1. Introduction
In order to adapt to a changing environment, all living organisms have to respond appropriately to circumstances. Unlike animals, plants are unable to move away from extremes in their surrounding environment or move toward a nutrition source. However, plants have a flexible pattern of development that allows them to adjust their organ architecture to the changing environment. The fundamental body plan of the mature plant is generated during the early stages of embryogenesis (Jurgens et al., 1991). This process involves the production of shoot and root meristem, cotyledons, radicle, and hypocotyl. Embryogenesis of Arabidopsis begins with the already heavily polarized zygote, which divides into the upper apical and the lower basal cell respectively. The basal daughter cell further differentiates into the suspensor and the hypophysis. The suspensor is needed to stabilize the embryo in the seed and later enter apoptosis. The hypophysis divides asymmetrically and then develops into the quiescent center of the root apical meristem and the columella the radicle (Kadereit et al., 2014). In animals, most organs are already present by the time the embryo is fully formed. On the contrary, most organs in plants are formed after embryogenesis is finished.
GABA is a four-carbon non-protein amino acid found in all life domains. It was discovered in plants some years ago (Steward et al., 1949). In plants, it was shown to play a critical role in pollen-tube guidance in the process of reproduction (Palanivelu et al. 2003). They reported that a gradient of GABA is formed from the stigmatic surface toward the ovary, which is essential for successful guidance of the pollen tube and fertilization (Palanivelu et al. 2003). One of the signaling roles of GABA was reported to be stomatal regulation under water deficiency (Mekonnen et al., 2016). The modulation of reactive oxygen species was also reported as one of the key functions of GABA (Bouché et al., 2003), recently various developmental effects of GABA modulation both exogenous application (Du et al., 2020) and genetic engineering (Xie et al., 2020) was reported. GABA was also suggested to function in plant communication with bacterial (Lang et al., 2016) interactions. Furthermore, GABA-binding sites have been detected on plant cell membranes (Yu et al., 2006, Wudick et al., 2018). These discoveries further suggest that GABA functions as a signaling molecule in plants.
In addition, GABA has been found in all organs in plants, including the embryo, cotyledon, roots, shoot, flowers, fruit, nodule, xylem, and phloem (Kinnersley and Turano, 2000; Hijaz and Killiny, 2020). Its concentrations vary significantly in different organs, tissues, and compartments (Ramesh et al., 2017), e.g., 100–150 μM in the xylem of soybean (Wallace et al., 1984) and up to 20 mM in tomato fruit (Yin et al., 2010). This broad range in GABA concentrations, from low micro- to millimolar, may indicate its function as a signaling molecule and primary metabolite, respectively. However, none of these studies provided solid proof of the occurrence of a GABA signaling system in plants. GABA also plays a role as a primary metabolite related to the balance between nitrogen and carbon metabolism (Fait et al., 2011). Combined transcriptomics and metabolomics of Arabidopsis seedlings exposed to exogenous GABA suggest its role in plants is predominantly metabolic (Batushansky et al., 2014). Light cues of varied intensity and quality cause plants to change their morphological traits (Yadav et al., 2020). Besides that, light also directs plant growth in a specific direction, shoots, for example, bend towards the light, which is called phototropism. Our studies show GABA’s effect on plants’ growth under different light conditions.
2. Results
2.1. Arabidopsis Thaliana in Different Light Conditions
2.1.1. Root-Shaded Light Conditions
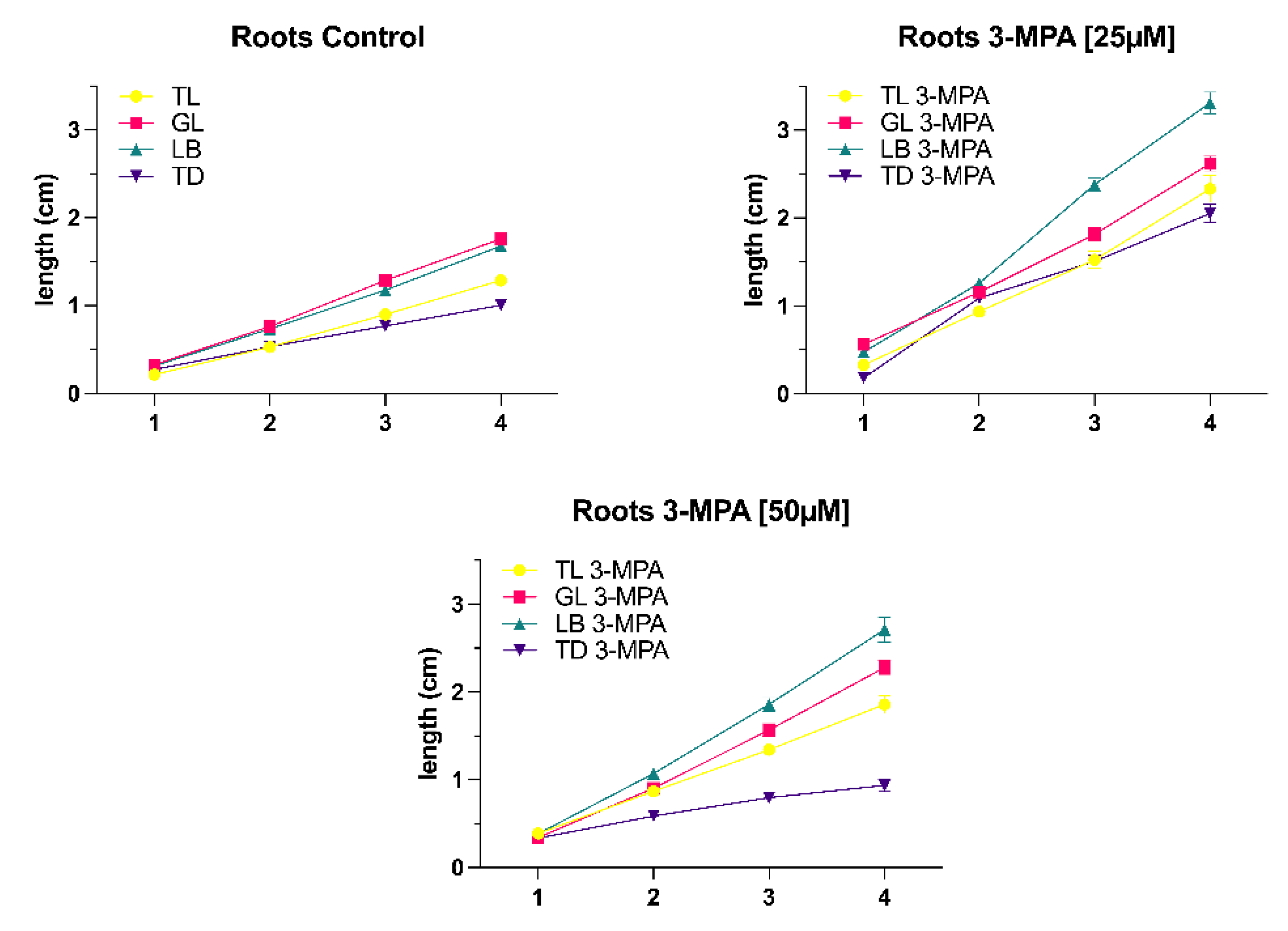
Comparing the control group and the 3-MPA [25μM] treatment group, it was noticeable that the root length of seedlings under the 3-MPA [25μM] treatment was longer than that of the control group, and the difference between the two treatments was most apparent under light blocker (LB) conditions (
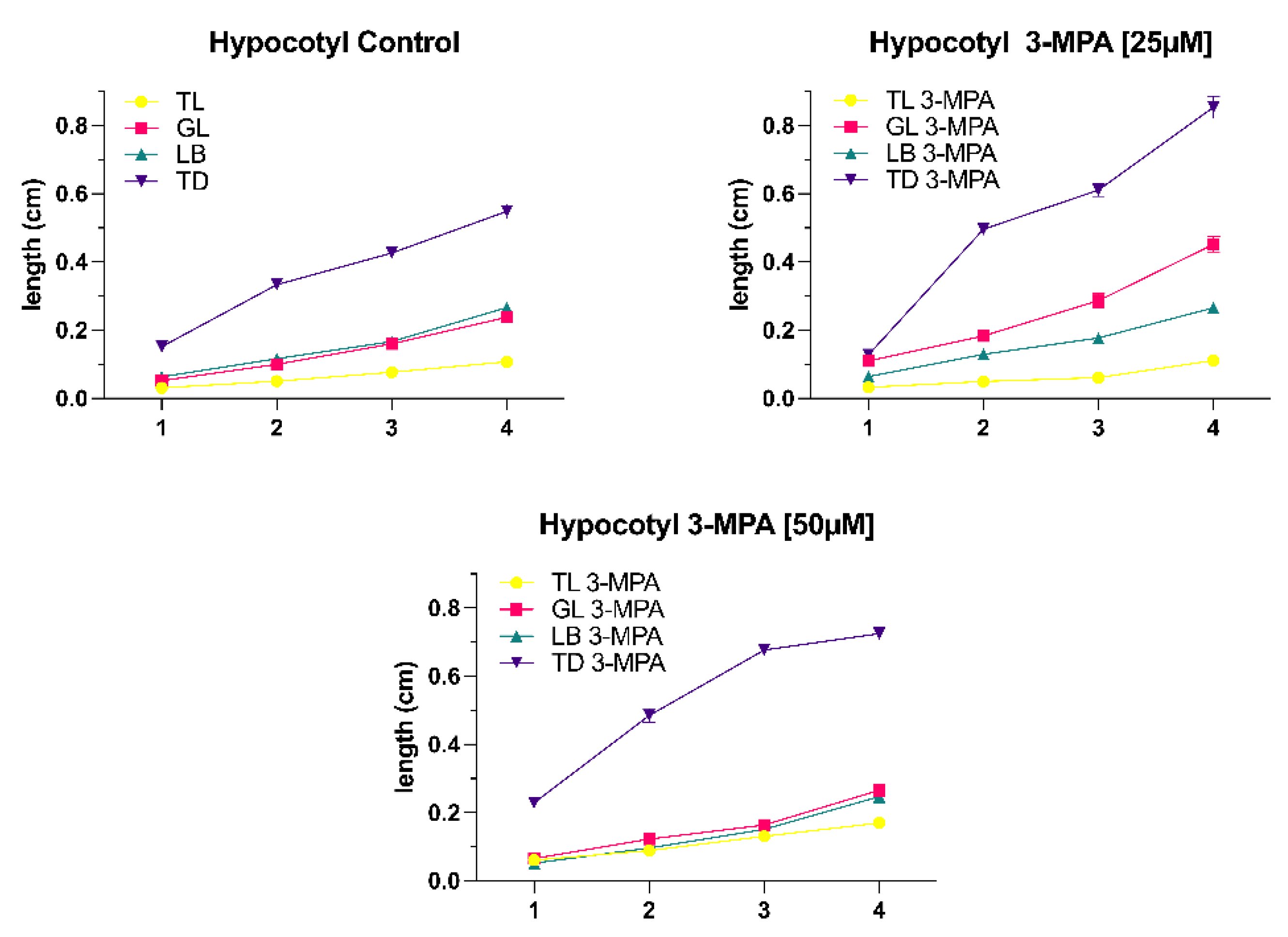
Figure 1). The hypocotyl development looked to be the same, with the 3-MPA treatment group hypocotyl being longer than the control group under the same light conditions (
Figure 2).
2.1.2. Shoot Shaded Light Conditions
After 24, 48, 72, and 96 hours, there was no significant difference in root development and hypocotyl development between the lower concentration of 3-MPA [25μM] and the control group (
Figure 2 and
Figure 3B). However, it has been discovered that when light intensity decreases, the rate of hypocotyl development increases (
Figure 3B).
The root length was longer than the control group under the same light when the 3-MPA concentration increased to 50μM (
Figure 3C). Comparing the four different light conditions, the root length of the
A. thaliana seedlings under light blocker (LB) condition varied the greatest under influence of 3-MPA, whereas the root length of the seedlings under total dark (TD) condition remained relatively constant. The length of the hypocotyl changes in the 3-MPA treatment group under varied lighting settings, as seen in
Figure 3D. Except in the total dark (TD) condition, hypocotyl length rose significantly when 3-MPA was promoted, and hypocotyl length in the other three light conditions did not alter remarkably in the control group or 3-MPA treatment.
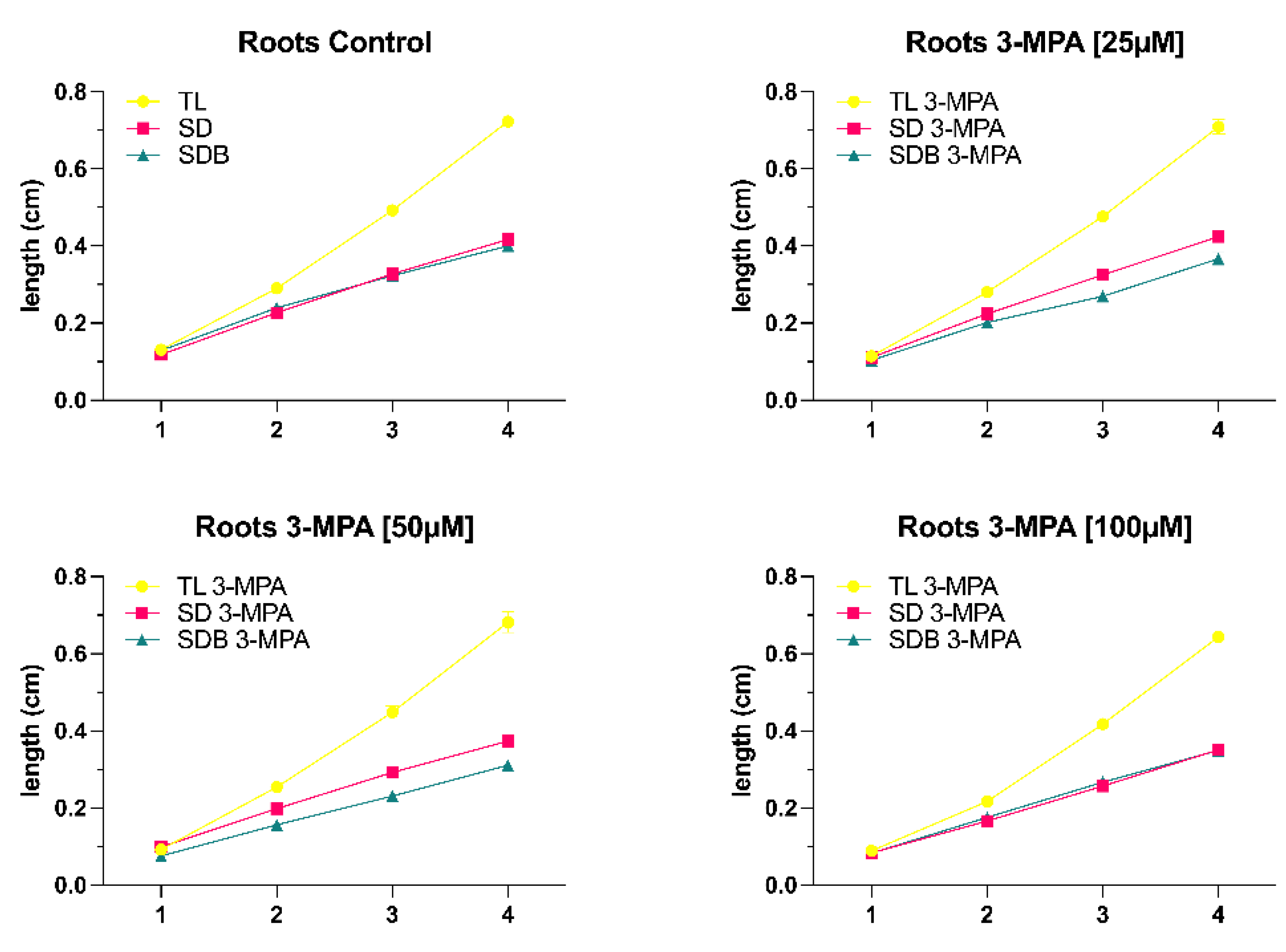
With the increase of 3-MPA concentration to [50μM], root development began to reduce under the same light conditions. However, as the time was increased from 24 to 72 hours, the difference in root growth between the control group and the 3-MPA treatment group becomes less noticeable (
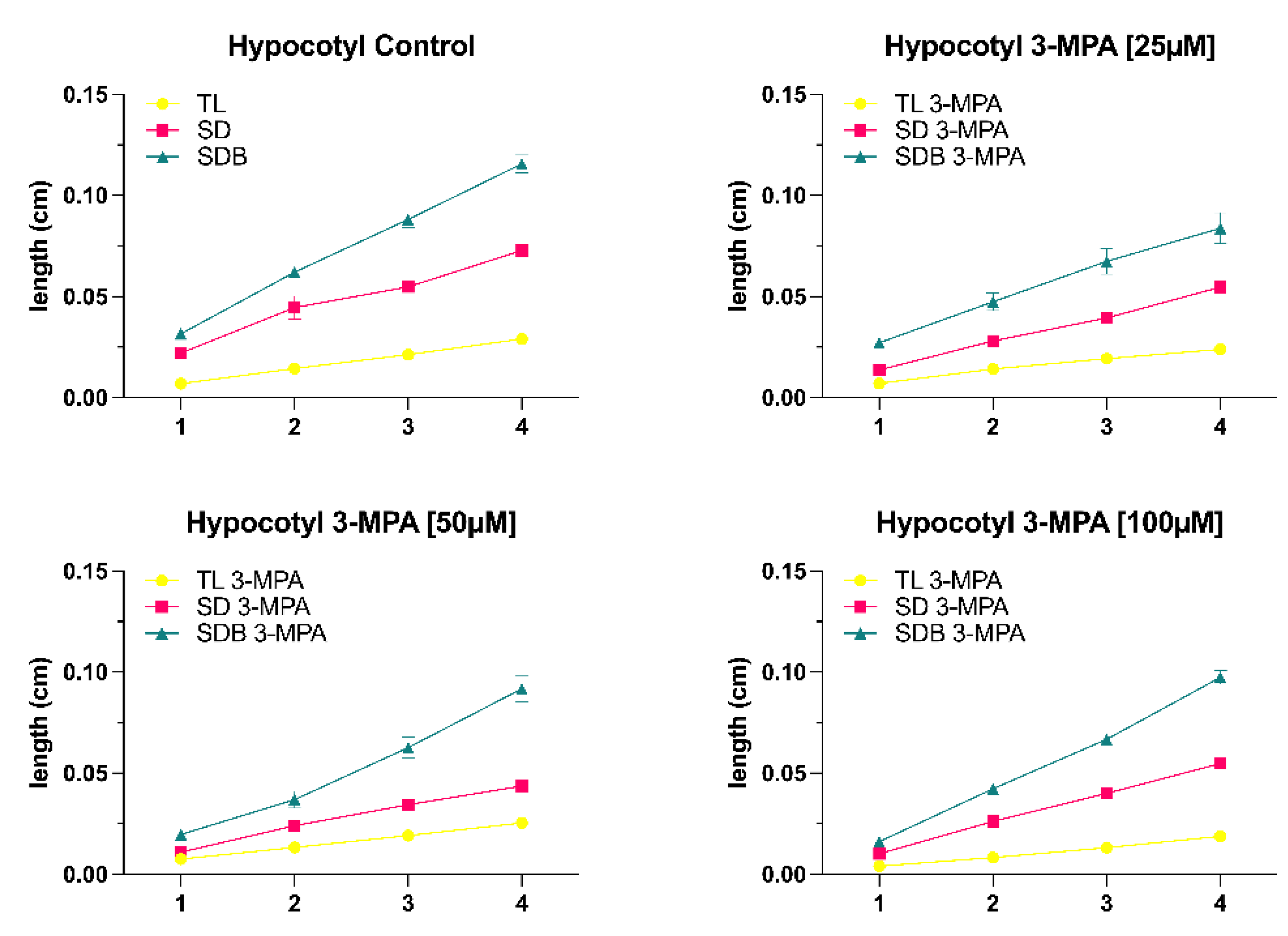
Figure 4A), and when the duration was increased to 96 hours, there was no significant difference in root growth in shoot dark (SD) and shoot dark with light blocker (SDB) conditions (Fig 4B).
The growth of hypocotyl, on the other hand, responds considerably to shoot dark (SD) and shoot dark with light blocker (SDB) conditions when the 3-MPA concentration was 50μM. Furthermore, the same phenomenon also occurred. With the increase of time, the effect of 3-MPA [50μM] on seedling hypocotyl growth was gradually less obvious (
Figure 4C).
As the 3-MPA concentration is increased to [100μM], the difference in seedling root development became increasingly apparent under the same light conditions. 3-MPA [100μM] clearly suppressed root development under the same light conditions. After 96 hours, the root length of the 3-MPA [100μM] group reached 0.6cm under total light conditions, and the root length of the other two light conditions was equally close to 0.4cm (
Figure 4D).
3. Discussion
Arabidopsis is commonly used as a model plant to study plant physiology due to its unique traits that made it ideal for laboratory research. Arabidopsis has been cultivated on Petri dishes since then, and the vast majority of root biology research has been done with the root system exposed to light. Some of these fail to point out the direct influence of light on root development and responses. Recently these influences became more visible according to recent research (Yokawa et al., 2014; Meng, 2015).
Our results have revealed a role for light in both root growth and hypocotyl growth. We set up six different light conditions (total light, gradient light, light blocker, total dark, shoot dark, and shoot dark with blocker) since the influence of light on seedling growth and differentiation can be divided into direct and indirect aspects. The light conditions (shoot dark and shoot dark with blocker) are the indirect ways to explore the influence of light on root growth via changing the light intensity on the shoot. Our investigations have found that the total root length under shoot dark and shoot dark with blocker circumstances were substantially less than under total light conditions. Within 24h and 48h, there was no significant difference in root length across the three lighting conditions, but from 72h to 96h, the total light condition had a considerably longer root length than the other two illumination conditions. This situation can be explained by the fact that photosynthesis mainly occurs in plant shoots, and plants are unable to produce enough organic matter (sucrose) to fulfill their growth requirements after a period of limited light.
Yokawa et al. (2011) discovered that the root-shoot ratio of Arabidopsis seedlings growing in the soil (whose roots are almost completely dark) is 1:1. The exposure of roots to light causes stress in the entire plant, and roots normally respond by increasing their growth. This indicates that illumination of the roots disturbs the balance of the root-shoot ratio, which is approximately 1:1 in a normal physiological situation. The analysis of our experimental data also supported this work. The gradient light condition had a higher light intensity than the simulated natural condition (light blocker), and the gradient light condition had a somewhat longer root length than the light blocker condition. Meanwhile, in the gradient light condition, the hypocotyl length was less than in the light blocker condition. Most Arabidopsis studies are conducted out in transparent Petri dishes, ignoring the extra effects that the additional light may have on the roots, as explained in the introduction. Our results reported the root length under total light conditions was significantly shorter than that under gradient light and simulated natural conditions (light blocker), confirming the shortcomings of the traditional plant-growing (TPG) technique. Silva-Navas et al. (2015) demonstrated root illumination shortens root length and increases the early development of lateral roots, promoting root system expansion. They discovered that roots grown under full light produce shorter roots and more emerged lateral roots than roots produced in nearly full darkness (simulated natural condition). This is also in exact agreement with the results of our experiments. This situation can be explained by the potential reasons: (1) light diminishes the accumulation of potassium, sodium, and molybdenum in roots while dramatically increasing the absorption of iron in roots and shoots (Silva-Navas et al., 2015). Because light photocatalyses reactive oxygen species (ROS) formation in roots (Yokawa et al., 2011) and iron solubilization happens via redox processes, iron buildup in roots under light might be due to ROS activation. All living organisms require iron, and it may be a growth-limiting resource for plants since it is a fundamental component of redox reactions in photosynthesis and respiration (Silva-Navas et al., 2015). (2) As root illumination induces the burst of ROS (Yokawa et al., 2011). Higher levels of ROS in roots may break the equilibrium between root development and lateral root emergence (Tsukagoshi et al., 2010; Passaia et al., 2014). Furthermore, ROS is a strong oxidant that may react with a wide range of biomolecules, causing significant damage to plant tissues (Petrov and Van Breusegem, 2012).
The hypocotyl is a highly plastic organ whose length is controlled by a network of interacting elements including light and plant hormones. In continuous darkness, the process of hypocotyl elongation differs significantly from that in uniform light. Figure 48 depicts an overview of the major signals that contribute to hypocotyl development in Arabidopsis. GA, BR, and auxin may induce hypocotyl elongation in etiolated plants via downstream actuators (Figure 48A).
It is well known that photoreceptors in light-grown plants suppress the biosynthesis of the hormones GA, BR, and auxin. Cytokinin’s stimulate ethylene synthesis, and ethylene influences actuators via regulation of the auxin or GA signal or directly on downstream actuators. Our results demonstrated that the length of the hypocotyl under dark conditions (total dark, shoot dark, and shoot dark with blocker) was considerably longer than the hypocotyl under light conditions (total light, light blocker, and gradient light), and the root length of the total dark condition was significantly shorter than other light conditions (total light, gradient light, and light blocker). Plants that do not have access to light will develop a skotomorphogenesis pattern, which leads to etiolation (Yokawa et al., 2011).
As described in the introduction, modulation of ALMT activity by GABA leads to altered root development and tolerance to alkaline pH, acid pH, and aluminum ions. AtALMT1, the first recognized Arabidopsis thaliana homologue of ALMTs, was likewise shown to be involved in Al-resistance (Hoekenga et al., 2006). Aluminum ions in acid soils are toxic to plants and excessive quantities of soluble aluminum in soil solutions result in poor plant growth (Matsumoto, 2000). Plants that are resistant to higher soluble aluminum concentrations in soil release malate anions from their root cells, which chelate the toxic Al3+ cations in the apoplast (Delhaize, Ryan and Randall, 1993). Thus, the efflux modulation of organic acids from plant roots, such as malate, plays a significant role in the aluminum resistance controlled by the ALMT1 gene (Ryan, Delhaize and Jones, 2001). According to Ramesh et al. (2017), the application of 3-MPA may interact with the predicted GABA-binding region in ALMTs, elucidating the molecular identity and basis of GABA control of ion fluxes in plants. We set up treatment groups with different concentrations of 3-MPA (25μM, 50μM and 100μM). Our experimental results show that when the 3-MPA concentration is 25μM compared to the control group, the development of Arabidopsis roots and hypocotyl under six distinct light conditions is increased. This situation may be explained as follows: 3-MPA inhibits the biosynthesis of GAD which catalyzes the formation of GABA, resulting in a negative regulating impact on ALMTs on the membrane. Low concentrations of GABA increase inward currents, which in root and hypocotyl growth used could have been malate efflux or cation influx. Interestingly, the promoting tendency of root and hypocotyl development became less noticeable as 3-MPA concentration increased. The higher the 3-MPA concentration, the less GABA is produced and the more malate effluxes. This excessive carbon loss pathway is damaging to plant growth and stress resistance.
4. Materials and Methods
The growth media was prepared by mixing the MS media salt (with vitamins), saccharose, and dH2O. For 1L, 2.2g of MS media salt (with Vitamins) and 10g of Saccharose were added. After adding each in a 3L container, the pH was constantly checked (when the pH was less than 5.8, KOH was added, and when it was higher, HCl was added). After determining the pH to be 5.8, 4g of Phytagel was added to it. The media was autoclaved at 120OC. The media was placed in Petri dishes of different sizes and prepared under a sterile bench, for further usage. For the growth experiment with A. thaliana seedlings, media was added on round Petri dishes with 3-MPA 2.17μL/1L [25μM], 3-MPA 5.14μL/1L [50μM] and 3-MPA 8.68μL/1L [100μM].
Arabidopsis thaliana seeds were sterilized in a plastic tube for 3 minutes with 1 ml of 70% ethanol. This was followed by a 5-minute treatment with 1ml hypochlorite solution. The plastic tube was inverted many times in each phase. The seeds were washed five times in distilled water. Sterilized seeds were sown on square Petri dishes with ½ MS medium under the sterile bench as further described in (Njimona et al. 2021). The square Petri dishes with sterilized seeds were stored in the fridge for stratification for 48h at 4°C and transferred to the growth chamber for 96h for seed germination.
To investigate the influence of 3-MPA on A. thaliana root growth and hypocotyl growth. A. thaliana seedlings were transferred to round Petri dishes of Phyto agar having different concentrations of 3-MPA (2.17μL/1L of 3-MPA [25μM], 5.14μL/1L of 3-MPA [50μM] and 8.68μL/1L of 3-MPA [100μM]). Control dishes for this experiment were treated in the same way but, only containing Phyto agar. After placing the seedlings, all dishes were sealed with parafilm and then transferred to different light conditions (Yan et al. 2022). Each treatment was repeated three times. The treatment of 3-MPA [100μM] was only carried out under total light and two kinds of shoot-shaded light conditions (SD and SDB). The six light conditions are as follows: Total light (TL): The round Petri dishes with A. thaliana seedlings were placed under the light of the growth chamber with the intensity of 100 μmol s-1 m-2. Total dark (TD): the plants were kept in total darkness. Moreover, shaded seedling roots create two forms of light: gradient light and light blocker. Shoot dark and shoot dark with a light blocker were two types of light conditions created by shading seedling shoots. Gradient light (GL): plants in the Petri dish were introduced in a black box resulting in a light gradient with a value of 39.74 μmol s-1 m-2. Light blocker (LB): a light blocker is placed on the plants in the Petri dish and then they were introduced into a black box, resulting in light intensity of 7.27 μmol s-1 m-2. Shoot dark (SD): The hypocotyls of A. thaliana were covered resulting in light intensity of 7.91 μmol s-1 m-2. Shoot dark with light blocker (SDB): a light blocker was placed on the seedlings in a round Petri dish and then the hypocotyls of the seedlings were covered, resulting in light intensity of 2.03 μmol s-1 m-2. Detail experimental set was described in our previous publication (Yan et al. 2022). After 24h, 48h, 72h, and 96h the round Petri dishes were scanned. On the basis of the digital images, the root length and hypocotyl length were measured via Fiji software
Collected data for the experiment were evaluated with Fiji ImageJ software. Root length and hypocotyl length were measured with ImageJ plugins The length standard for these measurements was set with the help of a ruler which was calibrated with the respective samples. Statistical analysis was performed using GraphPad Prism (version 9.1.1) for the light experiment.
5. Conclusions
Plant roots can sense light and assess spectrum and light intensity using various photoreceptors, thus integrating the growth of aboveground and underground organs. The roots of laboratory-grown Arabidopsis seedlings should be kept in darkened Petri plates (Yokawa et al., 2011, 2014; Xu et al., 2013). The illumination of roots influences not only the roots but also the morphology and physiology of the whole seedlings. Moreover, TOR has been shown to be involved in auxin signaling. However, there is no evidence that the TOR complex is a major regulator of cell proliferation and expansion. Further studies are needed to understand the functions of ROP2 in TOR activation and to identify other TOR effectors in plants (Schepetilnikov et al., 2017). Additionally, the recent discovery that plant GABA can regulate ion channels (ALMTs) has promoted GABA research, although there are still many gaps in the regulation of GABA on plant physiology and development. Meanwhile, further research has revealed that plant hormones (ethylene, ABA) and ROS production can alter GABA metabolism in plants (Ramesh et al., 2017), and some experimental evidence has shown that high GABA concentrations inhibit root growth (Ramesh et al., 2015). In our work, we have provided further clarity on this topic. However future experiments on which young plants shall be treated with exogenous GABA will further provide more evidence of the role of GABA in plant growth
6. Summary
We set up six light conditions (TL, TD, GL, LB, SD, SDB) to investigate the changes in Arabidopsis thaliana hypocotyl and root development. A. thaliana seedlings developed under absolute darkness (TD) with shorter roots and longer hypocotyls. Shoots were shaded in SD and SDB conditions, and seedlings were unable to carry out photosynthesis, resulting in insufficient stored nutrients for root development. In the three groups of different light intensities on the root (TL, GL, LB), light causes stress in the entire plant under total light, the length of the root and hypocotyl in TL condition was shorter than GL and LB conditions. The stimulated natural condition, LB, had a shorter root but a longer hypocotyl than the GL condition. Different light treatments did significantly affect root growth and hypocotyl growth. We developed four treatment groups of 3-MPA [5μM], 3-MPA [25μM], 3-MPA [50μM], and 3-MPA [100μM]). Root and hypocotyl growth was promoted at the concentration of 3-MPA [25μM], and the development of root and hypocotyl was suppressed gradually as the 3-MPA concentration increased.
Author Contributions
Conceptualization, analysis, and writing, F.Y., X.Y., I.N., and F.B.; Investigation and resources, F.Y. X.Y. and F.B.; writing—review and editing, F.Y., X.Y., I.N., and F.B.; supervision, F.B. All authors read and agreed to the published version of the manuscript.
Funding
This research was funded by Stiftung Zukunft Jetzt! (Munich, Germany).
Data Availability Statement
Not applicable.
Acknowledgments
The authors acknowledge Stiftung Zukunft Jetzt! (Munich, Germany) for the scholarships to F.Y..
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bouché, N.; Fait, A.; Bouchez, D.; Møller, S.G.; Fromm, H. Mitochondrial succinic-semialdehyde dehydrogenase of the γ-aminobutyrate shunt is required to restrict levels of reactive oxygen intermediates in plants. Proc. Natl. Acad. Sci. 2003, 100, 6843–6848. [Google Scholar] [CrossRef]
- Batushansky, A.; Kirma, M.; Grillich, N.; Toubiana, D.; Pham, P.A.; Balbo, I.; Fromm, H.; Galili, G.; Fernie, A.R.; Fait, A. Combined Transcriptomics and Metabolomics of Arabidopsis thaliana Seedlings Exposed to Exogenous GABA Suggest Its Role in Plants Is Predominantly Metabolic. Mol. Plant 2014, 7, 1065–1068. [Google Scholar] [CrossRef]
- Delhaize, E.; Ryan, P.R.; Randall, P.J. Aluminum tolerance in wheat (Triticum aestivum 1.). Plant Physiology 1993, 103, 695–702. [Google Scholar] [CrossRef]
- Du, C.; Chen, W.; Wu, Y.; Wang, G.; Zhao, J.; Sun, J.; Ji, J.; Yan, D.; Jiang, Z.; Shi, S. Effects of GABA and Vigabatrin on the Germination of Chinese Chestnut Recalcitrant Seeds and Its Implications for Seed Dormancy and Storage. Plants 2020, 9, 449. [Google Scholar] [CrossRef]
- Fait, A.; Nesi, A.N.; Angelovici, R.; Lehmann, M.; Pham, P.A.; Song, L.; Haslam, R.P.; Napier, J.A.; Galili, G.; Fernie, A.R. Targeted Enhancement of Glutamate-to-γ-Aminobutyrate Conversion in Arabidopsis Seeds Affects Carbon-Nitrogen Balance and Storage Reserves in a Development-Dependent Manner. Plant Physiol. 2011, 157, 1026–1042. [Google Scholar] [CrossRef]
- Hijaz, F.; Killiny, N. The use of deuterium-labeled gamma-aminobutyric (D6-GABA) to study uptake, translocation, and metabolism of exogenous GABA in plants. Plant Methods 2020, 16, 1–9. [Google Scholar] [CrossRef]
- Hoekenga, O.A.; et al. AtALMT1, which encodes a malate transporter, is identified as one of several genes critical for aluminum tolerance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2006, 103, 9738–9743. [Google Scholar] [CrossRef]
- Kinnersley, A.M.; Turano, F.J. Gamma Aminobutyric Acid (GABA) and Plant Responses to Stress. Crit. Rev. Plant Sci. 2000, 19, 479–509. [Google Scholar] [CrossRef]
- Lang, J.; Gonzalez-Mula, A.; Taconnat, L.; Clement, G.; Faure, D. The plant GABA signaling downregulates horizontal transfer of the Agrobacterium tumefaciens virulence plasmid. New Phytol. 2015, 210, 974–983. [Google Scholar] [CrossRef]
- Matsumoto, H. Cell biology of aluminum toxicity and tolerance in higher plants. International Review of Cytology, 2000; 200, 1–46. [Google Scholar] [CrossRef]
- Meng, L.S. Transcription coactivator Arabidopsis ANGUSTIFOLIA3 modulates anthocyanin accumulation and light-induced root elongation through transrepression of Constitutive Photomorphogenic1. Plant Cell and Environment 2015, 38, 838–851. [Google Scholar] [CrossRef]
- Mekonnen, D.W.; Flügge, U.-I.; Ludewig, F. Gamma-aminobutyric acid depletion affects stomata closure and drought tolerance of Arabidopsis thaliana. Plant Sci. 2016, 245, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Njimona, I.; Baluška, F. The role of N-terminal module of PhyB in modulating root and hypocotyl growth length in Arabidopsis. Afr. J. Biotechnol. 2022, 21, 287–291. [Google Scholar] [CrossRef]
- Palanivelu, R.; Brass, L.; Edlund, A.F.; Preuss, D. Pollen Tube Growth and Guidance Is Regulated by POP2, an Arabidopsis Gene that Controls GABA Levels. Cell 2003, 114, 47–59. [Google Scholar] [CrossRef]
- Passaia, G.; Queval, G.; Bai, J.; Margis-Pinheiro, M.; Foyer, C.H. The effects of redox controls mediated by glutathione peroxidases on root architecture in Arabidopsis thaliana. J. Exp. Bot. 2014, 65, 1403–1413. [Google Scholar] [CrossRef] [PubMed]
- Petrov, V.D.; Van Breusegem, F. Hydrogen peroxide—a central hub for information flow in plant cells. AoB PLANTS 2012, 2012, pls014. [Google Scholar] [CrossRef]
- Ramesh, S.A.; et al. γ-Aminobutyric acid (GABA) signalling in plants. Cellular and Molecular Life Sciences 2017, 74, 1577–1603. [Google Scholar]
- Ramesh, S.A.; et al. GABA signalling modulates plant growth by directly regulating the activity of plant-specifific anion transporters. Nature Communications 2015, 6, 7879. [Google Scholar] [CrossRef]
- Ryan, P.; Delhaize, E.; Jones, D. Function and mechanism of organic anion exudation from plant roots. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 527–560. [Google Scholar] [CrossRef]
- Steward, F.C.; Thompson, J.F.; Dent, C.E. γ-Aminobutyric acid: a constituent of the potato tuber? Science 1949, 110, 439–440. [Google Scholar]
- Silva-Navas, J.; Moreno-Risueno, M.A.; Manzano, C.; Pallero-Baena, M.; Navarro-Neila, S.; Téllez-Robledo, B.; Garcia-Mina, J.M.; Baigorri, R.; Gallego, F.J.; del Pozo, J.C. D-Root: a system for cultivating plants with the roots in darkness or under different light conditions. Plant J. 2015, 84, 244–255. [Google Scholar] [CrossRef]
- Schepetilnikov, M.; et al. GTPase ROP2 binds and promotes activation of target of rapamycin, TOR, in response to auxin. The EMBO Journal 2017, 36, 886–903. [Google Scholar] [PubMed]
- Tsukagoshi, H.; Busch, W.; Benfey, P.N. Transcriptional Regulation of ROS Controls Transition from Proliferation to Differentiation in the Root. Cell 2010, 143, 606–616. [Google Scholar] [CrossRef] [PubMed]
- Wallace, W.; Secor, J.; Schrader, L.E. Rapid accumulation of γ-aminobutyric acid and alanine in soybean leaves in response to an abrupt transfer to lower temperature, darkness, or mechanical manipulation. Plant Physiology 1984, 75, 170–175. [Google Scholar] [PubMed]
- Wudick, M.M.; Michard, E.; Oliveira Nunes, C.; Feijo, J.A. Comparing plant and animal glutamate receptors: common traits but different fates? Journal of Experimental Botany 2018, 69, 4151–4163. [Google Scholar]
- Xie, T.; Ji, J.; Chen, W.; Yue, J.; Du, C.; Sun, J.; Chen, L.; Jiang, Z.; Shi, S. GABA negatively regulates adventitious root development in poplar. J. Exp. Bot. 2019, 71, 1459–1474. [Google Scholar] [CrossRef]
- Xu, W.; et al. An improved agar-plate method for studying root growth and response of Arabidopsis thaliana. Scientific Reports 2013, 3, 1273. [Google Scholar] [CrossRef]
- Yan, X.; Yamashita, F.; Njimona, I.; Baluška, F. Root and hypocotyl growth of Arabidopsis seedlings grown under different light conditions and influence of TOR kinase inhibitorAZD. International Journal of Biotechnology and Molecular Biology Research 2022, 12, 22–30. [Google Scholar] [CrossRef]
- Yadav, A.; et al. Light signaling and UV-B-mediated plant growth regulation. Journal of Integrative Plant Biology 2020, 62, 1270–1292. [Google Scholar]
- Yin, Y.G.; Tominaga, T.; Iijima, Y.; Aoki, K.; Shibata, D.; Ashihara, H.; Nishimura, S.; Ezura, H.; Matsukura, C. . Metabolic alterations in organic acids and gamma-aminobutyric acid in developing tomato (Solanum lycopersicum L.) fruits. Plant & Cell Physiology 2010, 51, 1300–1314. [Google Scholar]
- Yokawa, K.; et al. Light as stress factor to plant roots – case of root halotropism. Frontiers in Plant Science 2014, 5, 718. [Google Scholar]
- Yokawa, K.; Kagenishi, T.; Kawano, T.; Mancuso, S.; Baluška, F. Illumination of Arabidopsis roots induces immediate burst of ROS production. Plant Signal. Behav. 2011, 6, 1460–1464. [Google Scholar] [CrossRef]
- Yu, G.; Liang, J.; He, Z.; Sun, M. Quantum Dot-Mediated Detection of γ-Aminobutyric Acid Binding Sites on the Surface of Living Pollen Protoplasts in Tobacco. Chem. Biol. 2006, 13, 723–731. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).