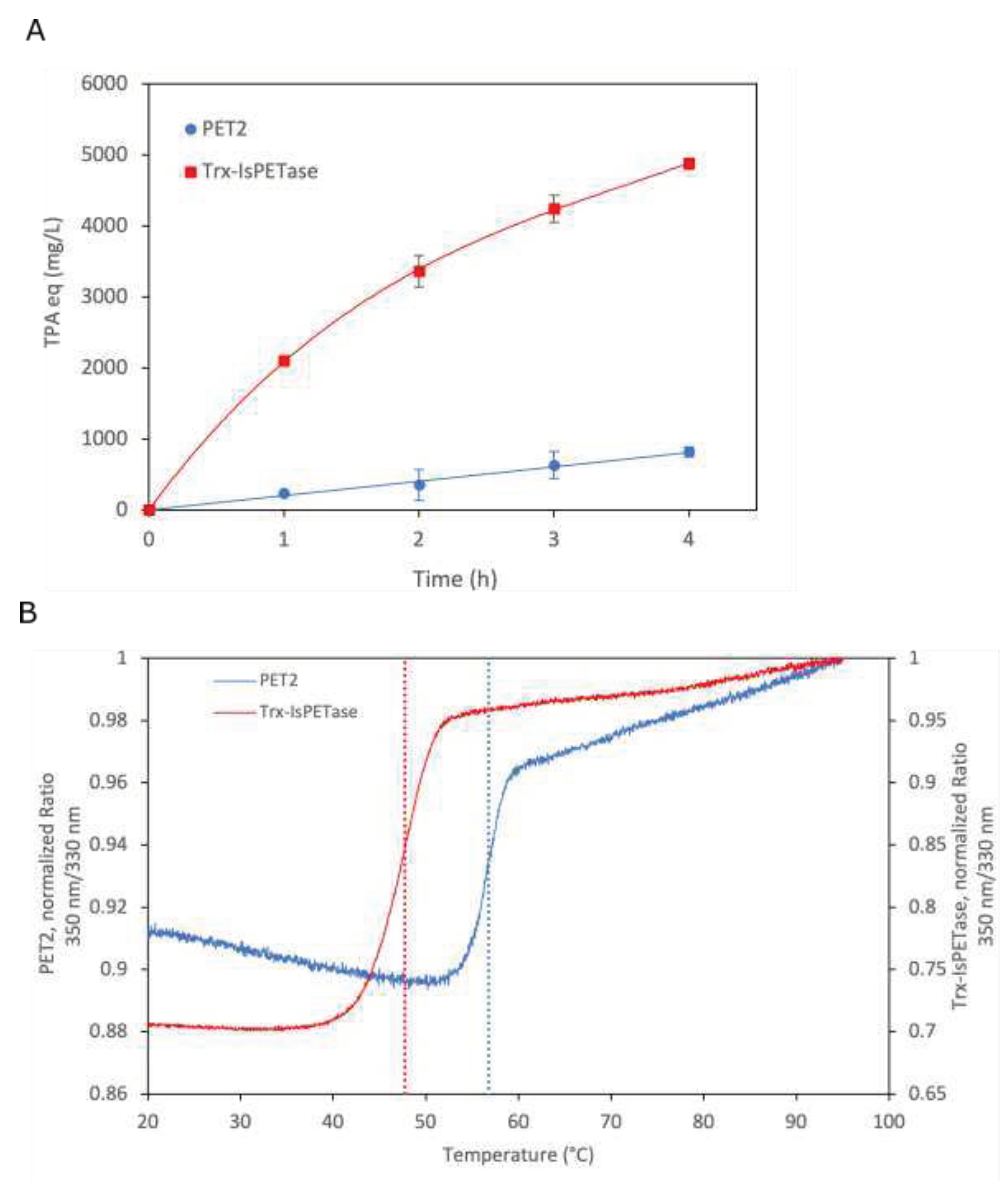
To further investigate the activity of PET2 and Trx-
IsPETase against PET, initial reaction rates were obtained with incubation of a high enzyme/substrate ratio (1.12 mg/mg) for 4 hours. The thioredoxin fusion domain (Trx) has allowed a significative higher yield production of Trx-
IsPETase than
IsPETase and a positive effect on PET depolymerization [
35]. Trx-
IsPETase reached significantly higher activity than PET2, around 5-fold (
Figure 4A). Furthermore, the thermostability of both enzymes was also compared. The DSF analyses have shown melting points (T
m) of 47.7 °C for Trx-
IsPETase, and 56.7 °C for PET2 (
Figure 4B). The T
m for PET2 determined here is significantly lower than the previously reported value in another work, 69°C, by circular dichroism [
37]. This difference can be attributed to several factors, mainly the use of different methods, additional amino acids from the histidine tag in the N-terminal of the construct (headline 2.1), and different buffer compositions, pH 9 here and 7 in the referenced work. The pH was chosen based on another work reporting PET2 preference in alkaline pH, from 8 to 9 [
9]. No effect of calcium was detected by DSF analysis for either of the enzymes, which was consistent with the circular dichroism analysis reported previously [
37]. The determined suitable temperature of 55 °C being so close to the T
m also indicates that the T
m determined by DSF might be underestimated, at least during operational conditions.
2.3. PET2 Activity on Indole-Based Polyesters
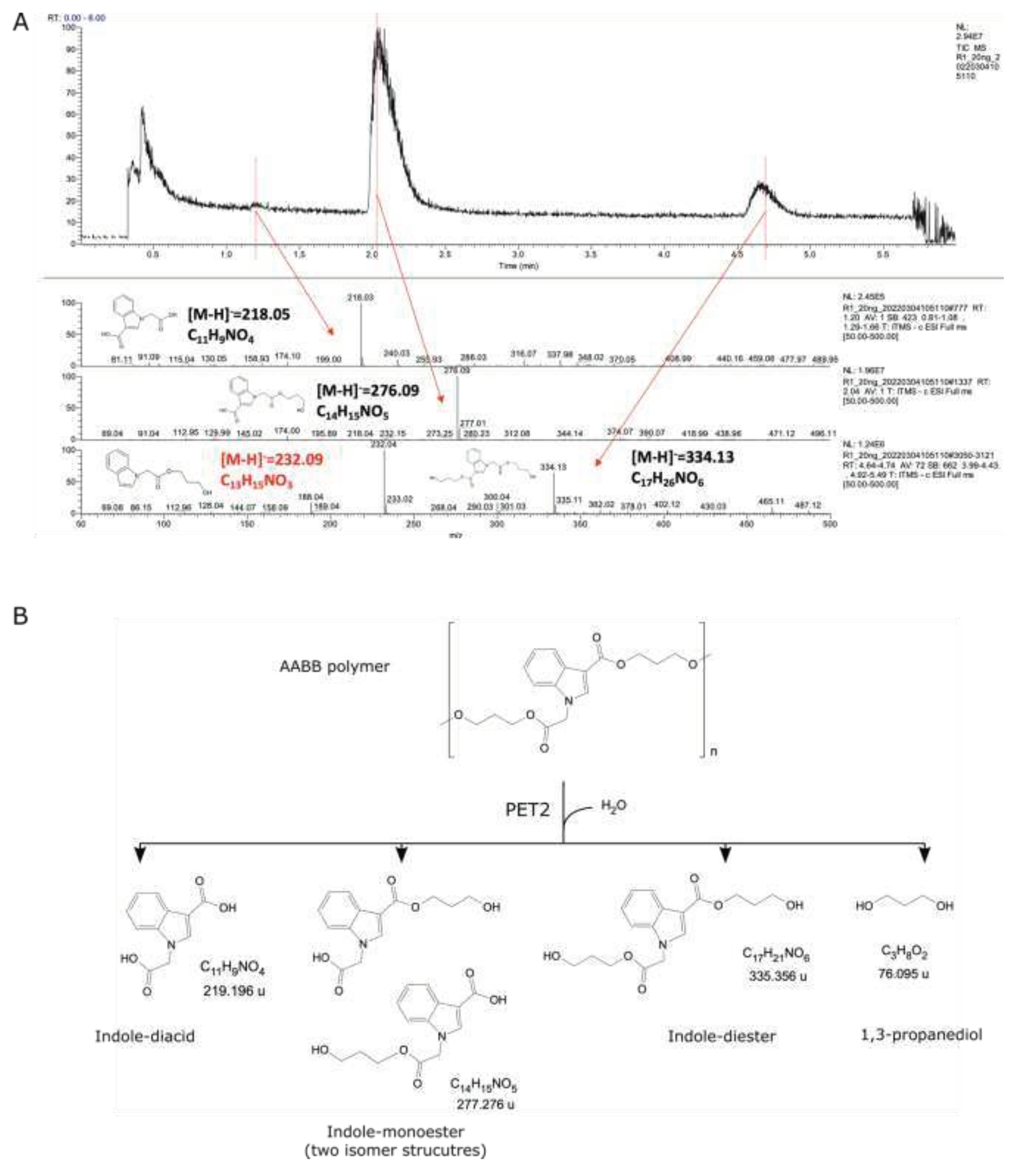
No PET2 activity was detected in polymer AB, while three depolymerization products were detected for AABB by LCMS (
Figure 5). The main product was an indole monoester [2-(3-((3-hydroxypropoxy) carbonyl)-1H-indol-1-yl) acetic acid], followed by an indole-diester [3-hydroxypropyl 1-(2-(3-hydroxypropoxy)-2-oxoethyl)-1H-indole-3-carboxylate], and an indole-diacid [1-(carboxymethyl) indole-3-carboxylic acid], which was under the quantification limit of the HPLC analysis. The depolymerization products were quantified as equivalents in methyl 1-(2-methoxy-2-oxoethyl)-1H-indole-3-carboxylate, assuming that the extinction coefficient of the chromophore tryptophane group is the same as in the AABB depolymerization products. Thus, PET2 activity yielded ~925 mg/L depolymerization products corresponding to ~7 % of the polymer used as a substrate (
Table 1).
2.4. Structural Analysis
PET2 thermostability and its thermostabilized forms were recently reported by Nakamura et al. [
37]. Therefore, the structural analysis performed in this work focuses on comparing the active sites between
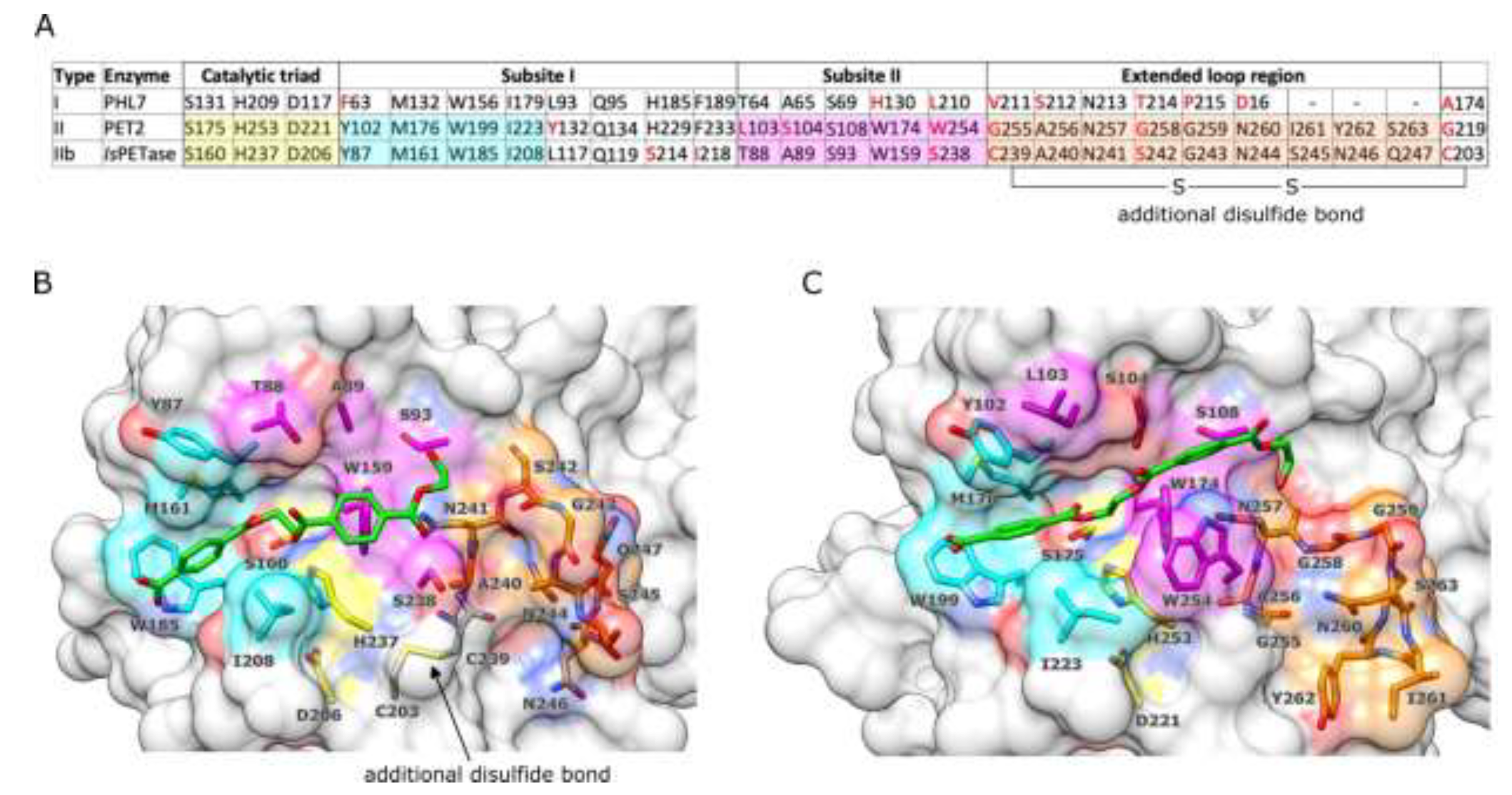
IsPETase and PET2. PET-like enzymes were previously classified into two types, where type II enzymes were further subdivided into IIa and IIb [
39]. This classification is based on a phylogenetic analysis of 69 putative PETases, and a comparative analysis of structures of
IsPETase and cutinases active on PET. The catalytic amino acids Ser, His and Asp are well conserved in all these enzymes, suggesting the same catalytic mechanism, independently of their thermal stability [
40]. Ser is the nucleophile, His is the base, and Asp is the acid. The substrate cleavage point divides the binding cavity into subsites I and II. Type I and II PETases share the same key residues in subsite I but differ in subsite II. Furthermore, type II enzymes have an extended loop region connecting the β-strand 8 with the adjacent α-helix 5 and an additional disulfide bond linking this loop with the neighbouring β-strand 7 [
40,
41].
IsPETase is classified as type IIb due to the presence of a Ser132, while type IIa enzymes have Phe or Tyr instead [
39]. Interestingly, PET2 does not fit very well whiting this classification system. PET2 has an extended loop but not an additional sulfide bond. In addition, PET2 has a Trp instead of Phe or Tyr in type IIa, or Ser in type IIb. It is also remarkable that the His229 of PET2 in the subsite I is also present in type I enzymes, such as PHL7, LCC and TfCut2, while in
IsPETase it is Ser214 (
Figure 6a). More structural and functional studies are needed to update the PETase classification system. Therefore, PET2 can be provisionally classified as a type II PETase.
Dockings of
IsPETase and PET2 with mono-(2-hydroxyethyl) terephthalate dimer (MHET)2 have shown different ligand conformations—the carboxyl-free end MHET unit bound to subsite I, while the glycol-free end MHET unit bound to subsite II. In
IsPETase, the OC-CO dihedral angle of the ethylene glycol (EG) moiety adopted a
gauche conformation. However, interestingly, in PET2, the OC-CO dihedral angle of the EG moiety adopted a
trans conformation (Figures 6b and 6c). NMR studies have shown that
gauche EG moieties predominate in amorphous PET while
trans do it in crystalline PET [
42]. These results, among others, could contribute to explaining the remarkably higher activity of the
IsPETase than PET2 using as a substrate a highly amorphous PET (11.3% crystallinity).
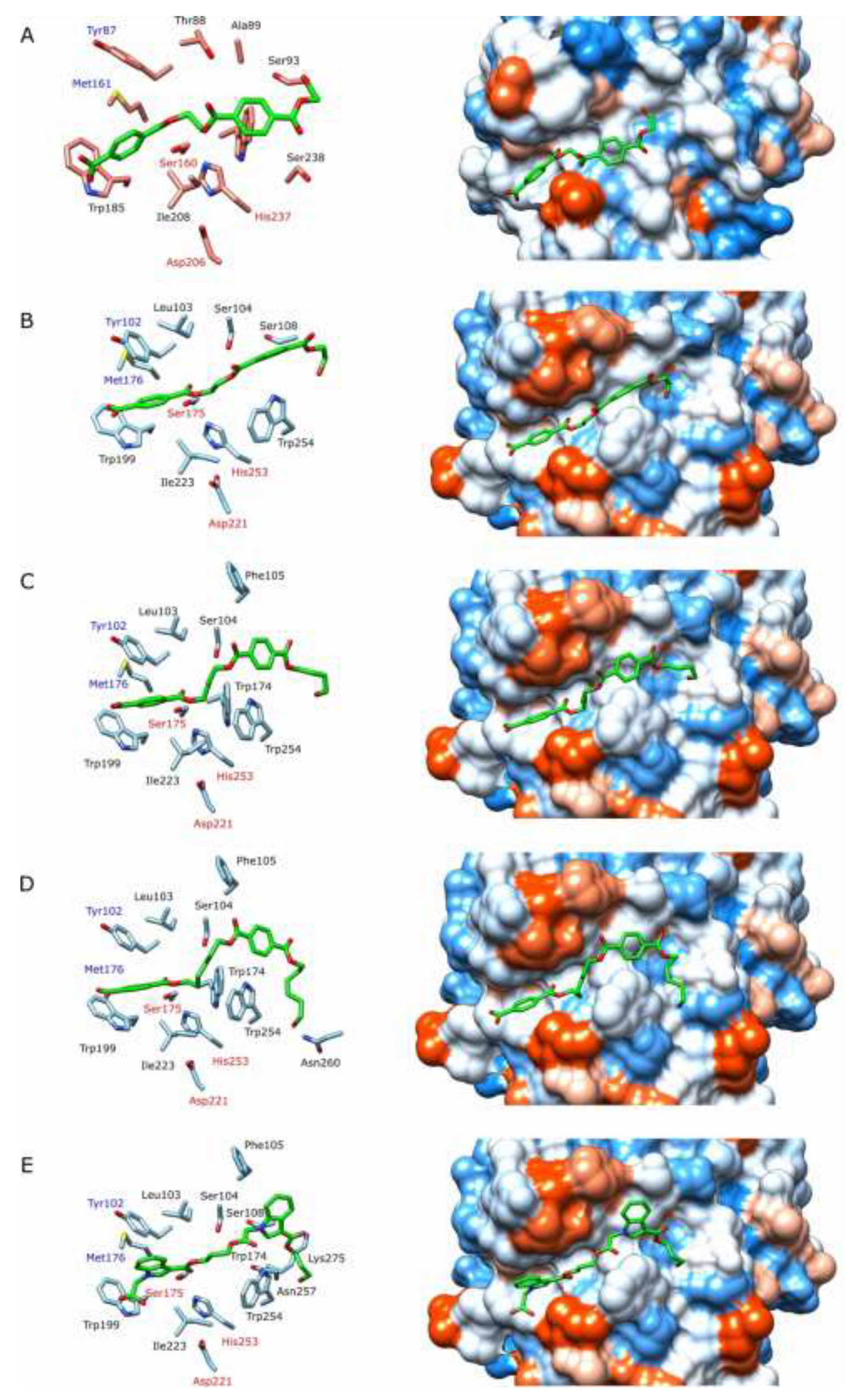
IsPETase and PET2 share 53% of identity when comparing their amino acid sequences, excluding the signal peptides. However, despite these differences, the active sites have several conserved amino acids (
Figure 7). Thus, the catalytic triad in PET2, Ser175, Hist237, and Asp212 corresponds well to the positions of Ser160, His237, and Asp206 in
IsPETase. The amino acids of the proposed oxyanion hole [
39] are also well conserved in both enzymes, Tyr102 and Met176 in PET2 and Tyr87 and Met161 in
IsPETase. Other amino acids which stabilize PET are Trp199 and Ile223 in PET2, corresponding to Trp185 and Ile 208 in
IsPETase [
32,
43]. The Trp has been proposed to make π-π interactions between its indole ring and the PET aromatic ring. These amino acids have shown similar interactions with other aromatic terephthalates (
Figure 7b-7d) and indole (
Figure 7e) dimeric esters used as ligands for docking. The presence of Trp254 and Leu103 in PET2 instead of Ser238 and Thr88 in
IsPETAse, respectively (Figures 5a and 5b), is the main difference between their active sites. The bulkier side group of Trp254 forms a narrower groove in the active site of PET2 than in
IsPETase, which could affect the access of the polymeric substrate giving a lower PET depolymerization yield, which was supported experimentally (
Table 1). It is also consistent with the non-detectable activity on Akestra
TM with PET2, which has a rigid spiroglycol terephthalate linker group (
Table 1). On the other hand, the Leu103 increases the hydrophobicity in PET2 remarkably (Figures 5a and 5b), which could favour the interactions with long flexible terephthalate linkers, such as the diol moieties of PBT, PHT, and AABB (Figures 7c to 7e). Indeed, PET2 gave a higher depolymerization yield than
IsPETase for these substrates (
Table 1). The Phe105 in PET could also contribute to additional hydrophobic interactions with large linker substrates, such as PBT and PHT (
Figure 7c and 6d), or bulkier aromatic groups, such as the indole group of AABB polymers (
Figure 7e). The corresponding amino acid in
IsPETase is Arg90. Although the docking studies can provide valuable information on PET hydrolases/ligands, more experimental support is still needed.
3. Materials and Methods
3.1. Production and Purification of Recombinant PET2 and Trx-IsPETase
A recombinant intracellular PET2-enzyme was produced in the
Escherichia coli BL21(DE3) strain. A synthetic gene encoding PET2 (GenBank accession number ON416993), with optimized codons, was inserted into the vector pET28b between the NdeI and XhoI restriction sites, giving the plasmid pET28b::PET2. The vector backbone provided a sequence encoding a histidine tag at the N-terminus of PET2.
E. coli BL21(DE3) harbouring the plasmid was grown, at 37°C, in 300 mL LB medium supplemented with kanamycin 35 mg/mL as a selection factor. The protein expression was induced with 1 mM IPTG and decreased to 20°C when the optical density (l=600nm) reached 0.6 for 20 hours. Then, the cell pellets were separated by centrifugation at 5600 x
g for 10 minutes and washed with 30 mL of binding buffer (BB). PET2 was purified by immobilized metal affinity chromatography (IMAC) as described in our previous work [
35], using a BB consisting of 100 mM Tris-HCl, 500 and mM NaCl adjusted to pH 7.4, and an elution buffer (EB) with the same composition as BB but with additional 500 mM imidazole, adjusted to pH 7.4. Recombinant Trx-
IsPETase was also produced similarly and purified as described in our earlier work [
35].
3.2. MALDI-TOF/TOF
MS spectrum of intact PET2 protein was acquired using an Autoflex Speed MALDI TOF/TOF mass spectrometer (Bruker Daltonics, Bremen, Germany) in positive linear mode. The protein sample was diluted 1:3 with 0.1 % TFA (Trifluoroacetic acid), then 1 µL of the sample was put on a MALDI target plate together with 0.5 µl matrix solution consisting of 5 mg/ml α-cyano-4-hydroxy cinnamic acid, 80% acetonitrile, 0.1% TFA. The spectrum was externally calibrated using intact BSA from Bruker Daltonics.
3.3. Polymer Preparation
Akestra
TM was a courtesy from Perstorp AB, PET (Ramapet N180) was purchased from Indorama, PBT was purchased from Sigma-Aldrich, and PHT was synthesized according to a previously published protocol [
46]. All the samples were prepared into powders by dissolution-precipitation as described previously [
31,
35]. Changes in crystallinity were assessed by differential scanning calorimetry [
35,
47]. Sample properties are summarized in
Table 2.
3.4. Enzymatic Reactions
The reaction mixtures were prepared in 2 mL vials. They consisted of 20 mg of prepared polymer, 50 mM buffer glycine/NaOH pH 9, 2 mM CaCl2, 10% DMSO, and 0.7 g/L of PET2. All reactions were performed in triplicates, for 72 h at 55°C and under shaking at 200 rpm. A negative control, including all components of the reaction except the enzyme, was incubated in the same conditions for each enzyme.
Reaction rates for PET2 and Trx-
IsPETase were determined over a period of 4 hours. The enzyme concentration was 0.12 g/L, and the substrate was 20 mg PET. The reaction conditions for PET2 were the same as described above, while for Trx-
IsPETase pH 7.4 and 37°C, as described in our previous work [
35]. The samples were taken every hour and analysed by HPLC.
3.5. Reaction Products Analysis
The reaction products were analysed by HPLC and LCMS following the methods previously developed [
35]. Samples of 500 mL were retrieved and diluted to 1:1 with DMSO. Afterwards, the dilutions were filtered and loaded in analytical vials. A hydrophobic column C18 (Kinetex® 1.7 µm XB-C18 100 Å, LC Column 50 x 2.1 mm) was used with operational conditions described in a published paper [
35]. The HPLC used was an Ultimate 3000 RS (Dionex), while the LCMS analysis was performed in an HPLC system (Thermo Fisher Scientific) coupled to an LTQ Velos Pro Ion trap mass spectrum (Thermo Fisher Scientific) using a heated electrospray ionization source (HESI-II) operated in negative mode.
3.6. Differential Scanning Fluorometry
The thermal stability of both PET2 and Trx- IsPETase was evaluated by Differential Scanning Fluorometry (DSF) using the Prometheus NT 48 nano DSF (NanoTemper Technologies GmbH, Munich, Germany). Samples, with or without 1 mM CaCl2, were prepared in 50 µL of a 50 mM buffer, Gly-NaOH for PET2 and phosphate buffer for Trx-IsPETase, and 1 mg/mL of the enzyme. Triplicates of 10 µL were loaded in the instrument capillaries and were heated from 20 to 90°C at a rate of 1°C per minute; then, to monitor protein unfolding, intrinsic fluorescence at emission wavelengths of 330 and 350 nm was analysed.
3.7. Molecular Docking
PET2 was docked with ligands consisting of dimers of mono-(2-hydroxyethyl) terephthalate (MHET)
2, mono-(4-hydroxybuthyl) terephthalate (MHBT)
2, mono-(6-hydroxyhexhyl) terephthalate (MHHT)
2, and (Indole-monoester)
2; and
IsPETase with (MHET)
2. The structure of the wild-type PET2 was obtained by reverse mutation of the crystal structure of the mutant 2M [
37] (PDB ID: 7EC8), using YASARA v21.12.19 [
48]; while the crystal structure 6EQE (PDB ID) of
IsPETase was used as the receptor. All ligands were prepared in the Avogadro program [
49] as described previously [
50]. The molecular geometries were optimized using the force field MMFF94, with the steepest descent algorithm, 5000 steps and convergence of 10
-7. Dockings were performed with AutoDock Vina [
51], considering the total flexibility of the ligands. The interactions between enzymes and ligands were analyzed in UCSF Chimera v.1.15 [
45].
4. Conclusions
Although PET2 is significantly more thermostable than IsPETase, the latter has shown remarkably higher PET-depolymerizing activity. However, interestingly, PET2 has yielded higher depolymerization products from PBT and PHT than IsPETase. While IsPETase was able to depolymerize AkestraTM, PET2 did not produce any detectable degradation products. The highest activity of IsPETase, and the different product profiles suggest that the differences between the active sites of PET2 and IsPETase are more determinant than the thermostability for the depolymerization processes. On the other hand, PET2 is more selective for indole-based polyesters, being able to attack the AABB-type indole-based polyester with 1:1 ratio of aromatic and aliphatic ester groups, while unable to attack the AB-type indole-based polyester with purely aromatic ester groups. Finally, understanding the enzymatic preferences of polymers will contribute to the effective use of enzymes to depolymerize different types of polyesters.
Author Contributions
Conceptualization, J.A.L.-P. and B.Z.; methodology, P.W.-E., L.A.-L., C.T., P.W., K.B. and C.G.; software, P.W.-E. and J.A.L.-P.; validation, P.W.-E., L.A.-L., C.T., P.W., K.B. and C.G.; formal analysis, J.A.L.-P., P.W.-E., L.A.-L., C.T., K.B., C.G. and B.Z.; investigation, J.A.L.-P., P.W.-E., L.A.-L., P.W., C.G. and B.Z.; resources, J.A.L.-P. and B.Z.; writing—original draft preparation, P.W.-E. and J.A.L.-P.; writing, review and editing, all authors; supervision, J.A.L.-P. and B.Z.; project administration, J.A.L.-P.; funding acquisition, J.A.L.-P and B.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by The Crafoord Foundation, grant number 2020-0774, the Swedish Foundation for Strategic Environmental Research (Mistra) through the “STEPS” project (No. 2016/1489), the Swedish Research Council for Sustainable Development FORMAS (No. 2021-01107), and the Royal Physiographic Society in Lund.
Data Availability Statement
The files of the models presented in this study are available on request from the corresponding author. Other data are available in the article.
Acknowledgments
We thank Dr. Maria Gourdon from the Lund Protein Production Platform (LP3) for her assistance in using the DSF instrument. We thank Åsa Halldén Björklund and Linda Zellner from Perstorp AB for providing AkestraTM samples.
Conflicts of Interest
The authors declare no conflict of interest.
Notes
| 1 |
Current affiliation: Department of Chemistry, Technische Universität München, Germany. |
References
- Wilcox, C.; Van Sebille, E.; Hardesty, B.D. Threat of plastic pollution to seabirds is global, pervasive, and increasing. Proceedings of the national academy of sciences 2015, 112, 11899-11904. [CrossRef]
- Kumar, R.; Sadeghi, K.; Jang, J.; Seo, J. Mechanical, chemical, and bio-recycling of biodegradable plastic: A review. Science of The Total Environment 2023, 163446. [CrossRef]
- Mankar, S.V.; Wahlberg, J.; Warlin, N.; Valsange, N.G.; Rehnberg, N.; Lundmark, S.; Jannasch, P.; Zhang, B. Short-Loop Chemical Recycling via Telechelic Polymers for Biobased Polyesters with Spiroacetal Units. ACS Sustain. Chem. Eng. 2023, 11, 5135–5146. [CrossRef]
- Arza, C.R.; Wang, P.; Linares-Pastén, J.; Zhang, B. Synthesis, thermal, rheological characteristics, and enzymatic degradation of aliphatic polyesters with lignin-based aromatic pendant groups. Journal of Polymer Science Part A: Polymer Chemistry 2019, 57, 2314-2323. [CrossRef]
- Nikiema, J.; Asiedu, Z. A review of the cost and effectiveness of solutions to address plastic pollution. Environ. Sci. Pollut. Res. 2022, 29, 24547–24573. [CrossRef]
- Müller, R.-J.; Schrader, H.; Profe, J.; Dresler, K.; Deckwer, W.-D. Enzymatic Degradation of Poly(ethylene terephthalate): Rapid Hydrolyse using a Hydrolase fromT. fusca. Macromol. Rapid Commun. 2005, 26, 1400–1405. [CrossRef]
- Kaabel, S.; Therien, J.P.D.; Deschênes, C.E.; Duncan, D.; Friščić, T.; Auclair, K. Enzymatic depolymerization of highly crystalline polyethylene terephthalate enabled in moist-solid reaction mixtures. Proc. Natl. Acad. Sci. 2021, 118. [CrossRef]
- Tournier, V.; Topham, C.M.; Gilles, A.; David, B.; Folgoas, C.; Moya-Leclair, E.; Kamionka, E.; Desrousseaux, M.-L.; Texier, H.; Gavalda, S.; et al. An engineered PET depolymerase to break down and recycle plastic bottles. Nature 2020, 580, 216–219. [CrossRef]
- Danso, D.; Schmeisser, C.; Chow, J.; Zimmermann, W.; Wei, R.; Leggewie, C.; Li, X.; Hazen, T.; Streit, W.R. New Insights into the Function and Global Distribution of Polyethylene Terephthalate (PET)-Degrading Bacteria and Enzymes in Marine and Terrestrial Metagenomes. Appl. Environ. Microbiol. 2018, 84. [CrossRef]
- Hufendiek, A.; Lingier, S.; Du Prez, F.E. Thermoplastic polyacetals: chemistry from the past for a sustainable future? Polymer Chemistry 2019, 10, 9-33. [CrossRef]
- Hatti-Kaul, R.; Nilsson, L.J.; Zhang, B.; Rehnberg, N.; Lundmark, S. Designing Biobased Recyclable Polymers for Plastics. Trends Biotechnol. 2020, 38, 50–67. [CrossRef]
- Wang, W.; Wu, F.; Lu, H.; Li, X.; Yang, X.; Tu, Y. A Cascade Polymerization Method for the Property Modification of Poly(butylene terephthalate) by the Incorporation of Isosorbide. ACS Appl. Polym. Mater. 2019, 1, 2313–2321. [CrossRef]
- De Vos, L.; Van de Voorde, B.; Van Daele, L.; Dubruel, P.; Van Vlierberghe, S. Poly(alkylene terephthalate)s: From current developments in synthetic strategies towards applications. Eur. Polym. J. 2021, 161, 110840. [CrossRef]
- Peng, B.-Y.; Zhang, X.; Sun, Y.; Liu, Y.; Chen, J.; Shen, Z.; Zhou, X.; Zhang, Y. Biodegradation and Carbon Resource Recovery of Poly (butylene adipate-co-terephthalate)(PBAT) by Mealworms: Removal Efficiency, Depolymerization Pattern, and Microplastic Residue. ACS Sustainable Chemistry & Engineering 2023, 11, 1774-1784. [CrossRef]
- Yang, Y.; Min, J.; Xue, T.; Jiang, P.; Liu, X.; Peng, R.; Huang, J.-W.; Qu, Y.; Li, X.; Ma, N.; et al. Complete bio-degradation of poly(butylene adipate-co-terephthalate) via engineered cutinases. Nat. Commun. 2023, 14, 1–8. [CrossRef]
- Guo, Z.; Warlin, N.; Mankar, S.V.; Sidqi, M.; Andersson, M.; Zhang, B.; Nilsson, E. Development of Circularly Recyclable Low Melting Temperature Bicomponent Fibers toward a Sustainable Nonwoven Application. ACS Sustain. Chem. Eng. 2021, 9, 16778–16785. [CrossRef]
- Qian, K.; Qiao, R.; Chen, S.; Luo, H.; Zhang, D. Enhanced permittivity in polymer blends via tailoring the orderliness of semiconductive liquid crystalline polymers and intermolecular interactions. J. Mater. Chem. C 2020, 8, 8440–8450. [CrossRef]
- Hall, I.; Ibrahim, B. The structure and properties of poly (hexamethylene terephthalate): 1. The preparation, morphology and unit cells of three allomorphs. Polymer 1982, 23, 805-816. [CrossRef]
- Pan, T.; Deng, J.; Xu, Q.; Zuo, Y.; Guo, Q.X.; Fu, Y. Catalytic Conversion of Furfural into a 2, 5-Furandicarboxylic Acid-Based Polyester with Total Carbon Utilization. ChemSusChem 2013, 6, 47-50. [CrossRef]
- Amarasekara, A.S.; Nguyen, L.H.; Okorie, N.C.; Jamal, S.M. A two-step efficient preparation of a renewable dicarboxylic acid monomer 5, 5′-[oxybis (methylene)] bis [2-furancarboxylic acid] from d-fructose and its application in polyester synthesis. Green Chemistry 2017, 19, 1570-1575. [CrossRef]
- Rosenboom, J.-G.; Hohl, D.K.; Fleckenstein, P.; Storti, G.; Morbidelli, M. Bottle-grade polyethylene furanoate from ring-opening polymerisation of cyclic oligomers. Nat. Commun. 2018, 9, 1–7. [CrossRef]
- Wang, G.; Xu, Y.; Jiang, M.; Wang, R.; Wang, H.; Liang, Y.; Zhou, G. Fully bio-based polyesters poly (ethylene-co-1, 5-pentylene 2, 5-thiophenedicarboxylate) s (PEPTs) with high toughness: Synthesis, characterization and thermo-mechanical properties. Polymer 2020, 204, 122800. [CrossRef]
- Nguyen, H.T.H.; Reis, M.H.; Qi, P.; Miller, S.A. Polyethylene ferulate (PEF) and congeners: polystyrene mimics derived from biorenewable aromatics. Green Chem. 2015, 17, 4512–4517. [CrossRef]
- Gioia, C.; Banella, M.B.; Marchese, P.; Vannini, M.; Colonna, M.; Celli, A. Advances in the synthesis of bio-based aromatic polyesters: novel copolymers derived from vanillic acid and ε-caprolactone. Polym. Chem. 2016, 7, 5396–5406. [CrossRef]
- Wurdemann, M.A.; Bernaerts, K.V. Biobased Pyrazine-Containing Polyesters. ACS Sustainable Chemistry & Engineering 2020, 8, 12045-12052. [CrossRef]
- Burgess, S.K.; Karvan, O.; Johnson, J.R.; Kriegel, R.M.; Koros, W.J. Oxygen sorption and transport in amorphous poly(ethylene furanoate). Polymer 2014, 55, 4748–4756. [CrossRef]
- Terzopoulou, Z.; Karakatsianopoulou, E.; Kasmi, N.; Tsanaktsis, V.; Nikolaidis, N.; Kostoglou, M.; Papageorgiou, G.Z.; Lambropoulou, D.A.; Bikiaris, D.N. Effect of catalyst type on molecular weight increase and coloration of poly(ethylene furanoate) biobased polyester during melt polycondensation. Polym. Chem. 2017, 8, 6895–6908. [CrossRef]
- Loos, K.; Zhang, R.; Pereira, I.; Agostinho, B.; Hu, H.; Maniar, D.; Sbirrazzuoli, N.; Silvestre, A.J.D.; Guigo, N.; Sousa, A.F. A Perspective on PEF Synthesis, Properties, and End-Life. Front. Chem. 2020, 8, 585. [CrossRef]
- Tsanaktsis, V.; Terzopoulou, Z.; Exarhopoulos, S.; Bikiaris, D.N.; Achilias, D.S.; Papageorgiou, D.G.; Papageorgiou, G.Z. Sustainable, eco-friendly polyesters synthesized from renewable resources: preparation and thermal characteristics of poly(dimethyl-propylene furanoate). Polym. Chem. 2015, 6, 8284–8296. [CrossRef]
- Ma, Q.; Zhang, X.; Qu, Y. Biodegradation and Biotransformation of Indole: Advances and Perspectives. Front. Microbiol. 2018, 9, 2625. [CrossRef]
- Wang, P.; Arza, C.R.; Zhang, B. Indole as a new sustainable aromatic unit for high quality biopolyesters. Polym. Chem. 2018, 9, 4706–4710. [CrossRef]
- Wang, P.; Linares-Pastén, J.A.; Zhang, B. Synthesis, Molecular Docking Simulation, and Enzymatic Degradation of AB-Type Indole-Based Polyesters with Improved Thermal Properties. Biomacromolecules 2020, 21, 1078–1090. [CrossRef]
- Arza, C.R.; Zhang, B. Synthesis, Thermal Properties, and Rheological Characteristics of Indole-Based Aromatic Polyesters. ACS Omega 2019, 4, 15012–15021. [CrossRef]
- Wang, P.; Zhang, B. Sustainable aromatic polyesters with 1,5-disubstituted indole units. RSC Adv. 2021, 11, 16480–16489. [CrossRef]
- Wagner-Egea, P.; Tosi, V.; Wang, P.; Grey, C.; Zhang, B.; Linares-Pastén, J.A. Assessment of IsPETase-Assisted Depolymerization of Terephthalate Aromatic Polyesters and the Effect of the Thioredoxin Fusion Domain. Applied Sciences 2021, 11, 8315. [CrossRef]
- Meilleur, C.; Hupé, J.-F.; Juteau, P.; Shareck, F. Isolation and characterization of a new alkali-thermostable lipase cloned from a metagenomic library. J. Ind. Microbiol. Biotechnol. 2009, 36, 853–861. [CrossRef]
- Nakamura, A.; Kobayashi, N.; Koga, N.; Iino, R. Positive Charge Introduction on the Surface of Thermostabilized PET Hydrolase Facilitates PET Binding and Degradation. ACS Catal. 2021, 11, 8550–8564. [CrossRef]
- Aristizábal-Lanza, L.; Mankar, S.V.; Tullberg, C.; Zhang, B.; Linares-Pastén, J.A. Comparison of the enzymatic depolymerization of polyethylene terephthalate and AkestraTM using Humicola insolens cutinase. Front. Chem. Eng. 2022, 4. [CrossRef]
- Joo, S.; Lee, S.; Kim, K. Structural insight into molecular mechanism of poly(ethylene terephthalate) degradation. FEBS J. 2017, 284, 309–309. [CrossRef]
- Boneta, S.; Arafet, K.; Moliner, V. QM/MM Study of the Enzymatic Biodegradation Mechanism of Polyethylene Terephthalate. J. Chem. Inf. Model. 2021, 61, 3041–3051. [CrossRef]
- Richter, P.K.; Blázquez-Sánchez, P.; Zhao, Z.; Engelberger, F.; Wiebeler, C.; Künze, G.; Frank, R.; Krinke, D.; Frezzotti, E.; Lihanova, Y.; et al. Structure and function of the metagenomic plastic-degrading polyester hydrolase PHL7 bound to its product. Nat. Commun. 2023, 14, 1–11. [CrossRef]
- Wei, R.; Song, C.; Gräsing, D.; Schneider, T.; Bielytskyi, P.; Böttcher, D.; Matysik, J.; Bornscheuer, U.T.; Zimmermann, W. Conformational fitting of a flexible oligomeric substrate does not explain the enzymatic PET degradation. Nat. Commun. 2019, 10, 1–4. [CrossRef]
- Sagong, H.-Y.; Seo, H.; Kim, T.; Son, H.F.; Joo, S.; Lee, S.H.; Kim, S.; Woo, J.-S.; Hwang, S.Y.; Kim, K.-J. Decomposition of the PET Film by MHETase Using Exo-PETase Function. ACS Catal. 2020, 10, 4805–4812. [CrossRef]
- Kyte, J.; Doolittle, R.F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 1982, 157, 105–132. [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera?A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [CrossRef]
- Warlin, N.; Gonzalez, M.N.G.; Mankar, S.; Valsange, N.G.; Sayed, M.; Pyo, S.-H.; Rehnberg, N.; Lundmark, S.; Hatti-Kaul, R.; Jannasch, P.; et al. A rigid spirocyclic diol from fructose-based 5-hydroxymethylfurfural: synthesis, life-cycle assessment, and polymerization for renewable polyesters and poly(urethane-urea)s. Green Chem. 2019, 21, 6667–6684. [CrossRef]
- Din, S.U.; Kalsoom; Satti, S.M.; Uddin, S.; Mankar, S.V.; Ceylan, E.; Hasan, F.; Khan, S.; Badshah, M.; Beldüz, A.O.; et al. The Purification and Characterization of a Cutinase-like Enzyme with Activity on Polyethylene Terephthalate (PET) from a Newly Isolated Bacterium Stenotrophomonas maltophilia PRS8 at a Mesophilic Temperature. Appl. Sci. 2023, 13, 3686. [CrossRef]
- Krieger, E.; Koraimann, G.; Vriend, G. Increasing the precision of comparative models with YASARA NOVA-a self-parameterizing force field. Proteins 2002, 47, 393–402. [CrossRef]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [CrossRef]
- Li, X.; İlk, S.; Linares-Pastén, J.A.; Liu, Y.; Raina, D.B.; Demircan, D.; Zhang, B. Synthesis, Enzymatic Degradation, and Polymer-Miscibility Evaluation of Nonionic Antimicrobial Hyperbranched Polyesters with Indole or Isatin Functionalities. Biomacromolecules 2021, 22, 2256–2271. [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [CrossRef]
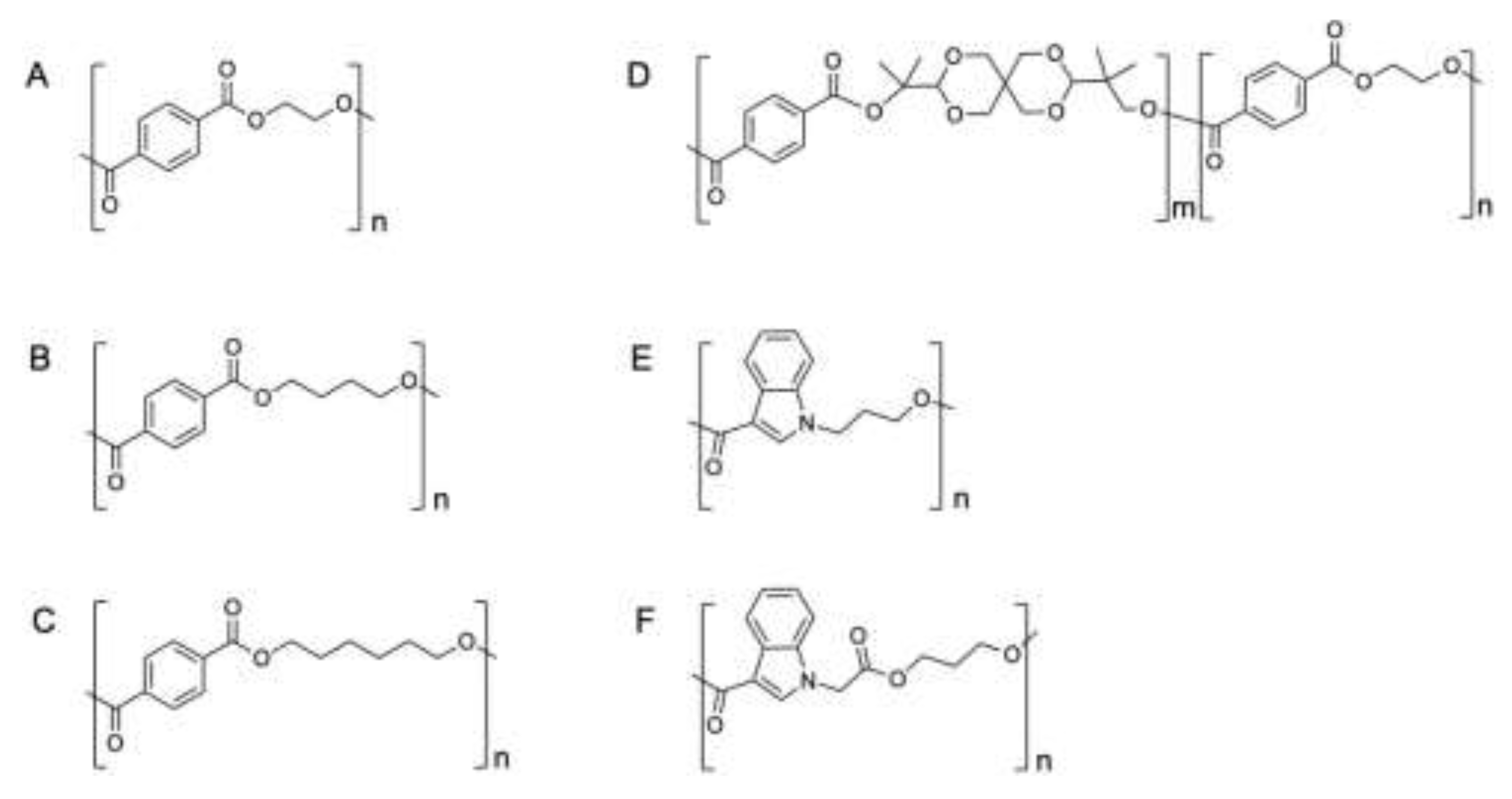
Figure 1.
Assessed polyesters as potential substrates for the PET2 depolymerase. (A) PET, (B) PBT, (C) PHT, (D) AkestraTM, (E) AB (aromatic) and (F) AABB (aliphatic aromatic).
Figure 1.
Assessed polyesters as potential substrates for the PET2 depolymerase. (A) PET, (B) PBT, (C) PHT, (D) AkestraTM, (E) AB (aromatic) and (F) AABB (aliphatic aromatic).
Figure 2.
Purification and molecular weight analysis of PET2. (A) SDS-PAGE, notice that PET2 looks a bit smaller than 25 kDa, which is attributed to an anomalous SDS-PAGE migration. (B) Linear MALDI Mass spectrometry analysis of the same sample, intact PET2. Notice that the molecular weight is 30 419 Da, close to the theoretical determined mass of 30 258 Da.
Figure 2.
Purification and molecular weight analysis of PET2. (A) SDS-PAGE, notice that PET2 looks a bit smaller than 25 kDa, which is attributed to an anomalous SDS-PAGE migration. (B) Linear MALDI Mass spectrometry analysis of the same sample, intact PET2. Notice that the molecular weight is 30 419 Da, close to the theoretical determined mass of 30 258 Da.
Figure 3.
PET2 depolymerization activity on terephthalate polyesters. (A) HPLC analysis of PET2 depolymerization of terephthalate polyesters after 72 h: polyethylene terephthalate (PET), polybutylene terephthalate (PBT), polyhexamethylene terephthalate (PHT), and Akestra™. Notice that the tiny peak corresponding to TPA in the Akestra-depolymerization chromatogram is also present in the control. (B) PET depolymerization reaction. Notice that the ethylene glycol was not analysed, but it is a predicted product.
Figure 3.
PET2 depolymerization activity on terephthalate polyesters. (A) HPLC analysis of PET2 depolymerization of terephthalate polyesters after 72 h: polyethylene terephthalate (PET), polybutylene terephthalate (PBT), polyhexamethylene terephthalate (PHT), and Akestra™. Notice that the tiny peak corresponding to TPA in the Akestra-depolymerization chromatogram is also present in the control. (B) PET depolymerization reaction. Notice that the ethylene glycol was not analysed, but it is a predicted product.
Figure 4.
Compared reaction rates and melting points of PET2 and Trx-IsPETase. (A) Reaction rates of PET2 at 55°C and Trx-IsPETase at 37°C. The enzyme/substrate ratio was 0.12 mg/mg. (B) Melting points of PET2 and Trx-IsPETase; dotted lines represent melting points at 56.7 and 47.7°C, respectively.
Figure 4.
Compared reaction rates and melting points of PET2 and Trx-IsPETase. (A) Reaction rates of PET2 at 55°C and Trx-IsPETase at 37°C. The enzyme/substrate ratio was 0.12 mg/mg. (B) Melting points of PET2 and Trx-IsPETase; dotted lines represent melting points at 56.7 and 47.7°C, respectively.
Figure 5.
PET2 depolymerization activity on AABB polyester. (A) LCMS analysis of depolymerization products. (B) Proposed reaction based on the LCMS analysis. Notice that the 1,3-propanediol was not analysed, but it is a predicted product from the AABB molecular structure. The molecule marked in red is not expected to be a main product based on the chemistry.
Figure 5.
PET2 depolymerization activity on AABB polyester. (A) LCMS analysis of depolymerization products. (B) Proposed reaction based on the LCMS analysis. Notice that the 1,3-propanediol was not analysed, but it is a predicted product from the AABB molecular structure. The molecule marked in red is not expected to be a main product based on the chemistry.
Figure 6.
Subsites of IsPETase and PET2. (A) Alignments of structurally equivalent amino acids of IsPETase, PET2, and PHL7 (PDB ID: 8BRA). The catalytic triad is conserved in all enzyme types. The amino acids in subsite I that directly interact with the ligand are conserved (highlighted in cyan), while those in subsite II vary in several positions (in red). The extended loop is present in IsPETase and PET2 (type II enzymes), while not in PHL7 (type I). PET2 lacks an additional disulfide bond. (B) Surface representation of the IsPETase/(MHET)2 complex and (C) PET2/(MHET)2. The ligand is represented by sticks (carbons in green and oxygens in red). Note that (MHET)2 is in a gauche conformation in IsPETase, whereas it is in trans in PET2. Catalytic amino acids are in yellow, subsite I amino acids are cyan, subsite II amino acids are magenta, and the extended loop is in orange.
Figure 6.
Subsites of IsPETase and PET2. (A) Alignments of structurally equivalent amino acids of IsPETase, PET2, and PHL7 (PDB ID: 8BRA). The catalytic triad is conserved in all enzyme types. The amino acids in subsite I that directly interact with the ligand are conserved (highlighted in cyan), while those in subsite II vary in several positions (in red). The extended loop is present in IsPETase and PET2 (type II enzymes), while not in PHL7 (type I). PET2 lacks an additional disulfide bond. (B) Surface representation of the IsPETase/(MHET)2 complex and (C) PET2/(MHET)2. The ligand is represented by sticks (carbons in green and oxygens in red). Note that (MHET)2 is in a gauche conformation in IsPETase, whereas it is in trans in PET2. Catalytic amino acids are in yellow, subsite I amino acids are cyan, subsite II amino acids are magenta, and the extended loop is in orange.
Figure 7.
Docking of different ligands (represented in green) into PET2 and
IsPETase. The hydrophobicity surfaces of the active sites are represented according to the Kyte-Doolittle scale, ranging from dodger blue for the most hydrophilic to white and to orange-red for the most hydrophobic [
44]. Catalytic amino acids are labelled in red, while the suggested amino acids that stabilize the oxyanion intermediate are in blue. (A) (MHET)
2/
IsPETase (PDB ID:6EQE). (B) (MHET)
2/PET2. (C) (MHBT)
2/PET2. (D) (MHHT)
2/PET2. (E) (Indole-monoester)
2/PET2. Figure elaborated in UCSF Chimera v.1.15 [
45].
Figure 7.
Docking of different ligands (represented in green) into PET2 and
IsPETase. The hydrophobicity surfaces of the active sites are represented according to the Kyte-Doolittle scale, ranging from dodger blue for the most hydrophilic to white and to orange-red for the most hydrophobic [
44]. Catalytic amino acids are labelled in red, while the suggested amino acids that stabilize the oxyanion intermediate are in blue. (A) (MHET)
2/
IsPETase (PDB ID:6EQE). (B) (MHET)
2/PET2. (C) (MHBT)
2/PET2. (D) (MHHT)
2/PET2. (E) (Indole-monoester)
2/PET2. Figure elaborated in UCSF Chimera v.1.15 [
45].
Table 1.
Depolymerization of different terephthalate and indole-based polyesters after 72 hours with PET2 at 55°C, pH 9, and IsPETase at 37°C, pH 7. The enzyme/substrate ratio was 0.07 mg/mg.
Table 1.
Depolymerization of different terephthalate and indole-based polyesters after 72 hours with PET2 at 55°C, pH 9, and IsPETase at 37°C, pH 7. The enzyme/substrate ratio was 0.07 mg/mg.
| |
PET2 |
IsPETase [35] |
| Polymer |
Degradation products (mg/L) |
Depolymerization (%) |
Degradation products (mg/L) |
Depolymerization (%) |
|
| PET |
1965 |
19.66 |
3934 |
39.34 |
|
| PBT |
138 |
1.38 |
25 |
0.25 |
|
| PHT |
974 |
9.74 |
12.5 |
0.13 |
|
| AkestraTM
|
N.D. |
N.D. |
13.3 |
0.13 |
|
| AB |
N.D. |
N.D. |
|
|
|
| AABB |
925 |
7.01 |
N.Q. |
N.Q. |
|
Table 2.
Properties of the polymers used before dissolution-precipitation, and crystallinity determined after dissolution-precipitation.
Table 2.
Properties of the polymers used before dissolution-precipitation, and crystallinity determined after dissolution-precipitation.
| Polymer |
IV* (dL/g) |
Tg (°C) |
Tm (°C) |
Mn (g/mol) |
Mw(g/mol) |
Crystallinity(%) |
Reference |
| PET |
~0.8 |
~78 |
~245 |
|
|
11.3 |
[35] |
| PBT |
|
|
~203 |
~38000 |
|
38.6 |
[35] |
| PHT |
~1.26 |
~17 |
~140 |
~26000 |
~43000 |
n.d. |
[35] |
| AkestraTM
|
~0.64 |
~95 |
- |
|
|
Amorphous |
[35] |
| AA |
|
102 |
- |
~8100 |
~14500 |
Amorphous |
[32] |
| AABB |
|
93 |
- |
~35800 |
~71700 |
Amorphous |
[31] |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).