Submitted:
30 August 2023
Posted:
31 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Preparation of samples and optimization of extraction for phenolic compounds
2.2. Quantification of phenolic contents in selected medicinal plants
2.2.1. Determination of Total Phenolic Content
2.2.2. Total Flavonoid Content
2.2.3. Total Tannin Content
2.3. Measurement of antioxidant and anti-diabetic potential of selected plants
2.4. LC-ESI-QTOF-MS/MS analysis
2.5. GC-HS-SPME-MS analysis of volatile compounds
2.6. Molecular docking of the most abundant phenolic compounds
2.7. Statistical analysis
3. Results and Discussion
3.1. Optimization of extraction process of total phenolic content from the selected plants
3.2. Estimation of polyphenols from culinary herbs and spices
3.2. Antioxidant and anti-diabetic activities of seleted medicinal plants
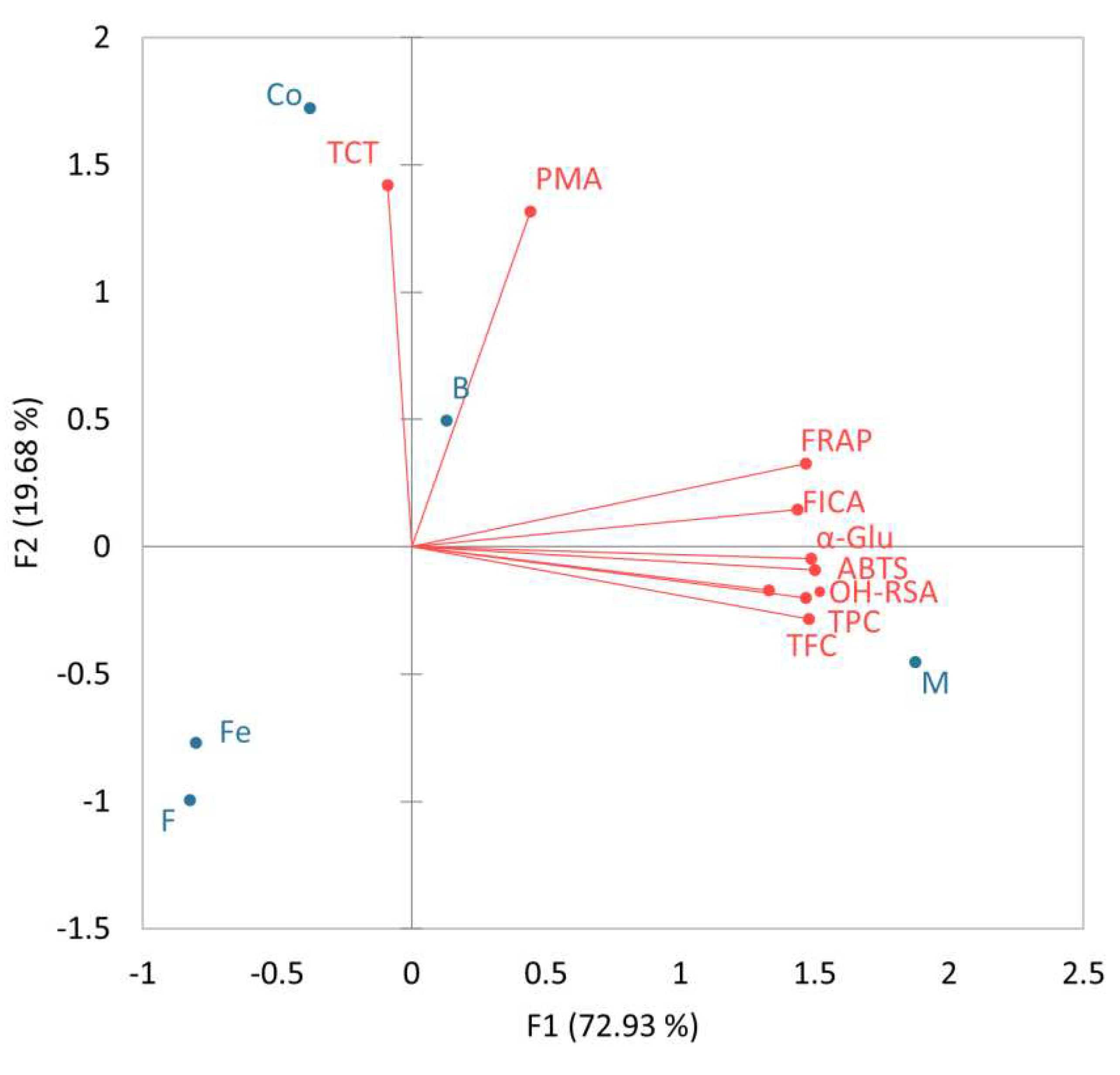
3.3. Correlation of biological activities and phenolics
3.4. LC-MS Characterization of Phenolic Compounds from Culinary Spices and Herbs
3.4.1. Flavonoids
Flavanols
Flavanones
Flavones
Flavonols
3.4.2. Isoflavonoids
3.4.3. Phenolic acids
Hydroxybenzoic acids
Hydroxycinnamic acids:
3.4.5. Other compounds
3.5. Quantification and Semi-quantification of volatile and non-volatile compounds
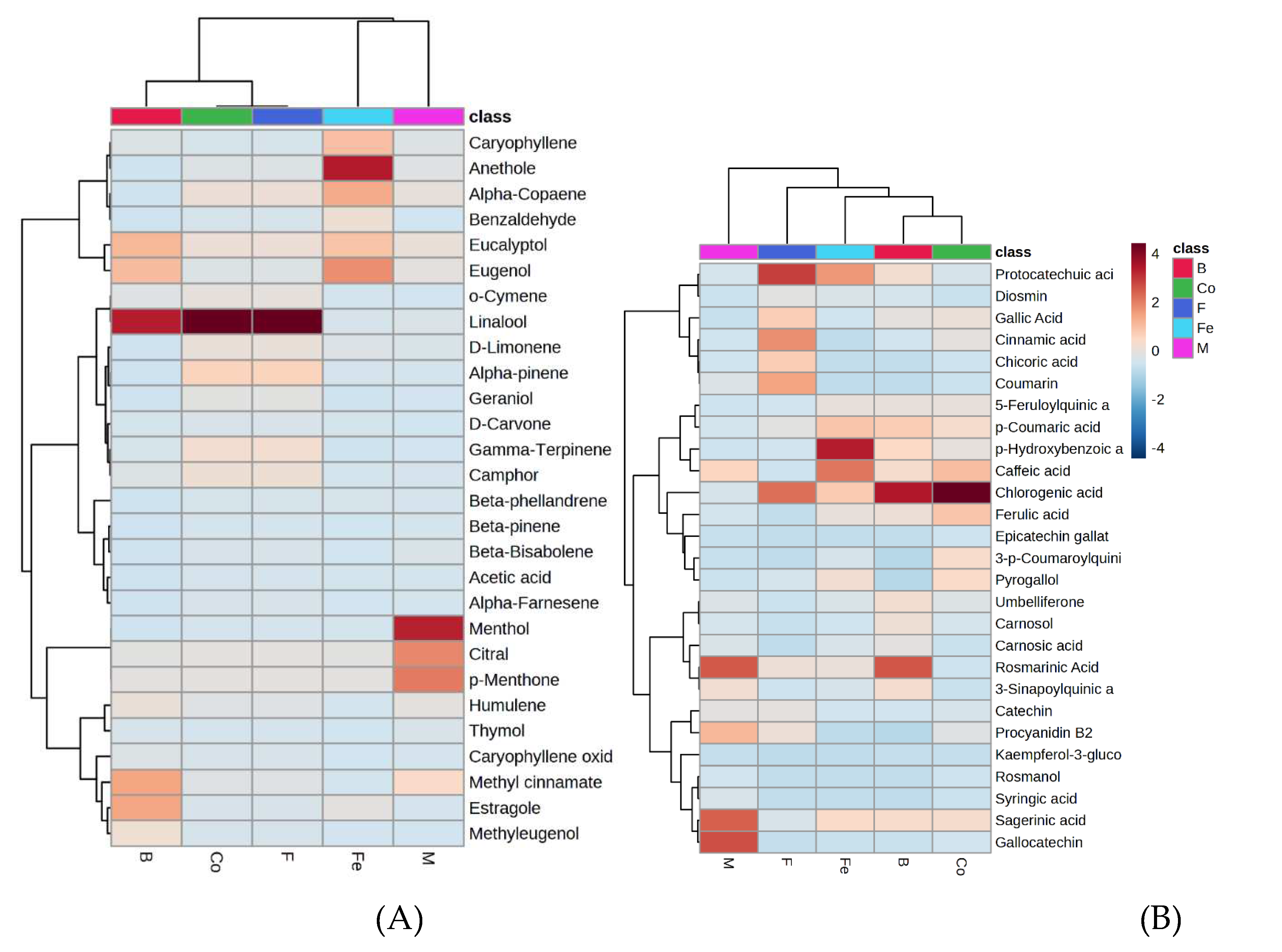
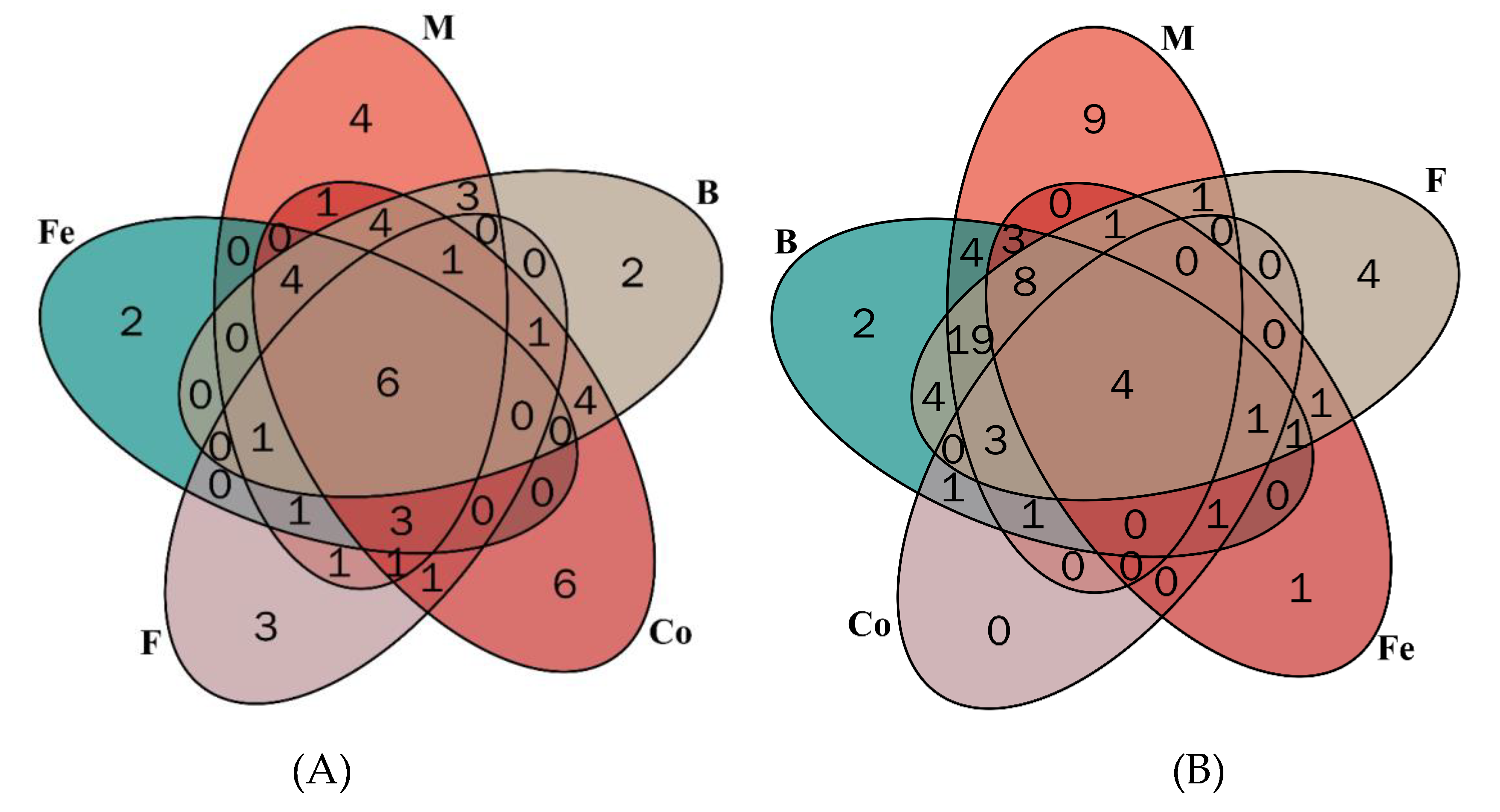
3.6. Distribution of phytochemicals in selected herbs and spices
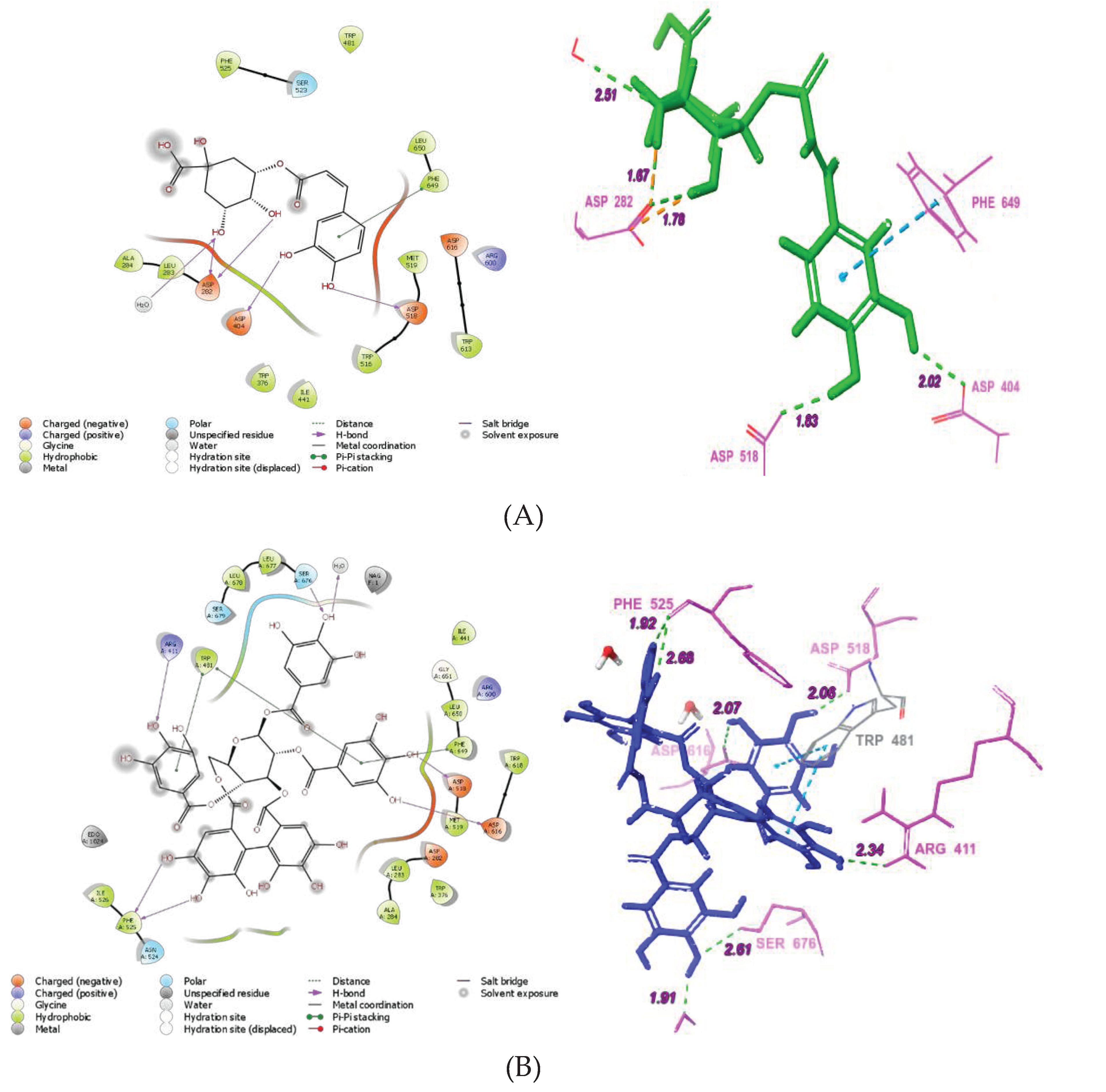
3.7. Molecular docking of abundant phenolic compounds
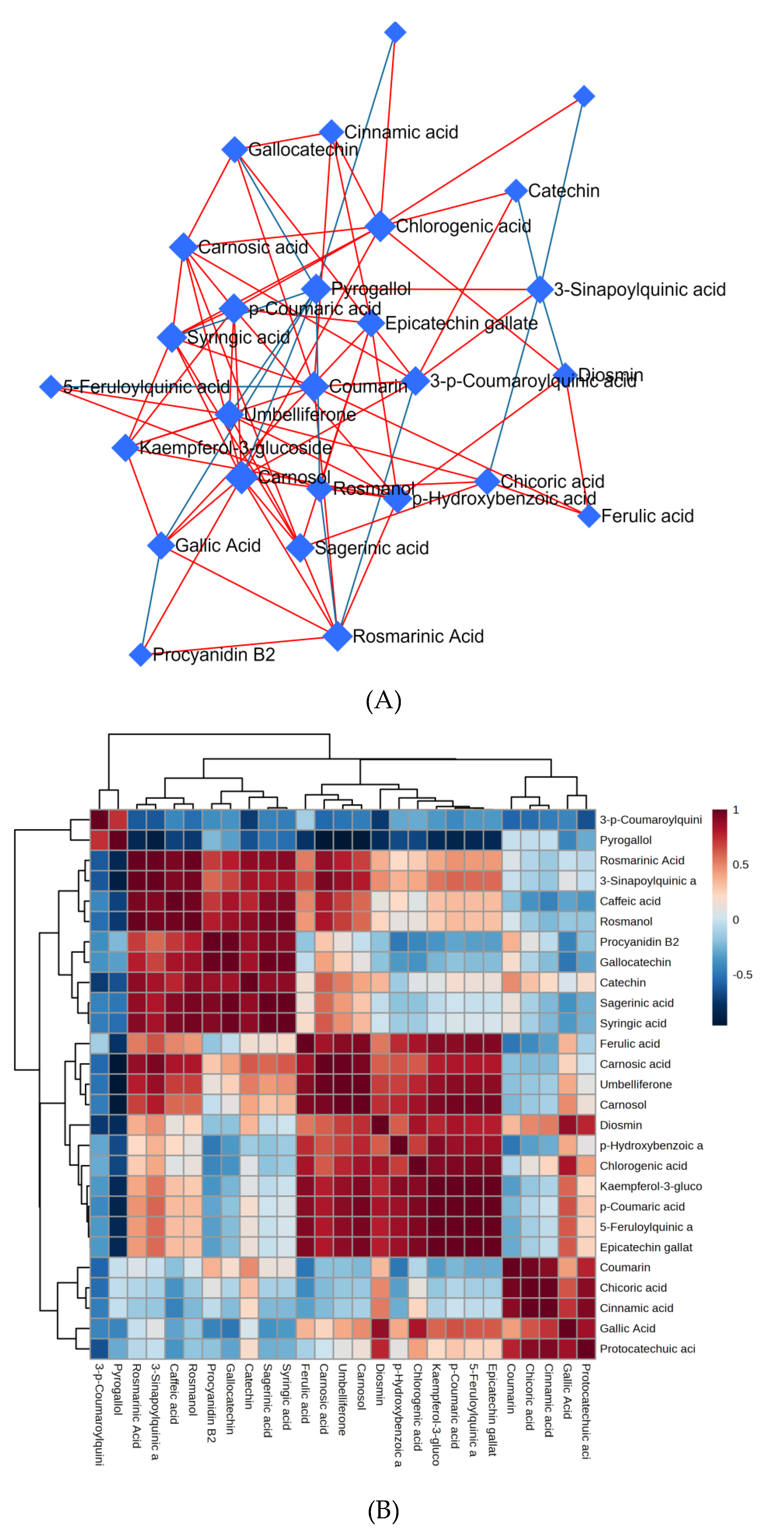
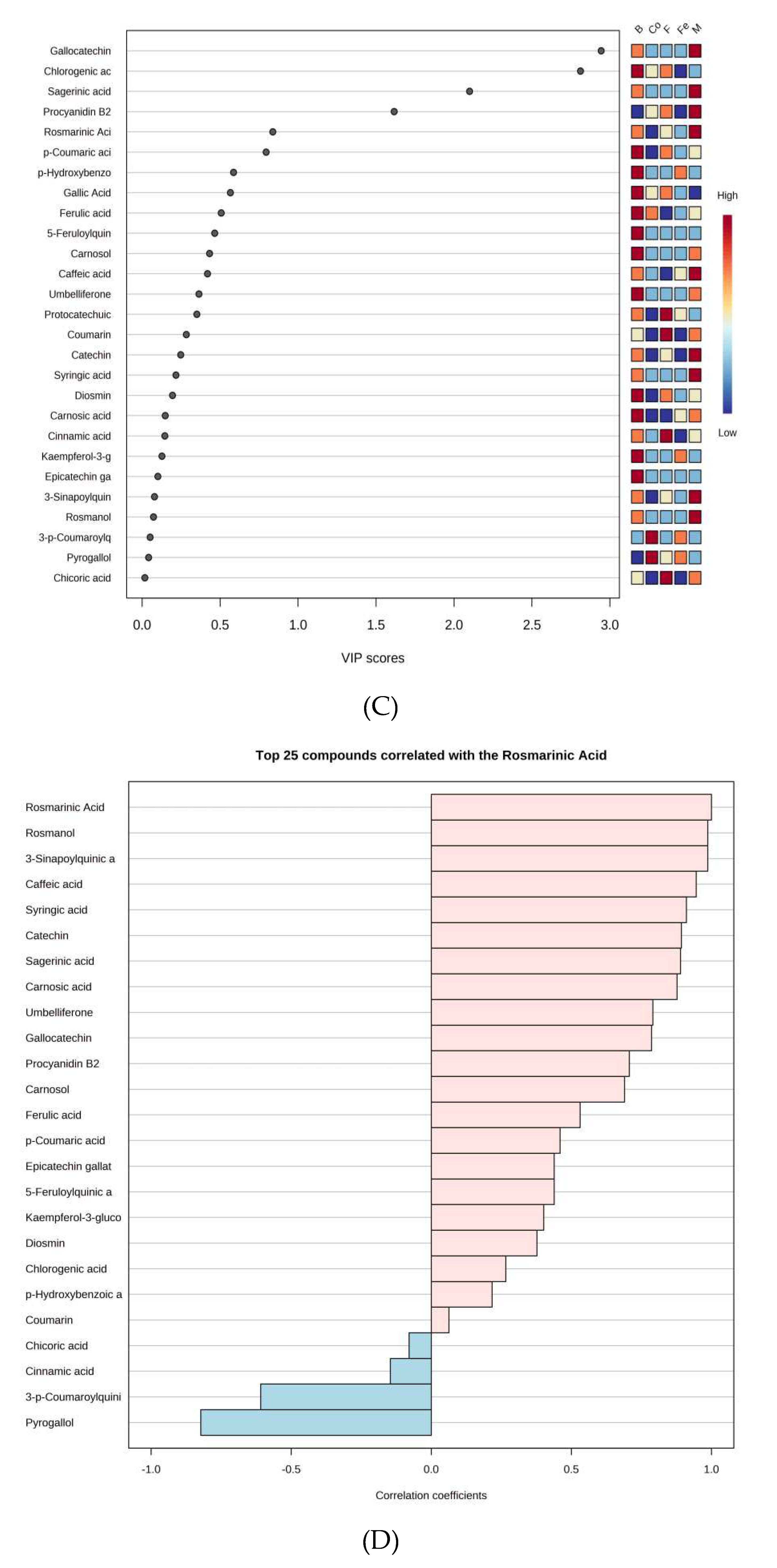
3.9. Chemometrics analysis of abundant metabolites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ali, A.; Cottrell, J.J.; Dunshea, F.R. Antioxidant, alpha-glucosidase inhibition activities, in silico molecular docking and pharmacokinetics study of phenolic compounds from native australian fruits and spices. Antioxidants 2023, 12, 254. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Cottrell, J.J.; Dunshea, F.R. Identification and characterization of anthocyanins and non-anthocyanin phenolics from australian native fruits and their antioxidant, antidiabetic, and anti-alzheimer potential. Food Research International 2022, 162, 111951. [Google Scholar] [CrossRef]
- Ali, A.; Wu, H.; Ponnampalam, E.N.; Cottrell, J.J.; Dunshea, F.R.; Suleria, H.A.R. Comprehensive profiling of most widely used spices for their phenolic compounds through lc-esi-qtof-ms2 and their antioxidant potential. Antioxidants (Basel) 2021, 10, 721. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Hinneburg, I.; Dorman, H.J.D.; Hiltunen, R. Antioxidant activities of extracts from selected culinary herbs and spices. Food Chem. 2006, 97, 122–129. [Google Scholar] [CrossRef]
- Leja, K.B.; Czaczyk, K. The industrial potential of herbs and spices? A mini review. Acta Scientiarum Polonorum Technologia Alimentaria 2016, 15, 353–365. [Google Scholar] [CrossRef]
- Gupta, M. Pharmacological properties and traditional therapeutic uses of important indian spices: A review. International Journal of Food Properties 2010, 13, 1092–1116. [Google Scholar] [CrossRef]
- Ali, A.; Bashmil, Y.M.; Cottrell, J.J.; Suleria, H.A.R.; Dunshea, F.R. Lc-ms/ms-qtof screening and identification of phenolic compounds from australian grown herbs and their antioxidant potential. Antioxidants (Basel) 2021, 10, 1770. [Google Scholar] [CrossRef]
- Ali, A.; Kiloni, S.M.; Cáceres-Vélez, P.R.; Jusuf, P.R.; Cottrell, J.J.; Dunshea, F.R. Phytochemicals, antioxidant activities, and toxicological screening of native australian fruits using zebrafish embryonic model. Foods 2022, 11, 4038. [Google Scholar] [CrossRef]
- Bashmil, Y.M.; Ali, A.; BK, A.; Dunshea, F.R.; Suleria, H.A.R. Screening and characterization of phenolic compounds from australian grown bananas and their antioxidant capacity. Antioxidants 2021, 10, 1521. [Google Scholar] [CrossRef]
- Ali, A.; Wu, H.; Ponnampalam, E.N.; Cottrell, J.J.; Dunshea, F.R.; Suleria, H.A.R. Comprehensive profiling of most widely used spices for their phenolic compounds through lc-esi-qtof-ms(2) and their antioxidant potential. Antioxidants (Basel, Switzerland) 2021, 10. [Google Scholar]
- Ali, A.; Bashmil, Y.M.; Cottrell, J.J.; Suleria, H.A.R.; Dunshea, F.R. Lc-ms/ms-qtof screening and identification of phenolic compounds from australian grown herbs and their antioxidant potential. Antioxidants (Basel, Switzerland) 2021, 10. [Google Scholar] [CrossRef]
- Ali, A.; Ahmadi, F.; Cottrell, J.J.; Dunshea, F.R. Comprehensive metabolite fingerprinting of australian black and green olives and their antioxidant and pharmacokinetics properties. Separations 2023, 10, 354. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Song, S.; Ali, A.; Subbiah, V.; Taheri, Y.; Suleria, H.A.R. Lc-esi-qtof-ms/ms characterization of phenolic compounds from pyracantha coccinea m. Roem. And their antioxidant capacity. Cellu. Molec. Biol. 2021, 67, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Chou, O.; Ali, A.; Subbiah, V.; Barrow, C.J.; Dunshea, F.R.; Suleria, H.A.R. Lc-esi-qtof-ms/ms characterisation of phenolics in herbal tea infusion and their antioxidant potential. Fermentation 2021, 7, 73. [Google Scholar] [CrossRef]
- Ali, A.; Cottrell, J.J.; Dunshea, F.R. Characterization, antioxidant potential, and pharmacokinetics properties of phenolic compounds from native australian herbs and fruits. Plants 2023, 12, 993. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Cottrell, J.J.; Dunshea, F.R. Characterization, antioxidant potential, and pharmacokinetics properties of phenolic compounds from native australian herbs and fruits. Plants 2023, 12, 993. [Google Scholar] [CrossRef]
- Ali, A.; Zahid, H.F.; Cottrell, J.J.; Dunshea, F.R. A comparative study for nutritional and phytochemical profiling of coffea arabica (c. Arabica) from different origins and their antioxidant potential and molecular docking. Molecules 2022, 27, 5126. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, M.A.; Ashraf, S.; Mahmood, S.; Hafiz, I.; Rehman, A.U.; Hussain, H.I.; Zakria, H.M.; Dilbar, G.H.; Riaz, M. Antioxidant and antibacterial potential of cinnamon bark, fennel seeds and peppermint leaves. PROGRESS IN NUTRITION 2021, 23. [Google Scholar]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. Journal of Agricultural and Food Chemistry 2001, 49, 5165–5170. [Google Scholar] [CrossRef]
- Aliakbarlu, J.; Mohammadi, S.; Khalili, S. A study on antioxidant potency and antibacterial activity of water extracts of some spices widely consumed in iranian diet. Journal of Food Biochemistry 2014, 38. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Sun, M.; Corke, H. Antioxidant capacity of 26 spice extracts and characterization of their phenolic constituents. J Agric Food Chem 2005, 53, 7749–7759. [Google Scholar] [CrossRef] [PubMed]
- Dvorackova, E.; Snoblova, M.; Chromcova, L.; Hrdlicka, P. Effects of extraction methods on the phenolic compounds contents and antioxidant capacities of cinnamon extracts. Food Sci. Biotec. 2015, 24, 1201–1207. [Google Scholar] [CrossRef]
- Chen, H.-Y.; Lin, Y.-C.; Hsieh, C.-L. Evaluation of antioxidant activity of aqueous extract of some selected nutraceutical herbs. Food Chem 2007, 104, 1418–1424. [Google Scholar] [CrossRef]
- Yang, D.; Dunshea, F.R.; Suleria, H.A.R. Lc-esi-qtof/ms characterization of australian herb and spices (garlic, ginger, and onion) and potential antioxidant activity. Journal of Food Processing and Preservation 2020, e14497. [Google Scholar] [CrossRef]
- Patra, K.; Jana, S.; Mandal, D.P.; Bhattacharjee, S. Evaluation of the antioxidant activity of extracts and active principles of commonly consumed indian spices. Journal of Environmental Pathology, Toxicology and Oncology 2016, 35. [Google Scholar] [CrossRef]
- Przygodzka, M.; Zielińska, D.; Ciesarová, Z.; Kukurová, K.; Zieliński, H. Comparison of methods for evaluation of the antioxidant capacity and phenolic compounds in common spices. LWT-Food Science and Technology 2014, 58, 321–326. [Google Scholar] [CrossRef]
- Iqbal, M.J.; Butt, M.S.; Saeed, I.; Suleria, H.A.R. Physicochemical and antioxidant properties of pizza dough-base enriched with black cumin (nigella sativa) extracts. Current Nutrition & Food Science 2019, 15, 508–516. [Google Scholar]
- Alqahtani, Y.S.; Mahnashi, M.H.; Alyami, B.A.; Alqarni, A.O.; Huneif, M.A.; Nahari, M.H.; Ali, A.; Javed, Q.; Ilyas, H.; Rafiq, M. Preparation of spice extracts: Evaluation of their phytochemical, antioxidant, antityrosinase, and anti-α-glucosidase properties exploring their mechanism of enzyme inhibition with antibrowning and antidiabetic studies in vivo. Biomed Res Int 2022, 2022, 9983124. [Google Scholar] [CrossRef]
- Adjimani, J.P.; Asare, P. Antioxidant and free radical scavenging activity of iron chelators. Toxicology Reports 2015, 2, 721–728. [Google Scholar] [CrossRef]
- Yang, W.-J.; Li, D.-P.; Li, J.-K.; Li, M.-H.; Chen, Y.-L.; Zhang, P.-Z. Synergistic antioxidant activities of eight traditional chinese herb pairs. Biological and Pharmaceutical Bulletin 2009, 32, 1021–1026. [Google Scholar] [CrossRef]
- Yoo, K.M.; Lee, C.H.; Lee, H.; Moon, B.; Lee, C.Y. Relative antioxidant and cytoprotective activities of common herbs. Food Chemistry 2008, 106, 929–936. [Google Scholar] [CrossRef]
- Tsai, M.-L.; Lin, C.-C.; Lin, W.-C.; Yang, C.-H. Antimicrobial, antioxidant, and anti-inflammatory activities of essential oils from five selected herbs. Biosci. Biotechnol. Biochem. 2011, 1108312632–1108312632. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Yang, C.-H.; Wu, P.-S.; Kwan, C.-C.; Chen, Y.-S. Antimicrobial, anti-tyrosinase and antioxidant activities of aqueous aromatic extracts from forty-eight selected herbs. J. Med. Plants Res. 2011, 5, 6203–6209. [Google Scholar]
- Garg, D.; Muley, A.; Khare, N.; Marar, T. Comparative analysis of phytochemical profile and antioxidant activity of some indian culinary herbs. Research Journal of Pharmaceutical, Biological and Chemical Sciences 2012, 3, 845–854. [Google Scholar]
- Chen, I.-C.; Chang, H.-C.; Yang, H.-W.; Chen, G.-L. Evaluation of total antioxidant activity of several popular vegetables and chinese herbs: A fast approach with abts/h2o2/hrp system in microplates. Journal of Food and Drug Analysis 2004, 12. [Google Scholar] [CrossRef]
- Kam, A.; Li, K.M.; Razmovski-Naumovski, V.; Nammi, S.; Chan, K.; Li, G.Q. Variability of the polyphenolic content and antioxidant capacity of methanolic extracts of pomegranate peel. Nat. Prod. Commu. 2013, 8, 707–710. [Google Scholar] [CrossRef]
- Liu, H.; Qiu, N.; Ding, H.; Yao, R. Polyphenols contents and antioxidant capacity of 68 chinese herbals suitable for medical or food uses. Food Research International 2008, 41, 363–370. [Google Scholar] [CrossRef]
- Lu, M.; Yuan, B.; Zeng, M.; Chen, J. Antioxidant capacity and major phenolic compounds of spices commonly consumed in china. Food Res. Int. 2011, 44, 530–536. [Google Scholar] [CrossRef]
- Kim, I.S.; Yang, M.R.; Lee, O.H.; Kang, S.N. Antioxidant activities of hot water extracts from various spices. Int J Mol Sci 2011, 12, 4120–4131. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. Journal of Nutritional Science 2016, 5, e47. [Google Scholar] [CrossRef]
- Embuscado, M.E. Spices and herbs: Natural sources of antioxidants–a mini review. Journal of functional foods 2015, 18, 811–819. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food & nutrition research 2017, 61, 1361779. [Google Scholar]
- Gülçin, İ.; Gören, A.C.; Taslimi, P.; Alwasel, S.H.; Kılıc, O.; Bursal, E. Anticholinergic, antidiabetic and antioxidant activities of anatolian pennyroyal (mentha pulegium)-analysis of its polyphenol contents by lc-ms/ms. Biocatalysis and agricultural biotechnology 2020, 23, 101441. [Google Scholar] [CrossRef]
- Tang, J.; Dunshea, F.R.; Suleria, H.A.R. Lc-esi-qtof/ms characterization of phenolic compounds from medicinal plants (hops and juniper berries) and their antioxidant activity. Foods 2019, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Plumb, G.W.; de Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C.; Williamson, G. Antioxidant properties of gallocatechin and prodelphinidins from pomegranate peel. Redox Rep. 2002, 7, 41–46. [Google Scholar] [CrossRef]
- Kuban-Jankowska, A.; Kostrzewa, T.; Musial, C.; Barone, G.; Lo-Bosco, G.; Lo-Celso, F.; Gorska-Ponikowska, M. Green tea catechins induce inhibition of ptp1b phosphatase in breast cancer cells with potent anti-cancer properties: In vitro assay, molecular docking, and dynamics studies. Antioxidants 2020, 9, 1208. [Google Scholar] [CrossRef]
- Valdés, A.; Mellinas, A.C.; Ramos, M.; Burgos, N.; Jiménez, A.; Garrigós, M.d.C. Use of herbs, spices and their bioactive compounds in active food packaging. RSC advances 2015, 5, 40324–40335. [Google Scholar] [CrossRef]
- Plumb, G.W.; de Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C.; Williamson, G. Antioxidant properties of gallocatechin and prodelphinidins from pomegranate peel. Redox Report 2002, 7, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Grzesik, M.; Naparło, K.; Bartosz, G.; Sadowska-Bartosz, I. Antioxidant properties of catechins: Comparison with other antioxidants. Food chemistry 2018, 241, 480–492. [Google Scholar] [CrossRef]
- Wang, Y.F.; Shao, S.H.; Xu, P.; Yang, X.Q.; Qian, L.S. Catechin-enriched green tea extract as a safe and effective agent for antimicrobial and anti-inflammatory treatment. African Journal of Pharmacy and Pharmacology 2011, 5, 1452–1461. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Li, D.; Ho, C.-T.; Li, J.; Wan, X. The absorption, distribution, metabolism and excretion of procyanidins. Food & function 2016, 7, 1273–1281. [Google Scholar]
- Makita, C.; Chimuka, L.; Steenkamp, P.; Cukrowska, E.; Madala, E. Comparative analyses of flavonoid content in moringa oleifera and moringa ovalifolia with the aid of uhplc-qtof-ms fingerprinting. South African Journal of Botany 2016, 105, 116–122. [Google Scholar] [CrossRef]
- Ledesma-Escobar, C.A.; Priego-Capote, F.; Robles-Olvera, V.J.; De Castro, M.D.L. Changes in the composition of the polar fraction of persian lime (citrus latifolia) during fruit growth by lc–qtof ms/ms analysis. Food chemistry 2017, 234, 262–268. [Google Scholar] [CrossRef]
- Nardarajah, D. Hesperidin-a short review. Research Journal of Pharmacy and Technology 2014, 7, 78–80. [Google Scholar]
- Hajialyani, M.; Hosein Farzaei, M.; Echeverría, J.; Nabavi, S.M.; Uriarte, E.; Sobarzo-Sánchez, E. Hesperidin as a neuroprotective agent: A review of animal and clinical evidence. Molecules 2019, 24, 648. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Avila, J.A.; Villa-Rodriguez, J.A.; Montiel-Herrera, M.; Pacheco-Ordaz, R.; Roopchand, D.E.; Venema, K.; González-Aguilar, G.A. Phenolic compounds promote diversity of gut microbiota and maintain colonic health. Digestive diseases and sciences 2021, 66, 3270–3289. [Google Scholar] [CrossRef]
- Barreca, D.; Gattuso, G.; Bellocco, E.; Calderaro, A.; Trombetta, D.; Smeriglio, A.; Laganà, G.; Daglia, M.; Meneghini, S.; Nabavi, S.M. Flavanones: Citrus phytochemical with health-promoting properties. BioFactors 2017, 43, 495–506. [Google Scholar] [CrossRef]
- Cirmi, S.; Ferlazzo, N.; Lombardo, G.E.; Ventura-Spagnolo, E.; Gangemi, S.; Calapai, G.; Navarra, M. Neurodegenerative diseases: Might citrus flavonoids play a protective role? Molecules 2016, 21, 1312. [Google Scholar] [CrossRef]
- Hussain, F.; Jahan, N.; Rahman, K.-u.; Sultana, B.; Jamil, S. Identification of hypotensive biofunctional compounds of <i>coriandrum sativum</i> and evaluation of their angiotensin-converting enzyme (ace) inhibition potential. Oxidative Medicine and Cellular Longevity 2018, 2018, 4643736. [Google Scholar]
- Gandhi, G.R.; Vasconcelos, A.B.S.; Wu, D.-T.; Li, H.-B.; Antony, P.J.; Li, H.; Geng, F.; Gurgel, R.Q.; Narain, N.; Gan, R.-Y. Citrus flavonoids as promising phytochemicals targeting diabetes and related complications: A systematic review of in vitro and in vivo studies. Nutrients 2020, 12, 2907. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Lin, M.-T.; Zhu, Y.-D.; Xu, H.-L.; Zhao, Y.-Z. Recent trends in potential therapeutic applications of the dietary flavonoid didymin. Molecules 2018, 23, 2547. [Google Scholar] [CrossRef]
- Guijarro-Díez, M.; Nozal, L.; Marina, M.L.; Crego, A.L. Metabolomic fingerprinting of saffron by lc/ms: Novel authenticity markers. Anal Bioanal Chem 2015, 407, 7197–7213. [Google Scholar] [CrossRef]
- Sebghatollahi, Z.; Ghanadian, M.; Agarwal, P.; Ghaheh, H.S.; Mahato, N.; Yogesh, R.; Hejazi, S.H. Citrus flavonoids: Biological activities, implementation in skin health, and topical applications: A review. ACS Food Science & Technology 2022, 2, 1417–1432. [Google Scholar]
- Kumar, S.; Singh, A.; Kumar, B. Identification and characterization of phenolics and terpenoids from ethanolic extracts of phyllanthus species by hplc-esi-qtof-ms/ms. J Pharm Anal 2017, 7, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Ranjit, A.; Sharma, K.; Prasad, P.; Shang, X.; Gowda, K.G.M.; Keum, Y.S. Bioactive compounds of citrus fruits: A review of composition and health benefits of carotenoids, flavonoids, limonoids, and terpenes. Antioxidants. 2022; 11 (2): 239. s Note: MDPI stays neu-tral with regard to jurisdictional claims in …: 2022.
- Zhang, L.; Tu, Z.-c.; Wang, H.; Fu, Z.-f.; Wen, Q.-h.; Chang, H.-x.; Huang, X.-q. Comparison of different methods for extracting polyphenols from ipomoea batatas leaves, and identification of antioxidant constituents by hplc-qtof-ms2. Food Res. Int. 2015, 70, 101–109. [Google Scholar] [CrossRef]
- Zhong, B.; Robinson, N.A.; Warner, R.D.; Barrow, C.J.; Dunshea, F.R.; Suleria, H.A.R. Lc-esi-qtof-ms/ms characterization of seaweed phenolics and their antioxidant potential. Marine drugs 2020, 18, 331. [Google Scholar] [CrossRef]
- Silva, A.S.; Tewari, D.; Sureda, A.; Suntar, I.; Belwal, T.; Battino, M.; Nabavi, S.M.; Nabavi, S.F. The evidence of health benefits and food applications of thymus vulgaris l. Trends in Food Science & Technology 2021, 117, 218–227. [Google Scholar]
- Lesjak, M.; Beara, I.; Simin, N.; Pintać, D.; Majkić, T.; Bekvalac, K.; Orčić, D.; Mimica-Dukić, N. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. Journal of Functional Foods 2018, 40, 68–75. [Google Scholar] [CrossRef]
- Muñoz-Reyes, D.; Casanova, A.G.; González-Paramás, A.M.; Martín, Á.; Santos-Buelga, C.; Morales, A.I.; López-Hernández, F.J.; Prieto, M. Protective effect of quercetin 3-o-glucuronide against cisplatin cytotoxicity in renal tubular cells. Molecules 2022, 27, 1319. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.; Guan, Y.-Y.; Zhang, Z.-L.; Rahman, K.; Wang, S.-J.; Zhou, S.; Luan, X.; Zhang, H. Isorhamnetin: A review of pharmacological effects. Biomedicine & Pharmacotherapy 2020, 128, 110301. [Google Scholar]
- Ali, A.; Cottrell, J.J.; Dunshea, F.R. Lc-ms/ms characterization of phenolic metabolites and their antioxidant activities from australian native plants. Metabolites 2022, 12, 1016. [Google Scholar] [CrossRef] [PubMed]
- Kiani, H.S.; Ali, A.; Zahra, S.; Hassan, Z.U.; Kubra, K.T.; Azam, M.; Zahid, H.F. Phytochemical composition and pharmacological potential of lemongrass (cymbopogon) and impact on gut microbiota. AppliedChem 2022, 2, 229–246. [Google Scholar] [CrossRef]
- Ali, A.; Zahid, H.F.; Cottrell, J.J.; Dunshea, F.R. A comparative study for nutritional and phytochemical profiling of coffea arabica (c. Arabica) from different origins and their antioxidant potential and molecular docking. Molecules 2022, 27, 5126. [Google Scholar] [CrossRef]
- Singh, A.; Bajpai, V.; Kumar, S.; Sharma, K.R.; Kumara, B. Profiling of gallic and ellagic acid derivatives in different plant parts of terminalia arjuna by hplc-esi-qtof-ms/ms. Nat Prod Commun 2016, 11, 239–244. [Google Scholar] [CrossRef]
- Yisimayili, Z.; Abdulla, R.; Tian, Q.; Wang, Y.; Chen, M.; Sun, Z.; Li, Z.; Liu, F.; Aisa, H.A.; Huang, C. A comprehensive study of pomegranate flowers polyphenols and metabolites in rat biological samples by high-performance liquid chromatography quadrupole time-of-flight mass spectrometry. Journal of Chromatography A 2019, 1604, 460472. [Google Scholar] [CrossRef] [PubMed]
- Lescano, C.H.; Freitas de Lima, F.; Caires, A.R.L.; de Oliveira, I.P. Chapter 25 - polyphenols present in campomanesia genus: Pharmacological and nutraceutical approach. In Polyphenols in plants (second edition), Watson, R.R., Ed. Academic Press: 2019; pp 407-420.
- Złotek, U.; Szymanowska, U.; Jakubczyk, A.; Sikora, M.; Świeca, M. Effect of arachidonic and jasmonic acid elicitation on the content of phenolic compounds and antioxidant and anti-inflammatory properties of wheatgrass (triticum aestivum l.). Food chemistry 2019, 288, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Srinivasulu, C.; Ramgopal, M.; Ramanjaneyulu, G.; Anuradha, C.M.; Kumar, C.S. Syringic acid (sa)‒a review of its occurrence, biosynthesis, pharmacological and industrial importance. Biomedicine & Pharmacotherapy 2018, 108, 547–557. [Google Scholar]
- Ríos, J.-L.; Giner, R.M.; Marín, M.; Recio, M.C. A pharmacological update of ellagic acid. Planta medica 2018, 84, 1068–1093. [Google Scholar] [CrossRef] [PubMed]
- Chroho, M.; Bouymajane, A.; Oulad El Majdoub, Y.; Cacciola, F.; Mondello, L.; Aazza, M.; Zair, T.; Bouissane, L. Phenolic composition, antioxidant and antibacterial activities of extract from flowers of rosa damascena from morocco. Separations 2022, 9, 247. [Google Scholar] [CrossRef]
- Bakr, R.O.; El-Naa, M.M.; Zaghloul, S.S.; Omar, M.M. Profile of bioactive compounds in nymphaea alba l. Leaves growing in egypt: Hepatoprotective, antioxidant and anti-inflammatory activity. BMC Complementary and Alternative Medicine 2017, 17, 1–13. [Google Scholar] [CrossRef]
- Zhang, X.-X.; Zhao, D.-S.; Wang, J.; Zhou, H.; Wang, L.; Mao, J.-L.; He, J.-X. The treatment of cardiovascular diseases: A review of ferulic acid and its derivatives. Die Pharmazie-An International Journal of Pharmaceutical Sciences 2021, 76, 55–60. [Google Scholar]
- Zduńska, K.; Dana, A.; Kolodziejczak, A.; Rotsztejn, H. Antioxidant properties of ferulic acid and its possible application. Skin pharmacology and physiology 2018, 31, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Peres, D.D.A.; Sarruf, F.D.; de Oliveira, C.A.; Velasco, M.V.R.; Baby, A.R. Ferulic acid photoprotective properties in association with uv filters: Multifunctional sunscreen with improved spf and uva-pf. Journal of Photochemistry and Photobiology B: Biology 2018, 185, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.M.; Zulfiqar, S.; Naeem, N.; Ibrahim, A.; Hassan, S.K.; Majeed, A.; Siddiqi, S.A. Cinnamon: A multifunctional medicinal plant. Lahore Garrison University Journal of Life Sciences 2017, 1, 196–202. [Google Scholar] [CrossRef]
- Alagawany, M.; Abd El-Hack, M.E.; Farag, M.R.; Gopi, M.; Karthik, K.; Malik, Y.S.; Dhama, K. Rosmarinic acid: Modes of action, medicinal values and health benefits. Animal Health Research Reviews 2017, 18, 167–176. [Google Scholar] [CrossRef]
- Magnani, C.; Isaac, V.L.B.; Correa, M.A.; Salgado, H.R.N. Caffeic acid: A review of its potential use in medications and cosmetics. Analytical Methods 2014, 6, 3203–3210. [Google Scholar] [CrossRef]
- Deng, H.; Xu, Q.; Guo, H.-Y.; Huang, X.; Chen, F.; Jin, L.; Quan, Z.-S.; Shen, Q.-K. Application of cinnamic acid in the structural modification of natural products: A review. Phytochemistry 2022, 113532. [Google Scholar] [CrossRef]
- Gunia-Krzyżak, A.; Słoczyńska, K.; Popiół, J.; Koczurkiewicz, P.; Marona, H.; Pękala, E. Cinnamic acid derivatives in cosmetics: Current use and future prospects. International Journal of Cosmetic Science 2018, 40, 356–366. [Google Scholar] [CrossRef]
- Gurib-Fakim, A. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Molecular aspects of Medicine 2006, 27, 1–93. [Google Scholar] [CrossRef]
- Jesus, M.; Martins, A.P.; Gallardo, E.; Silvestre, S. Diosgenin: Recent highlights on pharmacology and analytical methodology. J Anal Methods Chem 2016, 2016, 4156293. [Google Scholar] [CrossRef]
- Sirtori, C.R. Aescin: Pharmacology, pharmacokinetics and therapeutic profile. Pharmacological Research 2001, 44, 183–193. [Google Scholar] [CrossRef]
- Zhang, L.; Xie, Q.; Li, X. Esculetin: A review of its pharmacology and pharmacokinetics. Phytotherapy Research 2022, 36, 279–298. [Google Scholar] [CrossRef] [PubMed]
- Ammar, S.; del Mar Contreras, M.; Belguith-Hadrich, O.; Bouaziz, M.; Segura-Carretero, A. New insights into the qualitative phenolic profile of ficus carica l. Fruits and leaves from tunisia using ultra-high-performance liquid chromatography coupled to quadrupole-time-of-flight mass spectrometry and their antioxidant activity. RSC Advances 2015, 5, 20035–20050. [Google Scholar] [CrossRef]
- Vallverdú-Queralt, A.; Regueiro, J.; Martínez-Huélamo, M.; Alvarenga, J.F.R.; Leal, L.N.; Lamuela-Raventos, R.M. A comprehensive study on the phenolic profile of widely used culinary herbs and spices: Rosemary, thyme, oregano, cinnamon, cumin and bay. Food Chem. 2014, 154, 299–307. [Google Scholar] [CrossRef]
- Peixoto, J.A.B.; Álvarez-Rivera, G.; Alves, R.C.; Costa, A.S.G.; Machado, S.; Cifuentes, A.; Ibáñez, E.; Oliveira, M. Comprehensive phenolic and free amino acid analysis of rosemary infusions: Influence on the antioxidant potential. Antioxidants 2021, 10, 500. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Trudler, D.; Oh, C.-K.; Lipton, S.A. Potential therapeutic use of the rosemary diterpene carnosic acid for alzheimer’s disease, parkinson’s disease, and long-covid through nrf2 activation to counteract the nlrp3 inflammasome. Antioxidants 2022, 11, 124. [Google Scholar] [CrossRef]
- Bahri, S.; Jameleddine, S.; Shlyonsky, V. Relevance of carnosic acid to the treatment of several health disorders: Molecular targets and mechanisms. Biomedicine & pharmacotherapy 2016, 84, 569–582. [Google Scholar]
- Miraj, S. An evidence-based review on herbal remedies of rosmarinus officinalis. Pharm. Lett 2016, 8, 426–436. [Google Scholar]
- Rubió, L.; Motilva, M.-J.; Romero, M.-P. Recent advances in biologically active compounds in herbs and spices: A review of the most effective antioxidant and anti-inflammatory active principles. Critical reviews in food science and nutrition 2013, 53, 943–953. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, C.; Yang, G.; Yang, Y. Biological properties of 6-gingerol: A brief review. Natural product communications 2014, 9, 1934578X1400900736. [Google Scholar] [CrossRef]
| Variables | Basil leaves | Fennel seeds | Coriander seeds | Mint leaves | Fenugreek leaves |
|---|---|---|---|---|---|
| 80% Methanol | 26.99 ± 1.28 a | 9.90 ± 0.27 a | 23.02 ± 1.53 a | 125.42 ± 10.89 a | 17.21 ± 1.08 a |
| 80% Ethanol | 23.02 ± 1.53 b | 9.61 ± 0.76 a | 21.39 ± 1.91 ab | 116.84 ± 8.47 a | 15.43 ± 1.21 a |
| 80% Chloroform | 17.02 ± 1.01 c | 6.33 ± 0.22 b | 19.32 ± 1.14 c | 88.98 ± 7.32 B | 7.86 ± 0.19 a |
| 80% Acetone | 25.02 ± 2.61 a | 9.32 ± 0.52 a | 22.15 ± 1.28 a | 123.47 ± 11.04 a | 17.01 ± 0.32 a |
| Water | 3.15 ± 0.03 d | 1.01 ± 0.01 c | 3.02 ± 0.04 d | 15.53 ± 1.37 c | 1.52 ± 0.02 a |
| Variables | TPC (mg GAE/g) | TFC (mg QE/g) | TCT (mg CE/g) |
|---|---|---|---|
| Basil leaves | 26.99 ± 1.28 b | 9.35 ± 0.16 b | 3.97 ± 0.10 b |
| Fennel seeds | 9.90 ± 0.27 d | 2.48 ± 0.21 d | 2.88 ± 0.10 bc |
| Coriander seeds | 23.02 ± 1.53 b | 3.35 ± 0.26 de | 11.51 ± 1.68 a |
| Mint leaves | 125.42 ± 10.89 a | 31.72 ± 1.22 a | 3.31 ± 0.08 c |
| Fenugreek leaves | 17.21 ± 1.08 c | 4.41 ± 0.08 c | 1.36 ± 0.08 d |
| Variables | Basil leaves | Fennel seeds | Coriander seeds | Mint leaves | Fenugreek leaves |
|---|---|---|---|---|---|
| ABTS (mg AAE/g) | 34.70 ± 1.55 b | 9.43 ± 0.35 e | 19.17 ± 0.57 c | 74.62 ± 0.77 a | 16.23 ± 0.13 d |
| FICA (mg EDTA/g) | 1.24 ± 0.08 bc | 0.70 ± 0.01 c | 1.88 ± 0.03 b | 4.25 ± 0.05 a | 0.75 ± 0.07 c |
| FRAP (mg AAE/g) | 2.43 ± 0.22 b | 0.54 ± 0.20 d | 1.71 ± 0.33 c | 4.35 ± 0.09 a | 0.14 ± 0.05 de |
| •OH-RSA mg (AAE/g) | 52.17 ± 3.46 b | 23.89 ± 0.17 c | 17.25 ± 1.36 de | 61.87 ± 2.85 a | 19.62 ± 0.46 d |
| PMA (mg AAE/g) | 8.39 ± 0.35 ab | 3.21 ± 0.93 c | 8.04 ± 0.55 ab | 8.64 ± 1.84 a | 3.44 ± 0.32 c |
| α-glucosidase inhibition activity IC50 (μg/mL) | 3.32 ± 0.47 d | 31.92 ± 2.07 a | 9.41 ± 1.82 c | 2.04 ± 0.03 e | 15.83 ± 1.13 b |
| Variables | TPC | TFC | TCT | ABTS | FICA | FRAP | •OH-RSA | PMA |
|---|---|---|---|---|---|---|---|---|
| TFC | 0.99 | |||||||
| TCT | -0.12 | -0.22 | ||||||
| ABTS | 0.97 | 0.99 | -0.13 | |||||
| FICA | 0.97 | 0.93 | 0.13 | 0.93 | ||||
| FRAP | 0.89 | 0.90 | 0.12 | 0.95 | 0.92 | |||
| •OH-RSA | 0.78 | 0.85 | -0.30 | 0.89 | 0.69 | 0.87 | ||
| PMA | 0.10 | 0.11 | 0.67 | 0.26 | 0.25 | 0.50 | 0.32 | |
| α-Glu | 0.98 | 0.97 | -0.04 | 0.96 | 0.96 | 0.95 | 0.83 | 0.21 |
| No. | Proposed compounds | Molecular Formula | RT (min) | ESI +/- | Theoretical (m/z) | Observed (m/z) | MS/MS | Mass error (ppm) | Samples |
|---|---|---|---|---|---|---|---|---|---|
| Flavonoids | |||||||||
| Flavanols | |||||||||
| 1 | (-)-Epigallocatechin 3'-O-glucuronide | C21H22O13 | 7.331 | [M−H]− | 481.0987 | 481.0989 | 305 | 0.4 | F, B |
| 3 | (+)−Catechin | C15H14O6 | 14.158 | [M−H]− | 289.0717 | 289.0701 | 245 | −4.5 | B, Ci, F, M |
| 2 | (+)−Gallocatechin | C15H14O7 | 16.244 | [M−H]− | 305.0667 | 305.0659 | 245 | −2.5 | M, Co, B |
| 4 | Procyanidin trimer C1 | C45H38O18 | 24.969 | [M−H]− | 865.1985 | 865.1972 | 739, 713, 695, 577, 451 | −1.5 | B, M, Fe |
| 5 | Procyanidin B2 | C30H26O12 | 27.396 | [M−H]− | 577.1351 | 577.1353 | 451, 425, 407, 289 | 0.3 | B, M, F |
| Flavanones | |||||||||
| 6 | Hesperidin | C28H34O15 | 31.486 | [M−H]− | 609.1825 | 609.1805 | 301 | −3.3 | B, M, Co, Fe, F |
| 7 | Neoeriocitrin | C27H32O15 | 36.849 | [M−H]− | 595.1668 | 595.1665 | 459, 287, 151 | −0.6 | M, F, B |
| 8 | Didymin | C28H34O14 | 41.407 | [M−H]− | 593.1876 | 593.1872 | 447, 285, 151 | −0.7 | Fe, M, B |
| 9 | 6-Geranylnaringenin | C25H28O5 | 54.360 | [M−H]− | 407.1864 | 407.1878 | 287, 243, 159, 119 | 3.4 | Co, B, Fe |
| Flavones | |||||||||
| 10 | Diosmin | C28H32O15 | 17.572 | [M−H]− | 607.1668 | 607.1697 | 300, 299 | 4.8 | Fe, F, B, M |
| 11 | Apigenin 6,8-di-C-glucoside | C27H30O15 | 21.708 | [M−H]− | 593.1512 | 593.1525 | 449, 287 | 2.2 | F, Co, M, B |
| 12 | Rhoifolin | C27H30O14 | 26.583 | [M−H]− | 577.1563 | 577.1554 | 431, 269 | −1.6 | B, M, F |
| 13 | Apigenin 7-O-glucuronide | C21H18O11 | 32.733 | [M−H]− | 445.0776 | 445.0770 | 269 | −1.8 | F, M, Fe |
| 14 | Apigenin 6-C-glucoside (Isovitexin) | C21H20O10 | 33.72 | [M−H]− | 431.0983 | 431.1001 | 269 | 4.2 | F |
| 15 | Chrysoeriol 7-O-glucoside | C22H22O11 | 35.196 | [M−H]− | 461.1089 | 461.1085 | 299 | −0.9 | F, B, Fe |
| 16 | Cirsilineol | C18H16O7 | 37.596 | [M−H]− | 343.0823 | 343.0815 | 327, 255, 241 | −2.3 | Fe, M |
| Flavonols | |||||||||
| 17 | Quercetin 3'-O-glucuronide | C21H18O13 | 17.998 | [M+H]+ | 479.0820 | 479.0801 | 303 | −4.5 | B |
| 18 | Rutin | C27H30O16 | [M−H]− | 609.1461 | 609.1466 | 301, 300, 271 | 0.9 | F, M, B | |
| 19 | Myricetin 3-O-rhamnoside | C21H20O12 | 28.52 | [M−H]− | 463.0882 | 463.0887 | 317 | 1.1 | B, M, Co |
| 20 | Quercetin 3-O-arabinoside | C20H18O11 | 30.94 | [M−H]− | 433.0776 | 433.0756 | 301 | −4.6 | B, M, F |
| 21 | Isorhamnetin | C16H12O7 | 36.254 | * [M−H]− | 315.0510 | 315.0523 | 300, 151, 107 | 4.1 | M |
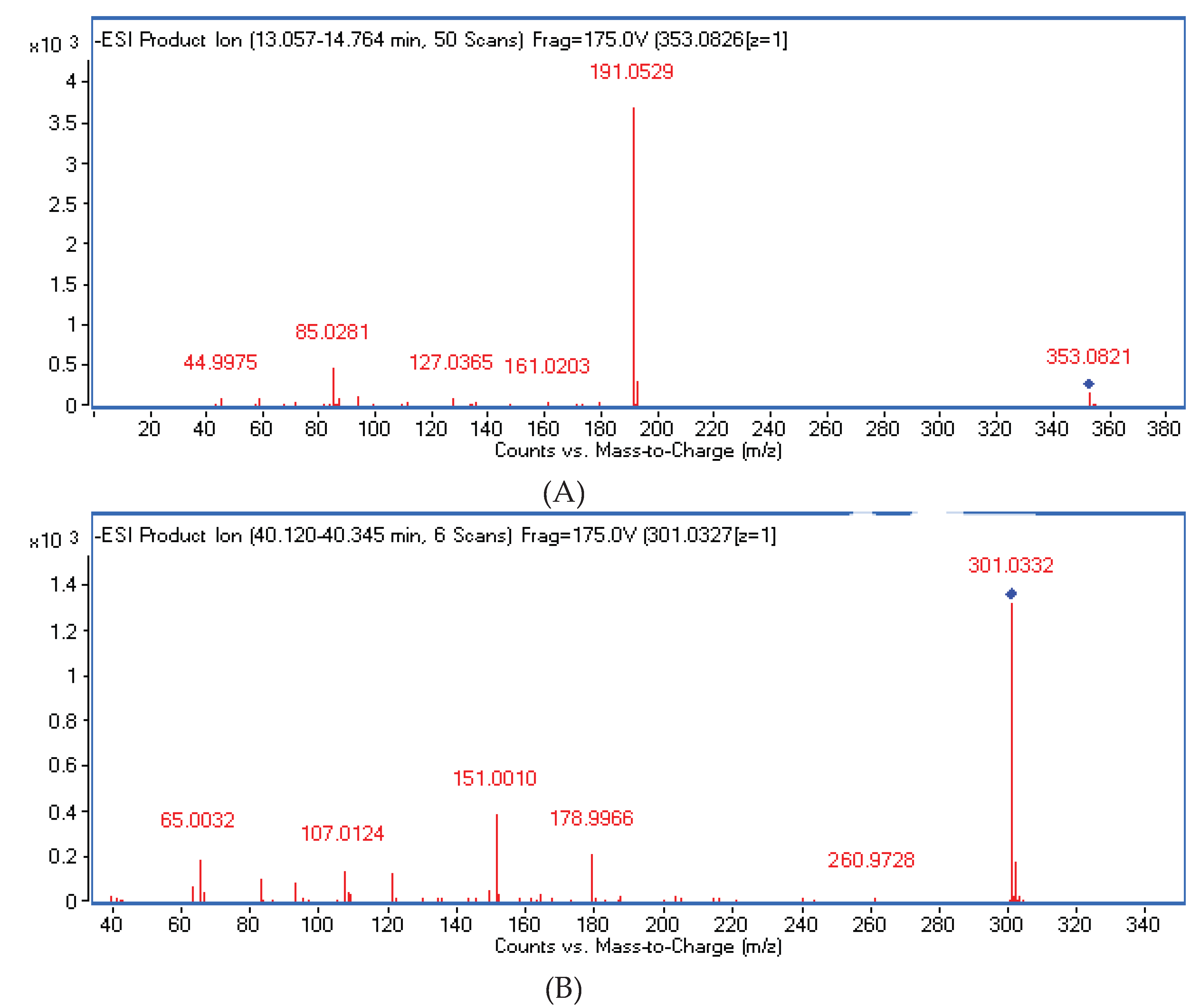
| 22 | Quercetin | C15H10O7 | 40.120 | [M−H]− | 301.0349 | 301.0332 | 179, 151 | −4.2 | F, B, M |
| 23 | 3,7-Dimethylquercetin | C17H14O7 | 43.569 | * [M−H]− | 329.0667 | 329.0658 | 314, 299, 271 | −2.7 | B |
| Isoflavonoids | |||||||||
| 24 | Equol 4'-O-glucuronide | C21H22O9 | 17.955 | [M−H]− | 417.1191 | 417.1191 | 399, 241 | 0.0 | M |
| 25 | 3'-O-Methylequol | C16H16O4 | 18.413 | [M−H]− | 271.0976 | 271.0964 | 255, 149, 121 | −4.4 | M |
| 26 | 6''-O-Acetylglycitin | C24H24O11 | 26.07 | [M−H]− | 487.1246 | 487.1248 | 283, 267, 59 | 0.6 | F, M |
| 27 | 3'-Hydroxygenistein | C15H10O6 | 32.762 | * [M−H]− | 285.0404 | 285.0418 | 267, 251, 201, 177 | 4.9 | M |
| 28 | 3'-Hydroxydaidzein | C15H10O5 | 36.949 | [M−H]− | 269.0455 | 269.045 | 241, 224, 213, 181 | −1.9 | B, F, M |
| 29 | 3',4',7-Trihydroxyisoflavanone | C15H12O5 | 47.982 | [M−H]− | 271.0612 | 271.0611 | 239, 135, 121 | −0.4 | M |
| 30 | Violanone | C17H16O6 | 48.32 | [M−H]− | 315.0874 | 315.0866 | 300, 285, 135 | −2.5 | M |
| Phenolic Acids | |||||||||
| Hydroxybenzoic acids | |||||||||
| 31 | Gallic acid 4-O-glucoside | C13H16O10 | 10.099 | [M−H]− | 331.067 | 331.0666 | 169, 125 | −1.2 | M, Co, F, B |
| 32 | Gallic acid | C7H6O5 | 10.544 | * [M−H]− | 169.0142 | 169.0144 | 125 | 1.2 | B, M, F, Fe, Co |
| 33 | Syringic acid | C9H10O5 | 12.284 | * [M−H]− | 197.0455 | 197.0459 | 182, 163, 138, 123, 95 | 2 | B, F, M, Fe |
| 34 | Protocatechuic acid 4-O-glucoside | C13H16O9 | 12.569 | [M−H]− | 315.0721 | 315.0727 | 153 | 1.9 | F, M, B |
| 35 | 3-O-Methylgallic acid | C8H8O5 | 14.587 | * [M−H]− | 183.0299 | 183.0292 | 139, 123 | −3.8 | M, F, B |
| 36 | Protocatechuic acid | C7H6O4 | 14.784 | * [M−H]− | 153.0193 | 153.0197 | 109 | 2.6 | F, M, B, Fe |
| 37 | 4-Hydroxybenzoic acid 4-O-glucoside | C13H16O8 | 16.589 | * [M−H]− | 299.0772 | 299.0786 | 255, 137 | 4.7 | *F, Ci, B |
| 38 | Punicafolin | C41H30O26 | 20.341 | [M−H]− | 937.0952 | 937.0967 | 169, 125 | 1.7 | F, B, M |
| 39 | Ellagic acid | C14H6O8 | 23.790 | [M−H]− | 300.999 | 300.9977 | 284, 257 | −4.3 | Fe, M, B, F |
| 40 | Valoneic acid dilactone | C21H10O13 | 30.649 | [M−H]− | 469.0048 | 469.0052 | 425, 301, 139 | 0.9 | M |
| 41 | p-Hydroxybenzoic acid | C7H6O3 | 37.172 | * [M−H]− | 137.0244 | 137.0242 | 93 | −1.5 | F, Co, B, M, Fe |
| Hydroxycinnamic acids | |||||||||
| 42 | 3-Caffeoylquinic acid | C16H18O9 | 13.005 | * [M−H]− | 353.0878 | 353.0867 | 191, 179, 161 | −3.1 | Fe, B, F, M |
| 43 | Ferulic acid | C10H10O4 | 13.817 | [M−H]− | 193.0506 | 193.0504 | 178, 149, 134 | −1.0 | Fe, Co, F, B, M |
| 44 | Caffeoyl glucose | C15H18O9 | 14.764 | [M−H]− | 341.0878 | 341.0878 | 179 | 0.0 | B, M |
| 45 | Caffeoyl aspartic acid | C13H13NO7 | 17.247 | [M−H]− | 294.0619 | 294.0631 | 179 | 4.1 | M |
| 46 | p-Coumaric acid 4-O-glucoside | C15H18O8 | 17.313 | [M−H]− | 325.0929 | 325.0923 | 163 | −1.8 | B, F, M |
| 47 | p-Coumaric acid | C9H8O3 | 18.32 | * [M−H]− | 163.04 | 163.0405 | 119 | 3.1 | F, Co, B, M |
| 48 | Caffeic acid | C9H8O4 | 21.084 | [M−H]− | 179.035 | 179.0346 | 135 | -2.2 | B, F, M |
| 49 | Sinapic acid | C11H12O5 | 21.339 | * [M−H]− | 223.0612 | 223.0615 | 193, 179, 149, 134 | 1.2 | M, Fe, F, B |
| 50 | 3-p-Coumaroylquinic acid | C16H18O8 | 23.499 | [M−H]− | 337.0929 | 337.0939 | 191, 119 | 3.0 | F, B, M |
| 51 | 3-Feruloylquinic acid | C17H20O9 | 24.897 | [M−H]− | 367.1034 | 367.1030 | 193, 191 | −1.6 | F, M, B |
| 52 | Chicoric acid | C22H18O12 | 30.115 | * [M−H]− | 473.0725 | 473.0736 | 293, 311 | 2.3 | B, M |
| 53 | 1,5-Dicaffeoylquinic acid | C25H24O12 | 30.921 | [M−H]− | 515.1195 | 515.1197 | 191, 179, 135 | 0.7 | B, F, M, Fe |
| 54 | Rosmarinic acid | C18H16O8 | 33.313 | [M−H]− | 359.0772 | 359.0776 | 197, 179, 161, 135 | 1.1 | M, B, F |
| 55 | Cinnamic acid | C9H8O2 | 38.22 | [M−H]− | 147.0451 | 147.046 | 103 | 6.1 | F, B, M, Fe |
| Other compounds | |||||||||
| 56 | Scopoletin | C10H8O4 | 8.681 | [M−H]− | 191.035 | 191.035 | 175, 147 | 0.0 | M |
| 57 | Pyrogallol | C6H6O3 | 8.573 | [M−H]− | 125.0244 | 127.0256 | 107, 97, 79 | 2.1 | B, M, F |
| 58 | Trigonelline | C7H7NO2 | 14.621 | [M−H]− | 138.0549 | 138.0553 | 120, 94, 92 | 5.0 | M, F |
| 59 | p-Anisaldehyde | C8H8O2 | 15.526 | * [M−H]− | 135.0451 | 135.0445 | 122, 109 | 1.7 | B, F, M |
| 60 | Diosgenin | C27H42O3 | 16.716 | [M+H]+ | 415.3212 | 415.3212 | 271, 253, 157 | 0.0 | F |
| 61 | Sagerinic acid | C36H32O16 | 26.198 | [M−H]− | 719.1612 | 719.1610 | 359, 197, 179, 161 | −1.2 | B, M, Fe |
| 62 | Esculin | C15H16O9 | 20.426 | [M−H]− | 339.0721 | 339.0728 | 177 | 2.2 | F, M |
| 63 | Esculetin | C9H6O4 | 20.473 | [M−H]− | 177.0193 | 177.0196 | 133, 105 | −4.4 | F, B |
| 64 | Coumarin | C9H6O2 | 22.521 | [M+H]+ | 147.0441 | 147.0441 | 103 | 0.0 | M, B, F |
| 65 | 2-Hydroxybenzaldehyde | C7H6O2 | 24.656 | * [M−H]− | 121.0295 | 121.0292 | 92, 77 | −2.5 | Ci, F, B |
| 66 | Umbelliferone | C9H6O3 | 26.399 | * [M−H]− | 161.0244 | 161.0242 | 133 | −1.6 | F, M |
| 67 | Rosmanol | C20H26O5 | 26.775 | [M−H]− | 345.1707 | 347.1708 | 301 | 0.2 | F, Fe |
| 68 | Carnosic acid | C20H28O4 | 44.680 | [M−H]− | 331.1915 | 331.1903 | 287 | −4.5 | B, M, F |
| 69 | Carnosol | C20H26O4 | 46.724 | [M−H]− | 329.1758 | 329.1760 | 285 | 0.6 | B, M |
| 70 | 3,4,5,4'-Tetramethoxystilbene | C26H44N4O2S | 47.334 | [M−H]− | 475.3112 | 475.3112 | 475 | 0.0 | M |
| 71 | [6]-Gingerol | C17H32O4 | 49.633 | [M−H]− | 293.1758 | 293.1769 | 299 | 3.6 | B, M |
| 72 | Carvacrol | C10H14O | 58.621 | [M−H]− | 149.0972 | 149.0966 | 131, 105 | 4.4 | B, M, F |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





