Submitted:
02 August 2023
Posted:
03 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
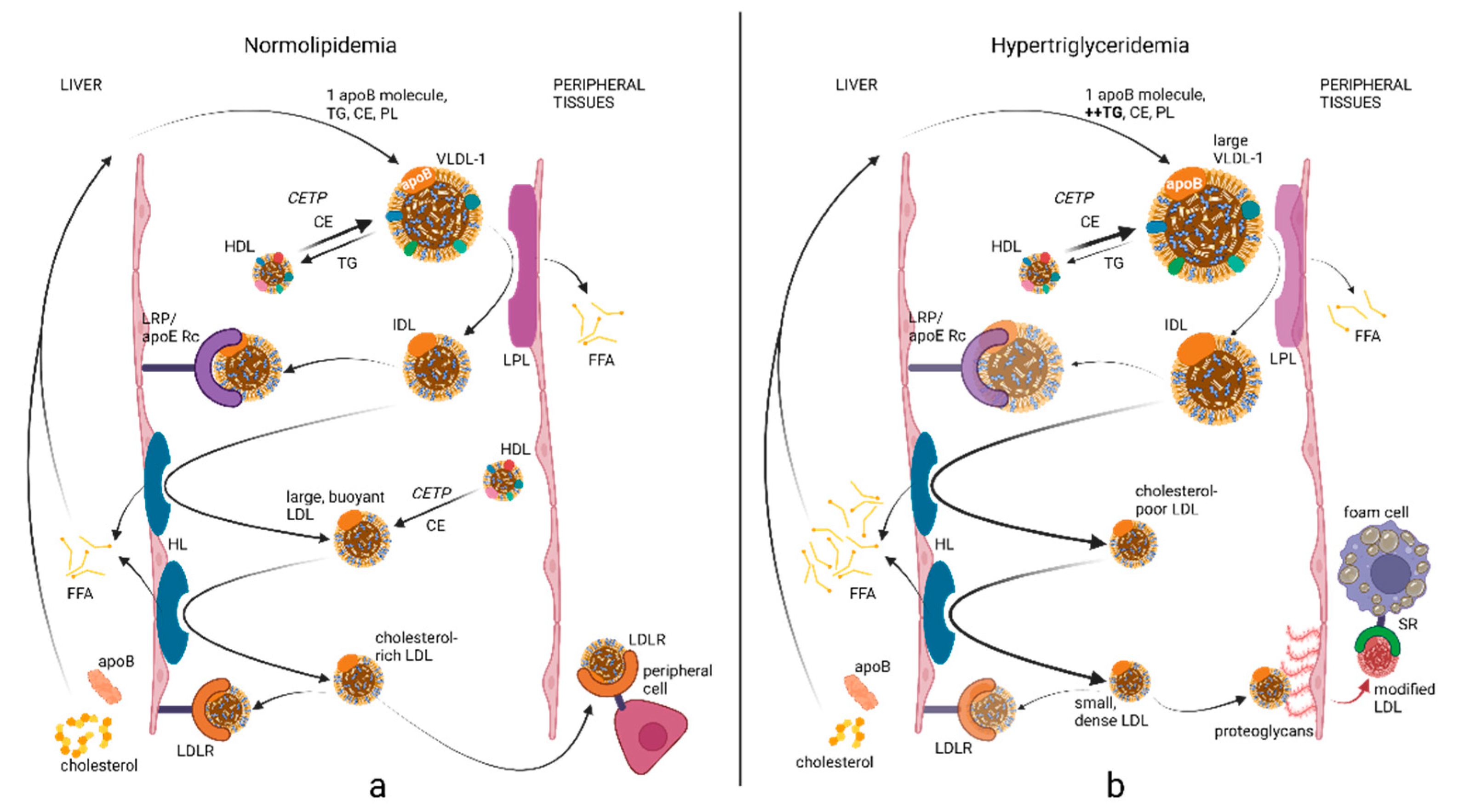
2. ApoB Biochemistry and Lipoprotein Metabolism
3. Clinical Utility of ApoB in Primary Prevention
4. Clinical Utility of ApoB in Secondary Prevention
5. Assay Standardization
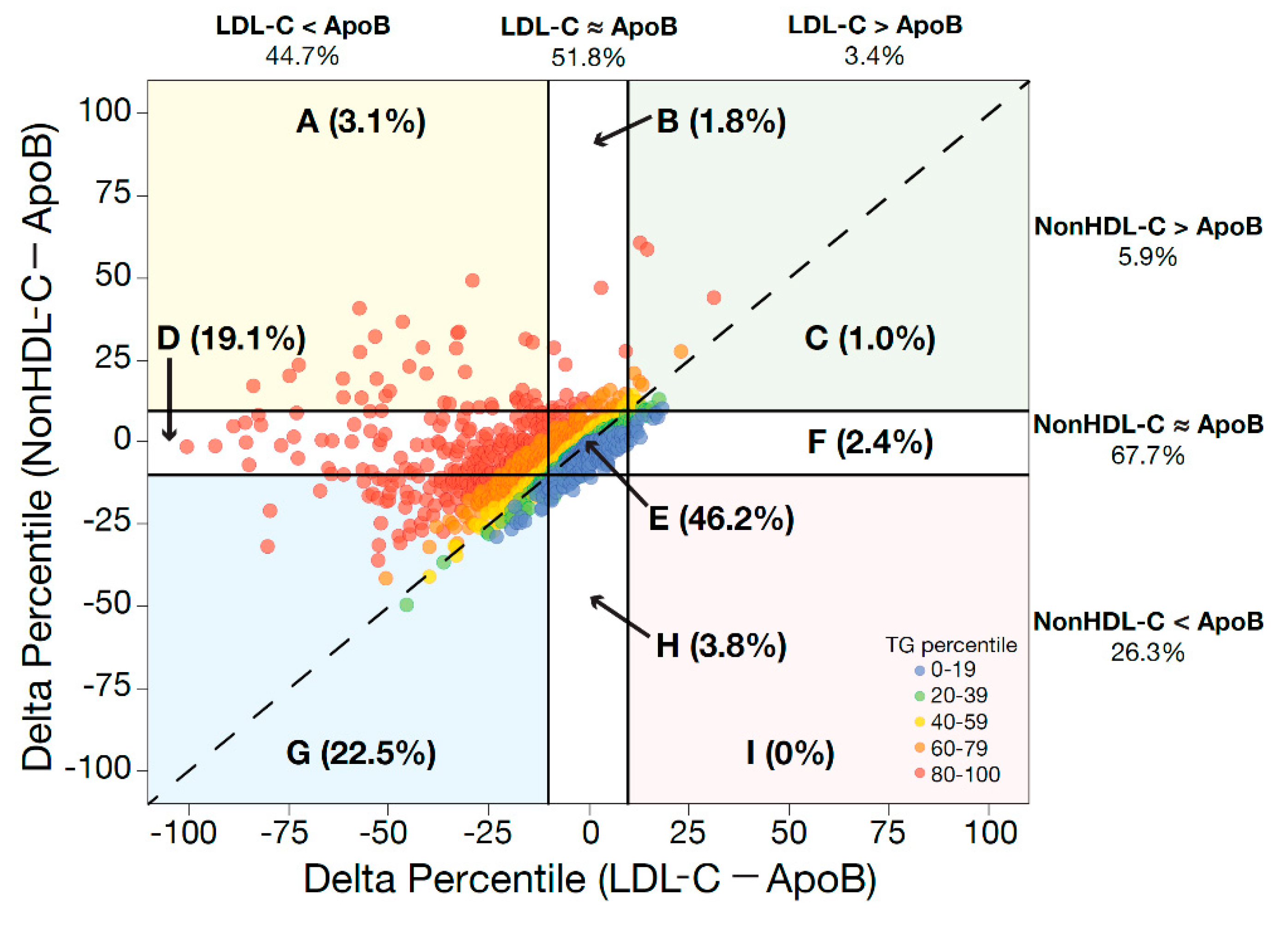
6. Utility of ApoB Versus Non-HDL-C as ASCVD Risk Markers
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jones, D.S.; Greene, J.A. The decline and rise of coronary heart disease: understanding public health catastrophism. American journal of public health 2013, 103, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Gofman, J.W.; Jones, H.B.; Lindgren, F.T.; Lyon, T.P.; Elliott, H.A.; Strisower, B. Blood lipids and human atherosclerosis. Circulation 1950, 2, 161–178. [Google Scholar] [CrossRef] [PubMed]
- Olson, R.E. Prevention and control of chronic disease. I. Cardiovascular disease--with particular attention to atherosclerosis. American journal of public health and the nation's health 1959, 49, 1120–1128. [Google Scholar] [CrossRef]
- Brown, M.S.; Goldstein, J.L. Familial hypercholesterolemia: defective binding of lipoproteins to cultured fibroblasts associated with impaired regulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity. Proceedings of the National Academy of Sciences of the United States of America 1974, 71, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. The Expert Panel. Archives of internal medicine 1988, 148, 36–69. [CrossRef]
- Contois, J.H.; McConnell, J.P.; Sethi, A.A.; Csako, G.; Devaraj, S.; Hoefner, D.M.; Warnick, G.R.; Lipoproteins, A.; Vascular Diseases Division Working Group on Best, P. Apolipoprotein B and cardiovascular disease risk: position statement from the AACC Lipoproteins and Vascular Diseases Division Working Group on Best Practices. Clin Chem 2009, 55, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Cromwell, W.C.; Otvos, J.D.; Keyes, M.J.; Pencina, M.J.; Sullivan, L.; Vasan, R.S.; Wilson, P.W.; D'Agostino, R.B. LDL Particle Number and Risk of Future Cardiovascular Disease in the Framingham Offspring Study - Implications for LDL Management. J Clin Lipidol 2007, 1, 583–592. [Google Scholar] [CrossRef]
- Kuller, L.; Arnold, A.; Tracy, R.; Otvos, J.; Burke, G.; Psaty, B.; Siscovick, D.; Freedman, D.S.; Kronmal, R. Nuclear magnetic resonance spectroscopy of lipoproteins and risk of coronary heart disease in the cardiovascular health study. Arterioscler Thromb Vasc Biol 2002, 22, 1175–1180. [Google Scholar] [CrossRef]
- Marston, N.A.; Giugliano, R.P.; Melloni, G.E.M.; Park, J.G.; Morrill, V.; Blazing, M.A.; Ference, B.; Stein, E.; Stroes, E.S.; Braunwald, E.; et al. Association of Apolipoprotein B-Containing Lipoproteins and Risk of Myocardial Infarction in Individuals With and Without Atherosclerosis: Distinguishing Between Particle Concentration, Type, and Content. JAMA Cardiol 2022, 7, 250–256. [Google Scholar] [CrossRef]
- Otvos, J.D.; Collins, D.; Freedman, D.S.; Shalaurova, I.; Schaefer, E.J.; McNamara, J.R.; Bloomfield, H.E.; Robins, S.J. Low-density lipoprotein and high-density lipoprotein particle subclasses predict coronary events and are favorably changed by gemfibrozil therapy in the Veterans Affairs High-Density Lipoprotein Intervention Trial. Circulation 2006, 113, 1556–1563. [Google Scholar] [CrossRef]
- Lipoproteins, A.; Vascular Diseases Division Working Group on Best, P.; Cole, T.G.; Contois, J.H.; Csako, G.; McConnell, J.P.; Remaley, A.T.; Devaraj, S.; Hoefner, D.M.; Mallory, T.; et al. Association of apolipoprotein B and nuclear magnetic resonance spectroscopy-derived LDL particle number with outcomes in 25 clinical studies: assessment by the AACC Lipoprotein and Vascular Diseases Division Working Group on Best Practices. Clin Chem 2013, 59, 752–770. [Google Scholar] [CrossRef]
- Caixas, A.; Ordonez-Llanos, J.; de Leiva, A.; Payes, A.; Homs, R.; Perez, A. Optimization of glycemic control by insulin therapy decreases the proportion of small dense LDL particles in diabetic patients. Diabetes 1997, 46, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.M.; Perez, A.; Calvo, F.; Bonet, R.; Castellvi, A.; Ordonez, J. Apolipoprotein(B) identifies dyslipidemic phenotypes associated with cardiovascular risk in normocholesterolemic type 2 diabetic patients. Diabetes Care 1999, 22, 812–817. [Google Scholar] [CrossRef]
- Choi, C.U.; Seo, H.S.; Lee, E.M.; Shin, S.Y.; Choi, U.J.; Na, J.O.; Lim, H.E.; Kim, J.W.; Kim, E.J.; Rha, S.W.; et al. Statins do not decrease small, dense low-density lipoprotein. Texas Heart Institute journal 2010, 37, 421–428. [Google Scholar]
- Nigon, F.; Lesnik, P.; Rouis, M.; Chapman, M.J. Discrete subspecies of human low density lipoproteins are heterogeneous in their interaction with the cellular LDL receptor. Journal of lipid research 1991, 32, 1741–1753. [Google Scholar] [CrossRef] [PubMed]
- Sniderman, A.D. Differential response of cholesterol and particle measures of atherogenic lipoproteins to LDL-lowering therapy: implications for clinical practice. J Clin Lipidol 2008, 2, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Pearson, G.J.; Thanassoulis, G.; Anderson, T.J.; Barry, A.R.; Couture, P.; Dayan, N.; Francis, G.A.; Genest, J.; Gregoire, J.; Grover, S.A.; et al. 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in Adults. Can J Cardiol 2021, 37, 1129–1150. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 139, e1082–e1143. [Google Scholar] [CrossRef]
- Meeusen, J.W.; Ueda, M.; Nordestgaard, B.G.; Remaley, A.T. Lipids and lipoproteins. In ., Elsevier eBooks+, Edition. ed.; Rifai, N., Ed.; Elsevier - OHCE: 2022; pp. 1214–1229. In Tietz Textbook of Laboratory Medicine, Elsevier eBooks+, 7th ed.; Elsevier - OHCE, 2022; pp. 1214–1229. [Google Scholar]
- Phillips, M.L.; Pullinger, C.; Kroes, I.; Kroes, J.; Hardman, D.A.; Chen, G.; Curtiss, L.K.; Gutierrez, M.M.; Kane, J.P.; Schumaker, V.N. A single copy of apolipoprotein B-48 is present on the human chylomicron remnant. Journal of lipid research 1997, 38, 1170–1177. [Google Scholar] [CrossRef]
- Elovson, J.; Chatterton, J.E.; Bell, G.T.; Schumaker, V.N.; Reuben, M.A.; Puppione, D.L.; Reeve, J.R., Jr.; Young, N.L. Plasma very low density lipoproteins contain a single molecule of apolipoprotein B. Journal of lipid research 1988, 29, 1461–1473. [Google Scholar] [CrossRef] [PubMed]
- Knott, T.J.; Pease, R.J.; Powell, L.M.; Wallis, S.C.; Rall, S.C., Jr.; Innerarity, T.L.; Blackhart, B.; Taylor, W.H.; Marcel, Y.; Milne, R.; et al. Complete protein sequence and identification of structural domains of human apolipoprotein B. Nature 1986, 323, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Lucero, D.; Wolska, A.; Aligabi, Z.; Turecamo, S.; Remaley, A.T. Lipoprotein Assessment in the twenty-first Century. Endocrinol Metab Clin North Am 2022, 51, 459–481. [Google Scholar] [CrossRef]
- Chen, S.H.; Habib, G.; Yang, C.Y.; Gu, Z.W.; Lee, B.R.; Weng, S.A.; Silberman, S.R.; Cai, S.J.; Deslypere, J.P.; Rosseneu, M.; et al. Apolipoprotein B-48 is the product of a messenger RNA with an organ-specific in-frame stop codon. Science 1987, 238, 363–366. [Google Scholar] [CrossRef]
- Berneis, K.K.; Krauss, R.M. Metabolic origins and clinical significance of LDL heterogeneity. Journal of lipid research 2002, 43, 1363–1379. [Google Scholar] [CrossRef] [PubMed]
- Meyers, N.L.; Larsson, M.; Olivecrona, G.; Small, D.M. A Pressure-dependent Model for the Regulation of Lipoprotein Lipase by Apolipoprotein C-II. J Biol Chem 2015, 290, 18029–18044. [Google Scholar] [CrossRef]
- Wolska, A.; Dunbar, R.L.; Freeman, L.A.; Ueda, M.; Amar, M.J.; Sviridov, D.O.; Remaley, A.T. Apolipoprotein C-II: New findings related to genetics, biochemistry, and role in triglyceride metabolism. Atherosclerosis 2017, 267, 49–60. [Google Scholar] [CrossRef]
- Nakajima, K.; Nakano, T.; Tokita, Y.; Nagamine, T.; Inazu, A.; Kobayashi, J.; Mabuchi, H.; Stanhope, K.L.; Havel, P.J.; Okazaki, M.; et al. Postprandial lipoprotein metabolism: VLDL vs chylomicrons. Clin Chim Acta 2011, 412, 1306–1318. [Google Scholar] [CrossRef]
- Hussain, M.M.; Kancha, R.K.; Zhou, Z.; Luchoomun, J.; Zu, H.; Bakillah, A. Chylomicron assembly and catabolism: role of apolipoproteins and receptors. Biochim Biophys Acta 1996, 1300, 151–170. [Google Scholar] [CrossRef]
- Wolska, A.; Remaley, A.T. Lipoproteins. In Handbook of Diagnostic Endocrinology, 3rd ed.; William Winter E, L.J.S., Brett Holmquist, Roger L. Bertholf, Ed.; Elsevier - Academic Press: Cambridge (MA), 2021; pp. 287–308. [Google Scholar]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 2017, 38, 2459–2472. [Google Scholar] [CrossRef]
- Segrest, J.P.; Jones, M.K.; De Loof, H.; Dashti, N. Structure of apolipoprotein B-100 in low density lipoproteins. Journal of lipid research 2001, 42, 1346–1367. [Google Scholar] [CrossRef] [PubMed]
- Sniderman, A.D.; Thanassoulis, G.; Glavinovic, T.; Navar, A.M.; Pencina, M.; Catapano, A.; Ference, B.A. Apolipoprotein B Particles and Cardiovascular Disease: A Narrative Review. JAMA Cardiol 2019, 4, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Sniderman, A.D.; Marcovina, S.M. Apolipoprotein A1 and B. Clin Lab Med 2006, 26, 733–750. [Google Scholar] [CrossRef] [PubMed]
- Austin, M.A.; Breslow, J.L.; Hennekens, C.H.; Buring, J.E.; Willett, W.C.; Krauss, R.M. Low-density lipoprotein subclass patterns and risk of myocardial infarction. JAMA 1988, 260, 1917–1921. [Google Scholar] [CrossRef] [PubMed]
- Hagstrom, E.; Steg, P.G.; Szarek, M.; Bhatt, D.L.; Bittner, V.A.; Danchin, N.; Diaz, R.; Goodman, S.G.; Harrington, R.A.; Jukema, J.W.; et al. Apolipoprotein B, Residual Cardiovascular Risk After Acute Coronary Syndrome, and Effects of Alirocumab. Circulation 2022, 146, 657–672. [Google Scholar] [CrossRef]
- Cole, J.; Otvos, J.D.; Remaley, A.T. A Translational Tool to Facilitate Use of Apolipoprotein B for Clinical Decision-Making. Clin Chem 2023, 69, 41–47. [Google Scholar] [CrossRef]
- Michos, E.D.; McEvoy, J.W.; Blumenthal, R.S. Lipid Management for the Prevention of Atherosclerotic Cardiovascular Disease. N Engl J Med 2019, 381, 1557–1567. [Google Scholar] [CrossRef]
- Leibowitz, M.; Cohen-Stavi, C.; Basu, S.; Balicer, R.D. Targeting LDL Cholesterol: Beyond Absolute Goals Toward Personalized Risk. Current cardiology reports 2017, 19, 52. [Google Scholar] [CrossRef]
- Hoogeveen, R.C.; Ballantyne, C.M. Residual Cardiovascular Risk at Low LDL: Remnants, Lipoprotein(a), and Inflammation. Clin Chem 2021, 67, 143–153. [Google Scholar] [CrossRef]
- Wolska, A.; Remaley, A.T. Measuring LDL-cholesterol: what is the best way to do it? Current opinion in cardiology 2020, 35, 405–411. [Google Scholar] [CrossRef]
- Wadstrom, B.N.; Wulff, A.B.; Pedersen, K.M.; Nordestgaard, B.G. Remnant cholesterol in the era of intensive lipid-lowering therapies. Eur Heart J 2023. [CrossRef] [PubMed]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972, 18, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Buring, J.E.; Ridker, P.M. Discordance of low-density lipoprotein (LDL) cholesterol with alternative LDL-related measures and future coronary events. Circulation 2014, 129, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, J.T.; Li, R.C.; Sniderman, A.; Chan, C.; Lloyd-Jones, D.M. Discordance Between Apolipoprotein B and LDL-Cholesterol in Young Adults Predicts Coronary Artery Calcification: The CARDIA Study. J Am Coll Cardiol 2016, 67, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Lawler, P.R.; Akinkuolie, A.O.; Ridker, P.M.; Sniderman, A.D.; Buring, J.E.; Glynn, R.J.; Chasman, D.I.; Mora, S. Discordance between Circulating Atherogenic Cholesterol Mass and Lipoprotein Particle Concentration in Relation to Future Coronary Events in Women. Clin Chem 2017, 63, 870–879. [Google Scholar] [CrossRef]
- Sniderman, A.D.; Islam, S.; Yusuf, S.; McQueen, M.J. Discordance analysis of apolipoprotein B and non-high density lipoprotein cholesterol as markers of cardiovascular risk in the INTERHEART study. Atherosclerosis 2012, 225, 444–449. [Google Scholar] [CrossRef]
- Pencina, M.J.; D'Agostino, R.B.; Zdrojewski, T.; Williams, K.; Thanassoulis, G.; Furberg, C.D.; Peterson, E.D.; Vasan, R.S.; Sniderman, A.D. Apolipoprotein B improves risk assessment of future coronary heart disease in the Framingham Heart Study beyond LDL-C and non-HDL-C. Eur J Prev Cardiol 2015, 22, 1321–1327. [Google Scholar] [CrossRef]
- Johannesen, C.D.L.; Mortensen, M.B.; Langsted, A.; Nordestgaard, B.G. Apolipoprotein B and Non-HDL Cholesterol Better Reflect Residual Risk Than LDL Cholesterol in Statin-Treated Patients. J Am Coll Cardiol 2021, 77, 1439–1450. [Google Scholar] [CrossRef]
- Cho, Y.; Lee, S.G.; Jee, S.H.; Kim, J.H. Hypertriglyceridemia is a major factor associated with elevated levels of small dense LDL cholesterol in patients with metabolic syndrome. Annals of laboratory medicine 2015, 35, 586–594. [Google Scholar] [CrossRef]
- Kang, H.S.; Gutin, B.; Barbeau, P.; Litaker, M.S.; Allison, J.; Le, N.A. Low-density lipoprotein particle size, central obesity, cardiovascular fitness, and insulin resistance syndrome markers in obese youths. International journal of obesity and related metabolic disorders : journal of the International Association for the Study of Obesity 2002, 26, 1030–1035. [Google Scholar] [CrossRef]
- Nikolic, D.; Katsiki, N.; Montalto, G.; Isenovic, E.R.; Mikhailidis, D.P.; Rizzo, M. Lipoprotein subfractions in metabolic syndrome and obesity: clinical significance and therapeutic approaches. Nutrients 2013, 5, 928–948. [Google Scholar] [CrossRef]
- Thanassoulis, G.; Williams, K.; Ye, K.; Brook, R.; Couture, P.; Lawler, P.R.; de Graaf, J.; Furberg, C.D.; Sniderman, A. Relations of change in plasma levels of LDL-C, non-HDL-C and apoB with risk reduction from statin therapy: a meta-analysis of randomized trials. Journal of the American Heart Association 2014, 3, e000759. [Google Scholar] [CrossRef] [PubMed]
- Emerging Risk Factors, C.; Danesh, J.; Erqou, S.; Walker, M.; Thompson, S.G.; Tipping, R.; Ford, C.; Pressel, S.; Walldius, G.; Jungner, I.; et al. The Emerging Risk Factors Collaboration: analysis of individual data on lipid, inflammatory and other markers in over 1.1 million participants in 104 prospective studies of cardiovascular diseases. European journal of epidemiology 2007, 22, 839–869. [Google Scholar] [CrossRef] [PubMed]
- Boekholdt, S.M.; Arsenault, B.J.; Mora, S.; Pedersen, T.R.; LaRosa, J.C.; Nestel, P.J.; Simes, R.J.; Durrington, P.; Hitman, G.A.; Welch, K.M.; et al. Association of LDL cholesterol, non-HDL cholesterol, and apolipoprotein B levels with risk of cardiovascular events among patients treated with statins: a meta-analysis. JAMA 2012, 307, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Sniderman, A.D.; Williams, K.; Contois, J.H.; Monroe, H.M.; McQueen, M.J.; de Graaf, J.; Furberg, C.D. A meta-analysis of low-density lipoprotein cholesterol, non-high-density lipoprotein cholesterol, and apolipoprotein B as markers of cardiovascular risk. Circulation. Cardiovascular quality and outcomes 2011, 4, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Soffer, G.; Pavlyha, M.; Ngai, C.; Thomas, T.; Holleran, S.; Ramakrishnan, R.; Karmally, W.; Nandakumar, R.; Fontanez, N.; Obunike, J.; et al. Effects of PCSK9 Inhibition With Alirocumab on Lipoprotein Metabolism in Healthy Humans. Circulation 2017, 135, 352–362. [Google Scholar] [CrossRef]
- Telford, D.E.; Sutherland, B.G.; Edwards, J.Y.; Andrews, J.D.; Barrett, P.H.; Huff, M.W. The molecular mechanisms underlying the reduction of LDL apoB-100 by ezetimibe plus simvastatin. Journal of lipid research 2007, 48, 699–708. [Google Scholar] [CrossRef]
- Toth, P.P.; Jones, S.R.; Monsalvo, M.L.; Elliott-Davey, M.; Lopez, J.A.G.; Banach, M. Effect of Evolocumab on Non-High-Density Lipoprotein Cholesterol, Apolipoprotein B, and Lipoprotein(a): A Pooled Analysis of Phase 2 and Phase 3 Studies. Journal of the American Heart Association 2020, 9, e014129. [Google Scholar] [CrossRef]
- Tremblay, A.J.; Lamarche, B.; Cohn, J.S.; Hogue, J.C.; Couture, P. Effect of ezetimibe on the in vivo kinetics of apoB-48 and apoB-100 in men with primary hypercholesterolemia. Arterioscler Thromb Vasc Biol 2006, 26, 1101–1106. [Google Scholar] [CrossRef]
- Waldmann, E.; Wu, L.; Busygina, K.; Altenhofer, J.; Henze, K.; Folwaczny, A.; Parhofer, K.G. Effect of PCSK9 inhibition with evolocumab on lipoprotein subfractions in familial dysbetalipoproteinemia (type III hyperlipidemia). PLoS One 2022, 17, e0265838. [Google Scholar] [CrossRef]
- Marcovina, S.M.; Albers, J.J.; Kennedy, H.; Mei, J.V.; Henderson, L.O.; Hannon, W.H. International Federation of Clinical Chemistry standardization project for measurements of apolipoproteins A-I and B. IV. Comparability of apolipoprotein B values by use of International Reference Material. Clin Chem 1994, 40, 586–592. [Google Scholar] [CrossRef] [PubMed]
- College of American Pathologists. Surveys and Anatomic Pathology Education Programs. Chemistry/Therapeutic, Drug Monitoring. Participant Summary; CAP, 2022.
- Cobbaert, C.; Weykamp, C.; Baadenhuijsen, H.; Kuypers, A.; Lindemans, J.; Jansen, R. Selection, preparation, and characterization of commutable frozen human serum pools as potential secondary reference materials for lipid and apolipoprotein measurements: study within the framework of the Dutch project "Calibration 2000". Clin Chem 2002, 48, 1526–1538. [Google Scholar] [CrossRef]
- Langlois, M.R.; Chapman, M.J.; Cobbaert, C.; Mora, S.; Remaley, A.T.; Ros, E.; Watts, G.F.; Boren, J.; Baum, H.; Bruckert, E.; et al. Quantifying Atherogenic Lipoproteins: Current and Future Challenges in the Era of Personalized Medicine and Very Low Concentrations of LDL Cholesterol. A Consensus Statement from EAS and EFLM. Clin Chem 2018, 64, 1006–1033. [Google Scholar] [CrossRef] [PubMed]
- Cobbaert, C.M.; Althaus, H.; Begcevic Brkovic, I.; Ceglarek, U.; Coassin, S.; Delatour, V.; Deprez, L.; Dikaios, I.; Dittrich, J.; Hoofnagle, A.N.; et al. Towards an SI-Traceable Reference Measurement System for Seven Serum Apolipoproteins Using Bottom-Up Quantitative Proteomics: Conceptual Approach Enabled by Cross-Disciplinary/Cross-Sector Collaboration. Clin Chem 2021, 67, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.G.; Myers, G.L.; Sakurabayashi, I.; Bachmann, L.M.; Caudill, S.P.; Dziekonski, A.; Edwards, S.; Kimberly, M.M.; Korzun, W.J.; Leary, E.T.; et al. Seven direct methods for measuring HDL and LDL cholesterol compared with ultracentrifugation reference measurement procedures. Clin Chem 2010, 56, 977–986. [Google Scholar] [CrossRef]
- van Deventer, H.E.; Miller, W.G.; Myers, G.L.; Sakurabayashi, I.; Bachmann, L.M.; Caudill, S.P.; Dziekonski, A.; Edwards, S.; Kimberly, M.M.; Korzun, W.J.; et al. Non-HDL cholesterol shows improved accuracy for cardiovascular risk score classification compared to direct or calculated LDL cholesterol in a dyslipidemic population. Clin Chem 2011, 57, 490–501. [Google Scholar] [CrossRef]
- Charlton-Menys, V.; Betteridge, D.J.; Colhoun, H.; Fuller, J.; France, M.; Hitman, G.A.; Livingstone, S.J.; Neil, H.A.; Newman, C.B.; Szarek, M.; et al. Targets of statin therapy: LDL cholesterol, non-HDL cholesterol, and apolipoprotein B in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS). Clin Chem 2009, 55, 473–480. [Google Scholar] [CrossRef]
- Pencina, M.J.; Navar, A.M.; Wojdyla, D.; Sanchez, R.J.; Khan, I.; Elassal, J.; D'Agostino, R.B., Sr.; Peterson, E.D.; Sniderman, A.D. Quantifying Importance of Major Risk Factors for Coronary Heart Disease. Circulation 2019, 139, 1603–1611. [Google Scholar] [CrossRef]
- Sniderman, A.D.; Pencina, M.; Thanassoulis, G. Limitations in the conventional assessment of the incremental value of predictors of cardiovascular risk. Curr Opin Lipidol 2015, 26, 210–214. [Google Scholar] [CrossRef]
- Benn, M.; Nordestgaard, B.G.; Jensen, G.B.; Tybjaerg-Hansen, A. Improving prediction of ischemic cardiovascular disease in the general population using apolipoprotein B: the Copenhagen City Heart Study. Arterioscler Thromb Vasc Biol 2007, 27, 661–670. [Google Scholar] [CrossRef]
- Langlois, M.R.; Sniderman, A.D. Non-HDL Cholesterol or apoB: Which to Prefer as a Target for the Prevention of Atherosclerotic Cardiovascular Disease? Current cardiology reports 2020, 22, 67. [Google Scholar] [CrossRef] [PubMed]
- Kohli-Lynch, C.N.; Thanassoulis, G.; Moran, A.E.; Sniderman, A.D. The clinical utility of apoB versus LDL-C/non-HDL-C. Clin Chim Acta 2020, 508, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.G.; Ray, K. Moving Toward the Next Paradigm for Cardiovascular Prevention. Circulation 2016, 133, 1533–1536. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.R.; Ridker, P.M. Calibration of the Pooled Cohort Equations for Atherosclerotic Cardiovascular Disease: An Update. Annals of internal medicine 2016, 165, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Kavousi, M.; Leening, M.J.; Nanchen, D.; Greenland, P.; Graham, I.M.; Steyerberg, E.W.; Ikram, M.A.; Stricker, B.H.; Hofman, A.; Franco, O.H. Comparison of application of the ACC/AHA guidelines, Adult Treatment Panel III guidelines, and European Society of Cardiology guidelines for cardiovascular disease prevention in a European cohort. JAMA 2014, 311, 1416–1423. [Google Scholar] [CrossRef]
- Ridker, P.M.; Cook, N.R. Statins: new American guidelines for prevention of cardiovascular disease. Lancet 2013, 382, 1762–1765. [Google Scholar] [CrossRef]
| β-Lipoprotein Cut-offs to Initiate Statins Defined in the 2021 Canadian Guideline (18) | |||
| Framingham Risk Score | LDL-C | ApoB | Non-HDL-C |
| <10% | ≥5.0 mmol/L | ≥1.45 g/L | ≥5.8 mmol/L |
| 5-9.9% with other CV risk factors | ≥3.5 mmol/L | ≥1.05 g/L | ≥4.2 mmol/L |
| 10-19.9% | ≥3.5 mmol/L | ≥1.05 g/L | ≥3.5 mmol/L |
| Statin Treatment Targets Recommended in the 2021 Canadian Guideline (18) | |||
| Statin indication | LDL-C | ApoB | Non-HDL-C |
| FH or genetic dyslipidaemia | <2.5 mmol/L | <0.85 g/L | <3.2 mmol/L |
| Intermediate or high risk, DM2, CKD | <2.0 mmol/L | <0.8 g/L | <2.6 mmol/L |
| ASCVD for ezetimibe | <1.8 mmol/L | <0.7 g/L | <2.4 mmol/L |
| ASCVD for PCSK9i | ≤2.2 mmol/L | ≤0.8 g/L | ≤2.9 mmol/L |
| Treatment Targets Recommended in the 2019 European Guideline (19) | |||
| Risk Group | LDL-C | ApoB | Non-HDL-C |
| Moderate | <2.6 mmol/L | <1.0 g/L | <3.4 mmol/L |
| High | <1.8 mmol/L | <0.8 g/L | <2.6 mmol/L |
| Very High | <1.4 mmol/L | <0.65 g/L | <2.2 mmol/L |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





