Submitted:
30 June 2023
Posted:
03 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
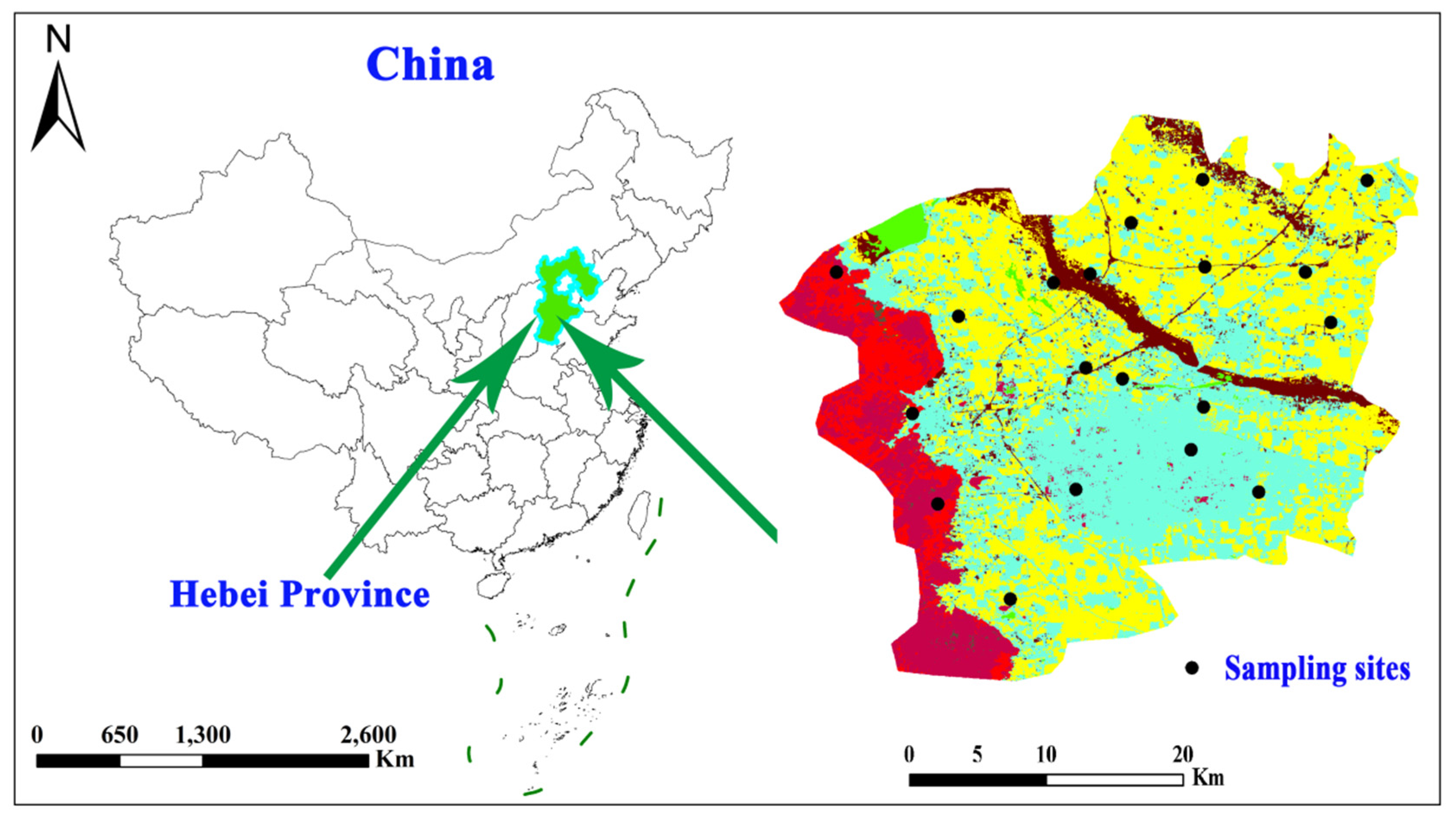
2.1. Study area overview
2.2. Geological and hydrogeological conditions
2.3. Data sources and research methods
2.3.1. Data sources
2.3.2. Research methods
3. Results and Discussion
3.1. Analysis of groundwater chemical composition characteristics
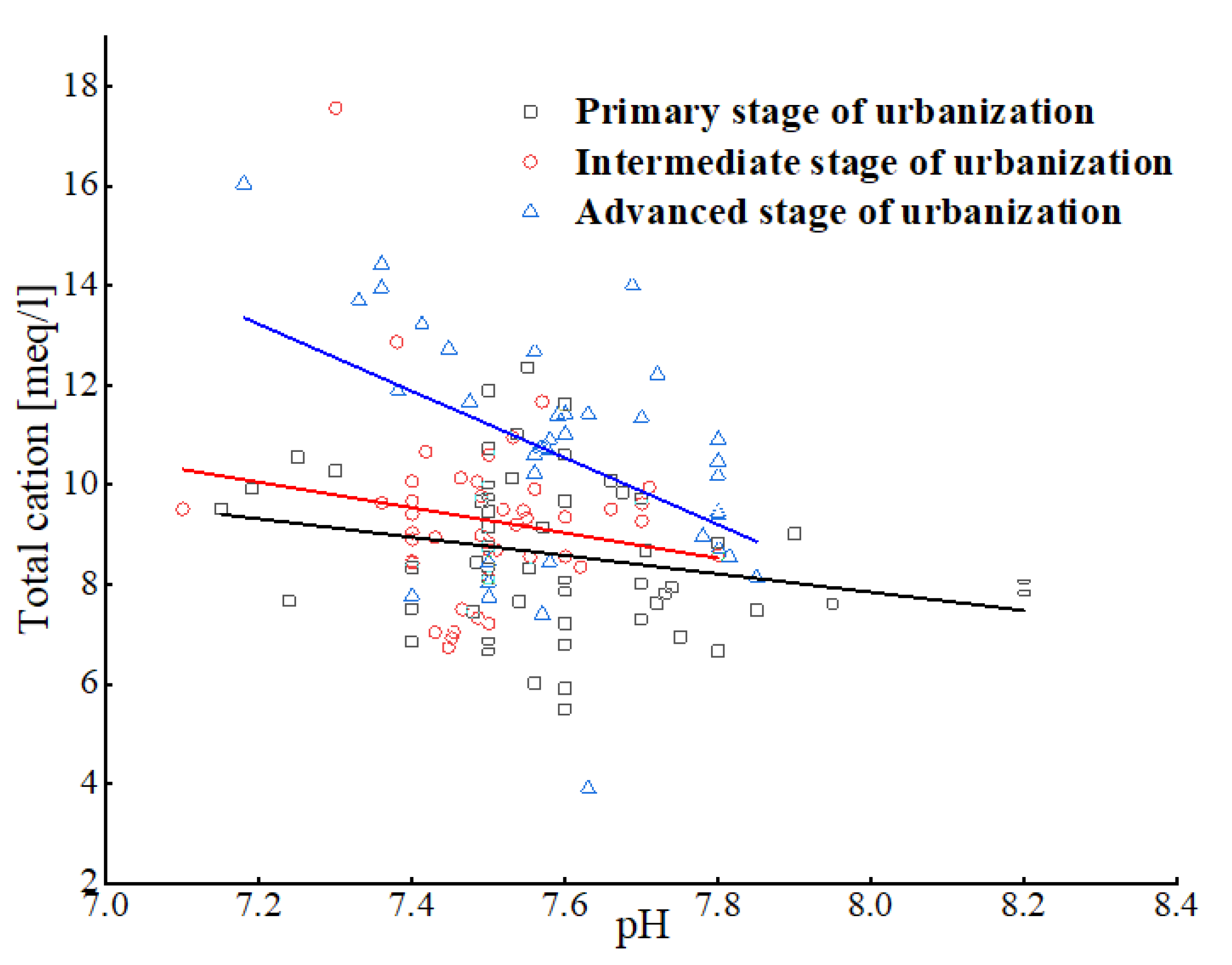
3.1.1. pH
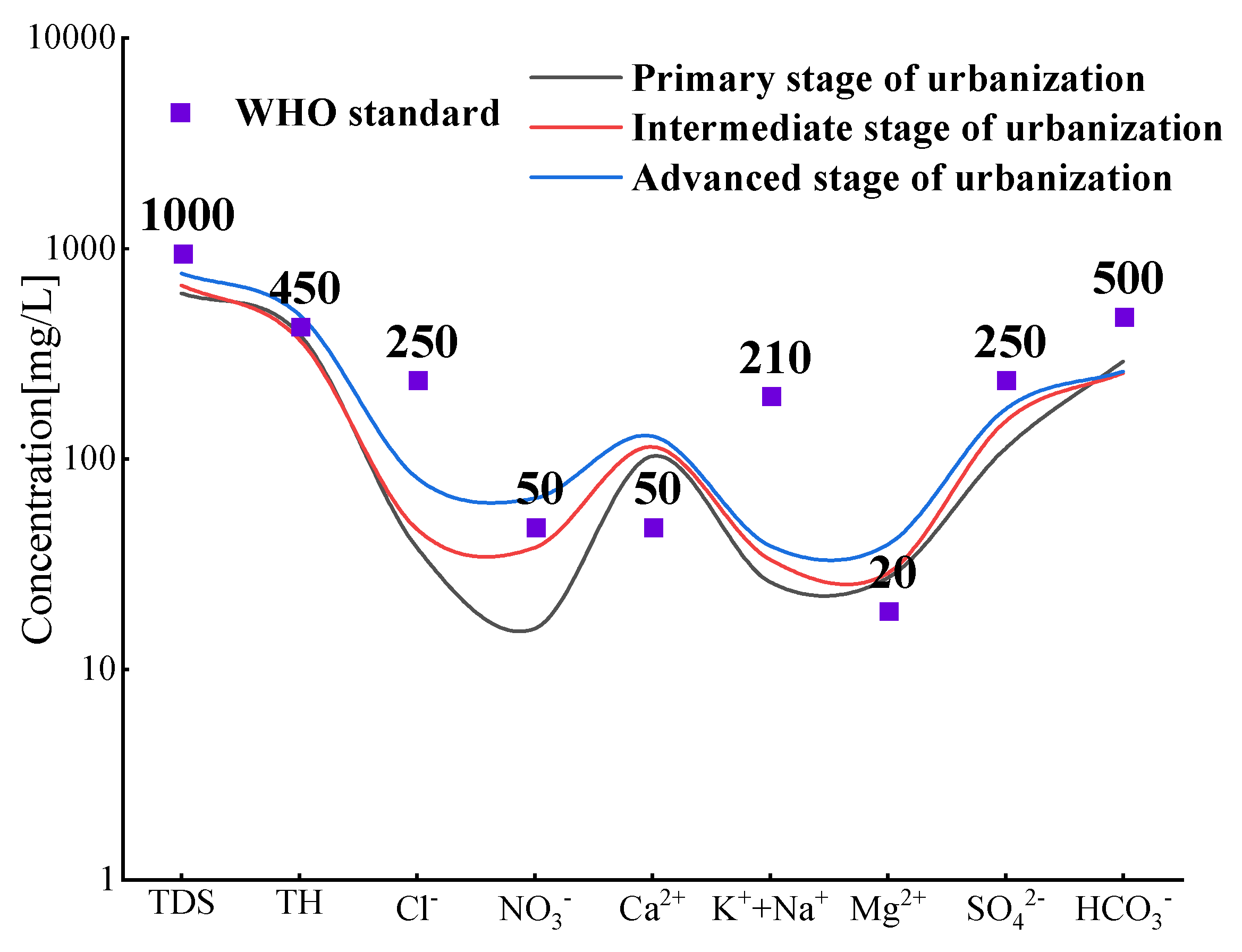
3.1.2. Ion characteristics analysis.
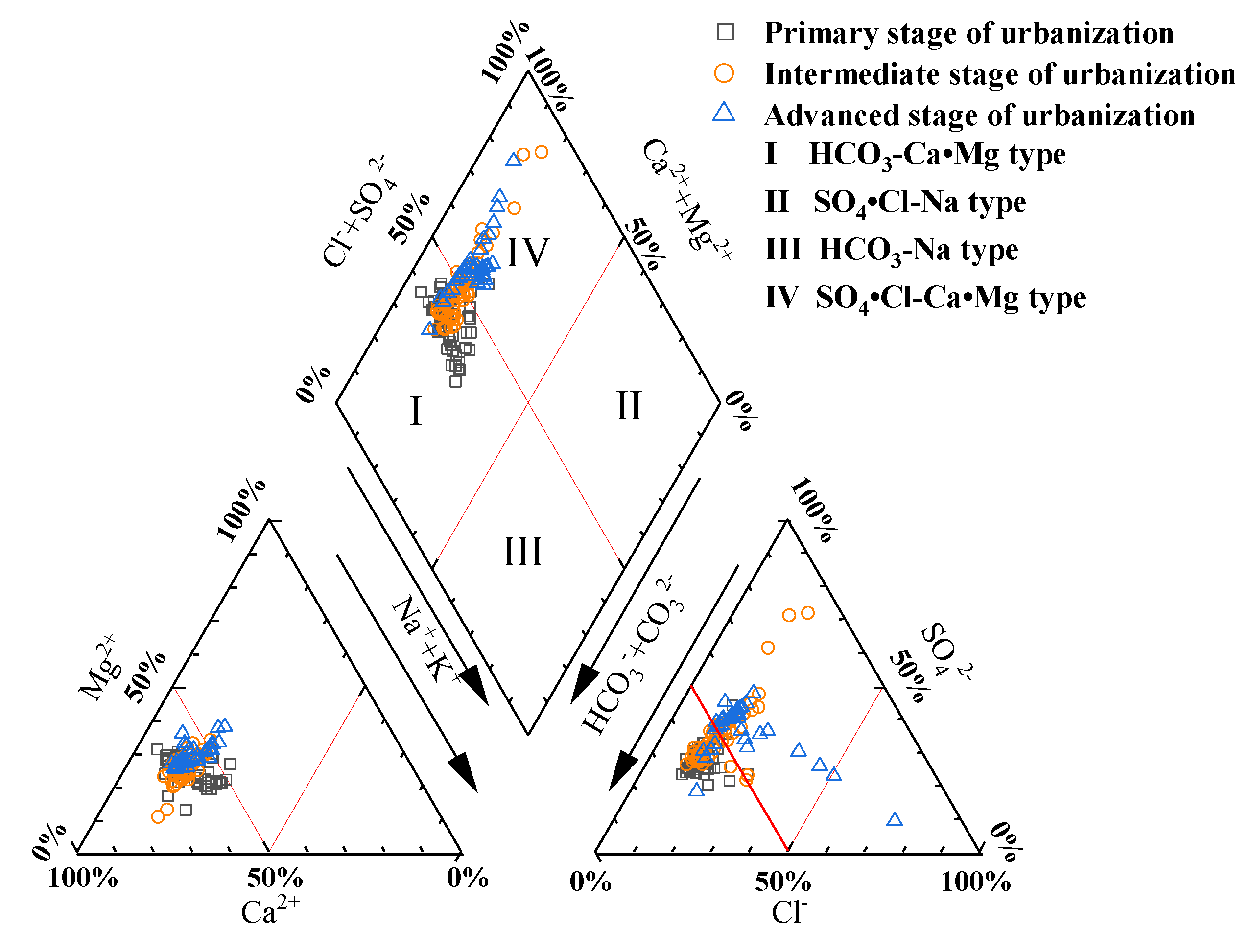
3.2. Hydrochemical type analysis
3.3. Factors driving hydrochemical evolution
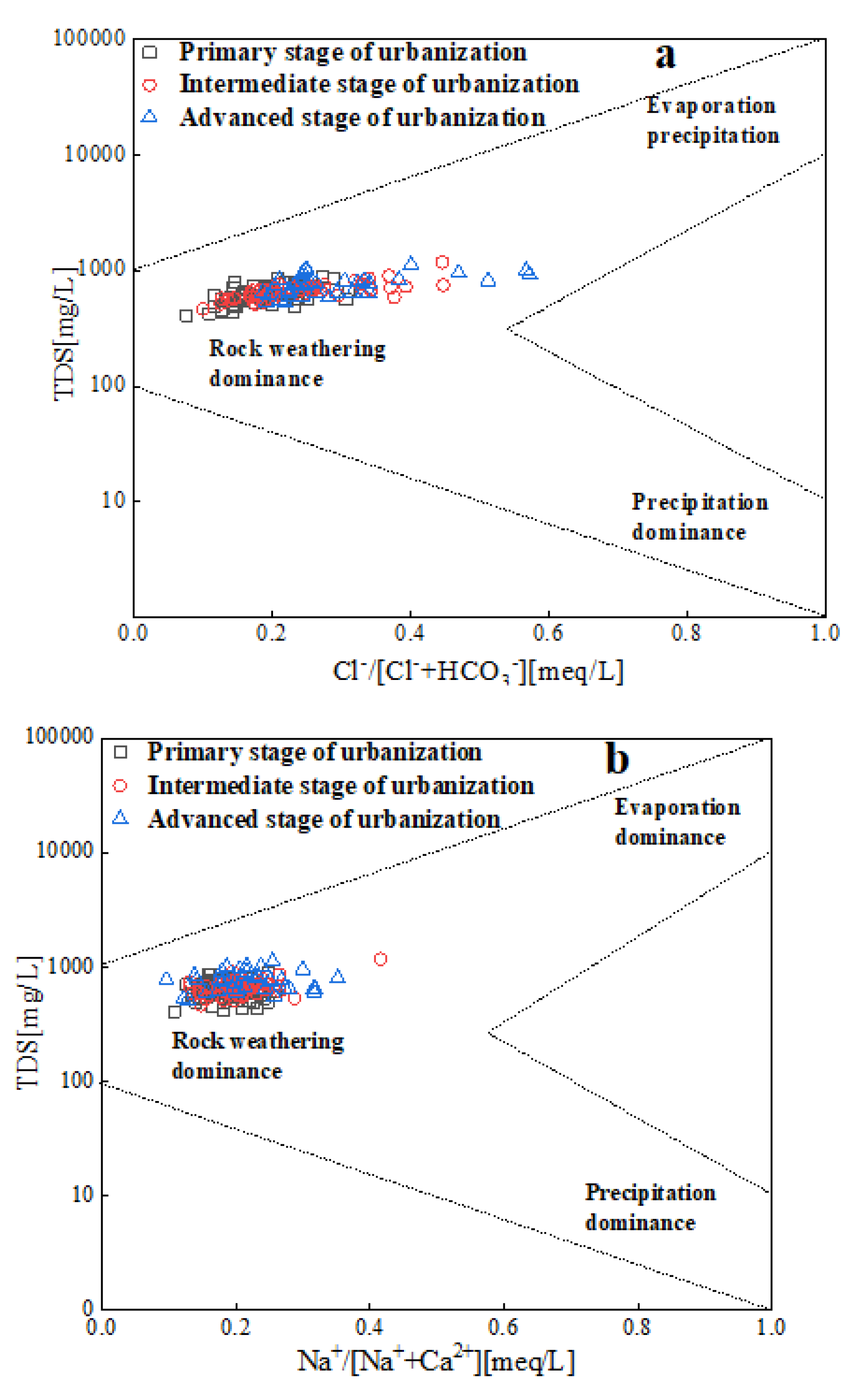
3.3.1. Analysis of controlling factors based on Gibbs diagram
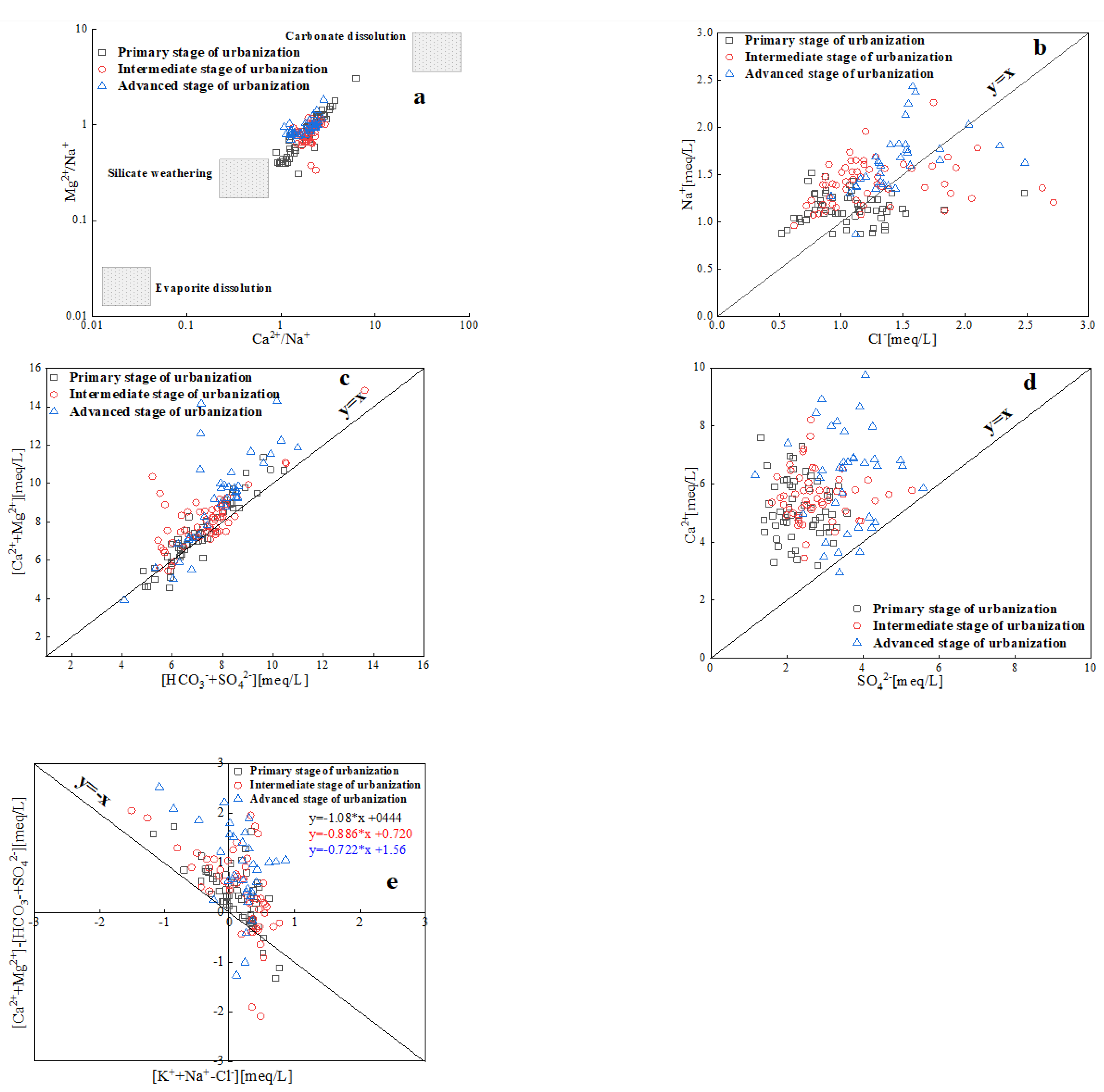
3.3.2. Water-rock interactions
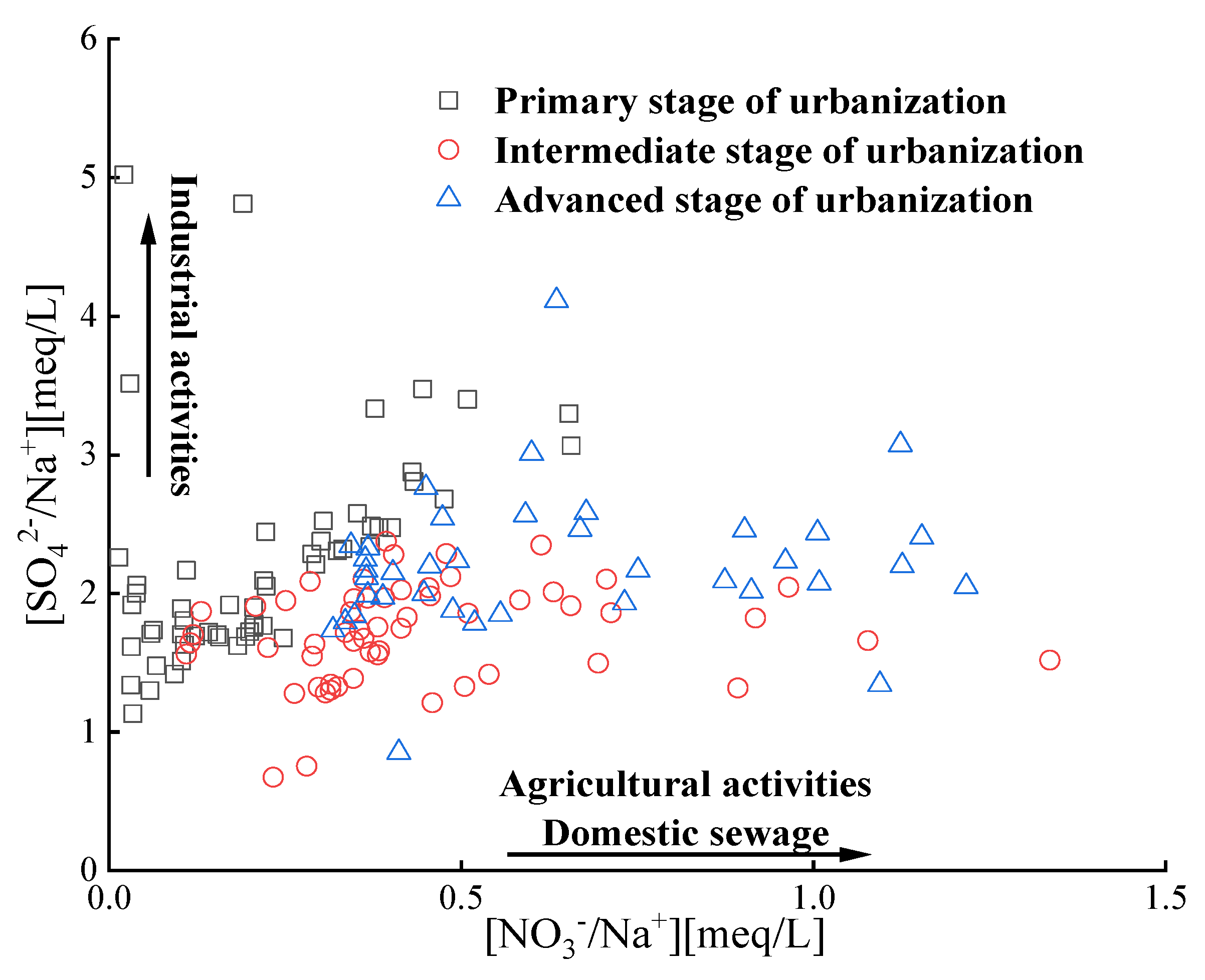
3.3.3. Impact of human activities on groundwater chemical composition
3.4. Driving mechanism of water chemistry temporal evolution
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Akakuru, O.; Eze, C.; Okeke, O. Hydrogeochemical evolution, water quality indices, irrigation suitability and pollution index of groundwater pig around eastern niger delta, nigeria. International Journal of Energy and Water Resources, 2022, 6, 389–411. [Google Scholar] [CrossRef]
- Barron, O. V.; Barr, A. D.; Donn, M. J. Evolution of nutrient export under urban development in areas affected by shallow watertable. Science of the Total Environment. 2013, 443, 491–504. [Google Scholar] [CrossRef]
- Cotruvo, Joseph, A. 2017 who guidelines for drinking water quality: first addendum to the fourth edition. Journal - American Water Works Association. 2017, 109, 44–51.
- Fei, L.; Jza, B.; Jza, B.; Pza, B. Factors controlling groundwater chemical evolution with the impact of reduced exploitation. Catena. 2022, 214, 106261. [Google Scholar]
- Huang, G.; Sun, J.; Zhang, Y.; Chen, Z.; Liu, F. Impact of anthropogenic and natural processes on the evolution of groundwater chemistry in a rapidly urbanized coastal area, south china. Science of the Total Environment. 2013, 463–464, 209-221.
- Huang, Z.; Pan, Y.; Gong, H.; Yeh, J.F.; Li, X.; Zhou, D.; et al. Subregional-scale groundwater depletion detected by grace for both shallow and deep aquifers in north china plain. Geophysical Research Letters 2015, 426, 1791–1799. [Google Scholar] [CrossRef]
- Israr, M.; Nazneen, S.; Raza, A.; Ali, N.; Khan, S.A.; Khan, H.; et al. Assessment of municipal solid waste landfilling practices on the groundwater quality and associated health risks: a case study of mardan-pakistan. Arabian Journal of Geosciences. 2022, 1517, 1–11. [Google Scholar] [CrossRef]
- Kai, L.; Zhong, P.; Raza, A. Impact of rapid urbanization on water quality index in groundwater fed Gomati River, Lucknow, India. Current Science, 2018, 3, 650–654. [Google Scholar]
- Li, P.; Karunanidhi, D.; Subramani, T.; Srinivasamoorthy, K. Sources and Consequences of Groundwater Contamination. Archives of environmental contamination and toxicology. 2021, 80, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, H.; Gosling, S.N.; Kummu, M.; Florke, M.; et al. Water scarcity assessments in the past, present, and future. Earth's Future. 2022, 5, 545–559. [Google Scholar] [CrossRef]
- Liu, J.; Yang, H.; Gosling, S.; Kummu, M.; Florke, M.; Pfister, S.; Hanasaki, N.; Wada, Y.; Zhang, X.; Zheng, C. Water scarcity assessments in the past, present, and future. Earth's Future, 2017, 5, 545–559. [Google Scholar] [CrossRef]
- Lü, X.; Liu, J.; Han, Z.; Zhu, L.; Chen, X. Effects of Yindaruqin irrigation project on groundwater chemical compositions in Qinwangchuan basin in Gansu Province Article. Transactions of the Chinese Society of Agricultural Engineering. 2020, 36, 166–174. [Google Scholar]
- Mukate, S.; Bhoominathan, S.; Solanky, V. Assessment of human health risk arising due to fluoride and nitrate in groundwater: a case study of bhokardan tehsil of maharashtra. Human and ecological risk assessment. 2022, 28, 594–620. [Google Scholar] [CrossRef]
- Pashaeifar, M.; Dehghanzadeh, R.; Ramazani, M.E.; Rafieyan, O.; Nejaei, A. Spatial and temporal assessment of groundwater quality and hydrogeochemical processes in urmia lake basin, iran. Water Supply, 2021, 21 8, 4328-4342.
- Peng, H.; Yang, W.; Ferrer, A.; Xiong, S.; Li, X.; Niu, G.; et al. Hydrochemical characteristics and health risk assessment of groundwater in karst areas of southwest china: a case study of bama, guangxi. Journal of Cleaner Production. 2022, 341, 130872. [Google Scholar] [CrossRef]
- Qin, R.; Song, Q.; Hao, Y.; Wu, G. Groundwater level declines in tianjin, north china: climatic variations and human activities. Environment, Development and Sustainability. 2023, 25, 1899–1913. [Google Scholar] [CrossRef]
- Quinn, N.; Oster, J.D. Innovations in sustainable groundwater and salinity management in california's san joaquin valley. Sustainability. 2021, 13, 6658. [Google Scholar] [CrossRef]
- Rai, S. C.; Saha, A. K. Impact of urban sprawl on groundwater quality: a case study of faridabad city, national capital region of delhi. Arabian Journal of Geoences. 2015, 810, 8039–8045. [Google Scholar] [CrossRef]
- Ren, C.; Zhang, Q.; Wang, H.; Wang, Y. Characteristics and source apportionment of polycyclic aromatic hydrocarbons of groundwater in hutuo river alluvial-pluvial fan, china, based on pmf model. Environmental science and pollution research. 2021, 288, 9647–9656. [Google Scholar] [CrossRef]
- Villegas, P.; Paredes, V.; Betancur, T.; Ribeiro, L. Assessing the hydrochemistry of the urabá aquifer, colombia by principal component analysis. Journal of Geochemical Exploration. 2013, 134, 120–129. [Google Scholar] [CrossRef]
- Saccò. ; Mattia.; Blyth, A.; Douglas, G.; et al. Stygofaunal diversity and ecological sustainability of coastal groundwater ecosystems in a changing climate: The Australian paradigm. Freshwater Biology. 2021, 6712, 2007–2023.
- Sinha, R.S.; Elango, L.J.W.g.C. Deterioration of groundwater quality: implications and management. Water Governance: Challenges and Prospects. 2019, 87-101.
- Stanley, Emily, H. ; Doyle, Martin. A geomorphic perspective on nutrient retention following dam removal. BioScience. 2002, 528, 693–693.
- Tiwari, A.K.; Singh, A.K.; Singh, A.K.; & Singh, M.P. ; & Singh, M. P. Hydrogeochemical analysis and evaluation of surface water quality of pratapgarh district, uttar pradesh, india. Applied Water Science. 2017, 74, 1–15. [Google Scholar]
- Wang, Q.; Dong, S.; Wang, H.; Yang, J.; Yu, B. Hydrogeochemical processes and groundwater quality assessment for different aquifers in the caojiatan coal mine of ordos basin, northwestern china. Environmental Earth Sciences. 2020, 79, 1–15. [Google Scholar] [CrossRef]
- Xiaohui, R.; Zhang, Z.; Yu, R.; Li, Y.; Li, Y.; et al. Hydrochemical variations and driving mechanisms in a large linked river-irrigation-lake system. Environmental Research, 2023, 225.
- Zhang, Q.; Wang, H. Assessment of sources and transformation of nitrate in the alluvial-pluvial fan region of north china using a multi-isotope approach. Journal of Environmental Sciences. 2020, 893, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, W.; Jin, X. Interaction analysis of magnaporthe grisea and rice varieties in heilongjiang province. Journal of Northeast Agricultural University. 2011, 421, 28–33(In Chinese). (In Chinese) [Google Scholar]
- Zheng, C.; Liu, J.; Cao, G.; Kendy, E.; Wang, H.; Jia, Y. Can China Cope with Its Water Crisis? —Perspectives from the North China Plain. Ground Water. 2010, 483, 350–354. [Google Scholar]
| Parameters | Primary stage of urbanization | Intermediate stage of urbanization | Advanced stage of urbanization | ||||
| PC1 | PC2 | PC1 | PC2 | PC1 | PC2 | PC3 | |
| TDS | 0.985 | -0.089 | 0.957 | 0.106 | 0.272 | 0.708 | 0.511 |
| Ca2+ | 0.952 | -0.205 | 0.925 | -0.113 | 0.796 | 0.523 | 0.183 |
| Mg2+ | 0.922 | -0.102 | 0.062 | 0.806 | 0.980 | 0.032 | 0.086 |
| NO3- | 0.882 | -0.167 | 0.016 | 0.599 | 0.139 | 0.925 | 0.185 |
| TH | 0.868 | 0.011 | 0.870 | 0.013 | 0.834 | 0.331 | 0.227 |
| SO42- | 0.868 | 0.061 | 0.934 | -0.188 | -0.074 | 0.243 | 0.916 |
| HCO3- | 0.859 | -0.183 | -0.401 | 0.646 | 0.036 | 0.851 | 0.239 |
| Cl- | 0.612 | -0.003 | 0.801 | 0.179 | 0.963 | -0.052 | -0.196 |
| K++Na+ | -0.039 | 0.827 | 0.765 | 0.014 | 0.744 | -0.077 | 0.583 |
| pH | -0.082 | 0.704 | 0.209 | 0.793 | -0.045 | -0.787 | 0.138 |
| Eigenvalue | 6.23 | 1.20 | 4.84 | 2.15 | 4.95 | 2.48 | 1.27 |
| Contribution rate% | 62.3 | 12.0 | 48.4 | 21.5 | 49.5 | 24.8 | 12.6 |
| Cumulative contribution rate% | 62.3 | 74.4 | 48.4 | 69.9 | 49.5 | 74.3 | 86.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




