Submitted:
06 August 2023
Posted:
08 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and methods
2.1. Chemicals
2.2. Soil Collection
2.3. Photodegradation experiment
2.4. Leaching experiment
| Category | Pesticide | CAS number | Molecular formula |
Molecular weight |
Water solubility* (mg/L) |
Vapor pressure# (Pa) |
Henry constant* (Pa·m3/mol) |
logKow* (pH=7) |
GUS | logKoa | Koc (mL/g) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Insecticide | Nitenpyram | 150824-47-8 | C₁₁H₁₅ClN₄O₂ | 270.72 | 5.90E+05 | 1.10E-06 | 3.54E-13 | -0.66 | 2.01 | 7.83 | 60 |
| Acetamiprid | 135410-20-7 | C₁₀H₁₁ClN₄ | 222.67 | 2.95E+03 | 1.73E-07 | 5.30E-08 | 0.8 | 0.94 | 6.35 | 343 | |
| Chlorantraniliprole | 500008-45-7 | C₁₈H₁₄BrCl₂N₅O₂ | 483.15 | 8.80E-01 | 6.30E-12 | 3.20E-09 | 2.86 | 3.51 | - | 330 | |
| Emamectin benzoate | 155569-91-8 | C56H81NO15 | 1008.24 | 2.40E+01 | 4.00E-10 | - | - | - | - | 283 | |
| Buprofezin | 69327-76-0 | C₁₆H₂₃N₃OS | 305.44 | 4.60E-01 | 4.20E-05 | 2.80E-02 | 4.93 | 0.45 | 8.7 | - | |
| Avermectin B1A | 65195-55-3 | C48H72O14 | 873.08 | 1.00E-02 | 2.00E-07 | - | 4.48 | - | 29.75 | 5638 | |
| Fungicide | Carbendazim | 10605-21-7 | C₉H₉N₃O₂ | 191.19 | 8.00E+00 | 9.00E-05 | 3.60E-03 | 1.48 | 2.21 | 10.54 | 350 |
| Oxadixyl | 77732-09-3 | C14H18N2O4 | 278.30 | 5.96E+02 | 2.70E-05 | 5.86E-07 | 1.4 | 3.89 | 10.3 | 169 | |
| Metalaxyl | 57837-19-1 | C₁₅H₂₁NO₄ | 279.33 | 8.40E+03 | 7.50E-04 | 1.60E-05 | 1.75 | 2.06 | 8.67 | 163 | |
| Diethofencarb | 87130-20-9 | C₁₄H₂₁NO₄ | 267.32 | 2.76E+01 | 9.94E-06 | 9.12E-05 | 2.89 | 1.09 | 7.36 | 271 | |
| Boscalid | 188425-85-6 | C₁₈H₁₂Cl₂N₂O | 343.21 | 4.60E+00 | 7.20E-07 | 5.18E-05 | 2.96 | 2.68 | 12.72 | 772 | |
| Tebuconazole | 107534-96-3 | C₁₆H₂₂ClN₃O | 307.82 | 3.60E+01 | 1.30E-06 | 1.00E-05 | 3.7 | 1.86 | 11.93 | 1000 | |
| Isoprothiolane | 50512-35-1 | C₁₂H₁₈O₄S₂ | 290.40 | 5.40E+01 | 1.88E-02 | 1.00E-01 | 3.3 | - | 8.69 | 1352 | |
| Hexaconazole | 79983-71-4 | C₁₄H₁₇Cl₂N₃O | 314.21 | 1.80E+01 | 1.80E-05 | 3.33E-04 | 3.9 | 2.31 | 10.77 | 1040 | |
| Difenoconazole | 119446-68-3 | C₁₉H₁₇Cl₂N₃O₃ | 406.26 | 1.50E+01 | 3.33E-08 | 9.00E-07 | 4.36 | 0.83 | 13.88 | 6120 | |
| Pyraclostrobin | 175013-18-0 | C₁₉H₁₈CIN₃O₄ | 387.82 | 1.90E+00 | 2.60E-08 | 5.31E-06 | 3.99 | 0.05 | 17.32 | 9300 | |
| Trifloxystrobin | 141517-21-7 | C₂₀H₁₉F₃N₂O₄ | 408.37 | 6.10E-01 | 3.40E-06 | 2.30E-03 | 4.5 | 0.15 | 9.86 | 2377 |
2.5. Determination of target pesticides
2.6. Quality assurance and quality control
2.7. Statistical analysis
3. Results and discussion
3.1. Degradation kinetics in soil and half-lives
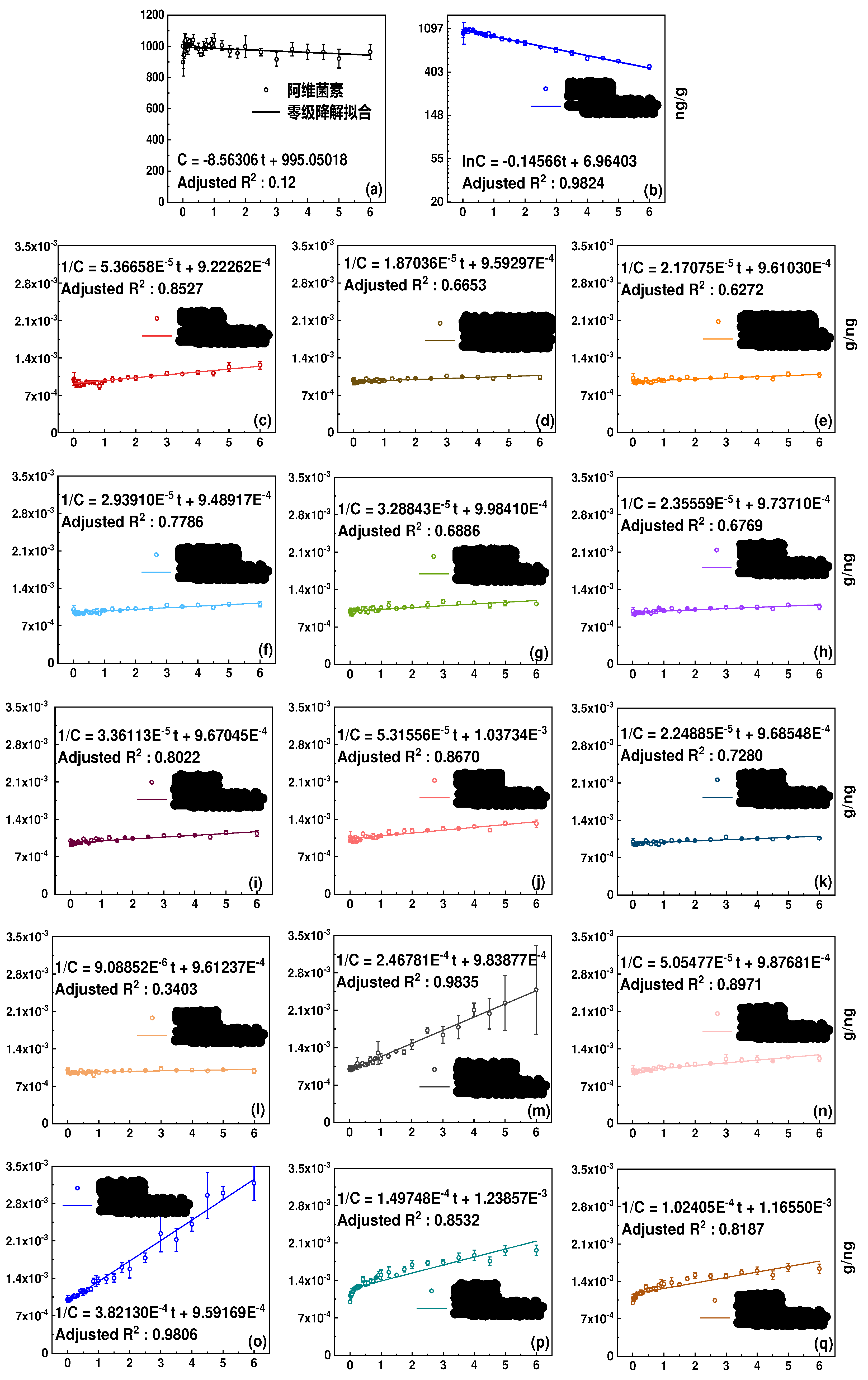
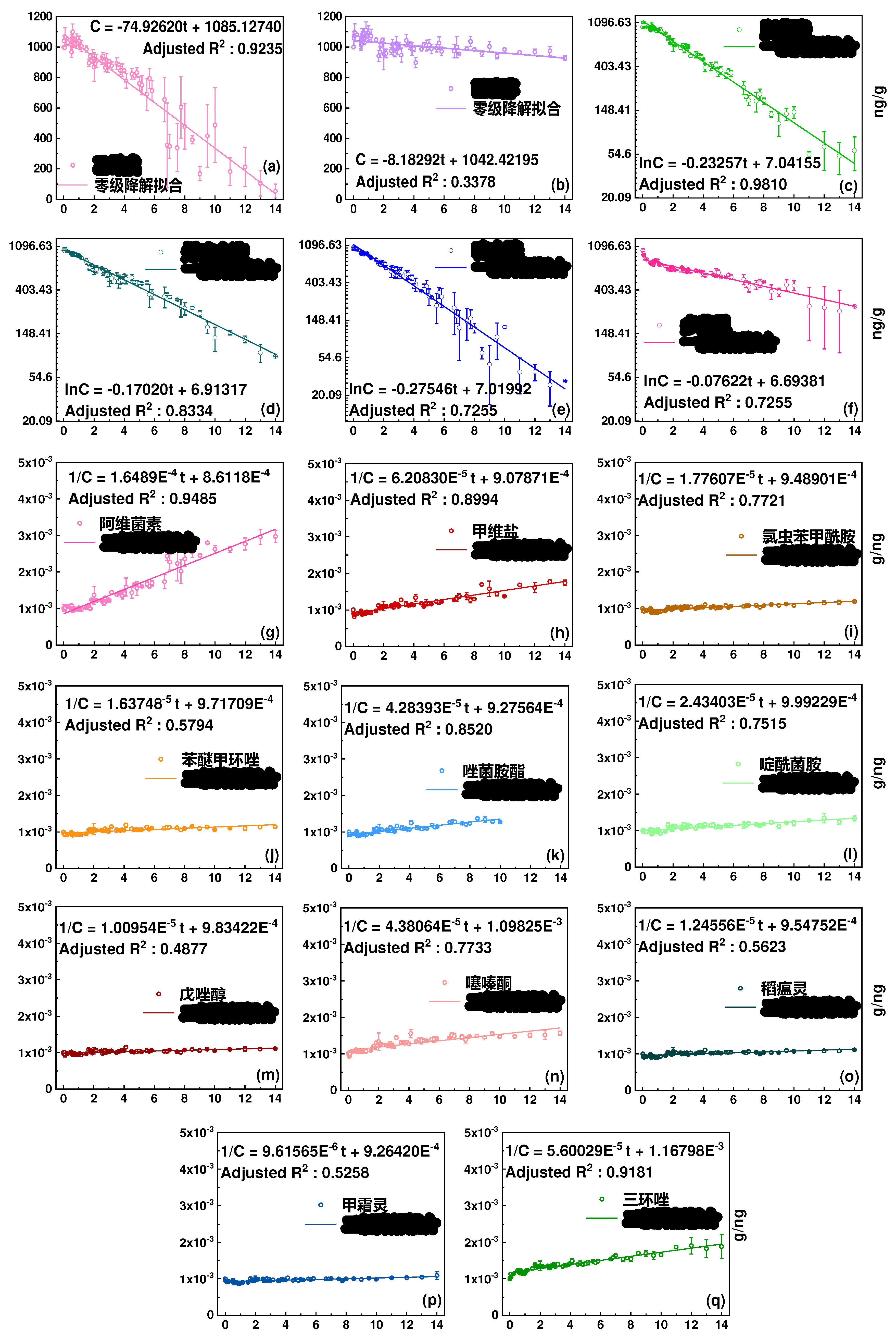
| Category | Pesticide | Dark | Light | ||||
|---|---|---|---|---|---|---|---|
| Dynamic level | Half-life (day) | Residual rate after 6 days |
Dynamic level | Half-life (day) | Residual rate after 14 days |
||
| Insecticide | Nitenpyram | Second-order | 4.1 | 40.3% | First-order | 4.1 | 8.8% |
| Acetamiprid | Second-order | 2.6 | 31.5% | First-order | 2.5 | 2.9% | |
| Chlorantraniliprole | Second-order | 53.5 | 96.3% | Second-order | 56.3 | 84.1% | |
| Emamectin benzoate | Second-order | 18.6 | 79.1% | Second-order | 16.1 | 57.6% | |
| Buprofezin | Second-order | 18.8 | 75.8% | Second-order | 22.8 | 63.7% | |
| Avermectin B1A | Zero-order | 58.4 | 96.4% | Second-order | 6.1 | 33.6% | |
| Fungicide | Carbendazim | Second-order | 6.7 | 50.9% | First-order | 9.1 | 30.9% |
| Oxadixyl | Second-order | 9.8 | 61.1% | Second-order | 17.9 | 53.1% | |
| Metalaxyl | Second-order | 110 | 101.4% | Second-order | 104 | 91.7% | |
| Diethofencarb | Second-order | 19.8 | 82.0% | Zero-order | 6.7 | 5.5% | |
| Boscalid | Second-order | 30.4 | 88.4% | Second-order | 41.1 | 74.9% | |
| Tebuconazole | Second-order | 29.8 | 88.1% | Second-order | 99.1 | 90.1% | |
| Isoprothiolane | Second-order | 44.4 | 93.8% | Second-order | 80.3 | 90.0% | |
| Hexaconazole | Second-order | 42.4 | 92.9% | Zero-order | 61.1 | 92.7% | |
| Difenoconazole | Second-order | 46.1 | 92.3% | Second-order | 61.1 | 87.6% | |
| Pyraclostrobin | Second-order | 34.0 | 90.8% | Second-order | 23.3 | 78.5% | |
| Trifloxystrobin | First-order | 4.8 | 45.7% | First-order | 3.0 | 5.9% | |
3.2. Leaching potential of pesticides in soil
| Solvent | Volume | Nitenpyram | Acetamiprid | Chlorantraniliprole | Emamectin benzoate |
Buprofezin | Avermectin B1A | Carbendazim | Oxadixyl | Metalaxyl |
|---|---|---|---|---|---|---|---|---|---|---|
| Water solution |
1 | 67±0.2 | 74.4±1 | 53.1±0.3 | 0±0 | 0±0 | 0±0 | 53.6±2.8 | 41.5±1.2 | 64±1.5 |
| 2 | 28.4±1.9 | 33.5±2.1 | 25.2±4.2 | 0±0 | 0±0 | 0±0 | 23.7±2.5 | 22.2±2.8 | 30.8±1.4 | |
| 3 | 5.1±2.6 | 8.5±3.4 | 7.9±3.2 | 0±0 | 0±0 | 0±0 | 8.2±2.7 | 11.6±1.1 | 6.9±3.8 | |
| 4 | 1.8±1.1 | 4±2.5 | 3.9±2.1 | 0±0 | 0±0 | 0±0 | 4.5±2.5 | 8±2.5 | 2.7±1.9 | |
| 5 | 0.6±0.3 | 1.7±0.8 | 1.9±0.9 | 0±0 | 0±0 | 0±0 | 2.3±1 | 5.2±1.4 | 1±0.6 | |
| 6 | 0.3±0.1 | 0.9±0.3 | 1.2±0.4 | 0±0 | 0±0 | 0±0 | 1.4±0.5 | 3.8±0.7 | 0.5±0.2 | |
| 7 | 0.2±0.1 | 0.6±0.2 | 0.8±0.3 | 0±0 | 0±0 | 0±0 | 1±0.3 | 3±0.4 | 0.3±0.1 | |
| Methanol | 8 | 0.1±0 | 0.5±0 | 1.1±0.3 | 3.3±0.7 | 24.3±9.8 | 6.6±3.3 | 0.8±0.2 | 5.5±2.4 | 0.2±0 |
| Acetonitrile | 9 | 0.1±0 | 0.8±0.2 | 1.3±0.4 | 2.5±0.5 | 17.7±6.4 | 4.4±2.1 | 1.1±0.4 | 6.6±2.7 | 0.2±0.1 |
| Isopropanol | 10 | 0±0 | 0.8±0.2 | 1.4±0.5 | 2±0.8 | 16.3±2.6 | 3.7±0.1 | 1.2±0.4 | 7.5±1.5 | 0.2±0 |
| 11 | 0±0 | 0.3±0.2 | 0.5±0.5 | 0.4±0.4 | 6.5±5.6 | 1.2±1 | 0.4±0.3 | 2.7±2.3 | 0.1±0.1 | |
| 12 | 0±0 | 0.2±0.2 | 0.3±0.3 | 0.1±0.1 | 3±3.3 | 0.6±0.7 | 0.2±0.2 | 1.5±1.7 | 0.1±0.1 | |
| 13 | 0±0 | 0.1±0.1 | 0.2±0.2 | 0±0 | 1.7±1.8 | 0.3±0.3 | 0.1±0.1 | 0.7±0.8 | 0±0 | |
| 14 | 0±0 | 0.1±0.1 | 0.1±0.2 | 0±0 | 1.6±1.9 | 0.4±0.4 | 0.1±0.1 | 0.6±0.7 | 0±0 | |
| Subtotal | Water solution | 103.4±2.1 | 123.8±6.2 | 94±2.4 | 0±0 | 0.1±0 | 0±0 | 94.5±1.6 | 95.3±2.1 | 106.1±6.5 |
| Organic solvent | 0.1±0 | 2.6±0.6 | 4.9±1 | 8.4±0.2 | 71.1±1 | 17.2±2.8 | 3.9±0.7 | 25±1.9 | 0.9±0.1 | |
| Total | 103.6±2.1 | 126.4±6.8 | 98.9±3.4 | 8.4±0.2 | 71.2±1 | 17.2±2.8 | 98.4±2.2 | 120.4±4 | 107±6.6 | |
| Water solution/organic solvent |
799.16 | 46.74 | 19.22 | 0 | 0 | 0 | 24.36 | 3.81 | 121 | |
| Solvent | Volume | Diethofencarb | Boscalid | Tebuconazole | Isoprothiolane | Hexaconazole | Difenoconazole | Pyraclostrobin | Trifloxystrobin |
|---|---|---|---|---|---|---|---|---|---|
| Water solution | 1 | 48.6±1 | 24.4±0.3 | 12.6±0.4 | 29.9±0.6 | 9±0.8 | 0±0 | 0±0 | 0±0 |
| 2 | 28.4±3.8 | 16.2±4.9 | 10.4±3.5 | 24.9±6.3 | 8.4±2.9 | 0±0 | 0±0 | 0±0 | |
| 3 | 10.3±4.3 | 9.3±1.5 | 7.5±2.2 | 14.5±3.7 | 7.7±1.4 | 0±0.1 | 0±0 | 0±0 | |
| 4 | 5±2.7 | 5.5±1.3 | 4.4±1 | 8.3±3.2 | 4.5±0.4 | 0±0 | 0±0 | 0±0 | |
| 5 | 2.3±1.1 | 3.7±0.7 | 3±0.6 | 5.1±1.9 | 3.5±0.2 | 0±0 | 0±0 | 0±0 | |
| 6 | 1.4±0.5 | 2.7±0.6 | 2.3±0.4 | 3.5±1.2 | 2.8±0.2 | 0±0 | 0±0 | 0±0 | |
| 7 | 1±0.4 | 2.2±0.4 | 1.7±0.4 | 2.5±0.9 | 2.3±0.4 | 0±0 | 0±0 | 0±0 | |
| Methanol | 8 | 1.2±0.3 | 6±2.6 | 4.9±1.4 | 4±0.6 | 7.8±2.7 | 16.7±7.8 | 15.4±7.8 | 11.3±4.3 |
| Acetonitrile | 9 | 1.4±0.3 | 6.1±2.2 | 4.9±1 | 4.2±0.4 | 7±1.4 | 13.8±5.2 | 12.5±5.6 | 8.1±2.1 |
| Isopropanol | 10 | 1.3±0.6 | 6.1±2.1 | 4.8±2.2 | 3.9±1.9 | 6.8±2.9 | 13.6±1.9 | 11.5±0.3 | 7.3±2 |
| 11 | 0.5±0.5 | 2.4±2.4 | 2.1±2.1 | 1.6±1.6 | 2.9±2.8 | 5.6±5.1 | 5.2±4.9 | 2.8±2.7 | |
| 12 | 0.3±0.3 | 1.4±1.6 | 1.2±1.4 | 0.9±1.2 | 1.5±1.8 | 2.7±3.1 | 2.8±3.3 | 1.5±1.7 | |
| 13 | 0.2±0.2 | 0.7±0.8 | 0.6±0.7 | 0.4±0.6 | 0.8±0.9 | 1.5±1.7 | 1.5±1.8 | 0.8±0.9 | |
| 14 | 0.1±0.1 | 0.6±0.7 | 0.5±0.7 | 0.4±0.5 | 0.7±0.9 | 1.4±1.8 | 1.5±2 | 0.8±1 | |
| Subtotal | Water solution | 97.1±4.2 | 64±0.8 | 41.9±1.4 | 88.7±3.9 | 38.1±0.5 | 0.1±0.1 | 0±0 | 0±0 |
| Organic solvent | 4.9±1 | 23.3±2.8 | 19±4.7 | 15.5±4.8 | 27.5±5.2 | 55.3±0.8 | 50.3±0.9 | 32.6±1.8 | |
| Total | 102±5.2 | 87.2±1.9 | 60.9±6.1 | 104.2±8.7 | 65.7±5.7 | 55.4±0.8 | 50.3±0.9 | 32.6±1.8 | |
| Water solution/organic solvent | 19.71 | 2.75 | 2.21 | 5.72 | 1.38 | 0 | 0 | 0 | |
3.3. Environmental behaviors of pesticides
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- CCPIA, Chinese Pesticide Industry Yearbook 2017. 2017, Beijing: ChinaCrop Protection Industry Association.
- Singh, S., et al., Toxicity, monitoring and biodegradation of the fungicide carbendazim. Environ. Chem. Lett., 2016. 14(3): p. 317-329. [CrossRef]
- Wang, Y., et al., Accumulation and toxicity of thiamethoxam and its metabolite clothianidin to the gonads of Eremias argus. Sci. Total Environ., 2019. 667: p. 586-593. [CrossRef]
- Song, Y., et al., Long-term plastic greenhouse cultivation changes soil microbial community structures: A case study. J. Agric. Food Chem., 2018. 66(34): p. 8941-8948. [CrossRef]
- Hu, W., et al., Soil environmental quality in greenhouse vegetable production systems in eastern China: Current status and management strategies. Chemosphere, 2017. 170: p. 183-195. [CrossRef]
- Li, Y., et al., Adsorption-desorption and degradation of insecticides clothianidin and thiamethoxam in agricultural soils. Chemosphere, 2018. 207: p. 708-714. [CrossRef]
- Silva, V., et al., Pesticide residues in European agricultural soils - A hidden reality unfolded. Sci. Total Environ., 2019. 653: p. 1532-1545. [CrossRef]
- Hvezdova, M., et al., Currently and recently used pesticides in Central European arable soils. Sci. Total Environ., 2018. 613-614: p. 361-370. [CrossRef]
- Tao, Y., et al., Occurrence and dietary risk assessment of 37 pesticides in wheat fields in the suburbs of Beijing, China. Food Chem., 2021. 350: p. 129245. [CrossRef]
- Rani, S. and D. Sud, Degradation of dimethoate pesticide in soil: Impact of soil moisture and enhanced sunlight intensity. Water. Air. Soil Pollut., 2022. 233: p. 24. [CrossRef]
- Wu, C., et al., Dissipation and enantioselective degradation of plant growth retardants paclobutrazol and uniconazole in open field, greenhouse, and laboratory soils. Environ. Sci. Technol., 2013. 47(2): p. 843-849. [CrossRef]
- Konstantinou, I., A. Zarkadis, and T. Albanis, Photodegradation of selected herbicides in various natural waters and soils under environmental conditions. J. Environ. Qual., 2001. 30(1): p. 121–130. [CrossRef]
- Rani, S. and D. Sud, Role of enhanced solar radiation for degradation of triazophos pesticide in soil matrix. Sol. Energy., 2015. 120: p. 494-504. [CrossRef]
- Houbraken, M., et al., Multi-residue determination and ecological risk assessment of pesticides in the lakes of Rwanda. Sci. Total Environ., 2017. 576: p. 888-894. [CrossRef]
- Menon, M., R. Mohanraj, and W. Sujata, Monitoring of neonicotinoid pesticides in water-soil systems along the agro-landscapes of the Cauvery delta region, South India. Bull. Environ. Contam. Toxicol., 2021. 106(6): p. 1065-1070. [CrossRef]
- Sultana, T., et al., Neonicotinoid pesticides in drinking water in agricultural regions of southern Ontario, Canada. Chemosphere, 2018. 202: p. 506-513. [CrossRef]
- Xi, N., et al., Elevated temperatures decrease the photodegradation rate of pyrethroid insecticides on spinach leaves: Implications for the effect of climate warming. Environ. Sci. Technol., 2021. 55(2): p. 1167-1177. [CrossRef]
- Wang, C., et al., Field dissipation of trifloxystrobin and its metabolite trifloxystrobin acid in soil and apples. Environ. Monit. Assess., 2015. 187(1): p. 4100. [CrossRef]
- Banerjee, K., A. Ligon, and M. Spiteller, Environmental fate of trifloxystrobin in soils of different geographical origins and photolytic degradation in water. J. Agric. Food Chem., 2006. 54: p. 9479-9487. [CrossRef]
- Shang, Q., et al., Pesticide-conjugated polyacrylate nanoparticles: novel opportunities for improving the photostability of emamectin benzoate. Polym. Adv. Technol. , 2012. 24: p. 137-143. [CrossRef]
- Gao, Y., et al., Dual stimuli-responsive fungicide carrier based on hollow mesoporous silica/hydroxypropyl cellulose hybrid nanoparticles. J Hazard Mater, 2021. 414: p. 125513. [CrossRef]
- Zeng, L., et al., Evaluation of photolysis and hydrolysis of pyraclostrobin in aqueous solutions and its degradation products in paddy water. J. Environ. Sci. Health, Part B, 2019. 54: p. 317-325. [CrossRef]
- Bermúdez-Couso, A., et al., Influence of different abiotic and biotic factors on the metalaxyl and carbofuran dissipation. Chemosphere, 2013. 90(10): p. 2526-2533. [CrossRef]
- Li, Y., Residue dynamics of typical pesticides in the water-soil-crop system of paddy field and their effects on soil microorganisms, in College of Urban and Environmental Sciences. 2020, Peking University: Beijing. p. 253.
- Radolinski, J., et al., Transport of a neonicotinoid pesticide, thiamethoxam, from artificial seed coatings. Sci. Total Environ., 2018. 618: p. 561-568. [CrossRef]
- Kookana, R., H. Di, and L. Aylmore, A field study of leaching and degradation of nine pesticides in a sandy soil Aust. J. Soil Res., 1995. 33: p. 1019-1030. [CrossRef]
- de Oliveira Ferreira, F., R. Porto, and S. Rath, Aerobic dissipation of avermectins and moxidectin in subtropical soils and dissipation of abamectin in a field study. Ecotoxicol. Environ. Saf., 2019. 183: p. 109489. [CrossRef]
- Takai, K., et al., Development of a water-soluble preparation of emamectin benzoate and its preventative effect against the wilting of pot-grown pine trees inoculated with the pine wood nematode, Bursaphelenchus xylophilus. Pest Manag. Sci., 2001. 57(5): p. 463-466. [CrossRef]
- Zhang, S., et al., Preparation and physicochemical characteristics of polylactide microspheres of emamectin benzoate by modified solvent evaporation/extraction method. J. Agric. Food Chem., 2013. 61(50): p. 12219-12225. [CrossRef]
- Kaur, R., et al., Pesticide residues degradation strategies in soil and water: a review. Int. J. Environ. Sci. Technol., 2021. 20(3): p. 3537-3560. [CrossRef]
- Fan, L., et al., Photolysis and photo-induced toxicity of pyraclostrobin to Vibrio fischeri: Pathway and toxic mechanism. Aquat. Toxicol., 2020. 220: p. 105417. [CrossRef]
- Fenoll, J., et al., Rate of loss of insecticides during soil solarization and soil biosolarization. J. Hazard. Mater., 2011. 185(2-3): p. 634-638. [CrossRef]
- Dionisio, A. and S. Rath, Abamectin in soils: Analytical methods, kinetics, sorption and dissipation. Chemosphere, 2016. 151: p. 17-29. [CrossRef]
- Liu, J., et al., Physiochemical assessment of environmental behaviors of herbicide atrazine in soils associated with its degradation and bioavailability to weeds. Chemosphere, 2021. 262: p. 127830. [CrossRef]
- Swartjes, F. and M. Van der Aa, Measures to reduce pesticides leaching into groundwater-based drinking water resources: An appeal to national and local governments, water boards and farmers. Sci. Total Environ., 2020. 699: p. 134186. [CrossRef]
- Li, Z., A health-based regulatory chain framework to evaluate international pesticide groundwater regulations integrating soil and drinking water standards. Environ. Int., 2018. 121: p. 1253-1278. [CrossRef]
- Aliste, M., et al., Mobility of insecticide residues and main intermediates in a clay-loam soil, and impact of leachate components on their photocatalytic degradation. Chemosphere, 2021. 274: p. 129965. [CrossRef]
- Gavrilescu, M., Fate of Pesticides in the Environment and its Bioremediation. Eng. Life Sci., 2005. 5(6): p. 497-526. [CrossRef]
- Liu, W., et al., DFT insights into the degradation mechanism of carbendazim by hydroxyl radicals in aqueous solution. J. Hazard. Mater., 2022. 431: p. 128577. [CrossRef]
- Wang, J., et al., Degradation of carbendazim in aqueous solution by dielectric barrier discharge cold plasma: Identification and toxicity of degradation products. Food Chem., 2023. 403: p. 134329. [CrossRef]
- Jornet, D., et al., Photodegradation of carbendazim sensitized by aromatic ketones. J. Photochem. Photobiol. A Chem., 2013. 256: p. 36-41. [CrossRef]
- Ma, C., et al., Kinetics, mechanisms and toxicity of the degradation of imidaclothiz in soil and water. J. Hazard. Mater., 2021. 403: p. 124033. [CrossRef]
- Chau, N., et al., Pesticide pollution of multiple drinking water sources in the Mekong Delta, Vietnam: evidence from two provinces. Environ. Sci. Pollut. Res. Int., 2015. 22(12): p. 9042-9058. [CrossRef]
- Zambito Marsala, R., et al., First evaluation of pesticides occurrence in groundwater of Tidone Valley, an area with intensive viticulture. Sci. Total Environ., 2020. 736: p. 139730. [CrossRef]
- Manjarres-Lopez, D., et al., Assessment of pesticide residues in waters and soils of a vineyard region and its temporal evolution. Environ. Pollut., 2021. 284: p. 117463. [CrossRef]
- Andrades, M., M. Sa´nchez-Martı´n, and M. Sa´nchez-Camazano, Significance of soil properties in the adsorption and mobility of the fungicide metalaxyl in vineyard soils. J. Agric. Food Chem., 2001. 49: p. 2363-2369. [CrossRef]
- Fisher, I., et al., Pesticides and their degradates in groundwater reflect past use and current management strategies, Long Island, New York, USA. Sci. Total Environ., 2021. 752: p. 141895. [CrossRef]
- Zhang, Y., et al., Occurrence and risk evaluation of organophosphorus pesticides in typical water bodies of Beijing, China. Environ. Sci. Pollut. Res. Int., 2021. 28(2): p. 1454-1463. [CrossRef]
- Wratten, S. and A. Forbes, Environmental assessment of veterinary avermectins in temperate pastoral ecosystems. Ann. appl. Biol., 1996. 128: p. 329-348. [CrossRef]
- Dusek, J., et al., Field leaching of pesticides at five test sites in Hawaii: study description and results. Pest Manag. Sci., 2010. 66(6): p. 596-611. [CrossRef]
- Wang, C., et al., Field dissipation of trifloxystrobin and its metabolite trifloxystrobin acid in soil and apples. Environ. Monit. Assess., 2015. 187: p. 4100. [CrossRef]
- Reddy, S., S. Gupta, and V. Gajbhiye, Adsorption-desorption and leaching of pyraclostrobin in Indian soils. J. Environ. Sci. Health, Part B, 2013. 48(11): p. 948-959. [CrossRef]
- Jiang, W., et al., Esterase TriH responsible for the hydrolysis of trifloxystrobin in Hyphomicrobium sp. B1. Int. Biodeterior. Biodegrad., 2022. 174: p. 105465. [CrossRef]
- Li, C., et al., Biodegradation of buprofezin by Rhodococcus sp. strain YL-1 isolated from rice field soil. J. Agric. Food Chem., 2012. 60(10): p. 2531-2537. [CrossRef]
- Chen, K., et al., Isolation of a buprofezin co-metabolizing strain of Pseudomonas sp. DFS35-4 and identification of the buprofezin transformation pathway. Biodegradation, 2011. 22(6): p. 1135-1142. [CrossRef]
- Chen, X., et al., Biodegradation of pyraclostrobin by two microbial communities from Hawaiian soils and metabolic mechanism. J. Hazard. Mater., 2018. 354: p. 225-230. [CrossRef]
- Wang, L., et al., Residues and dissipation of trifloxystrobin and its metabolite in tomatoes and soil. Environ. Monit. Assess., 2014. 186(11): p. 7793-7799. [CrossRef]
- Halley, B., W. VandenHeuvel, and P. Wislocki, Environmental effects of the usage of avermectins in livestock. Vet. Parasitol., 1993. 48: p. 109-125. [CrossRef]
- Ge, J., et al., Uptake and translocation of imidacloprid, thiamethoxam and difenoconazole in rice plants. Environ. Pollut., 2017. 226: p. 479-485. [CrossRef]
- Chen, S., et al., Deposition distribution, metabolism characteristics, and reduced application dose of difenoconazole in the open field and greenhouse pepper ecosystem. Agr. Ecosyst Environ., 2021. 313: p. 107370. [CrossRef]
- Li, J., et al., Transcriptomic and physiological properties reveal the tolerance mechanism to difenoconazole toxicity in wheat (Triticum aestivum L.). Ecotoxicol. Environ. Saf., 2023. 255: p. 114787. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




