Submitted:
20 July 2023
Posted:
20 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Focused Question
2.2. Search Strategies
2.3. Eligibility Criteria
2.4. Study Selection and Data Extraction
2.6. Quality Assessment of Studies
2.7. Risk-of-Bias Assessment
2.8. Statistical Methods
3. Results
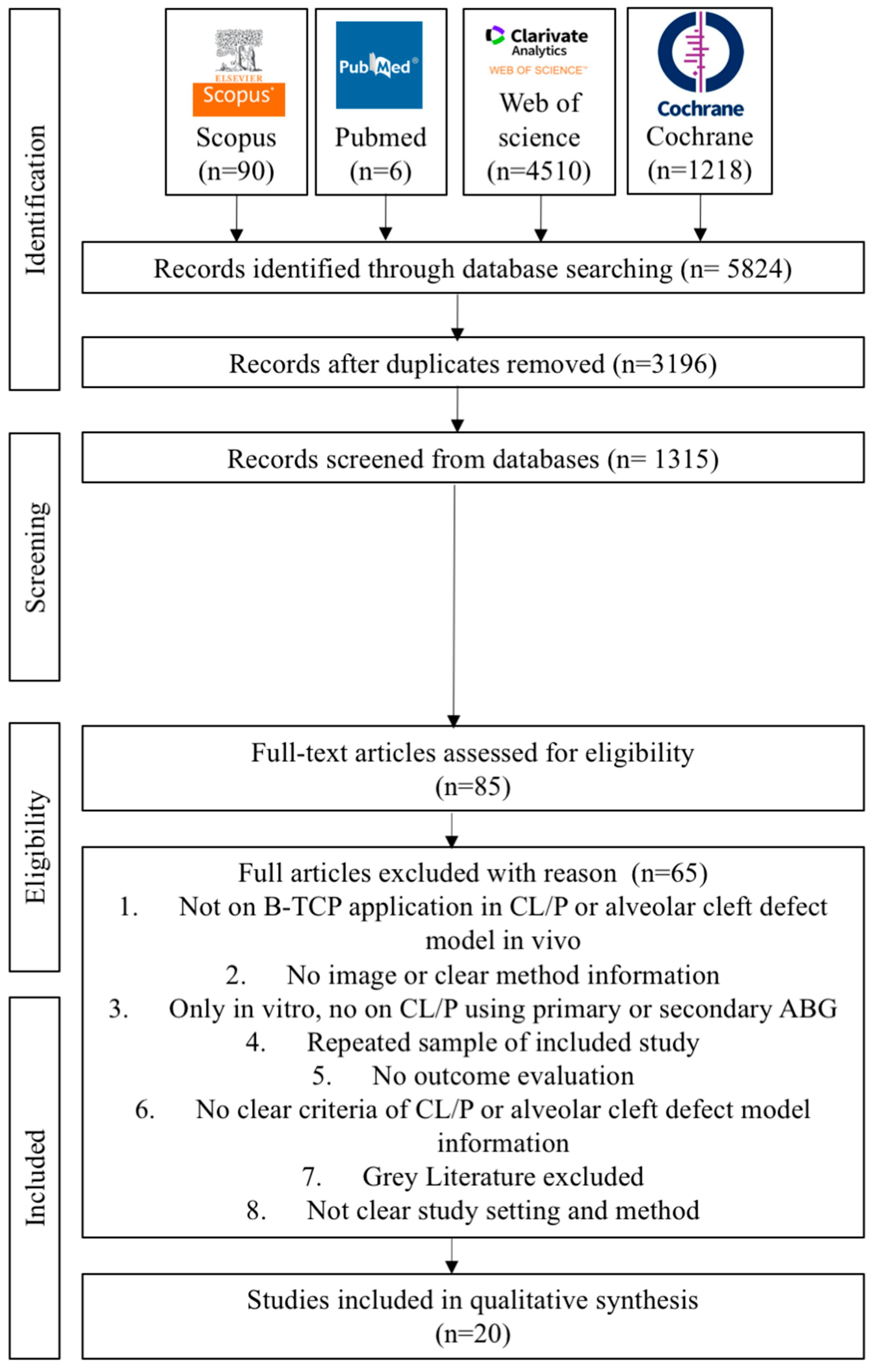
3.1. Study Selection, Data Extraction, and Quality Assessment
3.2. Assessment of the Risk of Bias and Quality
3.3. Qualitative Analysis
4. Discussion
5. Conclusions
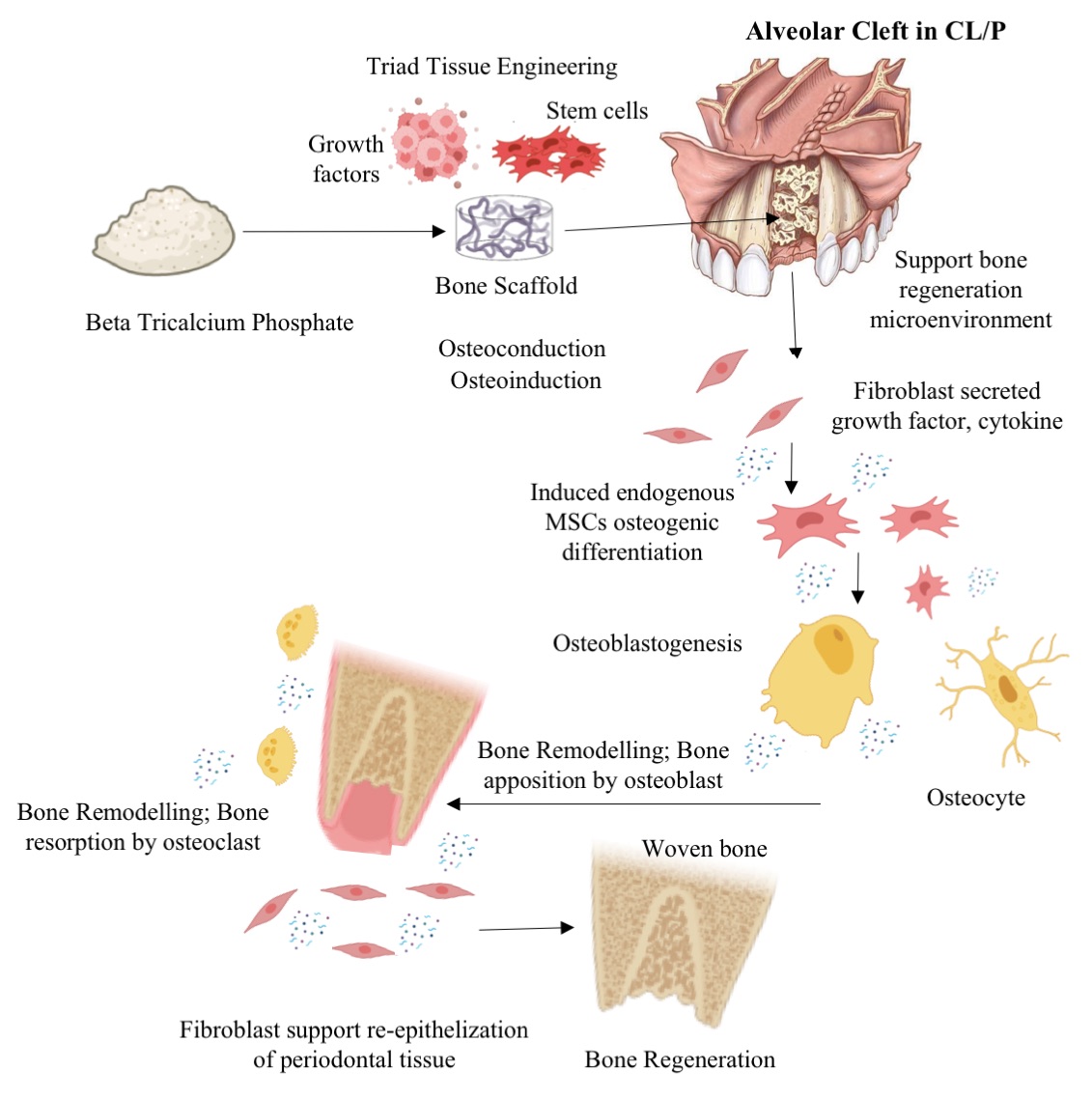
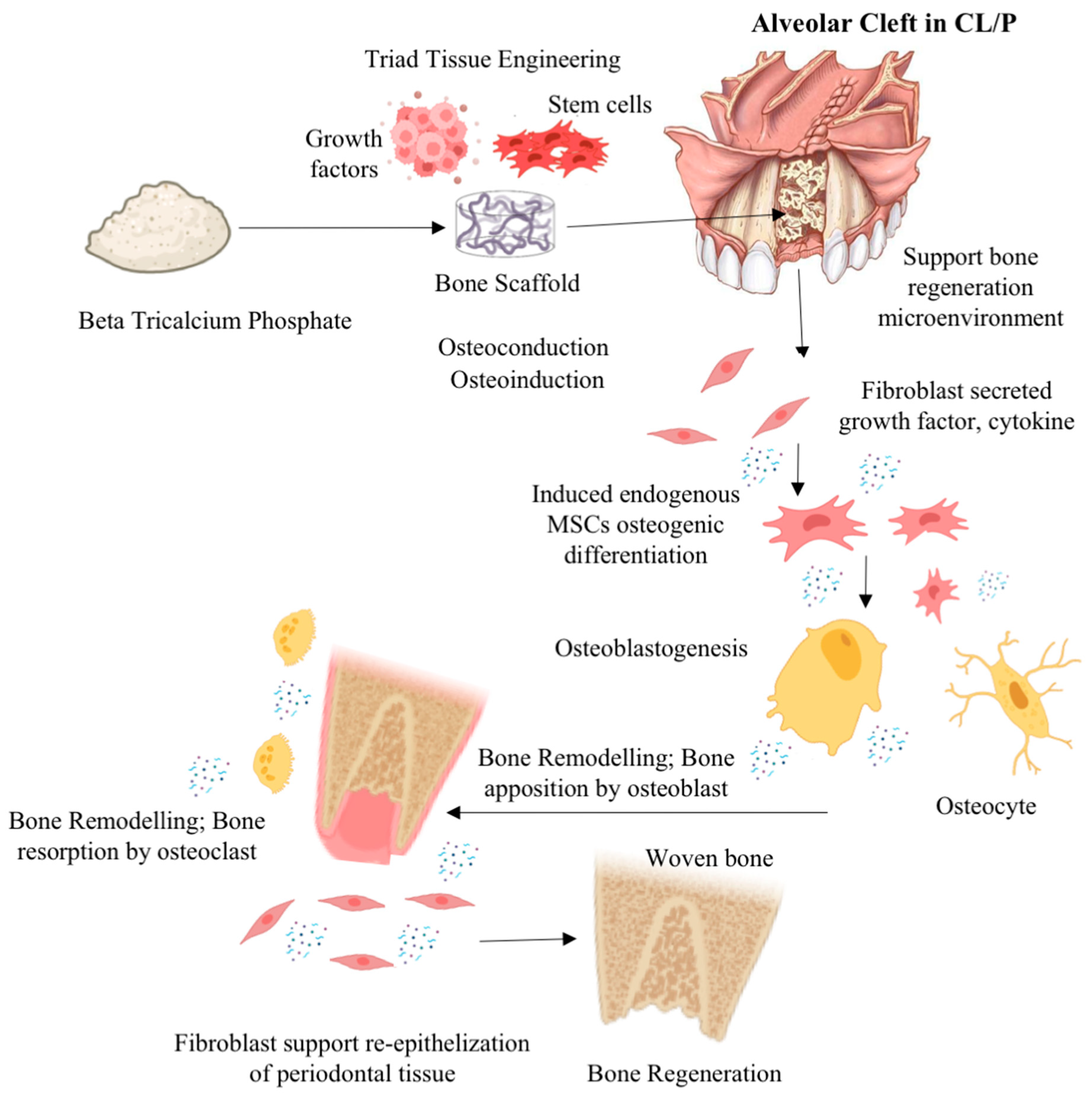
- β-TCP as ABG is biocompatible, more visible and practical, offers a less invasive procedure, and does not interfere with orthodontic treatment.
- β-TCP as synthetic ABG can be the alternative to autologous bone grafts with several terms and conditions, such as if autografts are hard to come by or there is donor site morbidity, and the size of the defect restricts the size of the autograft.
- Enhancement of osteoinductive and osteoconductive abilities for improvement of β-TCP efficacy for ABG in CL/P or alveolar bone cleft defects can be done by a tissue engineering approach combining β-TCP with growth factor, mesenchymal stem cells, or other graft materials and modification of β-TCP physical properties.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Competing Interests
References
- Shkoukani MA, Lawrence LA, Liebertz DJ, Svider PF. Cleft palate: a clinical review. Birth Defects Res C Embryo Today. 2014;102(4):333-42. [CrossRef]
- Mossey PA, Little J, Munger RG, Dixon MJ, Shaw WC. Cleft lip and palate. Lancet. 2009;374(9703):1773-85. [CrossRef]
- Dixon MJ, Marazita ML, Beaty TH, Murray JC. Cleft lip and palate: understanding genetic and environmental influences. Nat Rev Genet. 2011;12(3):167-78. [CrossRef]
- Massenburg BB, Hopper RA, Crowe CS, Morrison SD, Alonso N, Calis M, Donkor P, Kreshanti P, Yuan J; Global Burden of Disease 2017 Orofacial Clefting Collaborators. Global Burden of Orofacial Clefts and the World Surgical Workforce. Plast Reconstr Surg. 2021;148(4):568e-580e. [CrossRef]
- Tanaka SA, Mahabir RC, Jupiter DC, Menezes JM. Updating the epidemiology of cleft lip with or without cleft palate. Plast Reconstr Surg. 2012;129(3):511e-518e. [CrossRef]
- Panamonta V, Pradubwong S, Panamonta M, Chowchuen B. Global Birth Prevalence of Orofacial Clefts: A Systematic Review. J Med Assoc Thai. 2015;98 Suppl 7:S11-21.
- Ariawan D, Vitria EE, Sulistyani LD, Anindya CS, Adrin NSR, Aini N, Hak MS. Prevalence of Simonart's band in cleft children at a cleft center in Indonesia: A nine-year retrospective study. Dent Med Probl. 2022 ;59(4):509-515. [CrossRef]
- Iskandar RPD, Proboningrat A, Fadholly A, Narmada IB, Nidom CA, Sudjarwo SA. The Densitometric Analysis of Protein Pattern in Cleft Lip and Palate Patients. J Int Soc Prev Community Dent. 2019 May-Jun;9(3):240-244. [CrossRef]
- Wehby GL, Cassell CH. The impact of orofacial clefts on quality of life and healthcare use and costs. Oral Dis. 2010;16(1):3-10. [CrossRef]
- Menezes C, de Arruda JA, Silva LV, Monteiro JL, Caribé P, Álvares P, Almeida MC, Coelli JC, Goldemberg F, Silveira M, Sobral AP, Bueno DF. Nonsyndromic cleft lip and/or palate: A multicenter study of the dental anomalies involved. J Clin Exp Dent. 2018 Aug 1;10(8):e746-e750. PMID: 30305871; PMCID: PMC6174020. [CrossRef]
- Wei S, Wang Y, Ma X, Pan Y, Zhang P, Jiang H, Du Y, Wan L. Comparison of Anatomical Features of Alveolar Cleft in Unilateral Cleft Lip and Palate Patients of Different Ages. J Craniofac Surg. 2020 Sep;31(6):1629-1632. [CrossRef] [PubMed]
- Semb, G. Alveolar bone grafting. Front Oral Biol. 2012;16:124-36. Epub 2012 Jun 25. [CrossRef] [PubMed]
- Ebraheim NA, Elgafy H, Xu R. Bone-graft harvesting from iliac and fibular donor sites: techniques and complications. J Am Acad Orthop Surg. 2001 May-Jun;9(3):210-8. [CrossRef] [PubMed]
- Shibuya N, Jupiter DC. Bone graft substitute: allograft and xenograft. Clin Podiatr Med Surg. 2015 Jan;32(1):21-34. PMID: 25440415. [CrossRef]
- Haugen HJ, Lyngstadaas SP, Rossi F, Perale G. Bone grafts: which is the ideal biomaterial?. J Clin Periodontol. 2019;46 Suppl 21:92-102. [CrossRef]
- Yu H, Özcan M, Yoshida K, Cheng H, Sawase T. Bonding to industrial indirect composite blocks: A systematic review and meta-analysis. Dent Mater. 2020 Jan;36(1):119-134. Epub 2019 Nov 27. [CrossRef] [PubMed]
- Yang H, Xu YL, Hong G, Yu H. Effects of low-temperature degradation on the surface roughness of yttria-stabilized tetragonal zirconia polycrystal ceramics: A systematic review and meta-analysis. J Prosthet Dent. 2021 Feb;125(2):222-230. Epub 2020 Feb 28. [CrossRef] [PubMed]
- Yang H, Yang S, Attin T, Yu H. Effect of Acidic Solutions on the Surface Roughness and Microhardness of Indirect Restorative Materials: A Systematic Review and Meta-analysis. Int J Prosthodont. 2023 Jan-Feb;36(1):81–90. [CrossRef] [PubMed]
- Yang H, Yang S, Attin T, Yu H. Effect of Acidic Solutions on the Surface Roughness and Microhardness of Indirect Restorative Materials: A Systematic Review and Meta-analysis. Int J Prosthodont. 2023 Jan-Feb;36(1):81–90. [CrossRef] [PubMed]
- Zhang L-x, Hong D-w, Zheng M, Yu H. Is the bond strength of zirconia-reinforced lithium silicate lower than that of lithium disilicate? - A systematic review and meta-analysis. Journal of Prosthodontic Research 2022;66(4):530-7.
- Möhlhenrich, S.C. , Kniha, K., Magnuska, Z. et al. Evaluation of different grafting materials for alveolar cleft repair in the context of orthodontic tooth movement in rats. Sci Rep 11, 13586 (2021). [CrossRef]
- Trujillo RL, Kadioglu O, Currier GF, Smith KS, Yetkiner E. Volumetric Cleft Changes in Treatment with Bone Morphogenic Protein/β-Tricalcium Phosphate Versus Grafts From the Iliac Crest or Symphysis. J Oral Maxillofac Surg. 2018;76(9):1991-1997. [CrossRef]
- Putri IL, Fatchiyah, Pramono C, Bachtiar I, Latief FDE, Utomo B, Rachman A, Soesilawati P, Hakim L, Rantam FA, Perdanakusuma DS. Alveolar Repair Using Cancellous Bone and Beta Tricalcium Phosphate Seeded With Adipose-Derived Stem Cell. Cleft Palate Craniofac J. 2022 Oct 13:10556656221132372. Epub ahead of print. [CrossRef] [PubMed]
- Janssen NG, Schreurs R, de Ruiter AP, Sylvester-Jensen HC, Blindheim G, Meijer GJ, Koole R, Vindenes H. Microstructured beta-tricalcium phosphate for alveolar cleft repair: a two-centre study. Int J Oral Maxillofac Surg. 2019 Jun;48(6):708-711. Epub 2018 Dec 26. [CrossRef] [PubMed]
- Kamal, M. , Andersson, L., Tolba, R. et al. Bone regeneration using composite non-demineralized xenogenic dentin with beta-tricalcium phosphate in experimental alveolar cleft repair in a rabbit model. J Transl Med 15, 263 (2017). [CrossRef]
- Du, F. , Wu, H., Li, H. et al. Bone Marrow Mononuclear Cells Combined with Beta-Tricalcium Phosphate Granules for Alveolar Cleft Repair: A 12-Month Clinical Study. Sci Rep 7, 13773 (2017). [CrossRef]
- Miyagawa, K. , Tanaka, S., Hiroishi, S. et al. Comparative evaluation of bone microstructure in alveolar cleft repair by cone beam CT: influence of different autologous donor sites and additional application of β-tricalcium phosphate. Clin Oral Invest 24, 2789–2797 (2020). [CrossRef]
- Shahnaseri S, Sheikhi M, Hashemibeni B, Mousavi SA, Soltani P. Comparison of autogenous bone graft and tissue-engineered bone graft in alveolar cleft defects in canine animal models using digital radiography. Indian J Dent Res. 2020 Jan-Feb;31(1):118-123. [CrossRef]
- Pourebrahim N, Hashemibeni B, Shahnaseri S, Torabinia N, Mousavi B, Adibi S, Heidari F, Alavi MJ. A comparison of tissue-engineered bone from adipose-derived stem cell with autogenous bone repair in maxillary alveolar cleft model in dogs. Int J Oral Maxillofac Surg. 2013 May;42(5):562-8. [CrossRef]
- de Ruiter A, Janssen N, van Es R, Frank M, Meijer G, Koole R, Rosenberg T. Micro-structured Beta-Tricalcium Phosphate for Repair of the Alveolar Cleft in Cleft Lip and Palate Patients: A Pilot Study. Cleft Palate Craniofac J. 2015 May;52(3):336-40. [CrossRef]
- Huang J, Tian B, Chu F, Yang C, Zhao J, Jiang X, Qian Y. Rapid maxillary expansion in alveolar cleft repaired with a tissue-engineered bone in a canine model. J Mech Behav Biomed Mater. 2015 Aug;48:86-99. Epub 2015 Apr 13. [CrossRef] [PubMed]
- Weijs WL, Siebers TJ, Kuijpers-Jagtman AM, Bergé SJ, Meijer GJ, Borstlap WA. Early secondary closure of alveolar clefts with mandibular symphyseal bone grafts and beta-tri calcium phosphate (beta-TCP). Int J Oral Maxillofac Surg. 2010 May;39(5):424-9. Epub 2010 Mar 29. [CrossRef] [PubMed]
- Ito M, Toriumi T, Imura H, Akiyama Y, Arai Y, Natsume N, Honda M. Rat Palatine Fissure: A Suitable Experimental Model for Evaluating Bone Regeneration. Tissue Eng Part C Methods. 2019 Sep;25(9):513-522. Epub 2019 Sep 11. [CrossRef] [PubMed]
- Möhlhenrich SC, Kniha K, Magnuska Z, Chhatwani S, Hermanns-Sachweh B, Gremse F, Hölzle F, Danesh G, Modabber A. Development of root resorption during orthodontic tooth movement after cleft repair using different grafting materials in rats. Clin Oral Investig. 2022 Sep;26(9):5809-5821. Epub 2022 May 14. PMID: 35567639; PMCID: PMC9474460. [CrossRef]
- Hossain MZ, Kyomen S, Tanne K. Biologic responses of autogenous bone and beta-tricalcium phosphate ceramics transplanted into bone defects to orthodontic forces. Cleft Palate Craniofac J. 1996 Jul;33(4):277-83. [CrossRef] [PubMed]
- de Ruiter A, Meijer G, Dormaar T, Janssen N, van der Bilt A, Slootweg P, de Bruijn J, van Rijn L, Koole R. β-TCP versus autologous bone for repair of alveolar clefts in a goat model. Cleft Palate Craniofac J. 2011 Nov;48(6):654-62. Epub 2010 Aug 11. [CrossRef] [PubMed]
- Zhang D, Chu F, Yang Y, Xia L, Zeng D, Uludağ H, Zhang X, Qian Y, Jiang X. Orthodontic tooth movement in alveolar cleft repaired with a tissue engineering bone: an experimental study in dogs. Tissue Eng Part A. 2011 May;17(9-10):1313-25. Epub 2011 Mar 17. [CrossRef] [PubMed]
- Janssen NG, de Ruiter AP, van Hout WMMT, van Miegem V, Gawlitta D, Groot FB, Meijer GJ, Rosenberg AJWP, Koole R. Microstructured β-Tricalcium Phosphate Putty Versus Autologous Bone for Repair of Alveolar Clefts in a Goat Model. Cleft Palate Craniofac J. 2017 Nov;54(6):699-706. Epub 2016 Oct 10. [CrossRef] [PubMed]
- Ekin O, Calis M, Aliyev A, Yar AS, Korkusuz P, Bilgic E, Aydin HM, Celik HH, Ozgur F, Vargel I. Poly(L-Lactide)/Poly(ε-Caprolactone) and Collagen/β-Tricalcium Phosphate Scaffolds for the Treatment of Critical-Sized Rat Alveolar Defects: A Microtomographic, Molecular-Biological, and Histological Study. Cleft Palate Craniofac J. 2016 Jul;53(4):453-63. Epub 2015 Oct 27. [CrossRef] [PubMed]
- Tokugawa Y, Kubota M, Nishimura M, Haruyama N, Igarashi K. Bone regeneration of canine artificial alveolar clefts using bone-marrow-derived mesenchymal stromal cells and β-tricalcium phosphate: A preliminary study. 5: Orthodontic Wave 2012;71, 2012. [CrossRef]
- Ducheyne P, Radin S, King L. The effect of calcium phosphate ceramic composition and structure on in vitro behavior. I. Dissolution. J Biomed Mater Res. 1993 Jan;27(1):25-34. [CrossRef] [PubMed]
- Schwartz C, Liss P, Jacquemaire B, Lecestre P, Frayssinet P. Biphasic synthetic bone substitute use in orthopaedic and trauma surgery: clinical, radiological and histological results. J Mater Sci Mater Med. 1999 Dec;10(12):821-5. [CrossRef] [PubMed]
- Oberoi S, Chigurupati R, Gill P, Hoffman WY, Vargervik K. Volumetric assessment of secondary alveolar bone grafting using cone beam computed tomography. Cleft Palate Craniofac J. 2009 Sep;46(5):503-11. Epub 2009 Feb 27. [CrossRef] [PubMed]
- Stasiak M, Wojtaszek-Słomińska A, Racka-Pilszak B. A novel method for alveolar bone grafting assessment in cleft lip and palate patients: cone-beam computed tomography evaluation. Clin Oral Investig. 2021 Apr;25(4):1967-1975. Epub 2020 Aug 15. Erratum in: Clin Oral Investig. 2020 Sep 15;: PMID: 32803441; PMCID: PMC7966225. [CrossRef]
- Kamperos G, Theologie-Lygidakis N, Tsiklakis K, Iatrou I. A novel success scale for evaluating alveolar cleft repair using cone-beam computed tomography. J Craniomaxillofac Surg. 2020 Apr;48(4):391-398. Epub 2020 Feb 17. [CrossRef] [PubMed]
- European Commission. Radiation protection No 172. Cone Beam CT for Dental and Maxillofacial Radiology. Evidence Based Guidelines; European Commission: Luxembourg, 2012. European Commission: Luxembourg.
- Francisco I, Paula AB, Oliveiros B, Fernandes MH, Carrilho E, Marto CM, Vale F. Regenerative Strategies in Cleft Palate: An Umbrella Review. Bioengineering (Basel). 2021 Jun 3;8(6):76. PMID: 34205126; PMCID: PMC8230219. [CrossRef]
| Database | Search Strategy |
|---|---|
| Scopus | β-tricalcium AND phosphate OR β-TCP or bone graft OR bone grafting OR alveolar AND bone AND graft AND alveolar AND bone AND cleft OR cleft AND palate AND (LIMIT-TO (PUBSTAGE, “final”)) AND (LIMIT-TO (OA, “all”)) AND ( LIMIT-TO ( DOCTYPE , "ar" ) ) AND ( LIMIT-TO ( LANGUAGE , "English" ) ) AND ( LIMIT-TO ( SRCTYPE , "j" ) ) |
| PubMed | β-tricalcium phosphate OR β-TCP OR bone graft OR bone grafting OR biomaterial OR alveolar bone graft AND alveolar bone cleft AND cleft palate Filters applied: Free full text, Clinical Trial, Randomized Controlled Trial, English, Exclude preprints, MEDLINE. |
| Web of Science | ((((((ALL= (β-tricalcium phosphate)) OR TS= (β-TCP)) OR TS= (bone graft)) OR TS= (bone grafting)) OR TS= (alveolar bone graft)) AND TS=(alveolar bone cleft )) OR TS=(cleft palate) Refined By: Open Access. Click to remove this refine from your search. Document Types: Article. Click to remove this refine from your search. Open Access: All Open Access. Click to remove this refine from your search. Languages: English. |
| Cochrane Library | β-tricalcium phosphate OR β-TCP OR bone graft OR bone grafting OR alveolar bone graft AND alveolar bone cleft OR cleft palate in Title Abstract Keyword - in Trials, Clinical Answers (Word variations have been searched) |
| Authors, Year, Country | Study Design | Sample / Subject criteria (n) |
Type of Cleft | Type of Tissue Engineering | Examinations and Variables | Outcome | Conclusion |
|---|---|---|---|---|---|---|---|
| Möhlhenrich et al., 2021, German [21] | True experimental, post-test only control group design | Twenty-one; 8-week-old male Wistar-HAN rats; average weight of 465 ± 34 g. |
1.7 mm alveolar clefts at the left side of the upper jaws with 0.14 N continuous orthodontic tooth movement application |
Autologous rat hip, xenograft human bone substitute material and Synthetic graft β-TCP and HA. | µCT: BMD, BV/TV. Histomorphometric analysis: lamellar bone and woven bone |
Autologous bone-based grafting materials more strongly promoted the formation of new bone compared to the synthetic bone substitute, which similarly looked to have less resorbed and integrated into the cleft defect. . |
The gold standard for cleft repair has been proven to be autologous bone grafts. Autografts and xenografts seemed to be interchangeable terms. After more tissue engineering adjustments, β-TCP and HA might be used as an alternative. |
| Trujillo et al., 2018, USA [22] | Retrospective cohort study | Twenty-five patient CL/P: 15 and 10 females |
Unilateral or bilateral clefts. | Iliac crest bone autograft Mandibular symphyseal bone graft, rhBMP-2/ β-TCP bone substitute. |
CBCT: Preoperative defect volume(cm3), Postoperative residual defect volume (cm3), Bone formation (%) |
The newest bone development was shown in the iliac crest group, which was followed by the rhBMP-2/ACS/ β-TCP group and the mandibular symphysis group. However, the differences were not statistically significant. |
In alveolar cleft patients, rhBMP2 administered in a β-TCP scaffold may be an effective substitute for autogenous iliac crest and symphysis grafts, preventing donor site morbidity. |
| Putri et al., 2022, Indonesia [23] | True experimental, post-test only control group design | Thirty-six male Wistar rats (Rattus novergicus) | 5 × 5mm alveolar defect of the upper jaws | Autologous rat alveolar bone graft (ABG), human cancellous freezedried graft (HCG)–human adipose stem cell (hADSC), β-TCP –hADSC |
Immunohistochemical analysis: runt-related transcription factor 2 (RUNX2), alkaline phosphatase (ALP), osterix (OSX), and bone morphogenetic protein 2 (BMP2) µCT: BV/TV (mm3), BF (%), and trabecular thickness of bone (TT, mm). |
When it came to the expression of RUNX2, OSX, ALP, and BMP2, HCG-hADSC was superior to the comparison, followed by β-TCP-hADSC and ABG. In terms of promoting bone growth, β-TCP-hADSC outperformed HCG-hADSC and ABG, which both performed about as well. | Exogenous hADSC improved the ability of HCG and β-TCP to enhance osteogenesis, osteoconduction, and osteoinduction. The latter proved successful at promoting bone formation after HCG and ABG. . |
| Janssen et al., 2019, Netherland [24] | Case control randomized clinical study, | Total 20 CL/P patients: 14 Males, 6 Female |
unilateral CL/P | microporous β-TCP |
CBCT: Residual calcified tissue, Spontaneous eruption of canine/lateral incisor, Continuous alveolar process, Residual oronasal fistula |
No significant granule loss, surgical infection, or wound dehiscence occurred. The operating patients, who had an average residual calcified tissue volume of 65% one year after surgery, had no orinasal fistulas left. All patients displayed a continuous alveolar process on the affected side. The canine close to the alveolar cleft failed to erupt on its own in two patients. Three months after the procedure, fixed appliance of orthodontic treatment was begun, and it progressed well. The grafted area allowed easy passage for teeth along the cleft. |
In the clinical setting, secondary alveolar grafting using microporous β-TCP is safe to utilize. At the recipient site, the rates of complications, canine eruption, and residual calcified tissue are comparable to those for autologous grafts. |
| Kamal et al., 2017 German [25] | True experimental, post-test only control group design | sixteen male New Zealand rabbits . |
Unilateral alveolar cleft defects | β-TCP, composite xenogenic dentin with β-TCP. |
µCT: Defect size (mm3) Hounsfield unit (HU) % BV/TV, BMD. histomorphometric analysis: % bone formation % residual graft. |
The difference between the Hounsfield unit (HU), BV/TV, and BMD between clefts filled with dentin and β-TCP is considerable. Dentin/β-TCPgroup showed significantly larger bone volume fraction (%) and residual graft (%) compared to β-TCP group. | When assessed by µCT and histomorphometry, alveolar cleft defects repaired with dentin/β-TCP resulted in larger graft residual volume and bone volume fraction. |
| Du et al., 2017; China [26] | Clinical study | Ten CL/P patients (5 males and 5 females) |
Unilateral alveolar cleft defects | autologous iliac crest bone graft (ICBG), bone marrow mononuclear cells (BMMNCs) combined with β-TCP granules |
CBCT and computer aided engineering technology: Bone Volume (mm3), Bone Formation Ratio (%), Bone Volume (mm3) Bone Formation Ratio (%). Bone Union; Chelsea Scale; Duration of Hospital Stay (days). |
Average defect volume, bone formation ratio (%), bone volume (mm3), and bone formation ratio (%) were not significantly different between the BMMNCs/ β-TCP group and the ICBG group, respectively. In both the BMMNCs/-TCP and ICBG groups, the majority of the patients experienced alveolar bone union. Between the BMMNCs/ β-TCP and ICBG groups, there was no discernible difference. When compared to the BMMNCs/ β-TCP group, the length of hospital stays in the ICBG group was much longer. |
Alveolar cleft repair with autologous BMMNCs and β-TCP granules was radiographically similar to using ICBG. Significant reductions were made in both surgical pain and hospital stay days. Similar to ICBG, significant resorption for BMMNCs/ β-TCP grafting was observed up to 6 months following surgery. Alveolar bone regeneration using BMMNCs/β-TCP grafting was both secure and efficient. |
| Miyagawa et al., 2020; Japan [27 | Clinical study | Thirty-one CL/P patients | Non-syndromic unilateral cleft lip and alveolus (UCLA) and cleft lip palate (UCLP) | iliac crest bone, Mandibular symphysis, Mandibular symphysis combined with β-TCP granules. |
CBCT: BV/TV, trabecular thickness (Tb.Th), trabecular number (Tb.N), trabecular separation (Tb.Sp), trabecular bone pattern factor (TBPf), structure model index (SMI), and fractal dimension (FD). |
TBPs revealed variations between the IC and MS groups, leading to higher bone volume density values and a lower TBPf value in the IC group when compared to the MS group. When compared to the group without β-TCP, using MS+ β-TCP or administering β-TCP granules into the donor site considerably enhanced the reconstructed or regenerated BV/TV and Tb.Th. | In comparison to secondary alveolar bone grafting (SABG), the use of β-TCP granules as a partial substitute with autologous bone from the mandibular symphysis could produce similar results in the microstructure of the bone bridge. |
| Shahnaseri et al., 2020; Iran [28 | True experimental, post-test only control group design | Four male canines | Unilateral alveolar bone cleft (15 mm) | Autologous AMSCs osteogenic differentiated seeded in HA/ β-TCP scaffold, Autologous tibia bone graft |
Densitometer software with digital radiography: bone density histomorphometric: Bone regeneration (%) |
Bone density and bone regeneration in autologous tibia bone grafts and autologous AMSCs with osteogenic differentiation planted in HA/ β-TCP scaffolds did not differ in a statistically meaningful way. |
As a suitable and clinically reliable technique, autologous AMSCs that are osteogenically differentiated and seeded in HA/ β-TCP scaffolds can be used to reconstruct bone defects in patients who are unable to receive autogenous bone grafting when the size of the defect restricts the size of the autograft. |
| de Ruiter et al., 2015; Netherland [29] | Prospective clinical study | Seven CL/P patients (5 males and 2 females) | Unilateral alveolar cleft | Micro-structured β-TCP |
CBCT: Cleft Volume Pre-operation; Graft Volume Post-operation; Bone Volume 6 Months Post-operation |
The bone volume thus gained was satisfactory six months following the surgical grafting of micro-structured β-TCP into the alveolar cleft. Comparing the average bone volume to the initial cleft volume, 73% to 6%. |
The therapeutic application of microstructured β-TCP bone replacement in alveolar cleft repair. |
| Pourebrahim et al., 2013; Iran [30] | True experimental, post-test only control group design | Four male mongrel dogs |
15 mm Alveolar cleft in the crest to nasal floor by removal of two of the three incisors bilaterally |
autogenous tibial graft, HA/ β-TCP loaded canine AMSCs |
Histomorphometric: bone regeneration (%) and collagen regeneration (%) | At 15 and 60 days, the autograft sides had more bone growth than the stem cell sides, at 45% and 96%, compared to 5% and 70%, respectively. There were noticeable differences between the two groups at 15 and 60 days. . |
Even though autografts are the best option for bone regeneration, tissue-engineered HA/ β-TCP -loaded cAMSCs might be a viable alternative, particularly if autografts are hard to come by or there is donor site morbidity. |
| Huang et al., 2015; China [31] | True experimental, post-test only control group design | Fourteen 24-week-old male beagles |
Unilateral alveolar bone defect with 15 mm size |
Autogenous iliac crest bone graft; tissue-engineered bone (TEB) BMSCs/ β-TCP with rapid maxillary expansion (RME) |
Occlusal radiograph: Height of the bonegraft; Histomorphometric: Bone formation (%) |
In comparison to untreated dogs or dogs just receiving autogenous iliac bone after 8 weeks of therapy with TEB BMSCs- β-TCP and RME, the dogs' new bone production and mineralization were dramatically accelerated. Bone resorption was also significantly reduced. The bone trabecula was definitely more plentiful, and the region of bone growth was bigger after treatment with both TEB and RME. The groups treated with autogenous bone and RME and the BMSCs/-TCP group did not differ significantly from one another. |
In addition to being an effective therapeutic treatment for individuals with alveolar clefts, BMSCs-β-TCP also have the capacity to replace autogenous bone, and their combination with RME may be another option for treating alveolar clefts. |
| Weijs et al., 2010; Netherland [32] |
Clinical study | Forty-seven CL/P (24 males and 23 females) patients | Unilateral alveolar cleft | Autogenous mandibular symphyseal bone only, mandibular symphyseal bone wrapped in / β-TCP granules |
Occlusal radiograph: alveolar height and eruption disturbance. | There was no discernible difference in alveolar height or eruption disruption between the β-TCP granule group and the mandibular symphysis bone alone. |
Autogenous mandibular symphyseal bone grafts enhanced with β-TCP granules can be utilized effectively in circumstances when the alveolar cleft is too big to be grafted with mandibular symphyseal bone alone. Orthodontic treatment is not impeded by the use of β-TCP since the cleft-sided canine may still be placed in its normal position. |
| Ito et al., 2019; Japan [33] | True experimental, post-test only control group design | Twenty male Sprague-Dawley rats | Alveolar bone cleft in the palatine | autogenous bone graft, β-TCP. | µCT: BV and BMD Histology analysis: Osteoblast, osteoclast, Alkaline phosphatase, Tartate Resistant Acid Phosphatase |
Autologous bone grafts had a considerably larger bone volume and BMD than β-TCP. An autologous bone transplant and β-TCP histological investigation indicated extensive braided bone ingrowth. The presence of osteoblasts at the leading margins of freshly produced bone was shown by ALP staining. TRAP staining indicated the presence of a few multinucleated large cells on the surface of the freshly produced bone. β-TCP resulted in lower BV and BMD than autologous bone transplants. |
β-TCP resulted in lower bone volume and BMD than autologous bone transplants. |
| Möhlhenrich et al., 2022 [34] | True experimental, post-test only control group design | Twenty-one male Wistar rats (R. novergius) | Alveolar bone cleft | Autografts, human xenografts and synthetic bone substitute β-TCP/HA | μCT and histopathological investigation: tooth movement, and root resorption. |
The differences in root resorption and tooth movement between the bone graft replacements, as well as autologous bone as the gold standard, were not statistically significant at any time. |
Autografts, human xenografts, and synthetic bone substitutes used for cleft repair all appear to have a comparable influence on later orthodontic tooth movement and root resorption. |
| Hossain et al., 1996; Bangladesh [35] | True experimental, post-test only control group design | Nine male beagles dog | Alveolar bone cleft | Autogenous particulate marrow and Cancellous bone (PMCB), β-TCP and combination with experimental tooth movement |
Radiograph analysis: Bone deposition histopathological investigation: bone regeneration |
Autogenous bone and β-TCP showed similar adaptive alterations to alveolar bone. β-TCP showed a more pronounced biodegradative reaction to orthodontic force in connection with the production of new cementum. Root resorption was considerably lower in the β-TCP region than in the PMCB zone. | β-TCP is a more biocompatible option to autogenous bone transplantation into alveolar bone cleft defects that support orthodontic tooth movement. |
| de Ruiter et al., 2011; Netherland [36] | True experimental, post-test only control group design | Ten adult Dutch milk goats (Capra hircus) | Alveolar bone cleft | β-TCP, autologous iliac crest bone graft |
Histologic assessment: new bone formation and bone graft resorption. radiographic measurement: orthodontic tooth movement . |
An average tooth movement of 43.2% was measured in clefts restored with iliac bone and 41% in clefts rebuilt with β-TCP. There was no statistically significant difference between groups. Nonetheless, equivalent bone volumes are generated in the alveolar clefts following grafting of either β-TCP or autologous iliac crest bone. After grafting with β-TCP, the total quantity of bone discovered in all ten goats was roughly 10% higher. This difference, however, was not statistically significant, and no association between percentage of bone volume and degree of orthodontic movement was found. |
The bone substitute β-TCP is at least as successful as autologous iliac crest bone in the healing of alveolar clefts in goats, according to surgical, orthodontic, histologic, and radiologic viewpoints. |
| Zhang et al., 2011; China [37] | True experimental, post-test only control group design | Six canines | Alveolar bone cleft | Porous β-TCP combined with osteogenically induced BMSCs and autologous iliac bone with experimental tooth movement |
occlusal radiographic: repaired alveolar cleft, residual alveolar height (%). Immunofluorescence: rate of bone formation and mineralization. Histological examination: area of the residual scaffold in the grafted region (%) and area of bone formation in the grafted region (%) |
When compared to β-TCP alone, which was absorbed severely, BMSC-porous b-TCP significantly encouraged new bone formation and mineralization and attained a satisfactory height of the repaired alveolar cleft. The tissue-engineered bone had the same overall effect as autologous bone; the physiological function of the alveolar bone was restored by permitting the surrounding teeth to slide into the newly created bone in the grafted location. | For patients with alveolar clefts and resultant orthodontic tooth movement, porous β-TCP in combination with osteogenically produced BMSCs may be a practical therapeutic technique as a replacement for bone transplants. |
| Janssen et al., 2017, Netherland [38] | True experimental, post-test only control group design | Ten Dutch milk goats |
Alveolar bone cleft defect | microstructured beta-tricalcium phosphate (β-TCP) putty with autologous iliac bone |
histomorphometric and μCT: bone quality and BV/TV |
There was no statistically significant difference between cleft sites and bone area percentages in β-TCP -CMCG and autologous bone grafts. |
In a goat model, β-TCP -CMCG putty provides superior surgical handling in the correction of alveolar cleft deformities. |
| Ekin et al., 2015; Turkey [39] | True experimental, post-test only control group design | Fifty-six Sprague-Dawley rats | critical-sized alveolar bone cleft defect | autograft, col/ β-TCP scaffolds, and PLLA/PCL scaffolds |
μCT: mineralized matrix formation, new bone formation, BV HPA: defect healing and new bone formation. RT-qPCR: Runx2, OSC, SPARC, BSP, ALP, and OSX |
The autograft group had the best new bone volume rate at 1 month and 4 months, with a significant difference. Even though the col/ β-TCP group had a greater new bone volume rate than the PLLA/PCL group, the difference was not significant. The BSP and ALP gene expression levels were considerably greater in the autograft and col/ β-TCP groups than in the blank defect group. |
Although autogenic bone grafts remain the gold standard treatment for alveolar defects, the synthetic tissue scaffolds reported herein have significant potential as an alternative treatment option when cost, donor region morbidity, and hospitalization time are considered. |
| Tokugawa et al., 2012; Japan [40] | True experimental, post-test only control group design | Ten female beagles dogs |
Alveolar bone cleft defect | Canine BMSCs cultured on β-TCP, β-TCP. | μCT: BMD (mg/cm3) Bone mineral content (mg/mm), histopathological investigation: bone regeneration. |
The regenerated bone in the MSCs/ β-TCP group exhibited a bone mineral density that was midway between that of normal bone and that of β-TCP only. The MSCs/b-TCP group had three times the bone mineral content of the cleft region as the other two groups. In the MSCs/ β-TCP group, histological investigation demonstrated that bone had replaced by the artificial bone. | cBMSCs-β-TCP based bone regeneration offer a less invasive alternative to standard cancellous iliac bone autografts for alveolar bone replacement. |
| Authors, Year, Country | CL/P or alveolar cleft defect | β-TCP utilization | Sample Preparation | Randomization | Blind Examiner | Test method clearly reported | Complete results | Risk of Bias |
|---|---|---|---|---|---|---|---|---|
| Möhlhenrich et al., 2021, German [21] | Y | Y | Y | Y | Y | Y | Y | Low |
| Trujillo et al., 2018, USA [22] | Y | Y | Y | N | N | Y | Y | Moderate |
| Putri et al., 2022, Indonesia [23] | Y | Y | Y | Y | Y | Y | Y | Low |
| Janssen et al., 2019, Netherland [24] | Y | Y | Y | N | N | Y | Y | Moderate |
| Kamal et al., 2017 German [25] | Y | Y | Y | Y | Y | Y | Y | Low |
| Du et al., 2017; China [26] | Y | Y | Y | Y | Y | Y | Y | Low |
| Miyagawa et al., 2020; Japan [27 | Y | Y | Y | N | N | Y | Y | Moderate |
| Shahnaseri et al., 2020; Iran [28 | Y | Y | Y | N | N | N | N | High |
| de Ruiter et al., 2015; Netherland [29] | Y | Y | Y | N | N | Y | Y | Moderate |
| Pourebrahim et al., 2013; Iran [30] | Y | Y | Y | Y | Y | Y | Y | Low |
| Huang et al., 2015; China [31] | Y | Y | Y | Y | Y | Y | Y | Low |
| Weijs et al., 2010; Netherland [32] |
Y | Y | Y | N | N | N | Y | Moderate |
| Ito et al., 2019; Japan [33] | Y | Y | Y | Y | Y | Y | Y | Low |
| Möhlhenrich et al., 2022 [34] | Y | Y | Y | Y | Y | Y | Y | Low |
| Hossain et al., 1996; Bangladesh [35] | Y | Y | Y | Y | N | N | Y | Moderate |
| de Ruiter et al., 2011; Netherland [36] | Y | Y | Y | Y | Y | Y | Y | Low |
| Zhang et al., 2011; China [37] | Y | Y | Y | Y | Y | Y | Y | Low |
| Janssen et al., 2017, Netherland [38] | Y | Y | Y | Y | Y | Y | Y | Low |
| Ekin et al., 2015; Turkey [39] | Y | Y | Y | Y | Y | Y | Y | Low |
| Tokugawa et al., 2012; Japan [40] | Y | Y | Y | Y | Y | Y | Y | Low |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





