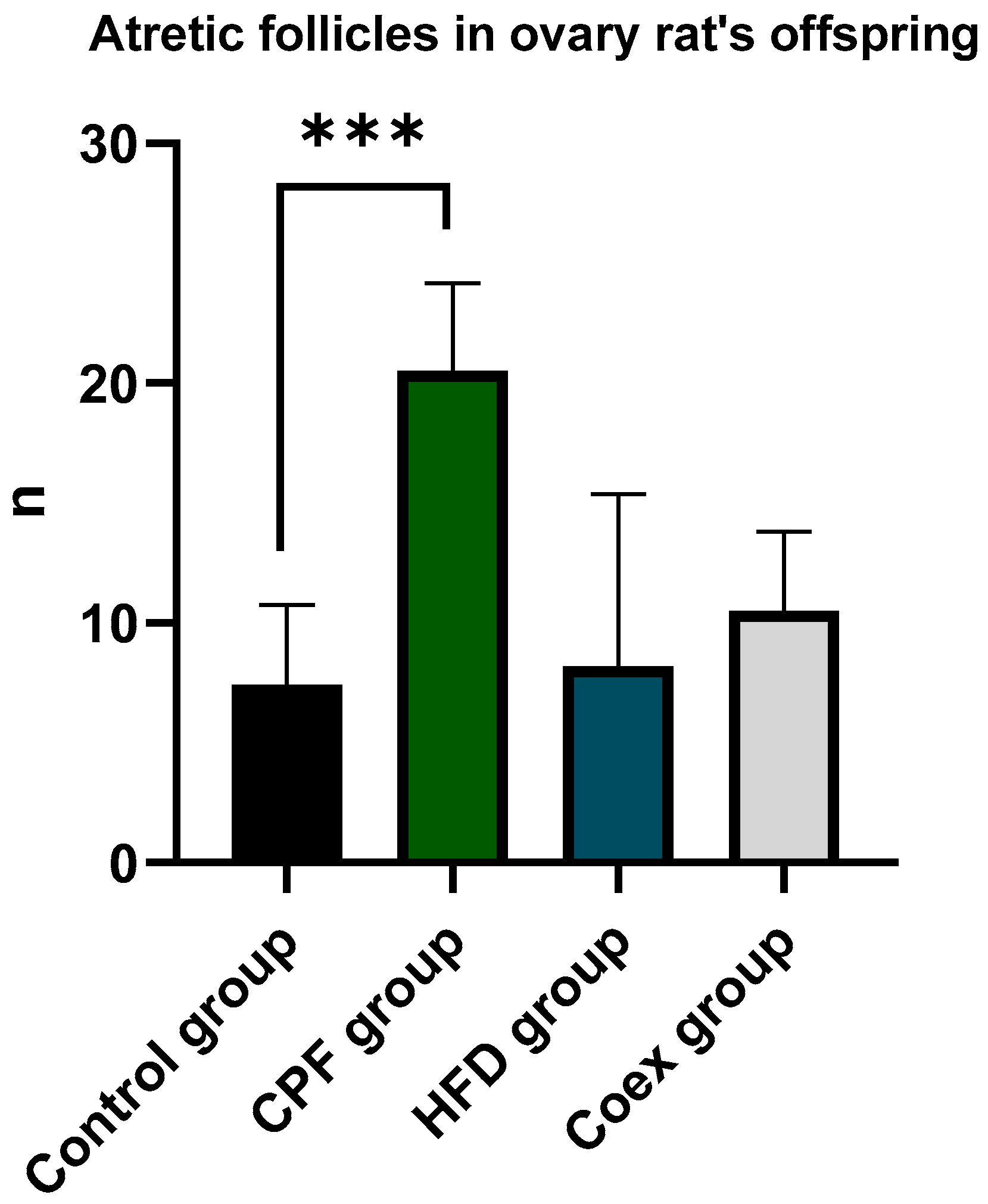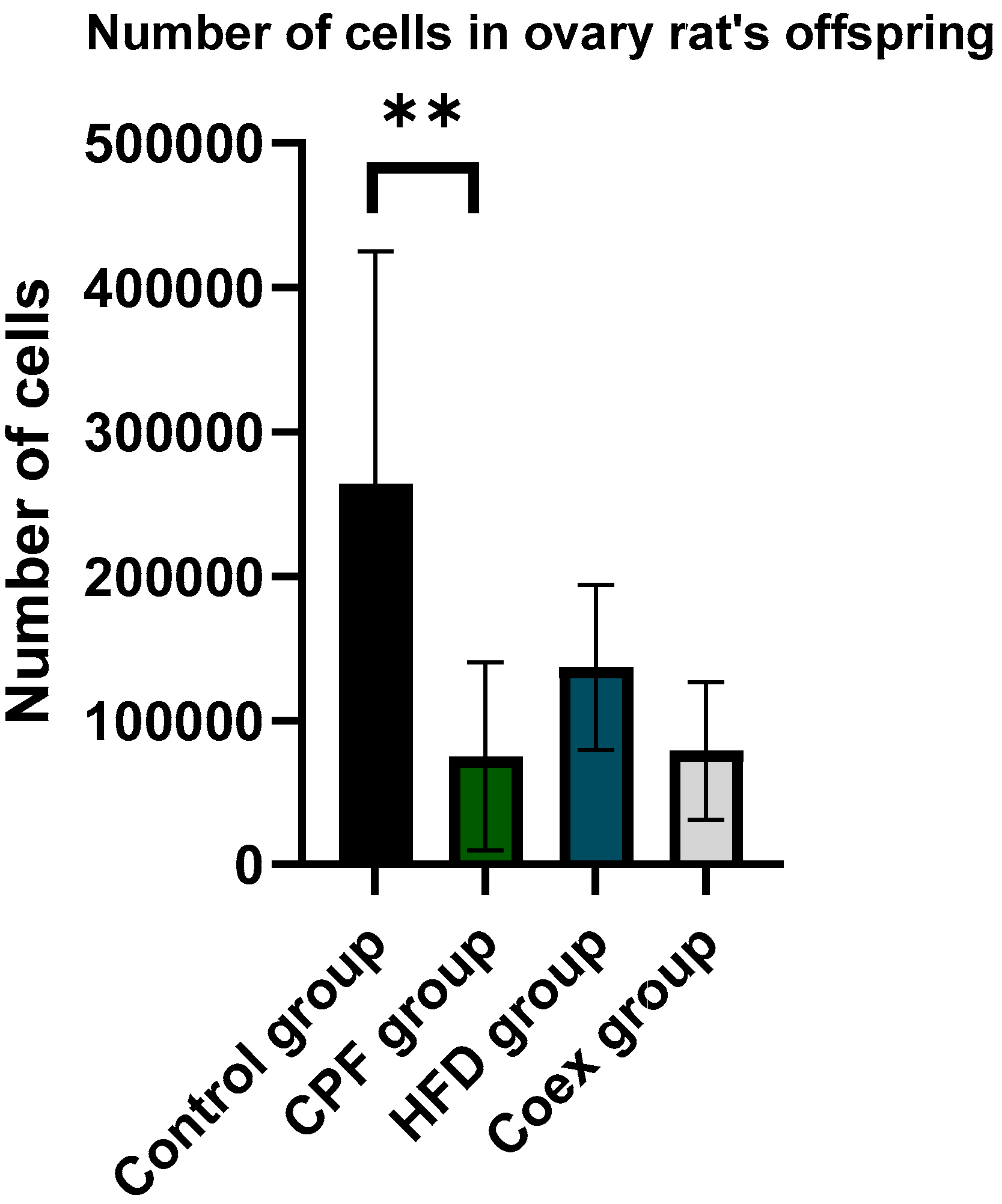1. Introduction
The World Health organization defines the infertility as the inability to achieve a successful pregnancy within 12 months of unprotected intercourse [
1,
2]. It affects approximately 17 to 20% of couples worldwide [
2]. According to a 2015 epidemiological study, the World Health Organization estimates that nearly around 186 million individuals worldwide affected with infertility globally [
3]. Numerous factors can affect fertility including environmental and lifestyle factors such as smoking, excessive alcohol intake and obesity [
4].
Since the last decade, the literature reported that the exposure to pesticides and the issue of obesity have emerged as pressing public health concerns. They are interconnected and have been associated with various adverse effects, including a decline in reproductive health and a detrimental impact on the effectiveness of assisted reproduction technology [
5]. The mechanisms behind this relationship are not yet fully elucidated, but it is believed that pesticide exposure and high fat diet disrupt metabolic processes [
6] and alter the reproductive health [
7]. Our previous studies have shown that pesticides exposure alters some sperm parameters including motility and vitality and increase the sperm DNA fragmentation [
8]. The Anses (L’Agence nationale de sécurité sanitaire de l’alimentation, de l’environnement et du travail) and EPA (United States Environmental Protection Agency) reported that certain pesticides are considered as an endocrine disruptor including, the chlorpyrifos polychlorinated bisphenyls (PCB), bisphenol A (BPA), dibutyl phthalates (DBP) and alkyl phenols, Di-(2-ethylhexyl) phthalate (DEHP), Glyphosate, Phthalates, dichlorodiphenyltrichloroethane DDT and Methoxychlor and chlorpyrifos (CPF) [
9,
10].
Chlorpyrifos is an organophosphate insecticide widely used in agriculture to control pests on crops [
11]. However, studies raised concerns about its potential effects on human health, including reproductive toxicity which includes both male and female reproductive organs as well as the development of embryos and fetuses [
12,
13]. CPF is an insecticide that disrupts the nervous system by inhibiting the activity of an enzyme called acetylcholinesterase (AChE) [
14,
15], which plays a critical role in transmitting nerve signals in target insects, and it can also affect non-target organisms, including humans [
16]. The Developmental Origins of Health and Disease (DOHaD) hypothesis suggests that environmental exposures and experiences during early life, particularly during fetal development and early childhood, can have a profound impact on an individual’s lifelong health and disease risk and can lead to long-term health consequences [
6,
17,
18]. The main aspects of DOHaD include Fetal Programming [
19], Critical Windows of Development [
20], Epigenetic Changes [
21], Long-Term Health Consequences [
22], Interactions with Genetics, Transgenerational Effects [
23]. Guidelines and regulations set by authorities like EFSA (European Food Safety Authority) and EPA (United States Environmental Protection Agency) regarding the use of pesticides like chlorpyrifos and to take necessary precautions to ban exposure, especially for pregnant women and young children [
11,
24].
Emerging evidence indicates that endocrine disruptors alter the synthesis and functions of androgens and estrogens, which are essential sex hormones responsible for the development and maintaining of the male reproductive system. It can disturb the hypothalamic and pituitary (HPG) axis function by altering synthesis of hormones regulating the reproduction including GnRH and kisspeptin [
25].
Kisspeptin, a neuropeptide with 145-aminoacid, encoded by the KISS1 gene. It considered as a central regulator of the hypothalamic-pituitary-gonadal (HPG) axis [
26]. It plays a crucial role in the regulation of reproductive function. It act as a potent stimulator of the release of gonadotropin-releasing hormone (GnRH) in the brain [
27]. GnRH serves as the key mediator, conveying these cues throughout the HPG axis [
28]. Over the past twenty years, significant research has been dedicated studying kisspeptin (KISS1) neurotransmitter, and a growing body of evidence supports their involvement in regulating the secretion of GnRH and gonadotropin hormones throughout the ovulatory cycle [
29].
It’s believed that kisspeptin neurotransmitters were only found in the hypothalamus. However, emerging evidence have revealed interesting findings indicates that kisspeptin-producing cells are also present in several extra-hypothalamic regions of the brain, as well as in peripheral tissues such as the placenta, ovary, uterus, testis [
30,
31], and gastrointestinal tract. It was detected for the first time in rat tissue [
32,
33,
34,
35]. Its implicated in the regulation of puberty onset, fertility, and pregnancy [
36]. Extra hypothalamic Kisspeptin has been found to modulate the secretion of reproductive hormones, influence the development and function of reproductive organs, and participate in the establishment of reproductive behaviors [
37,
38]. Siminara et al. 2003 has reported that mutation in kisspeptin receptor knock out (kiss1r
-/- or GPR54
-/-) in mice lead to an idiopathic hypogonadotropic hypogonadism (small testes in male mice and an absence of follicular maturation in female mice) [
39]. Furthermore, Feng et al. 2021 have reported that the expression of the KISS-1/GPR54 system in the testes of rats is influenced by a high-fat diet, leading to a decrease in the expression level of the KISS-1 protein [
30]. A case-control study published by Abdelkareem et al. 2023, included a total of 30 patients (10 patients with unexplained recurrent pregnancy loss, 10 patients with pregnancy loss due to aneuploidy and 10 patients for control group). The study has investigated the role of KISS1 and KISS1R in early placentation. The outcome revealed a significant decrease in KISS1R expression in the chorionic tissues of (euploid and aneuploid) recurrent pregnancy loss compared with control group [
40].
Several studies reported that GnRHR was founded not only in the hypothalamus but also in various extra-hypothalamic tissues, including ovary, oviduct [
41], testis [
42], mammary gland [
43], placenta and endometrium [
44]. While the specific mechanisms and the exact function of kisspeptin and GnRHR of extra-hypothalamic origin in mammals remains unknown and still under investigation. Additional research is required to better understand the relationship between environmental factors, diet and the implication of Kisspeptin in reproductive function.
The primary aim of this research is to identify the presence of Kisspeptin and GnRHR expression within the reproductive organs of rat offspring using Immunohistochemistry. Additionally, the study aims to evaluate how exposure to chlorpyrifos, and a high-fat diet impacts the expression of these proteins through in silico quantification. Furthermore, the study investigates the morpho-pathological effects on the reproductive organs using histological analysis.
2. Materials and Methods
a) Chlorpyrifos preparation and High Fat Diet
Chlorpyrifos, O, O-diethyl-O-(3,5,6-trichloro-2-pyridinyl) phosphorothioate, was purchased from LGC Standards (Molsheim, France). A quantity of 250 mg of Chlorpyrifos was combined with 25 ml of rapeseed oil (MP Biomedicals, Illkirch, France) as a vehicle, resulting in a concentration of 1 mg/ml. This mixture was then administered to the rats (the CPF group and the co-exposed group) via gavage at a dosage of 1 mg/kg body weight per day [
45]. The HFD group and the co-exposed group were fed a high fat diet (60% Kcal as fat) until PND21.
b) Experimental design and ethical approval
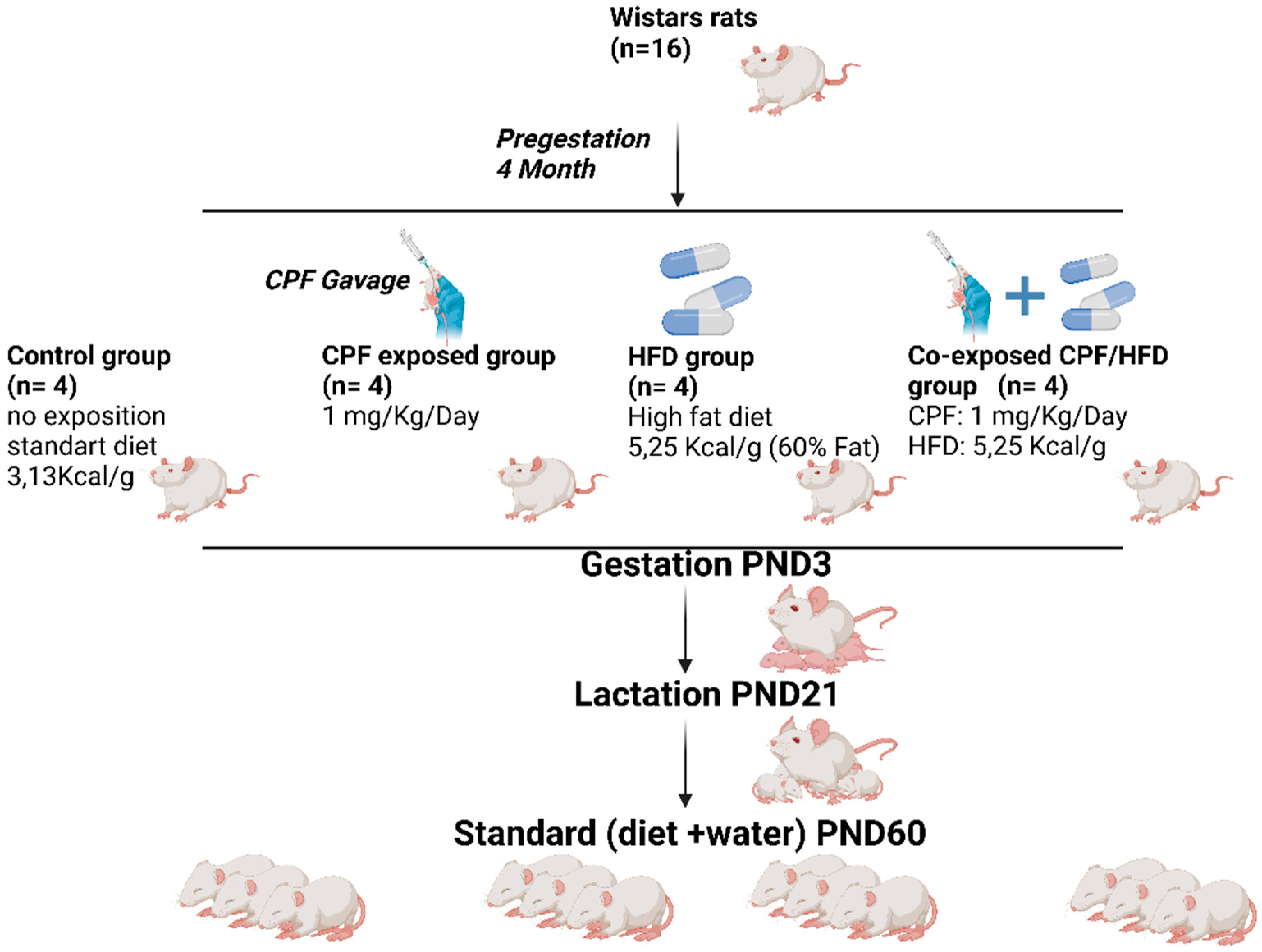
16 female Wistar rats (Janvier Labs, Le Genest Saint Isle, France,), upon arrival, aged 7 weeks, were placed in cages and kept under consistent conditions in a controlled environment. The room was maintained at a temperature of 23°C to ensure a stable and regulated environment for the rats. Then, rats were housed in NexGen Max cage system with 81 in2 /523 cm2 floor area mounted on EcoFlow rack system (Allentown Inc, Bussy Saint Georges, France). Following a week-long acclimation period, the female rats, with an average body weight of 225 ± 4.9 g, were randomly assigned to four groups. To facilitate social interaction, two rats were housed together in each cage. This housing arrangement allowed for appropriate socialization among the animals during the study (
Figure 1).
Figure 1.
Study design and animal’s preparation (personal source).
Figure 1.
Study design and animal’s preparation (personal source).
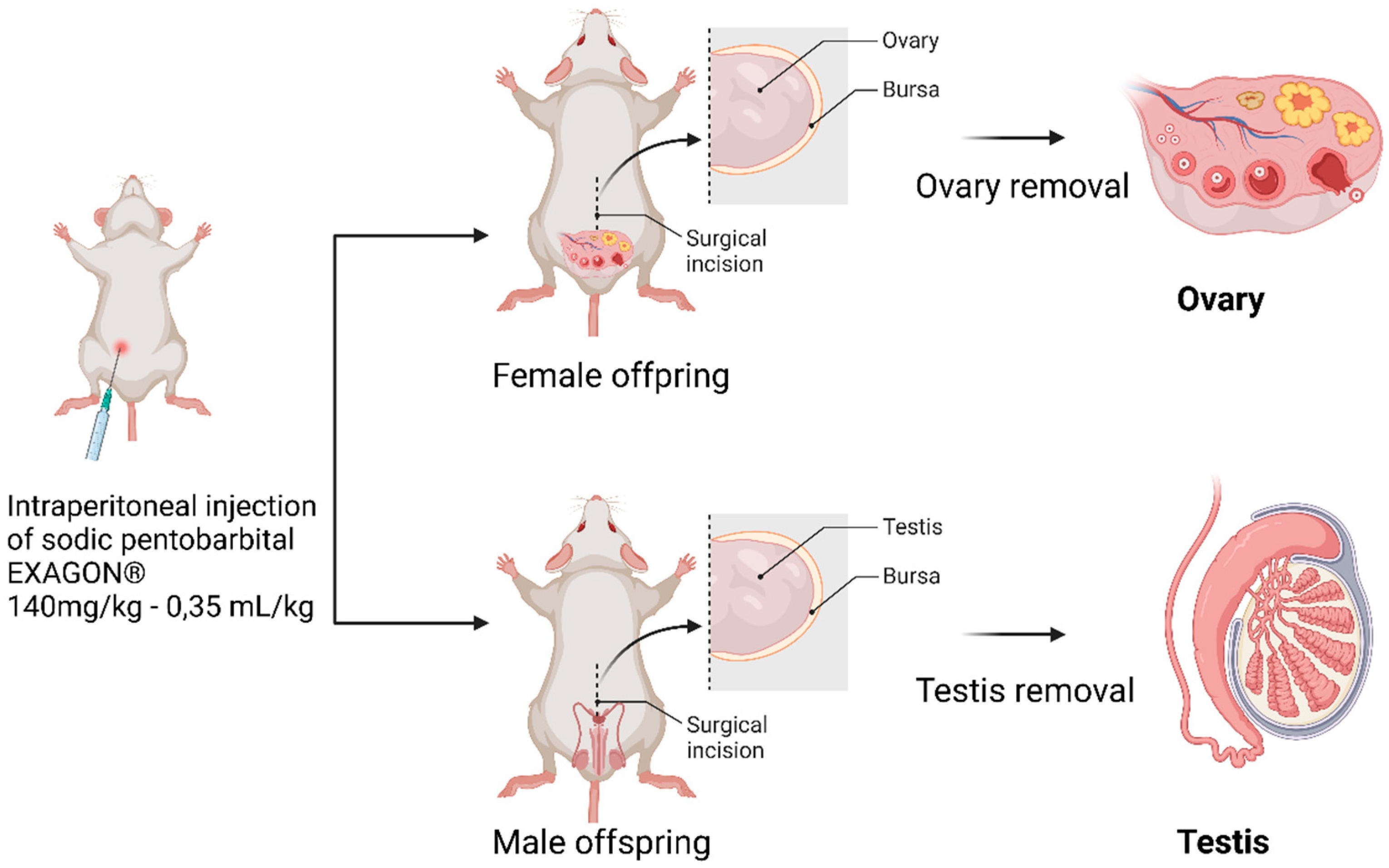
At PND60, the offspring rats were euthanized using an injection of (1 ml.kg-1; 200 mg.ml-1 solution) sodium pentobarbital from EXAGON (Axience, France). After euthanasia, the reproductive organs were dissected and placed directly into Krebs Henseleit solution from Sigma Aldrich (Saint Quentin Fallavier, France). This solution is commonly used to maintain the viability and functionality of isolated tissues during experimental procedures (
Figure 2).
Figure 2.
offspring’s sacrifice and reproductive organ dissection (personal source).
Figure 2.
offspring’s sacrifice and reproductive organ dissection (personal source).
The study protocol obtained approval from the Regional Directorate for Health, Animal and Environment Protection, an accredited organization in France. Additionally, it was also approved by the French Ministry of Research under the reference number APAFIS#8207-2016121322563594 v2. These regulatory approvals ensured that the study adhered to the necessary ethical and scientific standards regarding animal welfare and research conduct.
c) Immunohistochemistry (kisspeptin and GnRHR)
Tissue sections were deparaffinized in xylene (Sigma Aldrich, Germany) and rehydrated through decreasing concentrations of ethanol (TechniSolv, France). After deparaffinization the slides were incubated in Tris EDTA buffer (10mM tris-base (Cat # 161-0719, Biorad, US), 1mM EDTA solution (Cat # 161-0729, Biorad, US), 0.05% tween20® (Sigma-Aldrich, UE), pH=9) 20 min (2*10 min) in a microwave (850W) followed by washing with cold water for 10 min. To quench the endogenous peroxidase activity, the slides were incubated in water peroxide H202 (Gilbert) for 30 min then then washed twice (each 5 min delicately) in TBS (ThermoScientific, Belgium) 0,025 Triton and blocked with 10% normal Goat Serum (ab7481) with 1% BSA (ThermoScientific, USA) in TBS for 2 hours [C] between 0.5 and 10 ug/ml. thereafter, the sections were incubated overnight in a humidity room at 4 °C with primary antibody:
- -
For kisspeptin detection: Primary rabbit polyclonal anti kisspeptin antibody (1:300 dilution; product no.251265; Abbiotec. Escondido, CA, USA)
- -
For GnRHR detection: Primary rabbit polyclonal to GnRHR (1:200 dilution; abcam, ab202848, UK)
The sections were washed in TBS/0,025 Triton 2*(5min) under gentle agitation, then incubated with goat anti-rabbit IgG-HRP (1:1000 dilution; product no.252237; Abbiotec. Escondido, CA, USA) for 1 hour at room temperature. After washing step 3*(5min) in TBS, labeling was visualized by incubating with DAB (3,3’Diaminobenzidine) substrate kit (ab64238, abcam, UK) for 4 min then washed in 5 min in TBS. Sections were counterstained with Mayer’s hematoxylin (VWR Q-Path Chemicals, France) for 2 min and mounted with Aqueous Mounting Medium (ab64230, Abcam, UK).
The slides were observed under a light microscope, specifically the Nikon ECLIPSE Ci model from Nikon Europe B.V. This microscope offers magnifications of X4, X20, and X40, allowing for different levels of details observation. To capture the images, the NIS Elements version 1.10.00 imaging software was used. This software provides a platform for acquiring and processing images obtained from the microscope, enabling researchers to analyze and document their observations.
d) Histological analysis
Histopathological analysis of the testicular and ovary sections (4um-thick) was performed using a hematoxyline-eosine staining. 5 slides for each group and each slide contains 2~3 sections of an organ.
The paraformaldehyde-fixed paraffin-embedded sections were incubated in Xylene 2*(5 min) then rehydrated using different concentrations of ethanol (2 min ethanol 100%, 2 min ethanol 95%, 6min ethanol 70% and 2min osmosed water, respectively). The slides were stained with hematoxylin of Mayer 5% (2min) and after a washing step for2 min in osmosed water. The slides were soaked 5 times in acid/alcohol 37%. After the washing step, an eosin 1% staining was realized for 1~3 min. Finally, the slides were washed and dehydrated (2 min ethanol 95%, 2 min ethanol 100%, 2min in xylene, respectively). Morphological Analysis was performed under a light microscopy (Nikon ECLIPSE Ci, Nikon Europe B.V) with X4, X20 and X40 magnifications. The image was captured using an imaging software NIS Elements version 1.10.00.
The morphometric measurements assess the number of cells per seminiferous tubule (average of 10 selected seminiferous tubules per section); area of seminiferous tubules (average in 10 randomly selected seminiferous tubules per section) [
46].
e) In silico quantification of expression
The C, CPF, HFD, CPF/HFD immunodetected sections were scanned and photographed usingTissue Faxs plus system (Tissue Gnostics Medical & Biotech Solutions, Vienna, Austria. At least 5 randomly selected slides per animal were used for immunostaining and scanned. Regions of interest (ROIs) were manually delineated using the zooming and mark-up tools available in the TissueFAXS viewer. This process allows us to focus on specific regions for further analysis or measurements. The ROIs were then digitized at 20× using HistoQuest® (TissueGnostics GmbH, Austria) and the expression of kisspeptin and GnRHR (the intensity of signals detected onto the sections) were quantified. The typical image was selected for presentation [
47,
48].
f) Statistical analysis
Statistical data was analyzed using StatView software version 5.0. The results were presented as mean values ± standard deviations (SD), number (n) and percentage (%). We perform a Two-way ANOVA to compare the difference between the groups. The null hypothesis of the Two-way ANOVA is that all four variables being compared have the same mean. The alternative hypothesis, on the other hand, suggests that at least one of the variables has a different average. The statistical Figure s were generated by GraphPad 9 software.
3. Results
1- Impacts of treatment on pup’s body weight
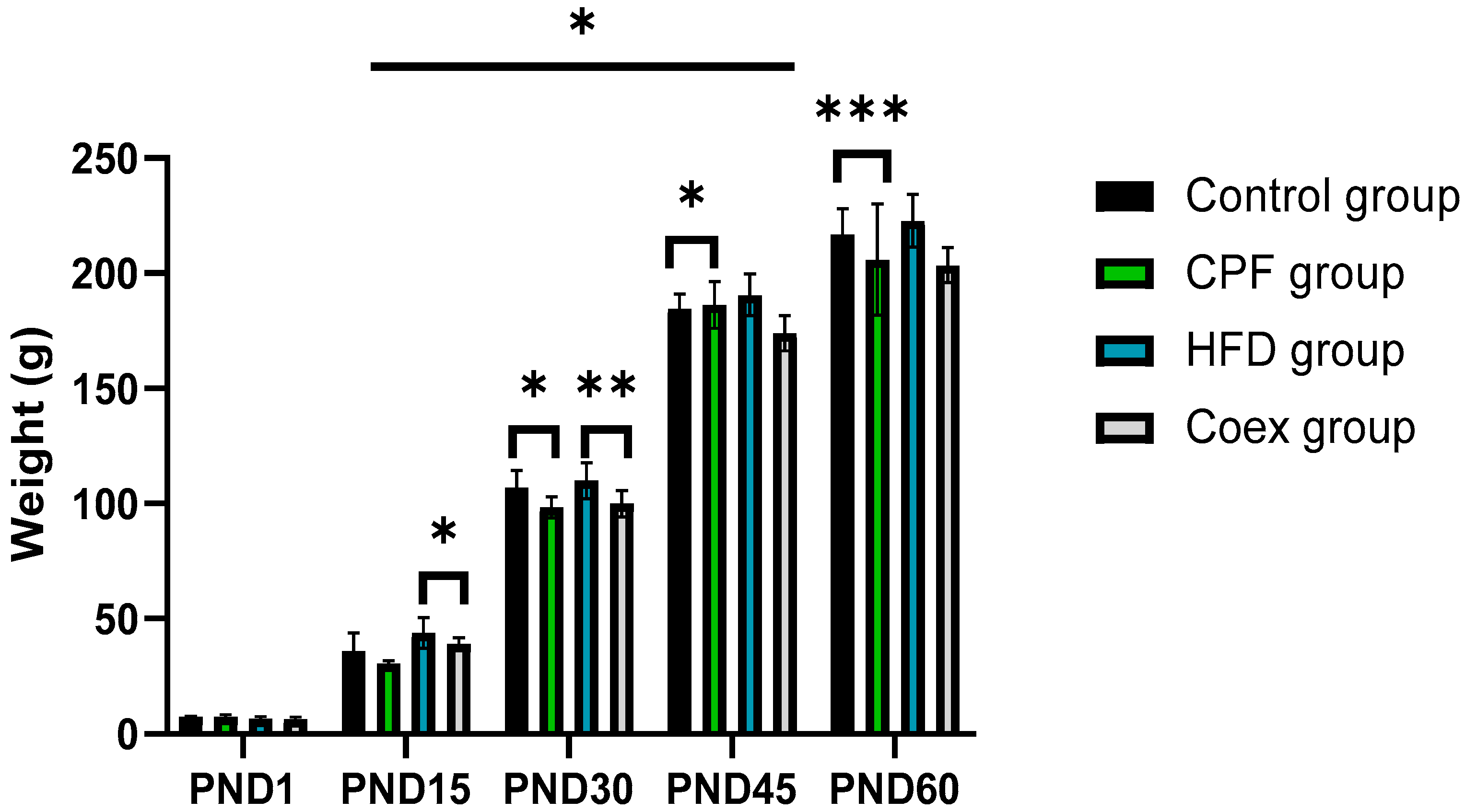
Statistical data analysis shows a significant increase in rat offspring’s body weight in postnatal period P=0.03. The high fat diet group presents the heaviest body weight groups in the different PND period.
The intergroup comparison between the control and the CPF exposed group shows a highly significant decrease in the body weight in the CPF exposed group at the period of (PND30 and PND 60) p<0.05 and p<0.001, respectively).
At PND 15 and PND 30, the body weight of rat offsprings shows a decrease in the co-exposed group to CPF/HFD compared to the HFD group (p<0.05 and p<0.01, respectively)
Figure 3.
No significant differences were observed between the different groups at PND1, between control group and CPF group at PND15 and between HFD group and Coex group at PND 45 and PND 60.
Figure 3.
Evolution of the pup’s body weight (male + female) at the postnatal periods.
Figure 3.
Evolution of the pup’s body weight (male + female) at the postnatal periods.
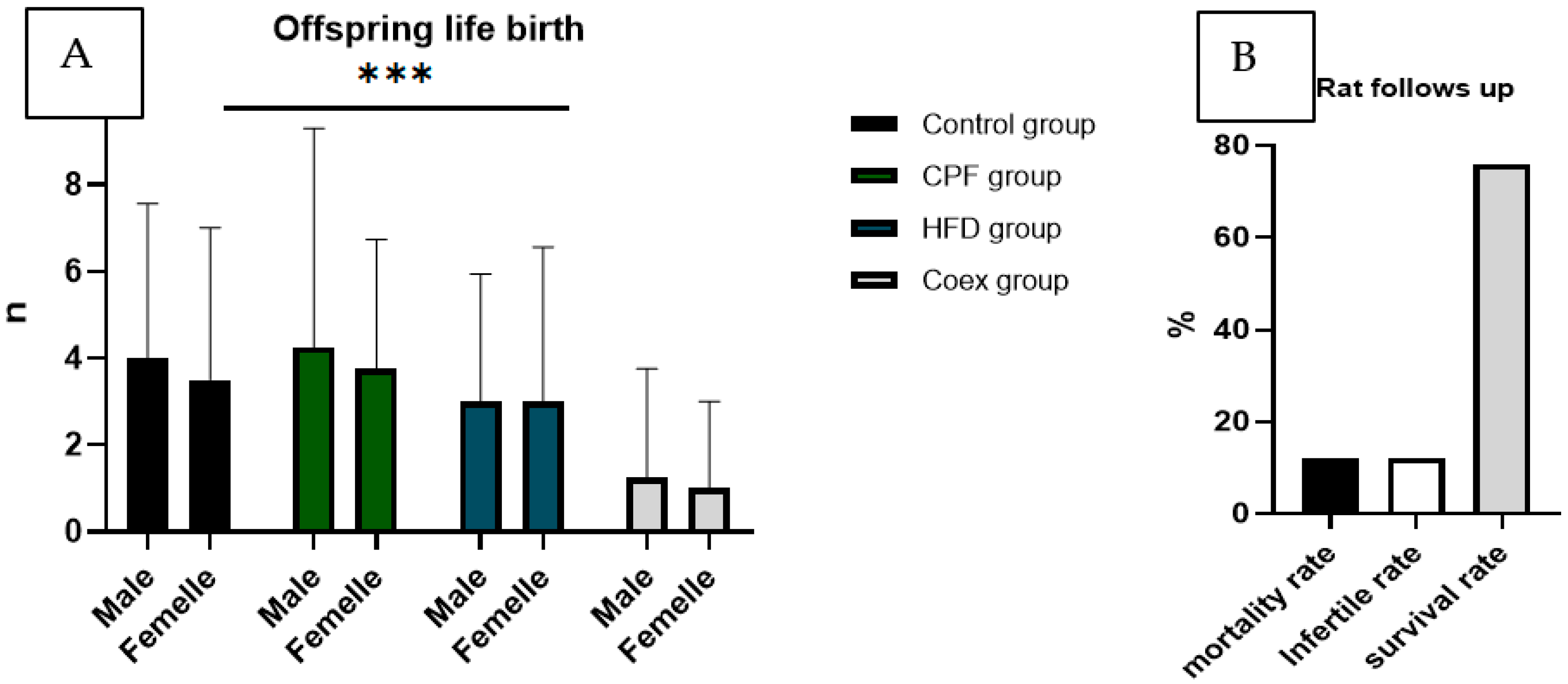
The outcomes of the rat study were showed in
Figure 4, illustrating the count of male and female offspring within each group, alongside the mortality rate of the rats
Figure 4.
Figure 4.
Rat follows up during the treatment (B) and gestational period (A).
Figure 4.
Rat follows up during the treatment (B) and gestational period (A).
2- Histological analysis
a. Ovary of rat offspring
Atresia in secondary and antral follicles was observed by eosin staining. Histological analysis of rat ovary was assessed in control group (n=5), CPF group (n=5), HFD group (n=5) and Coex group (n=5). Statistical data show a significant difference in atretic follicle number between CPF group and control group (p=0,0008)
Figure 5. Follicles with fractured oocytes were observed in some sections of CPF exposed group and Coex group
Figure 6.
Figure 5.
Measurement of atretic follicle number in the ovary of the rat offspring.
Figure 5.
Measurement of atretic follicle number in the ovary of the rat offspring.
Figure 6.
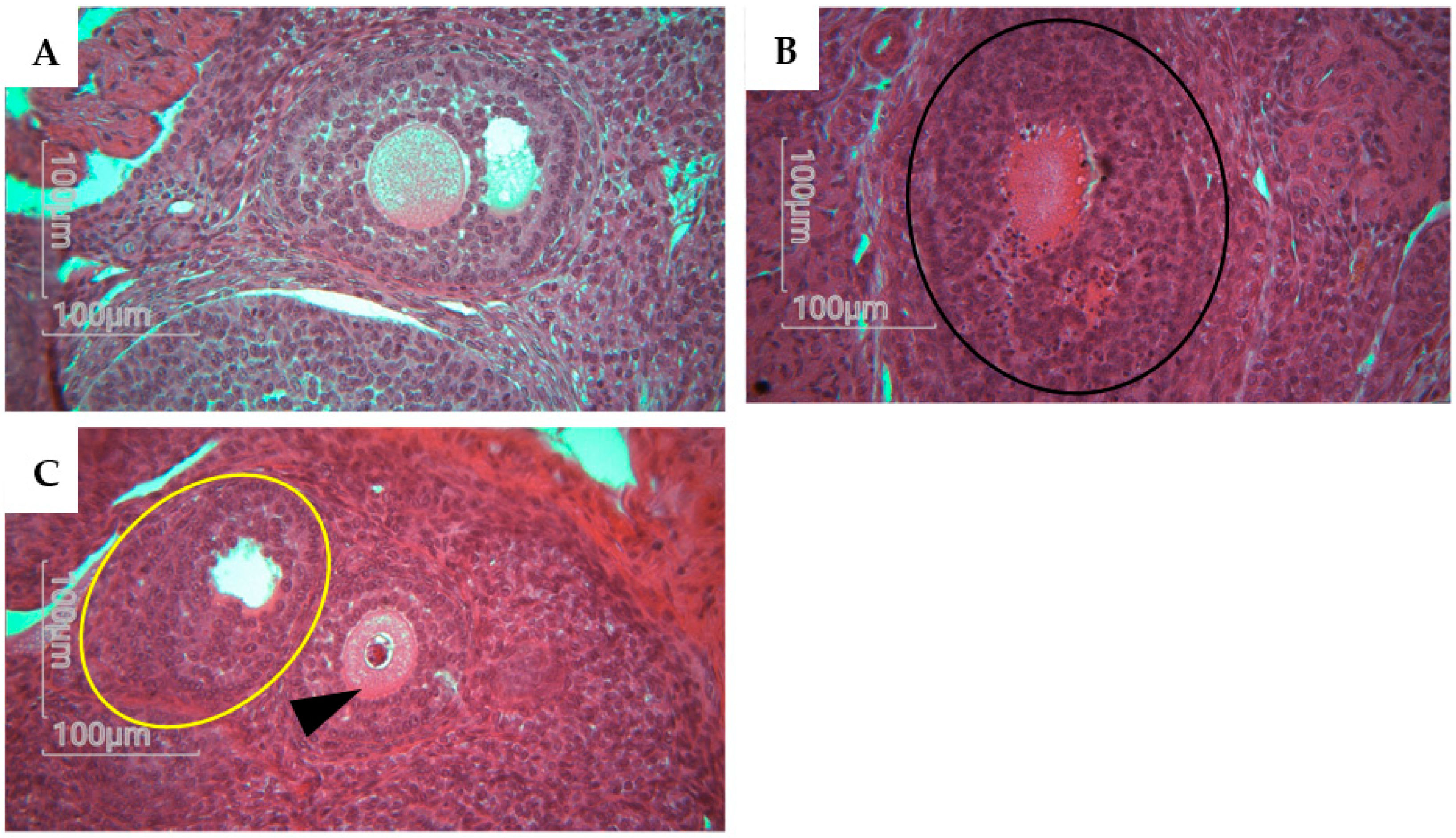
Histological analysis of rat offsping follicules showing alterations Magnification X40. A represents a normal morphology of antral follicle with normal oocytes and normal cumulus cells and theca cells. B represents an atresia in follicle with color change to dark red and absence of oocyte (yellow cercle). C represents a degenerative follicle, with no oocytes (black cercle) and a follicle with fractured oocyte that colored into dark red (head arrow).
Figure 6.
Histological analysis of rat offsping follicules showing alterations Magnification X40. A represents a normal morphology of antral follicle with normal oocytes and normal cumulus cells and theca cells. B represents an atresia in follicle with color change to dark red and absence of oocyte (yellow cercle). C represents a degenerative follicle, with no oocytes (black cercle) and a follicle with fractured oocyte that colored into dark red (head arrow).
b. Testis of rat offspring
Based on the histological analysis of testis from rat offspring’s the control group displayed typical and normal histological features of the seminiferous tubes (STs). However, in contrast, the CPF and the HFD subgroups exhibited a clear desquamation of the seminiferous epithelium
Figure 6. This desquamation resulted in the release of cellular debris and a large number of germinal lineage cells, predominantly primary spermatocytes, Cell in degeneration and round spermatids presented in the tubular lumen (
Figure 8 B, C, D). The Coex group presents a disorganized structure of ST (
Figure 7 D).
Figure 7.
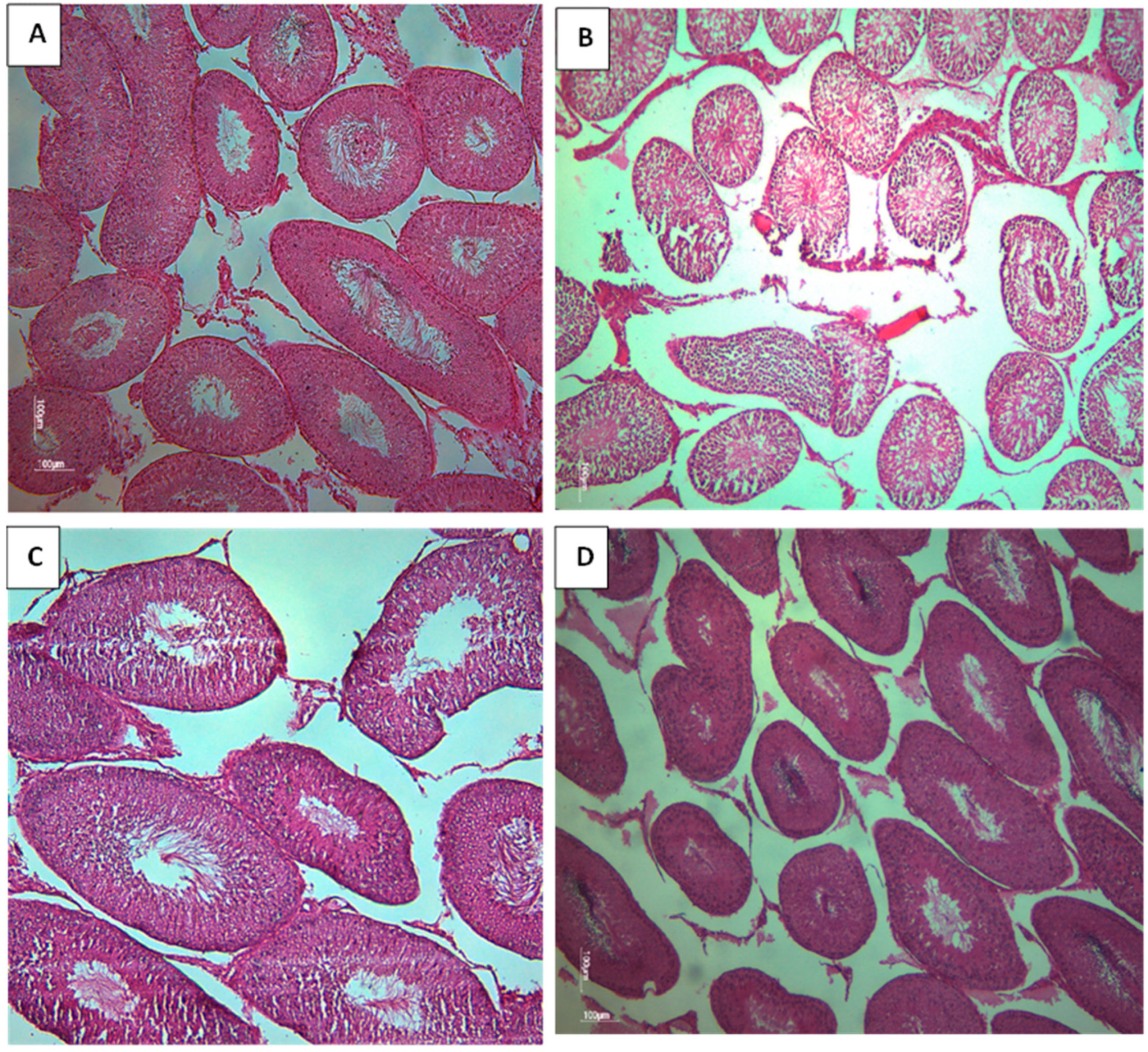
Histological testicular section of rat offsprings exposed to different treatments (Magnification X 10). A represents the normal morphology of rat testis. B represents a testis morphology of CPF exposed group, The observation reveals a disordered structure of ST, a notable absence of germ cells and a partial reduction in the ST. C represents the HFD exposed group, note reduction of some germ cells. D represents the coexposed group, the germ cells found in the tubular lumen.
Figure 7.
Histological testicular section of rat offsprings exposed to different treatments (Magnification X 10). A represents the normal morphology of rat testis. B represents a testis morphology of CPF exposed group, The observation reveals a disordered structure of ST, a notable absence of germ cells and a partial reduction in the ST. C represents the HFD exposed group, note reduction of some germ cells. D represents the coexposed group, the germ cells found in the tubular lumen.
Figure 8.
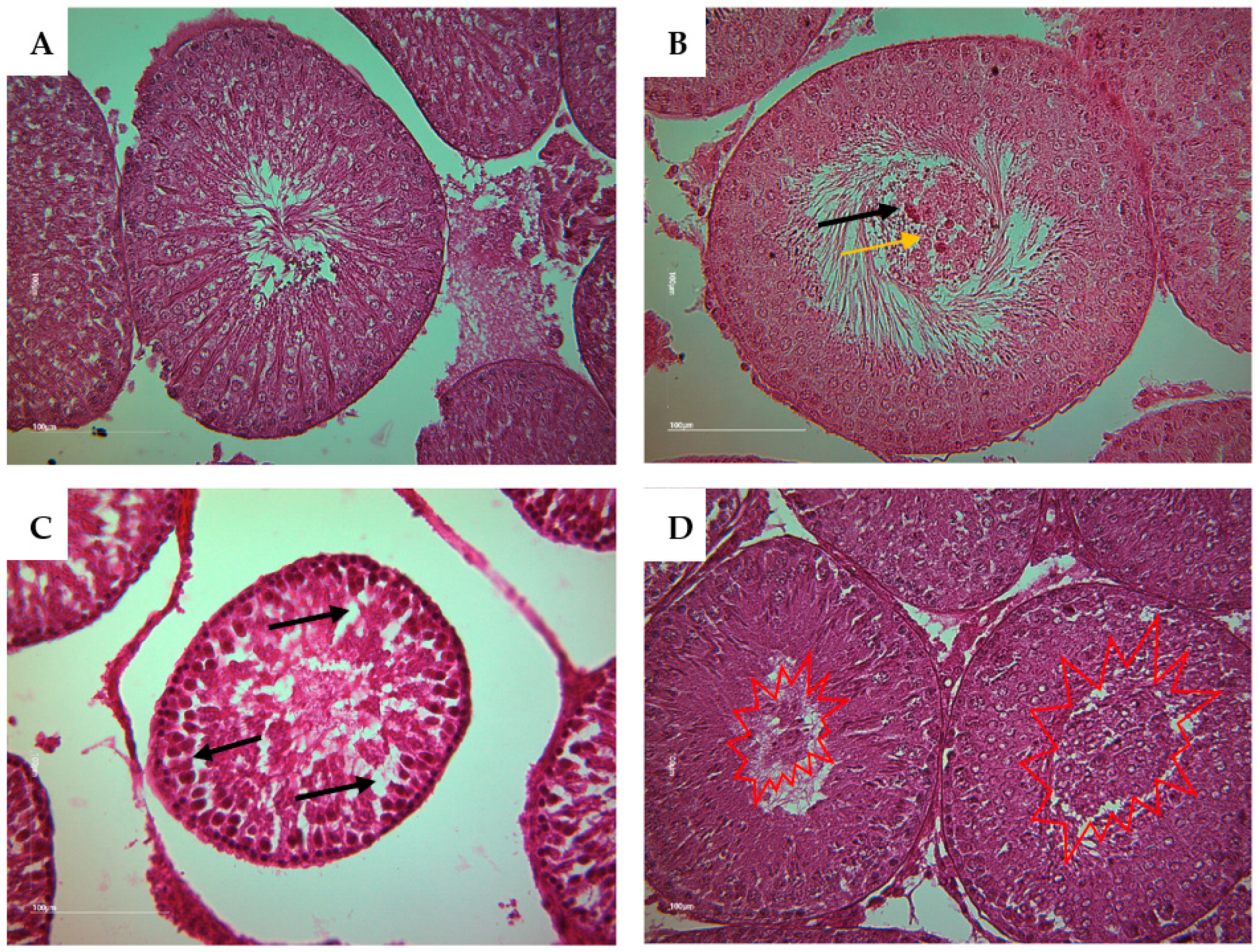
Histological detection of tubular section alteration of rat offsprings (Magnification X 40). A show a normal ST containing various cell types including spermatogonia Ad, spermatogonia B, primary spermatocytes, round and elongated spermatids with Sertoli cells and Leydig cells. B shows the presence of some germinal lineage cells in the tubular lumen including primary spermatocytes (yellow arrow) and cell in degeneration (black arrow). C shows an abnormal structure of ST with partial depletion or an absence of some germ cell (black arrow). D shows an accumulation of a large quantity of germinal lineage cells in the tubular lumen including cells in degeneration, round spermatids and primary spermatocytes (red explosion).
Figure 8.
Histological detection of tubular section alteration of rat offsprings (Magnification X 40). A show a normal ST containing various cell types including spermatogonia Ad, spermatogonia B, primary spermatocytes, round and elongated spermatids with Sertoli cells and Leydig cells. B shows the presence of some germinal lineage cells in the tubular lumen including primary spermatocytes (yellow arrow) and cell in degeneration (black arrow). C shows an abnormal structure of ST with partial depletion or an absence of some germ cell (black arrow). D shows an accumulation of a large quantity of germinal lineage cells in the tubular lumen including cells in degeneration, round spermatids and primary spermatocytes (red explosion).
Morphometric analysis of tubule sections did not exhibit significant statistical differences in ST area between the different groups, as indicated in
Table 1.
Table 1.
Area (μm2) of seminiferous tubule sections observed in rat offspring in different groups; Control, CFP group, HFD group and Coexp group.
Table 1.
Area (μm2) of seminiferous tubule sections observed in rat offspring in different groups; Control, CFP group, HFD group and Coexp group.
3- Kisspeptin and GnRHR immunodetection
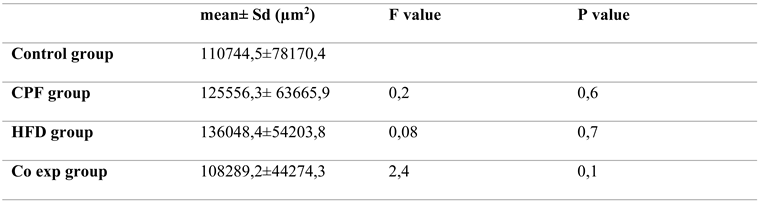
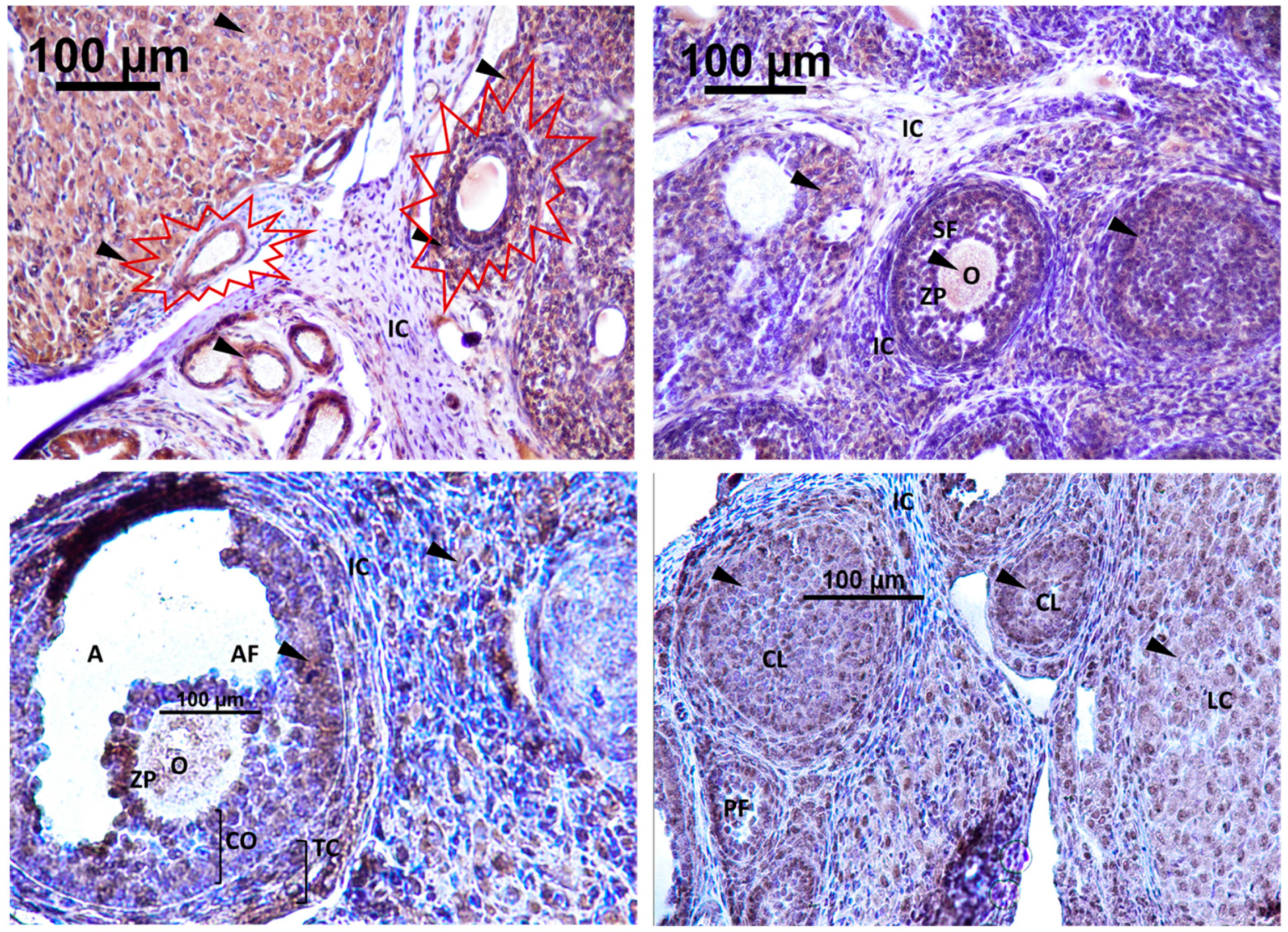
a. Localization of immunoreactive kisspeptin and GnRHR in the ovary of rat offspring
The expression of Kisspeptin was detected in different localizations within the rat ovary. Strong immunoreactivity for GnRH was exhibited in the CL: corpus luteum, LC: luteal cells, O: oocyte, TC: theca cell of atretic follicle and in the cytoplasm of GC: granulosa cell. No detection was observed in interstitial tissue and theca cells of secondary follicle
Figure 9.
Figure 9.
immunodetection of Kisspeptin in rat ovary offspring. IC: interstitial cell, CL: corpus luteum cell, A: antrum, O: oocyte, ZP: zona pellucida, SF: secondary follicle, PF: primary follicle, AF: antral follicle, CO: cumulus oophorus, TC: theca cell, GC: granulosa cell. The first section of the Figure shows the atretic follicle (surrounded in red) Magnification X40.
Figure 9.
immunodetection of Kisspeptin in rat ovary offspring. IC: interstitial cell, CL: corpus luteum cell, A: antrum, O: oocyte, ZP: zona pellucida, SF: secondary follicle, PF: primary follicle, AF: antral follicle, CO: cumulus oophorus, TC: theca cell, GC: granulosa cell. The first section of the Figure shows the atretic follicle (surrounded in red) Magnification X40.
Strong immunodetection for GnRHR was noted in the interstitial cell (IC) and O: oocyte within the rat ovary offspring
Figure 9. No detection of GnRHR in SF: secondary follicle, PF: primary follicle, AF: antral follicle, CO: cumulus oophorus, TC: theca cell, GC: granulosa cell
Figure 10.
Figure 10.
immunodetection of GnRHR in rat ovary offspring; IC: interstitial cell, CL: corpus luteum, A: antrum, O: oocyte, ZP: zona pellucida, SF: secondary follicle, PF: primary follicle, AF: antral follicle Magnification X40.
Figure 10.
immunodetection of GnRHR in rat ovary offspring; IC: interstitial cell, CL: corpus luteum, A: antrum, O: oocyte, ZP: zona pellucida, SF: secondary follicle, PF: primary follicle, AF: antral follicle Magnification X40.
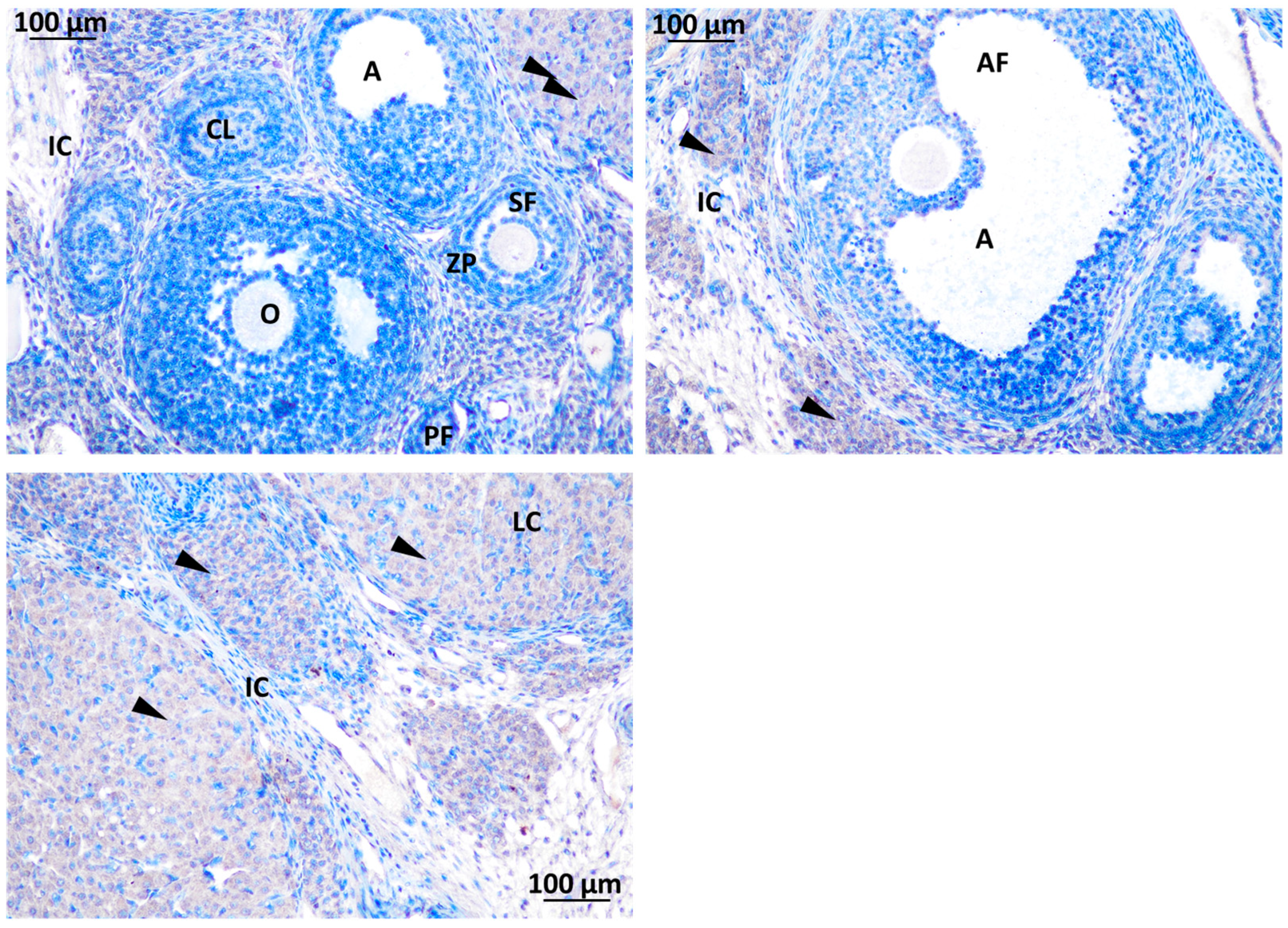
b. Localization of immunoreactive kisspeptin and GnRHR in the testis of rat offspring
Immunodetection of kisspeptin has revealed its presence in specific areas within the testis of rat offspring. Kisspeptin was observed in two main locations in the testis: Interstitial Leydig Cells (LC) in the interstitial spaces between the seminiferous tubules suggesting a potential role in regulating testosterone production and Inside the Seminiferous Tubules within the seminiferous tubules, indicates a possible direct influence on spermatogenesis Figure 11.
Figure 11.
immunodetection of Kisspeptin in seminiferous tubules of rat offsprings testis; S.Ad: spermatogonia Ad, SpI: spermatocyte I, SCN: Sertoli cell nuclei, ST: seminiferous tube, S.B: Spermatogonia B, Sp C: Spermatogonia C.
Figure 11.
immunodetection of Kisspeptin in seminiferous tubules of rat offsprings testis; S.Ad: spermatogonia Ad, SpI: spermatocyte I, SCN: Sertoli cell nuclei, ST: seminiferous tube, S.B: Spermatogonia B, Sp C: Spermatogonia C.
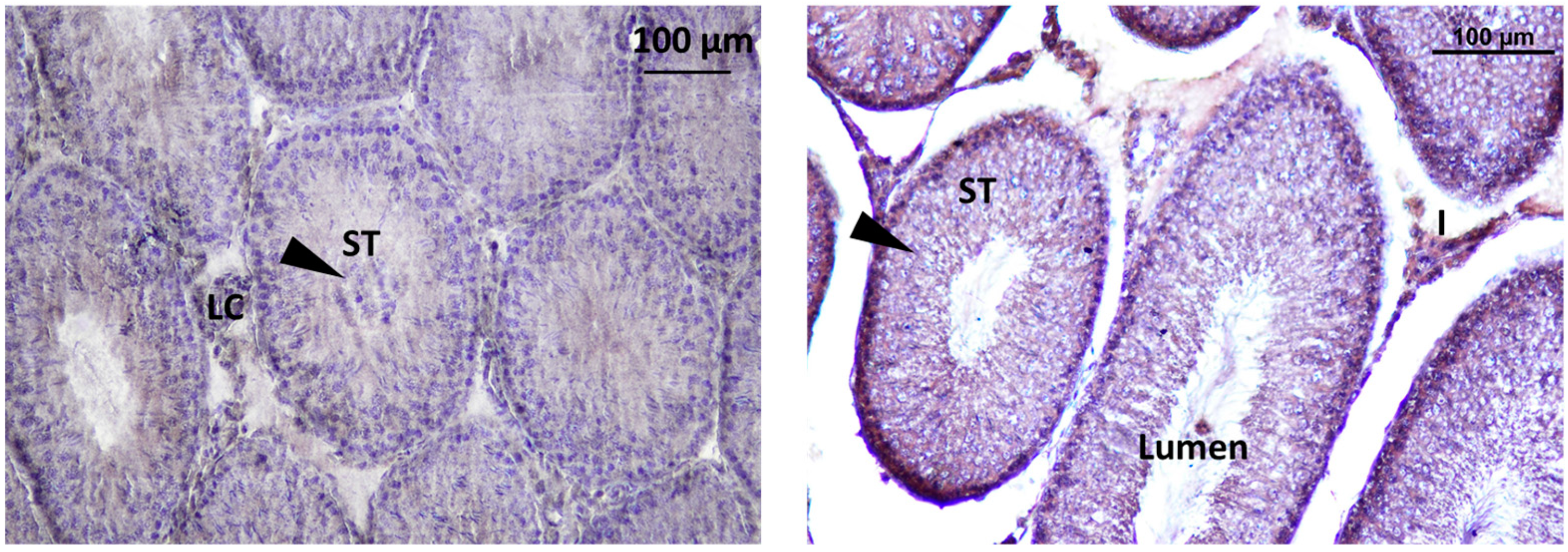
Localization of GnRHR was observed in the interstitial Leydig cells (LC) and inside the seminiferous tubule. Localization of immunoreactive kisspeptin and GnRHR in the testis of rat offsprings Figure 12.
Figure 12.
immunodetection of Kisspeptin in seminiferous tubules of rat offsprings testis; LC: Leydig cell, ST: seminiferous tube, I: intertubular.
Figure 12.
immunodetection of Kisspeptin in seminiferous tubules of rat offsprings testis; LC: Leydig cell, ST: seminiferous tube, I: intertubular.
4- In silico quantification of the Expression of Kisspeptin and GnRHR
The levels of Kisspeptin and GnRHR expression in the reproductive organs (testis and ovary) of rat offsprings were measured using HistoQuest®.
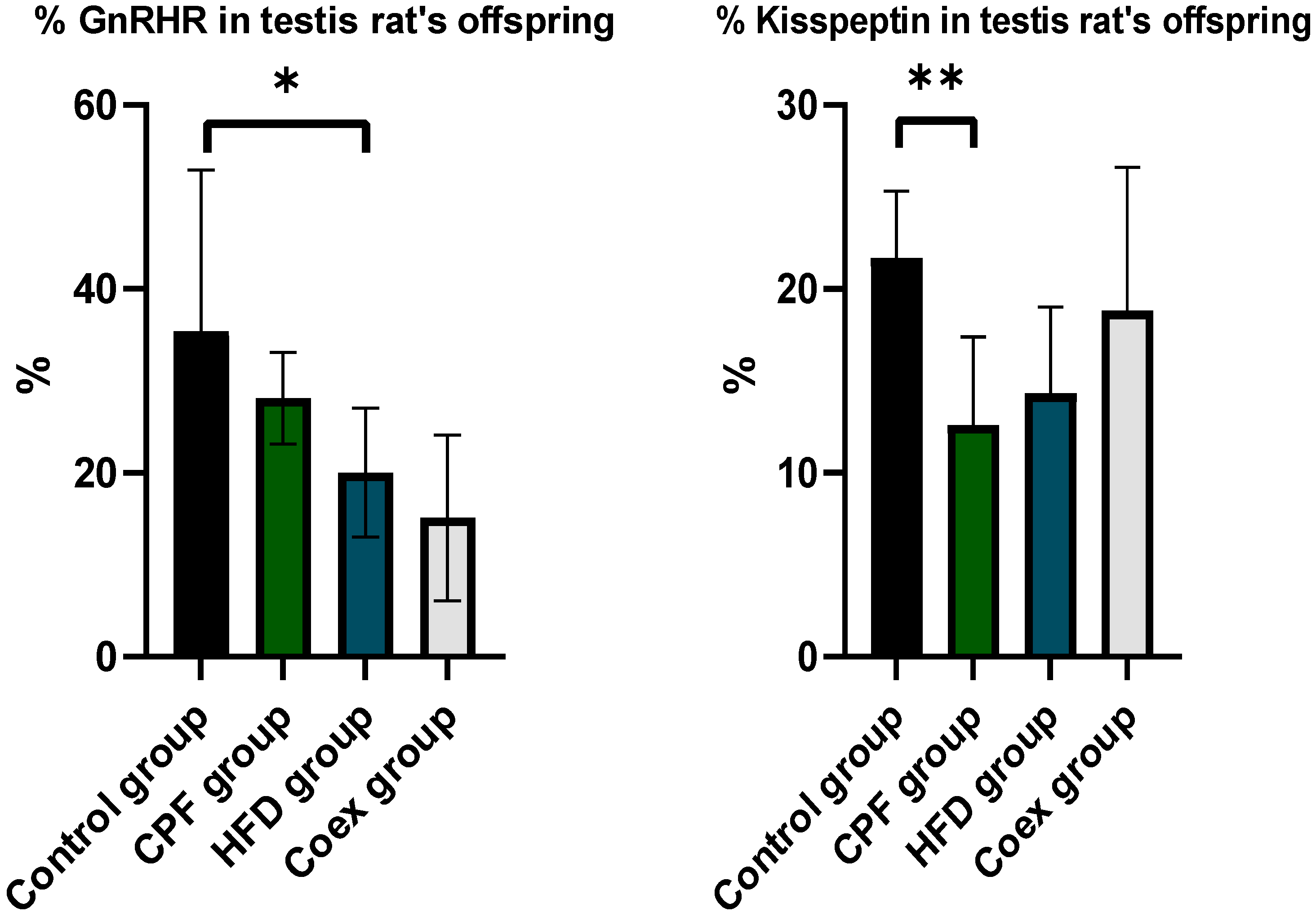
The statistical analysis revealed a significant decrease in the percentage of GnRHR (P=0.01) in the offspring from the high-fat diet (HFD) group compared to the control group. However, there were no significant differences observed between the control group, the group exposed to CPF (chlorpyrifos, a pesticide), and the coexposure group (p<=0,05) (
Figure 13 A).
The data analysis demonstrated a significant reduction in the percentage of Kisspeptin (P=0.002) in the testis rat offspring exposed to CPF, compared to the control group. No significant difference found in the other groups (p<=0,05) (
Figure 13 B).
The quantification of levels of Kisspeptin and GnRHR in ovary rat offspring show no significant difference between the different groups (p<=0,05).
Figure 13.
Effect of different treatments on GnRHR and kisspeptin expression in testis rat Offspring.
Figure 13.
Effect of different treatments on GnRHR and kisspeptin expression in testis rat Offspring.
The comparison of the number of cells in ovary rat offspring revealed a significant decrease in cell number shown in the CPF group compared to the control group (p=0,002). No significant difference found in the other groups (p<=0,05) Figure 14.
Data analysis of testis’ cell number in different treated group show no significant differences in cell number between the groups (p<=0,05).
Figure 11.
Effect of Different Treatments on the number of cell in ovary rat Offspring.
Figure 11.
Effect of Different Treatments on the number of cell in ovary rat Offspring.
4. Discussion
The present study investigates the adverse effects of chlorpyrifos pesticide and high fat diet on fertility and reproduction function in rat offspring. Chlorpyrifos pesticide was chosen in this study because it is widely used in agriculture especially in France and it’s considered as an endocrine disruptor [
9,
10].
In vivo studies have explored the effects of CPF treatment on body weight and birth weight, some showing an increase in body weight and birth weight with CPF exposure, while others have shown a decrease [
18,
49].
The current study revealed a significant gain in body weight from PND1 to PND60 with the highest body weight observed in HFD group. The intergroup comparison at PND30 and PND60 shows a significantly lower body weight in the CPF-treated groups compared to the other groups. Similar results were observed by Condette et al. 2014, which revealed that rats exposed to CPF exhibited a notable increase in weight (averaging 13 g; p<0.01) and length (averaging 2 cm; p<0.01) compared to non-exposed groups [
50]. In addition, the body weight of rat offspring decreases in the co-exposed (Coex) group compared to the HFD group at PND 15 and PND 30. This finding suggests the toxic effect of CPF. Several studies corroborate the observed results [
18,
45]. Similarly, Akhtar et al. 2009 revealed a significant body weight decrease at high dose (9 mg kg-1d -1) of chlorpyrifos [
12]. Conversely, Silva in 2021 reported that male exposed to CPF showed increased body weights [
51]. The perinatal exposure to CPF cause an indirect toxicity of rat offspring by leading to a hormonal disorder through the endocrine disrupting property [
52]. CPF can block or alter growth hormones including GnRH which contribute to a development decline [
53]. The morphological analysis of rat testis shows an abnormal testis structure including a depletion of seminiferous epithelium and some germinal lineage cells and absence specially in CPF exposed group and HFD exposed group. Similar outcome was found in the study of spooner in 2015 [
54,
55]. It seems that exposure to CPF may have detrimental effects on spermatogenesis and the production of viable sperm [
46].
No significant difference in seminiferous tube area was observed but histopathological sign was detected. This alteration is defined by the presence of some germinal lineage cells in the tubular lumen including primary spermatocytes and cells in degeneration and an accumulation of large quantities of germinal lineage cells in the tubular lumen. The study of Gabriel et al. 2014 confirms the current results, they reported that oral ingestion of Permethrin lead to abnormalities such as disruption of the normal architecture, decrease of mature sperm cells, and reduction of luminal diameter and reduced interstitial spaces [
56]. Histological analysis of ovary rat offspring shows significant increase in atretic follicle in CPF exposed group compared to control group. It seems that CPF disrupts the folliculogenesis and leads to follicle and oocyte degeneration [
57]. Several studies demonstrate similar result [
58,
59]
Our study demonstrated the immunodetection of kisspeptin and GnRHR for the first time in the reproductive organs of rat offspring. This finding suggests the presence and potential role of kisspeptin and GnRHR in the reproductive development of rats as reported in other studies [
31,
34,
35,
42]. In ovary rat offspring, the immunodetection of kisspeptin was found in the different stages of follicles (from primordial to antral follicle) and in the corpus luteum cells. Indeed, the results of the study strongly suggest that kisspeptin plays a significant role in the regulation of folliculogenesis and suggests its involvement in regulating the development and maturation of ovarian follicles [
60]. Similarly, the immunodetection of kisspeptin in testis rat offspring’s including seminiferous tubes and Leydig cells reveal the function of kisspeptin in spermatogenesis, the initiation of puberty, regulation of the male reproductive system, and overall control of testosterone secretion [
30,
61].
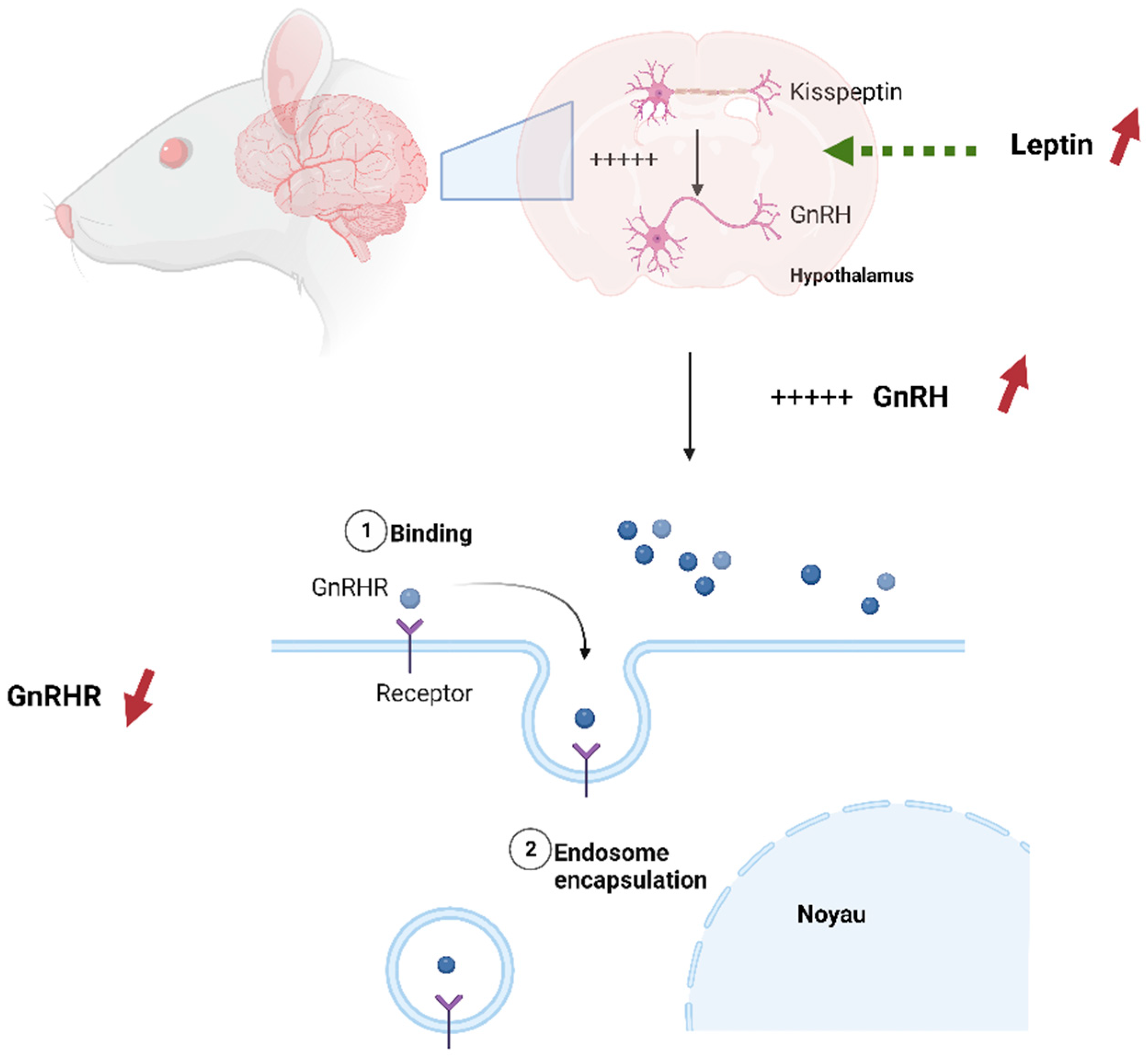
This study is the first to assess the impact of pesticide exposure and HFD on kisspeptin and GnRHR presence in the reproductive organs of rat offspring. Our findings demonstrate a reduction in the expression of GnRHR in the testis of rat offspring’s exposed to HFD. In the context of a high-fat diet, excessive intake of dietary fats can lead to an increased release of leptin, a hormone produced by adipose (fat) cells in response to the body’s energy stores [
62]. Leptin receptors are present on kisspeptin neurons in the hypothalamus. When leptin binds to these receptors, it stimulates the activity of kisspeptin neurons, leading to an increase in kisspeptin expression. Kisspeptin, in turn, stimulates the release of GnRH from the hypothalamus [
63]. However, over time, the constant exposure to high fat diet leads to an increase of leptin expression and as a result the increase of GnRH [
63]. It’s widely known that the responsiveness of cells to specific signaling hormones includes different mechanisms to regulate homeostasis [
64]. These mechanisms include receptor downregulation and receptor desensitization [
65]. It seems that as a response to excessive stimulation of GnRH after the mother’s high fat diet exposure, testis cells applied a downregulation mechanism to reduce the number of GnRHR receptors Figure 12.
Figure 12.
GnRHR downregulation as a response to High fat diet (personal source).
Figure 12.
GnRHR downregulation as a response to High fat diet (personal source).
Hiller-Sturmhöfel et Bartke, 1998 defined this process by internalization of some receptors, effectively removing them from the cell surface [
64]. According to CPF exposure, our result shows a negative impact of the expression of kisspeptin in the rat offspring’s testis. Chlorpyrifos, as an endocrine disruptor, alters the expression of extrahypothalamic kisspeptin in rat ‘s testis. This result is similar to the study of Johanna that reported the decrease of kisspeptin expression in cells exposed to dioxin [
66,
67]. Conversely it was revealed that
Kiss1 expression was not affected by endocrine disruptors [
68].
In ovary rat offspring, the comparison of kisspeptin and GnRHR does not exhibit persistent changes after perinatal exposure to CPF, HFD and coex. Whereas, several studies have shown a negative link between pesticide exposure and kisspeptin expression [
30,
36,
69,
70].
The strategy of this study is a chronic low dose perigestational exposure to understand the indirect effect on the progeny. Thus, our study highlights the concept “window of vulnerability” of the “Developmental Origin of Health and Disease” [
17]. In other terms, exposure to food contaminants such as pesticides during the first 1000 days of life increases vulnerability to chronic diseases [
71].
In the current study, the perinatal exposure of female rats to chlorpyrifos and/or a high-fat diet had no effect on the expression level of kisspeptin and GnRH receptor in the female offspring, however, it significantly impacted the male offspring. The differential response of the male and female offspring to the exposure raises questions about the underlying mechanisms and biological differences between the sexes [
72]. It may indicate that there are sex-specific pathways or hormone signaling networks involved in the regulation of kisspeptin and GnRH receptor expression [
73]. The DOHaD hypothesis suggests that early life exposures lead to the development of chronic diseases such as obesity, diabetes, cardiovascular disease, and even certain neurodevelopmental disorders [
22]. These periods of development (prenatal and early postnatal periods) are considered as critical windows of vulnerability[
20]. During these windows, cells and tissues are rapidly differentiating and developing, making them more susceptible to disruptions caused by environmental exposures [
20]. The chronic low-dose exposure to CPF can have indirect effects on offspring when it occurs through maternal exposure during pregnancy[
18]. CPF can cross the placental barrier and reach the developing fetus and leads to an indirect health alteration of offspring which can develop in long-term health Impact[
74]. In addition, our findings suggest the implication of non-genetic mechanisms “epigenetics” that might explain these changes in male offspring in response to maternal environment during early development and postnatal period [
21]. Exposure to CPF during pregnancy can also influence gene expression patterns in the fetus through epigenetic mechanisms [
21]. Understanding these sex-specific differences could be crucial for developing targeted interventions or therapies to mitigate the potential negative impacts of environmental factors on reproductive health [
75].
A significant severe reduction in cell number was demonstrated in the offspring’s ovary of the rat exposed to CPF. It seems that the CPF induces an oxidative stress in ovary tissue leading to an increase in ROS levels within the ovarian tissue. These ROS are highly reactive and can cause damage to cellular components, such as lipids, proteins, and DNA. As a result, the delicate balance within the ovarian cells is disrupted. Apoptosis process is initiated as a protective response to eliminate cells that are severely affected by the ROS-induced damage[
76,
77,
78,
79].